1. Introduction
Intercellular communication in a multicellular
organism is essential for its proper functionality. Traditionally,
soluble factors released by different types of cells were
considered to serve such a role in a localized environment
(1). With the emergence and
discovery of small membrane sacs, later termed as extracellular
vesicles (EVs), that are released by various cell types, the
concept of ‘cell-to-cell talk’ has been re-evaluated (2,3).
It is now widely accepted that EVs have a substantial contribution
to intercellular communication by shuttling bioactive molecules
(transmembrane receptors, mRNAs, miRNAs and signaling molecules)
that are able to modulate the extracellular environment (4–9).
The term EV implies to all shed membrane vesicles, which can be
further classified on the basis of their size, origin and their
cargo (10). In general, however,
EVs have been divided into two groups, exosomes and microvesicles
(5,10,11). Exosomes are formed through
invagination into endosomes to form multivesicular bodies (MVBs)
and are thought to be somewhat unique in their protein and lipid
composition (10). Due to their
endosomal origin, exosomes contain membrane transport and fusion
proteins (GTPases, Annexins and flotillin), tetraspannins (CD9,
CD63, CD81 and CD82), heat shock proteins (Hsc70 and Hsp90),
proteins involved in multivesicular body biogenesis (Alix and
TSG101), as well as lipid-related proteins and phospholipases
(10,12,13), although other markers have also
been associated with them (14).
On the other hand, microvesicles are particles shed from the plasma
membrane following stimulation and are often a hallmark of cell
apoptosis (15–17). Upon cell activation, as a result
of stimuli, such as shear stress or cytokine/endotoxin release, the
cytosolic calcium concentration increases, leading to the
activation of a number of enzymes (calpains, gelsolins, scramblases
and kinases) (16). This, in
turn, leads to the inhibition of enzymes, such as translocases and
phosphatases, resulting in cytoskeletal reorganization, the loss of
membrane asymmetry and membrane blebbing, which causes microvesicle
formation and release (16,18). Microvesicles have a rich
phospholipid bilayer consisting of phosphatidyl serine on the outer
leaflet and their membrane proteins reflect those of the cell they
originate from (15,17,18).
2. Concept of EV isolation
The concept of EV isolation begins with the nature
of the sample itself. First of all, one has to realize that
different samples present unique obstacles when it comes to EV
isolation, which is greatly related to the origin of the EV. Thus,
with respect to the source, EVs can be divided into two groups: i)
those isolated from cell culture media, and ii) those isolated from
body fluids (plasma, urine, spinal fluid, saliva, etc.).
3. Cell culture medium
When isolating EVs from conditioned cell culture
media one has to consider the presence of an additional,
‘artificial’ EV source, namely fetal bovine serum (FBS) or any
other supplement for that matter, which is routinely added to cell
cultures (19,20). Our results obtained by
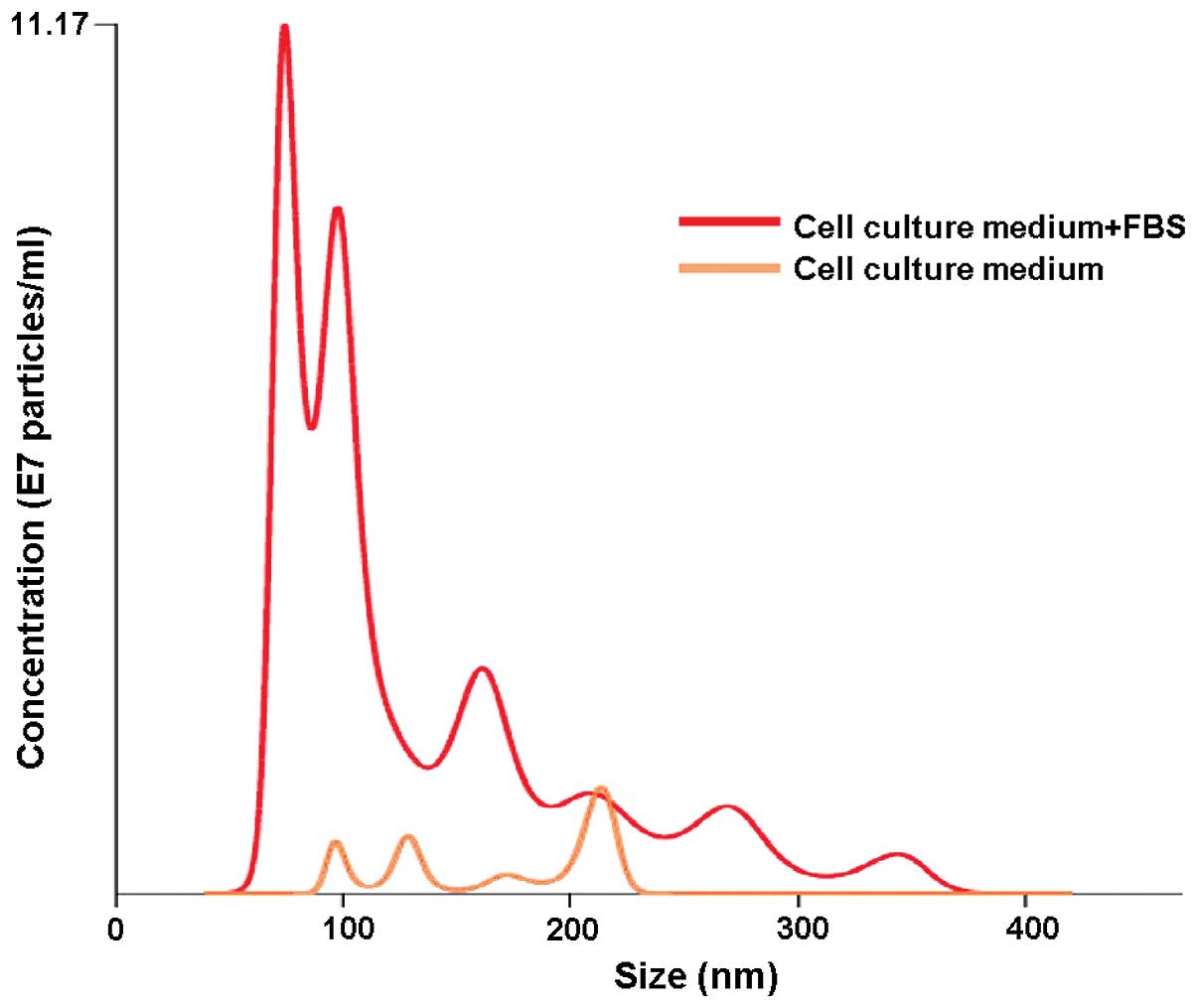
nanoparticle tracking analysis (NTA) showed freshly prepared cell
culture medium supplemented with FBS to have a substantial particle
population (mostly <100 nm in size) already present before the
actual use (Fig. 1). This
observation alone clearly demonstrates that the downstream EV
isolation from conditioned cell culture media carries along the
risk of obtaining, apart from proper EVs, also those from FBS,
which obviously, obscures the final results. This observation is in
accordance with observations of other groups, which have also shown
FBS to contain vesicles that may be later isolated along the actual
EV (19–21). To counter that problem, many
groups apply filters to remove EVs from the media/FBS or use a long
ultracentrifugation step. In an elegant study, Lötvall et al
showed that the removal of FBS-originated EVs is critical for
further downstream experiments, since these vesicles are capable of
inducing effects similar to those of EVs isolated from the actual
cell line culture media (21).
Thus, it has been proposed that a 16 h-ultracentrifugation step at
