Introduction
Oral cavity and oropharyngeal squamous cell
carcinoma is the eighth most common type of cancer among men and
fourteenth most common among women in the U.S. according to
recently reported data (1). Oral
squamous cell carcinoma (OSCC) is the most common head and neck
cancer worldwide, with a high morbidity and mortality, and is
generally defined as squamous cell carcinoma originating in the
lips, tongue, gums, cheeks, palate and mouth (2). Prognosis and treatment planning is
typically assessed based on tumor staging according to the TNM
classification and Union for International Cancer Control system
(3-6). However, the biological heterogeneity
of OSCC is not taken into consideration by these staging systems.
Therefore, more specific bio-markers for OSCC are required.
The majority of OSCC cases are attributed to
smoking, alcohol consumption, chewing betel nut and human
papillomavirus infection (7).
However, the precise mechanisms underlying OSCC development remain
poorly understood. In recent decades, little improvement has been
achieved to the overall prognosis for advanced-stage OSCC patients,
with a heavy disease burden remaining for patients and their
families (8). Thus, achieving a
better understanding of the mechanisms underlying oral
carcinogenesis continues to be urgent in order to aid the
identification of effective therapeutic targets (9-11).
Long non-coding RNA (lncRNA) in the genome is a type
of RNA that is >200 nt in length and lacks an open reading frame
(12). The information contained
in lncRNA is rich and serves a role in normal physiological
processes, including cell growth, development, metabolism and
apoptosis. In addition, a large number of abnormally expressed
lncRNAs are involved in tumor metabolism, occurrence and
development (13). Therefore, the
study of lncRNAs associated with tumor formation, invasion and
metastasis, as well as their underlying molecular mechanisms,
serves an extremely important role in the prevention and treatment
of cancer.
It has been estimated that >70% of DNA sequences
in the human genome can be transcribed into RNA; however, only 2%
of these transcripts are protein-coding, while the majority of
transcripts are categorized as non-coding RNAs (ncRNAs) (14). With the exception of housekeeping
ncRNAs, such as transfer RNAs and ribosomal RNAs, certain mRNA-like
transcripts also exist that can be subdivided by length, including
small ncRNAs (<200 nt) and long ncRNAs (lncRNA; >200 nt)
(15). Small ncRNAs, such as
microRNAs, have been extensively studied, and there is evidence to
suggest that they may serve an important role in several types of
cancer, including OSCC (16).
However, the role of lncRNAs in cancer has only been investigated
in the last decade (17,18). To date, a small amount of research
has been conducted on the function of lncRNAs, indicating that they
are involved in transcription and serve as post-transcriptional
regulators. Furthermore, lncRNAs can reshape chromatin states
(8,18-21), stabilize proteins and protein
complexes (22), and compete with
endogenous RNAs to regulate their function (23,24).
At present, the classification according to the
tumor size and presence of distant metastasis of cancer cells in
regional lymph nodes is not sufficient to accurately predict the
prognosis of OSCC. Therefore, new prognostic parameters that are
sufficiently specific and sensitive are urgently required to
optimize patient stratification, treatment options and prognosis in
clinical practice.
To better understand the role of lncRNAs in the
carcinogenesis of OSCC and gain an insight into the identification
of potential clinically relevant targets, the current study
investigated the role and underlying mechanism of the lncRNA
C5orf66 antisense RNA 1 (C5orf66-AS1) in OSCC tissues and
cells.
Materials and methods
Clinical specimens
A total of 30 paired OSCC and adjacent normal
tissues were collected from 30 OSCC patients (male, 21; female, 9;
age, 35-67 years) at the Stomatological Hospital of Guizhou Medical
University (Guiyang, China) between July 2016 and July 2017. None
of the patients received radiotherapy, chemotherapy or other
special treatment. All patients were diagnosed with oral squamous
cell carcinoma by histological examination. All tissue samples were
immediately flash-frozen in liquid nitrogen and stored at −80°C.
The present study was approved by the Human Ethics Committee Review
Board at the Stomatological Hospital of Guizhou Medical University.
Informed consent was provided by each patient.
Cell culture and treatment
The OSCC cell line SCC9 and the primary normal human
oral keratinocyte (HOK) cells were originally acquired from the
American Type Culture Collection (Manassas, VA, USA) and cultured
in our institute. OSCC cells were incubated in RPMI 1640 medium
(Gibco; Thermo Fisher Scientific, Inc., Waltham, MA, USA)
containing 10% fetal bovine serum and 1% penicillin-streptomycin
solution (Sigma-Aldrich; Merck KGaA, Darmstadt, Germany) at 37°C
with 5% CO2. Cells were passaged every 2-3 days.
Following two passages, SCC9 cells (3×104
cells/well) were seeded into 6-well plates and then transiently
transfected with the negative control, lncRNA C5orf66-AS1-small
interfering RNA (siRNA), lncRNA C5orf66-AS1-plasmid or lncRNA
C5orf66-AS1-siRNA + cytochrome c1 (CYC1)-siRNA, respectively. For
transfection, Lipofectamine 2000 (Invitrogen; Thermo Fisher
Scientific, Inc.) was used, according to the manufacturer’s
protocol.
MTT assay
In the present study, the cell proliferation ability
was measured by performing an MTT assay (Sigma-Aldrich; Merck
KGaA). Briefly, cells were seeded into 96-well plates
(3×103 cells per well) and incubated for 0, 12, 24 or 48
h, after which 20 µl MTT was added to each well and then incubated
for a further 4 h at 37°C. Cell viability was assessed by detecting
the absorbance at 450 nm using a microplate reader. All tests were
performed in quadruplicate.
Reverse transcription-quantitative
polymerase chain reaction (RT-qPCR)
TRIzol reagent (Thermo Fisher Scientific, Inc.) was
used to extract the total RNA from the cells. The total RNA
concentration was detected by Nanodrop 2000 (Thermo Fisher
Scientific, Inc.), and then stored at −80°C prior to use. Next,
cDNA was generated using the PrimeScript™ RT reagent kit (Takara
Bio, Inc.) in line with the manufacturer’s protocol. SYBR Premix Ex
Taq (Takara Bio, Inc.) was then used to analyze the synthesized
cDNAs according to the manufacturer’s instructions. Amplification
conditions were as follows: 95°C for 10 min, followed by 40 cycles
at 95°C for 15 sec, 72°C for 30 sec and 78°C for 1.5 min. Primer
sequences used for qPCR were as follows: LncRNA C5orf66-AS1,
forward 5′-GCTTCGCGTCAAGAGGGTAT-3′ and reverse
5′-GACCGACGTCTGCTGCTTTT-3′; CYC1, forward
5′-GAGGTGGAGGTTCAAGACGG-3′ and reverse 5′-TAGCTCGCACGATGTAGCTG-3′;
B cell lymphoma 2 (Bcl-2), forward 5′-TTGGATCAGGGAGTTGGAAG-3′ and
reverse 5′-TGTCCCTACCAACCAGAAGG-3′; Bcl-2-associated X protein
(Bax), forward 5′-CGTCCACCAAGAAGCTGAGCG-3′ and reverse
5′-CGTCCACCAAGAAGCTGAGCG-3′; matrix metalloproteinase (MMP)-9,
forward 5′-GAGGCGCTCATGTACCCTATGTAC-3′ and reverse
5′-GTTCAGGGCGAGGACCATAGA G-3′; Caspase-3, forward
5′-GGCTTGCCAGAAGATACCGGT-3′ and reverse
5′-GCATAAATTCTAGCTTGTGCGCGT-3′; Caspase-7, forward
5′-GATCAGCCTTGTGGGATGGCAGA-3′ and reverse
5′-GTACTGATATGTAGGCACTCG-3′; Caspase-9, forward
5′-CAGAATGCGCTCCTTTCACTTTG-3′ and reverse
5′-AAGTAGCTCACGATTCTCTCTAC-3′; and GAPDH, forward
5′-CTTTGGTATCGTGGAAGGACTC-3′ and reverse
5′-GTAGAGGCAGGGATGATGTTCT-3′. The 2−ΔΔCq method
(25) was applied for the
calculation of the relative expression of the genes. GAPDH was used
as an internal control.
