Introduction
Vitamin D, which comprises a group of fat-soluble
secosterols found naturally in very few foods, is photosynthesized
in the skin of vertebrates by the action of solar ultraviolet-B
(UV-B) radiation (1). The major
biological function of vitamin D is to maintain normal serum
concentrations of calcium and phosphorus by enhancing the ability
of the small intestine to absorb these minerals from food (1). The original active form of vitamin D
is derived from cholesterol. Hepatic hydroxylation enzymes and
possibly other enzymes, convert cholesterol into the major
circulating metabolite 25-hydroxyvitamin D (25D), which is a
relatively long-lived metabolite, as well as a marker of the
vitamin D status. 25D is hydroxylated at 1α in the kidneys and
peripheral tissue to produce the hormonally active form of vitamin
D, 1,25-dihydroxyvitamin D3 (VD3) (2–4).
The majority of the pleiotropic, long-term effects of VD3 are
mediated by its binding to a nuclear high-affinity receptor
[vitamin D receptor (VDR)], which is principally located in the
nuclei of target cells, in complex with 9-cis-retinoic acid, a
retinoid X receptor (RXR) (5).
The activated VDR-RXR complex binds to a vitamin D response element
(VDRE) located in the promoters of target genes that are involved
in calcium absorption, such as transient receptor potential cation
channel, subfamily V (TRPV)5 and TRPV6 genes (6).
It has now become clear that vitamin D has
additional physiological functions beyond calcium absorption.
Previous research has provided evidence indicating that vitamin D
signaling regulates several physiological responses unrelated to
mineral ion homeostasis, including both innate and adaptive immune
responses (7). Moreover,
epidemiological data have established associations between a lack
of UV-B exposure, vitamin D defficiency and the prevalence of
certain types of cancer (8–10).
Vitamin D is also related to lipid metabolism and obesity. Indeed,
epidemiologically, obesity has been found to be associated with
lower serum levels of 25D and sun exposure (11,12).
According to previous data, VD3 is known to inhibit
mouse 3T3-L1 pre-adipocyte differentiation, which is mediated by
the induction of the expression of a number of genes, such as
CCAAT/enhancer-binding protein-α (C/EBP-α), peroxisome
proliferator-activated receptor-γ (PPAR-γ) and sterol regulatory
element-binding protein-1c (SREBP-1c), which are characteristic of
mature adipocytes. However, the effects of VD3 on mouse
pre-adipocytes are limited in blocking fat cell differentiation and
gene expression within the first 48 h of initiating differentiation
(13).
Lipogenesis and adipose tissue development are
regulated by female sex steroid hormones. The adipocytes of female
mice have increased lipogenic capacities compared with those of
male mice, although adipocytes the of females are smaller than
those of males (14). In
addition, the female sex hormone, estrogen, regulates glucose
homeostasis, which is a crucial factor in fatty acid metabolism
(14,15).
During gestation, women have a special endocrine
environment which helps to maintain pregnancy, and thereby lipid
metabolism is regulated differently from that of a non-pregnant
women. Since more nutrients are required, the endocrine system
tends to ncrease fat accumulation in the pregnant woman (16). The proliferation of adipose tissue
is often accompanied by a state of insulin resistance during
pregnancy. This pregnancy-specific adaptive response allows glucose
to be transferred across the placenta more efficiently and thus
fetal growth is promoted (17).
However, this response may also result in the mother and fetus
developing elevated blood glucose and insulin levels, which in turn
means a greater risk of obesity. Certain studies have indicated
that birth weight is directly associated with the BMI of the baby,
which implies that gestational diabetes in the mother contributes
to obesity in her child (18–20). Furthermore, maternal obesity
increases the risk of other pregnancy complications, including
pre-eclampsia, gestational diabetes, hypertension, thrombosis and
labor issues (21–23).
Certain studies have suggested that the serum
concentration of vitamin D differs between pregnant and
non-pregnant women, indicating that vitamin D may play a specific
role during pregnancy (24,25). However, correlations between
lipogenesis and vitamin D during pregnancy have not yet been
established. Therefore, in the present study, we examined the
effects of VD3 on lipogenesis and fat accumulation in the liver and
adipose tissue of pregnant rats.
Materials and methods
Reagents and chemicals
VD3 was purchased from Merck Millipore Co.
(Billerica, MA, USA). Corn oil and mouse anti-β actin antibody were
purchased from Sigma-Aldrich (St. Louis, MO, USA). Rabbit anti-FAS
(sc-20140), mouse anti-SREBP-1c (sc-133551) and mouse anti-PPAR-γ
(sc-7273) antibodies, and horseradish peroxidase (HRP)-conjugated
anti-rabbit (sc-2313) and anti-mouse (sc-2005) IgG were all
purchased from Santa Cruz Biotechnology, Inc. (Santa Cruz, CA,
USA).
Animals and treatments
Pregnant Sprague-Dawley (SD; n=12) female rats were
purchased from Samtako (Osan, Korea) and handled at the Pusan
National University Laboratory Animal Resources Center (Busan,
Korea), which is accredited by the Korea FDA according to the
National Institutes of Health guidelines. The rats were housed in
cages under a 12-h light/dark cycle, with a constant temperature of
23±1°C. The rats were fed a diet of soy-free pellets (Samtako). All
experimental animal procedures were approved by the Ethics
Committee of Pusan National University (Busan, Korea; approval no.
PNU-2013-0401). From gestational days (GD) 11–16, the rats were
administered daily subcutaneous injections of VD3 (1
μg/kg/day) or corn oil as the vehicle control. The dosage
was adjusted according to changes in body weight (BW). All the
pregnant female rats were sacrificed on GD 17 using CO2
gas. Tissue samples (liver and abdominal adipose) and fetus were
removed using scissors from each individual pregnant rat and stored
at −70°C or fixed in 10% formalin until analysis.
Reverse transcription-quantitative PCR
(RT-qPCR)
Total RNA was extracted using TRIzol reagent
(Invitrogen, Carlsbad, CA, USA) according to the manufacturer's
instructions. The concentration of total RNA was measured using a
spectrophotometer (Biospec-nano; Shimadzu, Kyoto, Japan).
First-strand complementary DNA (cDNA) was prepared from total RNA
(3 μg) by reverse transcription using Moloney murine
leukemia virus (M-MLV) reverse transcriptase (Invitrogen) and
random primers (9-mers; Takara Bio Inc., Shiga, Japan).
