Introduction
Mesenchymal stem cells (MSCs) are multipotent
stromal cells that have been isolated from both adult and fetal
tissues and are defined as adherent, fibroblast-like cells. In the
early 1970s, Friedenstein et al described the existence of
multipotent mesenchymal cells in mouse bone marrow (BM) with the
ability to form colonies [fibroblast colony-forming units (CFU-F)]
and differentiate into adipocytes, chondrocytes and osteocytes
(1). It was only 20 years later
that Caplan defined the terminology, MSCs (2). Subsequently, approximately 10 years
later, MSCs were finally identified in human adult BM (3,4).
MSCs are initially isolated and characterized from BM, but can be
also obtained from other sources, such as the amniotic membrane,
skin, hair follicles, dental pulp, adipose tissue (AT), cord blood,
umbilical cord Wharton’s jelly (WT), the endometrium, amniotic
fluid, fetal liver, the placenta (PL) and the synovium (5). Among these sources, AT, WJ from the
umbilical cord and PL are considered to be valuable alternatives to
BM as a rich source of MSCs (6,7).
MSCs possess 2 major properties, a self-renewal
ability and the potential for multilineage differentiation. MSCs
can differentiate into a variety of cell types, including
osteoblasts, chondrocytes, adipocytes and myocytes (8–12).
It has been reported that the most important characteristics of
MSCs are their potential for differentiation into bone and
cartilage cell lineages (13,14). The differential ability of MSCs
raises the hope for treating some types of bone or cartilage
injuries which can be treated by general medication practices
(15,16). In vitro and in vivo
studies have also demonstrated that MSCs can differentiate into
cells of non-mesodermal origin, such as neurons, skin and gut
epithelial cells, hepatocytes and pneumocytes (17).
It has been demonstrated that MSCs have both
immunosuppressive and immunomodulatory functions (18–20). Although, the mechanisms underlying
the behavior of MSCs during an immune response and their
immunomodulatory effects remain unclear, tissue-derived MSCs have
potent immunomodulatory properties and suppress T lymphocyte, B
lymphocyte and natural killer (NK) cell functions (21–24). Members of the human leukocyte
antigen (HLA) family and immunoregulatory factors are of importance
in determining the nature of the response generated by MSCs and T
lymphocyte interactions. Thus, establishing and comparing the
immunological profiles of MSCs isolated from different types of
tissue may facilitate the determination of the best
immune-privileged MSCs for clinical therapy.
Materials and methods
Isolation and expansion of MSCs
MSCs were isolated from 4 different sources: BM, AT,
WJ and PL tissue. The BM and AT samples were obtained from healthy
volunteer donors, while the WJ and PL samples were from tissue
following normal caesarean birth. All individuals provided written
informed consent and the study was approved by the Ethics Committee
of the China-Japan Union Hospital, Jilin University, Changchun,
China. The age range of the donors was as follows: the BM was from
individuals aged 18 to 43 years, the AT was from individuals aged
23 to 50 years, and the WJ and PL tissue were from individuals aged
23 to 38 years. The MSCs derived from BM, AT, WJ and PL were
isolated according to previously described methods (25–27) with some modifications. The
enzymatic digestion method was used to isolate the MSCs from the
tissues. Briefly, collagenase and hyaluronidase were used to digest
the umbilical cord after the outside skin was removed. The PL and
AT were digested by collagenase only. BM-MSCs were obtained by BM
adherence culture. The MSCs were cultured in α-MEM supplemented
with 10% FBS (Invitrogen Australia Pty Ltd., Mount Waverley,
Victoria, Australia). The culture was maintained at 37°C with
saturated humidity and 5% CO2. After 48 h, the
non-adherent cells were removed by washing, and the medium was
changed twice a week. The MSCs were subcultured at 80% confluence
following treatment with 0.05% trypsin and 0.02% EDTA (Sigma, St.
Louis, MO, USA) for 3 min at 37°C. The cells were washed and
harvested by centrifugation at 1000 rpm for 5 min, then replanting
at a lower density (1,000 cells/cm2).
Flow cytometric characterization of
MSCs
The MSCs were removed from the culture flasks by
incubation in 0.05% trypsin-EDTA at 37°C and then washed twice with
PBS. The cell suspensions were then incubated with antibodies
against CD44-phycoerythrin (PE), CD73-PE, CD14-PE, CD34-PE,
CD90-fluorescein isothiocyanate (FITC), CD45-FITC and CD105-PerCP
(BD Biosciences, Franklin Lakes, NJ, USA) and protected from light
for 30 min at 4°C. Following incubation, the cells were washed
twice with PBS. The fluorescence intensity of the cells was
evaluated using a flow cytometer (FACScan; BD Biosciences) and the
data were analyzed using CellQuest software (BD Biosciences).
MSC differentiation potential
Adipogenesis
A total of 5x104 MSCs/cm2 was
seeded into a 12-well plate and incubated in MSC growth medium
(α-MEM with 10% FBS) at 37°C in a humidified atmosphere with 5%
CO2 for a minimum of 2 h or up to 4 days (to near or
complete confluence) before re-seeding with adipogenic
differentiation medium (StemPro® Adipogenesis
Differentiation kit; Invitrogen Australia Pty Ltd.). The MSCs will
continue to undergo limited expansion as they differentiate under
adipogenic conditions. The cultures were re-fed every 3–4 days.
After 21 days under differentiating conditions, the medium was
removed from the wells, and the cells were rinsed once with PBS,
and fixed with 4% formaldehyde solution for 30 min. After fixation,
the cells were rinsed twice with PBS, stained with Oil Red O
(Sigma) for 15 min, rinsed twice with PBS and visualized under a
light microscope (Olympus, Tokyo, Japan). Images were captured for
qualitative and quantitative analyses. Fifteen fields at x400
magnification under the microscope were randomly selected and the
stained cell and total cell numbers were counted. The sample number
was 3 each for all 4 populations of MSCs.
Osteogenesis
The MSCs were seeded into a 12-well plate at
5x104 cells/cm2. The cells were fed in α-MEM
with 10% FBS and incubated at 37°C in a humidified atmosphere with
5% CO2. Osteogenic differentiation medium
(StemPro® Osteogenesis Differentiation kit; Invitrogen
Australia Pty Ltd.) was added to the cells the second day after the
cells attached to the bottom of the petri dish. MSCs will undergo
limited expansion during differentiation under osteogenic
conditions. The cultures were re-fed every 3–4 days. On day 35, the
medium was removed from the 12-well plate and the cells were rinsed
twice with PBS. The cells were fixed with 4% formaldehyde solution
for 30 min. After fixation, the cells were rinsed with distilled
water and stained with 2% Alizarin Red S solution (Alizarin
Staining kit; Genmed, Shanghai, China) for 5 min. The cells were
rinsed 3 times with distilled water and visualized under a light
microscope (Olympus). Images were captured for qualitative or
quantitative analyses. Bone nodule numbers of a total of 3 wells
were counted for each sample. Three samples from each population of
MSCs were analyzed.
Proliferative potential of MSCs
The 4 populations of MSCs beginning from passage 1
were trypsinized from the culture discs before plating into the
6-well plates at a density of 105/well. Each population
of MSCs was seeded in triplicate. Every 4 days, the cells were
harvested with trypsin/EDTA and counted using a hemacytometer for
up to 8 passages. The mean value of the cell number counts was
calculated from 7 tissue samples from each cell population and the
mean population doubling time was obtained for each passage
according to the following formula: population doubling time = T x
lg2/(lgNt - lgN0), where T is the culture time, N0 is the initial
cell number and Nt is the harvested cell number.
Reverse transcription PCR (RT-PCR)
Total cellular RNA was extracted using TRIzol
reagent (Invitrogen Life Technologies, Grand Island, NY, USA)
according to the manufacturer’s instructions. RNA integrity was
electrophoretically verified by ethidium bromide staining and a
OD260/OD280 nm absorption ratio (OD >1.9). Total RNA (500 ng)
was reverse transcribed using AWV reverse trancriptase, oligo(dT)15
(~20 mer, Takara, Dalian, China) according to the manufacturer’s
instructions. No amplification control (NAC) was included without
adding reverse transcriptase. Reactions were incubated at 42°C for
30 min, then 95°C for 5 min and 5°C for 5 min.
Twenty five nanograms of cDNA was used in a 25-μl
reaction volume with 0.15 μl Takara Ex Taq HS (Takara), 5 μM of
gene-specific forward and reverse primers (Sangon Biotech,
Shanghai, China), 5 μl 5X PCR buffer and sterile water. Reactions
were incubated at 94°C for 30 sec, 56°C for 30 sec and 72°C for 30
sec for total 30 cycles.
