Introduction
Hepatitis B virus (HBV) infection remains a global
health concern and is associated with a great economic burden,
affecting approximately 400 million people worldwide despite
decades of research. HBV infection leads to a broad spectrum of
clinical manifestations, including cirrhosis, fulminant hepatic
failure and hepatocellular carcinoma (1). Almost 20% of chronic HBV patients
(approximately 2.6 million) in China alone developed cirrhosis and
hepatocellular carcinoma. Chronic HBV infection is a consequence of
complex interactions between the replicating noncytopathic virus
and the host immune system, which plays a critical role in viral
clearance. Deficient host immunity subsequently augments the
chronic status (2).
Host antiviral innate immunity is initiated via
viral pathogen-associated molecular patterns (PAMP), such as viral
double-stranded RNA (dsRNA). All RNA and most DNA viruses,
including HBV (3), generate dsRNA
molecules at the transcription or replication level (4,5).
Retinoic acid-inducible gene I (RIG-I), a cytoplasmic RNA helicase,
plays an essential role in host antiviral immunity by activating
type-I interferon (IFN-α/β) (6,7) in
response to intracellular viral RNA recognition. The RIG-I
antiviral response is cell-type specific (8) and is enhanced by type-I interferon
following an interaction between TLR7-9 and their ligands (9). RIG-I mediated antiviral immunity and
its downstream signaling pathway have been well illustrated in HCV
infection (10). Interferon-β
(IFN-β) production is suppressed in the liver following viral
invasion, which directly represses RIG-I and its downstream adaptor
IFN-β promoter stimulator 1 (IPS-1) (11). However, it is unclear whether the
RIG-I-mediated signaling pathway contributes to host antiviral
immunity in HBV. In this study, we investigated the correlation
between RIG-I expression and downstream signaling in
monocyte-derived dendritic cells (moDCs) from HBV-infected patients
as well as in HBV-transformed cell lines in response to vesicular
stomatitis virus (VSV) challenge. This insight may lead to basic
research and/or clinical treatment of this disease.
Materials and methods
HBV patients and healthy subjects
Chronic hepatitis B patients (CHB; n=21) and acute
hepatitis B patients (AHB; n=7) were recruited for the current
study with the following criteria: all patients were HBV surface
antigen (HBsAg) positive but negative for other viruses (e.g., HCV,
HDV and HIV) and treatment-naïve for 6 months prior to sample
collection. Age-, gender- and ethnic background-matched
HBsAg-negative healthy controls (HCs; n=18) were selected. The
clinical characteristics of patients are listed in Table I. Informed consent was obtained
from all subjects. All experimental procedures adhered to the
guidelines of the Helsinki Declaration of 1975 and were approved by
the Human Ethics Committee, Shanghai Jiaotong University School of
Medicine.
 | Table ICharacteristics of patients. |
Table I
Characteristics of patients.
| Groups | Age | Gender | HBV DNA (log10
copies/ml) | ALT | AST |
|---|
| CHB | 39.47±2.84 | 14/7 | 6.74±0.24 | 370.18±95.34 | 217.94±64.91 |
| AHB | 40.14±3.37 | 4/3 | 2.04±0.11 | 1053.00±261.44 | 550.00±186.02 |
| HC | 37.67±2.71 | 11/7 | - | - | - |
| P-value | 0.843a | 0.594a | <0.001 | 0.005 | 0.043 |
Virological assessment
Serum HBsAg, HBeAg and anti-HBe were determined
using commercial enzyme immunoassay kits (AXSYM System; Abbott,
Wiesbaden, Germany). HBV DNA levels were quantified by the Applied
Biosystems PCR system (Prism 7500; Applied Biosystems, Inc., USA)
with the lowest detection limit at 1000 copies/ml as previously
described (12,13).
Monocyte collection and moDC
induction
Peripheral blood mononuclear cells (PBMC) were
isolated from 20 ml of heparinized peripheral blood by
Ficoll-Hypaque gradient centrifugation. Monocytes were obtained
using a MACS CD14 isolation kit (Miltenyi Biotec, Bergisch
Gladbach, Germany) and >90% purity was confirmed with flow
cytometry. The purified monocytes were seeded in 6-well plates
(1.5×106 cells/ml) in 2 ml RPMI-1640 supplemented with
10% heat-inactivated 10% fetal calf serum, 2 mmol/l L-glutamine,
100 U/ml penicillin and 100 μg/ml streptomycin. moDCs were
generated by adding GM-CSF (500 U/ml) and IL-4 (250 U/ml)
(Peprotech, Inc., Rocky Hill, NJ, USA) in the culture medium for 7
days as previously described (14). The moDCs obtained were challenged
with wild-type VSV (15) at a
multiplicity of infection (MOI) of 0.1 for activation of
RIG-I-mediated signaling.
