Introduction
Several reports were recently published on the
potential beneficial effects of Citrullus colocynthis
extracts in animal models of diabetes mellitus (1–4).
In light of these previous findings, the present study aimed mainly
at assessing the effects of five distinct extracts from
Citrullus colocynthis seeds, i.e. a crude and a defatted
aqueous extract, an H2O-methanol extract, an ethyl
acetate extract and a n-butanol extract on a number of metabolic
variables in streptozotocin-induced diabetic rats. For the purpose
of comparison, a comparable study was conducted in normal rats,
whilst a further group of STZ rats was exposed to glibenclamide.
Both the short-term effects recorded over 180 min after the first
intraperitoneal injection of each tested material and the long-term
effects observed after daily injection up to three weeks after the
onset of the experiments were examined in the present study.
Materials and methods
Fresh fruit of Citrullus colocynthis were
collected from desert area of Aïn sefra (Algeria). The species was
botanically authenticated. A voucher specimen of the plant is
deposited in the herbarium of the Laboratory of Ecology. Box
Cucurbitaceae. Code 2686 (University Es Senia-Oran, Algeria). Five
extracts from Citrullus colocynthis seeds were used in the
present experiments, namely a crude untreated aqueous extract, a
defatted aqueous extract, an H2O-methanol extract, an
ethyl acetate extract and a n-butanol extract. The aqueous extracts
of Citrullus colocynthis L. seeds were prepared as described
elsewhere (2). For the
hydromethanolic extract, 50 g of seeds were ground and degreased in
hexane. This material was heated and stirred 3 times for 3 h in
water:methanol mixture (30:70). Then, after filtration and
centrifugation, the recovered solution was divided into two
volumes; one was evaporated to dryness and provided a hygroscopic
red-orange residue (H2O-methanol extract; 4.5% dry
matter). The second volume was used, after liquid-liquid extraction
3 times with ethyl acetate and n-butanol respectively, for the
preparation of the ethyl acetate (orange powder 1.1% dry matter)
and n-butanol (brown powder 1.2% dry matter) extracts.
Male Wistar rats weighing 208±3 g (n=72) at the
onset of the present experiments were given free access to tap
water and a commercial diet containing (w/w) 49.5% carbohydrate,
15.7% protein and 7.8% lipid. Forty-two of these rats were injected
intraperitoneally with streptozotocin (65 mg/kg body wt.) dissolved
in a citrate buffer (0.1 M, pH 4.5) one week before the onset of
the experiments after overnight starvation.
A first blood sample was obtained on day zero after
overnight fasting by puncture of the retro-orbital sinus. The
animals were then injected intraperitoneally either with saline (5
ml/kg), glibenclamide (10 mg/kg), the crude or defatted aqueous
extract (90 mg/kg), the H2O-methanol extract (100
mg/kg), the ethyl acetate extract (10 mg/kg) or the n-butanol
extract (70 mg/kg). Further blood samples were obtained from the
tail tip 30, 60, 120 and 180 min after such an intraperitoneal
injection (short-term experiments).
The same rats were then injected daily over a period
of 3 weeks with the same material always administered
intraperitoneally. Blood samples were obtained after overnight
starvation by retro-orbital puncture on day 7, day 14 and day 21
(long-term experiments). An oral glucose tolerance test was then
performed, a solution of D-glucose being administered by a gastric
sound to deliver 2.0 g of D-glucose per kg body wt. and blood
samples being collected from the tail tip 30, 60, 120 and 180 min
thereafter. The next day (day 22), the rats were sacrificed under
chloral anesthesia after overnight starvation and the pancreas,
liver, kidneys, testes, epididymal adipose tissue and diaphragm
muscle removed and weighed.
The glycemia was measured in blood samples using a
glucometer (Accu-check). The serum concentration of cholesterol,
triglycerides and urea were measured by enzymatic Kit Spinreact,
S.A. Ctra. Santa Coloma, Sant Esteve de Bas (GI) Spain; creatinine
is measured by end point Jaffé method (Kit Prochima, Prochima
Sigma, Sarl, Tlemcen, Algeria), glutamate-pyruvate and
glutamate-oxaloacetate transaminase and alkaline phosphatase were
measured by enzymatic Kit Spinreact, S.A. Ctra. Santa Coloma.
All results, including those already mentioned, are
presented as mean values (± SEM) together with the number of
individual observations (n). The statistical significance of
differences between mean values was assessed by use of Student’s
t-test.
Results
Short-term experiments
In the short-term experiments, overnight fasted rats
were injected intraperitoneally with various extracts, their
glycemia being measured before and at intervals after such an
injection. The primary results of these experiments are listed in
Table I. As expected, the initial
measurements of glycemia yielded a much higher mean value
(p<0.001) in STZ rats (19.62±0.55 mM; n= 42) than in normal
animals (5.25±0.09 mM; n=30). In order to assess the overall
changes in glycemia, the incremental (or decremental) area was
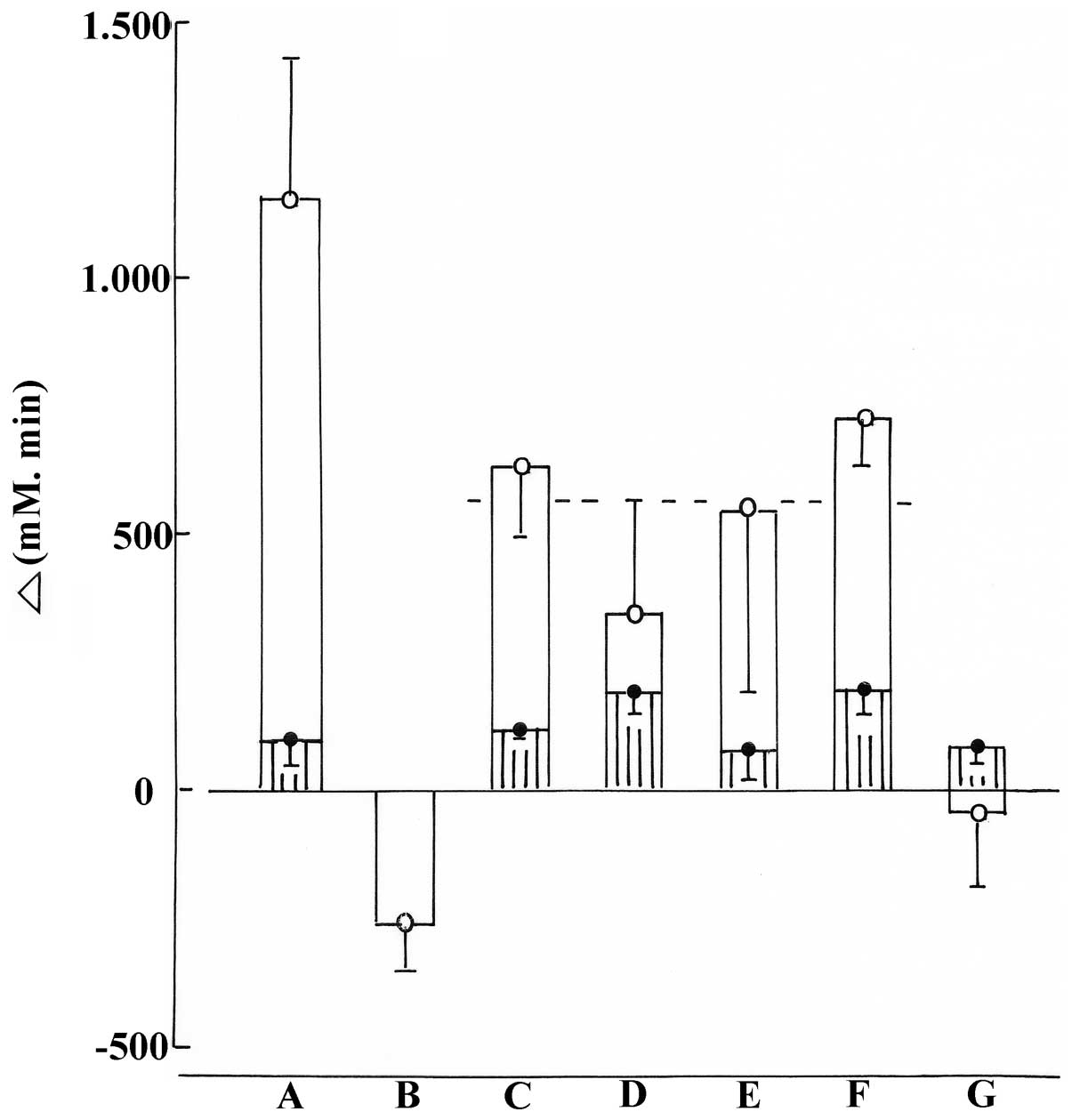
calculated in each individual experiment. As illustrated in
Fig. 1, in the normal animals, no
significant difference was observed (p>0.19 or more) for such an
incremental area between control rats and any of the five
experimental groups. In the control animals injected with saline,
the incremental area was significantly higher (p<0.01) in the
STZ rats (1,156±275 mM.min; n=7) than in the normal animals (99±48
mM.min; n=5). In the STZ rats injected with glibenclamide, a
decremental area (−258±97 mM.min; n=5; p<0.03) prevailed, as
distinct from the incremental area recorded in the control STZ
rats. In the STZ rats injected with the n-butanol extract, the mean
area below time zero value (−42±145 mM.min; n=5) was also
significantly lower (p<0.008) than the mean incremental area
found in the control STZ rats. Although such was no more the case
in the other four groups of experimental STZ rats, which yielded
mean values not significantly different from one another (p>0.15
or more), the overall mean incremental area recorded in these four
groups (566 ±109 mM. min; n=20) was again significantly lower
(p<0.025) than that found in the control STZ rats (Fig. 1).