Quantitative reverse transcription PCR
(RT-qPCR)
RT-qPCR was carried out to assess the expression of
genes, including HLA-DMA, HLA-DRA, HLA-DPB1, JAG1, TLR4, TLR3,
NOTCH2 and NOTCH3 in the MSCs derived from the 4 different tissue
sources. Primers were designed using primer3 input (http://flypush.imgen.bcm.tmc.edu/primer/primer3_www.cgi)
and synthesized with the temperature at 60°C (Sangon Biotech;
Table I). RNA extraction and
reverse transcription were carried out as described above. RT-qPCR
was carried out using the ABI PRISM 7900 Sequence Detection System
(Perkin-Elmer/Applied Biosystems, Foster City, CA, USA). Twenty
five nanograms of cDNA was used in the qRT-PCR reactions with
SYBR-Green PCR Master Mix (Applied Biosystems, Warrington, UK) and
5 μM of gene-specific forward and reverse primers (Sangon Biotech).
All PCR products demonstrated a single band by a dissociation curve
and gel electrophoresis. The thermal cycler parameters for the
amplification of these genes were as follows: 1 cycle at 95°C for
10 min followed by 40 cycles at 95°C for 15 sec, 55°C for 15 sec
and 72°C for 30 sec. The 18S RNA gene was used to normalize the
cDNA amounts used in RT-qPCR. Amplification using 18S RNA primers
consistently yielded similar CT levels among all types of tissue.
No template control (NTC) and NAC were included in each reaction.
Gene expression was presented using a modification of the
2−ΔΔCt method, first described by Livak and Schmittgen
in the PE Biosystems Sequence Detector User Bulletin 2 (28).
 | Table IPrimers used for RT-qPCR. |
Table I
Primers used for RT-qPCR.
| Gene name | Primers | Sequences | Product size
(bp) |
|---|
| HLA-DMA | Forward |
AAAATCCCGGTGTCCAGAG | 167 |
| Reverse |
GTAGGCCCAAATCCTTCCA | |
| HLA-DRA | Forward |
CCTGACTGTGGGTCTGGTG | 81 |
| Reverse |
CGTTCTGCTGCATTGCTTT | |
| HLA-DPB1 | Forward |
CCTGGTGATGCTGGAAATG | 105 |
| Reverse |
GACTGTGCCTTCCACTCCA | |
| JAG1 | Forward |
CGGCCTCTGAAGAACAGAAC | 82 |
| Reverse |
TCACCAAGCAACAGATCCAA | |
| TLR4 | Forward |
TTTCACCTGATGCTTCTTGCT | 103 |
| Reverse |
TCCTTACCCAGTCCTCATCCT | |
| TLR3 | Forward |
AGGATTGGGTCTGGGAACA | 95 |
| Reverse |
AAAAACACCCGCCTCAAAG | |
| NOTCH2 | Forward |
TGGGCTACACTGGGAAAAAC | 107 |
| Reverse |
TAGGCACTGGGACTCTGCTT | |
| NOTCH3 | Forward |
CTCATCCGAAACCGCTCTAC | 101 |
| Reverse |
TCTTCCACCATGCCCTCTAC | |
Co-culture of MSCs and T lymphocytes
The co-culture of the MSCs and T cells was carried
out using 24-well plates (contact culture) and the Transwell system
(isolated culture) in which the T cells and MSCs were physically
separated by a membrane permeable for soluble factors. The inner
hanging cell culture insets with 0.4-μm pore size membrane of a
24-well plate were purchased from Millipore (Billerica, MA, USA).
The BM-, AT-, WJ- and PL-MSCs were seeded at 105
cells/well in regular 24-well plates and 24-well Transwell plates
containing α-MEM, 10% FBS and 100 U/ml penicillin/streptomycin.
After 24 h, 10 μg/ml mitomycin C (MMC; Sigma) were added to inhibit
MSC proliferation, and the cells were incubated for 2 h at 37°C
followed by 5 extensive washes with medium. A total of
105 T cells/well was added and stimulated with 10 g/ml
phytohemagglutinin (PHA; Sigma) and 10 ng/ml interleukin (IL)-2
(Sigma). IL-2/PHA-activated T cells were cultured in the presence
or absence of MSCs. The cultures were plated in triplicate and
incubated for 5 days before the addition of 5-bromo-20-deoxyuridine
(BrdU). After 18 h, proliferation was assessed using the BrdU-Assay
kit (Roche Applied Science, Penzberg, Germany) according to the
manufacturer’s instructions.
Statistical analysis
Statistical analyses were performed using SPSS
software version 17.0 (SPSS Inc., Chicago, IL, USA). The
differences among the treatment groups were analyzed by one-way
analysis of variance (ANOVA) and two-way ANOVA with post-hoc
analysis using the Dunnett’s test. Parametric data are expressed as
the means ± standard deviation (SD). A value of P<0.05 was
considered to indicate a statistically significant difference.
Results
General characterization of MSCs
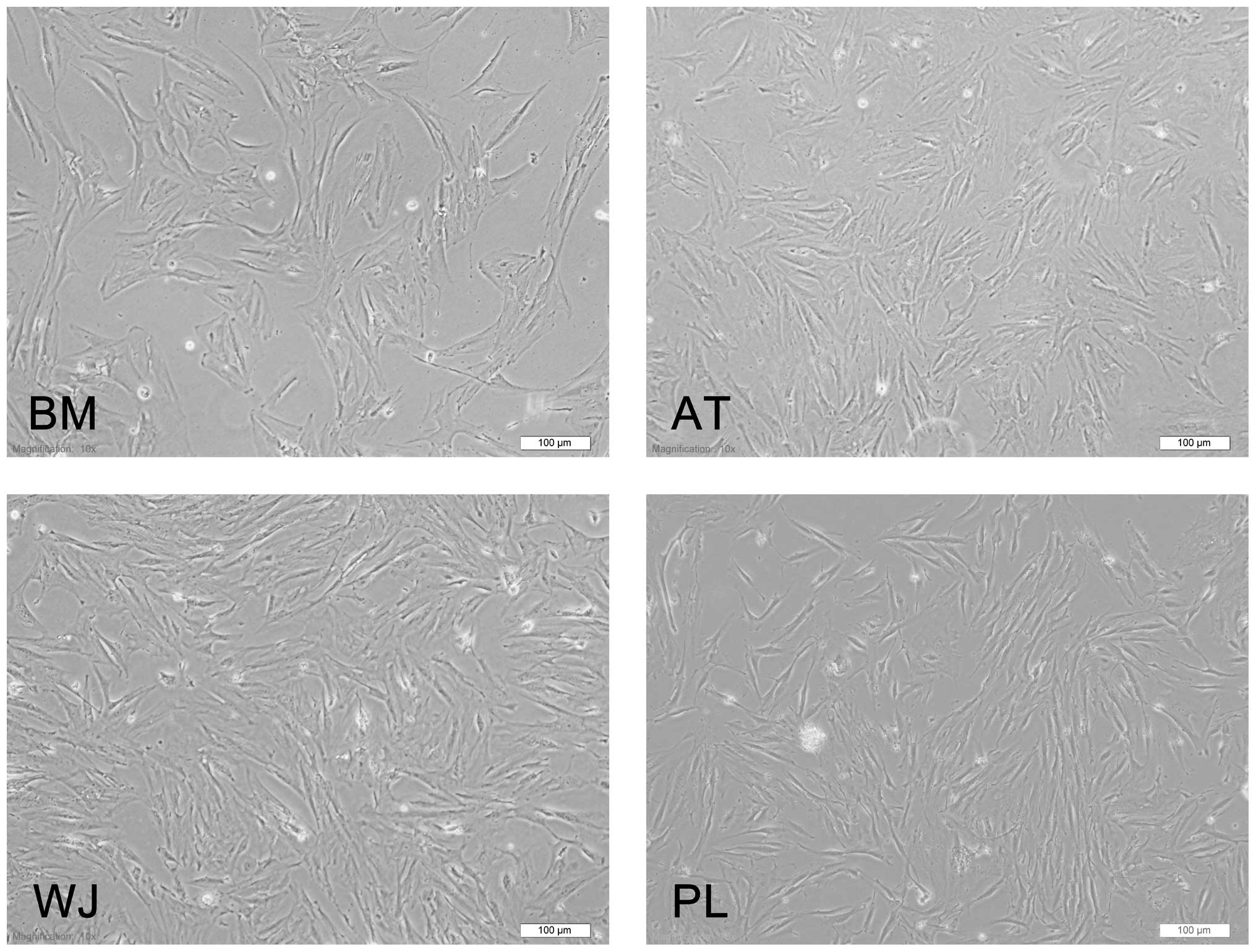
The MSCs derived from 4 different sources (BM, AT,
WJ and PL) presented the typical morphology of fibroblastic cells
and displayed a high capacity to adhere to the plastic disc
(Fig. 1). Following subculture,
they showed a strong proliferative ability and they adhered rapidly
and expanded without visible changes in either their growth
patterns or morphology. The cells reached confluence after
approximately 3 days.
