Introduction
Influenza is an infectious viral disease that causes
mild to severe respiratory complications. The mild symptoms
associated with influenza include fever, cough and muscle pains.
The severe symptoms include lethal pneumonia due to the virus or
secondary bacterial infection (1,2).
Influenza viruses are enveloped negative-sense single-strand RNA
viruses, which encode viral glycoproteins [haemagglutinin (HA) and
neuraminidase (NA)] that facilitate viral entry. The HA and NA
viral proteins are the main targets for human antibodies. These
proteins also serve as the primary sites for the accumulating
mutations in the virus (3,4). This antigenic drift requires frequent
updates of influenza vaccines. Therefore, antivirals are essential
for treating virus-infected patients and preventing infection in
virus-exposed individuals (3,4).
There are currently five Food and Drug Administration-approved
drugs for influenza: Oseltamivir phosphate, zanamivir, baloxavir,
marboxil and peramivir (5).
However, the high prevalence of drug resistance is a major
challenge (6).
Herbal medicines and natural products exhibit
numerous advantages in early intervention, combination therapies
and uncomplicated diseases due to their multi-components exerting
synergistic effects (7). Eucommiae
Cortex (EC) is a traditional medicine used in Asia to tonify the
liver and kidney, and also known to be effective in osteoporosis
(8). Chemical characterization
studies of EC have shown that lignans, iridoids, phenols, steroids
and flavonoids are the main bioactive components of EC, which have
anti-inflammatory, antioxidant, antitumor and antiviral effects
(9,10).
Identifying the molecular mechanisms of plant
extracts is complicated due to the synergistic effects of the
multiple active compounds and targets involved (11). The concept of bioinformatics
analysis using network pharmacology emphasizes ‘multi-target,
multi-component therapeutics’ which indicates synergistic and
holistic approach (12).
Bioinformatics research is time-saving research compared with
conventional research and may identify novel compounds and targets
of unresolved infectious diseases (13).
In the present study, in an attempt to identify
active compounds and targets of EC in influenza, common targets of
EC and influenza were retrieved. The active compounds were
identified by analyzing the compound-target (C-T) networks, and
potential targets were examined by generating a protein-protein
interaction (PPI) network. Molecular docking was applied to observe
the binding affinity of each ligand and target to identify the main
compounds and targets of EC. Further in vitro experiments
were conducted to measure antiviral effects to identify the effect
of EC on influenza.
Materials and methods
In silico analyses. Active compounds
of EC and potential targets of influenza
The active compounds of EC were screened using the
Traditional Chinese Medicine Systems Pharmacology (TCMSP) database
(https://www.tcmsp-e.com/), a systems pharmacology
platform for herbal medicines that integrates pharmacokinetic
properties and targets associated with herbal drugs. Potential
target genes of EC were investigated using DrugBank (https://go.drugbank.com/) to access disease target in
association with the natural compounds. Influenza target genes
(Homo sapiens) were retrieved from Kyoto Encyclopedia of
Genes and Genomes (KEGG; https://www.genome.jp/kegg/pathway.html) and GeneCards
(https://www.genecards.org/) to retrieve
gene functions and underlying biological mechanisms. The common
intersecting targets were retrieved using Venny-2.1.0 web
(https://bioinfogp.cnb.csic.es/tools/venny/).
Network construction and analysis. Network
pharmacology is a computational drug design method to identify
active compounds and targets through the connection of nodes in a
biological network, and further clarify the mechanism of compound
actions. The active compounds were screened by analyzing the C-T
network constructed using Cytoscape (https://cytoscape.org/, version 3.9.0) to visualize
and analyze the networks. The PPI network was generated using the
Search Tool for the Retrieval of Interacting Genes/Proteins
(STRING) database (https://string-db.org/, version 12.0) to interpret the
raw lists of screened targets and determine the functional
connections among proteins with a high confidence (0.700) setting.
The intersecting core target datasets were imported into Cytoscape
(version 3.9.0), and then analyzed to determine hub targets.
Gene Ontology (GO) and pathway enrichment
analysis. GO and pathway analyses were performed using the
Database for Annotation, Visualization and Integrated Discovery
(DAVID; https://david.ncifcrf.gov).
Molecular docking analysis. Molecular docking
is a computational modeling method to study and predict the
interaction between a compound (or ligand) and a protein target.
The method aims to identify the binding sites and evaluate the
binding affinity using the scoring system using the AutoDock tool
(https://vina.scripps.edu/, ver. 1.1.2)
(14). Binding affinity is defined
as the strength of the interaction between the ligand and its
target based on Gibbs free energy. The smaller the equilibrium
dissociation constant value, the greater the binding affinity
(15). Active compounds were
docked into hub targets obtained from the network for C-T
interaction analysis. 3D structures of potential compounds were
downloaded from the PubChem database (https://pubchem.ncbi.nlm.nih.gov/) and converted into
Protein Data Bank (PDB) files using Biovia Discovery Studio
(https://www.3ds.com/products/biovia/discovery-studio,
version 20.1.0.19295). The main targets in the 3D version were
obtained from the Research Collaboratory for Structural
Bioinformatics PDB [https://www.rcsb.org/; for the targets IL-6 (PDB ID:
1P9M), BCL2 (PDB ID: 6GL8), IL1B (PDB ID: 1ITB) and TNF (PDB ID:
1TNF)]. Molecular docking analysis was performed with the AutoDock
Vina (ver.1.1.2) option in Pyrx (https://pyrx.sourceforge.io/, ver.0.9.6) based on
scoring functions. The 2D interactions of ligands and targets were
analyzed using Biovia Discovery Studio.
In vitro analyses. Plant material,
cells and viral infection
EC extract was purchased from the National
Development Institute of Korean Medicine (https://nikom.or.kr/). The plant source is the stem
bark of Eucommia ulmoides Oliver. A total of 3.5 liters of
70% ethanol was added to EC and the sample was extracted for 3 h at
room temperature. The solvent was then filtered and separated using
filter paper (Hyundai No. 2; cat. no. HD2-090). The filtrate was
collected and concentrated under reduced pressure using a rotary
vacuum evaporator, followed by freeze-drying to obtain the
extract.
Madin-Darby canine kidney (MDCK) cells were
purchased from American Type Culture Collection (cat. no. CCL 34)
and cultured at 37˚C and 5% CO2 in Minimum Essential
Medium (BYLABS) supplemented with 10% fetal bovine serum (Gibco;
Thermo Fisher Scientific, Inc.) and 1% penicillin/streptomycin.