RIG-I transfection and VSV challenge
VSV was propagated in Wish cells and quantified by
standard plaque assay. HepG2 (hepatoma cell line) and HepG2.2.15
cells (hepatoma cell line transformed with HBV) were transfected
with RIG-I plasmids (16) by
Lipofectamine™ 2000 (Invitrogen Life Technologies, Shanghai, China)
in DMEM culture medium for 24 h, following the manufacturer’s
instructions. The RIG-I-transfected cells were cultured with fully
supplemented growth medium for 0, 8, 16 and 24 h, after a half-hour
challenge with VSV (17) at 0.1
MOI in free serum medium. IFN-β production in the challenged cells
and supernatants was determined by qRT-PCR and ELISA.
RNA isolation, reverse transcription and
quantitative real-time PCR
RNA was extracted with an RNAprep pure Micro kit
(Tiangen Biotech Co., Ltd., Beijing, China). Total-RNA (0.5
μg) was reverse transcribed with the SuperScript
first-strand synthesis system for RT-PCR (Invitrogen Life
Technologies). RNA expression was quantified by SYBR-Green-based
qRT-PCR using a sequence detection system (Prism 7500; Applied
Biosystems, Inc.) with GAPDH serving as an endogenous control. The
sequences of gene-specific primers are listed in Table II. The threshold cycle (Ct) for
each gene was determined as the cycle number at which the reaction
crossed an arbitrarily placed threshold, and the amount of each
target gene relative to GAPDH was described using the formula
2−ΔCt where ΔCt = (Cttarget
gene−CtGAPDH).
 | Table IISequences of primers used for PCR or
qPCR of mRNA. |
Table II
Sequences of primers used for PCR or
qPCR of mRNA.
| Gene name | Primer sequences
(5′→3′) |
|---|
| RIG-I | Sense |
TGTGCTCCTACAGGTTGTGGA |
| Antisense |
CACTGGGATCTGATTCGCAAAA |
| IPS-1 | Sense |
CAGCAAGAGACCAGGATCGAC |
| Antisense |
CGTCCGCGAGATCAACTAGC |
| INF-β | Sense |
GCGACACTGTTCGTGTTGTCA |
| Antisense |
CCAAGCAAGTTGTAGCTCATGGA |
| GAPDH | Sense |
GAAGGTGAAGGTCGGAGTC |
| Antisense |
GAAGATGGTGATGGGATTTC |
Western blot analysis
HepG2 and HepG2.2.15 cells with different treatments
were homogenized in RIPA buffer (20 mM Tris, pH 7.4, 2.5 mM EDTA,
1% Triton X-100, 10% glycerol, 1% deoxycholic acid, 0.1% SDS, 1 mM
PMSF, 10 ng/ml aprotinin). Western blot analyses were performed as
previously described (18).
Briefly, extracted protein (50 μg) was resolved by SDS-PAGE
(10% gel) and transferred to nitrocellulose membranes. The blots
were blocked and labeled with primary antibodies [rabbit anti-human
RIG-I, 1:2,000 (Biodesign, USA); or mouse anti-human β-actin
1:5,000 (Sigma, USA)] overnight. Membranes were incubated with the
secondary antibody (goat anti-rabbit-HRP or goat anti-mouse-HRP,
1:5,000 each; Amersham Pharmacia Biotech, Saclay, France). The
labeled proteins were detected using the ECL Plus kit (Amersham
Pharmacia Biotech).
Enzyme-linked immunosorbent assay
The HepG2 and HepG2.2.15 cell supernatant IFN-β
concentrations were determined using an ELISA kit (R&D Systems,
Minneapolis, MN, USA) at different time points following various
treatments.
Statistical analysis
All data are expressed as the mean ± standard
deviation (SD). Comparisons between the 2 groups were made using
the Student’s t-test. Non-parametric statistical analysis was
performed using the Mann-Whitney U-test between 2 independent
groups with SPSS 13.0 for Windows (SPSS, Inc., Chicago, IL, USA).
Spearman’s rho was used for correlation analysis. Differences were
considered statistically significant at P<0.05.
