1. Introduction
As one of the most common and severe acute abdomen
diseases, intra-abdominal infections (IAIs) have become the main
factor for the high mortality rate in diverse and complicated
refractory disease (1). IAIs are
mainly associated with abdominal traumatic surgery in the form of
post-operative complications (2).
The mortality rate from complicated intra-abdominal infections
(CIAIs) varies between different countries and regions and ranges
from 3-42% (3). In addition, the
associated morbidity also exhibits an increasing trend. A previous
multinational observational cohort study demonstrated that
secondary peritonitis accounted for the vast majority of cases
(68.4%), followed by biliary tract infection (12.2%),
intra-abdominal abscess (6.9%) and pancreatic infection (6.3%)
(4). IAI is a risk prognostic
factor for complications of these diseases, and despite advances in
treatment over the last few decades, the morbidity and mortality
remain at high levels.
The definition of IAI is a general term for a class
of diseases and represents a wide array of pathological conditions,
including cholecystitis, pancreatitis, appendicitis,
diverticulitis, and abdominal abscess and sepsis (5). Classified by its severity, IAI can be
divided into two categories as follows: i) Uncomplicated
intra-abdominal infection, which emerges in a single organ and is
accompanied by mild symptoms; this can be treated using antibiotics
therapy; and ii) CIAI, which not only affects the hollow organs,
but also penetrates the pathological organs to reach the peritoneum
(6), resulting in secondary
peritonitis and even in severe abdominal abscess. Of note,
peritonitis is typical of CIAI, and is mainly divided into primary
and secondary peritonitis (Fig.
1). Moreover, CIAI can be further categorized into two types:
Community CIAI that may be severe and occurring in the community,
and hospital CIAI, which is also termed healthcare-acquired CIAI,
usually occurring in patients who were hospitalized or who were
admitted to other medical care institutions; the latter type is
more common than the former (7),
particularly as regards post-operative infections. IAI is a severe
post-operative complication, which can lead to systemic
inflammatory response syndrome, sepsis and even in multiple organ
dysfunction syndrome. It induces pseudoaneurysm, sepsis and septic
shock, which not only exacerbate patient suffering, but also
lengthen hospital duration and increase medical costs; in addition,
IAIs decreases the quality of life of patients and may even result
in mortality (8).
Probiotics are live bacteria that, when consumed,
modify the gut microbial flora and provide the host with health
benefits (9). The use of
probiotics has been examined in inflammatory bowel disease,
irritable bowel syndrome and antibiotic-associated diarrhea
(10-12).
In the present review article, the current literature on the use of
probiotic therapy in the treatment of IAIs is summarized.
2. Literature search strategy
The following electronic databases were used to
search potentially relevant studies: PubMed (1995 to October,
2021), China National Knowledge Infrastructure (1995 to October,
2021) and Wan Fang Data (1995 to October, 2021).
The key words used included probiotics,
intra-abdominal infections, intestinal barrier dysfunction (IBD),
immune imbalance, mechanism, source control, antibiotics, bacterial
translocation and intestinal mucosal permeability.
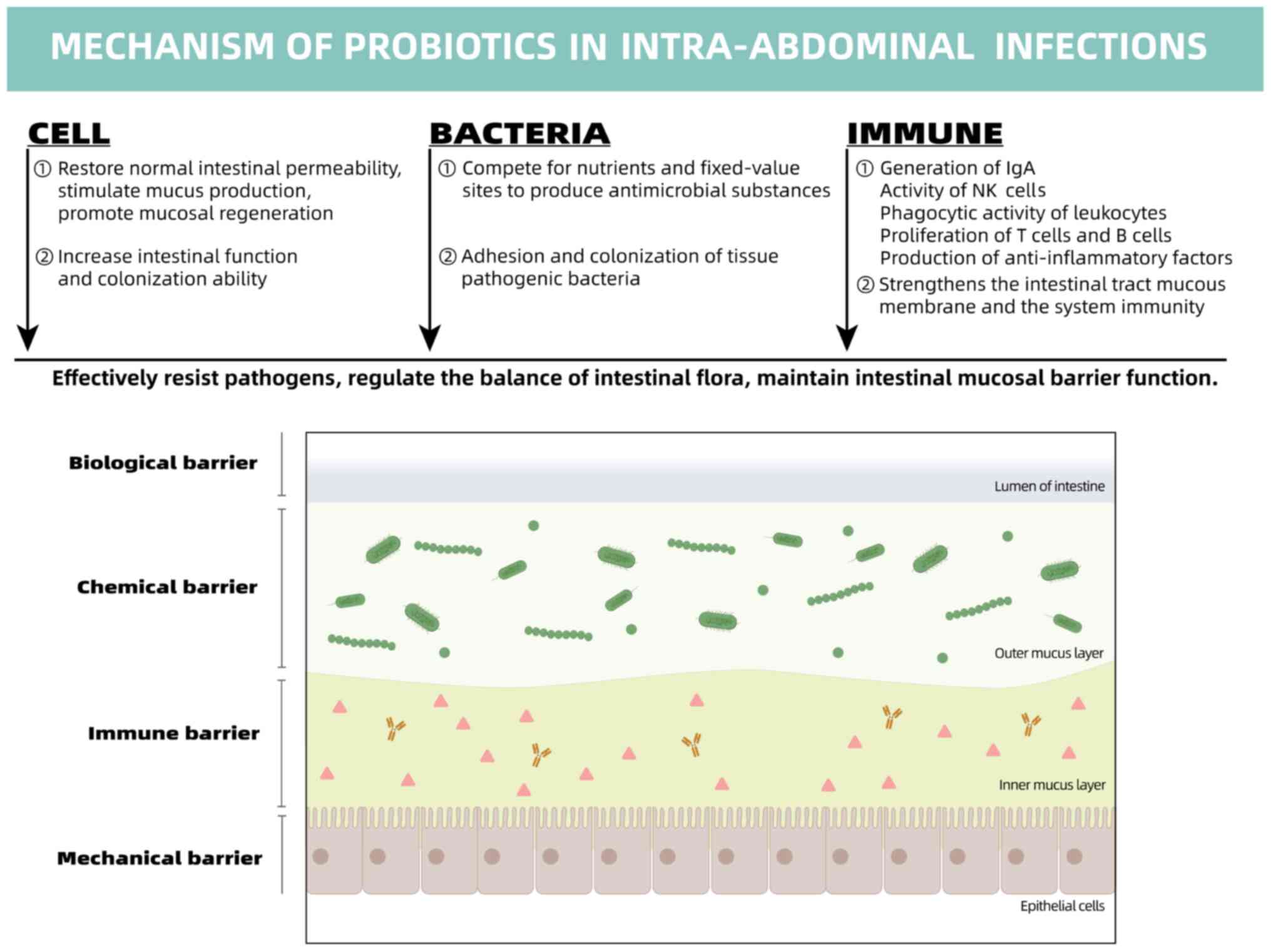
3. Mechanisms of action of probiotics
As is already known, there is a collection of
microorganisms in nature. For the most part, this collection of
microorganisms and bacteria has a negative impact on the daily life
of humans. However, decades of research have revealed that
bacteria, which is a subset of microorganisms that also includes
viruses, fungi and other protists, plays a critical and useful role
in the human body. All these microbes combine to form a chemical
that some researchers have termed the microbiota. The microbiota
can be found in a variety of tissues, including the mouth cavity,
gastrointestinal tract, nasal cavity, and others. In particular,
~80% of the normal microbiota is concentrate in the gut (13). It has been reported that almost
1,014 bacteria are present in adult intestinal cells, which is
10-fold larger than the amount present in normal cells in the human
body (14). Consequently, some
researchers have even indicated that the intestinal microbiota is
one of the organs in the human body (15).
