Introduction
Nuclear receptor subfamily 2, group E member 3
(NR2E3) serves an important function in retinal photoreceptor cell
development and maintenance (1).
Mutations in the human nr2e3 gene have been reported to
cause several retinal degenerative diseases, such as enhanced
S-cone syndrome and retinitis pigmentosa (2). In recent years, research on the
molecular function of NR2E3 in other tissues and its role in
various diseases, such as liver injury and breast cancer, has been
attracting attention (3,4). In estrogen receptor (ER)+
breast cancer, the level of nr2e3 expression has been found
to be positively associated with recurrence-free survival. In
addition, patients with higher nr2e3 expression tended to be
more sensitive to tamoxifen treatment, which in turn confers more
positive clinical outcomes compared with those with lower
nr2e3 expression (5).
Nr2e3 is typically expressed at low levels in ER-
breast cancer tissues and its overexpression induces cancer cell
growth, invasion and metastasis (6). In addition, elevated levels of
nr2e3 expression have been associated with improved clinical
prognosis in patients with hepatic carcinoma, and with the
occurrence and progression of lung carcinoma and pancreatic cancer
(7-9).
These findings suggest that outside the retina, NR2E3 can serve
biological functions in cancers.
The cancer stem cell (CSC) theory hypothesizes that
a subgroup of malignant cancer cells is endowed with stem-like
properties that are undifferentiated, express self-renewing
capacities and can replenish other differentiated bulk tumor cells
(10). Clinically, conventional
radiotherapy and drug treatments can terminate the majority of
differentiated tumor cells, but they have limited efficacy against
CSCs. CSCs also appear to form the basis of tumor heterogeneity and
the pathological cause of tumor growth, drug resistance, metastasis
and recurrence (11,12). In this regard, promoting the
differentiation of CSCs into drug-sensitive cancer cells, known as
differentiation therapy, has been proposed to be a promising
treatment strategy for eradicating cancer cells (13-15).
Therefore, identifying novel biomarkers that match the specific
molecular signatures of cancer cells to broaden the target spectrum
may prove beneficial.
The present study hypothesized that nr2e3 is
expressed at higher levels in differentiated tumor cells in
ER+ breast cancer compared with breast cancer stem cells
based on the following considerations: i) CSCs form a fraction of
the tumor cell population that can differentiate into the majority
of the bulk tumor cell types, which can then contribute to poor
prognosis and drug resistance (16); ii) patients with breast cancer with
higher nr2e3 expression levels tend to have superior
clinical outcomes and exhibit favorable responses to tamoxifen
treatment (5); and iii)
Nr2e3 expression was previously found to be increased in
ER+ breast tumor tissues compared with that in normal
and ER- breast tissues (17). In addition, it was hypothesized
that NR2E3 may facilitate the differentiation of breast CSCs into
bulk tumor cells, not too dissimilar to its activity in promoting
the differentiation of rod photoreceptors from retinal pluripotent
cells in the retina (18).
Therefore, the present study aimed to investigate the relationship
between nr2e3 expression and the stem-like characteristics
of ER+ breast cancer cells to evaluate the suitability
of NR2E3 as a diagnostic and therapeutic biomarker for
ER+ breast cancer.
Materials and methods
Cell culture
Human normal mammary epithelial cells MCF10A (cat.
no. CL-0525) and the ER+ cell line MCF7 (cat. no.
CL-0149) were purchased from Procell Life Science & Technology
Co., Ltd. The two cell lines were cultured in their cell-specific
complete medium (cat. nos. CM-0525 for MCF10A and CM-0149 for MCF7;
Procell Life Science & Technology Co., Ltd.,). When MCF10A
cells were cultured, 5% horse serum, 20 ng/ml epidermal growth
factor, 0.5 µg/ml hydrocortisone, 1% non-essential amino acid
(NEAA), 10 µg/ml insulin and 1% penicillin-streptomycin solution
(P/S) were added to DMEM/F12 medium. When MCF7 cells were cultured,
10% fetal bovine serum (FBS), 1% NEAA, 10 µg/ml insulin and 1% P/S
were added to the MEM. According to a previous study (19), paclitaxel-resistant MCF7 cells were
cultured with 10 µg/ml paclitaxel (cat. no. ab120143; Abcam) over 6
months. All cells were cultured at 37˚C with 5% CO2.
Plasmids and transfection
The negative control (NC) and nr2e3
short-hairpin RNAs (shRNAs) were designed and purchased from
RiboBio Co., Ltd. The target sequences (Table I) were cloned into the
pRNAT-U6.1/Neo plasmid (RiboBio Co., Ltd.). The full-length cDNA
sequence of human nuclear receptor subfamily 2 group C member 2
(nr2c2) was cloned into the pEGFP-N1 vector (Miaoling
Biotechnology Co., Ltd.). Lipofectamine™ 3000 reagents
(cat. no. L300000; Invitrogen; Thermo Fisher Scientific, Inc.) was
used to transfect the shRNA-carrying plasmids and/or the
nr2c2-overexpression plasmids into MCF7 cells. Plasmids were
incubated with Lipofectamine™ 3000 reagents and
Opti-MEM™ (cat. no. 11058021; Invitrogen; Thermo Fisher
Scientific, Inc.) for 20 min at room temperature. Dishes with a
diameter of 35 mm and 15 mm were transfected with 6 and 4 µg of
plasmids, respectively. For the nr2c2 overexpression
experiment, MCF7 cells were co-transfected with nr2e3 shRNA
plasmids and the nr2c2-overexpression vector. 36 h after
transfection, cells were harvested for subsequent experiments.
 | Table IshNC and NR2E3-specific shRNA
sequences. |
Table I
shNC and NR2E3-specific shRNA
sequences.
| shRNA used | shRNA sequence
(5'-3') |
|---|
| shNC | Sense:
CAACAAGATGAAGAGCACCAA |
| | Antisense:
TTGGTGCTCTTCATCTTGTTG |
| shRNA1 | Sense:
GAAGGATCCTGAGCACGTA |
| | Antisense:
TACGTGCTCAGGATCCTTC |
| shRNA2 | Sense:
GGGAAGCACTATGGCATCT |
| | Antisense:
AGATGCCATAGTGCTTCCC |
| shRNA3 | Sense:
CATGGCCAGCCTTATAACA |
| | Antisense:
TGTTATAAGGCTGGCCATG |
The Cancer Genome Atlas (TCGA) data
analysis
According to our previous study (19), the expression data of nr2e3
and nr2c2 in 808 ER+ breast tumor and 113 normal
tissues were downloaded from the TCGA database (https://portal.gdc.cancer.gov/; version 32.0) for
bioinformatics analysis. Furthermore, the expression profile of
nr2e3 in 1,109 breast tumor samples that were not
categorized according to ER content was also downloaded from the
TCGA database. BRB-Arra/Tools (http://brb.nci.nih.gov/BRB-ArrayTools/download.html,
version 4.6.2) was used to analyze these data. Briefly, data
collation and gene labeling modules were used, differentially
expressed genes in the dataset were screened with P<0.05 and
logFC >1 as criteria, and hierarchical cluster analysis was
performed.
MTT assay
The MTT cytotoxicity assay was conducted using an
MTT Cell Proliferation and Cytotoxicity Detection kit (cat. no.
C0009S; Beyotime Institute of Biotechnology). Briefly,
~2x103 MCF7 cells transfected with either NR2E3 shRNAs
or shNC were seeded into 96-well plates, before 10 µl MTT reagent
was added. After incubation at 37˚C for 4 h, 100 µl formazan
solvent (DMSO) was added. After incubation for another 4 h (at
37˚C), absorbance at 570 nm was measured.
Wound healing assay
MCF7 cells were cultured in complete medium.
shRNA-transfected MCF7 cell monolayers were scratched when the
confluence reached ~80%. Scratched cells were serum starved, and
incubated at 37˚C with 5% CO2. Images were captured at 0
and 24 h post-scratching using an inverted fluorescent microscope
(cat. no. CKX53; Olympus Corporation) under the same
magnification.