100,000 × g or greater for FBS is absolutely obligatory for a
complete FBS vesicle depletion, since shorter centrifugation steps
are insufficient (19,21). An alternative may be the use of an
exosome-free FBS, which is already commercially available, but is
still rather expensive. Some also postulate the use of bovine serum
albumin (BSA) instead of the standard FBS (20).
Another major issue involving EV isolation from
conditioned cell culture media regards the culture medium itself.
Our NTA results revealed that even a newly opened cell culture
medium contains a trace of particles that resemble EVs size-wise
(Fig. 1). The presence of these
background particles itself puts tremendous strain on the integrity
of the final EV isolation results. Similar results were obtained by
Jeppesen et al, who compared two different cell culture
media and showed that one type had more particle background than
the other (19). Moreover, the
storage temperature also seems to have an effect on the occurrence
of the particles in a cell culture medium. A cell culture medium
stored at room temperature showed more background particles than
the one stored at 4°C as observed by NTA, which was even more
apparent with time passage (own data). It has been suggested that
in such cases, ultracentrifugation for an extended period of time
(16 h or longer) is necessary for the removal of particle
background from the cell culture media (19).
There seems to be a strong demand for establishing a
uniform protocol for EV isolation, particularly for EVs which are
being isolated from cell culture media; however, this may not be so
simple. Studies have shown that different cell lines generate
unique EVs, suggesting that they should be considered individually
with respect to the isolation approach. In their study, Jeppesen
et al showed that the optimal isolation conditions for EVs
obtained from embryonic kidney HEK293 and bladder carcinoma FL3
cells differed from each other (19). Their study indicated that the
optimal vesicle-to-protein yield for HEK293 cells was obtained at
67,000 × g, while that for FL3 cells was at 100,000 × g.
Furthermore, it was demonstrated that specific g-force/k-factor
usage during differential centrifugation greatly influences the
purity and yield of exosomes (19).
4. Body fluids
EV isolation from body fluids seems to be even more
complex. Although the problem of the FBS vesicle presence obviously
does not apply in this case, there are other factors (lipoproteins,
DNA, RNA, protein aggregates and microbes) that need to be
addressed for proper isolation (22–26). The majority of the available data
concerns the overall EV population isolated from blood samples;
however, there are attempts to ‘select out’ from that EV pool a
certain type of EV depending on its origin (i.e., platelet,
epithelial, leukocyte and tumor). Again, a careful approach needs
to be undertaken for the isolation of the ‘wanted’ EV fraction(s).
For example, if the isolation of leukocyte-origin EV is desired,
the sample needs to be depleted of platelets. For that to be
accomplished, platelets need to be centrifuged at a certain speed
and under appropriate conditions (discussed in further detail
below) to avoid platelet activation and, consequently, the
generation of EVs of platelet origin (27,28). It has to be kept in mind that a
blood sample (or any other body fluid sample for that matter) is a
source of many types of EVs. Realizing the complexity of such
samples puts another perspective on the EV fraction isolation
approaches.
5. Storage and isolation conditions
There are no strictly defined conditions for
storing/isolating EVs. One exception, however, are platelet-derived
EVs, where firm guidelines for their proper acquisition have been
laid down by the Vascular Biology group of the Scientific and
Standardization Committee of the International Society on
Thrombosis and Hemostasis over a decade ago (27–29). Although some basic framework has
been established for other EVs, many groups have still
independently developed their own storage/isolation protocols that
are suitable to their individual laboratory settings (30). There is a general understanding
that freshly acquired and processed samples guarantee the best EV
yield; however, that is, for the most part, not possible (30,31).
