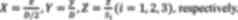Introduction
Restoring the motor function of the paralyzed limbs
has always been an important aim of neuroscience research. Motor
dysfunction of the limbs and the trunk below an injured segment
after spinal cord injury (SCI) caused by falling from high
altitude, traffic accidents and disease (1), severely affects the quality of life
of individuals (2).
Spinal neural networks serve an important role in
the execution of locomotion (3).
Grill (4) pursued an approach to
activate spinal neural networks: Restoration of function by
electrical activation of higher-order interneurons directly instead
of using synaptic activation of spinal neurons indirectly. These
networks of spinal neurons, known as spinal locomotor central
pattern generators (CPGs), are normally modulated by supraspinal
and peripheral inputs, but retain the intrinsic ability to regulate
the rhythmical and reciprocal activation pattern of hindlimb
muscles after SCI, even in the absence of supraspinal and/or
afferent input (5,6). Thus, the locomotor CPG is a dedicated
neural network that generates regulatory signals for movements and
contains signals that activate different motor neurons in the
appropriate sequence and intensity to generate motor patterns
(7-10).
The coordination and control of damaged muscles can be effectively
restored by electrical stimulation of the CPG. Thus, intraspinal
microstimulation (ISMS), known as an electrical stimulation therapy
for motor function recovery consisting of stimulation through fine,
hair-like microwires targeted at the ventral horn of the
lumbosacral enlargement, has been proved effective for restoring
standing and walking following SCI (11-13).
The implantation of intraspinal microwires and chronic applications
of ISMS is generally well tolerated by spinal cord tissue (14). The mechanisms of action of ISMS and
the clinical application prospect of ISMS have been widely
discussed (15,16). Focal ISMS has been observed to
activate sets of interneurons that projected to, and activated,
motor neuron pools to generate synergistic motion (17-19).
Gaunt et al (20) found
that ISMS at a single point in the grey matter of the spinal cord
could activate afferent terminals along the entire length of the
lumbosacral enlargement. By activating the neural networks within
the lumbosacral spinal cord, ISMS can generate inherently
synergistic movements that are weight-bearing and fatigue-resistant
(21,22). Through targeted activation of
hindlimb locomotor-related networks, ISMS has been used to restore
walking in SCI cats (23).
Accurate and robust control of the multi-joint movement can be
achieved by focally stimulating the target muscle motor pools
within the spinal cord (24).
Holinski et al (25)
developed a feedback-driven ISMS system to enhance the
functionality of stepping by reducing muscle fatigue and to produce
synergistic movements by activating neural networks in the spinal
cord. A fully intradural intraspinal microstimulation implant based
on wireless floating microelectrode arrays has been exploited and
the long-term stability of the implant in the spinal cord assessed
(26). Barthélemy et al
(27) focused on the use of the
same polarity of the pulse for scanning stimulation of various loci
of the spinal cord and described the distribution of various types
of hindlimb responses evoked by ISMS in spinal cats. However, they
did not consider the effect of the pulse polarity of the stimulus
on the movement patterns of the bilateral hindlimbs. If different
movement patterns can be induced by changing the polarity of the
stimulation signal, then combinations of movement patterns can be
generated by editing the positive and negative pulse signals to
achieve a reduction both the number of stimulation electrodes and
secondary injury to the spinal cord.
The present study aimed to investigate the effects
of the different pulse polarity of stimulus on the movement
patterns of the bilateral hindlimbs when applied to the same site
and further developed a gait function reconstruction training with
a single site stimulation strategy. It also conducted a 4-week
evaluation of the effects of a single site stimulation training
protocol.
Materials and methods
Experimental animals. All rats were purchased from
Laboratory Animal Center, Nantong University School of Medicine
(Nantong, China). Rats were kept in a temperature- and
humidity-controlled environment with a 12:12-h light-dark cycle and
allowed free access to food and water. The housing conditions
adhered to specific requirements, including a temperature range of
18-26˚C and relative humidity of 40-70%. Typically, the rat housing
temperature was 1-2˚C warmer than the ambient temperature, while
the humidity was 5-10% higher. The noise level did not exceed 85
decibels, and the concentration of ammonia was <20 ppm. The lab
was kept dry and ventilated. A total of 60 Sprague Dawley rats
(age, 10 weeks; weight, 220-280 g) that provided valid experimental
data were used. They were randomly divided into four experimental
groups: Intact group (n=6; 3 males; 3 females), test group (n=30;
15 males, 15 females; exploration of the location of those special
sites with the ability to activate the CPG neural network and
appropriate stimulus parameters, not used for comparison), SCI
group (n=12; 6 males, 6 females incomplete SCI due to weight
impact) and SCI + ISMS (n=12; 6 males, 6 females; incomplete SCI
followed by ISMS training: 20 min/day, 5 days/week, 4 weeks;). At
the end of all experiments, 62 rats were sacrificed, including two
rats in the SCI + ISMS group that were excluded from the present
study due to electrode detachment problems.
Determination of the location of
special sites in the spinal cord
All survival surgical procedures were performed
under aseptic conditions. The animal's body temperature and
respiratory rate were monitored simultaneously. The operating table
was kept at a constant temperature of 37˚C to maintain the
physiologic body temperature of the rats. A total of 30 rats in the
test group were anesthetized with an intraperitoneal injection of
sodium pentobarbital (30 mg/kg) (28). A midline incision was made over the
skin in the back, exposing the thoracic vertebra (VT)9 and
VT12-lumbar vertebra (VL1). The L1-L5 spinal segments (VT12-VL1
vertebral segments) were identified and the skin was cut along the
direction of the spine. The corresponding segments of the spinal
cord and vertebrae are shown in Fig.
1A. The lamina surface muscles were removed by a hemostat to
expose the lamina. The length of each vertebra segment which was
denoted as Si(i=1,2,3) and the transverse
diameter of the spinal lumbar enlargement (L2 spinal segment) which
was denoted as D were measured. The locations of the motor
function points in the lumbosacral spinal cord are described as the