Two-dimensional colony formation
assay
A total of 1,000 transfected MCF7 cells were seeded
into 6-cm dishes, maintained in complete medium, and incubated for
2 weeks at 37˚C with 5% CO2. Colonies were fixed with 4%
paraformaldehyde (cat. no. P0099; Beyotime Institute of
Biotechnology) for 30 min and stained with crystal violet (cat. no.
C0121; Beyotime Institute of Biotechnology) for 10 min, all at room
temperature. Images were taken using an Ordinary camera (cat. no.
VlogR7; Canon Corporation). Colonies that contain more than 50
cells were counted manually.
Transwell assays
Transwell chambers (cat. no. 3422; BD Biosciences)
were used to investigate cell migration capacities. In every well,
a total of ~2x103 cells were resuspended in 200 µl
serum-free DMEM and placed into the upper chamber, whereas 600 µl
complete medium with 10% FBS was added to the lower chamber. After
incubation for 24 h at 37˚C, the cells were fixed in 4%
formaldehyde for 30 min and stained with crystal violet for 10 min,
both at room temperature. The number of cells on the underside of
the membrane was counted under an Olympus CKX53 inverted microscope
at x200 magnification, before cells in three randomly selected
fields were counted.
Quantitative PCR (qPCR)
MCF7 cells that were transfected with nr2e3
shRNAs were harvested when the confluence reached ~90%. RNAiso Plus
Reagent (cat. no. 9108; Takara Bio Inc.) was used to extract the
total RNA. cDNA was synthesized through reverse transcription using
the BeyoRT™ II kit (cat. no. D7168M; Beyotime Institute
of Biotechnology). Briefly, total RNA, oligo(dT)18 primer, reaction
buffer, RNase inhibitor, dNTP Mix, BeyoRT™ II M-MLV and
DEPC-treated water formed a reaction system. Following the
manufacturer's instruction, this system was incubated at 42˚C for
60 min, and then for 10 min at 80˚C. For the qPCR experiment,
AceQ™ qPCR SYBR® Green Master Mix (cat. no.
Q111-02; Vazyme Biotechnology Co., Ltd.) was used. According to the
manufacturer's instructions, thermocycling conditions for PCR were
95˚C for 10 sec and then 60˚C for 30 sec. The relative mRNA
expression was calculated using the 2-ΔΔCq method. The
primer sequences used for qPCR are listed in Table II. GAPDH was used as the
normalization control.
 | Table IIPrimers used for reverse
transcription-quantitative PCR. |
Table II
Primers used for reverse
transcription-quantitative PCR.
| Gene (accession
number) | Primer sequence (5'
to 3') |
|---|
| Nr2e3 | F:
GATCCTGAGCACGTAGAGGC |
| (NM_016346.4) | R:
GCAATTTCCCAAACCTCACGG |
| Nr2c2 | F:
GGCGCCAAATCCTGAGGTAA |
| (NM_003298.5) | R:
GGTGAGGCTACAGCAGAGTC |
| Nanog | F:
TCCTCCTCTTCCTCTATACTAAC |
| (NM_024865.4) | R:
CCCACAAATCACAGGCATAG |
| Klf4 | F:
ATCTCGGCCAATTTGGGGTT |
| (NM_004235.6) | R:
CCAGGTGGCTGCCTCATTA |
| Oct4 | F:
ATCGAGAACCGAGTGAGA |
| (NM_002701.6) | R:
ACACTCGGACCACATCCTT |
| Sox2 | F:
GGGAAATGGGAGGGGTGCAAAAGAGG |
| (NM_003106.4) | R:
TTGCGTGAGTGTGGATGGGATTGGTGT |
|
E-cadherin | F:
CCTCCAGAGTTTACTGCCATGAC |
| (NM_001792.5) | R:
GTAGGATCTCCGCCACTGATTC |
|
N-cadherin | F:
GGCGCCACCTGGAGAGA |
| (NM_004360.5) | R:
TGTCGACCGGTGCAATCTT |
|
Vimentin | F:
TACAGGAAGCTGCTGGAAGG |
| (NM_003380.5) | R:
ACCAGAGGGAGTGAATCCAG |
| GAPDH | F:
GGAGCGAGATCCCTCCAAAAT |
| (NM_002046.7) | R:
GGCTGTTGTCATACTTCTCATGG |
Western blotting
MCF7 cells that were transfected with nr2e3
shRNAs were harvested when the confluence reached ~90%. Total
proteins were extracted using cell lysis buffer for western
blotting (cat. no. P0013; Beyotime Institute of Biotechnology). The
protein concentration was determined with a bicinchoninic acid kit
(cat. no. P0010; Beyotime Institute of Biotechnology). A 10% gel
was used for electrophoresis and each lane was loaded with 20 µg
protein. Proteins were then transferred to a polyvinylidene
fluoride membrane (cat. no. FFP78; Beyotime Institute of
Biotechnology), and incubated in blocking buffer (cat. no. P0023B;
Beyotime Institute of Biotechnology) for 2 h at room temperature.
After washing with TBST buffer (containing 20% Tween), the
membranes were then incubated with the primary and secondary
antibodies (primary antibodies, 1:500; secondary antibodies,
1;10,000). A visualization reagent (cat. no. KF8005; Affinity
Biosciences) was the applied. The primary and secondary antibodies
used for western blotting are listed in Table III. GAPDH was used as the
normalization control.
 | Table IIIAntibodies used for WB and FCM. |
Table III
Antibodies used for WB and FCM.
| Protein | Experiment | Company | Cat. no. |
|---|
| NR2E3 | WB | Proteintech Group,
Inc. | 14246-1-AP |
| NR2E3 | WB | Santa Cruz
Biotechnology, Inc. | sc-374513 |
| E-cadherin | WB | ProteinTech Group,
Inc. | 20874-1-AP |
| N-cadherin | WB | ProteinTech Group,
Inc. | 22018-1-AP |
| VIMENTIN | WB | Affinity
Biosciences, Ltd. | BF8006 |
| SLUG | WB | Affinity
Biosciences, Ltd. | AF4002 |
| NR2C2 | WB | ABclonal Biotech
Co., Ltd. | A6422 |
| LSD1 | WB | ABclonal Biotech
Co., Ltd. | A1156 |
| H3K4me2 | WB | ABclonal Biotech
Co., Ltd. | A2356 |
| GAPDH | WB | ProteinTech Group,
Inc. | 60004-1-Ig |
| Goat anti-rabbit
IgG | WB | Proteintech Group,
Inc. | SA00001-2 |
| Goat anti-mouse
IgG | WB | Proteintech Group,
Inc. | SA00001-1 |
| CD44-FITC | FCM | Invitrogen; Thermo
Fisher Scientific, Inc. | 11-0441-82 |
| CD24-PE | FCM | Invitrogen; Thermo
Fisher Scientific, Inc. | 12-0247-42 |
JASPAR prediction
The online prediction software JASPAR (https://jaspar.genereg.net) was used to predict the
NR2E3 binding site on the nr2c2 gene promoter. The human
nr2c2 gene promoter sequence (https://www.ncbi.nlm.nih.gov/nuccore/NC_000003.12?from=14947583&to=15049273&report=fasta)
with FASTA format was scanned at 2 kbp length.
Flow cytometry
A total of ~1x106 nr2e3-silenced
MCF7 cells and the negative control cells, as well as the
nr2c2-overexpressed MCF7 cells were incubated with 5 µl FC
receptor blocker (cat. no. abs9476; Absin Bioscience, Inc.) at 4˚C
for 10 min, and then incubated on ice with 0.25 µg FITC-conjugated
CD44 and 0.25 µg PE-conjugated CD24 antibodies for 30 min. The
percentages of CD44+CD24-/low subgroup cells
were detected using flow cytometry with the Beckman CytoFlex system
(Beckman Coulter, Inc.) and analyzed using FlowJo software (version
10.8.1). The antibodies used for flow cytometry are listed in
Table III.
