Introduction
The human brain undergoes structural and functional
changes throughout the aging process. Among these changes, the
corpus callosum, the largest white matter structure in the brain,
plays a notable role in interhemispheric communication and exhibits
age-related structural and microstructural alterations (1-4).
In particular, the splenium of the corpus callosum is involved in
the integration of visual, auditory and somatosensory information,
and its morphological and signal intensity alterations have been
recognized as potential imaging biomarkers of brain aging (1).
Fluid-attenuated inversion recovery (FLAIR)
hyperintensity in the splenium has been observed in a variety of
neurological disorders, including ischemia, metabolic disorders and
demyelinating diseases (5,6). However, limited research has
investigated the presence and significance of splenial
hyperintensity in otherwise healthy individuals as part of the
normal aging process. Given the growing elderly population and the
increasing utilization of magnetic resonance (MR) imaging in
clinical practice, the understanding of the normal variations of
splenial FLAIR hyperintensity across different age groups is
essential for distinguishing physiological changes from
pathology.
Previous studies have suggested that splenial signal
abnormalities may be linked to changes noted in myelin density,
axonal integrity and vascular supply, which naturally evolve with
age (7-9).
In addition, radiotherapy and white matter disease have been
implicated as factors contributing to increased splenial
hyperintensity; however, their precise impact remains unclear
(10). The identification of the
threshold at which these changes become statistically significant
can provide a valuable reference for radiologists and clinicians
interpreting brain MR imaging scans.
The present study aimed to fill this gap by
systematically analysing splenial FLAIR hyperintensity in a large
cohort of patients, establishing age-related reference values and
assessing the influence of sex, radiation therapy and white matter
disease on these findings. By using a comprehensive statistical
approach, the present study aimed to refine the interpretation of
MR imaging findings and contribute to the standardization of
diagnostic criteria for aging-related changes in the splenium.
The subsequent sections detail the methodology used
in the current retrospective cohort study, present the results
derived from a thorough examination of cerebral MR imaging and
discuss the broader implications of these findings in clinical
practice. By elucidating the normal aging dynamics of the splenium,
the current research study sought to provide a foundation for more
accurate and reliable neuroradiological assessments, ultimately
improving patient care and diagnostic accuracy.
Patients and methods
Study design and population
The present study was approved by the Ethics
Committee of Dr Abdurrahman Yurtaslan Ankara Oncology Training and
Research Hospital (approval no. 2022-01/27) in accordance with
institutional guidelines and The Declaration of Helsinki. The
present study analysed cerebral MR imaging, which was performed
between January 2018 and December 2021. The most common clinical
indication for MR imaging referral was headache. The patients'
neurological history was evaluated from the information recorded in
the hospital and exclusion criteria were applied. Initially, 1,176
patients were screened and following application of strict
inclusion and exclusion criteria, 975 patients were included in the
final analysis (Fig. 1).
Inclusion and exclusion criteria
The inclusion criteria were as follows: Availability
of high-quality axial FLAIR MR imaging, adults aged ≥18 years with
no history of neurological disorders and no contraindications to MR
imaging. The exclusion criteria were as follows: Presence of
intracranial mass lesions or stroke that could alter the structure
and signal of the splenium, diagnosis of white matter diseases such
as multiple sclerosis or leukoencephalopathy, prior traumatic brain
injury with reported MR imaging abnormalities, use of medications
known to affect FLAIR signal intensity such as antiepileptics and
corticosteroids and severe metabolic disorders such as uncontrolled
diabetes and hepatic encephalopathy (11).
MR imaging protocol
All MR imaging was performed on a 1.5T MR imaging
scanner (SIGNA™ Experience; GE HealthCare) with the following
standardized FLAIR protocol: Repetition time, 9,000 msec; echo
time, 85 msec; inversion time, 2,500 msec; slice thickness, 5 mm
(no interslice gap); field of view, 240 mm.
