Introduction
Inflammatory lung injury is a notable cause of acute
respiratory disease and mortality commonly induced by bacterial
pathogens [such as Streptococcus pneumoniae (SP) and
Staphylococcus aureus], viral pathogens (such as SARS-CoV-2
and influenza) or mixed infections (1,2). SP
remains the leading bacterial pathogen in community-acquired
pneumonia (CAP). CAP is frequently associated with severe
complications, such as parapneumonic effusion, empyema, necrotizing
pneumonia and acute respiratory distress syndrome (ARDS) (3,4). In
veterinary practice, SP is increasingly recognized as a pathogen
contributing to feline lower respiratory tract infections (5). Despite the effectiveness of
antibiotics in pathogen clearance, residual lung injury and
secondary inflammation often persist post-infection, as exemplified
by post-COVID-19 pulmonary syndromes (2,6). The
sequelae as a result of bacterial pneumonia are characterized by
endothelial and epithelial cell death, junctional disruption and
leukocyte infiltration, which ultimately impair nutrient and gas
exchange (7-10).
These outcomes highlight a notable gap in the understanding of host
recovery mechanisms beyond microbial eradication.
Although extensive studies have elucidated the
pathogenesis of pneumonia and antimicrobial strategies, the
resolution phase, particularly the mechanisms of host cellular
recovery following infection, has received less attention. Alveolar
epithelial regeneration, macrophage turnover and metabolic
reprogramming in tissue repair are essential in ARDS,
ventilator-induced lung injury and post-viral pneumonia (6,11,12).
However, to the best of our knowledge, specific investigations upon
SP-induced epithelial or mesenchymal injury and the ensuing
host-driven reparative responses remain scarce. Notably, systematic
analysis of post-SP metabolic reprogramming and membrane repair,
especially in felines, is lacking.
Glutamine (GLU) is a conditionally essential amino
acid that plays notable roles in redox homeostasis, nucleotide
biosynthesis and immunoregulation under stress or injury. GLU
preserves epithelial barrier integrity and prevents oxidative
damage in pulmonary and intestinal models (13-16);
however, its efficacy in bacterial pneumonia, especially its role
in coordinating metabolic recovery, redox balance and cell survival
after SP-induced lung damage, remains undefined.
Fish collagen peptides (FCP) are bioactive
oligopeptides derived from enzymatically hydrolyzed marine
byproducts possessing promising regenerative and anti-inflammatory
properties. FCP acts by inhibiting MAPK pathways, activating
mannose receptors and modulating Wnt/β-catenin signaling, and has
demonstrated benefits in epithelial repair, skin regeneration and
macrophage polarization (17-21).
Nonetheless, to the best of our knowledge, their role in bacterial
lung infections, including their reparative or synergistic effects
in SP-induced epithelial damage, is unexplored.
Pulmonary interstitial cells of mesenchymal lineage
play a notable role in lung tissue repair, extracellular matrix
(ECM) remodeling and epithelial recovery following injury (22). Epithelial cells are the primary
targets of bacterial infection, but mesenchymal cells are
indispensable in the subsequent repair processes. Mesenchymal cells
synthesize ECM components, such as collagen, cadherins, claudins
and fibronectin, which are essential for tissue regeneration and
structural restoration after damage (23). Moreover, they promote epithelial
recovery through paracrine signaling, secreting growth factors and
cytokines that stimulate epithelial cell proliferation, migration
and restoration of barrier integrity (22,24,25).
The interaction between mesenchymal and epithelial cells during the
repair process is crucial for maintaining lung function after
infection. Mesenchymal cells thus serve as an ideal in vitro
model for investigating reparative mechanisms during the
post-infectious recovery phase following bacterial lung injury
because of their central role in tissue regeneration and ECM
remodeling. Their ability to modulate inflammation, ECM composition
and cell-cell interactions directly influences the resolution of
infection-induced damage and supports lung tissue recovery.
In the present study, a feline pulmonary mesenchymal
cell model (FCA-L2) of SP infection was established, followed by
integrative transcriptomic and metabolomic analyses to delineate
the molecular landscape of infection-induced injury to address the
existing knowledge gaps. This study aimed to delineate SP-induced
injury patterns and evaluate the efficacy of GLU and FCP in
promoting recovery by modulating metabolism, inflammation and
structural restoration.
Materials and methods
Materials and reagents
L-glutamine (GLU, purity >99%) and doxycycline
hydrochloride (Dox, purity >98%) were sourced from Shanghai
Macklin Biochemical Co., Ltd. Fish scale-derived collagen peptides
(FCP), with a molecular weight of <5,000 Da and peptide content
>98%, were purchased from Hainan Huayan Collagen Technology Co.,
Ltd. Supplementary Tables SI and
SII details the molecular weight
distribution and fully hydrolyzed amino acid composition of FCP. SP
(Klein) Chester (ATCC 6305) was acquired from the American Type
Culture Collection. All compounds (GLU, FCP and Dox) were dissolved
in phenol red-free Minimum Essential Medium (MEM, Procell, Life
Science & Technology Co., Ltd.) before use.
Cell culture and viability assay
Feline pulmonary interstitial cells (FCA-L2), which
represented fibroblast-like stromal cells of mesenchymal lineage,
were obtained from the PETCC cell bank (https://www.petcc.org/). The cells were cultured in
Dulbecco's modified Eagle's medium (DMEM) supplemented with 10%
fetal bovine serum (FBS; Gibco; Thermo Fisher Scientific, Inc.) and
maintained in a humidified incubator at 37.5˚C and 5%
CO2. Cell viability was confirmed using trypan blue
exclusion prior to experiments.
Based on published literature (26-28)
and preliminary laboratory screenings, concentration ranges for GLU
(0-80 mM) and FCP (0-1,000 µg/ml) were selected to determine the
optimal working doses. The cells were then seeded into 96-well
plates at a density of 3x105 cells/ml for adherence for
24 h at 37.5˚C, after which various concentrations of GLU and FCP
were added for an additional 24 h at 37.5˚C. Treatment media were
subsequently removed, and 110 µl of phenol red-free MEM containing
10% Cell Counting Kit-8 (CCK-8; Beyotime Biotechnology) was added
to each well. The plates were gently shaken for 30 sec, then
incubated for 2 h at 37˚C. Absorbance of the cells was measured at
450 nm using a microplate reader. Cells cultured in MEM alone
(without treatment) served as the negative control (NC).
Lactate dehydrogenase (LDH) release
assay for SP-infected lung cells
The infection duration and bacterial concentration
were optimized to establish a stable SP infection model in feline
lung cells. LDH release was employed as a reliable indicator of
membrane damage due to the interference of bacteria with CCK-8
detection (29). SP (ATCC 6305)
was cultured in brain heart infusion broth (Qingdao Hope
Bio-technology Co., Ltd.) at 37˚C and 5% CO2 for 24 h.
The bacteria were harvested by centrifugation at 3,000 x g for 10
min at 4˚C, and the resulting pellet was resuspended in MEM. The
suspension was then adjusted to a turbidity of 0.0-0.05 McFarland
(McF) units using a calibrated turbidimeter (cat. no. WGZ-1M16100;
Shanghai Precision Instruments, Co., Ltd.). FCA-L2 cells were
seeded in 96-well plates (3x104 cells/well) and
incubated at 37.5˚C for 24 h. A total of two control groups were
established: A NC group (cells incubated with MEM only) and an NC +
Dox group (cells treated with 7.5 µg/ml Dox without SP infection)
to evaluate potential antibiotic-induced toxicity. To optimize
infection conditions, cells were exposed to SP at 0.02 or 0.05 McF
for 2 or 4 h. Following infection, the medium was replaced with MEM
containing 7.5 µg/ml Dox (2x) to inhibit further bacterial growth,
and cells were incubated for an additional 18 h at 37.5˚C.
Furthermore, to characterize the temporal progression of
post-antibiotic injury, a time-course experiment was performed.
Cells were infected with 0.05 McF SP for 4 h, followed by Dox
treatment (7.5 µg/ml) for 12, 18, 24 or 30 h at 37.5˚C before
supernatant collection. At each designated time point, plates were
centrifuged at 1,000 x g for 5 min at 4˚C, and 100 µl of
supernatant was collected.
LDH release was determined using a commercial kit
(C0017; Beyotime Biotechnology). Briefly, LDH working solution (50
µl), containing lactate, iodonitrotetrazolium chloride and
lipoamide dehydrogenase, was added to each well containing 100 µl
of supernatant and incubated at room temperature for 30 min.
Absorbance at 490 nm was measured using a microplate reader
(SpectraMax; Molecular Devices, LLC).
Evaluation of pharmacological
intervention
The effects of GLU and FCP on SP-induced injury in
FCA-L2 cells were evaluated based on the LDH release. Briefly, 2x
concentrations of GLU and FCP were prepared in MEM containing 7.5
µg/ml Dox (2x Dox-MEM). FCA-L2 cells were seeded in 96-well plates
(3x104 cells/well) and incubated for 24 h at 37.5˚C,
after which 100 µl of 0.05 McF SP suspension was added and the
cells incubated for 4 h at 37.5˚C for infection to take place. GLU-
or FCP-containing Dox-MEM (100 µl) was then added, and the cells
were incubated for an additional 18 h at 37.5˚C. Cells treated with
MEM alone served as the NC, whereas those treated with Dox-MEM
alone served as the antibiotic control. Supernatants were
subsequently collected for LDH measurement. The concentration with
the greatest reduction in LDH release (GLU, 40 mM; FCP, 500 µg/ml)
was used in subsequent experiments.
Collection of whole cell lysates of
differentially treated FCA-L2 cells
A total of four treatment groups were established:
NC (MEM only), SP infection (SP), GLU treatment (SP + GLU, 40 mM)
and FCP treatment (SP + FCP, 500 µg/ml). FCA-L2 cells were seeded
into 6-well plates (6x105 cells/well), with and without
glass coverslips, and incubated at 37.5˚C for 24 h. The cells were
subsequently treated with 1 ml of 0.05 McF SP suspension for 4 h,
followed by 1 ml of Dox-MEM containing 80 µM GLU or 1,000 µg/ml FCP
at 37.5˚C for 18 h. The SP group received Dox-MEM without
functional compounds, whereas the NC group received MEM only. Cell
supernatants were directly collected after treatment for subsequent
assays. The supernatant was centrifuged at 9,400 x g for 10 min to
pellet detached cells, which were then resuspended in their
original wells. Total protein of the FCA-L2 cells was isolated
using M-PER mammalian protein extraction reagent (Thermo Fisher
Scientific, Inc.), followed by incubation at 4˚C for 20 min to
obtain whole-cell lysates. Alternatively, FCA-L2 cells were
digested with trypsin without EDTA (0.25%) and centrifuged together
with the supernatant at 400 x g for 5 min to obtain whole-cell
lysates. The resulting cell pellet was used for apoptosis
detection.
Measurement of cytokines in culture
supernatants
Cytokines, including interleukin-1β (IL-1β; cat. no.
ml023097-2), interleukin-8 (IL-8/CXCL8; cat. no. ml023094-2) and
tumor necrosis factor-α (TNF-α; cat. no. ml035852-2) in the culture
supernatants, were quantified using specific ELISA kits (Shanghai
Enzyme-linked Biotechnology Co., Ltd.). A sandwich ELISA protocol
was employed; briefly, 50 µl of each culture supernatant was added
to 96-well plates pre-coated with capture horseradish
peroxidase-conjugated detection antibodies and subsequently
incubated at 37˚C for 60 min. The plates were subsequently washed
five times with PBST, followed by the addition of
3,3',5,5'-tetramethylbenzidine substrate. After incubation for 15
min in the dark at 37˚C, the reaction was terminated by adding 2 M
H2SO4. The absorbance of the mixture was
recorded at 450 nm using a microplate reader (SpectraMax; Molecular
Devices, LLC).
Measurement of tight junction
proteins
The expression levels of tight junction-associated
proteins, including E-cadherin (cat. no. ml680120), occludin (cat.
no. ml847159), claudin (cat. no. ml847222) and zonula occludens-1
(ZO-1; cat. no. ml847150), were determined in whole-cell lysates
using ELISA kits (Shanghai Enzyme-linked Biotechnology Co., Ltd.),
following the aforementioned procedure.
Measurement of oxidative stress
markers
Oxidative stress was evaluated by quantifying
malondialdehyde (MDA), superoxide dismutase (SOD) activity,
glutathione peroxidase (GPX) activity, total antioxidant capacity
(T-AOC) and total oxidant status (TOS) in whole-cell lysates.
Protein concentration was determined using the Pierce BCA protein
assay kit (Thermo Fisher Scientific, Inc.). Whole-cell lysate
samples (25 µl) were mixed with 200 µl of BCA working solution,
incubated at 37˚C for 30 min, and the absorbance subsequently
measured at 562 nm using a microplate reader (SpectraMax; Molecular
Devices, LLC). All oxidative stress assays were strictly performed
in accordance with the manufacturer's reaction protocols. MDA
levels were measured using a commercial thiobarbituric acid
(TBA)-based kit (cat. no. S0131S; Beyotime Biotechnology) by
incubating the samples at 100˚C for 15 min, with absorbance
recorded at 532 nm. SOD activity was assessed using a commercial
WST-8 detection reagent containing WST-8 and xanthine oxidase (cat.
no. S0101S; Beyotime Biotechnology). Briefly, whole-cell lysate
samples (20 µl) were incubated with the detection reagent at 37˚C
for 30 min, followed by measurement of the absorbance read at 450
nm. GPX activity was measured using a commercial DTNB
colorimetric-based kit (cat. no. BC1195; Beijing Solarbio Science
& Technology Co., Ltd.). Briefly, 20 µl of cell lysate was
mixed with the reaction working solution according to the
manufacturer's instructions and incubated at 37˚C for 5 min,
followed by the addition of DTNB for color development. The
absorbance was recorded at 412 nm using a microplate reader. T-AOC
was determined using the Fe³+-tripyridyltriazine
(TPTZ)-based FRAP method (cat. no. BC1315-100T/96S; Beijing
Solarbio Science & Technology Co., Ltd.). Briefly, whole-cell
lysate samples (6 µl) were mixed with 180 µl of detection reagent,
incubated at 25˚C for 10 min, followed by measurement of the
absorbance at 593 nm. TOS was quantified using a commercial kit
(cat. no. BC6245-100T/96S; Beijing Solarbio Science &
Technology Co., Ltd.). Briefly, 30 µl of sample was mixed with 160
µl of reaction reagent 1, and the baseline absorbance at 560 nm was
recorded. Subsequently, 10 µl of xylenol orange reagent (reagent 2)
was added and the mixture was incubated at 37˚C for 5 min. The
absorbance was then measured at 560 nm, and TOS levels were
calculated according to the manufacturer's instructions.
Analysis of cell morphology and
apoptosis
Cells on coverslips from each group were fixed with
4% paraformaldehyde (Beyotime Biotechnology) at 25˚C for 15 min,
followed by staining with crystal violet solution (Qingdao Hope
Bio-technology Co., Ltd.) at 25˚C for 5 min. The cells were then
rinsed thrice using PBS (2 min for each rinse), and the
morphological features were subsequently examined under a light
microscope (Olympus CX33; Olympus Corporation). Cell apoptosis was
detected by staining the cell pellets from each group with 5 µl
Annexin V-FITC and 15 µl propidium iodide (PI) solution (Beyotime
Biotechnology) in the dark for 15 min at room temperature. The
cells were then centrifuged at 1,000 x g for 4 min, after which a
20 µl aliquot of the stained suspension was mounted on a concave
glass slide (Nantong Mevid Life Science Co., Ltd.) and examined
under a fluorescence microscope (BDS400; Chongqing Aote Optical
Instrument, Co., Ltd.). To quantify cell density and apoptosis
intensity, the obtained images were analyzed using ImageJ software
(version 1.54P; National Institutes of Health). For each group, the
mean gray value was determined from at least three randomly
selected fields of view. The quantitative results were then
expressed as a percentage relative to the NC group, which was
defined as 100%.
RNA sequencing. RNA isolation and
library preparation
Total RNA was extracted from cell pellets of each
treatment group for transcriptome sequencing. Briefly, the cells
were lysed in TRIzol reagent (Beyotime Biotechnology), followed by
phase separation using chloroform (Shanghai Macklin Biochemical
Co., Ltd.) and RNA precipitation using isopropanol (Shanghai
Macklin Biochemical Co., Ltd.). RNA quantity and integrity were
assessed using the Agilent 5400 Bioanalyzer. Samples with an RNA
Integrity Number (RIN) ≥4.0 were considered qualified for
subsequent analysis. Polyadenylated mRNA was subsequently enriched
from total RNA using oligo(dT)-conjugated magnetic beads (cat. no.
RK20257; ABclonal Biotech Co., Ltd.). cDNA libraries were
constructed using the Fast RNA-seq Lib Prep Kit V2 (cat. no.