Statistical analysis
All data were analyzed using GraphPad Prism (version
6; Dotmatics). One-way ANOVA followed by Tukey's post hoc test was
used for all comparisons. Data are presented as the mean ± SD. All
data were obtained from at least three independent experiments.
P<0.05 was used to indicate a statistically significant
difference.
Results
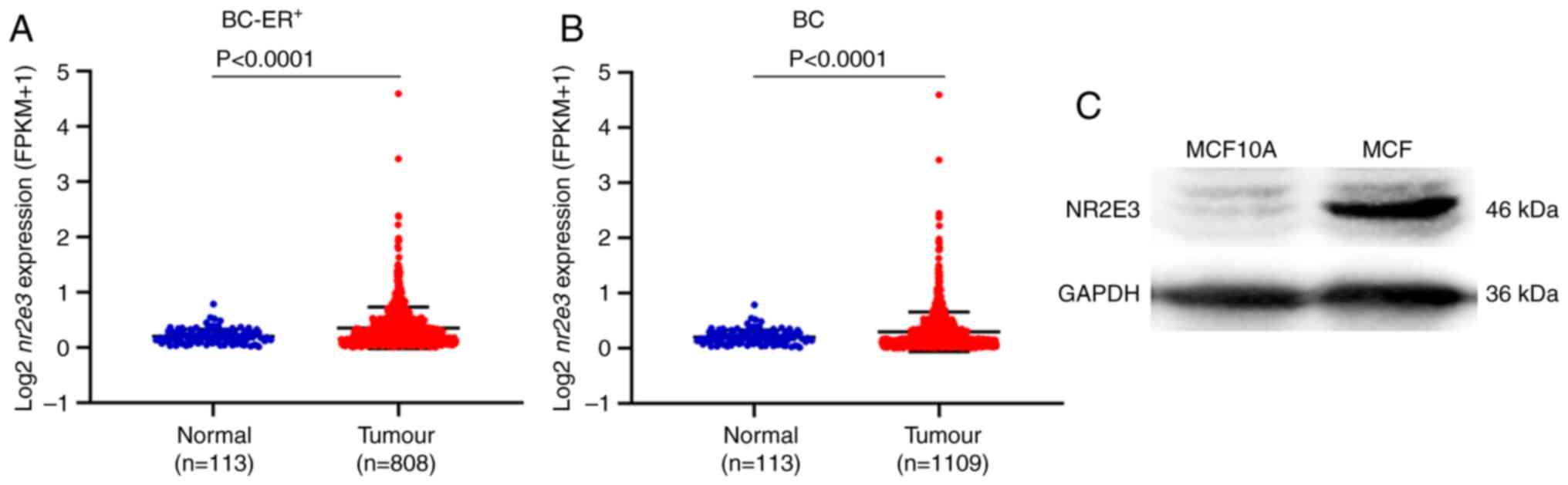
Expression of nr2e3 is increased in
ER+ breast cancer tissues and cell lines
To investigate the biological roles of NR2E3 in
ER+ breast cancer, its expression was first investigated
in ER+ breast cancer tissues compared with that in
normal breast tissue samples. Data from 808 ER+ breast
tumors and 113 normal samples were downloaded from TCGA database,
where the subsequent analysis revealed that the expression of
nr2e3 was significantly higher in the tumor samples
(Fig. 1A). When the tumor types
were not categorized according to the ER content, the expression of
nr2e3 remained significantly higher in breast cancer tissues
(Fig. 1B). The protein expression
level of NR2E3 was found to be elevated in the ER+ cell
line MCF7 compared with that in the MCF10A normal human breast
epithelial cell line (Fig. 1C).
These results suggest that nr2e3 expression is increased in
ER+ breast cancer and tumor types not categorized
according to the ER content.
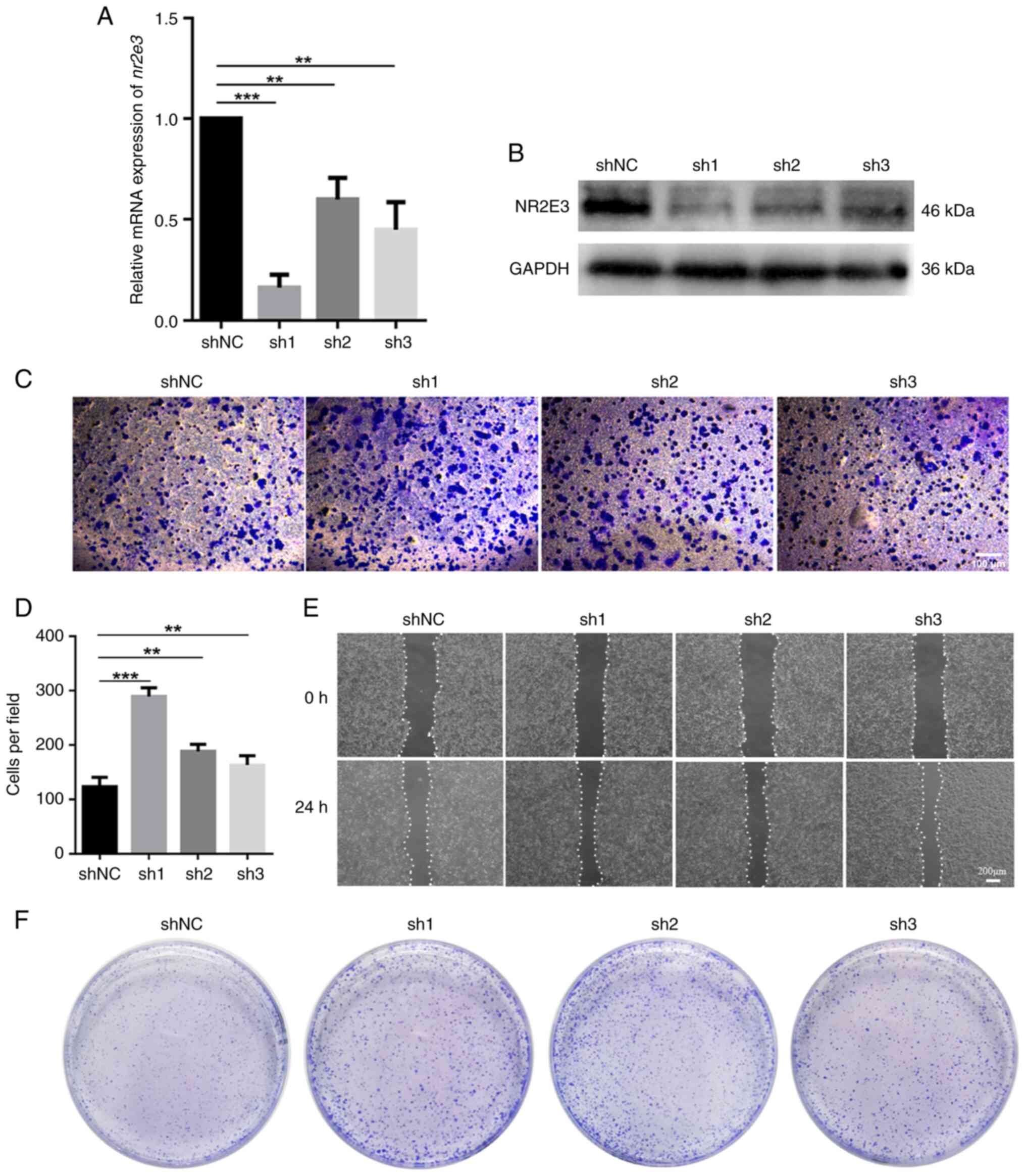
Nr2e3 silencing promotes the
migration, invasion and colony-formation by MCF7 cells
The association between nr2e3 expression and
the migration, invasion and colony-formation of MCF7 cells was next
assessed as an indication of the stemness property of CSCs in
vitro. In total, three shRNAs targeting nr2e3 were
designed and transfected into MCF7 cells. None of the three
nr2e3-specific shRNAs, sh1, sh2 and sh3, nor the shNC,
exhibited cytotoxicity towards MCF7 cells according to the MTT
assay (Fig. S1A). In addition,
mRNA and protein expression levels of nr2e3 were found to be
significantly decreased in shRNA-transfected cells compared with
those in the shNC group (Fig. 2A
and B). Among them, sh1 exerted
the highest silencing effect and so was selected for use in further
experiments. The results of the Transwell and wound healing assays
showed that nr2e3 knockdown markedly increased both their
migratory and invasive capabilities (Figs. 2C-E and S1B). Two-dimensional colony formation
tests showed that silencing nr2e3 expression markedly
promoted colony-formation (Figs.