Measurement of splenial FLAIR
hyperintensity
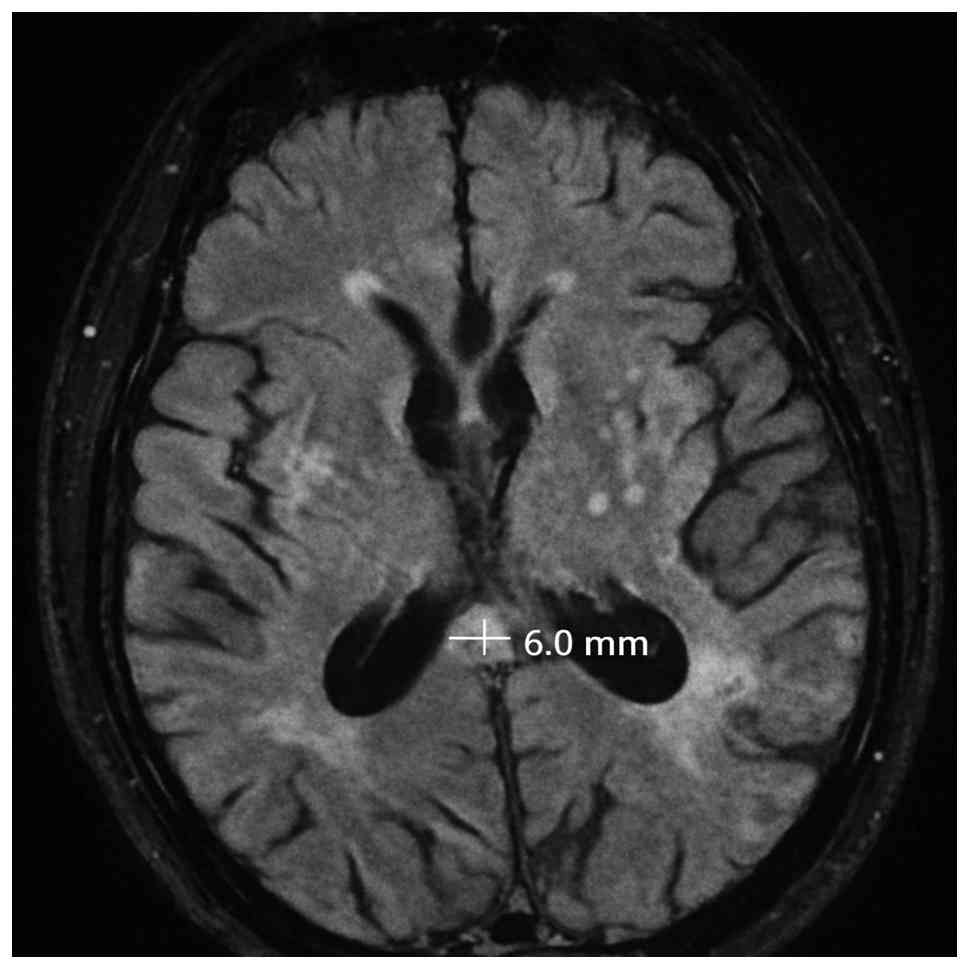
Splenial FLAIR hyperintensity thickness was measured
in the anteroposterior direction on the axial FLAIR slice where the
hyperintensity appeared most prominent, using the outer margins of
the hyperintense area as anatomical reference points; a sample case
is presented in Fig. 2. Two
independent observers (one with 2 years and another with 10 years
of neuroradiology experience) performed the measurements in a
blinded manner to minimize bias. The interobserver agreement was
assessed using the intraclass correlation coefficient (ICC).
Assessment of white matter
hyperintensities
White matter hyperintensities were evaluated using
the Fazekas scale, which classifies lesions into four categories:
0, absent; I, punctate foci; II, beginning confluence of foci; and
III, large confluent areas (12).
Statistical analysis
Statistical analysis was performed using IBM SPSS
Statistics for Windows (version 25.0; IBM Corp.). The
Kolmogorov-Smirnov test was applied to assess normality of
continuous variables. The Mann-Whitney U test was used to compare
sex differences in splenial hyperintensity thickness. The
Kruskal-Wallis test followed by Dunn's multiple comparison test
with Bonferroni correction was conducted for age group differences.