Cell migration and invasion assays
An in vitro invasion assay was performed
using Transwell plates (BD Biosciences, Franklin Lakes, NJ, USA)
with 8-µm pores. SCC9 cells (1×104 cells/ml) in RPMI
1640 medium were added to the upper chamber of the Transwell
24-well plates, while RPMI-1640 medium containing 20% fetal bovine
serum as a chemoattractant was added to the lower chamber. After
48-h incubation, cells remaining in the upper chamber were removed
using cotton wool, and the invading cells in the upper surface were
fixed with methanol at room temperature for 30 min and stained with
0.5% crystal violet. Images were captured at ×200 magnification,
and the cells were counted using a photomicroscope (Olympus
Corporation, Tokyo, Japan).
For the wound healing assay, at 48 h after
transfection, confluent monolayers of SCC9 cells cultured in
24-well plates (5×105 cells/ml) were mechanically
wounded using a 10-µl pipette tip. The wells were washed to remove
any cellular debris, and the cells were allowed to migrate for 24
h. Representative images were captured at ×100 magnification under
an inverted microscope (Olympus Corporation, Tokyo, Japan). The
experiments were repeated at least three times.
Cell apoptosis detection
Following treatment, OSCC cells were collected and
washed with cold PBS for at least three times. OSCC cell apoptosis
was then measured by a cell apoptosis assay. Briefly, OSCC cells
(1×106 cells/well) in a 6-well plate from different
groups were first resuspended in binding buffer, and then labeled
with Annexin V-FITC and propidium iodide (BD Pharmingen, San Diego,
CA, USA), in line with the manufacturer’s protocol. Flow cytometry
(BD FACSAria; BD Biosciences, Franklin Lakes, NJ, USA) was applied
to analyze the cell apoptosis. The experiment was repeated at least
three times.
Western blot analysis
Following treatment, total cellular proteins from
OSCC cells were extracted using radioim-munoprecipitation assay
buffer (OriGene Technologies, Inc., Beijing, China). A BCA protein
quantitative kit (Thermo Fisher Scientific, Inc.) was then used to
measure the concentration of protein samples. Next, equal amounts
of protein samples were resolved by 12% SDS-PAGE and then
transferred onto polyvinylidene difluoride membranes. The membranes
were blocked with 5% non-fat milk at room temperature for 1 h,
followed by overnight incubation at 4°C with primary antibodies,
including anti-CYC1 (ab224044; 1:1,000 dilution; Abcam, Cambridge,
UK), anti-Bcl-2 (no. 4223; 1:1,000 dilution; Cell Signaling
Technology, Inc., Danvers, MA, USA), anti-Bax (no. 5023; 1:1,000
dilution; Cell Signaling Technology, Inc.), anti-Caspase-3 (no.
9665; 1:1,000 dilution; Cell Signaling Technology, Inc.),
anti-Caspase-7 (no. 9492; 1:1,000 dilution; Cell Signaling
Technology; Inc.), anti-Caspase-9 (no. 9502; 1:1,000 dilution; Cell
Signaling Technology, Inc.), anti-cleaved Caspase-3 (no. 9664;
1:1,000 dilution; Cell Signaling Technology, Inc.), anti-cleaved
Caspase-7 (no. 9491; 1:1,000 dilution; Cell Signaling Technology,
Inc.), anti-cleaved Caspase-9 (no. 9505; 1:1,000 dilution; Cell
Signaling Technology, Inc.), and anti-MMP9 (no. 13667; 1:1,000
dilution; Cell Signaling Technology, Inc.). Subsequently, membranes
were incubated with a horseradish peroxidase-conjugated anti-rabbit
IgG secondary antibody (no. 7074; 1:5,000 dilution; Cell Signaling
Technology, Inc.) at room temperature for 2 h. To visualize the
protein blots, an ECL kit (Applygen Technologies, Inc., Beijing,
China) was used according the manufacturer’s protocol. Results were
quantified using Quantity One version 4.6 software (Bio-Rad
Laboratories, Inc., Hercules, CA, USA).
Statistical analysis
All data are displayed as the mean ± standard
deviation. SPSS statistical software (version 17.0; SPSS, Inc.,
Chicago, IL, United States) was performed for statistical analyses.
Comparison between groups was performed by using Student’s t-test
or analysis of variance. P<0.05 was considered to denote
differences that were statistically significant.
Results
Expression of lncRNA C5orf66-AS1 in SCC9
cells
The present study first detected the level of the
lncRNA C5orf66-AS1 in OSCC and adjacent normal tissues, as well as
in the OSCC cell line SCC9 and primary normal HOK cells, using
RT-qPCR. The results demonstrated that, compared with the adjacent
normal tissues, lncRNA C5orf66-AS1 expression was significantly
decreased in OSCC tissues (Fig.
1A). It was also observed that, compared with normal HOK cells,
the expression of lncRNA C5orf66-AS1 was significantly reduced in
SCC9 cells (Fig. 1B). Given the
limited understanding of the role of lncRNA C5orf66-AS1 in SCC9
cells, the cells were then transiently transfected with lncRNA
C5orf66-AS1-siRNA, the negative of lncRNA C5orf66-AS1-siRNA (NC) to
further examine the effect of this lncRNA on SCC9 cells, and cells
without any treatment were used as the control group. Besides,
lncRNA C5orf66-AS1-plasmid was also transfected into SCC9 cells and
empty plasmid as the control. The results indicated that the
expression of lncRNA C5orf66-AS1 was significantly increased in the
lncRNA C5orf66-AS1-plasmid-transfected cells, indicating successful
overexpression by plasmid transfection (Fig. 1C). However, in the lncRNA
C5orf66-AS1-siRNA group, the expression on lncRNA C5orf66-AS1 was
significantly reduced, indicating successful silencing by siRNA
transfection (Fig. 1D).