RK20306; ABclonal Biotech Co., Ltd.) according to the
manufacturer's instructions. First-strand cDNA synthesis was
carried out using M-MuLV reverse transcriptase, followed by RNA
template removal using RNase H and second-strand synthesis using
DNA polymerase I. The resulting double-stranded cDNA was subjected
to end repair, A-tailing and ligation with Illumina-compatible
adapters, followed by purification with AMPure XP magnetic beads
(cat. no. RK20257; ABclonal Biotech Co., Ltd.). The cDNA templates
were amplified by PCR using the high-fidelity PCR master mix and
universal primers provided in the kit (cat. no. RK20306; ABclonal
Technology Co., Ltd.). The thermocycling conditions were as
follows: Initial denaturation at 98˚C for 45 sec; 12-15 cycles
comprised of denaturation at 98˚C for 10 sec), annealing at 60˚C
for 15 sec and extension at 72˚C for 30 sec; and a final extension
at 72˚C for 1 min. The generated libraries finally underwent
paired-end sequencing on an Illumina NovaSeq 6000 platform
(Novogene Co., Ltd.).
Differential expression analysis. Raw reads
were processed using fastp (v2.3; https://github.com/OpenGene/fastp/blob/master/README.md)
to remove adapter sequences, undetermined bases and low-quality
reads, yielding high-quality clean data. The clean reads were
subsequently aligned to the Felis catus reference genome
(Felis_catus_9.0; https://ftp.ncbi.nlm.nih.gov/genomes/all/GCF/000/181/335/GCF_000181335.3_Felis_catus_9.0/)
using HISAT2 (v2.2; https://daehwankimlab.github.io/hisat2/). Gene-level
read counts were obtained using featureCounts (v2.0; https://subread.sourceforge.net/). Gene
expression levels were normalized and quantified using fragments
per kilobase of transcript per million mapped (FPKM) reads.
Principal component analysis (PCA) was performed based on FPKM
values. Differentially expressed genes (DEGs) between groups were
identified using the DESeq2 R package (v1.42.0; https://bioconductor.org/packages/release/bioc/html/DESeq2.html).
Raw P-values were calculated and adjusted for multiple testing
using the Benjamini-Hochberg method. Genes with an adjusted P-value
(padj)≤0.05 and absolute fold change (FC)≥1.2 were considered
significantly differentially expressed.
Gene set enrichment analysis (GSEA). GSEA was
performed to identify biological pathways affected by various
treatments. Notably, gene sets were annotated based on the Kyoto
Encyclopedia of Genes and Genomes database (KEGG; https://www.kegg.jp/). All genes were ranked based on
expression differences between groups using the Signal2Noise
metric. The analysis was conducted using the GSEA desktop
application (Broad Institute; http://www.broadinstitute.org/gsea). Enrichment scores
were calculated via permutation testing and normalized to account
for gene set size. Pathways with a false discovery rate <0.25
were considered significantly enriched.
Untargeted metabolomics analysis
Untargeted metabolomics profiling was performed on
whole-cell lysates from each treatment group using liquid
chromatography-tandem mass spectrometry (LC-MS/MS). Cells were
extracted with 80% methanol, flash-frozen in liquid nitrogen and
ground into powder. The ground samples were reconstituted in 10%
methanol after lyophilization. Equal aliquots from all samples were
pooled to generate a quality control (QC) sample, while 53%
methanol was used as a blank control. Chromatographic separation
was conducted on a ultra-high-performance-LC system equipped with a
Hypersil GOLD C18 column (Thermo Fisher Scientific, Inc.), followed
by MS detection using a Q Exactive™ HF instrument
(Thermo Fisher Scientific, Inc.) operating in both positive and
negative ionization modes. Chromatographic separation was performed
on a Hypersil GOLD C18 column maintained at 40˚C with a constant
flow rate of 0.2 ml/min. The mobile phases consisted of 0.1% formic
acid in water (A) and methanol (B). A linear gradient elution was
applied as follows: 98% A at 0-1.5 min, decreased to 15% A at 3
min, followed by 0% A at 10 min. The system returned to initial
conditions (98% A) at 10.1 min and was re-equilibrated until 12
min. Mass spectrometric detection was performed in both positive
and negative electrospray ionization modes with full MS scan ranges
set from 100 to 1,500 m/z. The spray voltage was set to 3.5 kV,
with a sheath gas flow rate of 35 psi and an auxiliary gas flow
rate of 10 l/min. The capillary temperature was maintained at
320˚C, and the auxiliary gas heater was set to 350˚C. The S-lens RF
level was configured at 60. Data acquisition was carried out in
data-dependent MS/MS mode.
Raw data were processed using the XCMS software
(V4.0; The Scripps Research Institute) for peak detection and
quantification. Metabolite identification was performed using
Novogene In-house Database (Novogene Co., Ltd.), which integrates
high-resolution MS data from established chemical standards and
internal reference materials. Data preprocessing, which involved
filtering of the background ions using blank samples and exclusion
of metabolites with a coefficient of variation >30% in QC
samples, was performed in Python (v3.5; Python Software
Foundation). Normalization was performed by calculating the ratio
of metabolite intensities in each sample to those in QC samples.
PCA was conducted using metaX (v1.4.16; BGI Genomics), followed by
a calculation of the variable importance in projection (VIP)
scores, FC and t-test P-values. Metabolites with VIP≥1, P<0.05
and FC≥1.5 or ≤0.67 were defined as significantly different.
Volcano plots were generated using the ggplot2 package in R
(v3.4.3; https://ggplot2.tidyverse.org/). Pathway enrichment
analysis of differential metabolites was performed using GSEA
software (gsea-3.0; Broad Institute; https://www.gsea-msigdb.org/gsea/index.jsp).
Integrated transcriptomic and
metabolomic analysis
Integrated analysis of transcriptomic and
metabolomic datasets was performed using the MetTranAnalysis module
on the NovoMagic cloud platform (Novogene Co., Ltd.) to investigate
post-transcriptional regulatory patterns. KEGG-based co-enrichment
analysis was conducted to identify pathways jointly affected by
DEGs and differential metabolites. Bubble plots of shared pathways
were generated, followed by a determination of their statistical
significance using Fisher's exact test (P<0.05). Gene-metabolite
correlation analysis was subsequently performed to strengthen the
integration at the molecular level. Genes and metabolites belonging
to the enriched core KEGG pathways in each comparison group (GLU_SP
vs. SP and FCP_SP vs. SP) were selected for correlation analysis.
Pearson correlation coefficients were calculated using the
NovoMagic platform based on log2-transformed normalized expression
values of transcripts and metabolites. Correlation matrices were
visualized as heatmaps to display the association patterns between
key genes and metabolites.
Statistical analysis
Data are presented as means ± standard deviation
(n≥4 biological replicates per group). Statistical analyses were
performed using SPSS software (IBM Corp., version 26). Statistical
differences among multiple groups were first evaluated using
Levene's test to assess the homogeneity of variances. For datasets
with equal variances, a one-way analysis of variance (ANOVA)
followed by Tukey's post-hoc test was performed. For datasets with
unequal variances, Welch's ANOVA followed by the Games-Howell
post-hoc test was employed. P<0.05 was considered to indicate a
statistically significant difference.
Results
Optimization of SP infection
conditions
The optimal SP infection condition in FCA-L2 cells
was determined based on LDH release. As shown in Fig. 1A, LDH levels in the NC and NC + Dox
groups were similar (100±10.10% vs. 99.54±3.90%), indicating that
Dox had no effect on LDH release. SP infection at a low dose (0.02
McF) for 2 and 4 h, followed by 18 h of Dox treatment, caused only
a slight increase in LDH (108.40-111.72%). By contrast, high-dose
SP infection (0.05 McF) for 4 h led to a sharp increase
(352.90±12.39%) despite Dox treatment, indicating persistent
cytotoxicity and damage. To further clarify the temporal
characteristics of post-antibiotic injury, LDH release was assessed
at different time points (12, 18, 24 and 30 h) after Dox treatment
following SP infection (0.05 McF, 4 h) (Fig. 1B). LDH levels remained consistently
elevated at 12 h (381.46±9.27%), 18 h (352.90±7.95%) and 24 h
(365.68±6.42%), but declined at 30 h (309.61±6.97%), suggesting
that the 12-24 h window represents a sustained injury phase. SP
infection at 0.05 McF for 4 h, followed by 18 h of Dox treatment,
was selected as the standard condition for subsequent experiments
to ensure feasibility.
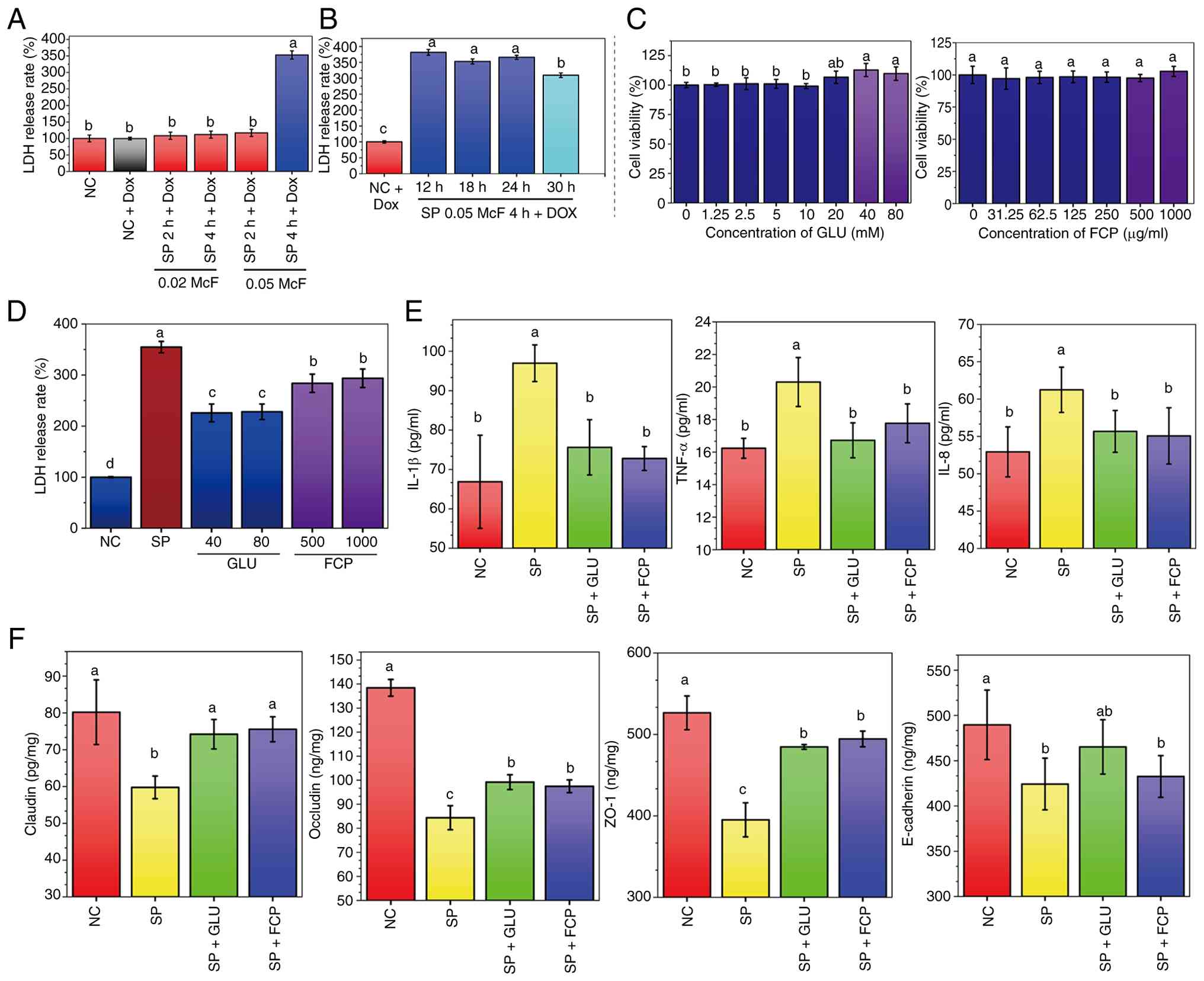 | Figure 1Effects of GLU and FCP on
cytotoxicity, cytokine levels and tight junction protein expression
in SP-infected feline lung FCA-L2 cells. (A) LDH release following
SP infection at different durations and bacterial concentrations.
NC: untreated cells in minimum essential medium; NC-Dox: cells
treated with 3.75 µg/ml Dox for 18 h; SP + 2 h + Dox and SP + 4 h +
Dox: SP infection for 2 or 4 h, followed by 3.75 µg/ml Dox
treatment for 18 h. (B) Time-course assessment of LDH release after
SP infection (0.05 McF, 4 h) followed by doxycycline treatment
(3.75 µg/ml) for 12, 18, 24 or 30 h. The NC + DOX group represents
uninfected cells treated with DOX for the corresponding duration.
(C) Cell viability of FCA-L2 cells after treatment with different
concentrations of GLU (left) or FCP (right), assessed using the
Cell Counting Kit-8 assay. (D) LDH release after SP infection (4 h)
followed by 18 h treatment with GLU or FCP in the presence of 3.75
µg/ml Dox. The SP group received Dox alone. (E) Levels of IL-1β,
TNF-α and IL-8 in the supernatant of SP-infected cells treated with
GLU (40 mM) or FCP (500 µg/ml). (F) Expression of tight junction
proteins, including E-cadherin, occludin, claudin and ZO-1, after
GLU or FCP treatment. All data are presented as mean ± standard
deviation (n≥4 per group). Different lowercase letters (a-d)
indicate significant differences (P<0.05), while groups sharing
at least one common letter show no statistically significant
difference. GLU, glutamine; FCP, fish collagen peptides; SP,
Streptococcus pneumoniae; ZO-1, zonula occludens-1; NC,
negative control; Dox, doxycycline; McF, McFarland; LDH, lactate
dehydrogenase. |
GLU and FCP reduced SP-induced LDH
release
As shown in Fig.
1C, CCK-8 assays demonstrated that GLU (1.25-20 mM) and FCP
(31.25-500 µg/ml) had no adverse effects on FCA-L2 cell viability.
Notably, higher concentrations (40-80 mM GLU and 1,000 µg/ml FCP)
demonstrated slight promotive effects on cell growth, confirming
their safety. Consequently, GLU (40 and 80 mM) and FCP (500 and
1,000 µg/ml) were selected for subsequent efficacy evaluations. SP
infection significantly increased LDH release to 354.80±11.12%
relative to NC (100%) (Fig. 1D);
however, treatment with GLU reduced LDH release to 225.86±17.42%
(40 mM) and 228.05±15.29% (80 mM), whereas FCP treatment reduced it
to 283.85±17.93% (500 µg/ml) and 293.67±18.07% (1,000 µg/ml). These
results indicate that both GLU and FCP alleviated SP-induced
cytotoxicity.
GLU and FCP reduced the levels of
SP-induced inflammatory cytokines
SP infection significantly elevated the secretion of
IL-1β, TNF-α and IL-8 in the supernatant of FCA-L2 cells compared
with the NC group (Fig. 1E).
Treatment with GLU (40 mM) and FCP (500 µg/ml) significantly
reduced the levels of all three cytokines compared with SP: IL-1β
decreased from 96.96±4.66 pg/ml (SP) to 75.61±7.01 pg/ml (GLU + SP)
and 72.78±3.04 pg/ml (FCP + SP); TNF-α decreased from 19.88±1.85
pg/ml to 16.72±1.07 and 17.77±1.19 pg/ml; and IL-8 decreased from
60.02±4.47 pg/ml to 55.67±2.80 and 55.07±3.77 pg/ml, respectively.
These results suggest that both GLU and FCP alleviate SP-induced
inflammatory responses in feline lung cells, with the most
prominent effect observed on IL-1β.
GLU and FCP restored junction protein
expression suppressed by SP
SP infection significantly reduced the expression of
claudin, ZO-1, occludin and E-cadherin in FCA-L2 cells compared
with NC (Fig. 1F). However,
treatment with GLU (40 mM) or FCP (500 µg/ml) partially reversed
these reductions. Claudin increased from 59.76 pg/mg (SP) to 74.22
(GLU + SP) and 75.56 pg/mg (FCP + SP), ZO-1 increased from 395.17
to 484.65 and 494.46 ng/mg and occludin increased from 84.39 to
99.20 and 97.49 ng/mg, respectively. Regarding E-cadherin, its
expression was significantly restored by GLU treatment, increasing
from 424.23 ng/mg in the SP group to 465.27 ng/mg. By contrast,
although FCP treatment slightly increased E-cadherin levels to
432.62 ng/mg, this change did not reach statistical
significance.
GLU and FCP attenuated apoptosis and
oxidative stress in SP-infected cells
Cell morphology and apoptosis were assessed via
crystal violet and Annexin V-FITC/PI staining, respectively.
Notably, FCA-L2 cells in the NC group exhibited dense and orderly
arrangement with tight intercellular connections. (Fig. 2A; upper and middle panels). By
contrast, cells infected with SP exhibited increased staining
intensity, reduced cell density and signs of shrinkage and rupture.