Body fluid samples (blood, serum, plasma, urine and
tumor ascites) are collected/stored in a number of ways. For
example, there are studies demonstrating that blood samples
collected on different anticoagulants, when processed further
downstream, exhibit different EV yields (27,28). It has been shown that platelet and
endothelial EV counts were substantially lower in blood samples
collected in citrate or EDTA than in the ones collected in protease
inhibitors, either hirudin and soybean trypsin inhibitor or heparin
(27–29,32). Some groups follow strict protocols
and process their blood samples within 1 h after collection
(33). Others believe that a
fasting period (of up to 12 h) prior to the sample collection is
necessary for proper EV acquisition (27,28,34). On the other hand, others have
indicated that storing blood samples, or any other body fluid
samples for that matter, at 4°C for up to 5 days does not affect
the final EV yield (20).
Observations have also been made that the EV diameter significantly
decreases with the duration of the storage period (e.g., within 2
days) and with the increasing temperature during storage (from
4–37°C) (35). Freezing and
thawing cycles are also critical. Most groups are in agreement that
multiple freezing-thawing cycles of a sample affect EV
characteristics/concentration, although some suggest that repeating
these cycles for up to 10 times has no influence on the size and
composition of EVs to any significant degree (29–32,35). Moreover, the thawing conditions
alone seem to play a crucial role in EV recovery. It has been
observed that EV samples thawed on ice showed a lower EV recovery
compared to the ones thawed at room temperature or 37°C (36). To resolve this issue, aliquots of
once obtained EV stocks are stored at −70 to −80°C until use for up
to a year, which then undergo only one freezing/thawing cycle
(20,34). Zhou et al reported that the
storage of EVs isolated from urine at −20°C resulted in a major
loss of EVs, while storage at −80°C had no effect on the EV
recovery (37).
In our opinion, the general rule for storage and
isolation conditions should be ‘the sooner, the better’. The
starting sample should be handled rapidly after collection,
avoiding extensive waiting periods between further processing
stages (i.e., centrifugation steps). In blood/plasma samples, all
the necessary precautions (i.e., processing temperature, upright
sample position for transport, no agitation) should be undertaken
to avoid platelet activation, and thus potential platelet-derived
EV generation. Moreover, once isolated, EV aliquots should be
prepared, and they should undergo only one freezing/thawing cycle
at an appropriate temperature to ensure optimal EV recovery.
6. Isolation
There are basically three major methodologies used
for EV purification/isolation that serve as the backbone for
potential method variations: i) differential
centrifugation/ultracentrifugation with/without a sucrose
gradient/cushion; ii) adsorption to magnetic/non-magnetic
microbeads; and iii) size exclusion chromatography.
Differential
centrifugation/ultracentrifugation
This is probably the most commonly used method for
EV isolation/purification (20,30,38). As with other methods, there are
variations depending on the laboratory setting; however, for the
most part, the protocols follow the scheme that was put forward in
the study by Raposo et al, who purified exosomes from the
conditioned culture media of transformed human B cell lines
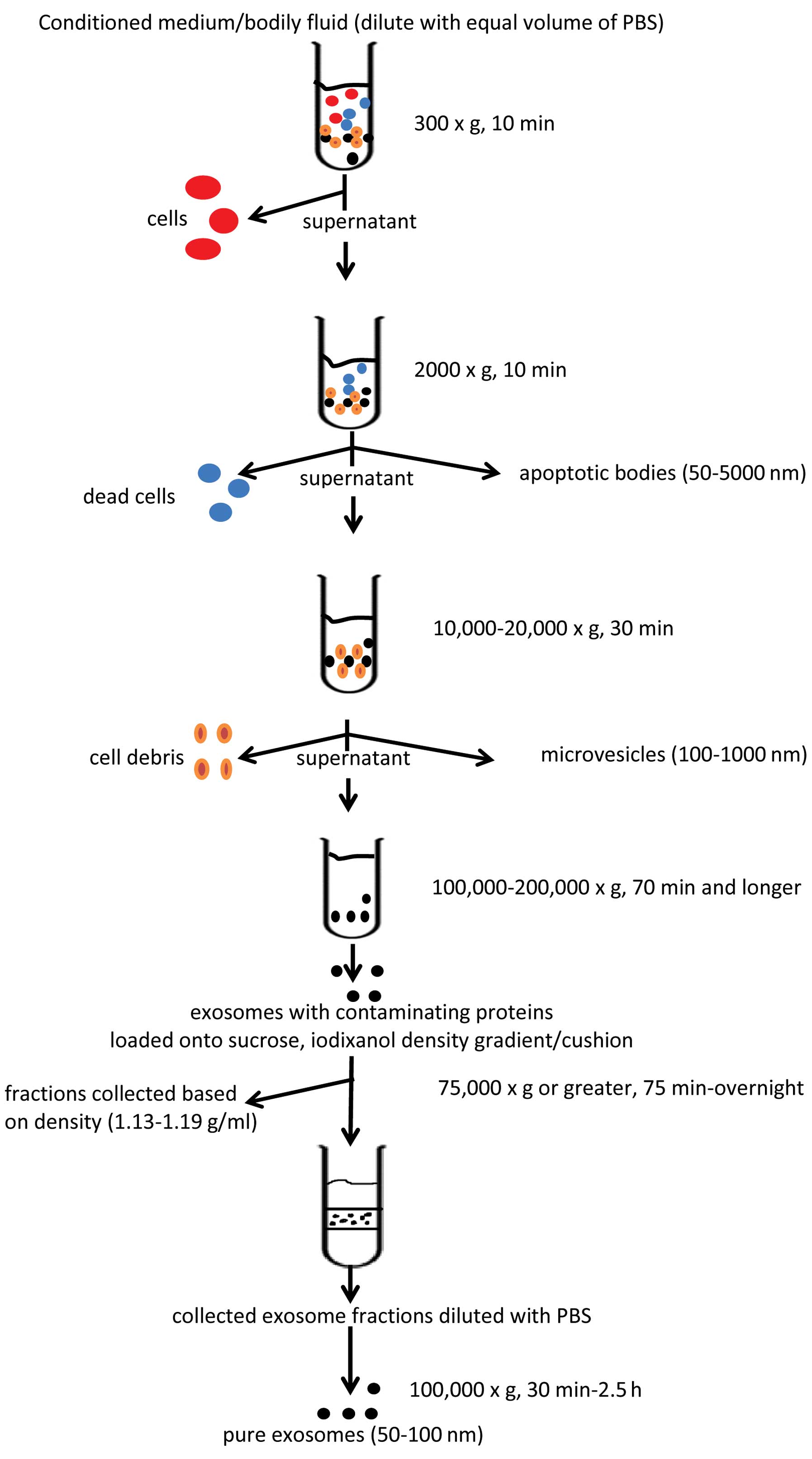
(38). The protocol involves a
number of sequential centrifugation steps at different centrifugal
forces (g) whose purpose is to remove unwanted components from the
actual exosomes. The first three steps of the protocol are designed
to remove intact cells, dead cells or cell debris using three
different centrifugal forces, that is 300 × g for 10 min, 2,000 × g
for 10 min and 10,000 × g for 30 min, respectively. After each
centrifugation, the supernatant is transferred into a new test tube
while the generated pellets are being discarded. After the 10,000 ×
g spin, the supernatant is then subjected to a final
ultracentrifugation at 100,000 × g for 70 min. The outcome of this
step is an exosome pellet that can be used for further studies. It
should be also noted that all the centrifugation steps are being
carried out at 4°C. The basic scheme for EV isolation is presented
in Fig. 2.