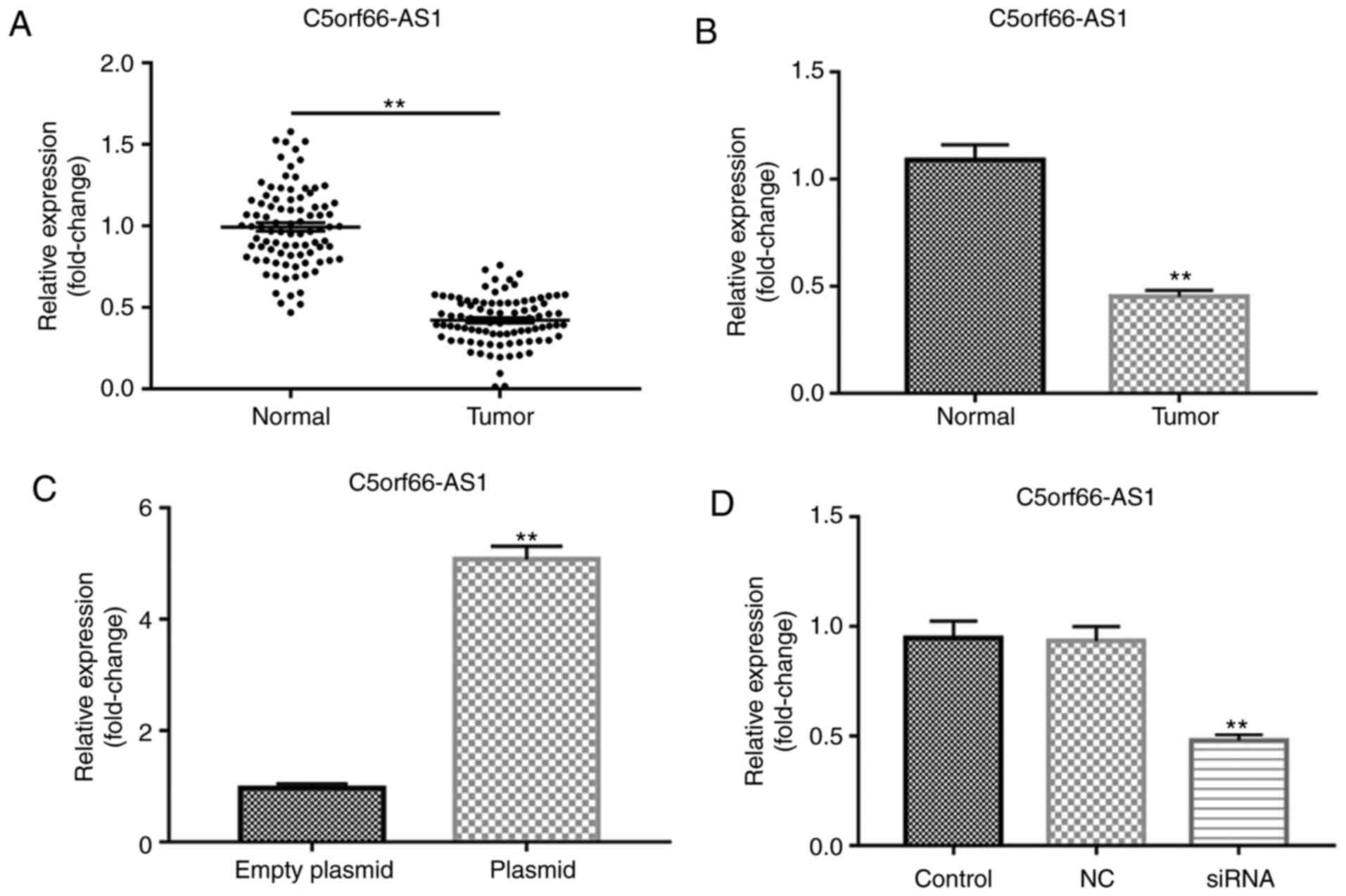 | Figure 1lncRNA C5orf66-AS1 expression in OSCC
tissues and cells was detected using RT-qPCR. Expression of lncRNA
C5orf66-AS1 in (A) OSCC and adjacent normal tissues, and (B) OSCC
cell line SCC9 and primary normal human oral keratinocyte cells.
Next, SCC9 cells were transfected with (C) lncRNA
C5orf66-AS1-plasmid (overexpression) or control plasmid, and with
(D) lncRNA C5orf66-AS1-siRNA (knockdown) or NC-siRNA. Cells without
any siRNA treatment were considered as the control group. After 48
h, lncRNA C5orf66-AS1 expression was detected using RT-qPCR.
**P<0.01 vs. corresponding control groups. OSCC, oral
squamous cell carcinoma; lncRNA, long non-coding RNAs; C5orf66-AS1,
C5orf66 antisense RNA 1; siRNA, small interfering RNA; NC, negative
control siRNA; RT-qPCR, reverse transcription-quantitative
polymerase chain reaction. |
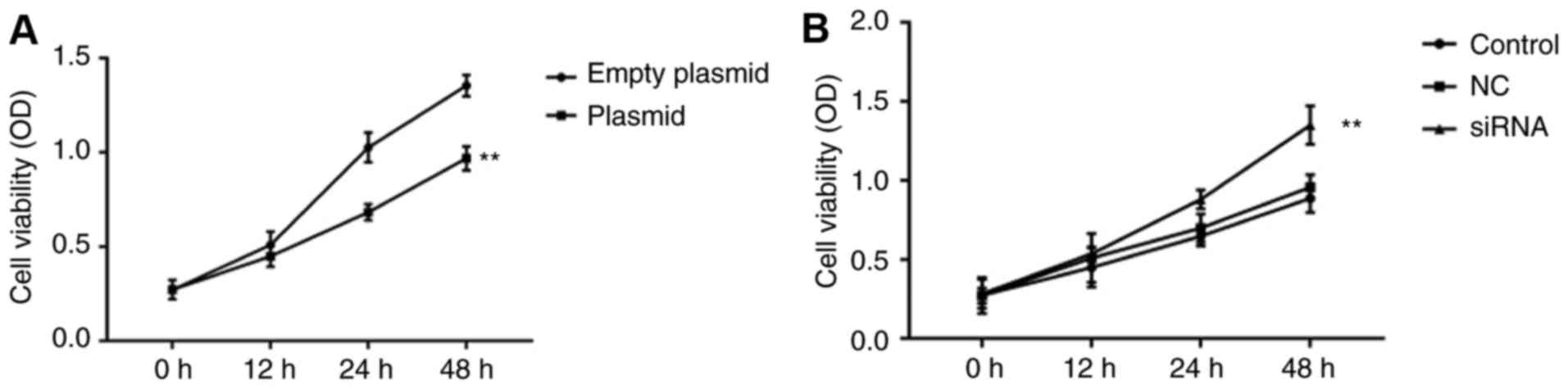
Effect of lncRNA C5orf66-AS1 on the
proliferation of SCC9 cells
Cells were transiently transfected with the negative
control, lncRNA C5orf66-AS1-siRNA, lncRNA C5orf66-AS1-plasmid and
empty plasmid serving as the control. Next, the cell proliferation
ability was detected using an MTT assay, and the results
demonstrated that lncRNA C5orf66-AS1-plasmid significantly
suppressed SCC9 cell proliferation (Fig. 2A), while lncRNA C5orf66-AS1-siRNA
increased the proliferation ability of SCC9 cells (Fig. 2B).
