Introduction
Medication reconciliation (MR) is defined as the
systematic process of comparing a current medication regimen of a
patient with prescribed orders to ensure consistency (1,2).
Through MR, clinical pharmacists can identify actual or potential
drug-related problems (DRPs), supervise and intervene in the
medication-taking behaviors of patients, and promptly negotiate and
adjust therapeutic plans with the multidisciplinary team to
safeguard the safety and efficacy of pharmacotherapy. Over the
years, the incidence of DRPs has been gradually increasing among
various diseases and populations, leading to adverse effects on the
therapeutic outcomes of diseases and individual health. For
example, Venugopal et al (3) conducted a systematic analysis of 11
prospective studies and six retrospective studies, and revealed
that the incidence of DRPs in patients with cancer ranged from
9.6-92.8%. Lau et al (4)
performed a meta-analysis of the incidence of DRPs in patients with
dementia by collecting data from six public datasets, and the
results indicated that the incidence of DRPs in patients with
dementia was 19%. In another study targeting home-dwelling
patients, it was revealed that the average number of DRPs per
individual could reach 4.16(5).
Regarding pediatric patients, Rashed et al (6) conducted a statistical analysis of
DRPs in children from the Hong Kong region and found that the
incidence of DRPs in pediatric patients was 21%, which was higher
than that in adult patients overall (6,7).
Clinical pharmacists, as patient-centered pharmaceutical care
professionals, are responsible for identifying, analyzing and
resolving DRPs (8-10).
Since the 1990s, pharmacists have employed classification systems
to achieve comprehensive and standardized qualitative management of
DRPs. At present, no universally accepted classification system
exists, but the most widely used schemes are the Pharmaceutical
Care Network Europe (PCNE) taxonomy (11) and the Strand classification system
(12). Research on DRPs in China
is still in its infancy. Nevertheless, in 2022, the Shanghai
Hospital Association released the first national classification
system, the Chinese Drug-Related Problem Classification System
(version 1.0) (13). However,
previous domestic studies have predominantly focused on adult
populations and have largely adopted foreign DRP classification
systems (14-18).
Investigations specifically addressing pediatric patients remain
limited.
The prevalence of chronic kidney disease (CKD) in
children globally is estimated to range from 14.9-118.8 per million
(19). Epidemiological research on
CKD in China is still developing; however, given the large
population base and the uneven distribution of medical resources
across different regions in China, CKD is characterized by high
prevalence, high mortality rates and low levels of awareness among
the general population (20). In
addition, patients with early-stage CKD often exhibit no notable
clinical symptoms, which renders them prone to being overlooked or
misdiagnosed during medical visits. This is particularly true for
pediatric patients, who are more likely to experience the
occurrence of DRPs (21). Ibrahim
et al (22) reported that,
in pediatric nephrology units, the incidence of DRPs among children
with CKD is substantial and markedly higher in inpatients than in
outpatients (51.2 vs. 32.0%, respectively). The treatment of
pediatric CKD is a prolonged, continuous process; complete recovery
during hospitalization is rare, necessitating sustained
post-discharge therapy (23,24).
Following discharge, the absence of a supervised environment often
leads to declining adherence over time. Moreover, omissions or
administration errors are common since young patients mainly depend
on caregivers for medication administration, further predisposing
them to DRPs. Additionally, patients referred from external
hospitals may have incomplete medication histories, resulting in
DRPs, such as therapeutic duplication or drug-drug interactions
(25,26). For pediatric patients with CKD, the
occurrence of DRPs can have adverse effects on their prognosis and
quality of life, such as disease progression, a decreased quality
of life, increased hospitalization rates, prolonged periods of
hospitalization and increased treatment costs (27-32).
Therefore, there is an urgent need to conduct research on DRPs in
pediatric patients with CKD, to identify common types and causes of
DRPs, and to provide references for the treatment and management of
CKD. At present, to the best of our knowledge, no studies have
systematically investigated DRPs in Chinese children with CKD.
The present study prospectively collected clinical
data on pediatric patients with CKD from the Guiyang Maternal and
Child Health Care Hospital (Guiyang, China) to determine the
incidence of DRPs in this population and to characterize their
types and underlying causes. Furthermore, the impact of
clinical-pharmacist interventions on DRPs was evaluated to inform
the development of evidence-based pharmaceutical care models for
chronic disease management in children.
