|
1
|
Mendell JR and Sahenk Z: Clinical
practice. Painful sensory neuropathy. N Engl J Med. 348:1243–1255.
2003. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Sawynok J: Topical and peripherally acting
analgesics. Pharmacol Rev. 55:1–20. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Hansson PT and Dickenson AH:
Pharmacological treatment of peripheral neuropathic pain conditions
based on shared commonalities despite multiple etiologies. Pain.
113:251–254. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Ciaramitaro P, Padua L, Devigili G, Rota
E, Tamburin S, Eleopra R, Cruccu G and Truini A; Neuropathic pain
special interest group of the Italian Neurological Society:
Prevalence of neuropathic pain in patients with traumatic brachial
plexus injury: A multicenter prospective hospital-based study. Pain
Med. March 3–2017.Epub ahead of print. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Anand P and Birch R: Restoration of
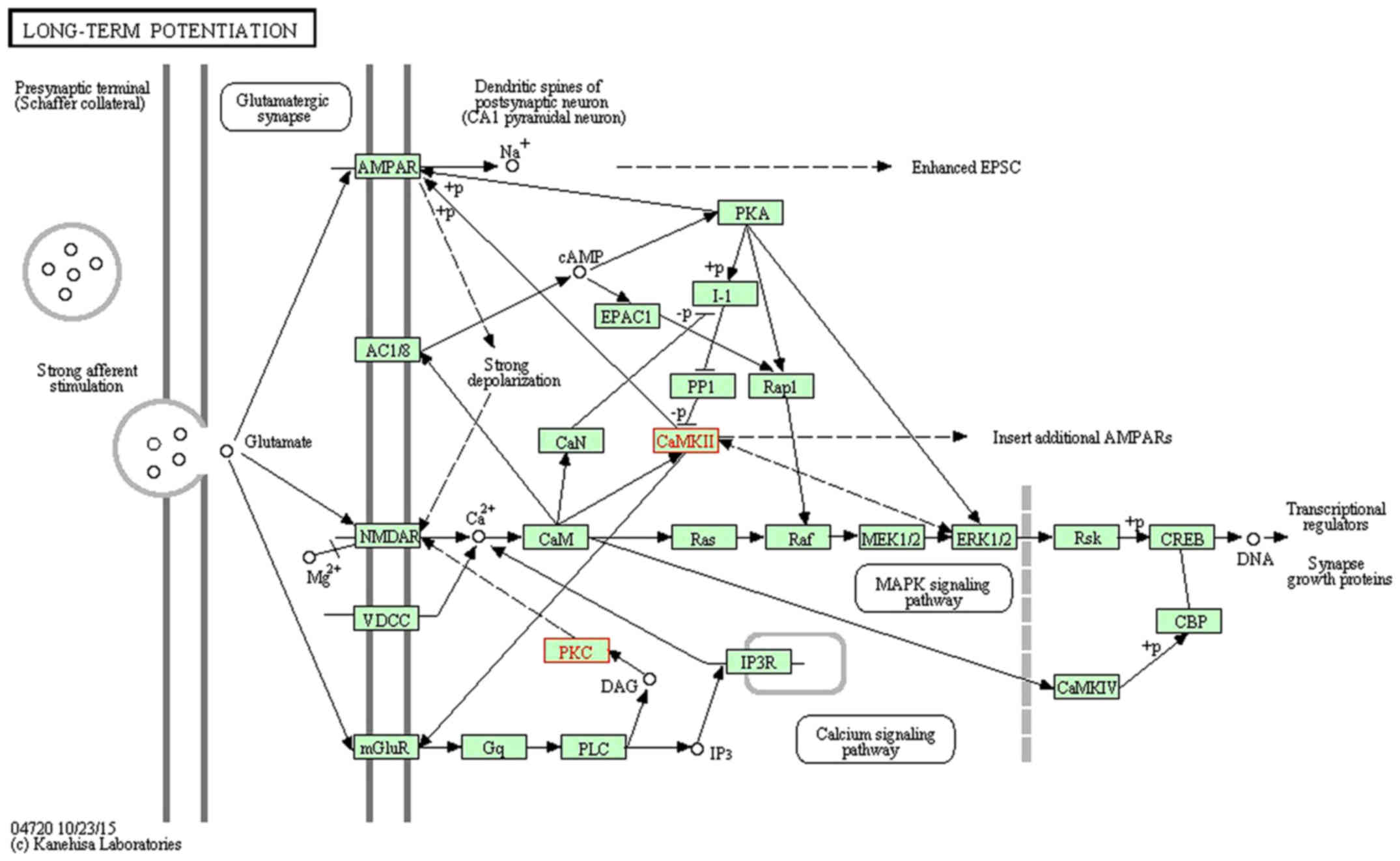
sensory function and lack of long-term chronic pain syndromes after
brachial plexus injury in human neonates. Brain. 125:113–122. 2002.
