Introduction
Dilated cardiomyopathy (DCM), which is clinically
characterized by progressive cardiac chamber enlargement and
contractile dysfunction with normal left ventricular wall
thickness, is the most prevalent form of primary myocardial
disease, with an estimated prevalence of 1/2,500 in the general
population (1). It is the third
most common cause of heart failure and the most frequent indication
for cardiac transplantation in adults and children (1). The entity of DCM has also been
recognized as a major cause of sudden cardiac death, accounting for
at least 30% of the overall mortality in DCM patients (2). The etiologies underlying DCM are
diverse, with both environmental and genetic risk factors involved
in the pathogenesis of DCM, although most cases remain idiopathic
(3). Approximately 25–50% of DCM
individuals had familial forms of the disease, highlighting the
important role of genetic defects in the pathogenesis of DCM
(4). Pathogenic mutations in
>50 genes have been associated with various types of DCM, of
which autosomal dominant inheritance is the most common type,
although other types, including X-linked, autosomal recessive and
mitochondrial inheritance, have also been reported (4–8).
Nevertheless, the DCM-related genes identified thus far explain
only a minority of DCM patients, and the genetic determinants
underpinning DCM in a large majority of cases remain elusive.
The GATA transcription factors are a family of zinc
finger-containing DNA-binding proteins, which preferentially bind
to a 5′-(A/T)GATA(A/G)-3′ motif within the regulatory region of
target genes (9). Six members of
the GATA family have been identified in vertebrates that are parsed
into two subfamilies based on their expression profiles. GATA1,
GATA2 and GATA3 are prominently expressed in hematopoietic cell
lineages, while GATA4, GATA5 and GATA6 are broadly expressed in
various mesoderm- and endoderm-derived tissues, particularly in the
heart (10). GATA4 and GATA6 are
highly expressed in the embryonic heart and continue the high
expression in the post-natal and adult myocardium, where they
function as crucial transcriptional regulators of various key
cardiac structural and regulatory genes, including the atrial
natriuretic factor (ANF), α- and β-myosin heavy chain, cardiac
troponin C, cardiac troponin I, cardiac-restricted ankyrin repeat
protein, sodium/calcium exchanger, A1 adenosine receptor,
m2 muscarinic receptor, and the myosin light chain 1/3
genes (10). In mice, the
overexpression of GATA4 or GATA6 was sufficient to induce cardiac
hypertrophy (11,12), whereas the cardiomyocyte-specific
conditional deletion of GATA6 significantly reduced the cardiac
hypertrophic response to pressure overload stimulation and rapidly
led to heart failure, similar to that observed in the mice with
heart-specific deletion of GATA4. Furthermore, the combinatorial
deletion of GATA4 and GATA6 from the adult heart resulted in DCM
and lethality by 16 weeks of age (12–14). In humans, mutations in GATA4 have
been associated with various cardiac phenotypes, including
congenital heart diseases, atrial fibrillation and DCM (15–25). Similarly, genetic variations in
GATA6 are also involved in the pathogenesis of congenital
cardiovascular malformations and atrial fibrillation (26–34), rendering it justifiable to screen
GATA6 as a prime candidate gene for DCM.
Materials and methods
Study participants
A cohort of 140 genetically unrelated patients with
DCM was prospectively enlisted from the Han Chinese population. The
available relatives of the index patients were also recruited. The
controls comprised 200 ethnically matched, unrelated healthy
individuals. Prior to enrollment in the study, all the subjects
were evaluated for detailed individual and familial histories,
underwent complete physical examination, chest radiography,
electrocardiogram, echocardiography, and exercise performance
testing. Cardiac catheterization, angiography, endomyocardial
biopsy, and cardiac magnetic resonance imaging were performed only
if there was a strong clinical indication requiring any these
procedures. Medical records were reviewed in all cases of deaths
thought to be related to DCM. Diagnosis of DCM was made based on
the criteria established by the World Health
Organization/International Society and Federation of Cardiology
Task Force on the Classification of Cardiomyopathy. The criteria
included a left ventricular end-diastolic diameter >27
mm/m2 and a left ventricular ejection fraction <40%
or fractional shortening <25% in the absence of abnormal loading
conditions, coronary artery disease, congenital heart lesions and
other systemic diseases (24,35). Exclusion criteria were
insufficient echocardiographic image quality, or coexistent
conditions that may lead to cardiac contractile dysfunction, such
as uncontrolled systemic hypertension, coronary artery or valvular
heart disease. A diagnosis of familial DCM was assigned when
occurring in at least two closely related family members (36). Individuals were classified as
healthy when found to be well with normal echocardiographic
parameters. Peripheral venous blood samples from all the
participants were collected. The clinical studies were conducted
with investigators blinded to the results of genetic testing. The
study was performed in accordance with the principles outlined in
the 1964 Declaration of Helsinki and its later amendments as well
as the ethics laws of China, and the study protocol was approved by
the local institutional ethics committee (of Shanghai Chest
Hospital, Shanghai Jiao Tong University, Shanghai, China). Written
informed consent was obtained from each participant prior to
enrollment in the study.
Mutational screening of GATA6
Genomic DNA was extracted from each subject from
whole blood leukocytes using a Wizard Genomic DNA Purification kit
(Promega, Madison, WI, USA). The coding regions and splice junction
sites of GATA6 were sequenced initially in 140 unrelated
patients with DCM and subsequently in the available relatives of
the index patients carrying the identified mutations and the 200
control individuals using the same method. The referential genomic
DNA sequence of GATA6 was derived from GenBank at the
National Center for Biotechnology Information (NCBI; accession no.
NT_010966; http://www.ncbi.nlm.nih.gov/nucleotide). The primer
pairs used to amplify the coding exons and flanking introns of
GATA6 by polymerase chain reaction (PCR) were designed as
described in a previous study (32). The PCR was performed using HotStar
Taq DNA Polymerase (Qiagen GmbH, Hilden, Germany) on a Veriti
Thermal Cycler (Applied Biosystems, Foster, CA, USA), with standard
conditions and concentrations of reagents. Amplified products were
analyzed on 1% agarose gels stained with ethidium bromide and
purified with QIAquick Gel Extraction kit (Qiagen GmbH). The two
strands of each PCR product were sequenced using a
BigDye® Terminator v3.1 Cycle Sequencing kit (Applied
Biosystems) on an ABI PRISM 3130 XL DNA Analyzer (Applied
Biosystems). The sequencing primers were identical to those
designed for the amplification of specific regions of GATA6.
