Introduction
Glioma, one of the most common primary brain tumors,
presents particularly aggressive activities and highly fatal
prognosis. The malignancy is the characteristic of glioma, which
including rapid tumor growth, metastasis and angiogenesis (1). Although accumulated advanced
techniques contribute to the diagnosis and treatment of glioma, the
median survival is still from 9 to 12 months (2). Given the high incidence,
refractoriness and poor prognosis, novel therapeutic strategies
based on molecular interaction networks must be achieved for glioma
(3).
Fms-related tyrosine kinase 1 (Flt1) is a
full-length tyrosine kinase receptor characterized by extracellular
domain, which is known to be expressed on vessel endothelial cells
and presents a critical point in angiogenesis and subsequent cancer
progression (4). Recent studies
revealed that Flt1 is also widly expressed on tumor cells (5) and the effect of these Flt1 on tumor
progression has been investigated (6–8).
However, the detailed mechanism of Flt1 in glioma progression are
still barely known. Wnt/β-catenin signaling pathway is a
conservative pathway, which has been observed to have a vital role
in many cancers containing glioma (9–12).
The Wnt/β-catenin is involved in tumor growth, metastasis and
angiogenesis, while the activation of Flt1 may be a basis of
Wnt/β-catenin activity in malignant glioma progression.
Recently, microRNAs (miRNAs) have been related to
participation in the biological process of tumor initiation and
progression. These small noncoding single-stranded RNAs serve as
tumor-suppressors or oncogenes by completely or incompletely
binding the 3′ untranslated region (3′UTR) of its target gene to
result in either the degradation of messenger RNA (mRNA) or the
reduction of protein expression in numerous cancers (13). Downregulation of certain miRNAs
correlated with various tumorigenic processes (14). Among these, miR-139-5p has been
validated to be decreased in several tumors (15,16) and restoration of miR-139-5p
contributes to the inhibitory impact on tumor malignancies. To
date, miR-139-5p was identified to be responsible for target genes,
such as NOTCH1, IGF1R, ZEB1, ZEB2 and Mcl-1 (15–18). However, whether Flt1 is drawn into
the epigenetic alteration in miR-139-5p-mediated modulation of
glioma progression remains unclear.
In the present study, we found miR-139-5p was
dysregulated and lead to abrogation of the malignant phenotypes of
glioma cells by directly pairing with the 3′UTR of Flt1.
Additionally, we proved that high expression of Flt1 may be
involved in modulating the malignancy of glioma in vitro.
Furthermore, miR-139-5p suppressed Flt1-mediated Wnt/β-catenin
signaling pathway in the progression of glioma. Together, our
finding may provide a potential supplementary therapeutic strategy
concerning the molecular mechanism of glioma.
Materials and methods
Patient samples and cell culture
Human glioma tissues and normal brain tissues were
obtained from Tianjin Huanhu Hospital, including 6 grade I-II
tumors, 6 grade III tumors, 14 grade IV tumors and 12 normal brain
tissues. Tissues were collected during surgery and immediately
snap-frozen in liquid nitrogen and storing at −80°C. This study was
approved by the hospital institutional review board and written
informed consent was obtained from all the patients.
Human glioma cell lines U87, SNB19, U251, LN308 and
LN229 were all obtained from the Peking Union Medical College cell
library (Beijing, China) which were cultured in complete medium
containing Dulbecco’s modified Eagle’s medium containing 10% fetal
bovine serum (both from Gibco, Los Angeles, CA, USA) and 100 U/ml
penicillin/streptomycin and maintained in a humidified atmosphere
with 5% CO2 at 37°C.
Plasmid construction, oligonucleotides
and cell transfection
Hsa-miR-139-5p mimics, Flt1 siRNA, were both
chemically artificialized by GenePharma (Shanghai, China) as well
as their relative negative control. The sequences are as follows:
miR-139-5p, 5′-UCUACAGUGCACGUGUCUCCAGU-3′; miR-NC,
5′-UUCUCCGAACGUGUCACGUTT-3′; si-Flt1, 5′-GGACGUAACUGAAGAGGAUTT-3′;
si-NC, 5′-UUCUCC GAACGUGUCACGUTT-3′. The cDNA encoding Flt1
(GV230-Flt1) was purchased from Genechem (Shanghai, China). Cell
transfection was with Lipofectamine 2000 (Invitrogen, Carlsbad, CA,
USA) per the manufacturer’s manual.
RNA extraction and qRT-PCR
TRIzol reagent (Invitrogen) was applied to extract
total RNA according to the manufacturer’s instructions. Specific
stem-loop primer and oligod(T) primer were respectively used to
measure the levels of miR-139-5p and Flt1 through PrimeScript RT
reagent kit (Invitrogen) as described previously (19). U6 and β-actin served as an
endogenous control. Then the cDNAs were further amplified by
qRT-PCR using SYBR Premix Ex Taq™ II kit (Takara, Dalian, China)
and the changes were symbolized by relative quantification. The
primers used are shown in Table
I.
 | Table IPrimers used in the polymerase chain
reaction. |
Table I
Primers used in the polymerase chain
reaction.
| Primer names | Primer
sequences |
|---|
| miR-139-5p RT
primer |
5′-GTCGTATCCAGTGCAGGGTCCGAGGTGCACTGGATACGACACTGGAG-3′ |
| U6 RT primer |
5′-GTCGTATCCAGTGCAGGGTCCGAGGTATTCGCACTGGATACGACAAAATATGGAAC-3′ |
| miR-139-5p
forward |
5′-TGCGGTCTACAGTGCACGTGTCTC-3′ |
| U6 forward |
5′-TGCGGGTGCTCGCTTCGGCAGC-3′ |
| U6 reverse |
5′-CCAGTGCAGGGTCCGAGGT-3′ |
| Flt1 forward |
5′-CCACCATCTGAACGTGGTTA-3′ |
| Flt1 reverse |
5′-GCTGCATCCTTGTTGAGAAA-3′ |
| β-actin
forward |
5′-CGTGACATTAAGGAGAAGCTG-3′ |
| β-actin
reverse |
5′-CTAGAAGCATTTGCGGTGGAC-3′ |
Proliferation assay and colony-formation
analysis
The description of
3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT)
assay and colony-formation assay were reported previously (19).
Cell cycle assay via flow cytometry
Flow cytometry for cell cycle assay were described
previously (16). The results
were analyzed with FlowJo (BD Pharmingen, San Diego, CA, USA).
Tumorigenicity in the nude mouse
model
All procedures were forced as the protocols approved
by the Animal Care and Use Committee of Tianjin Medical University.
The U87 subcutaneous model was constructed as mentioned previously
(19). Ten days later the male
nude mice (4 weeks old) were randomly divided into two treatment
groups, which were injected with 400 pmol miR-139-5p or miR-NC
mimics (20 nmol/l×20 μl) in Lipofectamine 2000 into
xenograft model via a multisite injection every 3 days as well as
the measurement of tumor volume [(volume=length x
width2)/2]. The xenograft tumor growth curves were
calculated and the tumors were removed for the immunohistochemical
assay after 18 days of treatment.
Migration and invasion assays
The Transwell assay was carried out with the
chambers (8 μm pore size; Corning, Cambridge, MA, USA)
covered with or without Matrigel (BD Biosciences, San Diego, CA,
USA) respectively. Detail processes of the Transwell assay were
previously described (15).
Vasculogenic mimicry formation assay
Growth factor-reduced Matrigel (50 μl/well;
BD Biosciences) was precoated on the 96-well plate at 37°C for 60
min to solidify, which was seeded with the transfected U87 cell
suspension (2×104/well) and incubated at 37°C for 12 h,
and then was analyzed directly under a microscope. Each closed tube
was numbered.
