Introduction
The occurrence of secondary pulmonary lymphoma (SPL)
in patients diagnosed with non-Hodgkin's lymphoma (NHL) is not
rare. Apart from the gastrointestinal tract, the lungs are one of
the most frequent extra-nodal sites invaded by NHL, with 25-40%
reported in the literature (1,2). SPL
is part of systemic lymphoma and can invade lung tissue through
blood dissemination or transfer to the lung through hilar and
mediastinal lymph nodes. Clinically, patients frequently present
with an occasional cough and hemoptysis, the same symptoms as those
of pneumonia (3). Therefore, when
CT scans demonstrate pneumonia-like imaging features,
distinguishing between SPL and infectious pneumonia poses notable
diagnostic challenges. The risk of opportunistic infections is
influenced by the degree of pathogen exposure, host immune status
and pathogen-host interactions (4). Patients with hematologic malignancies
are prone to pulmonary opportunistic infections, which often
deteriorate rapidly and evolve into respiratory failure (5). However, diagnosis is often delayed
and occasionally completely overlooked, especially with invasive
bronchopulmonary aspergillosis. The commonly used diagnostic
methods for SPL include transbronchial lung biopsy or CT-guided
lung biopsy and surgical interventions. The present report outlines
2 cases of coexistent SPL and opportunistic infections in patients
with NHL diagnosed by minimal residual disease (MRD) detected in
bronchoalveolar lavage fluid (BALF), as well as a review of the
relevant literature.
Case report
Case 1
A 63-year-old woman with no past medical history of
lymphoma was diagnosed with peripheral T cell lymphoma following a
skin biopsy within the Department of Dermatology (Union Hospital,
Tongji Medical College, Huazhong University of Science and
Technology, Hubei, China), based on the symptoms of a skin rash and
pruritus 1 year prior to admission in October 2024. The patient
completed six cycles of 100 mg thalidomide + 50 mg/m2
etoposide + 750 mg/m2 cyclophosphamide + 50
mg/m2 hydroxydaunorubicin + 1.4 mg/m2
vincristine sulfate + 100 mg prednisone, and acquired complete
remission 2 months prior to hospitalization. Furthermore, 20 days
before admission to the Department of Critical Care Medicine, the
patient was diagnosed with a pulmonary infection and received 6
days of mechanical ventilation due to severe respiratory failure.
Following combination treatment with antibiotics, the patient was
successfully weaned and discharged. Subsequently, 1 week later, the
patient was re-admitted, presenting with a worsening cough, white
sputum and an intermittent fever (maximum of 39.4˚C). Compared with
the complete blood count 1 week ago, the white blood cell count of
the patient was higher (201.57x109/l; normal range,
3.5-9.5x109/l), with 84.2% lymphocytes. Additionally,
results of liver and kidney function, coagulation function and
echocardiography appeared similarly normal compared with 1 week
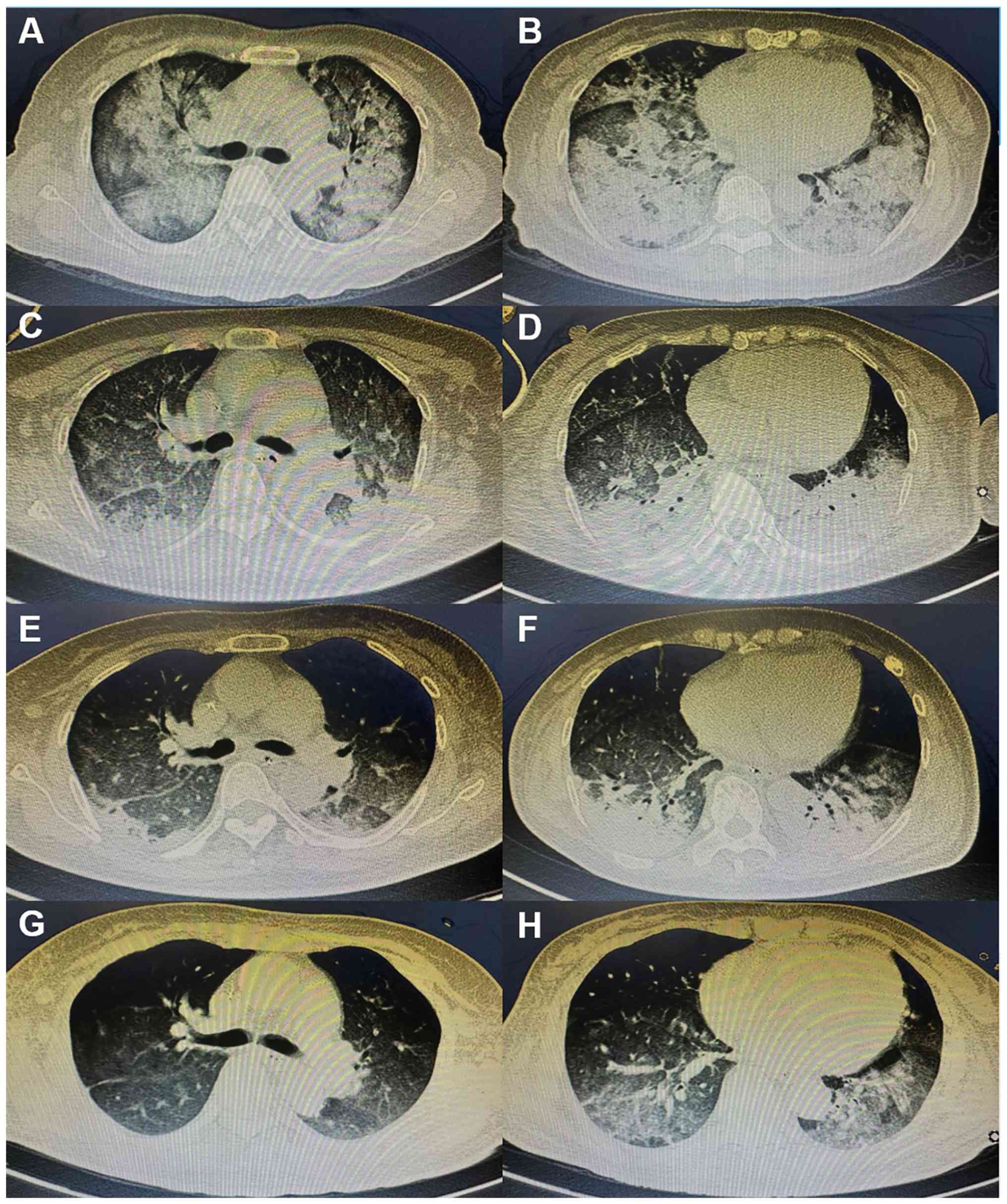
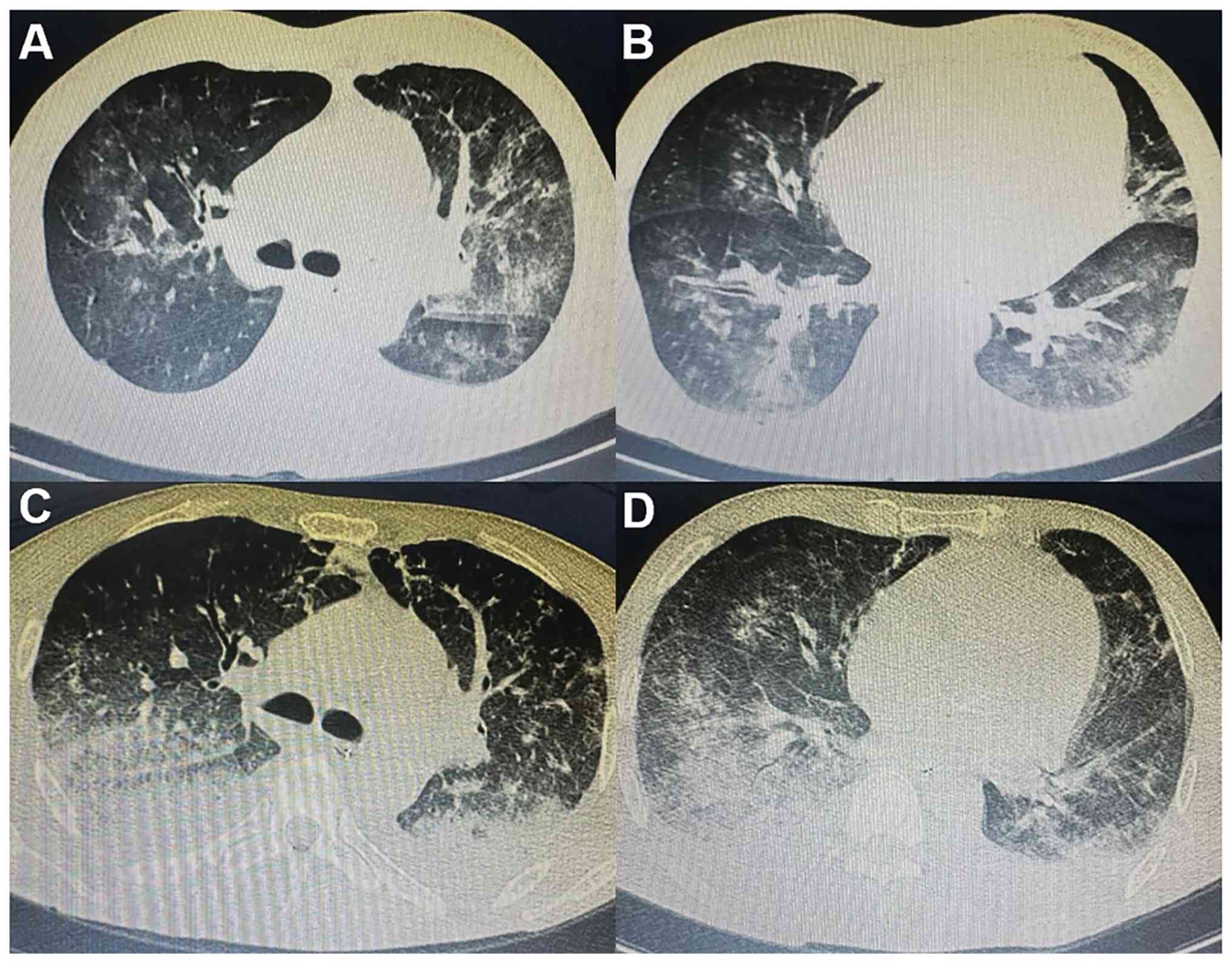
ago. Chest CT scans revealed enlarged consolidation, segmental
atelectasis and mediastinal lymph nodes (Fig. 1A-D). The patient therefore received
mechanical ventilation, due to respiratory failure and empirical
anti-infection treatment with cefoperazone sodium and sulbactam
sodium. Simultaneously, the patient underwent bronchoscopy and
bronchoalveolar lavage, which revealed thin, white secretions. BALF
was sent for metagenomic next-generation sequencing (mNGS)
(Table SI), revealing the
presence of human γ-herpesvirus 4 (sequence no. 118) and
Streptococcus pneumoniae (sequence no. 15) which did not
align with clinical symptoms or CT findings. Meanwhile, to rule out
bloodstream infection, peripheral blood specimens were also
collected and sent for mNGS testing. The results revealed that 217
sequences of Torque teno virus and 160 sequences of human
β-herpesvirus 6B were detected.