Patients and methods
Participants and baseline
characteristics
The present single-center study was conducted at the
Guiyang Maternal and Child Health Care Hospital. Clinical
encounters of pediatric patients with CKD between January 1 and
December 31 2023 were collected. The inclusion criteria were as
follows: i) Aged 0-18 years; ii) patients with CKD at stages 1-5;
and iii) the use of ≥1 medication (medications used for therapeutic
purposes, including vitamin supplements) prior to admission for the
purpose of comparing pre- and post-admission differences in
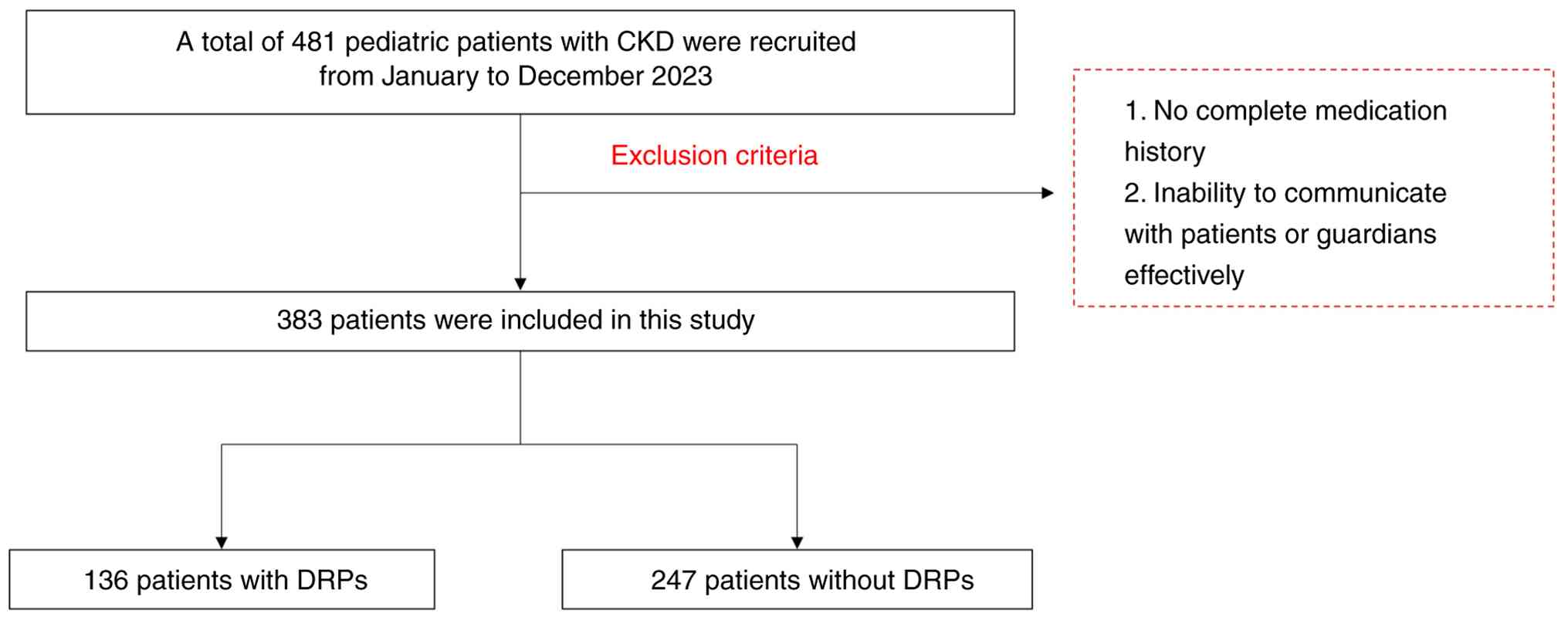
medication administration and identifying possible DRPs. A total of
481 patients initially met these criteria. However, the following
exclusion criteria were also applied: i) an incomplete medication
history; and ii) the inability to communicate effectively with the
patient or legal guardian. The final analytical cohort included 383
individuals through the exclusion process. A flow diagram of
participant recruitment is provided in Fig. 1. The study protocol was approved by
the Guiyang Maternal and Child Health Care Hospital Ethics
Committee (approval no. 2022-25) and was conducted in accordance
with The Declaration of Helsinki (World Medical Association,
2002).
Within 24 h of admission to the pediatric nephrology
ward, a structured data-collection form (admission medication
reconciliation/review sheet) was completed for each patient with
CKD. The following information was prospectively recorded:
demographic data (name, sex, age, date of admission and diagnosis
upon admission), previous medical history and complete
pre-admission medication history.
PCNE classification
The present study employed the PCNE DRP
classification system [version 9.1 (v9.1)] as the primary taxonomy.
To enhance its applicability to the study population, the original
PCNE v9.1 was modified following a careful review of physician
order-check records and documented clinical-pharmacist
interventions. Revisions consisted chiefly of the addition of
several causative sub-categories for DRPs. In brief, codes C1.7,
C1.8, C7.11-C7.17 and C8.2 were newly appended to the PCNE
(v9.1).
Identification and classification of
DRPs
MR was performed for all in-patients with pediatric
CKD. Potential DRPs were identified, their underlying causes were
analyzed, and targeted interventions were proposed to both
prescribers and caregivers; each identified DRP was subsequently
coded using the modified PCNE v9.1 framework.
Statistical analysis
Continuous variables that conformed to a normal
distribution were presented as mean ± standard deviation (SD),
whereas those that did not were expressed as median and
interquartile range (IQR). Categorical variables were expressed as
percentages. Variables with a normal distribution were analyzed
using the two-tailed, paired Student's t-test, whereas those
failing normality were compared using the Mann-Whitney U test.
Categorical variables were analyzed using the χ² test by default;
for categorical variables that did not meet the assumptions of the
χ² test, the Fisher's exact test was employed for statistical
analysis. Two-tailed P<0.05 was considered to indicate a
statistically significant difference. All statistical analyses were
performed using R software version 4.5.1 (RStudio, Inc.).