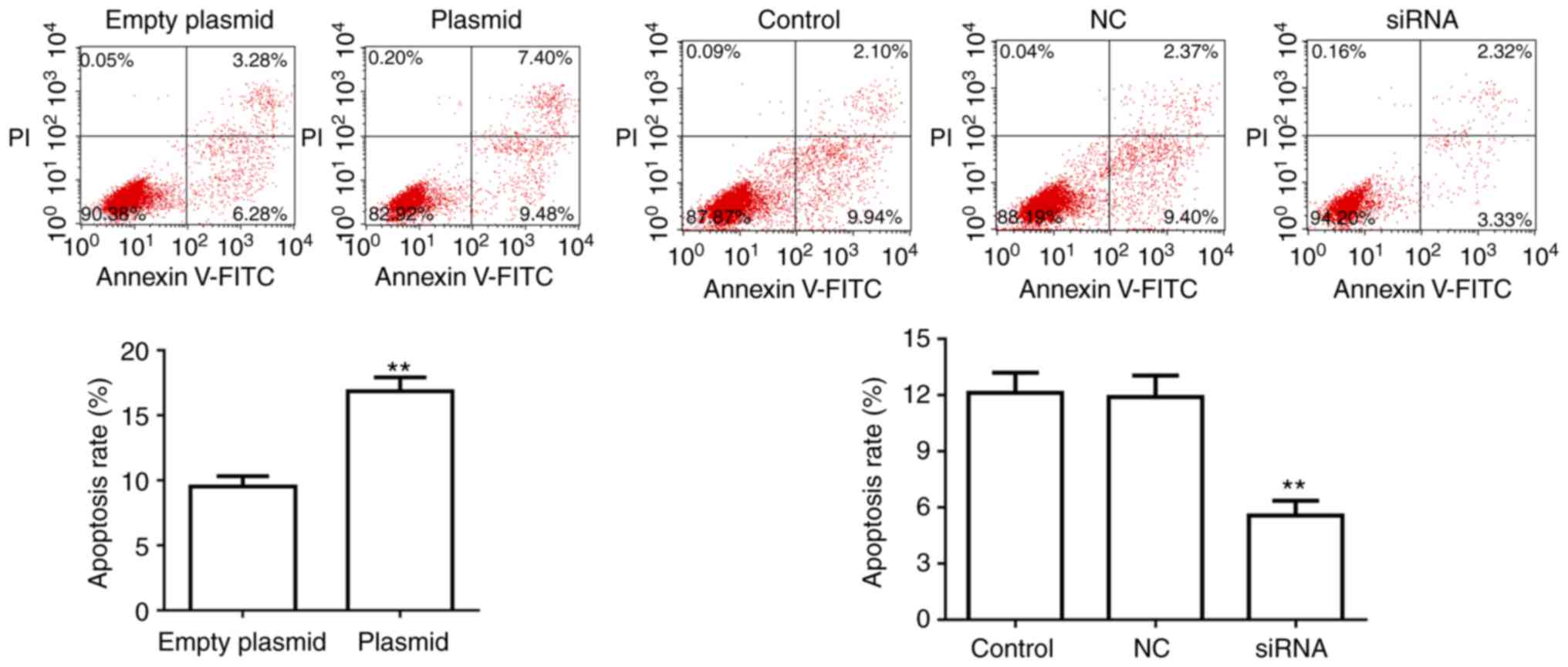
Effect of lncRNA C5orf66-AS1 on apoptosis
of SCC9 cells
SCC9 cell apoptosis was also detected at 48 h after
transfection. Flow cytometry analysis revealed that, compared with
the control groups, the apoptosis of SCC9 cells was significantly
upregulated in cells transfected with lncRNA C5orf66-AS1-plasmid.
By contrast, the apoptosis rate was markedly decreased in lncRNA
C5orf66-AS1-siRNA-transfected SCC9 cells compared with the control
cells (Fig. 3).
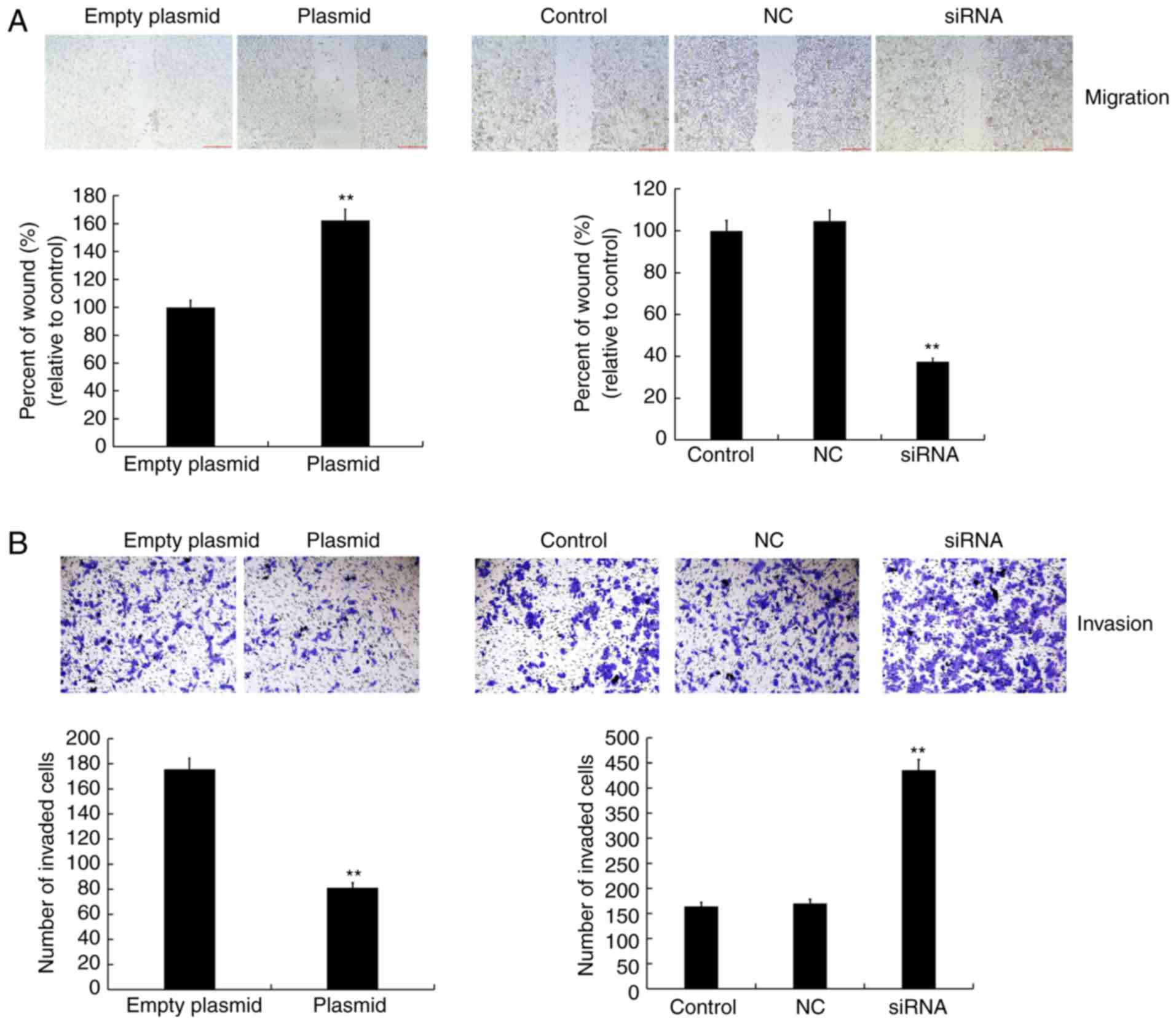
Effect of lncRNA C5orf66-AS1 on migration
and invasion abilities of SCC9 cells
A wound healing assay was conducted to detect cell
migration, while cell invasion was detected using a transwell
assay. It was observed that the cell migration and invasion
abilities were inhibited in the lncRNA C5orf66-AS1-plasmid
transfection group as compared with the control group. By contrast,
in the lncRNA C5orf66-AS1-siRNA transfection group, the cell
migration and invasion abilities were increased and there was no
statistically significant changes between the control group and the
NC group (Fig. 4).