Luciferase reporter assay
The 3′UTR of Flt1 reporter was created containing
one putative miR-139-5p targeting site and one randomly scrambled
sequence inserted into the pGL3 vector (Promega, Madison, WI, USA).
U87 cells were cotransfected with the wild or mutant plasmid and
miR-139-5p or miR-NC. Following 48 h cultivation, luciferase
activity was determined with the Dual-Luciferase reporter system
(Promega).
Immunocytochemical staining
Immunocytochemical analysis was used to visualize
the expression of Flt1 in glioma tissues, isolated tumors and cells
as described previously (19,20).
Western blot analysis
The detailed process of western blot analysis can be
found in a previous study (19).
The antibodies used were: Flt1, cyclin D1, p21, matrix
metalloproteinase 2 (MMP2), MMP9, E-cadherin, vimentin,
VE-cadherin, Wnt1, β-catenin, GSK-3β, p-GSK-3β and β-actin were all
from Santa Cruz Biotechnology, Inc. (Santa Cruz, CA, USA).
Statistical analysis
Each experiment was executed three times and
quantitative data are shown as mean ± SD and all statistical
analysis were carried out with SPSS 17.0. The significance was
calculated through two-tailed Student’s t-test. P<0.05 was
defined as statistical significance.
Results
miR-139-5p expression is frequently
downregulated in human glioma tissues and cell lines
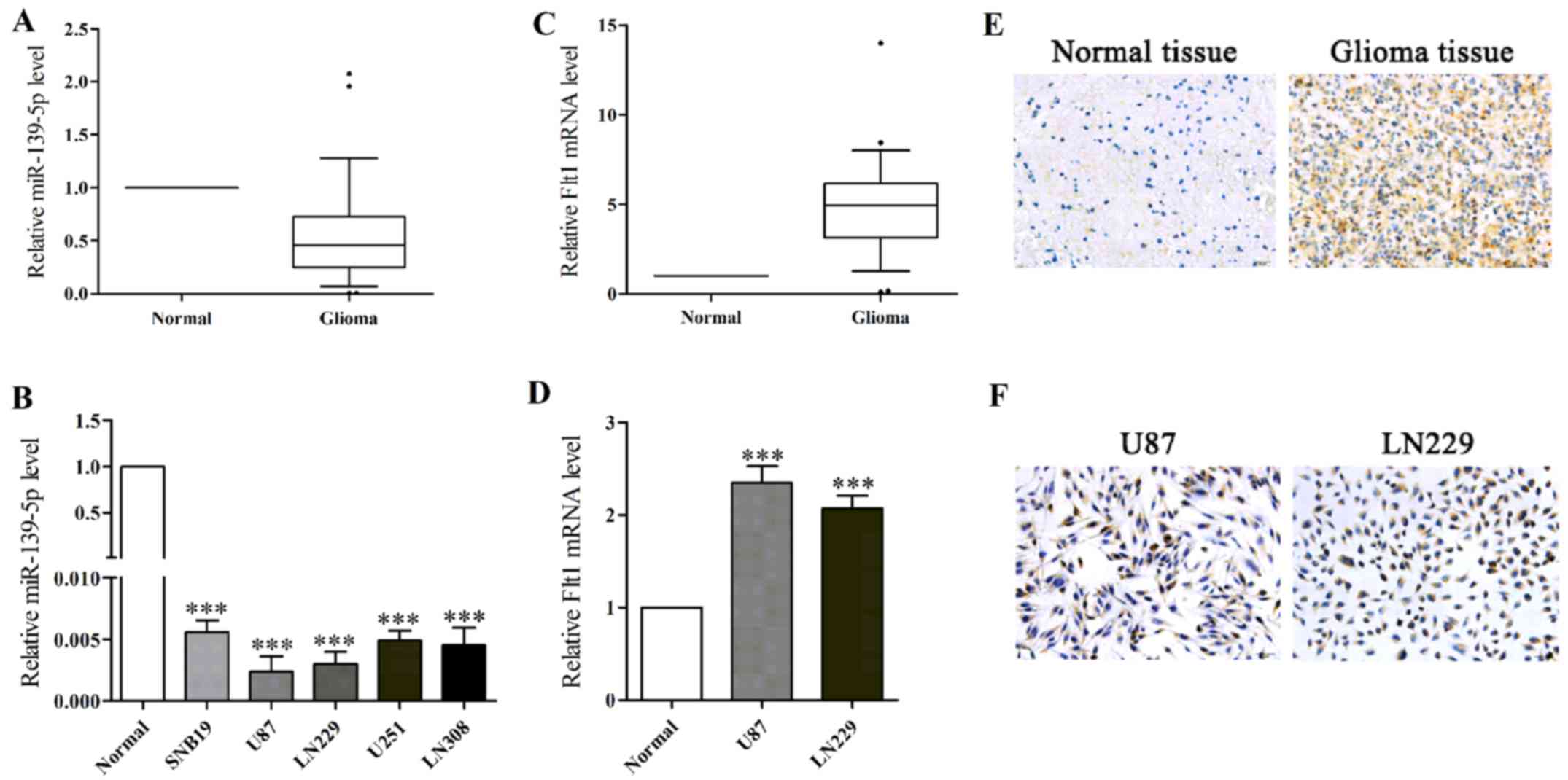
To evaluate the action of miR-139-5p in glioma, we
first analyzed the expression levels of normal brain specimens and
glioma specimens as well as five glioma cell lines via qRT-PCR. The
results showed miR-139-5p expression levels were generally lower in
glioma specimens and glioma cell lines (Fig. 1A and B). According to results, the
two lowest level glioma cells (U87 and LN229 cells) were selected
for the following experiments.
In accordance with previous studies, we found the
average expression of Flt1 mRNA was notably higher in human glioma
tissues and in glioma cell lines (Fig. 1C and D). Moreover, we performed
immunohistochemical examination to observe the expression of Flt1,
and the results showed that Flt1 expression was more commonly
positive in glioma tissues compared normal brain tissues (Fig. 1E). Immunohistochemistry (ICH) was
performed to further locate the expression of Flt1 in U87 and LN229
cells, and as shown in Fig. 1F,
Flt1 was found highly located on cytomembrane and in the cytoplasm.
Then we observed the inverse relevance between the levels of
miR-139-5p and Flt1 based on the data above.
