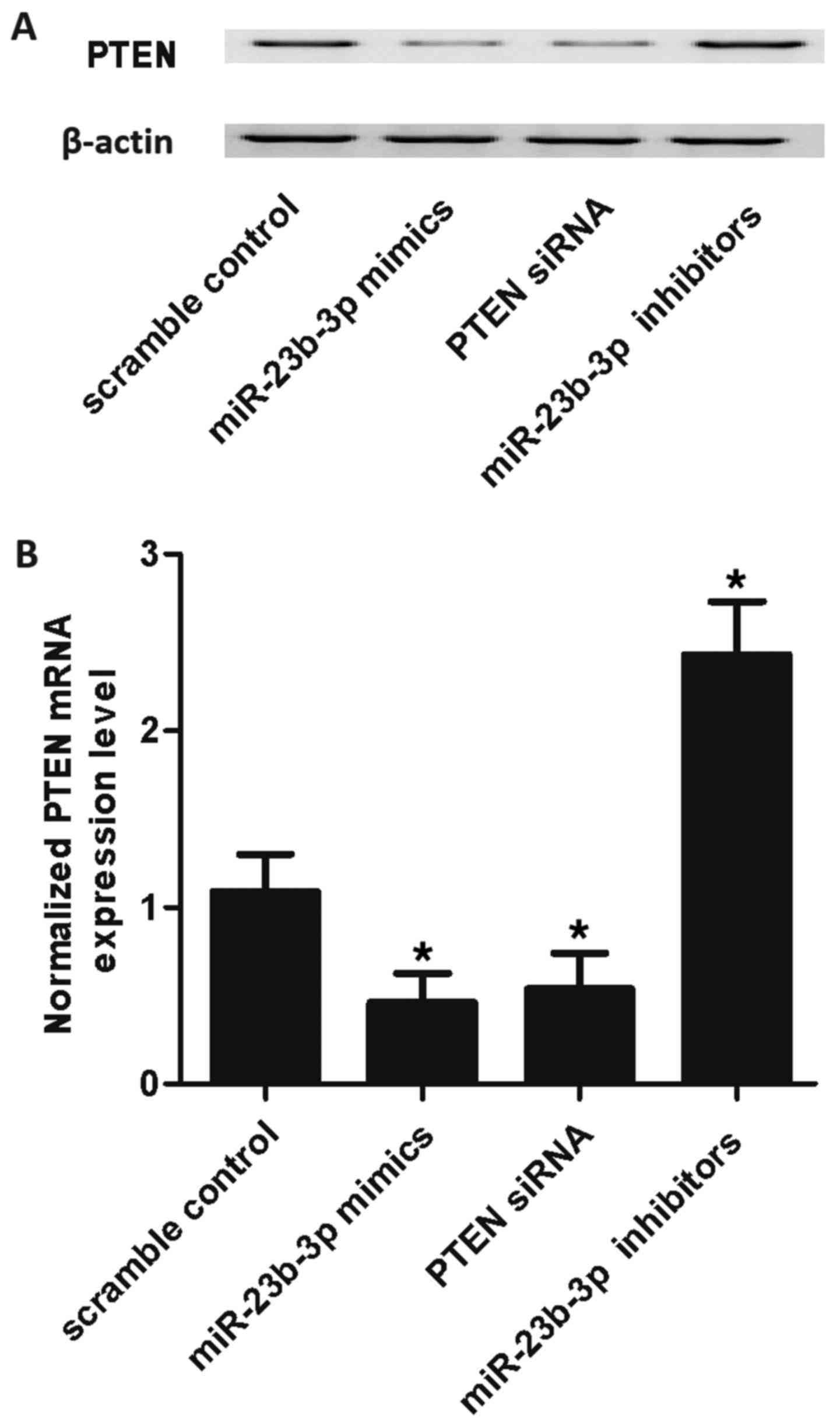
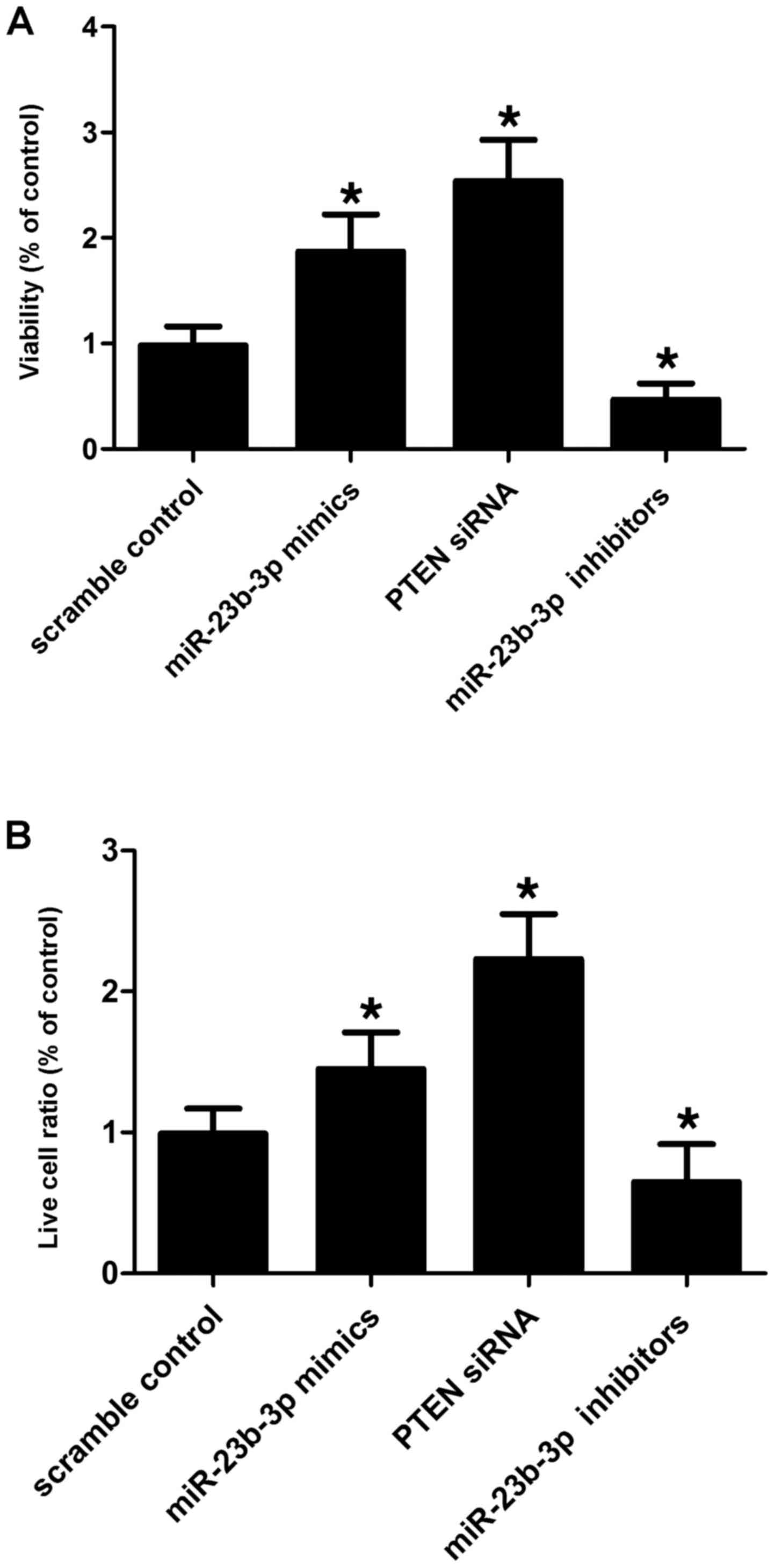
|
1
|
Fergusen S and Macdonald RL: Predictors of
cerebral infarction in patients with aneurysmal subarachnoid
hemorrhage. Neurosurgery. 60:658–667; discussion 667. 2007.
View Article : Google Scholar
|
|
2
|
Schievink WI: Intracranial aneurysms. N
Engl J Med. 336:28–40. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Connolly ES Jr, Choudhri TF, Mack WJ,
Mocco J, Spinks TJ, Slosberg J, Lin T, Huang J and Solomon RA:
Influence of smoking, hypertension, and sex on the phenotypic
expression of familial intracranial aneurysms in siblings.
Neurosurgery. 48:64–68; discussion 68–69. 2001.PubMed/NCBI
|
|
4
|
Bonita R: Cigarette smoking, hypertension
and the risk of subarachnoid hemorrhage: a population-based
case-control study. Stroke. 17:831–835. 1986. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Kaźmierski M, Michalewska-Włudarczyk A and
Krzych LJ: Intima-media thickness and flow-mediated dilatation in
the diagnosis of coronary artery disease in perimenopausal women.