 | Table IGlycemia (mM) in short-term
experiments. |
Table I
Glycemia (mM) in short-term
experiments.
| Rats | Extract | Zero | 30 min | 60 min | 120 min | 180 min |
|---|
| Normal | Control | 5.43±0.28 (5) | 6.02±0.14 (5) | 6.87±0.29 (5) | 5.56±0.23 (5) | 5.77±0.27 (5) |
| Untreated
aqueous | 5.34±0.10 (5) | 6.39±0.34 (5) | 6.49±0.09 (5) | 5.90±0.22 (5) | 5.60±0.31 (5) |
| Defatted aqueous | 5.27±0.18 (5) | 6.55±0.26 (5) | 7.08±0.41 (5) | 6.07±0.23 (5) | 5.00±0.23 (5) |
|
H2O-methanol | 5.02±0.34 (5) | 5.08±0.40 (5) | 6.51±0.21 (5) | 5.34±0.33 (5) | 4.64±0.41 (5) |
| Ethyl acetate | 5.41±0.16 (5) | 6.62±0.48 (5) | 6.79±0.72 (5) | 6.43±0.58 (5) | 6.63±0.59 (5) |
| n-butanol | 5.06±0.14 (5) | 5.36±0.22 (5) | 5.61±0.14 (5) | 5.83±0.34 (5) | 5.48±0.36 (5) |
| STZ | Control | 21.32±1.26 (7) | 26.44±0.54 (7) | 28.25±0.49 (7) | 29.13±1.04 (7) | 28.33±1.09 (7) |
| Glibenclamide | 22.01±1.04 (10) | 23.75±1.42 (10) | 21.13±1.73 (10) | 19.54±1.38 (10) | 17.81±1.36 (10) |
| Untreated
aqueous | 19.03±0.55 (5) | 22.32±1.02 (5) | 23.82±0.69 (5) | 22.52±0.97 (5) | 22.71±1.15 (5) |
| Defatted aqueous | 20.00±0.82 (5) | 25.32±0.68 (5) | 24.26±1.27 (5) | 20.30±1.00 (5) | 19.26±0.87 (5) |
|
H2O-methanol | 18.52±1.46 (5) | 18.43±1.56 (5) | 20.91±2.96 (5) | 22.89±2.40 (5) | 24.69±3.12 (5) |
| Ethyl acetate | 17.06±2.50 (5) | 19.86±2.64 (5) | 21.42±2.80 (5) | 22.48±3.46 (5) | 21.12±1.62 (5) |
| n-butanol | 16.38±0.26 (5) | 17.37±0.46 (5) | 17.91±1.34 (5) | 15.98±1.36 (5) | 12.49±1.21 (5) |
Long-term experiments
Glycemia
The same animals as those used in the short-term
experiments were injected daily for 3 weeks with the different
extracts, the glycemia being measured on day 7, 14 and 21 after
overnight starvation. The primary data collected in these long-term
experiments are listed in Table
II. The overall changes in glycemia over this 3-week period
were assessed by calculating, in each individual experiment, the
incremental (or decremental) area above (or below) the day zero
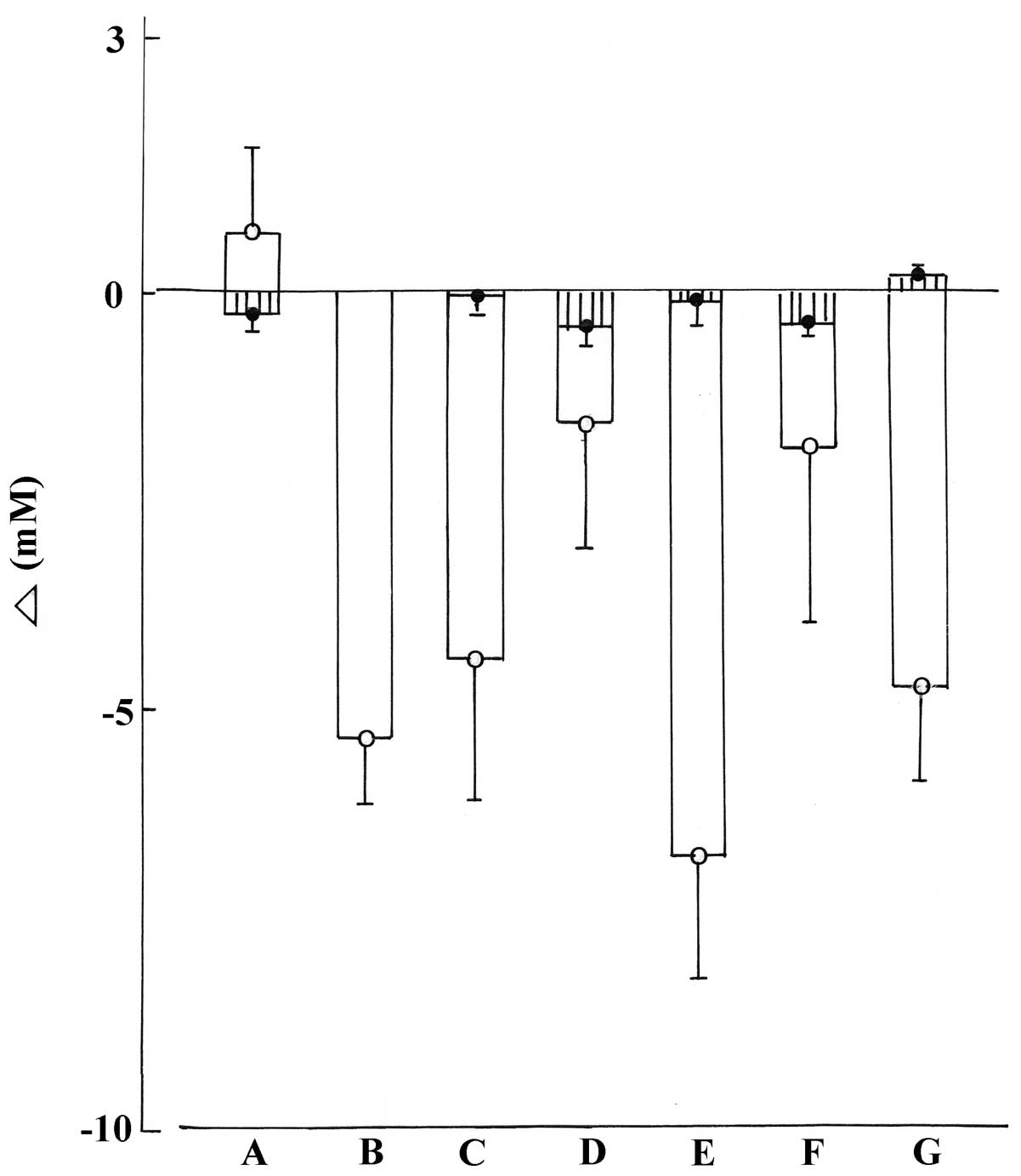
measurement. As documented in Fig.