Human Influenza A subtype H1N1 (Ca/07/09) was supplied by the
Centers for Disease Control and Prevention. The virus titer,
calculated according to the Reed-Muench endpoint method, was
7x107 50% tissue culture infectious dose
(TCID50)/ml (16). All
experiments were performed in a biosafety level 2 facility.
Cytotoxicity and antiviral assay
To calculate the 50% cytotoxic concentration
(CC50) of the EC extract, the viability of treated cells
was determined by a WST assay using an EZ-Cytox kit (Daeil Lab
Service, http://www.daeillab.co.kr). MDCK
cells were seeded at density 1.5x104 cells/well in a
96-well plate for 24 h, and treated with a series of 2-fold diluted
extracts for 48 h. The CC50 value of the extract was
calculated by regression analysis based on spectrophotometric
measurements at 450 nm performed after 1 h incubation of reagent
(10 µl/well) with the cells.
The antiviral properties of the EC extract against
H1N1 Influenza A infection at a dose of 100 TCID50/well
were characterized by four assays: Pretreatment, attachment
inhibition, co-treatment and post-treatment assays. In the
pre-treatment assay, cells were treated with non-toxic doses (2-250
µg/ml) of extract and incubated for 1 h at 37˚C, followed by a 1 h
infection at 37˚C (100 TCID50/well). In the attachment
inhibition and co-treatment experiments, cells were simultaneously
co-incubated for 1 h with the virus and different doses (2-250
µg/ml) at 4˚C (attachment inhibition assay) or 37˚C (co-treatment
assay). In the post-treatment assay, cells were infected with the
virus for 1 h at 37˚C. Following viral removal, cells were treated
with EC extract at concentrations ranging from 2-250 µg/ml for 2
days at 37˚C. After 48 h, cell viability was examined using a WST
cell viability assay.
Time-of-addition assay
MDCK cells were seeded at density 1.5x104
cells/well and incubated for 24 h at 37˚C. The cell monolayer was
infected with the virus at a dose of 100 TCID50/well for
1 h (37˚C). Subsequently, cells were treated (37˚C) with 100 or 200
µg/ml EC extract at 2, 4, 8 and 12 h post-infection. After 48 h,
cell viability was evaluated based on spectrophotometric
measurements (450 nm) using a WST assay kit (10 µl/well).
Statistical analysis
To evaluate the dose-dependent antiviral effects of
the EC extract, significant differences between treatment groups
were determined by one-way analysis of variance. Dunnett's test was
used to characterize the effective dose of the extract by comparing
treated groups with the infected-untreated control group. An
unpaired t-test was used to compare the viability of cells treated
with an extract at a specific concentration to the negative control
(NTC) in cytotoxicity experiment. All statistical analyses were
performed using GraphPad Prism 8.0.2 (Dotmatics). P<0.05 was
considered to indicate a statistically significant difference. The
results reflect an average ± standard deviation of three or more
repetitions.
Results
Active compounds and target
prediction
A total of 28 active compounds were retrieved from
TCMSP based on the criteria of oral bioavailability (OB) ≥30 and
drug-likeness (DL) ≥0.18, following database suggestions. Based on
other studies of EC, genistein (OB, 17.93; DL, 0.21) was included
as an active compound in the present study (17,18).
The potential targets regulated by these compounds were determined
using the TCMSP and DataBank databases (https://go.drugbank.com/). The targets of influenza
were searched in KEGG and GeneCards databases, with the species
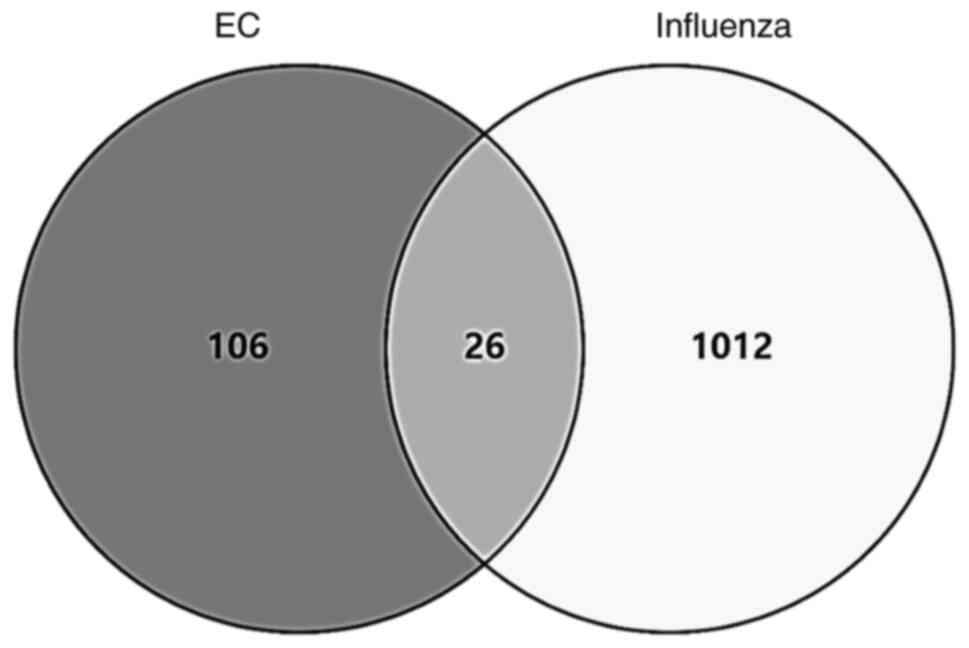
‘Homo sapiens’. Overall, 26 intersecting targets were
identified (Fig. 1).
