1. Introduction
Polymyalgia rheumatica (PMR) is a chronic
inflammatory disease which affects the connective vascular tissue,
characterized by pain and accompanied by morning stiffness,
predominantly of the neck muscles, hip and shoulder girdle. The
main characteristics included in the majority of definitions are
pain and morning stiffness of the hip and shoulder girdle and/or
the neck muscles, lasting for >30 min, with a disease onset of
>1 month. Usually, patients are aged >50 years and the
biological inflammatory syndrome is present, with an increase in
both the erythrocyte sedimentation rate (ESR) and C-reactive
protein (CRP) levels, aspects similar to giant cell arteritis (GCA)
(1).
PMR predominantly affects the elderly, and the
median age of disease onset is 73 years. The prevalence is
estimated at 700/100.000 individuals aged >50 years. The
incidence increases with age and varies depending on the
geographical region, with an increased incidence observed in
Scandinavian countries. The disease affects females 2-3-fold more
frequently than males, as well as individuals of Caucasian
ethnicity, as compared with Asian, Latin-American and
African-American populations (2).
PMR is frequently associated with GCA in ~30% of
cases. From a clinical point of view, 40-60% of patients with GCA
can present with symptoms of PMR at the time of diagnosis. PMR and
GCA bear multiple similarities, including age at disease onset, an
increased prevalence among females and geographical distribution,
suggesting that these clinical entities may represent subtypes of
the same pathology (3).
The aim of the present review was to depict the
current pathogenic hypothesis, the diagnostic and treatment
approaches for PMR patients, and novelties since the development of
the currently used 2012 European League Against Rheumatism (EULAR)
and American College of Rheumatology (ACR) provisional
classification criteria.
2. Pathogenesis
To date, the etiology and pathogenesis of PMR are
not clearly understood. This can be attributed to earlier studies,
which were conducted in mixed cohorts presenting with both PMR and
GCA, impeding the successful evaluation of the patterns involved in
the pathogenesis of isolated PMR, as reviewed by Guggino et
al (4).
HLA-DRB1*04 allele is usually associated
with PMR in conjunction with GCA. However, when assessing genotypes
and susceptibility to PMR alone, the data presented in literature
is controversial. Salvarani et al (5) revealed a high incidence of
HLA-DRB1*04 alleles in a cohort of patients of Italian
descent with ‘pure’ PMR. Furthermore, Gonzalez-Gay et al
(6) described an association of
HLA-DRB1*04 with more severe disease activity and
increased synovial inflammation in patients with PMR from a patient
cohort of Spanish descent.
Since PMR is associated with inflammation of the
bursae, the cytokines implicated in the inflammatory process may be
responsible for some of the pathogenic traits of this disease. It
has been suggested that PMR is associated with various TNF
polymorphisms. Also, higher levels of interleukin (IL)-1, IL-6 and
intercellular adhesion molecule-1 (ICAM-1) have been associated
with increased risk of disease development or increased disease
severity in PMR (7).
The role of infectious and environmental factors has
been postulated in PMR pathogenesis. Among several of the
investigated infectious factors, Mycoplasma pneumoniae,
parvovirus B19 and Chlamydia pneumoniae have been more
frequently incriminated in the development of PMR (8-10).
Additionally, Cimmino and Zaccaria (11) indicated that antibodies to
adenovirus and respiratory syncytial virus may also trigger PMR,
due to their high prevalence in the bloodstream of PMR
patients.
Another factor involved in the development of PMR is
the use of immune check-point inhibitors in cancer patients, due to
their antagonizing effect on cytotoxic T lymphocyte associated
antigen-4 (CTLA-4) and programmed death cell protein 1 (PD-1). The
therapeutics that have been implicated in the pathogenesis of PMR
are ipilimumab, nivolumab and pembrolizumab (12).
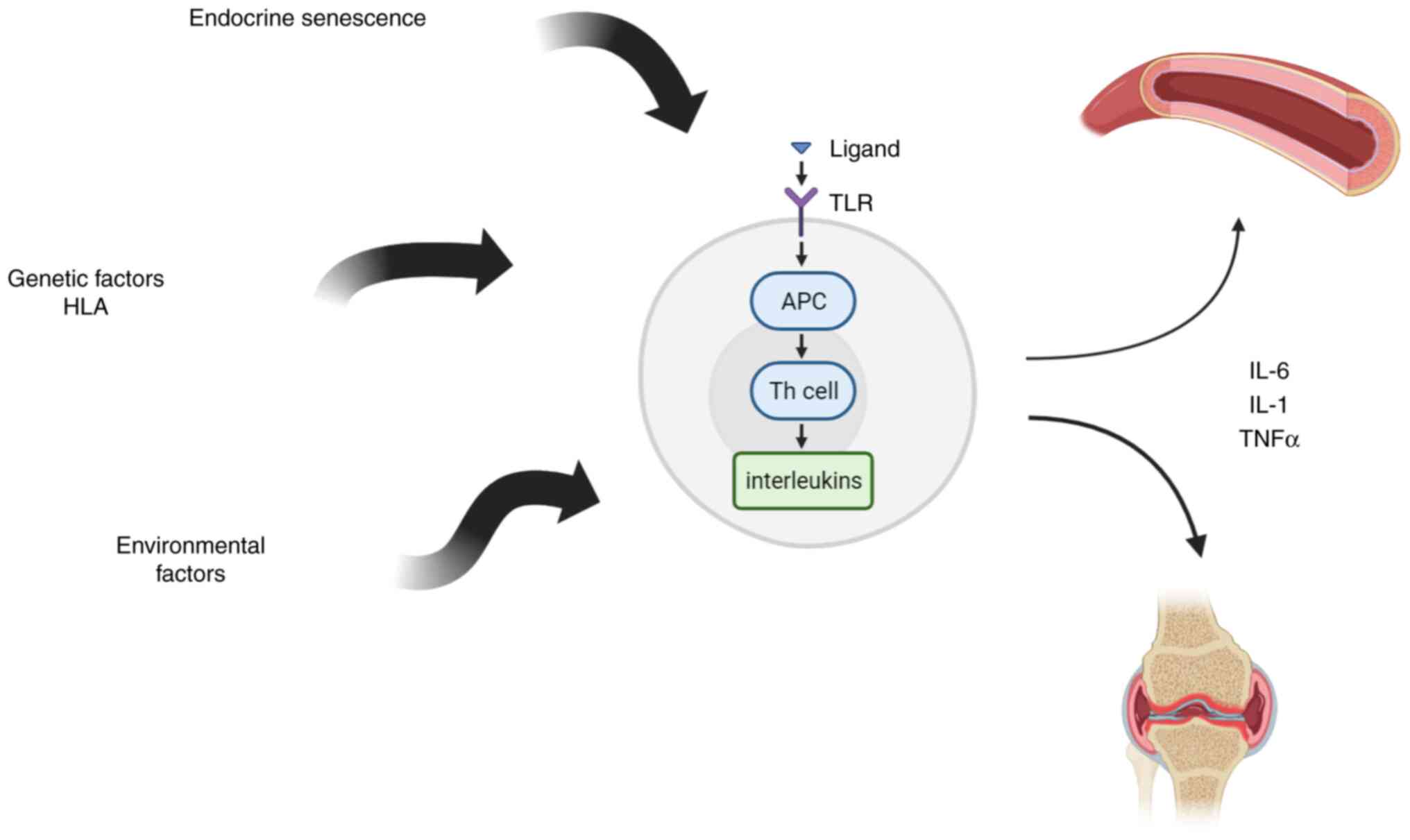
The pathophysiology of PMR may entail an abnormal
immune response, particularly one involving T cells. An increase in
T-helper 17 (Th17) cells was observed in a group of individuals
with PMR and/or GCA, and a concurrent decrease was also discovered
in regulatory T cells. Also, an increase of memory-effector T cells
was noted, revealing an alteration in T-cell subpopulation no
longer expressing the co-stimulatory molecule
(CD4+CD28- and
CD8+CD28-). These subtypes of T cells are
known to be increased in elderl; however, as compared to sex and
age-matched controls, the observed levels of T cells were increased
in those with PMR/GCA. Memory-effector T lymphocytes contribute to
the pro-inflammatory cascade in PMR, due to the ability to produce
interferon (IFN)-γ and tumor necrosis factor (TNF)-α in large
quantities (13,14) (Fig.
1).
IL-17 has also been recently linked to PMR and GCA,
due to the subsequently incited activation of Th17 responses.
Additionally, there is a correlation between higher IL-6 levels and
PMR disease activity. IL-6 inhibitors are presently being trialed
for the treatment of PMR after demonstrating effectiveness in GCA
(15).
van der Geest et al (16) also demonstrated a decrease in the
numbers of B lymphocytes which presented an inverse association
with ESR, CRP, and B-cell activating factor (BAFF) levels.
Pro-inflammatory cytokines could be markedly
implicated in PMR pathogenesis as well. When comparing symptomatic
vastus lateralis and trapezius muscles of PMR patients to healthy
individuals included as a control, higher interstitial
concentrations of IL-1α, IL-1β, IL-1 receptor antagonist, IL-6,
IL-8, TNF-α, and monocyte chemoattractant protein-1 have been
detected in the serum of the PMR population. The etiology of the
illness may thus be influenced by the elevated interstitial
concentrations of pro-inflammatory cytokines in the affected
muscles (17,18). JAK/STAT signaling pathway has been
studied in GCA and PMR. The inhibition of JAK 1 and JAK2 may lead
to the downregulation of Th1 and Th17 pathways and also
IL-6(19).
The clinical symptoms of PMR may be attributed to
immune cell infiltration in the muscles and periarticular areas.
Patients with PMR and GCA were demonstrated to present immune
complexes in their muscles. PMR has also been associated with
synovitis. In comparison to healthy individual controls, deltoid
muscle biopsies from patients with PMR exhibited increased
microvascularization. The evaluation of arthroscopic samples used
to study synovitis of the shoulder, it was revealed that only
macrophages and T cells, infiltrate the extracted fragments,
whereas B cells, NK cells, or γ/δ T cells were not detected
(20,21). A strong adhesion molecule
expression, including vascular cell adhesion molecule (VCAM)-1 and
ICAM-1, has been observed in PMR subjects with synovitis and may be
important for the recruitment of several immune system components
in PMR synovial infiltration (22).
Also, the process of endocrine senescence that
produces decreased levels of dehydropiandrosterone and alterations
of hypothalamic-pituitary-gonadal axis with adrenal cortex
insufficiency and decreased cortisol secretion in response to the
inflammatory status was incriminated as an etiopathogenically
important mechanism (23).
Deregulation of the immune system may result in a
vicious cycle, with the immune system remaining activated and in a
permanent state of inflammation, as is frequently observed in
inflammatory autoimmune diseases. However, it should also be
considered that chronic inflammation gradually causes dysregulation
of the immune system (24).
The immune system dysregulation may lead to a higher
risk of cancer occurrence in PMR patients. Nevertheless, the data
in literature regarding the development of cancer in PMR patients
is controversial. According to a follow-up study in Sweden, there
was discovered a link between malignant diseases and PMR.
Furthermore, some specific types of cancers such as skin cancers
and hematologic malignancies such as acute myeloid leukemia,
multiple myeloma or myeloproliferative diseases have been
associated with PMR (25). A more
recent study on 80 patients diagnosed with PMR, which were observed
for >40 weeks and screened using positron emission
tomography/computed tomography (PET/CT) revealed a higher
prevalence of cancer in PMR patients, in comparison to the general
population (26). Also, the
treatment for cancer such as immune checkpoint inhibitors may
trigger PMR (27).