The patient did not undergo CT-guided percutaneous
lung biopsy due to coagulation dysfunction, but BALF was tested to
detect MRD and malignant cells were identified by abnormal
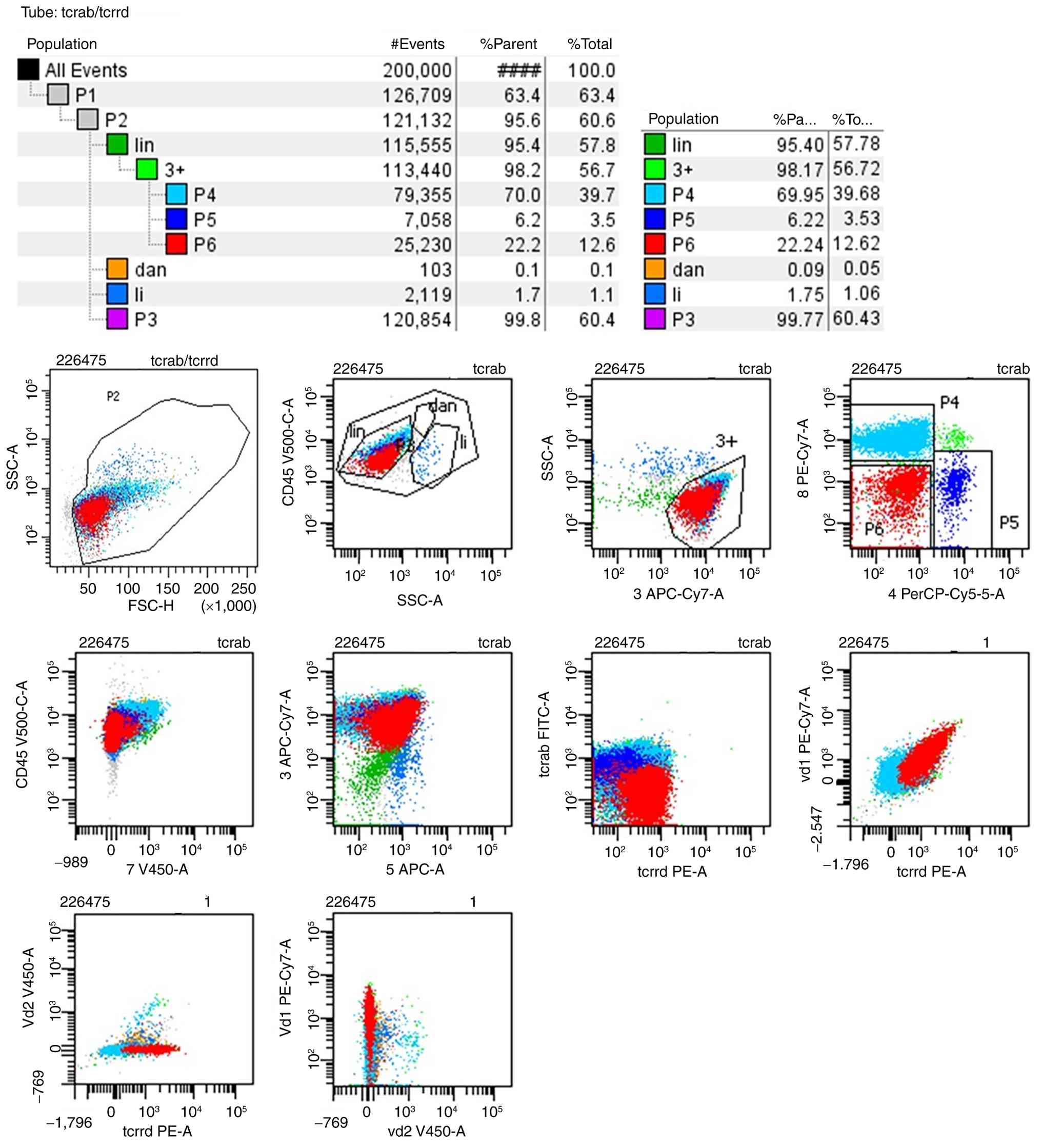
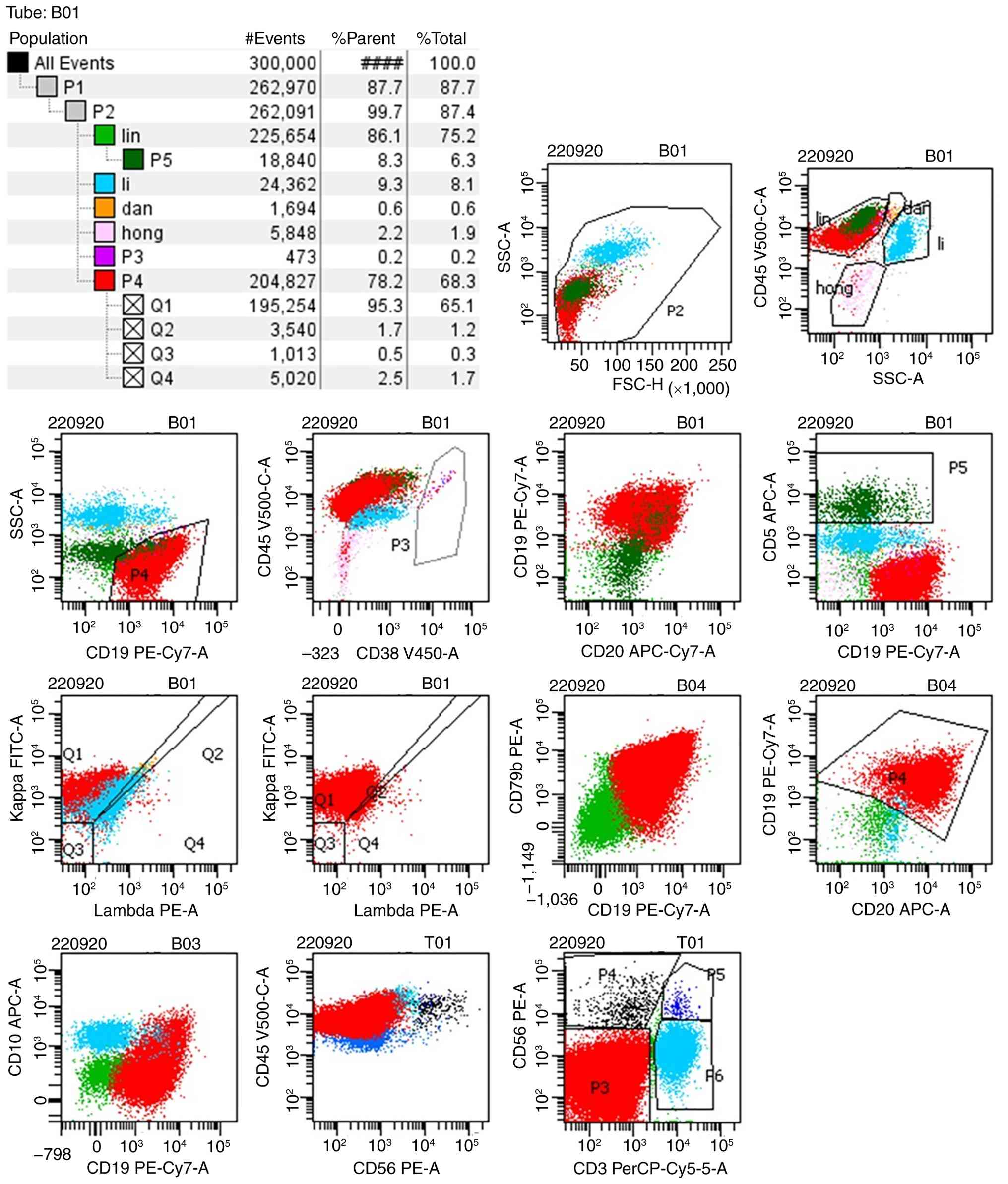
immunophenotypic molecules. The BALF MRD test results revealed that
94.21% karyocytes were lymphocytes and matured γδ T cells accounted
for 20.91% karyocytes exhibiting the phenotypes CD3+,
CD5+, T cell receptor (TCR)-γδ+, TCR
Vδ1+, CD4-, CD8-, CD7-
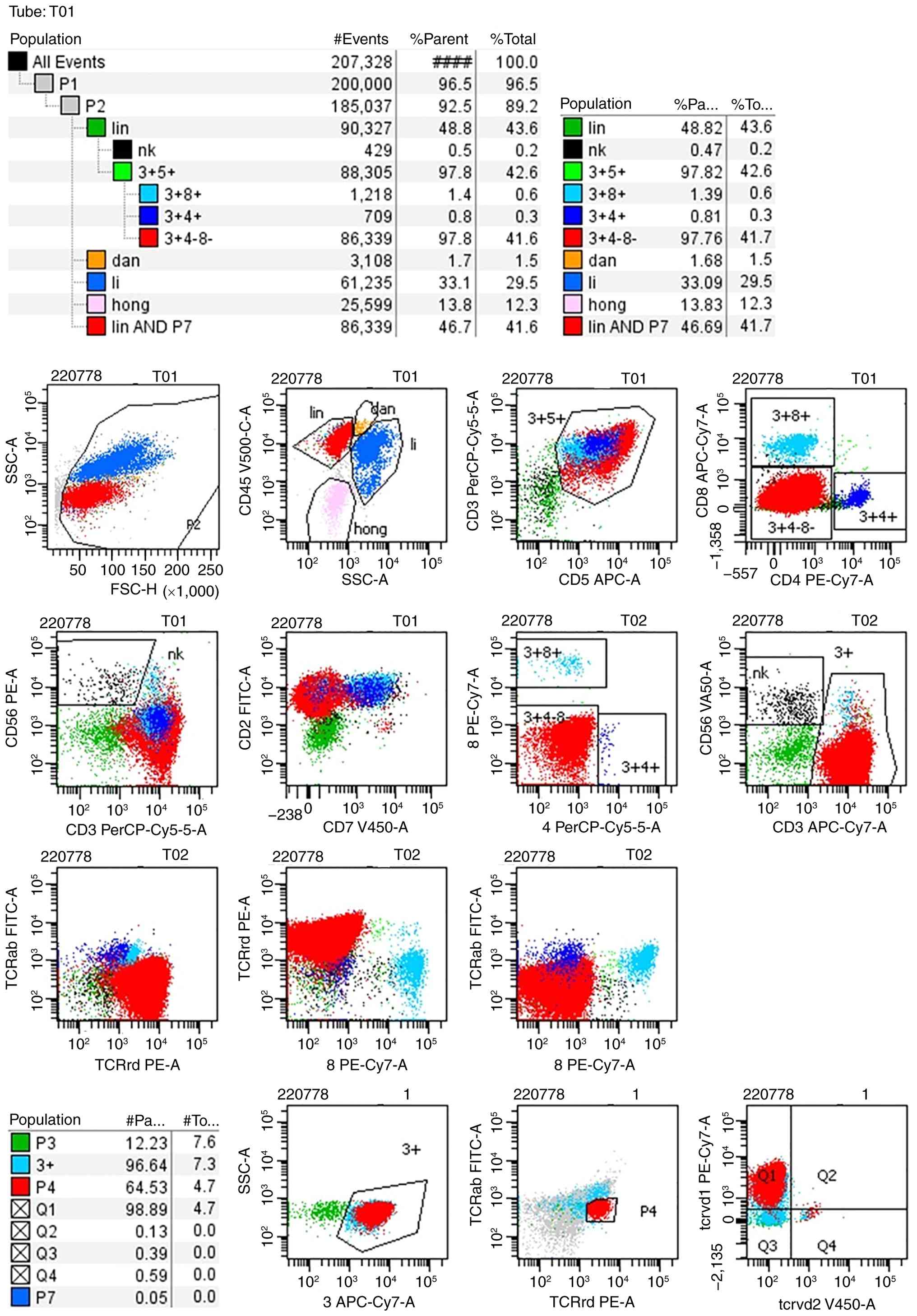
and TCRαβ- (Fig. 2),
consistent with the immunophenotypic profile observed in bone
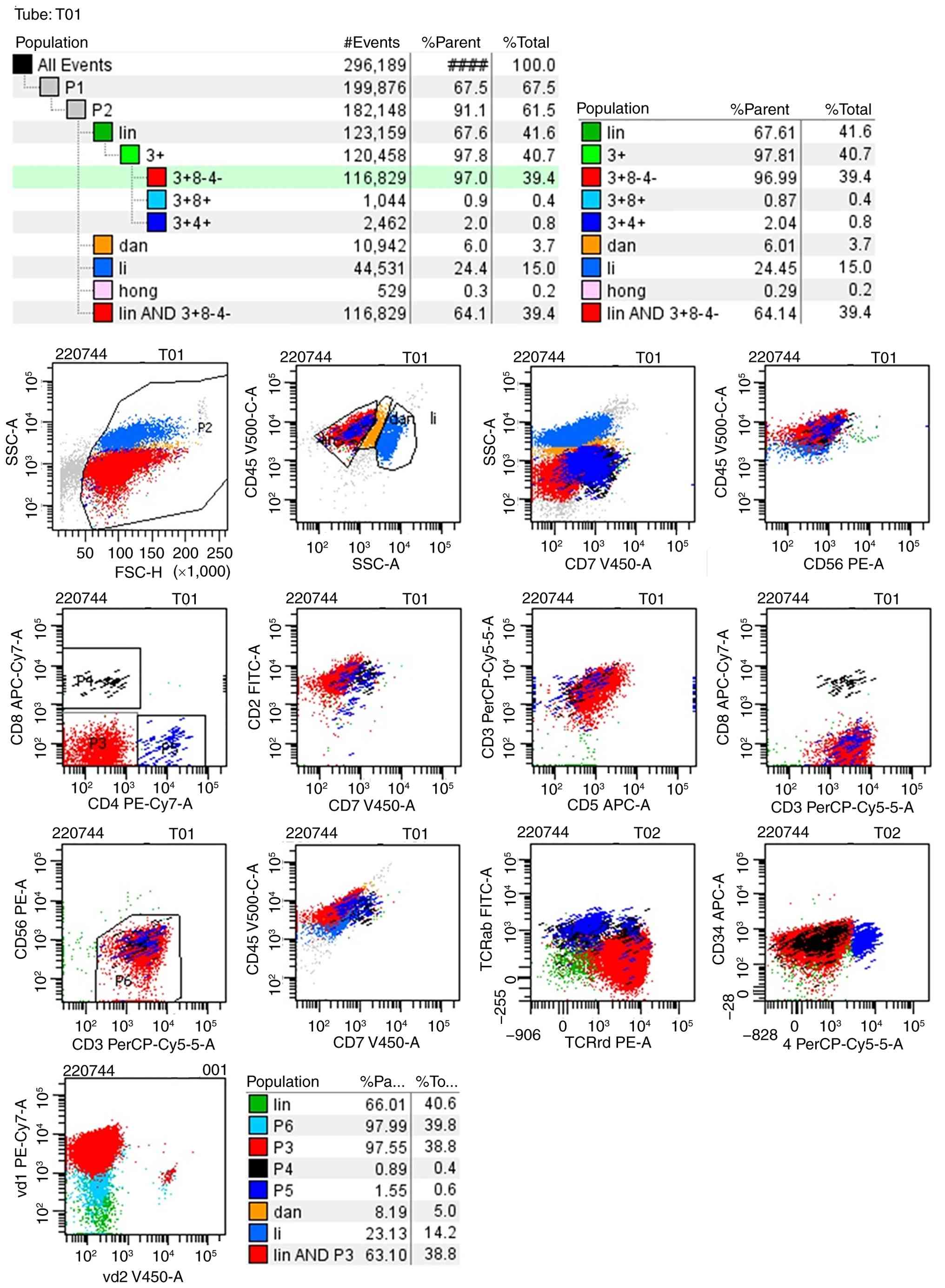
marrow (Fig. 3) and peripheral
blood samples (Fig. 4) from
peripheral T cell lymphoma. Until then, the patient had been
diagnosed with SPL and accordingly, had received methylprednisolone
(80 mg/day) and responded well. The oxygenation index of the
patient had markedly improved and the white blood cells count had
notably decreased. CT scans showed that the lung disease was
locally alleviated (Fig. 1G and
H). The patient was successfully
extubated on day 8 of mechanical ventilation. However, the patient
declined further antitumor treatment. Eventually the respiratory
failure continued to progress, leading to mortality 20 days
later.