2F and S2A). Considering that
CSCs contribute to the migration and invasion ability of tumor
cells (12), while nr2e3
knockdown increases the migration and invasion ability of MCF7
cells and promotes the colony formation of MCF7 cells, it can be
inferred that nr2e3 knockdown enhances the stem cell-like
properties of ER+ tumor cells.
In addition, paclitaxel treatment terminated most
differentiated tumor cells, whereas the ratio of stem-like cells
was elevated in paclitaxel-resistant (PR) breast tumor cells
(20). In the present study, it
was found that nr2e3 was expressed at lower levels in
MCF7-PR cells (Fig. S2B). This
finding supported the notion that nr2e3 is mainly expressed
in differentiated tumor cells, whereas in stem-like breast tumor
cells nr2e3 expression is low.
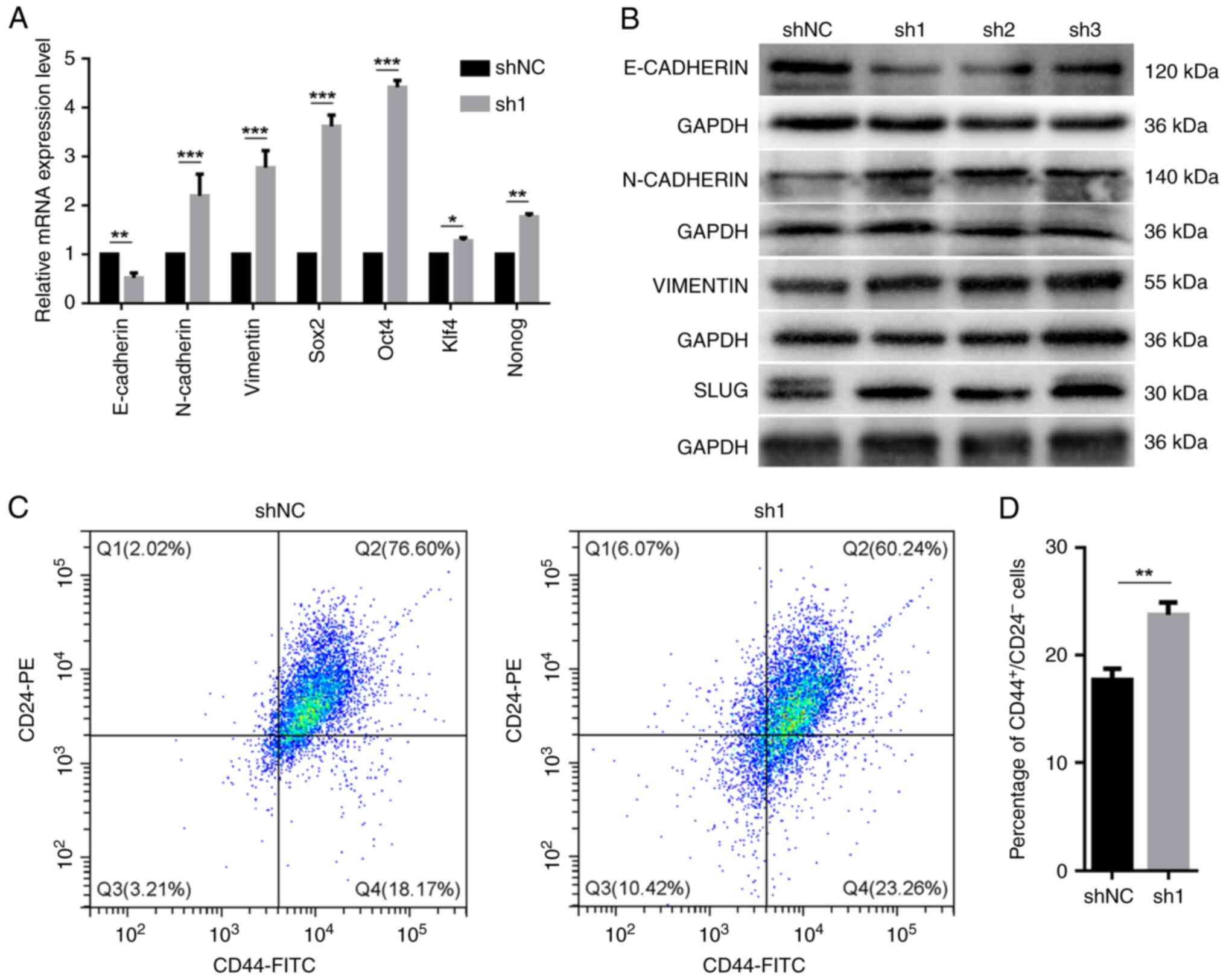
nr2e3 silencing promotes
epithelial-mesenchymal transition (EMT), enhances the expression of
stem cell-related transcription factors and increases the
proportion of CD44+CD24-/low cells
Tumor cells that undergo epithelial-mesenchymal
transition (EMT) usually have enhanced migration, invasion and drug
resistance. Additionally, EMT gives carcinoma cells the capacity to
renew themselves and increases the ratio of tumor stem cells
(21). Thus, the relationship
between nr2e3 expression and the expression of EMT-related
marker genes was next evaluated. It was shown that nr2e3
knockdown significantly reduced the mRNA expression levels of
E-cadherin, but increased the expression of
N-cadherin and vimentin mRNA (Fig. 3A). Similar directions of changes in
the protein expression levels of E-cadherin, N-cadherin and
vimentin were found using western blotting (Figs. 3B and S3). The protein expression of slug, a
transcription factor that can promote EMT (22), was also shown to be increased when
nr2e3 expression was knocked down (Figs. 3B and S3). These results support the hypothesis
that nr2e3 knockdown can promote the EMT process.