positions in the corresponding vertebral segment, denoted as
x, y and z. The intersection of the posterior
median sulcus of the spinal cord and the cephalic side of the
spinal segment was taken as the coordinate origin (29). The mediolateral direction X,
dorsoventral direction Y, and rostrocaudal direction
Z were normalized by
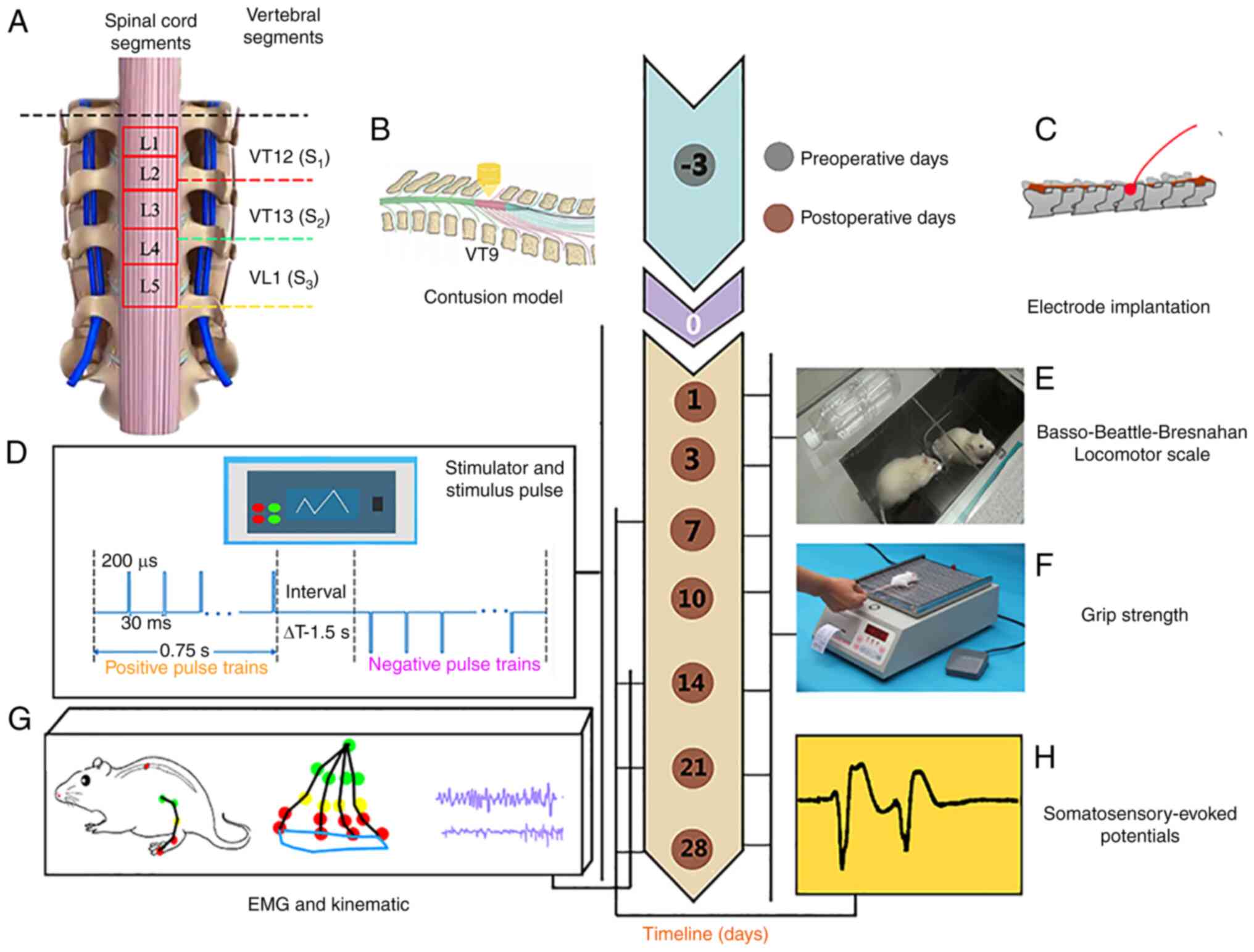 | Figure 1Experimental design and timeline. (A)
The corresponding segments of the spinal cord and vertebrae. The
length of each vertebra segment (VT12, VT13, VT 1) was denoted as
Si(i=1,2,3). (B) A schematic of preparing
the contusion model. (C) Implanted electrodes at the special site
right of the midline in the SCI + ISMS group three days before the
training. (D) A constant alternating positive and negative pulse
was delivered to the special site right of the midline in the SCI +
ISMS group for four weeks. During the ISMS training, (E) the
Basso-Beattie-Bresnahan scale, (F) grip strength, (G) detailed
kinematics and EMG and (H) somatosensory-evoked potentials analyses
were used to assess the recovery of motor function. Color markers
were attached bilaterally to bony landmarks at the iliac crest
(green), hip (green), knee (yellow), ankle (red) and fifth
metatarsal (red) to collect the hindlimb movements. VT, thoracic
vertebra; VT, lumbar vertebra; SCI, spinal cord injury; ISMS,
intra-spinal micro-stimulation; EMG, electromyography. |
ISMS using a stimulus isolator (ISO-Flex; A.M.P.I.)
was applied to the spinal cord with a PFA-Coated Tungsten Wire (A-M
Systems, LLC) electrode except for the tip and the electrode that
served as a reference in the electrical stimulation, which was
inserted into the adjacent muscle. The stimulation was applied to
one point at a time in different parts of the L1-L5 spinal cord
segments and from the midline to 2.5 or 3 mm on either side. The
electrode was held in a microdrive and was mounted on a stereotaxic
manipulandum with X and Z coordinates. Stimulation was performed on
the mediolateral displacements (where 0 was on the midline) along
the X axis at intervals of 200 µm and dorsoventral (where 0 was on
the dorsal surface) along the Z axis at intervals of 500 µm.
Stimulation tracks went from the dorsal surface to 1.8-2.1 mm deep
in 300 µm intervals along the Y axis. Two patterns of movement were
defined here: Pattern 1, left hindlimb extension and right hindlimb
flexion; pattern 2, left hindlimb flexion and right hindlimb
extension (The combination of the two patterns can realize a
complete gait cycle movement.) A constant pulse of positive or
negative currents (200 µs pulse width, 33 Hz frequency) was
delivered. Only when the reversal of the pulse polarity of the
stimulus was performed and the two patterns switched, can the point
be confirmed as a special site. The duration of the entire surgery
was 2-3 h. At the end of the surgery, euthanasia was performed by
intraperitoneal injection of sodium pentobarbital (150 mg/kg) to
reduce the pain of the animal. At five minutes after the
administration of anesthesia, the rats' pupils were observed with a
flashlight, and the absence of a constriction response was an
indication that the animal had succumbed. To confirm that the rats
were deceased, professionals were consulted to ensure compliance
with ethical and standard procedures.
Efficacy evaluation of special sites
activation with ISMS after SCI
Preparation of SCI model. To establish an
incomplete SCI model, laminectomy was performed at the selected
level (VT9). The vertebra was stabilized using a clamp system and
an impactor (MASCIS Impactor Model-III; W.M. Keck Center for
Collaborative Neuroscience, The State University of New Jersey,
Piscataway, NJ, USA) was used to apply injury of desired severity.
In the experiment, injury of moderate severity was applied by using
the stimulus parameters described below. The diameter of the impact
head was 2.5 mm, the weight 10 g and the height 6.25 mm.
Electrode implantation
Rats in the SCI + ISMS group were implanted with
electrodes to chronically record muscle activity [electromyography
(EMG)]. Pairs of PFA-Coated Tungsten Wires were sewn into the
selected hindlimb muscles [the lateral gastrocnemius (LG) and the
tibialis anterior (TA)] for dual electrode recordings. The
stimulating electrode was attached to the special point right of
the midline in the spinal cord by biocompatible dental cement [Root
Canal Fitting Materials MTA; Longly Biotechnology (Wuhan) Co.,
Ltd.]. One additional wire that served as a reference electrode for
use in electrical stimulation was sewn into the adjacent muscle. At
the end of the experiment, the muscles and skin were sutured and
thoroughly washed with saline. After the surgery, the animal was
placed in an insulated incubator until it regained consciousness.
The rats were manually urinated and injected with gentamicin
sulfate (3 mg/kg) for anti-inflammatory treatment three days after
surgery. Careful postoperative care was given for the next four
weeks. However, two rats were excluded from this group because
their implanted electrodes became dislodged on the first and third
days, respectively. The SCI + ISMS group consisted of 12 rats and
was euthanized four weeks later via intraperitoneal injection of
sodium pentobarbital (150 mg/kg). The method used for verifying
mortality is similar to the one aforementioned.
Locomotor training with ISMS
The day after the spinal cord surgery (i.e.,
post-operative day 1), rats in the SCI + ISMS groups underwent a
four-week locomotor training period with ISMS. A constant
alternating positive and negative pulse was delivered according to
the following pulse train stimulation parameters: 200 µsec pulse
width; 33 Hz frequency; 30 msec interval of the pulse of the same
polarity; 1.5 sec interval between positive and negative pulse
trains and 3 sec duration of stimulation.
Assessment methods
Evaluation of motor function recovery combined
behavioral, kinesiological and physiological approaches (Fig. 1). The initial assessment for
movement characteristics used the Basso-Beattie-Bresnahan (BBB)
scale (30), EMG and kinematics
(29) and grip strength (31,32)]
to precisely assess hindlimb function rebuilding.
Somatosensory-evoked potentials (SEPs) (33-35)
were used to detect electrophysiological function for evaluating
the recovery of spinal pathways.
BBB locomotor scale
To evaluate the motor function of hindlimbs, a
21-point BBB locomotion scale was used based on the movement of
joints and the placement of paws and coordination of forepaw and
hindlimbs on days 1, 3, 7, 10, 14, 21 and 28 after surgery.
EMG and kinematic testing
procedures
EMG was collected using a Keypoint Portable
apparatus (Dantec™ Keypoint® Focus; NEUROLITE
AG). The EMG data set was obtained from bilateral recordings in the
TA (ankle flexor) and the LG (ankle extensor). TA and LG with the
best signal-to-noise ratio were retained for analysis.