Spearman correlation test was used to determine relationships
between continuous variables. Receiver operating characteristic
(ROC) curve analysis and Youden index were utilized to define an
optimal age cut-off for hyperintensity presence [57 years; area
under the curve (AUC)=0.79; P<0.001]. Interobserver reliability
was assessed using the κ coefficient for categorical data and ICC
for continuous measurements (ICC=0.977; P<0.001).
Results
Study population and demographics
Following application of the inclusion and exclusion
criteria, 975 patients were included in the final analysis. The
present study population consisted of 653 females (67%) and 322
males (33%) with an age range of 18 to 91 years (mean, 49.47±16.30
years). There were no statistically significant differences in the
demographic distribution between sex (P=0.929).
Presence and thickness of splenial
hyperintensity
Splenium hyperintensity was detected in 165 (16.9%)
of the 975 patients. Patients with hyperintensity exhibited a
significantly higher mean age compared with those without
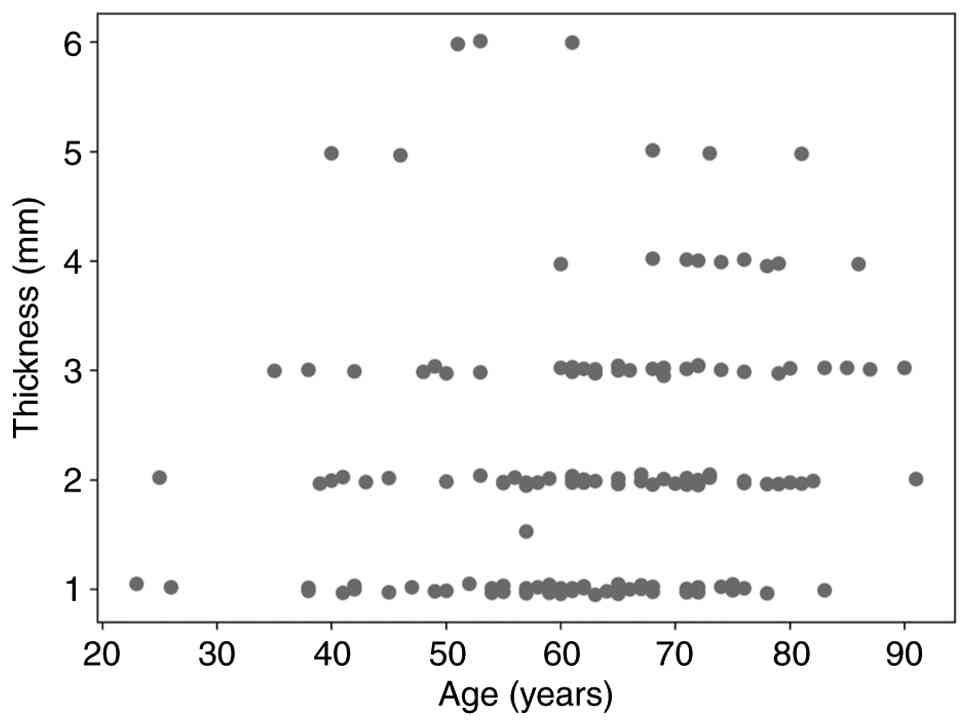
hyperintensity (Z=-11.855; P<0.001; Tables I and II). A statistically significant but weak
positive correlation was observed between age and splenial FLAIR
hyperintensity thickness (Spearman's r=0.214; P=0.006). This
relationship is visually demonstrated in the dot plot shown in
Fig. 3.
 | Table IPresence of splenium hyperintensity
according to age and sex. |
Table I
Presence of splenium hyperintensity
according to age and sex.
| Variable | Total | Yes | No | Test value | P-value |
|---|
| All | 975(100) | 165 (16.9) | 810 (83.1) | | |
| Age, years | 49.47±6.30 | 62.92±12.78 | 46.73±5.57 | -11.855a | <0.001 |
| Sex | | | | 0.008b | 0.929 |
|
Male | 322(33) | 54 (16.8) | 268 (83.2) | | |
|
Female | 653(67) | 111 (17.0) | 542 (83.0) | | |
 | Table IIThickness of splenium hyperintensity
according to patient characteristics. |
Table II
Thickness of splenium hyperintensity
according to patient characteristics.