miR-139-5p potently targets Flt1 gene to
decrease its expression
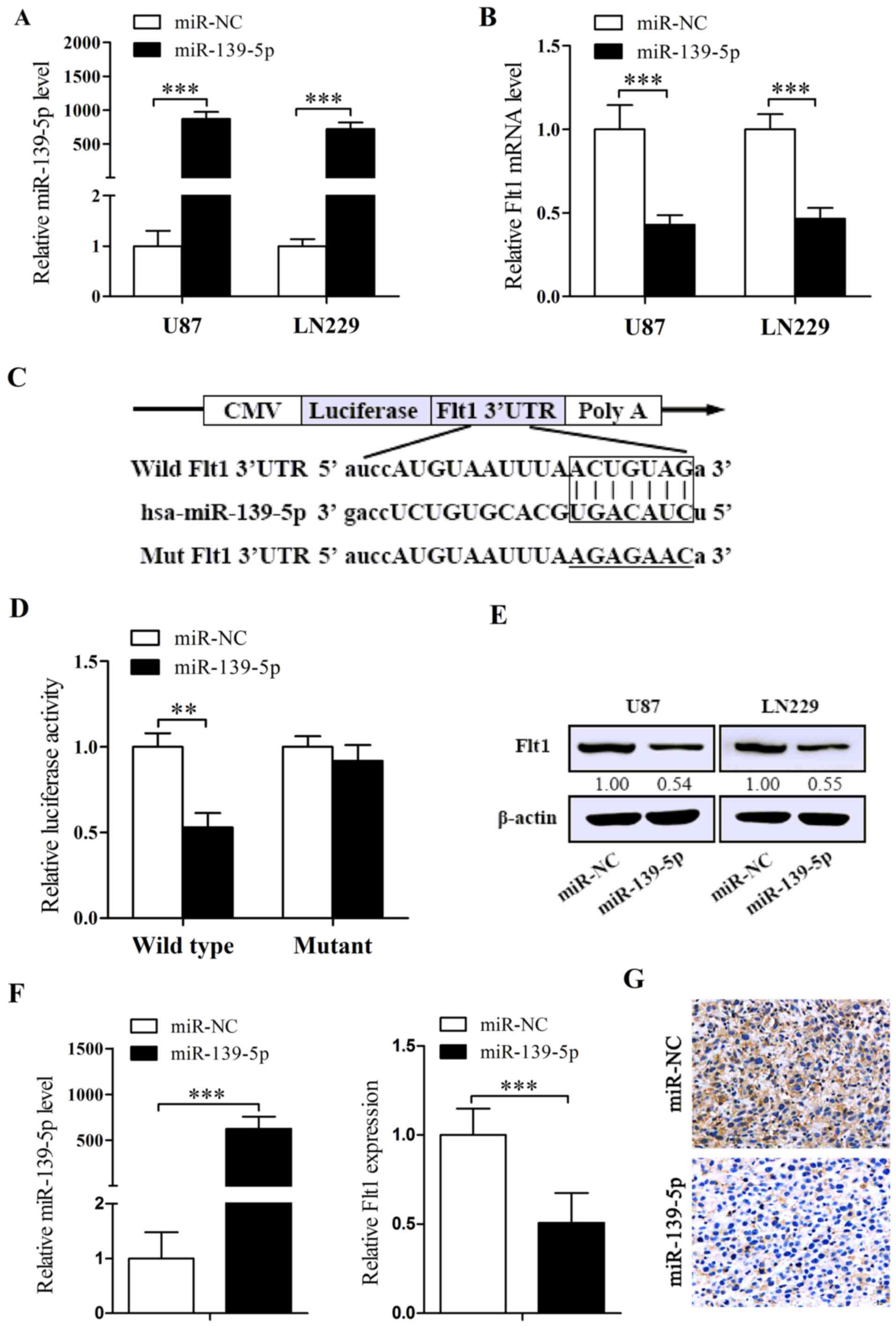
In order to explain whether the lower levels of
endogenous miR-139-5p implies that miR-139-5p is a potential tumor
suppressor, chemosynthesised miR-139-5p mimics were transiently
transfected into U87 and LN229 cells. The efficiency was checked by
qRT-PCR. As revealed in Fig. 2A,
the levels of miR-139-5p are significantly increased after the
transient transfection of miR-139-5p mimics.
To comprehensively explore the mechanism of
miR-139-5p in glioma, considerate analysis of 2 bioinformatics
algorithms, namely miRanda and RNA22, were performed to predict a
putative target gene. Next we detected Flt1 expression levels in
cells treated with miR-139-5p mimics and found reducted expression
of Flt1 both in mRNA and in protein levels (Fig. 2B and E). To further confirm
whether the decrease of Flt1 is related to the agent that
miR-139-5p directly targets its binding sites (Fig. 2C), we cotransfected U87 cells with
the vectors containing wild-type or mutant Flt1 3′UTR binding areas
together with miR-139-5p or miR-NC. The results indicated that
miR-139-5p induced a remarkable depression in luciferase intensity
of the wild-type Flt1 3′UTR, while the mutant-3′UTR showed no
distinct changes (Fig. 2D).
Furthermore, we detected the expression of miR-139-5p and Flt1 in
the tumor tissues removed from the xenograft models dosed with
miR-139-5p mimics or miR-NC by qRT-PCR and ICH, and the results
indicated evident higher levels of miR-139-5p and apparent lower
levels of Flt1 in miR-139-5p mimics injecting tumors (Fig. 2F and G). To conclude, we
demonstrated that miR-139-5p directly target Flt1 transcription,
and potentially induce the downregulation of Flt1.
Overexpression of miR-139-5p affects
glioma cell growth in vitro and in vivo and blocks the cell cycle
in G0/G1 progress
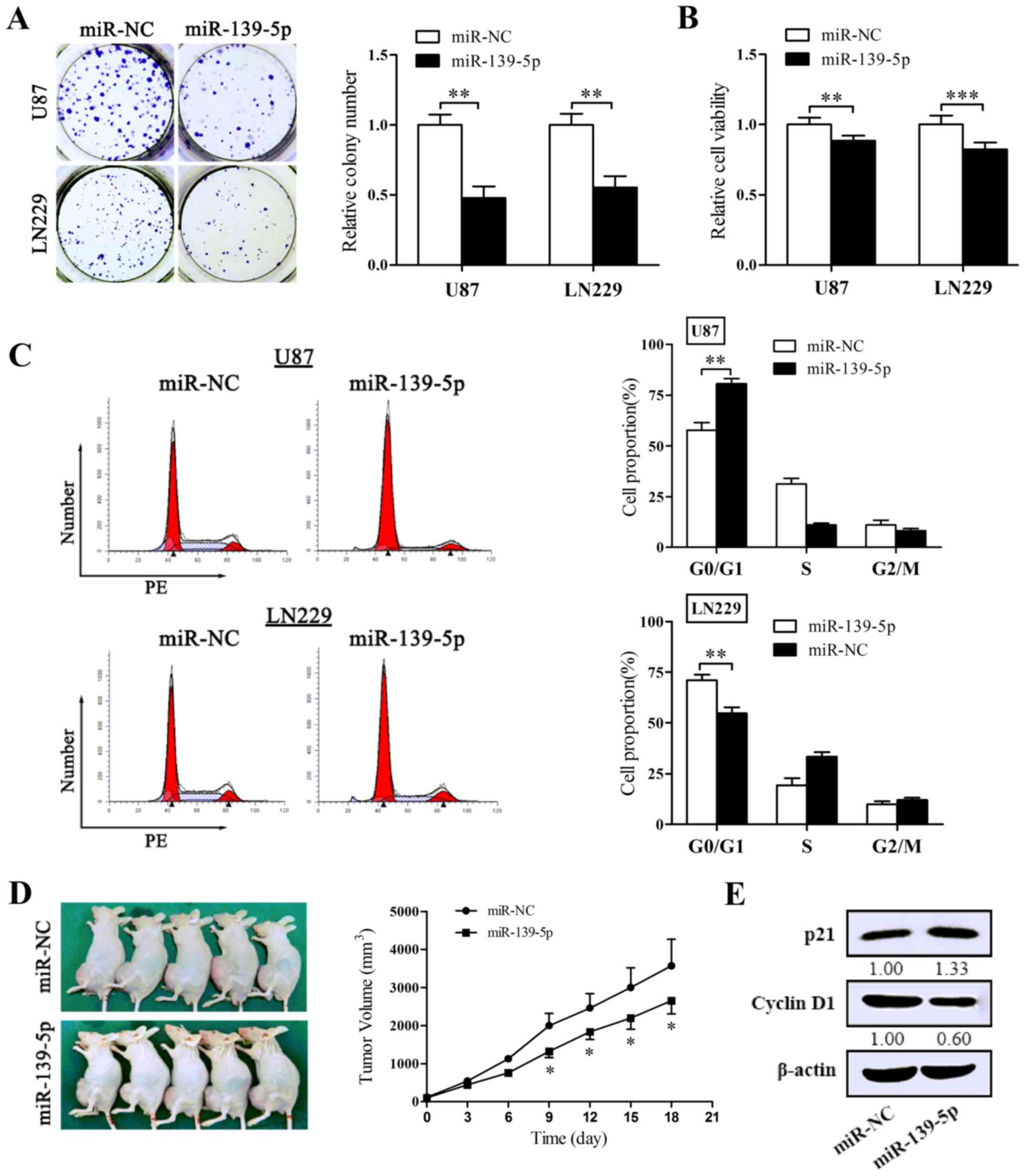
To identify the effect on the proliferation of
miR-139-5p, colony formation assay and MTT assay were performed in
U87 and LN229 cells. Reintroduction of miR-139-5p degraded colony
formation numbers of U87 and LN229 cells (Fig. 3A). Coincidentally, forced
expression of miR-139-5p induced a remarkable diminution in cell
viability (Fig. 3B). To affirm
the inhibitory impact of miR-139-5p on tumorigenesis in
vivo, the xenograft tumor models were conducted. As shown in
Fig. 3D, the multilocus delivery
of miR-139-5p mimics led to a prohibitive reaction to xenograft
tumor growth.
