Introduction
Periodontal diseases, periapical diseases, jaw
osteomyelitis, and oral and maxillofacial tumors can cause alveolar
bone tissue loss, which eventually affects chewing function
(1-3). Therefore, there is much interest in
the use of periodontal tissue engineering to repair bone defects
and promote bone regeneration. Human periodontal ligament stem
cells (hPDLSCs) are a subtype of mesenchymal stem cells (MSCs) that
possess multi-potency to differentiate into cells of chondrogenic,
osteogenic, adipogenic and neurogenic lineages in vitro
(4,5). These cells have been shown to have
pluripotent capacity to differentiate into osteoblasts, osteocytes
and cementoblasts, and have been used in the regeneration of
periodontal tissue (6-8). Therefore, hPDLSCs are a promising
cell population for periodontal tissue engineering. It is necessary
to have a deep understanding of the underlying regulatory
mechanisms that regulate target stem cell proliferation and
differentiation prior to practical applications.
1α, 25-dihydroxyvitamin D3 (1,25-D3), an active form
of vitamin D, is one of the key factors that regulates bone
metabolism. 1,25-D3 functions mainly through binding to its nuclear
vitamin D receptor (VDR) (9).
1,25-D3 and VDR are essential for regulation of the osteogenic
differentiation of stem/progenitor cells (10,11). Studies have shown that 1,25-D3 can
stimulate the mineralization of human osteoblasts and induce the
osteogenic differentiation of human MSCs (12-16). Similarly, increasing evidence
suggests that 1,25-D3 is essential in promoting osteogenic activity
in hPDLSCs (17,18). Further investigations are required
to understand the specific regulatory molecular mechanisms of
1,25-D3-induced osteogenic differentiation in hPDLSCs.
The Hippo signaling pathway was initially found to
be implicated in organ size control, and subsequent studies have
found that the Hippo signaling pathway is highly conserved and may
have a similar important role in the context of tissue regeneration
(19-21). Transcriptional coactivator with
PDZ-binding motif (TAZ) is a key mediator of Hippo signaling that
regulates stem cell self-renewal and differentiation in different
contexts (22). Similar to other
transcriptional coactivators, TAZ shuttles between the cytoplasm
and the nucleus in response to different signaling molecules;
through this translocation, TAZ regulates its potential target gene
(23). It has been reported that
TAZ is a critical mediator for regulating MSC differentiation
towards osteoblasts (24,25).
As TAZ has an important function during the
osteogenic differentiation of stem cells, it may be possible to
capitalize on its osteogenic role in stem cell differentiation to
enhance bone regeneration. TAZ-mediated activation is involved in
osteoblast differentiation in various cell types upon stimulation
with growth factors, cytokines and/or chemical compounds (26-29). Cross-talk between 1,25-D3 and TAZ
may also represent an important mechanism to mediate the
tissue-specific expression of osteogenic genes. Therefore, the
present study aimed to investigate whether the effect of 1,25-D3 on
the osteogenic differentiation of hPDLSCs involves the action of
TAZ.
Materials and methods
Isolation and culture of hPDLSCs
The present study was approved by the Medical
Ethical Committee of the School of Stomatology, Shandong University
(Shandong, China; protocol no. 20170303). It has been shown that
aging has significantly negative effects on hPDLSC proliferation
and differentiation (2).
Therefore, a relative narrow age range of subjects (12-16 years
old) was selected to avoid the effects of aging on PDLSCs. Prior to
commencement of the study, the patients and their parents were
informed verbally and in writing. The parents provided written
informed consent in accordance with the Declaration of Helsinki.
The isolation and culture methods were as reported previously
(30,31). Briefly, 10 premolars, extracted
for orthodontic reasons from four otherwise healthy patients at the
Stomatological Hospital of Shandong University (two girls and two
boys, the girls are 13 and 15 years old and underwent teeth
extraction in May 2017, the boys are 12 and 16 years old and
underwent teeth extraction in October 2017), were used for cell
isolation. The harvested teeth were placed in α-MEM (Gibco; Thermo
Fisher Scientific, Inc., Waltham, MA, USA) with penicillin (400
U/ml, Gibco; Thermo Fisher Scientific, Inc.) and streptomycin (400
mg/ml, Gibco; Thermo Fisher Scientific, Inc.) on ice, and were
transported to the laboratory immediately. Human periodontal
ligament tissue from the middle third of the tooth root was scraped
off and minced into small pieces with an aseptic scalpel. The
minced tissues were incubated with collagenase type I (3 mg/ml,
Sigma-Aldrich; Merck KGaA, Darmstadt, Germany) and dispase (4
mg/ml, Sigma-Aldrich; Merck KGaA) in α-MEM at 37˚C for 1 h,
followed by filtering through a 70-µm cell strainer to
obtain a single cell suspension. The cells were grown in α-MEM
supplemented with 10% fetal bovine serum (FBS; Gibco; Thermo Fisher
Scientific, Inc.), 2 mM L-glutamine, 100 U/ml penicillin and 100
mg/ml streptomycin in an incubator at 37°C and 5% CO2.
Cells at passages 3-5 were used.
Phenotype analysis of isolated hPDLSCs by
flow cytometry
After being trypsinized and washed with
phosphate-buffered saline (PBS; Thermo Fisher Scientific, Inc.),
the cells were incubated with fluorescent dye-conjugated monoclonal
antibodies at 4°C for 20 min in the dark, followed by analysis
using a BD FACSCalibur flow cytometer (BD Biosciences, Franklin
Lakes, NJ, USA) with a BD Stemflow™ hMSC Analysis kit. The
following antibodies (included in the kit) were used:
hMSC+ cocktail (CD90-FITC, CD105-PerCP-Cy5.5, CD73-APC
and CD44-PE), and hMSC− cocktail (CD34-PE, CD11b-PE,
CD19-PE, CD45-PE and HLA-DR-PE). Non-specific staining was
controlled using isotype-matched antibodies following the
manufacturer’s protocol (BD Biosciences).
Multipotent differentiation of
hPDLSCs
The hPDLSCs were cultured in 6-well dishes at
1×105 cells/well. At 90% confluence, the culture medium
was replaced according to the type of differentiation. For
osteogenic differentiation, the cells were exposed to osteogenic
induction medium (OIM) comprising α-MEM supplemented with 10% FBS),
100 U/ml penicillin G, 0.1 mg/ml streptomycin, 10 nmol/l
dexamethasone (Beijing Solarbio Science & Technology Co., Ltd.,
Beijing, China), 10 mmol/l β-glycerophosphate (Beijing Solarbio
Science & Technology Co., Ltd.), and 50 mg/l ascorbic acid
(Beijing Solarbio Science & Technology Co., Ltd.). The medium
was replaced every 3 days. Following 4 weeks of induction, the
formation of mineralized nodules was stained with Alizarin Red (AR;
Sigma-Aldrich; Merck KGaA) for 5 min at room temperature and
detected by an inverted microscope (Olympus Corp., Tokyo,
Japan).
For adipogenic differentiation, the cells were
exposed to adipogenic induction medium comprising α-MEM containing
10% FBS, 100 U/ml penicillin G, 0.1 mg/ml streptomycin, 2
µmol/l dexamethasone (Beijing Solarbio Science &
Technology Co., Ltd.), 0.2 mmol/l indomethacin (Sigma-Aldrich;
Merck KGaA), 0.01 g/l insulin (Sigma-Aldrich; Merck KGaA) and 0.5
mmol/l isobutyl-methylxanthine (Sigma-Aldrich; Merck KGaA).
Following 4 weeks of induction, the lipid droplets in the cells
were stained with Oil Red O (Cyagen Biosciences, Suzhou, China) for
30 min at room temperature and detected by an inverted microscope
(Olympus Corp.).