Flow cytometric analysis of MSC
makers
The cell-surface antigen profiles of these cells
after 3 passages in culture were analyzed by flow cytometry. These
cells were strongly positive for MSC-specific surface markers, such
as CD44, CD73, CD90 and CD105, but negative for CD14, CD34 and CD45
(Fig. 2). As regards the
percentage of cells expressing each angigen, our data showed that
>99% of the MSCs from all 4 tissue sources were positive for
CD44, CD73, CD90 and CD105, while only ≤1.8% of the cells were
positive for CD14, CD34 and CD45 (Fig. 2).
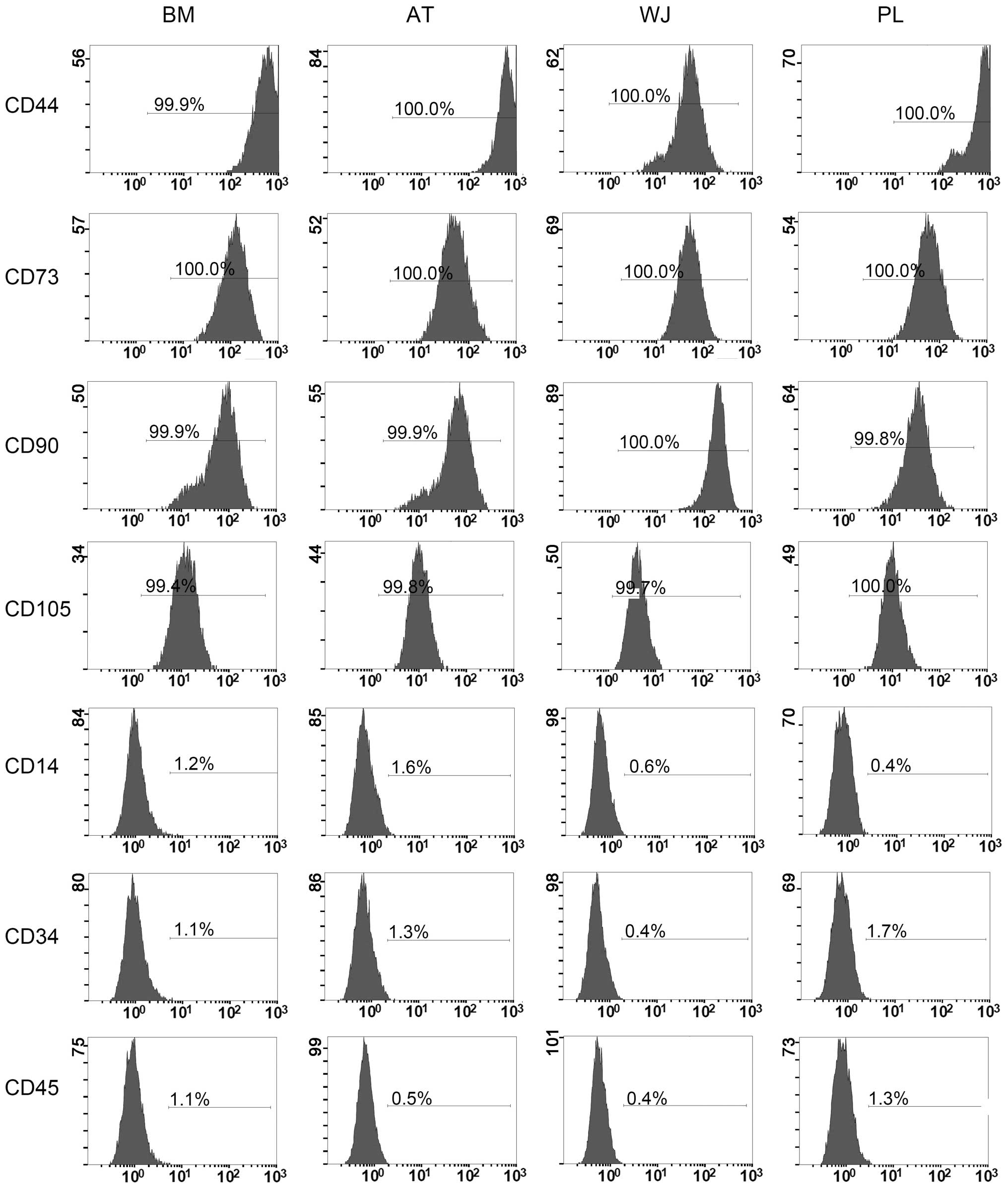 | Figure 2Flow cytometric analysis of
mesenchymal stem cell (MSC) makers. Cells at passage 3 were used.
BM, bone marrow; AT, adipose tissue; WJ, umbilical cord Wharton’s
jelly; PL, placenta. The conjugated fluorescent dyes are: CD44-PE,
CD73-PE, CD14-PE, CD34-PE, CD90-FITC, CD45-FITC and CD105-PerCP.
The positively stained cells are expressed as a percentage in the
middle of the frame. BM, bone marrow; AT, adipose tissue; WJ,
umbilical cord Wharton’s jelly; PL, placenta. |
Adipogenesis and osteogenesis
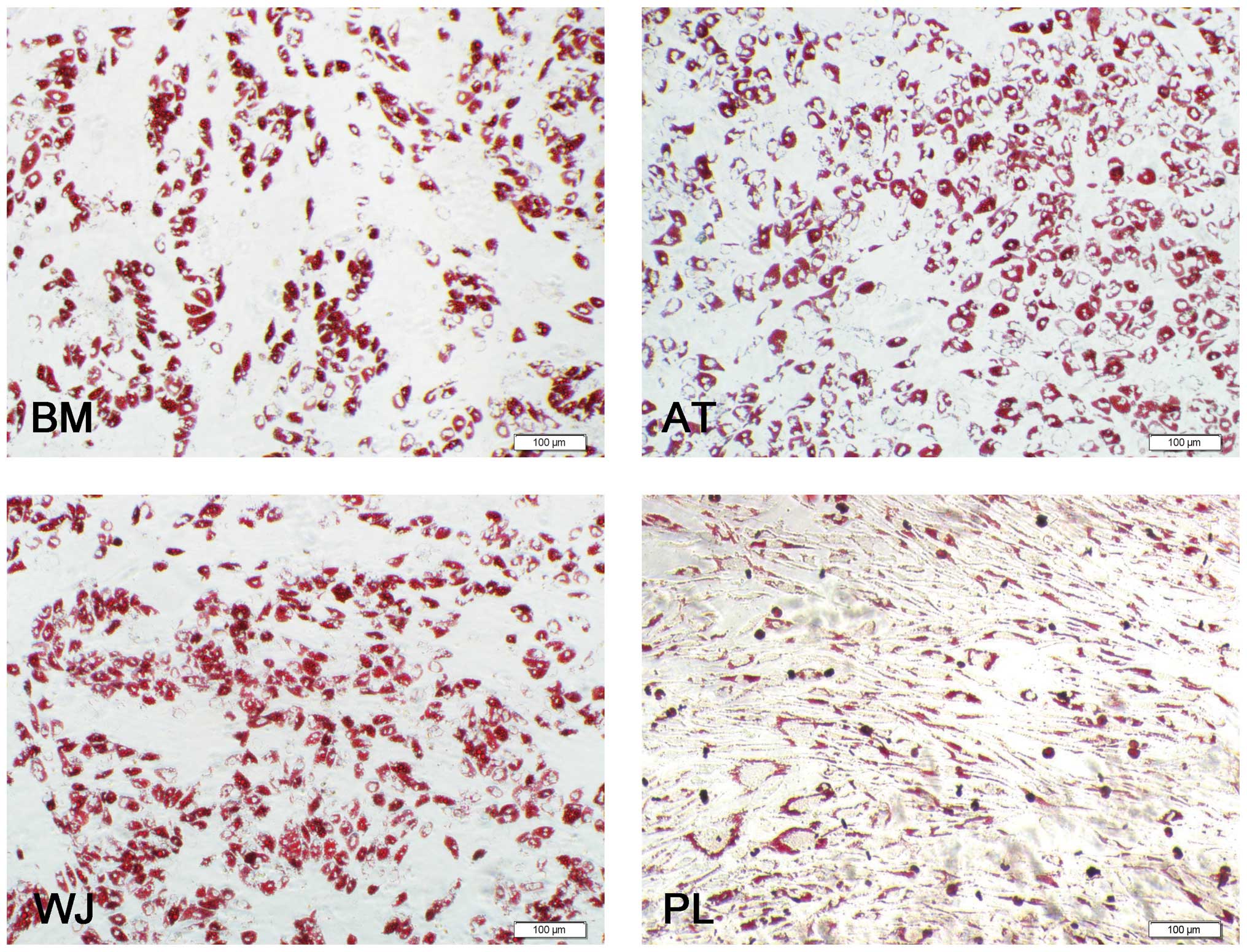
Oil Red O staining revealed that the MSCs were
positive for lipid vesicle-forming adipocytes (Fig. 3), and calcium deposits were
observed by Alizarin red staining (Fig. 4). For adipogenesis, lipid droplets
began to be observed 3 days after the addition of the induction
reagents. The time periods by which the lipids appeared were as
follows: AT-MSCs, 3 days; BM-MSCs, 6 days; WJ-MSCs and PL-MSCs, 8
days. Subsequently, the size and number of the droplets began to
increase until 3 weeks. Although the speed of adipocyte