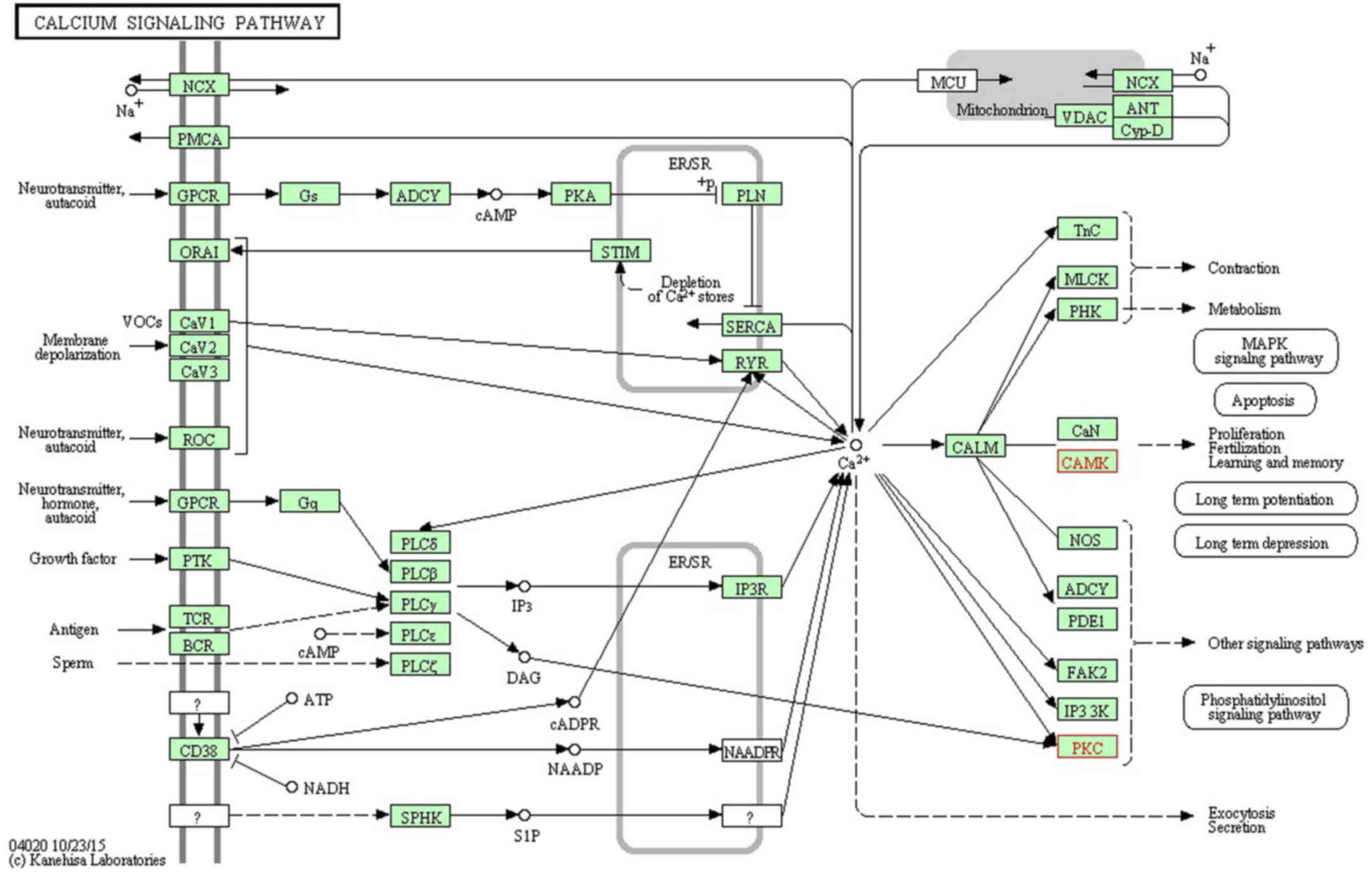
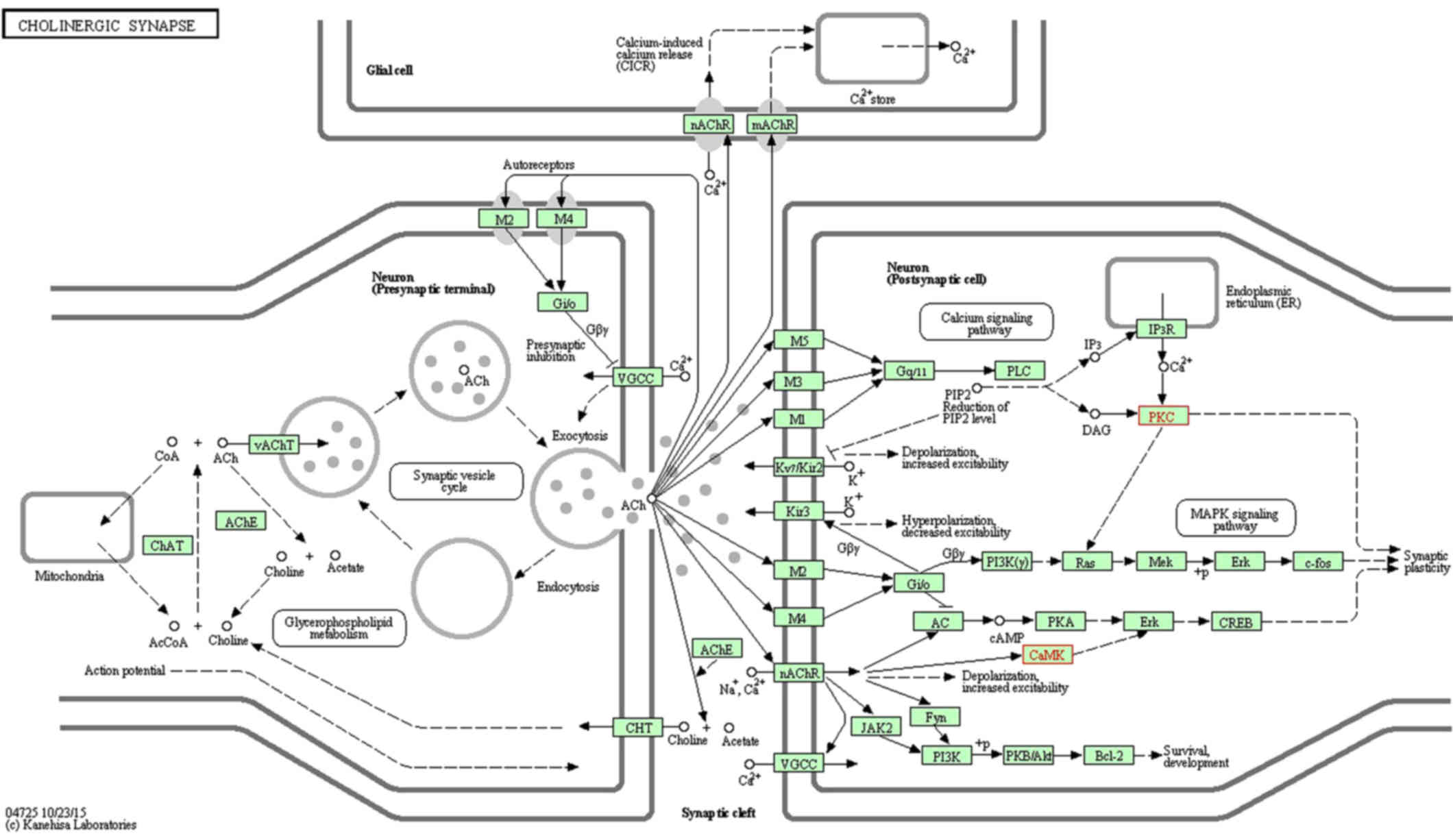
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Berman JS, Birch R and Anand P: Pain
following human brachial plexus injury with spinal cord root
avulsion and the effect of surgery. Pain. 75:199–207. 1998.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Baruah S, Devi BI, Bhat DI and Shukla D:
Drezotomy in the management of post brachial plexus injury
neuropathic pain: Preliminary results. Indian J Neurotrauma.
11:27–29. 2014. View Article : Google Scholar
|
|
8
|
Aley KO and Levine JD: Different
peripheral mechanisms mediate enhanced nociception in
metabolic/toxic and traumatic painful peripheral neuropathies in
the rat. Neuroscience. 111:389–397. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Erichsen HK and Blackburn-Munro G:
Pharmacological characterisation of the spared nerve injury model
of neuropathic pain. Pain. 98:151–161. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Aldrich BT, Frakes EP, Kasuya J, Hammond
DL and Kitamoto T: Changes in expression of sensory organ-specific
microRNAs in rat dorsal root ganglia in association with mechanical
hypersensitivity induced by spinal nerve ligation. Neuroscience.