3. Clinical manifestations
PMR manifests in patients >50 years of age,
leading to discomfort, a reduced range of motion and stiffness of
the shoulder girdle may, which is a fundamental clinical hallmark
of PMR. Furthermore, neck, hip girdle and thigh symptoms may also
occur. Patients also frequently complain about difficulties in
movement, with the symptoms being bilateral in most cases (28).
In total, up to 40-50% of patients may exhibit
established symptoms, including low-grade fever, lethargy,
asthenia, anorexia and weight loss. In some cases, the first sign
of isolated PMR is a fever >38˚C (3).
The onset of symptoms is frequently unforeseen,
occurring typically within a few days; however, in rare cases,
symptoms may develop suddenly overnight. Aching and early morning
stiffness lasting for >30 min are two of the most common
symptoms occurring in the musculoskeletal areas that are involved
in the inflammatory process. The symptoms of inflammatory pain and
stiffness are often most aggravated in the morning, gradually
improving during the course of the day, and then relapsing to their
baseline level after the patient has rested or has been inactive
(also known as ‘gelling’) for an extended period of time.
Hip girdle symptoms are described as pain in the
groin area and lateral sides of the hip and often radiate to the
posterior thigh region (29).
Performing tasks necessary for the activities of
daily living, including clothing, combing hair, getting out of bed,
or getting up from a chair are examples of actions that become
challenging and are often coupled with debilitating pain. Nocturnal
pain is also common, and patients frequently face difficulties in
falling or staying asleep (30).
At disease onset, symptoms may be unilateral,
rapidly becoming symmetrical and bilateral. During a physical
examination, the active mobility of a patient, particularly
concerning the abduction of the shoulders, may be restricted due to
tenderness. In addition, there is no clinically apparent joint
swelling. A passive range of motion that is facilitated by the
examiner may, in certain cases, approximate a healthy phenotype.
The discomfort in the shoulder is widespread, and it is not
localized in specific shoulder structures (31).
Under normal circumstances, a painful limitation in
the active range of motion of the neck and hips also occurs. Even
though pain in the muscles manifests, it is not typical for the
muscles to exhibit any weakness, despite the existence of muscular
discomfort (32).
There is a possibility that other joint symptoms may
also be present. Clinical signs of peripheral synovial inflammation
can be observed in various cases, approximating to ≤23-39% of
patients. Arthritis is characterized by asymmetrical presentation,
a non-erosive character, mainly affecting the knees and wrists.
Following the initially administered therapy with glucocorticoids
(GCs), the symptoms appear to subside in the majority of patients.
Inflammation of the periarticular structures, including tendons and
bursae, may also be present in patients with peripheral synovitis.
Tenosynovitis and bursitis can be evidenced by musculoskeletal
ultrasound (MSUS) and other imaging techniques, including magnetic
resonance imaging (MRI). It has been reported that ~15% of patients
with PMR exhibit ultrasonographic evidence of carpal tunnel
syndrome, and 3% of patients have been reported to exhibit distal
tenosynovitis (33).
PMR may, in certain instances, manifest clinically
as distal swelling and edema, which may be analogous to the
symptoms experienced by individuals diagnosed with remitting
seronegative symmetrical synovitis with pitting edema syndrome
(34).
4. Laboratory features
Laboratory analyses are non-specific. The increase
in acute phase reactants is dominant from a paraclinical point of
view, with values of ESR that vary from moderate to high, often
>100 mm/h, with <20% of patients presenting with values below
40 mm/h. By contrast, CRP levels are constantly increased,
representing a reliable inflammatory monitoring marker, normal
values being incompatible with the diagnosis of PMR (35,36).
The study by Cantini et al which evaluated 177 patients with
PMR, revealed that 6% of the patients presented with normal ESR
values at the time of diagnosis, while CRP levels were normal in
only 1% of cases. Even in several cases of relapse, ESR levels were
normal in 68% of cases, whereas CRP levels were elevated in 62% of
cases (37).
Increased levels of markers of non-specific
inflammation, including α2 and α1-globulins, gamma globulins,
fibrinogen, α1-antitrypsin, α1-antichemotrypsin and haptoglobin may
also be detected (38).
Blood count changes indicate an inflammatory
biological profile with the presence of mild or moderate normocytic
normochromic anemia, reactive leukocytosis or thrombocytosis
(38).
Rheumatoid factors (RFs), anticitrullinated protein
antibodies and antinuclear antibodies are usually absent. However,
a weak positivity of RFs must be considered in ~10% of elderly
population, without any clinical significance (39).
On occasion, anticardiolipin antibodies may be
detected in increased titers as an independent predictive marker
for the risk of vascular complications (40).
Indications of hepatic damage are often present with
increases in the levels of alkaline phosphatase, γ-glutamyl
transpeptidase, 5'-nucleotidase, and occasionally, moderate
increases in transaminase levels. Serum levels of creatine kinase
and lactate dehydrogenase are within a normal range and exclude
myositis-type involvement. IL-6 and von Willebrand factor levels
are increased, with significant decreases following treatment
administration (36,41).
The examination of the synovial fluid may reveal
mild inflammation, including an increase in the total number of
leukocytes with levels ≤20,000/mm3, 40-50% of which
being polymorphonuclear (42).
Also, neuropeptides such as vasoactive intestinal peptide were
found in the synovial fluid of PMR patients which may contribute to
the immunomodulation of synovial fluid inflammation, but also
extraarticular manifestations such as cardiac rhythm dysregulations
(43,44).
5. Imaging
Probably the most abundantly available data and type
of evidence in the literature over the past decade have been
acquired through musculoskeletal imaging, with applications in
diagnosis, disease monitoring and relapse, prognosis and change
with treatment. Currently, none of the various sets of
classification criteria for PMR are fully validated in clinical
practice. The simultaneous presence of inflammation in articular
and periarticular structures of the bilateral shoulder or in one
shoulder and the hips, as identified by ultrasound, aided in the
improvement of the sensitivity and specificity of the clinical
criteria in 2012, with the introduction of the ultrasound criteria.
Multiple imaging techniques, having advantages and disadvantages,
from conventional radiology, scintigraphy to MSUS, MRI and 18-FDG
PET/CT have improved the diagnosis of PMR and have made it possible
to differentiate this particular pathology from other similar
disease, such as elderly onset rheumatoid arthritis (RA; EORA), and
to provide a prompt therapeutic intervention (1,33).
Conventional radiology
The use of conventional radiology is considered
outdated for the diagnosis of PMR. Due to the inflammatory features
of joint and periarticular structures characteristic of the disease
and to the non-erosive aspect of the arthritis, the use of this
method does not provide useful information. In this setting, it
could be used only for differentially diagnosing PMR from other
inflammatory, erosive or degenerative joint disease or concomitant
diseases. The last guidelines of the British Society for
Rheumatology and the Health Professionals in Rheumatology for the
management of PMR include a chest X-ray as the bare minimum for the
establishment of the diagnosis, being useful for the exclusion of
alternative conditions that may mimic the disease (45).
Scintigraphy
The advances in nuclear medicine imaging techniques
over the past decade have surpassed the capabilities of
conventional scintigraphy. The lack of the high specificity of the
method and the use of new nuclear medicine imaging modalities
justify the absence of recent publications on this topic over the
past decade. Gallium-67 scintigraphy reports in PMR demonstrate
intense uptake in both shoulders (46). The high sensitivity of the
Technetium pertechnetate scintigraphy was reported by O'Duffy et
al (47) since 1976. That
study, reported that 24 out of 25 patients exhibited positive PMR
characteristics with abnormal uptake in both shoulders, as compared
with the lack of the PMR characteristics in 26 controls.
Nevertheless, the lack of discriminative power currently justifies
the absence of recent data regarding the use of the method.
Ultrasound
MSUS has recently become a preferred technique,
mainly due to its capacity to visualize in a multi-planar and
dynamic way both articular and extra-articular synovial structures,
with a relative low-cost and wide availability. Using standardized
scanning techniques and defined ultrasound pathology, together with
the addition of power- and colour-Doppler, MSUS has improved the
ability to detect and assess inflammatory activity in PMR with
excellent reliability. In addition, MSUS has been demonstrated to
have high intraobserver (k=0.96) and interobserver (k=0.99)
reproducibility (48).
Diagnostic accuracy. As a result of several
ultrasound studies performed in Europe, with regard to the
detection of inflammatory lesions in PMR mostly using B-mode, and
to a lesser extent, power Doppler examination, the most frequent
ultrasound abnormalities described are bursitis of the
subacromial/subdeltoid (SASD) bursae and tenosynovitis of the long
head of the biceps tendon (LHBT), ranging from 6.2 to 100% at the
shoulder level, with a higher prevalence of SASD bursitis, and less
frequently, trochanteric bursitis and synovitis at the hip level
(49,50). The importance of this data
determined the inclusion of an ultrasound criteria for the first
time in rheumatology in the 2012 EULAR/ACR Provisional
Classification Criteria for PMR, increasing the specificity of the
clinical diagnosis to 81% (51).
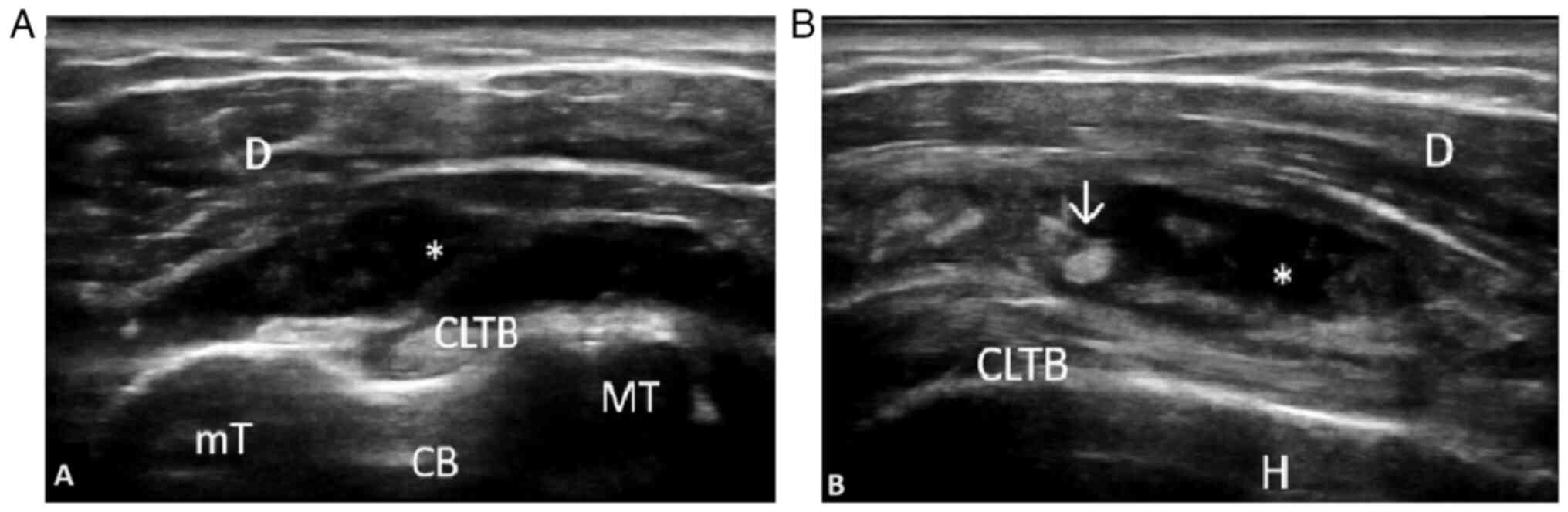
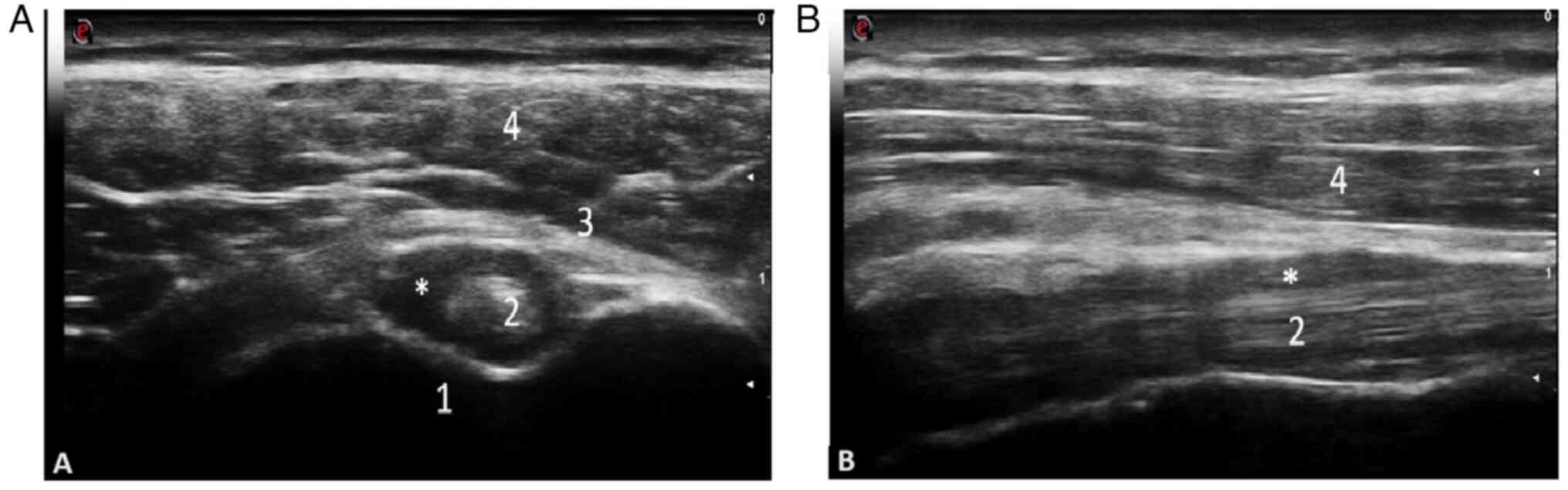
Subsequently, Macchioni et al (52) revealed that the addition of
ultrasound to clinical criteria increased the diagnostic
performance from 81.5 to 91.3% in patients with PMR, while
comparing PMR to other types of inflammatory arthritis, including
RA. The diagnostic specificity in this case increased from 79.9 to
89.9% (Figs. 2 and 3). The images were obtained by examining
a patient with PMR at the Emergency Clinical County Hospital of
Craiova.