Active compound and target network
analysis
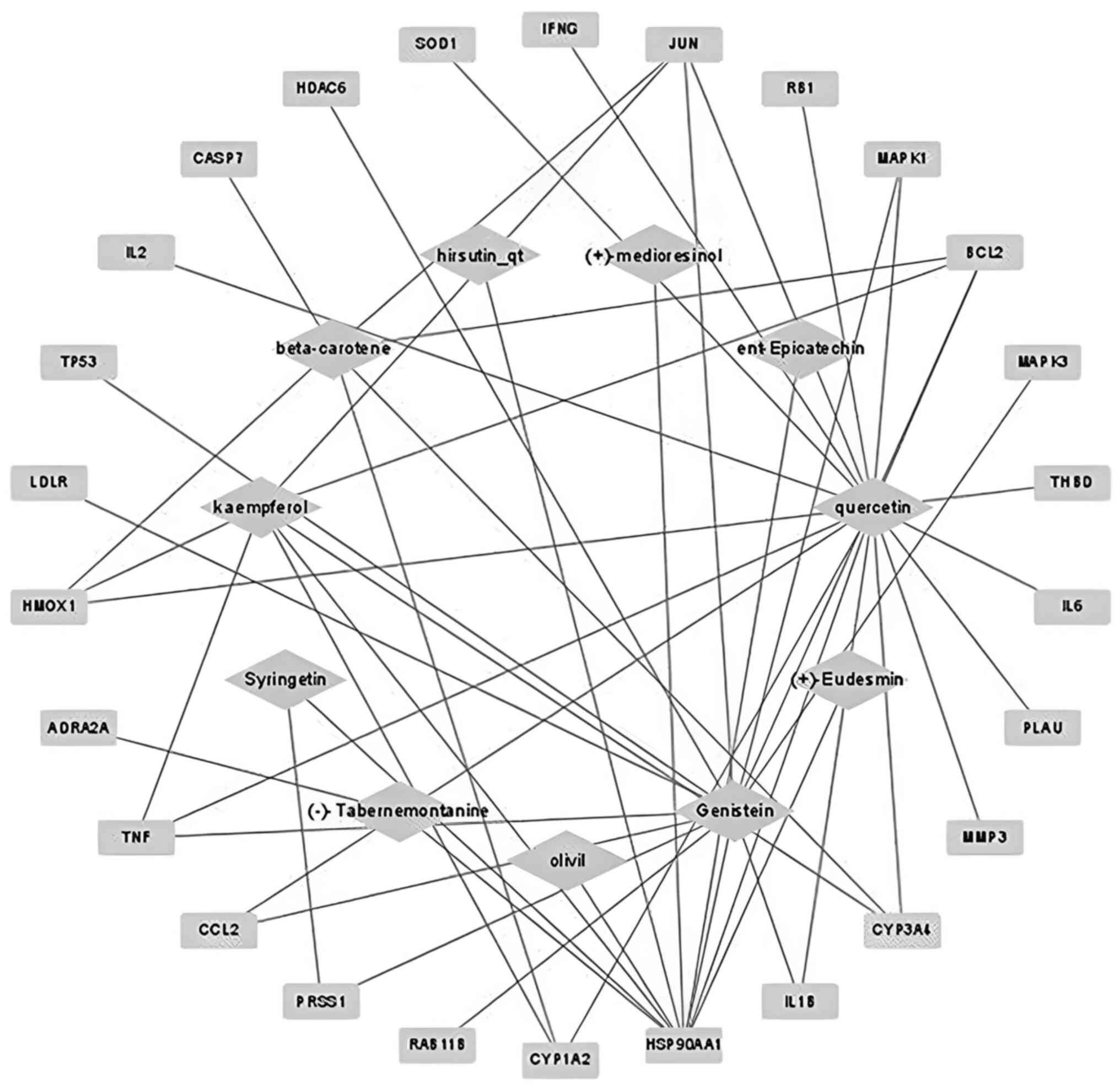
For the 28 active compounds, 132 targets were
identified using the DrugBank database. The C-T network was
constructed using Cytoscape and indicated that 11 active compounds
were linked to 26 hub targets. The network contained 37 nodes and
53 edges (Fig. 2). Based on the
network, it was identified that quercetin showed the highest degree
(degree of 18), followed by genistein (13) and kaempferol (9).
Hub target PPI network analysis
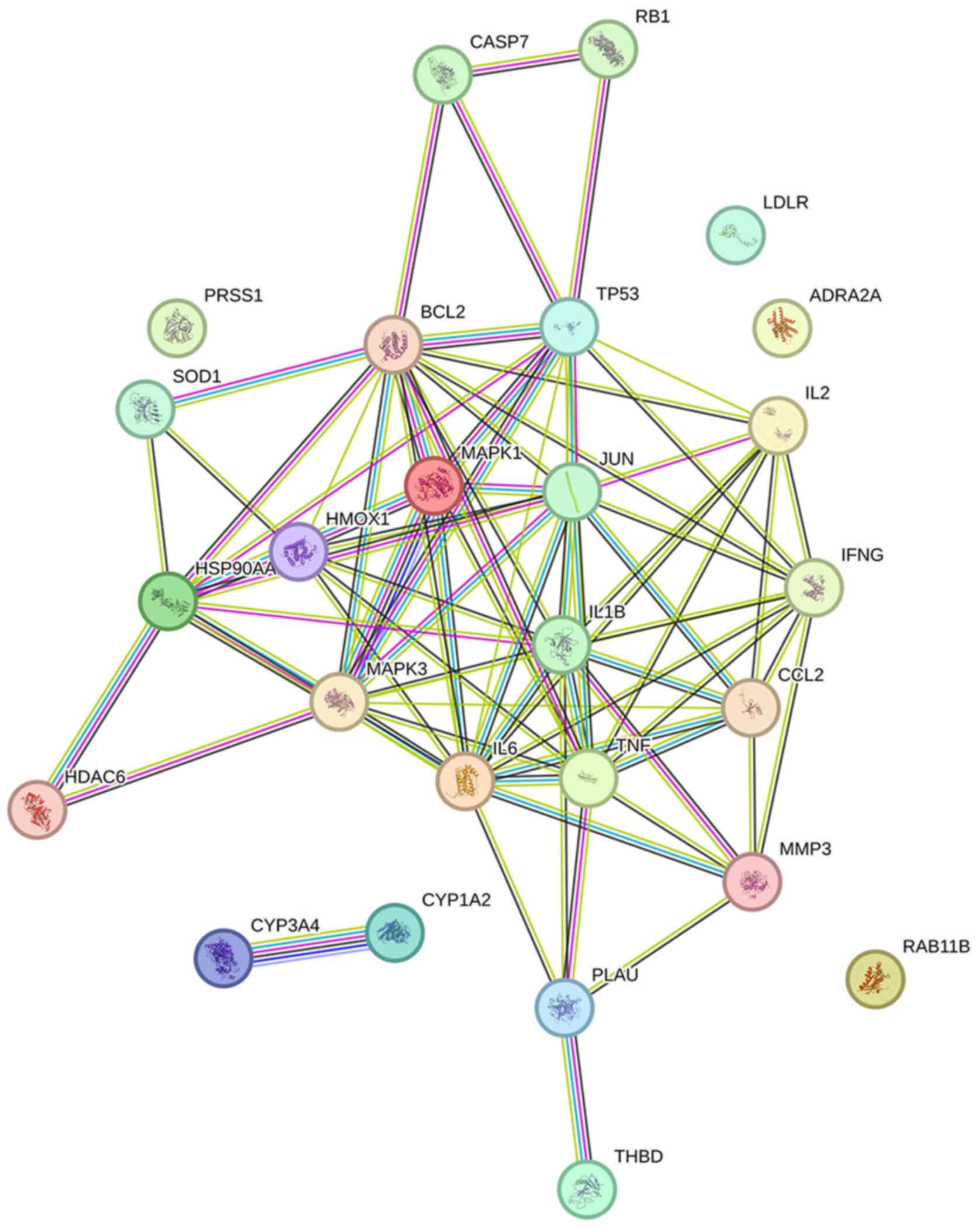
The dataset of 26 hub targets of EC associated with
influenza was used as a dataset to construct the PPI network in
STRING and imported into Cytoscape for network analysis (Fig. 3). According to the database, PPI
enrichment showed a P-value <1.0-16 and the average
local clustering coefficient was 0.66. The network contained 26
nodes and 80 edges. Based on the network, IL-6 had the highest
degree of 14, followed by BCL2, IL1B and TNF (degree of 13).