For chondrogenic differentiation, the cells were
exposed to chondrogenic induction medium containing 2 ng/ml
transforming growth factor-β1 (R&D Systems, Inc., Minneapolis,
MN, USA), 50 mg/ml L-ascorbic acid, 100 mg/ml sodium pyruvate
(Sigma Aldrich; Merck KGaA) and 100 nM dexamethasone (Beijing
Solarbio Science & Technology Co., Ltd.). After 4 weeks,
chondrogenic differentiation was assessed via Alcian Blue (Beijing
Solarbio Science & Technology Co., Ltd.) staining for 5 min at
room temperature and detected by an inverted microscope (Olympus
Corp.).
Detection of the expression of VDR in
hPDLSCs by immunofluorescence
hPDLSCs were plated on 24-well chamber slides at a
density of 5,000 cells/well and then treated with or without 10 nM
1,25-D3 (Sigma-Aldrich; Merck KGaA) for 48 h at 37°C. The hPDLSCs
were fixed in 4% paraformaldehyde (Beijing Solarbio Science &
Technology Co., Ltd.) for 30 min at room temperature, permeabilized
with 0.1% Triton X-100 (Beijing Solarbio Science & Technology
Co., Ltd.) for 10 min and washed with PBS. The slides were then
blocked in goat serum (Cell Signaling Technology, Inc.) for 60 min
at room temperature, followed by incubation with primary mouse
anti-human VDR monoclonal antibody (1:100; cat. no. sc-13133; Santa
Cruz Biotechnology, Inc., Dallas, TX, USA) at 4°C overnight.
Following washing with PBS, the cells were incubated with goat
anti-mouse secondary antibody (1:200; cat. no. SP-9000; OriGene
Technologies, Inc., Beijing, China) for 1 h in the dark at room
temperature. The cells were then stained with
4′,6-diamidino-2-phenylindole (Cell Signaling Technology, Inc.) for
5 min at room temperature and viewed under a fluorescence
microscope (Olympus Corp.).
Proliferation assay of hPDLSCs
The cell proliferation ability of the hPDLSCs under
different conditions was assayed using a Cell Counting Kit-8
(CCK-8; Dojindo Molecular Technologies, Inc., Kumamoto, Japan). The
cells were seeded in 96-well plates with 3,000 cells/well and were
treated with different concentrations of 1,25-D3 (0, 0.1, 1 and 10
nM). The culture media was refreshed every 48 h for each media
concentration. Every 24 h, 10 µl CCK-8 reagent was added to
the wells. Following a 1.5-h incubation period, the plates were
measured for spectrophotometric absorbance at 450 nm. Cell
proliferation was also evaluated by western blotting to detect
changes in the expression of proliferating cell nuclear antigen
(PCNA), a marker essential for cell proliferation (32), following incubation with or
without 1,25-D3 for 72 h.
Osteogenic differentiation of
hPDLSCs
The hPDLSCs (1×105 cells/well) were
seeded in 6-well plates and cultured in OIM with or without 1,25-D3
(10 nM). For alkaline phosphatase (ALP) staining, the cells were
cultured for 7 days. The cells were then washed with PBS and fixed
with 4% paraformaldehyde at 4°C for 30 min. A BCIP/NBT alkaline
phosphatase staining kit (Beyotime Institute of Biotechnology,
Haimen, China) was used for ALP staining, and the stained samples
were then observed with an inverted microscope (Olympus Corp.). For
the AR staining, the hPDLSCs were stained with 1% AR for 5 min at
room temperature on day 14. The osteogenesis-related proteins,
including ALP and osteopontin (OPN), were also detected by western
blotting.
Viral transfection and establishment of
TAZ-overexpressing stable cell clones of hPDLSCs
Lentiviral vectors overexpressing TAZ were
constructed and produced by Shanghai Genechem Company (Shanghai,
China). At the third passage, hPDLSCs were seeded in 6-well plates
(0.5×105 cells/well) and cultured to 40% confluence; the
cells were then transfected with culture medium containing 8
µg/ml Polybrene (Shanghai Genechem Company) and 100
µl TAZ-overexpression lentivirus particle LV5-homo-TAZ
(NM_000116.4) (109/ml) as the TAZ-overexpression group.
hPDLSCs transfected with 100 µl empty vector lentivirus
LV5-Nc (NM_000116.4) (109/ml) and 8 µg/ml
Polybrene were used as controls. The transfection efficiency of TAZ
was measured by reverse transcription-quantitative polymerase chain
reaction (RT-qPCR) and western blot analyses after 72 h of
transfection. All experiments were performed in triplicate and
repeated three times.
RT-qPCR analysis
TRIzol (1 ml) was added to the wells and total RNA
was extracted from the cells according to the manufacturer’s
protocol (Invitrogen; Thermo Fisher Scientific, Inc.). The total
RNA (1 µg) was reverse transcribed to cDNA using a
SuperScript™ II Reverse Transcriptase kit (Invitrogen; Thermo
Fisher Scientific, Inc.). The temperature protocol was as follows:
42°C for 2 min, and 4°C for 30 min; followed by 37°C for 15 min at
85°C for 5 sec, and 4°C for 30 min. These generated cDNA samples
were then amplified with RT-qPCR in 20 µl of the reaction
system, which contained the 10 µl SYBR® Primix Ex
Taq™ (Takara Bio, Inc., Otsu, Japan), 2 µl cDNA, 0.4
µl each primer and 7.2 µl RNase-free H2O,
following the manufacturer’s protocol. RT-qPCR analysis was
performed on a Roche Light Cycler® 480 (Hoffmann-La
Roche Ltd., Basel, Switzerland). The thermal cycling conditions
were as follows: 95°C for 30 sec, followed by 40 cycles of 95°C for
5 sec and 60°C for 20 sec. The relative gene expression was
calculated using the 2−ΔΔCq method (33), normalizing with GADPH levels. The
primers used were as follows: GAPDH, forward 5′-GCA CCG TCA AGG CTG
AGA AC-3′ and reverse 5′-TGG TGA AGA CGC CAG TGG A-3′; TAZ, forward
5′-CCT CTT CAA TGA TGT AGA GTC TGC-3′ and reverse 5′-AGT GAT TAC
AGC CAG GTT AGA AAG-3′.
Total protein isolation and western
blotting
The primary monoclonal antibodies used included
rabbit anti-human TAZ (cat. no. cst70148; Cell Signaling
Technology, Inc.; 1:1,000), rabbit anti-human ALP (cat. no.
ab108337; Abcam, Cambridge, UK; 1:20,000), mouse anti-human OPN
(cat. no. sc-21742; Santa Cruz Biotechnology, Inc; 1:500), rabbit
anti-human PCNA (cat. no. WL0341c; Wanlei Biotechnology Co., Ltd.,
Shanghai, China; 1:1,000), and anti-human GAPDH monoclonal (cat.
no. HRP-60004, ProteinTech Group, Inc., Chicago, IL, USA;
1:20,000). The cells were lysed in radioimmunoprecipitation assay
buffer (Beijing Solarbio Science & Technology Co., Ltd.)
containing 1% phenylmethylsulfonyl fluoride (Beijing Solarbio
Science & Technology Co., Ltd.). The protein concentration was
quantified using the bicin-choninic acid (Beijing Solarbio Science
& Technology Co., Ltd.) method. Subsequently, 20 µg of
protein were separated electrophoretically with 10% SDS-PAGE and
was then elec-troblotted onto a PVDF membrane (Bio-Rad
Laboratories, Inc., Hercules, CA, USA). Following blocking with
nonfat milk (5%; Beijing Solarbio Science & Technology Co.,
Ltd.), the membranes were incubated with the primary monoclonal
antibodies against TAZ, ALP, OPN or GAPDH (loading control) at 4°C
overnight. The cells were then labeled with horseradish peroxidase
(HRP)-labeled goat anti-mouse IgG (cat. no. 7076S) or goat
anti-rabbit IgG (cat. no. 7074S) from Santa Cruz Biotechnology,
Inc. The protein bands were detected using the chemiluminescent HRP
substrate (EMD Millipore, Billerica, MA, USA) and protein levels
were analyzed using ImageJ 1.47V software (National Institutes of
Health, Bethesda, MD, USA).