Case 2
A 59-year-old man was diagnosed with follicular
lymphoma 6 years prior to the present hospitalization and achieved
complete remission following six cycles of rituximab CHOP
chemotherapy. The patient then received single-drug maintenance
therapy with rituximab. No follow-up treatment was conducted in the
previous 4 years before admission to the Department of Lymphoma of
Union Hospital (Tongji Medical College, Huazhong University of
Science and Technology, Hubei, China) in November 2024 with
systemic lymph node enlargement. The neck lymph node and bone
marrow biopsy both showed lymphoma infiltration. A regimen of 10
mg/m2 dexamethasone, 500 mg/m2
cyclophosphamide and 90 mg/m2 bendamustine were
administered as antitumor treatment. After 1 week, the patient
exhibited sudden respiratory distress with decreased blood oxygen
saturation (minimum 68%) and moist rale at the bottom of the lung.
The patient was then transferred to the intensive care unit for
further management, including mechanical ventilation. Laboratory
test results revealed a white blood cell count of
134.80x109/l with 16.42% neutrophils, 81.4% lymphocytes,
0.45% basophils, 0.22% eosinophils, 1.48% monocytes, an erythrocyte
count of 3.03x1012/l (normal range,
3.8-5.1x1012/l) and a platelet count of
128x109/l (normal range, 125-350x109/l). The
C-reactive protein level was 96.60 mg/l (normal range, 0-4 mg/l),
procalcitonin (PCT) was 4.32 ng/ml (normal range, 0-0.5 ng/ml) and
lactate dehydrogenase was 203 U/l (normal range, 109-245 U/l).
The initial sputum culture was extended-spectrum
β-lactamase (+), Escherichia coli and the sputum mNGS
revealed Moraxella catarrhalis (sequence no. 134),
Pneumocystis jiroveci (sequence no. 4) and Torque teno virus
(sequence no. 50). In addition, cytomegalovirus presence in whole
blood cells was negative whereas the Epstein-Barr virus was
positive (copy no. 6.82x103). Chest CT scans (Fig. 5A and B) showed large areas of ground-glass
opacity and patchy shadows. The patient was further diagnosed with
pulmonary infection and received empirical treatment with 1,000 mg
meropenem every 8 h, 50 mg caspofungin every day, 15 mg/kg
trimethoprim/sulfamethoxazole every day and 5 mg/kg ganciclovir
every 12 h combined with methylprednisolone (80 mg/day). After 2
weeks of anti-infection treatment, white blood cell counts and PCT
levels gradually reduced to normal, as did the oxygenation index.
However, the respiratory distress exhibited by the patient could
not be relieved, requiring continuous analgesia, sedation and
mechanical ventilation.
A large amount of pale-yellow thin secretions was
continually aspirated from the lung. CT imaging (Fig. 5C and D) showed that the lung-infiltrating
shadow was progressing. However, no pathogens were identified in
the subsequent sputum culture (which was repeated nine times) and
mNGS (which was repeated twice) of BALF. Specifically, the patient
underwent two rounds of BALF mNGS testing since the results of the
first test could not explain the pulmonary condition of the
patient. BALF was immediately collected for re-testing on the day
after the initial results were obtained. However, the two negative
results prompted the conclusion that the patient did not have a
pulmonary infection, but rather an alternative diagnosis such as
SPL. SPL was not confirmed until numerous monoclonal B cells with
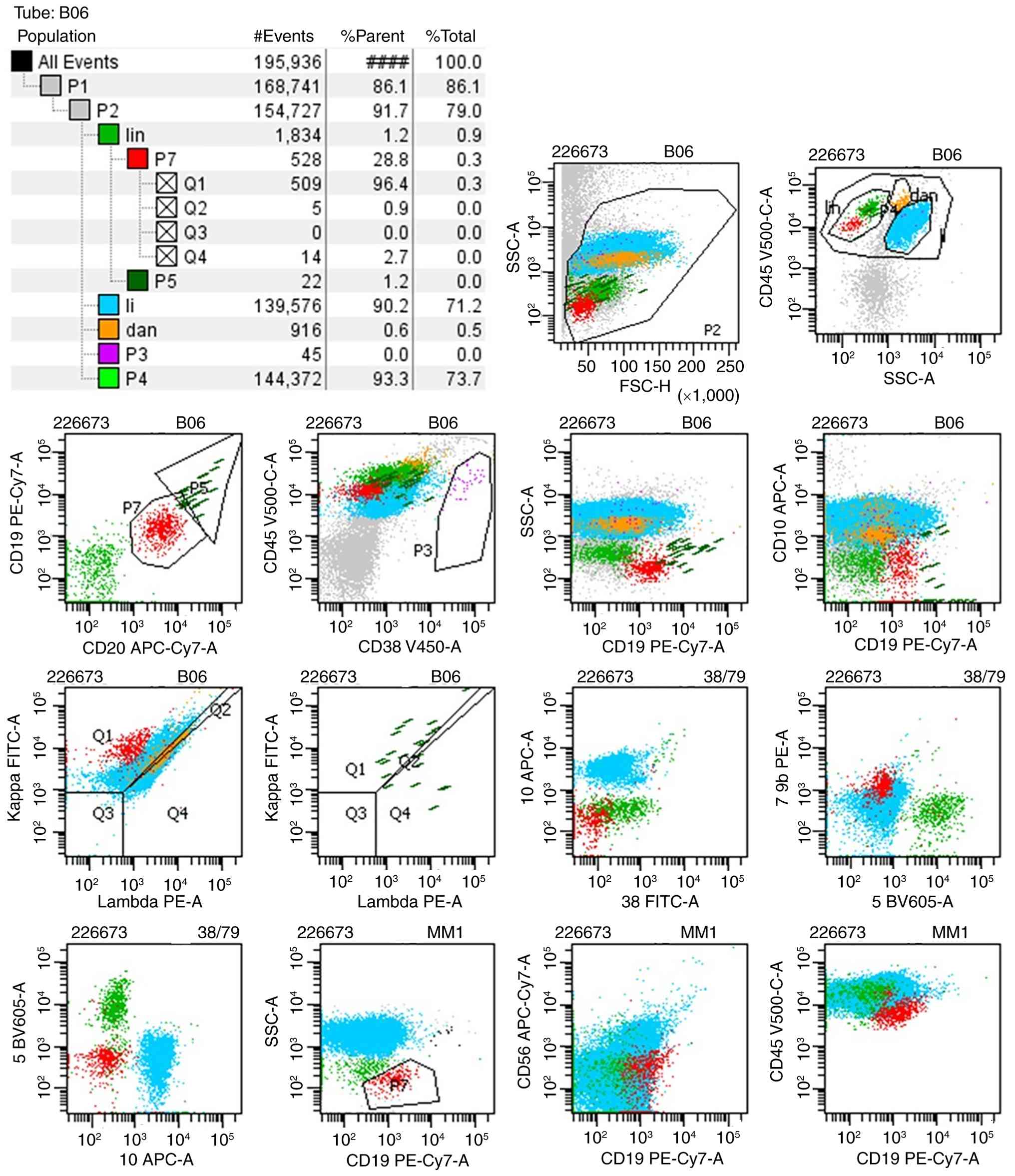
an abnormal phenotype were identified by MRD in BALF. Subsequently,
~140,000 karyocytes were collected from the BALF, of which 0.28%
were monoclonal B cells with an abnormal immunophenotype:
CD19dim, κ+, CD20+,
CD45dim, CD79b+, λ-,
CD5-, CD10-, CD38-,
CD56- and small forward and side scatter (Fig. 6). This profile was consistent with
the immunophenotyping results of bone marrow cells (Fig. 7). The antitumor treatment of the
patient was ineffective, resulting in mortality due to respiratory
failure on day 15 of mechanical ventilation.