Results
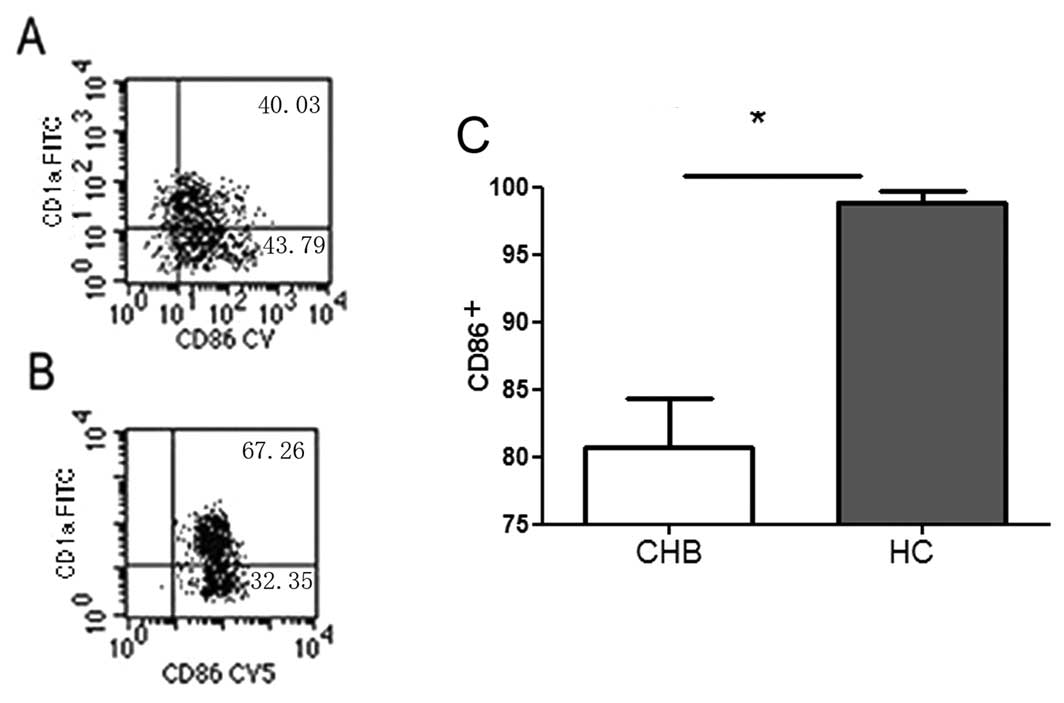
Reduced maturity of moDCs in HBV
patients
Mature CD86+ moDCs derived from the
VSV-stimulated and GM-CSF/IL-4-stimulated CD14+
monocytes were confirmed by flow cytometry (Fig. 1). The proportion of
CD86+ moDC cells from CHB patients was 17% lower than in
HCs (80.77±8.51 vs. 96.89±2.12%, P<0.05).
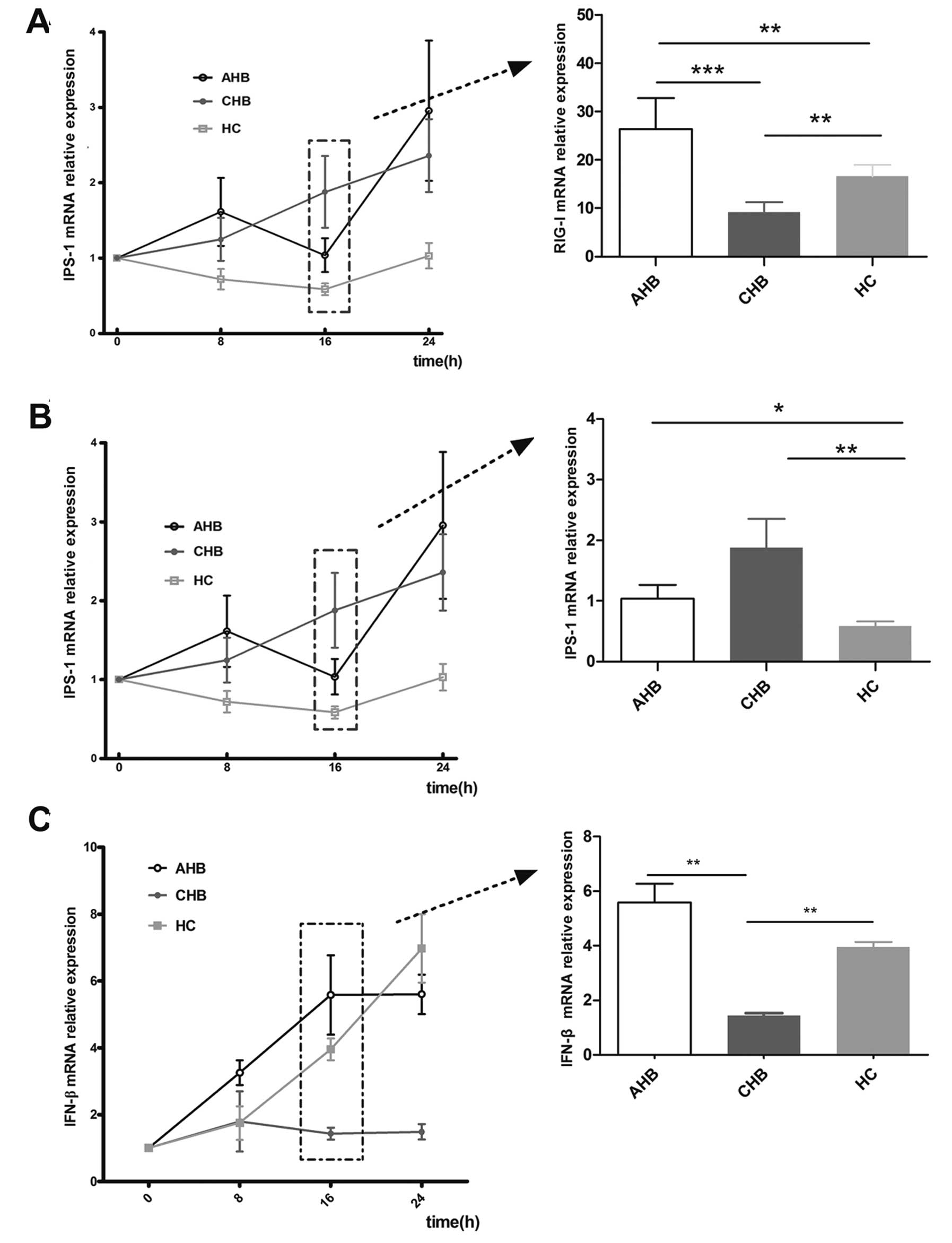
Expression of RIG-I, IPS-1 and IFN-β mRNA
in the moDC
The expression of RIG-1 mRNA was elevated ∼9-fold at
8 h post-VSV stimulation in all 3 groups (CHB, AHB and HC; Fig. 2) compared to their unchallenged
counterparts. No significant difference was observed among these 3
groups in the absence of stimulation. RIG-I expression peaked at 16
h in the CHB, AHB and HC groups, at which point RIG-I expression
was increased 9-, 27- and 17-fold relative to the unstimulated
counterparts, respectively (P<0.01). A similar pattern was
observed at 24 h.
Expression of IPS-1 was induced in the moDCs of both
AHB and CHB patients following VSV challenge, but not in the HCs
(Fig. 2B). No significant
induction was observed at 8 h post-challenge. However, at 16 and 24
h post-stimulation the level of IPS-1 induction in AHB moDCs was
1.1- and 2.9-fold higher than the unstimulated controls and the
level of IPS-1 induction in CHB patients was 1.8- and 2.4-fold
higher than the unstimulated controls.
Following VSV stimulation, IFN-β expression was
detected in the moDCs of AHB and CHB patients as well as HCs at
various time points. There were no significant changes observed in
the moDCs from CHB patients at various time points. However, IFN-β
expression in the moDCs from AHB patients was induced 3.3-, 5.8-
and 5.6-fold at 8, 16 and 24 h compared with matched unstimulated
moDCs (Fig. 2C). IFN-β expression