A recent study by Kobayashi et al (53) demonstrated that ultrasound of the
shoulder and knee, improves the accuracy of the 2012 EULAR/ACR
Provisional Classification Criteria for PMR; however, this does not
apply for the hip. Considering that the assessment of the hip joint
by ultrasound is not a patient or physician-friendly procedure, due
to limited sensitivity in the detection of abnormalities, in
comparison to MRI and that inflammatory knee lesions are frequently
detected in PMR using MRI and PET/CT, particularly in tendons and
ligaments besides bursas and synovia, it was concluded that
bilateral involvement of the shoulder (LHBT, supraspinatus or
subscapularis tendon) and the bilateral involvement of the knee
[popliteus tendon (PopT) or medial or lateral collateral ligament]
provided numerically increased sensitivity (90 vs. 87%),
specificity (83 vs. 68%), positive predictive value (79 vs. 67%)
and negative predictive value (92 vs. 87%) compared with the 2012
EULAR/ACR criteria without ultrasound. In the PMR-definite group
the dominant ultrasound lesions were the tenosynovitis of LHBT and
that of PopT, with 85% exhibiting both abnormalities (53).
In a systematic review by Mackie et al
(54) in 2015 regarding the
accuracy of musculoskeletal imaging for the diagnosis of PMR, the
use of ultrasound was associated with several strengths. It is
worth mentioning that according to that review, control patients
with other inflammatory diseases were included in order to estimate
the diagnostic accuracy compared to MRI and PET/CT studies only in
ultrasound-related studies. Bilateral SASD bursitis had the most
discriminative value for PMR diagnosis, with a specificity of 89%
and sensitivity of 66%, superior to glenohumeral synovitis,
according to data from four ultrasound-related studies. The
ultrasound detection of trochanteric bursitis demonstrated a
sensitivity ranging from 21 to 100% (54).
Ultrasound in PMR may be of particular assistance in
establishing positive diagnosis in cases with normal ESR, as
recorded in 7-22% of patients at time of diagnosis (3). Manzo et al (55) suggested a 4-point guidance on how
to investigate a suspicion of PMR, ultrasound being of real
positive value when bilateral SASD bursitis, LHBT tenosynovitis or
trochanteric bursitis are present.
Differential diagnosis. The role of
ultrasound in differential diagnosis of PMR is supported by several
studies. When analyzing the diagnostic outcome in patients with
polymyalgic symptoms, Falsetti et al (56) suggested the importance of
ultrasound in the identification of the most predictive ultrasound
model for PMR. This particular model is represented by the
detection of the presence of bilateral SASD bursitis, a low
frequency of wrist, metacarpophalangeal and metatarsophalangeal
effusion/synovitis, a low frequency of knee menisci
chondrocalcinosis or tendinous calcaneal calcifications, Achilles
enthesitis and low-power Doppler ultrasound (PDUS) scores at wrist
level.
Ruta et al (49) compared shoulder ultrasound
abnormalities in patients with PMR and RA and detected bilateral
SASD bursitis in 36% of patients with PMR and only in 3% of
patients with RA, with a similar difference noted for LHBT
tenosynovitis, which was observed in 30% of patients with PMR and
was not observed in the RA control group.
Furthermore, the presence of moderate to severe
proliferative synovitis of the shoulder bursae, particularly in the
subacromial bursa, is a key ultrasound feature for discriminating
EORA from PMR-like onset EORA (pm-EORA) from PMR. Higher scores of
gray scale and the power Doppler evaluation of synovitis were
obtained by Suzuki et al (57) in 2017 in patients with PMR compared
to those with pm-EORA. The same authors further extended the
comparison between pm-EORA and PMR by proposing a semi-quantitative
PD scoring system for the hyperemia on the subscapularis tendon,
with good intraobserver and interobserver reproducibility,
demonstrating that inflammation in PMR is predominantly localized
in extrasynovial soft tissue or shoulder bursa, as compared to
pm-EORA (58).
In a recent study by Ottaviani et al
(59) which analyzed 94 patients
with polymyalgic syndrome, it was concluded that the screening of
the acromioclavicular joint may help distinguish PMR from calcium
pyrophosphate disease (CPPD), as patients with CPPD demonstrated
humeral bone erosions, synovitis and CPPD of the AC joint more
frequently, with a sensitivity of 85.2% and specificity of 97.1%.
By contrast, despite a low specificity, the most sensitive US
features for PMR diagnosis were subacromial-subdeltoid bursitis
(96.3%) and biceps tenosynovitis (85.2%).
Treatment efficacy. Consistent information to
support the role of ultrasound in monitoring the response to
treatment in PMR is still lacking. Jiménez-Palop et al
(48) performed a prospective
study in a cohort of 53 patients with PMR treated with
corticosteroids, assessing as main objective the ultrasound
inflammatory changes at the shoulder and hip level. Their study
concluded that an ultrasound may be a useful additional tool for
monitoring the response to corticosteroid treatment, due to the
significant decrease in the ultrasound inflammatory parameters
having been detected at week 4, whereas after 4 and 12 weeks of
treatment were more prone to the alteration of their levels in
comparison with clinical and laboratory markers of the disease
activity (48). However, according
to another study by Miceli et al (60) in 2017 on 66 patients with PMR that
underwent ultrasound evaluation at baseline and after 12 months of
GC therapy, the presence of subdeltoid bursitis and/or biceps
tenosynovitis at baseline was not a predictive marker either for GC
response or for the requirement for the administration of an
increased GC dose to maintain remission at 12 months. Nevertheless,
in the prospective open-label outcomes and treatment regimens
(TENOR) study that included 18 patients with PMR treated with
tocilizumab infusions without corticosteroids, ultrasound and MRI
demonstrated notable improvements in inflammatory lesions. At week
12, ultrasound examinations proved that bursitis improved
significantly in all four joints (P=0.029), although
intra-articular effusions/synovitis exhibited less improvement
(P=0.001). By the end of week 12, 37% of ultrasound-detected
abnormalities improved (61).
MRI
MRI has extensive applications in rheumatology and
its use in PMR is not an exception to this. Due to the accurate
visualization of deep structures, including spine, peripheral
joints, tendons, bursae and periarticular tissue, several studies
over the past decade have provided novel insight into the
anatomical origin of inflammation in PMR, with emphasis on
extra-articular involvement of enthesis, bursae or periarticular
tissues.
Diagnostic accuracy. Several MRI studies have
facilitated the diagnosis of PMR. Fruth et al (62) in 2018 investigated the presence of
disease-specific patterns in 40 patients with PMR using
contrast-enhanced MRI (ceMRI) of the pelvis. The predominantly
occurring characteristic for all patients with PMR was the
peritendinous enhancement of pelvic girdle tendons. All cases
exhibited bilateral involvement of the common ischiocrural tendon,
gluteus medius and minimus tendons, proximal rectus femoris origin
and in 90% of cases enhancement of the adductor muscles at the
inferior pubic bone. Therefore, bilateral involvement of at least
four extracapsular sites in the pelvic region detected in patients
with PMR by using ceMRI suggests that it may be relevant for
diagnostic purposes (62). The
same authors performed in 2020 pelvis ceMRI in 40 patients with
confirmed diagnosis of PMR, including 80 individual healthy
controls. That study confirmed a distinct pattern of extracapsular
inflammation including bilateral peritendinitis and pericapsulitis
of the proximal origins of the rectus femoris muscle and adductor
longus muscle, characteristic for PMR, with significant diagnostic
capability of the method, an excellent sensitivity of 95.8% and a
specificity of 97.1% (63).
MRI has been proven to be useful for the diagnosis
and the identification of inflammatory sites difficult to evaluate,
including lumbar interspinous bursae in patients with PMR, as
demonstrated by Salvarani et al (64). The authors of that study reported
evidence of interspinous lumbar bursitis found in 9/10 patients
with PMR and that lumbar pain may be supportive of predominantly
extra-articular synovial involvement (64). Although the use of MRI aids in
identifying additional areas of inflammation in the spine and
pelvis, the number of controls with inflammatory disease was
insufficient for precise specificity estimates, as demonstrated by
Mackie et al (65) in a
systematic review of the literature regarding the accuracy of
musculoskeletal imaging for the diagnosis of PMR. Although MRI
appears to be of particular interest in identifying deep structures
with a limited acoustic window for ultrasound examination,
including the spine and pelvis, its use may be limited by increased
costs and limited availability, particularly for repeated
evaluations in patients with symptom resolution following GC
treatment, a limited area of imaging and a longer examination time,
as well as limited access to whole-body MRI.
Instead, according to Mackie et al (65), whole-body MRI in PMR can identify a
distinct subset of patients who are more likely to respond to GC
therapy according to the MRI pattern of extracapsular inflammation
and high IL-6 and CRP levels. The same study was designed for
distinguishing PMR from RA according to the patterns of
inflammation. In patients with PMR, extracapsular features of
inflammation, including periacetabular inflammation without the
involvement of the hip joint, extended from the anterior hip
capsule, medial to the gluteus muscle and lateral to the iliac
bone, distinct from iliopectineal bursitis, may help distinguishing
between PMR and RA. Additionally, it is considered a predictor of
response to glucocorticoid therapy. In this particular subset of
patients with PMR, the entheseal involvement resembled a
seronegative spondyloarthropathy (65).