The DNA sequences were viewed and assessed using DNA Sequencing
Analysis Software v5.1 (Applied Biosystems). A sequence variation
was verified by re-sequencing an independent PCR-generated amplicon
from the same subject and met our quality control thresholds with a
call rate of >99%. Additionally, for an identified sequence
variant, the single nucleotide polymorphism (SNP; http://www.ncbi.nlm.nih.gov/SNP) and human gene
mutation (HGM; http://www.hgmd.org) databases were
queried to confirm its novelty.
Comparison of amino acid sequences of
GATA6 proteins from various species
Amino acid sequences of GATA6 protein from human
(NP_005248.2) were aligned with those from rhesus monkey
(XP_002800933.1), cattle (XP_002697773.1), mouse (NP_034388.2), rat
(NP_062058.1), fowl (NP_990751.1), and zebrafish (NP_571632.1)
using the online program of MUSCLE, version 3.6 (http://www.ncbi.nlm.nih.gov).
Prediction of the disease-causing
potential of the GATA6 sequence variations
The causative potential of a GATA6 sequence
variation was assessed using MutationTaster (an online program at
http://www.mutationtaster.org), which
provides a probability for the variation to be a pathogenic
mutation or a benign polymorphism. The p-value utilized at this
point is the probability of the correct prediction as opposed to
the probability of error as used in t-test statistics (i.e. a value
close to 1 indicates a high ‘security’ of the prediction). The
online program PolyPhen-2 (http://genetics.bwh.harvard.edu/pph2) was also used to
assess the pathogenic similarity of an amino acid variation.
Plasmids and site-directed
mutagenesis
The expression vector pcDNA3-hGATA6 used in the
present study was generously provided by Dr Angela
Edwards-Ghatnekar, from the Division of Rheumatology and
Immunology, Medical University of South Carolina (Charleston, SC,
USA). The ANF-luciferase reporter plasmid, which contains the
2600-bp 5′-flanking region of the ANF gene, i.e.,
ANF(-2600)-Luc, was kindly provided by Dr Ichiro Shiojima, from the
Department of Cardiovascular Science and Medicine, Chiba University
Graduate School of Medicine (Chuo-ku, Chiba, Japan). Each
identified mutation was introduced into the plasmid containing the
wild-type human GATA6 cDNA to generate the mutant expression
vector using a QuickChange II XL Site-Directed Mutagenesis kit
(Stratagene, La Jolla, CA, USA) with a complementary pair of
primers, which was verified by direct sequencing.
Dual-luciferase reporter assay
HEK-293 cells plated onto 12-well plates at an
initial density of 2×104 cells/well were cultured in
Dulbecco’s modified Eagle’s medium supplemented with 10% fetal
bovine serum and 1% penicillin/streptomycin at 37°C with 5%
CO2, and grown to a confluence of ~80%. Transient
transfection was subsequently performed using
Lipofectamine® 2000 Transfection Reagent (Invitrogen
Life Technologies, Carlsbad, CA, USA) in accordance with the
manufacturer’s instructions. The ANF(-2600)-Luc reporter vector and
an internal control reporter plasmid pGL4.75 (hRluc/CMV; Promega)
were used in transfection assays to evaluate the transcriptional
activation function of the GATA6 mutants. HEK-293 cells were
transfected with 0.4 μg of empty vector pcDNA3, or 0.4 μg of
wild-type or mutant pcDNA3-hGATA6 expression plasmid, together with
0.4 μg of ANF(-2600)-Luc reporter construct and 0.04 μg of pGL4.75
control reporter vector. For the co-transfection experiments, 0.2
μg of wild-type pcDNA3-hGATA6, 0.2 μg of mutant pcDNA3-hGATA6, 0.4
μg of ANF(-2600)-Luc, and 0.04 μg of pGL4.75 were used. Firefly
luciferase and Renilla luciferase activities were measured
with the Dual-Glo luciferase assay system (Promega) 48 h after
transfection and normalized to the cells transfected with empty
vector. The activity of the ANF promoter was presented as
the fold activation of normalized Firefly luciferase relative to
normalized Renilla luciferase. Three independent experiments
were performed at minimum for wild-type and mutant GATA6.
Experiments were performed in triplicate
Statistical analysis
Data are expressed as means ± SD. Continuous
variables were tested for normality of distribution, and the
Student’s unpaired t-test was used for comparison of numeric
variables between two groups. Comparison of the categorical
variables between two groups was performed using Pearson’s
χ2 test or Fisher’s exact test when appropriate. A
two-tailed p<0.05 was considered to indicate a statistically
significant result.
Results
Clinical characteristics of the study
population
A cohort of 140 genetically unrelated patients with
DCM (76 males, mean age 53.2±12.8 years) was clinically evaluated
in contrast to a total of 200 ethnically matched, unrelated healthy
individuals (105 males, mean age 54.6±10.7 years) that were used as
controls. Of the 140 DCM patients, 51 had a positive family history
of DCM, while no family history of DCM was confirmed in the 200
control individuals. Compared with those in the control group,
blood pressure and left ventricular ejection fraction were
statistically decreased whereas the heart rate, left ventricular
end-diastolic diameter and left ventricular end-systolic diameter
were significantly increased in the patient group. The baseline
clinical characteristics of the 140 unrelated DCM patients are
shown in Table I.
 | Table IBaseline clinical characteristics of
the study subjects. |
Table I
Baseline clinical characteristics of
the study subjects.