Quantitative PCR (qPCR) was performed using a cDNA template (2
μl) and 2X Power SYBR-Green (6 μl; Toyobo Co., Ltd.,
Osaka, Japan) containing specific primers. The primer sequences for
fatty acid synthase (FAS), stearoyl-CoA desaturase 1 (SCD1),
acetyl-CoA carboxylase 1 (ACC1), SREBP-1c, PPAR-γ, C/EBP-α and
insulin-induced gene 2 (INSIG2) are listed in Table I. qPCR was carried out for 40
cycles using the following parameters: denaturation at 95°C for 15
sec, followed by annealing and extension at 70°C for 60 sec. The
fluorescence intensity was measured at the end of the extension
phase of each cycle. The threshold value for the fluorescence
intensity of all samples was set manually. The reaction cycle at
which the PCR products exceeded this fluorescence intensity
threshold during the exponential phase of PCR amplification was
considered to be the threshold cycle (CT). The expression of the
target gene was quantified relative to that of β-actin, an
ubiquitous housekeeping gene, based on a comparison of CTs at a
constant fluorescence intensity.
 | Table IPrimer sequences used for
RT-qPCR. |
Table I
Primer sequences used for
RT-qPCR.
| Gene name | Primer sequence
(5′→3′) | Fragment (bp) |
|---|
| FAS | F:
CCAGGGTTTGGAATTATTTC
R: GAAGATAAACCCTAAGGCTC | 293 |
| SCD1 | F:
AAGAGCATTTTTCAACCCTA
R: GTCTCAGAATTTGCTTTATT | 314 |
| ACC1 | F:
CTTGGCCTACTGGCTCTGAC
R: GGGCAGGTAGTTCTCCTCCT | 216 |
| SREBP-1c | F:
TTCTTCGTGCAGATGTGGAG
R: TAACGAGCAGAGCAGCAGAA | 234 |
| PPAR-γ | F:
TGAAAAGCGGTGTGAGACTG
R: TGATGGGTCCAAAATTCCAT | 220 |
| C/EBP-α | F:
ACCTGTGAGACGACGCTCTT
R: GCCGGTGACTCAGAGATAGC | 160 |
| INSIG2 | F:
CCTTGGTGTTGGAATTGCTT
R: TTGCTAGCTGACGACCAATG | 167 |
Western blot analysis
Protein samples were extracted from the liver and
adipose tissue using PRO-PREP solution (Intron Biotechnology,
Seoul, Korea) following the manufacturer's instructions. A total of
40 μg cytosolic proteins was separated by 8–10% sodium
dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE) and
transferred onto nitrocellulose membranes (Daeil Lab Service Co.,
Ltd., Seoul, Korea). The membranes were then blocked for 1 h with
5% skim milk (Difco, Sparks, MD, USA) in phosphate-buffered saline
(PBS) with 0.05% Tween-20 (PBS-T). After blocking, the membranes
were incubated o/n with antibodies specific for FAS (dilution
1:500), SREBP-1c (dilution 1:500) and PPAR-γ (dilution 1:500), as
well as with HRP-conjugated anti-rabbit and anti-mouse secondary
antibodies (dilution 1:2,000) in 5% skim milk with PBS-T for 1 h.
Luminol reagent (Bio-Rad Laboratories Inc., Hercules, CA, USA) was
used to visualize antibody binding. Each blot was then stripped by
incubation with 2% SDS and 100 mM 2-mercapto-ethanol in 62.5 mM
Tris-HCl (pH 6.8) for 30 min at 50–60°C. The membranes were
subsequently probed with an antibody against β-actin (diluted
1:2,000; Santa Cruz Biotechnology, Inc.) as an internal control.
The blots were scanned using Gel Doc 1000, version 1.5 (Bio-Rad
Laboratories Inc.) and the band intensities were normalized to
β-actin levels.
Histological analysis
Adipose tissue was removed from the rats using
scissors, immediately fixed with 10% formalin, embedded in paraffin
wax, routinely processed, and then sectioned into 4-μm-thick
sections. The tissue sections were then stained with hematoxylin
and eosin (H&E; Sigma-Aldrich). Five different areas from each
animal were analyzed to determine the histological changes. Images
of the tissues were captured at ×20 magnification using an optical
microscope (model BX50F-3; Olympus, Tokyo, Japan).
Statistical analyses
The results are presented as the means ± standard
deviation (SD). Data were analyzed using one-way analysis of
variance (ANOVA) (SPSS for Windows, 10.10, standard version; SPSS
Inc., Chicago, IL, USA). P-values <0.05 were considered to
indicate statistically significant differences.
Results
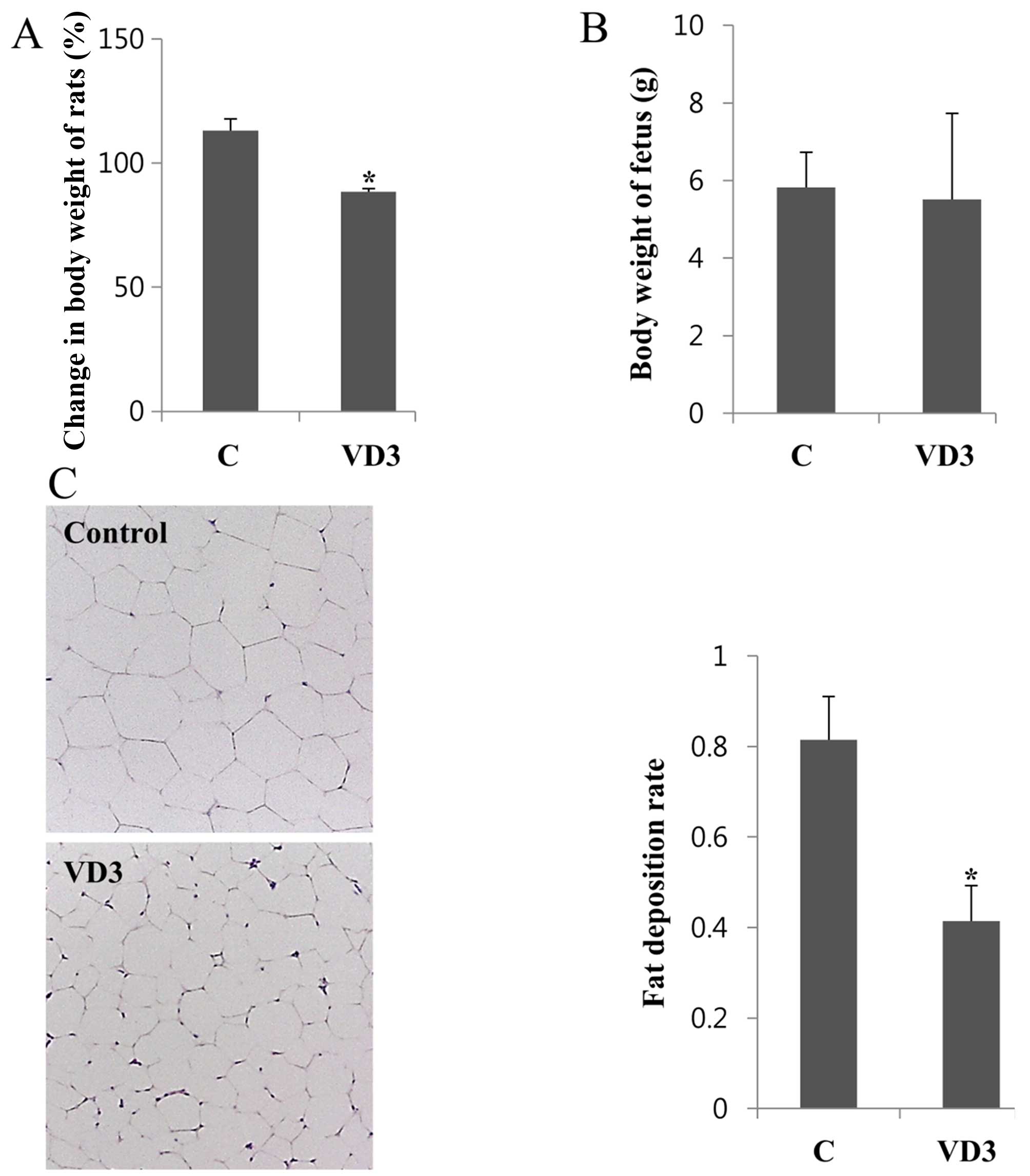
Changes in body weight in the pregnant
rats
To determine the effects of VD3 on fat accumulation
during pregnancy, the rats were subcutaneously injected with 1
μg/kg/day of VD3, from GD11 to GD16, and were then
sacrificed on GD17. The body weights of the rats were measured on
GD17 and normalized to the values obtained on GD11; the changes in
body weight are shown in Fig. 1A.