To determine whether miR-139-5p accommodates the
cell cycle processes, we performed flow cytometry and obtained
results that miR-139-5p accumulated the number of cells in G0/G1
phase and suppressed the G1/S transition both in U87 and LN229
cells (Fig. 3C). Then we tested
the expression of p21 and cyclin D1 in U87 cells to explain the
molecular levels in proliferation. As pictured in Fig. 3E, the levels of cyclin D1 declined
in cells overexpressing miR-139-5p and in contrast p21 increased.
Thus, these data suggest that miR-139-5p prohibits the
proliferation of glioma cells both in vitro and in
vivo via blocking the transition process of G1/S of glioma
cells.
Forced-expression of miR-139-5p
suppresses the migration, invasion and vasculogenic mimicry (VM) of
glioma cells in vitro and altered the expression of EMT-associated
protein
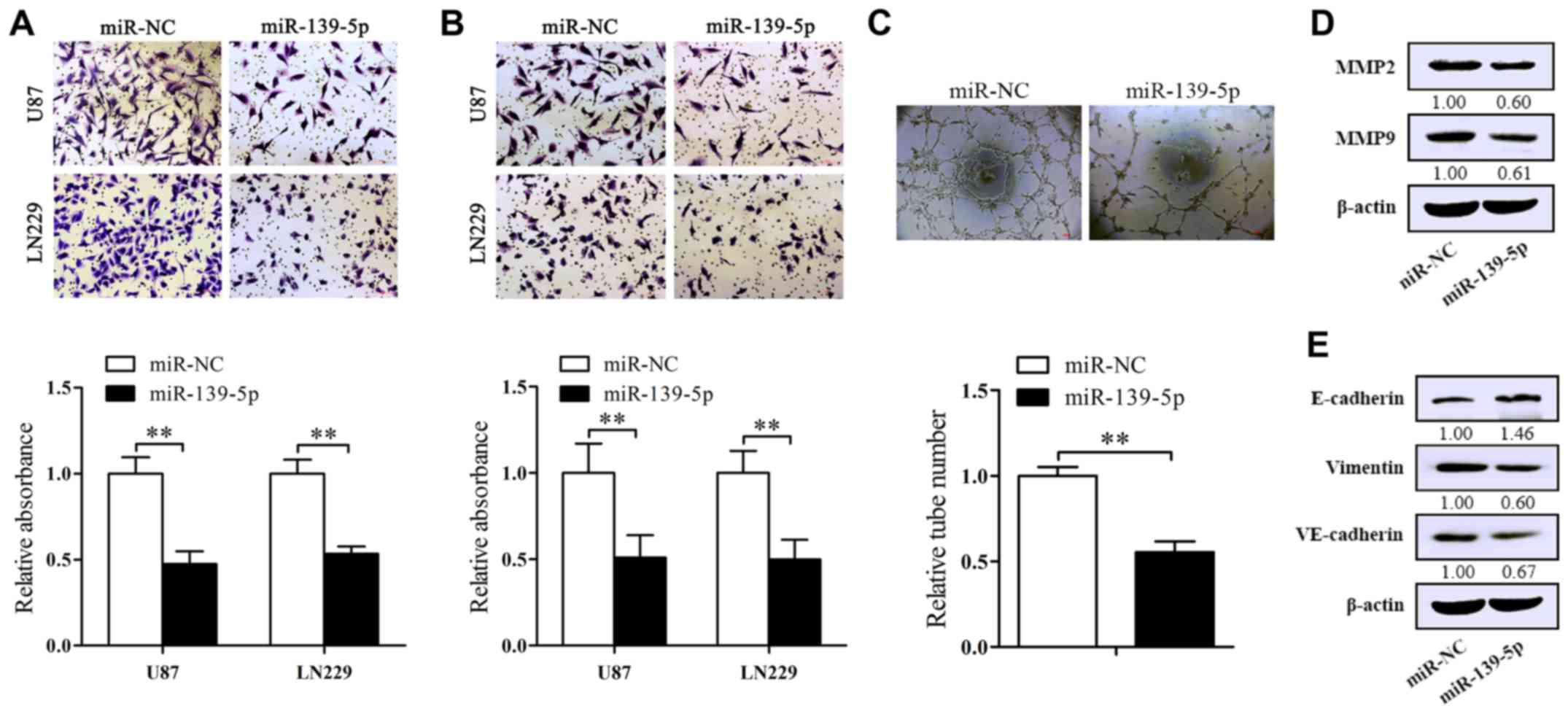
To identify the influence of miR-139-5p on glioma
cell migration and invasion qualities, the inserts were covered
with or without Matrigel. As expected, forced-expression of
miR-139-5p suppressed the migrated cells compared to control ones
(Fig. 4A). At equal place,
decreased invasion of U87 and LN229 cells were also obtained in the
inserts covered with Matrigel (Fig.
4B). To further explore the mechanism behind the inhibition, we
examined the levels of MMP2 and MMP9 which both are related to
migration/invasion processes. The results of western blot analysis
show a significantly decrease of MMP2 and MMP9 levels in miR-139-5p
overexpression cells (Fig.
4D).
VM, a kind of non-endothelial cell blood vessels
composed by solid tumor cells, has already been reported in human
glioblastoma tissues (21). Here
we used U87 cells to assess whether miR-139-5p affects the
formation of VM. The results we attained proclaimed that the
tube-formed number was decreased in miR-139-5p overexpressing U87
cells (Fig. 4C). Furthermore,
western blot analysis was executed to explore the vital molecular
markers in VM and EMT influenced by miR-139-5p mimics. As shown in
Fig. 4E, the elevated level of
miR-139-5p resulted in the reduction of VE-cadherin and vimentin
and the increase of E-cadherin in U87 cells. These data revealed
the inhibitory impression of miR-139-5p on EMT and VM of glioma
cells.