Statistical analysis
All experiments were repeated at least three times
and data are presented as the mean ± standard error of mean. Data
were statistically analyzed using GraphPad Prism 5 (GraphPad
Software, Inc., La Jolla, CA, USA) with Student’s two-tailed t-test
for two groups of data or one-way analysis of variance with Tukey’s
post hoc test for multiple groups of data. P<0.05 was considered
to indicate a statistically significant difference.
Results
Characterization of hPDLSCs
The hPDLSCs exhibited a typical spindle-shaped
morphology (Fig. 1A). In flow
cytometric analysis, the hPDLSCs negatively expressed CD34, CD11b,
CD19, CD45, HLA-DR, but positively expressed CD90, CD44, CD105,
CD73 (Fig. 1B). This indicated
that hPDLSCs exhibit a similar phenotypic characterization as MSCs.
Furthermore, the hPDLSCs demonstrated multi-potency, as evidenced
by the formation of mineralized nodules, lipid droplets and
cartilage following induction (Fig.
1C-E).
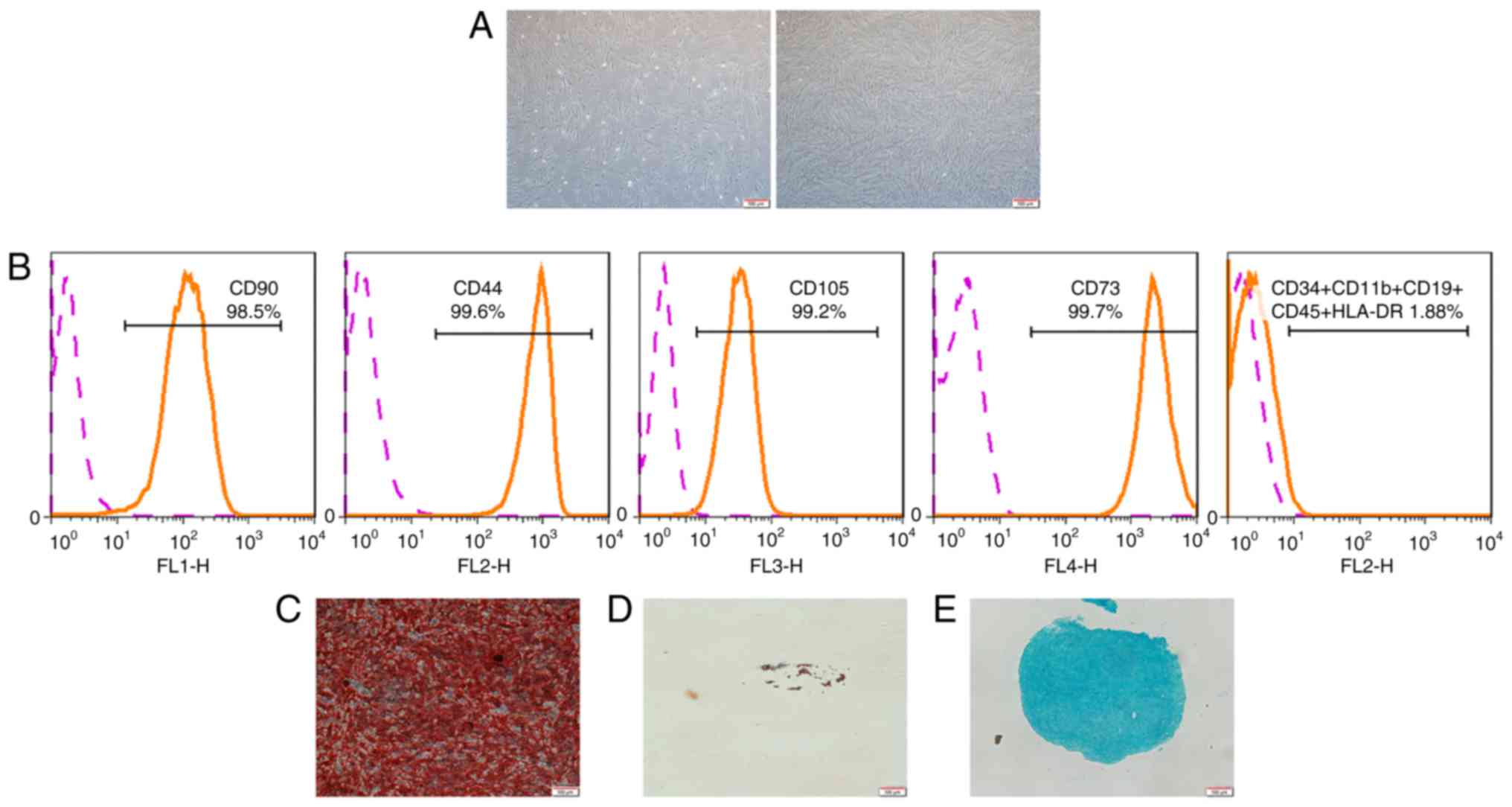 | Figure 1Characterization of hPDLSCs. (A)
Morphological characterization of hPDLSCs. Primary culture (left)
and culture at passage 4 (right) are shown. (B) Flow cytometric
analysis showed that PDLSCs were positive for CD90, CD44, CD105,
CD73, and negative for CD34, CD11b, CD19, CD45 and HLA-DR (as
demonstrated by the orange line). Pink line represents non-specific
staining with isotype-matched antibodies. (C) Following 4 weeks of
culture in osteogenic induction medium, the cells were stained with
Alizarin Red. (D) Following 4 weeks of culture in adipogenic
induction medium, the cells were stained with Oil Red O. (E)
Following 4 weeks of culture in chondrogenic induction medium, the
cells were stained with Alcian Blue (scale bar, 100 µm).