2, in the normal rats, such incremental (or decremental) area
yielded mean values not significantly different (p>0.09 or more)
in the control animals and in the five groups of experimental
rats.
 | Table IIGlycemia (mM) in long-term
experiments. |
Table II
Glycemia (mM) in long-term
experiments.
| Rats | Extract | Day zero | Day 7 | Day 14 | Day 21 |
|---|
| Normal | Control | 5.43±0.28 (5) | 5.14±0.21 (5) | 5.07±0.21 (5) | 5.02±0.14 (5) |
| Untreated
aqueous | 5.34±0.10 (5) | 5.01±0.19 (4) | 5.43±0.07 (4) | 5.56±0.23 (3) |
| Defatted aqueous | 5.27±0.18 (5) | 4.62±0.27 (5) | 4.94±0.15 (5) | 4.43±0.24 (5) |
|
H2O-methanol | 5.02±0.34 (5) | 4.90±0.24 (5) | 4.70±0.29 (5) | 5.16±0.17 (5) |
| Ethyl acetate | 5.41±0.16 (5) | 4.83±0.25 (5) | 5.01±0.17 (5) | 4.92±0.11 (5) |
| n-butanol | 5.06±0.14 (5) | 5.17±0.15 (5) | 5.46±0.14 (5) | 5.16±0.34 (5) |
| STZ | Control | 21.32±1.26 (7) | 19.41±2.54 (7) | 25.19±2.11 (7) | 21.44±0.95 (7) |
| Glibenclamide | 22.01±1.04
(10) | 13.77±1.40 (6) | 14.17±3.04 (5) | 13.40±3.97 (5) |
| Untreated
aqueous | 19.03±0.55 (5) | 11.70±1.14 (5) | 13.09±3.03 (5) | 7.98±1.38 (5) |
| Defatted
aqueous | 20.00±0.82 (5) | 15.79±2.07 (5) | 18.14±2.85 (4) | 25.98±2.51 (3) |
|
H2O-methanol | 18.52±1.46 (5) | 15.94±1.76 (4) | 6.93±1.32 (4) | 7.02±1.84 (3) |
| Ethyl acetate | 17.06±2.50 (5) | 18.97±3.11 (5) | 10.91±5.33 (5) | 9.85±3.64 (4) |
| n-butanol | 16.38±0.26 (5) | 11.17±1.21 (5) | 10.98±1.65 (5) | 9.04±0.97 (5) |
As judged from the incremental (or decremental)
area, no significant difference (p>0.47) was observed between
the mean increment in glycemia over the 3 weeks period in the
control STZ rats (+0.68±1.04 mM; n=7) and the mean decrement in
glycemia over the same period in the control normal rats
(−0.28±0.23 mM; n=5). Except in the STZ rats injected with either
the defatted aqueous extract (p>0.22) or ethyl acetate extract
(p>0.26), the mean decreases in glycemia were significantly
different (p<0.007 or less) in the experimental STZ rats from
the mean increment recorded in the control STZ rats (Fig. 2).
Oral glucose tolerance test
(OGTT)
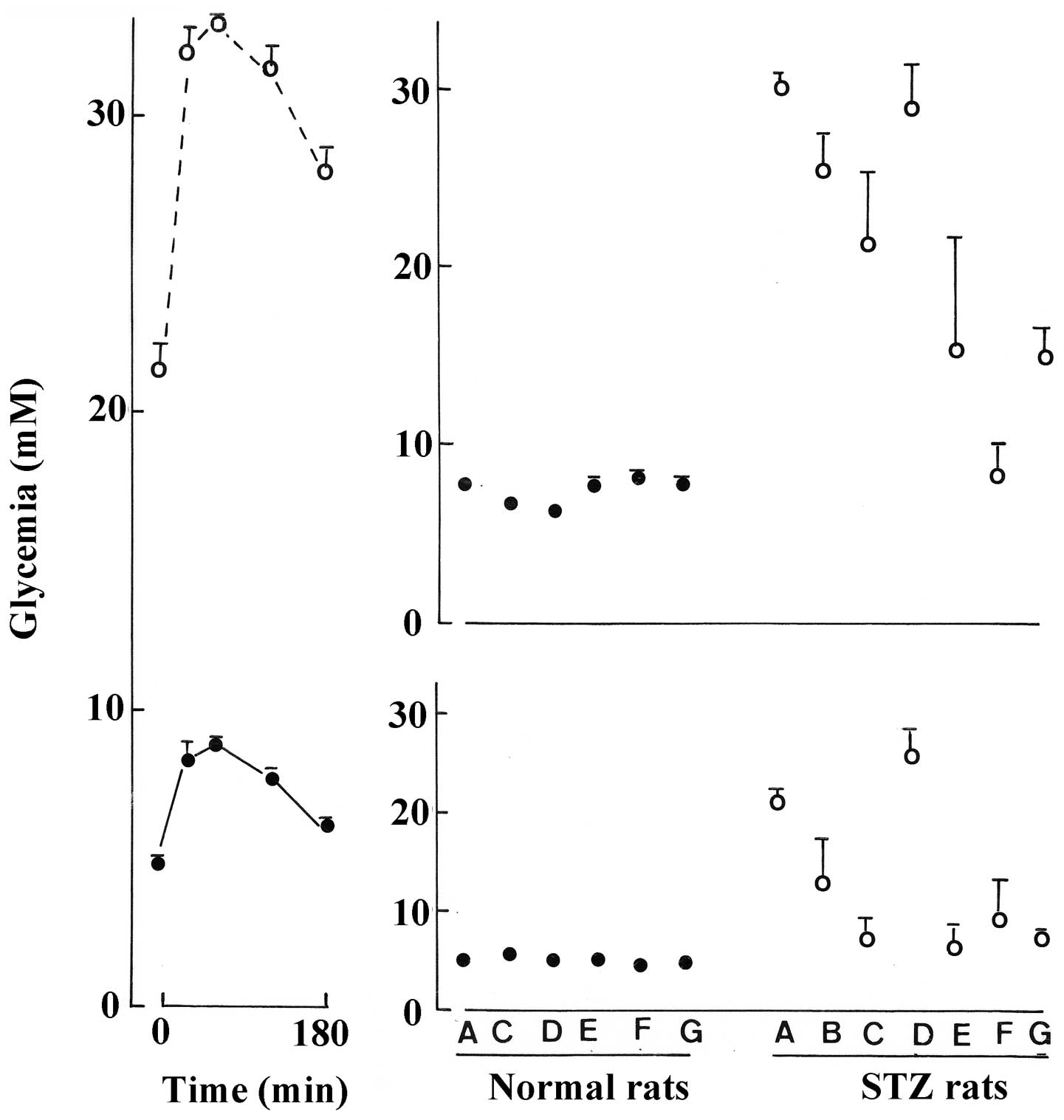
The vastly different pattern of the OGTT in the
control normal and STZ rats is illustrated in the left panel of
Fig. 3. In the normal rats, there
was little to distinguish between the 6 groups of animals in terms
of the time zero values for glycemia at the onset of the OGTT.