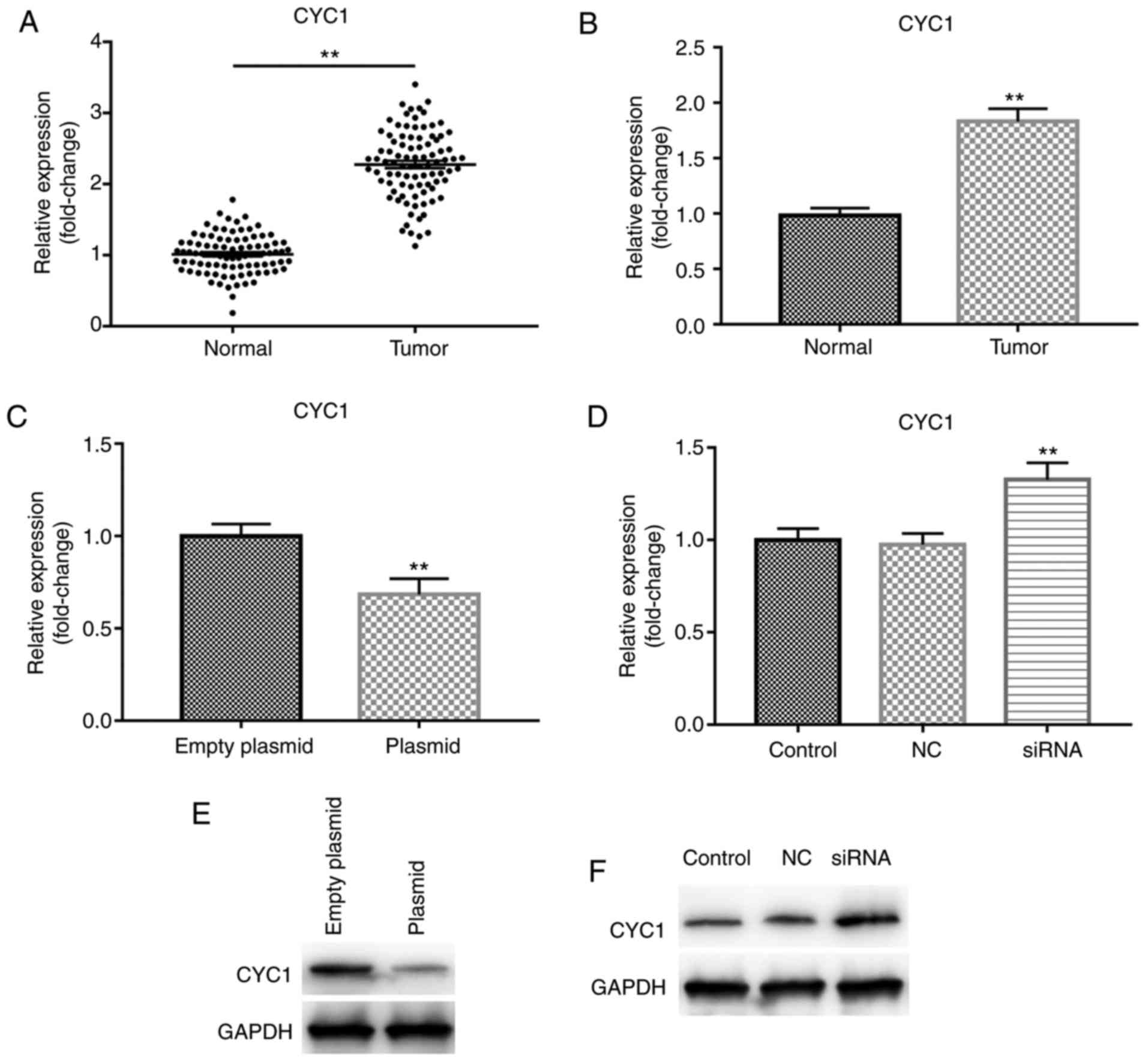
CYC1 is upregulated in OSCC tissues and
cells, and is downregulated by lncRNA C5orf66-AS1
overexpression
The mRNA level of CYC1 in OSCC and adjacent normal
tissues, and in the OSCC cell line SCC9 and primary normal HOK
cells was detected using RT-qPCR. The results indicated that,
compared with the adjacent normal tissues, CYC1 was significantly
increased in OSCC tissues (Fig.
5A). It was also demonstrated that, compared with HOK cells,
the expression of CYC1 was significantly increased in SCC9 cells
(Fig. 5B). Subsequently, to
explore the association between lncRNA C5orf66-AS1 and CYC1, SCC9
cells were trans-fected with the lncRNA C5orf66-AS1-plasmid or
lncRNA C5orf66-AS1-siRNA for 48 h and the mRNA level of CYC1 was
detected. The results revealed that overexpression of lncRNA
C5orf66-AS1 significantly decreased the mRNA level of CYC1, while
lncRNA C5orf66-AS1 silencing by siRNA transfection significantly
enhanced the mRNA level of CYC1 (Fig.
5C and D). Furthermore, the protein level of CYC1 in SCC9 cells
was detected following transfection with lncRNA C5orf66-AS1-plasmid
or -siRNA. As expected, overexpression of lncRNA C5orf66-AS1
downregulated the protein level of CYC1 in SCC9 cells, while
silencing of lncRNA C5orf66-AS1 significantly enhanced the protein
level of CYC1, as compared with the corresponding control groups
(Fig. 5E and F).
Effect of lncRNA C5orf66-AS1 on the
expression levels of associated genes in SCC9 cells
Subsequently, the effects of lncRNA C5orf66-AS1 on
the expression levels of genes associated with cell proliferation,
apoptosis and metastasis, including Bcl-2, Bax, cleaved
Caspase-3/7/9, Caspase-3/7/9 and MMP9, were investigated. The
results demonstrated that lncRNA C5orf66-AS1-plasmid transfection
significantly reduced the protein levels of Bcl-2 and MMP9, while
the expression levels of Bax, cleaved Caspase-3/Caspase-3, cleaved
Caspase-7/Caspase-7 and cleaved Caspase-9/Caspase-9 were
significantly increased (Fig.
6A). The mRNA levels of Bcl-2 and MMP9 were also reduced by
lncRNA C5orf66-AS1-plasmid transfection, while mRNA levels of Bax,
Caspase-3, Caspase-7 and Caspase-9 were enhanced (Fig. 6B-G). However, lncRNA
C5orf66-AS1-siRNA transfection significantly increased the protein
levels of Bcl-2 and MMP9, while it markedly decreased the
expression levels of Bax, cleaved Caspase-3/Caspase-3, cleaved
Caspase-7/Caspase-7, and cleaved Caspase-9/Caspase-9 (Fig. 7). Taken together, these data
indicated that lncRNA C5orf66-AS1 overexpression may induce SCC9
cell apoptosis through regulating the expression of
apoptosis-associated proteins (Bcl-2 and Bax) and increasing the
expression and activation of Caspase-3/7/9, while it may regulate
SCC9 cell migration and invasion by regulating MMP9 expression.