differentiation for the WJ-MSCs and PL-MSCs was slower than that of
the BM-MSCs, the final induction ratio differed: the WJ-MSC
induction ratio was higher than that of the BM-MSCs, and that of
the PL-MSCs was lower than that of the BM-MSCs. For a more detailed
comparison, 15 fields at x400 magnification under a microscope were
selected and the stained adipocytes were counted. The ratio of
differentiated adipocytes from the total cells in the 15
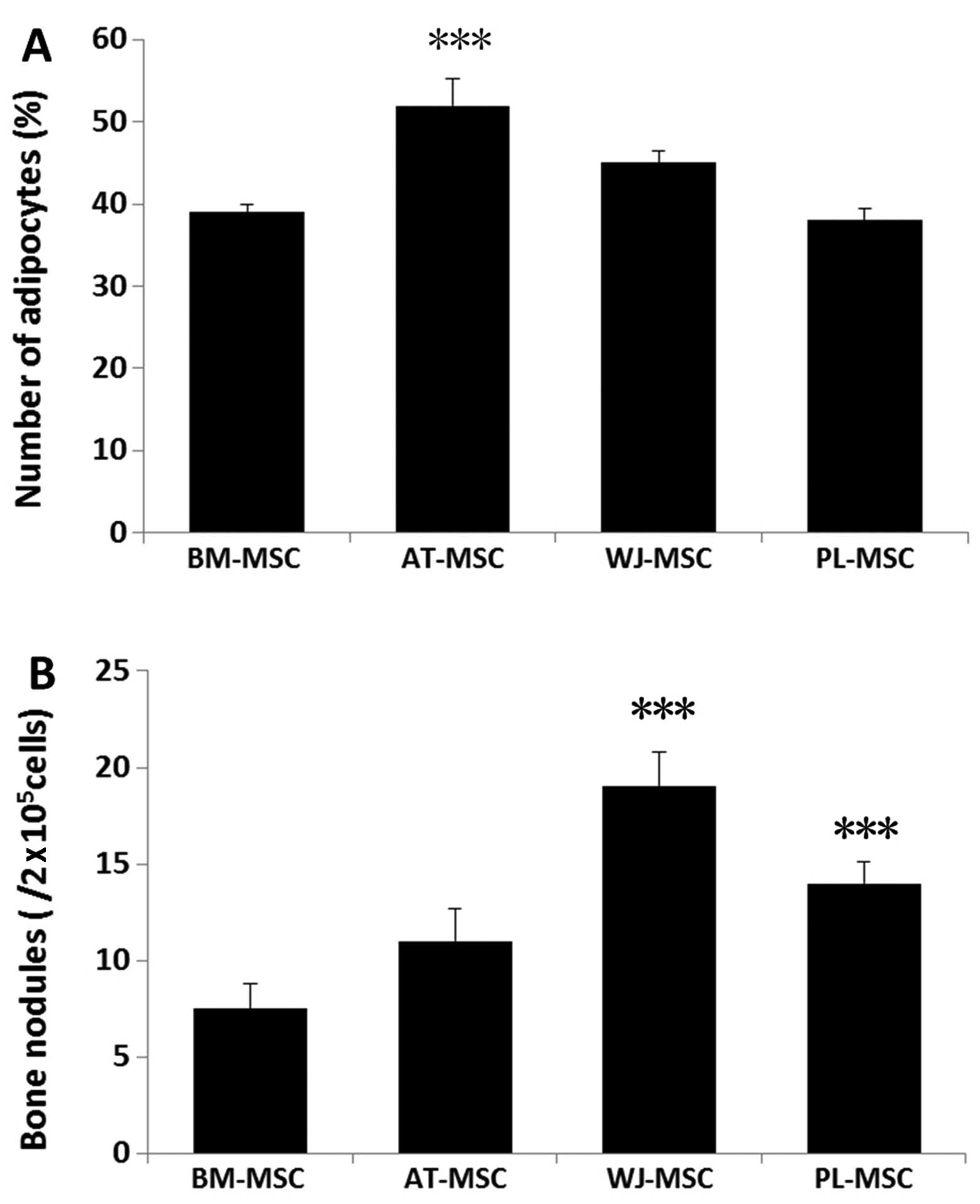
microscopic fields was 52±3.2% for the AT-MSCs, 45±1.5% for the
WJ-MSCs, 39±1% for the BM-MSCs and 38±1.4% for the PL-MSCs. Three
samples of each population of MSCs were counted (Fig. 5A).
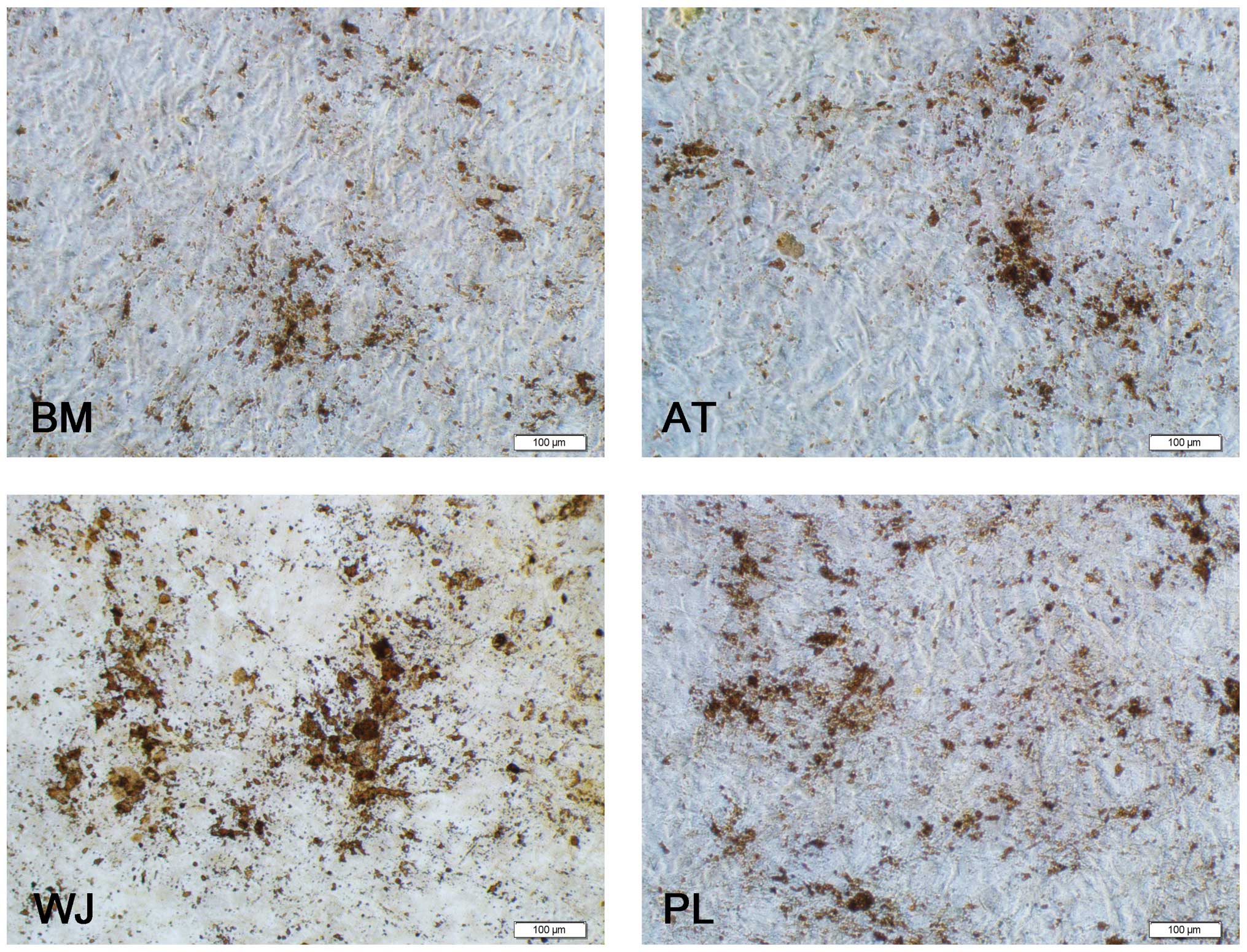
As regards osteogenesis, the cell volume began to
increase after 1 week of induction. The shape of the cells
gradually became triangular or polygonal and granular matter
appeared in the cytoplasm. After 3 weeks of induction, yellow
substances accumulated in the MSCs from all 4 tissue sources,
particulary in the WJ-MSCs, and gradually, these sediments
increased in size to form round or polygonal nodules (Fig. 4). A total of 9 wells from 3
samples of each population of MSCs was analyzed. The average
numbers of bone nodules from 1 well were 19±1.8 for the WJ-MSCs
(0.01±0.001%), 14±1.1 for the PL-MSCs (0.007±0.0006%), 11±1.7 for
the AT-MSCs (0.006±0.0009%) and 7.5±1.3 for the BM-MSCs
0.004±0.0007%). Three samples of each population of MSCs were
counted (Fig. 5B). These results
demonstrate the multipotent nature of the MSCs. The
undifferentiated MSCs cultured in the growth medium did not show
any staining for Oil Red O or Alizarin red.