Nevertheless, the area under the curve (AUC) was significantly
lower in the normal rats injected with either the untreated aqueous
extract (p<0.01) or defatted aqueous extract (p<0.001) than
in the control normal rats. Such was not the case in the normal
rats injected with either the H2O-methanol extract
(p>0.56), ethyl acetate extract (p>0.61) or n-butanol extract
(p>0.73). In the seven groups of STZ rats, obvious differences
between control and experimental animals were observed whether for
the time zero values for glycemia or AUC during the OGTT. As a
matter of fact, there was a significant positive correlation (r=
+0.7912; n=7; p<0.04) between the mean values for the latter two
variables in the seven groups of STZ rats. Except in the STZ rats
injected with the defatted aqueous extract, the mean values for the
time zero glycemia were significantly lower (p<0.05 or less) in
the experimental groups of STZ rats than in the control STZ rats.
Likewise, except in the STZ rats injected with the defatted aqueous
extract, the mean values for the AUC during the OGTT were
significantly lower (p<0.02 or less) in the experimental groups
of STZ rats than in the control STZ rats (Fig. 3, right panels).
Body weight
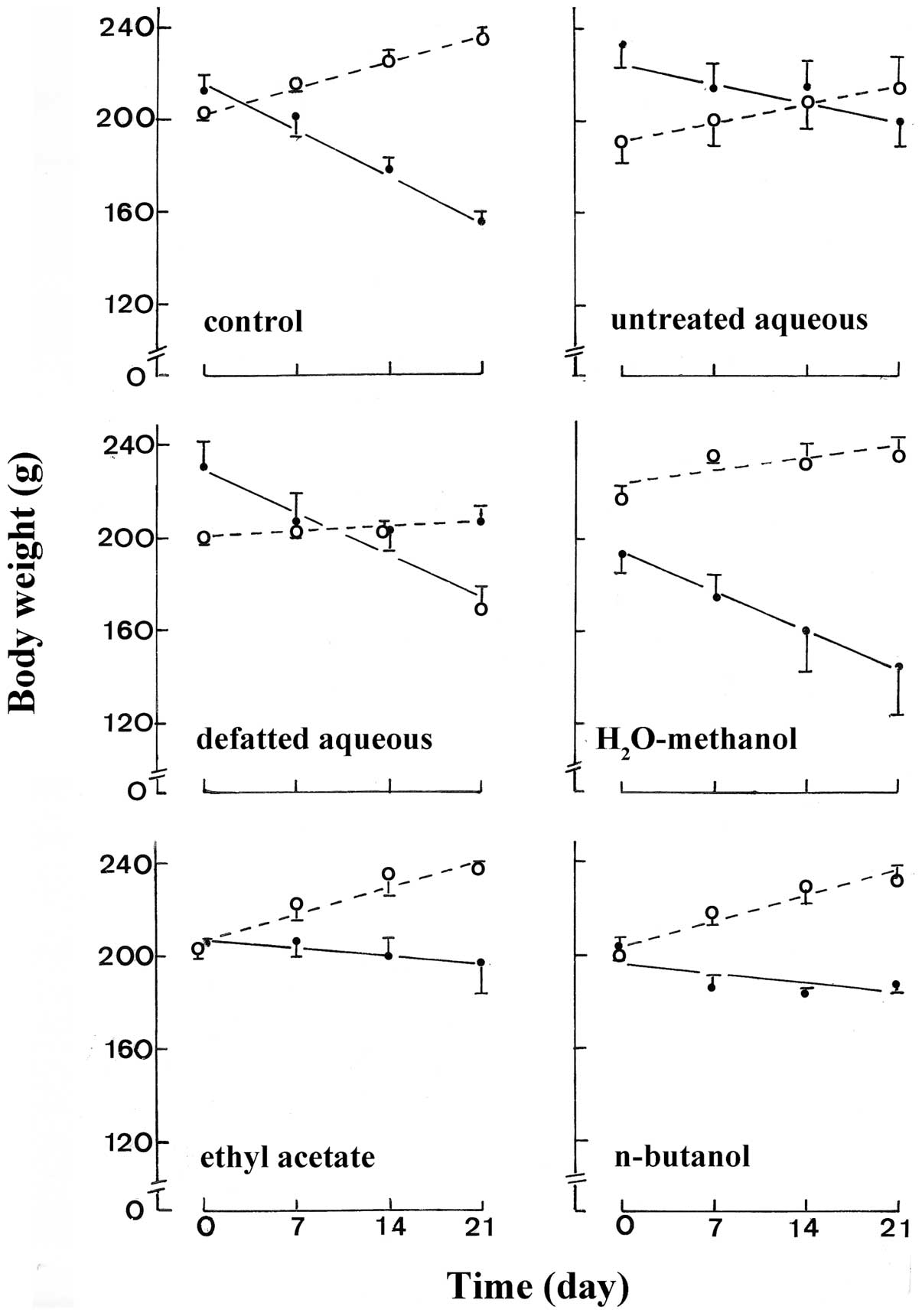
The body weight of the control normal rats
progressively increased (n=5 in all cases) from 201.5±1.8 g to
214.0±2.1 g, 226.3±3.6 g and 236.5±3.5 g, respectively one, two and
three weeks after the start of the experiments (Fig. 4). The day 21 minus day zero paired
difference in body weight failed to differ significantly in the
control normal rats (34.9±4.3 g; n=5) and the normal rats injected
with either the untreated aqueous extract (25.2±4.2 g; n=4), the
H2O-methanol extract (18.3±7.8 g; n=5), the ethyl
acetate extract (35.0±5.2 g; n=5) or the n-butanol extract
(32.2±7.3 g; n=5). Such a paired difference was significantly lower
(p<0.02), however, in the normal rats injected with the defatted
aqueous extract (7.5±7.5 g; n=5) than in the control normal
rats.
The body weight of the control STZ rats
progressively decreased (n=7 in all cases) from 211.6±8.2 g to
201.6±8.7, 177.0±7.5 and 155.8±4.9 g, respectively one, two and
three weeks after the onset of the experiments, i.e. one week after
the administration of STZ. The day 21 minus day zero paired
difference in body weight did not differ significantly in the
control rats (−55.8±4.7 g; n=7) and the STZ rats injected with
glibenclamide (−48.9±19.6 g; n=5). Likewise, such a decrease in
body weight also failed to differ significantly from that found in
the control STZ rats in the diabetic animals injected either with
the defatted aqueous extract (−63.3±6.8 g; n=3) or the
H2O-methanol extract (−42.3±13.8 g; n=3). However, the
same decrease in body weight did not exceed −33.7±6.8 g (n=5),
−6.5±10.6 g (n=4) and −15.0±4.9 g (n=5) in the STZ rats injected
with the untreated aqueous extract, ethyl acetate extract and
n-butanol extract, respectively, the latter three mean decreases in
body weight being significantly less marked (p<0.025 or less)
than that recorded in the control STZ rats.