Kinematic data were obtained using the Open MV CAM
(OpenMV4 CAM H7; OpenMV, LLC) system without electrical
stimulation. Color markers were attached bilaterally to bony
landmarks at the iliac crest, hip, knee, ankle and fifth metatarsal
to collect the hindlimb movements.
Grip strength testing procedures
The grip strength in all groups was determined
without electrical stimulation by a grip strength meter (Stoelting
Co.). The maximum grip strength was determined as the force
recorded just before the rat released the bar. This was repeated
three times a day and the maximum value was recorded.
SEPs testing procedures
Electromyograph and evoked potential equipment
(Dantec™ Keypoint® Focus; NEUROLITE AG) was
used to measure SEPs. The negative electrode of the stimulation
electrode was inserted into the common peroneal nerve of the left
hind limb of the rat (lateral gastrocnemius muscle) and the
positive electrode was inserted ~0.8 cm distal to the negative
electrode. Electrical stimulation was conducted at a frequency of 3
Hz, pulse intensity between 2 and 4 mA, a pulse width of 0.1 msec
and 200 repetitions. The stimulation intensity was set to make the
toes of the bilateral hindlimbs twitch slightly. The stimulation
induced electrical activity from the lateral gastrocnemius muscle
passing through the peripheral and central nervous system when
nerve fiber action potential transmission, until the synaptic
potentials. The recording electrode was placed in the sensory
cortex of the rat brain and the reference electrode was placed
under the mucosa of the hard palate of the rat. The ground
electrode was placed between the stimulating electrode and the
recording electrode. SEPs collected in the present study were
composed of a forward wave (P) and a negative wave (N). The latency
and amplitude of SEPs were recorded before the operation and on
days 7, 14, 21 and 28 after the operation. Spinal cord conduction
capacity (latency) and synchronous discharge quantity (amplitude)
of the sensory or motor nervous system can indirectly reflect the
recovery ability of motor function after SCI (35).
Statistical analysis
Statistical tests were performed with SPSS 24.0
software (IBM Corp.). To compare the effects of special site
stimulation using ISMS on the recovery of hindlimbs movement
two-way mixed [group (SCI + ISMS/SCI) x time (days 1, 3, 7, 10, 14,
21 and 28 after surgery)] ANOVA with Bonferroni's multiple
comparison adjustment method was performed on BBB locomotor scales
following ISMS. Two-way mixed [group (SCI + ISMS/SCI) x time (days
3, 7, 10, 14, 21 and 28 after surgery)] ANOVA with Bonferroni's
multiple comparison adjustment methods was performed on grip
strength after ISMS. Two-way mixed [group (SCI + ISMS/SCI) x time
(days 7, 14, 21 and 28 after surgery)] ANOVA with Bonferroni's
multiple comparison adjustment methods was performed on SEPs
following ISMS. Two-way mixed [group (SCI + ISMS/SCI) x iEMG data
(TA, LG)] ANOVA with Bonferroni's multiple comparison adjustment
methods was performed. Paired t-tests were used to assess the
activation current threshold of flexion and extension muscles and
negative and positive pulses which evoked two movement patterns.
Group data in the graphs were the mean ± the standard deviation.
Data in Table I are presented as
the mean. The collected gait data was processed by processing
software (Processing 3.5.3; Processing Foundation) to generate
stick diagrams and trajectories.
 | Table IiEMG of the intact, SCI + ISMS and
SCI groups of TA and LG of right legs in rats when the negative and
positive pulses were applied to the right special site. |
Table I
iEMG of the intact, SCI + ISMS and
SCI groups of TA and LG of right legs in rats when the negative and
positive pulses were applied to the right special site.
| Time | iEMG (mV) | Intact | SCI + ISMS | SCI |
|---|
| Day 14 | TA | 7.16 | 1.53b | 1.12b |
| | LG | 7.83 | 0.95a,b | 0.31a,b |
| Day 21 | TA | 7.13 | 3.26a,b | 2.25a,b |
| | LG | 7.84 | 4.15a,b | 1.13a,b |
| Day 28 | TA | 7.17 | 7.13a,b | 5.97a,b |
| | LG | 7.81 | 7.72a,b | 6.02a,b |
Results
Special sites in the spinal cord
A total of 30 rats in the test group were used to
explore and verify the special sites.
Location of the special sites
Multiple sites were found on each side of the lumbar
enlargement on which electrical stimulation evoked alternating
activity in groups of flexors and extensors of the bilateral
hindlimbs. However, two optimal sites could be identified as
special sites, as the movement pattern immediately switched once
the pulse polarity of the stimulus was switched. Longitudinal and
transverse sections of VT12-VL1 vertebral segments were drawn, with
grey dots representing the points of ISMS stimulation, purple dots
representing the locations that can evoke alternating left-right
movement and red and green dots representing the locations where
the special site appeared. The red dot was the optimal site in the
left side of the spinal cord and the green dot was the optimal site
in the right side of the spinal cord (Fig. 2A and B). The three-dimensional map depicting
the location of special sites which could activate movement pattern
1 and pattern 2 of the bilateral hindlimbs was also drawn (Fig. 2C). According to the normalized
coordinates of intact rats (Fig.
2D), the special sites were located in the L2 spinal segment
and described as the left special site (PL) and the
right special site (PR). PR was determined as
(X, Y, Z)=(0.377±0.196, 0.619±0.019,
0.780±0.143) and PL was determined as (X,
Y, Z)=(-0.385±0.182, 0.638±0.020, 0.779±0.147).
Furthermore, when negative pulse was applied to PR,
pattern 1 was induced and when the polarity of the stimulus was
switched from negative pulse to positive pulse and application
continued to the same site, pattern 2 was induced. Therefore,
considering the above phenomena and the criteria defined in the
methods, PR and PL were identified as optimal
sites and the target special sites.
Moreover, it was found that the position of the site
did not change when electrical stimulation was applied to the
special site by the stimulating electrode in the SCI + ISMS groups
in the following four weeks after the incomplete SCI caused by
weight impact.