| Variables | Patients
(n=140) | Controls
(n=200) |
|---|
| Age (years) | 53.2±12.8 | 54.6±10.7 |
| Male (%) | 76 (54.3) | 105 (52.5) |
| Family history of
DCM (%) | 51 (36.4)a | 0 (0) |
| SBP (mmHg) | 115.8±14.6a | 126.2±11.5 |
| DBP (mmHg) | 75.2±8.7a | 82.5±7.3 |
| HR (bpm) | 92.6±10.1a | 75.8±9.6 |
| LVEDD (mm) | 62.1±8.5a | 48.1±6.2 |
| LVESD (mm) | 51.5±7.2a | 32.7±6.0 |
| LVEF (%) | 38.9±5.3a | 64.2±6.5 |
| NYHA function class
(%) | | |
| I | 25 (17.9) | NA |
| II | 47 (33.6) | NA |
| III | 52 (37.1) | NA |
| IV | 16 (11.4) | NA |
Identification of GATA6 mutations in DCM
patients
By analysis of the protein coding sequence of
GATA6 in the 140 unrelated DCM patients, two heterozygous
missense mutations were identified in 2 of 140 patients,
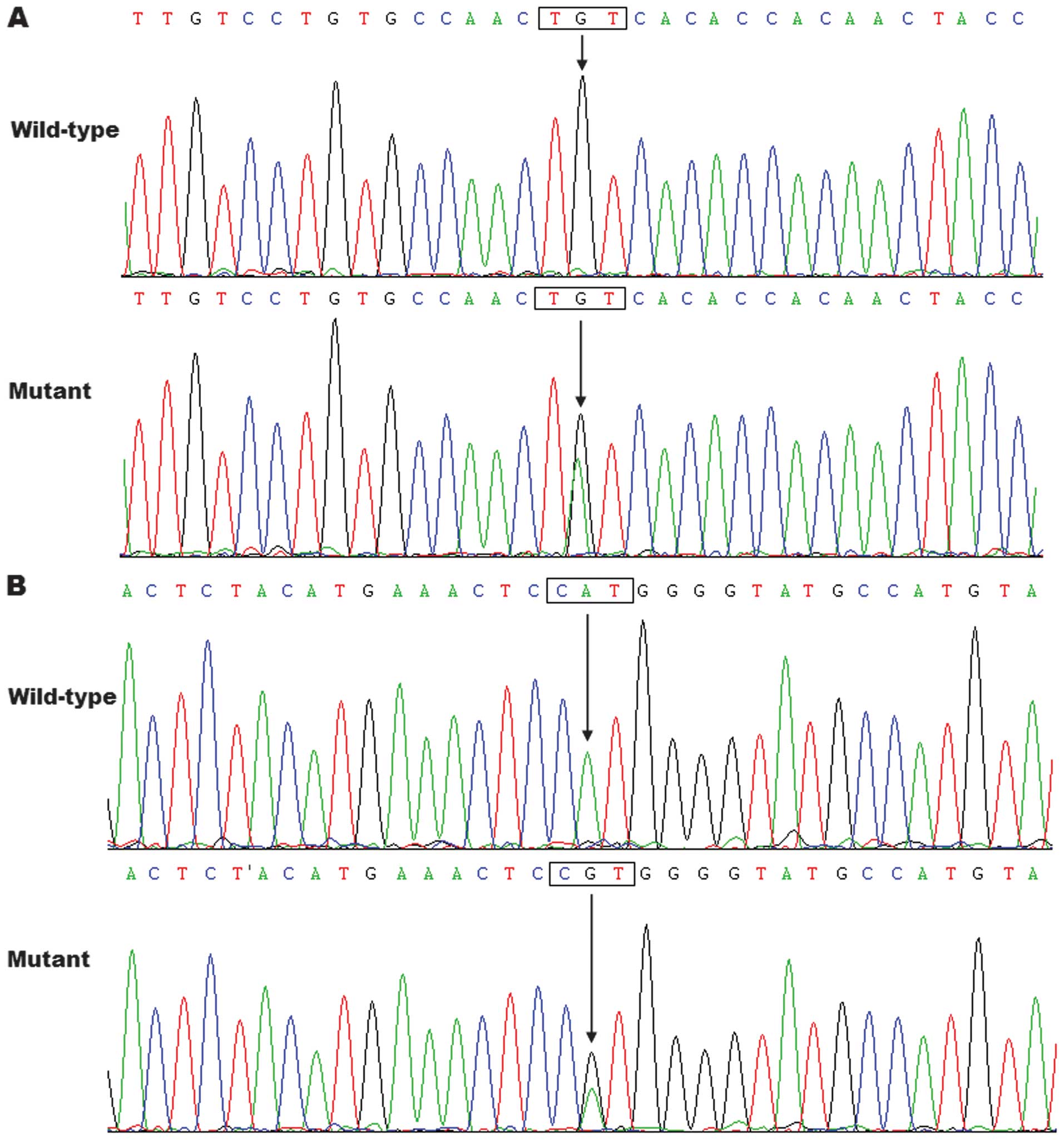
respectively, with a mutational prevalence of ~1.43%. Specifically,
a substitution of adenine for guanine in the second nucleotide of
codon 447 of the GATA6 gene (c.1340G>A), equivalent to
the transition of cysteine to tyrosine at amino acid position 447
(p.C447Y), was identified in the proband from family 1. A
transversion of adenine into guanine at coding nucleotide 1424
(c.1424A>G), predicting the change of histidine into arginine at
amino acid 475 (p.H475R), was identified in the index patient from
family 2. The sequence chromatograms showing the detected
heterozygous GATA6 variations in contrast to the
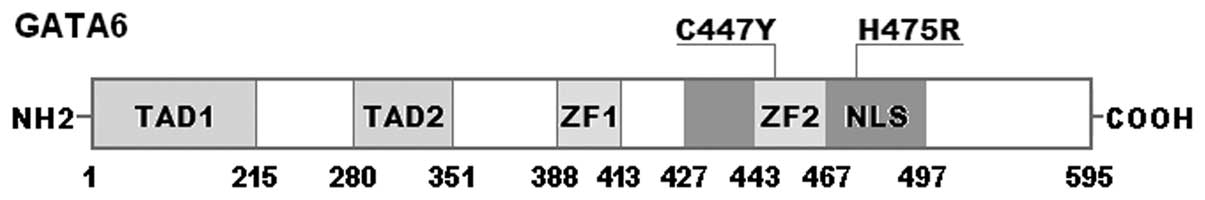
corresponding control sequences are shown in Fig. 1. A schematic diagram of GATA6
showing the structural domains and the locations of the identified
mutations is presented in Fig. 2.