| Variable | n | Thickness (mean ±
SD) | Test value | P-value |
|---|
| Age | 165 | | 0.214a | 0.006 |
| Sex | | | -0.579b | 0.562 |
|
Male | 54 | 2.24±1.30 | | |
|
Female | 111 | 2.06±1.10 | | |
| Radiation
therapy | | | -1.982b | 0.048 |
|
Yes | 46 | 2.50±1.45 | | |
|
No | 119 | 1.98±1.01 | | |
| Fazekas scale | | | 10.026c | 0.018 |
|
0 | 59 | 1.86±1.16 | | |
|
I | 63 | 2.12±1.07 | | |
|
II | 25 | 2.28±1.03 | | |
|
III | 18 | 2.78±1.48 | | |
Age threshold analysis
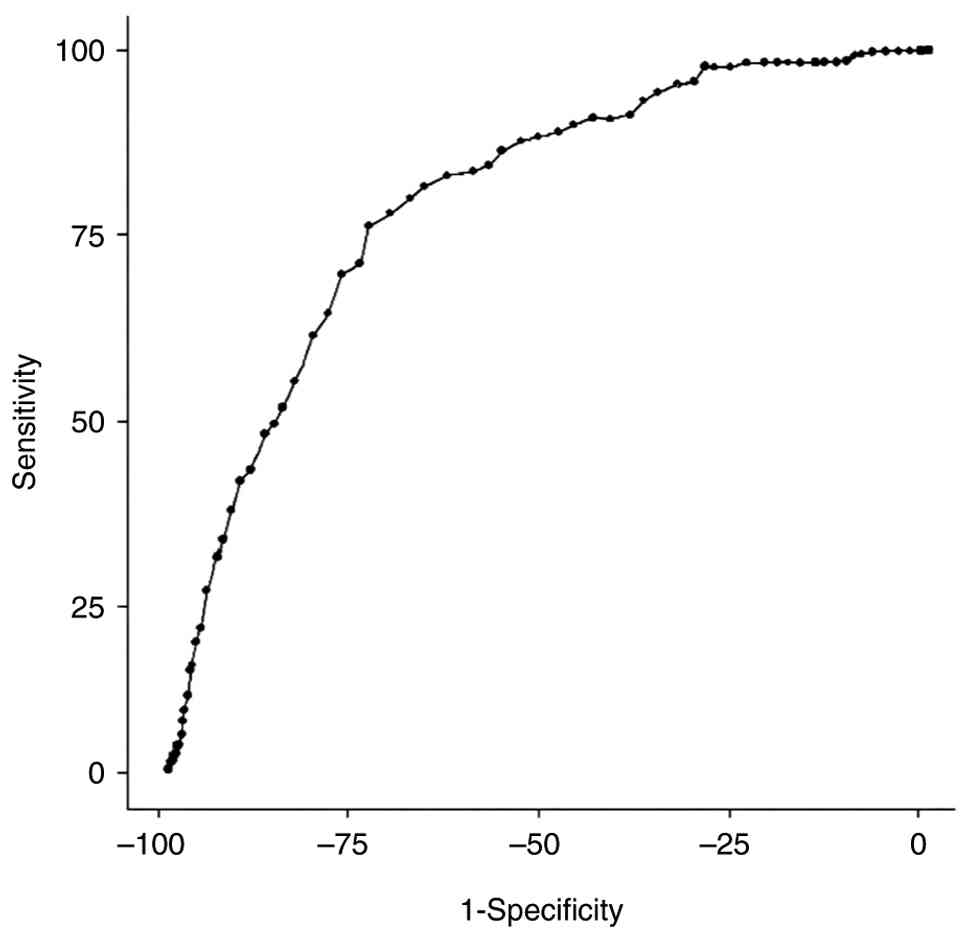
ROC analysis was performed to evaluate the potential
of age to predict the presence of splenial FLAIR hyperintensity.
The analysis identified an optimal age cut-off value of 57 years,
with an AUC of 0.79 (95% confidence interval: 0.76-0.83;
P<0.001). At this threshold, the sensitivity and specificity
were 76 and 73%, respectively (Fig.
4).