KEGG signaling pathway analysis
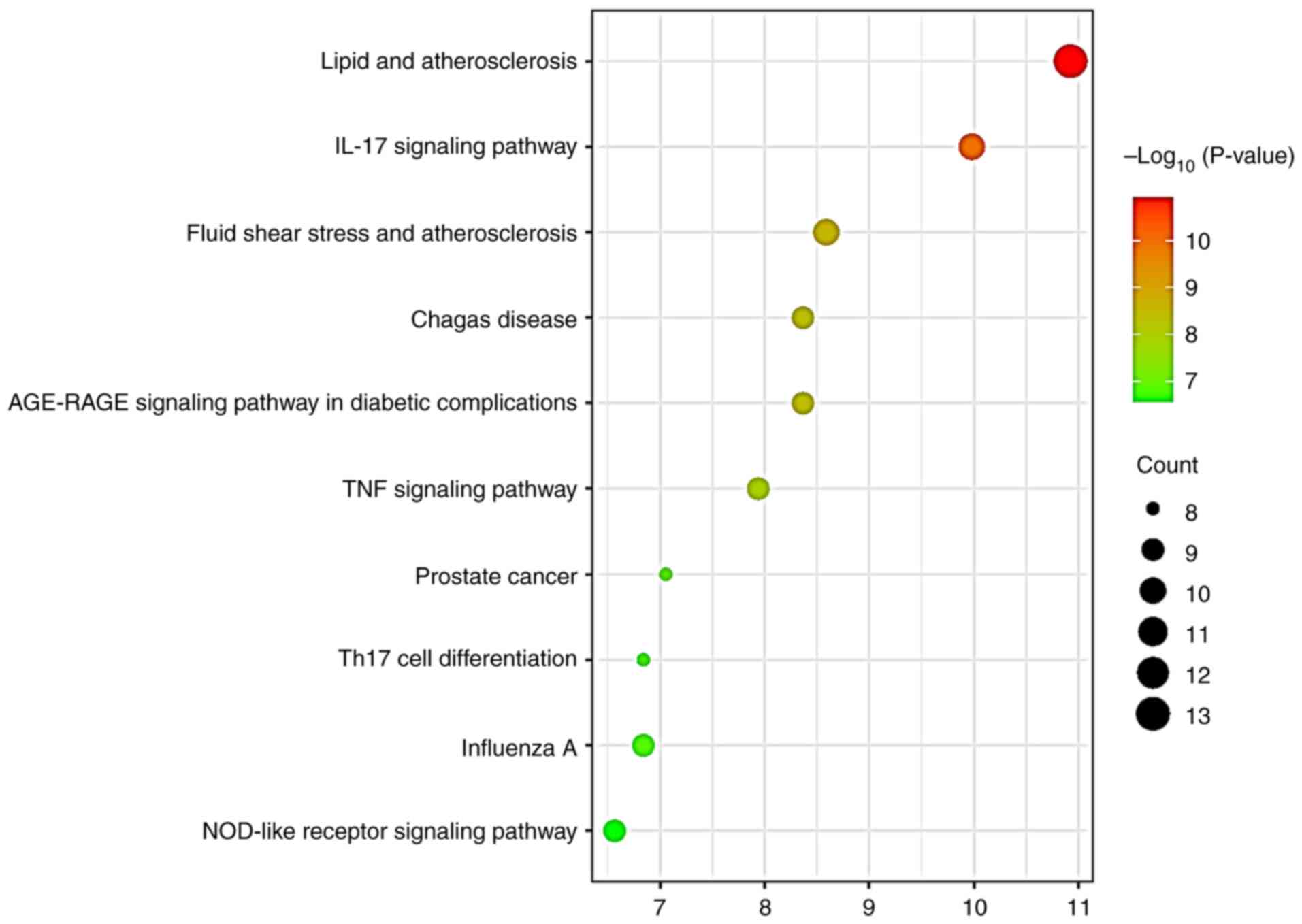
Based on the 26 hub targets, top 10 KEGG signaling
pathways were identified from DAVID and plotted in bubble diagrams
(Fig. 4; Table SI). The analysis revealed that
‘influenza A’ was ranked ninth highest in the pathway analysis, and
two inflammation-associated pathways (‘IL-17 signaling pathway’ and
‘TNF signaling pathway’) were identified.
Molecular docking analysis
A total of three active compounds (quercetin,
kaempferol and genistein) and four targets (IL-6, BCL2, IL1B and
TNF) were selected as main hub targets for EC in influenza based on
the highest degree results in two networks. The four main targets
were docked with the main compounds using the AutoDock Vina option
in Pyrx. The results indicated that kaempferol had the strongest
binding with TNF (Table I).
 | Table IBinding affinity of Eucommiae Cortex
active compounds with influenza targets. |
Table I
Binding affinity of Eucommiae Cortex
active compounds with influenza targets.