Results
Baseline characteristics of pediatric
patients with CKD
A total of 383 children met the predefined inclusion
criteria. The cohort comprised 205 male patients (53.52%) and 178
female patients (46.48%). Distribution by CKD stage was as follows:
Stage 1, 303 patients (79.11%); stage 2, 17 (4.44%); stage 3, 21
(5.48%); stage 4, 3 (0.78%); and stage 5, 39 (10.18%) patients. A
total of 78 children (20.37%) were receiving ≥8 concurrent drugs.
The median of hospitalization was 4 days and 62 patients (16.19%)
were hospitalized for ≥10 days. In addition, 292 (76.24%) patients
had been previously admitted for CKD management at least once
during the study period.
Occurrence of DRPs
Among the 383 enrolled patients, 136 (35.51%)
experienced at >1 DRP (Table
I). Within this subgroup, 71 patients were male (52.21%) and 65
were female (47.79%); the median of ages in these patients was
11.1, the median duration of hospitalization was 4.0 days, and the
median number of prescribed medications was 6.0. Notably, all 136
patients who developed DRPs had been readmitted at least once.
Among these variables, age, comorbidity, the number of medications
and hospital readmission were significantly associated with the
occurrence of DRPs.
 | Table IGeneral information of participants in
the present study. |
Table I
General information of participants in
the present study.
| Parameter | No DRPs (n=247) | DRPs (n=136) | P-value |
|---|
| Age, years | 9.1 (6.0, 11.9) | 11.1 (7.3, 13.0) | 0.005 |
| Age, years | | | 0.123 |
|
≤6 | 64 (25.9) | 25 (18.4) | |
|
>6 | 183 (74.1) | 111 (81.6) | |
| Sex, | | | 0.782 |
|
Female | 113 (45.7) | 65 (47.8) | |
|
Male | 134 (54.3) | 71 (52.2) | |
| Hospital time,
days | 4.0 (3.0, 8.0) | 4.0 (3.0, 7.0) | 0.952 |
| Hospital time,
days | | | 0.191 |
|
<10 | 202 (81.8) | 119 (87.5) | |
|
≥10 | 45 (18.2) | 17 (12.5) | |
| CKD | | | 0.712 |
|
1 | 199 (80.6) | 104 (76.5) | |
|
2 | 11 (4.5) | 6 (4.4) | |
|
3 | 13 (5.3) | 8 (5.9) | |
|
4 | 1 (0.4) | 2 (1.5) | |
|
5 | 23 (9.3) | 16 (11.8) | |
| Number of
comorbidities | 0.0 (0.0, 1.0) | 0.0 (0.0, 1.0) | 0.009 |
| Number of
medications | 6.0 (4.0, 7.0) | 6.0 (5.0, 7.2) | 0.035 |
| Number of
medications | | | 0.124 |
|
<8 | 203 (82.2) | 102 (75.0) | |
|
≥8 | 44 (17.8) | 34 (25.0) | |
| Hospital
readmission | | | <0.001 |
|
No | 91 (36.8) | 0 (0.0) | |
|
Yes | 156 (63.2) | 136 (100.0) | |
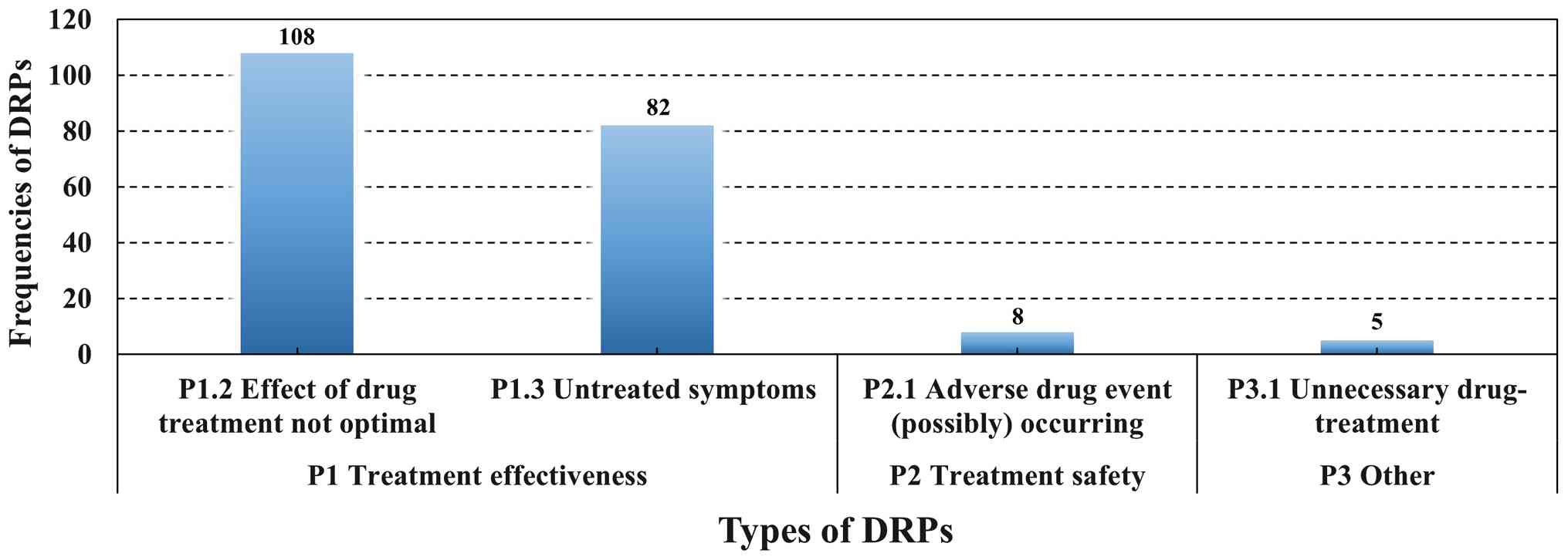
The classification of identified DRPs is presented
in Fig. 2. According to the PCNE
Classification for DRPs v9.1, DRPs are grouped into three primary
domains: P1 (treatment effectiveness), P2 (treatment safety) and P3
(other). P1 indicates a (potential) issue arising from the absence
or insufficiency of the pharmacotherapeutic effect; P2 indicates
that the patient is experiencing or is at risk of developing an
adverse drug event; and P3 indicates miscellaneous issues, such as
unnecessary drug therapy or an unclear/unspecified issue or
complaint. In the present study, the majority of DRPs fell within
domain P1 (treatment effectiveness). Specifically, 108 DRPs were
coded as P1.2 (sub-optimal drug effect) and 82 as P1.3 (untreated
symptom or indication). In addition, eight DRPs were categorized as
P2.1 [(potential) adverse drug event)] and five as P3.1
(unnecessary drug treatment).
Causes of DRPs
The related factors underlying the identified DRPs
are presented in Table II.
Regarding C1.7, ‘Drug is not discontinued promptly despite
resolution of the indication’, continuation of a drug following a
