1. Introduction
Epithelial-to-mesenchymal transition (EMT) is an
evolutionarily conserved process that regulates the expression
levels of various genes in epithelial cells that assume the
mesenchymal phenotype. During the early phase of human development,
EMT is involved in stem cell plasticity and morphogenesis necessary
for proper gastrulation and organ development (1,2).
In the adult organism, tissue conservation may be regulated via EMT
[and the reverse process, mesenchymal-to-epithelial transition
(MET)], and may lead to tissue reconstruction and the restoration
of cell homeostasis following inflammatory insults. It is known
that the EMT/MET process is important in chronic inflammatory and
degenerative diseases (for example, organ fibrosis), which may
result in organ insufficiency followed by organ failure (3). EMT under physiological conditions is
similar to pathological EMT, in that it is controlled by similar
regulators, signaling pathways and effectors.
During tumor development, various types of cell,
such as circulating tumor cells (CTCs) and CTC clusters, may be
observed in the patient’s bloodstream. In addition, another type of
cell, disseminated tumor cells (DTCs), which are present in the
bone marrow, are important during metastasis development (4). CTCs and CTC clusters are released
from primary tumors after significant changes in cell morphology
due to EMT (5). It has been
demonstrated that EMT is crucial in the development of metastasis,
as follows: i) Changes in cell polarity from apical-basal to
antero-posterior and loss of adherens junctions; ii) changes from
the epithelial to the mesenchymal phenotype; and, iii) higher
mobility and invasiveness of cancer cells (6). One of the most common
characteristics of epithelial cells is the strong cell-cell
adhesion integrity via various components, such as E-cadherin and
cytokeratins, with desmosomes and adherens, and tight and gap
junctions. Furthermore, the activity of matrix metalloproteinases
(MMPs) is highly increased during EMT. These enzymes are
responsible for extracellular matrix (ECM) degradation and,
therefore, for increased mobility of mesenchymal-like cells, which
is necessary for invasion and metastasis (7). CTCs and CTC clusters represent an
attractive alternative to tissue biopsy of metastatic lesions, due
to the fact that they may be non-invasively obtained. Current
research on CTCs and CTC clusters may lead to the discovery of
novel potential diagnostic procedures that will improve knowledge
of the molecular mechanisms and perseverance of tumor cells in the
bloodstream, during and following drug therapy. CTC clusters are
aggregates consisting of various types of cell, such as tumor,
stroma and immune cells, as well as platelets. This combination of
cells is referred to as a microembolus (8). The niche, the inner microenvironment
of CTC clusters, may protect them from lysis in the bloodstream by
minimizing immune attacks and shear stress, and also facilitate
cells by promoting colonization (9).
Due to the complexity of the EMT process, special
transcription factors and signaling pathways are activated in
epithelial cells to regulate all the molecular and morphological
changes. These include the Snail family zinc finger transcriptional
factors (SNAIL), Twist family BHLH transcriptional factor (TWIST)
and zinc finger E-box binding homeobox (ZEB) factors, and the
transforming growth factor (TGF)-β and Wnt/β-catenin signaling
pathways, which are highly conserved among species (10). Notably, cancer cells must avoid
anoikis before escaping from the primary tumor. This type of
programmed cell death is observed in anchorage-dependent cells,
which are detached from the ECM (11,12). In addition, EMT is regulated by
epigenetic mechanisms, such as DNA methylation, histone methylation
and acetylation, and by microRNA binding (10). Epigenetic regulation is
particularly important due to the reversibility of EMT and the
flexibility of tumor cells to react to different internal and
external stimuli. Taken together, currently available information
may provide a complete understanding of the mechanisms underlying
EMT, and may indicate methods to develop novel drugs that are able
to target this transition process during tumorigenesis. Therefore,
a review of the present knowledge of EMT in human cancer was
prepared, focusing on the following: i) Its regulation via the
TGF-β signaling pathway, epigenetic modifications and microRNAs;
and ii) the biological properties of different types of CTCs and
their role in tumorigenesis.
2. EMT in embryogenesis
EMT and MET are crucial during embryogenesis, as
they contribute to the induction of implantation and gastrulation
(13,14).
Epithelial cells form a sheet with cells connected
by specific junctions and adherent molecules, such as desmosomes,
and tight, gap or adherens junctions. The latter are important for
constructing and assembling lateral intercellular connections in
the sheet of cells in the epithelium (15). Furthermore, epithelial cells with
apical-basal orientation are connected to the basement membrane.
Under physiological conditions, the association with the basement
membrane allows only lateral movement of epithelial cells (14). Therefore, maintaining their
organization within the epithelium prevents cell admittance into
the ECM.
The majority of mesenchymal cells of metazoans are
derived from the primitive epithelium during early embryo-genesis.
Unlike epithelial cells, mesenchymal cells display a front-rear
polarity and very rarely exhibit tight cell-cell connections
(16). Furthermore, mesenchymal
cells migrate through the ECM as a single cell. Although
epithelial, as well as mesenchymal, cells have long been
characterized in the developing embryo, EMT was only identified as
a cellular process in the 1980s. Greenburg and Hay (17) performed a set of in vitro
experiments that focused on the culture of epithelial cells in 3D
gels. The authors demonstrated that these cells lost their
apical-basal polarity and assumed a mesenchymal-like phenotype.
Furthermore, the presence of pseudopodia and filopodia in
mesenchymal cells supported the hypothesis that epithelial cells
transition to the mesenchymal phenotype via the EMT process.
Multiple steps are involved in activating EMT during embryogenesis
to enable the conversion of epithelial into mesenchymal cells
(14).
It is necessary to study the EMT/MET process during
embryogenesis, as the derived knowledge may be useful for
elucidating pathological processes, such as chronic diseases and
tumor development. Furthermore, this knowledge may be helpful for
the development of novel cancer therapeutic agents.
3. EMT in chronic diseases
Physiological regeneration shares the same molecular
principle of EMT/MET as embryonic development. EMT/MET are
important during chronic conditions caused by inflammation and
upregulated regeneration.
In fibrotic tissues, myofibroblasts produce an
excessive quantity of collagen. This protein may compromise organ
function and lead to its failure. It has been hypothesized that
fibrosis occurs via the activation of interstitial fibroblasts,
which may be transformed to myofibroblasts during pathological
processes. It was experimentally demonstrated that certain
myofibroblasts were originally epithelial cells that underwent EMT
(18–20).
Transition of endothelial cells into
mesenchymal-like cells was also observed in renal and cardiac
fibrosis (21,22). Notably, mesothelial cells may
transform into mesenchymal cells in patients who undergo ambulant
dialysis, who may develop peritoneal fibrosis, a process involving
the mitogen-activated protein kinases (MAPK) signaling pathway and
SNAIL activation (23). In
addition, EMT may occur in the epithelial cells of the lens, where
it contributes to the development of capsular opacity following
cataract surgery. It was demonstrated that SNAIL activation via
TGF-β in the adult kidney may be implicated in the induction of
renal fibrosis followed by renal failure (24). Elevated SNAIL expression levels
have been identified in fibrotic kidneys of patients subjected to
nephrectomy. Based on this observation, higher expression levels of
the TGF-β protein may be either a part of the physiological
reaction to an insult, or a pathological response. As SNAIL
transduces the detrimental effect of TGF-β, inhibition of SNAIL may
be a preferable alternative to treating kidney disease, as that
would preserve the beneficial effect on TGF-β secretion (18). Initially demonstrated in
differentiated renal ducts and tubules, it is obvious that cells of
the endothelium, epithelium and lens, cardiomyocytes and
hepatocytes may be transformed via EMT, leading to the progression
of tissue fibrosis (21,22).
These observations may be useful for future
therapeutic methods, protecting against organ fibrosis and avoiding
end-stage organ failure.
4. EMT in tumorigenesis
The process of cell de-differentiation via EMT is
currently accepted as one of the hallmarks of cancer (25,26). EMT is crucial in the initiation of
tumor cell migration and metastasis development. Once the cancer
cells begin to metastasize, they must first overcome anoikis.
Cancer cells may avoid anoikis via different methods associated
with EMT. E-cadherin and cytokeratins are proteins typically found
in epithelial cells and their decreased expression is an important
feature of EMT. In mesenchymal cells, these proteins are replaced
by mesenchymal-specific factors, including fibronectin, vimentin,
or neural cadherin (N-cadherin) (12). The changes in the expression of
E-cadherin/N-cadherin are positively correlated with the avoidance
of anoikis and an increase in cell invasiveness (27). Furthermore, it has been
demonstrated that dysregulation of growth factor receptors may
result in resistance to anoikis. Before cancer cells begin to
migrate, they must activate the genes that are necessary in various
processes, such as cell differentiation, proliferation, activation
of anti-apoptotic pathways, alteration of cellular characteristics
from the epithelial to the mesenchymal phenotype, proteolytic
digestion of the receptors that are involved in cell-cell
junctions, increased activity of adhesion molecules that assist in
cell movement, and the activation of proteases on the cell surface,
which digest components of the ECM (10). Due to the heterogeneity of cancer
cells, various epigenetic patterns may support these cellular
changes (7). In addition, all
cells cannot enter the EMT process simultaneously, and only a few
at a time may successfully initiate metastasis and progression.
Cancer progenitor cell phenotype, intracellular and extracellular
signaling, epigenetic modifications, and environmental factors all
strongly affect cells entering the EMT process and metastasis.
Studies using different types of cancer cell lines or animal tumor
models have demonstrated the relevance of EMT to metastasis. By
contrast, EMT during tumorigenesis in humans is difficult to
identify, as cancer cells that exhibit the mesenchymal phenotype
share a number of similar molecular and morphological
characteristics with stromal fibroblasts (10,28).
Regardless, the research and clinical results on
solid tumors, including colorectal, ovarian and breast cancer, have
verified the increased expression levels of typical EMT
transcription factors, such as SNAIL1 and SNAIL2. Furthermore,
these results are positively correlated with a worse prognosis in
terms of survival or relapse (29–31). The inhibition of EMT may improve
the efficiency of traditional curative therapy based on the data
from pancreatic, lung and hepatic cancer cells (32–34).
5. Molecular mechanisms of EMT
regulation
The molecular steps that regulate EMT are highly
evolutionarily conserved. The key factors in EMT are
transcriptional factors (TF), such as SNAIL, ZEB and TWIST, and
their target, E-cadherin. Various signaling pathways involved in
the induction and modification of EMT during different steps of
embryogenesis or tumorigenesis, including TGF-β/bone morphogenetic
protein, Wnt/β-catenin, tyrosine kinase receptor, T-lymphoma
invasion and metastasis inducing protein 1/Rac family small GTPase
1, and Hedgehog signaling, have been identified. These signaling
pathways regulate EMT in a context-dependent manner (35).