The two variants were not observed in 400 control alleles or
identified in the SNP and HGM databases, which were consulted again
on January 21, 2014.
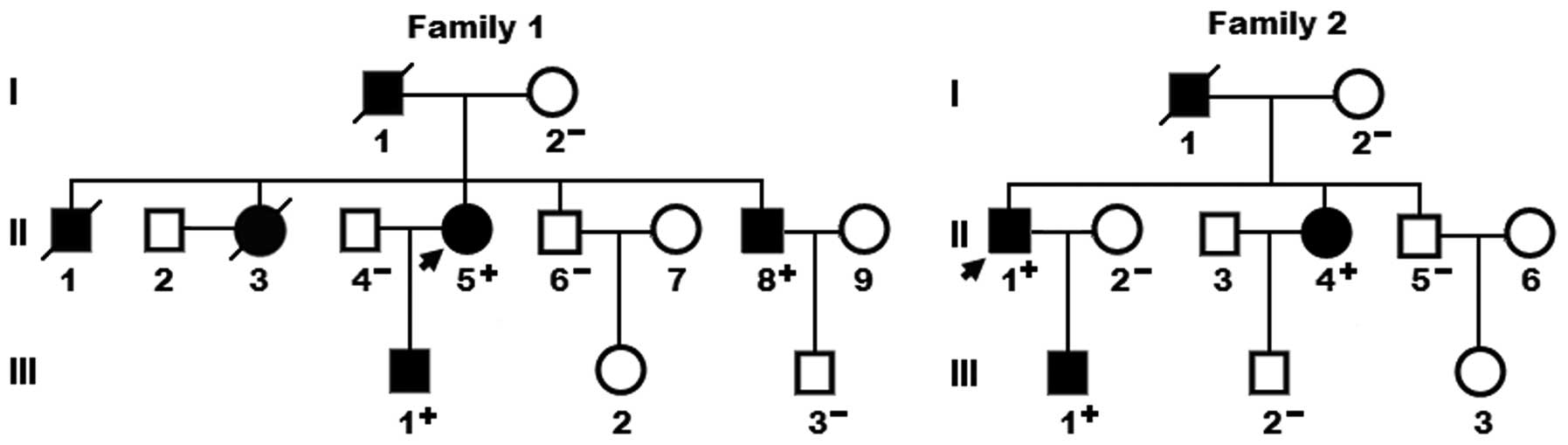
A genetic screen of family members of the mutation
carriers demonstrated that in each family, the variation was
present in all the affected family members available, but absent in
the unaffected family members examined. Analysis of the pedigrees
showed that in each family the variation co-segregated with DCM
transmitted in an autosomal dominant pattern with complete
penetrance. The pedigree structures of the two families are shown
in Fig. 3. In family 1, the
proband’s father (I-1) and two brothers (II-3 and II-8) also had an
electrocardiogram documented atrial fibrillation. The phenotypic
characteristics and results of genetic screening of the affected
pedigree members are shown in Table
II.
 | Table IIPhenotypic characteristics and status
of GATA6 mutations of the affected pedigree members. |
Table II
Phenotypic characteristics and status
of GATA6 mutations of the affected pedigree members.
| Subject
information | | Phenotypes | Echocardiogram | | Genotypes |
|---|
| |
|
| |
|
|---|
| Identity | Gender | Age (years) | Cardiac
phenotypes | LVEDD (nm) | LVESD (nm) | LVEF (%) | GATA6
mutations |
|---|
| Family 1 | | | | | | | C447Y |
| I-1 | M | 60a | DCM, AF | NA | NA | NA | NA |
| II-1 | M | 21a | DCM | NA | NA | NA | NA |
| II-3 | F | 53a | DCM, AF | NA | NA | NA | NA |
| II-5 | F | 55 | DCM | 74 | 66 | 28 | +/− |
| II-8 | M | 50 | DCM, AF | 60 | 48 | 35 | +/− |
| III-1 | M | 29 | DCM | 52 | 37 | 45 | +/− |
| Family 2 | | | | | | | H475R |
| I-1 | M | 62a | DCM | NA | NA | NA | NA |
| II-1 | M | 58 | DCM | 68 | 58 | 37 | +/− |
| II-4 | F | 55 | DCM | 56 | 44 | 40 | +/− |
| III-1 | M | 32 | DCM | 47 | 36 | 46 | +/− |
Multiple alignments of GATA6 protein
sequences among various species
A cross-species alignment of multiple GATA6 protein
sequences showed that the affected amino acids were highly
conserved from human to zebrafish, indicating that these amino
acids are functionally important (Fig. 4).
Disease-causing potential of GATA6
sequence variations
The GATA6 sequence variations of c.1340G>A
and c.1424A>G were predicted to be disease-causing, with an
identical p-value of 1.00000. No SNPs in the altered regions were
identified in the MutationTaster database. The amino acid
substitutions of C447Y and H475R in GATA6 were also predicted by
PolyPhen-2 to be damaging, with scores of 1.000 (sensitivity 0.00;
specificity 1.00) for p.C447Y and 0.972 (sensitivity 0.77;
specificity 0.96) for p.H475R, respectively.
