1. Introduction
Tumors cover both benign and malignant types and are
a serious concern. Various anticancer treatments, such as surgery,
chemoradiotherapy, immunotherapy, gene therapy, phototherapy and
targeted therapy, have been introduced to overcome this problem
(1). However, the adaptive
evolution of cancer cells and the complex construction of the tumor
microenvironment (TME) still largely constrain the further
enhancement of therapeutic effects (2). The TME is a complex ecosystem
composed of diverse cellular constituents, including immune and
stromal cells, alongside an acellular milieu of cytokines, growth
factors, hormones and the extracellular matrix (ECM) (3). Through intricate interactions with
tumor cells, these components collectively foster an environment
that promotes cancer cell survival, proliferation and invasion
(4). Additionally, the TME can
create a protective ecological niche for tumor cells, allowing them
to evade conventional treatment and leading to treatment failure
(5,6). Therefore, a comprehensive
understanding of the metabolic dynamics within the TME is of
paramount importance for the advancement of cancer therapy.
The concentration of lactic acid in the TME is one
of its key features and reflects the metabolic adaptive evolution
of tumor cells. Far from being a simple metabolic waste product, a
substantial body of research has revealed that lactic acid is a
multifaceted signaling molecule within the TME (7-9).
Its influence extends beyond regulating the metabolism and
proliferation of cancer cells to profoundly reshaping the tumor
immune microenvironment, and it is strongly associated with adverse
patient outcomes (10).
Numerous researchers and scholars have made
remarkable breakthroughs in tumor therapy by regulating lactate
metabolism within the TME (11,12).
In this article, the current research on the metabolic regulation
of lactate within the TME and the emergence of new therapies in
this field were reviewed. An in-depth discussion of these studies
and therapies was provided to comprehensively dissect the field and
provide useful guidance for the development of lactate metabolism
modulation approaches for tumor therapy.
2. Sources of lactate in the TME
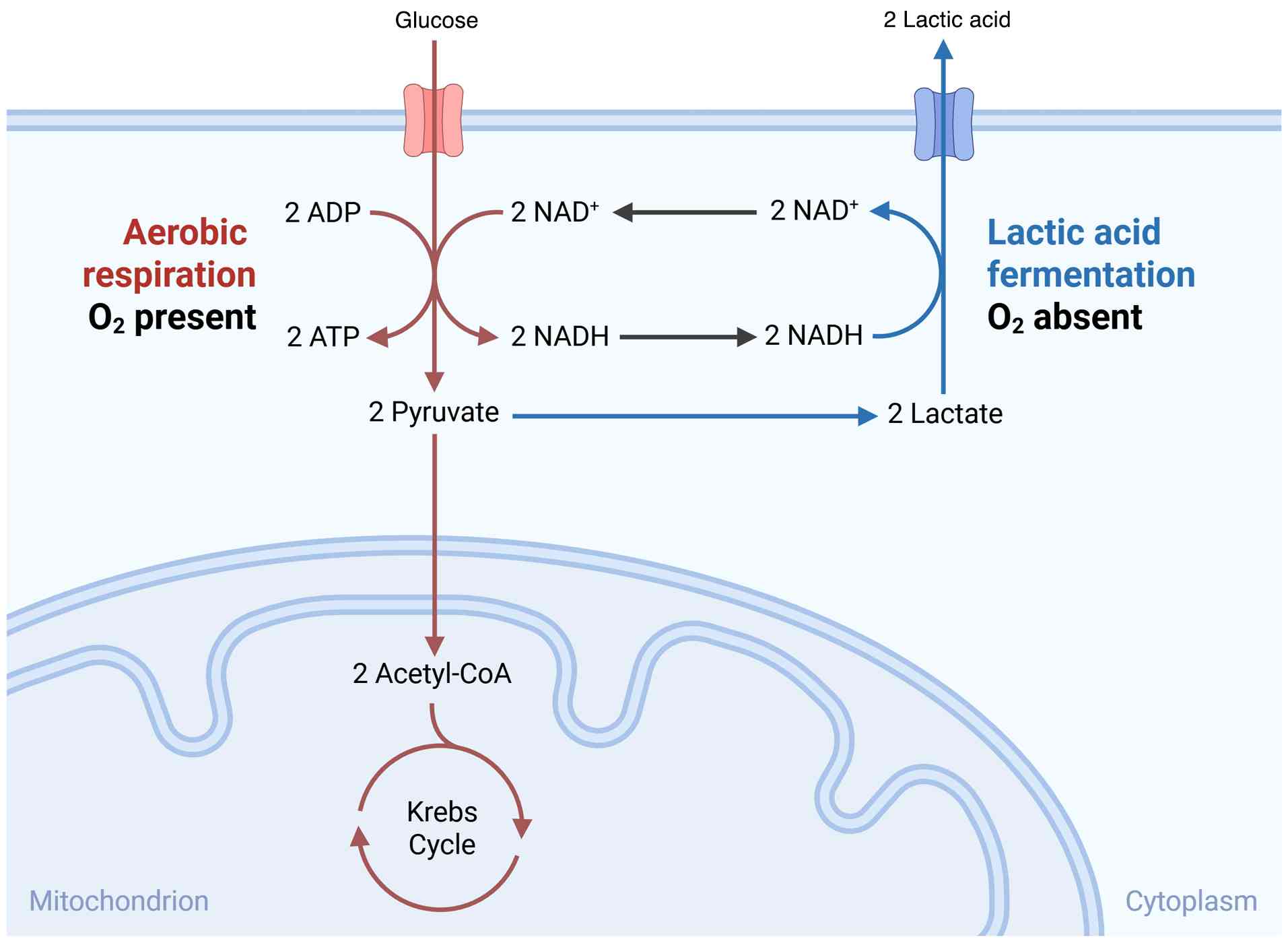
As early as the 1920s, Otto Warburg and his team
reported that cancer cells have special metabolic characteristics;
tumor cells still consume substantial quantities of glucose and
generate high levels of lactate under conditions of sufficient
oxygen (13), unlike normal cells,
tumor cells, instead of choosing the normal metabolic pathway, tend
to rely on the glycolytic transformation of glucose into lactate, a
phenomenon termed the ‘Warburg effect’ (14), which is a major source of lactic
acid production in the TME.