Inducers and TFs
TGF-β is a cytokine that is considered to be a
primary inducer of EMT (Fig. 1)
(36). It is also considered a
facilitator of metastasis with a complex role in cancer
development. The TGF-β signaling pathway is crucial to the
regulation of cell proliferation, differentiation, invasion,
migration, apoptosis and modification of the microenvironment, as
well as to cancer metastasis (37). TGF-β binds to two different
serine/threonine kinase receptors, referred to as TGF-β receptor
type I (TβRI) and type II (TβRII), which may activate SMAD and
non-SMAD signaling pathways, respectively. Activated TβRI
phosphorylates the receptor-specific SMAD2/3 elements that are
associated with SMAD4. Together, these elements form a heterogenic
complex, which is transported into the nucleus and is responsible
for the regulation of the transcription of a number of genes,
including those involved in EMT activation (38). The ability of the TGF-β/SMAD
signaling cascade to activate EMT is dependent on the collaboration
with other signaling pathways, such as the Ras kinase cascade,
through the activation of tyrosine kinase receptors and the
cooperation of the Wnt/β-catenin/lymphoid enhancer-binding factor-1
signaling pathway (39). The
TGF-β signaling pathway may regulate, and be regulated by,
different signals, as aforementioned. Furthermore, metabolic and
mechanical stresses, such as tissue hypoxia, inflammatory cytokines
and ECM stiffness, may act as powerful inducers of EMT followed by
invasion of cancer cells (40,41). It was demonstrated that one of the
main sources of TGF-β in carcinomas are the stromal fibroblasts,
which were observed in the tumor niche (25). Notably, the TGF-β cytokine exerts
different effects at various cancer stages. It maintains cell
proliferation and differentiation under physiological conditions or
in early-stage cancer; however, it also promotes cancer cell
invasion and metastasis in late-stage cancer (42).
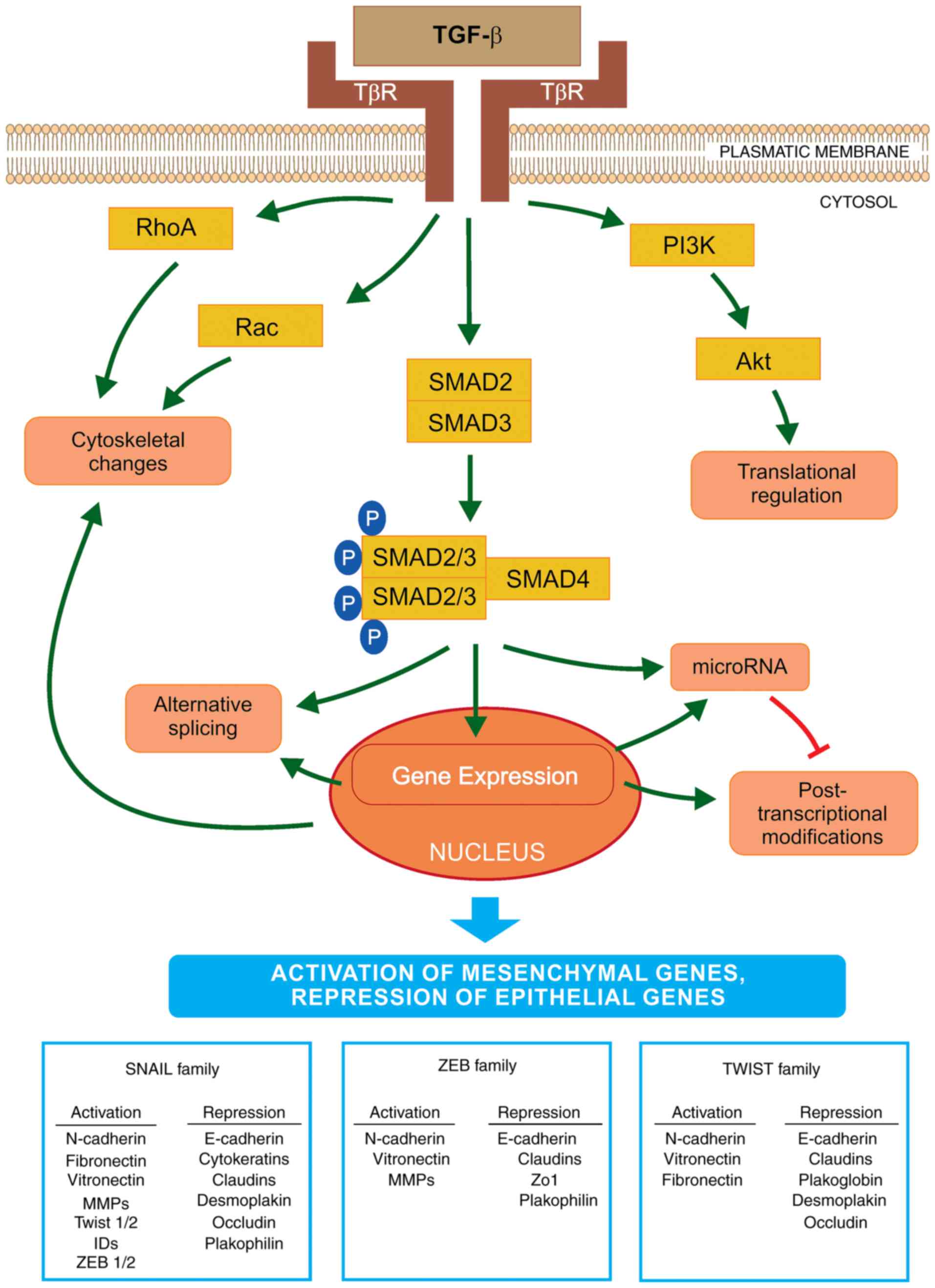 | Figure 1Schematic representation of EMT
activation via TGF-β. The initiation and progression of EMT is
regulated at all levels of macromolecule synthesis
(transcription/post-transcriptional modifications and
translation/post-translational modifications). TGF-β binds to its
receptors (TβR) and activates EMT via the SMAD signaling pathway.
SMAD2/3 are bound to the SMAD4 protein and this phosphorylated
complex is translocated into the nucleus where it interacts with
transcriptional factors to regulate the expression levels of
EMT-specific genes. The key transcriptional factors of EMT are
SNAIL, ZEB and TWIST. These are important for the activation or
repression of various genes that encode proteins involved in the
transition. Furthermore, TGF-β activates microRNA expression that
regulates gene expression at the post-transcriptional level. By
contrast, EMT transcription regulators may decrease the expression
levels of specific microRNAs that affect the EMT factors important
in the mesenchymal-like phenotype. Furthermore, TGF-β may induce
EMT via a non-SMAD pathway, by activation of the PI3K-AKT signaling
cascade, which may lead to translational regulation of EMT factors.
TGF-β also initiates a decrease in the number of cell junctions and
activates the cytoskeletal reorganization via RHO-GTPases. EMT,
epithelial-to-mesenchymal transition; TGF-β, transforming growth
factor-β; SNAIL, Snail family zinc finger transcriptional factors;
TWIST, Twist family BHLH transcriptional factor; ZEB, zinc finger
E-box binding homeobox; PI3K, phosphoinositide 3-kinase. |
TGF-β directly activates EMT core TFs (SNAIL, ZEB
and TWIST), which are the key regulators of this process. The first
group of TFs is the SNAIL zinc finger family, consisting of SNAIL1
and SNAIL2 (also referred to as SLUG). The two are able to bind to
the E-box sequences in the promoter region of the E-cadherin gene
leading to its transcriptional repression. The elevated expression
of SNAIL1 in breast cancer cell lines resulted in the loss of
cell-cell junctions due to a decrease in E-cadherin production,
followed by significant changes from the epithelial to the
mesenchymal spindle cell phenotype, with an accompanying increase
in cell mobility and invasiveness (43). Although TGF-β is crucial in the
activation of SNAIL1 expression, other factors, such as Wnt family
proteins, neurogenic locus notch homolog protein (Notch) and
certain growth factors (via tyrosine kinase receptors) also
activate the expression of SNAIL1, depending on the physiological
or pathological conditions (44).
SNAIL1 and SNAIL2 work synergistically with other TFs and regulate
the expression of various genes involved in EMT. By contrast, TGF-β
may activate the apoptotic pathway, thus, tumor cells must avoid
this type of cell death. SNAIL1 increases the expression levels of
B-cell lymphoma-extra large (Bcl-xL) and Akt, which leads to
the inhibition of apoptosis mediated by TGF-β (45). Furthermore, SNAIL downregulates
the expression of cyclin D2, which negatively affects the
cell cycle. When tumor cells begin to differentiate, their
proliferation is reduced. In the tumor niche, expression of SNAIL
may be activated via numerous factors, including: i) The cytokine
tumor necrosis factor (TNF)-α via nuclear factor (NF)-κB activation
in response to inflammation and ii) hypoxia-inducible factor
(HIF)-1, HIF-2 and Notch in response to hypoxia (46).
The second group of TFs is the zinc finger
E-box-binding family proteins, ZEB1 and ZEB2, which also
downregulate E-cadherin expression. ZEB expression is
downregulated as follows: i) Post-transcriptionally via miR-200 (a
double-negative feedback loop) and ii) post-translationally by
SUMOylation of ZEB2, which prevents its nuclear translocation and
attenuates ZEB2-mediated gene expression (47,48). ZEB expression is often followed by
activation of SNAILs and direct targeting of the expression of the
ZEB1 gene by SNAIL1. The activity of ZEB is regulated by
certain signaling pathways, such as TGF-β and Wnt, as well as by
the Ras-MAPK signaling cascade (49). It was also demonstrated that
members of the ZEB and SNAIL families may decrease the expression
of factors involved in tight junctions, including connexins
junctional adhesion molecule 1/A, or zonula occludens-1 (50).
The third group is a typical helix-loop-helix family
of TFs, including TWIST1, TWIST2, inhibitor of differentiation
proteins (ID) and E12/E47. These factors induce EMT alone or by
acting synergistically (51).
Therefore, similar to SNAIL and ZEB, TWIST binds to the E-boxes of
the promoter region of the E-cadherin gene and acts as
transcriptional repressor or activator, leading to the repression
of genes involved in the epithelial phenotype and to the activation
of EMT genes. In tumor cells, TWIST1 decreases E-cadherin
and increases N-cadherin expression levels independent of
SNAIL activity (52). TNF-α
induces the expression of TWIST1 via the NF-κB signaling pathway
(53). In the tumor niche, other
cytokines may activate Stat3 via Janus kinase and lead to the
induction of TWIST1 activity (54). In addition, the regulation of
interaction between TWIST1 and HIF-1α was analyzed. Hypoxia or
overexpression of HIF-1α leads to the activation of EMT and
metastatic phenotypes. HIF-1 may regulate the expression level of
TWIST by binding directly to the hypoxia-response element in the
proximal promoter region of TWIST (55). Furthermore, TWIST1 activates
invadopodia-mediated matrix degradation, followed by degradation of
the basement membrane during EMT (41).
Recently, other TFs contributing to and controlling
the EMT/MET cell plasticity were identified, including members of
the activator protein-1 (Jun/Fos) family, forkhead box protein C2,
Kruppel-like factor 4, paired related homeobox 1, p53, SRY-box
(Sox)4, Sox9 and TEA domain transcription factor 2 (56). Based on their specificity, the
same EMT TFs may serve as molecular markers of EMT during cancer
progression.
Effectors
Activation of EMT TFs lead to reduction in the
transcription of specific genes encoding proteins involved in
adherens and tight junctions, desmosomes, and maintaining the
apical-basal cell polarity (6).
These junctions support the epithelial phenotype and control
various signaling pathways through their associated proteins.