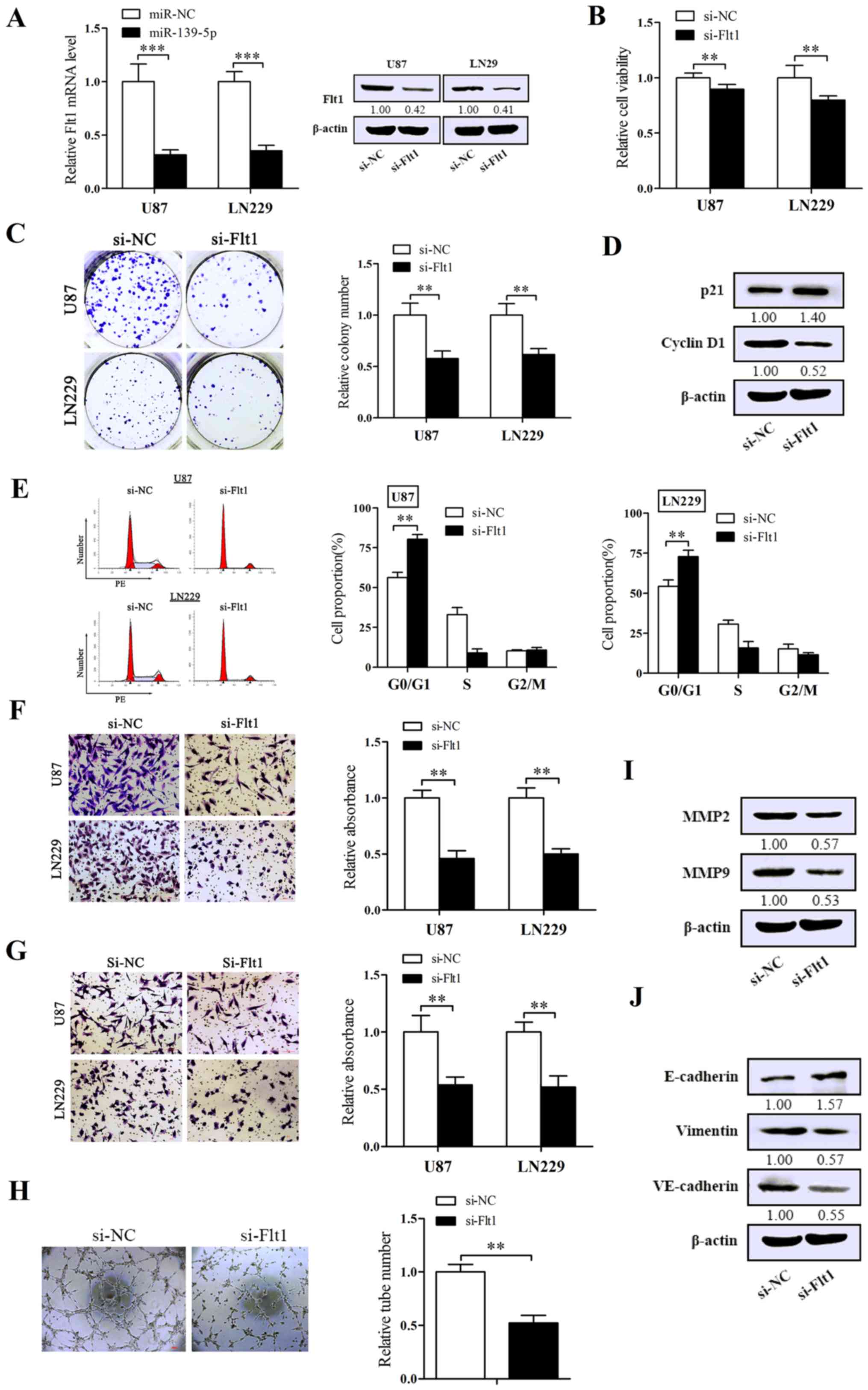
Suppression of Flt1 performs the
inhibitory effect on the malignant phenotype of glioma cells
To further investigate the concrete biological
impression of Flt1 on glioma cells, we chemically synthesized a
specific siRNA against Flt1. U87 and LN229 cells was transfected
with the siRNA of Flt1, then we validated the efficiency of
silencing Flt1. As expected, the expression of Flt1 was obviously
decreased in both mRNA and protein levels (Fig. 5A). As shown in Fig. 5B and C, knockdown of Flt1 induced
the suppression of cell viability and the capacity of
colony-formation. Flow cytometry was performed to assess the impact
of Flt1 on cell cycle. As shown in Fig. 5E, downregulation of Flt1 markedly
arrested cell cycle in G0/G1 phase. The migration/invasion assay
pointed out that silencing of Flt1 remarkably abated both migration
and invasion (Fig. 5F and G).
Moreover, silencing Flt1 also reduced the shaping of VM in U87
cells (Fig. 5H).
Furthermore, U87 cells were used to examine the
representative molecules related to the malignant phenotypes
following the silencing of Flt1 by western blot analysis. Cell
proliferation ability showed lower level of cyclin D1 and higher
level of p21 (Fig. 5D) in
si-Flt1-transfected U87 cells, in which we also obtained the
upregulation levels of MMP2 and MMP9 (Fig. 5I). Coincidentally with the effect
in EMT and VM, interfering Flt1 decreased the expression levels of
VE-cadherin and vimentin and increased the E-cadherin level
compared with control cells (Fig.
5J). In conclusion, Flt1 may serve as an oncogene in
glioma.
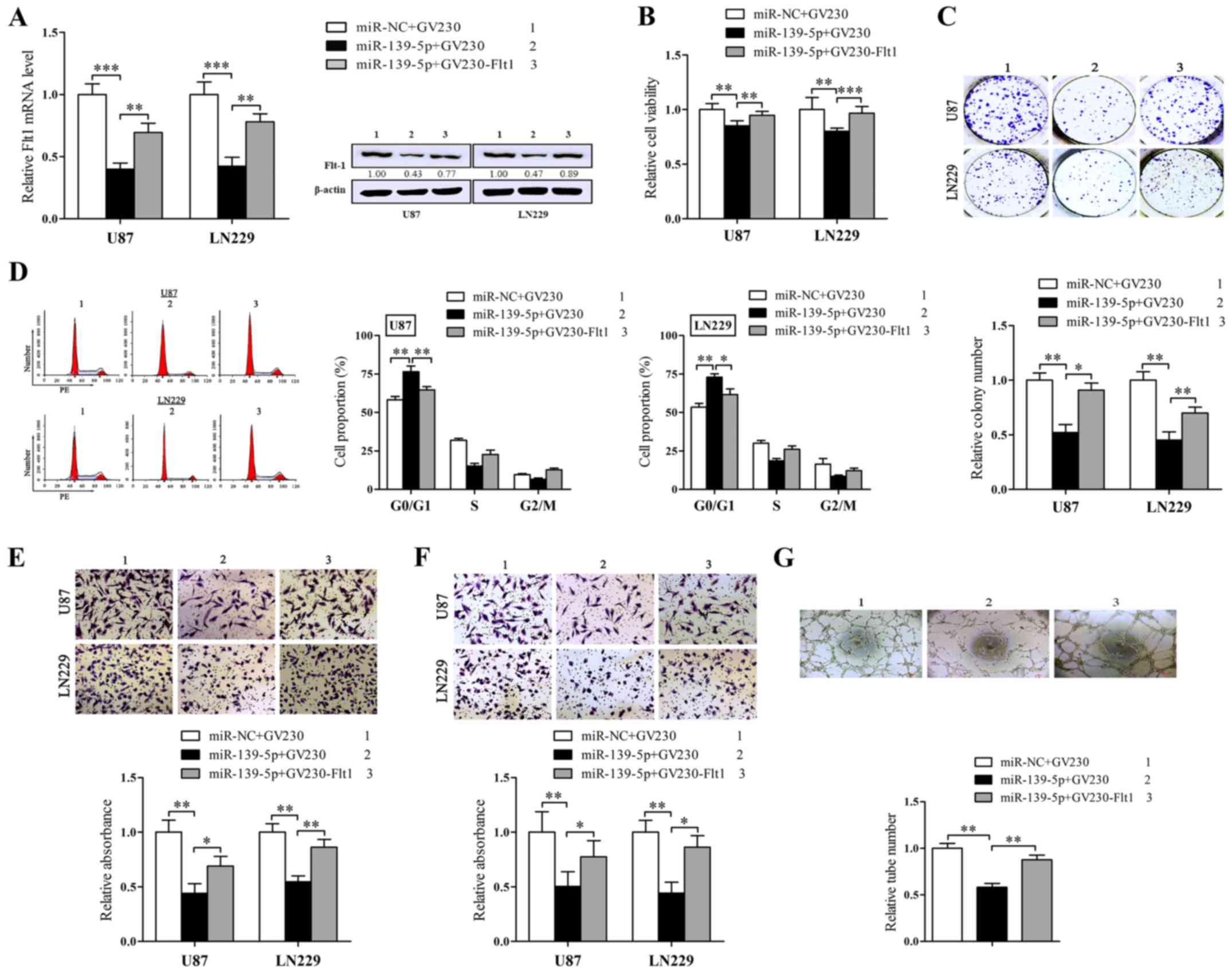
Restoration of Flt1 neutralizes the
function of miR-139-5p in glioma cells in vitro
Given the observations above uncovered that
miR-139-5p and Flt1 served as converse agents in regulating
malignant phenotypes of human glioma cells, we doubted whether the
function of miR-139-5p on glioma cells was mediated through its
inhibitory impression on Flt1 expression. Then the rescue
experiments were conducted to dispose it. We constructed an Flt1
expression plasmid without its 3′UTR and cotransfected it with
miR-139-5p mimics. Western blot analysis was forced to validate
whether the Flt1 protein levels reduced by miR-139-5p may be
rescued via the overexpression of Flt1 (Fig. 6A).