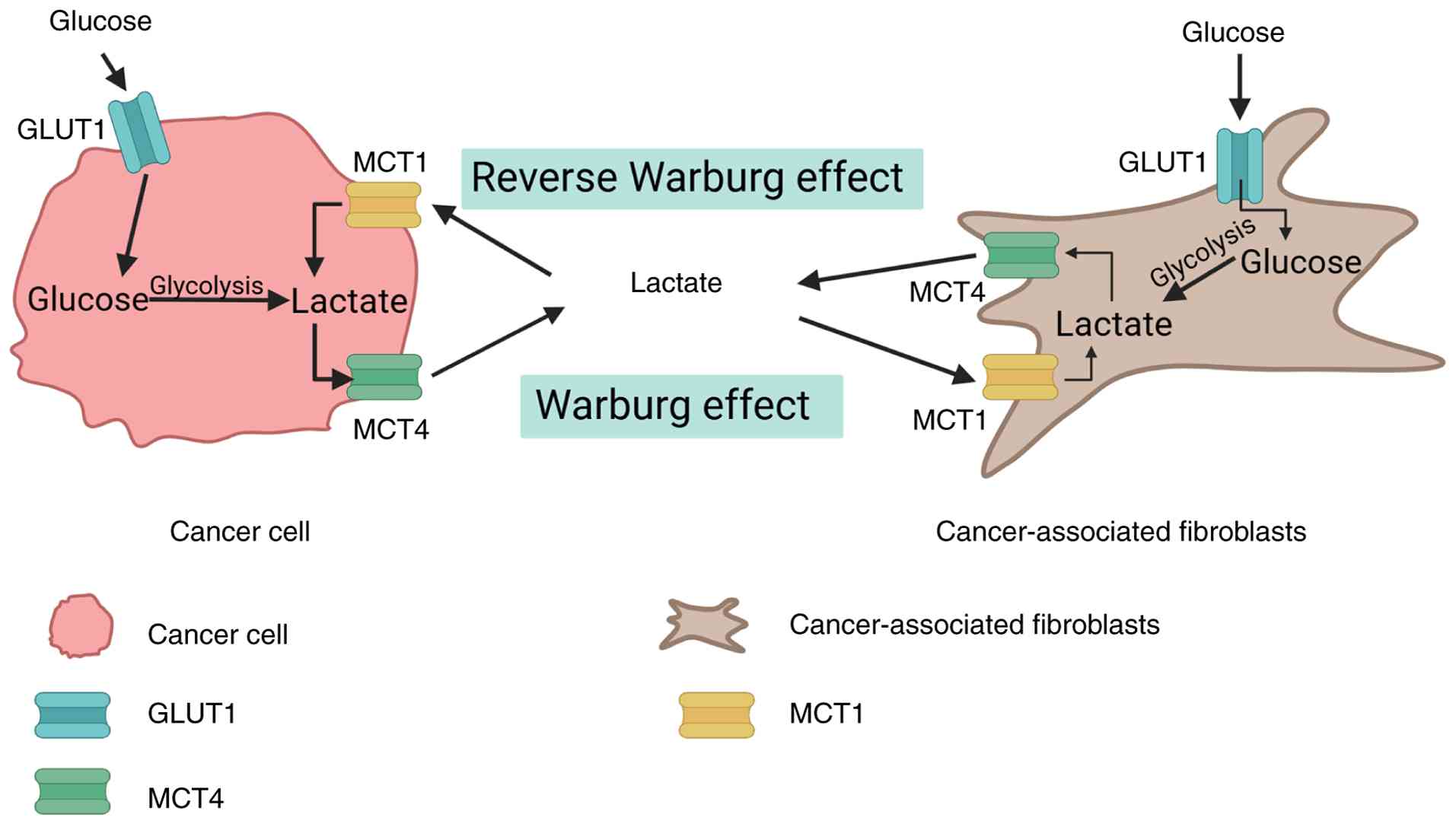
The TME is a multicellular system driven by
intricate tumor-stroma interactions, in which cancer-associated
fibroblasts (CAFs) are a major and stroma-rich cell type. Notably,
in contrast to neighboring cancer cells, CAFs in the tumor stroma
are more prone to activating glycolysis and autophagy, thereby
triggering the reverse Warburg effect (15-17).
Through the reverse Warburg effect, CAFs can deliver nutrients such
as lactate to cancer cells and the TME (Fig. 1). Additionally, in tumors, high
expression of the lactate dehydrogenase A (LDHA) isoform drives the
efficient transformation of pyruvate into lactic acid, a reaction
coupled with the regeneration of nicotinamide adenine dinucleotide
(oxidized form; NAD+) from NADH (reduced form), thereby leading to
substantial lactate accumulation (18,19).
In addition, cancer cells and CAFs express lactate
transporters such as monocarboxylate transporter proteins (MCTs),
which transport a vast amount of lactic acid produced
intracellularly into the microenvironment where it accumulates
within the TME (20).
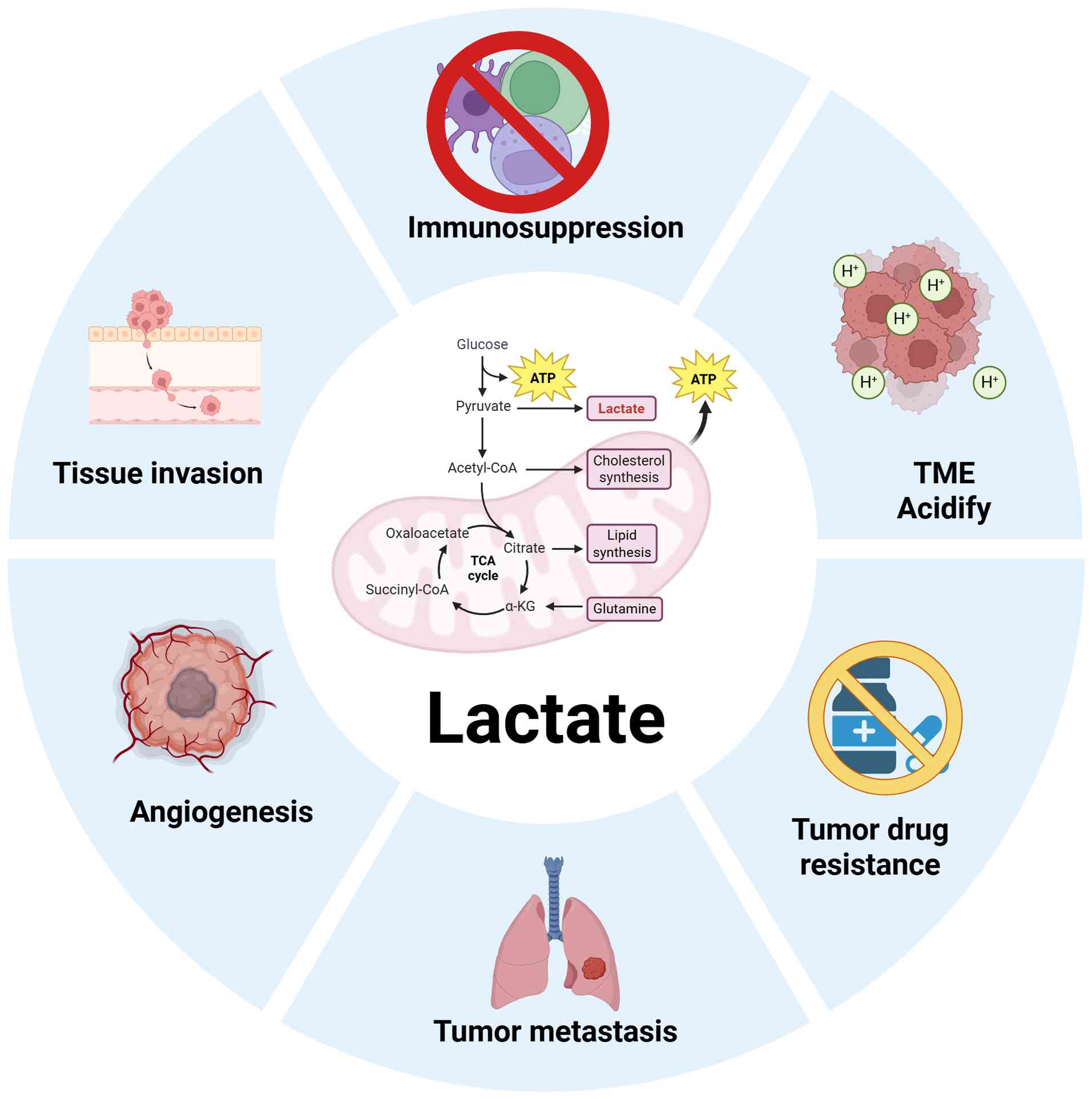
Together, these sources contribute to the creation
of a lactate-rich TME. The increase in lactate and acidification in
the TME have significant implications for tumor development,
including immune escape, tissue invasion, tumor metastasis,
angiogenesis and tumor resistance (21-23)
(Fig. 2).
3. Role of lactic acid in the TME
Lactate's influence extends past the tumor cells
themselves to actively remodel the stromal landscape of the TME
(24,25). It weakens the effector functions of
key immune cells (such as CD8+ T cells), governs the metabolism of
cancer-associated fibroblasts and fosters an intercellular
‘metabolic symbiosis’. Notably, the role of lactate is not limited
to promoting tumor development, but also appears to supply immune
cells with a carbon source, thus contributing to cancer immunity to
a certain extent, and this ‘dual’ role deserves to be explored in
depth (26). Within the TME,
lactate plays a dual role: It not only contributes to
microenvironmental acidification through lactic acidosis but also
functions as a crucial metabolic shuttle, moving between diverse
cell populations such as tumor cells, cancer-associated stromal
cells, tumor-associated macrophages (TAMs) and tumor-infiltrating
lymphocytes, thus creating immune crosstalk between different cell
populations (27) (Fig. 3). All in all, lactic acid is
important in the TME.