The positive bacterial component is constituted by
probiotics. Probiotics are microorganisms that may be of net
benefit to humans when consumed. The administration of sufficient
doses of probiotics, which are defined as live microorganisms,
primarily modifies the balance of the intestinal microflora of the
host (16). Probiotics are live
microorganisms that can alter the intestinal microbial flora when
ingested, and confer beneficial health effects to the host. Their
use has been studied in various gastrointestinal conditions,
including inflammatory bowel disease, irritable bowel syndrome and
antibiotic-associated diarrhea. Metchnikff, who worked at the
Pasteur Institute at the turn of the century, was the first to
bring forth the hypothesis that individuals could benefit from the
consumption of fermented milk (17). As a result, the World Health
Organization (WHO) and the United Nations Food and Agriculture
Organization (FAO) designated it as a food-grade ingredient used in
dairy products that would promote the host's health. In one of his
articles from 1954, Vergin (18)
discussed the mechanisms thorough which antibiotics damage the
healthy gut microorganisms. Since then, the general comprehension
of probiotics and related applications have become increasingly
thorough, and further in-depth research has been conducted.
Probiotics are currently categorized into three
groups: Lactobacilli, Bifidobacteria, and Gram-positive
cocci, including certain yeasts and enzymes. However, depending
on the source and mode of action, they are divided into symbiotic
bacteria, native bacteria and fungi (19). Lactobacilli,
Bifidobacterium, butyric acid-producing bacteria,
Escherichia coli (E. coli), Streptococcus
thermophilus and other bacteria are all considered to be
applicable to the human body on a global scale. Currently, the
mechanisms of action of probiotics, such as lactic acid bacteria,
Bifidobacteria and yeast, are relatively well understood,
and previous research has been performed in this field. For
instance, it has been demonstrated that the combined use of
Lactobacillus rhamnosus GG (LGG) with conventional
antibiotic therapy reduces bacterial translocation, preventing bile
duct obstruction (20).
Lactobacillus strains have also been shown to significantly
enhance the phagocytic activity of macrophages (21). In recurrent respiratory tract
infections, the abundance of Bifidobacterium has been shown
to be reduced (21).
Probiotics mainly restore the balance of the flora
of the body via a mechanism termed the flora-host interaction
(22). A multitude of biological
reactions are also involved in this process. Probiotics have been
shown to function via a variety of mechanisms. For example, through
competitive inhibition, they can limit the ability of pathogens to
adhere to and colonize mucous membranes. Some probiotics secrete
proteases which aid in the breakdown of toxins. Probiotics have
also been shown to alter host immune functions by inhibiting the
release of pro-inflammatory cytokines by T-cells, and some
probiotic strains have been shown to inhibit mitogen-induced
lymphocyte proliferation in vitro in a similar manner to
dexamethasone (23). In addition,
probiotic bacteria exert a potential protective effect against
enteropathogens through various mechanisms, including the
production of antimicrobial compounds, the reduction of pathogenic
bacterial adhesion and the competition for host cell binding sites.
The probiotics principle can be characterized by the following
mechanisms: Immunoregulation, competitive inhibition, signal
transduction and metabolic alterations are all examples of barrier
function (24-27).
Immunoregulation can be further divided into innate and specific
immunity (25). It can also
inhibit the adhesion and colonization of pathogens through
competitive inhibition (28). The
oral administration of probiotics can increase the number of IgA
cells in the intestinal lamina propria (29). Moreover, probiotics can also induce
IgA circulation and significantly increase the activity of
macrophages distal to the gastrointestinal tract (30).
Regulatory effects of probiotics on
the disruption of the bacterial flora in IAIs
Similar to the endogenous microbiota, probiotics can
improve mucosal barrier function, limiting the ability of viral
particles to cross the barrier, thus maintaining this physical
barrier during viral disease (31). The adhesion of viruses to the
mucosal surface is the first step in infection. Extracellular
polysaccharides produced by lactic acid bacteria have been shown to
interfere effectively with this step, as for example adenoviruses
(32). However, the most effective
antiviral effect of probiotics is the regulation of the immune
system. Selected probiotics have been reported to increase natural
killer cell activity and cytotoxic activity (33). In addition, responses to antiviral
cytokines, such as interferon γ (IFN-γ), interleukin (IL)-2, IL-12
and IL-18, as well as improved antibody responses have been
reported with the use of probiotics (34). It is thus possible that several of
these mechanisms and others operate in parallel and/or
consecutively.
The incidence of IAIs, CIAIs in particular, is one
of the most common reasons for the increased health costs observed
$25,000 per case in the USA (35).