An MRI study by Cimmino et al (66) regarding hand involvement in PMR
also demonstrated the prevalent inflammation of extra-articular
structures, presenting with extensor and flexor tendons
tenosynovitis rather than joint synovitis. Of note, the authors of
that study did not identify an association between clinical
presentation and MRI, supporting the presence of extensor
tenosynovitis as an epiphenomenon suggestive for subclinical
disease (66).
Differential diagnosis. In support of the use
of MRI in differential diagnosis, Ochi et al (67) evaluated shoulder and hip joints in
PMR and RA patients. The MRI parameters analyzed were thickness and
abnormalities of the supraspinatus tendon, effusion around the
glenohumeral joint, subacromial-subdeltoid bursa, the biceps tendon
in the shoulder and effusion around the acetabulofemoral joint,
iliopsoas bursa and trochanteric bursa in the hip (67). The supraspinatus tendon was
significantly thicker in PMR patients than in RA and control
patients (P<0.05). Patients with PMR exhibited increased scores
for effusions (joint, bursa, and tendon sheath in the shoulder and
bursa in the hip), as well as more frequent periarticular soft
tissue edema (P<0.05) as compared with RA cases.
A recent article by Nakamura et al (68) analyzed whether gadolinium-enhanced
MRI in shoulders of patients with PMR could increase the diagnostic
value and predict recurrence. Supporting the findings of
extra-synovial involvement detected at hip level by Ochi et
al (67), MRI abnormalities,
including capsulitis, rotator cuff tendinitis and focal bone edema
in the shoulder improved diagnostic accuracy in PMR with 76%
sensitivity and 85% specificity. In addition, in patients with
recurrence of the disease, rotator cuff tendinitis and synovial
hypertrophy were predictive signs (68).
Treatment efficacy. In a previous study, the
response to treatment with tocilizumab was evaluated in a post hoc
MRI analysis of the data from the TENOR study, at baseline,
following 2 and 12 weeks of treatment. Myofascial lesions were
characteristic for recent onset PMR in the shoulder and hip.
Resolution of inflammatory lesions was observed at week 12 in 41.7%
of the 103 groups of muscles studied, while improvements were
depicted in 64.1% of the examined muscle groups (69).
PET/CT
PET/CT scans using an analogue of glucose known as
2-[fluorine-18]-fluoro-2-deoxy-D-glucose
(18F-FDG-PET/CT), are a type of imaging technique that
use a radioactive isotope, often implemented in the diagnosis and
monitoring of oncological patients. By contrast, other clinical
applications excluding cancer diagnosis are currently being used in
clinical practice, since FDG accumulates in tissues that are not
exclusively malignant. FDG also accumulates in inflammatory areas
of the tissues, due to elevated activity levels in cells involved
in inflammation, including lymphocytes, neutrophils and macrophages
(70). In 2018, Slart et al
(71) established recommendations
for the application of PET/CT in improving the diagnostic and
monitorization process in individuals with large vessel vasculitis
(LVV), as well as PMR.
PET/CT can be used for the detection of mural
inflammation and/or luminal changes in extra-cranial arteries to
support the diagnosis of large vessel-GCA, as stated in the EULAR
recommendations for the use of imaging in LVV, also revealing PMR
lesions that remain elusive when other techniques are used. Even
though it is not routinely used in PMR, PET/CT can reveal PMR
lesions that are difficult to detect using other methods (72).
Numerous studies have been conducted in an effort to
define a particular pattern of 18F-FDG uptake that may aid in the
diagnostic process. Yuge et al (73) conducted a study on 60 individuals
who initially diagnosed with PMR, enthesitis, arthritis, or
myopathy. However, after applying the criteria established by the
ACR/EULAR in 2012, the total number of patients diagnosed with PMR
was limited to 16 individuals. In the final PMR group, the highest
incidence of 18F-FDG was detected in the glenohumeral and
sternoclavicular joints (88%), followed by the spinous processes
and greater trochanters, ischial tuberosities and the last
acromioclavicular joints, wrists and elbows. An enhanced ‘Y-shaped’
uptake along the interspinous bursae was a characteristic pattern
for patients with PMR (73,74).
In the study by Kaneko et al (75), 20 patients with PMR were enrolled,
detecting isotope accumulation specifically in the proximal joint
structures (glenohumeral, coxofemural and sternoclavicular joints)
and in the extra-articular synovial structures (greater trochanter,
ischial tuberosity, and the area anterolateral to the rim of
acetabulum). Furthermore, another study conducted by Rehak et
al (76) discovered an
accumulation of the isotope in the prepubic region in specific
individuals. This finding was most likely the result of pectineus
and adductor longus enthesitis. In addition to this, the authors of
that study demonstrated that the areas with high accumulation of
the tracer revealed no uptake after PMR therapy (76). This provides support to the
utilization of 18F-FDG-PET/CT in the management of PMR not only for
the diagnosis but also for the monitoring of treatment.
Sondag et al (77) demonstrated that considerable
absorption in three or more sites in the joints, bursae, or
entheses (acromioclavicular, sternoclavicular, glenohumeral;
ischial, trochanteric, iliopectineal, and interspinous; pubic
symphysis, respectively) was related to the diagnosis of PMR with a
sensitivity of 74%. This method also assists in the process of
differentially diagnosing PMR and RA, particularly EORA (77).
In a previous study by Takahashi et al
(78), a typical pattern for PMR
and EORA was established. In patients with PMR, a high sensitivity
(92.6%) and a high specificity (90%) was observed when three out of
five characteristic regions exhibited either an increased or an
absent 18F-FDG accumulation. An increase in uptake was
detected in the ischial tuberosities, vertebral spinous processes,
glenohumeral joints, and iliopectineal bursitis, and was not
observed in the wrists (78).
Moreover, a retrospective study was conducted by
Wendling et al (79) at a
single center on patients diagnosed with PMR according to the
criteria established by the ACR and EULAR in 2012. A control group
of individuals who did not present with rheumatological symptoms,
but were tested as part of neoplastic research, or patients with
neoplastic disorders who were followed-up were also analyzed. A
total number of 201 cases were investigated, including 101 patients
with PMR and 100 healthy individual controls. Overall, PET muscle
injury was observed in 34% of patients with PMR, as compared with
10% of the individuals in the control group. In total, 19, 14, 13
and six afflicted muscle sites were detected in the spinal region,
the scapular girdle, the pelvic girdle and the thighs,
respectively. On three occasions, fasciitis was also observed. In
individuals diagnosed with PMR, age, CRP levels, or an overall PMR
PET score were not linked to muscle involvement detected by PET
(79).
In conclusion, although PET/CT is not a routine
investigation as this imaging method exposes patients to increased
levels of radiation, PET/CT may prove to be a useful diagnostic and
monitoring tool for patients with PMR.
Role of imaging in PMR
The use of modern imaging techniques provides novel
information regarding the anatomical and pathophysiological basis
of PMR. Novel sites of inflammation were discovered with the use of
MRI and PET/CT as compared to the use of MSUS alone. Thus, in
addition to SASD bursitis and biceps tenosynovitis, inflammation of
the peritendon of muscle insertions at the hip and interspinsous
bursae are findings that may aid clinicians in differentially
diagnosing PMR from other elderly-onset inflammatory diseases.
Additional studies on larger patient cohorts are
required; however, these imaging techniques may be valuable for the
diagnosis and monitoring the response to treatment in patients with
PMR.
6. Diagnosis
When common signs and symptoms, as well as increased
levels of inflammatory markers occur, the diagnosis of PMR is not a
difficult process for a clinician with an extensive knowledge in
this research field. However, there is a certain risk for less
experienced clinicians to over- or underdiagnose PMR, particularly
in situations involving illnesses that mimic PMR or in patients
with many comorbidities, due to the absence of a diagnostic gold
standard and the lack of specificity of the signs, symptoms, and
laboratory data associated with PMR.
Over the years, several classification criteria have
been proposed for PMR, the latest being the 2012 European League
Against Rheumatism and American College of Rheumatology provisional
classification criteria (Table I)
(80).
 | Table IEULAR/ACR 2012 provisional
classification criteria for polymyalgia rheumatica. |
Table I
EULAR/ACR 2012 provisional
classification criteria for polymyalgia rheumatica.
|
Criteria/symptoms | Points without
MSUS | Points with
MSUS |
|---|
| Morning stiffness
lasting for >45 min | 2 | 2 |
| Hip pain or limited
mobility | 1 | 1 |
| Absence of RF or
ACPA | 2 | 2 |
| Absence of other
joint involvement | 1 | 1 |
| At least one
shoulder with subdeltoid bursitis and/or bicep tenosynovitis and/or
glenohumeral synovitis (either posterior or axillary) and at least
one hip with synovitis and/or trochanteric bursitis | Not applicable | 1 |
| Both shoulders with
subdeltoid bursitis, biceps tenosynovitis or glenohumeral
synovitis | Not applicable | 1 |
The required inclusion criteria are the following:
An age ≥50 years, bilateral shoulder pain and abnormal CRP and/or
ESR levels. A score ≥4 strongly indicates PMR manifesting without
MSUS, whereas a score ≥5 is indicates the presence of PMR with
MSUS.
Prior to the development of the aforementioned
criteria, other four research groups developed classification
criteria for PMR, as follows: i) Bird criteria in 1979; ii) Jones
and Hazleman criteria in 1981; iii) Chuang and Hunder criteria in
1982; and iv) Healey criteria in 1984 (Table II) (80-83).
 | Table IIClassification criteria for PMR. |
Table II
Classification criteria for PMR.
| Bird criteria 1979
(at least 3 out of 6 criteria stated below are required for
classification as PMR) | Jones and Hazleman
criteria 1981 (all criteria stated below are required for PMR
classification) | Chuang and Hunder
criteria 1982 (all criteria stated below are required for PMR
classification) | Healey criteria
1984 (required criteria: Age ≥50 years and at least 3 out of 5 of
the remaining criteria required for classification as PMR) |
| Bilateral shoulder
pain and/or stiffness | Shoulder or pelvic
girdle pain without muscle weakness | Age ≥50 years | Age ≥50 years |
| Disease onset <2
weeks Initial ESR ≥40 mm/h | Morning stiffness
lasting for more than 1 h Disease duration of more than 2 months
per year | Bilateral pain and
stiffness persisting for 1 month or more involving two of the
following areas: neck or torso, shoulders or upper arms, | Persistent pain for
1 month or more involving two of the following areas: neck,
shoulders, or pelvic girdle |
| Morning stiffness
duration >1 h | ESR >30 mm/h or
CRP >6 mg/l | hips or thighs | Morning stiffness
lasting for more than 1 h |
| Age >65
years | Absence of
rheumatoid arthritis | ESR >40
mm/h | Rapid response to
prednisolone (≤20 mg per day) |
| Depression and/or
loss of weight | Absence of
objective signs of muscle disease | Exclusion of other
diagnoses with the exception of giant cell arteritis | Absence of other
joint or musculoskeletal diseases |
| Bilateral upper arm
tenderness | Fast and dramatic
response to systemic glucocorticoids | | ESR >40
mm/h |
7. Differential diagnosis
Conditions that afflict adults aged >50 years and
are linked with bilateral shoulder pain should be considered for
the differential diagnosis of PMR, since it is also a condition
that causes discomfort in the neck and shoulders. This is
important, considering the fact that there are no specific
diagnostic tests for PMR. A misinterpretation of any disease as PMR
may lead to inappropriate exposure to GCs for extended periods of
time. Rheumatic diseases and non-rheumatic diseases should also be
included in the differential diagnosis. With the emerging of new
diagnostic criteria and the use of MSUS, PMR is easier to detect,
making the differential diagnosis less complicated (84).