Corn oil was used as a negative control. We observed that VD3
significantly decreased body weight by up to 25% compared with the
controls. To examine the possible adverse effects of VD3 on the
fetus, fetal body weight was also measured. The body weight of the
fetuses was not altered significantly (Fig. 1B).
Histological changes in adipose tissues
following treatment with VD3
As VD3 was shown to reduce body weight, we then
analyzed the histological changes in adipose tissue following
treatment with VD3 in order to determine whether the decrease in
body weight was due to the loss of fatty tissue. The adipose tissue
was fixed in paraffin and stained with H&E. Images of the
stained adipocytes were then captured using an optical microscope,
and the number of cells in the diagram was counted. The fat
deposition rate was calculated based on the number of adipocytes.
Five separate areas from each animal were analyzed, and the results
are shown in Fig. 1C. The fat
deposition rate in adipose tissue was decreased following treatment
with VD3 compared with the vehicle-treated controls (Fig. 1C). We also noted that the size of
the adipocytes was smaller in the VD-treated tissues than those
treated with the vehicle control. These results suggest that VD3
treatment reduces fat deposition, resulting in a decrease in total
body weight of the pregnant rats.
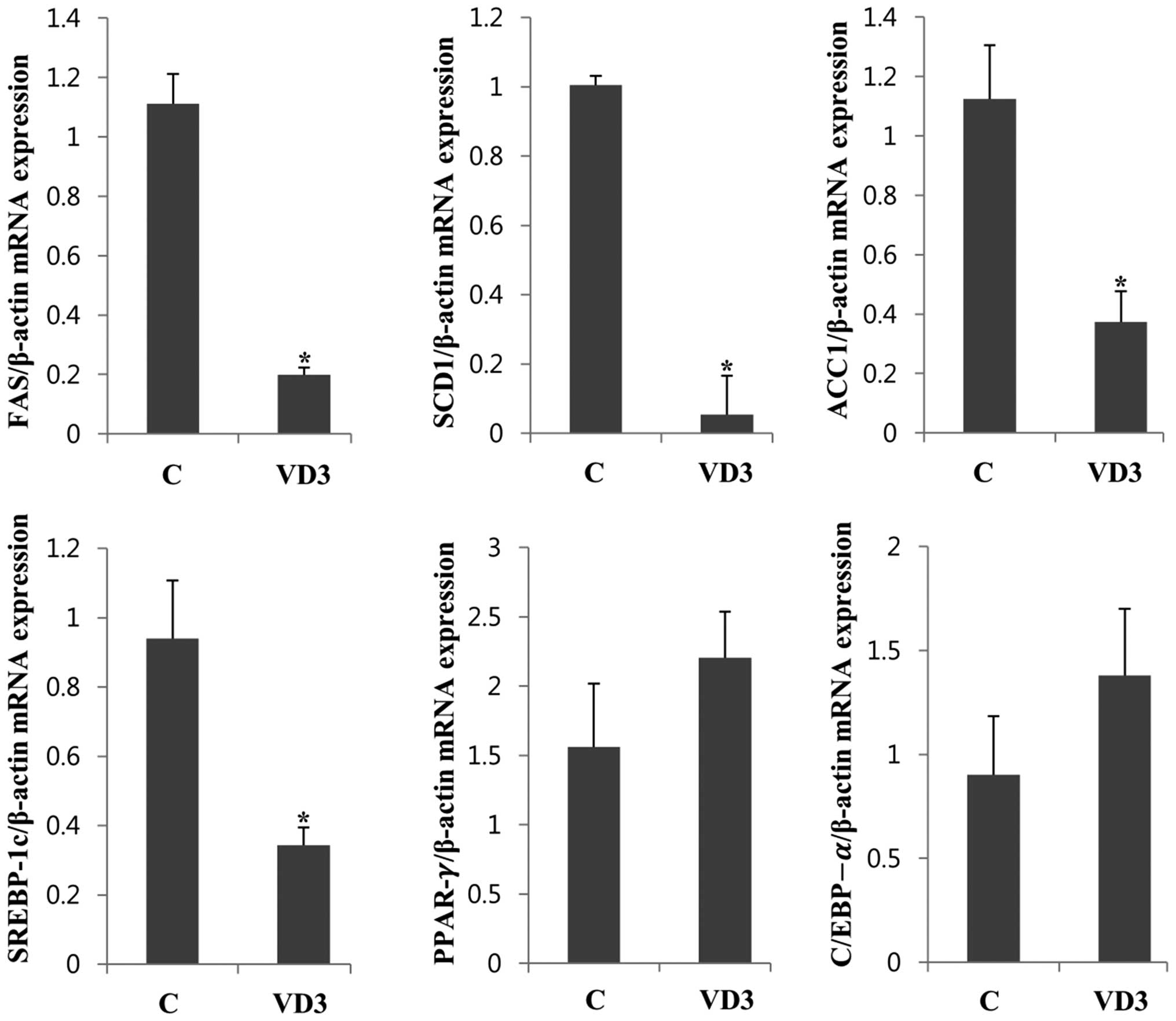
Regulation of lipogenesis-associated gene
expression in the liver
To determine the mechanisms through which VD3
regulates fat deposition and body weight, the expression levels of
genes associated with lipogenesis, so-called lipogenic enzymes,
were measured in the liver. The liver is an organ that
biosynthesizes and catalyzes triacylglycerol (TG), which is the
typical form of animal fat (26).