Although the protocol presented in the study by
Raposo et al (38) served
as the backbone for others, it focused only on the purification of
exosomes, a portion of the EV population with a size of <100 nm,
and their isolation from conditioned cell culture media. With the
discovery of EVs being present in all types of body fluids, there
is an increasing need to adopt the protocol for appropriate samples
with the incorporation of a step(s) enabling the isolation of EVs
with a larger size (>100 nm). The EVs that can be obtained from
a body fluid sample are divided into four distinct EV populations:
exosomes, microvesicles, apoptotic bodies and microsomes (19). Exosomes (40–100 nm), the most
extensively studied EV population, are usually isolated by
centrifugation at 100,000–200,000 × g (20,39), whereas microvesicles (100–1,000
nm) are isolated by centrifugation at 10,000–20,000 × g (30,40). Apoptotic bodies (50–5,000 nm) are
obtained at a g-force of approximately 2,000 × g (19), whereas microsomes are 80–120 nm in
size and their isolation/identification needs to be confirmed by
additional means (19,41,42). For the isolation of EVs from
plasma, lymph fluid, urine, bronchiolar lavage fluid and tumor
ascites, Théry et al proposed diluting the samples with an
equal volume of PBS before further processing due to the viscosity
of the respective fluids (20).
Moreover, due to the complexity of the viscous fluid samples the
time and centrifugation speeds have been increased/adapted to
obtain the appropriate EV populations. The major difference
compared to the protocol presented in the study by Raposo et
al (38) was made at the
beginning of the modified protocol. Here, after the initial
centrifugation step at 300 × g, another additional centrifugation
at 2,000–3,000 × g for up to 30 min is performed, followed by the
12,000–15,000 × g step for up to 1 h. The purpose of the first step
is to deplete the sample of cells/cellular debris. The second step
enables the acquiring of apoptotic bodies, whereas the third one
eliminates platelets/platelet EVs (plasma samples) and/or isolates
microvesicles. Some groups at this point check whether
platelets/platelet EV have been removed from the centrifuged
samples by anti-CD41, -CD61 monoclonal antibody staining and flow
cytometry analysis (40). Only
platelet-free plasma (PFP) and platelet EV-free plasma samples
undergo further processing. It should be noted that, although some
groups recommend centrifugation at 4°C at all times, that should
not be the case when dealing with plasma samples. The processing of
these samples at this temperature leads to platelet activation,
which in turn causes the generation of platelet EVs that may become
difficult to remove downstream.
A variation of the EV isolation method by
centrifugation that has been adopted by some groups includes an
additional sucrose gradient/cushion step (43–45). Some argue that during EV isolation
by centrifugation, aggregates of large proteins and/or proteins
that were non-specifically associated with EVs are also being
sedimented (20,45). A sucrose gradient (20–60%)/cushion
(30% sucrose) step incorporated into the centrifugation protocol
supposedly eliminates this contamination, and the resulting EV
population seems to be of a greater purity. The diluted (with PBS)
EV suspension obtained by the protocol discussed above is gently
loaded onto the Tris/sucrose/D2O solution containing
tube and ultracentrifuged for a period of time (75 min up to
overnight at 75,000 × g or greater) (20,44–47). Following centrifugation, the EV
fractions are collected based on their density, that ranges between
1.13–1.19 g/ml, diluted with PBS and centrifuged again for 30
min–2.5 h, at 100,000 × g (20,44–47). Although the incorporation of the
density gradient step into the EV isolation protocol has been
designed to purify and isolate specific EV fractions, it has been
recently reported that certain high-density lipoproteins (HDLs) can
also be isolated using this method (48). An alternative to these problems
may be the OptiPrep velocity gradient. In this method, 5–40%
iodixanol gradient is used instead of sucrose, showing, reportedly,
an improved separation of EVs from viral particles and small
apoptotic bodies (47). Moreover,
unlike sucrose, iodixanol is capable of forming iso-osmotic
solutions at all densities, thus better preserving the size of the
vesicles in the gradient (46).