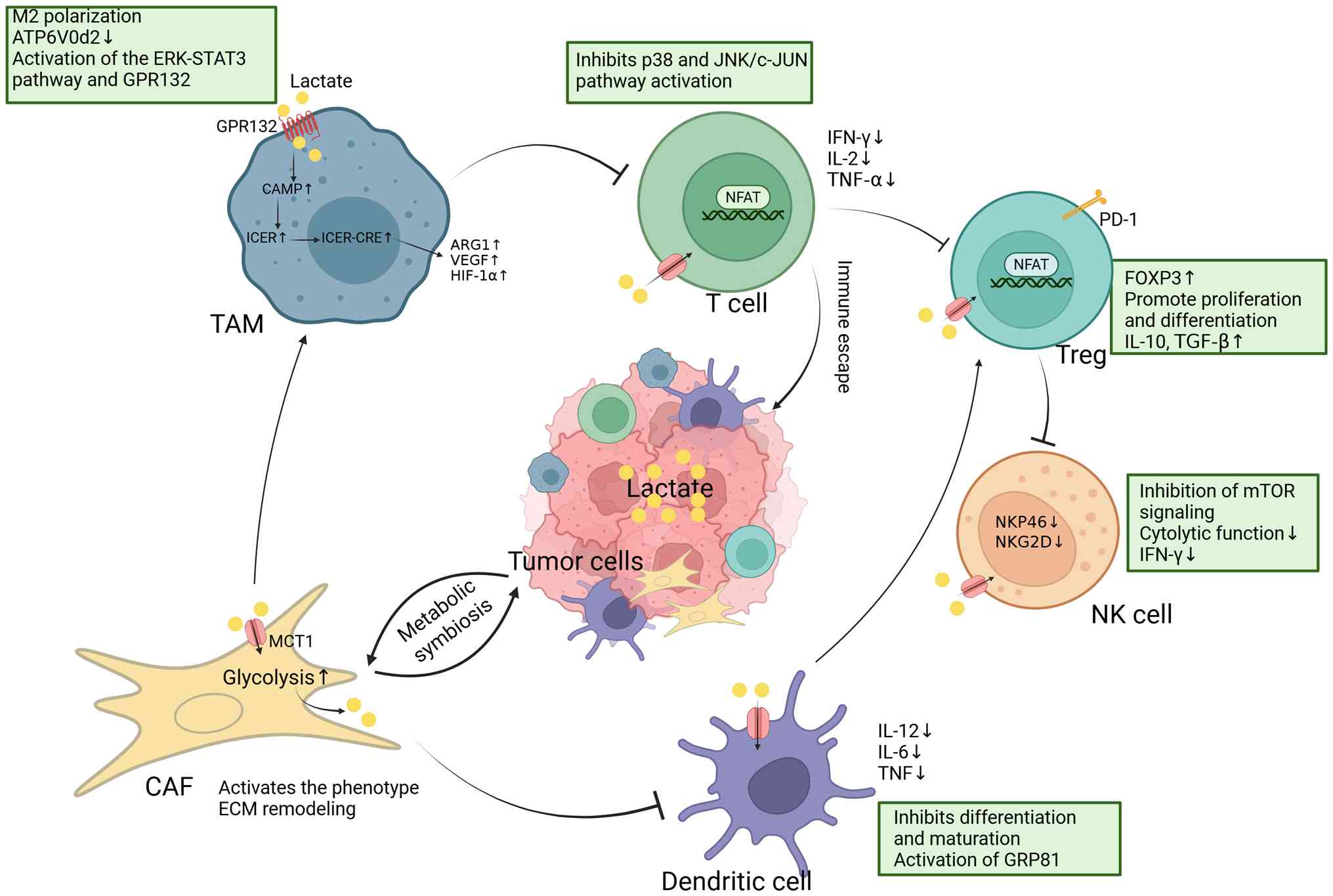 | Figure 3Crosstalk between lactic acid and
various immune cells. TAM, tumor-associated macrophage; T cell, T
lymphocyte; Treg, regulatory T cell; NK cell, natural killer cell;
CAF, cancer-associated fibroblast; DC, dendritic cell; ERK,
extracellular signal-regulated kinase; STAT, signal transducer and
activator of transcription; GRP, G protein-coupled receptor; CAMP,
cyclic adenosine monophosphate; ICER, inducible cAMP early
repressor; ARG, arginase; VEGF, vascular endothelial growth factor;
NFAT, nuclear factor of activated T cells; PD-1, programmed cell
death protein 1; mTOR, mechanistic target of rapamycin; NKP46,
natural cytotoxicity triggering receptor 1; NKG2D, natural killer
group 2, member D; p38, p38 mitogen-activated protein kinase; JNK,
c-Jun N-terminal kinase; c-Jun, Cellular Jun (proto-oncogene),
component of AP-1 transcription factor; HIF, hypoxia-inducible
factor. |
Lactate promotes malignant growth and
therapeutic resistance
When in an environment of hypoxia and a low energy
supply, cancer cells rely on lactate to maintain their
proliferative capacity. At different levels, lactic acid is a
metabolic fuel for tumor growth and acts as a signaling molecule.
Cancer cells maintain high glycolytic activity with the help of
monocarboxylic acid transport proteins (MCTs) and take up large
amounts of lactate from the surrounding environment (28). Adequate amounts of NADH produced by
oxidation can enter the mitochondria via the malate-aspartate
shuttle mechanism, which in turn fuels the respiratory chain. This
mechanism is beneficial to the autophagy process in cancer cells.
In humans, lactate predominantly exists as the L-isomer, which
stands in contrast to the far less common D-lactate. The D-isomer
originates from the metabolic activity of gut microorganisms and is
specifically catabolized by the mitochondrial enzyme D-lactate
dehydrogenase. The efficiency of this process is significantly more
pronounced in cancerous tissues compared to their healthy
counterparts, a phenomenon that consequently fuels malignant cell
proliferation. Within tumor cells, the pivotal lactate receptor G
protein-coupled receptor 81 (GPR81) orchestrates a state of
mitochondrial hyperactivity by modulating the expression of
peroxisome proliferator-activated receptor γ coactivator 1-α, CD147
and MCT, an effect that culminates in enhanced cancer cell
proliferation (29).
Recent investigations have revealed that lactate
accumulation is closely correlated with the insensitivity of
cancers to drugs (30). Lactic
acid can enhance the drug resistance of tumor cells (31). In cancer cells, the aberrant
activation of Pl3K stimulates its downstream effector, PKB/AKT,
which in turn drives a signaling cascade that ultimately enhances
glucose uptake (32). Inhibitors
that specifically target AKT, a key downstream node in the Pl3K
signaling pathway, are extensively employed in both preclinical
research and clinical trials (33). However, the accumulation of lactic
acid in cancer cells may lead to evasion of Pl3K blockade. Lactate
supplementation by itself has been reported to induce insensitivity
to Akt inhibitors within colon cancer cells, which can be reversed
when MCT-mediated transport is inhibited or eliminated by OXPHOS
treatment (34). Another study
confirmed that the expression of telomerase complex genes in breast
cancer cells is promoted when the lactate concentration is high. In
addition, cells that are chronically exposed to exogenous lactic
acid show the activation of EGFR, which correlates with the
emergence of acquired tamoxifen resistance. Additionally, the
literature indicates a direct link between lactic acid
acidification and tumor drug resistance. Recent investigations have
revealed that in lung cancer, the upregulation of aldo-keto
reductase drives the lactonization process of H4K12, which
ultimately triggers chemoresistance (35). In view of the continuing challenges
associated with drug resistance in tumors, the association between
lactic acid and drug insensitivity of tumor cells still needs to be
explored in depth.
Lactic acid activates the CAF
phenotype
CAFs regulate multiple functions by secreting
signaling molecules and inflammatory cytokines, altering their
intrinsic bioenergetics to ensure the provision of vital metabolic
substrates and factors that promote cell survival, and affecting
tumor epithelial cells by remodeling the ECM. The collaborative
effects of CAFs and epithelial cells drive malignant tumor
progression and may also adversely affect cancer therapy by
promoting tumor cell aggregation. In conclusion, activation of the
CAF phenotype facilitates tumor cell malignant growth and tissue
infiltration, which in turn endangers the health of the organism.