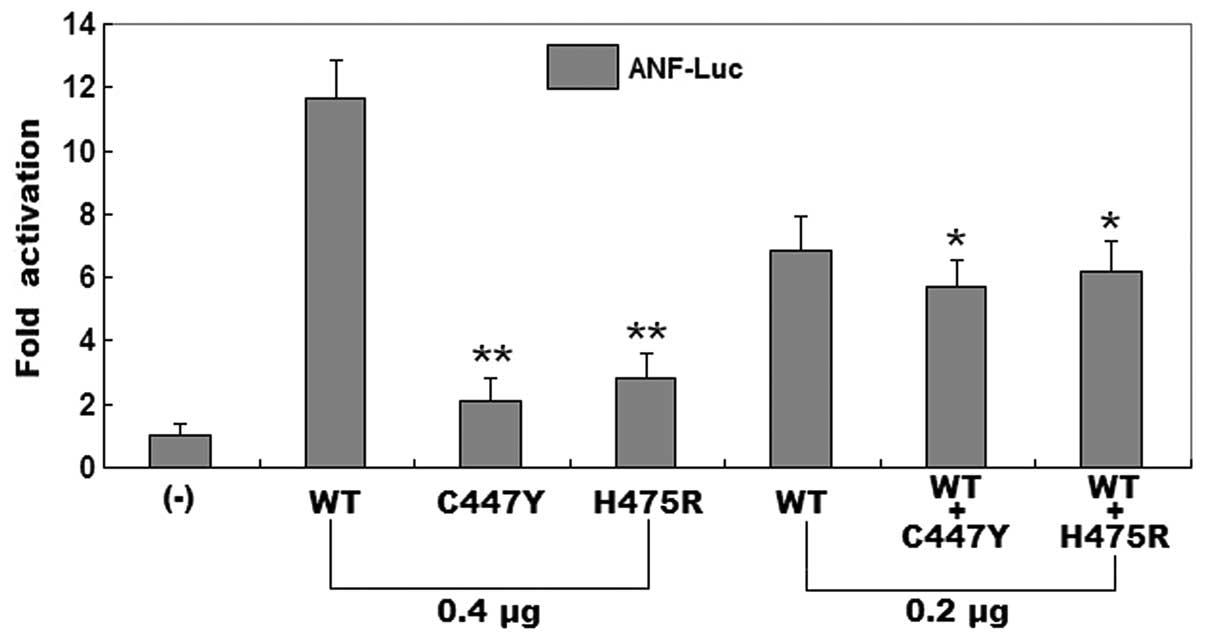
Reduced transcriptional activity of the
GATA6 mutants
The wild-type GATA6, C447Y-mutant GATA6 and
H475R-mutant GATA6 activated the ANF promoter by ~12-, ~2- and
~3-fold, respectively. When wild-type GATA6 was co-expressed with
the same amount of C447Y-mutant GATA6 or H475R-mutant GATA6, the
induced activation of the ANF promoter was the same as ~6-fold.
These results showed that the two GATA6 mutants are associated with
significantly reduced transcriptional activity compared with their
wild-type counterpart (Fig.
5).
Discussion
In the current study, two novel heterozygous GATA6
mutations, p.C447Y and p.H475R, were identified in two families
with DCM. In each family the mutation co-segregated with DCM
transmitted in an autosomal dominant mode with complete penetrance.
The two mutations, which were absent in 400 referential chromosomes
from an ethnically matched control population, altered the amino
acids that were completely conserved evolutionarily and were
predicted to be pathogenic. Functional assays showed that the
mutant GATA6 proteins were associated with significantly reduced
transcriptional activity. Therefore, it is likely that functionally
compromised GATA6 contribute to DCM in these families.
Human GATA6 gene maps to chromosome
18q11.1-q11.2, coding for a protein with 595 amino acids (37). The functional domains of GATA6
comprise two transcriptional activation domains (TAD1, amino acids
1–215; TAD2, amino acids 280–351), two adjacent zinc fingers (ZF1,
amino acids 388–413; ZF2, amino acids 443–467), and one nuclear
localization signal (NLS, amino acids 427–497). The two TADs are
important for the appropriate transcriptional activity of GATA6.
The N-terminal ZF2 is responsible for DNA sequence recognition and
binding to the consensus motif, while the C-terminal ZF1 is crucial
for the sequence specificity and stability of protein-DNA binding.
The NLS is required for the sub-cellular trafficking and
distribution of GATA6 (33). The
GATA6 mutations p.C447Y and p.H475R identified in the present study
are located in ZF2 and NLS, respectively, and may therefore exert
impact on the transcriptional activity of GATA6 by interfering with
the binding to target DNA or nuclear distribution of GATA6.
GATA6 has been found to mediate the expression of
several target genes during embryogenesis and cardiac
morphogenesis, including the genes that encode atrial natriuretic
factor (ANF), brain natriuretic peptide, α- and β-myosin heavy
chain, and gap junction protein Cx40 (10,38). Thus, the functional effect of the
GATA6 mutation may be investigated by assay of the
transcriptional activity of a target gene promoter in tool cells.
In the present study, the functional characteristics of the novel
GATA6 mutations identified in the DCM patients were determined by
transcriptional activation analysis and the results showed a
significantly decreased transcriptional activity on the downstream
gene, ANF. These findings suggest that haploinsufficiency or
a dominant-negative effect resulting from GATA6 loss-of-function
mutation is potentially an alternative molecular mechanism
underpinning DCM.
Association of genetically defective GATA6
with increased vulnerability to DCM has been established in
animals. In zebrafish, embryos depleted of GATA6 developed
variable cardiac morphogenetic defects including cardia
bifida, partially fused tube, and fused but non-looping tube
(39). By contrast, zebrafish
embryos depleted of GATA4 and GATA6 were heartless
and restoring either gene product was sufficient to rescue
cardiomyocyte specification (40). In mice, among the mammalian GATA
factors that have been identified thus far (GATA1-GATA6), GATA6 was
the earliest expressed during embryonic development, and
GATA6-deficient embryos died shortly after implantation (41). Although the mice heterozygous for
a GATA4 or GATA6 null allele were normal, compound heterozygosity
of GATA4 and GATA6 resulted in embryonic lethality accompanied by a
spectrum of cardiovascular defects, including thin-walled
myocardium, ventricular septal defect, persistent truncus
arteriosis, double outlet right ventricle, myocardial hypoplasia,
reduced proliferation of cardiomyocytes, and the impaired
differentiation of vascular smooth muscle cells (42,43). The mice with a
cardiomyocyte-specific conditional deletion of GATA6, which
lacked >95% of GATA6 protein in the heart, were viable and
survived into adulthood, but they were predisposed to progressive
deterioration in cardiac function and enlargement of heart in
adulthood, a phenotype similar to that of GATA4 heart-specific
deleted mice. Furthermore, the combinatorial deletion of GATA4 and
GATA6 from the adult murine heart led to DCM (12). By contrast, the overexpression of
GATA6 was sufficient to induce myocardial hypertrophy in
vitro and in vivo, both alone and in combination with
GATA4 (11,12). These experimental results
emphasize the pivotal role of GATA6 in the development and
remodeling of the heart.