in the moDCs from HCs was 1.7-, 4.3- and 7.2-fold higher than
unstimulated controls at 8, 16 and 24 h. Notably, IFN-β expression
in the moDCs from CHB patients at 16 and 24 h was significantly
lower than from the other 2 groups (P<0.05).
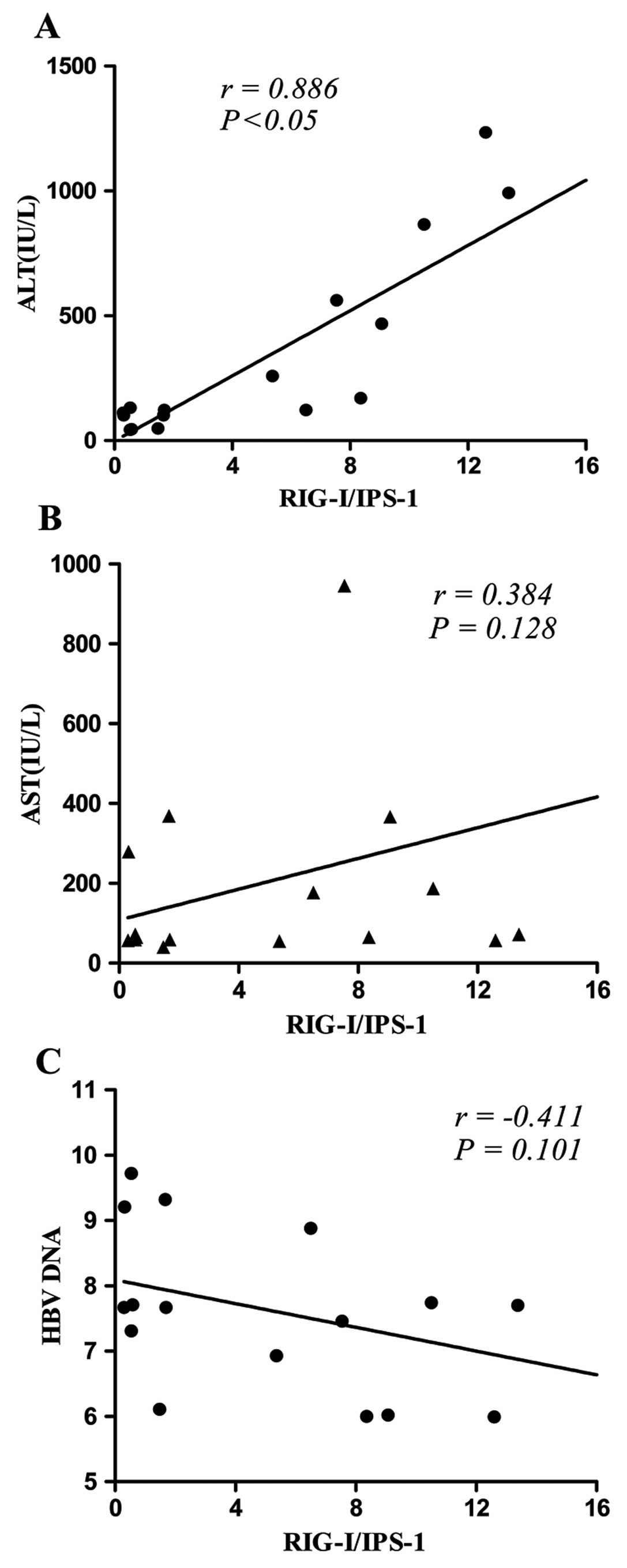
Correlation between RIG-I/IPS-1 and ALT,
AST and HBV DNA
RIG-I and IPS-1 are involved in antiviral immunity,
whereas alanine aminotransferase (ALT), aspartate amino-transferase
(AST) and HBV DNA are important markers for liver injury and viral
replication in HBV infection. The relationship among these factors
is unclear. There was a significant correlation between ALT and
RIG-I/IPS-1 in CHB patients (P<0.05) (Fig. 3A). No comparison was performed in
AHB patients due to insufficient patient number. No significant
correlation between AST and RIG-I/IPS-1 (Fig. 3B) or between HBV DNA and
RIG-I/IPS-1 (Fig. 3C) was found
in CHB patients.
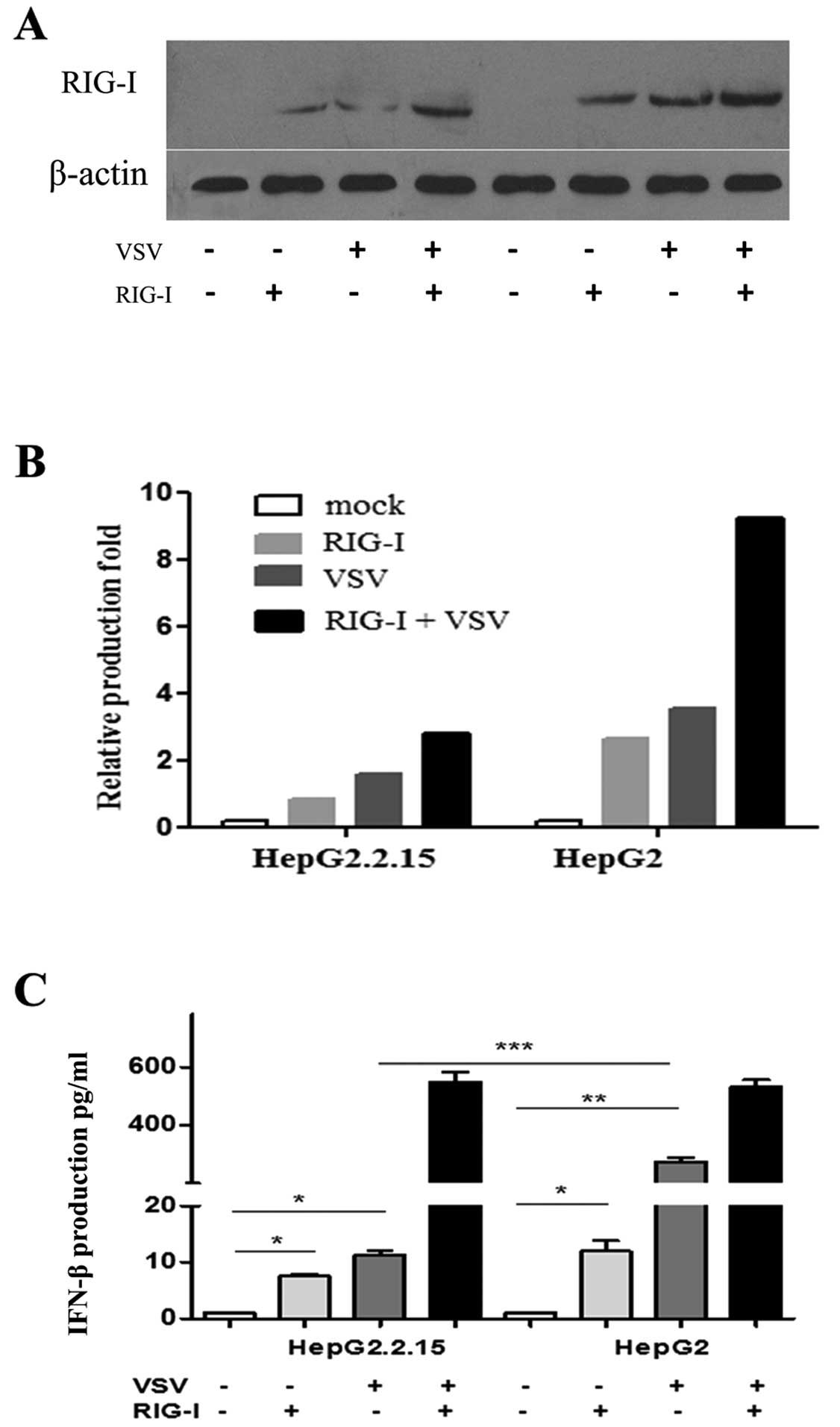
RIG-I elevates IFN-β secretion in
HBV-transformed cells
RIG-I induces type-I interferon production, which
also upregulates RIG-I expression during viral infection in a
positive feedback loop (18). To
determine the relationship between HBV infection and RIG-I
production, the production of RIG-I and IFN-β by RIG-I-transfected,
HBV-transformed HepG2.2.15 cells was determined 24 h after VSV
stimulation. There was a constant level of IFN-β in the supernatant
from both unstimulated and untransfected HepG2.2.15 and HepG2 cells
(Fig. 4). There was a 5- or
6-fold elevation of IFN-β production in the RIG-I-transfected
HepG2.2.15 cells regardless of VSV treatment. Markedly, there was
an almost 100-fold induction in IFN-β production in the supernatant
from RIG-I-transfected, VSV-stimulated cells. Following RIG-I
transfection, IFN-β production in HepG2 cells was ∼10-fold higher