In addition, there is a metabolic symbiosis between tumor cells and
tumor stromal cells. An example is the reverse Warburg effect,
which refers to the ability of CAFs to undergo glycolysis under
aerobic conditions and supply energy to cancer cells by secreting
lactate (36). Lactic acid
secreted by CAFs has been reported to increase the invasive
potential of breast cancer cells through the stimulation of the
TGF-β1/p38MAPK/MMP2/9 signaling axis and by bolstering
mitochondrial bioenergetics (37).
Besides, lactic acid enrichment in the TME can activate fibroblasts
around tumors, which allows them to acquire a CAF phenotype
(24). Low NAD+ levels were found
to be a key factor in the inhibition of p62 and its CAF phenotypic
activation in fibroblasts by lactate (38). These findings further point to a
possible link among lactic acid metabolism, TME cells and
epigenetic reprogramming in tumors.
Regulation of immune cells by lactic
acid
Lactate can also influence other TME immune cells
via specific mechanisms, which in turn modulate the immune
response. These changes, in turn, contribute to a specific new
homeostatic scenario for tumor progression.
TAMs. TAMs found in TME participate in cancer
progression (39). Depending on
the microenvironmental signals they receive, macrophages can
differentiate into two main subtypes: Classically activated (M1) or
alternatively activated (M2), where the M2-type plays a pro-tumor
progression role. Lactic acid suppresses M1 polarization and
contributes to their differentiation toward an M2-like phenotype
(40). Lactic acid-induced
acidosis can inhibit M1 macrophage function by decreasing the
production of C-C motif chemokine ligand 2, inducible nitric oxide
synthase and IL-6. In addition, lactic acid can suppress the
production of ATPase H+ transporting V0 subunit d2 in
TAMs, which in turn promotes the progression of cancer.
Lactate-induced polarization of M2 macrophages is associated with
hypoxia-inducible factor (HIF)-1α stabilization, the signaling
pathway of ERK-STAT3 and GPR132 activation (27,41).
As a highly abundant receptor on the macrophage cell surface,
GPR132 is a key mediator in inducing a proinflammatory phenotype in
macrophages. Extracellular lactate sensing through GPR132 induces
cyclic AMP production, which in turn induces inducible cAMP early
repressor and ultimately upregulates the expression of numerous
cytokines, contributing to the change of macrophages to a
proangiogenic phenotype (42).
T cells and natural killer (NK) cells. In the
TME, lactate accumulation triggers tumor lactatemia, which in turn
inhibits cytotoxic T-lymphocyte function by suppressing the
signaling of p38 and JNK/c-Jun (43). It also interferes with the
signaling pathway of mTOR, resulting in impaired NK T-cell function
(44). Lactate-mediated
intracellular acidification hampers the activation of nuclear
factor of activated T cells (NFAT) in T cells and NK cells, thereby
inhibiting IFN-γ secretion and their function and survival, which
in turn weakens their immunosurveillance (12). The action of lactic acid on
cytotoxic cells is complex and cannot be unilaterally categorized
as a single effect. On one hand, the effects produced by lactic
acid on cells may vary with the concentration or pH. On the other
hand, lactate affects numerous aspects of the physiological
functions of cells, such as cellular respiration, metabolic
processes and immune functions.
Studies have shown that lactic acid is a crucial
carbon source in physiological activities and plays a vital role in
physiological processes such as energy metabolism. Some scholars
have noted that the suppression of CD8+ immune function is related
mainly to the acidic environment induced by lactic acid, but it can
also serve as an energy source for T-cell metabolism, which can
stimulate cellular vitality to a certain extent (45). Notably, the upregulation of LDHB
enhances cellular respiration and partially reverses the
suppression of T-cell function by lactate by counteracting with the
inhibitory effect of lactic acid on the expression of intracellular
cytokines (46). These
observations suggest that enhancing the lactate metabolism of
immune cells may promote immune-cell function to a certain
extent.
Dendritic cells (DCs). DCs are pivotal parts
in antigen presentation in antitumor immunity. In the TME, lactate
is a known regulator of interfering with the effective
antigen-presenting function of DCs by preventing lysosome formation
via a process reliant on Chop that regulates endoplasmic reticulum
stress (47). Tumor-secreted
lactate is a key regulator of DC phenotype in the TME (48). According to reports related to lung
cancer, lactic acid impacts DCs' role in orchestrating adaptive
immune responses, accelerating antigen breakdown and interfering
with cross-presentation (47). In
the case of breast cancer, lactic acid influences DC activity
through the activation of GPR81, which hinders the recognition of
tumor antigens by other immune cells. In addition, lactic acid can
restrict the process of IFN-α and IFN-γ induction in plasmacytoid
DCs, thus restricting the antitumor immune response (49).
T-regulatory (Treg) cells. Tregs promote
tumor immune tolerance by suppressing the growth and activation of
immune cells and secreting immunosuppressive cytokines. Notably,
Tregs utilize lactate to increase function and growth (50,51).
In a low-glycemic and lactic acid-rich environment, Tregs have an
advantage in metabolism and rely on the uptake and metabolism of
lactic acid to maintain their potent inhibitory functions (52-54).
At the mechanistic level, lactate may be able to regulate Treg cell
activity by mediating the lactation of MOESIN and enhancing TGF-β
signaling, thereby promoting Treg cell maturation and
differentiation (55). In
addition, in the glycolysis-dominant TME, lactate functions as a
key metabolic regulator, modulating the activity of Tregs by
enhancing programmed cell death (PD)-1 expression. The
monocarboxylate transporter 1 is instrumental in this process,
importing lactate into Tregs. This lactate influx then promotes the
nuclear accumulation of NFAT1, ultimately leading to increased
expression of the key immunosuppressive receptor PD-1(21). Some investigators have suggested
that the induction of Tregs in the TME may be enhanced via a
lactate-dependent pathway (56,57).
These findings imply that increased lactate from tumor glycolysis
may impact immune tolerance in the TME via Treg checkpoint
pathways.
Neutrophils. In a preclinical model of
sepsis, lactic acid reportedly contributes to the upregulation of
the PD-L1 immune checkpoint molecule on neutrophils via an
MCT1-dependent mechanism, giving rise to a decrease in apoptosis
and exacerbation of the morbidity of the disease (58). In mouse experiments, prolonged,
high-intensity exercise yielded decreased levels of lactate-driven
neutrophil extracellular traps (NETs). Conversely, lactate drives
NET release from human neutrophils through NADPH oxidase
(NOX)-mediated and NOX-independent NETotic processes (59).
Myeloid-derived suppressor cells (MDSCs). In
the TME, MDSCs drive immunosuppression effects, including T-cell
suppression and congenital immunomodulation. As early as 2013, it
was shown that lactate from tumors boosted the count of MDSCs,
which in turn inhibited NK cytotoxicity (60). Follow-up studies have confirmed
that lactic acid recruits MDSCs and regulates their development,
influencing the dynamic relationship between anti-tumor immunity
and malignant progression (61,62).
Another pancreatic cancer study indicated that lactic acid
activates MDSCs via the pathway of GPR81/mTOR/HIF-1α/STAT3 and
enhances their immunosuppression (63).
In conclusion, lactic acid is crucial for the TME
and is no longer considered ‘metabolic waste’. It regulates
cellular metabolism and immunity in the TME through various
signaling molecules and mechanisms, and these findings indicate a
promising avenue in the targeting of lactate metabolism in tumor
therapy.
4. Lactate metabolism combined with targeted
therapy
A wealth of research has revealed that lactate
occupies an indispensable position in the TME, and its metabolism
has been increasingly studied with the aim of enhancing the
therapeutic effects on tumors. The process of lactate metabolism
can be divided into three parts: Lactate anabolism (Fig. 4), lactate catabolism and lactate
transmembrane transport. By regulating the three phases of lactate
metabolism, researchers aim to find increasingly effective ways to
treat tumors.