The treatment regimen for CIAIs, according to the most recent World
Society of Emergency Surgery (WSES) management guideline (3), is simply a combination of various
antibiotics. It is also for this reason that, as a result of the
overuse of antibiotics, resistance to medication is becoming a
major concern for all clinicians and patients (36). Thus, it is necessary to search for
a promising prevention and treatment strategy for CIAIs with
limited side-effects. Probiotics appear to meet this requirement.
As previously indicated by a clinical questionnaire in 2011, a
number of gastroenterologists and surgeons in the UK have recommend
the daily oral use of probiotics to their patients; in some cases
probiotics have been directly prescribed (37). In a previous systematic review and
meta-analysis of randomized controlled trials, 1,354 patients were
administered probiotics in the treatment group, while 1,369
patients were administered the placebo in the control group. The
outcome manifested that the use of probiotics decreased the risk of
developing IAIs and related complications post-operatively
(38). Additionally, it has been
demonstrated that probiotics can reduce the levels of
pro-inflammatory cytokines in rodents with post-infectious
irritable bowel syndrome (39). As
already aforementioned, IAIs are associated with the emergence of
bacterial translocation. However, bacterial translocation is caused
by IBD. The intestinal tract serves as a repository for bacteria
and endotoxins, and the colonic mucosal epithelium is tightly
connected to this tract, which provides a strong barrier function,
so that bacteria (or endotoxins) cannot easily transfer to other
normal sterile tissues through the barrier (40). Probiotics can boost the expression
of transmembrane binding protein (occludin) and IgA, allowing the
barrier function to be improved and bacterial translocation to be
reduced (41). Probiotics perform
a similar coordinating function by exerting immunomodulatory
effects. In animal models, oral Lactobacillus strains have
been shown to activate the mucosal immune system and to affect the
distal intestinal mucosa (42). A
previous study discussed the role of probiotics in the prevention
and treatment of intestinal infections (25).
Intestinal mucosal barrier (IMB)
function of probiotics in IAIs
Intestinal barrier function (IBF) is one of the most
essential barriers of the human body. Intestinal epithelial cells
(IECs) separate the inner and outer intestinal cavity so that
bacteria, endotoxins or other related pathogens, which may cause a
variety of infections, are unable to move across the IMB to other
tissues outside the intestinal lumen, preventing the body from
invasion by endogenous microorganisms and their toxins (43). IBD can accelerate endotoxin and
bacterial translocation. IBD is caused by a number of factors, such
as inflammation, stress, trauma, hypovolemia and
ischemia-reperfusion injury. In particular, infection, which is
caused by the invasion of a pathogen into the body, plays a major
role in IBD. Infection can lead to the activation and release of
inflammatory factors in human organs, and neutrophil aggregation
and adhesion result in damage to the intestinal mucosa. This can
subsequently reduce intestinal mucosa permeability, thus lead to a
series of pathological processes (44). Thus, the intestinal tract is not
only the initiator of infection, but also the ‘victim’ of
infection, and both play a causal role. Similarly, the occurrence
of abdominal infection aggravates IBD, and the further aggravation
of IBD further increases the occurrence of abdominal infections,
forming a vicious cycle. The core of the IBF is IMB, which includes
a mechanical barrier, chemical barrier, immune barrier and
biological barrier (Fig. 2).
The intestinal barrier can prevent harmful
substances in the intestinal cavity, such as pathogens and toxins,
from entering other tissues and organs in the body and blood
circulation through the intestinal mucosa. It represents a direct
physical barrier for the host. Injuries to the intestinal barrier
increase intestinal permeability to bacteria and their metabolites
(45). The IMB is responsible for
the majority of negative effects on IBF. When the IMB is
compromised, bacteria and their derivatives in the intestines can
break through and enter the bloodstream, causing bacteria or
endotoxins to be transferred to other organs, thus promoting the
spread of enterogenous illness (46). A number of studies have evaluated
the efficacy of probiotics in IAIs (Table I). For example, some lactic acid
bacteria and Bifidobacterium can upregulate the expression
of tight junction proteins, improving intestinal integrity and
protecting the IMB (47,48). Miele et al (48) demonstrated that damage to the tight
junctions of IECs may lead to increased intestinal permeability,
leading to the transposition of a large number of bacteria and
bacterial endotoxins into the blood circulation. Probiotics can
help prevent the invasion of pathogenic bacteria, forming a
microbial membrane with intestinal mucosal epithelial cells, and
competitively inhibiting the adhesion and colonization of
pathogenic bacteria (47).
 | Table IStudies evaluating the efficacy of
probiotics in intra-abdominal infections. |
Table I
Studies evaluating the efficacy of
probiotics in intra-abdominal infections.
| Authors | No. of
subjects | Methods | Study duration | Patients | Year | (Refs.) |
|---|
| Poonyam et
al | 100 | Double-blind
randomized placebo-controlled | ≥4 weeks | Aged ≥18 years
(with non-ulcer dyspepsia) | 2019 | (77) |
| Çekin et
al | 159 | Randomized placebo-
controlled | 2 weeks | Patients with H.
pylori infection receiving sequential H. pylori
eradication therapy | 2017 | (78) |
| Shukla et
al | 46 | Prospective
randomized controlled trials | 12 weeks | Patients were
children aged 6-20 years with diagnosed IBD | 2016 | (26) |
| Pande et
al | 110 | Double-blind,
randomized- controlled trials | 6 months | Consecutive
patients with cirrhosis | 2012 | (79) |
| Diepenhorst et
al | 30 | Prospective
randomized controlled trials | 4 weeks | Patients scheduled
for a PPPD with curative intent for a periampullary or ampullary
pancreatic malignancy | 2011 | (80) |
Immune regulatory mechanisms of
probiotics in IAIs
The interaction with IECs and the generation of
certain immune regulatory factors to increase the body's immune
function and manufacture immunoglobulin to resist harmful
microorganisms represent the regulatory effects of the immune
system. When activated by antigens, macrophages, B lymphocytes and
natural killer cells initiate an immunological response,
stimulating the intestinal mucosa to release anti-inflammatory
cytokines, increasing intestinal immunological performance and
enhancing immunological barrier function (49). Some probiotics can boost phagocytic
or natural killer cell activity and interact with dendritic cells
directly. Bharwani et al treated mice with oral
Lactobacillus rhamnosus (JB-1) for 28 days, and the results
revealed that treatment with JB-1 increased IL-10+
regulatory T-cells and reduced the activation of stress-related
dendritic cells, affecting the regulation of stress on immunity
(50). Furthermore, to limit
inflammation, Bifidobacterium relies on T-cell control, and
it can significantly reduce the inflammatory phenotype and
histopathology produced by cytotoxic T-lymphocyte antigen 4
(CTLA-4) antibodies (50).