Pol Arch Med Wewn. 120:181–188. 2010.
|
|
6
|
Nakajima N, Nagahiro S, Sano T, Satomi J
and Satoh K: Phenotypic modulation of smooth muscle cells in human
cerebral aneurysmal walls. Acta Neuropathol. 100:475–480. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Aoki T, Kataoka H, Morimoto M, Nozaki K
and Hashimoto N: Macrophage-derived matrix metalloproteinase-2 and
-9 promote the progression of cerebral aneurysms in rats. Stroke.
38:162–169. 2007. View Article : Google Scholar
|
|
8
|
Page-McCaw A, Ewald AJ and Werb Z: Matrix
metalloproteinases and the regulation of tissue remodelling. Nat
Rev Mol Cell Biol. 8:221–233. 2007. View
Article : Google Scholar : PubMed/NCBI
|
|
9
|
Parks WC, Wilson CL and López-Boado YS:
Matrix metallo-proteinases as modulators of inflammation and innate
immunity. Nat Rev Immunol. 4:617–629. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Meng H, Metaxa E, Gao L, Liaw N, Natarajan
SK, Swartz DD, Siddiqui AH, Kolega J and Mocco J: Progressive
aneurysm development following hemodynamic insult. J Neurosurg.
114:1095–1103. 2011. View Article : Google Scholar
|
|
11
|
Aoki T, Kataoka H, Ishibashi R, Nozaki K,
Morishita R and Hashimoto N: Reduced collagen biosynthesis is the
hallmark of cerebral aneurysm: contribution of interleukin-1beta
and nuclear factor-kappaB. Arterioscler Thromb Vasc Biol.
29:1080–1086. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Bartel DP: MicroRNAs: genomics,
biogenesis, mechanism, and function. Cell. 116:281–297. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Griffiths-Jones S: The microRNA registry.
Nucleic Acids Res. 32:D109–D111. 2004. View Article : Google Scholar :
|
|
14
|
Pandey AK, Agarwal P, Kaur K and Datta M:
MicroRNAs in diabetes: tiny players in big disease. Cell Physiol
Biochem. 23:221–232. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Porter KE and Riches K: The vascular
smooth muscle cell: a therapeutic target in type 2 diabetes? Clin
Sci (Lond). 125:167–182. 2013. View Article : Google Scholar
|
|
16
|
Jiang Y, Zhang M, He H, Chen J, Zeng H, Li
J and Duan R: MicroRNA/mRNA profiling and regulatory network of
intra-cranial aneurysm. BMC Med Genomics. 6:362013. View Article : Google Scholar
|
|
17
|
Furgeson SB, Simpson PA, Park I, Vanputten
V, Horita H, Kontos CD, Nemenoff RA and Weiser-Evans MC:
Inactivation of the tumour suppressor, PTEN, in smooth muscle
promotes a pro-inflammatory phenotype and enhances neointima
formation. Cardiovasc Res. 86:274–282. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Livak and Schmittgen: Analysis of relative
gene expression data using real-time quantitative PCR and the
2(-Delta Delta C(T)) Method. Methods. 25:402–408. 2001. View Article : Google Scholar
|
|
19
|
Hartl DL and Ochman H: Inverse polymerase
chain reaction. Methods Mol Biol. 58:293–301. 1993.
|
|
20
|
Chen D, Wu X, Xia M, Wu F, Ding J, Jiao Y,
Zhan Q and An F: Upregulated exosomic miR-23b-3p plays regulatory
roles in the progression of pancreatic cancer. Oncol Rep.
38:2182–2188. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Zhang H, Hao Y, Yang J, Zhou Y, Li J, Yin
S, Sun C, Ma M, Huang Y and Xi JJ: Genome-wide functional screening
of miR-23b as a pleiotropic modulator suppressing cancer
metastasis. Nat Commun. 2:5542011. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Zaman MS, Thamminana S, Shahryari V,
Chiyomaru T, Deng G, Saini S, Majid S, Fukuhara S, Chang I, Arora
S, et al: Inhibition of PTEN gene expression by oncogenic
miR-23b-3p in renal cancer. PLoS One. 7:e502032012. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Li B, Sun M, Gao F, Liu W, Yang Y, Liu H,
Cheng Y, Liu C and Cai J: Up-regulated expression of miR-23a/b
targeted the pro-apoptotic Fas in radiation-induced thymic
lymphoma. Cell Physiol Biochem. 32:1729–1740. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
An Y, Zhang Z, Shang Y, Jiang X, Dong J,
Yu P, Nie Y and Zhao Q: miR-23b-3p regulates the chemoresistance of
gastric cancer cells by targeting ATG12 and HMGB2. Cell Death Dis.