cure offers no further benefit and exposes the patient to needless
adverse effects and economic cost; a typical example is the
prolonged use of hepatoprotective agents following liver-enzyme
normalization. Concerning C1.8, ‘Incorrect prescription due to
similar medicine names’, names that look or sound alike frequently
mislead prescribers; for example, a child on vitamin A + D drops
may erroneously receive a prescription for vitamin D drops alone.
Categories C7.11-C7.17 capture patient-related factors that are not
explicitly itemized in the PCNE framework, such as the following:
Self-administration of the wrong product due to name similarity,
the unintentional omission of drugs amid polypharmacy, and
medication errors or non-adherence resulting from inadequate
caregiver supervision. Similarly, C8.2 refers to disruptions caused
by a change of healthcare setting (such as transfer from an
outpatient clinic to an inpatient ward), leading to erroneous or
missed dosing. By incorporating these additional descriptors, the
PCNE system can be refined to permit more precise identification
and classification of DRPs in the present pediatric CKD cohort.
 | Table IICauses of drug-related problems based
on the revised PCNE classification. |
Table II
Causes of drug-related problems based
on the revised PCNE classification.
| Primary domain | Category | Number of
cases | Proportion, % |
|---|
| C1, Drug
selection | C1.4 Inappropriate
duplication of therapeutic group or active ingredient | 1 | 0.45% |
| | C1.5 No or
incomplete drug treatment in spite of existing indication | 66 | 29.60% |
| | C1.7 Drug is not
discontinued promptly despite resolution of the
indicationa | 2 | 0.90% |
| | C1.8 Incorrect
prescription due to similar medicine namea | 10 | 4.48% |
| C3, Dose
selection | C3.1 Drug dose too
low | 17 | 7.62% |
| | C3.2 Drug dose of a
single active ingredient too high | 9 | 4.04% |
| | C3.3 Dosage regimen
not frequent enough | 23 | 10.31% |
| | C3.4 Dosage regimen
too frequent | 8 | 3.59% |
| C6, Drug use
process | C6.1 Inappropriate
timing of administration or dosing intervals by a health
professional | 7 | 3.14% |
| | C6.2 Drug
under-administered by a health professional | 2 | 0.90% |
| | C6.5 Wrong drug
administered by a health professional | 1 | 0.45% |
| C7, Patient
related | C7.1 Patient
intentionally uses/takes less drug than prescribed or does not take
the drug at all for whatever reason | 21 | 9.42% |
| | C7.2 Patient
uses/takes more drug than prescribed | 9 | 4.04% |
| | C7.4 Patient
decides to use unnecessary drug | 6 | 2.69% |
| | C7.7 Inappropriate
timing or dosing intervals | 11 | 4.93% |
| | C7.11 Incorrect
medication due to the patient self-purchasinga | 6 | 2.69% |
| | C7.12 Medication
not administered due to depletion of the supplya | 7 | 3.14% |
| | C7.13 Incorrect
medication due to similar medicine namea | 4 | 1.79% |
| | C7.14 Non-adherence
to prescription due to excessive medication usea | 1 | 0.45% |
| | C7.15 Medications
not taken regularly due to the lack of the medication
supervisora | 5 | 2.24% |
| | C7.16 Medication
supervisor forgets to supply medicationa | 1 | 0.45% |
| | C7.17 Incorrect
medication due to the change of medication supervisora | 1 | 0.45% |
| C8, Patient
transfer related | C8.2 Patient takes
an incorrect medication or does not take medication due to a change
in care institutiona | 5 | 2.24% |
Drug-selection issues constituted the largest
category (n=79), followed by patient-related factors (n=72),
dose-selection issues (n=57), drug-use process errors (n=10) and
care-transition failures (n=5).