Furthermore, nr2e3 knockdown was found to
significantly increase the mRNA expression levels of stem
cell-associated transcription factors sox2, oct4,
nanog and Kruppel-like factor 4 (Fig. 3A). The association between NR2E3
and the proportion of the CD44+CD24-/low
population was investigated, where it was found that nr2e3
knockdown significantly enhanced the proportion of
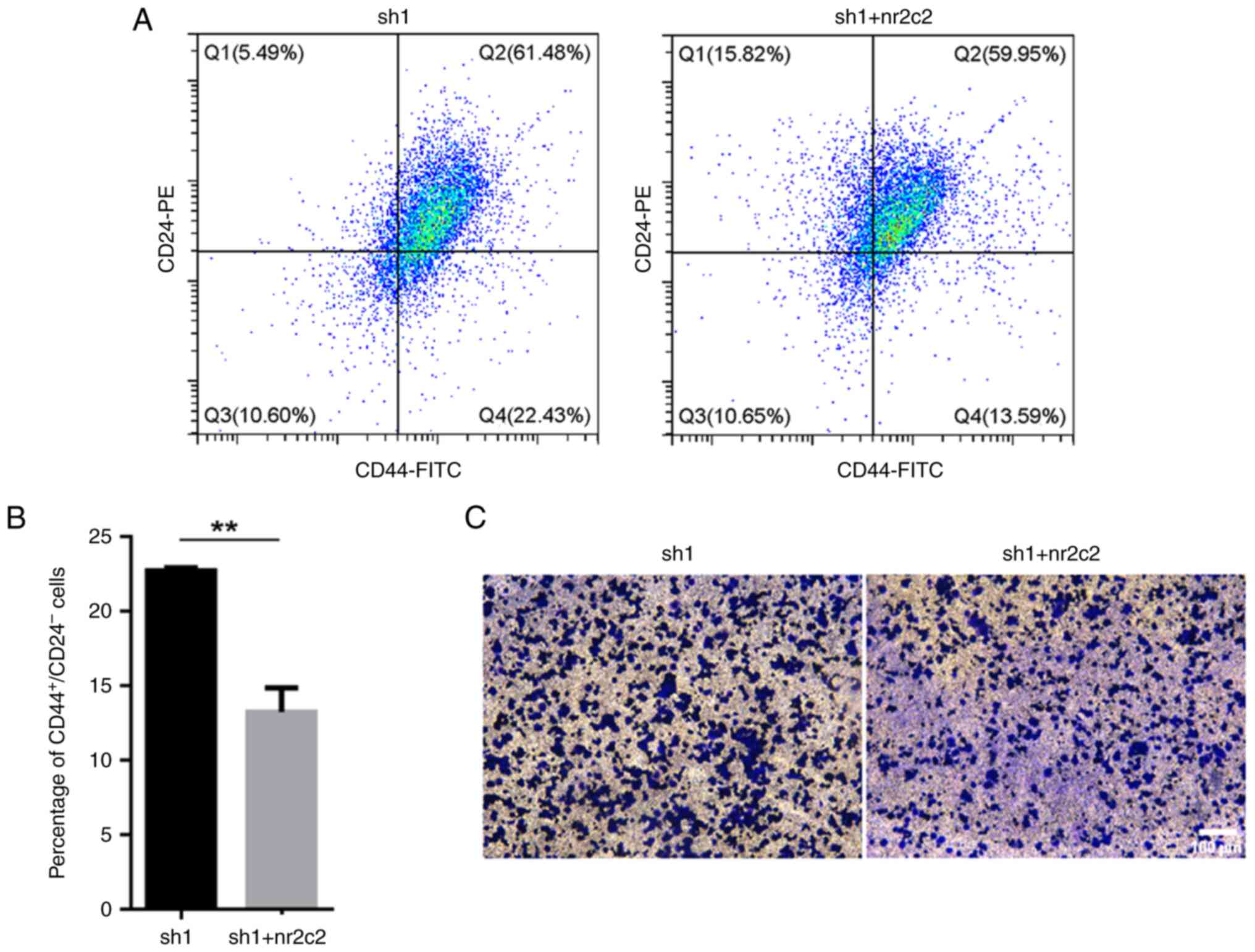
CD44+CD24-/low in MCF7 tumor cells (Fig. 3C and D).
NR2C2 is a potential downstream target
of NR2E3
Using genome-wide chromatin immunoprecipitation
assays and publicly available database analysis (NCI-60 database),
Park et al (5) previously
showed that NR2E3 can directly regulate the expression of the ERα
(esr1) gene. In addition to ESR1, NR2E3 also has the highest
number of correlated genes encoding nuclear receptors, such as
peroxisome proliferator activated receptor α (PPARA), thyroid
hormone receptor α (THRA), estrogen related receptor α (ESRRA),
hepatocyte nuclear factor 4α (HNF4A) and NR2C2, suggesting that
there may be further cross-talk between NR2E3 and these nuclear
receptors (5). In the present
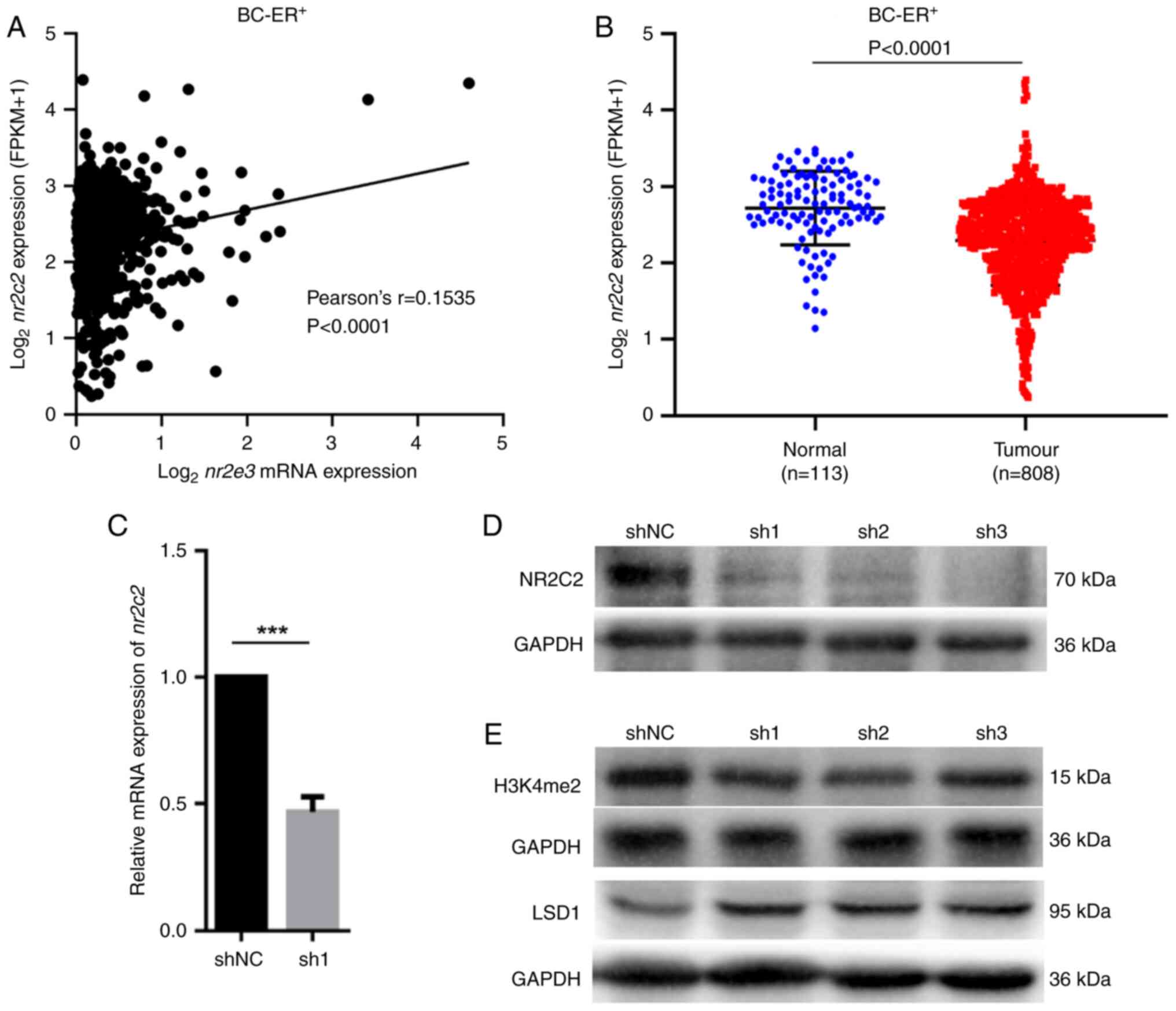
study, using TCGA database, the correlation between mRNA expression
of nr2e3 and ppara, thra, esrra, hnf4a
and nr2c2 in ER+ breast tumor cells were
evaluated, where it was found that there was little or no
correlation between nr2e3 expression and the expression of
ppara, thra, esrra and hnf4a (Fig. S4). By contrast, a significant
positive correlation between the mRNA expression of nr2e3
and nr2c2 was found (Fig.
4A). Compared with that in the normal breast samples,
nr2c2 expression was significantly lower in ER+
breast cancer tissues compared with that in normal tissues
(Fig. 4B).