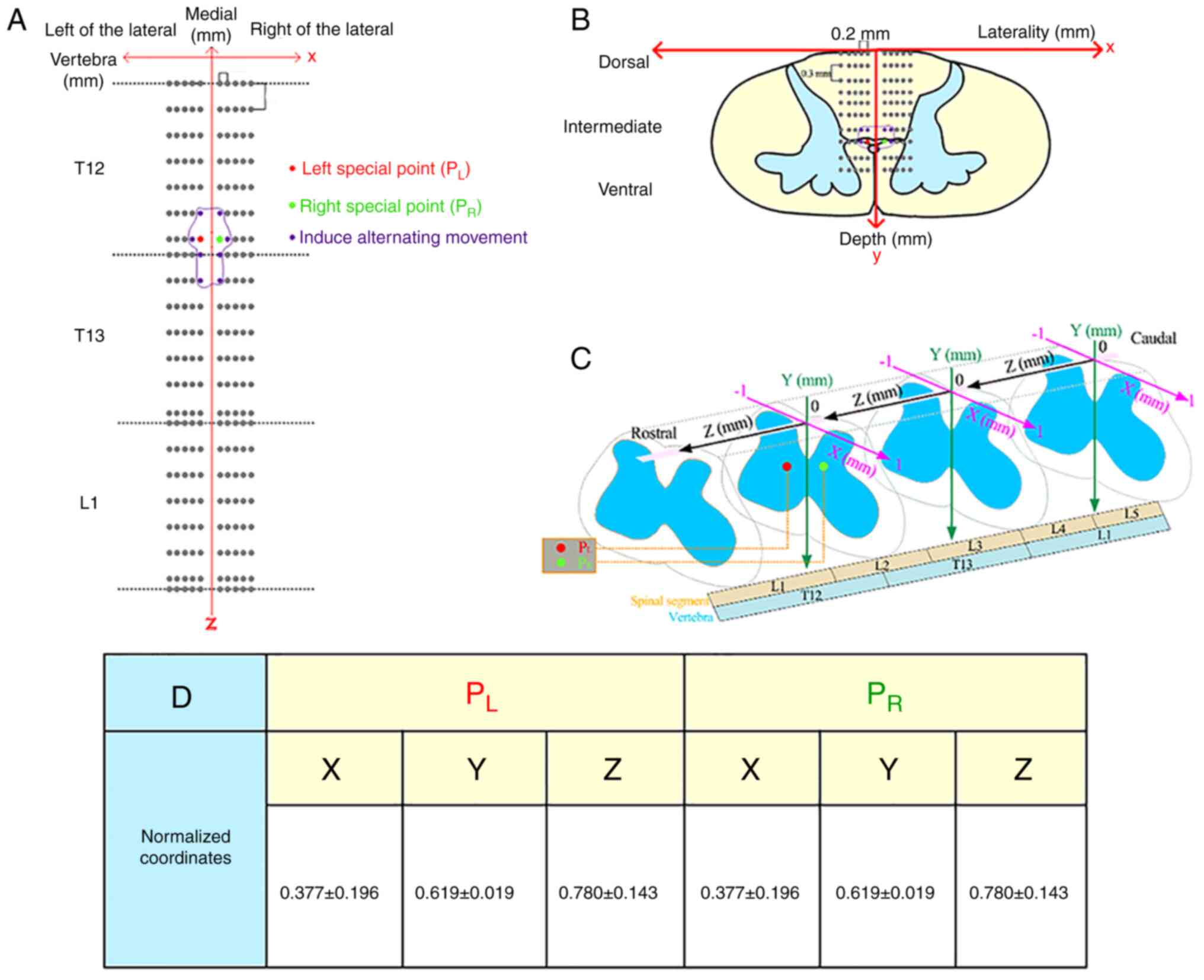
Amplitude of stimulus current
The amplitudes of the current used in 30 rats to
induce alternation of hindlimbs by stimulating the special site
were measured as was the changed current when the polarity of the
current was switched to reverse the movement patterns of the
bilateral hindlimbs. Paired t-tests were used to assess the
activation current threshold of flexor and extensor muscles and
negative and positive pulses which evoked two movement patterns
(Fig. 3B and D). Fig.
3B depicted the statistical results of the threshold currents
for activating the flexor neurons (blue box bubble plot) and
extension neurons (purple box bubble plot). Fig. 3D depicted the statistical results
of the threshold current for negative pulses (brown box bubble
plot) and positive pulses (darker brown box bubble plot) when the
movement patterns were switched. The activation threshold current
of the flexor was 210±55 µA. The activation threshold current of
the extensor was 113±26 µA. The results showed that the activation
threshold currents of flexor and extensor muscles were
significantly different [t=12.179; P=0.001; 95% confidence interval
(CI) 80.66-113.49] through the measurement analysis. There were
also significant differences between the negative and positive
pulse amplitudes which evoked two movement patterns. The activation
threshold current of the negative pulse was -228±44 µA. The
activation threshold current of the positive pulse was 282±57 µA.
The results showed that the activation threshold currents of
evoking two movement patterns induced by negative pulse and
positive pulse were significant (t=-26.088; P=0.001; 95% Cl-550.35-
-469.81).
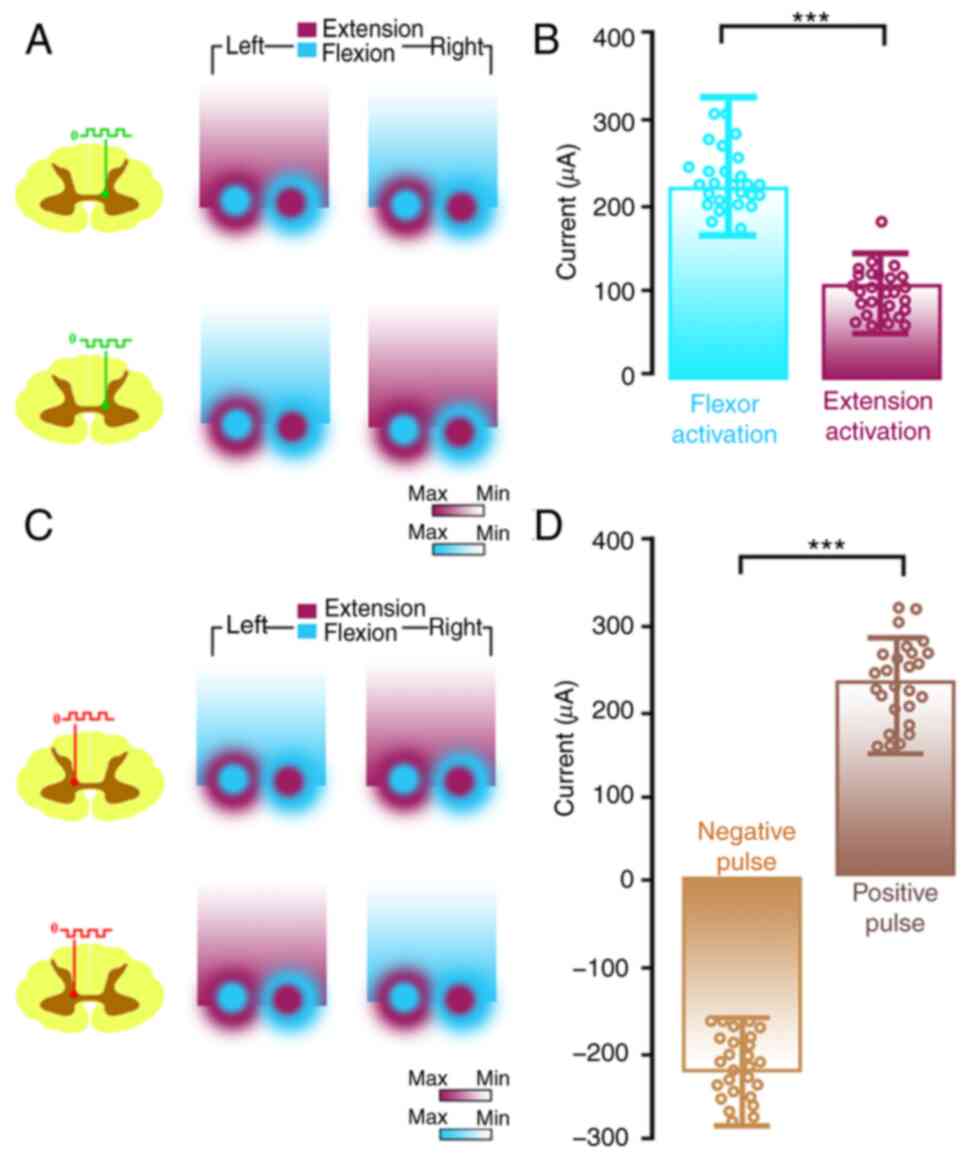
Relationship between stimulation
signal/site and the bilateral hindlimbs movement patterns
Combined with ISMS, four specific properties were
found indicating the relationships between the polarity of
stimulus, the special sites and the movement patterns.
First, the coordinates of the two special sites
indicated that they were symmetrical on the posterior median sulcus
and yet triggered opposite movement patterns in the left and right
hindlimbs once the polarity of the stimulus was switched.
Second, there was a reciprocal characteristic
between the two symmetrical special sites. That is, when the
negative pulse is applied to PL, pattern 2 was induced
(Fig. 4A); then when the same
pulse polarity of stimulus was applied to PR, pattern 1
is induced (Fig. 4B). When the
polarity of the stimulus signal was positive, the movement patterns
exchanged correspondingly, as shown in Fig. 4C and F.
Third, there was another reciprocal characteristic
of both special sites. When the opposite pulse polarity of the
stimulus was applied to the same special site, it induced the
opposite movement pattern (Fig. 3A
and C). When the negative pulse
was applied to PL, pattern 2 was induced (Fig. 4D) and when the reverse polarity was
applied to PL, pattern 1 was induced (Fig. 4G). That was the same for
PR (Fig. 4E and
H).