hPDLSCs, human periodontal ligament stem cells. |
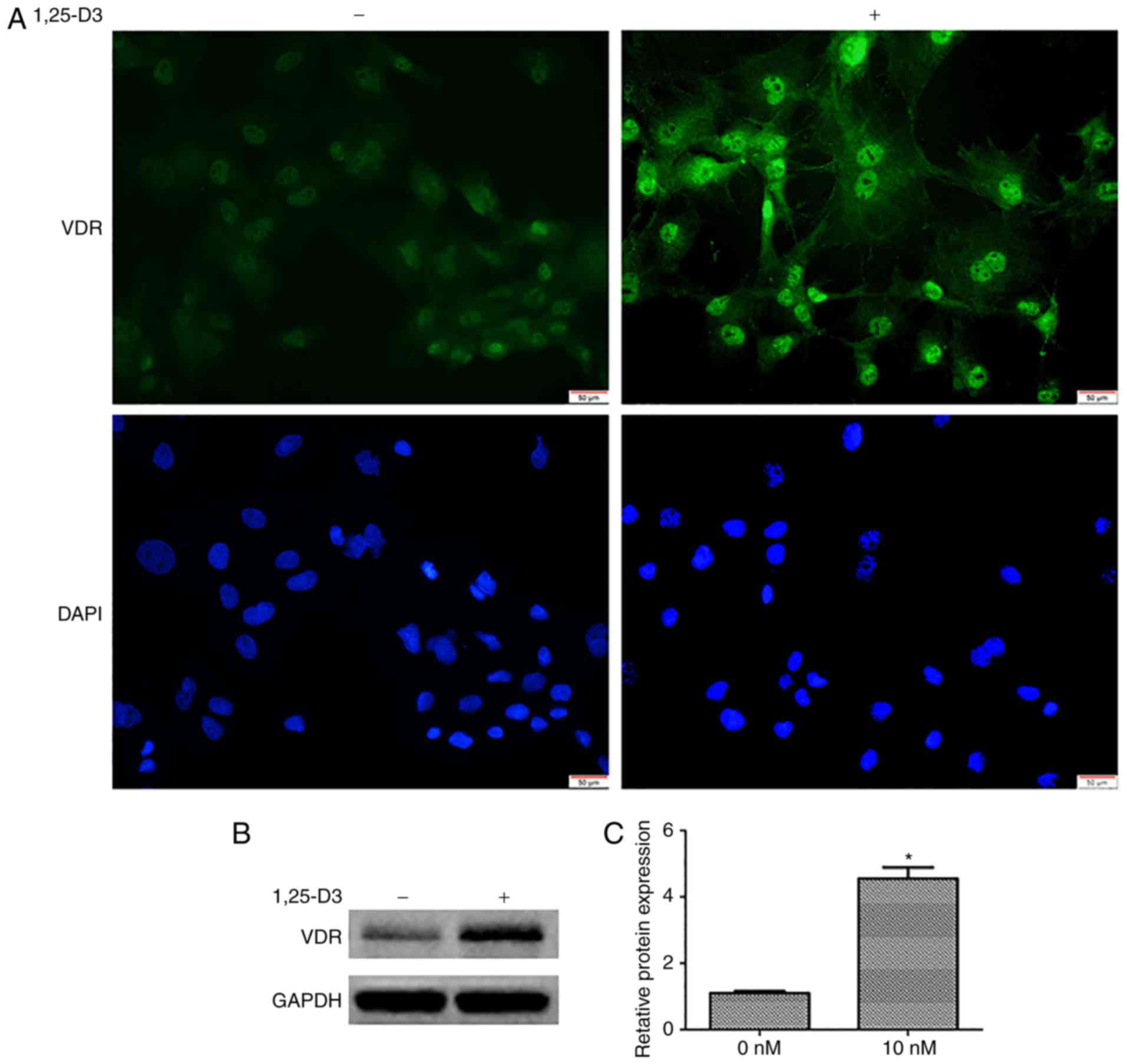
1,25-D3 upregulates the expression of VDR
in hPDLSCs
To determine the expression profile of the VDR in
hPDLSCs and further assess whether its expression is induced by
1,25-D3, the expression of VDR was measured by immunofluorescence
and western blotting. The cells were incubated in the presence or
absence of 10 nM 1,25-D3 for 48 h. Under basal conditions (0 nM),
the specific immunofluorescence signal for VDR (green) was weak.
However, the expression of VDR was upregulated following 48 h
incubation with 10 nM 1,25-D3 (Fig.
2A).
The upregulation of VDR in response to 1,25-D3 was
further validated by western blotting. Following culture with 10 nM
1,25-D3 for 48 h, the quantitative results from the western
blotting revealed an increase in the expression of VDR relative to
the controls (0 nM) (Fig. 2B and
C).
1,25-D3 inhibits the proliferation of
hPDLSCs
To evaluate the effect of 1,25-D3 on hPDLSC
proliferation, the CCK-8 proliferation assay was performed. The
hPDLSCs were treated with different concentrations of 1,25-D3 (0,
0.1, 1 and 10 nM) for 24, 48, and 72 h. Starting at 48 h, 1 and 10
nM 1,25-D3 induced a significant reduction in cell proliferation
compared with the control cells (Fig.
3A). To further corroborate these findings, the expression of
PCNA was examined by western blotting, and decreased expression of
PCNA was confirmed at the protein level (Fig. 3B).
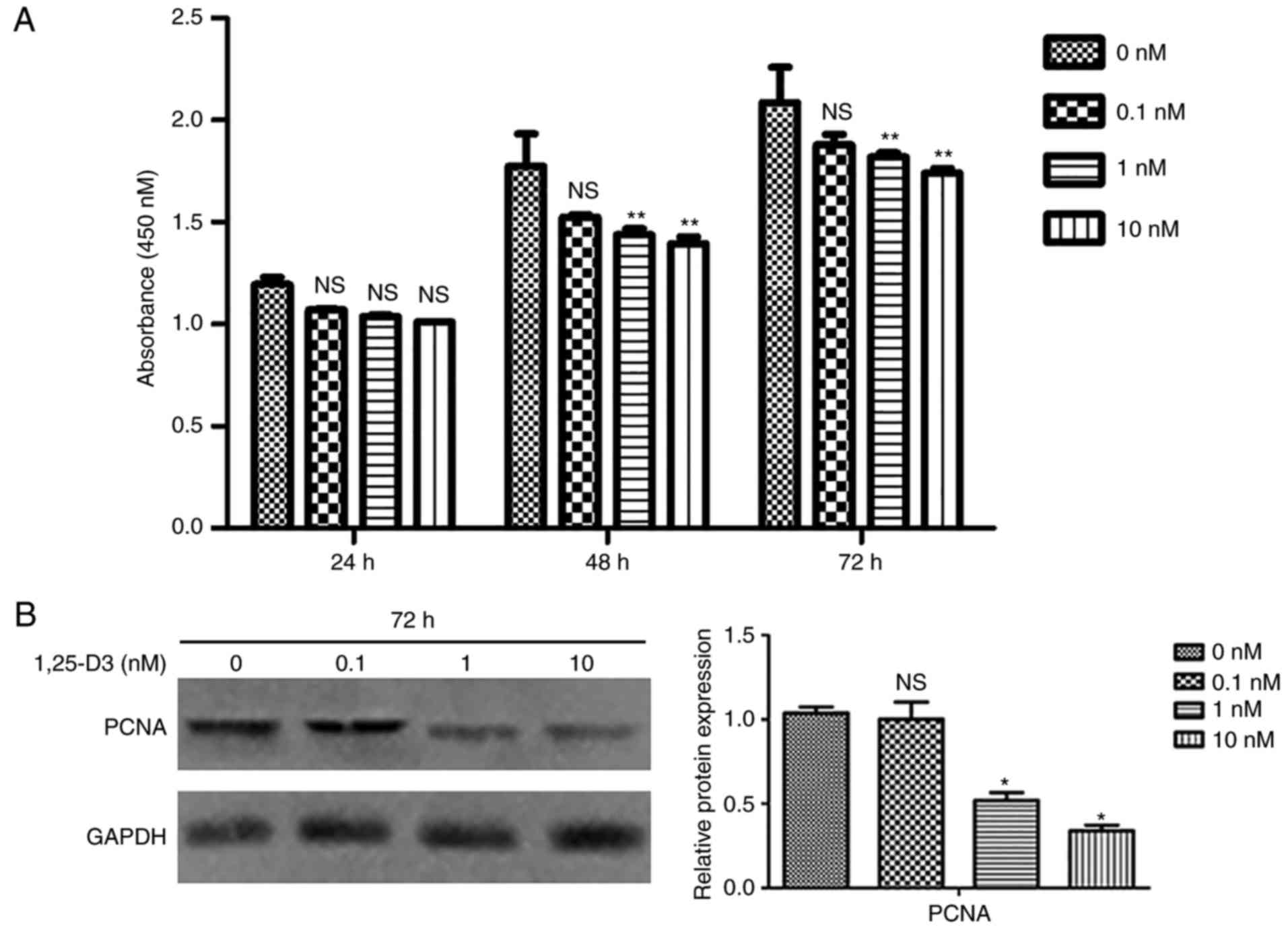 | Figure 31,25-D3 inhibits the proliferation of
hPDLSCs. (A) Effects of different concentrations of 1,25-D3 (0,
0.1, 1 and 10 nM) on the proliferation of hPDLSCs were analyzed by
CCK-8 at 24, 48 and 72 h following treatment. Cell proliferation
was significantly inhibited by 1,25-D3 (1 and 10 nM) in hPDLSCs at
48 and 72 h. (B) Western blotting showed that 1,25-D3 downregulated
the protein expression of PCNA in hPDLSCs following treatment.