Regulation of the gut barrier by
probiotic metabolites in IAIs
Probiotics lead to the formation of secondary
metabolites, such as bacteriocins, extracellular vesicles, short
fatty acids chain, indole and extracellular proteins (secreted
proteins). Bacteriocins are a class of ribosomally synthesized
antimicrobial peptides produced by bacteria (51,52).
Bacteriocins can function as colonizing peptides of certain
intestinal micro-organisms, promoting these bacteria to obtain a
competitive advantage over other strains and to thus occupy
established niches in the intestines. Alternatively, bacteriocins
can function as ‘killing’ peptides, since they can interfere with
the growth of pathogens (particularly Gram-negative bacteria) by
penetrating the inner membrane or disrupting cell wall synthesis
(53). Extracellular vesicles are
involved in bacteria-host communications and in the maintenance of
the gut homeostasis. It has been reported that the oral application
of Akkermansia muciniphila-derived extracellular vesicles
can alleviate dextran sulfate sodium-induced colitis by preventing
inflammatory cell infiltration into the colon wall and alterations
in colon length (54). Short-chain
fatty acids, which comprise mainly butyrate, propionate and
acetate, are metabolites secreted by intestinal microbiota from
undigested dietary carbohydrates and proteins (55). As butyrate, among all short-chain
fatty acids, is the preferred source of energy for colonic
epithelial cells, the association between butyrate and the
intestinal epithelial barrier has been previously studied (56). Butyrate can promote gut epithelium
O2 consumption, resulting in the stabilization of
hypoxia-inducible factor (HIF) and can increase the expression of
barrier-protective HIF target genes, connecting microbes and
epithelial barriers (57). Indole
is usually produced by bacteria that contain tryptophanase and has
been reported to function as an intestinal symbiotic bacteria
signal (58,59). It has been demonstrated that indole
produced by symbiotic E. coli can inhibit the chemotaxis of
pathogenic E. coli. E. coli-secreted indole can also
inhibit the attachment of pathogens to the epithelium by increasing
the expression of genes involved in intestinal epithelial function,
such as actin cytoskeleton, adhesion junctions and tight junctions
(60). The Lactiplantibacillus
plantarum BMCM12 secretes extracellular proteins and
considerably reduces the adhesion of microbes or strengthens the
gut barrier. It has been shown that two proteins secreted by
p40, p75 and LGG improve IEC homeostasis. Moreover,
p40 and p75 maintain gut homeostasis by activating
the EGFR/PIK3/Akt signaling pathway. It has been confirmed that
probiotic-secreted proteins are involved in the host and symbiotic
bacterial association (61).
These secondary metabolites can protect the gut
epithelial barrier, while integrating with certain antimicrobial
peptides, instantly enhancing mucus secretion via goblet cells and
promoting the tight junction protein expression. They thus
participate in the metabolism of human nutrients.
4. Safety of the use of probiotics in
intra-abdominal infection
Approximately 230 million surgical procedures are
performed worldwide each year. Some can lead to complications
post-surgery, including surgical site infections, urinary tract
infections, pneumonia and sepsis. Standard practice is prophylactic
antibiotic treatment; however, this is complicated by the increase
in antibiotic resistance. A recent network meta-analysis of 2,952
patients undergoing abdominal surgery in 31 studies revealed a
beneficial effect of synbiotics on surgical site infection
(62). In that meta-analysis, it
was found that synbiotics (successful trials focused on
Lactiplantibacillus plantarum, Lacticaseibacillus
casei and Bifidobacterium breve combined with
galacto-oligosaccharides) were the most effective interventions for
reducing pneumonia, urinary tract infections, sepsis, hospital
duration and antibiotic use, whereas they had no effect on
mortality (62). It was concluded
that surgeons should consider the use of synbiotics as an adjuvant
treatment for the prevention of post-operative complications
(54). Another meta-analysis
documented similar effects (63),
although it was concluded that these observations should be treated
with caution due to possible publication bias. In a recent
randomized controlled clinical trial, in which 55 patients who
underwent liver transplants received four probiotics or polylactic
acid-placebo treatments prior to the scheduled transplantation, it
was shown that at 90 days following the intervention, the infection
rate was significantly lower in the patients receiving the
probiotics compared with the control group (5 vs. 48%, P=0.002)
(64). In addition, in another
randomized controlled clinical trial with 18 patients with head and
neck cancer, no symbiotic effects of Lacticaseibacillus
paracase, Bifidobacterium lactis, Lactobacillus
acidophilus and Lactobacillus plus
fructooligosaccharides were observed compared with the 18 placebo
controls (65). Several clinical
trials on human milk oligosaccharides (HMOs) have been registered,
and HMOs may well represent a novel trend in prebiotics (66-68).
Chemically defined metabolites or cell wall compounds released by
probiotics may also become more important in the future (69). Probiotics may affect the gut-brain
axis, with implications for patients with irritable bowel syndrome,
mood disorders and anxiety (70).
5. Conclusions and future perspectives
In recent years, studies have demonstrated that the
intestinal flora of patients with abdominal cavity infections
exhibits certain dysfunctions, and the resulting intestinal
microecological imbalance and the impaired IMB function may be the
starting point of the inflammatory reaction of abdominal cavity
infection. When the intestinal flora is unbalanced, the IMB
function is impaired, and the impaired IMB function is the most
important link for the occurrence of intestinal inflammatory
reactions and abdominal infections. As a result, preserving the
intestinal microecological balance is crucial in the treatment of
abdominal infections (15,42,47,71).