than in HepG2.2.15 cells (P<0.01), whereas more profound IFN-β
production was only observed following VSV challenge. There was a
∼500-fold induction of IFN-β production in both HepG2.2.15 and
HepG2 cells in response to combined RIG-1 transfection and VSV
challenge compared with the mock-treated counterparts. However, no
significant difference was observed between the HepG2.2.15 and
HepG2 cells.
Discussion
HBV is a member of the Hepadnaviridae family, which
consists of hepatotropic non-cytopathic DNA viruses. Host immunity
plays a critical role in determining the outcome following HBV
infection (19). Mature dendritic
cells (DC) are competent antigen-presenting cells that play key
roles in priming and directing the viral-specific T-cell response.
Dendritic cells induce inflammation following challenge from an
invading microorganism, a critical step in the initiation of the
innate immune response. Due to difficulty in obtaining mature DCs
from peripheral circulation in humans (<1% of human PBMCs)
(14), moDCs were generated in
vitro as described (20). Our
data demonstrated that the proportion of CD86+ mature
moDCs was significantly reduced in CHB patients compared with HCs
following VSV stimulation, suggesting that chronic HBV infection
blocks the maturation of moDCs and subsequently compromises the
activation of specific anti-HBV immunity in CHB patients (21,22).
Activation of RIG-I by viral RNA stimulation results
in the activation of the IFN system and thus plays a pivotal role
in the antiviral response. However, in susceptible individuals,
viruses can still circumvent the IFN response through unknown
mechanisms (23), resulting in
chronic viral infection.
Here, we demonstrated that the induction of RIG-I
and IFN-β expression was compromised in moDCs isolated from CHB