GATA6 has been confirmed to regulate the expression
of multiple key cardiac genes alone or in cooperation with its
transcriptionally synergistic partners, such as GATA4, NKX2–5 and
TBX20, and in humans, an increasing number of mutations in such
target molecules as α-actin, α-myosin heavy chain, troponin C, and
troponin I, as well as in the transcriptional cooperative partners
of GATA6, including GATA4, NKX2–5 and TBX20, are involved in the
pathogenesis of DCM (10,24,25,44,45). These findings suggest that
functionally compromised GATA6 predisposes to DCM probably by
reducing the expression of genes essential for cardiac structure
and function.
Atrial fibrillation was documented in three DCM
patients from family 1, consistent with previous studies on the
association of GATA6 mutations with atrial fibrillation (32–34). Similarly, mutations in other
cardiac transcriptional factor genes, such as GATA4,
GATA5, NKX2–5 and PITX2c, were also associated
with atrial fibrillation (21–23,46–53). These observations support the
hypothesis that a subset of atrial fibrillation may have
developmental origin.
In conclusion, to the best of our knowledge, this is
the first study to connect GATA6 loss-of-function mutations with
enhanced susceptibility to DCM, providing novel insight into the
molecular mechanism of DCM, suggesting potential implications for
the antenatal prophylaxis and allele-specific treatment of DCM.
Acknowledgements
We are grateful to the participants for their
dedication to the study. This study was supported by a grant from
the National Natural Science Fund of China (no. 81270161) and a
grant from the Natural Science Fund of Shanghai, China (no.
14ZR1438000).
References
|
1
|
Maron BJ, Towbin JA, Thiene G,
Antzelevitch C, Corrado D, Arnett D, Moss AJ, Seidman CE and Young
JB: American Heart Association; Council on Clinical Cardiology,
Heart Failure and Transplantation Committee; Quality of Care and
Outcomes Research and Functional Genomics and Translational Biology
Interdisciplinary Working Groups; Council on Epidemiology and
Prevention: Contemporary definitions and classification of the
cardiomyopathies: an American Heart Association Scientific
Statement from the Council on Clinical Cardiology, Heart Failure
and Transplantation Committee; Quality of Care and Outcomes
Research and Functional Genomics and Translational Biology
Interdisciplinary Working Groups; and Council on Epidemiology and
Prevention. Circulation. 113:1807–1816. 2006.
|
|
2
|
Sen-Chowdhry S and McKenna WJ: Sudden
death from genetic and acquired cardiomyopathies. Circulation.
125:1563–1576. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Towbin JA, Lowe AM, Colan SD, Sleeper LA,
Orav EJ, Clunie S, Messere J, Cox GF, Lurie PR, Hsu D, Canter C,
Wilkinson JD and Lipshultz SE: Incidence, causes, and outcomes of
dilated cardiomyopathy in children. JAMA. 296:1867–1876. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
McNally EM, Golbus JR and Puckelwartz MJ:
Genetic mutations and mechanisms in dilated cardiomyopathy. J Clin
Invest. 123:19–26. 2013. View
Article : Google Scholar : PubMed/NCBI
|
|
5
|
Refaat MM, Lubitz SA, Makino S, Islam Z,
Frangiskakis JM, Mehdi H, Gutmann R, Zhang ML, Bloom HL, MacRae CA,
Dudley SC, Shalaby AA, Weiss R, McNamara DM, London B and Ellinor
PT: Genetic variation in the alternative splicing regulator RBM20
is associated with dilated cardiomyopathy. Heart Rhythm. 9:390–396.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Disertori M, Quintarelli S, Grasso M,
Pilotto A, Narula N, Favalli V, Canclini C, Diegoli M, Mazzola S,
Marini M, Del Greco M, Bonmassari R, Masè M, Ravelli F, Specchia C
and Arbustini E: Autosomal recessive atrial dilated cardiomyopathy
with standstill evolution associated with mutation of Natriuretic
Peptide Precursor A. Circ Cardiovasc Genet. 6:27–36. 2013.
View Article : Google Scholar
|
|
7
|
Paavola J, Schliffke S, Rossetti S, Kuo
IY, Yuan S, Sun Z, Harris PC, Torres VE and Ehrlich BE:
Polycystin-2 mutations lead to impaired calcium cycling in the
heart and predispose to dilated cardiomyopathy. J Mol Cell Cardiol.
58:199–208. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Wahbi K, Béhin A, Bécane HM, Leturcq F,
Cossée M, Laforêt P, Stojkovic T, Carlier P, Toussaint M, Gaxotte
V, Cluzel P, Eymard B and Duboc D: Dilated cardiomyopathy in
patients with mutations in anoctamin 5. Int J Cardiol. 168:76–79.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Oka T, Xu J and Molkentin JD:
Re-employment of developmental transcription factors in adult heart
disease. Semin Cell Dev Biol. 18:117–131. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Molkentin JD: The zinc finger-containing
transcription factors GATA-4, -5, and -6. Ubiquitously expressed
regulators of tissue-specific gene expression. J Biol Chem.