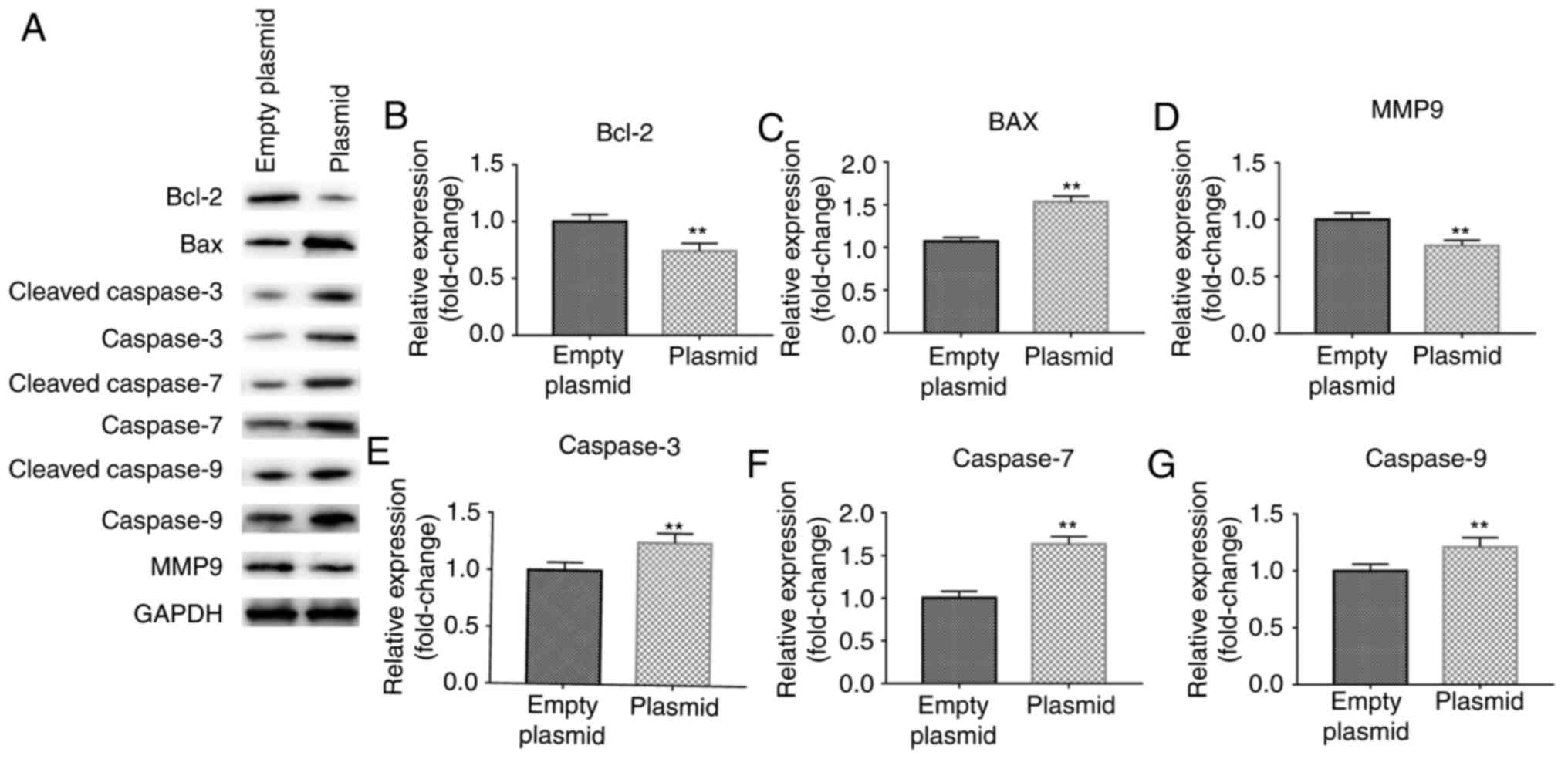 | Figure 6Effect of lncRNA C5orf66-AS1-plasmid
on Bcl-2, Bax, MMP9, Caspase-3, Caspase-7 and Caspase-9 expression
levels in SCC9 cells. (A) Western blot analysis was performed at 48
h after cell transfection to detect the protein levels. (B) Bcl-2,
(C) Bax, (D) MMP9, (E) cleaved Caspase-3/Caspase-3, (F) cleaved
Caspase-7/Caspase-7 and (G) cleaved Caspase-9/Caspase-9 in SCC9
cells were detected using reverse transcription-quantitative
polymerase chain reaction. **P<0.01 vs. control
groups. lncRNA, long non-coding RNAs; C5orf66-AS1, C5orf66
antisense RNA 1; Bcl-2, B-cell lymphoma 2; Bax, Bcl-2-associated X
protein; MMP9, matrix metalloproteinase 9. |
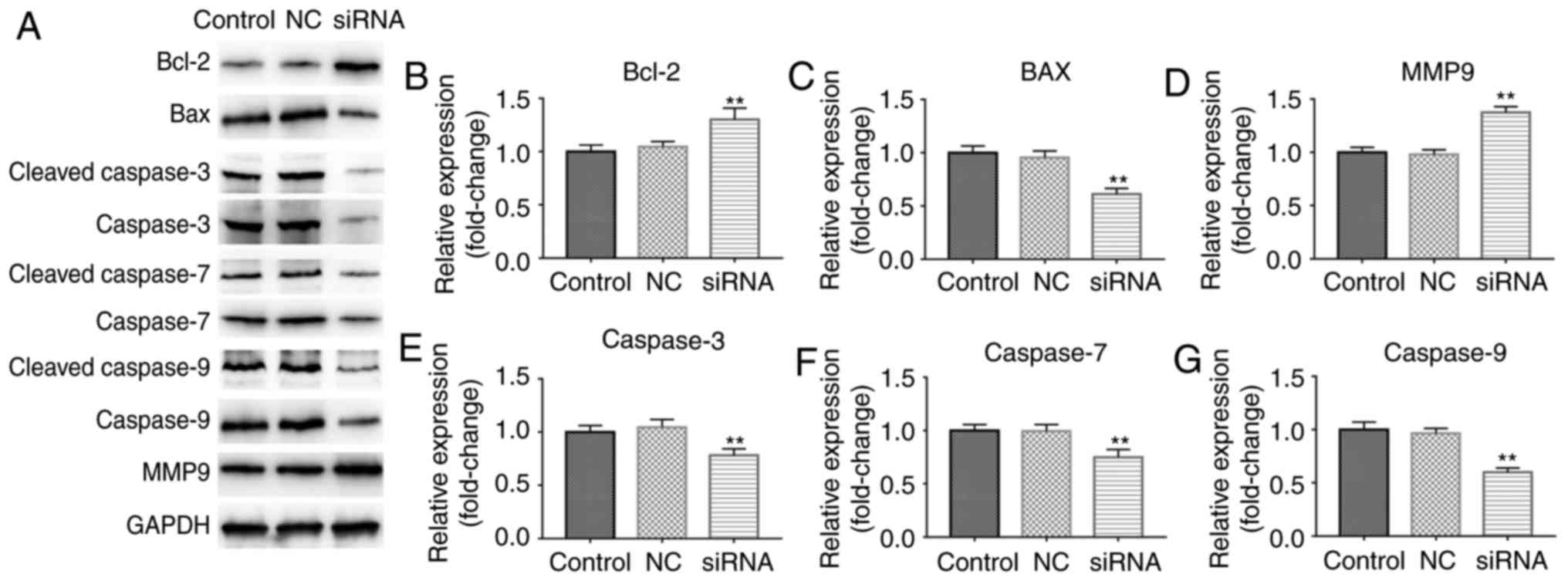 | Figure 7Effect of lncRNA C5orf66-AS1-siRNA on
Bcl-2, Bax, MMP9, Caspase-3, Caspase-7 and Caspase-9 expression
levels in SCC9 cells. (A) Western blot analysis was performed at 48
h after cell transfection to detect the protein levels. (B) Bcl-2,
(C) Bax, (D) MMP9, (E) cleaved Caspase-3/Caspase-3, (F) cleaved
Caspase-7/Caspase-7 and (G) cleaved Caspase-9/Caspase-9 in SCC9
cells were detected using transcription-quantitative polymerase
chain reaction. **P<0.01 vs. control groups. lncRNA,
long non-coding RNAs; C5orf66-AS1, C5orf66 antisense RNA 1; siRNA,
small interfering RNA; NC, negative control; Bcl-2, B-cell lymphoma
2; Bax, Bcl-2-associated X protein; MMP9, matrix metalloproteinase
9. |
Low expression of CYC1 reverses the
effect of lncRNA C5orf66-AS1-siRNA on the biological behavior of
SCC9 cells
Next, to explore whether CYC1 was involved in the
effect of lncRNA C5orf66-AS1 on SCC9 cells, the cells were
transiently transfected with lncRNA C5orf66-AS1-siRNA or lncRNA
C5orf66-AS1-siRNA + CYC1-siRNA. The findings suggested that lncRNA
C5orf66-AS1-siRNA transfection significantly enhanced the
proliferation, migration and invasion ability of SCC9 cells, and
inhibited cell apoptosis. However, these changes were significantly
reversed by CYC1-siRNA (Fig.
8).