Therefore, the amount of lipogenic enzymes in the liver is
important for fat synthesis. Constitutive lipogenic enzymes in the
liver include FAS, SCD1 and ACC1 (27). We found that VD3 significantly
reduced the mRNA expression of FAS in the liver by up to 80%
(Fig. 2). The levels of the other
lipogenic enzymes, SCD1 and ACC1, were also markedly decreased
following treatment with VD3. It is known that lipogenic enzymes
are controlled at the transcriptional level and that proadipogenic
transcription factors, such as SREBP-1c, PPAR-γ and C/EBP-α are
involved in the regulation of adipocyte formation (28). Therefore, in this study, we
measured the mRNA expression of SREBP-1c, PPAR-γ and C/EBP-α in the
liver (Fig. 2). The mRNA
expression levels of SREBP-1c were significantly decreased
following treatment with VD3, and a similar result was noted in
relation to the expression levels of its target genes, FAS, SCD1
and ACC1. However, the mRNA levels of PPAR-γ and C/EBP-α were
slightly upregulated following treatment with VD3. The transcripts
of PPAR-γ and C/EBP were slightly elevated following treatment with
VD3 without significance. These results suggest that the expression
levels of lipogenic enzymes in the liver is reduced by VD3
treatment in pregnant rats, and this is possibly mediated by
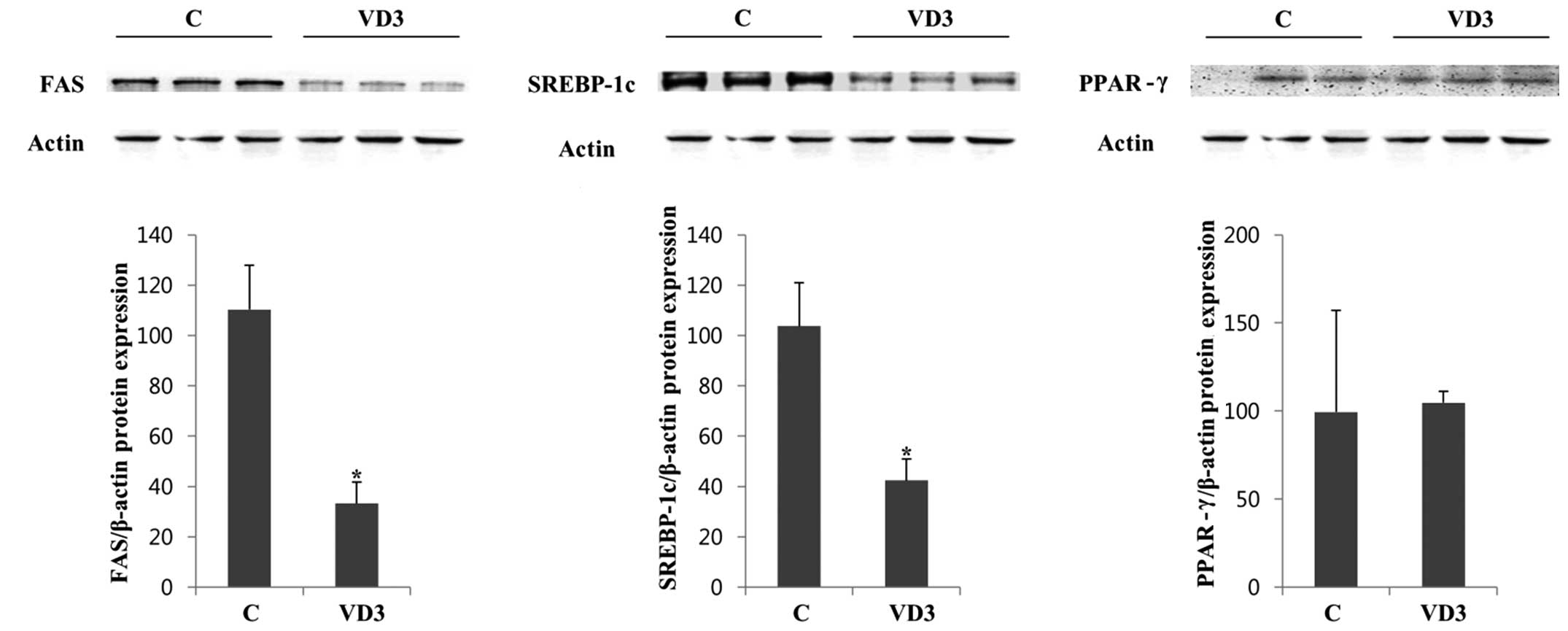
SREBP-1c. To confirm the regulation of these genes at the
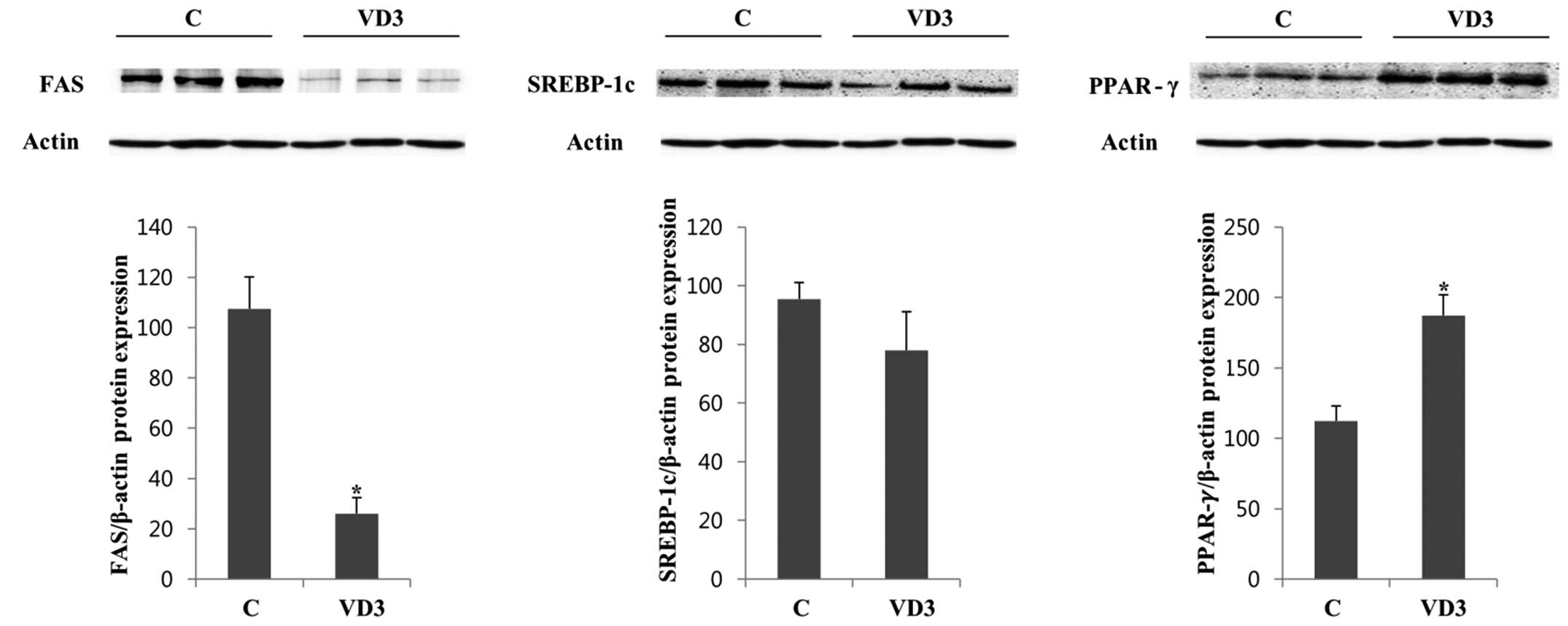
translational level, we examined the protein expression levels of
FAS, SREBP-1c and PPAR-γ by western blot analysis in the absence
(vehicle control) and presence of VD3 (Fig. 3). We selected 3 independent
protein samples from the control and VD3 groups for western blot
analysis, and the results are represented in (Fig. 3). The protein expression of FAS
and SREBP-1c in the liver was significantly decreased following
treatment with VD3 compared with the control. These results were in
agreement with the mRNA results (Fig.