275:38949–38952. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Liang Q, De Windt LJ, Witt SA, Kimball TR,
Markham BE and Molkentin JD: The transcription factors GATA4 and
GATA6 regulate cardiomyocyte hypertrophy in vitro and in vivo. J
Biol Chem. 276:30245–30253. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
van Berlo JH, Elrod JW, van den Hoogenhof
MM, York AJ, Aronow BJ, Duncan SA and Molkentin JD: The
transcription factor GATA-6 regulates pathological cardiac
hypertrophy. Circ Res. 107:1032–1040. 2010.PubMed/NCBI
|
|
13
|
Bisping E, Ikeda S, Kong SW, Tarnavski O,
Bodyak N, McMullen JR, Rajagopal S, Son JK, Ma Q, Springer Z, Kang
PM, Izumo S and Pu WT: Gata4 is required for maintenance of
postnatal cardiac function and protection from pressure
overload-induced heart failure. Proc Natl Acad Sci USA.
103:14471–14476. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Oka T, Maillet M, Watt AJ, Schwartz RJ,
Aronow BJ, Duncan SA and Molkentin JD: Cardiac-specific deletion of
Gata4 reveals its requirement for hypertrophy, compensation, and
myocyte viability. Circ Res. 98:837–845. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Garg V, Kathiriya IS, Barnes R,
Schluterman MK, King IN, Butler CA, Rothrock CR, Eapen RS,
Hirayama-Yamada K, Joo K, Matsuoka R, Cohen JC and Srivastava D:
GATA4 mutations cause human congenital heart defects and reveal an
interaction with TBX5. Nature. 424:443–447. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Rajagopal SK, Ma Q, Obler D, Shen J,
Manichaikul A, Tomita-Mitchell A, Boardman K, Briggs C, Garg V,
Srivastava D, Goldmuntz E, Broman KW, Benson DW, Smoot LB and Pu
WT: Spectrum of heart disease associated with murine and human
GATA4 mutation. J Mol Cell Cardiol. 43:677–685. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Wang J, Fang M, Liu XY, Xin YF, Liu ZM,
Chen XZ, Wang XZ, Fang WY, Liu X and Yang YQ: A novel GATA4
mutation responsible for congenital ventricular septal defects. Int
J Mol Med. 28:557–564. 2011.PubMed/NCBI
|
|
18
|
Liu XY, Wang J, Zheng JH, Bai K, Liu ZM,
Wang XZ, Liu X, Fang WY and Yang YQ: Involvement of a novel GATA4
mutation in atrial septal defects. Int J Mol Med. 28:17–23.
2011.PubMed/NCBI
|
|
19
|
McCulley DJ and Black BL: Transcription
factor pathways and congenital heart disease. Curr Top Dev Biol.
100:253–277. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Yang YQ, Gharibeh L, Li RG, Xin YF, Wang
J, Liu ZM, Qiu XB, Xu YJ, Xu L, Qu XK, Liu X, Fang WY, Huang RT,
Xue S and Nemer G: GATA4 loss-of-function mutations underlie
familial tetralogy of Fallot. Hum Mutat. 34:1662–1671. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Posch MG, Boldt LH, Polotzki M, Richter S,
Rolf S, Perrot A, Dietz R, Ozcelik C and Haverkamp W: Mutations in
the cardiac transcription factor GATA4 in patients with lone atrial
fibrillation. Eur J Med Genet. 53:201–203. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Jiang JQ, Shen FF, Fang WY, Liu X and Yang
YQ: Novel GATA4 mutations in lone atrial fibrillation. Int J Mol
Med. 28:1025–1032. 2011.PubMed/NCBI
|
|
23
|
Wang J, Sun YM and Yang YQ: Mutation
spectrum of the GATA4 gene in patients with idiopathic atrial
fibrillation. Mol Biol Rep. 39:8127–8135. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Li RG, Li L, Qiu XB, Yuan F, Xu L, Li X,
Xu YJ, Jiang WF, Jiang JQ, Liu X, Fang WY, Zhang M, Peng LY, Qu XK
and Yang YQ: GATA4 loss-of-function mutation underlies familial
dilated cardiomyopathy. Biochem Biophys Res Commun. 439:591–596.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Zhao L, Xu JH, Xu WJ, Yu H, Wang Q, Zheng
HZ, Jiang WF, Jiang JF and Yang YQ: A novel GATA4 loss-of-function
mutation responsible for familial dilated cardiomyopathy. Int J Mol
Med. 33:654–660. 2014.PubMed/NCBI
|
|
26
|
Kodo K, Nishizawa T, Furutani M, Arai S,
Yamamura E, Joo K, Takahashi T, Matsuoka R and Yamagishi H: GATA6
mutations cause human cardiac outflow tract defects by disrupting
semaphorin-plexin signaling. Proc Natl Acad Sci USA.
106:13933–13938. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Lin X, Huo Z, Liu X, Zhang Y, Li L, Zhao
H, Yan B, Liu Y, Yang Y and Chen YH: A novel GATA6 mutation in
patients with tetralogy of Fallot or atrial septal defect. J Hum
Genet. 55:662–667. 2010. View Article : Google Scholar
|
|
28
|
Maitra M, Koenig SN, Srivastava D and Garg
V: Identification of GATA6 sequence variants in patients with
congenital heart defects. Pediatr Res. 68:281–285. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Wang J, Luo XJ, Xin YF, Liu Y, Liu ZM,
Wang Q, Li RG, Fang WY, Wang XZ and Yang YQ: Novel GATA6 mutations
associated with congenital ventricular septal defect or tetralogy
of fallot. DNA Cell Biol. 31:1610–1617. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Zheng GF, Wei D, Zhao H, Zhou N, Yang YQ
and Liu XY: A novel GATA6 mutation associated with
congenital ventricular septal defect. Int J Mol Med. 29:1065–1071.
2012.
|
|
31
|
Huang RT, Xue S, Xu YJ and Yang YQ:
Somatic mutations in the GATA6 gene underlie sporadic
tetralogy of Fallot. Int J Mol Med. 31:51–58. 2013.
|
|
32
|
Yang YQ, Wang XH, Tan HW, Jiang WF, Fang
WY and Liu X: Prevalence and spectrum of GATA6 mutations associated
with familial atrial fibrillation. Int J Cardiol. 155:494–496.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Li J, Liu WD, Yang ZL and Yang YQ: Novel
GATA6 loss-of-function mutation responsible for familial atrial
fibrillation. Int J Mol Med. 30:783–790. 2012.PubMed/NCBI
|
|
34
|
Yang YQ, Li L, Wang J, Zhang XL, Li RG, Xu
YJ, Tan HW, Wang XH, Jiang JQ, Fang WY and Liu X: GATA6
loss-of-function mutation in atrial fibrillation. Eur J Med Genet.