Effect of sex
No significant differences were noted between males
and females with regard of the splenium hyperintensity presence
(P=0.929) or thickness (P=0.562), indicating that sex does not
significantly influence these findings (Tables I and II).
Impact of radiation therapy and white
matter disease
The patients who had undergone cranial radiation
therapy exhibited significantly increased splenial hyperintensity
thickness compared with those without radiation therapy (RT)
history (Z=-1.982; P=0.048). In addition, a significant difference
was observed between the Fazekas score groups (χ²=10.026, P=0.018).
Post-hoc analysis revealed that patients with a Fazekas score of
III exhibited significantly higher splenial hyperintensity
thickness compared with those with a score of 0 (P<0.05).
Information regarding the radiation therapy
subgroup, including primary diagnoses, radiation dose parameters
and the time interval since treatment, was not consistently
available due to the retrospective design of the study; therefore,
the observed association between cranial radiation therapy and
increased splenial hyperintensity thickness should be interpreted
with caution.
Interobserver agreement
The interobserver agreement was found to be high for
the presence of splenial hyperintensity (κ=0.821; P<0.001) and
excellent for thickness measurements (ICC=0.977; P<0.001),
confirming the reliability of the measurement methodology used in
the present study.
Discussion
The findings of the present study provide compelling
evidence that splenial FLAIR hyperintensity is a common feature of
the aging brain, with a significant increase observed in
individuals >57 years. This threshold, determined via ROC
analysis, underscores the requirement for age-adjusted
interpretation of MR imaging findings to differentiate normal
age-related changes from pathological conditions.
There are certain studies in the literature
explaining the causes of splenium involvement, but the specific
cause of involvement of this region and pathophysiological changes
have not yet been explained. Cytotoxic oedema, focal inflammatory
changes, electrolyte changes in the cell membrane and focal
demyelination due to antiepileptic drugs are considered responsible
for the involvement of this area (3,11,13).
It has been postulated that isolated involvement may be due to the
histopathological difference between the other parts of the corpus
callosum or the difference in vascular circulation (3,11,13).
Studies have further highlighted the importance of age-adjusted
interpretation of white matter signal changes on MR imaging,
emphasizing that age-related FLAIR hyperintensities may overlap
with imaging features of subclinical pathology, notably in older
adults. The distinction of physiological aging from early or
subclinical disease therefore requires careful consideration of
patient age, lesion distribution and associated imaging findings,
underscoring the clinical value of age-specific reference
thresholds in routine neuroradiological practice.
Splenium abnormalities on MR imaging have been
reported commonly and unexpectedly in various cases. Various
studies have been conducted on this topic; however, the majority of
the studies are limited to case reports (7,9,14,15).
Age-related white matter alterations have been widely studied, with
previous literature suggesting a decline in myelin density and
axonal integrity contributing to increased signal intensities in
FLAIR imaging (1-3).
The significant association between splenial hyperintensity and
Fazekas grade III further reinforces the link between white matter
degeneration and hyperintensity formation in the corpus callosum.
These findings align with earlier reports that have associated deep
white matter disease with microvascular dysfunction and gliosis,
both of which may play a role in splenial hyperintensity
progression (6,7). The absence of significant sex
differences in the current findings suggests that these age-related
alterations are a universal aspect of human ageing rather than
being influenced by sex-specific factors. This universality
necessitates a nuanced approach to interpret MR imaging, where
age-related changes are considered alongside potential pathological
findings.
Due to the retrospective and cross-sectional design
of the present study, the observed association between age and
splenial FLAIR hyperintensity reflects differences across age
groups rather than longitudinal progression within the same
individuals; therefore, prospective longitudinal studies are
required to confirm the development and temporal progression of
these changes. Given the retrospective and cross-sectional design
of the present study, the observed association between age and
splenial FLAIR hyperintensity represents inter-individual
differences across distinct age groups rather than a true temporal
progression within the same individuals. Therefore, these findings
should not be interpreted as evidence of progressive development of
splenial hyperintensity over time. Confirmation of the natural
evolution and progression of these changes would require
well-designed longitudinal studies with follow-up imaging of the
same subjects.