To determine if NR2E3 can directly regulate
nr2c2 expression, the mRNA and protein levels of
nr2c2 were detected in nr2e3-silenced MCF7 cells. It
was shown that nr2e3 silencing markedly reduced nr2c2
expression (Fig. 4C and D). JASPAR (https://jaspar.genereg.net) was used to scan the 2 kb
promoter sequence upstream from the transcriptional start site of
the nr2c2 gene to identify an NR2E3 binding site. In total,
two predicted NR2E3 binding sites were identified on the proximal
promoter of the nr2c2 gene (-1748 to -1742 bp, and -1516 to
-1510 bp), with the predicted binding sequence being xAAGCTT (x
represents nucleotide A, T, C or G; Fig. S5). This suggested that NR2E3 can
directly bind to the nr2c2 promoter to regulate its
transcription. Furthermore, nr2e3 knockdown was found to
decrease the expression levels of the active histone marker histone
H3 lysine 4 dimethylation (H3K4me2), in addition to markedly
increasing the protein expression level of lysine-specific histone
demethylase 1A (lsd1) (Fig. 4E),
which usually retains the suppressive histone status (9). These results suggest that NR2E3 may
also serve a role as an epigenetic modification factor that can
sustain nr2c2 promoter chromatin accessibility. Taken
together, these findings suggest that the orphan nuclear receptor
NR2C2 may be implicated in the NR2E3 signaling pathway upstream of
the regulation of ER+ breast cancer cell physiology.
Nr2c2 overexpression decreases the
proportion of CD44+CD2-/low cells and
suppresses migratory activity
To determine if NR2E3 can modulate the stem-like
characteristics of ER+ breast carcinoma cells through
NR2C2, vectors containing the full-length coding sequence of the
human nr2c2 cDNA were transfected into the
nr2e3-silenced MCF7 cells. The protein expression levels of
nr2c2 were found to be increased following nr2c2
overexpression (Fig. S6). It was
then observed that nr2c2 overexpression reversed the
elevated ratio of both CD44+CD24-/low cells
and the increased number of migratory cells caused by nr2e3
silencing (Fig. 5A-C). These
findings suggest that an NR2E3/NR2C2 network can modulate the
stem-like activities of ER+ breast tumor cells.
Discussion
Nr2e3 was initially thought to be uniquely
expressed in the retinal photoreceptor cells, it was therefore also
called the photoreceptor-specific nuclear receptor (23). However, nr2e3 has also been
reported to be expressed in other tissues, such as liver,
mammary-glands, adrenal gland, thyroid gland, prostate, testis,
uterus, trachea, digestive tract and salivary glands (4,24,25).
In addition, its expression has been reported in several cancer
cell lines, including the Y79, HepG2, MCF7, T47D, HeLa and HCT116.
Nr2e3 expression has been associated with the occurrence,
progression and drug sensitivity depending on the cancer type
(5-9,26,27).
Proteins specifically designed for the development
of photoreceptors in the retina are employed to direct the
proliferation of tumor cells. Neuroretinal leucine zipper protein
and cone-rod homeobox transcription factor, two pivotal
transcription factors that can form functional complexes with NR2E3
to regulate photoreceptor differentiation, are closely associated
with the growth of the medulloblastoma (28). In patients with liver cancer and
ER+ breast carcinoma, high levels of nr2e3
expression are associated with favorable clinical outcomes and
higher sensitivity to tamoxifen treatment (5,9). In
ER- breast cancer, nr2e3 overexpression has been
previously found to induce migration and metastasis (6), suggesting that NR2E3 serves a
tumorigenic and antineoplastic function influenced by the molecular
environment.
In the present study, nr2e3 expression was
found to be increased in ER+ breast cancer tissues and
cell lines, which is consistent with previously reported data from
TaqMan PCR assays and data re-elaboration (4,17).
In the present study, nr2e3 silencing promoted EMT
progression, increased the ratio of
CD44+CD24-/low cells and promoted the
expression of stem cell-related transcription factors. By contrast,
knocking down nr2e3 expression enhanced the ability of
migration, invasion and colony formation of ER+ MCF7
cells. Data in the present study also verified that nr2e3
expression is inversely associated with the stem-like properties of
ER+ breast tumor cells. In addition, changes to the
stem-like properties of the MCF7 cells appeared to be in part
mediated by the regulation of nr2c2 expression. Therefore,
the present study provided a novel finding that the NR2E3/NR2C2
nuclear receptor network can modulate the physiological behaviors
of breast cancer cells.
In retinal cells, NR2E3 mediates the expression of
photoreceptor genes such as rhodopsin and gnat1 on the
transcriptional level (29). In
tumor cells, NR2E3 can function as an epigenetic modulator to
regulate the chromatin accessibility of target genes, such as
esr1, aryl hydrocarbon receptor and long non-coding RNA
damage-induced noncoding (3,9,30).
NR2E3 can also regulate protein activity through post-translational
modifications. NR2E3 has been reported to enhance the stability of
p53 proteins by increasing acetylation, thereby strengthening p53
signaling (27), These results
suggest that NR2E3 can modulate signal transduction on
pre-transcriptional, transcriptional and post-translational
levels.
The expression of nr2e3 and nr2c2
mRNA was positively correlated in ER+ breast carcinoma,
although not as high as the correlation between nr2e3 and
esr1 (5). In the present
study, nr2e3 knockdown markedly downregulated the mRNA and
protein expression of nr2c2. Mechanistically, several
predicted binding sequences (for example, xAAGCTT) of NR2E3 were
predicted at the proximal promoter of the nr2c2 gene (-1748
to -1742 bp, and -1516 to -1510 bp), suggesting that NR2E3 may
directly activate nr2c2 transcription. nr2c1, a
homologous gene that is associated with nr2c2 and with high
degrees of sequence homology (the overall structural identity is
65%, and the DNA binding domain is 82%), was previously identified
as a direct target of NR2E3 (31,32).
The consensus sequence AAGTCA recognized by NR2E3 proteins in
retinal photoreceptors is also present on the nr2c2 promoter
(33). Further studies to
determine the binding sequences of NR2E3 in ER+ breast
cancer cells are warranted using specific antibodies in chromatin
immunoprecipitation experiments. Another potential mechanism by
which NR2E3 can regulates nr2c2 expression could be by the
modulation of chromatin accessibility, since nr2e3 knockdown
was found to increase the expression of LSD1 whilst decreasing that
of the active histone marker H3K4me2, consistent with previous
findings (3,9,30).
Nr2c2 is ubiquitously expressed in the human
brain, lung, kidney, skeletal muscle, prostate, ovary and testis,
where they serve as a factor in neuronal development, glucose
metabolism, hematogenesis and spermatogenesis (34). Since it is abundantly expressed in
testicular tissues, it is also called testicular orphan nuclear
receptor 4(35). Depending on the
tumor type, NR2C2 may function as a tumorigenic or
tumor-suppressive factor. In prostatic carcinoma, non-small-cell
lung carcinoma and malignant neuroglioma, NR2C2 was found to
enhance the migratory and infiltrative capabilities of tumor cells
(35-37).
In hepatocellular carcinoma and bladder cancer, the opposite effect
is observed (38,39). Consistent with a previously
reported TaqMan array analysis (17), the present study showed that
nr2c2 was expressed at lower levels in ER+ breast
tissues. In ER+ breast carcinoma, NR2C2 breaks the ER
homodimers by binding to monomeric ESR1, thereby reducing cell
proliferation (40). In addition,
NR2C2 can alter the oxygen state of MCF7 cells by decreasing the
expression of oncogenic microRNAs (miR)-526b and miR-655, which
then suppresses tumor migration and invasion (41). These data suggest that NR2C2 may
inhibit the tumorigenicity of ER+ breast cancer
cells.
The molecular mechanism underlying the
NR2E3-mediated regulation of the characteristics of ER+
breast cancer cells can be complex. In addition to the
aforementioned NR2C2, NR2E3 can enhances esr1 transcription
by interacting with protein inhibitor of activated STAT protein 3
(PIAS3), a representative inhibitor of STAT3 (5,42).