Therefore, dysregulation of cell-cell junctions significantly
affects a number of molecular pathways that further activate EMT
and cancer invasion. For example, β-catenin released from the
complex with E-cadherin crosses into the nucleus and promotes the
transcription of Wnt-target genes to initiate EMT. Furthermore, the
transcription of genes involved in the mesenchymal phenotype, such
as fibronectin, vimentin or N-cadherin, is increased
(57). Various non-epithelial
cadherins (N-cadherin), cell surface proteins (CD44) and integrin
β6, are induced and may be crucial for the migration of cancer
cells (58). In epithelial cells,
the cytoskeletal network connected to desmosomes is destroyed.
Expression of vimentin is upregulated, which results in the actin
and intermediated filament reorganization (59). The transformation into the
mesenchymal phenotype promotes cell migration and the formation of
actin-rich membrane protrusions (invadopodia). In addition, actin
fibers increase cell contractility. The formation of invadopodia
and increased activity of MMPs results in the degradation of
adherens junctions and other cell surface proteins, as well as ECM
fibers, which increases cancer cell motility and promotes breaking
through the basement membrane and the invasion of neighboring
tissues (26).
E-cadherin is considered a gatekeeper of the
epithelial phenotype. It is one of the main targets for MMP
digestion and its degradation allows tissue reorganization into
single cells and the activation of other signals important in EMT
induction. Cleavage of the E-cadherin ectodomain creates soluble
E-cadherin fragments that are important in EMT induction, invasion
and proliferation via estimated Glomerular Filtration Rate
signaling (60).
It is known that certain epithelial markers, such as
epithelial cell adhesion molecule (EpCAM) and cytokeratins, are
downregulated during EMT (61).
EpCAM, a transmembrane protein, is important for intercellular
connections in the epithelium. It has oncogenic potential, as it
upregulates the activity of cyclin A, cyclin E and c-Myc (62). Cytokeratins are proteins of the
keratin-containing intermediate filaments. EpCAM and cytokeratins
may be used as biomarkers for CTC detection in the blood of
patients with cancer (63).
Epigenetics
Embryogenesis is a process of cell differentiation
and growth, controlled predominantly via epigenetic events.
Similarly, epigenetic modification was found to be a key event in
cancer cells and cancer progenitor cells that undergo EMT followed
by metastasis initiation (10).
The development of cancer has long been characterized based upon
genetic regulation, although it has been demonstrated that
epigenetic modifications perform a critical role (7,10).
Epigenetics is the study of the variations and changes in the
expression levels of genes that are independent from alterations in
the DNA sequence. These changes include methylation of DNA and
histones, acetylation and phosphorylation, or their reverse
processes, as well as variations in microRNA expression.
Hypermethylation of CpG islands in the DNA sequence leads to gene
silencing and affects transcription (64). Epigenetic changes are variable and
grade-specific. They occur in different cell processes and at
different time-points, for example cell growth is decreased and
cell differentiation is activated. When cancer cell differentiation
is accomplished, cell growth is activated and novel mutations
appear. The reverse reactions occur during MET. Epigenetic
modifications that support the EMT/MET process are variable and
dynamic; their role in metastasis development and cancer
progression has been extensively investigated (7,10).
TβRs are highly active during EMT. It has been
hypothesized that the differentiation of cancer cells is not
required at the terminal level of metastasis and, thus, TβR
transcription is silenced by methylation (10). Inhibition of HDAC activity leads
to the suppression of EMT, which is mediated by the epigenetic
modification of TβRI in human renal epithelial cells (65).
The epigenetic changes are present in cancer cells,
as well as in stromal cells. However, this finding does not clarify
whether the differentiation of metastatic cells occurs
simultaneously or at different and specific time-points. Epigenetic
drifts have recently attracted attention in cancer research; these
include age-dependent alterations in DNA methylation patterns.
Epigenetic drifts may be tissue-specific. Furthermore, these drifts
may affect the differentiation of stem cells and lead to a decrease
of their stemness during aging. Furthermore, the expression of
certain microRNAs is controlled by methylation (66). The expression of another family of
regulatory RNAs, long non-coding RNAs (lncRNAs), may also be
controlled by epigenetics. For example, lncRNA H19 suppresses
E-cadherin expression in bladder cancer via hypomethylation,
resulting in enhanced metastatic progression (67).
Epigenetic changes may affect cell differentiation
and proliferation. In various cancer types, cell cycle regulator
proteins, such as p16, p21, p27 and p53, have been controlled by
methylation silencing (68). A
DNA repair enzyme, O6-methylguanine DNA methyltransferase (MGMT),
is another example of epigenetic silencing. It was demonstrated
that augmented methylation of the promoter region of the
MGMT gene in cancer cells increases the sensitivity of DNA
to damage by alkylating agents, such as temozolomide (69).
DNA methylation is generally catalyzed by DNA
methyltransferases (DNMT). DNMT3a, as well as DNMT3b, regulate
de novo methylation of DNA in the developing embryo, and
DNMT1 is responsible for DNA methylation between cell divisions.
Additionally, DNMT1 is significantly overexpressed in tumor cells
(70). Two groups of proteins,
histone deacetylases (HDAC) and methyl-binding domain proteins, are
important in downregulation of methylation of the promoter region
of the CpG islands. These proteins bind close to the promoter
region, thus inhibiting the binding of RNA polymerase II, followed
by inhibition of transcription. The levels of HDACs 1, 2 and 6 are
highly elevated in cancer cells (71). Furthermore, during the increased
acetylation of histones, HDAC inhibitors demethylate CpG residues
by decreasing DNMT1 activity (72). The changes to histone
modifications are important in gene silencing in cancer cells. The
expression of tumor suppressor genes may be regulated by epigenetic
silencing, which subsequently leads to inhibition of the apoptotic
pathway and to cancer development. Apoptosis is a precisely
controlled pathway of cell death that occurs during growth, and
regulates the cell population in adult organisms. The dysregulation
of apoptosis is a typical feature of cancer cells (73).
Alternative splicing
Alternative splicing may enable the encoding of
various protein isoforms, with possible antagonistic functions, by
one gene. It was recently demonstrated that this
post-transcriptional modification may exert an important effect on
EMT regulation (74).
The invasion isoform of the Mena
(MenaINV) protein is produced in aggressive cancer
cells. MenaINV promotes invasiveness via the
stabilization of invadopodia maturation (75). Two RNA-binding proteins, termed
epithelial splicing regulatory proteins (ESRP)1 and ESRP2, regulate
the splicing of EMT-associated genes, such as fibroblast growth
factor receptor 2, Mena, p120-catenin and erythrocyte membrane
protein band 4.1 like 5. In a study of pancreatic adenocarcinoma,
metastatic cancer cells displayed specific splice variants of CD44
that were not present in the primary cancer cells. ESRP1 acts as an
inhibitor of the CD44 isoform, which is important during EMT.
Furthermore, SNAIL inhibits ESRP1, leading to elevated expression
levels of the CD44 isoform, which is associated with the
de-differentiation and increased invasion of cancer cells (76). Alternative splicing of various
genes encoding proteins responsible for invasion was observed in
breast cancer cells undergoing EMT (74).
MicroRNAs
MicroRNAs are a group of non-coding, highly
conserved, single-stranded 19–25 nucleotide-long RNA molecules.
MicroRNAs control gene expression post-transcriptionally via
interaction with the 3′ untranslated region (UTR) of mRNA,
resulting in its degradation or inhibition of translation. Despite
the 3′UTR specificity, microRNAs interact with a number of
different mRNAs (7). MicroRNAs
are able to regulate transcription in development, as well as in
tumor cells, and they have been associated with germline, muscle
and neuronal development (77).
The regulatory role of microRNAs and EMT key inducers in cancer
cells was recently demonstrated (66). However, despite the fact that the
associations between the expression levels of certain microRNAs in
specific tumors have been extensively investigated, little is known
regarding the mechanisms underlying these associations. It is
important to identify specific microRNA targets and determine how
microRNAs initiate metastasis development and progression.
Numerous studies have analyzed the association
between the miR-200 family of microRNAs and ZEB regulators
(78). miR-200 interacts with the
3′UTR region of ZEB mRNA, followed by the downregulation of ZEB
expression and increased epithelial differentiation. Furthermore,
ZEB1/ZEB2 TFs may interact, via their ZEB binding sites, with the
promoter region of the miR-200 family and reverse control miR-200
transcription. This feedback regulation of ZEB1/ZEB2 and miR-200
family members controls cell morphology, as well as cell migration
and invasion (79). Different
double-negative feedback loops between microRNAs and key EMT TFs
have been identified, and they are important underlying mechanisms
of EMT regulation, reversibility and cell plasticity (80). Apart from its regulatory role in
EMT, the ZEB/miR-200 feedback loop is important in stem-like
phenotypes (81). The Notch
pathway has been identified downstream of ZEB/miR-200 signaling.
The Notch cascade regulates various processes in cells, such as EMT
or stemness. Dysregulation of the Notch pathway has been observed
in various types of cancer. Members of the miR-200 family affect
the mRNA of Notch pathway factors, including Jagged1 and
Mastermind-like co-activators (Maml2 and Maml3). Furthermore, Notch
stimulates ZEB1 expression and promotes the mesenchymal stem-like
cancer cell phenotype (82).
It has been demonstrated that aberrant expression of
p53 may increase the activity of certain microRNAs, including
members of the miR-192 and miR-200 families, leading to the
downregulation of factors, such as B lymphoma Moloney murine
leukemia virus insertion region 1 and ZEB, which are involved in
EMT and cell stemness (81,83). Other members of the miR-200 family
(miR-10b, miR-373 and miR-520c) have been associated with the
progression of breast cancer. In addition, the expression level of
miR-21 was found to be highly overregulated in EMT mediated by the
TGF-β signaling pathway (66).
MicroRNAs regulate the expression of E-cadherin
directly or indirectly. This transmembrane glycoprotein is a
crucial factor of the adherens junctions in the epithelium, and is
involved in structural and intracellular signaling. E-cadherin is
post-transcriptionally downregulated by miR-9. It was demonstrated
that miR-9 is highly overexpressed in the primary breast tumors of
patients with metastases, compared with non-metastatic patients. An
aberrant expression of miR-9 activates EMT, and results in ~70%
reduction in E-cadherin expression and the activation of vimentin
expression (84).
In conclusion, numerous EMT TFs, microRNAs,
epigenetic modifications, lncRNAs and alternative splicing factors
appear to control the process of EMT at various molecular levels.
However, the epistatic hierarchy of these regulatory networks must
be established.