The different diseases that should be considered for
differential diagnosis are the following: i) Rheumatic diseases,
including rheumatoid arthritis, particularly the seronegative form,
spondyloarthritis, microcrystalline arthritis, systemic lupus
erythematosus, vasculitis and inflammatory myopathies; ii)
non-inflammatory musculoskeletal pathologies, including
fibromyalgia, osteoarthritis-glenohumeral and coxofemoral
osteoarthritis, rotator cuff pathologies, subacromial/subdeltoid
bursitis and adhesive capsulitis; iii) remitting symmetrical
seronegative synovitis with puffy edema; iv) endocrinopathies,
including thyroid pathologies and pathologies of the parathyroid
glands; v) viral, bacterial infections and infectious or
mycobacterial endocarditis; vi) solid or hematological neoplasia;
and vii) other pathologies, including Parkinson's disease,
depression, hypovitaminosis D and medication-induced myopathy (e.g.
statin-induced myopathy) (85,86).
8. Treatment
The treatment of PMR is currently based on the 2015
EULAR/ACR recommendations. There is no validated definition of
remission and/or relapse for patients with PMR. However, the
majority of definitions encountered in the literature comprise a
combination of the absence of clinical
symptoms/myalgias/improvement of clinical symptoms with ESR levels
<20-40 mm/h and CRP levels <0.5-1 mg/dl. Regarding the
therapy, the patients should have discontinued the GCs or these
should be administered at a reduced dose (87).
Thus, the use of GCs is recommended instead of
non-steroidal anti-inflammatory drugs (NSAIDs) in patients with
PMR, with the exception of the short-term use of NSAIDs and/or
analgesics for the improvement of the symptoms of other associated
pathologies, including coexisting osteoarthritis. According to the
guidelines, a minimum effective dose of equivalent of prednisone
ranging from 12.5 to 25 mg/day is recommended. Dose tapering is
required to be individualized, according to the clinical and
biological profile of each patient. The following principles for
dose tapering are recommended to be administered: i) initial
tapering ≤10 mg/day equivalent of prednisone in 4-8 weeks; ii) for
relapse therapy, the dose of GCs is increased to the previous dose
before the relapse, followed by its gradual tapering in 4-8 weeks
up to the dose at which the relapse occurred; and iii) when
tapering the dose in the case of remission, the dose of prednisone
should be decreased by 1 mg every 4 weeks until the discontinuation
of therapy, as long as remission is maintained.
The administration of intramuscular
methylpredisolone should be considered as an alternative to
administering GCs orally; however, this decision remains at the
discretion of the attending physician (88). Concerning recently diagnosed
patients, Dejaco et al (88) compared the efficacy of the oral
administration of prednisolone (initial dose of 15 mg/day gradually
reduced to 10 mg/day) with the administration of intramuscular
methylprednisolone acetate (120 mg every 2 weeks for 12 weeks
followed by injections every month, with dose tapering by 20 mg
every 3 months). The prednisolone dosage was gradually decreased at
levels <10 mg per day at a rate of 1 mg every 8 weeks. Both
courses of treatment successfully induced and maintained the
patients with PMR in remission. By contrast, oral prednisolone
administration trended towards managing symptoms more rapidly and
effectively than intramuscular injections of methylprednisolone
(88).
Patients to whom oral prednisolone was administered
received a larger cumulative GC dose, being prone to more
GC-related adverse events than those who were administered
injectable methylprednisolone; however, higher rates of stopping
the medication were observed (88).
A single dose of prednisone per day is recommended,
except for cases in which nocturnal pain is severe following the
reduction the dose of GCs administered to <5 mg/day equivalent
of prednisone (88).
The early introduction of synthetic disease
modifying therapy with methotrexate (MTX) in doses of 7.5-10
mg/week is conditionally recommended, particularly in patients with
an increased risk of relapse, as well as in cases with risk
factors, comorbidities and/or with concomitant treatments that
predispose to adverse reactions in combination with GCs (78). In the study by Ruediger et
al (89) in 2020 conducted on
70 patients with PMR, out of which 31% were prescribed MTX in
combination with GCs, MTX was associated with a reduction in
steroidal anti-inflammatory drugs use and an improvement in
inflammatory biological profile. A multicenter randomized,
double-blind, placebo-controlled trial performed by Caproali et
al (90) on 72 patients newly
diagnosed with PMR proved that the administration of 10 mg/week of
MTX in combination with GCs compared to GCs alone was associated
with the earlier cessation of prednisone therapy, rendering it
useful in patients at a high risk for steroid use. Furthermore, an
ongoing multicenter double-blind placebo-controlled clinical trial
is currently conducted by Marsman et al (91) aiming to evaluate the efficacy of
the administration of 25 mg/week MTX in patients with PMR in an
early disease phase.
Studies on other conventional synthetic
immunosuppressive drugs are limited, usually based on small study
groups or case series. Hydroxychloroquine and azathioprine have
been tested in patients with PMR. The study by de Silva et
al (92) involving 31 patients
with PMR and/or GCA tested the efficacy of azathioprine, suggesting
that patients who received azathioprine required a reduced GC
dosage. However, the majority of the patients fulfilled the
criteria for GCA and the number of patients was limited (92); thus further extensive studies are
required in order to attest the efficacy of azathioprine.
Hydroxychloroquine was also tested in a retrospective study
performed by Lee et al (93), demonstrating no benefits for
patients with PMR.
The use of anti-TNFα biological therapy is not
recommended for the treatment of PMR as it has not proven to be
beneficial to the patients. The administration of the antagonist of
the receptor for IL-6, tocilizumab, has been demonstrated to
improve symptoms and attenuate the inflammatory syndrome in
patients with PMR in several series of cases and retrospective
studies. In the study performed by Lally et al (94) on 10 patients with PMR, with only 9
patients having been assessed at the time of the primary endpoint,
it was concluded that tocilizumab may be an efficient,
well-tolerated drug, with a good safety profile and a great
steroid-sparing effect. All the patients did not present relapse
without GC therapy at the primary endpoint (94). Overall, 20 patients with active PMR
of recent onset were included in a prospective open-label study
performed by Devauchelle-Pensec et al (95). These patients received three
tocilizumab infusions at 4-week intervals, without receiving GC
therapy, followed by the administration of oral prednisone. At the
end of the 12th week, all of the patients reported clinical
improvement in their PMR symptoms (95). Furthermore, in a more recent
randomized, double-blind, placebo-controlled trial on 101 patients
with PMR, steroid-dependent patients were treated with tocilizumab.
GC therapy was terminated by week 24 in 49% of patients in the
tocilizumab group as compared to the placebo group, of which only
9% terminated the GC (96). In a
series of cases presented by Mori and Koga (97), three patients presenting with
GC-resistant PMR were administered tocilizumab in addition to GCs,
with all of the patients achieving remission. A phase 2/3
randomized controlled trial on 36 patients with new-onset PMR
conducted by Bonelli et al (98) proved that tocilizumab was superior
to the placebo when attesting to the sustained GC-free remission,
time to relapse and the cumulative GC dose. Out of the 36 patients
enrolled in that study, 19 received subcutaneous tocilizumab in
doses of 162 mg per week, while 17 were administered the placebo.
All the patients received prednisone doses tapered from 20 mg to 0
mg over the course of 11 weeks (98).
Limited research has been conducted on the
administration of other biological therapeutics in individuals
diagnosed with PMR. In a proof-of-concept, single-blind, three-arm
study, 16 patients with PMR were administered either secukinumab or
canakinumab, as a single dose of 3 mg/kg/body weight, or oral
prednisone at a dose of 20 mg per day (99). Patients were randomly assigned
1:1:1 to receive either secukinumab or canakinumab or GCs. Patients
who were administered GCs demonstrated significant reductions in
their levels of pain, whereas those who were treated with
secukinumab and canakinumab only exhibited a slight improvement in
their range of motion. On day 15, none of the patients who were
receiving biological treatment and only one of the patients who
were receiving GCs obtained a full response. In the group that
received secukinumab, in 4 patients, this was replaced by GCs. A
dose of GCs that was 40% lower on a monthly basis was then
required, as compared with individuals who were not treated with
biological therapeutics. Additionally, this also applied for 3
patients who were treated with canakinumab prior to changing the
treatment to GCs. Overall, it was suggested that the application of
these biological therapeutics in patients with PMR requires further
investigation (99).
A prospective open-label 52-week pilot study
investigated the efficacy of baricitinib, which is a JAK1 and JAK2
inhibitor, in treating relapsing forms of GCA in patients (100). Baricitinib was well-tolerated and
the majority of patients were able to terminate GCd administration
as a consequence. It is probable that JAK inhibition may be also
important for the treatment of PMR (100).
The BRIDGE-PMR, a double-blind, randomized,
placebo-controlled, proof-of-concept trial included 47 patients
with PMR randomized 1:1 to a single intravenous infusion of
rituximab 1000 mg or the placebo (101). All the patients received a
17-week GC tapering scheme. That study revealed that rituximab was
more efficient in combination with GCs than the placebo and GCs
(101). In extension of that
study, the 47 patients included in the original study were
followed-up from 2019 to 2021, and it was proven that the patients
treated with rituximab were in GC-free remission at 1 year after
the infusion (92). Thus,
rituximab may be considered a valid treatment option for PMR,
although studies on larger groups of patients are required
(102).
Sarilumab, a recently approved drug for the
treatment of PMR, was studied the SAPHYR trial which compared
sarilumab and 14-week GC tapering with placebo and 52-week GC
tapering. The arm treated with sarilumab demonstrated improved
clinical status than the GC arm (103).
There are several ongoing studies evaluating the
efficacy of certain conventional synthetic and/or biological agents
for PMR treatment (Table III).
The website https://clinicaltrials.gov/ was used as research for
the ongoing studies evaluating treatment options in PMR.
 | Table IIIOngoing clinical studies in PMR. |
Table III
Ongoing clinical studies in PMR.
| Clinical study | Accession
no./year | Type | Primary
outcome | Secondary
outcome | Status/results |
|---|
| A study to evaluate
the change in disease state and adverse events in adult
participants with PMR, dependent on glucocorticoid treatment, and
receiving subcutaneous injections of ABBV-154 (AIM-PMR). |
NCT0497296/2021 | A phase 2,
randomized, double-blind, placebo-controlled clinical trial | Time to flare (time
frame 52 weeks) | Percentage of
patients who achieve flare free-state; cumulative glucocorticoid
dose change from baseline in glucocorticoid dose. | Recruiting |
| Clinical trial of
prednisolone in combination with SPI-62 or placebo in subjects with
PMR. |
NCT05436652/2022 | Interventional | Erythrocyte
sedimentation rate; CRP; Plasma fibrinogen; Timeframe, baseline to
day 28. | | Not yet
recruiting |
| Treatment with
leflunomide in patients with PMR (PMRLEFRCT). |
NCT0357679/2018 | Prospective,
randomized, double-blind, placebo-controlled treatment study during
12 months (part I) and an additional open-label follow-up of 12
months (part II) | PMR relapse. | Time until first
relapse within the first 24 months; Percentage of patients with at
least one relapse in the first 12 or 24 months; number of relapsing
patients within the first 24 months; time until glucocorticoid free
remission; glucocorticoid-sparing effect; number of participants
with adverse events and serious adverse events as assessed by
MedDRA V21.0. | Recruiting |
| Abatacept in earLy
onset PMR: Study ALORS (ALORS) |
NCT03632187/2018/2018 | Multicenter double
blinded randomized placebo controlled trial. | Following of one
biological parameter (CRP) for a time frame of 12 weeks. | Emergence of
adverse events; following of the PMR activity score; medical
resource evaluation; following of the cumulative dosages of
glucocorticoids; The flare of the PMR (PMR-AS); following of the
medical exam using the ultrasound Scoring; evaluation of FDG uptake
using TEP scanner in regions of interest; following the proportion
of patient relapse; biological marker levels (interleukin,
cytokines, immune cells); following of patient quality of life | Active, not
recruiting |
| BAriCitinib Healing
Effect in earLy pOlymyalgia Rheumatica (BACHELOR) |
NCT0402710/2019 | Multicenter double
blinded randomized placebo-controlled trial. | Following of the
PMR activity score (time frame 12 weeks) | Following of the
PMR activity score (time frame 36 weeks); Emergence of adverse
events (safety and tolerability); following of the cumulative
dosages of glucocorticoids; ultrasound of synovitis and
tenosynovitis; levels of biological markers (interleukin,
cytokines, immune cells); following of the quality of life using
SF36; following of the quality of life according to HAD; following
of the quality of life according to the scale EuroQol 5
dimensions. | Recruiting |
The optimization of the benefit-to-risk ratio of
GCs in order to achieve durable remission, while minimizing the
occurrence of side-effects is an ongoing issue. Subsequently, the
creation of novel GC preparations and/or GC receptor ligands may be
able to improve the benefit-to-risk ratio of GCs. Accordingly,
selective GC receptor agonists and modulators may be potential
therapeutics targeted at selectively enhancing anti-inflammatory
cellular pathways. As consequence, the undesirable effects
associated with these medications would not be triggered by the
pathways that these pharmaceuticals could prevent from being
activated (104).