DRP interventions and acceptance
In response to the 223 identified causal factors,
254 discrete interventions were proposed; 243 (95.67%) were fully
accepted and executed (Table
III). These were stratified by the intervention level as
follows: 112 (44.10%) were addressed to prescribers, of which 107
were fully implemented (acceptance rate 95.54%); 49 (19.29%)
targeted patients or caregivers, with 45 fully implemented
(91.84%); and 93 (36.61%) pertained to the drug product, with 91
fully implemented (97.85%).
 | Table IIIInterventions made by pharmacists,
acceptance and outcomes of intervention proposals classified by the
Pharmaceutical Care Network Europe version 9.1. |
Table III
Interventions made by pharmacists,
acceptance and outcomes of intervention proposals classified by the
Pharmaceutical Care Network Europe version 9.1.
| Primary domain | Types of
intervention/implementation/outcomes | Number of cases,
n | Proportion, % |
|---|
| I1, At prescriber
level | I1.1 Prescriber
informed only | 36 | 14.17% |
| | I1.3 Intervention
proposed to prescriber | 69 | 27.17% |
| | I1.4 Intervention
discussed with prescriber | 7 | 2.76% |
| I2, At patient
level | I2.2 Written
information provided (only) | 3 | 1.18% |
| | I2.4 Spoken to
family member/caregiver | 46 | 18.11% |
| I3, At drug
level | I3.1 Drug changed
to … | 9 | 3.54% |
| | I3.2 Dosage changed
to … | 29 | 11.42% |
| | I3.4 Instructions
for use changed to … | 29 | 11.42% |
| | I3.5 Drug paused or
stopped | 9 | 3.54% |
| | I3.6 Drug
started | 17 | 6.69% |
| A1, Intervention
accepted | A1.1 Intervention
accepted and fully implemented | 243 | 95.67% |
| | A1.2 Intervention
accepted, partially implemented | 3 | 1.18% |
| | A1.3 Intervention
accepted but not implemented | 6 | 2.36% |
| A2, Intervention
not accepted | A2.2 Intervention
not accepted: no agreement | 2 | 0.79% |
| O1, Solved | O1.1 Problem
totally solved | 243 | 95.67% |
| O3, Not solved | O3.1 Problem not
solved, lack of cooperation of patient | 3 | 1.18% |
| | O3.2 Problem not
solved, lack of cooperation of prescriber | 8 | 3.15% |
Post-intervention outcomes
Among the 136 patients with identified DRPs, the
underlying causes were completely resolved in 243 instances
(95.67%); 11 cases (4.33%) remained unresolved.
Discussion
Pediatric patients often have difficulty adhering
accurately to prescribed regimens due to limited swallowing
capacity and under-developed autonomy; consequently, medication
administration typically depends on a caregiver. In a previous
study, Zhang et al (33)
applied the PCNE taxonomy to 914 patients with CKD and reported
that ‘Treatment safety’ was the predominant DRP domain, followed by
‘Treatment effectiveness’. This hierarchy diverges from the
findings of the present study and is probably attributable to the
distinct demographic profile of their study cohort. In the present
study, MR identified DRPs that were predominantly attributable to
drug selection, patient-related factors and dose selection;
specifically, 66 DRPs arose from untreated or incompletely treated
indications, 23 from sub-optimal dosing frequency and 21 from
under-dosing or non-administration of prescribed doses. In a
previous study, Zafar et al (34), employing the PCNE framework in
patients from Pakistan with CKD, identified ‘inappropriate drug
selection’ as the most common root cause of DRPs; an identical
pattern was also reported in a separate cohort that consisted of
269 pre-dialysis patients recruited from the nephrology ward of
Ibni Sina Hospital, Ankara University School of Medicine (Ankara,
Turkey) between October 2019 and March 2020(35). These findings suggest that lapses
in caregiver or self-supervision serve a key role; compared with
adults, children exhibit a poorer comprehension of medication use
and, in the event that the caregiver lacks adequate medication
literacy or no consistent caregiver is available, safety and
effectiveness cannot be assured (36,37).
Patients with chronic diseases are frequently
re-hospitalized and among the 383 children enrolled in the present
study, 292 were repeat patients, of which 136 experienced DRPs;
conversely, no DRPs were observed in first-time patients.
Additionally, the ‘hospital readmission’ variable displayed a
significant association with the occurrence of DRPs, indicating
that repeated admissions may introduce gaps in medication
histories, particularly in pediatric patients, leading to
incomplete information at admission and subsequent DRPs. Moreover,
patients who experienced DRPs were significantly older, had more
comorbidities and were exposed to more concomitant medications. By
contrast, the duration of hospitalization and CKD stage did not
exert significant effects on the risks of developing DRPs.