6. Different types of metastatic cancer
cells
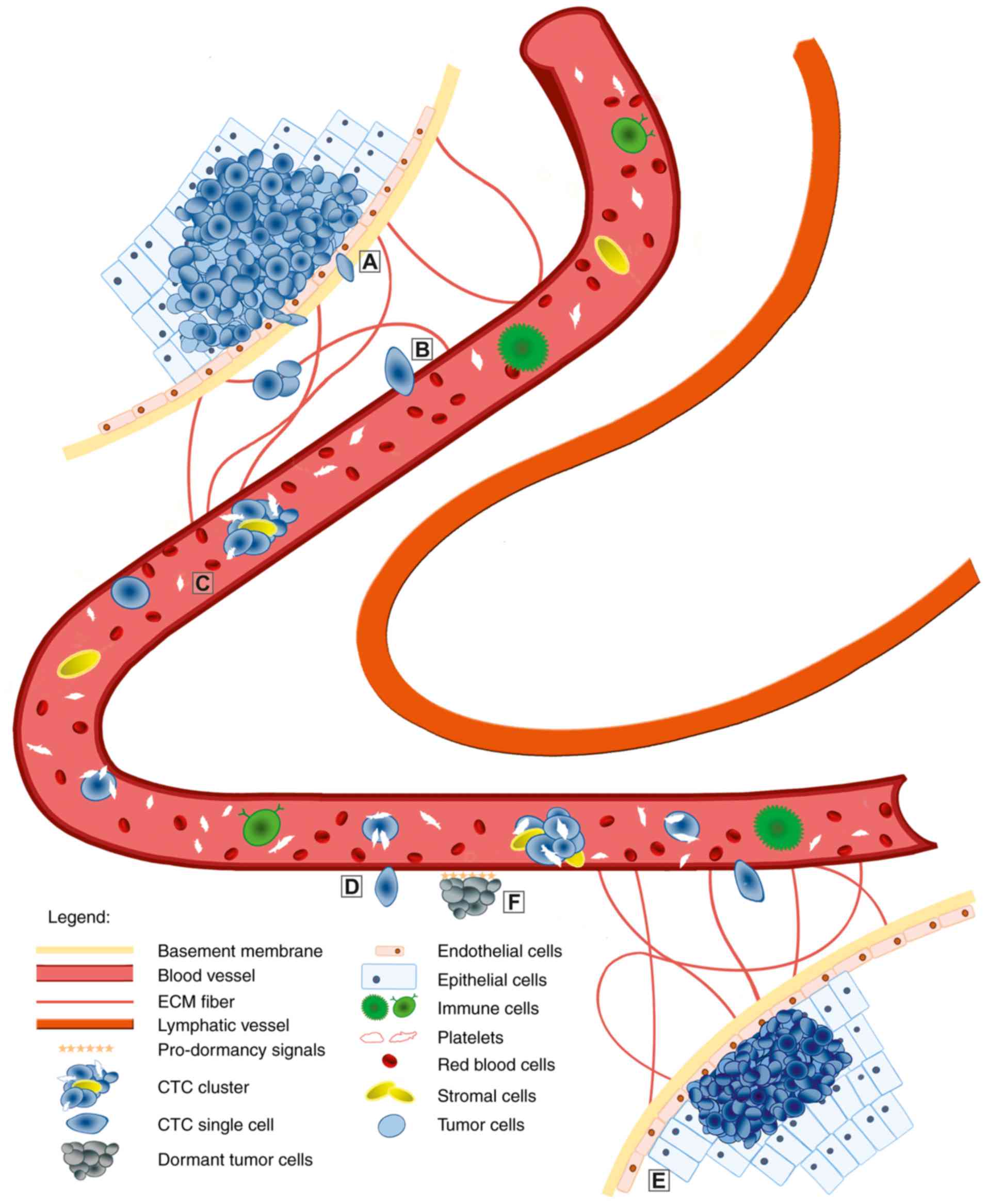
CTCs (Fig. 2) have
attracted increasing attention due to their crucial function in
metastasis development. CTCs were first detected by a pathologist,
Thomas Ashworth, in the blood of a male patient with metastatic
disease >150 years ago (85).
However, for more than a century, research focused on CTCs was not
possible due to technological limitations. Recently, CTC
characterization has become more widespread with the technological
advances in detection and separation methods.
Another milestone in cancer research was the
identification of CTC clusters (Fig.
2). CTC clusters represent a class of >2 to <100 cancer
cells with efficient cell-cell interactions found in the
circulation. In 1954, Watanabe demonstrated the function of CTC
clusters in metastasis formation by proving that cancer cells in
clusters were able to form metastases, while individual cells were
not (86). Research over the last
20 years has verified this observation, using various cancer cell
types and colon cancer-derived liver metastasis or melanoma-derived
lung metastasis. It was demonstrated that metastasis development is
dependent on the quantity, as well as on the size, of the CTC
clusters (9).
Another group of tumor cells, referred to as dormant
tumor cells (DTCs), was identified to be important in metastatic
progression (Fig. 2). These cells
may remain dormant for numerous years prior to re-growth. However,
due to insufficient specific markers for the detection and
isolation of DTCs in the epithelium of cancer patients or animal
models, current knowledge of this process is solely derived from
bone marrow micrometastasis. Watson et al (87) isolated and analyzed the molecular
profile of micrometastases from the bone marrow, and identified
TWIST1 as a factor in the detection of early tumor relapse in
breast cancer patients. However, further studies focusing on EMT
during cancer dormancy are required.
CTCs and CTC clusters in the
circulation
CTCs do not migrate through the bloodstream alone,
and also receive support from other non-tumor cells. Platelets
rapidly coat CTCs, protecting them from violent shear forces. Tumor
cells interact with the platelet’s adhesive proteins (fibronectin
and von Willebrand factor) through integrins, supporting cluster
formation (88). Coagulates of
tumor cells and platelets create microemboli, which become
entrapped in small vessels and, thus, require a longer time to
enter tissues. Furthermore, platelets support adhesion to the
luminal side of the endothelium. Notably, platelets also form a
shield against immune cells via the inactivation of natural killer
(NK) group 2D receptors on NK cells and T lymphocytes (89). Immune attenuation arises via the
transfer of major histocompatibility complex I complexes from
granulated platelets to CTCs, obtaining a new ‘selfʼ identity and
preventing NK cell-mediated cytolytic attacks. However, CTCs
survive only for 1–2.4 h in the circulation based on in vivo
experiments; the majority of CTCs die in the circulation due to
shear stresses and/or anoikis (90).
Recently, CTC clusters were identified to possess
different properties compared with single CTCs, including for
phenotype, gene expression profile and dissemination activity. Our
current knowledge indicates that metastasis arising from CTC
clusters is equally important to that arising from CTCs. Apart from
tumor cells, CTC clusters contain non-tumor cells, such as
mesenchymal, epithelial and immune cells, platelets, pericytes or
fibroblasts (91). These
non-tumor elements help CTC clusters to survive and effectively
metastasize. For example, tumor-associated stromal cells
(endothelial fibroblasts and tumor-infiltrated myeloid cells)
promote the survival of cancer cells in CTC clusters, and promote
the growth of metastases (Strnádel; unpublished data). Furthermore,
following a reduction in the number of tumor-derived fibroblasts in
CTC clusters, decreased metastatic activity was observed. When
mouse mammary tumor cells were co-cultured with endothelial cells
in 3D spheroids, promotion of angiogenesis was observed in cancer
cells, resulting in greater numbers and larger sizes of metastatic
lesions (92). Platelets in the
CTC clusters, as in CTCs, may protect cancer cells from immune
attacks and blood shear damage by physical shielding and ‘platelet
mimicry’. In addition, laboratory evidence indicates that activated
platelets interact with cancer cells within the tumor
microenvironment via paracrine signaling and direct contact
(93). Other unspecified cells,
including cytokeratin-positive dendritic cell-like cells have been
detected in CTC clusters, although their biological function
remains unknown (9).
Metastatic activity of CTCs and CTC
clusters
Research into the biological properties of CTCs may
improve our understanding of their metastatic activity in humans.
The presence of CTCs in colorectal, breast, pancreatic, lung and
prostate cancer support their role in cancer cell dissemination and
invasion during metastasis development, which is associated with a
poor clinical outcome (94–98). The co-expression of the two sets
of genes, namely mesenchymal and epithelial, was confirmed in CTCs
from various cancer types, while not in the corresponding primary
tumor cells. Furthermore, high numbers of CTCs with increased
expression levels of EMT factors (e.g., SNAIL, TWIST or vimentin)
were detected in patients with advanced-stage cancer, compared with
in those with early-stage cancer. This may indicate that CTCs
should prevail during cancer progression and contribute to
metastatic outgrowth (26). Mego
et al (99) analyzed the
expression levels of the EMT regulators, TWIST and SNAIL in CTCs
from breast cancer patients, and demonstrated a positive
correlation between the increased expression levels of these
factors and the prediction of early disease relapse. In
hepatocellular carcinoma patients, the overexpression of vimentin
and TWIST in CTCs was positively correlated with cancer progression
and increased metastasis development (100). Furthermore, CTCs with a
mesenchymal-like phenotype were associated with decreased
therapeutic efficacy in lung, colorectal and breast cancer
patients.
Various recent studies have focused on the reasons
why CTC clusters exhibit a higher metastatic potential when
compared with CTCs (101,102).
It was demonstrated that interactions between cancer
cell-associated mucin 1 and circulating galectin-3 assist by
prolonging CTC cluster survival and preventing anoikis, as well as
by increasing adhesions between tumor and endothelial cells in CTC
clusters (103). These findings
may improve our knowledge of the molecular signaling pathways
involved in the dissemination of CTC clusters, which may be useful
in designing novel therapeutic approaches for the treatment of
metastases.
Using an animal model of breast cancer, Aceto et
al (8) demonstrated that the
metastatic potential of CTC clusters is higher (23–50-fold)
compared with single CTCs. Two years later, Cheung et al
(104) validated this
observation and reported that ~97% of metastases originate from CTC
clusters. However, there remains the question of the origin of CTC
clusters: Whether they are actively separated from the primary
tumor as a group of cells and migrate into the bloodstream, or
whether they are released as single tumor cells and form aggregates
once in the bloodstream. Using differentially labeled cancer cells,
which were injected into the mammary fat pad of mice, it was
demonstrated that CTC clusters could not be formed in the
bloodstream, but that they arise from polyclonal primary tumors
(8,104). Notably, these CTC clusters form
polyclonal metastases at distant sites. Furthermore, Au et
al (105) observed that CTC
clusters with <20 cells reorganize into single chain-like
formations, which helps to decrease hydrodynamic resistance and
simplifies their passage through small vessels. The plasticity of
CTC clusters is particularly high, and chain-like cells are
reorganized to spheroid-shaped clusters after passing through the
small blood vessels. These results improved our understanding of
CTC cluster flexibility during invasion. The EMT phenotype in CTCs
is not particularly stable, and may change as part of the response
to therapy. It was demonstrated that CTC clusters may express more
mesenchymal factors in patients during anticancer therapy. The CTC
cluster phenotype is a mixture of epithelial and mesenchymal
characteristics, to which the high plasticity of this complex is
attributed (8). Recently, Cima
et al (106) demonstrated
the presence of non-cancer tumor-derived circulating endothelial
cell clusters in colorectal cancer. Clusters of endothelial cells
expressed mesenchymal, as well as epithelial factors, although they
did not display the genetic variations of the primary tumor. The
presence of benign endothelial clusters may provide novel insights
into tumor vasculature during therapy (107).
7. Methods for CTC and CTC cluster
detection
Although aggressive tumors release thousands of CTCs
into the blood stream daily, CTCs and CTC clusters are rarely
identified in the circulation. In the peripheral blood, a count of
one single CTC per 105–107 mononuclear cells
may be expected, while CTC clusters are even more rare. These cells
represent only 3% of detected circulating cells (8). For the detection of these cells in
the blood, methods allowing the enrichment of their fraction prior
to detection are required. In recent years, significant efforts
have been made to develop appropriate methods for the enrichment
and identification of CTCs. Methods for CTC and CTC cluster
detection, and isolation must be highly specific to distinguish
CTCs from blood elements. A number of techniques, which are focused
on CTC enrichment, have used specific markers to separate CTCs from
leukocytes. Cytokeratins and EpCAM are predominantly applied as
common epithelial markers (63,107).