It has been shown that H2O2, bacteriocin and
biosurfactant secreted by Lactobacillus can kill pathogenic
microorganisms, so as to further control the inflammatory response
and enhance the therapeutic effect (47).
The metabolites of probiotics play a crucial role in
regulating host-disease interactions. Extracellular polysaccharide
(EPSCG11) from Lactobacillus plantarum BGCG11 has been shown
to alleviate inflammatory pain in rats, reducing the expression of
pro-inflammatory factor, and increasing that of anti-inflammatory
factors (IL-10 and IL-6), thus exerting potent anti-hyperalgesic
and anti-edema effects (72).
Therefore, it can be concluded that the application of probiotics
for the clinical treatment of abdominal cavity infections may aid
in the suppression of the inflammatory response and in relieving
associated pain or discomfort.
Despite reports of microbial research in high-impact
scientific journals (73-76),
a broad definition of probiotics should be avoided. In the case
that a probiotic is found to exert a medical effect, it should be
noted that this effect is associated with specific strains or
specific prebiotics, providing specific health effects in a
specific patient or in a specific population. Although this
statement is trivial, it is still difficult to distill these
positive conclusions from the complex and sometimes contradictory
research literature.
In conclusion, probiotics and their metabolites may
improve the prognosis of IAIs by regulating IBF, immune regulation
and dysbacteriosis. However, further large-sample, multi-center
clinical trials are required to further explore the use of
probiotics in the treatment of IAIs.
Acknowledgements
Not applicable.
Funding
Funding: The present study was supported by the key Discipline
of Liaoning Traditional Chinese Medicine Clinical Ability Promote
Construction Project (grant no. 2019JH-8), the development Guidance
Plan Projects in Liaoning Province (grant no. 10300028) and the
National Science Foundation of China Project (grant no.
82174136).
Availability of data and materials
Not applicable.
Authors' contributions
ST and JL conceived the review and drafted the
manuscript. CX was responsible for the preparation of the figures
and the literature search. DS and HC revised, edited and finalized
the manuscript for submission. GZ reviewed the manuscript. All
authors have read and approved the final manuscript. Data
authentication is not applicable.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Shirah GR and O'Neill PJ: Intra-abdominal
Infections. Surg Clin North Am. 94:1319–1333. 2014.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Mazuski JE, Tessier JM, May AK, Sawyer RG,
Nadler EP, Rosengart MR, Chang PK, O'Neill PJ, Mollen KP, Huston
JM, et al: The Surgical Infection Society Revised Guidelines on the
Management of Intra-Abdominal Infection. Surg Infect (Larchmt).
18:1–76. 2017.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Lalisang TJM, Usman N, Hendrawidjaya I,
Handaya AY, Nasution S, Saunar RY, Loho T, Karuniawati A, Moenadjat
Y and Widyahening IS: Clinical Practice Guidelines in Complicated
Intra-Abdominal Infection 2018: An Indonesian Perspective. Surg
Infect (Larchmt). 20:83–90. 2019.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Blot S, Antonelli M, Arvaniti K, Blot K,
Creagh-Brown B, de Lange D, De Waele J, Deschepper M, Dikmen Y,
Dimopoulos G, et al: Abdominal Sepsis Study (AbSeS) group on behalf
of the Trials Group of the European Society of Intensive Care
Medicine: Epidemiology of intra-abdominal infection and sepsis in
critically ill patients: ‘AbSeS’, a multinational observational
cohort study and ESICM Trials Group Project. Intensive Care Med.
45:1703–1717. 2019.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Menichetti F and Sganga G: Definition and
classification of intra-abdominal infections. J Chemother. 21
(Suppl 1):3–4. 2009.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Zhang J, Di Y, Wang Z and Liu C:
Re-Thinking the Definition of Complicated Intra-Abdominal
Infection. Surg Infect (Larchmt). 18(374)2017.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Pieracci FM and Barie PS: Management of
severe sepsis of abdominal origin. Scand J Surg. 96:184–196.
2007.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Allegranzi B, Zayed B, Bischoff P, Kubilay
NZ, de Jonge S, de Vries F, Gomes SM, Gans S, Wallert ED, Wu X, et
al: WHO Guidelines Development Group: New WHO recommendations on
intraoperative and postoperative measures for surgical site
infection prevention: An evidence-based global perspective. Lancet
Infect Dis. 16:e288–e303. 2016.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Salminen S, Collado MC, Endo A, Hill C,
Lebeer S, Quigley EMM, Sanders ME, Shamir R, Swann JR, Szajewska H
and Vinderola G: The International Scientific Association of
Probiotics and Prebiotics (ISAPP) consensus statement on the
definition and scope of postbiotics. Nat Rev Gastroenterol Hepatol.
18:649–667. 2021.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Karban A: Effect of Probiotics on
Inflammatory Bowel Diseases. J Inflam Bowel Dis Disorder.
6(e108)2021.
|
|
11
|
Agamennone V, Krul CAM, Rijkers G and Kort
R: A practical guide for probiotics applied to the case of
antibiotic-associated diarrhea in The Netherlands. BMC
Gastroenterol. 18(103)2018.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Simon E, Călinoiu LF, Mitrea L and Vodnar
DC: Probiotics, Prebiotics, and Synbiotics: Implications and
Beneficial Effects against Irritable Bowel Syndrome. Nutrients.
13(2112)2021.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Kundu P, Blacher E, Elinav E and
Pettersson S: Our Gut Microbiome: The Evolving Inner Self. Cell.
171:1481–1493. 2017.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Sommer F and Bäckhed F: The gut microbiota
- masters of host development and physiology. Nat Rev Microbiol.
11:227–238. 2013.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Sánchez B, Delgado S, Blanco-Míguez A,
Lourenço A, Gueimonde M and Margolles A: Probiotics, gut
microbiota, and their influence on host health and disease. Mol
Nutr Food Res. 61(1600240)2017.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Pan SJ, Kuo CH, Lam KP, Chu YT, Wang WL
and Hung CH: Probiotics and allergy in children - an update review.
Pediatr Allergy Immunol. 21:e659–e666. 2010.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Vanderhoof JA and Young RJ: Pediatric
applications of probiotics. Gastroenterol Clin North Am.