Although ESR1 functions in cancer progression (43,44),
its high expression has been associated with superior
recurrence-free survival in ER+ breast cancer (5). Furthermore, patients with higher
levels of expression of both nr2e3 and esr1 tended to
show the optimal recurrence-free survival (5). Esr1 expression was no longer
associated with prognosis when patients were treated with
tamoxifen. However, nr2e3 expression was still relevant
(5), suggesting that NR2E3 can
modulates the characteristics of breast tumor cells through
distinct pathways in patients who received hormonal therapy. PIAS3
acts as an essential protein that recruits NR2E3 to the esr1
promoter (5). Although PIAS3 is an
inhibitor of STAT3, a transcription factor that facilitates
self-renewal and metastasis of breast cancer cells (13,14),
ectopic expression of PIAS3 was shown to enhance the proliferation
of MCF7 cells, attenuate the cytotoxicity of tamoxifen and decrease
the survival time of patients with ER+ breast cancer
(42). Therefore, according to the
ER content, further studies are needed to investigate the molecular
association of NR2E3 with these factors.
In conclusion, results from the present study
suggest that nr2e3 expression is inversely associated with
the migratory and invasive capability of ER+ breast
cancer cells. Nr2e3 silencing reinforced the EMT process,
enhanced the expression of stem cell-related transcription factors
and elevated the proportion of CD44+CD24-/low
cells. In addition, NR2E3 may perform its function by targeting
NR2C2. Therefore, NR2E3/NR2C2 signaling may represent a target to
eliminate stem-like cells in this type of breast cancer.
Supplementary Material
(A) Cytotoxicity of sh1, sh2 and sh3
targeting nuclear receptor subfamily 2 group E member 3 to MCF7
cells was detected using the MTT assay. (B) Statistical analysis of
wound healing rate of the data presented in Fig. 2E. Nc, negative control; ns, not
significant; sh, short hairpin RNA. *P<0.05 and
**P<0.01.
(A) Statistical analysis of colony
formation of the data presented in Fig. 2F. (B) Protein expression levels of
nr2e3 were detected in parental and paclitaxel-resistant
MCF7 cells by western blotting. GAPDH was used as the endogenous
control. NR2E3, nuclear receptor subfamily 2 group E member 3; PR,
paclitaxel-resistant; sh, short hairpin RNA; *P<0.05
and **P<0.01.
Statistical analysis of the relative
protein content of (A) E-cadherin, (B) N-cadherin, (C) VIMENTIN and
(D) SLUG in Fig. 3B.
*P<0.05, **P<0.01 and
***P<0.001. sh, short hairpin RNA.
Evaluation of the correlation of
nr2e3 mRNA expression with the mRNA levels of (A)
thra, (B) esrra, (C) hnf4a and (D)
ppara in ER+ breast adenoma samples. Data was
provided by The Cancer Genome Atlas database. BC, breast cancer;
ER, estrogen receptor; NR2E3, nuclear receptor subfamily 2 group E
member 3; THRA, thyroid hormone receptor α; ESRRA, estrogen related
receptor α; HNF4A, hepatocyte nuclear factor 4αß; PPARA, peroxisome
proliferator activated receptor α.
Predicted binding sites of nuclear
receptor subfamily 2 group E member 3 on the proximal promoter of
the nuclear receptor subfamily 2 group C member 2 gene.
Protein expression of nr2c2 was
detected in parental and nr2c2 over-expressed MCF7 cells
using a western blotting experiment. GAPDH served as the endogenous
control. Untransfected, parental MCF7 cells. NR2C2, nuclear
receptor subfamily 2 group C member 2.
Acknowledgements
Not applicable.
Funding
Funding: The present study was supported by The Key Project of
Anhui Educational Committee (grant nos. KJ2020A0573 and
KJ2021A0748).
Availability of data and materials
The datasets used and/or analyzed during the
current study are available from the corresponding author on
reasonable request.
Authors' contributions
SX was responsible for the study conceptualization,
investigation, and writing and editing of the manuscript. YH was
responsible for performing experiments, writing and reviewing of
the manuscript. JJ was responsible for performing experiments and
methodology carried out. LF was responsible for performing
experiments. CZ was responsible for the methodology and data
analysis. QY was responsible for the project administration and
data interpretation. YN was responsible for the methodology. ZS was
responsible for the conceptualization, funding acquisition and
reviewing of the manuscript. SX and YH confirm the authenticity of
all the raw data. All authors read and approved the final version
of the manuscript.
Ethics approval and consent to
participate
The present study was approved by the Ethics
Committee of Bengbu Medical College (approval no. 2022-138).
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Aísa-Marín I, López-Iniesta MJ, Milla S,
Lillo J, Navarro G, de la Villa P and Marfany G: Nr2e3 functional
domain ablation by CRISPR-Cas9D10A identifies a new isoform and
generates retinitis pigmentosa and enhanced S-cone syndrome models.
Neurobiol Dis. 146(105122)2020.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Li S, Datta S, Brabbit E, Love Z,
Woytowicz V, Flattery K, Capri J, Yao K, Wu S, Imboden M, et al:
Nr2e3 is a genetic modifier that rescues retinal degeneration and
promotes homeostasis in multiple models of retinitis pigmentosa.
Gene Ther. 28:223–241. 2021.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Khanal T, Leung YK, Jiang W, Timchenko N,
Ho SM and Kim K: NR2E3 is a key component in p53 activation by
regulating a long noncoding RNA DINO in acute liver injuries. FASEB
J. 33:8335–8348. 2019.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Garattini E, Bolis M, Paroni G, Fratelli M
and Terao M: Lipid-sensors, enigmatic-orphan and orphan nuclear
receptors as therapeutic targets in breast-cancer. Oncotarget.
7:42661–42682. 2016.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Park YY, Kim K, Kim SB, Hennessy BT, Kim
SM, Park ES, Lim JY, Li J, Lu YL, Gonzalez-Angulo AM, et al:
Reconstruction of nuclear receptor network reveals that NR2E3 is a
novel upstream regulator of ESR1 in breast cancer. EMBO Mol Med.
4:52–67. 2012.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Zhao Z, Wang L and Xu W: IL-13Rα2 mediates
PNR-induced migration and metastasis in ERα-negative breast cancer.
Oncogene. 34:1596–1607. 2015.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Bracci PM, Zhou M, Young S and Wiemels J:
Serum autoantibodies to pancreatic cancer antigens as biomarkers of
pancreatic cancer in a San Francisco Bay Area case-control study.
Cancer. 118:5384–5394. 2012.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Eichen JG, Dalmau J, Demopoulos A, Wade D,
Posner JB and Rosenfeld M: The photoreceptor cell-specific nuclear
receptor is an autoantigen of paraneoplastic retinopathy. J
Neuroophthalmol. 21:168–172. 2001.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Khanal T, Choi K, Leung YK, Wang J, Kim D,
Janakiram V, Cho SG, Puga A, Ho SM and Kim K: Loss of NR2E3
represses AHR by LSD1 reprogramming, is associated with poor
prognosis in liver cancer. Sci Rep. 7(10662)2017.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Zhang L, Chen W, Liu S and Chen C:
Targeting breast cancer stem cells. Int J Biol Sci. 19:552–570.
2023.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Taurin S and Alkhalifa H: Breast cancers,
mammary stem cells, and cancer stem cells, characteristics, and
hypotheses. Neoplasia. 22:663–678. 2020.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Lim JR, Mouawad J, Gorton OK, Bubb WA and
Kwan AH: Cancer stem cell characteristics and their potential as
therapeutic targets. Med Oncol. 38(76)2021.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Rahmati M, Johari B, Kadivar M, Rismani E
and Mortazavi Y: Suppressing the metastatic properties of the
breast cancer cells using STAT3 decoy oligodeoxynucleotides: A
promising approach for eradication of cancer cells by
differentiation therapy. J Cell Physiol. 235:5429–5444.
2020.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Johari B, Rahmati M, Nasehi L, Mortazavi
Y, Faghfoori MH and Rezaeejam H: Evaluation of STAT3 decoy
oligodeoxynucleotides' synergistic effects on radiation and/or
chemotherapy in metastatic breast cancer cell line. Cell Biol Int.