Treatment with 40 mM GLU or 500 µg/ml FCP restored cellular
morphology, reduced staining intensity and enhanced cell-to-cell
contact compared with SP infected cells. Quantitative analysis of
the staining showed that the relative cell density decreased from
100% (NC) to 75.57% after SP infection; however, treatment with 40
mM GLU or 500 ug/ml FCP restored cell morphology and increased the
relative density to 85.89 and 88.46%, respectively.
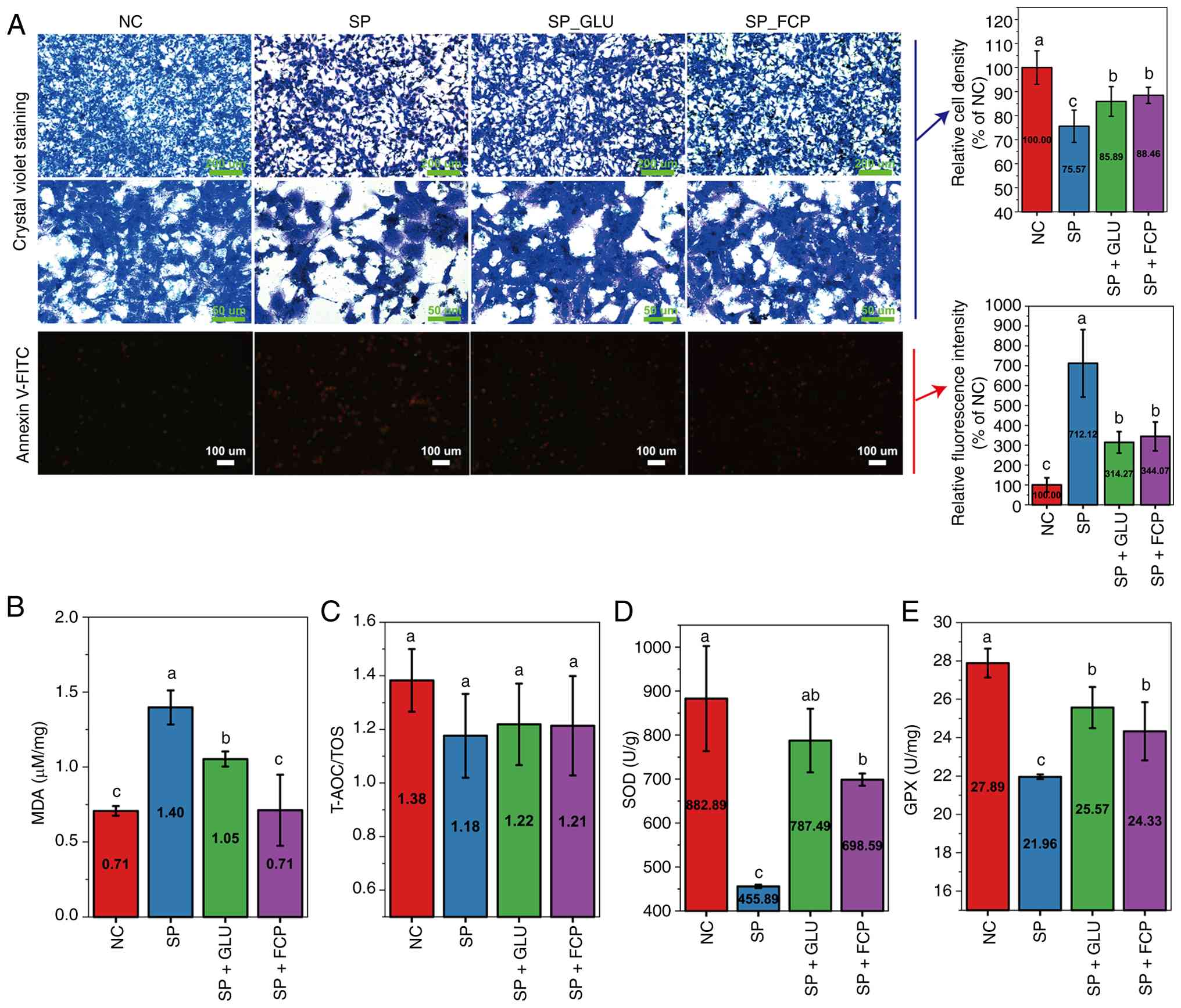 | Figure 2Effects of GLU and FCP on morphology,
apoptosis and oxidative stress in SP-infected FCA-L2 cells. (A)
Morphological changes (crystal violet staining, scale bars, 200 and
50 µm) and apoptosis (Annexin V-FITC/PI staining, scale bar, 100
µm) in each group following intervention with GLU (40 mM) or FCP
(500 µg/ml). The right panels show the corresponding quantitative
bar charts for relative cell density and the relative percentage of
apoptotic cells following intervention with GLU (40 mM) or FCP (500
ug/ml). (B) MDA levels; (C) Ratio of T-AOC/TOS; (D) SOD levels; and
(E) GPX levels. All data are expressed as mean ± standard deviation
(n≥4 per group). Different lowercase letters (a-c) indicate
significant differences (P<0.05), while groups sharing at least
one common letter show no statistically significant difference.
GLU, glutamine; FCP, fish collagen peptides; SP, Streptococcus
pneumoniae; MDA, malondialdehyde; SOD, superoxide dismutase;
MDA, malondialdehyde; T-AOC, total anti-oxidant capacity; TOS,
total oxidant capacity; GPX, glutathione peroxidase. |
Annexin V-FITC/PI staining (Fig. 2A, lower panel) revealed a increase
in early and late apoptotic cells following SP infection, which was
reduced by GLU and FCP treatment. The relative fluorescence
intensity of apoptotic cells spiked from 100% (NC) to 712.12% (SP
group). Notably, GLU and FCP treatment effectively mitigated this
effect, reducing the intensity to 314.27 and 344.07%, respectively.
Regards oxidative stress markers (Fig.
2B-E), SP infection significantly elevated MDA levels and
reduced SOD and GPX activities compared with the NC group. These
alterations were significantly reversed by both GLU and FCP
treatment. Although the T-AOC/TOS ratio followed a downward trend
after SP infection and a slight upward trend following GLU or FCP
treatment, these changes did not reach statistical
significance.
Transcriptomic analysis and DEG
screening
RNA sequencing was performed to assess global
transcriptomic changes among NC, SP, GLU_SP and FCP_SP groups. PCA
revealed distinct clustering across all four groups; there was a
clear separation between SP and NC, whereas GLU_SP and FCP_SP
showed partial shifts in different directions (Fig. 3A). The DEG clustering heatmap
demonstrated substantial transcriptional alterations after SP
infection and notable expression changes following GLU or FCP
treatment (Fig. 3B). Venn analysis
revealed 13,052 commonly expressed genes across groups, along with
subsets of uniquely regulated genes in each treatment (Fig. 3C). SP vs. NC had 3,980 DEGs (2,045
upregulated, 1,935 downregulated), GLU_SP vs. SP had 3,321 DEGs
(1,679 upregulated, 1,642 downregulated) and FCP_SP vs. SP had
2,312 DEGs (1,257 upregulated, 1,055 downregulated) (Fig. 3D). This finding suggested that both
GLU and FCP modulated SP-induced transcriptomic responses.
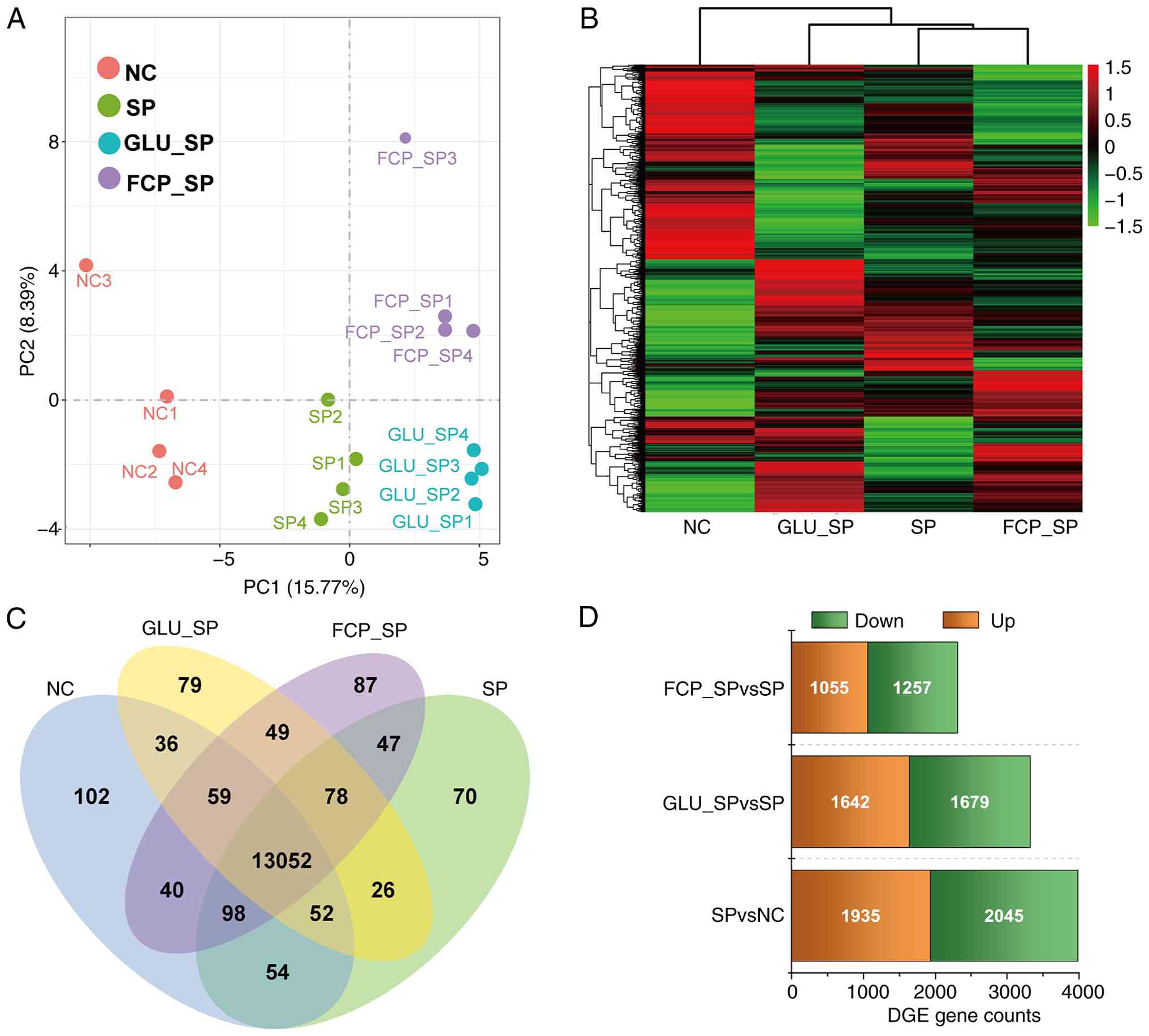 | Figure 3Transcriptomic profiling and
identification of DEGs. (A) PCA showing distinct clustering among
NC, SP, GLU_SP and FCP_SP groups. (B) Heatmap of DEGs based on
FPKM-normalized values; columns represent sample groups and rows
represent individual genes. (C) Venn diagram showing the number of
unique and overlapping DEGs among groups. (D) Bar plot summarizing
the number of upregulated and downregulated DEGs in each
comparison. Data were based on reference-guided RNA sequencing (n=4
per group). DGES, differentially expressed genes; PCA, Principal
component analysis; GLU, glutamine; FCP, fish collagen peptides;
SP, Streptococcus pneumoniae; SOD, superoxide dismutase;
MDA, malondialdehyde; NC, negative control. |
SP activated endocytosis and
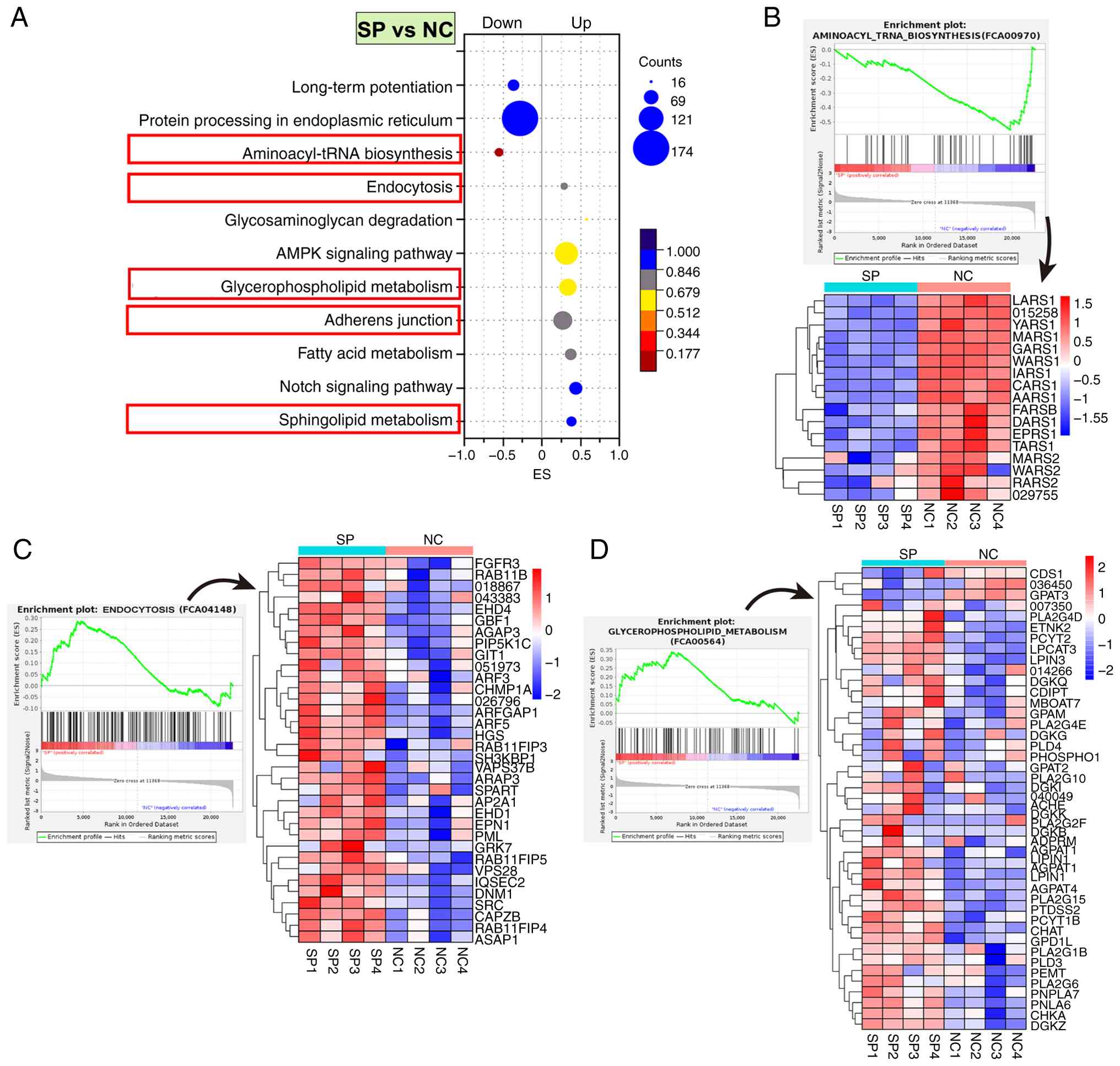
suppressed aminoacyl-tRNA biosynthesis in lung cells
GSEA was performed to explore the transcriptional
mechanisms of SP infection in FCA-L2 cells. SP significantly
upregulated ‘endocytosis’, ‘glycerophospholipid metabolism’,
‘sphingolipid metabolism’ and ‘adherens junction’ pathways compared
with NC. Alternatively, SP significantly downregulated the
‘aminoacyl-tRNA biosynthesis’ pathway compared with NC (Fig. 4A).
SP infection also significantly downregulated 14 out
of 17 core genes in the aminoacyl-tRNA biosynthesis pathway
(Fig. 4B). These genes included
LARS1, YARS1, MARS1, GARS1, WARS1, IARS1, CARS1, AARS1, FARSB,
DARS1, EPRS1 and TARS1. Notably, these genes encode
cytoplasmic aminoacyl-tRNA synthetases responsible for conjugating
leucine, tyrosine, methionine, glycine, tryptophan, isoleucine,
cysteine, alanine, phenylalanine, aspartate, glutamate, proline and
threonine to their respective tRNAs (30). Mitochondrial synthetases MARS2,
WARS2 and RARS2 were also suppressed, suggesting that SP
impairs both cytosolic and mitochondrial protein translation. This
widespread inhibition of aminoacylation potentially disrupted
translational fidelity and efficiency, contributing to cellular
injury and stress responses upon infection (31).
By contrast, SP infection significantly upregulated
a cluster of core genes in the endocytosis pathway. The notable
genes that were upregulated included RAB11B, RAB11FIP3,
RAB11FIP4 and RAB11FIP5, which encode notable regulators
of recycling endosomes (32)
(Fig. 4C). Notably, RAB11B,
a master GTPase that controls the slow recycling of membrane
proteins and receptors (33), was
notably induced, suggesting it may act as a central target
mediating SP-induced endocytic activation. Its interacting
partners, RAB11FIP3-5, which facilitate cargo sorting and
endosomal trafficking (34), were
also elevated, indicating enhanced membrane recycling dynamics.