Fourth, there is a clear and certain relationship
between the pulse polarity and the movement patterns of the
bilateral hindlimbs. That is, the positive pulse applied to
PR/PL faithfully induced the movement of
pattern 2/pattern 1. The negative pulse applied to
PL/PR faithfully induced the movement of
pattern 1/pattern 2. Under these circumstances, gait control can be
obtained by applying the relevant pulse polarity of the stimulus to
the corresponding special site(PR/PL).
Evaluation of motor function
recovery
Rehabilitation training was designed to evaluate the
effect of motor function regeneration and a four-week training for
rats in the SCI + ISMS group was performed to restore the motor
function of the bilateral hindlimbs using ISMS applied to the
special site. Alternating positive and negative pulse trains were
continuously delivered to the PR during the training
sessions using a signal generator to produce the movements of
pattern 1 and pattern 2. Meanwhile, the motor speed of the
bilateral hindlimbs of rats was artificially controlled by the time
interval of the stimulus pulse. Evaluation of motor function
recovery combined the BBB scale, EMG and kinematics, grip strength
and SEPs (Fig. 5A-E).
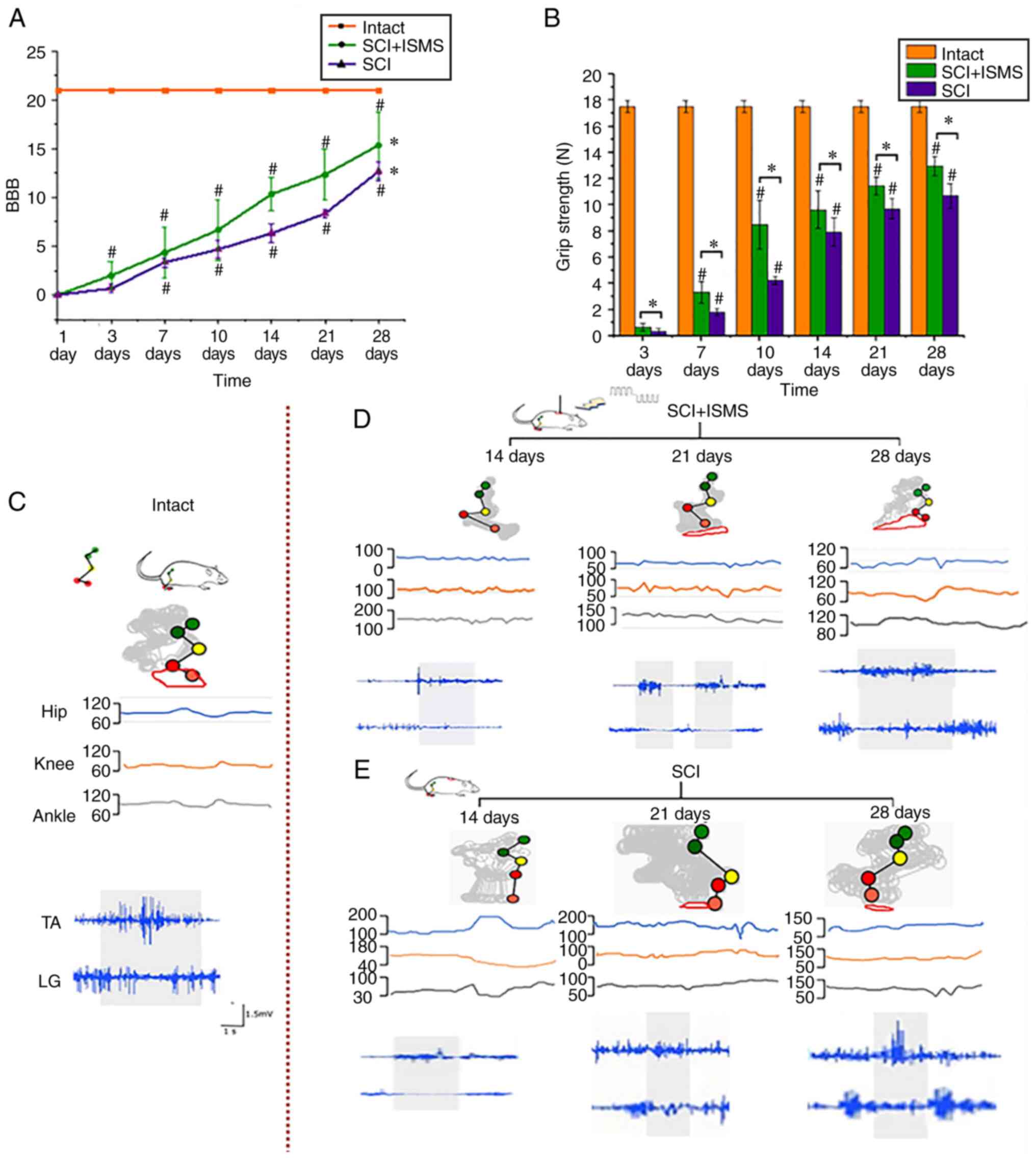 | Figure 5Results of the assessment of motor
function after SCI based on BBB scale, EMG, kinematics, grip
strength. (A) Effects of ISMS training on SCI rats assessed by BBB
scale on days 1, 3, 7, 10, 14, 21 and 28 days after surgery. In
each measurement, the simple effect was significant in both groups
and became more significant over time (P<0.001). The final
recovery effect in the SCI + ISMS was superior to that in the SCI
group *P<0.05, vs. group; #P<0.05, vs.
previous time point. (B) Time course of grip strength of the SCI +
ISMS and SCI groups. *P<0.05, vs. group;
#P<0.05, vs. previous time point. (C) Representative
illustrations of EMG and kinematic features in the intact group.
(D) Representative illustrations of EMG and kinematic features at
14, 21 and 28 days in the intact SCI + ISMS group. (E)
Representative illustrations of EMG and kinematic features at 14,
21 and 28 days in the SCI group. The toe trajectories recorded
overall session are displayed for different experimental data
including during pre-lesion locomotion, illustrating the marked
improvement of foot movements. The range of hindlimb joint angles
also shows the recovery ability of all groups. SCI, spinal cord
injury; BBB, Basso-Beattie-Bresnahan; EMG, electromyography; SEPs,
somatosensory-evoked potentials; ISMS, intra-spinal
micro-stimulation; TA, the tibialis anterior; LG, lateral
gastrocnemius. |
BBB locomotor scale
Two-way mixed ANOVA was applied to analyze the
effect and interaction of group and time on the BBB locomotor
scale, considering a Greenhouse-Geisser correction (data violated
the sphericity assumption, with Mauchly's W=0.022 and P<0.001).
Significant effects were observed for group (F=32.972;
P<0.001; partial η2=0.6), time (F=481.843;
P<0.001; partial η2=0.956) and interaction
between time and group (F=18.153; P<0.001; partial
η2=0.452). In the SCI group, pairwise comparisons of the
BBB scale showed significant differences (P<0.001) except for
day 1 and day 3 measurements (P=0.071), whereas the BBB scale in
the SCI + ISMS group increased significantly (P<0.001). It was
observed that two weeks after surgery, animals in the SCI + ISMS
group were able to slightly move their hindlimb joints, whereas
slight movement of a few joints of the hindlimbs was occasionally
seen in the SCI group (Fig.
5A).
EMG and kinematics assessments
To precisely assess the recovery of motor function,
the kinematics and EMG characteristics of stepping movements was
extensively quantified in the intact, SCI + ISMS and SCI groups
(Fig. 5C-E). The red circles
showed the movement trajectory of the toes and, as the training
time increased, the movement trajectory showed that the gait
function of rats in both SCI and SCI + ISMS groups gradually
rebuilt. The angles of hindlimb joint and the size of toe endpoint
trajectories in the SCI + ISMS group were bigger than those of the
rats in the SCI group and after 4 weeks of training, the range of
motion was significantly similar to that of the intact rats.