Organs weight
As shown in Table
III, the mean wet weight of the pancreatic gland did not differ
significantly in the control normal rats and in the five other
groups of normal rats. Such was also the case in the STZ rats. A
significantly difference between normal and STZ rats (p<0.02)
was only observed in the animals injected with the
H2O-methanol extract. As a matter of fact, the
pancreatic wet weight averaged, in the STZ rats, 92.5±5.6% (n=22;
p>0.27) of the mean corresponding values (100.0±4.1%; n=29)
found under the same experimental conditions in the normal
animals.
 | Table IIITissue wet weight (mg). |
Table III
Tissue wet weight (mg).
| Rats | Extract | Pancreas | Liver | Kidneys | Testes | Epididymal fat | Diaphragm
muscle |
|---|
| Normal | Control | 733±51 (5) | 8,008±318 (5) | 1,512±100 (5) | 3,882±149 (5) | 2,322±296 (5) | 514±29 (5) |
| Untreated
extract | 575±91 (5) | 6,385±328 (5) | 1,025±33 (5) | 3,540±120 (5) | 605±42 (5) | 530±45 (5) |
| Defatted
aqueous | 754±78 (5) | 7,164±308 (5) | 1,200±27 (5) | 3,397±74 (5) | 608±40 (5) | 412±35 (5) |
|
H2O-methanol | 759±48 (5) | 10,943±720 (5) | 1,693±67 (5) | 3,920±72 (5) | 1,682±221 (5) | 699±67 (5) |
| Ethyl acetate | 730±90 (5) | 7,734±488 (5) | 1,571±120 (5) | 3,708±146 (5) | 2,800±332 (5) | 781±62 (5) |
| n-butanol | 647±73 (4) | 10,083±615 (4) | 1,549±85 (4) | 3,664±109 (4) | 1,625±417 (4) | 535±89 (4) |
| STZ | Control | 665±91 (5) | 7,686±516 (5) | 2,059±112 (5) | 3,336±350 (5) | 66±14 (5) | 270±77 (5) |
| Glibenclamide | 505±37 (5) | 7,419±379 (5) | 2,070±111 (5) | 3,675±231 (5) | 221±65 (5) | 457±42 (5) |
| Untreated
aqueous | 635±42 (4) | 8,493±201 (4) | 1,820±84 (4) | 2,743±142 (4) | 438±112 (4) | 580±57 (4) |
| Defatted
aqueous | 820±35 (3) | 8,297±415 (3) | 1,700±80 (3) | 3,247±113 (3) | 520±111 (3) | 405±15 (2) |
|
H2O-methanol | 415±115 (2) | 7,300±1,100
(2) | 2,440±540 (2) | 2,680±790 (2) | 820±30 (2) | 940±70 (2) |
| Ethyl acetate | 578±136 (4) | 7,595±860 (4) | 2,030±224 (4) | 2,755±546 (4) | 718±124 (4) | 888±93 (4) |
| n-butanol | 628±37 (4) | 6,820±363 (4) | 1,654±49 (4) | 3,139±253 (4) | 582±69 (4) | 574±54 (4) |
The wet weight of the liver was significantly lower
(p<0.01) in the normal rats injected with the untreated aqueous
extract than in the control normal rats, whilst being significantly
higher (p<0.02 or less) in the normal rats injected with either
the H2O-methanol or n-butanol extract than in the
control animals. In the STZ rats, none of the mean values found in
the six experimental groups differed significantly from the mean
control value.
In the normal rats, only those animals injected with
the untreated or defatted aqueous extract yielded a mean kidneys
wet weight significantly different from (p<0.02), i.e. lower
than, that found in the control animals. In the STZ rats, only
those animals injected with the n-butanol extract displayed a
significantly different (p<0.02), i.e. lower, mean kidney weight
than that found in the control STZ rats. A salient finding
consisted in the higher weight of the kidneys in the STZ rats than
in the normal rats. In the former STZ rats, it averaged 136.6±6.1%
(n=22; p<0.001) of the mean corresponding values found in the
normal rats injected with the same material (100.0±1.9%; n=29).
In the normal rats, the wet weight of the testes was
lower (p<0.01) in the rats injected with the defatted aqueous
extract than in the control rats. This was the sole significant
difference between control and experimental normal rats. No
significant difference was observed between the six mean values for
testes weight in the experimental STZ rats and the mean value
recorded in the control STZ rats. The testes weight of the STZ rats
only averaged 82.0±4.0% (n=22; p<0.001) of the mean
corresponding values found in normal rats injected with the same
material (100.0±1.2%; n=29).
The wet weight of the epididymal fat was much lower
(p<0.001) in the normal rats injected with either the untreated
or defatted aqueous extract than in the control normal rats. The
difference between the latter control normal rats and the other
three groups of normal rats failed to achieve statistical
significance. In the STZ rats, all experimental mean values were
higher (p<0.05 or less) than the mean control value. The wet
weight of the epididymal fat was dramatically lower (p<0.001) in
the control STZ rats (66±14 mg; n=5) than in the control normal
rats (2,322±296 mg; n=5). In the experimental groups, it also
remained lower in the STZ rats than in the normal animals, the mean
values recorded in the STZ rats averaging 52.3±7.5% (n=17;
p<0.001) of the mean corresponding values recorded in normal
rats injected with the same material (100.0±5.4%; n=24).
Last, the wet weight of the diaphragm muscle was
higher (p<0.05 or less) in the normal rats injected either with
the H2O-methanol or ethyl acetate extract than in the
control normal rats. Likewise, in the STZ rats, the diaphragm
weight was higher (p<0.05 or less) in the rats injected with
either the untreated aqueous extract, H2O-methanol,
ethyl acetate or n-butanol extract than in the control STZ rats. As
a matter of fact, the values recorded in each group of STZ rats
averaged 97.6±7.5% (n=21; p>0.76) of the corresponding mean
values recorded in normal rats (100.0±3.1%; n=29), documenting the
absence of any obvious difference between normal and STZ
animals.
Serum cholesterol
In the normal control rats, no significant
correlation (r= +0.037; n=20) was observed between the serum total
cholesterol concentration (g/l) and time (day zero, 7, 14 and 21).
Such was also the case in the normal rats injected with either the
defatted aqueous extract (r= +0.244; n=19; p>0.1),
H2O-methanol extract (r= +0.275; n=20; p>0.1), ethyl
acetate extract (r= −0.0776; n=19; p>0.1) or n-butanol extract
(r= +0.150; n=20; p>0.1). However, in the normal rats injected
with the untreated aqueous extract, a highly significant negative
correlation (r= −0.643; n=19; p<0.005) prevailed between the two
variables under consideration (Table
IV).
 | Table IVSerum cholesterol and triglyceride
concentrations. |
Table IV
Serum cholesterol and triglyceride
concentrations.