Immunoaffinity isolation
Another method used for EV isolation involves
microbeads, usually magnetic, that are coated with an antibody that
recognizes certain markers present on the EV surface. This
technology can be used for EV isolation from either cell culture
media or body fluids. After mixing the EV sample with the
antibody-coated microbeads, a magnetic force is applied (i.e., to a
column, microplate) which retains the EV-covered microbeads, while
the rest of the sample is discarded (www.systembio.com/exosomes). Next, the microbeads with
attached EV are eluted using appropriate buffers and used for
further analysis. The advantage of this isolation method is its
ability to select a specific EV population based on a marker
expression regardless of its size. Many groups put in a lot of
effort into selecting an EV fraction (i.e., exosomes and
microvesicles) based on EV size, but not on the surface marker
profile. Although this approach may result in obtaining valuable
information on such EV fractions, it limits, however, the overall
scope on the impact that the whole EV population, regardless of
size, may exert on the surroundings. This is particularly evident
in an in vivo setting where different types of cells may
generate EVs of different sizes, but still carry the same surface
marker (10). Another important
advantage is the ability of coupling this method with other methods
(i.e., flow cytometry, western blotting and ‘real-time’ PCR) to
characterize even further the already selected/specific EV
fraction. At the same time, however, it needs to be pointed out
that the beads, due to their physical binding surface area, can
only bind a certain number of EVs, which, in turn, can lead to a
substantial population of EVs being still left out from the sample
or lost during the purification process.
An alternative to magnetic microbeads are
surfactant-free latex beads typically made of polystyrene (49), which are stabilized against
aggregation by covalently linked charge groups (sulfate, carboxyl,
amidine, carboxyl/sulfate, aldehyde/sulfate, chloromethyl and
aldehyde/amidine) and have ~95% of their surface available for
passive adsorption of proteins (www.Invitrogen.com). They can be either hydrophobic or
hydrophilic and can covalently bind EVs regardless of their size or
surface marker composition. Due to this feature, the bound EVs can
be further characterized by, for example, flow cytometry using
multi-antibody staining, thus providing valuable information on the
EVs. Although this method can lead to the acquisition of
informative data on EVs, their elution from the beads is difficult.
This is a major disadvantage to this method, since the bound EVs
cannot be used in downstream experiments.
Size exclusion chromatography
This method is usually coupled with a low-speed
centrifugation step that allows the removal of larger objects from
the sample (cells, cellular debris, organelles, etc.) that is
followed by a filtration step (0.8 and 0.2 µm pore size
filter) to pre-concentrate the EVs. The filtered EV sample is then
subjected to size exclusion chromatography (usually gel filtration
column) where small volume fractions (1 ml) of the filtrate are
collected and ultracentrifuged (100,000 × g, 1 h and longer) to
pellet down the EVs (50–52). The principal behind this technique
is that particles in a sample, depending on their size, will move
through the filtration column at different rates. Thus, larger
particles will elute more rapidly, while the smaller ones more
slowly, due to their ability to penetrate the stationary phase
(gel) of the column. In theory, the obtained eluted fraction at a
certain time should contain a population of particles of the same
particle size. The pelleted EVs are resuspended in PBS and used in
downstream assays.
Although this method is generally established as one
of the methods for EV isolation, it poses some concerns that need
to be addressed. Forcing EV passage through filters used to
pre-concentrate the sample may lead to EV deformation and eventual
rapture into smaller particles (30). To avoid this, it has been
suggested that size exclusion chromatography should be performed by
gravity or with the application of the smallest possible force
(23,30). Moreover, the selection of the
appropriate gel type is crucial to the recovery of EVs, rather than
proteins or lipoproteins. Additionally, the short isolation time
and relatively low cost are also beneficial.
7. Polymeric precipitation
Although polymeric precipitation methods do not fall
under the isolation method classification proposed above, it is
worthwhile to mention them as an interesting and promising
alternative. The concept behind this method is the formation of a
meshlike polymeric web that captures EVs of a certain size, usually
between 60–180 nm, which are later pelleted at low centrifugal
speeds. It can be used to obtain EVs from both cell culture medium
and/or body fluid samples. What is very appealing about this EV
isolation method is the fact that it is relatively quick, enables
high EV recoveries and does not require laborious
ultracentrifugation (53). A
previous study showed the superiority of this method over others;
however, it only assessed the RNA yield and protein purity/quantity
(54); thus, its possible
application in the overall EV recovery still needs to be addressed.
Others have raised a concern regarding the contaminants, such as
lipoproteins, that may be isolated along with the actual EVs
(30). Moreover, since polymeric
precipitation isolates EVs of 60–180 nm in size, it cannot be used
for the assessment of larger EVs present in a sample.
8. Biological activities of EVs and
associated challenges
It is rather difficult to evaluate how the different
isolation protocols affect the potential biological activities of
EVs. The main reason for this is that the majority of available
data concerns EVs isolated by a single method, and this mostly
involves differential centrifugation/ultracentrifugation. At the
same time, studies focusing on the comparison of the biological
effects of EVs isolated by more than one protocol simply are
unavailable, at least to the best of our knowledge. Most groups
choose one isolation protocol through which EVs are obtained and
their effect is then evaluated depending on the experimental
design. For instance, exosomes isolated from red blood cells by
ultracentrifugation are able to induce the release of
pro-inflammatory cytokine in monocytes (55). Another study demonstrated that
density-gradient isolated glioblastoma-derived EVs modified the
phenotype of monocytic cells (56). It has also been shown that RNA
transferred by macrophage-derived microvesicles isolated by
ultracentrifugation is biologically active and induces macrophage
differentiation (57). Although
there are attempts to compare two or more EV isolation methods, the
results are limited to the correlation of EV physical properties
(e.g., number, size distribution, phenotype, protein and/or RNA
expression), and do not show the biological effects. For example,
in a study on EVs present in blood and urine, it was suggested that
polymeric precipitation yielded the highest EV concentration,
although CD133 and CD63 protein expression in these EVs was
difficult to interpret compared to EVs obtained by other methods
(58). What makes the assessment
of EV biological activities even more difficult is that there is no
single isolation protocol to refer to as the ‘golden standard’
guaranteeing not just the complete recovery of EVs (all the
fractions), but also the recovery of EVs that retain their native
form/shape and function. Thus, it is simply unknown as to whether
the obtained EVs, using either one of the protocols discussed
above, comprise the original EVs released by the parent cells. Due
to these discrepancies, additional simultaneous studies on the
effects of different isolation methods are warranted for the proper
evaluation of EV biological activities.