2), indicating that VD3 reduced the expression of FAS and
SREBP-1c at both the transcriptional and translational level. The
basal expression of PPAR-γ was relatively low and was not altered
significantly following treatment with VD3 (Fig. 3).
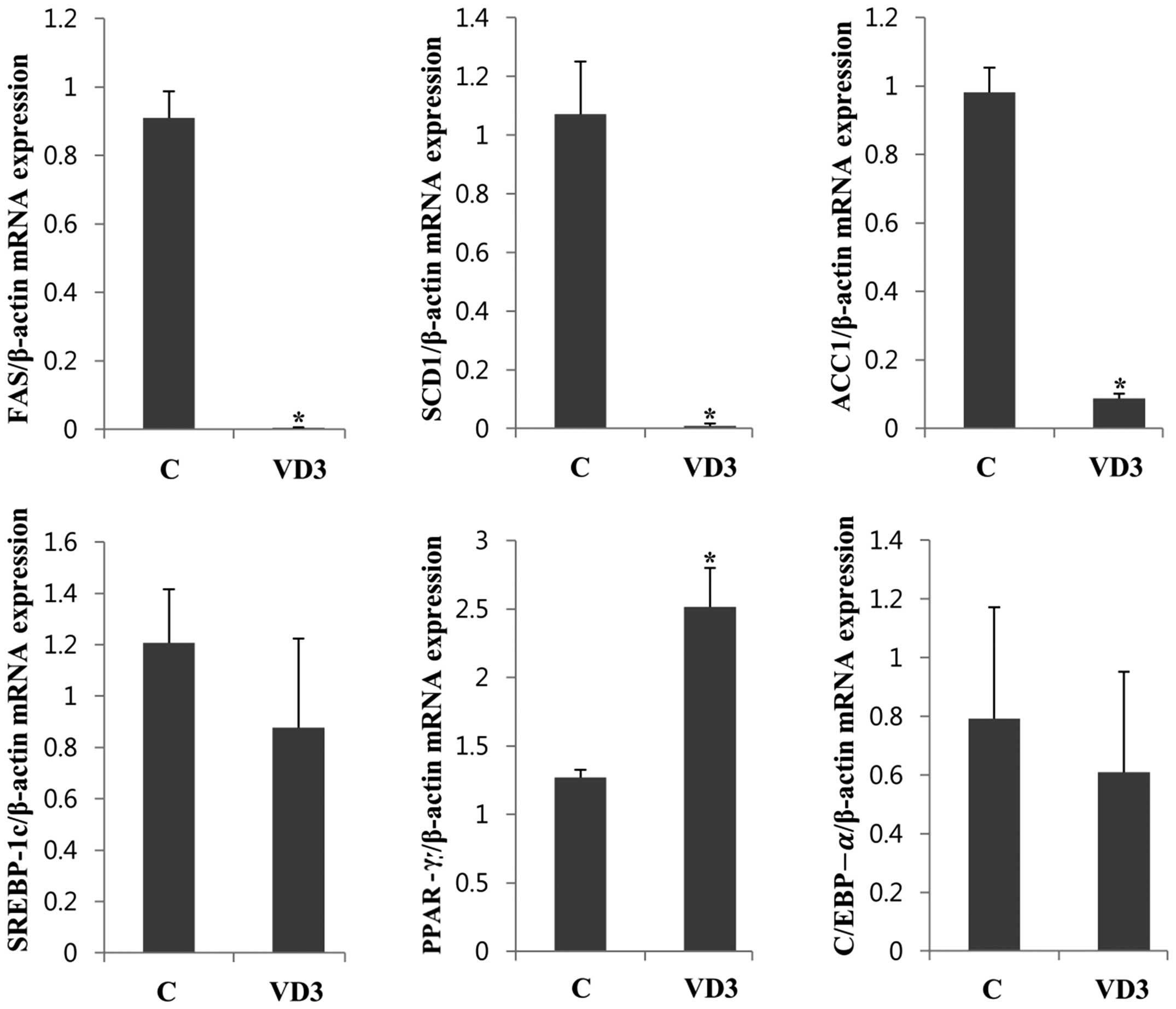
Regulation of lipogenesis-associated gene
epxression in adipose tissue
We then examined the effects of VD3 on the
expression of lipogenic enzymes, as well as their transcriptional
regulators in adipose tissue, where fat is mainly stored (Fig. 4). Treatment with VD3 markedly
downregulated the lipogenic enzymes, FAS, SCD1 and ACC1, in adipose
tissue, decreasing their expresion by up to 90% (Fig. 4). These results are similar to
those involving the liver (Fig.
2), suggesting that VD3 modulates the expression of lipogenic
enzymes in both the liver and adipose tissue. The transcriptional
levels of PPAR-γ were significantly upregulated following treatment
with VD3, which was slightly different to the results obtained with
the liver (the mRNA levels of PPAR-γ were only slightly increased
in the liver). The regulation of SREBP-1c expression in adipose
tissue also differed from that in the liver. The mRNA expression of
SREBP-1c in adipose tissue was not significantly altered following
treatment with VD3, whereas it was greatly reduced in the liver.