| Rats | Extract | Cholesterol (g/l)
|
| Day 0 | Day 7 | Day 14 | Day 21 |
|
| Normal | Control | 1.49±0.18 (5) | 1.39±0.19 (5) | 1.47±0.12 (5) | 1.50±0.16 (5) |
| Untreated
aqueous | 1.35±0.12 (5) | 1.17±0.29 (5) | 0.94±0.11 (5) | 0.90±0.08 (4) |
| Defatted
aqueous | 1.23±0.19 (5) | 1.26±0.06 (5) | 1.43±0.23 (5) | 1.42±0.12 (5) |
|
H2O-methanol | 1.29±0.20 (5) | 1.24±0.07 (5) | 1.19±0.18 (5) | 1.06±0.10 (5) |
| Ethyl acetate | 1.42±0.07 (5) | 1.63±0.18 (5) | 1.62±0.38 (5) | 1.28±0.11 (4) |
| n-butanol | 1.48±0.21 (5) | 1.25±0.21 (5) | 1.46±0.26 (5) | 1.43±0.26 (5) |
| STZ | Control | 2.24±0.07 (5) | 3.29±0.26 (5) | 3.67±0.44 (5) | 3.55±0.13 (5) |
| Glibenclamide | 2.07±0.25 (10) | 2.33±0.47 (6) | 2.09±0.50 (5) | 2.09±0.61 (5) |
| Untreated
aqueous | 1.85±0.04 (5) | 1.26±0.16 (5) | 1.14±0.18 (5) | 1.08±0.11 (4) |
| Defatted
aqueous | 1.51±0.15 (5) | 1.44±0.06 (5) | 1.63±0.09 (4) | 1.62±0.06 (3) |
|
H2O-methanol | 2.16±0.28 (5) | 2.01±0.24 (4) | 1.18±0.03 (4) | 0.94±0.02 (2) |
| Ethyl acetate | 2.95±0.26 (5) | 3.18±0.18 (5) | 3.12±0.36 (5) | 3.00±0.39 (5) |
| n-butanol | 1.92±0.24 (5) | 1.62±0.21 (5) | 1.61±0.14 (5) | 1.61±0.38 (4) |
|
| Rats | Extract | Triglycerides (g/l)
|
| Day 0 | Day 7 | Day 14 | Day 21 |
|
| Normal | Control | 1.52±0.10 (5) | 2.33±0.10 (5) | 2.41±0.21 (5) | 2.52±0.23 (5) |
| Untreated
aqueous | 1.40±0.12 (5) | 1.22±0.13 (5) | 0.93±0.14 (5) | 0.59±0.10 (4) |
| Defatted
aqueous | 1.45±0.14 (5) | 1.12±0.13 (5) | 1.43±0.21 (5) | 1.44±0.13 (5) |
|
H2O-methanol | 1.32±0.10 (5) | 1.22±0.10 (5) | 1.40±0.11 (5) | 1.27±0.15 (5) |
| Ethyl acetate | 1.65±0.16 (5) | 1.62±0.10 (5) | 1.44±0.14 (5) | 1.56±0.25 (4) |
| n-butanol | 1.23±0.08 (5) | 1.10±0.09 (5) | 1.18±0.12 (7) | 1.40±0.19 (4) |
| STZ | Control | 2.09±0.21 (5) | 2.18±0.19 (5) | 1.46±0.18 (5) | 1.58±0.17 (5) |
| Glibenclamide | 1.49±0.11 (10) | 1.38±0.20 (6) | 1.11±0.11 (5) | 0.91±0.03 (5) |
| Untreated
aqueous | 1.53±0.35 (5) | 1.21±0.18 (5) | 1.02±0.19 (5) | 1.03±0.16 (4) |
| Defatted
aqueous | 1.58±0.15 (5) | 1.16±0.13 (5) | 0.90±0.12 (5) | 1.07±0.08 (3) |
|
H2O-methanol | 1.86±0.06 (5) | 1.04±0.01 (4) | 0.82±0.06 (4) | 0.67±0.05 (2) |
| Ethyl acetate | 2.76±0.36 (5) | 2.63±0.18 (4) | 1.95±0.16 (4) | 2.20±0.10 (3) |
| n-butanol | 1.58±0.17 (5) | 1.36±0.14 (5) | 1.48±0.18 (5) | 1.90±0.18 (4) |
In the control STZ rats, the serum total cholesterol
contribution reached between day 7 and day 21 an equilibrium value
(3.62±0.16; n=15) significantly higher (p<0.001) than that
recorded on day zero (2.24±0.07; n=5). The latter value was already
significantly higher (p<0.005) than that found at the same time
in control normal rats. The injection of either glibenclamide, the
defatted aqueous extract, the ethyl acetate extract or the
n-butanol extract to the STZ rats prevented the further rise in
cholesterol concentration otherwise observed in the control STZ
rats. Thus, in the former three groups of rats, no significant
difference was observed between the day zero values and those
recorded thereafter. A significant decrease in cholesterol
concentration was even observed over the same 3 weeks period in the
STZ rats injected either with the untreated aqueous extract (r=
−0.6738; n=19; p<0.003) or H2O-methanol extract (r=
−0.7450; n=15; p<0.003).
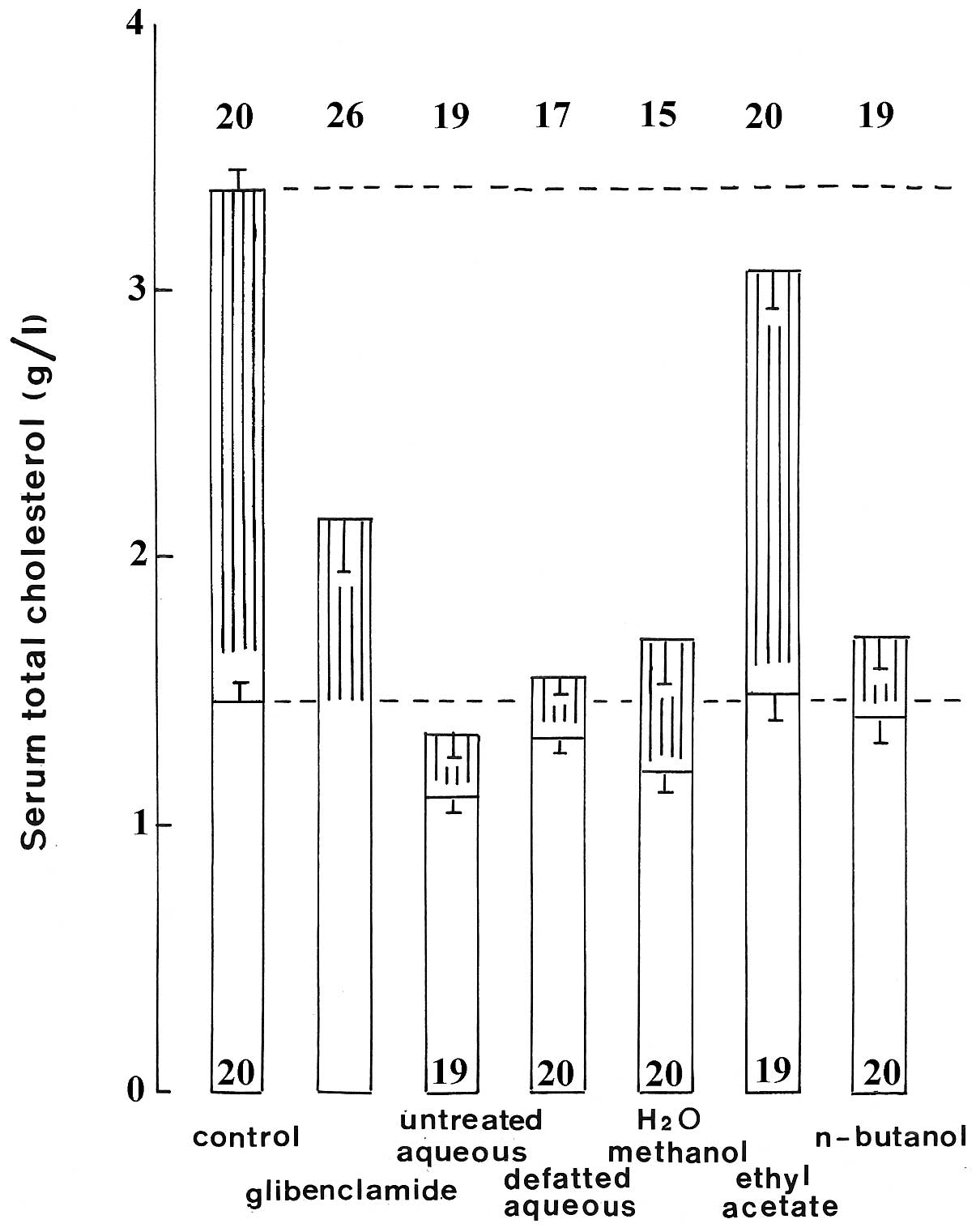
Fig. 5 provides a
further illustration of the beneficial effects of the various
extracts, as well as glibenclamide, on cholesterol concentration in
STZ rats. It refers to the mean of all available measurements made
between day zero and day 21 in either normal or STZ rats, and
emphasizes the close-to-normalization found in STZ rats injected
with the untreated or defatted aqueous extracts,
H2O-methanol and n-butanol extracts.
Serum triglycerides
In the control normal rats, the serum triglyceride
concentration (g/l) increased (p<0.001) from an initial value of
1.52±0.10 (n=5) on day zero to a plateau value averaging 2.41±0.10
(n=15) during the period of 3 weeks during which these rats were
injected daily with saline. Such an increase was not observed in
the normal rats injected with either the defatted aqueous extract,
H2O-methanol extract, ethyl acetate extract or n-butanol
extract (Table IV). Thus, in the
latter four groups, no significant change in serum triglyceride
concentration was detected over the same period of 3 weeks. In the
normal rats injected with the untreated aqueous extract, a
progressive decrease in triglyceride concentration was even
observed (r= −0.506; n=19; p<0.04).