9. Conclusion
The past decade has witnessed an extensive research
in the EV field. As we learn more about EVs and their role under
normal and/or pathological conditions, there is an increasing
temptation of trying to use that knowledge in the treatment of
certain diseases. Although elevated numbers of EVs or EVs bearing
certain proteins, RNAs, miRNAs, etc. have been tagged as diagnostic
and prognostic markers in a number of diseases, there still seems
to be a question pending as to the validity of this information.
The answer to this question begins with the establishment of firm
EV isolation protocol(s) that would enable the assessment of the
full spectrum of EVs present in a sample instantaneously. Available
isolation protocols only in part meet this goal, as they tend to
focus on the selection of a certain type(s) of EV, whether based on
their size, density or marker expression. The outcomes of these
approaches produce either inadequate EV numbers/concentration or
contaminated EV population(s)/fraction(s), whose usefulness in
downstream tests may be questionable. The isolation methods
presented in this review comprise the current knowledge on the
topic, shedding a light on the complexity of the EV ‘world’.
Acknowledgments
This review was supported by the grant no.
UMO-2012/07/B/NZ6/03499 financed by the National Science Centre of
Poland.
References
|
1
|
D’Souza-Schorey C and Clancy JW:
Tumor-derived microvesicles: Shedding light on novel
microenvironment modulators and prospective cancer biomarkers.
Genes Dev. 26:1287–1299. 2012. View Article : Google Scholar
|
|
2
|
Ratajczak J, Miekus K, Kucia M, Zhang J,
Reca R, Dvorak P and Ratajczak MZ: Embryonic stem cell-derived
microvesicles reprogram hematopoietic progenitors: Evidence for
horizontal transfer of mRNA and protein delivery. Leukemia.
20:847–856. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Mause SF and Weber C: Microparticles:
Protagonists of a novel communication network for intercellular
information exchange. Circ Res. 107:1047–1057. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Ratajczak J, Wysoczynski M, Hayek F,
Janowska-Wieczorek A and Ratajczak MZ: Membrane-derived
microvesicles: Important and underappreciated mediators of
cell-to-cell communication. Leukemia. 20:1487–1495. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Cocucci E, Racchetti G and Meldolesi J:
Shedding microvesicles: Artefacts no more. Trends Cell Biol.
19:43–51. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
van der Pol E, Hoekstra AG, Sturk A, Otto
C, van Leeuwen TG and Nieuwland R: Optical and non-optical methods
for detection and characterization of microparticles and exosomes.
J Thromb Haemost. 8:2596–2607. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Valadi H, Ekström K, Bossios A, Sjöstrand
M, Lee JJ and Lötvall JO: Exosome-mediated transfer of mRNAs and
microRNAs is a novel mechanism of genetic exchange between cells.
Nat Cell Biol. 9:654–659. 2007. View
Article : Google Scholar : PubMed/NCBI
|
|
8
|
Baj-Krzyworzeka M, Szatanek R, Weglarczyk
K, Baran J, Urbanowicz B, Brański P, Ratajczak MZ and Zembala M:
Tumour-derived microvesicles carry several surface determinants and
mRNA of tumour cells and transfer some of these determinants to
monocytes. Cancer Immunol Immunother. 55:808–818. 2006. View Article : Google Scholar
|
|
9
|
Al-Nedawi K, Meehan B, Micallef J, Lhotak
V, May L, Guha A and Rak J: Intercellular transfer of the oncogenic
receptor EGFRvIII by microvesicles derived from tumour cells. Nat
Cell Biol. 10:619–624. 2008. View
Article : Google Scholar : PubMed/NCBI
|
|
10
|
Vlassov AV, Magdaleno S, Setterquist R and
Conrad R: Exosomes: Current knowledge of their composition,
biological functions, and diagnostic and therapeutic potentials.
Biochim Biophys Acta. 1820:940–948. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Simpson RJ, Jensen SS and Lim JW:
Proteomic profiling of exosomes: Current perspectives. Proteomics.
8:4083–4099. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Conde-Vancells J, Rodriguez-Suarez E,
Embade N, Gil D, Matthiesen R, Valle M, Elortza F, Lu SC, Mato JM
and Falcon-Perez JM: Characterization and comprehensive proteome
profiling of exosomes secreted by hepatocytes. J Proteome Res.