patients compared with both AHB and HC subjects following VSV
stimulation in vitro, suggesting that the RIG-I-IFN pathway
was disrupted in the CHB patients. Consistent with these findings,
there was also a 90% reduction in IFN-β production in
HBV-transformed HepG2.2.15 cells relative to HepG2 cells following
VSV stimulation, which further indicated that HBV interferes with
IFN-β production. Compromised IFN-β production was restored in
HepG2.2.15 cells by RIG-1 overexpression, demonstrating the
critical role of RIG-I in the production of IFN-β in response to
HBV infection. The mechanisms underlying the differences in RIG-I
and IFN-β production among CHB and AHB patients remain unclear, but
may involve the complicated host anti-HBV immunity and/or
differential regulation of other receptors (such as TLRs) and
pathways at various stages: immune tolerance phase, immune
clearance phase and inactive phase (19). The different host immunities in
CHB and AHB patients may also be due to the immune response against
AHB infection being polyclonal, multi-specific and non-tolerant,
that is, the host immune system adopts a highly sensitive status
(24), whereas in the chronic
stage, host immunity is immune-tolerant. This hypothesis is
supported by the finding that more IFN-β was produced in the moDCs
from AHB patients following VSV infection, which provides positive
feedback to boost RIG-I expression. Increased IFN-β production may
also explain why RIG-I levels in AHB moDCs were the highest at 16 h
(representing a highly sensitive status). This observation is in
line with the findings that IFN-β production in AHB patients was
higher than in CHB patients and suggests that an impaired RIG-I
pathway contributes to compromised IFN-β production in CHB
patients.