In the control STZ rats, the triglyceride
concentration decreased over the 3 weeks period (r= −0.510; n=20;
p<0.03). Except in the STZ rats injected with the n-butanol
extract, in which no significant change was recorded over the 3
weeks period (r= +0.279; n=19; p>0.1), the triglyceride
concentration decreased during the same period, whether in the STZ
rats injected with glibenclamide (r= −0.571; n=24; p<0.04), with
the untreated or defatted aqueous extract (r= −0.458; n=37;
p<0.007), with the H2O-methanol extract (r= −0.912;
n=15; p<0.001) or with the ethyl acetate extract (r= −0.490;
n=16; p<0.06). Moreover, except in the STZ rats injected with
either the ethyl acetate or n-butanol extract, the mean values
recorded at each time point were always lower in the experimental
STZ rats than in the control STZ rats (Table IV).
On day zero, i.e. one week after the administration
of STZ, the serum triglyceride concentration averaged in the STZ
rats 1.80±0.10 (n=40), as distinct (p<0.005) from 1.43±0.05
(n=30) in the normal animals. The patterns of changes recorded
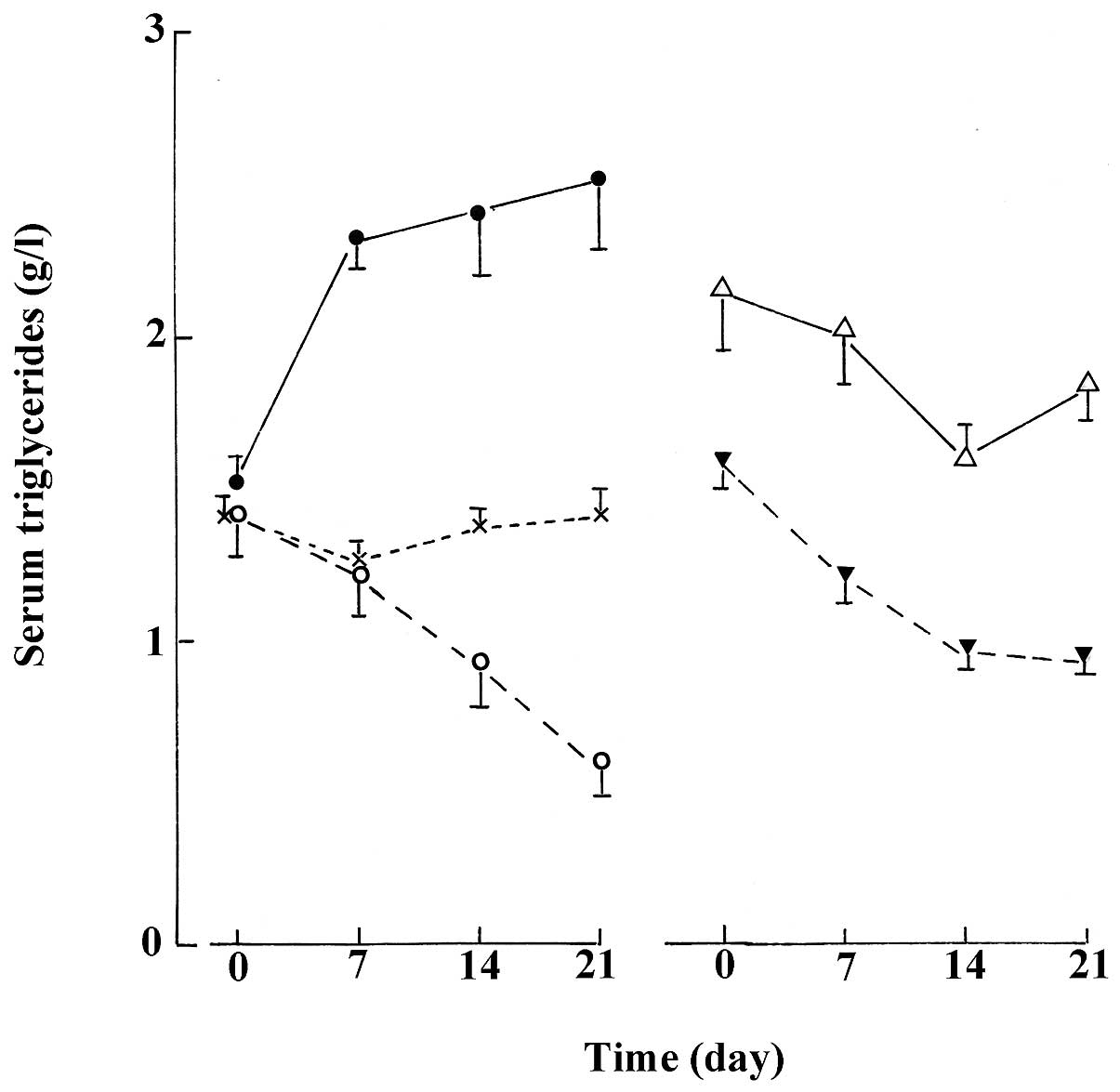
thereafter are illustrated in Fig.
6. For instance, in the normal animals, it compares the control
group, the rats injected with the untreated aqueous extract, which
displayed a progressive decrease in triglyceride concentration, and
the four other experimental groups, in which no obvious change in
triglyceride concentration was observed. Likewise, in the STZ rats,
Fig. 6 compares those groups of
rats in which no obvious change in triglyceridemia was observed
over the 3-week period, to those groups of rats which displayed a
sizeable lowering of triglyceridemia during the same period.
Other serum variables
The serum concentrations of urea, creatinine,
glutamate-pyruvate transaminase (TGP), glutamateoxaloacetate
transaminase (TGO) and alkaline phosphatase was also measured at
the onset (day zero) and end (day 21) of the 3 weeks experimental
period (Table V). Two findings
merit to be underlined. First, in the normal rats injected with the
untreated aqueous extract, the TGP value (UI/l) decreased
(p<0.003) from 1,123±44 (n=5) on day zero to 853±31 (n=4) on day
21. Second, in the STZ rats, a striking increase (p<0.001) in
creatinine concentration (mg/dl) was observed in both control and
experimental animals from a mean value of 19.3±0.8 (n=35) at day
zero to 29.6±1.1 (n=35) on day 21.
 | Table VSerum urea, creatinine,
glutamate-pyruvate (TGP) and glutamate-oxaloacetate (TGO)
transaminase and alkaline phosphatase concentrations. |
Table V
Serum urea, creatinine,
glutamate-pyruvate (TGP) and glutamate-oxaloacetate (TGO)
transaminase and alkaline phosphatase concentrations.