55:520–526. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Elliott P, O’Mahony C, Syrris P, Evans A,
Rivera Sorensen C, Sheppard MN, Carr-White G, Pantazis A and
McKenna WJ: Prevalence of desmosomal protein gene mutations in
patients with dilated cardiomyopathy. Circ Cardiovasc Genet.
3:314–322. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Mestroni L, Rocco C, Gregori D, Sinagra G,
Di Lenarda A, Miocic S, Vatta M, Pinamonti B, Muntoni F, Caforio
AL, McKenna WJ, Falaschi A, Giacca M and Camerini: Familial dilated
cardiomyopathy: evidence for genetic and phenotypic heterogeneity.
Heart Muscle Disease Study Group. J Am Coll Cardiol. 34:181–190.
1999. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Suzuki E, Evans T, Lowry J, Truong L, Bell
DW, Testa JR and Walsh K: The human GATA-6 gene: structure,
chromosomal location, and regulation of expression by
tissue-specific and mitogen-responsive signals. Genomics.
38:283–290. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Rémond MC, Iaffaldano G, O’Quinn MP,
Mezentseva NV, Garcia V, Harris BS, Gourdie RG, Eisenberg CA and
Eisenberg LM: GATA6 reporter gene reveals myocardial phenotypic
heterogeneity that is related to variations in gap junction
coupling. Am J Physiol Heart Circ Physiol. 301:H1952–H1964.
2011.PubMed/NCBI
|
|
39
|
Peterkin T, Gibson A and Patient R: GATA-6
maintains BMP-4 and Nkx2 expression during cardiomyocyte precursor
maturation. EMBO J. 22:4260–4273. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Holtzinger A and Evans T: Gata5 and Gata6
are functionally redundant in zebrafish for specification of
cardiomyocytes. Dev Biol. 312:613–622. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Koutsourakis M, Langeveld A, Patient R,
Beddington R and Grosveld F: The transcription factor GATA6 is
essential for early extraembryonic development. Development.
126:723–732. 1999.
|
|
42
|
Xin M, Davis CA, Molkentin JD, Lien CL,
Duncan SA, Richardson JA and Olson EN: A threshold of GATA4 and
GATA6 expression is required for cardiovascular development. Proc
Natl Acad Sci USA. 103:11189–11194. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Zhao R, Watt AJ, Battle MA, Li J, Bondow
BJ and Duncan SA: Loss of both GATA4 and GATA6 blocks cardiac
myocyte differentiation and results in acardia in mice. Dev Biol.
317:614–619. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Costa MW, Guo G, Wolstein O, Vale M,
Castro ML, Wang L, Otway R, Riek P, Cochrane N, Furtado M,
Semsarian C, Weintraub RG, Yeoh T, Hayward C, Keogh A, Macdonald P,
Feneley M, Graham RM, Seidman JG, Seidman CE, Rosenthal N, Fatkin D
and Harvey RP: Functional characterization of a novel mutation in
NKX2–5 associated with congenital heart disease and adult-onset
cardiomyopathy. Circ Cardiovasc Genet. 6:238–247. 2013.
|
|
45
|
Kirk EP, Sunde M, Costa MW, Rankin SA,
Wolstein O, Castro ML, Butler TL, Hyun C, Guo G, Otway R, Mackay
JP, Waddell LB, Cole AD, Hayward C, Keogh A, Macdonald P, Griffiths
L, Fatkin D, Sholler GF, Zorn AM, Feneley MP, Winlaw DS and Harvey
RP: Mutations in cardiac T-box factor gene TBX20 are associated
with diverse cardiac pathologies, including defects of septation
and valvulogenesis and cardiomyopathy. Am J Hum Genet. 81:280–291.
2007. View
Article : Google Scholar : PubMed/NCBI
|
|
46
|
Yang YQ, Wang J, Wang XH, Wang Q, Tan HW,
Zhang M, Shen FF, Jiang JQ, Fang WY and Liu X: Mutational spectrum
of the GATA5 gene associated with familial atrial fibrillation. Int
J Cardiol. 157:305–307. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Gu JY, Xu JH, Yu H and Yang YQ: Novel
GATA5 loss-of-function mutations underlie familial atrial
fibrillation. Clinics (Sao Paulo). 67:1393–1399. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Wang XH, Huang CX, Wang Q, Li RG, Xu YJ,
Liu X, Fang WY and Yang YQ: A novel GATA5 loss-of-function mutation
underlies lone atrial fibrillation. Int J Mol Med. 31:43–50.
2013.PubMed/NCBI
|
|
49
|
Huang RT, Xue S, Xu YJ, Zhou M and Yang
YQ: A novel NKX2.5 loss-of-function mutation responsible for
familial atrial fibrillation. Int J Mol Med. 31:1119–1126.
2013.PubMed/NCBI
|
|
50
|
Xie WH, Chang C, Xu YJ, Li RG, Qu XK, Fang
WY, Liu X and Yang YQ: Prevalence and spectrum of Nkx2.5 mutations
associated with idiopathic atrial fibrillation. Clinics (Sao
Paulo). 68:777–784. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Yang YQ, Xu YJ, Li RG, Qu XK, Fang WY and
Liu X: Prevalence and spectrum of PITX2c mutations associated with
familial atrial fibrillation. Int J Cardiol. 168:2873–2876. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Zhou YM, Zheng PX, Yang YQ, Ge ZM and Kang
WQ: A novel PITX2c loss-of-function mutation underlies lone atrial
fibrillation. Int J Mol Med. 32:827–834. 2013.PubMed/NCBI
|
|
53
|
Wang J, Zhang DF, Sun YM and Yang YQ: A
novel PITX2c loss-of-function mutation associated with familial
atrial fibrillation. Eur J Med Genet. 57:25–31. 2014. View Article : Google Scholar : PubMed/NCBI
|