Blocking lactate anabolism
Blocking lactate anabolism can decrease lactate
enrichment in the TME from the root and break down the metabolic
adaptations of tumors, thus curbing their survival and development
in a hostile environment. Oncogenic lesions in tumors increase the
levels and activity of enzymes involved in glycolysis, which drive
the metabolic shift to aerobic glycolysis and then generate lactic
acid. Malignant tumor cells often exhibit obvious aerobic
glycolysis, which leads to excessive dependence on glucose,
referred to as ‘glucose addiction’. To meet the demand for glucose
for rapid proliferation, tumor cells overexpress glucose
transporter protein (GLUT) to accelerate the transmembrane
transport of glucose (64). After
glucose is transported to cancer cells, it is transformed into
lactate through a cascade of enzymes. Therefore, it may be
hypothesized that directly blocking tumor cell glycolysis through
inhibition of glucose transport and inactivating enzymes of
glycolysis could effectively reduce lactate levels in the TME.
Currently, numerous drugs targeting GLUT and enzymes
of the glycolytic pathway, such as hexokinase 2 inhibitors
(3-bromopyruvate), LDHA suppressors (GNE-140), etc., are receiving
extensive attention (65-67).
However, these drugs face serious challenges in clinical
translation, such as high body-wide adverse effects and poor
therapeutic efficacy. The fundamental properties of nanoparticles,
such as tumor-targeting capacity, blood circulation steadiness and
ability to modulate enzyme activities, render them appropriate for
targeted suppression of tumor cell glycolysis. Researchers have
engineered various nanoplatforms to suppress lactic acid generation
in tumor cells via diverse intervention approaches, including
blockade of glucose uptake (68),
alleviation of tumor oxygen deprivation (69) and reduction in enzymatic activities
in the glycolytic pathway (70,71).
These nano-systems have been demonstrated to disrupt tumor cell
energy metabolism and remodel the TME, thus inhibiting tumor
expansion.
Promoting lactic acid catabolism
Owing to elevated glycolytic activity in tumor
cells, lactic acid levels in tumor tissues markedly increase over
normal tissue levels. The exocytosis of proton-coupled lactate from
tumor and stromal cells promotes the creation of an acidic
tumor-promoting milieu that facilitates tumor proliferation,
invasion, angiogenesis, metastasis, drug resistance and immune
escape. Therefore, enhancing lactate catabolism within tumors is an
attractive strategy to increase the efficacy of tumor therapy.
Currently, a range of micro/nano-systems have been engineered to
promote lactic acid catabolism, which could be categorized into
three main groups: Native bio-enzyme nano-systems, man-made
nano-enzymes and living bacteria. Lactate oxidase (LOX) and LDHB
are common natural enzymes involved in lactate catabolism (72,73).
Researchers have engineered a range of nano-enzymes with LOX- or
LDHB-like catalytic activities (74), for example, SnSe nanosheets,
NiO@Au nanocomposites and
Co4N/C (75). In
addition to nano-enzymes, lactic acid can be used as a metabolic
substrate by some microorganisms in nature, such as Acidobacter
oxidans (76), Snapper
(77), Fusobacterium
harzianum (78),
Anaerobacter spp. and Veillonella atypica (79). Compared with natural enzymes, live
bacteria have better in vivo stability and higher catalytic
efficiency than artificial nanozymes (80). Critically, some bacteria are
naturally tumorophilic and can actively penetrate into the tumor
and enrich the tumor site, thus enhancing the antitumor effect.
Blocking lactate transmembrane
transport
Abnormally high expression of MCT on tumor cell
membranes efficiently mediates lactate transmembrane transport and
enhances the interaction among cancer cells and the adjacent
microenvironment. In solid malignancies, internal hypoxic tumor
cells accelerate glycolysis to generate substantial lactate, which
is exported to the extracellular compartment through overexpressed
MCT4. By contrast, peripheral oxygen-enriched tumor cells highly
express MCT1, which takes up lactic acid and converts it to pyruvic
acid, which acts as an energy source for energy metabolism. This
metabolic heterogeneity triggered by differences in oxygen and
nutrient supplies ensures the fuel sustenance of neoplastic cells
from different ecological niches under unfavorable conditions and
facilitates metabolic symbiosis among tumor cells. Current studies
have confirmed that a number of tumor cells overexpress MCT. For
instance, in tumors such as glioblastoma and colon and breast
cancers, MCT1/MCT4 expression in terms of mRNA and protein are
significantly elevated, which contributes to driving tumor
progression, enhancing chemotherapy resistance and inducing
immunosuppression (81). From a
therapeutic perspective, blocking lactate transmembrane transport
to disrupt metabolic symbiosis stands out as a powerful and
innovative method for treating tumors. Specifically, the lactate
supply between cancer cells can be blocked by targeting
MCT4-mediated lactate outflow in hypoxic zones or MCT1-mediated
lactate inflow in aerobic tumor regions. This approach can disrupt
the competitive metabolic edge of tumoral cells in a harsh
environment and thereby suppress tumor expansion.
Over the past few years, various nanoparticle-based
formulations that target lactate transmembrane transport have been
published, and these drugs have exhibited significant antitumor
effects. For example, several small-molecule inhibitors of MCT4,
including lonidamine (LND), pyrazole derivatives and singolapine,
have been developed. To optimize the pharmacokinetic properties of
LND and facilitate its precise tumor targeting, Zhao et al
(82) developed a self-assembled
nanodrug system named TerBio, which is formed by Ce6, SB505124 and
LND. In addition, to enable regulated drug release, a
smart-responsive nanoplatform ‘HMON@HCPT-BSA-PEI-CDM-PEG’ was
constructed, which was able to provide good stability, safety and
targeting ability for therapeutic agents by progressively reacting
to the low-pH TME and glutathione-dependent intracellular redox
environments to achieve controlled drug release (83).
Although inhibiting MCT4 is a highly promising
antitumor strategy, the complex structural composition of solid
tumors makes it hard for nanotherapeutic agents to access the
target organ and release the drug. Therefore, well-designed
nano-system structures are still needed to increase the efficacy of
MCT4 inhibitors.
5. Conclusions and outlook
The current review presents the processes of lactic
acid production, transport and accumulation in cancerous tissues
and describes the effects of lactate on tumor progression through
multifaceted mechanisms. In addition, it provides insights into the
associations among lactic acid, drug insensitivity and the immune
response in tumors and analyzes the therapeutic potential of
lactate in clinical applications. With the continuous progress in
modern disciplines, the research and development of small-molecule
and biomacromolecule antineoplastic agents has entered a phase of
rapid development. To date, a variety of small molecule inhibitors
and gene drugs that target lactic acid metabolism in tumors have
arisen as potential tumor therapies, showing significant
advantages. However, these drugs generally have several
shortcomings, such as poor circulatory stability, poor
bioavailability, difficulty in effectively reaching the tumor site
and off-target effects, which severely restrict their clinical
translational applications. In recent years, nanoscience and
nanotechnology have made rapid progress and are revolutionizing the
delivery of antitumor drugs. Studies have shown that
nano-formulations can effectively optimize the pharmacokinetic
properties of lactic acid metabolism-targeted drugs, significantly
enhance the agent's pharmacokinetic profile, mitigate off-target
toxicity and enable a theranostic approach that combines both
therapy and diagnostics.
In conclusion, although the regulation of lactate
metabolism in antitumor therapy is challenging, various types of
nano-systems represent a promising class of materials that offer a
viable pathway to boost bioavailability and improve the safety
profile, thereby increasing their clinical viability. After a
comprehensive review and a deep dive into numerous recent advances
in this domain, the present review expects to establish a
foundation for understanding the future development of
high-performance nanomaterials that interfere with lactic acid
metabolism and develop more effective nano-formulations that
enhance cancer therapy. Furthermore, dissecting the role of lactate
metabolism in the immunoregulation of the TME may be helpful for
the clinical application of immunotherapy.
Acknowledgements
The figures were generated using BioRender
(https://biorender.com).
Funding
Funding: Financial support for the publication of this article
was provided by Zhongshan Municipal Science and Technology Bureau
(grant no. 2024B1042).
Availability of data and materials
Not applicable.