9. Conclusions and future perspectives
Although the present review was a narrative one,
which could be considered a limitation, it provides important
insight into the new diagnostic techniques and treatment options
for PMR. In conclusion, PMR is a prevalent disease that can
occasionally impose marked diagnostic and therapeutic difficulty.
Further research into its pathophysiology is required in order to
elucidate the underlying processes further, which will serve as the
foundation for future tailored treatments. In addition, there is a
demand for improved techniques of diagnosis, which should include
the further improvement of various imaging modalities, in order to
assist in accurate diagnosis and appropriate therapy. Other
potential therapeutic agents including JAK inhibitors have to be
further evaluated in PMR.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
Not applicable.
Authors' contributions
MMF, FB, AF, VP, AB, PLC, CC, LMF and AEM
contributed equally to the acquisition, analysis and
systematization of data, manuscript writing and critical revisions
for important intellectual content. All authors have read and
approved the final version of the manuscript. Data authentication
is not applicable.
Ethics approval and consent to
participate
Written informed consent was acquired from the
patients associated with the images presented in Figs. 2 and 3.
Patient consent for publication
Patients provided consent for the publication of
their personal data in Figs. 2 and
3.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Mahmood S, Bin Nelson E, Padniewski J and
Nasr R: Polymyalgia rheumatica: An updated review. Cleve Clin J
Med. 87:549–556. 2020.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Raheel S, Shbeeb I, Crowson CS and
Matteson EL: Epidemiology of polymyalgia rheumatica 2000-2014 and
examination of incidence and survival trends over 45 years: A
population-based study. Arthritis Care Res (Hoboken). 69:1282–1285.
2017.PubMed/NCBI View Article : Google Scholar
|
|
3
|
González-Gay MA, Matteson EL and Castañeda
S: Polymyalgia rheumatica. Lancet. 390:1700–1712. 2017.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Guggino G, Ferrante A, Macaluso F, Triolo
G and Ciccia F: Pathogenesis of polymyalgia rheumatica. Reumatismo.
70:10–17. 2018.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Salvarani C, Boiardi L, Mantovani V, Ranzi
A, Cantini F, Olivieri I, Bragliani M, Collina E and Macchioni PL:
HLA-DRB1 alleles associated with polymyalgia rheumatica in Northern
Italy: Correlation with disease severity. Ann Rheum Dis.
58:303–308. 1999.PubMed/NCBI View Article : Google Scholar
|
|
6
|
González-Gay MA, Amoli MM, Garcia-Porrua C
and Ollier WER: Genetic markers of disease susceptibility and
severity in giant cell arteritis and polymyalgia rheumatica. Semin
Arthritis Rheum. 33:38–48. 2003.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Carvajal Alegria G, Boukhlal S, Cornec D
and Devauchelle-Pensec V: The pathophysiology of polymyalgia
rheumatica, small pieces of a big puzzle. Autoimmun Rev.
19(102670)2020.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Elling P, Olsson AT and Elling H:
Synchronous variations of the incidence of temporal arteritis and
polymyalgia rheumatica in different regions of Denmark; association
with epidemics of Mycoplasma pneumoniae infection. J Rheumatol.
23:112–119. 1996.PubMed/NCBI
|
|
9
|
Caruana G, Are R, Mastrandrea S, Fiore V,
Peruzzu F, Porqueddu E and Mura MS: Human Parvovirus B19 and
polymyalgia rheumatica: A case report and short review of the
literature. Infect Dis Trop Med. 1(e186)2015.
|
|
10
|
Uddhammar A, Boman J, Juto P and Rantapää
Dahlqvist S: Antibodies against Chlamydia pneumoniae,
cytomegalovirus, enteroviruses and respiratory syncytial virus in
patients with polymyalgia rheumatica. Clin Exp Rheumatol.
15:299–302. 1997.PubMed/NCBI
|
|
11
|
Cimmino MA and Zaccaria A: Epidemiology of
polymyalgia rheumatica. Clin Exp Rheumatol. 18 (4 Suppl 20):S9–S11.
2000.PubMed/NCBI
|
|
12
|
Manzo C, Isetta M, Natale M and Castagna
A: Identification and classification of polymyalgia rheumatica
(PMR) and PMR-like syndromes following immune checkpoint inhibitors
(ICIs) therapy: Discussion points and grey areas emerging from a
systematic review of published literature. Medicines (Basel).
7(68)2020.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Samson M, Audia S, Fraszczak J, Trad M,
Ornetti P, Lakomy D, Ciudad M, Leguy V, Berthier S, Vinit J, et al:
Th1 and Th17 lymphocytes expressing CD161 are implicated in giant
cell arteritis and polymyalgia rheumatica pathogenesis. Arthritis
Rheum. 64:3788–3798. 2012.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Dejaco C, Duftner C, Klauser A and
Schirmer M: Altered T-cell subtypes in spondyloarthritis,
rheumatoid arthritis and polymyalgia rheumatica. Rheumatol Int.
30:297–303. 2010.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Toussirot É, Régent A, Devauchelle-Pensec
V, Saraux A and Puéchal X: Interleukin-6: A promising target for
the treatment of polymyalgia rheumatica or giant cell arteritis?
RMD Open. 2(e000305)2016.PubMed/NCBI View Article : Google Scholar
|
|
16
|
van der Geest KSM, Abdulahad WH, Chalan P,
Rutgers A, Horst G, Huitema MG, Roffel MP, Roozendaal C, Kluin PM,
Bos NA, et al: Disturbed B cell homeostasis in newly diagnosed
giant cell arteritis and polymyalgia rheumatica. Arthritis
Rheumatol. 66:1927–1938. 2014.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Alvarez-Rodríguez L, Lopez-Hoyos M, Mata
C, Marin MJ, Calvo-Alen J, Blanco R, Aurrecoechea E, Ruiz-Soto M
and Martínez-Taboada VM: Circulating cytokines in active
polymyalgia rheumatica. Ann Rheum Dis. 69:263–269. 2010.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Kreiner F, Langberg H and Galbo H:
Increased muscle interstitial levels of inflammatory cytokines in
polymyalgia rheumatica. Arthritis Rheum. 62:3768–75.
2010.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Zhang H, Watanabe R, Berry GJ, Tian L,
Goronzy JJ and Weyand CM: Inhibition of JAK-STAT signaling
suppresses pathogenic immune responses in medium and large vessel
vasculitis. Circulation. 137:1934–1948. 2018.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Weyand CM, Hicok KC, Hunder GG and Goronzy
JJ: Tissue cytokine patterns in patients with polymyalgia
rheumatica and giant cell arteritis. Ann Intern Med. 121:484–491.
1994.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Kreiner F, Langberg H and Galbo H:
Increased muscle interstitial levels of inflammatory cytokines in
polymyalgia rheumatica. Arthritis Care Res (Hoboken). 62:3768–3775.
2010.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Buttgereit F, Dejaco C, Matteson EL and
Dasgupta B: Polymyalgia rheumatica and giant cell arteritis a
systematic review. JAMA. 315:2442–2458. 2016.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Cutolo M, Montecucco CM, Cavagna L,
Caporali R, Capellino S, Montagna P, Fazzuoli L, Villaggio B,
Seriolo B and Sulli A: Serum Cytokines and steroidal hormones in
polymyalgia rheumatica and elderly-onset rheumatoid arthritis. Ann
Rheum Dis. 65:1438–1443. 2006.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Floris A, Piga M, Cauli A, Salvarani C and
Mathieu A: Polymyalgia rheumatica: An autoinflammatory disorder?
RMD Open. 4(e000694)2018.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Ji J, Liu X, Sundquist K, Sundquist J and
Hemminki K: Cancer risk in patients hospitalized with polymyalgia
rheumatica and giant cell arteritis: A follow-up study in Sweden.
Rheumatology (Oxford). 49:1158–1163. 2010.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Emamifar A, Hess S, Ellingsen T, Due Kay
S, Christian Bang J, Gerke O, Syrak Hansen P, Ahangarani Farahani
Z, Petersen H, Marcussen N, et al: Prevalence of newly diagnosed
malignancies in patients with polymyalgia rheumatica and giant cell
arteritis, comparison of 18F-FDG PET/CT scan with chest X-ray and
abdominal ultrasound: Data from a 40 week prospective, exploratory,
Single Centre study. J Clin Med. 9(3940)2020.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Bobircă A, Bobircă F, Ancuta I, Florescu
A, Pădureanu V, Florescu DN, Pădureanu R, Florescu A and Mușetescu
AE: Rheumatic immune-related adverse Events-A consequence of immune
checkpoint inhibitor therapy. Biology (Basel).
10(561)2021.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Salvarani C, Cantini F and Hunder GG:
Polymyalgia rheumatica and giant-cell arteritis. Lancet.
372:234–245. 2008.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Salvarani C, Pipitone N, Versari A and
Hunder GG: Clinical features of polymyalgia rheumatica and giant
cell arteritis. Nat Rev Rheumatol. 8:509–521. 2012.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Prior JA, Muller S, Helliwell T, Hider SL,
Barraclough K, Dasgupta B and Mallen CD: The association of pain
and stiffness with fatigue in incident polymyalgia rheumatica:
Baseline results from the polymyalgia rheumatica cohort study. Prim
Health Care Res Dev. 20(e46)2019.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Gazitt T, Zisman D and Gardner G:
Polymyalgia rheumatica: A common disease in seniors. Curr Rheumatol
Rep. 22(40)2020.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Clinical Manifestations and Diagnosis of
Polymyalgia Rheumatica-UpToDate Available online: https://www.uptodate.com/contents/clinical-manifestations-and-diagnosis-of-polymyalgia-rheumatica
(Accessed on 19 August 2022).
|
|
33
|
Iagnocco A, Finucci A, Ceccarelli F,
Scirocco C and Rutigliano IM: Musculoskeletal ultrasound in the
evaluation of Polymyalgia Rheumatica. Med Ultrason. 17:361–366.
2015.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Manzo C and Natale M: Polymyalgia
rheumatica in association with remitting Seronegative Sinovitis
with pitting edema: A Neoplastic Warning. Can Geriatr J. 20:94–96.