34:451–463, viii-ix. 2005.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Vergin F: Antibiotics and probiotics.
Hippokrates. 25:116–119. 1954.PubMed/NCBI(In Undetermined language).
|
|
19
|
Sanders ME: Overview of Functional Foods:
Emphasis on Probiotic Bacteria. Int Dairy J. 8:341–347. 1998.
|
|
20
|
Celikkaya ME, Akcora B, Hakverdi S, Ozer
B, Ulutas KT and Duran N: Effects of Probiotic Use on Bacterial
Translocation in Created Rat Models with Biliary Obstructions.
Eurasian J Med. 51:106–111. 2019.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Li L, Wang F, Liu Y and Gu F: Intestinal
microbiota dysbiosis in children with recurrent respiratory tract
infections. Microb Pathog. 136(103709)2019.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Maslowski KM: Metabolism at the centre of
the host-microbe relationship. Clin Exp Immunol. 197:193–204.
2019.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Liu Q, Tian H, Kang Y, Tian Y, Li L, Kang
X, Yang H, Wang Y, Tian J, Zhang F, et al: Probiotics alleviate
autoimmune hepatitis in mice through modulation of gut microbiota
and intestinal permeability. J Nutr Biochem.
98(108863)2021.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Narula N and Marshall JK: Role of
probiotics in management of diverticular disease. J Gastroenterol
Hepatol. 25:1827–1830. 2010.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Iqbal Z, Ahmed S, Tabassum N, Bhattacharya
R and Bose D: Role of probiotics in prevention and treatment of
enteric infections: A comprehensive review. 3 Biotech.
11(242)2021.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Shukla A, Gaur P and Aggarwal A: Effect of
probiotics on clinical and immune parameters in enthesitis-related
arthritis category of juvenile idiopathic arthritis. Clin Exp
Immunol. 185:301–308. 2016.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Gupta V and Garg R: Probiotics. Indian J
Med Microbiol. 27:202–209. 2009.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Monteagudo-Mera A, Rastall RA, Gibson GR,
Charalampopoulos D and Chatzifragkou A: Adhesion mechanisms
mediated by probiotics and prebiotics and their potential impact on
human health. Appl Microbiol Biotechnol. 103:6463–6472.
2019.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Yurong Y, Ruiping S, Shimin Z and Yibao J:
Effect of probiotics on intestinal mucosal immunity and
ultrastructure of cecal tonsils of chickens. Arch Anim Nutr.
59:237–246. 2005.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Zeng W, Shen J, Bo T, Peng L, Xu H, Nasser
MI, Zhuang Q and Zhao M: Cutting Edge: Probiotics and Fecal
Microbiota Transplantation in Immunomodulation. J Immunol Res.
2019(1603758)2019.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Bron PA, Kleerebezem M, Brummer R-J, Cani
PD, Mercenier A, MacDonald TT, Garcia-Ródenas CL and Wells JM: Can
probiotics modulate human disease by impacting intestinal barrier
function? Br J Nutr. 117:93–107. 2017.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Biliavska L, Pankivska Y, Povnitsa O and
Zagorodnya S: Antiviral Activity of Exopolysaccharides Produced by
Lactic Acid Bacteria of the Genera Pediococcus, Leuconostoc and
Lactobacillus against Human Adenovirus Type 5. Medicina
(Kaunas). 55(519)2019.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Miller LE, Lehtoranta L and Lehtinen MJ:
Short-term probiotic supplementation enhances cellular immune
function in healthy elderly: Systematic review and meta-analysis of
controlled studies. Nutr Res. 64:1–8. 2019.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Lehtoranta L, Latvala S and Lehtinen MJ:
Role of Probiotics in Stimulating the Immune System in Viral
Respiratory Tract Infections: A Narrative Review. Nutrients.
12(3163)2020.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Lytvyn L, Quach K, Banfield L, Johnston BC
and Mertz D: Probiotics and synbiotics for the prevention of
postoperative infections following abdominal surgery: A systematic
review and meta-analysis of randomized controlled trials. J Hosp
Infect. 92:130–139. 2016.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Adámková V: The role of new antibiotics in
intra-abdominal infections in the era of multi-resistant bacteria.
Rozhl Chir. 98:145–151. 2019.PubMed/NCBI
|
|
37
|
Cordina C, Shaikh I, Shrestha S and
Camilleri-Brennan J: Probiotics in the management of
gastrointestinal disease: Analysis of the attitudes and prescribing
practices of gastroenterologists and surgeons. J Dig Dis.
12:489–496. 2011.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Chowdhury AH, Adiamah A, Kushairi A,
Varadhan KK, Krznaric Z, Kulkarni AD, Neal KR and Lobo DN:
Perioperative Probiotics or Synbiotics in Adults Undergoing
Elective Abdominal Surgery: A Systematic Review and Meta-analysis
of Randomized Controlled Trials. Ann Surg. 271:1036–1047.
2020.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Hong KB, Seo H, Lee JS and Park Y: Effects
of probiotic supplementation on post-infectious irritable bowel
syndrome in rodent model. BMC Complement Altern Med.
19(195)2019.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Liu Z, Qin H, Yang Z, Xia Y, Liu W, Yang
J, Jiang Y, Zhang H, Yang Z, Wang Y, et al: Randomised clinical
trial: The effects of perioperative probiotic treatment on barrier
function and post-operative infectious complications in colorectal
cancer surgery - a double-blind study. Aliment Pharmacol Ther.
33:50–63. 2011.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Michail S and Abernathy F:
Lactobacillus plantarum reduces the in vitro secretory
response of intestinal epithelial cells to enteropathogenic
Escherichia coli infection. J Pediatr Gastroenterol Nutr.
35:350–355. 2002.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Paone P and Cani PD: Mucus barrier, mucins
and gut microbiota: The expected slimy partners? Gut. 69:2232–2243.
2020.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Shen TY, Qin HL, Gao ZG, Fan XB, Hang XM
and Jiang YQ: Influences of enteral nutrition combined with
probiotics on gut microflora and barrier function of rats with
abdominal infection. World J Gastroenterol. 12:4352–4358.