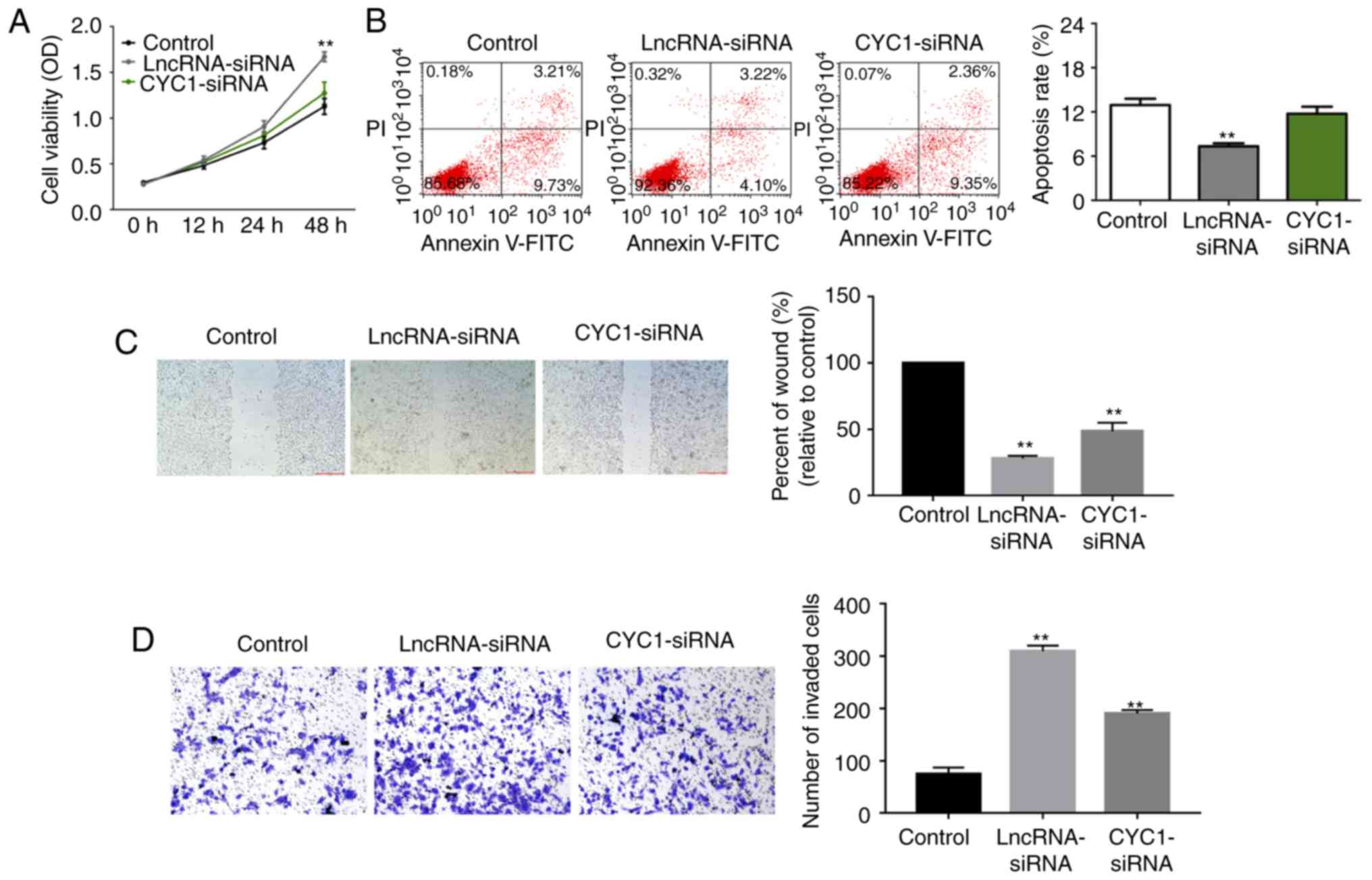 | Figure 8Effect of CYC1 on SCC9 cells. At 48 h
after cell transfection with lncRNA C5orf66-AS1-siRNA or lncRNA
C5orf66-AS1-siRNA + CYC1-siRNA, the (A) proliferation, (B)
apoptosis, (C) migration and (D) invasion of SCC9 cells were
determined using an MTT assay, flow cytometry, wound healing assay
and transwell assay, respectively. Wound closure was quantified and
the number of invasive cells was counted. **P<0.01
vs. control groups. Magnification, ×100 (migration) and ×200
(invasion). lncRNA, long non-coding RNAs; C5orf66-AS1, C5orf66
antisense RNA 1; siRNA, small interfering RNA; NC, negative
control; CYC1, cytochrome c1. |
Discussion
OSCC is a malignant tumor with poor prognosis that
occurs in the head and neck region, and accounts for 90% of oral
cancer cases (26). Despite the
current advances in treatment strategies, the 5-year and 10-year
survival rates of patients with OSCC have not significantly
improved; therefore, OSCC severely impacts human health (27). Previous studies have demonstrated
that dysregulation of oncogenes and tumor suppressor genes is an
important factor in tumorigenesis and development (28-30). Previous studies have reported that
lncRNAs have a complex biological function, are able to regulate
the progression of a variety of diseases, and they are closely
associated with tumor cell proliferation and metastasis (31). There are a large number of lncRNAs
with an unknown function that have yet to be studied. While the
genome-wide study of lncRNAs is a good approach to unveil more
lncRNAs that may be involved in carcinogenesis or may represent
potential therapeutic targets, performing direct comparisons of
results from various studies remains challenging.
To the best of our knowledge, the present study is
the first to evaluate the role of lncRNA C5orf66-AS1 in OSCC.
First, the expression levels of lncRNA C5orf66-AS1 in OSCC and
adjacent normal tissues, as well as in the OSCC cell line SCC9 and
a normal oral cell line, were compared. The findings of the current
study suggested that lncRNA C5orf66-AS1 was significantly
downregulated in OSCC. Next, in order to assess the effect of
lncRNA C5orf66-AS1 on OSCC cells, lncRNA C5orf66-AS1 was
upregulated or downregulated in SCC9 cells through transfection
with lncRNA C5orf66-AS1-plasmid or -siRNA, respectively. The
results revealed that overexpression of lncRNA C5orf66-AS1 in OSCC
significantly inhibited cell proliferation, invasion and migration,
and promoted apoptosis, while lncRNA C5orf66-AS1 downregulation
presented the opposite effects. These results suggested that lncRNA
C5orf66-AS1 inhibits the malignant behavior of SCC9 cells,
providing a basis for the treatment of OSCC.
CYC1 is an important subunit of mitochondrial
complex III. It is well known that mitochondria are indispensable
for energy metabolism. Approximately 90% of cellular adenosine
triphosphate is generated in mitochondria through the oxidative
phosphorylation pathway. Recent studies indicated that CYC1 serves
a key role in the development of tumors, including breast cancer
and osteosarcoma among others (32,33). To the best of our knowledge, the
role of CYC1 in the progression of OSCC remains unclear. In the
present study, it was observed that CYC1 was upregulated in OSCC
tissues and cells. In addition, the data revealed that
overexpression of lncRNA C5orf66-AS1 in OSCC cells inhibited the
expression of CYC1, while knockdown of lncRNA C5orf66-AS1 in SCC9
cells promoted the expression of CYC1. This process may involve the
mitochondrial pathway; however, the specific mechanism involved is
not clear. Furthermore, it was observed that CYC1 silencing
partially offset the promotion of lncRNA C5orf66-AS1 silencing on
OSCC cells.
In conclusion, this is the first study to report
that lncRNA C5orf66-AS1 was downregulated in OSCC, and
overexpres-sion of lncRNA C5orf66-AS1 was able to prevent OSCC
progression by inhibiting OSCC cell growth and metastasis via
regulating CYC1 expression. Therefore, lncRNA C5orf66-AS1 may be a
novel and promising target for OSCC treatment.
Funding
No funding was received.
Availability of data and materials
The analyzed data sets generated during the present
study are available from the corresponding author on reasonable
request.
Authors’ contributions
TL designed the study. TL and HL were responsible
for data access and analysis. GY interpreted results. All authors
collaborated to develop the manuscript.