The grey shading in the EMG indicates the presence
of complementary phase in the EMG bursts of a pair of flexor and
extensor muscles (Fig. 5C-E) and
the corresponding EMG integral values are shown in Table I. At 14 days after the injury, rats
in the SCI group could only move their hindlimbs slightly and no
significant EMG bursts were observed in the antagonistic pair of
muscles. By contrast, rats in the SCI + ISMS group showed improved
hindlimb joint mobility, with weak bursts of muscle activity. On
day 21, all rats in the SCI + ISMS group demonstrated a substantial
increase in EMG bursting patterns or tonic activity in their
hindlimb muscles and exhibited stepping movements. After four
weeks, the EMG activity in the muscles of rats in the SCI + ISMS
group was greatly improved compared with the rats in the SCI group.
The EMG in rat muscle reached levels similar to those recorded
during spontaneous movement in intact rats.
Grip strength assessment
At three days postoperatively, hindlimb grip
strength was particularly weak in both the SCI and SCI + ISMS
groups, at <0.5 N. At 1-2 weeks postoperatively, there was a
significant increase in hindlimb grip strength in the SCI + ISMS
group, approaching 10 N, while the hindlimb grip strength in the
SCI group improved more slowly. After 4 weeks of training, the
maximum hindlimb grip strength of the rats in the SCI + ISMS group
was close to 14 N, which was closer to the maximum hindlimb grip
strength of the rats in SCI group (11 N) than to that of the intact
group (18N). Two-way mixed ANOVA was applied to analyze the effect
and interaction of group and time on the grip strength, considering
a Greenhouse-Geisser correction (data violated the sphericity
assumption, with Mauchly's W=0.06 and P<0.001). Significant
effects were observed for group (F=28.356; P<0.001;
partial η2=0.563), time (F=1631.072; P<0.001;
partial η2=0.987) and interaction between time and group
F=4.881; P=0.007; partial η2=0.182). Grip strength
increased significantly over time in both groups (P<0.001). The
grip strength of the SCI + ISMS group was significantly greater
than that of the SCI group at each measurement (P<0.001;
Fig. 5B).
SEPs assessment
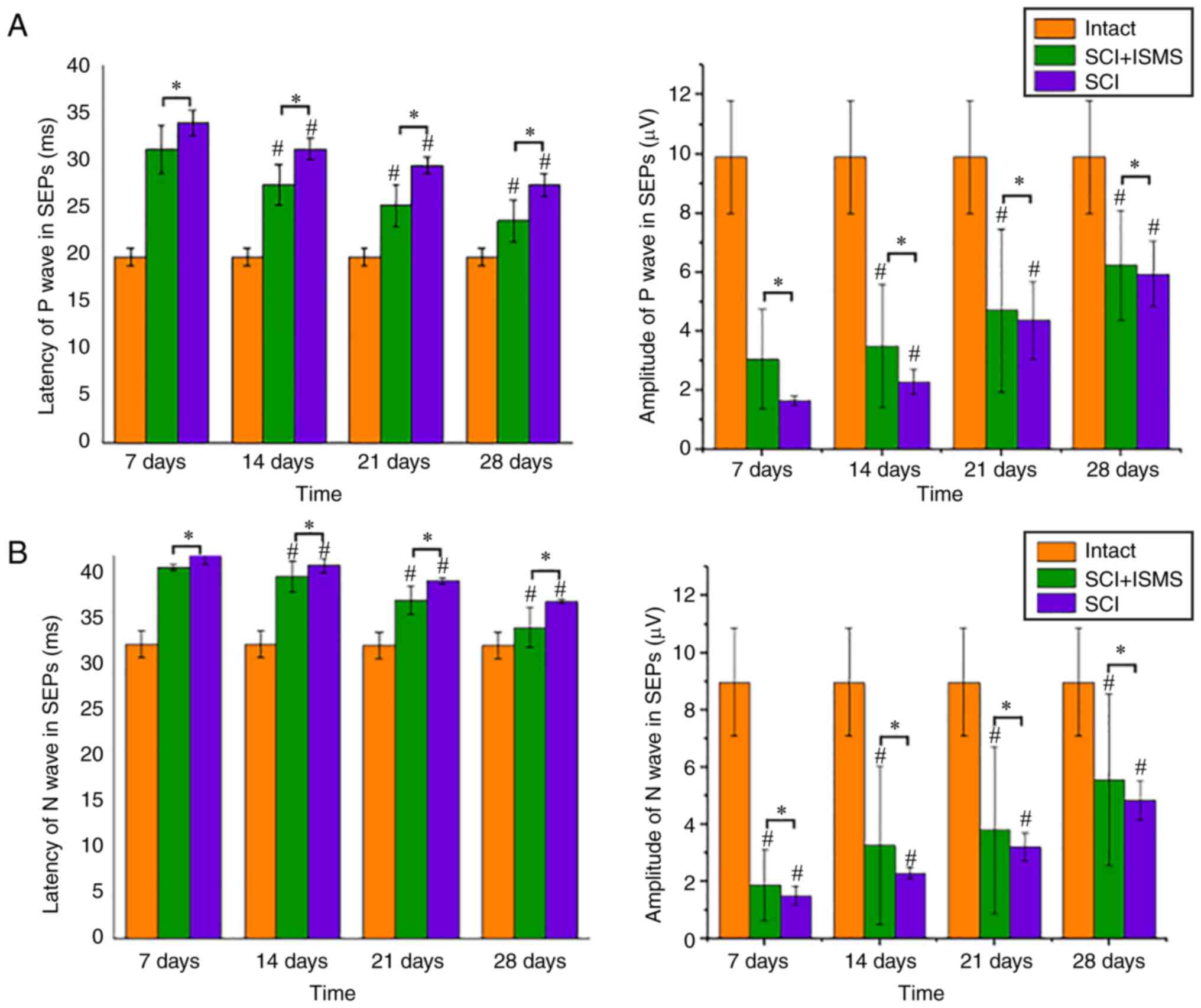
At 7 days postoperatively, the SEPs (P and N waves)
changed due to the injury: The latency was somewhat prolonged and
the amplitude correspondingly reduced. The main reason for this was
the blockage of the upstream pathway following SCI, which resulted
in the inability of the upstream nerve signals to transmit signals
through the spinal cord when the nerve impulses transmitted them to
the cerebral cortex, drastically reducing the number of nerve
conduction and therefore significantly reducing the speed of nerve
conduction in the upstream pathway. With ISMS following injury, the
latency and amplitude would gradually approach that of SEPs in
intact rats. As shown in Fig. 6A
and B, the latency of the P-wave
in the SCI group (34 msec) was much longer than that of the P-wave
in intact rats (20 msec) at 7 days postoperatively, whereas the
latency of the P-wave in the SCI + ISMS group (31 msec) was reduced
compared with that of the SCI group. The amplitude of the P waves
in the SCI group (1.6 mV) was much smaller than that of the intact
rats (10 mV), whereas the amplitude of the P waves in the SCI +
ISMS group (3 mV) increased compared with that of the SCI group.
The latency and amplitude of the N waves also showed the same
trend. After 3-4 weeks of stimulation, the latency of the P and N
waves in the SCI + ISMS group gradually decreased and the amplitude
became higher. By the fourth week, the latency and amplitude more
closely resembled the SEPs of the intact rats and the rats in the
SCI + ISMS group improved compared with than the rats in the SCI
group in terms of overall improvement in SEPs. As represented in
Fig. 6A and B, an improvement in the function of the
sensory pathways was inferred based on the increase in amplitude
and reduction of the latency in SEPs. The latency and amplitude
values of P and N showed a gradual improvement in the SEPs of all
rats from the first week to the end of the study. The application
of two-way mixed ANOVA showed significant differences among the SCI
+ ISMS and SCI groups in latency and amplitude of SEPs at every
time point (P<0.001). With the ISMS training, rats in the SCI +
ISMS group exhibited significant improvement in the latency and the
amplitude of SEPs over time (P<0.001).
All outcome measures revealed that a four-week
period of ISMS locomotor training was sufficient to improve walking
symmetry and correct the residual hindlimb deficits on the side of
the lesion in SCI models.