By contrast, previous studies using cell lines or
patient samples demonstrated that ~20% of cells exhibited decreased
EpCAM expression levels. In these cells, the increased expression
levels of mesenchymal markers and the decreased expression levels
of epithelial markers have been verified. It was hypothesized that
the detection of mesenchymal-like cells may hinder or underestimate
the number of CTCs based upon the expression level of EpCAM
(108). This observation may be
significant for prognosis, as EpCAM-CTCs from the blood samples of
breast cancer patients may produce a specific population of CTCs
(positive for human epidermal growth factor receptor 2/epidermal
growth factor receptor/heparanase/Notch1), which metastasize to
brain tissue (109).
Furthermore, the selection of CTCs based on the presence of EpCAM
may falsely identify normal cells as cancer cells. It was
demonstrated that even erythroid progenitor cells from the bone
marrow may temporarily express EpCAM. However, prostate cancer
cells disseminated in the bone marrow, which show dormancy
signatures, also exhibited EpCAM positivity (110).
CTC clusters display a combination of epithelial and
mesenchymal features. Therefore, the detection methods that are
based upon the presence of typical epithelial markers may
underestimate the number of CTC clusters (111). Techniques for CTC isolation and
detection use their specific characteristics to distinguish them
from blood cells, including: i) Physical properties, such as cell
density, size and electrical charge; and ii) biological properties,
including the expression of specific surface markers and invasion
potential. Therefore, a number of techniques are based on the
immunoaffinity properties, using specific antibodies bound to
magnetic beads to enrich the CTC fraction and avoid leukocyte
contamination (112). Only one
method, the CellSearch system, is currently validated by the USA
Food and Drug Administration for clinical use (113). A good prognostic outcome for
cancer patients is considered to be the detection of <5
circulating cells per 7.5-ml blood sample. CellSearch was used for
CTC detection in various types of cancer, including colorectal,
prostate and breast cancer. The benefit of this technique is its
reproducibility, while its disadvantage is a lower level of CTC
detection due to their epithelial-mesenchymal plasticity. Other
detection methods, such as Parsortix and ScreenCell, are based upon
the physical properties, with size-based enrichment platforms.
These methods take advantage of the slightly bigger size of CTCs
compared with white (WBCs) and red blood cells (RBCs; WBCs are
sized approximately 7–15 μm; RBCs are ~8 μm; and
single CTCs are approximately 12–25 μm) (114). Another detection method for CTCs
and CTC clusters was recently employed, using a combination of
hydrodynamic cell separation and immunomagnetic depletion of
antibody-tagged WBCs to isolate larger CTCs. These are recently
developed microfluidic devices, such as the spiral biochip or the
CTC-iChip (115). Furthermore,
based on the rare presence of CTCs, GILUPI Nanomedizin developed a
novel method for in vivo CTC enrichment using EpCAM-coated
wire that may be inserted into the antecubital vein (116).
8. Future perspectives
EMT is a highly complex, dynamic and precisely
regulated developmental process that is important during
embryogenesis. It is also crucial during metastasis development and
the progression of chronic diseases. Thus, EMT/MET are reverse
processes, and they represent a complex, highly controlled
reversible reaction at different stages during cellular transition.
Epithelial cells usually enter EMT simultaneously and express
epithelial and mesenchymal markers. It is necessary to investigate
the dynamic plasticity and heterogeneity of cancer cells that
undergo EMT. Over the past two decades, an intense EMT study has
been focused on the EMT/MET process that occurs during in
vivo metastasis development, as well as in circulating cells
exhibiting the EMT phenotype (117–119). Research data derived from
various studied models, including cancer cell lines, animal cancer
and human cancer samples, have demonstrated the importance of EMT
for successful metastasis development. Due to the lack of detection
of the typical EMT markers in CTCs, it is possible that certain
tumor cells disseminate and metastasize using a mechanism other
than EMT (26). Due to the
inability for CTC or CTC cluster detection, the presence of EMT
during dissemination cannot be excluded. This may also be due to
the limitations of the detection methods. However, there may be
different mechanisms underlying invasiveness and metastasis
development, and further research is required to identify novel and
effective EMT markers for the assessment of the EMT process in
tumorigenesis.
One of the accepted theories for cancer relapse is
based on the presence of cancer progenitor cells in the tumor, or
also in the CTC clusters, as well as the presence of progenitor
CTCs in the circulation (7,10,94). The occurrence of progenitor cells
in patients who are in remission should alert physicians to the
risk of potential cancer relapse. Various clinical studies
demonstrated that the combination of standard chemotherapy and
epigenetic drugs may be a powerful treatment model for reducing
relapse in various cancer types (120). The improvement in cancer
treatment may also focus on the increased efficiency of therapy
against cancer progenitor cells. However, further research and
clinical studies are required to achieve in-depth understanding of
the mechanisms underlying epigenetic-based therapies. The
hypermethylation of numerous genes is likely to be associated with
cancer (121). It has been
revealed that the combination of inhibitors, DNMT and HDAC, may
represent a promising approach to the treatment of myelodysplastic
syndrome (122). This provides
the potential to investigate the effect of hypermethylated DNA
regions associated with higher DNMT1 activity, which may affect
epigenetic modifications of enhancers and TFs, as well as gene
expression during carcinogenesis. Modification of histones by
acetylation or deacetylation is crucial in epigenetics and
chromatin remodeling. Therefore, inhibition of the HDAC activity
exerts a strong effect on cancer progression and pathogenesis. The
activity of HDAC is markedly increased in various types of
carcinoma (123,124). Therefore, HDAC inhibition may
negatively affect tumor progression and support the apoptotic
process in cancer cells, while healthy tissues would not be
affected. HDAC inhibitors have an impact on DNA-histone structure,
as well as on the acetylation level of non-histone proteins.
Studies analyzing the effect of HDAC inhibitors on cancer
progression demonstrated their strong anti-tumor activity. For
example, suberoylanilide hydroxamic acid/vorinostat and romidepsin
are used in peripheral and cutaneous T-cell lymphoma therapy
(125–127). In addition, panobinostat has
been clinically successful in the treatment of multiple myeloma
(128). In addition, it has been
demonstrated that the HDAC inhibitors applied in breast cancer
studies display a strong activity in combination with aromatase
inhibitors, cytotoxic drugs, pro-drugs and radiation therapy
(120). A number of inhibitors
are currently in the advanced stages of clinical testing.
MicroRNAs are also implicated in carcinogenesis and
disease progression. Their role in the transition process is
obvious and further elucidates EMT regulation. The identification
of microRNA downstream and upstream EMT targets may present novel
possibilities for biomarker determination during cancer
progression, leading to improvements in prognosis and therapy.
There are several questions regarding the biology of
CTCs and CTC clusters, including: i) Establishing a standardized
method for isolating CTCs or CTC clusters with conservation of
their molecular and morphological status; ii) understanding the
biological pathways underlying CTC cluster composition and
acquiring knowledge on cancer cells that are transformed into CTCs;
and, iii) investigating the molecular mechanisms underlying CTC
cluster formation, their migration into the circulation and
metastasis development at distant sites. Answering these questions
may reveal novel areas in CTC and CTC cluster research, and should
be considered in future diagnostic and therapeutic approaches.
A deeper understanding of the biology and molecular
mechanisms of DTC activation may help to identify specific factors
responsible for metastatic progression (129). Future analysis of the dormant
niche using mass spectrometry and metabolomics may help to identify
proteins and metabolites that lead to the induction and activation
of cancer cell dormancy. Development of tissue-specific models that
may simulate the native DTC niche is crucial for understanding the
dynamic changes that occur in the tumor niche following
chemotherapy treatment. Novel markers or molecular pathways
involved in tumor cell dormancy initiation and progression are
required for further studies (90). Therefore, the optimal therapy may
be a specific treatment that is applied directly following
detection of the primary tumor, in order to avoid the dissemination
and dormancy of cancer cells.
Research and clinical studies support the
significance of EMT in tumorigenesis. Therefore, novel treatments
focused on CTC clusters and DTC elimination, in combination with
conventional therapies, should decrease metastasis, progression and
resistance to specific drugs. For example, certain preclinical
studies have focused on testing the effect of low-molecular-weight
compounds on EMT-inducing factors and their signaling cascades
(130). Based on the present
knowledge of cellular and molecular variability during EMT,
therapeutic interventions based on anti-EMT treatment may be
precisely planned and applied. Therefore, preventing the transition
process may be useful during treatment of primary tumor cells that
are able to change their epithelial phenotype to a mesenchymal
phenotype. Additionally, the novel markers, including transcriptome
analysis of EMT characteristics from primary tumors and from CTCs
or CTC clusters, may facilitate with understanding the response to
treatment and with improving patient prognosis, as well as in
designing novel cancer treatments.
Abbreviations:
|
CTCs
|
circulating tumor cells
|
|
DNMT
|
DNA methyltransferase
|
|
DTCs
|
disseminated tumor cells
|
|
ECM
|
extracellular matrix
|
|
E-cadherin
|
epithelial cadherin
|
|
EMT
|
epithelial-to-mesenchymal
transition
|
|
EpCAM
|
epithelial cellular adhesion
molecule
|
|
ESRP1
|
epithelial splicing regulatory protein
1
|
|
HDAC
|
histone deacetylase
|
|
HIF
|
hypoxia-inducible factor
|
|
ID
|
inhibitor of differentiation
proteins
|
|
lncRNA
|
long non-coding RNA
|
|
MET
|
mesenchymal-to-epithelial
transition
|
|
MMP
|
matrix metalloproteinase
|
|
N-cadherin
|
neural cadherin
|
|
NF-κB
|
nuclear factor κ-light-chain-enhancer
of activated B cells
|
|
NK cells
|
natural killer cells
|
|
Notch
|
neurogenic locus notch homolog
protein
|
|
SNAIL1
|
Snail family zinc 1 finger
|
|
TIAM1
|
T-lymphoma invasion and metastasis
inducing protein 1
|
|
TF
|
transcriptional factor
|
|
TβR
|
transforming growth factor β
receptor
|
|
TGF-β
|
transforming growth factor-β
|
|
TNF-α
|
tumor necrosis factor-α
|
|
TWIST1
|
Twist family BHLH transcriptional
factor 1
|
|
ZEB
|
zinc finger E-box binding
homeobox
|
Acknowledgments
The authors would like to thank Mrs. Zuzana
Necpalová (TJTN Matúš, Slovakia) and Mr. Miroslav Škoviera
(Lutheran Academy in Martin, Slovakia) for figure preparation. The
current study was supported by the following grants: Slovak
Scientific Grant Agency (grant no. VEGA 1/0178/17), Research and
Development Support Agency (grant no. APVV-15-0217) and the project
‘Biomedical Center Martin’, (ITMS code 26220220187) co-financed
from EU sources.
References
|
1
|
Nakaya Y and Sheng G: Epithelial to
mesenchymal transition during gastrulation: An embryological view.