6:e17662015. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Oudit GY, Sun H, Kerfant BG, Crackower MA,
Penninger JM and Backx PH: The role of phosphoinositide-3 kinase
and PTEN in cardiovascular physiology and disease. J Mol Cell
Cardiol. 37:449–471. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Huang J, Niu XL, Pippen AM, Annex BH and
Kontos CD: Adenovirus-mediated intraarterial delivery of PTEN
inhibits neointimal hyperplasia. Arterioscler Thromb Vasc Biol.
25:354–358. 2005. View Article : Google Scholar
|
|
27
|
Mitra AK, Jia G, Gangahar DM and Agrawal
DK: Temporal PTEN inactivation causes proliferation of saphenous
vein smooth muscle cells of human CABG conduits. J Cell Mol Med.
13:177–187. 2009. View Article : Google Scholar :
|
|
28
|
Mourani PM, Garl PJ, Wenzlau JM, Carpenter
TC, Stenmark KR and Weiser-Evans MC: Unique, highly proliferative
growth phenotype expressed by embryonic and neointimal smooth
muscle cells is driven by constitutive Akt, mTOR, and p70S6K
signaling and is actively repressed by PTEN. Circulation.
109:1299–1306. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Nemenoff RA, Horita H, Ostriker AC,
Furgeson SB, Simpson PA, VanPutten V, Crossno J, Offermanns S and
Weiser-Evans MC: SDF-1α induction in mature smooth muscle cells by
inactivation of PTEN is a critical mediator of exacerbated
injury-induced neointima formation. Arterioscler Thromb Vasc Biol.
31:1300–1308. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Ji R, Cheng Y, Yue J, Yang J, Liu X, Chen
H, Dean DB and Zhang C: MicroRNA expression signature and
antisense-mediated depletion reveal an essential role of microRNA
in vascular neointimal lesion formation. Circ Res. 100:1579–1588.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Nelen MR, van Staveren WC, Peeters EA,
Hassel MB, Gorlin RJ, Hamm H, Lindboe CF, Fryns JP, Sijmons RH,
Woods DG, et al: Germline mutations in the PTEN/MMAC1 gene in
patients with Cowden disease. Hum Mol Genet. 6:1383–1387. 1997.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Marsh DJ, Dahia PL, Zheng Z, Liaw D,
Parsons R, Gorlin RJ and Eng C: Germline mutations in PTEN are
present in Bannayan-Zonana syndrome. Nat Genet. 16:333–334. 1997.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Suzuki A, de la Pompa JL, Stambolic V,
Elia AJ, Sasaki T, del Barco Barrantes I, Ho A, Wakeham A, Itie A,
Khoo W, et al: High cancer susceptibility and embryonic lethality
associated with mutation of the PTEN tumor suppressor gene in mice.
Curr Biol. 8:1169–1178. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Freeman D, Lesche R, Kertesz N, Wang S, Li
G, Gao J, Groszer M, Martinez-Diaz H, Rozengurt N, Thomas G, et al:
Genetic background controls tumor development in PTEN-deficient
mice. Cancer Res. 66:6492–6496. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Sun JF, Phung T, Shiojima I, Felske T,
Upalakalin JN, Feng D, Kornaga T, Dor T, Dvorak AM, Walsh K, et al:
Microvascular patterning is controlled by fine-tuning the Akt
signal. Proc Natl Acad Sci USA. 102:128–133. 2005. View Article : Google Scholar
|
|
36
|
Phung TL, Ziv K, Dabydeen D, Eyiah-Mensah
G, Riveros M, Perruzzi C, Sun J, Monahan-Earley RA, Shiojima I,
Nagy JA, et al: Pathological angiogenesis is induced by sustained
Akt signaling and inhibited by rapamycin. Cancer Cell. 10:159–170.
2006. View Article : Google Scholar : PubMed/NCBI
|