Upregulation of AP2A1 (clathrin adaptor complex),
DNM1 (dynamin GTPase for vesicle scission), SH3KBP1
(endocytic scaffold protein) and EPN1 (epsin, membrane
curvature modulator) indicated activation of clathrin-mediated
endocytosis. Moreover, upregulation of ARF3, ARF5,
ARFGAP1, EHD1 and EHD4 provided additional
evidence for the widespread activation of vesicle budding,
trafficking and recycling machinery. These transcriptional changes
collectively suggest that SP infection enhances endocytic
trafficking and membrane turnover in lung mesenchymal cells,
potentially disrupting intracellular homeostasis and facilitating
pathogen-host interactions.
SP infection also significantly upregulated a
cluster of genes involved in glycerophospholipid metabolism,
including GPAT3 (glycerol-3-phosphate acyltransferase),
AGPAT1 and AGPAT4 (1-acylglycerol-3-phosphate
O-acyltransferases) and LPIN1 (phosphatidate phosphatase).
Upregulation of these genes denoted accelerated de novo
phosphatidic acid synthesis and triacylglycerol precursor
production (35). The upregulation
of PCYT1B and PTDSS2, which encode CDP-choline
synthase and phosphatidylserine synthase, respectively, as well as
CHKA (choline kinase) and PEMT
(phosphatidylethanolamine N-methyltransferase), reflected enhanced
phosphatidylcholine and phosphatidylserine biosynthesis (36). Upregulation of DGKZ, DGKB
and GPD1L suggested increased diacylglycerol-phosphatidic
acid interconversion and redox-linked glycerolipid flux (37,38).
Enzymes involved in phospholipid hydrolysis, including PLA2G15,
PLA2G6, PLA2G1B and PLD3, were also elevated, supporting
intensified membrane lipid turnover. Moreover, ADPRM
(ADP-ribose pyrophosphatase) and CHAT (choline
acetyltransferase) were upregulated, implying altered nucleotide
and acetylcholine-linked phospholipid metabolism. These
transcriptional changes collectively indicated that SP infection
activates glycerophospholipid remodeling through coordinated
upregulation of lipid biosynthetic, hydrolytic and signaling
enzymes, potentially disrupting membrane integrity and promoting
inflammatory stress responses in lung mesenchymal cells.
GLU suppressed cell cycle-related
pathways in SP-infected cells
GLU intervention (GLU_SP vs. SP) led to significant
pathway remodeling (Fig. 5A),
leading to downregulation of pathways, including the ‘cell cycle’,
‘aminoacyl-tRNA biosynthesis’, ‘p53 signaling’ and ‘apoptosis’,
suggesting inhibition of proliferation and stress responses. By
contrast, the ‘biosynthesis of nucleotide sugars’ and ‘biosynthesis
of amino sugars’, as well as ‘amino acid and nucleotide sugar
metabolism’ were significantly upregulated (Fig. 5E-F), indicating a metabolic shift
toward glycan precursor generation. These changes suggested that
GLU suppressed SP-induced proliferative stress while reprogramming
intracellular carbohydrate metabolism.
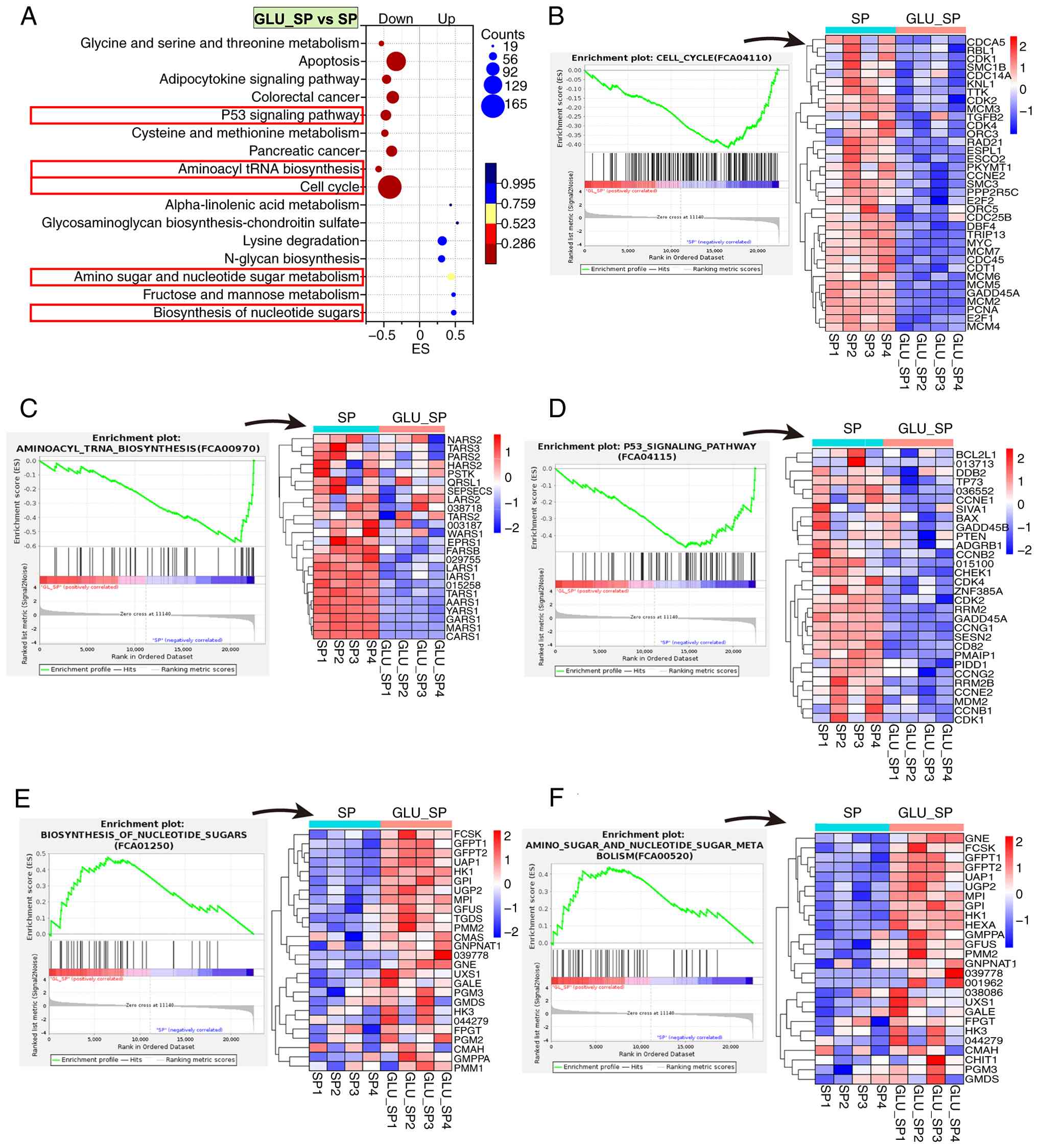 | Figure 5KEGG-based GSEA of transcriptomic
changes in FCA-L2 cells following GLU treatment after SP infection.
(A) KEGG enrichment bubble plot of pathways significantly altered
in the GLU_SP vs. SP group. The x-axis indicates ES, bubble size
represents the number of core genes and bubble color corresponds to
FDR values. (B-F) GSEA enrichment plots and heatmaps of core genes
enriched in representative pathways, including (B) cell cycle, (C)
aminoacyl-tRNA biosynthesis, (D) p53 signaling pathway, (E)
biosynthesis of nucleotide sugars and (F) amino sugar and
nucleotide sugar metabolism. Heatmaps display Z-score normalized
expression levels (n=4 per group). KEGG, Kyoto Encyclopedia of
Genes and Genomes; GSEA, gene set enrichment analysis; GLU,
glutamine; SP, Streptococcus pneumoniae; NC, negative
control; ES, enrichment score; FDR, false discovery rate. |
GLU treatment led to broad suppression of core
regulatory genes in the cell cycle pathway (Fig. 5B). Cyclin-dependent kinases and
regulators, including CDK1, CDK2, CDK4, CCNE2 and
CDC25B, were downregulated, indicating inhibition of G1/S
and G2/M transitions (39).
Multiple DNA replication licensing and elongation factors, such as
MCM2-7, CDT1, CDC45 and PCNA, were also suppressed,
reflecting S-phase arrest (40).
In parallel, mitotic checkpoint and chromosome segregation
components, including SMC1B, SMC3, RAD21 and TTK,
were downregulated, suggesting impaired spindle assembly and
progression into mitosis (41,42).
These changes collectively indicated that GLU inhibited SP-induced
aberrant cell cycle progression by targeting DNA replication,
checkpoint signaling and mitotic machinery in lung mesenchymal
cells.
Moreover, GLU treatment suppressed a panel of
cytoplasmic aminoacyl-tRNA synthetases involved in the
aminoacyl-tRNA biosynthesis pathway (Fig. 5C). They included LARS1, MARS1,
GARS1, AARS1, CARS1, TARS1, FARSB and IARS1, which are
responsible for charging tRNAs with leucine, tyrosine, methionine,
glycine, alanine, cysteine, threonine, phenylalanine and
isoleucine, respectively. This coordinated repression indicated a
global inhibition of tRNA aminoacylation, suggesting translational
suppression and reduced protein synthesis demand under GLU
treatment in SP-infected lung mesenchymal cells.
GLU treatment also led to coordinated downregulation
of multiple core genes involved in DNA damage response, cell cycle
arrest and apoptosis regulation in the p53 signaling pathway
(Fig. 5D). Notable
cyclin-dependent kinases (CDK1, CDK2 and CDK4) and
their regulatory cyclins (CCNB1, CCNB2, CCNE2, CCNG1 and
CCNG2) were suppressed, indicating a blockade of G1/S and
G2/M transitions. Checkpoint kinase CHEK1 and tumor
suppressor GADD45A, which mediate DNA damage-induced arrest,
were also downregulated. In addition, GLU reduced the expression of
RRM2 and RRM2B (ribonucleotide reductase subunits),
PMAIP1 and PIDD1 (pro-apoptotic mediators) and
MDM2 (a p53 feedback regulator). These transcriptional
changes suggested that GLU dampens p53-mediated cell cycle
checkpoint activation and apoptotic signaling in SP-infected lung
mesenchymal cells, potentially contributing to cellular recovery
and reduced stress response.
GLU activated nucleotide sugar
metabolism
GLU intervention significantly upregulated a cluster
of core genes involved in nucleotide and amino sugar biosynthesis
and nucleotide sugar metabolism (Fig.
5E and F). Key upregulated
genes included GFPT1 and GFPT2
(glutamine-fructose-6-phosphate transaminases), UAP1
(UDP-N-acetylglucosamine pyrophosphorylase), GNE (UDP-GlcNAc
2-epimerase/ManNAc kinase) and PGM3
(phosphoacetylglucosamine mutase). Notably, these genes constitute
the core enzymatic machinery of the hexosamine biosynthetic pathway
(43). UGP2, GPI, HK1 and
HK3, which are involved in glucose phosphorylation and
UDP-glucose generation (44), were
also upregulated. The enhanced expression of GALE, GMD and
UXS1, which catalyze interconversion among UDP-glucose,
UDP-galactose, UDP-xylose and GDP-fucose (45), underscored the broad activation of
nucleotide sugar biosynthesis. These transcriptional changes
suggested that GLU stimulates nucleotide sugar supply and amino
sugar metabolism, potentially supporting glycoprotein synthesis and
redox homeostasis in lung mesenchymal cells during SP-induced
stress.
FCP suppressed amino acid biosynthesis
in SP-infected cells
FCP treatment (FCP_SP vs. SP) induced significant
transcriptional remodeling (Fig.
6A), which involved downregulation of notable pathways,
including ‘aminoacyl-tRNA biosynthesis’, ‘biosynthesis of amino
acids’, ‘insulin resistance’, ‘p53 signaling pathway’, ‘mTOR
signaling pathway’ and ‘apoptosis (multiple species)’. This
phenomenon indicated inhibition of amino acid production, protein
synthesis and stress signaling. By contrast, the ‘biosynthesis of
nucleotide sugars’ and ‘biosynthesis of amino sugars’, as well as
‘amino sugar and nucleotide sugar metabolism’ pathways were
significantly upregulated, reflecting enhanced glycan precursor
generation. These changes suggested that FCP attenuates metabolic
and apoptotic stress while promoting glycosylation-associated
recovery processes in SP-infected lung mesenchymal cells.
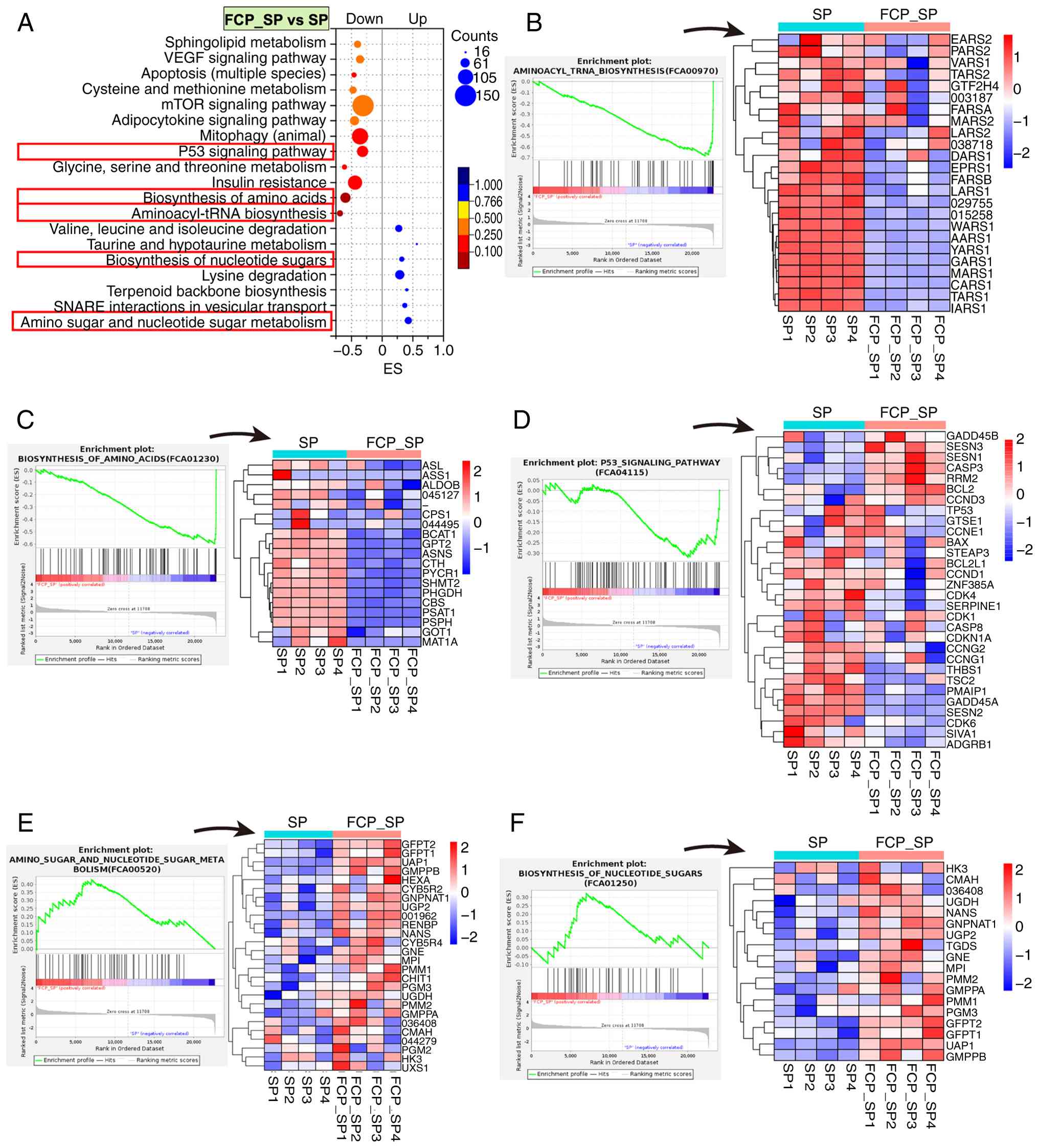 | Figure 6KEGG-based GSEA of transcriptomic
changes in FCA-L2 cells following FCP treatment after SP infection.