2017.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Kermani TA and Warrington KJ: Polymyalgia
rheumatica. Lancet. 381:63–72. 2013.PubMed/NCBI View Article : Google Scholar
|
|
36
|
González-Gay MA, Rodríguez-Valverde V and
Blanco R: Polymyalgia rheumatica without significantly increased
erythrocyte sedimentation rate: A more benign syndrome. Arch Intern
Med. 157:317–320. 1997.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Cantini F, Salvarani C, Olivieri I,
Macchioni L, Ranzi A, Niccoli L, Padula A and Boiardi L:
Erythrocyte sedimentation rate and C-Reactive protein in the
evaluation of disease activity and severity in polymyalgia
rheumatica: A prospective follow-up study. Semin Arthritis Rheum.
30:17–24. 2000.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Salvarani C, Cantini F, Niccoli L,
Macchioni P, Consonni D, Bajocchi G, Vinceti M, Catanoso MG,
Pulsatelli L, Meliconi R, et al: Acute-phase reactants and the risk
of relapse/recurrence in polymyalgia rheumatica: A prospective
Follow-up study. Arthritis Rheum. 53:33–38. 2005.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Cutolo M, Cimmino MA and Sulli A:
Polymyalgia rheumatica vs Late-onset rheumatoid arthritis.
Rheumatology. 48:93–95. 2009.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Espinoza LR, Jara LJ, Silveira LH,
Martínez-Osuna P, Zwolinska JB, Kneer C and Aguilar JL:
Anticardiolipin antibodies in polymyalgia rheumaticagiant cell
arteritis: Association with severe vascular complications. Am J
Med. 90:474–478. 1991.PubMed/NCBI
|
|
41
|
Uddhammar AC: Von Willebrand factor in
polymyalgia rheumatica and giant cell arteritis. Clin Exp
Rheumatol. 18 (4 Suppl 20):S32–S33. 2000.PubMed/NCBI
|
|
42
|
Henderson DRF, Tribe CR and Dixon ASTJ:
Synovitis in polymyalgia rheumatica. Rheumatology. 14:244–250.
1975.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Gherghina FL, Tica AA, Deliu E, Abood ME,
Brailoiu GC and Brailoiu E: Effects of VPAC1 activation in nucleus
ambiguus neurons. Brain Res. 1657:297–303. 2017.PubMed/NCBI View Article : Google Scholar
|
|
44
|
Pulsatelli L, Dolzani P, Silvestri T, De
Giorgio R, Salvarani C, Macchioni P, Frizziero L and Meliconi R:
Synovial expression of vasoactive intestinal peptide in polymyalgia
rheumatica. Clin Exp Rheumatol. 24:562–566. 2006.PubMed/NCBI
|
|
45
|
Dasgupta B, Borg FA, Hassan N, Barraclough
K, Bourke B, Fulcher J, Hollywood J, Hutchings A, Kyle V, Nott J,
et al: BSR and BHPR guidelines for the management of polymyalgia
rheumatica. Rheumatology. 49:186–190. 2010.PubMed/NCBI View Article : Google Scholar
|
|
46
|
Friedmann R, Feldman H and Nesher G:
Gallium-67 Scintigraphy in polymyalgia rheumatica. Clin Exp
Rheumatol. 25 (Suppl):S34–S35. 2007.PubMed/NCBI
|
|
47
|
O'Duffy JD, Wahner HW and Hunder GG: Joint
imaging in polymyalgia rheumatica. Mayo Clin Proc. 51:519–524.
1976.PubMed/NCBI
|
|
48
|
Jiménez-Palop M, Naredo E, Humbrado L,
Medina J, Uson J, Francisco F, García-Yebenes MJ and Garrido J:
Ultrasonographic monitoring of response to therapy in polymyalgia
rheumatica. Ann Rheum Dis. 69:879–882. 2010.PubMed/NCBI View Article : Google Scholar
|
|
49
|
Ruta S, Rosa J, Navarta DA, Saucedo C,
Catoggio LJ, Monaco RG and Soriano ER: Ultrasound assessment of new
onset bilateral painful shoulder in patients with polymyalgia
rheumatica and rheumatoid arthritis. Clin Rheumatol. 31:1383–1387.
2012.PubMed/NCBI View Article : Google Scholar
|
|
50
|
Cantini F, Niccoli L, Nannini C, Padula A,
Olivieri I, Boiardi L and Salvarani C: Inflammatory changes of hip
synovial structures in polymyalgia rheumatica. Clin Exp Rheumatol.
23:462–468. 2005.PubMed/NCBI
|
|
51
|
Dasgupta B, Cimmino MA, Kremers HM,
Schmidt WA, Schirmer M, Salvarani C, Bachta A, Dejaco C, Duftner C,
Jensen HS, et al: 2012 provisional classification criteria for
polymyalgia rheumatica: A European league against
Rheumatism/American College of Rheumatology Collaborative
Initiative. Arthritis Rheum. 64:943–954. 2012.PubMed/NCBI View Article : Google Scholar
|
|
52
|
Macchioni P, Boiardi L, Catanoso M,
Pazzola G and Salvarani C: Performance of the New 2012 EULAR/ACR
classification criteria for polymyalgia rheumatica: Comparison with
the previous criteria in a Single-Centre study. Ann Rheum Dis.
73:1190–1193. 2014.PubMed/NCBI View Article : Google Scholar
|
|
53
|
Kobayashi K, Nakagomi D, Kobayashi Y,
Ajima C, Hanai S, Koyama K and Ikeda K: Ultrasound of shoulder and
knee improves the accuracy of the 2012 EULAR/ACR provisional
classification criteria for polymyalgia rheumatica. Rheumatology
(Oxford). 61:1185–1194. 2022.PubMed/NCBI View Article : Google Scholar
|
|
54
|
Mackie SL, Koduri G, Hill CL, Wakefield
RJ, Hutchings A, Loy C, Dasgupta B and Wyatt JC: Accuracy of
musculoskeletal imaging for the diagnosis of polymyalgia
rheumatica: Systematic review. RMD Open. 13(e000100)2015.PubMed/NCBI View Article : Google Scholar
|
|
55
|
Manzo C, Milchert M, Isetta M, Natale M
and Castagna A: Early diagnosis in patients with polymyalgia
rheumatica: Is a fast track clinic a viable solution? Comment on:
‘Fast track clinic for early diagnosis of polymyalgia rheumatica:
Impact on symptom duration and prednisolone initiation’ by Frølund
et al. Joint Bone Spine 2021;88:105185. Joint Bone Spine.
88(105262)2021.PubMed/NCBI View Article : Google Scholar
|
|
56
|
Falsetti P, Acciai C, Volpe A and Lenzi L:
Ultrasonography in early assessment of elderly patients with
polymyalgic symptoms: A role in predicting diagnostic outcome?
Scand J Rheumatol. 40:57–63. 2011.PubMed/NCBI View Article : Google Scholar
|
|
57
|
Suzuki T, Yoshida R, Hidaka Y and Seri Y:
Proliferative Synovitis of the Shoulder Bursae is a key feature for
discriminating elderly onset rheumatoid arthritis mimicking
polymyalgia rheumatica from polymyalgia rheumatica. Clin Med
Insights Arthritis Musculoskelet Disord.
10(179544117745851)2017.PubMed/NCBI View Article : Google Scholar
|
|
58
|
Suzuki T, Yoshida R, Okamoto A and Seri Y:
Semiquantitative evaluation of extrasynovial soft tissue
inflammation in the shoulders of patients with polymyalgia
rheumatica and elderly-onset rheumatoid arthritis by power Doppler
ultrasound. Biomed Res Int. 2017(4272560)2017.PubMed/NCBI View Article : Google Scholar
|
|
59
|
Ottaviani S, Goossens J, Demaria L, Forien
M, Palazzo E and Dieudé P: Ultrasound shoulder assessment of
calcium pyrophosphate disease with suspected polymyalgia
rheumatica. Clin Exp Rheumatol. 38:1170–1175. 2020.PubMed/NCBI
|
|
60
|
Miceli MC, Zoli A, Peluso G, Bosello S,
Gremese E and Ferraccioli G: Baseline shoulder ultrasonography is
not a predictive marker of response to glucocorticoids in patients
with polymyalgia rheumatica: A 12-month followup study. J
Rheumatol. 44:241–247. 2017.PubMed/NCBI View Article : Google Scholar
|
|
61
|
Huwart A, Garrigues F, Jousse-Joulin S,
Marhadour T, Guellec D, Cornec D, Gouillou M, Saraux A and
Devauchelle-Pensec V: Ultrasonography and magnetic resonance
imaging changes in patients with polymyalgia rheumatica treated by
tocilizumab. Arthritis Res Ther. 20(11)2018.PubMed/NCBI View Article : Google Scholar
|
|
62
|
Fruth M, Buehring B, Baraliakos X and
Braun J: Use of Contrast-enhanced magnetic resonance imaging of the
pelvis to describe changes at different anatomic sites which are
potentially specific for polymyalgia rheumatica. Clin Exp
Rheumatol. 36 (Suppl):S86–S95. 2018.PubMed/NCBI
|
|
63
|
Fruth M, Seggewiss A, Kozik J,
Martin-Seidel P, Baraliakos X and Braun J: Diagnostic capability of
Contrast-Enhanced pelvic girdle magnetic resonance imaging in
polymyalgia rheumatica. Rheumatology (Oxford). 59:2864–2871.
2020.PubMed/NCBI View Article : Google Scholar
|
|
64
|
Salvarani C, Barozzi L, Boiardi L,
Pipitone N, Bajocchi GL, Macchioni PL, Catanoso M, Pazzola G,
Valentino M, De Luca C and Hunder GG: Lumbar interspinous bursitis
in active polymyalgia rheumatica. Clin Exp Rheumatol. 31:526–531.
2013.PubMed/NCBI
|
|
65
|
Mackie SL, Pease CT, Fukuba E, Harris E,
Emery P, Hodgson R, Freeston J and McGonagle D: Whole-body MRI of
patients with polymyalgia rheumatica identifies a distinct subset
with complete patient-reported response to glucocorticoids. Ann
Rheum Dis. 74:2188–2192. 2015.PubMed/NCBI View Article : Google Scholar
|
|
66
|
Cimmino MA, Parodi M, Zampogna G, Barbieri
F and Garlaschi G: Polymyalgia Rheumatica is associated with
extensor tendon tenosynovitis but not with synovitis of the hands:
A magnetic resonance imaging study. Rheumatology (Oxford).
50:494–499. 2011.PubMed/NCBI View Article : Google Scholar
|
|
67
|
Ochi J, Nozaki T, Okada M, Suyama Y,
Kishimoto M, Akaike G, Tasaki A, Ohde S, Saida Y and Yoshioka H:
MRI findings of the shoulder and hip joint in patients with
polymyalgia rheumatica. Mod Rheumatol. 25:761–767. 2015.PubMed/NCBI View Article : Google Scholar
|
|
68
|
Nakamura H, Kamada K, Tarumi M, Tanimura
S, Shibata Y and Horita T: Gadolinium-enhanced magnetic resonance
imaging in shoulders contributes accurate diagnosis and predicting
recurrence to patients with polymyalgia rheumatica. Clin Exp
Rheumatol. 39:84–90. 2021.PubMed/NCBI View Article : Google Scholar
|
|
69
|
Laporte JP, Garrigues F, Huwart A,
Jousse-Joulin S, Marhadour T, Guellec D, Cornec D,
Devauchelle-Pensec V and Saraux A: Localized Myofascial
inflammation revealed by magnetic resonance imaging in recent-onset
polymyalgia rheumatica and effect of tocilizumab therapy. J
Rheumatol. 46:1619–1626. 2019.PubMed/NCBI View Article : Google Scholar
|
|
70
|
Camellino D, Duftner C and Dejaco C: New
insights into the role of imaging in polymyalgia rheumatica.
