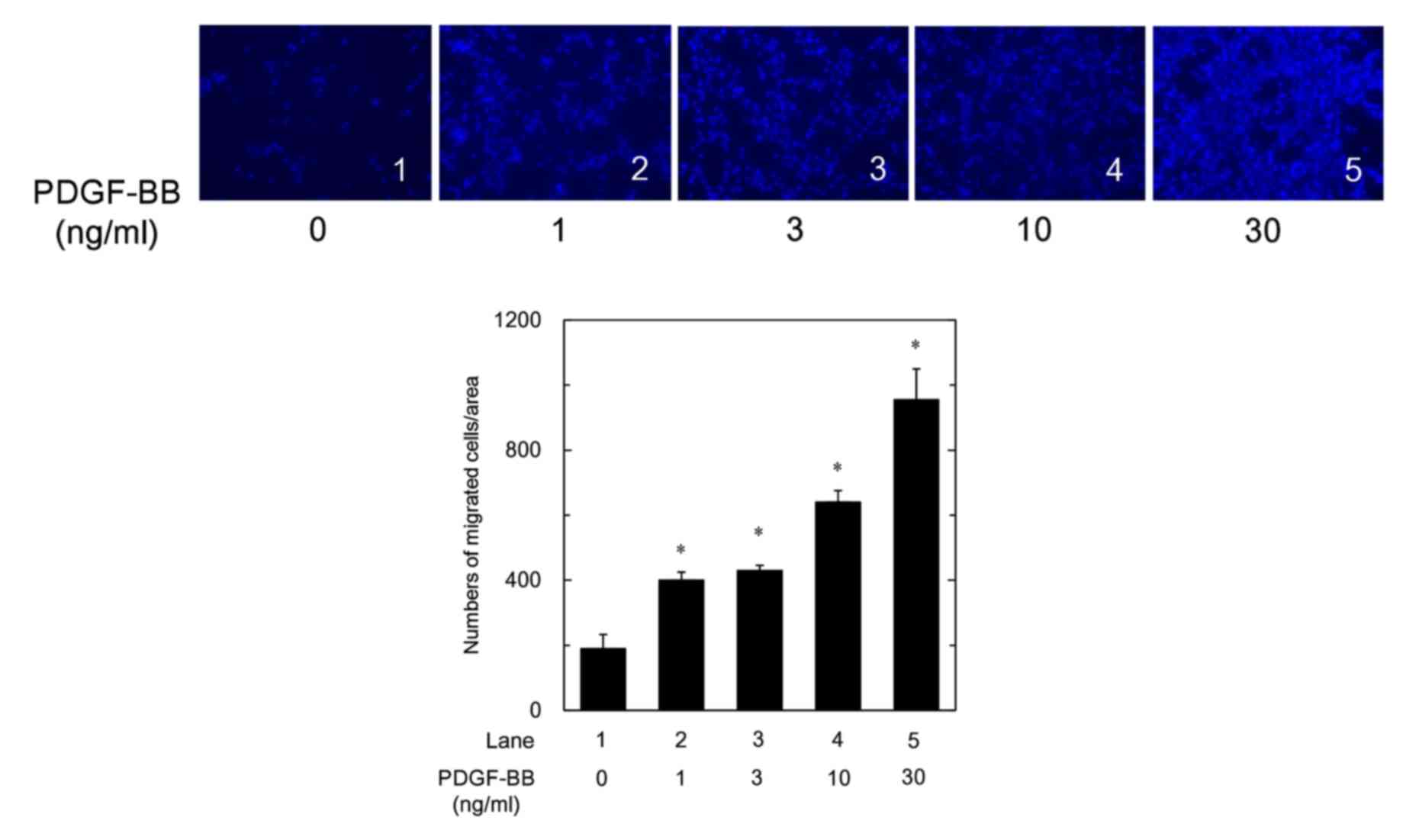
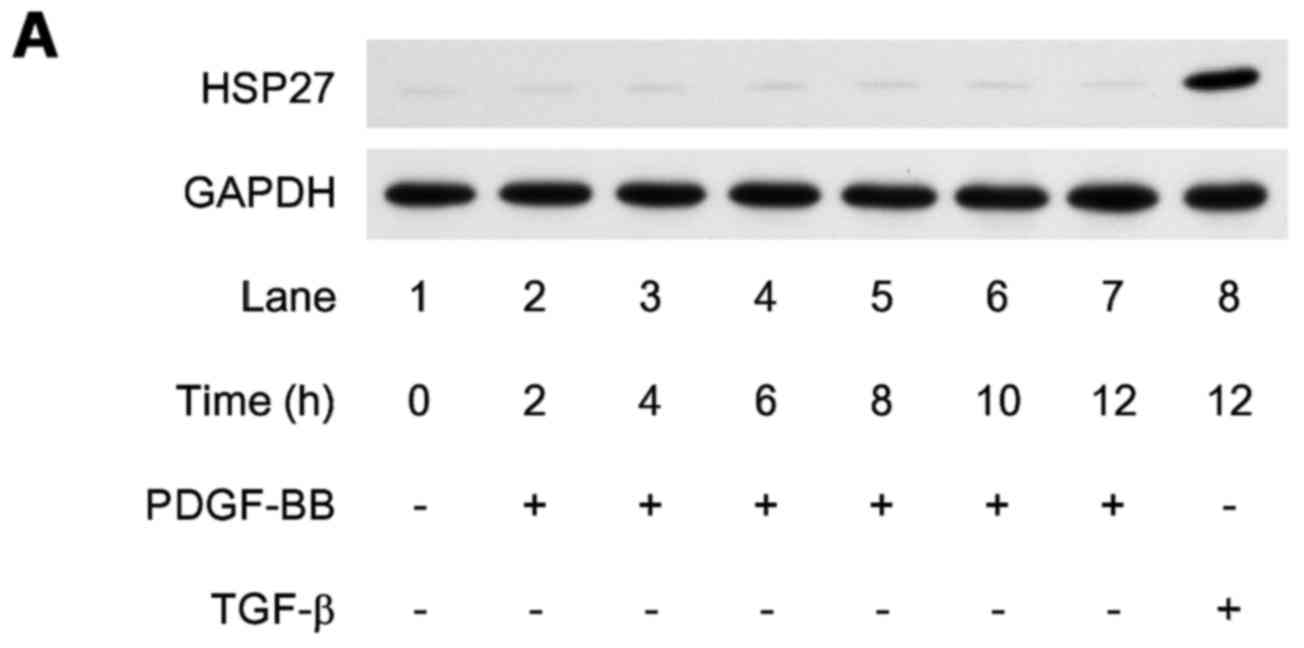
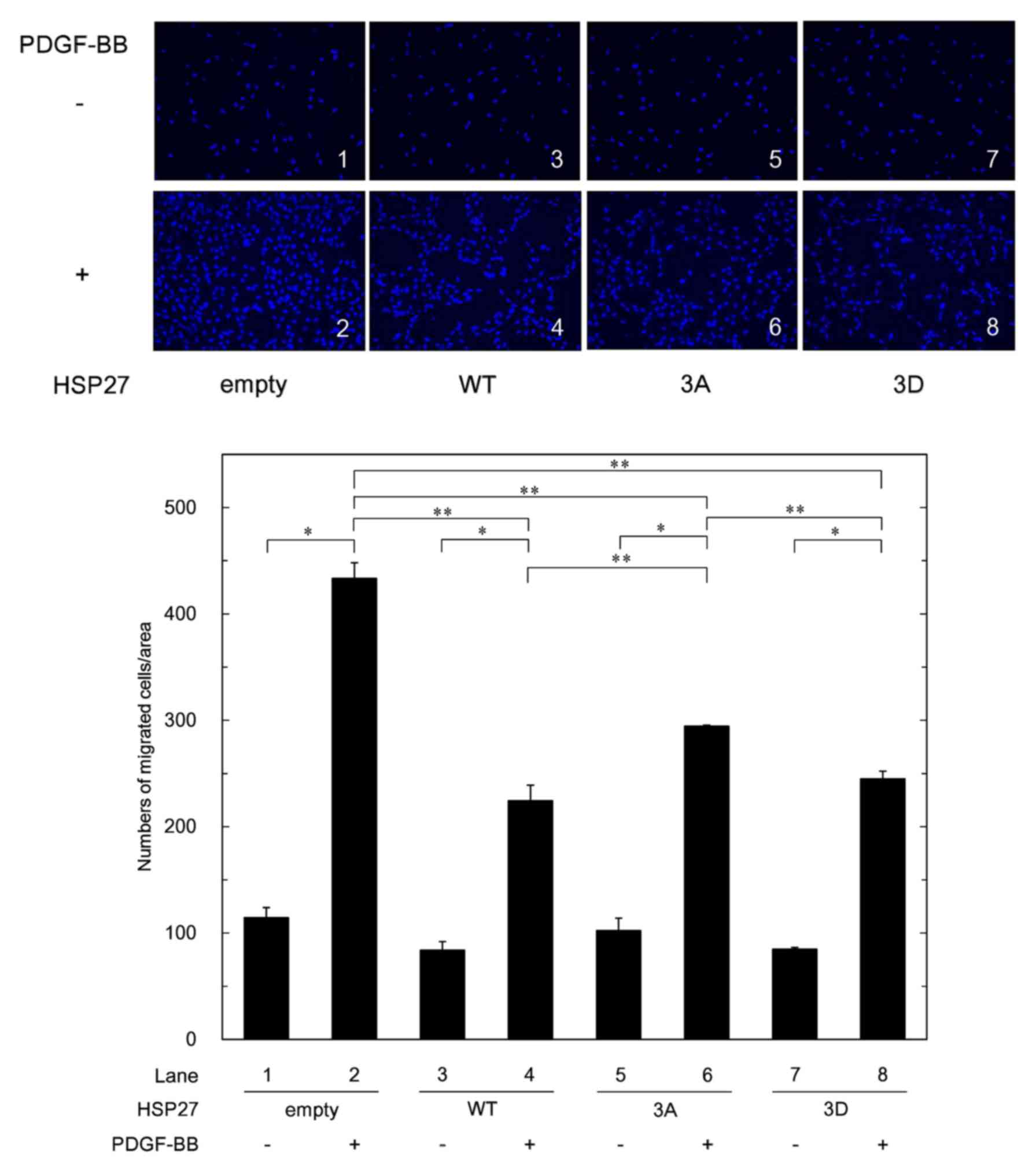
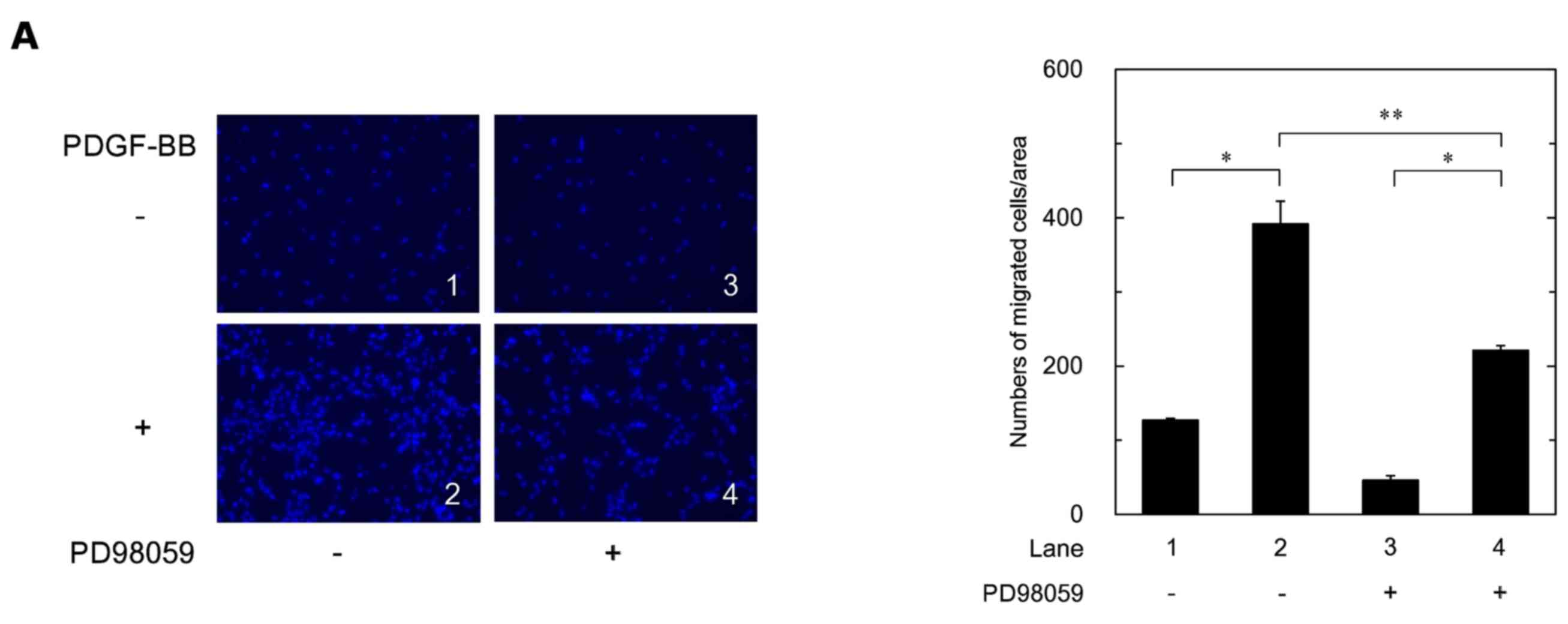
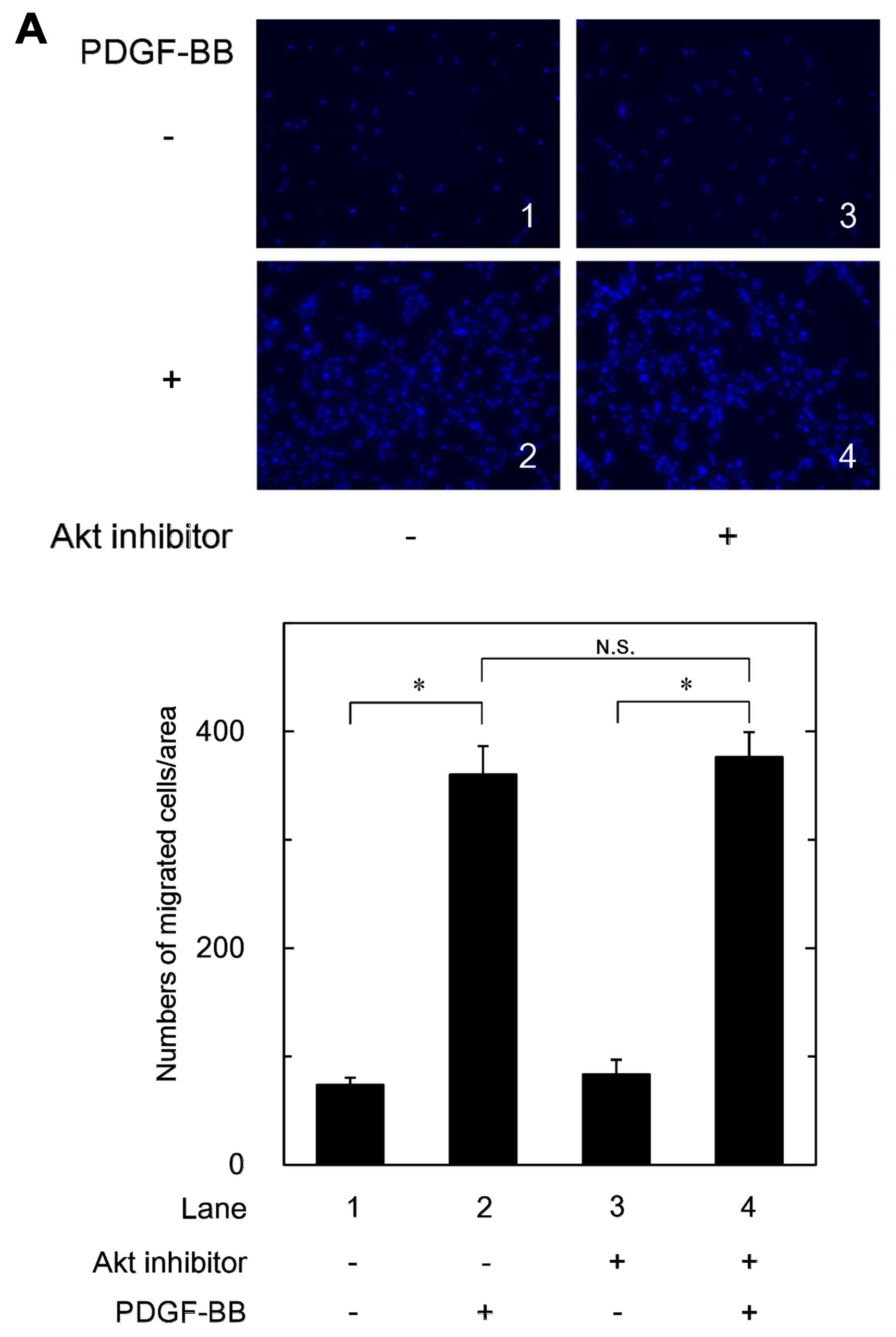
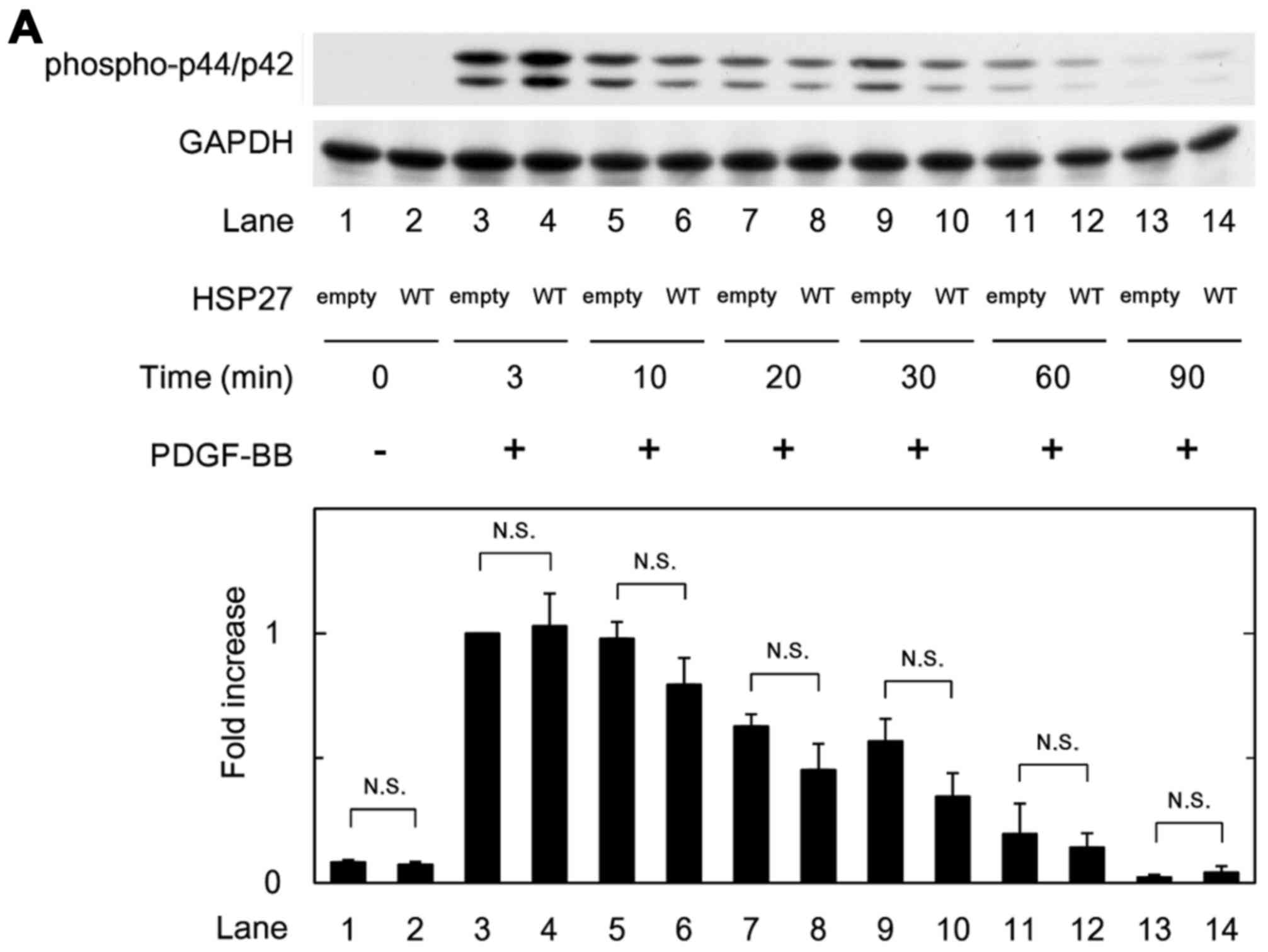
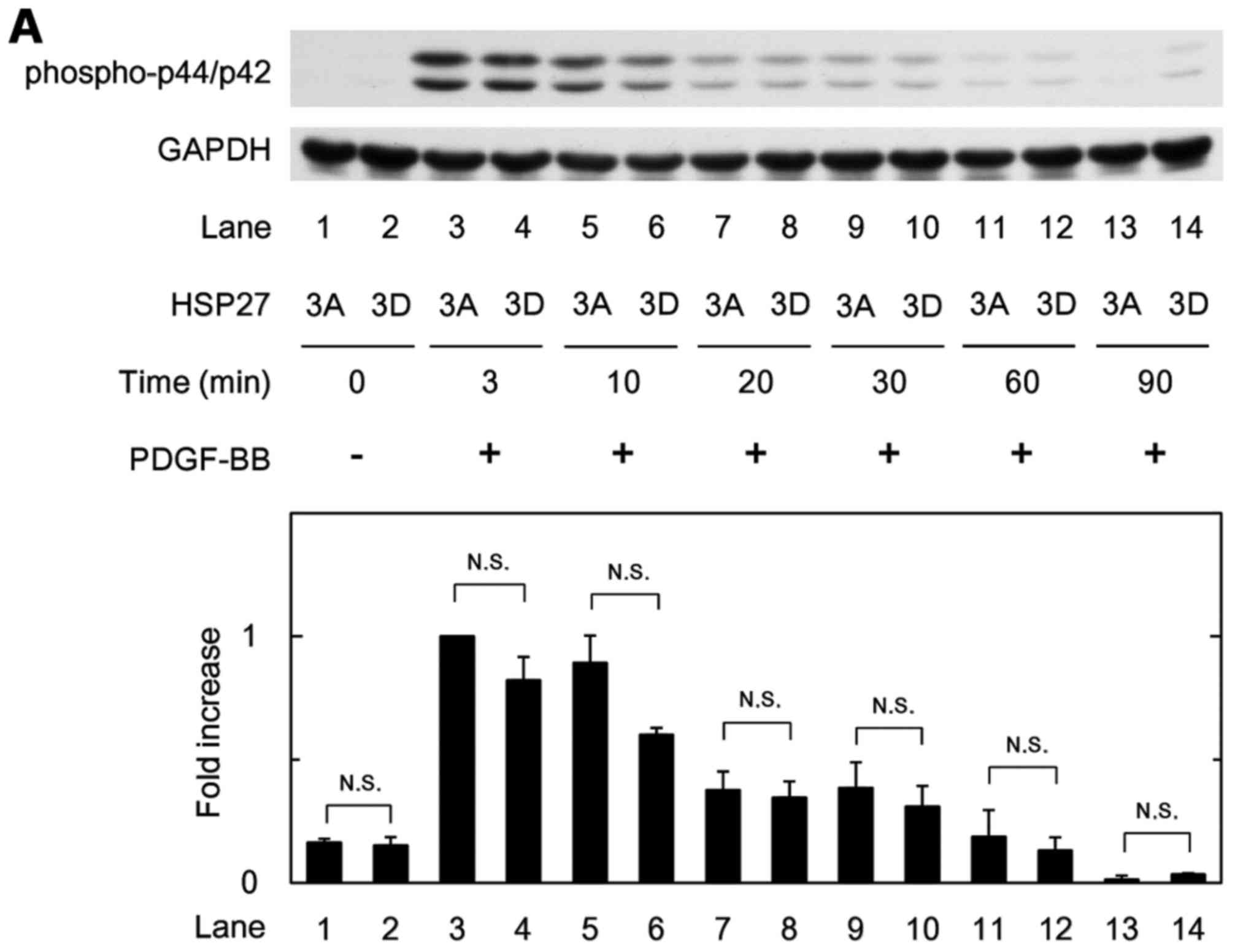
|
1
|
Heldin CH and Westermark B: Mechanism of