Discussion
The present study first reported that two special
sites were symmetrical on the posterior median sulcus and that ISMS
can induce bilateral alternation of two movement patterns of the
bilateral hind limbs after stimulating either of these two special
sites once the polarity of the stimulus is changed. Based on this
finding, four weeks of ISMS locomotor training was designed to
examine the efficacy of stimulation at the special site on the
reconstruction of lower limbs. Male and female rats did not differ
significantly at the biological level during the experiment. The
present study illustrated that the implantation of a pair of
microelectrodes on the special site in the lumbosacral spinal cord
in combination with the training of the bilateral hind limbs could
be used to restore locomotor-like activity following SCI. ISMS is
commonly used as a means of recovery motor function in animal
experiments following SCI, activating the motor networks in the
spinal cord distal to the site of injury (36). The present study also found that
the threshold of stimulation amplitude increases slightly over time
and that the position of the two special sites did not migrate with
time, no matter which segment was damaged. It was observed in the
present study that, before the identification of the optimal sites
that induce alternating flexion and extension movements, a double
rhythmic fast-jitter phenomenon of the bilateral hind limbs may be
the specific manifestation of the neuronal network oscillation.
Moreover, there is compelling evidence that neuronal connections
between the left and right sides and those between the cervical and
lumbar levels coordinate the activities of spinal CPGs (37). The optimal sites on the left and
right of the spinal cord that control the stepping movement of the
bilateral hind limbs are symmetrical concerning the posterior
median sulcus, but the movement patterns generated by the
stimulation using the same polarity pulses are complementary. These
phenomena indicated that these specific sites may be closely
related to CPGs. The induced body movement phenomenon met the
characteristics of the CPG network responsible for walking,
controlling the rhythm and the coordinated movement patterns of the
bilateral hind limbs (38-40).
In the process of transection injury of the VT9 vertebra segment,
cutting off from the cerebral cortex neural control signaling
pathways, the hind limbs spontaneously produced rhythmic movements
uncontrolled by the upper central nervous system (5-7).
Nevertheless, the two points identified using the method described
were consistent with the stimulation positions of the bilateral
alternating movements of the hind limbs induced by ISMS as
mentioned in a number of published studies, both located in the L2
segment of the spinal cord (41-43).
Therefore, it was hypothesized that these two optimal points were
the activation sites of the CPG network responsible for hind limb
walking and the special sites henceforth referred to as A-CPG
sites.
Our studies have also indicated that
micro-stimulation via a microelectrode did not merely activate an
individual ‘motor pool’, but rather activated a complex spinal
neural network that could generate coordinated, multi-joint flexion
or extension movements (44,45).
As an exploration of the motor mechanism by which unilateral
stimulation can produce bilateral activation, the lower limb motor
neural network was modelled as a CPG model in which the ipsilateral
flexion and extension neurons inhibit each other via inhibitory
interneurons, whereas the two contralateral extensor (or flexor)
neurons produce excitatory activation primarily via excitatory
interneurons. When an electrical stimulus was applied to the A-CPG
site, it was equivalent to activating the entire neuronal network.
Excitatory and inhibitory effects were transmitted between flexion
and extension neurons via synapses, culminating in a bilateral
pattern of motor activation. Compared with Saigal et al
(23), in which a single
micro-wire stimulating motor neuron pool of the cat produced
single-joint movements as well as coordinated multi-joint
synergies, the present study used fewer electrodes to activate the
movement patterns and, when switching the pulse polarity of the
stimulus, it could realize the switch of the movement pattern and
thus finally form a complete gait movement. To further explore the
mechanism behind this finding, the authors will next investigate
from the perspective of electrophysiology or physical engineering
to explain the relationship between the polarity of stimulus, the
A-CPG sites and the movement patterns.
In conclusion, based on the A-CPG sites that are
found to control the switch of two movement patterns, the present
study assessed recovery based on electrical stimulation using
mono-polar ISMS. It was possible to recover the gait function of
paralyzed hind limbs after SCI by controlling one pair of
electrodes on the spinal cord. It has been demonstrated that ISMS
has several advantages over direct muscle stimulation (46-48).
The disadvantage of the intramuscular stimulation was rapid muscle
fatigue and ISMS was able to elicit prolonged and stable force
generation (49). Compared with
muscle stimulation, the present study greatly reduced surgical
complexity, the number of implanted electrodes required and muscle
fatigue. It is hoped that the present study will provide some
insight into the motor function recovery of paralyzed limbs
following SCI.
Acknowledgements
Not applicable.
Funding
Funding: The present study was funded by the National Natural
Science Foundation of China (grant nos. 81371663 and 61534003), the
Six talents peaks Project (grant no. SWYY-116), and the
Post-graduate Research and Practice Innovation Program of Jiangsu
Province, China (grant no. KYCX21_3085).
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
XS was responsible for conceiving and designing the
current study, definition of English abbreviations for special
phrases in the paper, manuscript editing and manuscript review. XS,
YW and ZL were responsible for experimental studies, manuscript
editing and data acquisition. TS was responsible for design,
literature search, experimental studies, statistical analysis,
manuscript preparation and manuscript editing. TS, YW and ZL
confirm the authenticity of all the raw data. All authors read and
approved the final manuscript.
Ethics approval and consent to
participate
All procedures were conducted according to the Guide
for the Care and Use of Experimental Animals [approval no. SYXK
(Su) 2017-0046], using protocols approved by the Ethics Committee
of Nantong University.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Wein AJ: Re: Traumatic spinal cord injury
in the United States, 1993-2012. J Urol. 195(685)2016.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Krause JS and Carter RE: Risk of mortality
after spinal cord injury: Relationship with social support,
education, and income. Spinal Cord. 47:592–596. 2009.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Golowasch J: Neuromodulation of central
pattern generators and its role in the functional recovery of
central pattern generator activity. J Neurophysiol. 122:300–315.
2019.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Grill WM: Electrical activation of spinal
neural circuits: Application to motor-system neural prostheses.
Neuromodulation. 3:97–106. 2000.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Grillner S, Perret C and Zangger P:
Central generation of locomotion in the spinal dogfish. Brain Res.
109:255–269. 1976.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Grillner S and Zangger P: On the central
generation of locomotion in the low spinal cat. Exp Brain Res.
34:241–261. 1979.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Marder E and Bucher D: Central pattern
generators and the control of rhythmic movements. Curr Biol.
11:R986–R996. 2001.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Gordon IT and Whelan PJ: Deciphering the
organization and modulation of spinal locomotor central pattern
generators. J Exp Biol. 209:2007–2014. 2006.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Minassian K, Hofstoetter US, Dzeladini F,
Guertin PA and Ijspeert A: The human central pattern generator for
locomotion: Does it exist and contribute to walking?
Neuroscientist. 23:649–663. 2017.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Steuer I and Guertin PA: Central pattern
generators in the brainstem and spinal cord: An overview of basic
principles, similarities and differences. Rev Neurosci. 30:107–164.
2019.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Mushahwar VK, Gillard DM, Gauthier MJA and
Prochazka A: Intraspinal micro stimulation generates locomotor-like
and feedback-controlled movements. IEEE Trans Neural Syst Rehabil
Eng. 10:68–81. 2002.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Guevremont L, Renzi CG, Norton JA,
Kowalczewski J, Saigal R and Mushahwar VK: Locomotor-related
networks in the lumbosacral enlargement of the adult spinal cat:
Activation through intraspinal microstimulation. IEEE Trans Neural
Syst Rehabil Eng. 14:266–272. 2006.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Zhang H, Feng L and Wang Y: Intraspinal
microstimulation A novel technique for the functional recovery of
spinal cord injury. Neural Regen Res. 5:1249–1255. 2010.
|
|
14
|
Bamford JA, Todd KG and Mushahwar VK: The
effects of intraspinal microstimulation on spinal cord tissue in
the rat. Biomaterials. 31:5552–5563. 2010.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Bamford JA and Mushahwar VK: Intraspinal
microstimulation for the recovery of function following spinal cord
injury. In: Brain Machine Interfaces: Implications for Science,
Clinical Practice and Society. Schouenborg J, Garwicz M and
Danielsen N (eds.) Elsevier Science Bv, Amsterdam, pp227-239,
2011.
|
|
16
|
Shu B, Yang F and Guan Y: Intra-spinal
microstimulation may alleviate chronic pain after spinal cord
injury. Med Hypotheses. 104:73–77. 2017.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Jankowska E, Lundberg A, Roberts WJ and
Stuart D: A long propriospinal system with direct effect on
motoneurones and on interneurones in the cat lumbosacral cord. Exp
Brain Res. 21:169–194. 1974.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Jankowska E, Padel Y and Tanaka R: The
mode of activation of pyramidal tract cells by intracortical
stimuli. J Physiol. 249:617–636. 1975.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Jankowska E: Interneuronal relay in spinal
pathways from proprioceptors. Prog Neurobiol. 38:335–378.