Dev Growth Differ. 50:755–766. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Qin Q, Xu Y, He T, Qin C and Xu J: Normal
and disease-related biological functions of Twist1 and underlying
molecular mechanisms. Cell Res. 22:90–106. 2012. View Article : Google Scholar :
|
|
3
|
Piera-Velazquez S, Li Z and Jimenez SA:
Role of endothelial- mesenchymal transition (EndoMT) in the
pathogenesis of fibrotic disorders. Am J Pathol. 179:1074–1080.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Fidler IJ: The pathogenesis of cancer
metastasis: The ‘seed and soil’ hypothesis revisited. Nat Rev
Cancer. 3:453–458. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Książkiewicz M, Markiewicz A and Zaczek
AJ: Epithelial-mesenchymal transition: A hallmark in metastasis
formation linking circulating tumor cells and cancer stem cells.
Pathobiology. 79:195–208. 2012. View Article : Google Scholar
|
|
6
|
Nieto M: Epithelial plasticity: A common
theme in embryonic and cancer cells. Science. 342:12348502013.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Sarkar S, Horn G, Moulton K, Oza A, Byler
S, Kokolus S and Longacre M: Cancer development, progression, and
therapy: An epigenetic overview. Int J Mol Sci. 14:21087–21113.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Aceto N, Bardia A, Miyamoto DT, Donaldson
MC, Wittner BS, Spencer JA, Yu M, Pely A, Engstrom A, Zhu H, et al:
Circulating tumor cell clusters are oligoclonal precursors of
breast cancer metastasis. Cell. 158:1110–1122. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Hong Y, Fang F and Zhang Q: Circulating
tumor cell clusters: What we know and what we expect (Review). Int
J Oncol. 49:2206–2216. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Heerboth S, Housman G, Leary M, Longacre
M, Byler S, Lapinska K, Willbanks A and Sarkar S: EMT and tumor
metastasis. Clin Transl Med. 4:62015. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Kim YN, Koo KH, Sung JY, Yun UJ and Kim H:
Anoikis resistance: An essential prerequisite for tumor metastasis.
Int J Cell Biol. 2012:3068792012. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Iwatsuki M, Mimori K, Yokobori T, Ishi H,
Beppu T, Nakamori S, Baba H and Mori M: Epithelial-mesenchymal
transition in cancer development and its clinical significance.
Cancer Sci. 101:293–299. 2010. View Article : Google Scholar
|
|
13
|
Chaffer CL, Thompson EW and Williams ED:
Mesenchymal to epithelial transition in development and disease.
Cells Tissues Organs. 185:7–19. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Yang J and Weinberg RA:
Epithelial-mesenchymal transition: At the crossroads of development
and tumor metastasis. Dev Cell. 14:818–829. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Yap AS, Brieher WM and Gumbiner BM:
Molecular and functional analysis of cadherin-based adherens
junctions. Annu Rev Cell Dev Biol. 13:119–146. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Nelson WJ: Remodeling epithelial cell
organization: Transitions between front-rear and apical-basal
polarity. Cold Spring Harb Perspect Biol. 1:a0005132009. View Article : Google Scholar
|
|
17
|
Greenburg G and Hay ED: Cytoskeleton and
thyroglobulin expression change during transformation of thyroid
epithelium to mesenchyme-like cells. Development. 102:605–622.
1988.PubMed/NCBI
|
|
18
|
Thiery JP, Acloque H, Huang RY and Nieto
MA: Epithelial-mesenchymal transitions in development and disease.
Cell. 139:871–890. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Kiesslich T, Pichler M and Neureiter D:
Epigenetic control of epithelial-mesenchymal-transition in human
cancer. Mol Clin Oncol. 1:3–11. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Radisky DC, Kenny PA and Bissell MJ:
Fibrosis and cancer: Do myofibroblasts come also from epithelial
cells via EMT? J Cell Biochem. 10:830–839. 2007. View Article : Google Scholar
|
|
21
|
Zeisberg EM, Tarnavski O, Zeisberg M,
Dorfman AL, McMullen JR, Gustafsson E, Chandraker A, Yuan X, Pu WT,
Roberts AB, et al: Endothelial-to-mesenchymal transition
contributes to cardiac fibrosis. Nat Med. 13:952–961. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Zeisberg EM, Potenta SE, Sugimoto H,
Zeisberg M and Kalluri R: Fibroblasts in kidney fibrosis emerge via
endothelial-to- mesenchymal transition. J Am Soc Nephrol.
19:2282–2287. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Strippoli R, Benedicto I, Pérez Lozano ML,
Cerezo A, López-Cabrera M and del Pozo MA:
Epithelial-to-mesenchymal transition of peritoneal mesothelial
cells is regulated by an ERK/NF-kappaB/Snail1 pathway. Dis Model
Mech. 1:264–274. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Boutet A, De Frutos CA, Maxwell PH, Mayol
MJ, Romero J and Nieto MA: Snail activation disrupts tissue
homeostasis and induces fibrosis in the adult kidney. EMBO J.
25:5603–5613. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Hanahan D and Weinberg RA: Hallmarks of
cancer: The next generation. Cell. 144:646–674. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Yeung KT and Yang J:
Epithelial-mesenchymal transition in tumor metastasis. Mol Oncol.
11:28–39. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Derksen PW, Liu X, Saridin F, van der
Gulden H, Zevenhoven J, Evers B, van Beijnum JR, Griffioen AW, Vink
J, Krimpenfort P, et al: Somatic inactivation of E-cadherin and p53
in mice leads to metastatic lobular mammary carcinoma through
induction of anoikis resistance and angiogenesis. Cancer Cell.
10:437–449. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Tsai JH and Yang J: Epithelial-mesenchymal
plasticity in carcinoma metastasis. Genes Dev. 27:2192–2206. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Jouppila-Mättö A, Tuhkanen H, Soini Y,
Pukkila M, Närkiö-Mäkelä M, Sironen R, Virtanen I, Mannermaa A and
Kosma VM: Transcription factor snail1 expression and poor survival
in pharyngeal squamous cell carcinoma. Histol Histopathol.
26:443–439. 2011.PubMed/NCBI
|
|
30
|
Francí C, Gallén M, Alameda F, Baró T,
Iglesias M, Virtanen I and García de Herreros A: Snail1 protein in
the stroma as a new putative prognosis marker for colon tumours.
PLoS One. 4:e55952009. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Bièche I, Lerebours F, Tozlu S, Espie M,
Marty M and Lidereau R: Molecular profiling of inflammatory breast
cancer: Identification of a poor-prognosis gene expression
signature. Clin Cancer Res. 10:6789–6795. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Sarkar FH, Li Y, Wang Z and Kong D:
Pancreatic cancer stem cells and EMT in drug resistance and
metastasis. Minerva Chir. 64:489–500. 2009.PubMed/NCBI
|
|
33
|
van Zijl F, Zulehner G, Petz M, Schneller
D, Kornauth C, Hau M, Machat G, Grubinger M, Huber H and Mikulits
W: Epithelial-mesenchymal transition in hepatocellular carcinoma.
Future Oncol. 5:1169–1179. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Halasova E, Adamkov M, Matakova T, Kavcova
E, Poliacek I and Singliar A: Lung cancer incidence and survival in
chromium exposed individuals with respect to expression of
anti-apoptotic protein survivin and tumor suppressor P53 protein.
Eur J Med Res. 15(Suppl 2): S55–S59. 2010.
|
|
35
|
Heuberger J and Birchmeier W: Interplay of
cadherin-mediated cell adhesion and canonical Wnt signaling. Cold
Spring Harb Perspect Biol. 2:a0029152010. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Katsuno Y, Lamouille S and Derynck R:
TGF-β signaling and epithelial-mesenchymal transition in cancer
progression. Curr Opin Oncol. 25:76–84. 2013. View Article : Google Scholar
|
|
37
|
Akhurst RJ and Padgett RW: Matters of
context guide future research in TGFβ superfamily signaling. Sci
Signal. 8:re102015. View Article : Google Scholar
|
|
38
|
Zavadil J and Böttinger EP: TGF-beta and
epithelial-to- mesenchymal transitions. Oncogene. 24:5764–5774.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Nawshad A, Lagamba D, Polad A and Hay ED:
Transforming growth factor-beta signaling during
epithelial-mesenchymal transformation: Implications for
embryogenesis and tumor metastasis. Cells Tissues Organs.
179:11–23. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Ricciardi M, Zanotto M, Malpeli G, Bassi
G, Perbellini O, Chilosi M, Bifari F and Krampera M:
Epithelial-to-mesenchymal transition (EMT) induced by inflammatory
priming elicits mesenchymal stromal cell-like immune-modulatory
properties in cancer cells. Br J Cancer. 112:1067–1075. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Wei SC, Fattet L, Tsai JH, Guo Y, Pai VH,
Majeski HE, Chen AC, Sah RL, Taylor SS, Engler AJ and Yang J:
Matrix stiffness drives epithelial-mesenchymal transition and
tumour metastasis through a TWIST1-G3BP2 mechanotransduction
pathway. Nat Cell Biol. 17:678–688. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Roberts AB and Wakefield LM: The two faces
of transforming growth factor beta in carcinogenesis. Proc Natl
Acad Sci USA. 100:8621–8623. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Cano A, Pérez-Moreno MA, Rodrigo I,
Locascio A, Blanco MJ, del Barrio MG, Portillo F and Nieto MA: The
transcription factor snail controls epithelial-mesenchymal
transitions by repressing E-cadherin expression. Nat Cell Biol.
2:76–83. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Peinado H, Olmeda D and Cano A: Snail, Zeb
and bHLH factors in tumour progression: An alliance against the
epithelial phenotype? Nat Rev Cancer. 7:415–428. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Grille SJ, Bellacosa A, Upson J,
Klein-Szanto AJ, van Roy F, Lee-Kwon W, Donowitz M, Tsichlis PN and
Larue L: The protein kinase Akt induces epithelial mesenchymal
transition and promotes enhanced motility and invasiveness of
squamous cell carcinoma lines. Cancer Res. 63:2172–2178.
2003.PubMed/NCBI
|
|
46
|
Zhang L, Huang G, Li X, Zhang Y, Jiang Y,
Shen J, Liu J, Wang Q, Zhu J, Feng X, et al: Hypoxia induces
epithelial-mesenchymal transition via activation of SNAI1 by
hypoxia-inducible factor-1α in hepatocellular carcinoma. BMC
Cancer. 13:1082013. View Article : Google Scholar
|
|
47
|
Park SM, Gaur AB, Lengyel E and Peter ME:
The miR-200 family determines the epithelial phenotype of cancer
cells by targeting the E-cadherin repressors ZEB1 and ZEB2. Genes
Dev. 22:894–907. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Long J, Zuo D and Park M: Pc2-mediated
sumoylation of Smad-interacting protein 1 attenuates
transcriptional repression of E-cadherin. J Biol Chem.
280:35477–35489. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Xu J, Lamouille S and Derynck R:
TGF-beta-induced epithelial to mesenchymal transition. Cell Res.
19:156–172. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Bax NA, Pijnappels DA, van Oorschot AA,
Winter EM, de Vries AA, van Tuyn J, Braun J, Maas S, Schalij MJ,
Atsma DE, et al: Epithelial-to-mesenchymal transformation alters
electrical conductivity of human epicardial cells. J Cell Mol Med.