*P<0.05 and **P<0.01, vs. control (0 nM
group). hPDLSCs, human periodontal ligament stem cells; PCNA,
proliferating cell nuclear antigen; NS, not significant. |
1,25-D3 promotes the osteogenic
differentiation of hPDLSCs
In order to elucidate the role of 1,25-D3 in
osteogenic differentiation and mineralization, ALP and AR staining
were performed on hPDLSCs cultured with OIM with or without 10 nM
1,25-D3 for 7 and 14 days, respectively. The ALP staining of
hPDLSCs treated with 10 nM 1,25-D3 for 7 days was more extensive
than that in the corresponding group cultured without the drug
(Fig. 4A). In the normal basic
medium group, no calcium nodules were observed following incubation
for 14 days; in the osteogenic induction group, there was notable
calcium nodule formation. The hPDLSCs treated with 10 nM 1,25-D3
had a high number of calcium nodules than those cultured without
the drug for 14 days (Fig. 4B).
Furthermore, the protein expression of osteogenic markers were
examined in hPDLSCs cultured in OIM with or without 10 nM 1,25-D3
for 7 and 14 days. Consistently, the cells cultured in OIM with 10
nM 1,25-D3 expressed significantly higher levels of ALP and OPN
than those cultured in OIM without the drug (Fig. 4C and D).
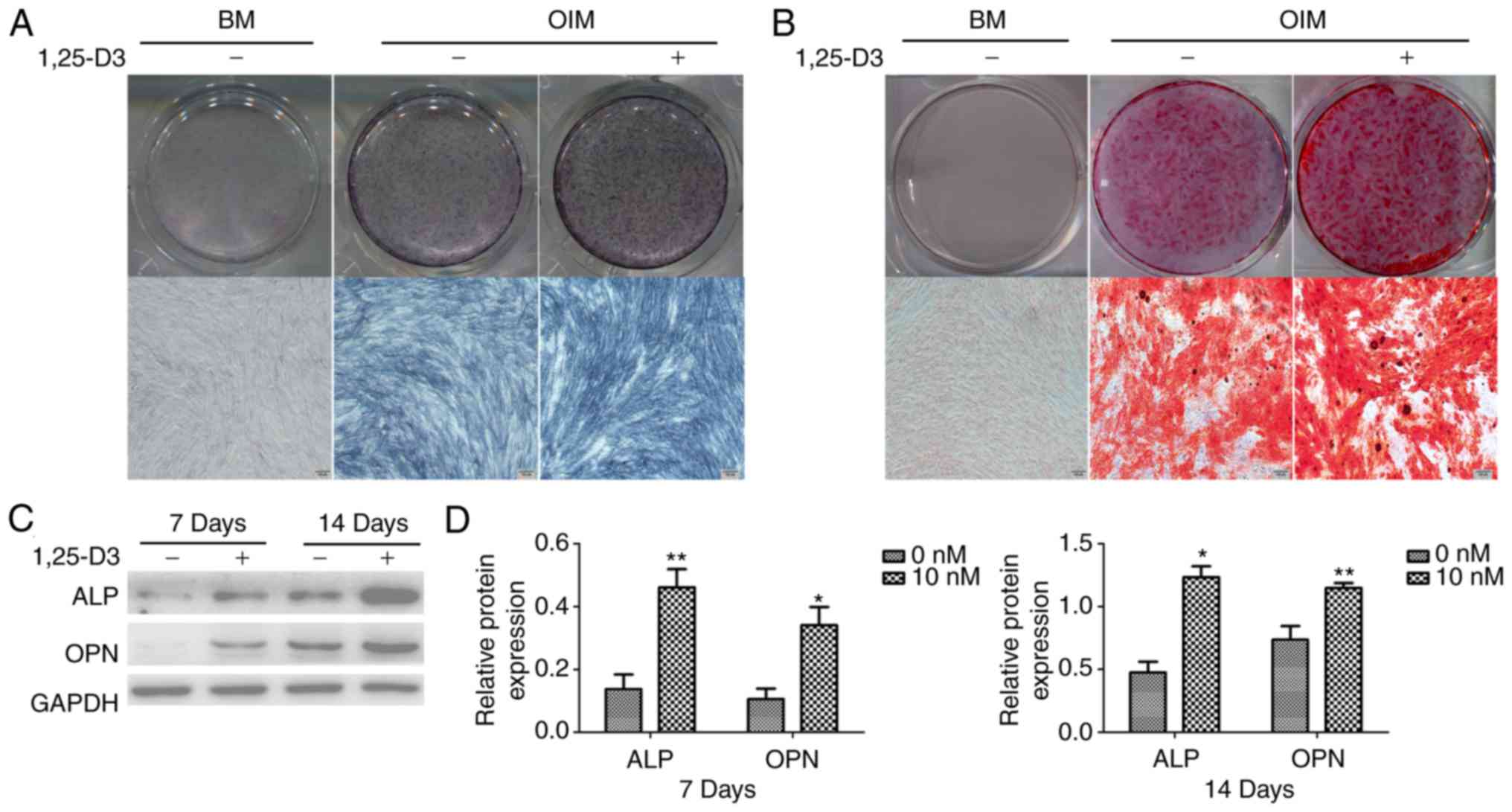 | Figure 41,25-D3 promotes the osteogenic
differentiation of hPDLSCs. (A) Osteogenic differentiation
following 7 days incubation was examined by ALP staining. In the BM
group, the expression was ALP was undetectable. The expression of
ALP in hPDLSCs cultured in OIM with 10 nM 1,25-D3 was more
extensive than in those cultured in OIM alone. (B) Mineralization
following 14 days incubation was examined by AR staining. No
calcium nodules were observed in the BM group. In the OIM group,
the presence of 10 nM 1,25-D3 significantly enhanced AR staining
(scale bar, 100 µm). (C) Expression levels of osteogenic
markers were detected by western blotting. The addition of 10 nM
1,25-D3 significantly upregulated the expression levels of ALP and
OPN in hPDLSCs. (D) Quantitative analysis of protein band density.