| Rats | Extracts | Urea (mg/dl)
| Creatinine (mg/dl)
| TGP (UI/l)
| TGO (UI/l)
| Phosphatase
alcaline (UI/l)
|
|---|
| Day 0 | Day 21 | Day 0 | Day 21 | Day 0 | Day 21 | Day 0 | Day 21 | Day 0 | Day 21 |
|---|
| Normal | Control | 62.2±5.9 (5) | 59.6±9.2 (5) | 17.8±1.5 (5) | 15.8±1.1 (5) | 1,133±37 (5) | 1,221±39 (4) | 1,215±51 (5) | 1,265±19 (4) | 1,217±29 (5) | 1,236±47 (3) |
| Untreated
aqueous | 50.1±7.2 (5) | 61.1±6.6 (4) | 16.6±0.7 (5) | 18.1±0.7 (5) | 1,123±44 (5) | 853±31 (4) | 1,071±75 (4) | 1,097±49 (5) | 1,161±75 (5) | 1,051±57 (5) |
| Defatted
aqueous | 50.6±4.3 (5) | 54.0±4.3 (5) | 17.1±2.1 (5) | 20.9±2.6 (5) | | | | | | |
|
H2O-methanol | 51.3±6.6 (5) | 52.2±5.9 (5) | 19.3±3.5 (5) | 19.3±3.1 (5) | | | | | | |
| Ethyl acetate | 50.0±5.9 (5) | 48.0±5.5 (5) | 17.6±1.4 (5) | 17.2±1.4 (5) | 1,129±29 (5) | 1,261±38 (3) | 1,160±22 (5) | 1,198±53 (3) | 1,223±39 (5) | 1,188±66 (3) |
| n-butanol | 49.7±1.4 (5) | 59.4±6.5 (5) | 18.9±2.9 (5) | 18.4±1.0 (5) | | | | | | |
| STZ | Control | 50.4±8.3 (7) | 62.5±4.7 (7) | 16.9±1.2 (5) | 23.1±1.6 (5) | 1,152±27 (5) | 1,118±12 (5) | 1,113±37 (5) | 1,044±28 (5) | 1,152±38 (5) | 1,269±94 (5) |
| Glibenclamide | 49.0±2.9 (10) | 58.7±4.3 (4) | 15.6±1.0 (5) | 26.8±3.7 (5) | 1,230±33 (5) | 1,097±41 (4) | 1,164±50 (5) | 1,092±44 (3) | 1,202±121 (5) | 1,387±176 (3) |
| Untreated
aqueous | 57.6±4.5 (5) | 54.2±3.3 (5) | 19.5±0.5 (5) | 31.1±4.3 (5) | 1,083±56 (5) | 990±30 (3) | 1,079±57 (5) | 1,106±25 (4) | 1,229±67 (5) | 1,286±116 (4) |
| Defatted
aqueous | 50.1±6.0 (5) | 55.8±4.0 (3) | 18.2±0.9 (5) | 33.5±2.6 (5) | | | | | | |
|
H2O-methanol | 51.6±3.4 (5) | 50.1±3.9 (3) | 19.0±1.3 (5) | 27.7±2.8 (5) | | | | | | |
| Ethyl acetate | 49.7±1.3 (5) | 58.6±5.4 (4) | 25.0±4.1 (5) | 33.0±1.3 (5) | 1,071±27 (5) | 999±26 (3) | 1,037±27 (5) | 1,121±61 (3) | 1,098±37 (5) | 1,029±112 (3) |
| n-butanol | 54.2±5.4 (5) | 49.7±5.4 (5) | 20.8±3.4 (5) | 31.6±3.1 (5) | | | | | | |
Discussion
In addition to the expected and converging
indications of a severe perturbation of glucose homeostasis in the
control STZ rats, when compared to the control normal rats and as
documented in Tables I and
II, as well as in Fig. 3 (left panel), several other
striking differences were observed between these two groups of
rats. For instance, the control, as well as the experimental,
normal rats displayed a progressive increase in body weight, with
the opposite situation in control, as well as experimental, STZ
rats (Fig. 4). Likewise, the wet
weight of the epididymal fat and, to a lesser relative extent that
of the testes, was lower in the STZ rats than in the normal
animals. Inversely, however, the wet weight of the kidney was
higher in the STZ rats than in the normal rats. This coincided with
a rise in serum creatinine concentration between day zero and day
21 from 19.3±0.8 to 29.6±1.1 mg/dl (n=35 in both cases; p<0.001)
in the STZ rats, whilst mean values of 17.9±0.8 and 18.3±0.7 mg/dl
(n=30 in both cases; p>0.69) were recorded on the same days in
the normal animals. Last, at the onset of the present experiments,
both the serum cholesterol and triglyceride concentrations were
higher (p<0.005 or less) in STZ rats than in normal animals,
with respective mean values of 2.10±0.10 and 1.80±0.10 g/l (n=40 in
both cases) in STZ rats, as distinct from 1.37±0.07 and 1.43±0.05
g/l (n=30 in both cases) in normal animals.
As a rule, there was little to distinguish, in the
normal rats, between control and experimental animals. This is
illustrated in Figs. 1, 2 and 3, which all refer to variables relevant
to glucose homeostasis. Likewise, the data illustrated in Fig. 4 indicate that the sole significant
effect of a Citrullus colocynthis L. extract, i.e. the
defatted aqueous extract, was to oppose the gain in body weight
otherwise recorded in the normal rats. The latter extract also
decreased, in the normal rats, the wet weight of the liver, kidney,
testes and epididymal fat. A modest beneficial effect was observed
in the normal rats injected with the untreated aqueous extract,
with a reduction of the AUC during the OGTT, of the epididymal fat
weight, and of the serum cholesterol, triglyceride and
glutamate-oxaloacetate transaminase concentration, when compared to
the corresponding values found in the control normal rats.
In contrast to the situation found in normal rats,
the daily administration of Citrullus colocynthis L. seed
extracts often exerted beneficial effects in the STZ rats. For the
purpose of comparison with the plant extracts, the experiments
conducted in STZ rats also included the administration of
glibenclamide. When compared to control STZ rats, the
glibenclamide-injected STZ rats displayed, as expected, lower
values of glycemia in the short-term experiments, as well as in the
long-term experiments and during the OGTT. Moreover, the daily
administration of glibenclamide prevented the increase in serum
cholesterol concentration otherwise found in control STZ rats, and
resulted in a higher epididymal fat weight than that found in the
latter control STZ rats.
The beneficial effects of the plant extracts in the
STZ rats included lower glycemia in the short-term experiments,
long-term experiments and during the OGTT, lesser decrease in body
weight, increase in epididymal fat weight, prevention of a
progressive increase in serum cholesterol concentration and lower
serum triglyceride concentration, when comparing the control STZ
rats to those injected with a Citrullus colocynthis extract.
When the relative magnitude of such beneficial effects was
expressed according to the number zero (no significant effect), one
(modest effect) and two (marked effect), the overall mean score for
the seven variables under consideration in each group of
experimental STZ rats yielded the highest value in the STZ rats
injected with the untreated aqueous extract (1.14±0.14) or
n-butanol extract (1.14±0.26) and the lowest value (0.57±0.20) in
the STZ rats injected with the defatted aqueous extract. Whilst the
latter value was indeed significantly lower (p<0.05) than that
recorded with the untreated aqueous extract, such was not the case
for the in-between values found in the STZ rats injected with
either the H2O-methanol extract (1.00±0.22) or ethyl
acetate extract (0.86±0.26). The difference observed in the STZ
rats between the untreated and defatted aqueous extract is
consistent with the findings made in the normal animals, in which
the untreated aqueous extract also exerted selected beneficial
effects, whilst the defatted aqueous extract caused several
undesirable effects.
In conclusion, the present study extends to distinct
extracts from Citrullus colocynthis L. seeds, and to a
number of distinct biological variables, the view that further
investigations dealing mainly with the identification of the most
active components might eventually lead to the selection and
development of a suitable agent in the perspective of the treatment
of diabetic subjects, including both type-1 and type-2 diabetic
patients.
Acknowledgements
We are grateful to Professor A. Sener
for help and hospitality in the Laboratory of Experimental
Hormonology (Université Libre de Bruxelles), and to C Demesmaeker
for secretarial help.
References
|
1.
|
AB ZareeF FallahhossiniR SharifabadyA
Norooz zadehH EmaniH GhoshooniThe effect of Citrullus
colocynthis L. extract on preventing/reducing
streptozotocin-induced diabetes in ratKawsar Med J1213202007
|
|
2.
|
N BenaribaR DjaziriBH ZerriouhK BoucheritK
LouchamiA SenerWJ MalaisseAntihyperglycemic effect of Citrullus
colocynthis seed aqueous extracts in streptozotocin-induced
diabetic ratsMet Funct Res Diab271762009
|
|
3.
|
R JayaramanA ShivakumarT AnithaVD JoshiNN
PaleiAntidiabetic effect of petroleum ether extract of Citrullus
colocynthis fruits against streptozotocin-induced hyperglycemic
ratsRom J Biol-Plant Biol541271342009
|
|
4.
|
M Al-KhateebM DallakF Al-HashemN BashirM
AbbasR ElessaM KhalilIn vivo, acute, normo-hypoglycemic,
antihyperglycemic, insulinotropic actions of orally administered
ethanol extract of Citrullus colocynthis (L.) schrab pulpAm
J Biochem Biotech5118125200910.3844/ajbbsp.2009.118.125
|