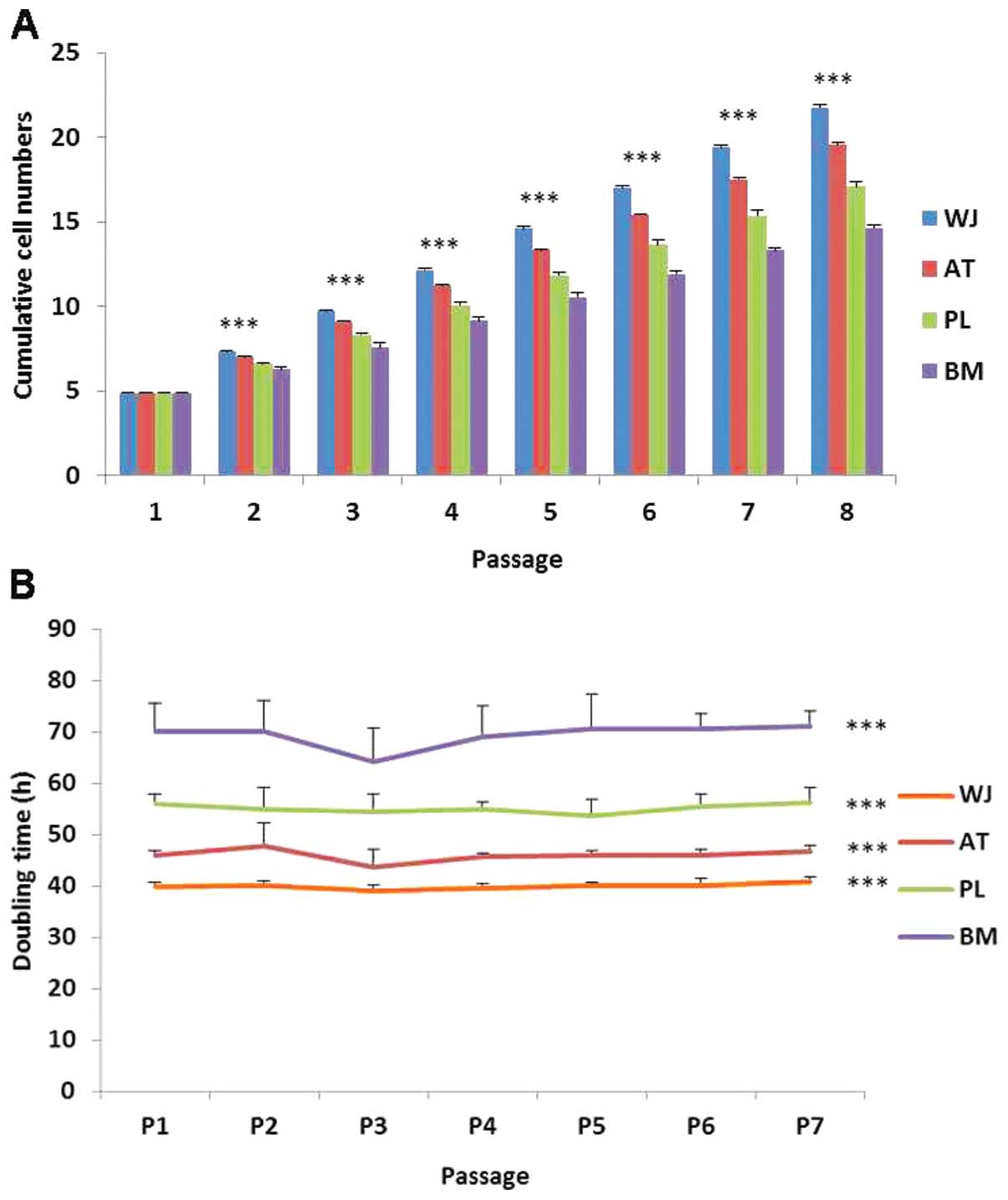
Proliferative differences in MSCs
During cell proliferation, the MSCs were cultured up
to passage 8. The WT-MSCs displayed the highest cumulative cell
population followed by the AT-, PL- and BM-MSCs (Fig. 6A). Based on the cell doubling time
calculation, the cell doubling time of the WJ-MSCs was
approximately 40 h, and that of the BM-MSCs was approximately 70 h
(Fig. 6B). The doubling times
among the 7 passages did not differ significantly. Thus, the order
of the growth rate of the cells was as follows (from the most rapid
to the least rapid): WJ-, AT-, PL- and BM-MSCs.
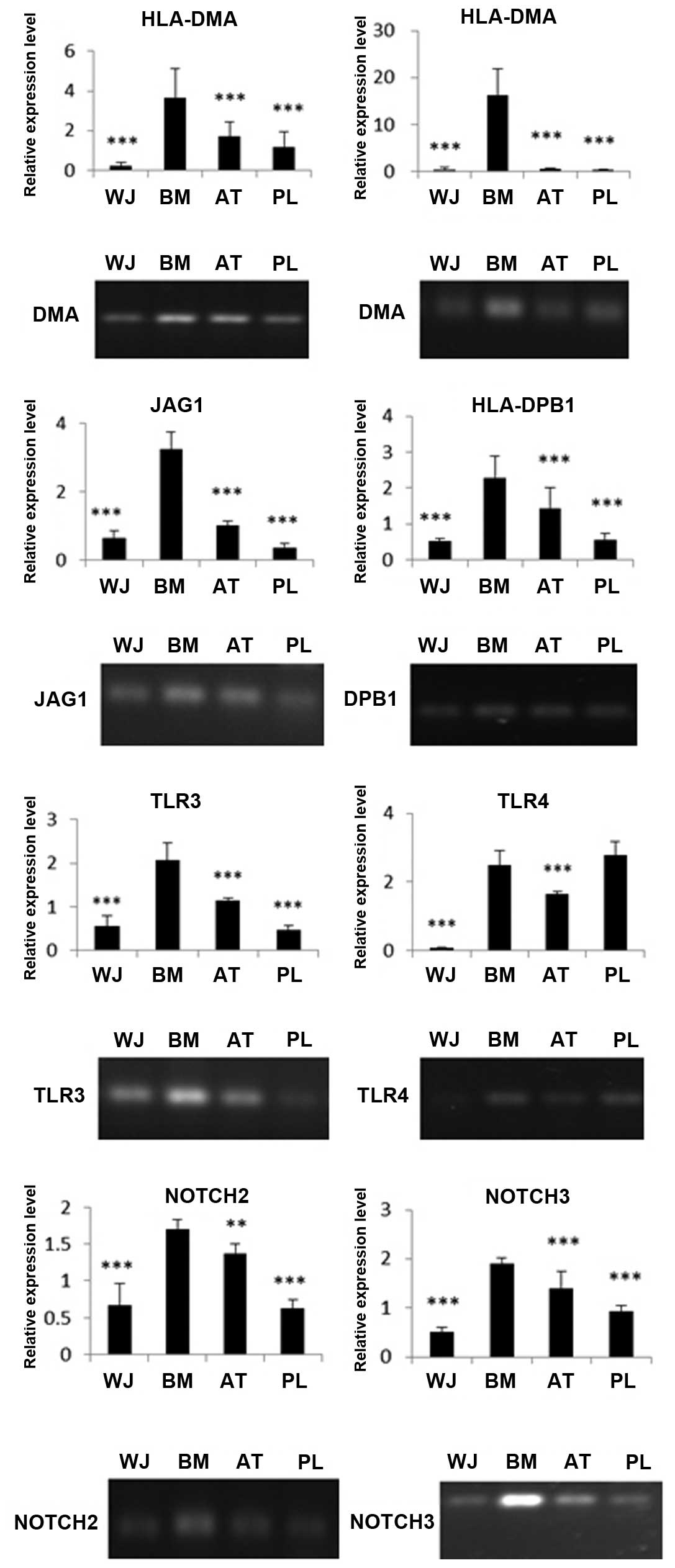
Expression of immune-related genes in
MSCs analyzed by RT-PCR and RT-qPCR
The MSCs were screened for their surface expression
of HLA antigens, co-stimulatory factors and immune tolerance
molecules. Through RT-qPCR of HLA-DMA, HLA-DPB1 and HLA-DRA, we
found that the BM-MSCs had the highest expression of MHCII
molecules than the other MSC populations. The WJ-MSCs had the
lowest expression of these molecules among the 4 cell populations.