| | Binding affinity
(kcal/mol) |
|---|
| Compound | IL6 (1P9M) | BCL2 (6GL8) | IL1B (1ITB) | TNF (1TNF) |
|---|
| Quercetin | -7.3 | -6.9 | -6.6 | -8.9 |
| Kaempferol | -6.9 | -6.6 | -6.2 | -9.1 |
| Genistein | -6.6 | -6.8 | -6.6 | -8.8 |
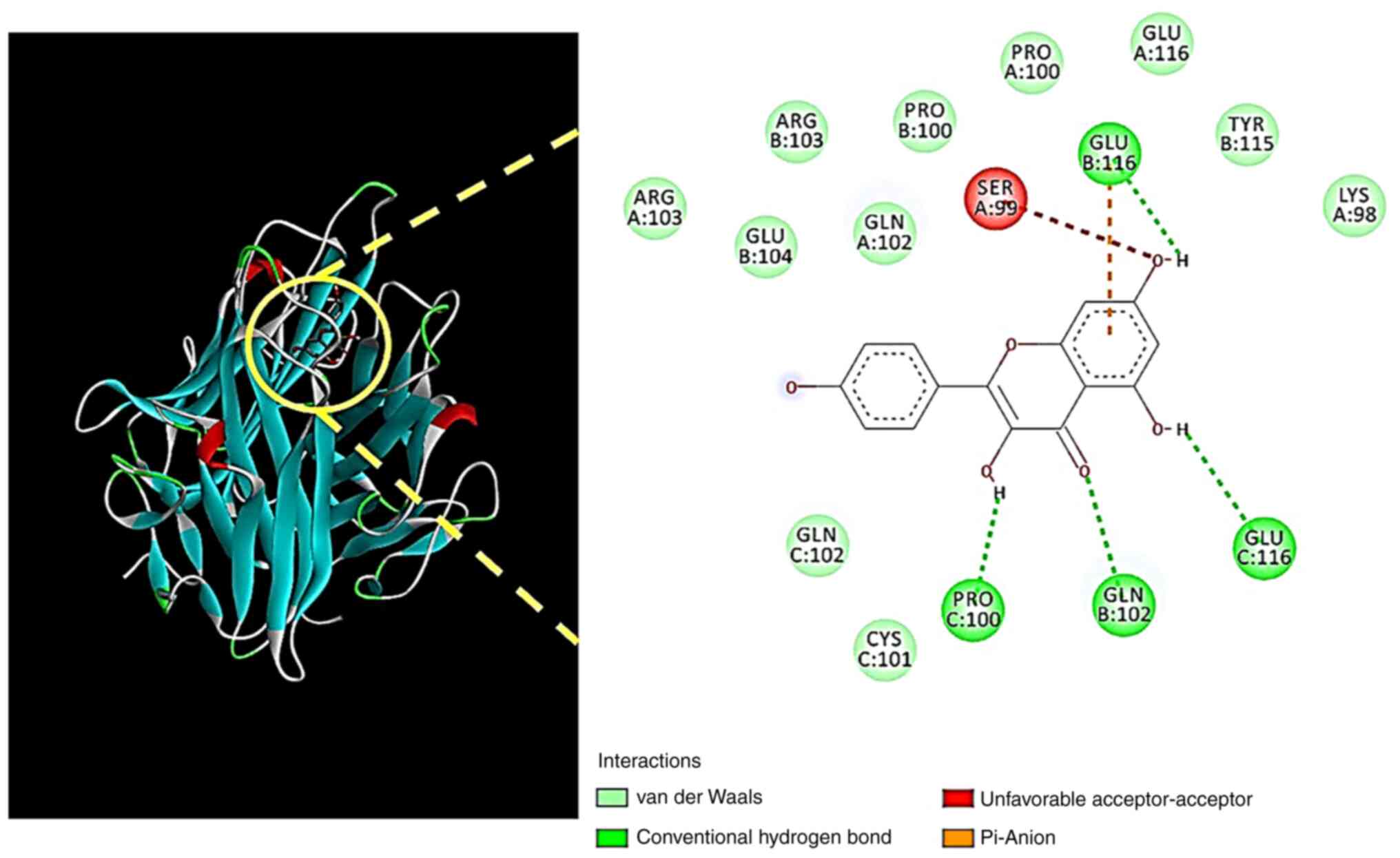
The 3D- and 2D-interactions of kaempferol and TNF
were analyzed using Biovia Discovery Studio (Fig. 5). Kaempferol was found to interact
with TNF amino acids PRO100, GLN102 and GLU116 via hydrogen bonds,
and GLU116 via a π-anion.
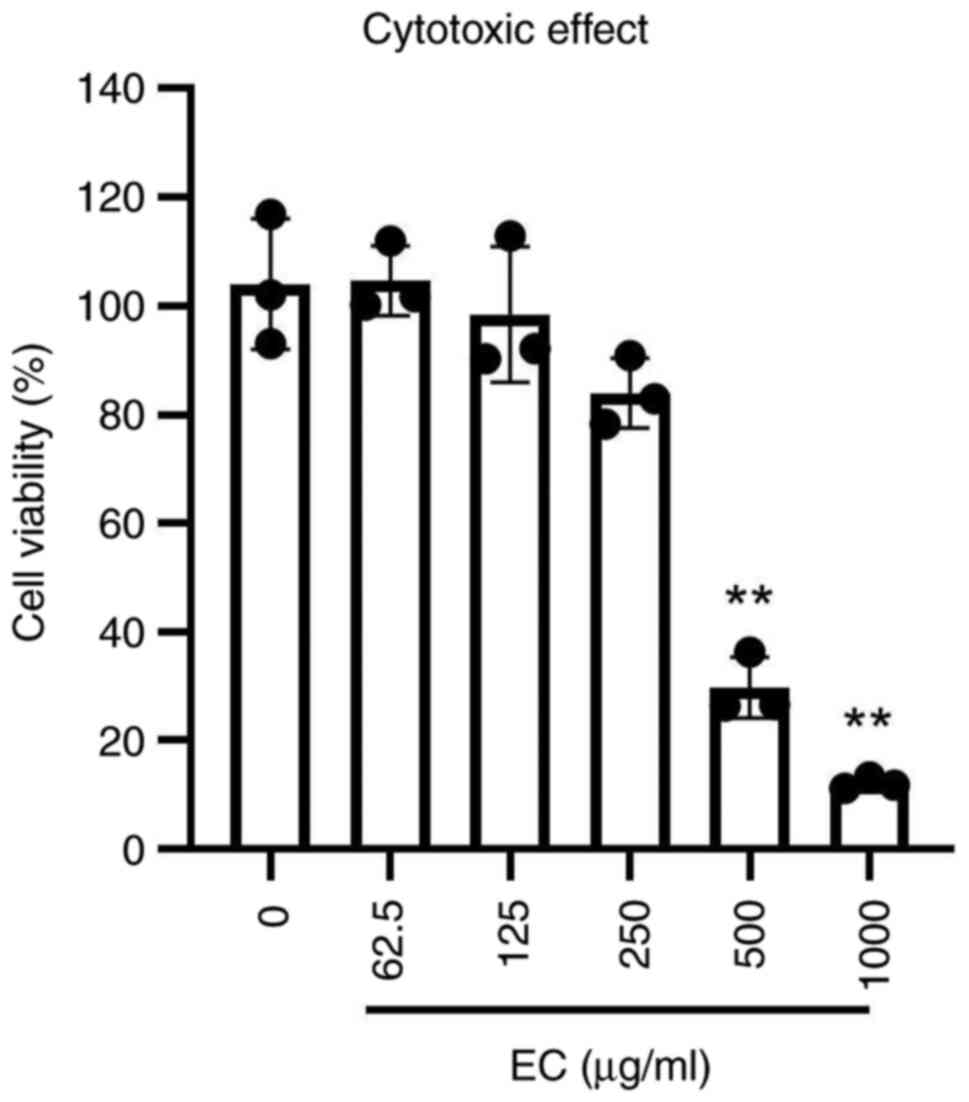
Cytotoxicity and antiviral assay
To determine the concentration range of EC extract
in vitro, a cytotoxicity assay was performed. Treatment with
the concentrations of 500 and 1,000 µg/ml significantly reduced
cell viability to 10-30% compared with that of non-treated cells
(Fig. 6).