Literature review
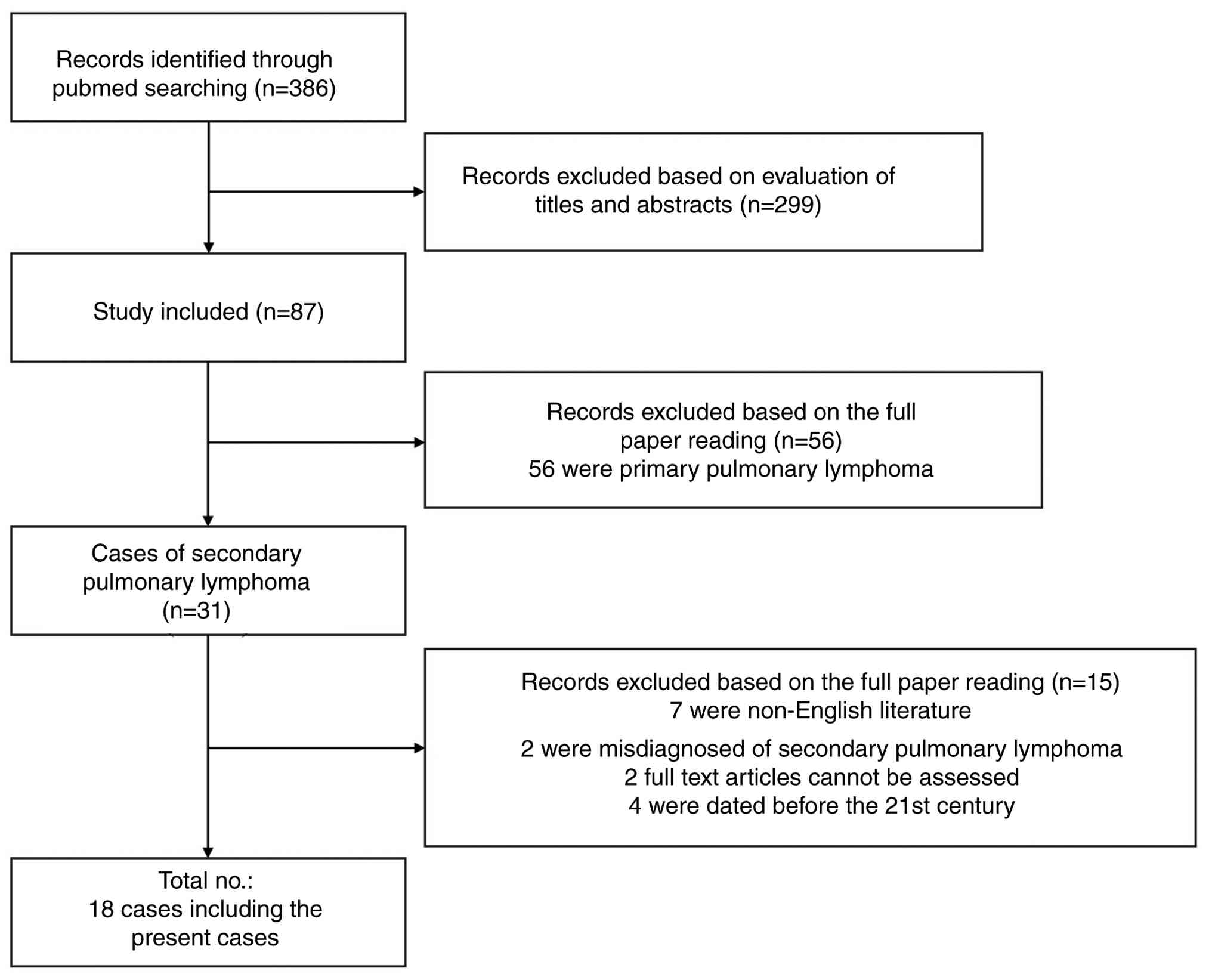
A combination of search terms (lymphoma and
pulmonary infiltrates) was used to screen abstracts and titles
under case reports in the PubMed database (https://pubmed.ncbi.nlm.nih.gov/). A total of 386
relevant studies were identified. After evaluating the titles and
abstracts, 299 irrelevant reports and duplicate cases were
excluded. Within the remaining 87 studies, 56 consisted of primary
pulmonary lymphoma and 31 contained SPL. Of these 31 cases, 11 were
excluded due to non-English language use or a misdiagnosis of SPL.
An additional four were excluded due to being dated before the 21st
century. Ultimately, 16 patient case reports were enrolled for
analysis (6-21).
Fig. 8 exhibits the screening
process and Table I outlines the
basic information of 18 patients who experienced SPL (including the
2 cases outlined in the present report).
 | Table IReported cases of secondary pulmonary
lymphoma. |
Table I
Reported cases of secondary pulmonary
lymphoma.
| Study no. | First author,
year | Age, years | Sex | Lymphoma type | Study title | (Refs.) |
|---|
| 1 | Miyahara et
al, 2001 | 27 | F | Diffuse large B
cell non-Hodgkin's malignant lymphoma | Pulmonary lymphoma
of large B cell type mimicking Wegener's granulomatosis | (6) |
| 2 | Wieker et
al, 2002 | 63 | F | MALT-lymphoma | Pulmonary low-grade
MALT-lymphoma associated with localized pulmonary amyloidosis. A
case report | (7) |
| 3 | Kusumoto et
al, 2004 | 35 | F | MALT-lymphoma | T(11;18)-bearing
pulmonary mucosa-associated lymphoid tissue lymphoma responding to
cladribine | (8) |
| 4 | Fujisawa et
al, 2007 | 62 | M | Peripheral T cell
lymphoma | Peripheral T cell
lymphoma with diffuse pulmonary infiltration and an increase in
serum KL-6 level | (9) |
| 5 | Jankipersadsing
et al, 2007 | 49 | F | Adult T cell
leukemia/lymphoma | Spontaneous
regression of pulmonary infiltration of adult T cell
leukemia/lymphoma | (10) |
| 6 | Hosseinnezhad et
al, 2011 | 82 | M | Chronic lymphocytic
leukemia | Diffuse pulmonary
infiltrates in an old man with chronic lymphocytic leukemia | (11) |
| 7 | Samiullah et
al, 2014 | 60 | M | MALT-lymphoma | Metastatic gastric
MALT lymphoma masquerading as pulmonary infiltrates, with a
dramatic response to chemotherapy | (12) |
| 8 | Sedgwick et
al, 2015 | 81 | F | Anaplastic large
cell lymphoma | Lesson of the month
1: A rash decision | (13) |
| 9 | Suzuki et
al, 2017 | 63 | M | Diffuse large B
cell lymphoma | EBV-positive
diffuse large B cell lymphoma as a secondary malignancy arising in
a myelodysplastic syndrome patient who was treated with
azacitidine | (14) |
| 10 | Kajimoto et
al, 2020 | 53 | F | Diffuse large B
cell lymphoma + T cell lymphoma | T cell lymphoma
with a granulomatous lesion of the lungs after autologous
hematopoietic stem cell transplantation for Epstein-Barr
virus-positive diffuse large B cell lymphoma: A unique rare case of
metachronous B cell and T cell lymphoma | (15) |
| 11 | Yagyu et al,
2020 | 82 | F | Follicular
lymphoma | Malignant lymphoma
mimics miliary tuberculosis by diffuse micronodular radiographic
findings | (16) |
| 12 | Miyaoka et
al, 2020 | 39 | M | T-acute
lymphoblastic leukaemia/lymphoblastic lymphoma | Aggressive lung
involvement in a patient with T-acute lymphoblastic
leukaemia/lymphoblastic lymphoma: A tricky and rare case
report | (17) |
| 13 | Tanaka et
al, 2022 | 69 | M | Intravascular large
B cell lymphoma | Rapid deterioration
of intravascular large B cell lymphoma with mass formation in the
trigeminal nerve and multiple organ infiltration: An autopsy case
report | (18) |
| 14 | Kobe et al,
2022 | 79 | F | Adult T cell
lymphoma/leukaemia | Cryobiopsy for
Pneumocystis jirovecii pneumonia secondary to adult T cell
lymphoma/leukaemia | (19) |
| 15 | Yamamoto et
al, 2022 | 73 | M | Angioimmunoblastic
T cell lymphoma | Use of
bronchoalveolar lavage in diagnosing angioimmunoblastic T cell
lymphoma: A case report | (20) |
| 16 | Tanaka et
al, 2022 | 76 | M | Intestinal T cell
lymphoma | Intestinal T cell
lymphoma with lung and lymph node involvement at relapse | (21) |
| 17 | The present
study | 63 | F | Peripheral T cell
lymphoma | Secondary pulmonary
lymphoma diagnosed by minimal residual disease detected in
bronchoalveolar lavage fluid: A case report | - |
| 18 | The present
study | 59 | M | Follicular
lymphoma | Secondary pulmonary
lymphoma diagnosed by minimal residual disease detected in
bronchoalveolar lavage fluid: A case report | - |
Within the 18 cases, SPL occurred in individuals of
both sexes and varying ages. The ratio of male to female occurrence
was 1:1 with a higher frequency among individuals aged >60 years
(Table II). The three most
prevalent clinical manifestations in these patients were dyspnea
(66.7%), cough (33.3%) and a fever (27.8%). These symptoms are
consistent with the 2 cases outlined in the present report.