The expression of DMA, DRA and DPB1 in the BM-MSCs was 16-, 36- and
4-fold higher, respectively compared with the WJ-MSCs (Fig. 7). The immune-related genes, JAG1,
TLR4, TLR3, NOTCH2 and NOTCH3, were also expressed at different
levels. The expression levels of TLR4, TLR3, JAG1, NOTCH2 and
NOTCH3 in the BM-MSCs were 38-, 4-, 5-, 3- and 4-fold higher,
respectively compared with the WJ-MSCs. The RT-PCR data coincided
well with our RT-qPCR results (Fig.
7).
MSC inhibition of T cell
proliferation
PHA/IL-2-activated T cells were incubated on a layer
of MSCs derived from BM, AT, WJ and PL tissue. T cell proliferation
decreased significantly following co-culture with the MSCs. The
ratio of growth retardation of the T lymphocytes in the contact
culture was: BM-MSCs, 41.4±3.2%; AT-MSCs, 35.5±1.7%; WJ-MSCs,
22.6±0.9% and PL-MSCs, 30.4±1.2% when compared to the T cells
cultured alone. The results from Transwell culture were similar to
those of the contact culture (P>0.05). The inhibitory effects of
the AT-MSCs, WJ-MSCs and PL-MSCs on T cell proliferation were more
prominent than those of the BM-MSCs (P<0.01). The inhibitory
effects of the WJ-MSCs on T cell proliferation were the most
prominent (P<0.01; Fig.
8).
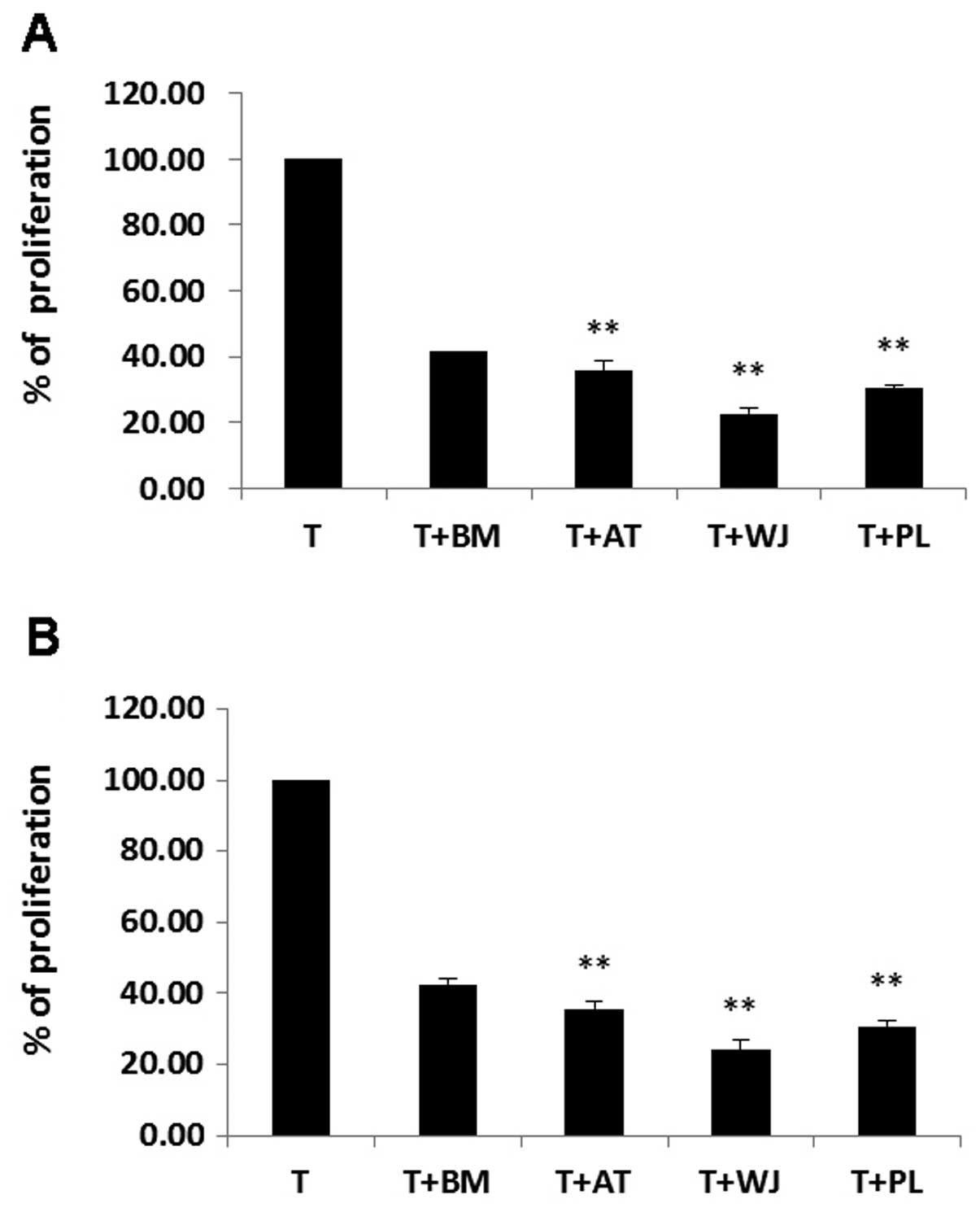 | Figure 8T cell proliferation was inhibited by
mesenchymal stem cells (MSCs) in co-culture. The first column is
the T cell only control. (A) Cell to cell contact co-culture. The T
cell proliferation ratio was reduced to 41.4±3.2%, 35.5±1.7%,
22.6±0.9% and 30.4±1.2%, respectively by MSCs derived from bone
marrow (BM), adipose tissue (AT), umbilical cord Wharton’s jelly
(WJ) and placenta (PL). (B) Transwell co-culture. The T cell
proliferation ratio was reduced to 42.6±1.6%, 35.7±1.8%, 24±2.7%
and 30.8±1.7%, respectively by co-culture with BM-MSCs, AT-MSCs,
WJ-MSCs, and PL-MSCs. The data are expressed as the means ± SD from
5 independent experiments. The inhibitory effect of the AT-, WJ-
and PL-MSC co-culture groups on T cell proliferation was more
prominent than that of the BM-MSC group. **P<0.01
when compared to the control. |
Discussion
BM is the earliest tissue source for MSC extraction
and represents the most commonly used source for MSCs in clinical
treatments. However, there are some limitations in using BM-MSCs
due to the high degree of viral exposure and the significant
decrease in cell number and proliferation/differentiation capacity
with the increasing age of the donor (29,30). It is also a painful and invasive
procedure to obtain BM from patients for MSC use. These
disadvantages have encouraged researchers to find a better tissue
source to replace BM for isolating MSCs. AT-MSCs have gained
increasing attention given the increasing problem posed by obesity
in recent years. In addition, AT-MSCs have a similar proliferative
ability and differentiation potential to BM-MSCs (31). MSCs derived from human WJ of the
umbilical cord also have the ability to differentiate into multiple
lineages, given the appropriate conditions. Kobayashi et al
also revealed that matrix cells from the WJ showed similar
characteristics with those of MSCs derived from BM (32). In addtion, progenitor cells or
stem cells may be isolated from the human term placenta (33). Human BM, AT, WJ and PL are the
main sources of MSCs, and these types of MSCs are frequently used
in both experimental and clinical studies. Although MSCs derived
from these 4 sources share global properties, such as morphology,
plastic adherence and multilineage potential, they diverge in terms
of their phenotypes. Up to now, there have been no detailed studies
comparing these MSCs derived from different sources. However, we do
know that they have different proliferative, differentiation and
immune properties.
In this study, we performed a side-by-side
comparison of 4 populations of MSCs derived from BM, AT, WJ and PL
simultaneously. We observed significant differences in the
proliferative potential among the 4 populations of MSCs; the
WJ-MSCs exhibited the highest growth rate. The growth curve showed
that the proliferative capacity of the WJ-, AT- and PL-MSCs was
significantly greater than that of the BM-MSCs. These data coincide
with those of previous studies (34–37).