Authors' contributions
ML performed the literature search, selection and
collation of information, writing-original draft and writing-review
and editing. KS was responsible for supervision, conceptualization,
project administration and funding acquisition. All authors have
read and agreed to the published version of the manuscript. Data
authentication is not applicable.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Debela DT, Muzazu SG, Heraro KD, Ndalama
MT, Mesele BW, Haile DC, Kitui SK and Manyazewal T: New approaches
and procedures for cancer treatment: Current perspectives. SAGE
Open Med. 9(20503121211034366)2021.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Lankadasari MB, Mukhopadhyay P, Mohammed S
and Harikumar KB: TAMing pancreatic cancer: Combat with a double
edged sword. Mol Cancer. 18(48)2019.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Balkwill FR, Capasso M and Hagemann T: The
tumor microenvironment at a glance. J Cell Sci. 125:5591–5596.
2012.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Anderson NM and Simon MC: The tumor
microenvironment. Curr Biol. 30:R921–R925. 2020.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Zhong H, Zhou S, Yin S, Qiu Y, Liu B and
Yu H: Tumor microenvironment as niche constructed by cancer stem
cells: Breaking the ecosystem to combat cancer. J Adv Res.
71:279–296. 2025.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Cukierman E and Bassi DE: The mesenchymal
tumor microenvironment. Cell Adhesion Migration. 6:285–296.
2014.
|
|
7
|
Mbah NE and Lyssiotis CA: Metabolic
regulation of ferroptosis in the tumor microenvironment. J Biol
Chem. 298(101617)2022.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Colegio OR, Chu NQ, Szabo AL, Chu T,
Rhebergen AM, Jairam V, Cyrus N, Brokowski CE, Eisenbarth SC,
Phillips GM, et al: Functional polarization of tumour-associated
macrophages by tumour-derived lactic acid. Nature. 513:559–563.
2014.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Hirschhaeuser F, Sattler UGA and
Mueller-Klieser W: Lactate: A metabolic key player in cancer.
Cancer Res. 71:6921–6925. 2011.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Wang ZH, Peng WB, Zhang P, Yang XP and
Zhou Q: Lactate in the tumour microenvironment: From immune
modulation to therapy. EBioMedicine. 73(103627)2021.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Le A, Cooper CR, Gouw AM, Dinavahi R,
Maitra A, Deck LM, Royer RE, Vander Jagt DL, Semenza GL and Dang
CV: Inhibition of lactate dehydrogenase A induces oxidative stress
and inhibits tumor progression. Proc Natl Acad Sci USA.
107:2037–2042. 2010.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Brand A, Singer K, Koehl GE, Kolitzus M,
Schoenhammer G, Thiel A, Matos C, Bruss C, Klobuch S, Peter K, et
al: LDHA-associated lactic acid production blunts tumor
immunosurveillance by T and NK cells. Cell Metab. 24:657–671.
2016.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Warburg O: On the origin of cancer cells.
Science. 123:309–314. 1956.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Vander Heiden MG, Cantley LC and Thompson
CB: Understanding the Warburg effect: The metabolic requirements of
cell proliferation. Science. 324:1029–1033. 2009.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Pavlides S, Whitaker-Menezes D,
Castello-Cros R, Flomenberg N, Witkiewicz AK, Frank PG, Casimiro
MC, Wang C, Fortina P, Addya S, et al: The reverse Warburg effect:
Aerobic glycolysis in cancer associated fibroblasts and the tumor
stroma. Cell Cycle. 8:3984–4001. 2009.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Martinez-Outschoorn UE, Trimmer C, Lin Z,
Whitaker-Menezes D, Chiavarina B, Zhou J, Wang C, Pavlides S,
Martinez-Cantarin MP, Capozza F, et al: Autophagy in cancer
associated fibroblasts promotes tumor cell survival: Role of
hypoxia, HIF1 induction and NFκB activation in the tumor stromal
microenvironment. Cell Cycle. 9:3515–3533. 2010.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Ganguly D, Chandra R, Karalis J, Teke M,
Aguilera T, Maddipati R, Wachsmann MB, Ghersi D, Siravegna G, Zeh
HJ III, et al: Cancer-associated fibroblasts: Versatile players in
the tumor microenvironment. Cancers (Basel).
12(2652)2020.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Fantin VR, St-Pierre J and Leder P:
Attenuation of LDH-A expression uncovers a link between glycolysis,
mitochondrial physiology, and tumor maintenance. Cancer Cell.
9:425–434. 2006.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Li X, Yang Y, Zhang B, Lin X, Fu X, An Y,
Zou Y, Wang JX, Wang Z and Yu T: Lactate metabolism in human health
and disease. Signal Transduct Target Ther. 7(305)2022.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Pereira-Nunes A, Afonso J, Granja S and
Baltazar F: Lactate and lactate transporters as key players in the
maintenance of the Warburg effect. Adv Exp Med Biol. 1219:51–57.
2020.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Kumagai S, Koyama S, Itahashi K,
Tanegashima T, Lin YT, Togashi Y, Kamada T, Irie T, Okumura G, Kono
H, et al: Lactic acid promotes PD-1 expression in regulatory T
cells in highly glycolytic tumor microenvironments. Cancer Cell.
40:201–218.e9. 2022.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Tasdogan A, Faubert B, Ramesh V,
Ubellacker JM, Shen B, Solmonson A, Murphy MM, Gu Z, Gu W, Martin
M, et al: Metabolic heterogeneity confers differences in melanoma
metastatic potential. Nature. 577:115–120. 2020.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Corbet C and Feron O: Tumour acidosis:
From the passenger to the driver's seat. Nat Rev Cancer.
17:577–593. 2017.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Bhagat TD, Von Ahrens D, Dawlaty M, Zou Y,
Baddour J, Achreja A, Zhao H, Yang L, Patel B, Kwak C, et al:
Lactate-mediated epigenetic reprogramming regulates formation of
human pancreatic cancer-associated fibroblasts. Elife.
8(e50663)2019.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Ippolito L, Duatti A, Iozzo M, Comito G,
Pardella E, Lorito N, Bacci M, Pranzini E, Santi A, Sandrini G, et
al: Lactate supports cell-autonomous ECM production to sustain
metastatic behavior in prostate cancer. EMBO Rep. 25:3506–3531.
2024.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Chen S, Xu Y, Zhuo W and Zhang L: The
emerging role of lactate in tumor microenvironment and its clinical
relevance. Cancer Lett. 590(216837)2024.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Certo M, Tsai CH, Pucino V, Ho PC and
Mauro C: Lactate modulation of immune responses in inflammatory
versus tumour microenvironments. Nat Rev Immunol. 21:151–161.
2021.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Hui S, Ghergurovich JM, Morscher RJ, Jang
C, Teng X, Lu W, Esparza LA, Reya T, Le Zhan Yanxiang Guo J, et al:
Glucose feeds the TCA cycle via circulating lactate. Nature.
551:115–118. 2017.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Roland CL, Arumugam T, Deng D, Liu SH,
Philip B, Gomez S, Burns WR, Ramachandran V, Wang H,
Cruz-Monserrate Z, et al: Cell surface lactate receptor GPR81 is
crucial for cancer cell survival. Cancer Res. 74:5301–5310.
2014.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Feng X, Li D, Wang P, Li X and Li G:
Lactylation in cancer: Unlocking the key to drug resistance and
therapeutic breakthroughs. Oncol Res. 33:3327–3346. 2025.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Li W, Zhou C, Yu L, Hou Z, Liu H, Kong L,
Xu Y, He J, Lan J, Ou Q, et al: Tumor-derived lactate promotes
resistance to bevacizumab treatment by facilitating autophagy
enhancer protein RUBCNL expression through histone H3 lysine 18
lactylation (H3K18la) in colorectal cancer. Autophagy. 20:114–130.