Rheumatology (Oxford). 60:1016–1033. 2021.PubMed/NCBI View Article : Google Scholar
|
|
71
|
Slart RHJA: Writing group; Reviewer group;
Members of EANM Cardiovascular; Members of EANM Infection &
Inflammation; Members of Committees, SNMMI Cardiovascular; Members
of Council, PET Interest Group; Members of ASNC and EANM Committee
Coordinator. FDG-PET/CT(A) imaging in large vessel Vasculitis and
polymyalgia rheumatica: Joint procedural recommendation of the
EANM, SNMMI, and the PET Interest Group (PIG), and endorsed by the
ASNC. Eur J Nucl Med Mol Imaging. 45:1250–1269. 2018.PubMed/NCBI View Article : Google Scholar
|
|
72
|
Prieto-Peña D, Martínez-Rodríguez I,
Loricera J, Banzo I, Calderón-Goercke M, Calvo-Río V, González-Vela
C, Corrales A, Castañeda S, Blanco R, et al: Predictors of positive
18F-FDG PET/CT-scan for large vessel vasculitis in
patients with persistent polymyalgia rheumatica. Semin Arthritis
Rheum. 48:720–727. 2019.PubMed/NCBI View Article : Google Scholar
|
|
73
|
Yuge S, Nakatani K, Yoshino K and Koyama
T: Diagnosing polymyalgia rheumatica on 18F-FDG PET/CT:
Typical uptake patterns. Ann Nucl Med. 32:573–577. 2018.PubMed/NCBI View Article : Google Scholar
|
|
74
|
van der Geest KSM, Treglia G, Glaudemans
AWJM, Brouwer E, Jamar F, Slart RHJA and Gheysens O: Diagnostic
Value of [18F]FDG-PET/CT in polymyalgia rheumatica: A
systematic review and meta-analysis. Eur J Nucl Med Mol Imaging.
48:1876–1889. 2021.PubMed/NCBI View Article : Google Scholar
|
|
75
|
Kaneko K, Suematsu E, Miyamura T and
Ishioka H: Differences of Articular and Extra-articular involvement
in polymyalgia rheumatica: A comparison by whole-body FDG-PET/CT.
Mod Rheumatol. 30:358–364. 2020.PubMed/NCBI View Article : Google Scholar
|
|
76
|
Rehak Z, Sprlakova-Pukova A, Kazda T,
Fojtik Z, Vargova L and Nemec P: 18F-FDG PET/CT in
polymyalgia rheumatica-a pictorial review. Br J Radiol.
90(20170198)2017.PubMed/NCBI View Article : Google Scholar
|
|
77
|
Sondag M, Verhoeven F, Prati C, Guillot X,
Blagosklonov O, Boulahdour H and Wendling D: Involvement of Pubic
Symphysis in polymyalgia rheumatica: 18-FDG PET/CT Evaluation.
Joint Bone Spine. 84:635–636. 2017.PubMed/NCBI View Article : Google Scholar
|
|
78
|
Takahashi H, Yamashita H, Kubota K, Miyata
Y, Okasaki M, Morooka M, Takahashi Y, Kaneko H, Kano T and Mimori
A: Differences in Fluorodeoxyglucose positron emission
Tomography/computed tomography findings between elderly onset
rheumatoid arthritis and polymyalgia rheumatica. Mod Rheumatol.
25:546–551. 2015.PubMed/NCBI View Article : Google Scholar
|
|
79
|
Wendling D, Sondag M, Giraud N, Chouk M,
Boulahdour H, Prati C and Verhoeven F: Muscle involvement on
18F-FDG PET-CT in polymyalgia rheumatica. A controlled
retrospective study of 101 patients. Joint Bone Spine. 87:225–228.
2020.PubMed/NCBI View Article : Google Scholar
|
|
80
|
Chuang TY, Hunder GG, Ilstrup DM and
Kurland LT: Polymyalgia rheumatica: A 10-year epidemiologic and
clinical study. Ann Intern Med. 97:672–679. 1982.PubMed/NCBI View Article : Google Scholar
|
|
81
|
Healey LA: Long-Term Follow-up of
polymyalgia rheumatica: evidence for Synovitis. Semin Arthritis
Rheum. 13:322–328. 1984.PubMed/NCBI View Article : Google Scholar
|
|
82
|
Jones JG and Hazleman BL: Prognosis and
management of polymyalgia rheumatica. Ann Rheum Dis. 40:1–5.
1981.PubMed/NCBI View Article : Google Scholar
|
|
83
|
Bird HA, Esselinckx W, Dixon SAJ, Mowat AG
and Wood PH: An evaluation of criteria for polymyalgia rheumatica.
Ann Rheum Dis. 38:434–439. 1979.PubMed/NCBI View Article : Google Scholar
|
|
84
|
Matteson EL and Dejaco C: Polymyalgia
rheumatica. Ann Intern Med. 166:ITC65–ITC80. 2017.PubMed/NCBI View Article : Google Scholar
|
|
85
|
Nothnagl T and Leeb BF: Diagnosis,
differential diagnosis and treatment of polymyalgia rheumatica.
Drugs Aging. 23:391–402. 2006.PubMed/NCBI View Article : Google Scholar
|
|
86
|
Nesher G: Polymyalgia rheumatica-diagnosis
and classification. J Autoimmun. 48-49:76–78. 2014.PubMed/NCBI View Article : Google Scholar
|
|
87
|
Dejaco C, Duftner C, Cimmino MA, Dasgupta
B, Salvarani C, Crowson CS, Maradit-Kremers H, Hutchings A,
Matteson EL and Schirmer M: International Work Group for PMR and
GCA. Definition of remission and relapse in polymyalgia rheumatica:
Data from a literature search compared with a Delphi-Based expert
consensus. Ann Rheum Dis. 70:447–453. 2011.PubMed/NCBI View Article : Google Scholar
|
|
88
|
Dejaco C, Singh YP, Perel P, Hutchings A,
Camellino D, Mackie S, Abril A, Bachta A, Balint P, Barraclough K,
et al: 2015 Recommendations for the management of polymyalgia
rheumatica: A European league against Rheumatism/American College
of Rheumatology Collaborative Initiative. Ann Rheum Dis.
74:1799–1807. 2015.PubMed/NCBI View Article : Google Scholar
|
|
89
|
Ruediger C, Nguyen L, Black R, Proudman S
and Hill C: Efficacy of methotrexate in polymyalgia rheumatica in
routine rheumatology clinical care. Intern Med J. 50:1067–1072.
2020.PubMed/NCBI View Article : Google Scholar
|
|
90
|
Caporali R, Cimmino MA, Ferraccioli G,
Gerli R, Klersy C, Salvarani C and Montecucco C: Prednisone plus
methotrexate for polymyalgia rheumatica: A randomized,
Double-blind, Placebo-controlled trial. Ann Intern Med.
141:493–500. 2004.PubMed/NCBI View Article : Google Scholar
|
|
91
|
Marsman DE, Bolhuis TE, den Broeder N, den
Broeder AA and van der Maas A: PolyMyalgia rheumatica treatment
with methotrexate in optimal dose in an early disease phase (PMR
MODE): Study protocol for a multicenter double-blind placebo
controlled trial. Trials. 23(318)2022.PubMed/NCBI View Article : Google Scholar
|
|
92
|
De Silva M and Hazleman BL: Azathioprine
in giant cell arteritis/polymyalgia rheumatica: A double-blind
study. Ann Rheum Dis. 45(136)1986.PubMed/NCBI View Article : Google Scholar
|
|
93
|
Lee JH, Choi ST, Kim JS, Yoon BY, Kwok SK,
Kim HS, Kim YS, Song JS, Lee SH and Kim HR: Clinical
characteristics and prognostic factors for relapse in patients with
polymyalgia rheumatica (PMR). Rheumatol Int. 33:1475–1480.
2013.PubMed/NCBI View Article : Google Scholar
|
|
94
|
Lally L, Forbess L, Hatzis C and Spiera R:
Brief report: A Prospective open-label phase IIa trial of
tocilizumab in the treatment of polymyalgia rheumatica. Arthritis
Rheumatol. 68:2550–2554. 2016.PubMed/NCBI View Article : Google Scholar
|
|
95
|
Devauchelle-Pensec V, Berthelot JM, Cornec
D, Renaudineau Y, Marhadour T, Jousse-Joulin S, Querellou S,
Garrigues F, de Bandt M, Gouillou M and Saraux A: Efficacy of
First-line tocilizumab therapy in early polymyalgia rheumatica: A
prospective longitudinal study. Ann Rheum Dis.
75(1506)2016.PubMed/NCBI View Article : Google Scholar
|
|
96
|
Antiochos B: Tocilizumab as a novel
therapy for steroid-dependent polymyalgia rheumatica. JAMA.
328:1047–1048. 2022.PubMed/NCBI View Article : Google Scholar
|
|
97
|
Mori S and Koga Y:
Glucocorticoid-resistant polymyalgia rheumatica: Pretreatment
characteristics and tocilizumab therapy. Clin Rheumatol.
35(1367)2016.PubMed/NCBI View Article : Google Scholar
|
|
98
|
Bonelli M, Radner H, Kerschbaumer A, Mrak
D, Durechova M, Stieger J, Husic R, Mandl P, Smolen JS, Dejaco C
and Aletaha D: Tocilizumab in patients with new onset polymyalgia
rheumatica (PMR-SPARE): A phase 2/3 randomised controlled trial.
Ann Rheum Dis. 81:838–844. 2022.PubMed/NCBI View Article : Google Scholar
|
|
99
|
A 2-Week Single-Blind, Randomized, 3-Arm
Proof of Concept Study of the Effects of Secukinumab (Anti-IL17
MAb), Canakinumab (Anti-IL-1 b MAb), or Corticosteroids on Initial
Disease Activity Scores in Patients with PMR, Followed By an
Open-Label Extension to Assess Safety and Effect Duration-ACR
Meeting Abstracts Available from: https://acrabstracts.org/abstract/a-2-week-single-blind-randomized-3-arm-proof-of-concept-study-of-the-effects-of-secukinumab-anti-il17-mab-canakinumab-anti-il-1-b-mab-or-corticosteroids-on-initial-disease-activity-scores-in-p/.
Accessed on 31 August 2022.
|
|
100
|
Koster MJ, Crowson CS, Giblon RE, Jaquith
JM, Duarte-García A, Matteson EL, Weyand CM and Warrington KJ:
Baricitinib for relapsing giant cell arteritis: A prospective
open-label 52-week pilot study. Ann Rheum Dis. 81:861–867.
2022.PubMed/NCBI View Article : Google Scholar
|
|
101
|
Marsman DE, den Broeder N, van den Hoogen
FHJ, den Broeder AA and van der Maas A: Efficacy of rituximab in
patients with polymyalgia rheumatica: A double-blind, randomised,
placebo-controlled, proof-of-concept trial. Lancet Rheumatol.
3:E758–E766. 2011.
|
|
102
|
Bolhuis T: 1-year results of treatment
with rituximab in polymyalgia rheumatica: an extension study of a
randomised double-blind placebo-controlled trial. Lancet
Rheumathol. 5:e208–e214. 2023.
|
|
103
|
Dasgupta B, Unizony S, Warrington KJ,
Lazar JS, Giannelou A, Nivens C, Akinlade B, Wong W, Lin Y,
Buttgereit F, et al: LB0006 SARILUMAB in patients with relapsing
polymyalgia rheumatica: A phase 3, multicenter, randomized, double
blind, placebo controlled trial (SAPHYR). Ann Rheumatic Diseases.
81:210–211. 2022.
|
|
104
|
Castañeda S, García-Castañeda N,
Prieto-Peña D, Martínez-Quintanilla D, Vicente EF, Blanco R and
González-Gay MA: Treatment of polymyalgia rheumatica. Biochem
Pharmacol. 165:221–229. 2019.PubMed/NCBI View Article : Google Scholar
|