However, the patients in the present report manifested a cough with
a large amount of pulmonary secretion, but did not exhibit
hemoptysis. According to the literature, the proportion of
asymptomatic patients was as high as 27.8%. It is worth noting that
within these 18 patients, only 2 (11.1%) were associated with
pulmonary infection. Among the chest CT findings, the majority of
SPL exhibited bilateral lung infiltration, with only 2 cases
showing unilateral infiltration (Table III). The majority of chest
imaging findings suggested ground glass density (50%), lymph node
enlargement (33.3%), multiple pulmonary nodules (44.4%) and lung
consolidation (27.8%). Other presentations included single lymph
node enlargement, pleural effusion and cavities.
 | Table IIAnalysis of information regarding
lung infiltration secondary to lymphoma. |
Table II
Analysis of information regarding
lung infiltration secondary to lymphoma.
| Feature | Cases, n (%) |
|---|
| Age, years | |
|
≤60 | 7 (38.9) |
|
>60 | 11 (61.1) |
| Sex | |
|
Male | 9 (50.0) |
|
Female | 9 (50.0) |
| Respiratory
symptoms | |
|
Asymptomatic | 5 (27.8) |
|
Cough | 6 (33.3) |
|
Dyspnea | 12 (66.7) |
|
Fever | 5 (27.8) |
|
Hemoptysis | 2 (11.1) |
|
Chest
pain | 0 (0.0) |
|
Coexisting
with pulmonary infection | 2 (11.1) |
| Outcome | |
|
Remission | 8 (44.4) |
|
Mortality | 10 (55.6) |
 | Table IIIImaging features of the chest. |
Table III
Imaging features of the chest.
| Feature | Cases, n (%) |
|---|
| Bilateral
disease | 16 (88.9) |
| Unilateral
disease | 2 (11.1) |
| Ground glass
opacity | 9 (50.0) |
| Consolidation | 5 (27.8) |
| Single nodule or
mass | 1 (5.6) |
| Multiple
nodules | 8 (44.4) |
| Pleural
effusion | 3 (16.7) |
|
Lymphadenopathy | 6 (33.3) |
| Cavity | 1 (5.6) |
Pathological characteristics were determined by a
pathologist in all 18 cases (Table
IV). There were no cases of Hodgkin's lymphoma. However, 4
cases were confirmed to contain diffuse large B cell lymphoma and
there were 3 cases of mucosa-associated lymphoid tissue (MALT) and
adult T cell lymphoma/leukemia as well as 2 cases of peripheral T
cell lymphoma and follicular lymphoma, which were confirmed
separately. The remaining cases were confirmed as
angioimmunoblastic T cell lymphoma, T-acute lymphoblastic
leukemia/lymphoblastic lymphoma, anaplastic large cell lymphoma and
chronic lymphocytic leukemia.
 | Table IVPathological types of the 18
cases. |
Table IV
Pathological types of the 18
cases.
| Pathological
type | Cases, n (%) |
|---|
| Hodgkin's
lymphoma | 0 (0.0) |
| Mucosa-associated
lymphoid tissue | 3 (16.7) |
| Diffuse large B
cell lymphoma | 4 (22.2) |
| Angioimmunoblastic
T cell lymphoma | 1 (5.6) |
| Peripheral T cell
lymphoma | 2 (11.1) |
| Adult T cell
lymphoma/leukemia | 3 (16.7) |
| Follicular
lymphoma | 2 (11.1) |
| T-acute
lymphoblastic leukemia/lymphoblastic lymphoma | 1 (5.6) |
| Anaplastic large
cell lymphoma | 1 (5.6) |
| Chronic lymphocytic
leukemia | 1 (5.6) |
There were a number of differential diagnoses of
secondary pulmonary infiltration in patients with lymphoma,
including SPL, drug-related lung injury, connective tissue disease
and pulmonary infections, especially pulmonary fungal infections
and tuberculosis. During the diagnostic process, the persistent
clinical symptoms and the radiographic manifestations served as key
indicators for clinicians to initiate therapeutic interventions.
Diagnostic methods often involved numerous types of invasive
methods, such as transbronchial lung biopsy (50%), surgery or
thoracoscopic lung biopsy (27.8%), autopsy (16.7%) and BALF
immunophenotyping test (27.8%) (Table
V). On occasion, multiple biopsy methods were involved in the
diagnostic process. Furthermore, it was observed that although
transbronchial lung biopsy was used more frequently, the success
rate was only 55.6%. Almost half of the patients needed to undergo
other methods at the same time due to negative pathological
findings or suboptimal biopsy specimen quality. In addition, unlike
traditional invasive methods, in the present report, lymphoma cells
were detected in BALF in only 5 cases, which has rarely been
reported before, despite 1 case initially failing to diagnose SPL
and requiring surgical biopsy for a definitive answer (case no.
11).
 | Table VDiagnostic methods and accuracy of
the diagnosis process. |
Table V
Diagnostic methods and accuracy of
the diagnosis process.
| Diagnostic
method | Cases, n (%) | Success rate,
% |
|---|
| CT-guided
percutaneous lung biopsy | 0 (0.0) | 0.0 |
| Transbronchial lung
biopsy | 9 (50.0) | 55.6 |
| Surgery or
thoracoscopy lung biopsy | 5 (27.8) | 100.0 |
| Autopsy | 3 (16.7) | 100.0 |
| Bronchoalveolar
lavage fluid | 5 (27.8) | 80.0 |
With regard to treatment and prognosis, 7 patients
received combination chemotherapy after SPL diagnosis, 2 patients
received single-agent chemotherapy with chlorambucil or cladribine
respectively, 2 patients received only glucocorticoid therapy, 1
patient received prednisolone followed by allogeneic hematopoietic
stem cell transplantation and 4 patients received no treatment or
only palliative care. The overall prognosis of SPL was poor, with
only 44.6% of enrolled patients exhibiting ongoing survival
(Table II). However, early
diagnosis and treatment has the potential to improve prognosis. As
for the 2 cases outlined in the present report, the first patient,
who had just achieved complete remission 2 months prior and
developed a pulmonary infection during the treatment course of
admission, received only methylprednisolone for antitumor therapy.
The second patient received antitumor therapy with dexamethasone,
cyclophosphamide and bendamustine after admission and continued
with methylprednisolone treatment after an SPL diagnosis was
confirmed. Both patients eventually progressed rapidly to
mortality. The marked differences in treatments among patients may
be attributed to individualized risk-benefit assessments based on a
number of key factors differing between each patient. These may
include disease characteristics (pathological confirmation, stage,
tumor burden and aggressiveness of the lesion) and patient-specific
factors (age, performance status, comorbidities, organ function and
previous treatment history). Additionally, the treatment
preferences of patients themselves also serve an important role in
the selection of treatment regimens. Between the 2 cases in the
present report, the first declined following further antitumor
treatment. By contrast, the second patient underwent combination
chemotherapy immediately upon confirmation of relapsed lymphoma
after admission.
Discussion
Within the present report, 2 cases of patients
diagnosed with NHL, but having achieved complete remission
following CHOP treatment, were outlined. The patients subsequently
developed respiratory distress. Due to the pneumonia-like symptoms,
signs and chest CT scan findings, the patients were initially
diagnosed with pulmonary infection. Following combination treatment
with multiple antibiotics, their sputum culture and mNGS of BALF
were both negative. However, the patients continued to develop a
fever and the imaging findings continued to progress in both lungs.
The MRD tests of BALF were performed to establish the existence of
SPL. Based on the diagnosis of SPL, a glucocorticoid-based
treatment regimen was implemented, a method consistent with a
number of reported cases (18,20).
However, both patients ultimately succumbed to respiratory failure
within 1 month. The lung is a common secondary site of involvement
in lymphoma, accounting for ~24% of cases in NHL (22). The clinical symptoms of SPL are
non-specific, except for respiratory and extrapulmonary symptoms.
However, common symptoms include dyspnea (66.7%), cough (33.3%) and
fever (27.8%). The proportion of asymptomatic patients was as high
as 27.8% in the reported literature. In previous reports, the
imaging findings of SPL have included ground glass density (50%),
lymph node enlargement (33.3%), multiple pulmonary nodules (44.4%)
and lung consolidation (27.8%). Due to the atypical clinical
manifestations, the majority of cases were initially misdiagnosed
as tuberculosis, pneumonia or deep fungal infections.
The lung is a predominant site for opportunistic
infections. The risk of such infections is determined by the extent
of pathogen exposure, host immunological competence and specific
pathogen-host interaction mechanisms (4). Among patients with hematological
malignancies, pulmonary opportunistic infections deteriorate
rapidly and evolve into respiratory failure. In the cases outlined
in the present report, infection was initially suspected based on
sputum culture and mNGS results of BALF, which revealed a number of
opportunistic pathogens. Following anti-infective treatment, the
clinical manifestations of the patients showed marked improvement
during the initial phase of treatment. No pathogens were found in
the subsequent sputum culture and mNGS of BALF. However, the
symptoms exhibited by the patients and the imaging findings
worsened afterwards, which prompted an alternative diagnosis.
Regardless of the ultimate etiology, prompt definitive diagnosis
and targeted therapeutic intervention are key in optimizing
clinical outcomes.
Histopathological workup is often required to
establish a correct diagnosis. In current clinical practice, the
diagnostic evaluation of pulmonary lesions primarily relies on
minimally invasive techniques, such as bronchoscopy and CT-guided
percutaneous lung biopsy, thereby markedly reducing the need for
more invasive surgical interventions (23-25).
However, it has been established that, although transbronchial lung
biopsy is conducted more frequently, the success rate lies at only
55.6%. Furthermore, although surgical lung biopsy and autopsy often
achieve an approximate diagnostic success rate of 100%, both are
associated with notably higher risks and cost, given that a lung
biopsy is often difficult to perform and requires patient
cooperation. In hematological patients, such as patients with
thrombocytopenia and coagulation disorders, these procedures may
notably increase the risk of complications associated with invasive
diagnostic interventions, and when sedation or general anesthesia
is required, patients are exposed to additional risks. The
literature demonstrates that a total of 27.8% of patients underwent
surgery or thoracoscopic lung biopsy. Therefore, non-invasive tools
to improve the differentiation of unclear pulmonary lesions are
desirable.