action and in vivo role of platelet-derived growth factor. Physiol
Rev. 79:1283–1316. 1999.PubMed/NCBI
|
|
2
|
Wang Z, Ahmad A, Li Y, Kong D, Azmi AS,
Banerjee S and Sarkar FH: Emerging roles of PDGF-D signaling
pathway in tumor development and progression. Biochim Biophys Acta.
1806:122–130. 2010.PubMed/NCBI
|
|
3
|
Kular J, Tickner J, Chim SM and Xu J: An
overview of the regulation of bone remodelling at the cellular
level. Clin Biochem. 45:863–873. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Canalis E: Growth factor control of bone
mass. J Cell Biochem. 108:769–777. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Caplan AI and Correa D: PDGF in bone
formation and regeneration: new insights into a novel mechanism
involving MSCs. J Orthop Res. 29:1795–1803. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Hollinger JO, Hart CE, Hirsch SN, Lynch S
and Friedlaender GE: Recombinant human platelet-derived growth
factor: biology and clinical applications. J Bone Joint Surg Am.
90(Suppl 1): 48–54. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Takai S, Tokuda H, Hanai Y and Kozawa O:
Limitation by p70 S6 kinase of platelet-derived growth
factor-BB-induced interleukin 6 synthesis in osteoblast-like
MC3T3-E1 cells. Metabolism. 56:476–483. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Hanai Y, Tokuda H, Ohta T,
Matsushima-Nishiwaki R, Takai S and Kozawa O: Phosphatidylinositol
3-kinase/Akt auto-regulates PDGF-BB-stimulated interleukin-6
synthesis in osteoblasts. J Cell Biochem. 99:1564–1571. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Hengartner NE, Fiedler J, Ignatius A and
Brenner RE: IL-1β inhibits human osteoblast migration. Mol Med.