7:5157–5166. 2008. View Article : Google Scholar
|
|
13
|
Subra C, Grand D, Laulagnier K, Stella A,
Lambeau G, Paillasse M, De Medina P, Monsarrat B, Perret B,
Silvente-Poirot S, et al: Exosomes account for vesicle-mediated
transcellular transport of activatable phospholipases and
prostaglandins. J Lipid Res. 51:2105–2120. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Mathivanan S and Simpson RJ: ExoCarta: A
compendium of exosomal proteins and RNA. Proteomics. 9:4997–5000.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Aharon A, Rebibo-Sabbah A, Tzoran I and
Levin C: Extracellular vesicles in hematological disorders. Rambam
Maimonides Med J. 5:e00322014. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Inal JM, Kosgodage U, Azam S, Stratton D,
Antwi-Baffour S and Lange S: Blood/plasma secretome and
microvesicles. Biochim Biophys Acta. 1834:2317–2325. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Piccin A, Murphy WG and Smith OP:
Circulating microparticles: Pathophysiology and clinical
implications. Blood Rev. 21:157–171. 2007. View Article : Google Scholar
|
|
18
|
Lynch SF and Ludlam CA: Plasma
microparticles and vascular disorders. Br J Haematol. 137:36–48.
2007.PubMed/NCBI
|
|
19
|
Jeppesen DK, Hvam ML, Primdahl-Bengtson B,
Boysen AT, Whitehead B, Dyrskjøt L, Orntoft TF, Howard KA and
Ostenfeld MS: Comparative analysis of discrete exosome fractions
obtained by differential centrifugation. J Extracell Vesicles.
3:250112014. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Théry C, Amigorena S, Raposo G and Clayton
A: Isolation and characterization of exosomes from cell culture
supernatants and biological fluids. Curr Protoc Cell Biol. Chapter
3: Unit 3. 22:2006. View Article : Google Scholar
|
|
21
|
Lötvall J, Hill AF, Hochberg F, Buzás EI,
Di Vizio D, Gardiner C, Gho YS, Kurochkin IV, Mathivanan S,
Quesenberry P, et al: Minimal experimental requirements for
definition of extracellular vesicles and their functions: A
position statement from the International Society for Extracellular
Vesicles. J Extracell Vesicles. 3:269132014. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Vickers KC, Palmisano BT, Shoucri BM,
Shamburek RD and Remaley AT: MicroRNAs are transported in plasma
and delivered to recipient cells by high-density lipoproteins. Nat
Cell Biol. 13:423–433. 2011. View
Article : Google Scholar : PubMed/NCBI
|
|
23
|
György B, Módos K, Pállinger E, Pálóczi K,
Pásztói M, Misják P, Deli MA, Sipos A, Szalai A, Voszka I, et al:
Detection and isolation of cell-derived microparticles are
compromised by protein complexes resulting from shared biophysical
parameters. Blood. 117:e39–e48. 2011. View Article : Google Scholar
|
|
24
|
Brinkmann V, Reichard U, Goosmann C,
Fauler B, Uhlemann Y, Weiss DS, Weinrauch Y and Zychlinsky A:
Neutrophil extracellular traps kill bacteria. Science.
303:1532–1535. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Williams JC and Mackman N: MPs or ICs?
Blood. 117:1101–1102. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Phillipson M and Kubes P: The neutrophil
in vascular inflammation. Nat Med. 17:1381–1390. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Gheldof D, Hardij J, Cecchet F, Chatelain
B, Dogné JM and Mullier F: Thrombin generation assay and
transmission electron microscopy: A useful combination to study
tissue factor-bearing microvesicles. J Extracell Vesicles. 2:22013.
View Article : Google Scholar
|
|
28
|
Lacroix R, Judicone C, Mooberry M,
Boucekine M and Key NS: Standardization of pre-analytical variables
in plasma microparticle determination: Results of the International
Society on Thrombosis and Haemostasis SSC Collaborative workshop. J
Thromb Haemost. Apr 2–2013.Epub ahead of print. View Article : Google Scholar
|
|
29
|
Yuana Y, Bertina RM and Osanto S:
Pre-analytical and analytical issues in the analysis of blood
microparticles. Thromb Haemost. 105:396–408. 2011. View Article : Google Scholar
|
|
30
|
Witwer KW, Buzás EI, Bemis LT, Bora A,
Lässer C, Lötvall J, Nolte-’t Hoen EN, Piper MG, Sivaraman S, Skog
J, et al: Standardization of sample collection, isolation and
analysis methods in extracellular vesicle research. Extracell
Vesicles. 2:203602013.
|
|
31
|
Ayers L, Kohler M, Harrison P, Sargent I,
Dragovic R, Schaap M, Nieuwland R, Brooks SA and Ferry B:
Measurement of circulating cell-derived microparticles by flow
cytometry: Sources of variability within the assay. Thromb Res.
127:370–377. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Jayachandran M, Miller VM, Heit JA and
Owen WG: Methodology for isolation, identification and
characterization of microvesicles in peripheral blood. J Immunol
Methods. 375:207–214. 2012. View Article : Google Scholar :
|
|
33
|
Chandler WL, Yeung W and Tait JF: A new
microparticle size calibration standard for use in measuring
smaller microparticles using a new flow cytometer. J Thromb
Haemost. 9:1216–1224. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Montoro-García S, Shantsila E, Tapp LD,
López-Cuenca A, Romero AI, Hernández-Romero D, Orenes-Piñero E,
Manzano-Fernández S, Valdés M, Marín F and Lip GY: Small-size
circulating microparticles in acute coronary syndromes: Relevance
to fibrinolytic status, reparative markers and outcomes.