15:2675–2683. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Lamouille S, Xu J and Derynck R: Molecular
mechanisms of epithelial-mesenchymal transition. Nat Rev Mol Cell
Biol. 15:178–196. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Yang MH, Hsu DS, Wang HW, Wang HJ, Lan HY,
Yang WH, Huang CH, Kao SY, Tzeng CH, Tai SK, et al: Bmi1 is
essential in Twist1-induced epithelial-mesenchymal transition. Nat
Cell Biol. 12:982–992. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Li CW, Xia W, Huo L, Lim SO, Wu Y, Hsu JL,
Chao CH, Yamaguchi H, Yang NK, Ding Q, et al:
Epithelial-mesenchymal transition induced by TNF-α requires
NF-κB-mediated transcriptional upregulation of Twist1. Cancer Res.
72:1290–1300. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Cheng GZ, Zhang WZ, Sun M, Wang Q, Coppola
D, Mansour M, Xu LM, Costanzo C, Cheng JQ and Wang LH: Twist is
transcriptionally induced by activation of STAT3 and mediates STAT3
oncogenic function. J Biol Chem. 283:14665–14673. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Yang MH, Wu MZ, Chiou SH, Chen PM, Chang
SY, Liu CJ, Teng SC and Wu KJ: Direct regulation of TWIST by
HIF-1alpha promotes metastasis. Nat Cell Biol. 10:295–305. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Diepenbruck M and Christofori G:
Epithelial-mesenchymal transition (EMT) and metastasis: Yes, no,
maybe? Curr Opin Cell Biol. 43:7–13. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Porta-de-la-Riva M, Stanisavljevic J,
Curto J, Francí C, Díaz VM, García de Herreros A and Baulida J:
TFCP2c/LSF/LBP-1c is required for Snail1-induced fibronectin gene
expression. Biochem J. 435:563–568. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Kuo YC, Su CH, Liu CY, Chen TH, Chen CP
and Wang HS: Transforming growth factor-beta induces CD44 cleavage
that promotes migration of MDA-MB-435s cells through the
up-regulation of membrane type 1-matrix metalloproteinase. Int J
Cancer. 124:2568–2576. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Beaty BT and Condeelis J: Digging a little
deeper: The stages of invadopodium formation and maturation. Eur J
Cell Biol. 93:438–444. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
David JM and Rajasekaran AK: Dishonorable
discharge: The oncogenic roles of cleaved E-cadherin fragments.
Cancer Res. 72:2917–2923. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Kalluri R: EMT: When epithelial cells
decide to become mesenchymal-like cells. J Clin Invest.
119:1417–1419. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Osta WA, Chen Y, Mikhitarian K, Mitas M,
Salem M, Hannun YA, Cole DJ and Gillanders WE: EpCAM is
overexpressed in breast cancer and is a potential target for breast
cancer gene therapy. Cancer Res. 64:5818–5824. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Wu S, Liu S, Liu Z, Huang J, Pu X, Li J,
Yang D, Deng H, Yang N and Xu J: Classification of circulating
tumor cells by epithelial-mesenchymal transition markers. PLoS One.
10:e01239762015. View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Tam WL and Weinberg RA: The epigenetics of
epithelial- mesenchymal plasticity in cancer. Nat Med.
19:1438–1449. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Yoshikawa M, Hishikawa K, Marumo T and
Fujita T: Inhibition of histone deacetylase activity suppresses
epithelial-to- mesenchymal transition induced by TGF-beta1 in human
renal epithelial cells. J Am Soc Nephrol. 18:58–65. 2007.
View Article : Google Scholar
|
|
66
|
Bullock MD, Sayan AE, Packham GK and
Mirnezami AH: MicroRNAs: Critical regulators of epithelial to
mesenchymal (EMT) and mesenchymal to epithelial transition (MET) in
cancer progression. Biol Cell. 104:3–12. 2012. View Article : Google Scholar
|
|
67
|
Luo M, Li Z, Wang W, Zeng Y, Liu Z and Qiu
J: Long non-coding RNA H19 increases bladder cancer metastasis by
associating with EZH2 and inhibiting E-cadherin expression. Cancer
Lett. 333:213–221. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
68
|
Neureiter D, Zopf S, Leu T, Dietze O,
Hauser-Kronberger C, Hahn EG, Herold C and Ocker M: Apoptosis,
proliferation and differentiation patterns are influenced by
Zebularine and SAHA in pancreatic cancer models. Scand J
Gastroenterol. 42:103–116. 2007. View Article : Google Scholar
|
|
69
|
Richterová R, Jurečeková J, Evinová A,
Kolarovszki B, Benčo M, De Riggo J, Sutovský J, Mahmood S, Račay P
and Dobrota D: Most frequent molecular and immunohistochemical
markers present in selected types of brain tumors. Gen Physiol
Biophys. 33:259–279. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Caplakova V, Babusikova E, Blahovcova E,
Balharek T, Zelieskova M and Hatok J: DNA methylation machinery in
the endometrium and endometrial cancer. Anticancer Res.
36:4407–4420. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
71
|
Bolden JE, Peart MJ and Johnstone RW:
Anticancer activities of histone deacetylase inhibitors. Nat Rev
Drug Discov. 5:769–784. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
72
|
Lapinska K, Housman G, Byler S, Heerboth
S, Willbanks A, Oza A and Sarkar S: The effects of histone
deacetylase inhibitor and calpain inhibitor combination therapies
on ovarian cancer cells. Anticancer Res. 36:5731–5742. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
73
|
Blahovcová E, Škovierová H, Strnádel J,
Mištuna D and Halašová E: Apoptosis in cancer cells. Information
technologies in medicine. Advances in intelligent systems and
computing. Piętka E, Badura P, Kawa J and Wieclawek W: 472.
Springer; Cham: pp. 207–213. 2016
|
|
74
|
Shapiro IM, Cheng AW, Flytzanis NC,
Balsamo M, Condeelis JS, Oktay MH, Burge CB and Gertler FB: An
EMT-driven alternative splicing program occurs in human breast
cancer and modulates cellular phenotype. PLoS Genet.
7:e10022182011. View Article : Google Scholar : PubMed/NCBI
|
|
75
|
Weidmann MD, Surve CR, Eddy RJ, Chen X,
Gertler FB, Sharma VP and Condeelis JS: MenaINV dysregulates
cortactin phosphorylation to promote invadopodium maturation. Sci
Rep. 6:361422016. View Article : Google Scholar :
|
|
76
|
Brown RL, Reinke LM, Damerow MS, Perez D,
Chodosh LA, Yang J and Cheng C: CD44 splice isoform switching in
human and mouse epithelium is essential for epithelial-mesenchymal
transition and breast cancer progression. J Clin Invest.
121:1064–1074. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
77
|
Stefani G and Slack FJ: Small non-coding
RNAs in animal development. Nat Rev Mol Cell Biol. 9:219–230. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
78
|
Brabletz S and Brabletz T: The ZEB/miR-200
feedback loop-a motor of cellular plasticity in development and
cancer? EMBO Rep. 11:670–677. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
79
|
Burk U, Schubert J, Wellner U, Schmalhofer
O, Vincan E, Spaderna S and Brabletz T: A reciprocal repression
between ZEB1 and members of the miR-200 family promotes EMT and
invasion in cancer cells. EMBO Rep. 9:582–589. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
80
|
Bracken CP, Li X, Wright JA, Lawrence DM,
Pillman KA, Salmanidis M, Anderson MA, Dredge BK, Gregory PA,
Tsykin A, et al: Genome-wide identification of miR-200 targets
reveals a regulatory network controlling cell invasion. EMBO J.
33:2040–2056. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
81
|
Chang CJ, Chao CH, Xia W, Yang JY, Xiong
Y, Li CW, Yu WH, Rehman SK, Hsu JL, Lee HH, et al: p53 regulates
epithelial-mesenchymal transition and stem cell properties through
modulating miRNAs. Nat Cell Biol. 13:317–323. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
82
|
Brabletz S, Bajdak K, Meidhof S, Burk U,
Niedermann G, Firat E, Wellner U, Dimmler A, Faller G, Schubert J
and Brabletz T: The ZEB1/miR-200 feedback loop controls Notch
signalling in cancer cells. EMBO J. 30:770–782. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
83
|
Adamkov M, Halasova E, Rajcani J, Bencat
M, Vybohova D, Rybarova S and Galbavy S: Relation between
expression pattern of p53 and survivin in cutaneous basal cell
carcinomas. Med Sci Monit. 17:BR74–BR80. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
84
|
Ma L, Young J, Prabhala H, Pan E, Mestdagh
P, Muth D, Teruya-Feldstein J, Reinhardt F, Onder TT, Valastyan S,
et al: miR-9, a MYC/MYCN-activated microRNA, regulates E-cadherin
and cancer metastasis. Nat Cell Biol. 12:247–256. 2010.PubMed/NCBI
|
|
85
|
Ashworth TR: A case of cancer in which
cells similar to those in the tumors were seen in the blood after
death. Australasian Med J. 14:146–149. 1869.
|
|
86
|
Watanabe S: The metastasizability of tumor
cells. Cancer. 7:215–223. 1954. View Article : Google Scholar : PubMed/NCBI
|
|
87
|
Watson MA, Ylagan LR, Trinkaus KM,
Gillanders WE, Naughton MJ, Weilbaecher KN, Fleming TP and Aft RL:
Isolation and molecular profiling of bone marrow micrometastases
identifies TWIST1 as a marker of early tumor relapse in breast
cancer patients. Clin Cancer Res. 13:5001–5009. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
88
|
Nierodzik ML, Plotkin A, Kajumo F and
Karpatkin S: Thrombin stimulates tumor-platelet adhesion in vitro
and metastasis in vivo. J Clin Invest. 87:229–236. 1991. View Article : Google Scholar : PubMed/NCBI
|
|
89
|
Kopp HG, Placke T and Salih HR:
Platelet-derived transforming growth factor-beta down-regulates
NKG2D thereby inhibiting natural killer cell antitumor reactivity.
Cancer Res. 69:7775–7783. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
90
|
Dasgupta A, Lim AR and Ghajar CM:
Circulating and disseminated tumor cells: Harbingers or initiators
of metastasis? Mol Oncol. 11:40–61. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
91
|
Ao Z, Shah SH, Machlin LM, Parajuli R,
Miller PC, Rawal S, Williams AJ, Cote RJ, Lippman ME, Datar RH and
El-Ashry D: Identification of cancer-associated fibroblasts in
circulating blood from patients with metastatic breast cancer.