*P<0.05 and **P<0.01, vs. 0 nM.
hPDLSCs, human periodontal ligament stem cells; BM, basic medium;
osteogenic induction medium; ALP, alkaline phosphatase; OPN,
osteopontin. |
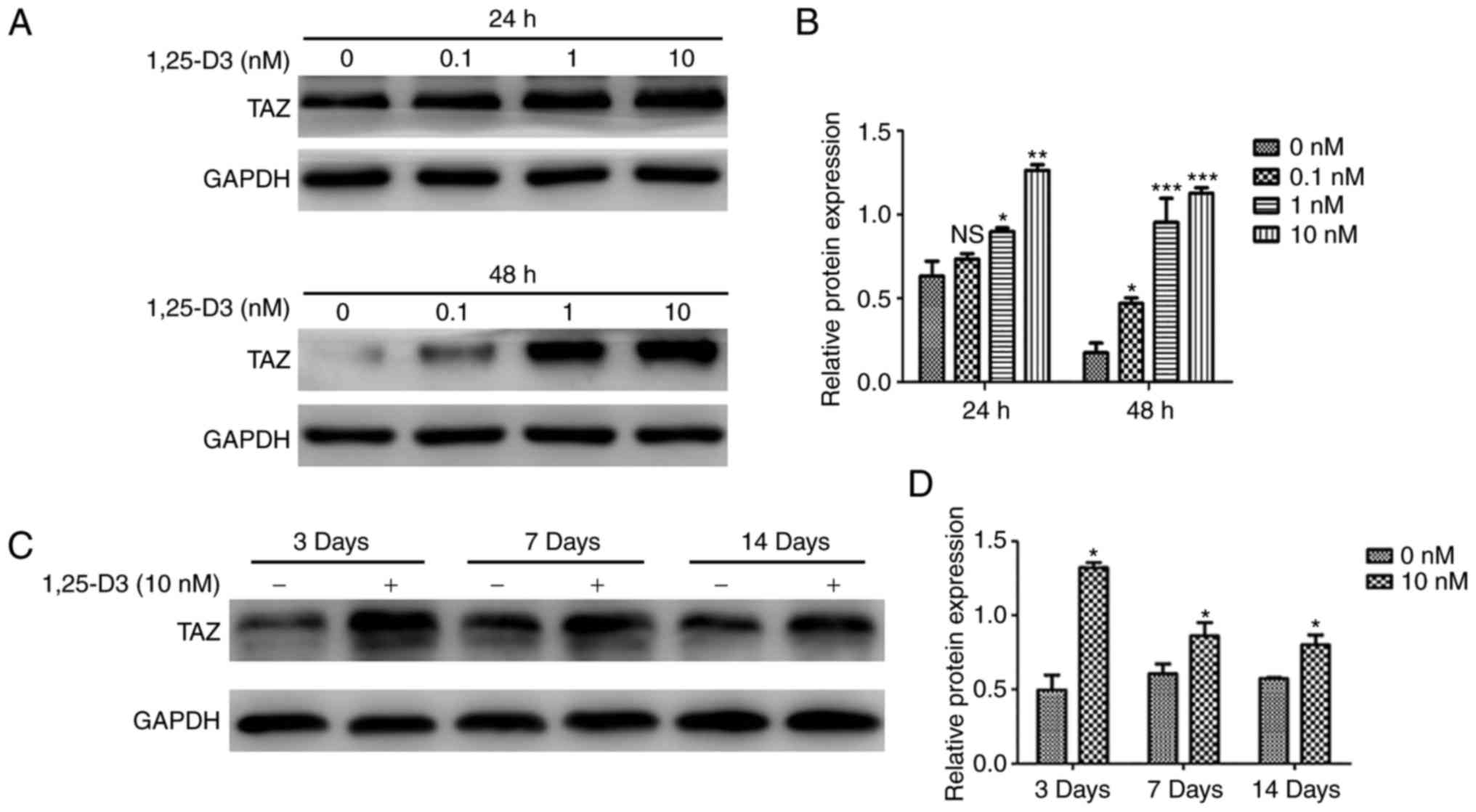
1,25-D3 treatment upregulates TAZ in
hPDLSCs
To determine whether the 1,25-D3-induced osteogenic
differentiation of hPDLSCs involves the modulation of TAZ, the
effects of 1,25-D3 on the expression of TAZ were examined in
hPDLSCs cultured with basic medium for 24, 48 h or OIM for 3, 7,
and 14 days. It was found that 1,25-D3 upregulated the expression
of TAZ in the hPDLSCs incubated with basic medium (Fig. 5A and B). Similarly, the protein
levels of TAZ were significantly upregulated by treatment with 10
nM 1,25-D3 in the hPDLSCs incubated with OIM for 3, 7 and 14 days
(Fig. 5C and D).
Potential interaction of 1,25-D3 and TAZ
in the osteogenic differentiation of hPDLSCs
To further elucidate the mechanism of 1,25-D3 on the
osteogenic differentiation of PDLSCs, the hPDLSCs were transfected
with lentiviral vectors containing the human TAZ cDNA sequence to
obtain stable cell clones. The overexpression of TAZ in hPDLSCs was
verified by RT-qPCR and western blot analyses (Fig. 6A and B). Subsequently, the hPDLSCs
overexpressing with TAZ were examined for their osteogenic
differentiation capacity in vitro. Following osteogenic
induction, the TAZ-overexpressing hPDLSCs exhibited marked ALP
staining on day 7 and enhanced mineralization, revealed by AR
staining, on day 14 (Fig.
6C).
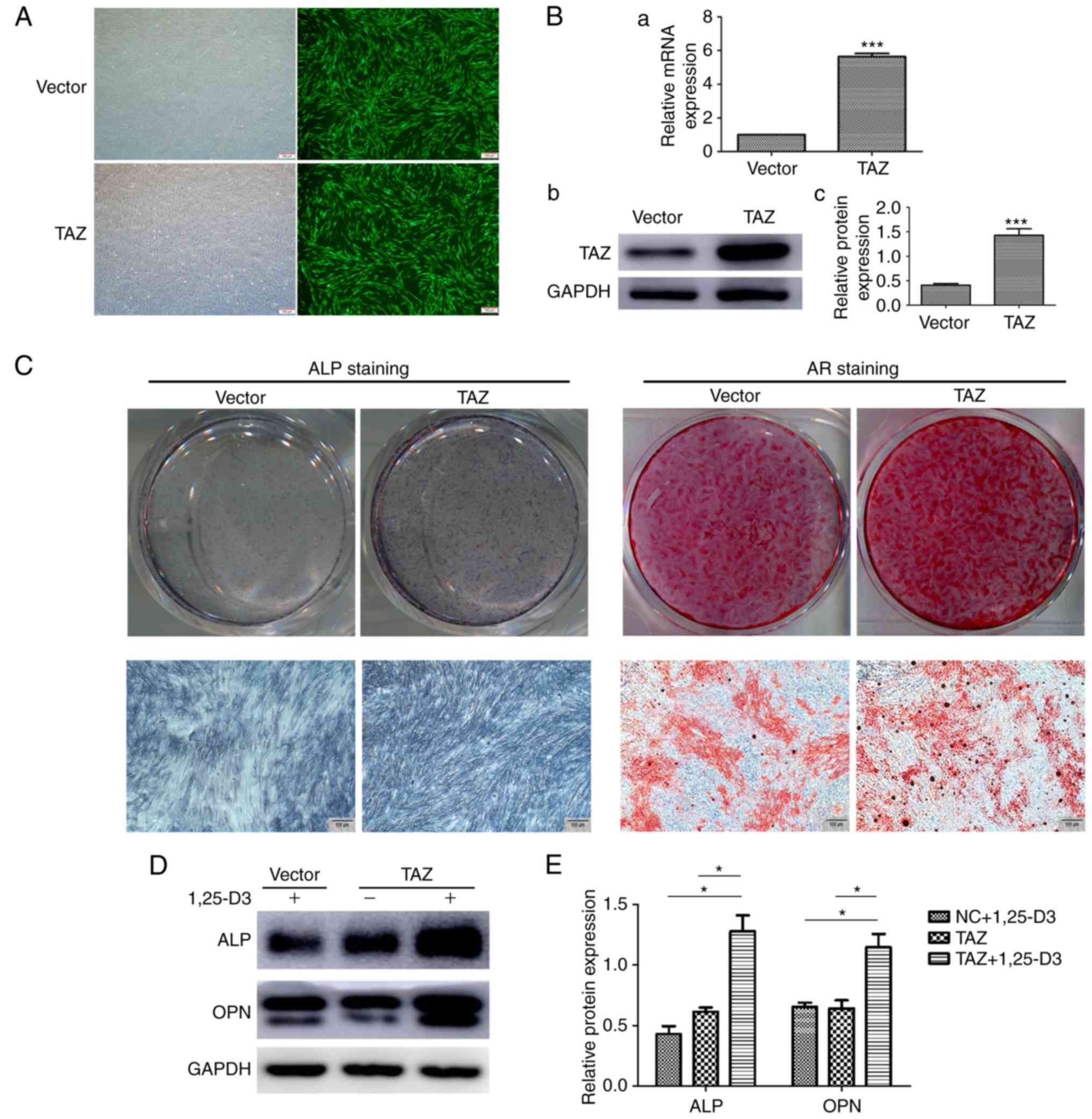 | Figure 6Potential interaction of 1,25-D3 and
TAZ on the osteogenic differentiation of hPDLSCs. (A) Fluorescence
detection of hPDLSCs transduced with lentiviral expression vectors.