19:36–42. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Mymrikov EV, Seit-Nebi AS and Gusev NB:
Large potentials of small heat shock proteins. Physiol Rev.
91:1123–1159. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Bakthisaran R, Tangirala R and Rao ChM:
Small heat shock proteins: role in cellular functions and
pathology. Biochim Biophys Acta. 1854:291–319. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Kostenko S and Moens U: Heat shock protein
27 phosphorylation: kinases, phosphatases, functions and pathology.
Cell Mol Life Sci. 66:3289–3307. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Katsogiannou M, Andrieu C and Rocchi P:
Heat shock protein 27 phosphorylation state is associated with
cancer progression. Front Genet. 5:3462014. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Leonardi R, Barbato E, Paganelli C and Lo
Muzio L: Immunolocalization of heat shock protein 27 in developing
jaw bones and tooth germs of human fetuses. Calcif Tissue Int.
75:509–516. 2004. View Article : Google Scholar
|
|
15
|
Shakoori AR, Oberdorf AM, Owen TA, Weber
LA, Hickey E, Stein JL, Lian JB and Stein GS: Expression of heat
shock genes during differentiation of mammalian osteoblasts and
promyelocytic leukemia cells. J Cell Biochem. 48:277–287. 1992.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Kozawa O, Niwa M, Matsuno H, Ishisaki A,
Kato K and Uematsu T: Stimulatory effect of basic fibroblast growth
factor on induction of heat shock protein 27 in osteoblasts: role
of protein kinase C. Arch Biochem Biophys. 388:237–242. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Kozawa O, Otsuka T, Hatakeyama D, Niwa M,
Matsuno H, Ito H, Kato K, Matsui N and Uematsu T: Mechanism of
prostaglandin D2-stimulated heat shock protein 27
induction in osteoblasts. Cell Signal. 13:535–541. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Hatakeyama D, Kozawa O, Niwa M, Matsuno H,
Kato K, Tatematsu N, Shibata T and Uematsu T: Inhibition by
adenylyl cyclase-cAMP system of ET-1-induced HSP27 in osteoblasts.
Am J Physiol Endocrinol Metab. 281:E1260–E1266. 2001.PubMed/NCBI
|
|
19
|
Tokuda H, Kozawa O, Niwa M, Matsuno H,
Kato K and Uematsu T: Mechanism of prostaglandin
E2-stimulated heat shock protein 27 induction in
osteoblast-like MC3T3- E1 cells. J Endocrinol. 172:271–281. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Tokuda H, Niwa M, Ishisaki A, Nakajima K,
Ito H, Kato K and Kozawa O: Involvement of stress-activated protein
kinase (SAPK)/c-Jun N-terminal kinase (JNK) in prostaglandin
F2α-induced heat shock protein 27 in osteoblasts.
Prostaglandins Leukot Essent Fatty Acids. 70:441–447. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Hayashi K, Takai S, Matsushima-Nishiwaki
R, Hanai Y, Kato K, Tokuda H and Kozawa O: (−)-Epigallocatechin
gallate reduces transforming growth factor β-stimulated HSP27
induction through the suppression of stress-activated protein
kinase/c-Jun N-terminal kinase in osteoblasts. Life Sci.
82:1012–1017. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Kato K, Adachi S, Matsushima-Nishiwaki R,
Minamitani C, Natsume H, Katagiri Y, Hirose Y, Mizutani J, Tokuda
H, Kozawa O, et al: Regulation by heat shock protein 27 of
osteocalcin synthesis in osteoblasts. Endocrinology. 152:1872–1882.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Kubisch C, Dimagno MJ, Tietz AB, Welsh MJ,
Ernst SA, Brandt-Nedelev B, Diebold J, Wagner AC, Göke B, Williams
JA, et al: Overexpression of heat shock protein Hsp27 protects
against cerulein-induced pancreatitis. Gastroenterology.