Atherosclerosis. 227:313–322. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Sokolova V, Ludwig AK, Hornung S, Rotan O,
Horn PA, Epple M and Giebel B: Characterisation of exosomes derived
from human cells by nanoparticle tracking analysis and scanning
electron microscopy. Colloids Surf B Biointerfaces. 87:146–150.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Trummer A, De Rop C, Tiede A, Ganser A and
Eisert R: Recovery and composition of microparticles after
snap-freezing depends on thawing temperature. Blood Coagul
Fibrinolysis. 20:52–56. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Zhou H, Yuen PS, Pisitkun T, Gonzales PA,
Yasuda H, Dear JW, Gross P, Knepper MA and Star RA: Collection,
storage, preservation, and normalization of human urinary exosomes
for biomarker discovery. Kidney Int. 69:1471–1476. 2006.PubMed/NCBI
|
|
38
|
Raposo G, Nijman HW, Stoorvogel W,
Liejendekker R, Harding CV, Melief CJ and Geuze HJ: B lymphocytes
secrete antigen-presenting vesicles. J Exp Med. 183:1161–1172.
1996. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Théry C, Ostrowski M and Segura E:
Membrane vesicles as conveyors of immune responses. Nat Rev
Immunol. 9:581–593. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Baran J, Baj-Krzyworzeka M, Weglarczyk K,
Szatanek R and Zembala M, Barbasz J, Czupryna A, Szczepanik A and
Zembala M: Circulating tumour-derived microvesicles in plasma of
gastric cancer patients. Cancer Immunol Immunother. 59:841–850.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Abas L and Luschnig C: Maximum yields of
microsomal-type membranes from small amounts of plant material
without requiring ultracentrifugation. Anal Biochem. 401:217–227.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Lavoie C, Lanoix J, Kan FW and Paiement J:
Cell-free assembly of rough and smooth endoplasmic reticulum. J
Cell Sci. 109:1415–1425. 1996.PubMed/NCBI
|
|
43
|
Bard MP, Hegmans JP, Hemmes A, Luider TM,
Willemsen R, Severijnen LA, van Meerbeeck JP, Burgers SA,
Hoogsteden HC and Lambrecht BN: Proteomic analysis of exosomes
isolated from human malignant pleural effusions. Am J Respir Cell
Mol Biol. 31:114–121. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Keller S, Ridinger J, Rupp AK, Janssen JW
and Altevogt P: Body fluid derived exosomes as a novel template for
clinical diagnostics. J Transl Med. 9:862011. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Poliakov A, Spilman M, Dokland T, Amling
CL and Mobley JA: Structural heterogeneity and protein composition
of exosome-like vesicles (prostasomes) in human semen. Prostate.
69:159–167. 2009. View Article : Google Scholar
|
|
46
|
Dettenhofer M and Yu XF: Highly purified
human immunodeficiency virus type 1 reveals a virtual absence of
Vif in virions. J Virol. 73:1460–1467. 1999.PubMed/NCBI
|
|
47
|
Cantin R, Diou J, Bélanger D, Tremblay AM
and Gilbert C: Discrimination between exosomes and HIV-1:
Purification of both vesicles from cell-free supernatants. J
Immunol Methods. 338:21–30. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Yuana Y, Levels J, Grootemaat A, Sturk A
and Nieuwland R: Co-isolation of extracellular vesicles and
high-density lipoproteins using density gradient
ultracentrifugation. J Extracell Vesicles. 3:32014. View Article : Google Scholar
|
|
49
|
Fitzner D, Schnaars M, van Rossum D,
Krishnamoorthy G, Dibaj P, Bakhti M, Regen T, Hanisch UK and Simons
M: Selective transfer of exosomes from oligodendrocytes to
microglia by macropinocytosis. J Cell Sci. 124:447–458. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Müller G: Novel tools study cell
type-specific exosomes microvesicles. J Bioanal Biomed. 4:46–60.
2012.
|
|
51
|
Taylor DD, Lyons KS and Gerçel-Taylor C:
Shed membrane fragment-associated markers for endometrial and
ovarian cancers. Gynecol Oncol. 84:443–448. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Böing AN, van der Pol E, Grootemaat AE,
Coumans FA, Sturk A and Nieuwland R: Single-step isolation of
extracellular vesicles by size-exclusion chromatography. J
Extracell Vesicles. 3:32014. View Article : Google Scholar
|
|
53
|
Alvarez ML, Khosroheidari M, Kanchi Ravi R
and DiStefano JK: Comparison of protein, microRNA, and mRNA yields
using different methods of urinary exosome isolation for the
discovery of kidney disease biomarkers. Kidney Int. 82:1024–1032.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Taylor DD, Zacharias W and Gerçel-Taylor
C: Exosome isolation for proteomic analyses and RNA profiling.
Methods Mol Biol. 728:235–246. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Danesh A, Inglis HC, Jackman RP, Wu S,
Deng X, Muench MO, Heitman JW and Norris PJ: Exosomes from red
blood cell units bind to monocytes and induce proinflammatory
cytokines, boosting T-cell responses in vitro. Blood. 123:687–696.
2014. View Article : Google Scholar :
|
|
56
|
de Vrij J, Maas SL, Kwappenberg KM,
Schnoor R, Kleijn A, Dekker L, Luider TM, de Witte LD, Litjens M,
van Strien ME, et al: Glioblastoma-derived extracellular vesicles
modify the phenotype of monocytic cells. Int J Cancer. Mar
20–2015.Epub ahead of print. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Ismail N, Wang Y, Dakhlallah D, Moldovan
L, Agarwal K, Batte K, Shah P, Wisler J, Eubank TD, Tridandapani S,
et al: Macrophage microvesicles induce macrophage differentiation
and miR-223 transfer. Blood. 121:984–995. 2013. View Article : Google Scholar :
|
|
58
|
Sáenz-Cuesta M, Arbelaiz A, Oregi A,
Irizar H, Osorio-Querejeta I, Muñoz-Culla M, Banales JM,
Falcón-Pérez JM, Olascoaga J and Otaegui D: Methods for
extracellular vesicles isolation in a hospital setting. Front
Immunol. 6:502015. View Article : Google Scholar : PubMed/NCBI
|