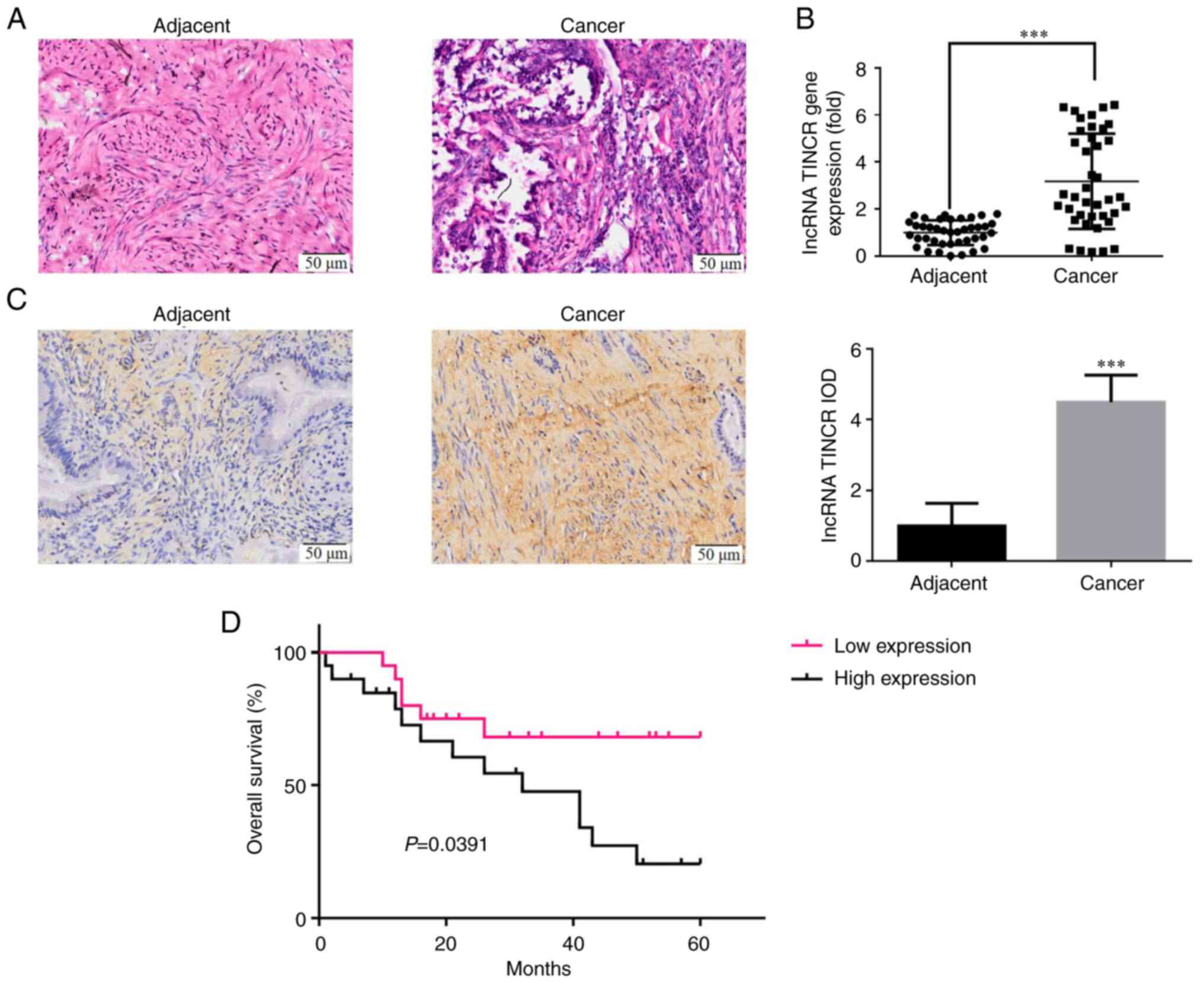
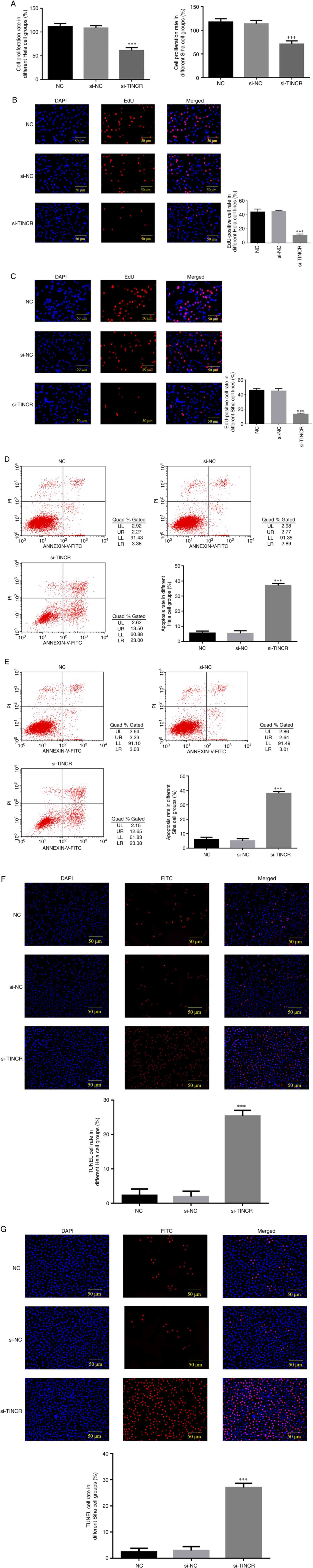
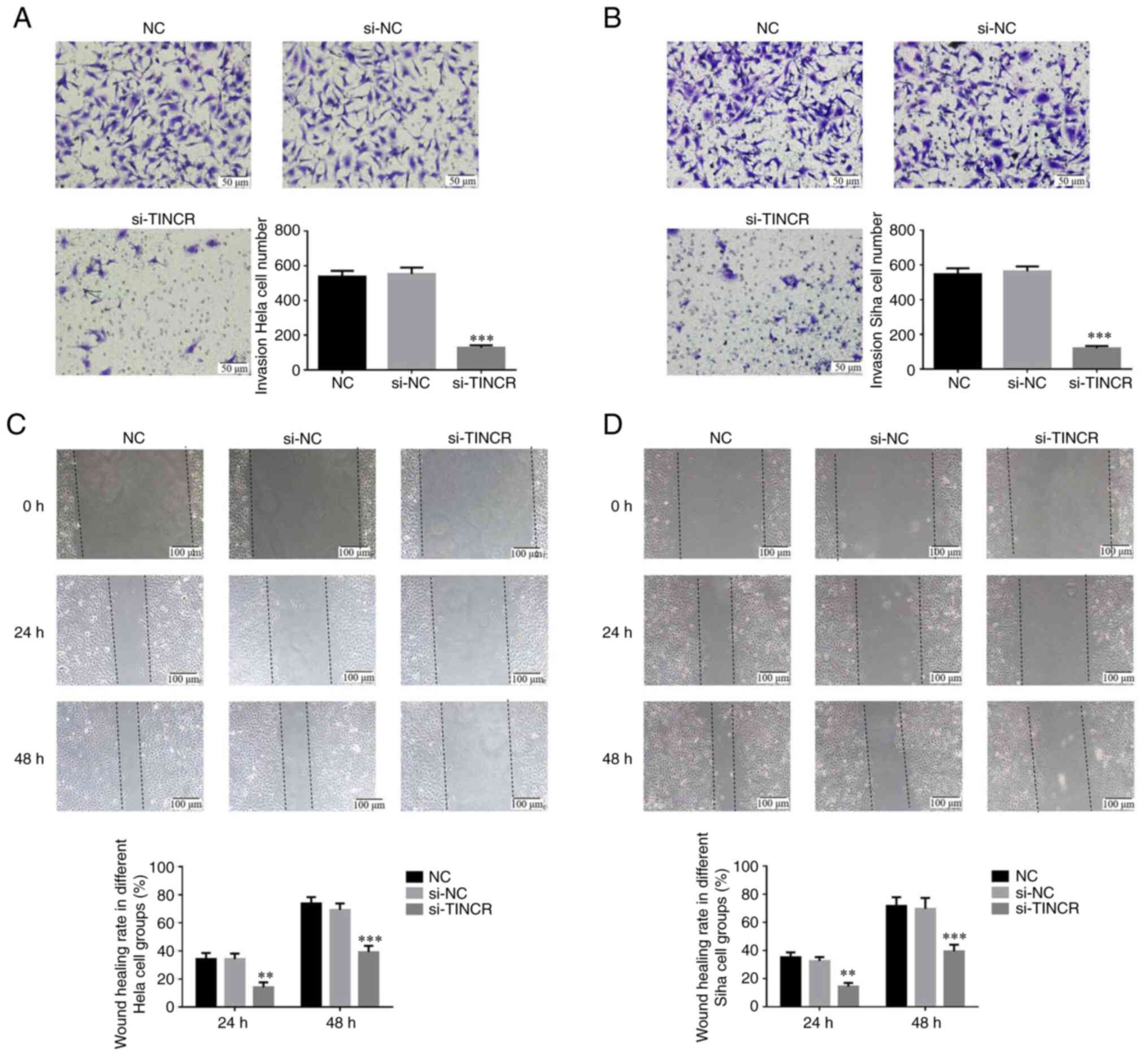
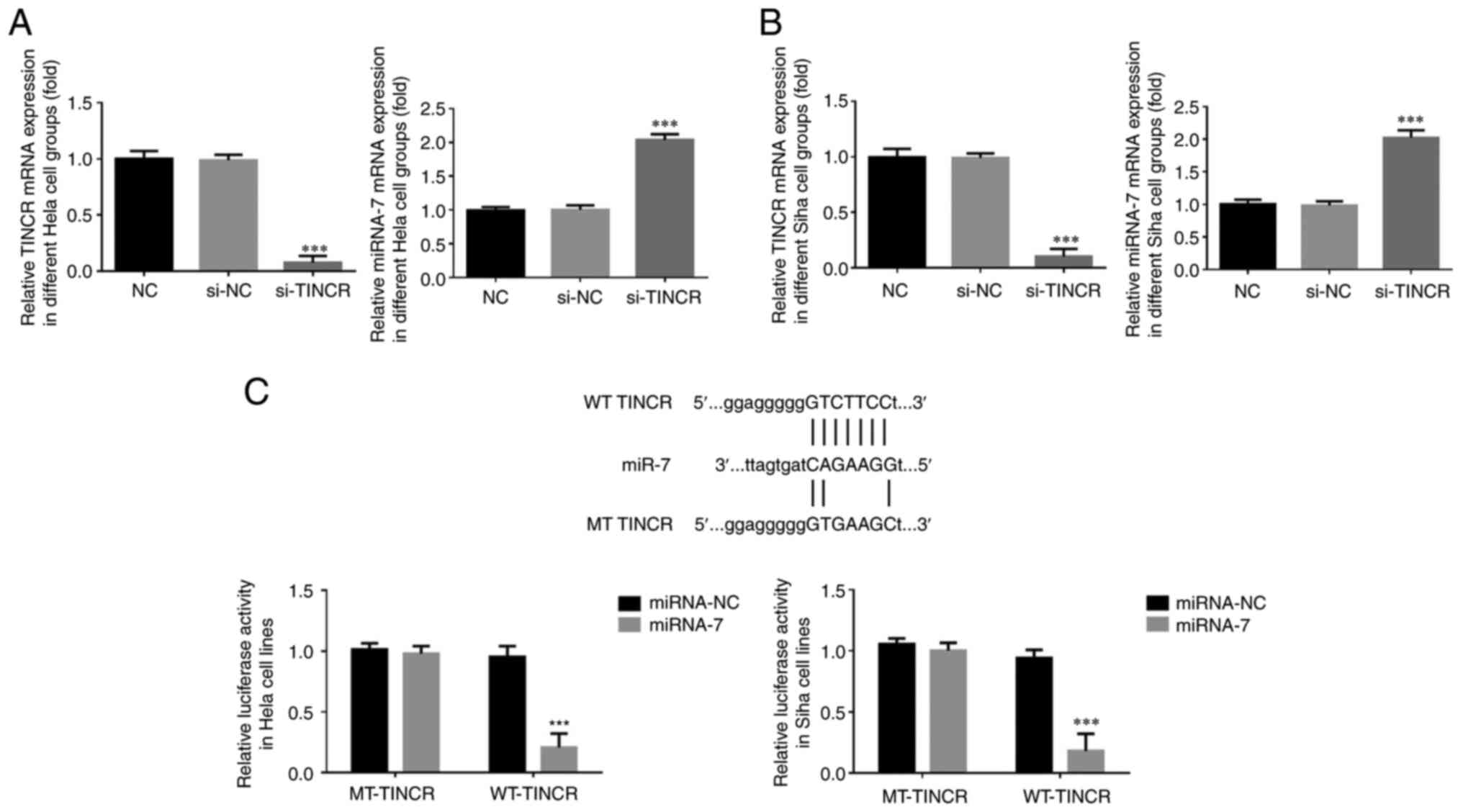
|
1
|
Small W Jr, Bacon MA, Bajaj A, Chuang LT,
Fisher BJ, Harkenrider MM, Jhingran A, Kitchener HC, Mileshkin LR,
Viswanathan AN and Gaffney DK: Cervical cancer: A global health
crisis. Cancer. 123:2404–2412. 2017.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Wardak S: Human Papillomavirus (HPV) and
cervical cancer. Med Dosw Mikrobiol. 68:73–84. 2016.PubMed/NCBI
|
|
3
|
Shah CA, Beck T, Liao JB, Giannakopoulos
NV, Veljovich D and Paley P: Surgical and oncologic outcomes after
robotic radical hysterectomy as compared to open radical
hysterectomy in the treatment of early cervical cancer. J Gynecol
Oncol. 28(e82)2017.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Schmitz SU, Grote P and Herrmann BG:
Mechanisms of long non-coding RNA function in development and