(A) KEGG enrichment bubble plot showing pathways significantly
regulated in the FCP_SP vs. SP group. The x-axis represents ES,
bubble size indicates the number of enriched genes and color
corresponds to the FDR. (B-F) GSEA enrichment plots and heatmaps of
core genes in selected pathways, including (B) aminoacyl-tRNA
biosynthesis, (C) biosynthesis of amino acids, (D) p53 signaling
pathway, (E) amino sugar and nucleotide sugar metabolism and (F)
biosynthesis of nucleotide sugars. Heatmaps show Z-score-normalized
gene expression (n=4 per group). KEGG, Kyoto Encyclopedia of Genes
and Genomes; GSEA, gene set enrichment analysis; FCP, fish collagen
peptides; SP, Streptococcus pneumoniae; NC, negative
control; ES, enrichment score; FDR, false discovery rate. |
In addition, FCP treatment resulted in widespread
suppression of both cytoplasmic and mitochondrial aminoacyl-tRNA
synthetases in the aminoacyl-tRNA biosynthesis pathway (Fig. 6B). They included LARS1, YARS1,
MARS1, CARS1, AARS1, GARS1, TARS1 and IARS1, which are
essential for accurate tRNA charging and translational fidelity.
Mitochondrial isoforms, such as LARS2, MARS2, TARS2, PARS2
and EARS2, were also downregulated, indicating reduced
capacity for mitochondrial protein synthesis. These changes
suggested a global attenuation of aminoacylation processes and
protein translation following FCP treatment.
In parallel, the biosynthesis of amino acids pathway
(Fig. 6C) exhibited coordinated
downregulation of multiple metabolic enzymes, including PHGDH,
PSAT1 and PSPH (serine synthesis), SHMT2
(serine-glycine interconversion), ASNS (asparagine synthetase),
BCAT1 (branched-chain amino acid metabolism) and
MAT1A (methionine activation) (46). Additional suppression of ALDOB,
CPS1, GOT1 and CTH further supports reduced amino acid
anabolism. These transcriptional changes collectively indicate that
FCP inhibits amino acid supply and protein synthesis pathways,
potentially contributing to translational downregulation and
metabolic reprogramming in SP-infected lung mesenchymal cells.
FCP treatment also downregulated a panel of
pro-apoptotic and cell cycle arrest-related genes in the p53
signaling pathway (Fig. 6D).
Notably, apoptotic genes, such as BAX, PMAIP1, CASP8, SIVA1
and THBS1, were suppressed, indicating reduced apoptotic
signaling (47). In addition,
genes associated with cell cycle inhibition, including CDKN1A
(p21), GADD45A, CCNG1, CCNG2, CDK1, CDK4 and CDK6, were
also downregulated, suggesting partial release from p53-induced
cell cycle arrest (47). By
contrast, FCP treatment upregulated CASP3 (a central
effector caspase), BCL2 (anti-apoptotic), CCND3 (G1/S
cyclin) and stress response mediators, such as GADD45B,
SESN1 and SESN3 (47). This phenomenon suggested a shift
toward adaptive stress recovery rather than p53-mediated apoptosis.
These changes collectively suggest that FCP inhibits excessive
p53-driven cytotoxicity while promoting moderate survival and
stress adaptation in SP-infected lung mesenchymal cells.
FCP activated nucleotide sugar
metabolism
FCP treatment significantly upregulated a set of
core genes involved in amino and nucleotide sugar metabolism and
nucleotide sugar biosynthesis, indicating enhanced flux through
hexosamine and UDP-sugar pathways (Fig. 6E and F). Notably, GFPT1, GFPT and
UAP1, which encode the rate-limiting enzymes for hexosamine
biosynthesis, were strongly induced, suggesting elevated UDP-GlcNAc
production (48). Concurrent
upregulation of GNE, MPI, PMM1, PMM2, GMPPA, GMPPB, PGM3 and
UGP2 further supported enhanced biosynthesis of diverse
nucleotide sugars, including UDP-GalNAc, GDP-mannose and
UDP-glucose (48). Genes essential
for sialic acid and GlcNAc-6-phosphate conversion, such as
NANS and GNPNAT1, were also elevated (49). These transcriptional changes
indicated that FCP stimulates nucleotide sugar biosynthetic
networks in SP-infected lung mesenchymal cells, potentially
facilitating glycosylation, membrane repair and metabolic
compensation under SP-induced stress.
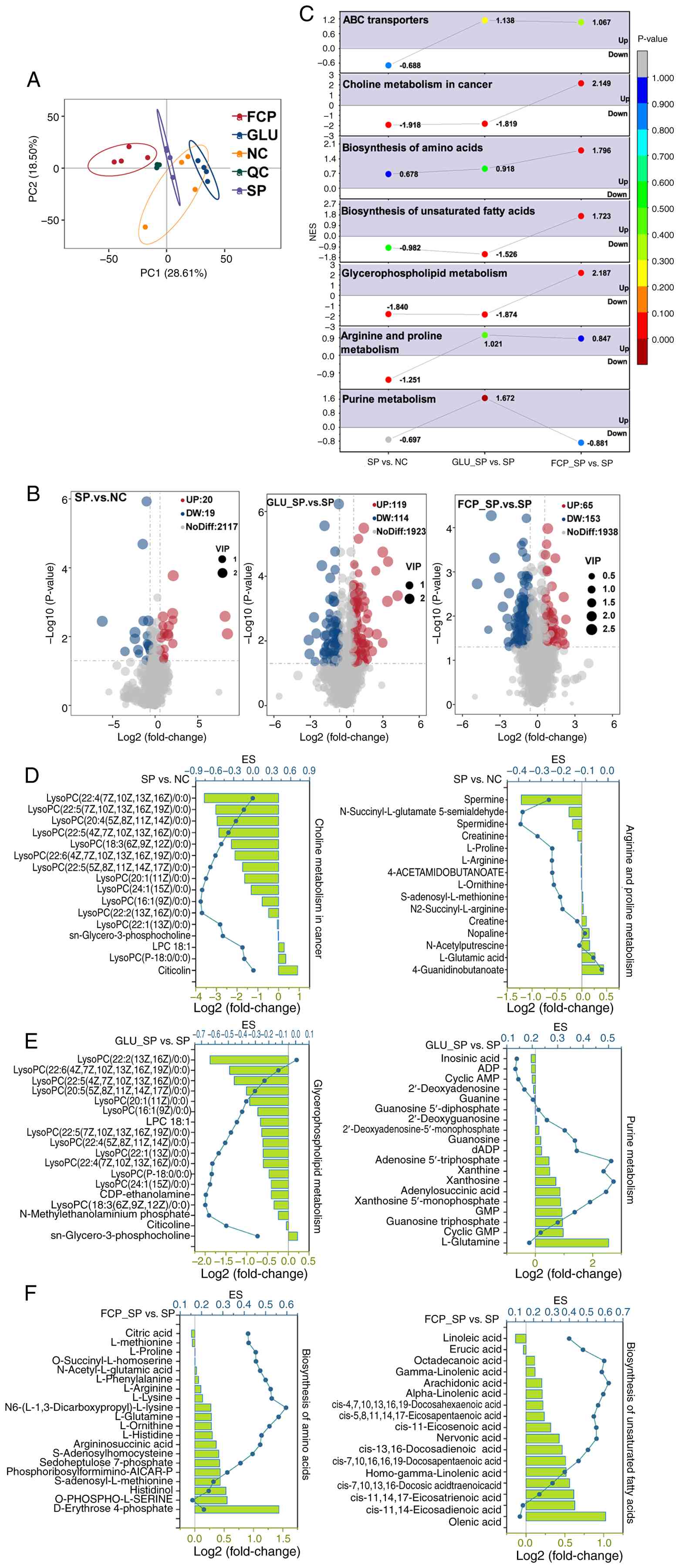
Metabolomic alterations
PCA analysis of metabolomic profiles (Fig. 7A) revealed clear separation among
SP, NC, GLU and FCP groups, indicating distinct metabolic states.
Notably, numerous metabolites were significantly altered in each
comparison (Fig. 7B). SP infection
induced significant elevation of lipids and lipid-like molecules,
including MG(0:0/i-13:0/0:0), MG(0:0/i-15:0/0:0) and
palmitoylcarnitine. By contrast, several amino acids and related
compounds, such as proline-arginine-leucine and
tyrosine-glutamine-phenylalanine, were strongly downregulated
(Table SIII).
GLU intervention (GLU_SP vs. SP; Table SIV) resulted in significant
upregulation of metabolites such as isoamyl isothiocyanate, lauroyl
diethanolamide and 4,5-dihydroxyhexanoic acid. Numerous amino acids
and peptides, including arginine-leucine-isoleucine,
l-lysyl-l-lysyl-l-lysine and tyrosine-glutamine-phenylalanine and
small oligopeptides, such as Ala-Leu-Leu-Thr, were also
significantly upregulated. By contrast, a broad range of lipids,
including lysophosphatidylcholine [22:2(13Z,16Z)/0:0] and
monoacylglycerol [MG(0:0/i-13:0/0:0)], were significantly
decreased.
The prominent upregulated metabolites in the FCP
intervention group (FCP_SP vs. SP; Table SV) included
cycloart-23-ene-3β,25-diol, ethyl linoleate and prolylproline,
suggesting enhanced lipid signaling and peptide metabolism.
Conversely, glutamylleucylarginine, N-benzoyl-D-arginine, PS
(20:4-OH (12S)/14:1) and homaxisterol B1 were among the most
significantly downregulated lipids and sterols.
Metabolite GSEA analysis
SP infection significantly downregulated
‘glycerophospholipid metabolism’, ‘choline metabolism in cancer’
and ‘arginine and proline metabolism’ (Fig. 7C), suggesting disrupted membrane
lipid turnover and amino acid-derived signaling (50,51).
GLU treatment significantly activated ‘purine metabolism’ and ‘ABC
transporters’ but did not affect ‘choline metabolism in cancer’,
‘glycerophospholipid metabolism’ and ‘biosynthesis of unsaturated
fatty acids’. By contrast, FCP treatment upregulated ‘choline
metabolism in cancer,’ ‘glycerophospholipid metabolism’ and
‘biosynthesis of amino acids’ and ‘biosynthesis of unsaturated
fatty acids’.
Notably, within the ‘choline metabolism in cancer’
pathway (Fig. 7D), a set of
SP-affected metabolites was identified, including LysoPC
(22:6/0:0), LysoPC (18:3/0:0), LysoPC (22:5/0:0) and LysoPC
(20:4/0:0) (Fig. 7D). The presence
of these polyunsaturated lysophosphatidylcholines denoted
SP-induced membrane remodeling and lipid depletion.
GLU treatment significantly elevated purine
metabolites (Fig. 7E), such as
cyclic GMP, GTP, GMP, xanthosine, xanthine, adenosine triphosphate,
adenylosuccinic acid and dADP, which reflect enhanced nucleotide
turnover and energy metabolism under SP infection stress (52). By contrast, FCP treatment increased
biosynthesis of amino acids (Fig.
7F) by upregulating sedoheptulose 7-phosphate,
S-adenosylhomocysteine, argininosuccinic acid, L-histidine,
L-ornithine, L-glutamine and N6-(L-1,3-dicarboxypropyl)-L-lysine,
which denoted strengthened amino acid regeneration and one-carbon
metabolism. In addition, FCP treatment enhanced biosynthesis of
unsaturated fatty acids, elevating nervonic acid, cis-11-eicosenoic
acid, EPA, DHA, alpha-linolenic acid and arachidonic acid. This
phenomenon suggested restored membrane lipid homeostasis and
anti-inflammatory lipid mediator production.
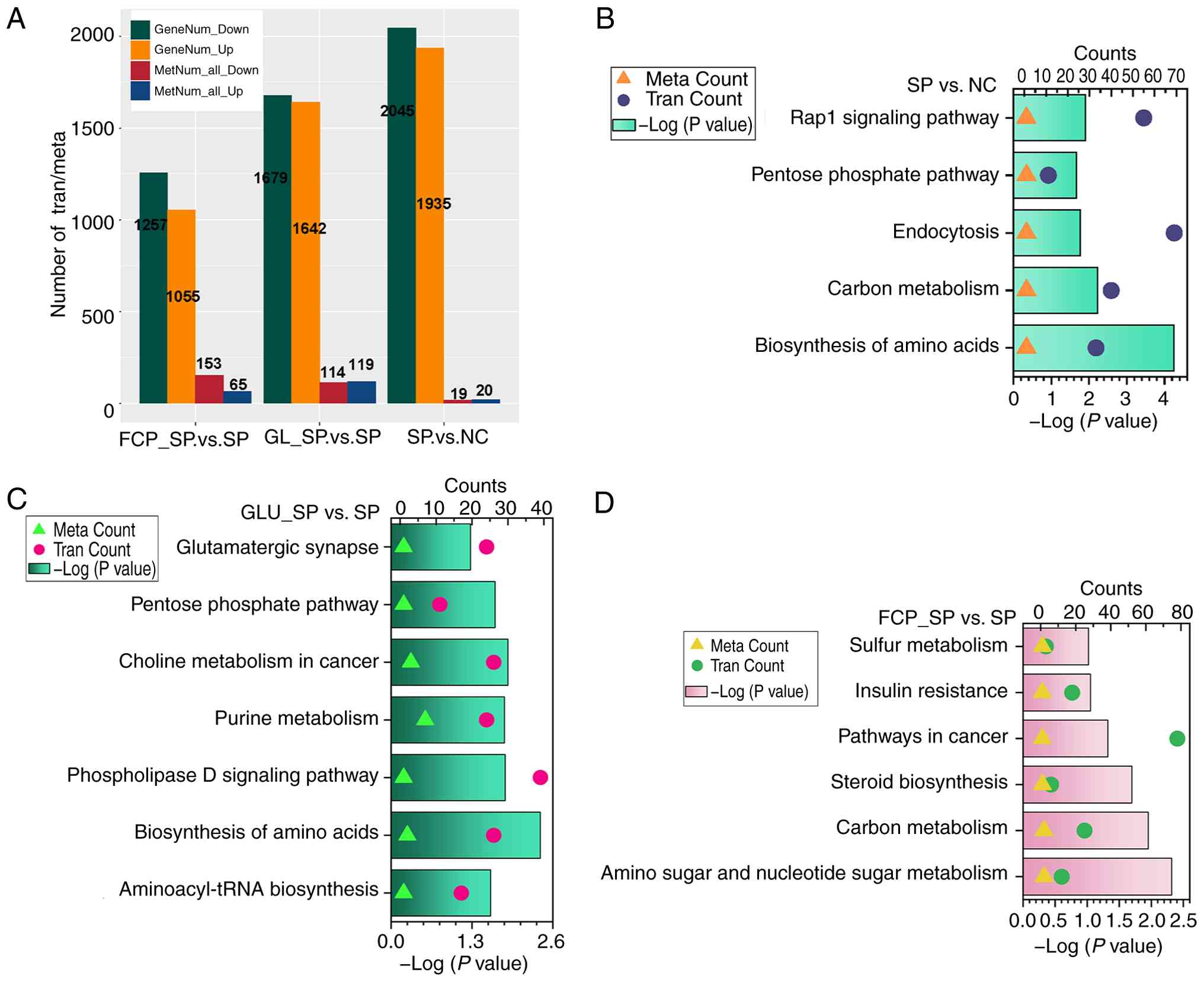
Integrated pathway analysis of
transcriptome and metabolome
The number of differential genes and metabolites
varied across comparisons, with SP infection causing the most
extensive changes (Fig. 8A).
Integrated enrichment analysis revealed that SP infection
significantly affected ‘biosynthesis of amino acids’, ‘carbon
metabolism’, ‘endocytosis’ and the ‘Rap1 signaling pathway’
(Fig. 8B). Notably, genes involved
in endocytic trafficking, including RAB11B, were strongly
upregulated (Fig. 4C), while
guanosine triphosphate (GTP), which is a crucial metabolite, was
significantly reduced (Table
SIII). This phenomenon reflected enhanced membrane recycling
activity driven by GTP-dependent GTPases under infection stress,
accompanied by energy depletion and disrupted purine metabolism,
resulting in intracellular GTP shortage despite increased demand
(53).
GLU enhanced nitrogen assimilation and
amino acid synthesis under SP infection
Integrated KEGG analysis revealed that GLU treatment
significantly modulated ‘aminoacyl-tRNA biosynthesis’,
‘biosynthesis of amino acids’, ‘phospholipase D signaling pathway’,
‘purine metabolism’ and ‘choline metabolism in cancer’ (Fig. 8C); the ‘biosynthesis of amino
acids’ pathway was prominently activated. The key upregulated genes
included ALDH18A1 (Δ¹-pyrroline-5-carboxylate synthase,
proline biosynthesis), IDH1 (isocitrate dehydrogenase, TCA
cycle and glutamate conversion), PYCR3
(pyrroline-5-carboxylate reductase, proline biosynthesis),
ENO1/ENO3, PFKL, ALDOA and GAPDH (glycolytic enzymes)
and ACO2 (aconitase, TCA cycle) (Table SVI). Metabolomic data further
confirmed significant elevation of pathway-relevant metabolites,
including L-histidine, L-lysine, L-proline, L-ornithine,
S-adenosylmethionine, argininosuccinic acid, L-arginine,
L-methionine, L-phenylalanine, L-asparagine, histaminol and citrate
(Table SVI). This phenomenon
supported the transcriptional activation of amino acid biosynthetic
machinery.