The protein levels of FAS, SREBP-1c and PPAR-γ were regulated in a
similar manner to the mRNA levels (Fig. 5). Treatment with VD3 markedly
downregulated the protein expression of FAS, whereas, it did not
significantly alter that of SREBP-1c. PPAR-γ protein expression
increased following treatment with VD3, which was similar to the
results of mRNA expression.
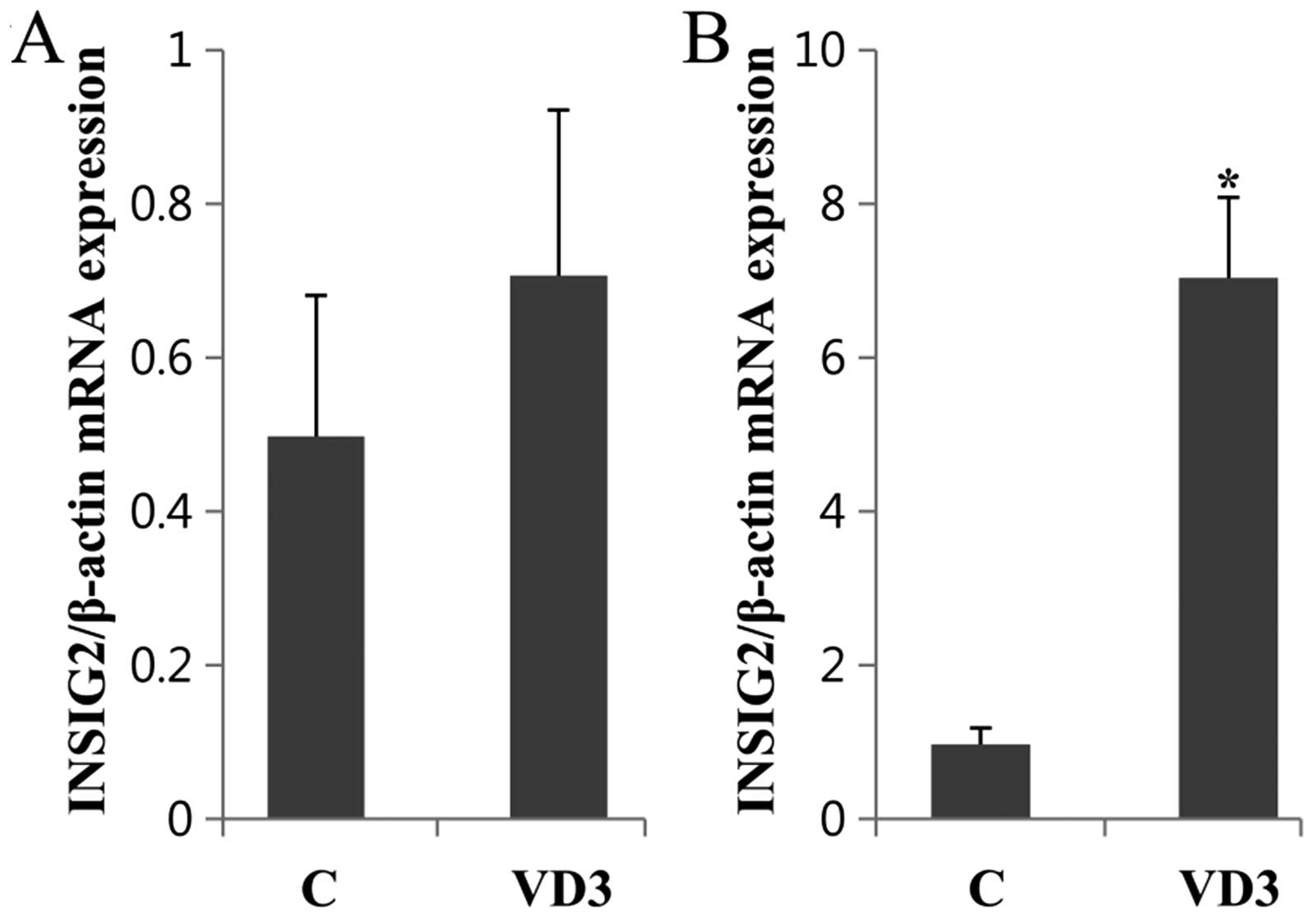
Tissue-specific regulation of INSIG2 by
VD3 in the liver and adipose tissue of pregnant rats
Since PPAR-γ and SREBP-1c were differentially
regulated by VD3, the expression of INSIG2, an intermediate
regulator between PPAR-γ and SREBP-1c, was investigated in the
liver and adipose tissue (Fig.
6). VD3 modulated the expression of INSIG2 in a tissue-specific
manner. In the adipose tissue, VD3 markedly enhanced the INSIG2
mRNA levels, whereas in the liver, these levels were not
significantly altered.
Discussion
The main natural source of vitamin D is synthesis
from cholesterol in the skin; the dermal synthesis of vitamin D
from cholesterol is dependent on exposure to sunlight. Full-body
exposure during the summer months for 10–15 min will provide 10,000
to 20,000 IU of VD3 within 24 h, for an adult (10). However, the amount of UV exposure
required for the synthesis of VD3 also depends on factors other
than just the amount of time spent in the sun (29). Vitamin D deficiency is defined as
a concentration of 25(OH)D <30 nmol/l. The Indoor Air Quality
Act of 1989 reported that Americans spent 93% of their time indoors
on average, thus contributing to a higher prevalence of lower
25(OH)D concentrations in the blood of adults. Vitamin D deficiency
among African American children has also been reported (30).
It is known that VD3 inhibits lipogenesis in 3T3-L1
pre-adipocytes (31,32). However, the majority of previous
studies were performed in vitro using mouse or human
pre-adipocytes, and only a few animal studies were in vivo.
Yin et al demonstrated that VD3 modulates lipid metabolism
by attenuating hepatic steatosis in the livers of male adult rats
(33). The protective effect of
VD3 against hepatic steatosis was mediated by downregulating
SREBP-1c and its target genes, ACC and FAS (33). However, they did not examine the
effect of VD3 in adipose tissue. In the present study, we, for the
first time and to the best of our knowledge, examined the effects
and role of VD3 in the adipose tissue and liver of pregnant
animals. VD3 significantly reduced the fat deposition rate in
adipose tissue and subsequently reduced the body weight of rats. As
fat deposition and the size of adipocytes were reduced, we propose
that VD3 administration alters lipogenesis.
In the present study, we found that the expression
levels of the lipogenic proteins, FAS, SCD1 and ACC1, were
downregulated by VD3 in both the liver and adipose tissue of
pregnant rats (Fig. 3 and
5). These results suggest that
the reduction of body weight and fat deposition following treatment
with VD3 may be attributed to modulation of genes associated with
lipogenesis. These lipogenic proteins are known to be regulated by
specific transcription factors. For instance, the estrogen-mediated
reduction of ACC1 and FAS gene expression is caused by the
downregulation of SREBP-1c, which is a transcription factor that
binds to the specific promoter sequence sterol regulatory element-1
(SRE1) (34). In addition,
SREBP-1c regulates genes required for glucose metabolism, as well
as fatty acid and lipid synthesis (35). PPAR-γ and C/EBP-α are also
transcription factors related to lipogenesis (36).