disease. Cell Mol Life Sci. 73:2491–2509. 2016.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Dong L, Ding H, Li Y, Xue D and Liu Y:
LncRNA TINCR is associated with clinical progression and serves as
tumor suppressive role in prostate cancer. Cancer Manag Res.
10:2799–2807. 2018.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Zhu ZJ and He JK: TINCR facilitates
non-small cell lung cancer progression through BRAF-activated MAPK
pathway. Biochem Biophys Res Commun. 497:971–977. 2018.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Tian F, Xu J, Xue F, Guan E and Xu X:
TINCR expression is associated with unfavorable prognosis in
patients with hepatocellular carcinoma. Biosci Rep.
37(BSR20170301)2017.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Xu J, Zeng W, Liu T, Wan Z, Yang X, Chen J
and Liu F: lncRNA TINCR knockdown inhibits colon cancer cells via
regulation of autophagy. Food Sci Nutr. 11:1965–1981.
2023.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Luo H, Xu C, Le W, Ge B and Wang T: lncRNA
CASC11 promotes cancer cell proliferation in bladder cancer through
miRNA-150. J Cell Biochem. 120:13487–13493. 2019.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Luan X and Wang Y: LncRNA XLOC_006390
facilitates cervical cancer tumorigenesis and metastasis as a ceRNA
against miR-331-3p and miR-338-3p. J Gynecol Oncol.
29(e95)2018.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Zhao W, Geng D, Li S, Chen Z and Sun M:
LncRNA HOTAIR influences cell growth, migration, invasion, and
apoptosis via the miR-20a-5p/HMGA2 axis in breast cancer. Cancer
Med. 7:842–855. 2018.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Peng CL, Zhao XJ, Wei CC and Wu JW: LncRNA
HOTAIR promotes colon cancer development by down-regulating
miRNA-34a. Eur Rev Med Pharmacol Sci. 23:5752–5761. 2019.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Thiel J, Alter C, Luppus S, Eckstein A,
Tan S, Führer D, Pastille E, Westendorf AM, Buer J and Hansen W:
MicroRNA-183 and microRNA-96 are associated with autoimmune
responses by regulating T cell activation. J Autoimmun. 96:94–103.
2019.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Kara M, Yumrutas O, Ozcan O, Celik OI,
Bozgeyik E, Bozgeyik I and Tasdemir S: Differential expressions of
cancer-associated genes and their regulatory miRNAs in colorectal
carcinoma. Gene. 567:81–86. 2015.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Yang Y, Li XJ, Li P and Guo XT:
MicroRNA-145 regulates the proliferation, migration and invasion of
human primary colon adenocarcinoma cells by targeting MAPK1. Int J
Mol Med. 42:3171–3180. 2018.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Zeng CY, Zhan YS, Huang J and Chen YX:
MicroRNA-7 suppresses human colon cancer invasion and proliferation
by targeting the expression of focal adhesion kinase. Mol Med Rep.
13:1297–1303. 2016.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Xu N, Lian YJ, Dai X and Wang YJ: MiR-7
increases cisplatin sensitivity of gastric cancer cells through
suppressing mTOR. Technol Cancer Res Treat. 16:1022–1030.