Unexpectedly, a non-significant trend toward a higher DRP rate was
observed among children with a shorter duration of hospitalization.
Pehlivanli et al (35)
reported that DRPs were associated with a prolonged duration of
hospitalization, which may reflect age-specific care dynamics:
Extended periods of hospitalization in pediatric units may
facilitate intensive pharmaceutical stewardship, thereby
attenuating the occurrence of DRPs. Nevertheless, the absence of a
statistically significant duration of hospitalization between
groups in the present study may stem from limited statistical
power; the multi-center enlargement of the cohort is warranted to
clarify the true association between the duration of
hospitalization and DRPs in children with CKD. Notably, patients
with CKD in the early stages (particularly stage 1) face
disproportionate medication-management challenges. The present
study hypothesized that these patients often perceive their illness
as trivial, resulting in an inadequate appreciation of treatment
complexity and the critical importance of adherence. Frequent
therapeutic adjustments, repeated admissions and inter-facility
transfer further magnify the probability of prescribing errors,
duplication or omissions. Thus, clinical pharmacists could compare
physician orders with patient-reported use, perform MR, identify
potential issues, rationalize dosing schedules, revise medication
lists, and thereby mitigate medication-related risks and enhance
the safety of patients with CKD.
In 2022, the Shanghai Hospital Association and the
Shanghai Pharmaceutical Association released the Chinese
Drug-Related Problem Classification System (version 1.0). Compared
with the PCNE taxonomy, this system adds a ‘DRP evaluation’ domain
requiring explicit coding of ‘E1 primary subject’ and ‘E2 DRP
severity’ and introduces ‘C7 information systems and devices’ under
causes, while adjusting several other items; however, its target
population is adults. As the cohort in the present study comprised
children, certain categories in the Chinese system were not
applicable. During the pilot phase of the present project, the
original PCNE classification system was first applied to 100
pediatric in-patients and it was revealed that several root causes
of DRPs could not be adequately coded. On the basis of these
preliminary data, a revised version of the PCNE system was
developed that could provide exhaustive categories for the present
study population. Accordingly, in the present study, the PCNE
(v9.1) was adapted to reflect local pediatric practice, focusing on
the causal domain and adding 10 new codes. Multicenter validation
of the modified PCNE system is nevertheless required in future work
to confirm its reliability and generalizability for detecting DRP
causes in Chinese children.
During the present MR study, several deficiencies
and misconceptions among medication caregivers were identified.
Typical examples included the following: i) the omission or
incorrect administration of drugs due to polypharmacy and the
absence of a dedicated caregiver (such as a patient prescribed nine
concurrent medications failed to take hydroxychloroquine and
mycophenolate mofetil); ii) the substitution of prescribed therapy
with nutraceuticals of uncertain composition (for example,
caregivers purchased unlabeled calcium supplements instead of the
prescribed calcium preparation, resulting in persistent
hypocalcemia); iii) dose discrepancies arising from transitions of
care (such as an outpatient instruction to taper prednisone
according to weekly urinalysis was misinterpreted; thus, the dose
on readmission differed from the intended regimen); iv) the
intentional omission of chronic therapy at admission (for example,
enalapril was withheld without justification); v) the
administration of doses exceeding the manufacturer's labeled
maximum; and vi) the continuation of therapy after the indication
had resolved. Consequently, clinical pharmacists could use the
study-specific, pediatric-adapted PCNE (v9.1) to rapidly classify
DRPs and their causes, and to design targeted interventions, such
as regimen simplification, caregiver education, written action
plans and psychological support, which could enhance both the
efficacy and the safety of pharmacotherapy.
The present MR initiative successfully identified
and addressed DRPs in children with CKD, providing a replicable
model for pediatric chronic disease management. Nevertheless,
several limitations should be acknowledged. First, the
modifications to the PCNE v9.1 were derived solely from a
single-center cohort, and external validity across institutions and
pediatric chronic conditions remains to be established through
multicenter studies. Second, long-term outcomes, particularly
sustained adherence and disease control, were not evaluated. Future
research is thus warranted to expand the research to diverse
geographic regions and hospital tiers to enhance generalizability
and incorporate prospective follow-up to assess the impact of
pharmaceutical care on long-term prognosis, including disease
progression and quality of life.
In conclusion, the implementation of comprehensive
pharmaceutical care throughout the continuum of chronic disease
management represents an inevitable trajectory for the advancement
of pharmacy practice. The present study explored a MR model
tailored to pediatric patients with CKD and further refined the
PCNE v9.1 taxonomy to enable more accurate classification of DRPs
in this population. These enhancements facilitate the development
of individualized interventions.
Acknowledgements
Not applicable.
Funding
Funding: The present research was supported by the Guizhou
Provincial Health Commission Science and Technology Fund Project
(grant no. gzwkj2023-080).