As SREBP-1c, PPAR-γ and C/EBP-α have been reported
to regulate the FAS, SCD1 and ACC1 genes (37,38), we examined the expression of these
transcription factors following treatment with VD3. The lipogenic
transcription factors were not as strongly regulated by VD3 as the
lipogenic enzymes. SREBP-1c was significantly downregulated by VD3
in the liver, but not significantly in the adipose tissue, whereas
C/EBP-α expression was not significantly altered by VD3 in the
liver or adipose tissue. Of note, PPAR-γ was significantly
upregulated by VD3 in adipose tissue, while its expression was not
significantly increased in the liver. These results differ from
those of previous studies on pre-adipocytes, which showed that VD3
markedly regulated SREBP-1c, C/EBP-α and PPAR-γ expression
(13–31,39). It is possible that the mechanism
of the VD3-mediated inhibition of lipogenesis is controlled in a
different manner during pregnancy, as a result of the unique
endocrine environment. According to a previous study, PPAR-γ
suppresses TG synthesis in rat hepatoma cells by reducing SREBP-1
activity through the upregulation of INSIG2 (40). INSIG2 is an endoplasmic reticulum
protein that blocks the activation of SREBPs by binding to SREBP
cleavage-activating protein (SCAP). Thus, in this study, we
examined the expression levels of INSIG2 in the liver and adipose
tissue following treatment with VD3. The INSIG2 mRNA levels were
significantly increased following treatment with VD3 only in the
adipose tissue. Taken together, our results suggest that VD3
reduces TG synthesis through the suppression of the expression of
lipogenic enzymes in the liver and adipose tissue. However, the
expression of the lipogenic transcription factors, SREBP-1c and
PPAR-γ, was regulated by VD3 in a tissue-specific manner, and
INSIG2 may be involved in this process.
Maternal obesity adversely affects the outcome of
pregnancy, mainly as it increases the risk of hypertensive diseases
(chronic hypertension and pre-eclampsia), gestational diabetes, the
need for cesarean sections and the risk of infection (41). Furthermore, maternal obesity is
related to a subsequent increased risk of childhood obesity and
associated morbidity (41).
Previous research has demonstrated the benefi-cial effects of
exercise during pregnancy, including a reduced risk of gestational
diabetes, pre-eclampsia, and a higher rate of operative vaginal
deliveries (42). In addition,
the number of obese women of reproductive age undergoing bariatric
surgery is increasing. It has been reported that pregnancies
following bariatric surgery are less likely to be complicated by
gestational diabetes mellitus, hypertension, pre-eclampsia and
macrosomia compared to obese women who have not undergone this
surgery (43). However, physical
activity during pregnancy is limited, and bariatric surgery is
related to complications such as gastrointestinal obstruction and
hemorrhage. Therefore, safe and easy methods to reduce maternal
obesity during pregnancy are urgently required.
Taken together, the results of the present study
demonstrated that VD3 downregulated lipogenesis-associated gene
expression, which triggered a reduction in fat synthesis in the
liver, suppressed the deposition of adipose tissue, and finally
reduced the body weight of pregnant rats. These results suggest
that the supplementation of VD3 or exposure to sunlight helps to
control weight gain during pregnancy, and thereby prevents
obesity-associated pregnancy complications.
Acknowledgments
This study was supported by the 2012 Specialization
Project Research Grant, funded by the Pusan National University and
Pusan National University Research Grant, 2013.
References
|
1
|
Holick MF: McCollum Award Lecture, 1994:
Vitamin D - new horizons for the 21st century. Am J Clin Nutr.
60:619–630. 1994.PubMed/NCBI
|
|
2
|
Loomis WF: Skin-pigment regulation of
vitamin-D biosynthesis in man. Science. 157:501–506. 1967.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Bouillon R, Okamura WH and Norman AW:
Structure-function relationships in the vitamin D endocrine system.
Endocr Rev. 16:200–257. 1995.PubMed/NCBI
|
|
4
|
Dusso AS, Thadhani R and Slatopolsky E:
Vitamin D receptor and analogs. Semin Nephrol. 24:10–16. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Carlberg C, Bendik I, Wyss A, Meier E,
Sturzenbecker LJ, Grippo JF and Hunziker W: Two nuclear signalling
pathways for vitamin D. Nature. 361:657–660. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Pike JW, Meyer MB and Martowicz ML: New
techniques in transcription research extend our understanding of
the molecular actions of the vitamin D hormone. IBMS BoneKEy.
6:169–180. 2009. View
Article : Google Scholar
|
|
7
|
White JH: Vitamin D signaling, infectious
diseases, and regulation of innate immunity. Infect Immun.
76:3837–3843. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Gorham ED, Garland CF, Garland FC, Grant
WB, Mohr SB, Lipkin M, Newmark HL, Giovannucci E, Wei M and Holick
MF: Vitamin D and prevention of colorectal cancer. J Steroid
Biochem Mol Biol. 97:179–194. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Giovannucci E, Liu Y, Rimm EB, Hollis BW,
Fuchs CS, Stampfer MJ and Willett WC: Prospective study of
predictors of vitamin D status and cancer incidence and mortality
in men. J Natl Cancer Inst. 98:451–459. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Holick MF: Vitamin D deficiency. N Engl J
Med. 357:266–281. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Wortsman J, Matsuoka LY, Chen TC, Lu Z and
Holick MF: Decreased bioavailability of vitamin D in obesity. Am J
Clin Nutr. 72:690–693. 2000.PubMed/NCBI
|
|
12
|
Parikh SJ, Edelman M, Uwaifo GI, Freedman
RJ, Semega-Janneh M, Reynolds J and Yanovski JA: The relationship
between obesity and serum 1,25-dihydroxy vitamin D concentrations
in healthy adults. J Clin Endocrinol Metab. 89:1196–1199. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Natarajan R: Vitamin D metabolites inhibit
adipocyte differentiation in 3T3-L1 preadipocytes. Master's thesis.