164:711–723. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Bhalala OG, Srikanth M and Kessler JA: The
emerging roles of microRNAs in CNS injuries. Nat Rev Neurol.
9:328–339. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Erson AE and Petty EM: MicroRNAs in
development and disease. Clin Genet. 74:296–306. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Tan PH, Pao YY, Cheng JK, Hung KC and Liu
CC: MicroRNA-based therapy in pain medicine: Current progress and
future prospects. Acta Anaesthesiol Taiwan. 51:171–176. 2013.
View Article : Google Scholar
|
|
14
|
Parry CB: Pain in avulsion lesions of the
brachial plexus. Pain. 9:41–53. 1980. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Narakas AO: The effects on pain of
reconstructive neurosurgery in 160 patients with traction and/or
crush injury to the brachial plexus. Phantom and Stump Pain.
Siegfried J and Zimmerman M: 1st edition. Springer-Verlag; Berlin:
pp. 126–147. 1981, View Article : Google Scholar
|
|
16
|
Bruxelle J, Travers V and Thiebaut JB:
Occurrence and treatment of pain after brachial plexus injury. Clin
Orthop Relat Res. 237:87–95. 1988.
|
|
17
|
von Schack D, Agostino MJ, Murray BS, Li
Y, Reddy PS, Chen J, Choe SE, Strassle BW, Li C, Bates B, et al:
Dynamic changes in the MicroRNA expression profile reveal multiple
regulatory mechanisms in the spinal nerve ligation model of
neuropathic pain. PLoS One. 6:e176702011. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Choi Y, Yoon YW, Na HS, Kim SH and Chung
JM: Behavioral signs of ongoing pain and cold allodynia in a rat
model of neuropathic pain. Pain. 59:369–376. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Li X, Kroin JS, Kc R, Gibson G, Chen D,
Corbett GT, Pahan K, Fayyaz S, Kim JS, van Wijnen AJ, et al:
Altered spinal microRNA-146a and the microRNA-183 cluster
contribute to osteoarthritic pain in knee joints. J Bone Miner Res.
28:2512–2522. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Wang L, Yuzhou L, Yingjie Z, Jie L and Xin
Z: A new rat model of neuropathic pain: Complete brachial plexus
avulsion. Neurosci Lett. 589:52–56. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2−ΔΔCT method. Methods. 25:402–408. 2001. View Article : Google Scholar
|
|
22
|
Ashburner M, Ball CA, Blake JA, Botstein
D, Butler H, Cherry JM, Davis AP, Dolinski K, Dwight SS, Eppig JT,
et al: The Gene Ontology Consortium: Gene ontology: Tool for the
unification of biology. Nat Genet. 25:25–29. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
The Gene Ontology Consortium: Expansion of
the Gene Ontology knowledgebase and resources. Nucleic Acids Res.
45:D331–D338. 2017. View Article : Google Scholar :
|
|
24
|
Kanehisa M and Goto S: KEGG: Kyoto
Encyclopedia of Genes and Genomes. Nucleic Acids Res. 28:27–30.
2000. View Article : Google Scholar
|
|
25
|
Kanehisa M, Furumichi M, Tanabe M, Sato Y
and Morishima K: KEGG: new perspectives on genomes, pathways,
diseases and drugs. Nucleic Acids Res. 45:D353–D361. 2017.
View Article : Google Scholar :
|
|
26
|
Kanehisa M, Sato Y, Kawashima M, Furumichi
M and Tanabe M: KEGG as a reference resource for gene and protein
annotation. Nucleic Acids Res. 44:D457–D462. 2016. View Article : Google Scholar :
|
|
27
|
Zhang L, Zhang Y and Zhao ZQ: Anterior
cingulate cortex contributes to the descending facilitatory
modulation of pain via dorsal reticular nucleus. Eur J Neurosci.
22:1141–1148. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Wei F, Li P and Zhuo M: Loss of synaptic
depression in mammalian anterior cingulate cortex after amputation.