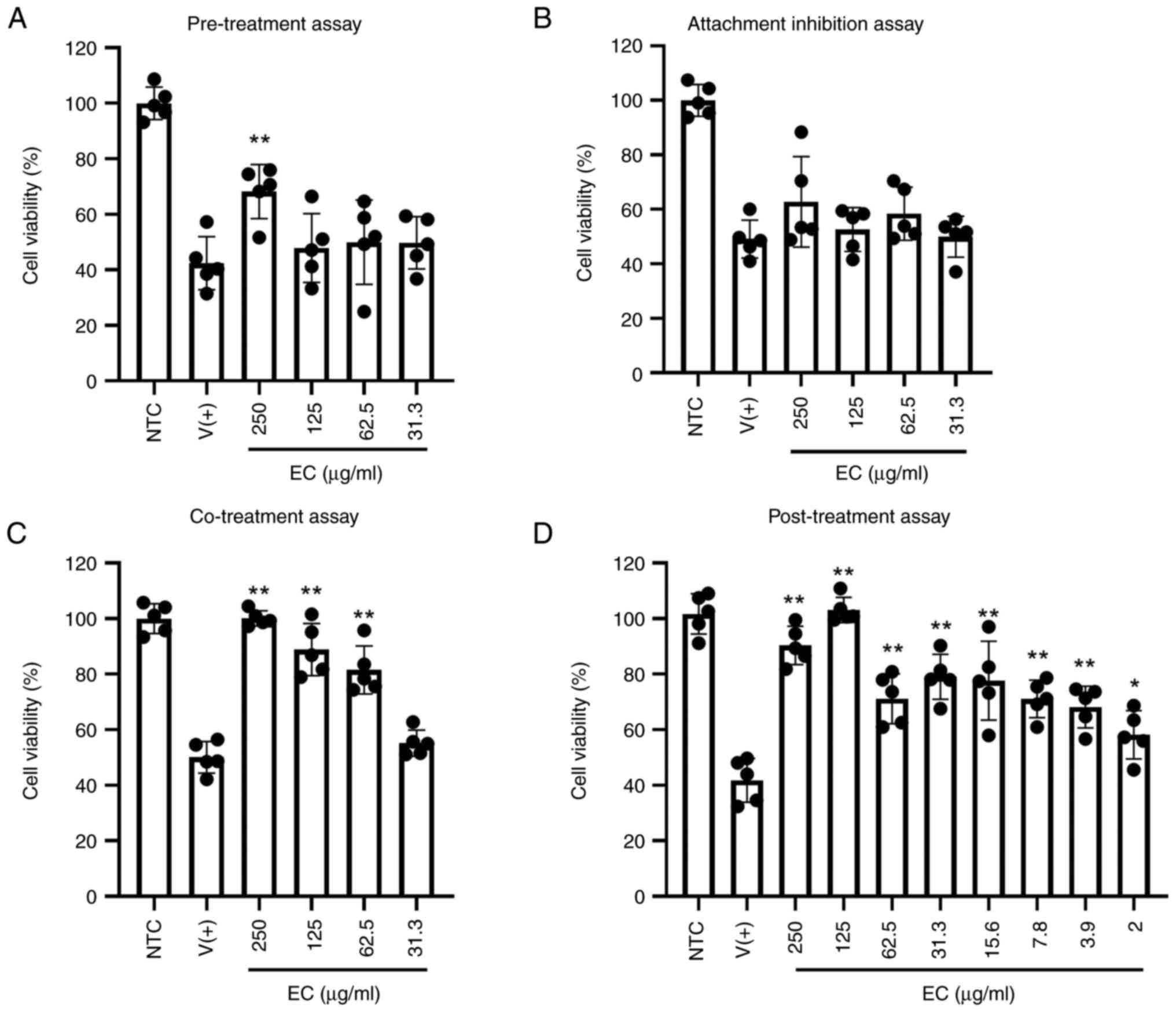
A total of four different in vitro antiviral
assays were performed to verify the anti-influenza potentials of EC
(Fig. 7). The results of
pre-treatment and attachment inhibition assays indicated low
antiviral activities at the earliest stages of infection. To assess
these phases of the Influenza virus infection cycle, experiments
were performed by adding the treatment at specific timepoints
representing initial virus-host cell interactions: before virus
infection (pre-treatment) and during virus adsorption to cells.
Only pre-treatment with 250 µg/ml resulted in post-infection cell
viability at a significant higher level compared with the other
infected groups. Only pre-treatment with 250 µg/ml resulted in
post-infection cell viability at a higher level compared with the
other infected groups. The co-incubation of the cells with the
virus and extract at a concentration between 62.5 and 250 µg/ml
effectively inhibited influenza infection and increased cell
survival to 80-100%. The anti-influenza properties were most
effective at the late stage of infection, which was confirmed by
the post-treatment experiment. Inoculation of extract after viral
pre-adsorption yielded the best results, showing that extract
dilutions down to 2 µg/ml successfully protected cells from
infection-induced death
To determine the Selectivity Index (SI), non-linear
regression analysis of cytotoxicity and pre-treatment assay
outcomes were calculated. The mean CC50 value was
>300 µg/ml, while the IC50 value was >13 µg/ml,
resulting in an SI index of 23 (Table
II).
 | Table IICC50, IC50 and
SI value of the Eucommiae Cortex ethanolic extract. |
Table II
CC50, IC50 and
SI value of the Eucommiae Cortex ethanolic extract.