2006.PubMed/NCBI View Article : Google Scholar
|
|
44
|
Tao Z, Hu FQ, Li CF, Zhang T, Cao BZ and
Cui LQ: Effect of ulinastatin, a human urinary protease inhibitor,
on heatstroke-induced apoptosis and inflammatory responses in rats.
Exp Ther Med. 13:335–341. 2017.PubMed/NCBI View Article : Google Scholar
|
|
45
|
Rohr MW, Narasimhulu CA, Rudeski-Rohr TA
and Parthasarathy S: Negative Effects of a High-Fat Diet on
Intestinal Permeability: A Review. Adv Nutr. 11:77–91.
2020.PubMed/NCBI View Article : Google Scholar
|
|
46
|
Nagpal K, Minocha VR, Agrawal V and Kapur
S: Evaluation of intestinal mucosal permeability function in
patients with acute pancreatitis. Am J Surg. 192:24–28.
2006.PubMed/NCBI View Article : Google Scholar
|
|
47
|
La Fata G, Weber P and Mohajeri MH:
Probiotics and the Gut Immune System: Indirect Regulation.
Probiotics Antimicrob Proteins. 10:11–21. 2018.PubMed/NCBI View Article : Google Scholar
|
|
48
|
Miele L, Valenza V, La Torre G, Montalto
M, Cammarota G, Ricci R, Mascianà R, Forgione A, Gabrieli ML,
Perotti G, et al: Increased intestinal permeability and tight
junction alterations in nonalcoholic fatty liver disease.
Hepatology. 49:1877–1887. 2009.PubMed/NCBI View Article : Google Scholar
|
|
49
|
Rose EC, Odle J, Blikslager AT and Ziegler
AL: Probiotics, Prebiotics and Epithelial Tight Junctions: A
Promising Approach to Modulate Intestinal Barrier Function. Int J
Mol Sci. 22(6729)2021.PubMed/NCBI View Article : Google Scholar
|
|
50
|
Bharwani A, Mian MF, Surette MG,
Bienenstock J and Forsythe P: Oral treatment with Lactobacillus
rhamnosus attenuates behavioural deficits and immune changes in
chronic social stress. BMC Med. 15(7)2017.PubMed/NCBI View Article : Google Scholar
|
|
51
|
Gillor O, Etzion A and Riley MA: The dual
role of bacteriocins as anti- and probiotics. Appl Microbiol
Biotechnol. 81:591–606. 2008.PubMed/NCBI View Article : Google Scholar
|
|
52
|
Chikindas ML, Weeks R, Drider D,
Chistyakov VA and Dicks LMT: Functions and emerging applications of
bacteriocins. Curr Opin Biotechnol. 49:23–28. 2018.PubMed/NCBI View Article : Google Scholar
|
|
53
|
Majeed H, Gillor O, Kerr B and Riley MA:
Competitive interactions in Escherichia coli populations:
The role of bacteriocins. ISME J. 5:71–81. 2011.PubMed/NCBI View Article : Google Scholar
|
|
54
|
Kang CS, Ban M, Choi EJ, Moon HG, Jeon JS,
Kim DK, Park SK, Jeon SG, Roh TY, Myung SJ, et al: Extracellular
Vesicles Derived from Gut Microbiota, Especially Akkermansia
muciniphila, Protect the Progression of Dextran Sulfate
Sodium-Induced Colitis. PLoS One. 8(e76520)2013.PubMed/NCBI View Article : Google Scholar
|
|
55
|
Hamer HM, Jonkers D, Venema K, Vanhoutvin
S, Troost FJ and Brummer RJ: Review article: The role of butyrate
on colonic function. Aliment Pharmacol Ther. 27:104–119.
2008.PubMed/NCBI View Article : Google Scholar
|
|
56
|
Donohoe DR, Garge N, Zhang X, Sun W,
O'Connell TM, Bunger MK and Bultman SJ: The microbiome and butyrate
regulate energy metabolism and autophagy in the mammalian colon.
Cell Metab. 13:517–526. 2011.PubMed/NCBI View Article : Google Scholar
|
|
57
|
Kelly CJ, Zheng L, Campbell EL, Saeedi B,
Scholz CC, Bayless AJ, Wilson KE, Glover LE, Kominsky DJ, Magnuson
A, et al: Crosstalk between Microbiota-Derived Short-Chain Fatty
Acids and Intestinal Epithelial HIF Augments Tissue Barrier
Function. Cell Host Microbe. 17:662–671. 2015.PubMed/NCBI View Article : Google Scholar
|
|
58
|
Shimada Y, Kinoshita M, Harada K, Mizutani
M, Masahata K, Kayama H and Takeda K: Commensal Bacteria-Dependent
Indole Production Enhances Epithelial Barrier Function in the
Colon. PLoS One. 8(e80604)2013.PubMed/NCBI View Article : Google Scholar
|
|
59
|
Beaumont M, Neyrinck AM, Olivares M,
Rodriguez J, de Rocca Serra A, Roumain M, Bindels LB, Cani PD,
Evenepoel P, Muccioli GG, et al: The gut microbiota metabolite
indole alleviates liver inflammation in mice. FASEB J.
32(fj201800544)2018.PubMed/NCBI View Article : Google Scholar
|
|
60
|
Bansal T, Alaniz RC, Wood TK and Jayaraman
A: The bacterial signal indole increases epithelial-cell
tight-junction resistance and attenuates indicators of
inflammation. Proc Natl Acad Sci USA. 107:228–233. 2010.PubMed/NCBI View Article : Google Scholar
|
|
61
|
Liu Q, Yu Z, Tian F, Zhao J, Zhang H, Zhai
Q and Chen W: Surface components and metabolites of probiotics for
regulation of intestinal epithelial barrier. Microb Cell Fact.
19(23)2020.PubMed/NCBI View Article : Google Scholar
|
|
62
|
Kasatpibal N, Whitney JD, Saokaew S,
Kengkla K, Heitkemper MM and Apisarnthanarak A: Effectiveness of
Probiotic, Prebiotic, and Synbiotic Therapies in Reducing
Postoperative Complications: A Systematic Review and Network
Meta-analysis. Clin Infect Dis. 64 (Suppl 2):S153–S160.