It has been reported that the RNA virus directly
downregulates RIG-I and its downstream adaptor IPS-1 (11), resulting in the inhibition of
IFN-β production. In the current study, IPS-1 expression in moDCs
from CHB patients was significantly higher than that of HCs and was
inversely correlated with IFN-β production. The high IPS-1 level in
moDCs from CHB patients may be due to compensation for compromised
IPS-1 function and subsequent induction of IFN-β production. The
underlying mechanisms leading to compromised IPS-1 function remain
to be explored, but interference from HBV may be a contributing
factor, as Lin et al (25)
have reported that the HCV protease NS3-4A degrades the IPS-1
protein.
To investigate the relationship between liver damage
(ALT) and immune regulation (RIG-1 signaling), the correlation
between ALT level and the ratio of RIG-I to IPS-1 (RIG-I/IPS-1) was
determined (26). The correlation
between RIG-I and IPS-1 levels was also evaluated as a measure of
RIG-I-IFN-β signaling pathway activity. We observed a positive
correlation between ALT level and RIG-I/IPS-1, indicating that
activation of RIG-I signaling by HBV infection in moDCs may induce
liver damage. The RIG-I signaling pathway activates antiviral
responses via IFN-β to clear invaded viruses and/or infected
hepatocytes. Liver damage in CHB patients with high ALT levels may
also result from other activated host lymphocytes, such as
HBV-specific cytotoxic lymphocyte (CTL) and natural killer (NK) T
cells (27,28).
Neither AST nor HBV DNA is a reliable indicator of
inflammation in CHB (29). Our
data showed no correlation between AST and the RIG-I/IPS-1 ratio or
between HBV DNA and the RIG-I/IPS-1 ratio in CHB patients,
suggesting that further investigation is required to elucidate the
relationship between AST, HBV DNA and RIG-I/IPS-1.
In summary, compromised moDC maturation and function
in CHB patients may result from a dysfunctional RIG-I-IFN-β
signaling pathway and consequent impairment of host viral
clearance, which we propose are contributing factors to HBV
persistence in a susceptible population.
Abbreviations:
|
AHB
|
acute hepatitis B;
|
|
ALT
|
alanine amino-transferase;
|
|
CARD
|
caspase recruitment domain;
|
|
CHB
|
chronic hepatitis B;
|
|
HC
|
healthy control;
|
|
IFN-β
|
interferon-β;
|
|
IPS-1
|
IFN-β promoter stimulator 1;
|
|
MDA-5
|
melanoma differentiation-associated
gene 5;
|
|
moDC
|
monocyte-derived dendritic cell;
|
|
PBMC
|
peripheral blood mononuclear cell;
|
|
RIG-I
|
retinoic acid-inducible gene I;
|
|
VSV
|
vesicular stomatitis virus
|
Acknowledgements
This study was supported by the
Natural Science Foundation of China (30671838, 81070334); the
National Eleven-Five Project of China (2008ZX10002-005,
2008ZX10002-007); and the Committee of Science and Technology of
Shanghai Municipal Government (08JC1403900).
References
|
1.
|
AS LokBJ McMahonChronic hepatitis B:
update 2009Hepatology50661662200910.1002/hep.2319019714720
|
|
2.
|
G MarinosF TorreS ChokshiInduction of
T-helper cell response to hepatitis B core antigen in chronic
hepatitis B: a major factor in activation of the host immune
response to the hepatitis B
virusHepatology2210401049199510.1002/hep.18402204057557849
|
|
3.
|
T HatakeyamaC NoguchiN HiragaSerum HBV RNA
is a predictor of early emergence of the YMDD mutant in patients
treated with
lamivudineHepatology4511791186200710.1002/hep.2158117465002
|
|
4.
|
G ChengJ ZhongJ ChungFV
ChisariDouble-stranded DNA and double-stranded RNA induce a common
antiviral signaling pathway in human cellsProc Natl Acad Sci
USA10490359040200710.1073/pnas.070328510417517627
|
|
5.
|
MS LeeYJ KimSignaling pathways downstream
of pattern-recognition receptors and their cross talkAnnu Rev
Biochem76447480200710.1146/annurev.biochem.76.060605.12284717328678
|
|
6.
|
V HornungJ EllegastS Kim5′-Triphosphate
RNA is the ligand for RIG-IScience3149949972006
|
|
7.
|
M YoneyamaM KikuchiT NatsukawaThe RNA
helicase RIG-I has an essential function in double-stranded
RNA-induced innate antiviral responsesNat
Immunol5730737200410.1038/ni108715208624
|
|
8.
|
H KatoS SatoM YoneyamaCell type-specific
involvement of RIG-I in antiviral
responseImmunity231928200510.1016/j.immuni.2005.04.01016039576
|
|
9.
|
T FujitaK OnoguchiK OnomotoR HiraiM
YoneyamaTriggering antiviral response by RIG-I-related RNA
helicasesBiochimie89754760200710.1016/j.biochi.2007.01.01317379377
|
|
10.