Then we further executed the rescue experiment in
different phenotypes of glioma cells. As anticipated (Fig. 6B), the restoration of Flt1 could
validly neutralize the suppressive influence of miR-139-5p on the
cell viability and colony formation (Fig. 6D) as well as the cell cycle block
in G0/G1 (Fig. 6C). In addition,
overexpression of Flt1 also overturned the inhibition of
migration/invasion and VM that miR-139-5p induced in glioma cells
(Fig. 6E–6G).
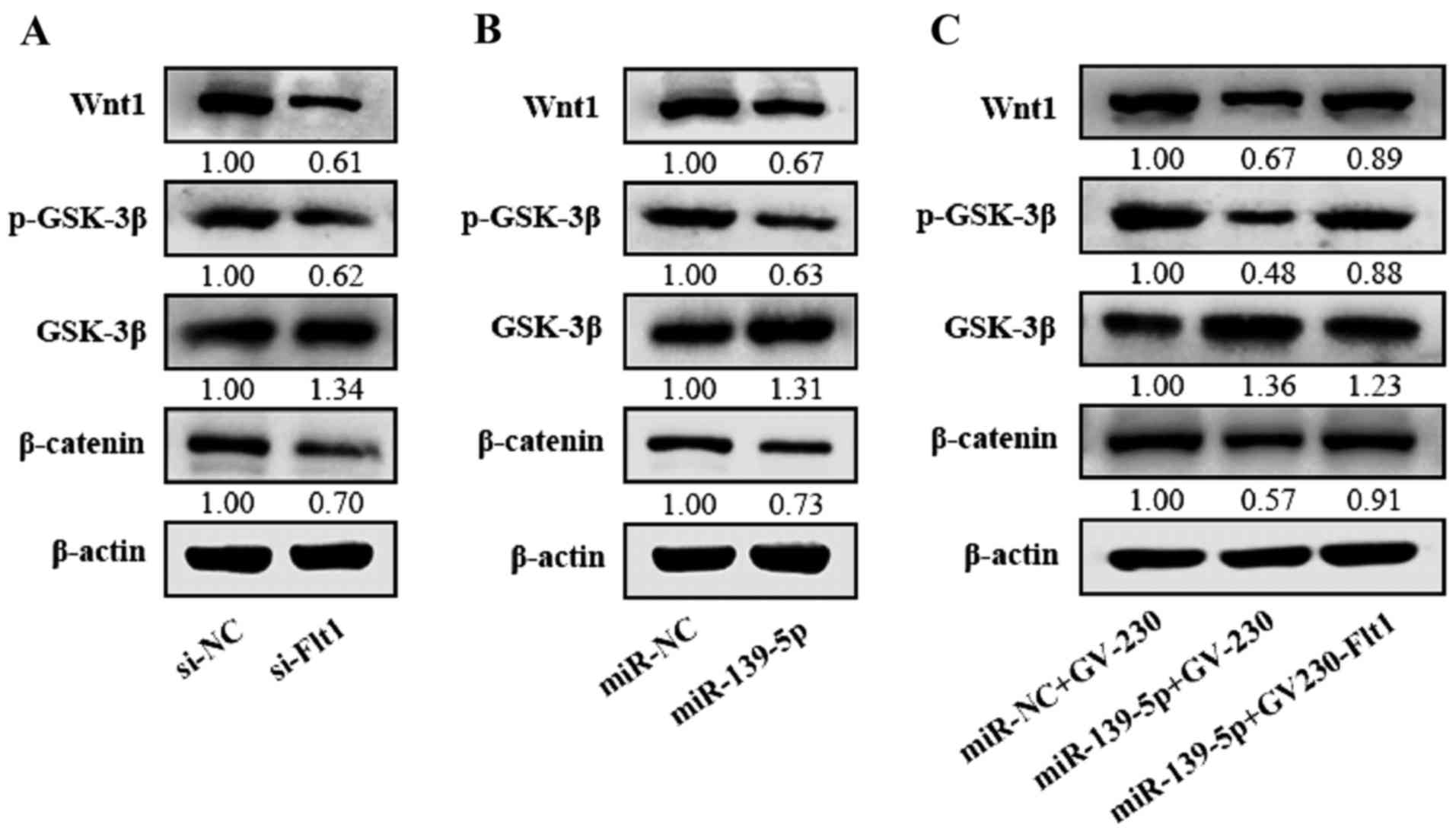
miR-139-5p inhibits the Wnt/β-catenin
pathway via targeting Flt1 in glioma cells
In spite of confirming the effect of Flt1 in the
malignance of glioma cells, we poorly clarified the extensional
mechanism among these. Flt1 is usually treated with VEGF as a whole
involved in VEGF/Flt1, which may serve as a potential synthetic
lethality to Wnt/β-catenin signaling pathway (22). In this study, we proved that
knockdown of Flt1 induced the restrain of the Wnt/β-catenin pathway
in glioma cells (Fig. 7A).
Together with our above results, we presumed that miR-139-5p may
suppress the Wnt/β-catenin signaling pathway by targeting Flt1 in
glioma. Then we verified the impression of miR-139-5p on the levels
of Wnt1, p-GSK-3β, GSK-3β and β-catenin, and the results indicated
that the Wnt1, β-catenin and the phosphorylated GSK-3β levels in
miR-139-5p-overexpressing cells were downregulated while the GSK-3β
was inversely upregulated (Fig.
7B), which indicates that miR-139-5p may inhibit the
Wnt/β-catenin pathway. To further verify the influence of
miR-139-5p on regulating the Wnt/β-catenin signaling pathway
mediated by Flt1, miR-139-5p mimics and Flt1 expression plasmid
without 3′UTR were cotransfected in U87 cells. Our results showed
that Flt1 can rescue the inhibition of Wnt/β-catenin pathway
induced by miR-139-5p (Fig. 7C).
All the data indicate that miR-139-5p may downregulate the
Wnt/β-catenin pathway through its target Flt1.
Discussion
Accumulated information concerning the molecular
mechanisms tightly interrelated to the initiation and progression
of glioma have been reported in recent years (23). MicroRNAs, mostly worked as a
tumor-suppressor agent in carcinoma, is commonly abrogated due to
the frequent hypermethylation of CpG islands of its primary
transcripts (24,25). miRNAs are involved in glioma
tumorigenesis, metastasis or angiogenesis, including that in glioma
cell biological processes in vitro. Recent studies have
indicated that miR-139-5p promotes apoptosis in cooperation with
TMZ (16) and inhibited the
migration and invasion (15) of
glioma cells in vitro. Our study further proved that
miR-139-5p worked as a tumor inhibitory factor, which mediated
inhibition of proliferation, migration, invasion, EMT and VM of
glioma cells. However, the molecular mechanism and the epigenetic
influence of miR-139-5p on human glioma remain unclear.