In addition to age-related changes, the present
study highlights the influence of RT on splenial hyperintensity
thickness. Patients with a history of RT exhibited significantly
increased hyperintensity, suggesting a potential impact of
radiation-induced demyelination or gliosis in the corpus callosum.
This aligns with previous studies that have identified RT-related
white matter changes, notably in patients undergoing cranial
irradiation for malignancies (10). Future research with longitudinal
imaging follow-ups is warranted to delineate the temporal
progression of RT-associated splenial changes.
From a clinical perspective, differentiating between
normal ageing processes and pathological changes is paramount. The
increased splenial FLAIR hyperintensity observed in older
individuals may mimic or obscure pathological hyperintensities,
potentially leading to misdiagnosis or overdiagnosis of conditions
such as mild cognitive impairment or early dementia. Therefore, the
findings advocate for incorporating age-specific reference ranges
in assessing splenial FLAIR hyperintensity, diagnostic accuracy and
patient management.
One of the key strengths of the present study is the
high interobserver agreement, ensuring the reliability of the
splenial hyperintensity measurements. This consistency strengthens
the validity of the present findings and supports the potential
integration of splenial hyperintensity as a neuroimaging biomarker
for age-related white matter changes. However, several limitations
should be acknowledged in the present study. Firstly, the
retrospective nature of the present study limits the ability to
establish causality between aging and hyperintensity development.
Future prospective studies with longitudinal assessments could
provide a clearer picture of the progression of splenial changes
over time. Secondly, while patients with known white matter
diseases were excluded, subclinical neurodegenerative processes may
have introduced a confounding factor despite exclusion criteria.
Incorporating cognitive assessments and advanced neuroimaging
techniques, such as diffusion tensor imaging or MR spectroscopy
could enhance the current understanding of the underlying
mechanisms.
While headache was the primary indication for MR
imaging referral, rigorous exclusion criteria were used to minimize
confounding pathology and this limitation inherent to
retrospective, clinically referred cohorts has been considered when
interpreting the results. In addition, the retrospective design
limited the availability of detailed radiation therapy-related
clinical parameters, which may have influenced the interpretation
of its effect on splenial hyperintensity.
A further limitation is the use of 1.5T MR imaging
scanners, which, while widely used in clinical practice, may not
capture the full extent of subtle signal changes compared with
higher-resolution 3T imaging. Future studies using higher-field MR
scanners could improve sensitivity in detecting microstructural
alterations in the splenium. In addition, expanding the study
population to include diverse ethnic backgrounds and larger
datasets from multiple institutions would enhance the
generalizability of these findings.
Despite these limitations, the findings of the
present study provide critical insights into the normal aging
process of the splenium and emphasize the importance of
contextualizing hyperintensity findings in older adults. The
integration of these imaging biomarkers with volumetric analysis
and machine learning-based pattern recognition may aid the
refinement of the diagnostic criteria and the improvement of the
differentiation between physiological aging and neurodegenerative
disorders.
In conclusion, the present study demonstrated that
splenial FLAIR hyperintensity was a frequent finding in aging
individuals, with a marked increase observed beyond the age of 57.
These findings could assist geriatricians and radiologists in
collaborative decision-making, notably in distinguishing benign
imaging findings from early neurodegenerative conditions. Future
research integrating multimodal imaging and longitudinal
assessments will be crucial in further understanding the clinical
implications of these changes and their potential role in
neurodegenerative disease prediction.
The present study demonstrated that splenial FLAIR
hyperintensity was a common finding in aging, significantly
increasing following 57 years. These hyperintensities likely
reflect normal aging rather than pathology, necessitating an
age-adjusted approach to MR imaging interpretation. Given the aging
global population, recognizing normative imaging changes in older
adults is essential to avoid overdiagnosis of white matter
pathology and unnecessary interventions in geriatric care. The
association between splenial hyperintensity and Fazekas grade III
suggests an association with white matter changes and vascular
factors. In addition, radiation therapy history was associated with
increased hyperintensity thickness, highlighting the requirement to
consider patient history in MR imaging interpretation.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
The data generated in the present may be requested
from the corresponding author.