Cancer Res. 75:4681–4687. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
92
|
Upreti M, Jamshidi-Parsian A, Koonce NA,
Webber JS, Sharma SK, Asea AA, Mader MJ and Griffin RJ:
Tumor-endothelial cell three-dimensional spheroids: New aspects to
enhance radiation and drug therapeutics. Transl Oncol. 4:365–376.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
93
|
Sharma D, Brummel-Ziedins KE, Bouchard BA
and Holmes CE: Platelets in tumor progression: A host factor that
offers multiple potential targets in the treatment of cancer. J
Cell Physiol. 229:1005–1015. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
94
|
Cristofanilli M, Budd GT, Ellis MJ,
Stopeck A, Matera J, Miller MC, Reuben JM, Doyle GV, Allard WJ,
Terstappen LW and Hayes DF: Circulating tumor cells, disease
progression, and survival in metastatic breast cancer. N Engl J
Med. 351:781–791. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
95
|
Goldkorn A, Ely B, Quinn DI, Tangen CM,
Fink LM, Xu T, Twardowski P, Van Veldhuizen PJ, Agarwal N, Carducci
MA, et al: Circulating tumor cell counts are prognostic of overall
survival in SWOG S0421: A phase III trial of docetaxel with or
without atrasentan for metastatic castration-resistant prostate
cancer. J Clin Oncol. 32:1136–1142. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
96
|
Mego M, Cierna Z, Janega P, Karaba M,
Minarik G, Benca J, Sedlácková T, Sieberova G, Gronesova P,
Manasova D, et al: Relationship between circulating tumor cells and
epithelial to mesenchymal transition in early breast cancer. BMC
Cancer. 15:5332015. View Article : Google Scholar : PubMed/NCBI
|
|
97
|
Kasimir-Bauer S, Hoffmann O, Wallwiener D,
Kimmig R and Fehm T: Expression of stem cell and
epithelial-mesenchymal transition markers in primary breast cancer
patients with circulating tumor cells. Breast Cancer Res.
14:R152012. View Article : Google Scholar : PubMed/NCBI
|
|
98
|
Weismann P, Weismanova E, Masak L, Mlada
K, Keder D, Ferancikova Z, Vizvaryova M, Konecny M, Zavodna K,
Kausitz J, et al: The detection of circulating tumor cells
expressing E6/E7 HR-HPV oncogenes in peripheral blood in cervical
cancer patients after radical hysterectomy. Neoplasma. 56:230–238.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
99
|
Mego M, Gao H, Lee BN, Cohen EN, Tin S,
Giordano A, Wu Q, Liu P, Nieto Y, Champlin RE, et al: Prognostic
value of EMT-circulating tumor cells in metastatic breast cancer
patients undergoing high-dose chemotherapy with autologous
hematopoietic stem cell transplantation. J Cancer. 3:369–380. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
100
|
Li YM, Xu SC, Li J, Han KQ, Pi HF, Zheng
L, Zuo GH, Huang XB, Li HY, Zhao HZ, et al: Epithelial-mesenchymal
transition markers expressed in circulating tumor cells in
hepatocellular carcinoma patients with different stages of disease.
Cell Death Dis. 4:e8312013. View Article : Google Scholar : PubMed/NCBI
|
|
101
|
Jansson S, Bendahl PO, Larsson AM,
Aaltonen KE and Rydén L: Prognostic impact of circulating tumor
cell apoptosis and clusters in serial blood samples from patients
with metastatic breast cancer in a prospective observational
cohort. BMC Cancer. 16:4332016. View Article : Google Scholar : PubMed/NCBI
|
|
102
|
Chang MC, Chang YT, Chen JY, Jeng YM, Yang
CY, Tien YW, Yang SH, Chen HL, Liang TY, Wang CF, et al: Clinical
significance of circulating tumor microemboli as a prognostic
marker in patients with pancreatic ductal adenocarcinoma. Clin
Chem. 62:505–513. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
103
|
Zhao Q, Barclay M, Hilkens J, Guo X,
Barrow H, Rhodes JM and Yu LG: Interaction between circulating
galectin-3 and cancer-associated MUC1 enhances tumour cell
homotypic aggregation and prevents anoikis. Mol Cancer. 9:1542010.
View Article : Google Scholar : PubMed/NCBI
|
|
104
|
Cheung KJ, Padmanaban V, Silvestri V,
Schipper K, Cohen JD, Fairchild AN, Gorin MA, Verdone JE, Pienta
KJ, Bader JS and Ewald AJ: Polyclonal breast cancer metastases
arise from collective dissemination of keratin 14-expressing tumor
cell clusters. Proc Natl Acad Sci USA. 113:E854–E863. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
105
|
Au SH, Storey BD, Moore JC, Tang Q, Chen
YL, Javaid S, Sarioglu AF, Sullivan R, Madden MW, O’Keefe R, et al:
Clusters of circulating tumor cells traverse capillary-sized
vessels. Proc Natl Acad Sci USA. 113:4947–4952. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
106
|
Cima I, Kong SL, Sengupta D, Tan IB, Phyo
WM, Lee D, Hu M, Iliescu C, Alexander I, Goh WL, et al:
Tumor-derived circulating endothelial cell clusters in colorectal
cancer. Sci Transl Med. 8:345ra892016. View Article : Google Scholar : PubMed/NCBI
|
|
107
|
Fabisiewicz A and Grzybowska E: CTC
clusters in cancer progression and metastasis. Med Oncol.
34:122017. View Article : Google Scholar
|
|
108
|
Punnoose EA, Atwal SK, Spoerke JM, Savage
H, Pandita A, Yeh RF, Pirzkall A, Fine BM, Amler LC, Chen DS and
Lackner MR: Molecular biomarker analyses using circulating tumor
cells. PLoS One. 5:e125172010. View Article : Google Scholar : PubMed/NCBI
|
|
109
|
Zhang L, Ridgway LD, Wetzel MD, Ngo J, Yin
W, Kumar D, Goodman JC, Groves MD and Marchetti D: The
identification and characterization of breast cancer CTCs competent
for brain metastasis. Sci Transl Med. 5:180ra482013. View Article : Google Scholar : PubMed/NCBI
|
|
110
|
Chéry L, Lam HM, Coleman I, Lakely B,
Coleman R, Larson S, Aguirre-Ghiso JA, Xia J, Gulati R, Nelson PS,
et al: Characterization of single disseminated prostate cancer
cells reveals tumor cell heterogeneity and identifies dormancy
associated pathways. Oncotarget. 5:9939–9951. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
111
|
Yu M, Bardia A, Wittner BS, Stott SL, Smas
ME, Ting DT, Isakoff SJ, Ciciliano JC, Wells MN, Shah AM, et al:
Circulating breast tumor cells exhibit dynamic changes in
epithelial and mesenchymal composition. Science. 339:580–584. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
112
|
Harouaka R, Kang Z, Zheng SY and Cao L:
Circulating tumor cells: Advances in isolation and analysis, and
challenges for clinical applications. Pharmacol Ther. 141:209–221.
2014. View Article : Google Scholar :
|
|
113
|
Beije N, Jager A and Sleijfer S:
Circulating tumor cell enumeration by the CellSearch system: The
clinician’s guide to breast cancer treatment? Cancer Treat Rev.
41:144–150. 2015. View Article : Google Scholar
|
|
114
|
Freidin MB, Tay A, Freydina DV, Chudasama
D, Nicholson AG, Rice A, Anikin V and Lim E: An assessment of
diagnostic performance of a filter-based antibody-independent
peripheral blood circulating tumour cell capture paired with
cytomorphologic criteria for the diagnosis of cancer. Lung Cancer.
85:182–185. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
115
|
Warkiani ME, Khoo BL, Wu L, Tay AK, Bhagat
AA, Han J and Lim CT: Ultra-fast, label-free isolation of
circulating tumor cells from blood using spiral microfluidics. Nat
Protoc. 11:134–148. 2016. View Article : Google Scholar
|
|
116
|
Saucedo-Zeni N, Mewes S, Niestroj R,
Gasiorowski L, Murawa D, Nowaczyk P, Tomasi T, Weber E, Dworacki G,
Morgenthaler NG, et al: A novel method for the in vivo isolation of
circulating tumor cells from peripheral blood of cancer patients
using a functionalized and structured medical wire. Int J Oncol.
41:1241–1250. 2012.PubMed/NCBI
|
|
117
|
Mani SA, Guo W, Liao MJ, Eaton EN, Ayyanan
A, Zhou AY, Brooks M, Reinhard F, Zhang CC, Shipitsin M, et al: The
epithelial-mesenchymal transition generates cells with properties
of stem cells. Cell. 133:704–715. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
118
|
Jolly MK, Jia D, Boareto M, Mani SA,
Pienta KJ, Ben-Jacob E and Levine H: Coupling the modules of EMT
and stemness: A tunable ‘stemness window’ model. Oncotarget.
6:25161–25174. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
119
|
Jolly MK, Tripathi SC, Jia D, Mooney SM,
Celiktas M, Hanash SM, Mani SA, Pienta KJ, Ben-Jacob E and Levine
H: Stability of the hybrid epithelial/mesenchymal phenotype.
Oncotarget. 7:27067–27084. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
120
|
Damaskos C, Garmpis N, Valsami S, Kontos
M, Spartalis E, Kalampokas T, Kalampokas E, Athanasiou A, Moris D,
Daskalopoulou A, et al: Histone deacetylase inhibitors: An
attractive therapeutic strategy against breast cancer. Anticancer
Res. 37:35–46. 2017. View Article : Google Scholar
|
|
121
|
Škovierová H, Vidomanová E, Mahmood S,
Sopková J, Drgová A, Červeňová T, Halašová E and Lehotský J: The
molecular and cellular effect of homocysteine metabolism imbalance
on human health. Int J Mol Sci. 17:17332016. View Article : Google Scholar :
|
|
122
|
Stintzing S, Kemmerling R, Kiesslich T,
Alinger B, Ocker M and Neureiter D: Myelodysplastic syndrome and
histone deacetylase inhibitors: ‘To be or not to be acetylated’? J
Biomed Biotechnol. 2011:2141432011. View Article : Google Scholar
|
|
123
|
Li A, Liu Z, Li M, Zhou S, Xu Y, Xiao Y
and Yang W: HDAC5, a potential therapeutic target and prognostic
biomarker, promotes proliferation, invasion and migration in human
breast cancer. Oncotarget. 7:37966–37978. 2016.PubMed/NCBI
|
|
124
|
Stojanovic N, Hassan Z, Wirth M, Wenzel P,
Beyer M, Schäfer C, Brand P, Kroemer A, Stauber RH, Schmid RM, et
al: HDAC1 and HDAC2 integrate the expression of p53 mutants in
pancreatic cancer. Oncogene. 36:1804–1815. 2017. View Article : Google Scholar
|
|
125
|
Mann BS, Johnson JR, He K, Sridhara R,
Abraham S, Booth BP, Verbois L, Morse DE, Jee JM, Pope S, et al:
Vorinostat for treatment of cutaneous manifestations of advanced
primary cutaneous T-cell lymphoma. Clin Cancer Res. 13:2318–2322.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
126
|
Marks PA: Discovery and development of
SAHA as an anticancer agent. Oncogene. 26:1351–1356. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
127
|
Barbarotta L and Hurley K: Romidepsin for
the treatment of peripheral T-Cell lymphoma. J Adv Pract Oncol.
6:22–36. 2015.PubMed/NCBI
|
|
128
|
Libby EN, Becker PS, Burwick N, Green DJ,
Holmberg L and Bensinger WI: Panobinostat: A review of trial
results and future prospects in multiple myeloma. Expert Rev
Hematol. 8:9–18. 2015. View Article : Google Scholar
|
|
129
|
Ghajar CM: Metastasis prevention by
targeting the dormant niche. Nat Rev Cancer. 15:238–247. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
130
|
Marcucci F, Stassi G and De Maria R:
Epithelial-mesenchymal transition: A new target in anticancer drug
discovery. Nat Rev Drug Discov. 15:311–325. 2016. View Article : Google Scholar : PubMed/NCBI
|