127:275–286. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Sudo H, Kodama HA, Amagai Y, Yamamoto S
and Kasai S: In vitro differentiation and calcification in a new
clonal osteogenic cell line derived from newborn mouse calvaria. J
Cell Biol. 96:191–198. 1983. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Kozawa O, Tokuda H, Miwa M, Kotoyori J and
Oiso Y: Cross-talk regulation between cyclic AMP production and
phosphoinositide hydrolysis induced by prostaglandin E2
in osteoblast-like cells. Exp Cell Res. 198:130–134. 1992.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Karagiosis SA, Chrisler WB, Bollinger N
and Karin NJ: Lysophosphatidic acid-induced ERK activation and
chemotaxis in MC3T3-E1 preosteoblasts are independent of EGF
receptor transactivation. J Cell Physiol. 219:716–723. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Laemmli UK: Cleavage of structural
proteins during the assembly of the head of bacteriophage T4.
Nature. 227:680–685. 1970. View
Article : Google Scholar : PubMed/NCBI
|
|
28
|
Kato K, Ito H, Hasegawa K, Inaguma Y,
Kozawa O and Asano T: Modulation of the stress-induced synthesis of
hsp27 and αB-crystallin by cyclic AMP in C6 rat glioma cells. J
Neurochem. 66:946–950. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Kozawa O, Niwa M, Matsuno H, Tokuda H,
Miwa M, Ito H, Kato K and Uematsu T: Sphingosine 1-phosphate
induces heat shock protein 27 via p38 mitogen-activated protein
kinase activation in osteoblasts. J Bone Miner Res. 14:1761–1767.
1999. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Hatakeyama D, Kozawa O, Niwa M, Matsuno H,
Ito H, Kato K, Tatematsu N, Shibata T and Uematsu T: Upregulation
by retinoic acid of transforming growth factor-β-stimulated heat
shock protein 27 induction in osteoblasts: involvement of
mitogen-activated protein kinases. Biochim Biophys Acta.
1589:15–30. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Kuroyanagi G, Tokuda H, Yamamoto N,
Matsushima-Nishiwaki R, Kozawa O and Otsuka T: Unphosphorylated
HSP27 (HSPB1) regulates the translation initiation process via a
direct association with eIF4E in osteoblasts. Int J Mol Med.
36:881–889. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Alessi DR, Cuenda A, Cohen P, Dudley DT
and Saltiel AR: PD 098059 is a specific inhibitor of the activation
of mitogen-activated protein kinase kinase in vitro and in vivo. J
Biol Chem. 270:27489–27494. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Cuenda A, Rouse J, Doza YN, Meier R, Cohen
P, Gallagher TF, Young PR and Lee JC: SB 203580 is a specific
inhibitor of a MAP kinase homologue which is stimulated by cellular
stresses and interleukin-1. FEBS Lett. 364:229–233. 1995.
View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Bennett BL, Sasaki DT, Murray BW, O'Leary
EC, Sakata ST, Xu W, Leisten JC, Motiwala A, Pierce S, Satoh Y, et
al: SP600125, an anthrapyrazolone inhibitor of Jun N-terminal
kinase. Proc Natl Acad Sci USA. 98:13681–13686. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Hu Y, Qiao L, Wang S, Rong SB, Meuillet
EJ, Berggren M, Gallegos A, Powis G and Kozikowski AP:
3-(Hydroxymethyl)-bearing phosphatidylinositol ether lipid
analogues and carbonate surrogates block PI3-K, Akt, and cancer
cell growth. J Med Chem. 43:3045–3051. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Price DJ, Grove JR, Calvo V, Avruch J and
Bierer BE: Rapamycin-induced inhibition of the 70-kilodalton S6
protein kinase. Science. 257:973–977. 1992. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Chun KH, Kosmeder JW II, Sun S, Pezzuto
JM, Lotan R, Hong WK and Lee HY: Effects of deguelin on the
phosphatidylinositol 3-kinase/Akt pathway and apoptosis in
premalignant human bronchial epithelial cells. J Natl Cancer Inst.
95:291–302. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Clarke JP and Mearow KM: Cell stress
promotes the association of phosphorylated HspB1 with F-actin. PLoS
One. 8:e689782013. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Lavoie JN, Hickey E, Weber LA and Landry
J: Modulation of actin microfilament dynamics and fluid phase
pinocytosis by phosphorylation of heat shock protein 27. J Biol
Chem. 268:24210–24214. 1993.PubMed/NCBI
|
|
40
|
Jiménez C, Portela RA, Mellado M,
Rodríguez-Frade JM, Collard J, Serrano A, Martínez-A C, Avila J and
Carrera AC: Role of the PI3K regulatory subunit in the control of
actin organization and cell migration. J Cell Biol. 151:249–262.
2000. View Article : Google Scholar : PubMed/NCBI
|