Regarding diagnostic choices for the included cases
from the literatures, the majority of patients underwent biopsy
procedures, including repeated transbronchial lung biopsy, surgical
lung biopsies and autopsies (Table
V). Immunohistochemical analysis of BALF was conducted in only
5 patients. Of these, 4 patients were directly diagnosed through
BALF immunohistochemistry, while the remaining patient still
required surgical biopsy for definitive diagnosis of SPL. This
corresponds with a diagnostic success rate of only 80% for BALF
immunohistochemistry. Biopsy is the well-established standard for
diagnosis, but if lymphoma cells can be identified through simple
flow cytometry-based immunohistochemistry of BALF, this would
potentially spare patients from the discomfort and risks associated
with invasive biopsies. For the 2 patients in the present report
who did not undergo biopsy, the lymphoma clinicians had initially
planned CT-guided lung biopsies. However, both patients exhibited
marked coagulation abnormalities and low oxygenation indices,
rendering the procedure too high-risk. Ultimately, the clinicians
decided against biopsy; therefore, to confirm the diagnosis,
alternative approaches had to be explored. Both of the cases
presented in the present study happened to produce large amounts of
daily secretion from the lungs, prompting the collection of BALF
for relevant diagnostic tests.
Flow cytometry analysis (Table SII) of the alveolar cell
composition in BALF, particularly the percentages of
CD4+ and CD8+ T cells, has long been applied
in the diagnosis of diseases, including hypersensitivity
pneumonitis, idiopathic non-specific interstitial pneumonia,
idiopathic pleuroparenchymal fibroelastosis and unclassifiable
idiopathic interstitial pneumonia (26). It may also predict the progression
and prognosis of fibrotic diseases such as interstitial lung
disease (27). Additionally,
literature has reported that flow cytometry-based analysis of
inflammatory cells in BALF can be indirectly used to assess the
degree of lung tissue damage and macrophage subtyping to evaluate
the pulmonary immune status (28).
The use of flow cytometry depends on the selection of antigens. In
the majority of lymphoproliferative disorders, lymphocyte subset
analysis of BALF demonstrates a T cell predominance (>90%) with
fewer B cell populations (<10%). Of note, lymphocytic alveolitis
is a characteristic feature of pulmonary lymphoma, which can be
examined by BALF cytological analysis (29). Particularly in pulmonary B cell
lymphomas, flow cytometry of BALF has revealed both a notable
elevation in B cell proportion (>10%) and immunophenotypic
evidence of clonal proliferation, which may contribute to a
diagnosis (30,31). Similar findings have been observed
in pulmonary T cell lymphomas (32).
The basic principle of flow cytometry has been
described for multiple purposes, including MRD detection, primarily
depending on the selection of antibodies labelled with
immunofluorescence (33). MRD
testing is a diagnostic tool for patients with hematological
diseases, mainly those with acute myeloid leukemia (AML). It refers
to the small number of residual leukemia cells in patients with
newly diagnosed or refractory/relapsed disease after achieving
complete hematological remission through treatment. This
encompasses the specific expertise in multiparameter flow cytometry
(MFC)-based MRD, molecular MRD, NGS and clinical considerations.
Methods for MRD detection using MFC include leukemia-associated
immunophenotype (LAIP) and ‘different from normal’ (D-F-N)
approaches. The former involves identifying the LAIP of the patient
at initial diagnosis and using it for subsequent MRD monitoring
during treatment. The latter is applicable to patients lacking an
initial LAIP and can also detect antigen shift occurring during
treatment. The combination of LAIP and D-F-N is suitable for both
patients without an initial LAIP and for detecting newly emerging
phenotypic abnormalities or antigen shift of existing LAIP
(34). With regard to antibody
selection, experts from diagnostic groups (34) recommend using MFC with ≥8-color
fluorescence labeling for MRD detection to improve specificity. The
antibody panels for MRD detection include: i) Core antibodies CD45,
CD117, CD34, CD13 and CD33; ii) other antibodies including CD4,
CD11b, CD14, CD64 and human leukocyte antigen-DR to assess MRD in
monocytic or myelomonocytic AML; iii) CD7, CD19 and CD56 to
evaluate cross-lineage antigen expression; and iv) CD133, CD38 and
CD123 to detect leukemia stem/progenitor cells. A number of
different marker panels have been used to assess MRD. To minimize
the number of panels employed, experts recommend (35) the design and validation of a single
common panel assay for all MRD studies. Each center should select
the appropriate antibody panel for MRD detection based on the
disease subtype. For the 2 cases outlined in the present report,
both patients underwent bone marrow flow immunophenotyping prior to
BALF MRD detection. Therefore, the BALF MRD detection in both cases
was performed using fluorochrome-conjugated antibodies specifically
tailored to their baseline B cell or T cell lineage.
Among the numerous cases of SPL diagnosed through
BALF reported in the literature, only 1 case was found to be
confirmed using flow cytometry of BALF, which showed λ-restricted B
cells with dim CD20 expression and co-expression of CD5 and CD23,
consistent with chronic lymphocytic leukemia. The majority of cases
are still diagnosed based on the fractionation results of
lymphocytes and neutrophils, cytological findings, combined with
clinical characteristics and other biopsy data. Therefore, to the
best of our knowledge, the present study was the first to report an
SPL diagnosis that relied on MRD testing in BALF. The premise of
this examination was that there must be sufficient living cells in
BALF for detection. Overall, the minimum number of cells needed for
accurate reporting of MRD is 500,000-1,000,000 excluding
CD45- cells and debris (35). These high numbers enable the
assessment of possible MRD <0.1%. Furthermore, except for the
definite MRD cell population identified during specimen collection,
other residual leukemia cell subsets must be confirmed by skilled
operators or personnel with professional knowledge of flow
cytometry. However, in the cases outlined in the preset report,
only 140,000 nucleated cells were found in Case 2 and even fewer in
Case 1, but a positive status was still exhibited based on the 0.1%
threshold.
To provide guidance for early detection and
diagnosis, the present report recommends the following clinical
strategies: i) In patients with a prior confirmed diagnosis of NHL,
when respiratory symptoms emerge, the possibility of secondary
pulmonary infection and SPL should both be considered in the
differential diagnosis; ii) when chest CT reveals pneumonia-like
changes, differentiating SPL from pneumonia is challenging,
therefore a comprehensive evaluation incorporating bronchoalveolar
lavage, serum testing and tissue biopsy is advised when necessary;
and iii) in certain cases, if pathological tissue biopsy cannot be
performed, MRD detection of BALF can serve as an alternative
diagnostic method.
The present study has certain limitations. First,
only 2 cases were reported in this study. From these, 1 patient did
not receive standardized treatment due to personal reasons, thereby
weakening the reliability and statistical power of the study
results. Second, the generalizability of the study conclusions is
limited as MRD detection in BALF requires collecting a sufficient
number of cells for staining, and most patients may not meet this
criterion. Third, this study excluded cases before the 21st
century, which may introduce temporal selection bias. However,
cases before the 21st century are relatively rare, which may
mitigate this type of bias.
Supplementary Material
Antibody list for flow
cytometry.a
List of kits used in the NGS
procedure.a
Acknowledgements
The authors would like to thank the Hematology and
Cytology Laboratory of Wuhan Union Hospital for performing all the
flow cytometry assays and Weiyuan Gene Technology Co., Ltd., for
performing all the NGS tests.
Funding
Funding: The present report was supported by The Project of
Department of Science and Technology of Hubei Province (grant no.
2024AFB690).
Availability of data and materials
The data generated in the present study may be found
in the National Center for Biotechnology Information sequence read
archive under accession number PRJNA1363855 or at the following
URL: https://www.ncbi.nlm.nih.gov/bioproject/PRJNA1363855.
Authors' contributions
YH and ZL contributed towards drafting and revising
the manuscript, as well as gathering the experimental data. LR and
XZ analyzed and interpreted the data. HL served a key role in
developing the conceptual framework and methodological approach of
the present report. YY was involved in study conceptualization and
oversight, taking lead responsibility for critically revising the
intellectual content of the manuscript. All authors read and
approved the final version of the manuscript. YH, ZL and YY confirm
the authenticity of all the raw data.
Ethics approval and consent to
participate
As a retrospective analysis, the present report
obtained an informed consent waiver with oversight and approval
from the Institutional Review Board at Union Hospital, Tongji
Medical College, Huazhong University of Science and Technology
(Wuhan, Hebei). No ethics approval number was obtained.
Patient consent for publication
Written informed consent was obtained from the
patients' spouses for publication of all images and clinical data
in the present case report.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Jaffe ES: Diagnosis and classification of
lymphoma: Impact of technical advances. Semin Hematol. 56:30–36.
2019.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Berkman N, Breuer R, Kramer MR and
Polliack A: Pulmonary involvement in lymphoma. Leuk Lymphoma.
20:229–237. 1996.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Zhang MC, Zhou M, Song Q, Wang S, Shi Q,
Wang L, Yan FH, Qu JM and Zhao WL: Clinical features and outcomes
of pulmonary lymphoma: A single center experience of 180 cases.
Lung Cancer. 132:39–44. 2019.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Kumar R and Ison MG: Opportunistic
infections in transplant patients. Infect Dis Clin North Am.
33:1143–1157. 2019.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Shao L, Jiang L, Wu S, Yu L, Wang L and
Huang X: Simultaneous occurrence of invasive pulmonary
aspergillosis and diffuse large B-cell lymphoma: Case report and
literature review. BMC Cancer. 20(15)2020.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Miyahara N, Eda R, Umemori Y, Murakami T,
Kunichika N, Makihata K, Aoe K, Murakami K, Takeyama H and Harada
M: Pulmonary lymphoma of large B-cell type mimicking Wegener's
granulomatosis. Intern Med. 40:786–790. 2001.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Wieker K, Röcken C, Koenigsmann M,
Roessner A and Franke A: Pulmonary low-grade MALT-lymphoma
associated with localized pulmonary amyloidosis. A case report.