Authors' contributions
EYB and YÖ designed the study and analysed the data.
ÖÜ and OŞ analysed the data and prepared the manuscript. EYB and YÖ
confirm the authenticity of all the raw data. All authors read and
approved the final version of the manuscript.
Ethics approval and consent to
participate
The present retrospective study was approved by the
local Ethics Committee (approval no. 2022-01/27). According to
institutional and national regulations, the requirement for
informed consent was waived.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Blaauw J and Meiners LC: The splenium of
the corpus callosum: Embryology, anatomy, function and imaging with
pathophysiological hypothesis. Neuroradiology. 62:563–585.
2020.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Wang HX, Li YD, Liang J, Xue YZ, Zhu L,
Xiong TW, Chen PD, Kang X, Huang JP, Gong ZL and Sun HL:
Altitude-related features and prognosis in patients with reversible
splenial lesion syndrome. Ann Med. 56(2401107)2024.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Wilson CA, Mullen MT, Jackson BP, Ishida K
and Messé SR: Etiology of corpus callosum lesions with restricted
diffusion. Clin Neuroradiol. 27:31–37. 2017.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Georgy BA, Hesselink JR and Jernigan TL:
MR imaging of the corpus callosum. AJR Am J Roentgenol.
160:949–955. 1993.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Lee SK, Kim DI, Kim J, Kim DJ, Kim HD, Kim
DS and Mori S: Diffusion-tensor MR imaging and fiber tractography:
A new method of describing aberrant fiber connections in
developmental CNS anomalies. Radiographics. 25:53–65.
2005.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Barkovich AJ and Norman D: Anomalies of
the corpus callosum: Correlation with further anomalies of the
brain. AJR Am J Roentgenol. 151:171–179. 1988.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Lo L, Tan AC, Umapathi T and Lim CC:
Diffusion-weighted MR imaging in early diagnosis and prognosis of
hypoglycemia. AJNR Am J Neuroradiol. 27:1222–1224. 2006.PubMed/NCBI
|
|
8
|
Serdenes R, Orr S, Trio P, Chandrasekhara
S and Musselman M: A rare case report of a corpus callosal splenial
lesion in the context of atypical neuroleptic malignant syndrome. J
Investig Med High Impact Case Rep: Jan-Dec 9, 2021 (Epub ahead of
print).
|
|
9
|
Li S, Hu D, Li P, Xiao W, Li H, Liu G,
Song Y, Ning S, Peng Q, Zhao D, et al: Parameters indicating
development of influenza-associated acute necrotizing
encephalopathy: Experiences from a single center. Med Sci Monit.
27(e930688)2021.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Katsura M, Sato J, Akahane M, Furuta T,
Mori H and Abe O: Recognizing radiation-induced changes in the
central nervous system: Where to look and what to look for.
Radiographics. 41:224–248. 2021.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Park MK, Hwang SH, Jung S, Hong SS and
Kwon SB: Lesions in the splenium of the corpus callosum: Clinical
and radiological implications. Neurology Asia. 19:79–88. 2014.
|
|
12
|
Fazekas F, Chawluk JB, Alavi A, Hurtig HI
and Zimmerman RA: MR signal abnormalities at 1.5 T in Alzheimer's
dementia and normal aging. AJR Am J Roentgenol. 149:351–356.
1987.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Marsala SZ, Antichi E, Pistacchi M,
Gioulis M, Candeago RM, Montemurro RT, Gentile M, D'Andrea P and
Ferracci F: Mild encephalitis with a reversible splenial lesion: A
clinically benign condition, often underrecognized - Clinical case
and literature review. J Neurosci Rural Pract. 8:281–284.
2017.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Balcik ZE, Senadim S, Keskek A, Ozudogru
A, Koksal A, Soysal A and Atakli D: Does restricted diffusion in
the splenium indicate an acute infarct? Acta Neurol Belg.
120:1085–1089. 2020.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Han J, Wang Y, Wu Y, Zhang J, Song X and
Ji G: A case of reversible splenial lesion syndrome secondary to
Fanconi syndrome with white matter swelling as the main
manifestation. J Int Med Res: Jan 20, 2021 (Epub ahead of
print).
|