Recent studies have demonstrated that VEGF pathway
significantly contributed to glioblastoma angiogenesis and tumor
growth (26) and anti-VEGF/VEGFRs
therapeutics may be a new potential treatment for glioma (27,28). Flt1, also named VEGFR-1, is one of
VEGF receptors, which have been previously regarded as a
vascular-endothelial ligand of VEGF (29). A recent study reported that Flt1
was also expressed in many tumor cells. Lee et al found that
Flt1 located on breast cancer cell membrane, which intracrined VEGF
blinding as a survival factor (30). Lichtenberger et al proved
that VEGF interacted with Flt1 on tumor cells to enhance tumor
metastasis (31). Flt1 has also
been detected on human ovarian carcinoma cells exerting a crucial
function in cancer cell invasion (6). Given that Flt1 has recently been
reported highly expressed in glioblastoma (26), the cumulative expression level of
Flt1 was examined in glioma tissues and two glioma cell lines in
our study. Then we further located the expression of Flt1 on U87
and LN229 cell membrane. The function of tumor cell membrane-bound
Flt1 can be observed in several previous studies. Wei et al
confirmed that Flt1 located in colorectal cancer cells was
important to CRC progression (7).
Studies enforced to evaluate the tumor cell effects of Flt1 in lung
adenocarcinoma cells revealed that Flt1 occupied a considerable
role in cell proliferation and migration/invasion (32). In the present study, the function
of Flt1 in glioma malignancy was affirmed with Flt1-siRNA. Based
upon in vitro research, we obtained similar results in the
suppression of aggressive phenotypes of glioma cells, including
proliferation, migration/invasion, EMT and VM. These outcomes
validated the inhibitive effect of Flt1-specific siRNA in glioma
progression, but the explicit regulation mechanisms of Flt1 require
exploration of the epigenetic aspects.
In the present study, we determined the expression
level of Flt1 at transcriptional level and found the level of
miR-139-5p was evidently decreased in negative correlation with
increased expression of Flt1 in glioma tissues and cells. Then we
predicted that miR-139-5p may serve as a significant agent
negatively regulated by Flt1 in the field of cell malignant
phenotypes in glioma. Subsequently, bioinformatics analysis was
used and luciferase reporter analysis to validate that miR-139-5p
explicitly binds the 3′UTR region of Flt1. As expected, we verified
that the level of Flt1 was decreased in cells treated with
miR-139-5p mimics in both mRNA and protein levels. Thereafter,
based on the data above we considered that miR-139-5p may restain
the malignant phenotypes via decreasing Flt1 level. Consistent with
our proposal, forced-expression of miR-139-5p suppressed the
proliferation, migration/invasion, EMT and VM in glioma cells.
Furthermore, the results that overexpression of Flt1 without the
3′UTR greatly reversed the suppressing influence of miR-139-5p on
the malignant phenotypes were likewise observed in cotransfected
glioma cells. Taken together, these results confirm that Flt1 is
the direct downstream miR-139-5p target to regulate the development
of glioma.
Our study observed the effects of Flt1 on the
malignancy on glioma cells, but the mechanism is poorly understood.
Flt1 is normally involved in VEGF/Flt1 pathway, which has been
reported in many studies to be a critical fraction interaction with
several signaling pathway, such as PI3K/Akt-Rac1 pathway (33) and ERK/MAPK pathway (34). PIGF (53% homology to VEGF) is
another ligand for Flt1 and PIGF/Flt1 plays a key role in
colorectal cancer progression through p38-MMP9 pathway (7). The Wnt signaling is activated in
numerous cancers and Wnt/β-catenin pathway is the canonical
function group of Wnt signals contributing to tumor progression
including proliferation, metastasis and angiogenesis (9–12).
β-catenin was accumulated in the cytoplasm when GSK-3β is
phosphorylated due to the activation of Wnt1, and then transfers
into the nucleus to interwork with the transcription factors of
TCF/Lef-1, which further modulate the levels of corresponding
target genes to regulate the malignancy of cancer cells, such as
proliferation, migration/invasion and VM. Inhibition of
Wnt/β-catenin has been administered to be part of cancer
therapeutic strategies, and Wnt/β-catenin inhibition may reverse
the resistance to the inhibitor of PI3K/Akt treatment (35). A previous study declared that the
excitation of Wnt/β-catenin relies on Flt1 activity in colorectal
cancer cells (36), which means
Flt1 may be one of the significant regulators of Wnt/β-catenin
signals. In this study, we proved that miR-139-5p inhibits the
Wnt/β-catenin pathway and decreased the phosphorylation of GSK-3β
through altering Flt1 expression, which can be rescued by the
overexpression of Flt1. Together with the impression of
miR-139-5p/Flt1 regulatory axis on the malignancy on glioma cells,
the Flt1/Wnt/β-catenin pathway may be a mechanism that is involved
in miR-139-5p regulating the development of glioma.
In conclusion, we have confirmed a connection
between miR-139-5p and Flt1, and further reveal that miR-139-5p
serves as a tumor-suppressor partially through downregulating the
expression of Flt1 and inhibiting the Wnt/β-catenin pathway in
glioma. Despite the gradual comprehension of the various molecular
mechanisms involved in glioma, the therapy of glioma remains a
difficult clinical challenge. Given that anti- VEGFR drugs have
been a novel trial therapeutic for glioma, our results contribute
to present an Flt1-associated miRNA for potential diagnostic and
therapeutic strategies in glioma.
Acknowledgments
The study was supported by the National Key
Technology Support Program (no. 2014BAI04B00), the National Key
disciplines Fund of the Ministry of Health of the People’s Republic
of China, the Foundation of Tianjin Science and Technology
Committee (no. 14JCZDJC35600).
Notes
[1] Competing
interests
The authors declare that they have no competing
interests.
References
|
1
|
Furnari FB, Fenton T, Bachoo RM, Mukasa A,
Stommel JM, Stegh A, Hahn WC, Ligon KL, Louis DN, Brennan C, et al:
Malignant astrocytic glioma: Genetics, biology, and paths to
treatment. Genes Dev. 21:2683–2710. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Maher EA, Furnari FB, Bachoo RM, Rowitch
DH, Louis DN, Cavenee WK and DePinho RA: Malignant glioma: Genetics
and biology of a grave matter. Genes Dev. 15:1311–1333. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Takano S, Yamashita T and Ohneda O:
Molecular therapeutic targets for glioma angiogenesis. J Oncol.
2010:3519082010. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Ferrara N, Gerber HP and LeCouter J: The
biology of VEGF and its receptors. Nat Med. 9:669–676. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Herold-Mende C, Steiner HH, Andl T, Riede
D, Buttler A, Reisser C, Fusenig NE and Mueller MM: Expression and
functional significance of vascular endothelial growth factor
receptors in human tumor cells. Lab Invest. 79:1573–1582. 1999.
|
|
6
|
Li J, Li L, Li Z, Gong G, Chen P, Liu H,
Wang J, Liu Y and Wu X: The role of miR-205 in the VEGF-mediated
promotion of human ovarian cancer cell invasion. Gynecol Oncol.
137:125–133. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Wei SC, Tsao PN, Weng MT, Cao Z and Wong
JM: Flt-1 in colorectal cancer cells is required for the tumor
invasive effect of placental growth factor through a p38-MMP9
pathway. J Biomed Sci. 20:392013. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Fragoso R, Pereira T, Wu Y, Zhu Z,
Cabeçadas J and Dias S: VEGFR-1 (FLT-1) activation modulates acute
lymphoblastic leukemia localization and survival within the bone
marrow, determining the onset of extramedullary disease. Blood.