Some believe that the growth differences may not
reflect compartment-specific characteristics, but most probably
reflect differences in newborn and adult donors (38). Alternatively, differences in
growth rate may reflect culture heterogeneity with variable
proportions of self-renewal versus lineage-committed cells in
different stromal cell compartments (39,40). In this study, the order of the
proliferation rate from the fastest to the slowest was: WJ-MSCs,
AT-MSCs, PL-MSCs and BM-MSCs. Basically, these data follow the age
trend except, that the AT was obtained from older individuals than
those from which the PL was obtained (6).
MSCs derived from different tissues have been
demonstrated in a large number of studies to differentiate into
cells in the mesodermal lineages, such as osteoblasts and
adipocytes (41–44). Our results demonstrated that there
are quantitative differences between different populations of MSCs
with respect to their differentiation potentials. Our data
demonstrated that of the 4 different populations of MSCs, the
AT-MSCs possessed the strongest adipogenic potential followed by
the WJ-MSCs, BM-MSCs and PL-MSCs. Although the BM-MSCs
differentiated into adipocytes earlier than the WJ-MSCs, the final
adipocyte ratio was lower in the BM-MSCs than in the WJ-MSCs.
Therefore, our data indicate that the WJ-MSCs have a greater
adipogenic potential than the BM-MSCs.
As regards osteogenesis, the results revealed that
the WJ-MSCs had the greatest potential in osteogenesis followed by
the PL-MSCs, AT-MSCs and MB-MSCs. Since the WJ and PL tisssues were
obtained from individuals who were much younger than the
individuals the AT and BM tissues were obtained from, we predicted
that the MSCs derived from younger tissue sources would
differentiate more easily into osteoblasts that those obtained from
older tissue sources. Baksh et al (35) also demonstrated that the
osteogenetic potential of WJ-MSCs was higher than that of
BM-MSCs.
It is a widely accepted fact that MSCs have
immunosuppressive and immunomodulatory functions. Due to their
ability to regulate immune responses, MSCs are a potential
candidate for treating a wide range of immune-mediated diseases
(45,46). Therefore, comparing the immune
properties of MSCs derived from different sources may have great
value in selecting the one which would be most effective in
clinical treatment.
HLA-DRA is one of the HLA class II α chain
paralogues. This class II molecule is a heterodimer consisting of
an α and a β chain, both anchored in the membrane. It plays a
central role in the immune system by presenting peptides derived
from extracellular proteins, such as HLA-DPB1. HLA-DMA is an
antigen processing and presentation-related gene. Our data
demonstrated that the WJ-MSCs expressed the lowest levels of the
HLA class II genes, HLA-DRA, HLA-DPB1 and HLA-DMA, than any other 3
populatinos of MSCs, while the BM-MSCs expressed the highest level
of these 3 genes. These data suggest that the WJ-MSCs may be the
most effective population of MSCs for clinical application.
We also analyzed the mRNA expression of JAG1, TLR4,
TLR3, NOTCH2 and NOTCH3 since they are expressed in MSCs and are
closely related to immune response (47). Our data demonstrated that the
expression levels of TLR4, TLR3, JAG1, NOTCH2 and NOTCH3 in the
BM-MSCs were 38-, 4-, 5-, 3- and 4-fold higher, respectively
compared with the WJ-MSCs. These data suggest that WJ-MSCs may have
the strongest immunosuppressive potential and produce only a
slight, if any immune response.
It has been shown that MSCs possess the intrinsic
homing ability to migrate to injured tissues and actively
participate in tissue repair. In addition, MSCs possess the unique
ability to suppress immune responses, both in vitro
(48) and in vivo
(49). In order to further assess
the immune characteristics of MSCs, we examined their direct effect
on mitogen (PHA/IL-2)-activated T cell proliferation using
co-culture experiments. PHA/IL-2-activated T cells were incubated
on a layer of MSCs derived from BM, AT, PL and WJ. The results
demonstrated that the suppressive effects of the MSCs derived from
AT, PL and WJ on T cell proliferation were more prominent than
those of the MSCs derived from BM. Our results are in accordance
with those of a previous study (50). Our data demonstrated that the
inhibitory effect of WJ-MSCs on T cell proliferation was the most
prominent. Previous studies have indicated that MSCs exert
immunosuppressive effects either through direct cell-cell contact
or by soluble factors (51).
Studies have implicated contact-dependent mechanisms, including the
expression of B7H1 on MSCs (51).
Other studies have reported that soluble factors secreted by MSCs,
or by immune cells in response to MSCs, play a major role in
MSC-mediated immune suppression (52). To determine this, we performed
co-culture experiments using both the contacted mix culture and the
Transwell system in which T cells and MSCs were physically
separated by a membrane permeable for soluble factors. Our data
revealed that the inhibitory effects of MSCs on T lymphocytes were
similar from both the contact culture and Transwell culture. This
illustrates that soluble factors may be involved in the
immunosuppressive properties of MSCs.
In this study, we demonstrate that the proliferative
potential of WJ-MSCs is higher than that of AT-MSCs, PL-MSCs and
BM-MSCs. WJ-MSCs have the strongest osteogenic ability followed by
the PL-MSCs, AT-MSCs and BM-MSCs. WJ-MSCs have the weakest
expression of MHC II genes, while BM-MSC have the highest. WJ-MSCs
have the weakest expression of the immune-related genes, TLR4,
TLR3, JAG1, NOTCH2 and NOTCH3. Furthermore, WJ-MSCs have the most
prominent suppressive effect on T lymphocytes among the 4
populations of MSCs in the co-culture experiments. Therefore,
WJ-MSCs may be one of the best sources of MSCs for tissue
regeneration in future clinical application.
References
|
1
|
Friedenstein AJ, Chailakhyan RK, Latsinik
NV, Panasyuk AF and Keiliss-Borok IV: Stromal cells responsible for
transferring the microenvironment of the hemopoietic tissues.
Cloning in vitro and retransplantation in vivo. Transplantation.
17:331–340. 1974. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Caplan AI: Mesenchymal stem cells. J
Orthop Res. 9:641–650. 1991. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Kopen GC, Prockop DJ and Phinney DG:
Marrow stromal cells migrate throughout forebrain and cerebellum,
and they differentiate into astrocytes after injection into
neonatal mouse brains. Proc Natl Acad Sci USA. 96:10711–10716.
1999. View Article : Google Scholar
|
|
4
|
Pittenger MF, Mackay AM, Beck SC, et al:
Multilineage potential of adult human mesenchymal stem cells.
Science. 284:143–147. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Beltrami AP, Cesselli D, Bergamin N, et
al: Multipotent cells can be generated in vitro from several adult
human organs (heart, liver, and bone marrow). Blood. 110:3438–3446.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Fan CG, Zhang QJ and Zhou JR: Therapeutic
potentials of mesenchymal stem cells derived from human umbilical
cord. Stem Cell Rev. 7:195–207. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Mosna F, Sensebe L and Krampera M: Human
bone marrow and adipose tissue mesenchymal stem cells: a user’s
guide. Stem Cells Dev. 19:1449–1470. 2010.
|
|
8
|
Friedenstein AJ, Petrakova KV, Kurolesova
AI and Frolova GP: Heterotopic of bone marrow. Analysis of
precursor cells for osteogenic and hematopoietic tissues.
Transplantation. 6:230–247. 1968.PubMed/NCBI
|
|
9
|
Tondreau T, Meuleman N, Delforge A, et al:
Mesenchymal stem cells derived from CD133-positive cells in
mobilized peripheral blood and cord blood: proliferation, Oct4
expression, and plasticity. Stem Cells. 23:1105–1112. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Zuk PA, Zhu M, Mizuno H, et al:
Multilineage cells from human adipose tissue: implications for
cell-based therapies. Tissue Eng. 7:211–228. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Campagnoli C, Roberts IA, Kumar S, Bennett
PR, Bellantuono I and Fisk NM: Identification of mesenchymal
stem/progenitor cells in human first-trimester fetal blood, liver,
and bone marrow. Blood. 98:2396–2402. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Keating A: Mesenchymal stromal cells. Curr
Opin Hematol. 13:419–425. 2006. View Article : Google Scholar
|
|
13
|
Friedenstein AJ, Chailakhjan RK and
Lalykina KS: The development of fibroblast colonies in monolayer
cultures of guinea-pig bone marrow and spleen cells. Cell Tissue
Kinet. 3:393–403. 1970.PubMed/NCBI
|
|
14
|
Friedenstein AJ, Gorskaja JF and Kulagina
NN: Fibroblast precursors in normal and irradiated mouse
hematopoietic organs. Exp Hematol. 4:267–274. 1976.PubMed/NCBI
|
|
15
|
Grassel S, Stockl S and Jenei-Lanzl Z:
Isolation, culture, and osteogenic/chondrogenic differentiation of
bone marrow-derived mesenchymal stem cells. Methods Mol Biol.