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
XJ contributed to the project administration,
conceptualization, and reviewed and edited the manuscript. WY
contributed to the study conceptualization, project administration,
methodology, and reviewed and edited the manuscript. XL wrote the
original draft, made substantial contributions to conception and
design, and was a project administrator. YL contributed to the
curation, formal analysis and investigation of data. ZM designed
the methodology and supervised the study. XW curated the data,
designed the methodology and created figures. SZ contributed to the
investigation, data curation and methodology. QH contributed to the
methodology, formal analysis of data and creating figures. JL
contributed to the data curation, methodology and investigation. YZ
reviewed and edited the manuscript and contributed to the curation
of data. SL contributed to the resources, curation of data and
investigation. BC reviewed and edited the manuscript, made
substantial contributions to conception and design, and contributed
to the methodology. YH contributed to statistical analysis,
supervision, and creating figures. XJ and XL confirm the
authenticity of all the raw data. All authors read and approved the
final version of the manuscript.
Ethics approval and consent to
participate
The present study protocol was approved by the
Guiyang Maternal and Child Health Care Hospital Ethics Committee
(approval no. 2022-25) and was conducted in accordance with The
Declaration of Helsinki (World Medical Association, 2002). The
parents or guardians of all participants provided their written
informed consent.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
Use of artificial intelligence tools
During the preparation of this work, an AI tool
(Kimi) was used to improve the readability and language of the
manuscript, and subsequently, the authors revised and edited the
content produced by the AI tool as necessary, taking full
responsibility for the ultimate content of the present
manuscript.
References
|
1
|
Ellingsen G, Hertzum M, Severinsen GH and
Wynn R: Medication reconciliation as repair work. Stud Health
Technol Inform. 310:104–108. 2024.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Muller LS: Meaningful medication
reconciliation. Prof Case Manag. 30:74–76. 2025.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Venugopal J, S S, A R and Karnan D:
Drug-related problems in cancer patients: A systematic review. J
Oncol Pharm Pract. 30:562–571. 2024.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Lau ECY, Wojt I, Jeon YH, Hilmer SN and
Tan ECK: Prevalence and risk factors for drug-related problems in
people with dementia living in the community: A systematic review
and meta-analysis. J Am Med Dir Assoc. 23:980–991.e10.
2022.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Placido AI, Herdeiro MT, Morgado M,
Figueiras A and Roque F: Drug-related Problems in Home-dwelling
older adults: A systematic review. Clin Ther. 42:559–572.e14.
2020.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Rashed AN, Wilton L, Lo CC, Kwong BY,
Leung S and Wong IC: Epidemiology and potential risk factors of
drug-related problems in Hong Kong paediatric wards. Br J Clin
Pharmacol. 77:873–879. 2014.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Mi X, Zeng L and Zhang L: Systematic
review of the prevalence and nature of drug-related problems in
paediatric patients. J Clin Pharm Ther. 47:776–782. 2022.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Christiansen M, Joensen N and Jacobsen R:
The clinical pharmacist-led medication review service at the
surgical department: A feasibility study. Scand J Caring Sci.
39(e70030)2025.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Altawalbeh SM, Sallam NM, Al-Khatib M,
Alshogran OY and Bani Amer MS: Clinical pharmacist-led medication
reconciliation supplemented with medication review in admitted
patients with chronic kidney disease: A cost-benefit analysis. BMJ
Open. 15(e087232)2025.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Simal I, Capiau A, De Spiegeleer A, Velghe
A, Van Den Noortgate N, Petrovic M and Somers A: Evaluating an
integrated clinical pharmacist model in a geriatric day hospital: A
prospective single-centre observational study. Int J Clin Pharm.
47:1375–1383. 2025.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Griese-Mammen N, Hersberger KE, Messerli
M, Leikola S, Horvat N, van Mil JWF and Kos M: PCNE definition of
medication review: Reaching agreement. Int J Clin Pharm.
40:1199–1208. 2018.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Strand LM, Morley PC, Cipolle RJ, Ramsey R
and Lamsam GD: Drug-related problems: Their structure and function.
DICP. 24:1093–1097. 1990.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Clinical Pharmaceutical Administration
Committee of Shanghai Hospital Association and Hospital Pharmacy
Professional Committee of Shanghai Pharmaceutical Association.
Standard for the use of the Chinese classification system for
drug-related problems. Zhonghua Yu Fang Yi Xue Za Zhi.
56:1042–1049. 2022.PubMed/NCBI View Article : Google Scholar : (In Chinese).
|
|
14
|
Wang Y, Zhu J, Shan L, Wang C, Dong Z and
Yang W: Chinese Obesity and Metabolic Surgery Collaborative.
Drug-related problems in bariatric surgery: A retrospective study.
Obes Surg. 32:3961–3972. 2022.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Tian L, Wu J, Qi Z, Qian S, Zhang S, Song
D, Chen B and Zhu D: Drug-related problems among community-dwelling
elderly with ischemic stroke in China. Adv Clin Exp Med.
32:423–432. 2023.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Gu G, Li Y, Hu Y, Zhao H, Wang X, Li X,
Zhang X, Zhu H, Dai X, Liu X, et al: Prevalence and influence
factor of drug-related problems in inpatients with kidney disease:
A prospective single central study. Front Pharmacol.