2017.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Chen WS, Yen CJ, Chen YJ, Chen JY, Wang
LY, Chiu SJ, Shih WL, Ho CY, Wei TT, Pan HL, et al: MiRNA-7/21/107
contribute to HBx-induced hepatocellular carcinoma progression
through suppression of maspin. Oncotarget. 6:25962–25974.
2015.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408.
2001.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Tang Y, Cheung BB, Atmadibrata B, Marshall
GM, Dinger ME, Liu PY and Liu T: The regulatory role of long
non-coding RNAs in cancer. Cancer Lett. 391:12–19. 2017.PubMed/NCBI View Article : Google Scholar
|
|
21
|
McCabe EM and Rasmussen TP: lncRNA
involvement in cancer stem cell function and epithelial-mesenchymal
transitions. Semin Cancer Biol. 75:38–48. 2021.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Morgado-Palacin L, Brown JA, Martinez TF,
Garcia-Pedrero JM, Forouhar F, Quinn SA, Reglero C, Vaughan J,
Heydary YH, Donaldson C, et al: The TINCR ubiquitin-like
microprotein is a tumor suppressor in squamous cell carcinoma. Nat
Commun. 14(1328)2023.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Zhang X, Yao J, Shi H, Gao B and Zhang L:
LncRNA TINCR/microRNA-107/CD36 regulates cell proliferation and
apoptosis in colorectal cancer via PPAR signaling pathway based on
bioinformatics analysis. Biol Chem. 400:663–675. 2019.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Zhu X, Li H, Wu Y, Zhou J, Yang G and Wang
W: LncRNA MEG3 promotes hepatic insulin resistance by serving as a
competing endogenous RNA of miR-214 to regulate ATF4. Int J Mol
Med. 43:345–357. 2019.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Sehgal SN: Sirolimus: Its discovery,
biological properties, and mechanism of action. Transplant Proc. 35
(3 Suppl):7S–14S. 2003.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Wiederrecht GJ, Sabers CJ, Brunn GJ,
Martin MM, Dumont FJ and Abraham RT: Mechanism of action of
rapamycin: New insights into the regulation of G1-phase progression
in eukaryotic cells. Prog Cell Cycle Res. 1:53–71. 1995.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Edinger AL and Thompson CB: Akt maintains
cell size and survival by increasing mTOR-dependent nutrient
uptake. Mol Biol Cell. 13:2276–2288. 2002.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Semenza GL: HIF-1 mediates metabolic
responses to intratumoral hypoxia and oncogenic mutations. J Clin
Invest. 123:3664–3671. 2013.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Ryan HE, Lo J and Johnson RS: HIF-1 alpha
is required for solid tumor formation and embryonic
vascularization. EMBO J. 17:3005–3015. 1998.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Li L, Qu Y, Li J, Xiong Y, Mao M and Mu D:
Relationship between HIF-1alpha expression and neuronal apoptosis
in neonatal rats with hypoxia-ischemia brain injury. Brain Res.
1180:133–139. 2007.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Chen Y, Peng GF, Han XZ, Wang W, Zhang GQ
and Li X: Apoptosis prediction via inhibition of AKT signaling
pathway by neogrifolin. Int J Clin Exp Pathol. 8:1154–1164.
2015.PubMed/NCBI
|
|
32
|
Kigure W, Fujii T, Sutoh T, Morita H,
Katoh T, Yajima RN, Yamaguchi S, Tsutsumi S, Asao T and Kuwano H:
The association of VEGF-C expression with tumor lymphatic vessel
density and lymph node metastasis in patients with gastric cancer
and gastrointestinal stromal tumor. Hepatogastroenterology.
60:277–280. 2013.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Lugano R, Ramachandran M and Dimberg A:
Tumor angiogenesis: Causes, consequences, challenges and
opportunities. Cell Mol Life Sci. 77:1745–1770. 2020.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Li W, Petrimpol M, Molle KD, Hall MN,
Battegay EJ and Humar R: Hypoxia-induced endothelial proliferation
requires both mTORC1 and mTORC2. Circ Res. 100:79–87.
2007.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Tsai SY, Yang LY, Wu CH, Chang SF, Hsu CY,
Wei CP, Leu SJ, Liaw J, Lee YH and Tsai MD: Injury-induced Janus
kinase/protein kinase C-dependent phosphorylation of
growth-associated protein 43 and signal transducer and activator of
transcription 3 for neurite growth in dorsal root ganglion. J
Neurosci Res. 85:321–331. 2007.PubMed/NCBI View Article : Google Scholar
|