2023.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Hoxhaj G and Manning BD: The PI3K-AKT
network at the interface of oncogenic signalling and cancer
metabolism. Nat Rev Cancer. 20:74–88. 2020.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Turner NC, Oliveira M, Howell SJ, Dalenc
F, Cortes J, Gomez Moreno HL, Hu X, Jhaveri K, Krivorotko P, Loibl
S, et al: Capivasertib in hormone receptor-positive advanced breast
cancer. N Engl J Med. 388:2058–2070. 2023.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Barnes EME, Xu Y, Benito A, Herendi L,
Siskos AP, Aboagye EO, Nijhuis A and Keun HC: Lactic acidosis
induces resistance to the pan-Akt inhibitor uprosertib in colon
cancer cells. Br J Cancer. 122:1298–1308. 2020.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Duan W, Liu W, Xia S, Zhou Y, Tang M, Xu
M, Lin M, Li X and Wang Q: Warburg effect enhanced by AKR1B10
promotes acquired resistance to pemetrexed in lung cancer-derived
brain metastasis. J Transl Med. 21(547)2023.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Kennedy KM, Scarbrough PM, Ribeiro A,
Richardson R, Yuan H, Sonveaux P, Landon CD, Chi JT, Pizzo S,
Schroeder T and Dewhirst MW: Catabolism of exogenous lactate
reveals it as a legitimate metabolic substrate in breast cancer.
PLoS One. 8(e75154)2013.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Sun K, Tang S, Hou Y, Xi L, Chen Y, Yin J,
Peng M, Zhao M, Cui X and Liu M: Oxidized ATM-mediated glycolysis
enhancement in breast cancer-associated fibroblasts contributes to
tumor invasion through lactate as metabolic coupling. EBioMedicine.
41:370–383. 2019.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Linares JF, Cid-Diaz T, Duran A, Osrodek
M, Martinez-Ordoñez A, Reina-Campos M, Kuo HH, Elemento O, Martin
ML, Cordes T, et al: The lactate-NAD+ axis activates
cancer-associated fibroblasts by downregulating p62. Cell Rep.
39(110792)2022.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Wang N, Liu W, Zheng Y, Wang S, Yang B, Li
M, Song J, Zhang F, Zhang X, Wang Q and Wang Z: CXCL1 derived from
tumor-associated macrophages promotes breast cancer metastasis via
activating NF-κB/SOX4 signaling. Cell Death Dis.
9(880)2018.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Zhao Y, Wang D, Xu T, Liu P, Cao Y, Wang
Y, Yang X, Xu X, Wang X and Niu H: Bladder cancer cells re-educate
TAMs through lactate shuttling in the microfluidic cancer
microenvironment. Oncotarget. 6:39196–39210. 2015.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Liu N, Luo J, Kuang D, Xu S, Duan Y, Xia
Y, Wei Z, Xie X, Yin B, Chen F, et al: Lactate inhibits ATP6V0d2
expression in tumor-associated macrophages to promote
HIF-2α-mediated tumor progression. J Clin Invest. 129:631–646.
2019.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Kes MMG, Van den Bossche J, Griffioen AW
and Huijbers EJM: Oncometabolites lactate and succinate drive
pro-angiogenic macrophage response in tumors. Biochim Biophys Acta
Rev Cancer. 1874(188427)2020.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Mendler AN, Hu B, Prinz PU, Kreutz M,
Gottfried E and Noessner E: Tumor lactic acidosis suppresses CTL
function by inhibition of p38 and JNK/c-Jun activation. Int J
Cancer. 131:633–640. 2012.PubMed/NCBI View Article : Google Scholar
|
|
44
|
Xie D, Zhu S and Bai L: Lactic acid in
tumor microenvironments causes dysfunction of NKT cells by
interfering with mTOR signaling. Sci China Life Sci. 59:1290–1296.
2016.PubMed/NCBI View Article : Google Scholar
|
|
45
|
Apostolova P and Pearce EL: Lactic acid
and lactate: Revisiting the physiological roles in the tumor
microenvironment. Trends Immunol. 43:969–977. 2022.PubMed/NCBI View Article : Google Scholar
|
|
46
|
Decking SM, Bruss C, Babl N, Bittner S,
Klobuch S, Thomas S, Feuerer M, Hoffmann P, Dettmer K, Oefner PJ,
et al: LDHB overexpression can partially overcome T cell inhibition
by lactic acid. Int J Mol Sci. 23(5970)2022.PubMed/NCBI View Article : Google Scholar
|
|
47
|
Caronni N, Simoncello F, Stafetta F,
Guarnaccia C, Ruiz-Moreno JS, Opitz B, Galli T, Proux-Gillardeaux V
and Benvenuti F: Downregulation of membrane trafficking proteins
and lactate conditioning determine loss of dendritic cell function
in lung cancer. Cancer Res. 78:1685–1699. 2018.PubMed/NCBI View Article : Google Scholar
|
|
48
|
Gottfried E, Kunz-Schughart LA, Ebner S,
Mueller-Klieser W, Hoves S, Andreesen R, Mackensen A and Kreutz M:
Tumor-derived lactic acid modulates dendritic cell activation and
antigen expression. Blood. 107:2013–2021. 2006.PubMed/NCBI View Article : Google Scholar
|
|
49
|
Raychaudhuri D, Bhattacharya R, Sinha BP,
Liu CSC, Ghosh AR, Rahaman O, Bandopadhyay P, Sarif J, D'Rozario R,
Paul S, et al: Lactate induces Pro-tumor reprogramming in
intratumoral plasmacytoid dendritic cells. Front Immunol.
10(1878)2019.PubMed/NCBI View Article : Google Scholar
|
|
50
|
Watson MJ, Vignali PDA, Mullett SJ,
Overacre-Delgoffe AE, Peralta RM, Grebinoski S, Menk AV,
Rittenhouse NL, DePeaux K, Whetstone RD, et al: Metabolic support
of tumour-infiltrating regulatory T cells by lactic acid. Nature.
591:645–651. 2021.PubMed/NCBI View Article : Google Scholar
|
|
51
|
Zhou J, Gu J, Qian Q, Zhang Y, Huang T, Li
X, Liu Z, Shao Q, Liang Y, Qiao L, et al: Lactate supports Treg
function and immune balance via MGAT1 effects on N-glycosylation in
the mitochondria. J Clin Invest. 134(e175897)2024.PubMed/NCBI View Article : Google Scholar
|
|
52
|
Angelin A, Gil-de-Gomez L, Dahiya S, Jiao
J, Guo L, Levine MH, Wang Z, Quinn WJ III, Kopinski PK, Wang L, et
al: Foxp3 reprograms T cell metabolism to function in low-glucose,
high-lactate environments. Cell Metab. 25:1282–1293.e7.
2017.PubMed/NCBI View Article : Google Scholar
|
|
53
|
Chattopadhyay A and Ferreira LMR: Why
regulatory T cells love lactic acid. Mol Ther Methods Clin Dev.
33(101647)2025.PubMed/NCBI View Article : Google Scholar
|
|
54
|
Gu XY, Yang JL, Lai R, Zhou ZJ, Tang D, Hu
L and Zhao LJ: Impact of lactate on immune cell function in the
tumor microenvironment: Mechanisms and therapeutic perspectives.
Front Immunol. 16(1563303)2025.PubMed/NCBI View Article : Google Scholar
|
|
55
|
Gu J, Zhou J, Chen Q, Xu X, Gao J, Li X,
Shao Q, Zhou B, Zhou H, Wei S, et al: Tumor metabolite lactate
promotes tumorigenesis by modulating MOESIN lactylation and
enhancing TGF-beta signaling in regulatory T cells. Cell Rep.
40(111122)2022.PubMed/NCBI View Article : Google Scholar
|
|
56
|
Tuomela K and Levings MK: Acidity promotes
the differentiation of immunosuppressive regulatory T cells. Eur J
Immunol. 53(e2350511)2023.PubMed/NCBI View Article : Google Scholar
|
|
57
|
Rao D, Stunnenberg JA, Lacroix R,
Dimitriadis P, Kaplon J, Verburg F, van van Royen PT, Hoefsmit EP,
Renner K, Blank CU and Peeper DS: Acidity-mediated induction of
FoxP3+ regulatory T cells. Eur J Immunol.
53(e2250258)2023.PubMed/NCBI View Article : Google Scholar
|
|
58
|
Wagner K, Sami MA, Norton C, McCoy J and
Hassan U: Profiling single-cell level phagocytic activity
distribution with blood lactate levels. RSC Adv. 11:21315–21322.