15(1436561)2024.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Liu P, Li G, Han M and Zhang C:
Identification and solution of drug-related problems in the
neurology unit of a tertiary hospital in China. BMC Pharmacol
Toxicol. 22(65)2021.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Li BX, Guo SM, Wang YP, Zhong KX and Wei
YH: Identification and resolution of drug-related problems in the
cardiovascular surgery unit of a tertiary hospital in China: A
retrospective study. J Cardiothorac Surg. 20(8)2025.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Kidney Disease: Improving Global Outcomes
(KDIGO) CKD Work Group. KDIGO 2024 clinical practice guideline for
the evaluation and management of chronic kidney disease. Kidney
Int. 105 (Suppl):S117–S314. 2024.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Yu X, Xiong H, Wang M, Chen Y, Zhang A and
Li Q: Protocol for development of clinical practice guideline for
early screening of chronic kidney disease in children from China
(Version 2021). J Pediatr Pharm. 28:1–5. 2022.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Shi XM, Liu BN, Zhong XH, Wang F and Ding
J: Epidemiology of chronic kidney disease in children. Zhonghua Er
Ke Za Zhi. 57:721–724. 2019.PubMed/NCBI View Article : Google Scholar : (In Chinese).
|
|
22
|
Ibrahim N, Wong IC, Tomlin S, Sinha MD,
Rees L and Jani Y: Epidemiology of medication-related problems in
children with kidney disease. Pediatr Nephrol. 30:623–633.
2015.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Harada R, Hamasaki Y, Okuda Y, Hamada R
and Ishikura K: Epidemiology of pediatric chronic kidney
disease/kidney failure: Learning from registries and cohort
studies. Pediatr Nephrol. 37:1215–1229. 2022.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Chadha V and Warady BA: Epidemiology of
pediatric chronic kidney disease. Adv Chronic Kidney Dis.
12:343–352. 2005.PubMed/NCBI View Article : Google Scholar
|
|
25
|
de Geus A, Koppen IJN, Flint RB, Benninga
MA and Tabbers MM: An update of pharmacological management in
children with functional constipation. Paediatr Drugs. 25:343–358.
2023.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Herziger B, Jeschke S, Muller RM,
Neininger MP, Bertsche T and Bertsche A: Drug-handling problems and
expectations of the ideal pediatric drug-reported by children and
their parents. Eur J Pediatr. 181:2161–2171. 2022.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Kaplan B, Shimp LA, Mason NA and Ascione
FJ: Chronic hemodialysis patients. Part II: Reducing drug-related
problems through application of the focused drug therapy review
program. Ann Pharmacother. 28:320–324. 1994.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Tozawa M, Iseki K, Iseki C, Oshiro S,
Higashiuesato Y, Yamazato M, Tomiyama N, Tana T and Takishita S:
Analysis of drug prescription in chronic haemodialysis patients.
Nephrol Dial Transplant. 17:1819–1824. 2002.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Mirkov S: Implementation of a pharmacist
medication review clinic for haemodialysis patients. N Z Med J.
122:25–37. 2009.PubMed/NCBI
|
|
30
|
Pai AB, Boyd A, Chavez A and Manley HJ:
Health-related quality of life is maintained in hemodialysis
patients receiving pharmaceutical care: A 2-year randomized,
controlled study. Hemodial Int. 13:72–79. 2009.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Pai AB, Boyd A, Depczynski J, Chavez IM,
Khan N and Manley H: Reduced drug use and hospitalization rates in
patients undergoing hemodialysis who received pharmaceutical care:
A 2-year, randomized, controlled study. Pharmacotherapy.
29:1433–1440. 2009.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Manley HJ and Carroll CA: The clinical and
economic impact of pharmaceutical care in end-stage renal disease
patients. Semin Dial. 15:45–49. 2002.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Zhang S, Zhang GB, Huang P, Ren Y, Lin B,
Shao YF and Ye XL: Drug-related problems in hospitalized patients
with chronic kidney diseases and clinical pharmacist interventions.
BMC Geriatr. 23(849)2023.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Zafar R, Rehman IU, Shah Y, Ming LC, Goh
KW, Suleiman AK and Khan TM: Impact of pharmacist-led intervention
for reducing drug-related problems and improving quality of life
among chronic kidney disease patients: A randomized controlled
trial. PLoS One. 20(e0317734)2025.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Pehlivanli A, Eyupoglu S, Basgut B, Erturk
S and Ozcelikay AT: Impact of a multidisciplinary approach
involving clinical pharmacist on resolving drug related problems in
chronic kidney patients: A prospective interventional study. BMC
Nephrol. 24(149)2023.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Triplett P, Dyer H and Pascoe J:
Partnership between parents/caregivers and pediatricians when
children are hospitalized: Parents' perspective. Curr Probl Pediatr
Adolesc Health Care. 51(100999)2021.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Goncalves KMM, Costa M, Silva DCB, Baggio
ME, Correa ADR and Manzo BF: Ludic strategy for promoting
engagement of parents and caregivers in the safety of pediatric
patients. Rev Gaucha Enferm. 41(e20190473)2020.PubMed/NCBI View Article : Google Scholar
|