| CC50 (µg/ml) | IC50
(µg/ml) | SI |
|---|
| >300 | >13 | 23 |
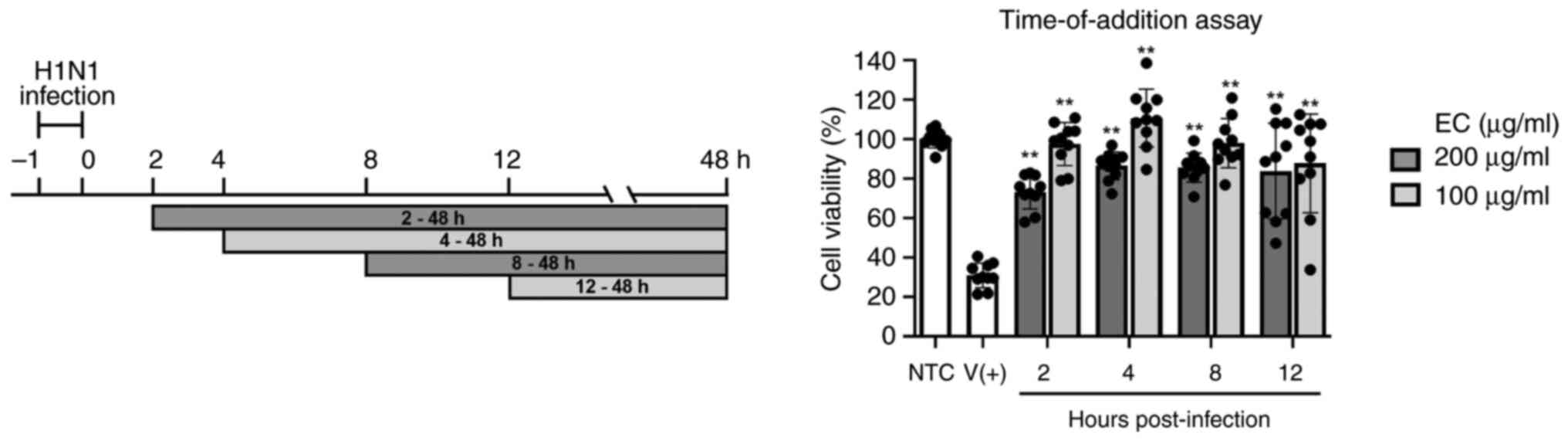
Time-of-addition assay
To determine the time-point of infection, EC extract
was added at different time-points (Fig. 8). The highest survival rate was
observed at 4 h post-infection, and all the sample-treated groups
showed significant improvements in cell conditions compared with
the infected-untreated group. These results verified the effective
therapeutic activity of EC extract against the H1N1 influenza
virus.
Discussion
With the emergence of influenza virus variants,
public health needs to acknowledge the need for the development of
novel antiviral treatments. In the present study, active compounds
of EC and targets associated with influenza were identified using
network pharmacology and molecular docking. Antiviral effects were
confirmed by in vitro experiments. Based on the
bioinformatics analysis, three main compounds (quercetin, genistein
and kaempferol) and four hub targets (IL6, BCL2, IL1B and TNF) were
identified. The C-T docking results indicated that kaempferol and
TNF showed the strongest binding. In the antiviral assays,
post-treatment with the EC extract had the most potent effect,
indicating its promising potential as a treatment.
Influenza virus initiates the activation of numerous
proinflammatory cytokines and chemokines (19). TNF-α is a cytokine that is
responsible for the immune response and has been reported to
increase susceptibility to H1N1 in humans (19). The upregulation of TNF-α expression
can cause an increase in cytokine production via the STAT3, MAPK
and NF-κB pathways, which leads to pulmonary edema and lung injury
(20). Based on the KEGG and
molecular docking results in the present study, the ‘TNF signaling
pathway’ was identified as a significant pathway and target, which
corresponds to previous research (20).
Kaempferol is a flavonoid found in various plants,
and possesses well-known antioxidant, anti-inflammatory and
antimicrobial properties (21).
Flavonoids, including kaempferol, are reported to be effective
viral NA inhibitors (22).
Kaempferol compounds from the extract of Eupatorium
perfoliatum L. have been reported to be effective against
influenza A (23). Another study
demonstrated that kaempferol extracted from Brazilian propolis
AF-08 suppressed viral growth in the respiratory tract (24). Furthermore, the orally administered
kaempferol group in mouse inhibited weight reduction and prolonged
survival compared with the non-administered group (24). In the present study, kaempferol was
identified as the main active compound in EC extract and it was
hypothesized that kaempferol exe5yhrts synergistic effects with
other compounds against influenza, which was verified by in
vitro experiments.
Network pharmacology is an evidence-based drug
discovery approach that explores novel compounds and targets.
However, its application to herbal formulas is limited by a lack of
standardization studies, and the parameters of OB and DL may vary
in clinical cases (25). While the
present study showed promising results, the adapted in vitro
model is a simplified representation of infection. Given that the
pathogenesis and influenza growth kinetics are well described in
MDCK cells, this cell line was used in the present investigation
(26,27). However, relying on a single cell
type may not capture the full spectrum of cellular interactions
involved in antiviral activity. Additionally, a fixed viral dose
(100 TCID50/well) and specific infection timing may not
accurately reflect the dynamics and complexity of the natural
course of infection. Furthermore, the current experiments focused
on the H1N1 virus, making it impossible to determine whether the
observed results apply to other influenza strains. Therefore,
further studies, including in vivo experiments, are required
in the future.
In conclusion, in silico and in vitro
studies were applied to identify novel compounds and targets of EC
due to complex matrices of plant extracts (28). Elucidating the underlying pathways
is difficult because of the synergistic effects of the active
compounds and multiple therapeutic targets (11). Computational research is a
promising method to identify novel compounds and targets in natural
product studies. It is suggested that the EC extract may possess
antiviral potential against influenza and can be developed as a
therapeutic application.
Supplementary Material
SRplot 081.
Acknowledgements
Not applicable.
Funding
Funding: The present study was supported by Konkuk
University.
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
AN and MK confirm the authenticity of all the raw
data. AN and MK contributed to the study conception and design. MK
performed material preparation, data collection and analysis, and
wrote the first draft of the manuscript. AN conducted the in
vitro study. Both authors commented on previous versions of the
manuscript. All authors read and approved the final version of the
manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Sellers SA, Hagan RS, Hayden FG and
Fischer WA: The hidden burden of influenza: A review of the
extra-pulmonary complications of influenza infection. Influenza
Other Respir Viruses. 11:372–393. 2017.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Krammer F: The human antibody response to
influenza A virus infection and vaccination. Nat Rev Immunol.