Ethics approval and consent to
participate
The present study was approved by the Human Ethics
Committee Review Board at the Stomatological Hospital of Guizhou
Medical University (Guiyang, China). Informed consent was provided
by each patient.
Patient consent for publication
All patients provided consent for publication.
Competing interests
The authors declare no competing interests.
Acknowledgements
Not applicable.
References
|
1
|
Ryerson AB, Eheman CR, Altekruse SF, Ward
JW, Jemal A, Sherman RL, Henley SJ, Holtzman D, Lake A, Noone AM,
et al: Annual report to the nation on the status of cancer,
1975–2012 featuring the increasing incidence of liver cancer.
Cancer. 122:1312–1337. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Ferlay J, Soerjomataram I, Dikshit R, Eser
S, Mathers C, Rebelo M, Parkin DM, Forman D and Bray F: Cancer
incidence and mortality worldwide: Sources, methods and major
patterns in GLOBOCAN 2012. Int J Cancer. 136:E359–E386. 2015.
View Article : Google Scholar
|
|
3
|
Gershenwald JE, Soong SJ and Balch CM;
American Joint Committee on Cancer (AJCC) Melanoma Staging
Committee: 2010.TNM staging system for cutaneous melanoma…and
beyond. Ann Surg Oncol. 17:1475–1477. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Kreppel M, Drebber U, Rothamel D, Eich HT,
Kübler A, Scheer M and Zöller JE: Prognostic impact of different
TNM-based stage groupings for oral squamous cell carcinoma. Head
Neck. 33:1467–1475. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Kolk A, Jubitz N, Mengele K, Mantwill K,
Bissinger O, Schmitt M, Kremer M and Holm PS: Expression of
Y-box-binding protein YB-1 allows stratification into long- and
short-term survivors of head and neck cancer patients. Br J Cancer.
105:1864–1873. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Götz C, Drecoll E, Straub M, Bissinger O,
Wolff KD and Kolk A: Impact of HPV infection on oral squamous cell
carcinoma. Oncotarget. 7:76704–76712. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Scully C and Bagan J: Oral squamous cell
carcinoma overview. Oral Oncol. 45:301–308. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Davidovich C and Cech TR: The recruitment
of chromatin modifiers by long noncoding RNAs: Lessons from PRC2.
RNA. 21:2007–2022. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Rogers SN, Brown JS, Woolgar JA, Lowe D,
Magennis P, Shaw RJ, Sutton D, Errington D and Vaughan D: Survival
following primary surgery for oral cancer. Oral Oncol. 45:201–211.
2009. View Article : Google Scholar
|
|
10
|
Nagtegaal ID, Quirke P and Schmoll HJ: Has
the new TNM classification for colorectal cancer improved care. Nat
Rev Clin Oncol. 9:119–123. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Piazzolla D, Palla AR, Pantoja C, Cañamero
M, de Castro IP, Ortega S, Gómez-López G, Dominguez O, Megías D,
Roncador G, et al: Lineage-restricted function of the pluripotency
factor NANOG in stratified epithelia. Nat Commun. 5:42262014.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Balch C and Nephew KP: The role of
chromatin, microRNAs, and tumor stem cells in ovarian cancer.
Cancer Biomark. 8:203–221. 2010–2011. PubMed/NCBI
|
|
13
|
Sun T: Long noncoding RNAs act as
regulators of autophagy in cancer. Pharmacol Res. 129:151–155.
2018. View Article : Google Scholar
|
|
14
|
Carvalho AL, Nishimoto IN, Califano JA and
Kowalski LP: Trends in incidence and prognosis for head and neck
cancer in the United States: A site-specific analysis of the SEER
database. Int J Cancer. 114:806–816. 2005. View Article : Google Scholar
|
|
15
|
Gutschner T and Diederichs S: The
hallmarks of cancer: A long non-coding RNA point of view. RNA Biol.
9:703–719. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Min A, Zhu C, Peng S, Rajthala S, Costea
DE and Sapkota D: MicroRNAs as important players and biomarkers in
oral carcinogenesis. Biomed Res Int. 2015.186904:2015.
|
|
17
|
Feng L, Houck JR, Lohavanichbutr P and
Chen C: Transcriptome analysis reveals differentially expressed
lncRNAs between oral squamous cellcarcinoma and healthy oral
mucosa. Oncotarget. 8:31521–31531. 2017.PubMed/NCBI
|
|
18
|
Maruyama R and Suzuki H: Long noncoding
RNA involvement in cancer. BMB Rep. 45:604–611. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Quinn JJ and Chang HY: Unique features of
long non-coding RNA biogenesis and function. Nat Rev Genet.
17:47–62. 2016. View Article : Google Scholar
|
|
20
|
Bartonicek N, Maag JL and Dinger ME: Long
noncoding RNAs in cancer: Mechanisms of action and technological
advancements. Mol Cancer. 15:432016. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Yang Q, Xu E, Dai J, Liu B, Han Z, Wu J,
Zhang S, Peng B, Zhang Y and Jiang Y: A novel long noncoding RNA
AK001796 acts as an oncogene and is involved in cell growth
inhibition by resveratrol in lung cancer. Toxicol Appl Pharmacol.
285:79–88. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Clemson CM, Hutchinson JN, Sara SA,
Ensminger AW, Fox AH, Chess A and Lawrence JB: An architectural
role for a nuclear noncoding RNA: NEAT1 RNA is essential for the
structure of paraspeckles. Mol Cell. 33:717–726. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Nie W, Ge HJ, Yang XQ, Sun X, Huang H, Tao
X, Chen WS and Li B: LncRNA-UCA1 exerts oncogenic functions in
non-small cell lung cancer by targeting miR-193a-3p. Cancer Lett.
371:99–106. 2016. View Article : Google Scholar
|
|
24
|
Peng W, Si S, Zhang Q, Li C, Zhao F, Wang
F, Yu J and Ma R: Long non-coding RNA MEG3 functions as a competing
endogenous RNA to regulate gastric cancer progression. J Exp Clin
Cancer Res. 34:792015. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(−Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar
|
|
26
|
SHahinas J and Hysi D: Methods and risk of
bias in molecular marker prognosis studies in oral squamous cell
carcinoma. Oral Dis. 24:115–119. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Peurala E, Tuominen M, Löyttyniemi E,
Syrjänen S and Rautava J: Eosinophilia is a favorable prognostic
marker for oral cavity and lip squamous cell carcinoma. APMIS.
126:201–207. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Lee EY and Muller WJ: Oncogenes and tumor
suppressor genes. Cold Spring Harb Perspect Biol. 2:a0032362010.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Iurlaro R, León-Annicchiarico CL and
Muñoz-Pinedo C: Regulation of cancer metabolism by oncogenes and
tumor suppressors. Methods Enzymol. 542:59–80. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Wang D, Qiu C, Zhang H, Wang J, Cui Q and
Yin Y: Human microRNA oncogenes and tumor suppressors show
significantly different biological patterns: From functions to
targets. PLoS One. 5:e130672010. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Geisler S and Coller J: RNA in unexpected
places: Long non-coding RNA functions in diverse cellular contexts.
Nat Rev Mol Cell Biol. 14:699–712. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Han Y, Sun S, Zhao M, Zhang Z, Gong S, Gao
P, Liu J, Zhou J, Ma D, Gao Q and Wu P: CYC1 predicts poor
prognosis in patients with breast cancer. Dis Markers.
2016.3528064:2016.
|
|
33
|
Li G, Fu D, Liang W, Fan L, Chen K, Shan
L, Hu S, Ma X, Zhou K and Cheng B: CYC1 silencing sensitizes
osteosarcoma cells to TRAIL-induced apoptosis. Cell Physiol
Biochem. 34:2070–2080. 2014. View Article : Google Scholar
|