J Neurosci. 19:9346–9354. 1999.PubMed/NCBI
|
|
29
|
Porreca F, Ossipov MH and Gebhart GF:
Chronic pain and medullary descending facilitation. Trends
Neurosci. 25:319–325. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Robinson D, Calejesan AA and Zhuo M:
Long-lasting changes in rostral ventral medulla neuronal activity
after inflammation. J Pain. 3:292–300. 2002. View Article : Google Scholar
|
|
31
|
Vogt BA: Pain and emotion interactions in
subregions of the cingulate gyrus. Nat Rev Neurosci. 6:533–544.
2005. View
Article : Google Scholar : PubMed/NCBI
|
|
32
|
Zhuo M: Cortical excitation and chronic
pain. Trends Neurosci. 31:199–207. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Wei F and Zhuo M: Potentiation of sensory
responses in the anterior cingulate cortex following digit
amputation in the anaesthetised rat. J Physiol. 532:823–833. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Xu H, Wu LJ, Wang H, Zhang X, Vadakkan KI,
Kim SS, Steenland HW and Zhuo M: Presynaptic and postsynaptic
amplifications of neuropathic pain in the anterior cingulate
cortex. J Neurosci. 28:7445–7453. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Waggener CT, Dupree JL, Elgersma Y and
Fuss B: CaMKIIβ regulates oligodendrocyte maturation and CNS
myelination. J Neurosci. 33:10453–10458. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Fang M, Feng C, Zhao YX and Liu XY: Camk2b
protects neurons from homocysteine-induced apoptosis with the
involvement of HIF-1α signal pathway. Int J Clin Exp Med.
7:1659–1668. 2014.
|
|
37
|
Miletic G, Hermes JL, Bosscher GL, Meier
BM and Miletic V: Protein kinase C gamma-mediated phosphorylation
of GluA1 in the postsynaptic density of spinal dorsal horn neurons
accompanies neuropathic pain, and dephosphorylation by calcineurin
is associated with prolonged analgesia. Pain. 156:2514–2520. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Yeh CY, Chung SC, Tseng FL, Tsai YC and
Liu YC: Biphasic effects of chronic intrathecal gabapentin
administration on the expression of protein kinase C gamma in the
spinal cord of neuropathic pain rats. Acta Anaesthesiol Taiwan.
49:144–148. 2011. View Article : Google Scholar
|
|
39
|
Kim CH, Oh Y, Chung JM and Chung K: The
changes in expression of three subtypes of TTX sensitive sodium
channels in sensory neurons after spinal nerve ligation. Brain Res
Mol Brain Res. 95:153–161. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Ma L, Uchida H, Nagai J, Inoue M, Chun J,
Aoki J and Ueda H: Lysophosphatidic acid-3 receptor-mediated
feed-forward production of lysophosphatidic acid: an initiator of
nerve injury-induced neuropathic pain. Mol Pain. 5:642009.
View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Campbell JN and Meyer RA: Mechanisms of
neuropathic pain. Neuron. 52:77–92. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Nickel FT, Seifert F, Lanz S and Maihöfner
C: Mechanisms of neuropathic pain. Eur Neuropsychopharmacol.
2:81–91. 2012. View Article : Google Scholar
|
|
43
|
Lu Y, Jian MY, Ouyang YB and Han RQ:
Changes in rat brain microRNA expression profiles following
sevoflurane and propofol anesthesia. Chin Med J (Engl).
128:1510–1515. 2015. View Article : Google Scholar
|
|
44
|
McAdams RM, McPherson RJ, Beyer RP,
Bammler TK, Farin FM and Juul SE: Dose-dependent effects of
morphine exposure on mRNA and microRNA (miR) expression in
hippocampus of stressed neonatal mice. PLoS One. 10:e01230472015.
View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Gong Q, Lu Z, Huang Q, Ruan L, Chen J,
Liang Y, Wang H, Yue Y and Feng S: Altered microRNAs expression
profiling in mice with diabetic neuropathic pain. Biochem Biophys
Res Commun. 456:615–620. 2015. View Article : Google Scholar
|
|
46
|
McKiernan RC, Jimenez-Mateos EM, Sano T,
Bray I, Stallings RL, Simon RP and Henshall DC: Expression
profiling the microRNA response to epileptic preconditioning
identifies miR-184 as a modulator of seizure-induced neuronal
death. Exp Neurol. 237:346–354. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Liu C, Teng ZQ, Santistevan NJ, Szulwach
KE, Guo W, Jin P and Zhao X: Epigenetic regulation of miR-184 by
MBD1 governs neural stem cell proliferation and differentiation.
Cell Stem Cell. 6:433–444. 2010. View Article : Google Scholar : PubMed/NCBI
|