107:1608–1616. 2006. View Article : Google Scholar
|
|
9
|
Pierzynski JA, Hildebrandt MA, Kamat AM,
Lin J, Ye Y, Dinney CP and Wu X: Genetic variants in the
Wnt/β-catenin signaling pathway as indicators of bladder cancer
risk. J Urol. 194:1771–1776. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Mir R, Pradhan SJ, Patil P, Mulherkar R
and Galande S: Wnt/beta-catenin signaling regulated SATB1 promotes
colorectal cancer tumorigenesis and progression. Oncogene.
35:1679–1691. 2016. View Article : Google Scholar
|
|
11
|
Wang G, Zhao Y and Zheng Y:
MiR-122/Wnt/β-catenin regulatory circuitry sustains glioma
progression. Tumour Biol. 35:8565–8572. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Yan Z, Che S, Wang J, Jiao Y, Wang C and
Meng Q: miR-155 contributes to the progression of glioma by
enhancing Wnt/beta-catenin pathway. Tumour Biol. 36:5323–5331.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Bartel DP: MicroRNAs: Target recognition
and regulatory functions. Cell. 136:215–233. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Sato F, Tsuchiya S, Meltzer SJ and Shimizu
K: MicroRNAs and epigenetics. FEBS J. 278:1598–1609. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Yue S, Wang L, Zhang H, Min Y, Lou Y, Sun
H, Jiang Y, Zhang W, Liang A, Guo Y, et al: miR-139-5p suppresses
cancer cell migration and invasion through targeting ZEB1 and ZEB2
in GBM. Tumour Biol. 36:6741–6749. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Li RY, Chen LC, Zhang HY, Du WZ, Feng Y,
Wang HB, Wen JQ, Liu X, Li XF, Sun Y, et al: miR-139 inhibits Mcl-1
expression and potentiates TMZ-induced apoptosis in glioma. CNS
Neurosci Ther. 19:477–483. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Zhang L, Dong Y, Zhu N, Tsoi H, Zhao Z, Wu
CW, Wang K, Zheng S, Ng SS, Chan FK, et al: microRNA-139-5p exerts
tumor suppressor function by targeting NOTCH1 in colorectal cancer.
Mol Cancer. 13:1242014. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Xu W, Hang M, Yuan CY, Wu FL, Chen SB and
Xue K: MicroRNA-139-5p inhibits cell proliferation and invasion by
targeting insulin-like growth factor 1 receptor in human non-small
cell lung cancer. Int J Clin Exp Pathol. 8:3864–3870.
2015.PubMed/NCBI
|
|
19
|
Wang X, Tong X, Gao H, Yan X, Xu X, Sun S,
Wang Q and Wang J: Silencing HIWI suppresses the growth, invasion
and migration of glioma cells. Int J Oncol. 45:2385–2392. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Pin AL, Houle F, Fournier P, Guillonneau
M, Paquet ÉR, Simard MJ, Royal I and Huot J: Annexin-1-mediated
endothelial cell migration and angiogenesis are regulated by
vascular endothelial growth factor (VEGF)-induced inhibition of
miR-196a expression. J Biol Chem. 287:30541–30551. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
El Hallani S, Boisselier B, Peglion F,
Rousseau A, Colin C, Idbaih A, Marie Y, Mokhtari K, Thomas JL,
Eichmann A, et al: A new alternative mechanism in glioblastoma
vascularization: Tubular vasculogenic mimicry. Brain. 133:973–982.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Naik S, Dothager RS, Marasa J, Lewis CL
and Piwnica-Worms D: Vascular endothelial growth factor receptor-1
is synthetic lethal to aberrant {beta}-catenin activation in colon
cancer. Clin Cancer Res. 15:7529–7537. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Marumoto T and Saya H: Molecular biology
of glioma. Adv Exp Med Biol. 746:2–11. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Lujambio A, Ropero S, Ballestar E, Fraga
MF, Cerrato C, Setién F, Casado S, Suarez-Gauthier A,
Sanchez-Cespedes M, Git A, et al: Genetic unmasking of an
epigenetically silenced microRNA in human cancer cells. Cancer Res.
67:1424–1429. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Zhang Z, Tang H, Wang Z, Zhang B, Liu W,
Lu H, Xiao L, Liu X, Wang R, Li X, et al: miR-185 targets the DNA
methyl-transferases 1 and regulates global DNA methylation in human
glioma. Mol Cancer. 10:1242011. View Article : Google Scholar
|
|
26
|
Huang H, Held-Feindt J, Buhl R, Mehdorn HM
and Mentlein R: Expression of VEGF and its receptors in different
brain tumors. Neurol Res. 27:371–377. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Kreisl TN, Kim L, Moore K, Duic P, Royce
C, Stroud I, Garren N, Mackey M, Butman JA, Camphausen K, et al:
Phase II trial of single-agent bevacizumab followed by bevacizumab
plus irinotecan at tumor progression in recurrent glioblastoma. J
Clin Oncol. 27:740–745. 2009. View Article : Google Scholar :
|
|
28
|
Batchelor TT, Mulholland P, Neyns B,
Nabors LB, Campone M, Wick A, Mason W, Mikkelsen T, Phuphanich S,
Ashby LS, et al: Phase III randomized trial comparing the efficacy
of cediranib as monotherapy, and in combination with lomustine,
versus lomustine alone in patients with recurrent glioblastoma. J
Clin Oncol. 31:3212–3218. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Vieira JM, Ruhrberg C and Schwarz Q: VEGF
receptor signaling in vertebrate development. Organogenesis.
6:97–106. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Lee TH, Seng S, Sekine M, Hinton C, Fu Y,
Avraham HK and Avraham S: Vascular endothelial growth factor
mediates intracrine survival in human breast carcinoma cells
through internally expressed VEGFR1/FLT1. PLoS Med. 4:e1862007.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Lichtenberger BM, Tan PK, Niederleithner
H, Ferrara N, Petzelbauer P and Sibilia M: Autocrine VEGF signaling
synergizes with EGFR in tumor cells to promote epithelial cancer
development. Cell. 140:268–279. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Roybal JD, Zang Y, Ahn YH, Yang Y, Gibbons
DL, Baird BN, Alvarez C, Thilaganathan N, Liu DD, Saintigny P, et
al: miR-200 inhibits lung adenocarcinoma cell invasion and
metastasis by targeting Flt1/VEGFR1. Mol Cancer Res. 9:25–35. 2011.
View Article : Google Scholar :
|
|
33
|
Wang F, Yamauchi M, Muramatsu M, Osawa T,
Tsuchida R and Shibuya M: RACK1 regulates VEGF/Flt1-mediated cell
migration via activation of a PI3K/Akt pathway. J Biol Chem.
286:9097–9106. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Takahashi M, Matsui A, Inao M, Mochida S
and Fujiwara K: ERK/MAPK-dependent PI3K/Akt phosphorylation through
VEGFR-1 after VEGF stimulation in activated hepatic stellate cells.
Hepatol Res. 26:232–236. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Arqués O, Chicote I, Puig I, Tenbaum SP,
Argilés G, Dienstmann R, Fernández N, Caratù G, Matito J,
Silberschmidt D, et al: Tankyrase inhibition blocks
Wnt/beta-catenin pathway and reverts resistance to PI3K and AKT
inhibitors in the treatment of colorectal cancer. Clin Cancer Res.
22:644–656. 2016. View Article : Google Scholar
|
|
36
|
Zeitlin BD, Ellis LM and Nor JE:
Inhibition of vascular endothelial growth factor
receptor-1/Wnt/{beta}-catenin crosstalk leads to tumor cell death.
Clin Cancer Res. 15:7453–7455. 2009. View Article : Google Scholar : PubMed/NCBI
|