FCP intervention enhanced amino and
nucleotide sugar metabolism
FCP treatment significantly regulated multiple
metabolic pathways, including ‘amino sugar and nucleotide sugar
metabolism’, ‘carbon metabolism’, ‘steroid biosynthesis’ and
‘pathways in cancer’ (Fig. 8D);
among them, ‘amino sugar and nucleotide sugar metabolism’ was
prominently activated. Metabolomic analysis revealed an increase in
galactose 1-phosphate and a reduction in cyclic
N-acetyl-D-mannosamine, which indicated altered hexosamine flux
(Table SVI). Transcriptomic data
revealed robust upregulation of GFPT1, GFPT2 and
UAP1, which encode key rate-limiting enzymes in UDP-GlcNAc
biosynthesis (Table SVI).
Additionally, GNE, MPI, PMM1, PMM2, GMPPA, GMPPB, PGM3 and
UGP2 were elevated (Table
SVI), supporting enhanced production of multiple nucleotide
sugars, including UDP-GalNAc, GDP-mannose and UDP-glucose. These
changes suggested that FCP promotes nucleotide sugar
biosynthesis.
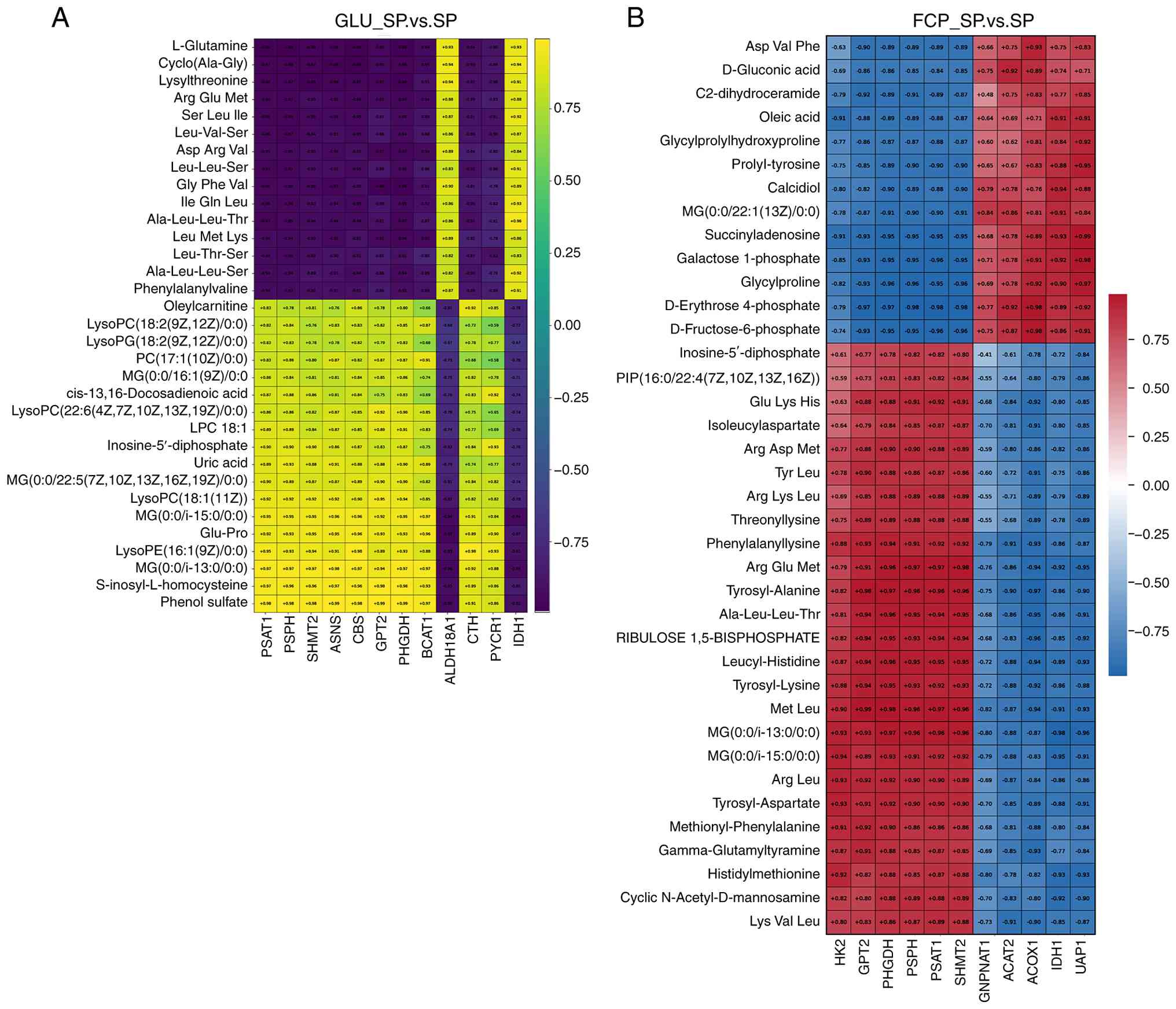
Correlation analysis between key genes
and metabolites in the GLU_SP vs. SP group
Pearson correlation analysis between key genes and
metabolites derived from the core pathways identified by joint KEGG
enrichment analysis was performed to characterize the functional
coupling between transcriptional changes and metabolite remodeling
induced by GLU supplementation (Fig.
9A). Notably, a structured correlation pattern was observed in
the GLU_SP vs. SP group. Genes belonging to the serine biosynthesis
and one-carbon metabolism axis, which included PHGDH,
PSAT1, PSPH, SHMT2 and ASNS, exhibited
consistently strong negative correlations with GLU and multiple
peptide-related metabolites (most |r|>0.95). In particular,
L-glutamine and a panel of di- and tripeptides, including
Ala-Leu-Leu-Thr, Gly-Phe-Val, Leu-Val-Ser and Ser-Leu-Ile,
clustered tightly and showed inverse correlations with the
PHGDH-PSAT1-PSPH-SHMT2 gene module. This pattern indicated
that GLU treatment enhanced the utilization of nitrogen substrates
and peptide-derived amino acids, rather than their accumulation,
suggesting accelerated amino acid flux into biosynthetic
pathways.
In parallel, a distinct metabolite cluster composed
of lysophospholipids (LysoPC, LysoPG), monoacylglycerols,
phosphatidylcholine species, acylcarnitines and unsaturated fatty
acid derivatives displayed strong positive correlations with the
PHGDH-PSAT1-PSPH-SHMT2 gene module. The coherence of these
lipid-associated metabolites in both hierarchical clustering and
correlation direction suggested that GLU treatment facilitates
concerted remodeling of membrane lipid composition and
mitochondrial lipid metabolism.
Notably, genes involved in transamination
(GPT2 and BCAT1), proline biosynthesis
(ALDH18A1 and PYCR1) and sulfur-containing amino acid
metabolism (CBS and CTH) exhibited correlation
patterns consistent with the central serine-one-carbon module.
These genes collectively formed a connected network regulating
amino acid interconversion, nitrogen redistribution and redox
balance, supporting the presence of a coordinated amino acid
metabolic reprogramming network in the GLU-treated cells.
The correlation architecture demonstrated that GLU
treatment induces a directional metabolic reorganization
characterized by active nitrogen consumption, enhanced amino acid
biosynthetic flux and coordinated lipid metabolic adaptation rather
than simply elevating metabolite abundance under SP infection
(Fig. 9).
Correlation analysis between key genes
and metabolites in the FCP_SP vs. SP group
Pearson correlation analysis between differential
metabolites and key genes derived from the core pathways identified
by joint KEGG enrichment analysis was performed to characterize the
association between transcriptional alterations and metabolic
changes induced by FCP treatment (Fig.
9B). Genes involved in the serine biosynthesis and one-carbon
metabolism pathway (PHGDH, PSAT1, PSPH and SHMT2)
exhibited notably consistent correlation directions across a broad
spectrum of metabolites. Notably, they had strong positive
correlations with multiple peptide-related metabolites, including
Ala-Leu-Leu-Thr, Met-Leu, Arg-Glu-Met, Leucyl-Histidine and
Lys-Val-Leu. By contrast, they were strongly negatively correlated
with several carbohydrate-related intermediates, such as
D-erythrose-4-phosphate, D-fructose-6-phosphate and
galactose-1-phosphate.
A similar correlation profile was observed for
GPT2, which displayed positive correlations with the
majority of peptide metabolites, including Met-Leu (and
Ala-Leu-Leu-Thr, but inverse correlations with sugar phosphate
intermediates, such as D-erythrose-4-phosphate and
D-fructose-6-phosphate. This coherence suggested that genes
involved in amino acid interconversion were tightly coupled to
peptide turnover and central carbon metabolism under FCP
treatment.
Notably, genes involved in amino and nucleotide
sugar metabolism, including UAP1 and GNPNAT1, had
distinct associations with carbohydrate-related metabolites.
UAP1 exhibited strong positive correlations with
galactose-1-phosphate, D-erythrose-4-phosphate and
D-fructose-6-phosphate. However, it exhibited negative correlations
with multiple peptide metabolites (such as Met-Leu;
Ala-Leu-Leu-Thr). These findings were consistent with the
transcriptional activation of the amino and nucleotide sugar
biosynthetic pathway in joint KEGG enrichment.
In addition, genes associated with central carbon
and lipid metabolism, including IDH1, ACAT2 and
ACOX1, were significantly and positively correlated with
both carbohydrate intermediates (e.g., D-fructose-6-phosphate) and
lipid-associated metabolites. These metabolites included
monoacylglycerols, oleic acid and C2-dihydroceramide, underscoring
the coordinated remodeling of energy and lipid metabolism in
response to FCP treatment.
Discussion
The present study investigated the molecular
responses of feline pulmonary interstitial cells (FCA-L2) following
SP infection and subsequent antibiotic treatment. Integrated
transcriptomic and metabolomic analyses revealed persistent cell
injury, sustained inflammatory activation and significant
remodeling of lipid metabolism and intracellular trafficking, which
provide mechanistic insights into post-infection
pathophysiology.
Notably, there was significant LDH release,
elevated IL-1β, TNF-α and IL-8 expression, increased MDA levels,
and disruption of junction proteins after 18 h despite antibiotic
administration at 4 h post-infection. These findings indicate that
bacterial clearance alone is insufficient to prevent secondary
damage, supporting the presence of a post-infectious inflammatory
syndrome. Notably, this phenomenon is consistent with clinical
reports in SP-related pneumonia, where excessive immune activation
and residual bacterial components, such as pneumolysin and cell
wall fragments, continue to trigger damage-associated signaling
even after pathogen eradication (54,55).
The reduction in junction proteins suggests compromised barrier
integrity due to cytokine-mediated cytoskeletal rearrangement and
oxidative injury. These observations reflect crucial features of
infection-associated acute lung injury, underscoring the need for
adjunctive interventions beyond antibiotics (56).
Notably, SP infection induced transcriptional and
metabolic remodeling of glycerophospholipid metabolism by
significantly upregulating key enzymes involved in de novo
phospholipid synthesis and hydrolysis, including GPAT3, AGPAT1,
LPIN1 and PLA2G6, which enhance membrane lipid turnover
(57,58). Metabolomic data confirmed
enrichment of the glycerophospholipid metabolism pathway. In
particular, there were reductions in multiple
lysophosphatidylcholine (LysoPC) species and increased levels of
CDP-ethanolamine and citicoline, which mediate PC biosynthesis.
These findings indicate active membrane remodeling, potentially
driven by stress and repair demands. Notably, the significant
depletion of multiple polyunsaturated LysoPC species, including
LysoPC 22:6/0:0, LysoPC 22:5/0:0 and LysoPC 20:4/0:0 potentially
compromises membrane stability and increases membrane fragility
(59). This finding is consistent
with the elevated LDH release and disruption of tight junction
proteins observed in SP-infected cells. LysoPC depletion may result
from increased utilization for membrane repair or altered PLA2
activity (60). Elevated
citicoline levels support compensatory PC synthesis to maintain
membrane integrity; however, excessive phospholipid turnover may
paradoxically destabilize membrane architecture under sustained
stress conditions. Alterations in phospholipids affect membrane
curvature, fluidity and raft formation, which modulate
host-pathogen interactions (61,62).
In the present study, enhanced membrane remodeling potentially
facilitates SP internalization or intracellular persistence by
disrupting immune receptor localization and amplifying inflammatory
signaling through lipid-derived mediators. Enhanced membrane
turnover may promote SP internalization or intracellular survival
by disrupting immune receptor localization. Excessive lipid
remodeling may also amplify inflammation via lipid mediators and
destabilization of membrane signaling platforms.
SP infection also activated endocytosis-related
pathways, further supporting membrane remodeling and persistent
intracellular stress (62).
Transcriptomic upregulation of RAB11B, RAB11FIP3, AP2A1 and
DNM1 indicated the activation of recycling endosomes and
clathrin-mediated endocytosis (63). Metabolomic analysis revealed
reduced intracellular GTP levels, suggesting high consumption
during vesicle trafficking, which is energetically costly and may
impose a substantial metabolic burden on infected cells (64). This finding was evidenced by the
increased oxidative stress and apoptosis observed in SP-treated
cells. Upregulated RAB11 signaling may enhance receptor recycling,
antigen processing and cytokine transport (65). The enhanced receptor recycling
capacity may contribute to prolonged surface availability of
cytokine receptors and pattern-recognition receptors, thereby
reinforcing inflammatory signaling. This phenomenon was observed in
the present study, as evidenced by the elevated levels of IL-1β,
TNF-α and IL-8. Notably, SP can exploit this machinery to evade
immune surveillance or promote intracellular persistence. These
alterations are closely linked to immune modulation through
potential activation of endosomal toll-like receptors (TLRs),
including TLR3 and TLR9, leading to cytokine
activation (61). Moreover,
excessive endosomal flux may overload the endosome-lysosome system
and impair autophagic homeostasis, leading to increased cellular
stress and apoptotic vulnerability (66,67).
This mechanism provides a plausible explanation for the elevated
apoptosis and cytotoxicity observed in the present study following
SP infection.
In the present study, GLU and FCP treatments
significantly alleviated SP-induced cellular injury and
inflammation. GLU and FCP reduced LDH release, suppressed
pro-inflammatory cytokine expression, restored tight junction
proteins and slowed apoptosis and cell death. They also enhanced
antioxidant capacity, which highlighted their dual cytoprotective
effect mediated by metabolic support and anti-inflammatory
modulation.
GLU treatment downregulated multiple cell cycle
regulators, including CDK1, CDK2, CCNE2 and MCM
family genes, and suppressed aminoacyl-tRNA synthetases, such as
EPRS1 and LARS1 (68,69). However, the amino acid
biosynthesis pathway was significantly upregulated at both
transcriptomic and metabolomic levels. Key upregulated genes
included ALDH18A1, PYCR3, IDH1, ACO2, ENO1, ENO3, PFKL,
ALDOA and GAPDH, translating to activation of proline
biosynthesis, glycolysis and the TCA cycle (69). Metabolomic profiling confirmed
increased levels of L-proline, L-lysine, L-ornithine,
S-adenosylmethionine, argininosuccinate, L-arginine and
L-histidine. This difference suggests that GLU induces the
preferential accumulation of free amino acids for stress defense,
rather than protein synthesis. Aminoacyl-tRNA synthetases are
essential components of the translational machinery; downregulation
indicates a deliberate reduction in protein synthesis, potentially
to prevent the accumulation of misfolded proteins and mitigate
endoplasmic reticulum stress under infection-induced damage
(70). In parallel, upregulated
amino acid biosynthesis supports antioxidant defenses and energy
metabolism. Notably, this metabolic shift directly correlates with
the decreased apoptosis and oxidative stress observed in the
present functional assays following GLU treatment. For example,
upregulation of ALDH18A1 and PYCR3 promotes proline synthesis,
contributing to redox buffering and mitochondrial protection
(71,72). IDH1 and ACO2 facilitate NADPH
production and maintain TCA cycle integrity. Elevated glycolytic
enzyme expression suggests increased glycolytic flux to support
biosynthetic demands and ATP supply (14). The enhancement of NADPH-dependent
redox buffering and ATP-generating pathways provides a mechanistic
explanation for the improved antioxidant capacity (SOD, GPX,
T-AOC/TOS) and the preservation of cell viability observed in
functional assays.
FCP treatment significantly activated amino and
nucleotide sugar metabolism. Transcriptomic analysis revealed
upregulation of GFPT1, GFPT2 and UAP1, indicating
increased synthesis of UDP-N-acetylglucosamine (UDP-GlcNAc)
(73). Upregulation of GNE,
MPI, PMM1, PMM2, GMPPA, GMPPB, PGM3 and UGP2 suggested
enhanced biosynthesis of multiple nucleotide sugars, including
UDP-GalNAc, GDP-mannose and UDP-glucose. Notably, genes involved in
sialic acid and GlcNAc-6-phosphate production (NANS and
GNPNAT1) were also elevated (74). These transcriptomic changes were
consistent with the metabolomic data, which exhibited increased
galactose 1-phosphate and decreased cyclic N-acetyl-D-mannosamine.