19:383–397. 2019.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Bouvier NM and Palese P: The biology of
influenza viruses. Vaccine. 26 (Suppl):D49–D53. 2008.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Kim H, Webster RG and Webby RJ: Influenza
virus: Dealing with a drifting and shifting pathogen. Viral
immunol. 31:174–183. 2018.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Li Y, Huo S, Yin Z, Tian Z, Huang F, Liu
P, Liu Y and Yu F: The current state of research on influenza
antiviral drug development: drugs in clinical trial and licensed
drugs. mBio. 14(e0127323)2023.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Krammer F, Smith GJD, Fouchier RAM, Peiris
M, Kedzierska K, Doherty PC, Palese P, Shaw ML, Treanor J, Webster
RG and García-Sastre A: Influenza. Nat Rev Dis Primers.
4(3)2018.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Zhao L, Yu X, Wu S, Xia K, Wang Y, Qin P,
Huang Z, Kang C, Yuan Z and Li Y: Pharmacokinetic profiling and
network pharmacology of honey-fried Licorice: An integrative
workflow to study traditional Chinese medicines (TCMs). J
Chromatogr B Analyt Technol Biomed Life Sci. 1:1248, 124353.
2024.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Huai Y, Zhang WJ, Wang W, Dang K, Jiang
SF, Li DM, Li M, Hao Q, Miao ZP, Li Y and Qian AR: Systems
pharmacology dissection of action mechanisms for herbs in
osteoporosis treatment. Chin Herb Med. 13:313–331. 2021.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Zhu MQ and Sun RC: Eucommia ulmoides
oliver: A potential feedstock for bioactive products. J Agric Food
Chem. 66:5433–5438. 2018.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Hussain T, Tan B, Liu G, Oladele OA, Rahu
N, Tossou MC and Yin Y: Health-promoting properties of eucommia
ulmoides: A review. Evid Based Complement Alternat Med.
2016(5202908)2016.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Kim M, Park KH and Kim YB: Identifying
active compounds and targets of Fritillariae thunbergii
against influenza-associated inflammation by network pharmacology
analysis and molecular docking. Molecules. 25(3853)2020.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Hopkins AL: Network pharmacology: The next
paradigm in drug discovery. Nat Chem Biol. 4:682–690.
2008.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Yu H, Chen J, Xu X, Li Y, Zhao H, Fang Y,
Li X, Zhou W, Wang W and Wang Y: A systematic prediction of
multiple drug-target interactions from chemical, genomic, and
pharmacological data. PloS One. 7(e37608)2012.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Agu PC, Afiukwa CA, Orji OU, Ezeh EM,
Ofoke IH, Ogbu CO, Ugwuja EI and Aja PM: Molecular docking as a
tool for the discovery of molecular targets of nutraceuticals in
diseases management. Sci Rep. 13(13398)2023.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Pantsar T and Poso A: Binding affinity via
docking: Fact and fiction. Molecules. 23(1899)2018.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Reed LJ and Muench H: A simple method of
estimating fifty per cent endpoints12. Am J Epidemiol. 27:493–497.
1938.
|
|
17
|
Tong X, Yang Y, Gong S, Han X, Wu Y and Ma
X, Lang S, Ma J and Ma X: Data mining and network pharmacology
analysis of kidney-tonifying herbs on the treatment of renal
osteodystrophy based on the theory of ‘kidney governing bones’ in
traditional Chinese medicine. Evid Based Complement Alternat Med.
30(1116923)2022.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Feng S, Wang T, Fan L, An X, Ding X, Wang
M, Zhai X, Cao Y, He J and Li Y: Exploring the potential
therapeutic effect of Eucommia ulmoides-Dipsaci Radix herbal
pair on osteoporosis based on network pharmacology and molecular
docking technology. RSC Adv. 12:2181–2195. 2022.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Ramos I and Fernandez-Sesma A: Modulating
the innate immune response to influenza a virus: Potential
therapeutic use of anti-inflammatory drugs. Front Immunol.
6(361)2015.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Shi X, Zhou W, Huang H, Zhu H, Zhou P and
Ju D: Inhibition of the inflammatory cytokine tumor necrosis
factor-alpha with etanercept provides protection against lethal
H1N1 influenza infection in mice. Crit Care.
17(R301)2013.PubMed/NCBI View
Article : Google Scholar
|
|
21
|
Periferakis A, Tsigas G, Periferakis AT,
Tone CM, Hemes DA, Periferakis K, Troumpata L, Badarau IA, Scheau
C, Caruntu A, et al: Kaempferol: A review of current evidence of
its antiviral potential. Int J Mol Sci. 24:2023.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Liu AL, Wang HD, Lee SM, Wang YT and Du
GH: Structure-activity relationship of flavonoids as influenza
virus neuraminidase inhibitors and their in vitro anti-viral
activities. Bioorg Med Chem. 16:7141–7147. 2008.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Derksen A, Kühn J, Hafezi W, Sendker J,
Ehrhardt C, Ludwig S and Hensel A: Antiviral activity of
hydroalcoholic extract from Eupatorium perfoliatum L.
against the attachment of influenza A virus. J Ethnopharmacol.
188:144–152. 2016.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Kai H, Obuchi M, Yoshida H, Watanabe W,
Tsutsumi S, Park YK, Matsuno K, Yasukawa K and Kurokawa M: In vitro
and in vivo anti-influenza virus activities of flavonoids and
related compounds as components of Brazilian propolis (AF-08). J
Funct Foods. 8:214–223. 2014.
|
|
25
|
Li Z, Qu B, Wu X, Chen H, Wang J, Zhou L,
Wu X and Zhang W: Methodology improvement for network pharmacology
to correct the deviation of deduced medicinal constituents and
mechanism: Xian-Ling-Gu-Bao as an example. J Ethnopharmacol.
289(115058)2022.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Chua S, Tan HQ, Engelberg D and Lim LHK:
Alternative experimental models for studying influenza proteins,
host-virus interactions and anti-influenza drugs. Pharmaceuticals
(Basel). 12(147)2019.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Tsai HC, Lehman CW, Lin CC, Tsai SW and
Chen CM: Functional evaluation for adequacy of MDCK-lineage cells
in influenza research. BMC Res Notes. 12(101)2019.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Kim M, Choi H, Kim S, Kang LW and Kim YB:
Elucidating the effects of curcumin against influenza using in
silico and in vitro approaches. Pharmaceuticals (Basel).
14(880)2021.PubMed/NCBI View Article : Google Scholar
|