2021.PubMed/NCBI View Article : Google Scholar
|
|
59
|
Fei M, Zhang H, Meng F, An G, Tang J, Tong
J, Xiong L, Liu Q and Li C: Enhanced lactate accumulation
upregulates PD-L1 expression to delay neutrophil apoptosis in
sepsis. View: 5, 2023 doi: 10.1002/VIW.20230053.
|
|
60
|
Husain Z, Huang Y, Seth P and Sukhatme VP:
Tumor-derived lactate modifies antitumor immune response: Effect on
myeloid-derived suppressor cells and NK cells. J Immunol.
191:1486–1495. 2013.PubMed/NCBI View Article : Google Scholar
|
|
61
|
Li W, Tanikawa T, Kryczek I, Xia H, Li G,
Wu K, Wei S, Zhao L, Vatan L, Wen B, et al: Aerobic glycolysis
controls myeloid-derived suppressor cells and tumor immunity via a
specific CEBPB isoform in triple-negative breast cancer. Cell
Metab. 28:87–103.e6. 2018.PubMed/NCBI View Article : Google Scholar
|
|
62
|
Zhao JL, Ye YC, Gao CC, Wang L, Ren KX,
Jiang R, Hu SJ, Liang SQ, Bai J, Liang JL, et al: Notch-mediated
lactate metabolism regulates MDSC development through the
Hes1/MCT2/c-Jun axis. Cell Rep. 38(110451)2022.PubMed/NCBI View Article : Google Scholar
|
|
63
|
Yang X, Lu Y, Hang J, Zhang J, Zhang T,
Huo Y, Liu J, Lai S, Luo D, Wang L, et al: Lactate-modulated
immunosuppression of myeloid-derived suppressor cells contributes
to the radioresistance of pancreatic cancer. Cancer Immunol Res.
8:1440–1451. 2020.PubMed/NCBI View Article : Google Scholar
|
|
64
|
Hsu PP and Sabatini DM: Cancer cell
metabolism: Warburg and beyond. Cell. 134:703–707. 2008.PubMed/NCBI View Article : Google Scholar
|
|
65
|
Martinez-Outschoorn UE, Peiris-Pages M,
Pestell RG, Sotgia F and Lisanti MP: Cancer metabolism: A
therapeutic perspective. Nat Rev Clin Oncol. 14(113)2017.PubMed/NCBI View Article : Google Scholar
|
|
66
|
Pelicano H, Martin DS, Xu RH and Huang P:
Glycolysis inhibition for anticancer treatment. Oncogene.
25:4633–4646. 2006.PubMed/NCBI View Article : Google Scholar
|
|
67
|
Luengo A, Gui DY and Vander Heiden MG:
Targeting metabolism for cancer therapy. Cell Chem Biol.
24:1161–1180. 2017.PubMed/NCBI View Article : Google Scholar
|
|
68
|
Ganapathy-Kanniappan S and Geschwind JF:
Tumor glycolysis as a target for cancer therapy: Progress and
prospects. Mol Cancer. 12(152)2013.PubMed/NCBI View Article : Google Scholar
|
|
69
|
Lee SY, Park JH, Ko SH, Shim JS, Kim DD
and Cho HJ: Mussel-inspired hyaluronic acid derivative
nanostructures for improved tumor targeting and penetration. ACS
Appl Mater Interfaces. 9:22308–22320. 2017.PubMed/NCBI View Article : Google Scholar
|
|
70
|
Dai Z, Wang Q, Tang J, Qu R, Wu M, Li H,
Yang Y, Zhen X and Yu C: A Sub-6 nm
MnFe2O4-dichloroacetic acid nanocomposite
modulates tumor metabolism and catabolism for reversing tumor
immunosuppressive microenvironment and boosting immunotherapy.
Biomaterials. 284(121533)2022.PubMed/NCBI View Article : Google Scholar
|
|
71
|
Huang J, Zhuang C, Chen J, Chen X, Li X,
Zhang T, Wang B, Feng Q, Zheng X, Gong M, et al: Targeted
drug/Gene/Photodynamic therapy via a stimuli-responsive
dendritic-polymer-based nanococktail for treatment of
EGFR-TKI-resistant non-small-cell lung cancer. Adv Mater.
34(e2201516)2022.PubMed/NCBI View Article : Google Scholar
|
|
72
|
Tian Z, Yang K, Yao T, Li X, Ma Y, Qu C,
Qu X, Xu Y, Guo Y and Qu Y: Catalytically selective chemotherapy
from tumor-metabolic generated lactic acid. Small.
15(e1903746)2019.PubMed/NCBI View Article : Google Scholar
|
|
73
|
Tseng SJ, Kempson IM, Huang KY, Li HJ, Fa
YC, Ho YC, Liao ZX and Yang PC: Targeting tumor microenvironment by
bioreduction-activated nanoparticles for light-triggered
virotherapy. ACS Nano. 12:9894–9902. 2018.PubMed/NCBI View Article : Google Scholar
|
|
74
|
Jiang D, Ni D, Rosenkrans ZT, Huang P, Yan
X and Cai W: Nanozyme: New horizons for responsive biomedical
applications. Chem Soc Rev. 48:3683–3704. 2019.PubMed/NCBI View Article : Google Scholar
|
|
75
|
Gao M, Wang Z, Zheng H, Wang L, Xu S, Liu
X, Li W, Pan Y, Wang W, Cai X, et al: Two-dimensional tin selenide
(SnSe) nanosheets capable of mimicking key dehydrogenases in
cellular metabolism. Angew Chem Int Ed Engl. 59:3618–3623.
2020.PubMed/NCBI View Article : Google Scholar
|
|
76
|
Dos Reis SA, da Conceicao LL, Siqueira NP,
Rosa DD, da Silva LL and Peluzio MD: Review of the mechanisms of
probiotic actions in the prevention of colorectal cancer. Nutr Res.
37:1–19. 2017.PubMed/NCBI View Article : Google Scholar
|
|
77
|
Sheng B, Xu J, Zhang Y, Jiang T, Deng S,
Kong J, Gao C, Ma C and Xu P: Utilization of D-Lactate as an Energy
Source Supports the Growth of Gluconobacter oxydans. Appl Environ
Microbiol. 81:4098–4110. 2015.PubMed/NCBI View Article : Google Scholar
|
|
78
|
Wang JW, Chen QW, Luo GF, Han ZY, Song WF,
Yang J, Chen WH and Zhang XZ: A Self-driven bioreactor based on
bacterium-metal-organic framework biohybrids for boosting
chemotherapy via cyclic lactate catabolism. ACS Nano.
15:17870–17884. 2021.PubMed/NCBI View Article : Google Scholar
|
|
79
|
Vacca M, Celano G, Calabrese FM,
Portincasa P, Gobbetti M and De Angelis M: The controversial role
of human gut lachnospiraceae. Microorganisms. 8(573)2020.PubMed/NCBI View Article : Google Scholar
|
|
80
|
Fernandez-Sanjurjo M, Fernández J,
Tomás-Zapico C, Fernández-García B, Villar CJ, Lombó F and
Iglesias-Gutiérrez E: Is physical performance (in mice) increased
by Veillonella atypica or decreased by Lactobacillus
bulgaricus? J Sport Health Sci. 9:197–200. 2020.PubMed/NCBI View Article : Google Scholar
|
|
81
|
Ullah MS, Davies AJ and Halestrap AP: The
plasma membrane lactate transporter MCT4, but not MCT1, is
up-regulated by hypoxia through a HIF-1α-dependent mechanism. J
Biol Chem. 281:9030–9037. 2006.PubMed/NCBI View Article : Google Scholar
|
|
82
|
Zhao LP, Zheng RR, Kong RJ, Huang CY, Rao
XN, Yang N, Chen AL, Yu XY, Cheng H and Li SY: Self-delivery
ternary bioregulators for photodynamic amplified immunotherapy by
tumor microenvironment reprogramming. ACS Nano. 16:1182–1197.
2022.PubMed/NCBI View Article : Google Scholar
|
|
83
|
Li K, Lin C, He Y, Lu L, Xu K, Tao B, Xia
Z, Zeng R, Mao Y, Luo Z and Cai K: Engineering of
Cascade-responsive nanoplatform to inhibit lactate efflux for
enhanced tumor chemo-immunotherapy. ACS Nano. 14:14164–14180.
2020.PubMed/NCBI View Article : Google Scholar
|