This response is closely associated with the pathogenic mechanism
of SP; SP expresses a range of glycosidases, such as neuraminidase
(NanA), β-galactosidase and β-N-acetylglucosaminidase, which cleave
host glycoproteins and glycolipids, thereby exposing hidden
epithelial receptors to facilitate bacterial adhesion (54,55,61,62).
NanA also degrades mucins, thereby reducing mucosal viscosity and
weakening the protective barrier (61). The activation of amino sugar and
nucleotide sugar metabolism by FCP potentially restored
glycosylation of membrane proteins and the structural integrity of
the glycocalyx, thereby counteracting bacterial deglycosylation and
promoting membrane repair. Mechanistically, FCP-derived peptides
potentially trigger the activation of nucleotide sugar biosynthesis
in a multifactorial manner. Small peptides can be transported into
cells through proton-coupled oligopeptide transporters, including
PEPT1 (SLC15A1) and PEPT2 (SLC15A2), which mediate
the uptake of di-/tripeptides and certain peptide-like substrates
(75-77).
Peptides and amino acids generated by intracellular peptidases upon
entry of the small peptides may enhance cellular nitrogen
availability and one-carbon metabolism, thereby supporting the
biosynthetic demand required for UDP-sugar production (78,79).
In parallel, peptide uptake and/or amino acid sensing could
activate nutrient-responsive signaling, such as mTOR/ATF4-related
adaptive programs (80,81), which coordinate anabolic pathways
under stress and may contribute to the observed upregulation of
GFPT1/2 and UAP1. Functionally, restoration of membrane
glycosylation stabilizes surface proteins and junctional complexes.
This phenomenon was exemplified in the present study by the
recovery of tight junction protein expression and reduced LDH
leakage following FCP treatment. FCP also enhanced amino acid and
polyunsaturated fatty acid (PUFA) metabolism. Metabolomic analysis
in the present study revealed elevated levels of key intermediates
in amino acid biosynthesis, including L-histidine, L-arginine,
L-glutamine, L-proline, L-lysine and S-adenosylmethionine.
Glycolytic and pentose phosphate pathway intermediates, such as
sedoheptulose 7-phosphate and erythrose 4-phosphate, were also
elevated. In parallel, there was an increase in the levels of
multiple long-chain PUFAs, including alpha-linolenic acid,
eicosapentaenoic acid, docosahexaenoic acid and nervonic acid.
Upregulation of amino acid and PUFA metabolism functionally
synergizes with nucleotide sugar biosynthesis. Specifically, the
accumulation of proline and other amino acids provides essential
precursors for cellular repair and redox homeostasis, while the
elevated long-chain PUFAs (e.g., EPA and DHA) act as
anti-inflammatory mediators to counteract SP-induced cytokine
production and membrane oxidative damage (71,72,82-84).
The coordinated metabolic reprogramming provides a mechanistic
explanation for the observed attenuation of inflammatory cytokine
production, reduced oxidative stress and improved cell survival
following FCP intervention. The coordinated metabolic programs
contributed to membrane structure repair, glycan renewal and
cellular recovery from infection-induced stress.
Alveolar epithelial cells are the primary sites of
SP adhesion and barrier disruption during the acute phase of
infection (85-87).
However, increasing evidence indicates that the resolution of lung
injury involves active participation of pulmonary interstitial
fibroblast populations during the post-infectious repair phase,
partly through paracrine signaling that supports alveolar and
parenchymal regeneration (88-90).
Mesenchymal cells contribute to tissue recovery by regulating ECM
remodeling, resolving inflammatory responses and re-establishing a
microenvironment conducive to epithelial regeneration following
bacterial clearance (25). In this
context, mesenchymal cells function as key coordinators of lung
repair rather than direct targets of bacterial invasion. The use of
mesenchymal cells in the present study is thus particularly
relevant for investigating reparative mechanisms that operate after
infection-induced damage, rather than the initial host-pathogen
interaction. The protective effects seen in mesenchymal cells in
the present study may influence epithelial recovery in vivo.
Mesenchymal cells influence epithelial repair through paracrine
signaling, including the secretion of growth factors, cytokines and
matrix-regulating molecules that promote epithelial cell
proliferation, migration and restoration of barrier integrity
(88-93).
The attenuation of inflammation and oxidative stress, along with
the preservation of junction-associated proteins observed in the
present study, suggests that the intervention may contribute to the
formation of a reparative microenvironment that indirectly supports
epithelial regeneration following pneumococcal injury.
Notably, the concentrations of GLU (40 mM) and FCP
(500 µg/ml) were determined via standardized in vitro
screenings to achieve an optimal balance between non-cytotoxicity
and maximal reparative efficacy. These pharmacological doses are
independent of systemic physiological plasma benchmarks because the
metabolic and structural requirements for cellular repair in an
in vitro environment represent a biological dimension
distinct from systemic homeostasis. Nevertheless, these doses
provide a theoretical foundation for targeted clinical strategies,
such as localized pulmonary nebulization and inhalation therapy.
However, it must be acknowledged that the current in vitro
monoculture model lacks the inherent complexity of the lung
microenvironment, which involves dynamic interactions between
various immune cells, including alveolar macrophages and
neutrophils and the three-dimensional ECM. These factors could
markedly influence the observed reparative effects in an in
vivo setting. For instance, GLU is a critical metabolic fuel
for macrophages (52). Its
supplementation could support fibroblast-mediated repair and
modulate macrophage polarization toward a pro-resolving M2
phenotype, potentially creating a synergistic effect that
accelerates tissue clearance and healing (94,95).
Similarly, FCP may interact with the native ECM architecture to
further enhance structural integrity (19). The simplified model established in
the present study essentially isolates and delineates the specific
metabolic and transcriptomic reprogramming within pulmonary
interstitial fibroblasts. However, future studies employing 3D
organoid models or in vivo animal infection trials should be
conducted to extensively capture the multicellular synergy during
the post-antibiotic lung recovery phase.
Despite the numerous insightful outputs of the
present study, it was limited by several factors. The use of a
mesenchymal cell model did not directly capture the initial
epithelial-pathogen interactions that occurred during SP infection.
The findings of the present study provide mechanistic insights into
stromal-mediated reparative responses following infection-induced
injury. However, the intercellular crosstalk and
epithelial-specific responses under infectious conditions should be
validated through future studies using epithelial cells or
mesenchymal-epithelial co-culture systems. In addition, a DOX-only
group was not included in the transcriptomic and metabolomic
analyses. The experimental contrasts were designed to characterize
molecular alterations associated with the post-infectious,
post-antibiotic state (SP + Dox), and their modulation by
nutritional interventions. Future studies incorporating a Dox-only
control will be valuable for fully disentangling
antibiotic-associated molecular effects from post-infectious host
responses.
In conclusion, SP induced sustained cell injury and
inflammation, accompanied by lipid remodeling, endocytic activation
and metabolic imbalance in FCA-L2 cells. However, SP-induced damage
was significantly alleviated by both GLU and FCP through
synergistic metabolic and anti-inflammatory effects. GLU
reprogrammed amino acid metabolism and redox homeostasis, whereas
FCP restored glycosylation and membrane integrity by enhancing
UDP-sugar, amino acid and PUFA metabolism. These results of the
present study underscore the importance of targeting host metabolic
and structural pathways to mitigate SP-related lung injury. They
also support the development of adjunctive therapies beyond
antibiotics.
Supplementary Material
Molecular weight distribution of fish
collagen peptide.
Amino acid composition of fully
hydrolysed fish collagen peptide.
Significantly altered metabolites
between SP and NC groUps.
Significantly altered metabolites
between GLU_SP and SP groUps.
Significantly altered metabolites
between FCP_SP and SP groups.
Key genes and metabolites annotated to
enriched pathways based on integrated transcriptomic and
metabolomic analysis.
Acknowledgements
The authors would like to acknowledge the Global
Pet Cell Resource Center PETCC for providing the cells and culture
technology support.
Funding
Funding: No funding was received.
Availability of data and materials
The raw RNA-sequencing data have been deposited in
the NCBI Sequence Read Archive (SRA) under BioProject accession
number PRJNA1416572. The raw metabolomics data have been deposited
in the Figshare open-access repository and are publicly available
under the DOI 10.6084/m9.figshare.31267111 (https://doi.org/10.6084/m9.figshare.31267111).
All other data generated in the present study may be requested from
the corresponding author.
Authors' contributions
HB contributed to the investigation, methodology
design, validation of experiments and drafting of the original
manuscript. TL contributed to data curation, formal analysis,
software processing and data visualization. HW contributed to the
investigation and validation. HJ contributed to conceptualization,
project administration and provision of resources. YL contributed
to resources and conceptualization of the study. ZW contributed to
conceptualization, supervision and critical review and editing of
the manuscript. All authors have read and approved the final
version of the manuscript. HB and ZW confirm the authenticity of
all the raw data.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Li Y, Jiang Y, Zhang H, Zhang J, Ma J,
Yang Z, Qiu M and Wang J: Research on acute lung injury
inflammatory network. Int J Clin Pharmacol Ther. 61:394–403.
2023.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Lanks CW, Musani AI and Hsia DW:
Community-acquired pneumonia and hospital-acquired pneumonia. Med
Clin North Am. 103:487–501. 2019.PubMed/NCBI View Article : Google Scholar
|
|
3
|
de Benedictis FM, Kerem E, Chang AB, Colin
AA, Zar HJ and Bush A: Complicated pneumonia in children. Lancet.
396:786–798. 2020.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Liu YN, Zhang YF, Xu Q, Qiu Y, Lu QB, Wang
T, Zhang XA, Lin SH, Lv CL, Jiang BG, et al: Infection and
co-infection patterns of community-acquired pneumonia in patients
of different ages in China from 2009 to 2020: A national
surveillance study. Lancet Microbe. 4:e330–e339. 2023.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Dear JD: Bacterial pneumonia in dogs and
cats: An update. Vet Clin North Am Small Anim Pract. 50:447–465.
2020.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Nguyen N, Xu S, Lam TYW, Liao W, Wong WSF
and Ge R: ISM1 suppresses LPS-induced acute lung injury and
post-injury lung fibrosis in mice. Mol Med. 28(72)2022.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Wang Y, Wang L, Ma S, Cheng L and Yu G:
Repair and regeneration of the alveolar epithelium in lung injury.
FASEB J. 38(e23612)2024.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Huang X, Zhang X, Machireddy N, Evans CE,
Trewartha SD, Hu G, Fang Y, Mutlu GM, Wu D and Zhao YY: Endothelial
FoxM1 reactivates aging-impaired endothelial regeneration for
vascular repair and resolution of inflammatory lung injury. Sci
Transl Med. 15(eabm5755)2023.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Morris SB, Ocadiz-Ruiz R, Asai N,
Malinczak CA, Rasky AJ, Lombardo GK, Velarde EM, Ptaschinski C,
Zemans RL, Lukacs NW and Fonseca W: Long-term alterations in lung
epithelial cells after EL-RSV infection exacerbate allergic
responses through IL-1β-induced pathways. Mucosal Immunol.
17:1072–1088. 2024.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Lucas R, Hadizamani Y, Gonzales J,
Gorshkov B, Bodmer T, Berthiaume Y, Moehrlen U, Lode H, Huwer H,
Hudel M, et al: Impact of bacterial toxins in the lungs. Toxins
(Basel). 12(223)2020.PubMed/NCBI View Article : Google Scholar
|
|
11
|
El Agha E and Thannickal VJ: The lung
mesenchyme in development, regeneration, and fibrosis. J Clin
Invest. 133(e170498)2023.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Zhang J, Wang T, Wang Y, Li Y, Wang L,
Wang J, Miao Y, Xu F and Yao Y: Bacterial pneumonia induces
senescence in resident alveolar macrophages that are outcompeted by
monocytes. Cell Rep. 44(115571)2025.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Wang S, Li X, Ma Q, Wang Q, Wu J, Yu H, Li
K, Li Y, Wang J, Zhang Q, et al: Glutamine metabolism is required
for alveolar regeneration during lung injury. Biomolecules.
12(728)2022.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Shaghaghi H, Para R, Tran C, Roman J,
Ojeda-Lassalle Y, Sun J, Romero F and Summer R: Glutamine restores
mitochondrial respiration in bleomycin-injured epithelial cells.
Free Radic Biol Med. 176:335–344. 2021.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Arribas-López E, Zand N, Ojo O, Snowden MJ
and Kochhar T: The effect of amino acids on wound healing: A
systematic review and meta-analysis on arginine and glutamine.
Nutrients. 13(2498)2021.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Stehle P, Ellger B, Kojic D, Feuersenger
A, Schneid C, Stover J, Scheiner D and Westphal M: Glutamine
dipeptide-supplemented parenteral nutrition improves the clinical
outcomes of critically ill patients: A systematic evaluation of
randomised controlled trials. Clin Nutr ESPEN. 17:75–85.
2017.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Kim Y, Lee JO, Lee JM, Lee MH, Kim HM,
Chung HC, Kim DU, Lee JH and Kim BJ: Low molecular weight collagen
peptide (LMWCP) promotes hair growth by activating the
Wnt/GSK-3β/β-catenin signaling pathway. J Microbiol Biotechnol.
34:17–28. 2024.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Bayan R, Tauseef I, Hussain M, Ahmed MS,
Haider A, Khalil AA, Islam SU and Subhan F: Fish collagen peptides'
modulating effect on human skin microbiota against pathogenic
Staphylococcus aureus. Future Microbiol. 18:795–807.
2023.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Czajka A, Kania EM, Genovese L, Corbo A,
Merone G, Luci C and Sibilla S: Daily oral supplementation with
collagen peptides combined with vitamins and other bioactive
compounds improves skin elasticity and has a beneficial effect on
joint and general wellbeing. Nutr Res. 57:97–108. 2018.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Rahabi M, Salon M, Bruno-Bonnet C, Prat M,
Jacquemin G, Benmoussa K, Alaeddine M, Parny M, Bernad J, Bertrand
B, et al: Bioactive fish collagen peptides weaken intestinal
inflammation by orienting colonic macrophages phenotype through
mannose receptor activation. Eur J Nutr. 61:2051–2066.
2022.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Hwang SB, Park HJ and Lee BH:
Hair-growth-promoting effects of the fish collagen peptide in human
dermal papilla cells and C57BL/6 mice modulating Wnt/β-catenin and
BMP signaling pathways. Int J Mol Sci. 23(11904)2022.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Ligresti G, Raslan AA, Hong J, Caporarello
N, Confalonieri M and Huang SK: Mesenchymal cells in the lung:
Evolving concepts and their role in fibrosis. Gene.
859(147142)2023.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Andersson-Sjöland A, Nihlberg K, Eriksson
L, Bjermer L and Westergren-Thorsson G: Fibrocytes and the tissue
niche in lung repair. Respir Res. 12(76)2011.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Doherty DF, Roets L and Krasnodembskaya
AD: The role of lung resident mesenchymal stromal cells in the
pathogenesis and repair of chronic lung disease. Stem Cells.
41:431–443. 2023.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Sveiven SN and Nordgren TM: Lung-resident
mesenchymal stromal cells are tissue-specific regulators of lung
homeostasis. Am J Physiol Lung Cell Mol Physiol. 319:L197–Ll210.
2020.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Thomas K, Zondler L, Ludwig N, Kardell M,
Lüneburg C, Henke K, Mersmann S, Margraf A, Spieker T, Tekath T, et
al: Glutamine prevents acute kidney injury by modulating oxidative
stress and apoptosis in tubular epithelial cells. JCI Insight.
7(e163161)2022.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Zhao Y, Albrecht E, Stange K, Li Z,
Schregel J, Sciascia QL, Metges CC and Maak S: Glutamine
supplementation stimulates cell proliferation in skeletal muscle
and cultivated myogenic cells of low birth weight piglets. Sci Rep.
11(13432)2021.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Huang CY, Chen JK and Kuo WT: Glutamine
induces remodeling of tight junctions in Caco-2 colorectal cancer
cell. Med Oncol. 40(32)2022.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Yang X, Zhong Y, Wang D and Lu Z: A simple
colorimetric method for viable bacteria detection based on cell
counting Kit-8. Anal Methods. 13:5211–5215. 2021.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Galindo-Feria AS, Notarnicola A, Lundberg
IE and Horuluoglu B: Aminoacyl-tRNA synthetases: On anti-synthetase
syndrome and beyond. Front Immunol. 13(866087)2022.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Rubio Gomez MA and Ibba M: Aminoacyl-tRNA
synthetases. RNA. 26:910–936. 2020.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Horgan CP and McCaffrey MW: The dynamic
Rab11-FIPs. Biochem Soc Trans. 37:1032–1036. 2009.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Wilson B, Flett C, Gemperle J, Lawless C,
Hartshorn M, Hinde E, Harrison T, Chastney M, Taylor S, Allen J, et
al: Proximity labelling identifies pro-migratory endocytic
recycling cargo and machinery of the Rab4 and Rab11 families. J
Cell Sci. 136(jcs260468)2023.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Horgan CP, Hanscom SR, Jolly RS, Futter CE
and McCaffrey MW: Rab11-FIP3 binds dynein light intermediate chain
2 and its overexpression fragments the Golgi complex. Biochem
Biophys Res Commun. 394:387–392. 2010.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Shindou H and Shimizu T:
Acyl-CoA:lysophospholipid acyltransferases. J Biol Chem. 284:1–5.