879:203–267. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Eslaminejad MB, Mirzadeh H, Mohamadi Y and
Nickmahzar A: Bone differentiation of marrow-derived mesenchymal
stem cells using beta-tricalcium phosphate-alginate-gelatin hybrid
scaffolds. J Tissue Eng Regen Med. 1:417–424. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Reyes M, Lund T, Lenvik T, Aguiar D,
Koodie L and Verfaillie CM: Purification and ex vivo expansion of
postnatal human marrow mesodermal progenitor cells. Blood.
98:2615–2625. 2001. View Article : Google Scholar
|
|
18
|
Tomic S, Djokic J, Vasilijic S, et al:
Immunomodulatory properties of mesenchymal stem cells derived from
dental pulp and dental follicle are susceptible to activation by
toll-like receptor agonists. Stem Cells Dev. 20:695–708. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Toubai T, Paczesny S, Shono Y, et al:
Mesenchymal stem cells for treatment and prevention of
graft-versus-host disease after allogeneic hematopoietic cell
transplantation. Curr Stem Cell Res Ther. 4:252–259. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Tse WT, Pendleton JD, Beyer WM, Egalka MC
and Guinan EC: Suppression of allogeneic T-cell proliferation by
human marrow stromal cells: implications in transplantation.
Transplantation. 75:389–397. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Ramasamy R, Tong CK, Seow HF, Vidyadaran S
and Dazzi F: The immunosuppressive effects of human bone
marrow-derived mesenchymal stem cells target T cell proliferation
but not its effector function. Cell Immunol. 251:131–136. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Che N, Li X, Zhou S, et al: Umbilical cord
mesenchymal stem cells suppress B-cell proliferation and
differentiation. Cell Immunol. 274:46–53. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Spaggiari GM, Capobianco A, Becchetti S,
Mingari MC and Moretta L: Mesenchymal stem cell-natural killer cell
interactions: evidence that activated NK cells are capable of
killing MSCs, whereas MSCs can inhibit IL-2-induced NK-cell
proliferation. Blood. 107:1484–1490. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Deuse T, Stubbendorff M, Tang-Quan K, et
al: Immunogenicity and immunomodulatory properties of umbilical
cord lining mesenchymal stem cells. Cell Transplant. 20:655–667.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Najar M, Raicevic G, Id Boufker H, et al:
Modulated expression of adhesion molecules and galectin-1: role
during mesenchymal stromal cell immunoregulatory functions. Exp
Hematol. 38:922–932. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
De Bruyn C, Najar M, Raicevic G, et al: A
rapid, simple, and reproducible method for the isolation of
mesenchymal stromal cells from Wharton’s jelly without enzymatic
treatment. Stem Cells Dev. 20:547–557. 2011.
|
|
27
|
Battula VL, Bareiss PM, Treml S, et al:
Human placenta and bone marrow derived MSC cultured in serum-free,
b-FGF-containing medium express cell surface frizzled-9 and SSEA-4
and give rise to multilineage differentiation. Differentiation.
75:279–291. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Young RG, Butler DL, Weber W, Caplan AI,
Gordon SL and Fink DJ: Use of mesenchymal stem cells in a collagen
matrix for Achilles tendon repair. J Orthop Res. 16:406–413. 1998.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Wakitani S, Saito T and Caplan AI:
Myogenic cells derived from rat bone marrow mesenchymal stem cells
exposed to 5-azacytidine. Muscle Nerve. 18:1417–1426. 1995.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Parker AM and Katz AJ: Adipose-derived
stem cells for the regeneration of damaged tissues. Expert Opin
Biol Ther. 6:567–578. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Kobayashi K, Kubota T and Aso T: Study on
myofibroblast differentiation in the stromal cells of Wharton’s
jelly: expression and localization of alpha-smooth muscle actin.
Early Hum Dev. 51:223–233. 1998.PubMed/NCBI
|
|
33
|
Wulf GG, Viereck V, Hemmerlein B, et al:
Mesengenic progenitor cells derived from human placenta. Tissue
Eng. 10:1136–1147. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Chen MY, Lie PC, Li ZL and Wei X:
Endothelial differentiation of Wharton’s jelly-derived mesenchymal
stem cells in comparison with bone marrow-derived mesenchymal stem
cells. Exp Hematol. 37:629–640. 2009.
|
|
35
|
Baksh D, Yao R and Tuan RS: Comparison of
proliferative and multilineage differentiation potential of human
mesenchymal stem cells derived from umbilical cord and bone marrow.
Stem Cells. 25:1384–1392. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Raynaud CM, Maleki M, Lis R, et al:
Comprehensive characterization of mesenchymal stem cells from human
placenta and fetal membrane and their response to osteoactivin
stimulation. Stem Cells Int. 2012:6583562012. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Zhu SF, Zhong ZN, Fu XF, et al: Comparison
of cell proliferation, apoptosis, cellular morphology and
ultrastructure between human umbilical cord and placenta-derived
mesenchymal stem cells. Neurosci Lett. 541:77–82. 2013. View Article : Google Scholar
|
|
38
|
Stenderup K, Justesen J, Clausen C and
Kassem M: Aging is associated with decreased maximal life span and
accelerated senescence of bone marrow stromal cells. Bone.
33:919–926. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Post S, Abdallah BM, Bentzon JF and Kassem
M: Demonstration of the presence of independent pre-osteoblastic
and pre-adipocytic cell populations in bone marrow-derived
mesenchymal stem cells. Bone. 43:32–39. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Larsen KH, Frederiksen CM, Burns JS,
Abdallah BM and Kassem M: Identifying a molecular phenotype for
bone marrow stromal cells with in vivo bone-forming capacity. J
Bone Miner Res. 25:796–808. 2010.PubMed/NCBI
|
|
41
|
De Ugarte DA, Morizono K, Elbarbary A, et
al: Comparison of multi-lineage cells from human adipose tissue and
bone marrow. Cells Tissues Organs. 174:101–109. 2003.PubMed/NCBI
|
|
42
|
Lorenz K, Sicker M, Schmelzer E, et al:
Multilineage differentiation potential of human dermal skin-derived
fibroblasts. Exp Dermatol. 17:925–932. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Kern S, Eichler H, Stoeve J, Kluter H and
Bieback K: Comparative analysis of mesenchymal stem cells from bone
marrow, umbilical cord blood, or adipose tissue. Stem Cells.
24:1294–1301. 2006. View Article : Google Scholar
|
|
44
|
Zhang X, Hirai M, Cantero S, et al:
Isolation and characterization of mesenchymal stem cells from human
umbilical cord blood: reevaluation of critical factors for
successful isolation and high ability to proliferate and
differentiate to chondrocytes as compared to mesenchymal stem cells
from bone marrow and adipose tissue. J Cell Biochem. 112:1206–1218.
2011.
|
|
45
|
Dazzi F and Krampera M: Mesenchymal stem
cells and autoimmune diseases. Best Pract Res Clin Haematol.
24:49–57. 2011. View Article : Google Scholar
|
|
46
|
Kebriaei P and Robinson S: Treatment of
graft-versus-host-disease with mesenchymal stromal cells.
Cytotherapy. 13:262–268. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Kode JA, Mukherjee S, Joglekar MV and
Hardikar AA: Mesenchymal stem cells: immunobiology and role in
immunomodulation and tissue regeneration. Cytotherapy. 11:377–391.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Krampera M, Glennie S, Dyson J, et al:
Bone marrow mesenchymal stem cells inhibit the response of naive
and memory antigen-specific T cells to their cognate peptide.
Blood. 101:3722–3729. 2003. View Article : Google Scholar
|
|
49
|
Polchert D, Sobinsky J, Douglas G, et al:
IFN-gamma activation of mesenchymal stem cells for treatment and
prevention of graft versus host disease. Eur J Immunol.
38:1745–1755. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Rubinstein P, Rosenfield RE, Adamson JW
and Stevens CE: Stored placental blood for unrelated bone marrow
reconstitution. Blood. 81:1679–1690. 1993.PubMed/NCBI
|
|
51
|
Augello A, Tasso R, Negrini SM, et al:
Bone marrow mesenchymal progenitor cells inhibit lymphocyte
proliferation by activation of the programmed death 1 pathway. Eur
J Immunol. 35:1482–1490. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Yang SH, Park MJ, Yoon IH, et al: Soluble
mediators from mesenchymal stem cells suppress T cell proliferation
by inducing IL-10. Exp Mol Med. 41:315–324. 2009. View Article : Google Scholar : PubMed/NCBI
|