1992.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Gaunt RA, Prochazka A, Mushahwar VK,
Guevremont L and Ellaway PH: Intraspinal microstimulation excites
multisegmental sensory afferents at lower stimulus levels than
local alpha-motoneuron responses. J Neurophysiol. 96:2995–3005.
2006.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Lau B, Guevremont L and Mushahwar VK:
Strategies for generating prolonged functional standing using
intramuscular stimulation or intraspinal microstimulation. IEEE
Trans Neural Syst Rehabil Eng. 15:273–285. 2007.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Mushahwar VK, Jacobs PL, Normann RA,
Triolo RJ and Kleitman N: New functional electrical stimulation
approaches to standing and walking. J Neural Eng. 4:S181–S197.
2007.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Saigal R, Renzi C and Mushahwar VK:
Intraspinal microstimulation generates functional movements after
spinal-cord injury. IEEE Trans Neural Syst Rehabil Eng. 12:430–440.
2004.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Asadi AR and Erfanian A: Adaptive
neuro-fuzzy sliding mode control of multi-joint movement using
intraspinal microstimulation. IEEE Trans Neural Syst Rehabil Eng.
20:499–509. 2012.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Holinski BJ, Mazurek KA, Everaert DG,
Toossi A, Lucas-Osma AM, Troyk P, Etienne-Cummings R, Stein RB and
Mushahwar VK: Intraspinal microstimulation produces over-ground
walking in anesthetized cats. J Neural Eng.
13(056016)2016.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Tawakol O, Mushahwar VK and Troyk PR: The
use of digital image correlation for measurement of strain fields
in a novel wireless intraspinal microstimulation interface. Artif
Organs. 46:2066–2072. 2022.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Barthélemy D, Leblond H, Provencher J and
Rossignol S: Nonlocomotor and locomotor hindlimb responses evoked
by electrical microstimulation of the lumbar cord in spinalized
cats. J Neurophysiol. 96:3273–3292. 2006.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Shen XY, Tao CL, Ma L, Shen JH, Li ZL,
Wang ZG and Lü XY: Influence of spinal cord injury on core regions
of motor function. Neural Regen Res. 16:567–572. 2021.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Islam R, Cuellar CA, Felmlee B, Riccelli
T, Silvernail J, Boschen SL, Grahn P and Lavrov I: Multifactorial
motor behavior assessment for real-time evaluation of emerging
therapeutics to treat neurologic impairments. Sci Rep.
9(16503)2019.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Basso DM, Beattie MS and Bresnahan JC:
Graded histological and locomotor outcomes after spinal cord
contusion using the NYU weight-drop device versus transection. Exp
Neurol. 139:244–256. 1996.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Alam M, Garcia-Alias G, Shah PK,
Gerasimenko Y, Zhong H, Roy RR and Edgerton VR: Evaluation of
optimal electrode configurations for epidural spinal cord
stimulation in cervical spinal cord injured rats. J Neurosci
Methods. 247:50–57. 2015.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Hanwright PJ, Rath JL, von Guionneau N,
Harris TGW, Sarhane KA, Kemp SWP, Hoke A, Cederna PS and Tuffaha
SH: Stimulated grip strength measurement: Validation of a novel
method for functional assessment. Muscle Nerve. 60:437–442.
2019.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Agrawal G, Kerr C, Thakor NV and All AH:
Characterization of graded multicenter animal spinal cord injury
study contusion spinal cord injury using somatosensory-evoked
potentials. Spine (Phila Pa 1976). 35:1122–1127. 2010.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Lewis MJ, Howard JF Jr and Olby NJ: The
relationship between trans-lesional conduction, motor neuron pool
excitability, and motor function in dogs with incomplete recovery
from severe spinal cord injury. J Neurotrauma. 34:2994–3002.
2017.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Cheng XH, Zhang L and Fu J: Somatosensory
evoked potential changes and decompression timing for spinal cord
function recovery and evoked potentials in rats with spinal cord
injury. Brain Res Bull. 146:7–11. 2019.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Jackson A and Zimmermann JB: Neural
interfaces for the brain and spinal cord-restoring motor function.
Nat Rev Neurol. 8:690–699. 2012.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Frigon A, Desrochers É, Thibaudier Y,
Hurteau MF and Dambreville C: Left-right coordination from simple
to extreme conditions during split-belt locomotion in the chronic
spinal adult cat. J Physiol. 595:341–361. 2017.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Cohen AH and Wallén P: The neuronal
correlate of locomotion in fish. ‘Fictive swimming’ induced in an
in vitro preparation of the lamprey spinal cord. Exp Brain Res.
41:11–18. 1980.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Cowley KC and Schmidt BJ: Regional
distribution of the locomotor pattern-generating network in the
neonatal rat spinal cord. J Neurophysiol. 77:247–259.
1997.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Ijspeert AJ and Kodjabachian J: Evolution
and development of a central pattern generator for the swimming of
a lamprey. Artif Life. 5:247–269. 1999.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Cazalets JR, Borde M and Clarac F:
Localization and organization of the central pattern generator for
hindlimb locomotion in newborn rat. J Neurosci. 15:4943–4951.
1995.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Zhang JM, Lanuza GM, Britz O, Wang Z,
Siembab VC, Zhang Y, Velasquez T, Alvarez FJ, Frank E and Goulding
M: V1 and v2b interneurons secure the alternating flexor-extensor
motor activity mice require for limbed locomotion. Neuron.
82:138–150. 2014.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Pujala A, Blivis D and O'Donovan MJ:
Interactions between dorsal and ventral root stimulation on the
generation of locomotor-like activity in the neonatal mouse spinal
cord. eNeuro. 3(ENEURO.0101-16.2016)2016.PubMed/NCBI View Article : Google Scholar
|
|
44
|
Shen XY, Du W, Huang W and Chen Y:
Rebuilding motor function of the spinal cord based on functional
electrical stimulation. Neural Regen Res. 11:1327–1332.
2016.PubMed/NCBI View Article : Google Scholar
|
|
45
|
Tao C, Shen X, Ma L, Li Z and Shen J:
Three-dimensional map of lumbar spinal cord motor function for
intraspinal microstimulation in rats. In: 2020 42nd Annual
International Conference of the IEEE Engineering in Medicine &
Biology Society (EMBC) IEEE, pp3525-3528, 2020.
|
|
46
|
Dalrymple AN, Roszko DA, Sutton RS and
Mushahwar VK: Pavlovian control of intraspinal microstimulation to
produce over-ground walking. J Neural Eng.
17(036002)2020.PubMed/NCBI View Article : Google Scholar
|
|
47
|
Tai C, Booth AM, Robinson CJ, de Groat WC
and Roppolo JR: Isometric torque about the knee joint generated by
microstimulation of the cat L6 spinal cord. IEEE Trans Rehabil Eng.
7:46–55. 1999.PubMed/NCBI View Article : Google Scholar
|
|
48
|
Tai C, Booth AM, Robinson CJ, de Groat WC
and Roppolo JR: Multimicroelectrode stimulation within the cat L6
spinal cord: Influences of electrode combinations and stimulus
interleave time on knee joint extension torque. IEEE Trans Rehabil
Eng. 8:1–10. 2000.PubMed/NCBI View Article : Google Scholar
|
|
49
|
Caldwell CW and Reswick JB: A percutaneous
wire electrode for chronic research use. IEEE Trans Biomed Eng.
22:429–432. 1975.PubMed/NCBI View Article : Google Scholar
|