Upper panels show cells transduced with normal lentivirus vectors
(vector), lower panels show cells transduced with TAZ
overexpressing lentiviral vectors (TAZ). Scale bar, 100 mM (B) (a)
mRNA and (b) protein levels of TAZ, (c) quantification of the
protein expression levels were significantly increased following
transfection with lentiviral vectors expressing TAZ, compared with
those in the control. (C) ALP staining of TAZ-overexpressing
hPDLSCs showed enhanced osteogenic differentiation following
osteogenic induction in vitro for 7 days. AR staining of the
TAZ-overexpressing hPDLSCs showed increased mineralization
following osteogenic induction for 14 days. (D) Following culture
with OIM for 7 days, co-treatment of 1,25-D3 with TAZ
overexpression significantly enhanced protein expression levels of
ALP and OPN compared with levels when treated with TAZ
overexpression or 1,25-D3 alone. (E) Quantification of the data
from D. *P<0.05, ***P<0.001 (scale bar,
100 µm). hPDLSCs, human periodontal ligament stem cells;
TAZ, transcriptional coactivator with the PDZ-binding motif; ALP,
alkaline phosphatase; AR, Alizarin Red. |
Taken together, the results obtained revealed that
TAZ was critical for driving the osteogenic differentiation of
hPDLSCs and was upregulated by 1,25-D3 treatment. Subsequently,
whether there is an interaction between 1,25-D3 and TAZ in the
osteogenic differentiation of hPDLSCs was investigated. Osteogenic
markers in the TAZ-overexpressing hPDLSCs treated with or without
10 nM 1,25-D3 were examined. Following culture with OIM for 7 days,
the cells overexpressing TAZ treated with 1,25-D3 expressed
markedly enhanced protein levels of ALP and OPN, compared with TAZ
overexpression or 1,25-D3 treatment alone (Fig. 6D). This data suggest that
overexpression of TAZ and treatment with 1,25-D3 synergistically
stimulate the expression of osteogenic markers in hPDLSCs.
Discussion
Alveolar bone defects due to periodontal diseases,
periapical diseases, jaw osteomyelitis and maxillofacial tumors are
associated with, not only functional, but also aesthetic and
psychological problems. A stem cell-based tissue-engineering
approach is perceived as a promising therapeutic alternative for
bone defects, which has revolutionized current treatment paradigms
(34). The periodontal ligament
itself contains PDLSCs, which allow for the self-renewal and
regeneration of other tissues, including cementum and alveolar
bone. It has been reported that PDLSCs are able to undergo
multilineage differentiation, for example to chondrogenic,
osteogenic and adipogenic lineages, when exposed to suitable
inductive conditions (35).
Additionally, the data obtained in the present study showed the
potential of PDLSCs to form calcified deposits in vitro.
However, it remains challenging to fully exploit the therapeutic
potential of hPDLSCs for bone repair and regeneration.
Skeletal development is controlled by multiple
osteogenic signals, and 1,25-D3 is one physiological regulator of
osteogenic differentiation. 1,25-D3 exerts its biological action
through binding to VDR, a member of the nuclear hormone receptor
superfamily (36). In the present
study, the addition of 1,25-D3 upregulated the expression of VDR in
hPDLSCs. This result is consistent with a study on H9c2 cardiac
cells, which showed that the expression of VDR increased upon
incubation with 1,25-D3 (37).
Through binding to VDR, 1,25-D3 has been found to inhibit the
proliferation of certain cells, including osteoblasts, osteoclasts
(38) and MSCs (36,39). Similarly, the present study study
revealed that treatment with 1,25-D3 inhibited the proliferation of
hPDLSCs.
As a systemic factor, 1,25-D3 is a key signaling
moiety that is well known for its roles in the regulation of bone
metabolism. For example, the expression of OPN is upregulated by
1,25-D3 in osteoblast cells (40). 1,25-D3 is also essential in
osteocalcin synthesis in bone cells in vitro (41). In addition, treatment with 1,25-D3
significantly increases the activity of ALP in hMSCs (36).
In the present study, it was hypothesized that
1,25-D3 may enhance the osteogenic differentiation of hPDLSCs in
vitro. The results revealed enhanced ALP staining of the
hPDLSCs treated with 10 nM 1,25-D3 in OIM relative to the control
groups. In addition, hPDLSCs treated with 10 nM 1,25-D3 exhibited
significantly more calcium nodules than those cultured without the
drug for 14 days. ALP is a key osteogenic maker expressed at the
early stage of osteogenic differentiation (42), whereas OPN is a marker reflecting
osteogenic maturation and bone formation (43). The results of the present study
confirmed that the expression of ALP and OPN were upregulated by
1,25-D3 in hPDLSCs. Although there is sufficient evidence
supporting the enhanced effects of 1,25-D3 on the osteogenic
differentiation of hPDLSCs, the mechanism of 1,25-D3 in hPDLSC
osteogenic differentiation remains to be fully elucidated.
The Hippo pathway is a key pathway in controlling
tissue regeneration. Inhibition of the Hippo pathway causes an
accumulation of TAZ and its eventual translocation into the nucleus
where it activates the transcription of its target genes, which are
required for osteogenic differentiation (44). Of note, canonical Wnt signaling
promotes the differentiation of MSCs into osteoblasts through TAZ
stabilization and upregulation (45). Zinc-finger transcriptional factors
Snail/Slug interact with TAZ to form complexes that induce the
osteogenesis of skeletal stem cells (46). Furthermore, the overexpression of
TAZ in osteoblasts significantly enhances bone formation in
transgenic mice (47). TAZ has
been found to drive osteogenic differentiation in stem cells from a
diverse source, and it has been reported that TAZ can regulate the
sensitivity of intra-hepatic cholangiocarcinoma cells to vitamin D
in vivo (48). Taking all
available data into consideration, it was hypothesized that the
osteogenic effect of 1,25-D3 in hPDLSCs involve the actions of TAZ.
The results of the present study indicated that 1,25-D3 enhanced
the protein levels of TAZ in hPDLSCs in the context of either basic
medium or OIM.
To further examine the underlying mechanism, the
present study generated stable TAZ-overexpressing cell clones of
hPDLSCs. The results revealed that the overexpression of TAZ
promoted the osteogenic differentiation of hPDLSCs in vitro.