University of Massachusetts; pp. 1642008
|
|
14
|
Macotela Y, Boucher J, Tran TT and Kahn
CR: Sex and depot differences in adipocyte insulin sensitivity and
glucose metabolism. Diabetes. 58:803–812. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Kuhl J, Hilding A, Östenson CG, Grill V,
Efendic S and Båvenholm P: Characterisation of subjects with early
abnormalities of glucose tolerance in the Stockholm Diabetes
Prevention Programme: the impact of sex and type 2 diabetes
heredity. Diabetologia. 48:35–40. 2005. View Article : Google Scholar
|
|
16
|
Catalano PM, Roman-Drago NM, Amini SB and
Sims EA: Longitudinal changes in body composition and energy
balance in lean women with normal and abnormal glucose tolerance
during pregnancy. Am J Obstet Gynecol. 179:156–165. 1998.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Rasmussen KM and Yaktine AL: Weight gain
during pregnancy: Reexamining the guidelines. National Academies
Press; 2009
|
|
18
|
Gillman MW, Rifas-Shiman S, Berkey CS,
Field AE and Colditz GA: Maternal gestational diabetes, birth
weight, and adolescent obesity. Pediatrics. 111:e221–e226. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Parsons TJ, Power C, Logan S and
Summerbell CD: Childhood predictors of adult obesity: a systematic
review. Int J Obes Relat Metab Disord. 23(Suppl 8): S1–S107.
1999.
|
|
20
|
Rogers I; EURO-BLCS Study Group: The
influence of birthweight and intrauterine environment on adiposity
and fat distribution in later life. Int J Obes Relat Metab Disord.
27:755–777. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Chu SY, Callaghan WM, Kim SY, Schmid CH,
Lau J, England LJ and Dietz PM: Maternal obesity and risk of
gestational diabetes mellitus. Diabetes Care. 30:2070–2076. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
O'Brien TE, Ray JG and Chan WS: Maternal
body mass index and the risk of preeclampsia: a systematic
overview. Epidemiology. 14:368–374. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Chu SY, Kim SY, Schmid CH, Dietz PM,
Callaghan WM, Lau J and Curtis KM: Maternal obesity and risk of
cesarean delivery: a meta-analysis. Obes Rev. 8:385–394. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Haddow JE, Neveux LM, Palomaki GE,
Lambert-Messerlian G, Canick JA, Grenache DG and Lu J: The
relationship between PTH and 25-hydroxy vitamin D early in
pregnancy. Clin Endocrinol (Oxf). 75:309–314. 2011. View Article : Google Scholar
|
|
25
|
Ginde AA, Sullivan AF, Mansbach JM and
Camargo CA Jr: Vitamin D insufficiency in pregnant and nonpregnant
women of childbearing age in the United States. Am J Obstet
Gynecol. 202:436.e1–8. 2010. View Article : Google Scholar
|
|
26
|
Haagsman H, De Haas C, Geelen M and Van
Golde L: Regulation of triacylglycerol synthesis in the liver.
Modulation of diacylglycerol acyltransferase activity in vitro. J
Biol Chem. 257:10593–10598. 1982.PubMed/NCBI
|
|
27
|
Lenhard JM: Lipogenic enzymes as
therapeutic targets for obesity and diabetes. Curr Pharm Des.
17:325–331. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Farmer SR: Transcriptional control of
adipocyte formation. Cell Metab. 4:263–273. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Wagner CL and Greer FR; American Academy
of Pediatrics Section on Breastfeeding; American Academy of
Pediatrics Committee on Nutrition: Prevention of rickets and
vitamin D deficiency in infants, children, and adolescents.
Pediatrics. 122:1142–1152. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Nesby-O'Dell S, Scanlon KS, Cogswell ME,
Gillespie C, Hollis BW, Looker AC, Allen C, Doughertly C, Gunter EW
and Bowman BA: Hypovitaminosis D prevalence and determinants among
African American and white women of reproductive age: third
National Health and Nutrition Examination Survey, 1988–1994. Am J
Clin Nutr. 76:187–192. 2002.PubMed/NCBI
|
|
31
|
Blumberg JM, Tzameli I, Astapova I, Lam
FS, Flier JS and Hollenberg AN: Complex role of the vitamin D
receptor and its ligand in adipogenesis in 3T3-L1 cells. J Biol
Chem. 281:11205–11213. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Lee S, Lee D-K, Choi E and Lee JW:
Identification of a functional vitamin D response element in the
murine Insig-2 promoter and its potential role in the
differentiation of 3T3-L1 preadipocytes. Mol Endocrinol.
19:399–408. 2005. View Article : Google Scholar
|
|
33
|
Yin Y, Yu Z, Xia M, Luo X, Lu X and Ling
W: Vitamin D attenuates high fat diet-induced hepatic steatosis in
rats by modulating lipid metabolism. Eur J Clin Invest.
42:1189–1196. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
D'Eon TM, Souza SC, Aronovitz M, Obin MS,
Fried SK and Greenberg AS: Estrogen regulation of adiposity and
fuel partitioning. Evidence of genomic and non-genomic regulation
of lipogenic and oxidative pathways. J Biol Chem. 280:35983–35991.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Ferré P and Foufelle F: Hepatic steatosis:
a role for de novo lipogenesis and the transcription factor
SREBP-1c. Diabetes Obes Metab. 12(Suppl 2): 83–92. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Brun RP, Kim JB, Hu E and Spiegelman BM:
Peroxisome proliferator-activated receptor gamma and the control of
adipogenesis. Curr Opin Lipidol. 8:212–218. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Horton JD, Shimomura I, Brown MS, Hammer
RE, Goldstein JL and Shimano H: Activation of cholesterol synthesis
in preference to fatty acid synthesis in liver and adipose tissue
of transgenic mice overproducing sterol regulatory element-binding
protein-2. J Clin Invest. 101:2331–2339. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Shimano H, Horton JD, Shimomura I, Hammer
RE, Brown MS and Goldstein JL: Isoform 1c of sterol regulatory
element binding protein is less active than isoform 1a in livers of
transgenic mice and in cultured cells. J Clin Invest. 99:846–854.
1997. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Kong J and Li YC: Molecular mechanism of
1,25-dihydroxyvitamin D3 inhibition of adipogenesis in 3T3-L1
cells. Am J Physiol Endocrinol Metab. 290:E916–E924. 2006.
View Article : Google Scholar
|
|
40
|
König B, Koch A, Spielmann J, Hilgenfeld
C, Hirche F, Stangl GI and Eder K: Activation of PPARalpha and
PPARgamma reduces triacylglycerol synthesis in rat hepatoma cells
by reduction of nuclear SREBP-1. Eur J Pharmacol. 605:23–30. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Castro LC and Avina RL: Maternal obesity
and pregnancy outcomes. Curr Opin Obstet Gynecol. 14:601–606. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Sui Z and Dodd JM: Exercise in obese
pregnant women: positive impacts and current perceptions. Int J
Womens Health. 5:389–398. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Sharara HA, Rhaman LNA, Ummunnisa F,
Aboubaker N, Abdullah MM and Shaikh N: Obese nulliparous women and
the risk for maternal and fetal complications. Open J Obstet
Gynecol. 4:239–242. 2014. View Article : Google Scholar
|