44:2499–2511. 2020.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Johari B and Moradi M: Application of
transcription factor decoy oligodeoxynucleotides (ODNs) for cancer
therapy. Methods Mol Biol. 2521:207–230. 2022.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Bai X, Ni J, Beretov J, Graham P and Li Y:
Cancer stem cell in breast cancer therapeutic resistance. Cancer
Treat Rev. 69:152–163. 2018.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Muscat GEO, Eriksson NA, Byth K, Loi S,
Graham D, Jindal S, Davis MJ, Clyne C, Funder JW, Simpson ER, et
al: Research resource: Nuclear receptors as transcriptome:
discriminant and prognostic value in breast cancer. Mol Endocrinol.
27:350–365. 2013.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Xie S, Han S, Qu Z, Liu F, Li J, Yu S,
Reilly J, Tu J, Liu X, Lu Z, et al: Knockout of Nr2e3 prevents rod
photoreceptor differentiation and leads to selective L-/M-cone
photoreceptor degeneration in zebrafish. Biochim Biophys Acta Mol
Basis Dis. 1865:1273–1283. 2019.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Wang W, Zhang L, Wang Y, Ding Y, Chen T,
Wang Y, Wang H, Li Y, Duan K, Chen S, et al: Involvement of miR-451
in resistance to paclitaxel by regulating YWHAZ in breast cancer.
Cell Death Dis. 8(e3071)2017.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Yan Y, Liu F, Han L, Zhao L, Chen J,
Olopade OL, He M and Wei M: HIF-2α promotes conversion to a stem
cell phenotype and induces chemoresistance in breast cancer cells
by activating Wnt and Notch pathways. J Exp Clin Cancer Res.
37(256)2018.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Gooding AJ and Schiemann WP:
Epithelial-mesenchymal transition programs and cancer stem cell
phenotypes: Mediators of breast cancer therapy resistance. Mol
Cancer Res. 18:1257–1270. 2020.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Recouvreux MV, Moldenhauer MR, Galenkamp
KMO, Jung M, James B, Zhang Y, Lowy A, Bagchi A and Commisso C:
Glutamine depletion regulates Slug to promote EMT and metastasis in
pancreatic cancer. J Exp Med. 217(e20200388)2020.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Venturini G, Kokona D, Steiner BL, Bulla
EG, Jovanovic J, Zinkernagel MS and Escher P: In vivo analysis of
onset and progression of retinal degeneration in the Nr2e3rd7/rd7
mouse model of enhanced S-cone sensitivity syndrome. Sci Rep.
11(19032)2021.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Choi MY, Romer AI, Hu M, Lepourcelet M,
Mechoor A, Yesilaltay A, Krieger M, Gray PA and Shivdasaniet RA: A
dynamic expression survey identifies transcription factors relevant
in mouse digestive tract development. Development. 133:4119–4129.
2006.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Nishimura M, Naito S and Yokoi T:
Tissue-specific mRNA expression profiles of human nuclear receptor
subfamilies. Drug Metab Pharmacokinet. 19:135–149. 2004.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Cai X, Conley SM, Cheng T, Al-Ubaidi MR
and Naash MI: A 350 bp region of the proximal promoter of Rds
drives cell-type specific gene expression. Exp Eye Res. 91:186–194.
2010.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Wen Z, Pyeon D, Wang Y, Lambert P, Xu W
and Ahlquist P: Orphan nuclear receptor PNR/NR2E3 stimulates p53
functions by enhancing p53 acetylation. Mol Cell Biol. 32:26–35.
2012.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Garancher A, Lin CY, Morabito M, Richer W,
Rocques N, Larcher M, Bihannic L, Smith K, Miquel C, Leboucher S,
et al: NRL and CRX define photoreceptor identity and reveal
subgroup-specific dependencies in medulloblastoma. Cancer Cell.
33:435–449.e6. 2018.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Al-Khuzaei S, Broadgate S, Halford S,
Jolly JK, Shanks M, Clouston P and Downes SM: Novel pathogenic
sequence variants in NR2E3 and clinical findings in three patients.
Genes (Basel). 11(1288)2020.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Khanal T, Kim D, Johnson A, Choubey D and
Kim K: Deregulation of NR2E3, an orphan nuclear receptor, by
benzo(a)pyrene-induced oxidative stress is associated with histone
modification status change of the estrogen receptor gene promoter.
Toxicol Lett. 237:228–236. 2015.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Lin SJ, Yang DR, Yang G, Lin CY, Chang HC,
Li G and Chang C: TR2 and TR4 orphan nuclear receptors: An
overview. Curr Top Dev Biol. 125:357–373. 2017.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Haider NB, Mollema N, Gaule M, Yuan Y,
Sachs AJ, Nystuen AM, Naggert JK and Nishina PM: Nr2e3-directed
transcriptional regulation of genes involved in photoreceptor
development and cell-type specific phototransduction. Exp Eye Res.
89:365–372. 2009.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Roduit R, Escher P and Schorderet DF:
Mutations in the DNA-binding domain of NR2E3 affect in vivo
dimerization and interaction with CRX. PLoS One.
4(e7379)2009.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Yao X, Zhang Y, Wu L, Cheng R, Li C, Qu C
and Ji H: Immunohistochemical study of NR2C2, BTG2, TBX19, and CDK2
expression in 31 paired primary/recurrent nonfunctioning pituitary
adenomas. Int J Endocrinol. 2019(5731639)2019.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Zhang L, Zhang J, Ma Y, Chen J, Dong B,
Zhao W, Wang X, Zheng Q, Fang F and Yang Y: Testicular orphan
receptor 4 (TR4) is a marker for metastasis and poor prognosis in
non-small cell lung cancer that drives the EMT phenotype. Lung
Cancer. 89:320–328. 2015.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Qiu X, Zhu J, Sun Y, Fan K, Yang DR, Li G,
Yang G and Chang C: TR4 nuclear receptor increases prostate cancer
invasion via decreasing the miR-373-3p expression to alter
TGFβR2/p-Smad3 signals. Oncotarget. 6:15397–15409. 2015.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Fan Z, Zheng J, Xue Y, Liu X, Wang D, Yang
C, Ma J, Liu L, Ruan X, Wang Z and Liu Y: NR2C2-uORF targeting
UCA1-miR-627-5p-NR2C2 feedback loop to regulate the malignant
behaviors of glioma cells. Cell Death Dis. 9(1165)2018.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Ren W, Hu J, Li H, Chen J, Ding J, Zu X
and Fan B: miR-616-5p promotes invasion and migration of bladder
cancer via downregulating NR2C2 expression. Front Oncol.
11(762946)2021.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Jin R, Lin H, Li G, Xu J, Shi L, Chang C
and Cai X: TR4 nuclear receptor suppresses HCC cell invasion via
downregulating the EphA2 expression. Cell Death Dis.
9(283)2018.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Shyr CR, Hu YC, Kim E and Chang C:
Modulation of estrogen receptor-mediated transactivation by orphan
receptor TR4 in MCF-7 cells. J Biol Chem. 277:14622–14628.
2002.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Gervin E, Shin B, Opperman R, Cullen M,
Feser R, Maiti S and Majumder M: Chemically induced hypoxia
enhances miRNA functions in breast cancer. Cancers (Basel).
12(2008)2020.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Yang SF, Hou MF, Chen FM, Ou-Yang F, Wu
YC, Chai CY and Yeh YT: Prognostic value of protein inhibitor of
activated STAT3 in breast cancer patients receiving hormone
therapy. BMC Cancer. 16(20)2016.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Lei JT, Gou X, Seker S and Ellis MJ: ESR1
alterations and metastasis in estrogen receptor positive breast
cancer. J Cancer Metastasis Treat. 5(38)2019.PubMed/NCBI View Article : Google Scholar
|
|
44
|
Matutino A, Joy AA, Brezden-Masley C, Chia
S and Verma S: Hormone receptor-positive, HER2-negative metastatic
breast cancer: Redrawing the lines. Curr Oncol. 25 (Suppl
1):S131–S141. 2018.PubMed/NCBI View Article : Google Scholar
|