|
M BinderG KochsR BartenschlagerV
LohmannHepatitis C virus escape from the interferon regulatory
factor 3 pathway by a passive and active evasion
strategyHepatology4613651374200710.1002/hep.2182917668876
|
|
11.
|
A KomuroD BammingCM HorvathNegative
regulation of cytoplasmic RNA-mediated antiviral
signalingCytokine43350358200810.1016/j.cyto.2008.07.01118703349
|
|
12.
|
W CaiQ XieB AnOn-treatment serum HBsAg
level is predictive of sustained off-treatment virologic response
to telbivudine in HBeAg-positive chronic hepatitis B patientsJ Clin
Virol482226201010.1016/j.jcv.2010.02.01420233672
|
|
13.
|
LY LinVW WongHJ ZhouRelationship between
serum hepatitis B virus DNA and surface antigen with covalently
closed circular DNA in HBeAg-negative patientsJ Med
Virol8214941500201010.1002/jmv.2186320648602
|
|
14.
|
M DauerB ObermaierJ HertenMature dendritic
cells derived from human monocytes within 48 hours: a novel
strategy for dendritic cell differentiation from blood precursorsJ
Immunol17040694076200310.4049/jimmunol.170.8.406912682236
|
|
15.
|
P NakhaeiT MespledeM SolisThe E3 ubiquitin
ligase Triad3A negatively regulates the RIG-I/MAVS signaling
pathway by targeting TRAF3 for degradationPLoS
Pathog5e1000650200910.1371/journal.ppat.100065019893624
|
|
16.
|
D SunM NassalStable HepG2- and Huh7-based
human hepatoma cell lines for efficient regulated expression of
infectious hepatitis B virusJ
Hepatol45636645200610.1016/j.jhep.2006.05.01916935386
|
|
17.
|
BR tenOeverS SharmaW ZouActivation of TBK1
and IKKvarepsilon kinases by vesicular stomatitis virus infection
and the role of viral ribonucleoprotein in the development of
interferon antiviral immunityJ
Virol781063610649200410.1128/JVI.78.19.10636-10649.2004
|
|
18.
|
Q HuangQ XieCC ShiExpression of
angiotensin-converting enzyme 2 in CCL4-induced rat liver
fibrosisInt J Mol Med23717723200919424597
|
|
19.
|
MC JungGR PapeImmunology of hepatitis B
infectionLancet Infect
Dis24350200210.1016/S1473-3099(01)00172-4
|
|
20.
|
D TerhorstBN KalaliS
WeidingerMonocyte-derived dendritic cells from highly atopic
individuals are not impaired in their pro-inflammatory response to
toll-like receptor ligandsClin Exp
Allergy37381390200710.1111/j.1365-2222.2006.02639.x17359388
|
|
21.
|
S PattersonH DonaghyP AmjadiB GazzardF
GotchP KelleherHuman BDCA-1-positive blood dendritic cells
differentiate into phenotypically distinct immature and mature
populations in the absence of exogenous maturational stimuli:
differentiation failure in HIV infectionJ
Immunol17482008209200510.4049/jimmunol.174.12.8200
|
|
22.
|
Y ShimizuLG GuidottiP FowlerFV
ChisariDendritic cell immunization breaks cytotoxic T lymphocyte
tolerance in hepatitis B virus transgenic miceJ
Immunol1614520452919989794377
|
|
23.
|
RE RandallS GoodbournInterferons and
viruses: an interplay between induction, signalling, antiviral
responses and virus countermeasuresJ Gen
Virol89147200810.1099/vir.0.83391-018089727
|
|
24.
|
CK HuiGK LauImmune system and hepatitis B
virus infectionJ Clin
Virol34S44S48200510.1016/S1386-6532(05)80009-516461223
|
|
25.
|
R LinJ LacosteP NakhaeiDissociation of a
MAVS/ IPS-1/VISA/Cardif-IKKepsilon molecular complex from the
mitochondrial outer membrane by hepatitis C virus NS3-4A
proteolytic cleavageJ
Virol8060726083200610.1128/JVI.02495-0516731946
|
|
26.
|
Y AsahinaN IzumiI HirayamaPotential
relevance of cytoplasmic viral sensors and related regulators
involving innate immunity in antiviral
responseGastroenterology13413961405200810.1053/j.gastro.2008.02.01918471516
|
|
27.
|
JN StoopRG van der MolenCC BaanRegulatory
T cells contribute to the impaired immune response in patients with
chronic hepatitis B virus
infectionHepatology41771778200510.1002/hep.2064915791617
|
|
28.
|
A ZerbiniM PilliC BoniThe characteristics
of the cell-mediated immune response identify different profiles of
occult hepatitis B virus
infectionGastroenterology13414701481200810.1053/j.gastro.2008.02.01718355815
|
|
29.
|
JJ ChangSR LewinImmunopathogenesis of
hepatitis B virus infectionImmunol Cell
Biol851623200710.1038/sj.icb.710000917130898
|