Amyloid. 9:190–193. 2002.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Kusumoto S, Kobayashi Y, Tanimoto TE,
Hasegawa T, Yokota Y, Tanimoto K, Sekiguchi N, Narabayashi M,
Watanabe T, Matsuno Y and Tobinai K: T(11;18)-bearing pulmonary
mucosa-associated lymphoid tissue lymphoma responding to
cladribine. Int J Hematol. 80:70–74. 2004.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Fujisawa T, Suda T, Matsuura S, Enomoto N,
Takeshita K, Ohnishi K and Chida K: Peripheral T-cell lymphoma with
diffuse pulmonary infiltration and an increase in serum KL-6 level.
Respirology. 12:452–454. 2007.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Jankipersadsing V, Tauchi T, Ohyashiki K,
Tanaka Y, Setoguchi Y and Mukai K: Spontaneous regression of
pulmonary infiltration of adult T-cell leukemia/lymphoma. Int J
Hematol. 86(207)2007.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Hosseinnezhad A, Seguel JM and Villanueva
AG: Diffuse pulmonary infiltrates in an old man with chronic
lymphocytic leukemia. Clin Pract. 1(e41)2011.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Samiullah S, Bhurgri H, Tufail M, Samad F,
Patel S, Marium M, Pliner L, Brelvi Z and Wang W: Metastatic
gastric MALT lymphoma masquerading as pulmonary infiltrates, with a
dramatic response to chemotherapy. J Gastrointest Cancer. 45 (Suppl
1):S151–S154. 2014.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Sedgwick CL, Hall PE, Ratnarajah A,
Natkunarajah J and Hogh L: Lesson of the month 1: A rash decision.
Clin Med (Lond). 15:206–207. 2015.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Suzuki N, Hiraga J, Kato H, Takagi Y,
Ujihira N, Narita M and Kagami Y: EBV-positive diffuse large B-cell
Lymphoma as a secondary malignancy arising in a myelodysplastic
syndrome patient who was treated with azacitidine. Intern Med.
56:1711–1713. 2017.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Kajimoto Y, Terasaki Y, Terasaki M, Kunugi
S, Okabe Y, Wakita S, Inokuchi K and Shimizu A: T-cell lymphoma
with a granulomatous lesion of the lungs after autologous
hematopoietic stem cell transplantation for Epstein-Barr
virus-positive diffuse large B-cell lymphoma: A unique rare case of
metachronous B-cell and T-cell lymphoma. Diagn Pathol.
15(125)2020.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Yagyu K, Kobayashi M, Ueda T, Uenishi R,
Nakatsuji Y and Matsushita H: Malignant lymphoma mimics miliary
tuberculosis by diffuse micronodular radiographic findings. Respir
Med Case Rep. 31(101239)2020.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Miyaoka C, Saraya T, Honda K, Fujiwara M,
Ishii H and Takizawa H: Aggressive lung involvement in a patient
with T-acute lymphoblastic leukaemia/lymphoblastic lymphoma: A
tricky and rare case report. Respirol Case Rep.
8(e00614)2020.PubMed/NCBI View
Article : Google Scholar
|
|
18
|
Tanaka Y, Momose S, Takayanagi N,
Tabayashi T, Tokuhira M, Tamaru JI and Kizaki M: Rapid
deterioration of intravascular large B-cell lymphoma with mass
formation in the trigeminal nerve and multiple organ infiltration:
An autopsy case report. J Clin Exp Hematop. 62:41–45.
2022.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Kobe H, Saito K, Arita M and Ishida T:
Cryobiopsy for Pneumocystis jirovecii pneumonia secondary to
adult T-cell lymphoma/leukaemia. Respirol Case Rep.
10(e0893)2021.PubMed/NCBI View
Article : Google Scholar
|
|
20
|
Yamamoto G, Takamura K, Ishida Y, Sato Y,
Sinozaki A, Kikuchi H, Yamamoto M, Kobayashi H, Hirose N and
Kikuchi K: Use of bronchoalveolar lavage in diagnosing
angioimmunoblastic T-cell lymphoma: A case report. Respirol Case
Rep. 10(e0924)2022.PubMed/NCBI View
Article : Google Scholar
|
|
21
|
Tanaka Y, Mishina T, Miyoshi H, Ohshima K
and Nohgawa M: Intestinal T-cell lymphoma with lung and lymph node
involvement at relapse. J Med Cases. 13:15–20. 2022.PubMed/NCBI View
Article : Google Scholar
|
|
22
|
William J, Variakojis D, Yeldandi A and
Raparia K: Lymphoproliferative neoplasms of the lung: A review.
Arch Pathol Lab Med. 137:382–391. 2013.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Borie R, Wislez M, Antoine M,
Copie-Bergman C, Thieblemont C and Cadranel J: Pulmonary
mucosa-associated lymphoid tissue lymphoma revisited. Eur Respir J.
47:1244–1260. 2016.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Swerdlow SH, Campo E, Pileri SA, Harris
NL, Stein H, Siebert R, Advani R, Ghielmini M, Salles GA, Zelenetz
AD and Jaffe ES: The 2016 revision of the World Health Organization
classification of lymphoid neoplasms. Blood. 127:2375–2390.
2016.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Zhu M, Chang Y, Fan H, Shi J, Zhu B and
Mai X: Primary pulmonary intravascular large B-cell lymphoma
misdiagnosed as pneumonia: Four case reports and a literature
review. Oncol Lett. 25(234)2023.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Yamagata A, Arita M, Tachibana H, Tokioka
F, Sugimoto C, Sumikawa H, Tanaka T, Yasui H, Fujisawa T, Nakamura
Y, et al: Impact of bronchoalveolar lavage lymphocytosis on the
effects of anti-inflammatory therapy in idiopathic non-specific
interstitial pneumonia, idiopathic pleuroparenchymal
fibroelastosis, and unclassifiable idiopathic interstitial
pneumonia. Respir Res. 22(115)2021.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Simons IA, Boerrigter BG, Hovestadt MCM,
Mooij-Kalverda KA, Zhang S, Boers LS, van der Zee AHM, Nossent EJ
and Duitman JW: Alveolar cell composition in interstitial lung
disease and the development of a pulmonary progressive fibrosing
phenotype: A retrospective cohort study. Respir Res.
26(164)2025.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Kwiecień I, Rutkowska E, Raniszewska A,
Rzeszotarska A, Polubiec-Kownacka M, Domagała-Kulawik J, Korsak J
and Rzepecki P: Flow cytometric analysis of macrophages and
cytokines profile in the bronchoalveolar lavage fluid in patients
with lung cancer. Cancers (Basel). 15(5175)2023.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Drent M, Wagenaar SS, Mulder PH, van
Velzen-Blad H, Diamant M and van den Bosch JM: Bronchoalveolar
lavage fluid profiles in sarcoidosis, tuberculosis, and
non-Hodgkin's and Hodgkin's disease. An evaluation of differences.
Chest. 105:514–519. 1994.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Borie R, Wislez M, Antoine M, Fleury-Feith
J, Thabut G, Crestani B, Monnet I, Nunes H, Delfau-Larue MH and
Cadranel J: Clonality and phenotyping analysis of alveolar
lymphocytes is suggestive of pulmonary MALT lymphoma. Respir Med.
105:1231–1237. 2011.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Philippe B, Delfau-Larue MH, Epardeau B,
Autran B, Clauvel JP, Farcet JP and Couderc LJ: B-cell pulmonary
lymphoma: Gene rearrangement analysis of bronchoalveolar
lymphocytes by polymerase chain reaction. Chest. 115:1242–1247.
1999.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Hanaka M, Yatera K, Itoh C, Kawanami T,
Nakanishi T, Katsuragi T, Shimajiri S, Ishimoto H, Tsukada J and
Mukae H: Case of adult T-cell leukemia/lymphoma with rapid
progression of pulmonary areas of ground-glass attenuation and
multiple nodules. Respir Investig. 51:40–45. 2013.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Kalina T, Flores-Montero J, van der Velden
VH, Martin-Ayuso M, Böttcher S, Ritgen M, Almeida J, Lhermitte L,
Asnafi V, Mendonça A, et al: EuroFlow standardization of flow
cytometer instrument settings and immunophenotyping protocols.
Leukemia. 26:1986–2010. 2012.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Heuser M, Freeman SD, Ossenkoppele GJ,
Buccisano F, Hourigan CS, Ngai LL, Tettero JM, Bachas C, Baer C,
Béné MC, et al: 2021 Update on MRD in acute myeloid leukemia: A
consensus document from the European LeukemiaNet MRD working party.
Blood. 138:2753–2767. 2021.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Schuurhuis GJ, Heuser M, Freeman S, Béné
MC, Buccisano F, Cloos J, Grimwade D, Haferlach T, Hills RK,
Hourigan CS, et al: Minimal/measurable residual disease in AML: A
consensus document from the European LeukemiaNet MRD working party.
Blood. 131:1275–1291. 2018.PubMed/NCBI View Article : Google Scholar
|