2009.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Gibellini F and Smith TK: The Kennedy
pathway-de novo synthesis of phosphatidylethanolamine and
phosphatidylcholine. IUBMB Life. 62:414–428. 2010.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Mérida I, Avila-Flores A and Merino E:
Diacylglycerol kinases: At the hub of cell signalling. Biochem J.
409:1–18. 2008.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Yu TCY, Zhao L, Sze KMF, Tian L, Zhang VX,
Lee E, Kam CS, Lee JMF, Ho DWH, Tsui YM, et al: GPD1L supports
glycerol-3-phosphate and triacylglycerol synthesis and promotes
tumor progression in HCC. Hepatology. 83:735–752. 2026.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Malumbres M and Barbacid M: Cell cycle,
CDKs and cancer: A changing paradigm. Nat Rev Cancer. 9:153–166.
2009.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Fragkos M, Ganier O, Coulombe P and
Méchali M: DNA replication origin activation in space and time. Nat
Rev Mol Cell Biol. 16:360–374. 2015.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Liu X and Winey M: The MPS1 family of
protein kinases. Annu Rev Biochem. 81:561–585. 2012.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Mannini L, Cucco F, Quarantotti V, Amato
C, Tinti M, Tana L, Frattini A, Delia D, Krantz ID, Jessberger R
and Musio A: SMC1B is present in mammalian somatic cells and
interacts with mitotic cohesin proteins. Sci Rep.
5(18472)2015.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Kroef V, Ruegenberg S, Horn M, Allmeroth
K, Ebert L, Bozkus S, Miethe S, Elling U, Schermer B, Baumann U and
Denzel MS: GFPT2/GFAT2 and AMDHD2 act in tandem to control the
hexosamine pathway. Elife. 11(e69223)2022.PubMed/NCBI View Article : Google Scholar
|
|
44
|
Wilson JE: Isozymes of mammalian
hexokinase: Structure, subcellular localization and metabolic
function. J Exp Biol. 206:2049–2057. 2003.PubMed/NCBI View Article : Google Scholar
|
|
45
|
Rodríguez-Díaz J and Yebra MJ: Enhanced
UDP-glucose and UDP-galactose by homologous overexpression of
UDP-glucose pyrophosphorylase in Lactobacillus casei. J Biotechnol.
154:212–215. 2011.PubMed/NCBI View Article : Google Scholar
|
|
46
|
Chen J, Cui L, Lu S and Xu S: Amino acid
metabolism in tumor biology and therapy. Cell Death Dis.
15(42)2024.PubMed/NCBI View Article : Google Scholar
|
|
47
|
Riley T, Sontag E, Chen P and Levine A:
Transcriptional control of human p53-regulated genes. Nat Rev Mol
Cell Biol. 9:402–412. 2008.PubMed/NCBI View Article : Google Scholar
|
|
48
|
Yang X and Qian K: Protein
O-GlcNAcylation: emerging mechanisms and functions. Nat Rev Mol
Cell Biol. 18:452–465. 2017.PubMed/NCBI View Article : Google Scholar
|
|
49
|
Morales MM and Pratt MR: The
post-translational modification O-GlcNAc is a sensor and regulator
of metabolism. Open Biol. 14(240209)2024.PubMed/NCBI View Article : Google Scholar
|
|
50
|
Wang B and Tontonoz P: Phospholipid
remodeling in physiology and disease. Annu Rev Physiol. 81:165–188.
2019.PubMed/NCBI View Article : Google Scholar
|
|
51
|
Morris SM Jr: Arginine metabolism
revisited. J Nutr. 146:2579S–2586S. 2016.PubMed/NCBI View Article : Google Scholar
|
|
52
|
Cruzat V, Macedo Rogero M, Noel Keane K,
Curi R and Newsholme P: Glutamine: Metabolism and immune function,
supplementation and clinical translation. Nutrients.
10(1564)2018.PubMed/NCBI View Article : Google Scholar
|
|
53
|
Prekeris R: Rabs, Rips, FIPs, and
endocytic membrane traffic. ScientificWorldJournal. 3:870–880.
2003.PubMed/NCBI View Article : Google Scholar
|
|
54
|
Loughran AJ, Orihuela CJ and Tuomanen EI:
Streptococcus pneumoniae: Invasion and inflammation.
Microbiol Spectr 7: 10.1128/microbiolspec.gpp3-0004-2018, 2019.
|
|
55
|
Bogaert D, De Groot R and Hermans PWM:
Streptococcus pneumoniae colonisation: The key to
pneumococcal disease. Lancet Infect Dis. 4:144–154. 2004.PubMed/NCBI View Article : Google Scholar
|
|
56
|
Ceccato A, Ferrer M, Barbeta E and Torres
A: Adjunctive therapies for community-acquired pneumonia. Clin
Chest Med. 39:753–764. 2018.PubMed/NCBI View Article : Google Scholar
|
|
57
|
Gonzalez-Baro MR and Coleman RA:
Mitochondrial acyltransferases and glycerophospholipid metabolism.
Biochim Biophys Acta Mol Cell Biol Lipids. 1862:49–55.
2017.PubMed/NCBI View Article : Google Scholar
|
|
58
|
Bradley RM, Marvyn PM, Aristizabal Henao
JJ, Mardian EB, George S, Aucoin MG, Stark KD and Duncan RE:
Acylglycerophosphate acyltransferase 4 (AGPAT4) is a mitochondrial
lysophosphatidic acid acyltransferase that regulates brain
phosphatidylcholine, phosphatidylethanolamine, and
phosphatidylinositol levels. Biochim Biophys Acta. 1851:1566–1576.
2015.PubMed/NCBI View Article : Google Scholar
|
|
59
|
Law SH, Chan ML, Marathe GK, Parveen F,
Chen CH and Ke LY: An Updated review of lysophosphatidylcholine
metabolism in human diseases. Int J Mol Sci.
20(1149)2019.PubMed/NCBI View Article : Google Scholar
|
|
60
|
Wang LM, Zhang WL, Lyu N, Suo YR, Yang L,
Yu B and Jiang XJ: Research advance of Chinese medicine in treating
atherosclerosis: Focus on lipoprotein-associated phospholipase A2.
Chin J Integr Med. 30:277–288. 2024.PubMed/NCBI View Article : Google Scholar
|
|
61
|
Subramanian K, Henriques-Normark B and
Normark S: Emerging concepts in the pathogenesis of the
Streptococcus pneumoniae: From nasopharyngeal colonizer to
intracellular pathogen. Cell Microbiol. 21(e13077)2019.PubMed/NCBI View Article : Google Scholar
|
|
62
|
Ogawa M, Shizukuishi S, Akeda Y and
Ohnishi M: Molecular mechanism of Streptococcus
pneumoniae-targeting xenophagy recognition and evasion:
Reinterpretation of pneumococci as intracellular bacteria.
Microbiol Immunol. 67:224–227. 2023.PubMed/NCBI View Article : Google Scholar
|
|
63
|
Xie X, Wang Y, Yu D, Xie R, Liu Z and
Huang B: DNM1, a dynamin-related protein that contributes to
endocytosis and peroxisome fission, is required for the vegetative
growth, sporulation, and virulence of Metarhizium robertsii. Appl
Environ Microbiol. 86:e01217–20. 2020.PubMed/NCBI View Article : Google Scholar
|
|
64
|
Martínez RAS, Pinky PD, Harlan BA and
Brewer GJ: GTP energy dependence of endocytosis and autophagy in
the aging brain and Alzheimer's disease. Geroscience. 45:757–780.
2023.PubMed/NCBI View Article : Google Scholar
|
|
65
|
Xu J, Liang Y, Li N, Dang S, Jiang A, Liu
Y, Guo Y, Yang X, Yuan Y, Zhang X, et al: Clathrin-associated
carriers enable recycling through a kiss-and-run mechanism. Nat
Cell Biol. 26:1652–1668. 2024.PubMed/NCBI View Article : Google Scholar
|
|
66
|
Settembre C, Fraldi A, Medina DL and
Ballabio A: Signals from the lysosome: A control centre for
cellular clearance and energy metabolism. Nat Rev Mol Cell Biol.
14:283–296. 2013.PubMed/NCBI View Article : Google Scholar
|
|
67
|
Okahashi N, Nakata M, Kuwata H and
Kawabata S: Streptococcus oralis induces lysosomal impairment of
macrophages via bacterial hydrogen peroxide. Infect Immun.
84:2042–2050. 2016.PubMed/NCBI View Article : Google Scholar
|
|
68
|
Wang Z: Cell cycle progression and
synchronization: An overview. Methods Mol Biol. 2579:3–23.
2022.PubMed/NCBI View Article : Google Scholar
|
|
69
|
Takénaka A and Moras D: Correlation
between equi-partition of aminoacyl-tRNA synthetases and amino-acid
biosynthesis pathways. Nucleic Acids Res. 48:3277–3285.
2020.PubMed/NCBI View Article : Google Scholar
|
|
70
|
Walter P and Ron D: The unfolded protein
response: From stress pathway to homeostatic regulation. Science.
334:1081–1086. 2011.PubMed/NCBI View Article : Google Scholar
|
|
71
|
Geng P, Qin W and Xu G: Proline metabolism
in cancer. Amino Acids. 53:1769–1777. 2021.PubMed/NCBI View Article : Google Scholar
|
|
72
|
Krishnan N, Dickman MB and Becker DF:
Proline modulates the intracellular redox environment and protects
mammalian cells against oxidative stress. Free Radic Biol Med.
44:671–681. 2008.PubMed/NCBI View Article : Google Scholar
|
|
73
|
Cao J, Li M, Liu K, Shi X, Sui N, Yao Y,
Wang X, Li S, Tian Y, Tan S, et al: Oxidative phosphorylation
safeguards pluripotency via UDP-N-acetylglucosamine. Protein Cell.
14:376–381. 2023.PubMed/NCBI View Article : Google Scholar
|
|
74
|
Nishikawa A, Karita S and Umekawa M: Ngk1
kinase-mediated N-acetylglucosamine metabolism promotes UDP-GlcNAc
biosynthesis in Saccharomyces cerevisiae. FEBS Lett. 598:1644–1654.
2024.PubMed/NCBI View Article : Google Scholar
|
|
75
|
Alteheld B, Evans ME, Gu LH, Ganapathy V,
Leibach FH, Jones DP and Ziegler TR: Alanylglutamine dipeptide and
growth hormone maintain PepT1-mediated transport in oxidatively
stressed Caco-2 cells. J Nutr. 135:19–26. 2005.PubMed/NCBI View Article : Google Scholar
|
|
76
|
Xu G, Zhao W and Yu Z: Intestinal
epithelial transport of bioactive di/tripeptides through PepT1:
Molecular mechanism and influencing factors. Food Chem.
496(146851)2025.PubMed/NCBI View Article : Google Scholar
|
|
77
|
Spanier B and Rohm F: Proton coupled
oligopeptide transporter 1 (PepT1) function, regulation, and
influence on the intestinal homeostasis. Compr Physiol. 8:843–869.
2018.PubMed/NCBI View Article : Google Scholar
|
|
78
|
Paneque A, Fortus H, Zheng J, Werlen G and
Jacinto E: The hexosamine biosynthesis pathway: Regulation and
function. Genes (Basel). 14(933)2023.PubMed/NCBI View Article : Google Scholar
|
|
79
|
Zhu Q, Cheng X, Cheng Y, Chen J, Xu H, Gao
Y, Duan X, Ji J, Li X and Yi W: O-GlcNAcylation regulates the
methionine cycle to promote pluripotency of stem cells. Proc Natl
Acad Sci USA. 117:7755–7763. 2020.PubMed/NCBI View Article : Google Scholar
|
|
80
|
Chen CL, Hsu SC, Ann DK, Yen Y and Kung
HJ: Arginine signaling and cancer metabolism. Cancers (Basel).
13(3541)2021.PubMed/NCBI View Article : Google Scholar
|
|
81
|
Wyant GA, Abu-Remaileh M, Wolfson RL, Chen
WW, Freinkman E, Danai LV, Vander Heiden MG and Sabatini DM: mTORC1
activator SLC38A9 is required to efflux essential amino acids from
lysosomes and use protein as a nutrient. Cell. 171:642–654.e12.
2017.PubMed/NCBI View Article : Google Scholar
|
|
82
|
Wu Z, Hou Y, Dai Z, Hu CA and Wu G:
Metabolism, nutrition, and redox signaling of hydroxyproline.
Antioxid Redox Signal. 30:674–682. 2019.PubMed/NCBI View Article : Google Scholar
|
|
83
|
Swanson D, Block R and Mousa SA: Omega-3
fatty acids EPA and DHA: Health benefits throughout life. Adv Nutr.
3:1–7. 2012.PubMed/NCBI View Article : Google Scholar
|
|
84
|
Kousparou C, Fyrilla M, Stephanou A and
Patrikios I: DHA/EPA (Omega-3) and LA/GLA (Omega-6) as bioactive
molecules in neurodegenerative diseases. Int J Mol Sci.
24(10717)2023.PubMed/NCBI View Article : Google Scholar
|
|
85
|
Kostadinova E, Chaput C, Gutbier B,
Lippmann J, Sander LE, Mitchell TJ, Suttorp N, Witzenrath M and
Opitz B: NLRP3 protects alveolar barrier integrity by an
inflammasome-independent increase of epithelial cell adherence. Sci
Rep. 6(30943)2016.PubMed/NCBI View Article : Google Scholar
|
|
86
|
Cui L, Yang R, Huo D, Li L, Qu X, Wang J
and Wang X, Liu H, Chen H and Wang X: Streptococcus
pneumoniae extracellular vesicles aggravate alveolar epithelial
barrier disruption via autophagic degradation of OCLN (occludin).
Autophagy. 20:1577–1596. 2024.PubMed/NCBI View Article : Google Scholar
|
|
87
|
Sura T, Gering V, Cammann C, Hammerschmidt
S, Maaß S, Seifert U and Becher D: Streptococcus pneumoniae
and influenza A virus co-infection induces altered
polyubiquitination in A549 cells. Front Cell Infect Microbiol.
12(817532)2022.PubMed/NCBI View Article : Google Scholar
|
|
88
|
Jones DL, Morley MP, Li X, Ying Y, Zhao G,
Schaefer SE, Rodriguez LR, Cardenas-Diaz FL, Li S, Zhou S, et al:
An injury-induced mesenchymal-epithelial cell niche coordinates
regenerative responses in the lung. Science.
386(eado5561)2024.PubMed/NCBI View Article : Google Scholar
|
|
89
|
Xin S, Ding Y, Yu T, Fu Y, Cui Y and Nie
H: Mesenchymal stem cell-secreted KGF ameliorates acute lung injury
via the Gab1/ERK/NF-κB signaling axis. Cell Mol Biol Lett.
30(79)2025.PubMed/NCBI View Article : Google Scholar
|
|
90
|
Ushakumary MG, Riccetti M and Perl AT:
Resident interstitial lung fibroblasts and their role in alveolar
stem cell niche development, homeostasis, injury, and regeneration.
Stem Cells Transl Med. 10:1021–1032. 2021.PubMed/NCBI View Article : Google Scholar
|
|
91
|
Kang H, Kim OH, Chang ES, Kim J, Kim JY,
Shin GS, Kim CH, Lim Y, Seo YJ, Kim JW and Lee HJ: Enhanced
engraftment and immunomodulatory effects of integrin
alpha-2-overexpressing mesenchymal stromal cells in
lipopolysaccharide-induced acute lung injury. Stem Cell Res Ther.
16(286)2025.PubMed/NCBI View Article : Google Scholar
|
|
92
|
van der Koog L, Boerrigter MJ, Gorter IC,
Gosens R and Nagelkerke A: Lung fibroblast-derived extracellular
vesicles and soluble factors alleviate elastase-induced lung
injury. Eur J Pharmacol. 974(176612)2024.PubMed/NCBI View Article : Google Scholar
|
|
93
|
van der Koog L, Woest ME, Gorter IC,
Verschut V, Elferink RAB, Zuidhof AB, Nugraha DF, Koloko Ngassie
ML, Bos SIT, Dhakad D, et al: Fibroblast-derived osteoglycin
promotes epithelial cell repair. NPJ Regen Med.
10(16)2025.PubMed/NCBI View Article : Google Scholar
|
|
94
|
Ren W, Xia Y, Chen S, Wu G, Bazer FW, Zhou
B, Tan B, Zhu G, Deng J and Yin Y: Glutamine metabolism in
macrophages: A novel target for obesity/type 2 diabetes. Adv Nutr.
10:321–330. 2019.PubMed/NCBI View Article : Google Scholar
|
|
95
|
Liu PS, Wang H, Li X, Chao T, Teav T,
Christen S, Di Conza G, Cheng WC, Chou CH, Vavakova M, et al:
α-ketoglutarate orchestrates macrophage activation through
metabolic and epigenetic reprogramming. Nat Immunol. 18:985–994.
2017.PubMed/NCBI View Article : Google Scholar
|