2017.PubMed/NCBI View Article : Google Scholar
|
|
63
|
Yang Z, Wu Q, Liu Y and Fan D: Effect of
Perioperative Probiotics and Synbiotics on Postoperative Infections
After Gastrointestinal Surgery: A Systematic Review With
Meta-Analysis. JPEN J Parenter Enteral Nutr. 41:1051–1062.
2017.PubMed/NCBI View Article : Google Scholar
|
|
64
|
Grąt M, Wronka KM, Lewandowski Z, Grąt K,
Krasnodębski M, Stypułkowski J, Hołówko W, Masior Ł, Kosińska I,
Wasilewicz M, et al: Effects of continuous use of probiotics before
liver transplantation: A randomized, double-blind,
placebo-controlled trial. Clin Nutr. 36:1530–1539. 2017.PubMed/NCBI View Article : Google Scholar
|
|
65
|
Lages PC, Generoso SV and Correia MITD:
Postoperative symbiotic in patients with head and neck cancer: A
double-blind randomised trial. Br J Nutr. 119:190–195.
2018.PubMed/NCBI View Article : Google Scholar
|
|
66
|
Sakanaka M, Gotoh A, Yoshida K, Odamaki T,
Koguchi H, Xiao JZ, Kitaoka M and Katayama T: Varied Pathways of
Infant Gut-Associated Bifidobacterium to Assimilate Human
Milk Oligosaccharides: Prevalence of the Gene Set and Its
Correlation with Bifidobacteria-Rich Microbiota Formation.
Nutrients. 12(71)2019.PubMed/NCBI View Article : Google Scholar
|
|
67
|
Bering SB: Human Milk Oligosaccharides to
Prevent Gut Dysfunction and Necrotizing Enterocolitis in Preterm
Neonates. Nutrients. 10(1461)2018.PubMed/NCBI View Article : Google Scholar
|
|
68
|
Donovan SM and Comstock SS: Human Milk
Oligosaccharides Influence Neonatal Mucosal and Systemic Immunity.
Ann Nutr Metab. 69 (Suppl 2):42–51. 2016.PubMed/NCBI View Article : Google Scholar
|
|
69
|
Aguilar-Toalá JE, Garcia-Varela R, Garcia
HS, Mata-Haro V, González-Córdova AF, Vallejo-Cordoba B and
Hernández-Mendoza A: Postbiotics: An evolving term within the
functional foods field. Trends Food Sci Technol. 75:105–114.
2018.
|
|
70
|
Mörkl S, Butler MI, Holl A, Cryan JF and
Dinan TG: Probiotics and the Microbiota-Gut-Brain Axis: Focus on
Psychiatry. Curr Nutr Rep. 9:171–182. 2020.PubMed/NCBI View Article : Google Scholar
|
|
71
|
Suez J, Zmora N, Zilberman-Schapira G, Mor
U, Dori-Bachash M, Bashiardes S, Zur M, Regev-Lehavi D, Ben-Zeev
Brik R, Federici S, et al: Post-Antibiotic Gut Mucosal Microbiome
Reconstitution Is Impaired by Probiotics and Improved by Autologous
FMT. Cell. 174:1406–1423.e16. 2018.PubMed/NCBI View Article : Google Scholar
|
|
72
|
Dinić M, Pecikoza U, Djokić J,
Stepanović-Petrović R, Milenković M, Stevanović M, Filipović N,
Begović J, Golić N and Lukić J: Exopolysaccharide Produced by
Probiotic Strain Lactobacillus paraplantarum BGCG11 Reduces
Inflammatory Hyperalgesia in Rats. Front Pharmacol.
9(1)2018.PubMed/NCBI View Article : Google Scholar
|
|
73
|
Allen SJ, Martinez EG, Gregorio GV and
Dans LF: Probiotics for treating acute infectious diarrhoea.
Cochrane Database Syst Rev. 2010(CD003048)2010.PubMed/NCBI View Article : Google Scholar
|
|
74
|
Zhong C, Qu C, Wang B, Liang S and Zeng B:
Probiotics for Preventing and Treating Small Intestinal Bacterial
Overgrowth: A Meta-Analysis and Systematic Review of Current
Evidence. J Clin Gastroenterol. 51:300–311. 2017.PubMed/NCBI View Article : Google Scholar
|
|
75
|
Suez J, Zmora N, Segal E and Elinav E: The
pros, cons, and many unknowns of probiotics. Nat Med. 25:716–729.
2019.PubMed/NCBI View Article : Google Scholar
|
|
76
|
The Integrative Human Microbiome Project.
The Integrative Human Microbiome Project. Nature. 569:641–648.
2019.
|
|
77
|
Poonyam P, Chotivitayatarakorn P and
Vilaichone RK: High Effective of 14-Day High-Dose PPI-
Bismuth-Containing Quadruple Therapy with Probiotics Supplement for
Helicobacter pylori Eradication: A Double Blinded-Randomized
Placebo-Controlled Study. Asian Pac J Cancer Prev. 20:2859–2864.
2019.PubMed/NCBI View Article : Google Scholar
|
|
78
|
Çekin AH, Şahintürk Y, Akbay Harmandar F,
Uyar S, Yolcular BO and Çekin Y: Use of probiotics as an adjuvant
to sequential H. pylori eradication therapy: Impact on
eradication rates, treatment resistance, treatment-related side
effects, and patient compliance. Turk J Gastroenterol. 28:3–11.
2017.PubMed/NCBI View Article : Google Scholar
|
|
79
|
Pande C, Kumar A and Sarin SK: Addition of
probiotics to norfloxacin does not improve efficacy in the
prevention of spontaneous bacterial peritonitis: A double-blind
placebo-controlled randomized-controlled trial. Eur J Gastroenterol
Hepatol. 24:831–839. 2012.PubMed/NCBI View Article : Google Scholar
|
|
80
|
Diepenhorst GM, van Ruler O, Besselink MG,
van Santvoort HC, Wijnandts PR, Renooij W, Gouma DJ, Gooszen HG and
Boermeester MA: Influence of prophylactic probiotics and selective
decontamination on bacterial translocation in patients undergoing
pancreatic surgery: A randomized controlled trial. Shock. 35:9–16.
2011.PubMed/NCBI View Article : Google Scholar
|