As it was found that the protein expression of TAZ was increased in
hPDLSCs following treatment with 1,25-D3, and that TAZ was found to
be associated with osteogenic differentiation in several previous
studies and the present study, the subsequent aim was to understand
the potential interaction between 1,25-D3 and TAZ on the osteogenic
differentiation of hPDLSCs. The osteogenic marker expression levels
of hPDLSCs were examined under the induction of osteogenic
differentiation following 1,25-D3 treatment and TAZ
overex-pression, alone or in combination. The results showed that
the combination of 1,25-D3 treatment and TAZ overexpression
potently enhanced the protein expression levels of ALP and OPN in
hPDLSCs cultured in OIM. These data showed that the treatment of
hPDLSCs overexpressing TAZ with 1,25-D3 synergistically stimulated
osteogenic marker expression. This synergistic stimulation may be
important in the osteogenic differentiation of hPDLSCs, however,
the precise underlying mechanism for this synergistic interaction
remains to be elucidated.
In conclusion, the present study confirmed that
1,25-D3 treatment enhanced the osteogenic differentiation of
hPDLSCs, and indicated that this pro-osteogenic effect involves, at
least in part, the actions of TAZ, a key Hippo signaling pathway
effector. These insights may be valuable for therapeutic
strategies, including preprogramming stem cells prior to clinical
use. However, the present study focused only on whether TAZ was
involved in the effect of 1,25-D3 on the osteogenic differentiation
of hPDLSCs. Future detailed investigations with an inhibitor of TAZ
or TAZ-knockdown are warranted to determine the underlying
mechanisms of TAZ in the osteogenic effect of 1,25-D3.
Acknowledgments
The authors would like to thank the Shandong
Provincial Key Laboratory of Oral Tissue Regeneration for their
support with the present study.
Funding
The present study was supported by grants from the
National Natural Science Foundation of China (grant nos. 81300885
and 81402150), the Shandong Provincial Key Research and Development
Program (grant nos. 2017GSF18117 and 2016GSF201115), the Shandong
Provincial Natural Science Foundation (grant nos. ZR2018MH018 and
ZR2017MH031), the China Postdoctoral Science Foundation (grant no.
2017M610432), the Young Scholars Program of Shandong University
(grant no. 2015WLJH53) and the Construction Engineering Special
Fund of Taishan Scholars (grant no. ts201511106).
Availability of data and materials
The datasets used during the present study are
available from the corresponding author upon reasonable
request.
Authors’ contributions
XX designed the experiments. YJ and PZ performed the
experiments. YX, LJ, YZ, XW and BZ analyzed the data and YJ wrote
the manuscript.
Ethics approval and consent to
participate
This study was approved by the Medical Ethical
Committee of School of Stomatology, Shandong University (protocol
no. 201 70303). Each participant provided written informed consent
in accordance with the Declaration of Helsinki.
Patient consent for publication
Each participant provided written informed
consent.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Martínez-Maestre MÁ, González-Cejudo C,
Machuca G, Torrejón R and Castelo-Branco C: Periodontitis and
osteoporosis: A systematic review. Climacteric. 13:523–529. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Zhang J, An Y, Gao LN, Zhang YJ, Jin Y and
Chen FM: The effect of aging on the pluripotential capacity and
regenerative potential of human periodontal ligament stem cells.
Biomaterials. 33:6974–6986. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Reddy MS and Morgan SL: Decreased bone
mineral density and periodontal management. Periodontol, 2000.
61:195–218. 2013. View Article : Google Scholar
|
|
4
|
Huang GT, Gronthos S and Shi S:
Mesenchymal stem cells derived from dental tissues vs. those from
other sources: Their biology and role in regenerative medicine. J
Dent Res. 88:792–806. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Lindroos B, Mäenpää K, Ylikomi T, Oja H,
Suuronen R and Miettinen S: Characterisation of human dental stem
cells and buccal mucosa fibroblasts. Biochem Biophys Res Commun.
368:329–335. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Liu Y, Zheng Y, Ding G, Fang D, Zhang C,
Bartold PM, Gronthos S, Shi S and Wang S: Periodontal ligament stem
cell-mediated treatment for periodontitis in miniature swine. Stem
Cells. 26:1065–1073. 2010. View Article : Google Scholar
|
|
7
|
Ding G, Liu Y, Wang W, Wei F, Liu D, Fan
Z, An Y, Zhang C and Wang S: Allogeneic periodontal ligament stem
cell therapy for periodontitis in swine. Stem Cells. 28:1829–1838.
2010. View
Article : Google Scholar : PubMed/NCBI
|
|
8
|
Chen FM, Sun HH, Lu H and Yu Q: Stem
cell-delivery therapeutics for periodontal tissue regeneration.
Biomaterials. 33:6320–6344. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Haussler MR, Haussler CA, Jurutka PW,
Thompson PD, Hsieh JC, Remus LS, Selznick SH and Whitfield GK: The
vitamin D hormone and its nuclear receptor: Molecular actions and
disease states. J Endocrinol. 154(Suppl): S57–S73. 1997.PubMed/NCBI
|
|
10
|
Amling M, Priemel M, Holzmann T, Chapin K,
Rueger JM, Baron R and Demay MB: Rescue of the skeletal phenotype
of vitamin D receptor-ablated mice in the setting of normal mineral
ion homeostasis: Formal histomorphometric and biomechanical
analyses. Endocrinology. 140:4982–4987. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Gurlek A, Pittelkow MR and Kumar R:
Modulation of growth factor/cytokine synthesis and signaling by 1α,
25-dihydroxyvitamin D3: Implications in cell growth and
differentiation. Endocr Rev. 23:763–786. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Ueno K, Katayama T, Miyamoto T and
Koshihara Y: Interleukin-4 enhances in vitro mineralization in
human osteoblast-like cells. Biochem Biophys Res Commun.
189:1521–1526. 1992. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Prince M, Banerjee C, Javed A, Green J,
Lian JB, Stein GS, Bodine PV and Komm BS: Expression and regulation
of Runx2/Cbfa1 and osteoblast phenotypic markers during the growth
and differentiation of human osteoblasts. J Cell Biochem.
80:424–440. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Jørgensen NR, Henriksen Z, Sørensen OH and
Civitelli R: Dexamethasone, BMP-2, and 1,25-dihydroxyvitamin D
enhance a more differentiated osteoblast phenotype: Validation of
an in vitro model for human bone marrow-derived primary
osteoblasts. Steroids. 69:219–226. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Driel MV, Koedam M, Buurman CJ, Hewison M,
Chiba H, Uitterlinden AG, Pols HA and van Leeuwen JP: Evidence for
auto/paracrine actions of vitamin D in bone: 1α-hydroxylase
expression and activity in human bone cells. FASEB J. 20:2417–2419.
2006. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Zhou YS, Liu YS and Tan JG: Is 1,
25-dihydroxyvitamin D3 an ideal substitute for dexamethasone for
inducing osteogenic differentiation of human adipose tissue-derived
stromal cells in vitro. Chin Med J. 119:1278–1286. 2006.
|
|
17
|
Nebel D, Svensson D, Arosenius K, Larsson
E, Jönsson D and Nilsson BO: 1α,25-dihydroxyvitamin D3 promotes
osteogenic activity and downregulates proinflammatory cytokine
expression in human periodontal ligament cells. J Periodontal Res.
50:666–673. 2015. View Article : Google Scholar
|
|
18
|
Uchiyama M, Nakamichi Y, Nakamura M,
Kinugawa S, Yamada H, Udagawa N and Miyazawa H: Dental pulp and
periodontal ligament cells support osteoclastic differentiation. J
Dent Res. 88:609–614. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Staley BK and Irvine KD: Hippo signaling
in Drosophila: Recent advances and insights. Dev Dyn. 241:3–15.
2012. View Article : Google Scholar :
|
|
20
|
Ramos A and Camargo FD: The Hippo
signaling pathway and stem cell biology. Trends Cell Biol.
22:339–346. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Yu FX and Guan KL: The Hippo pathway:
Regulators and regulations. Genes Dev. 27:355–371. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Zhao B, Tumaneng K and Guan KL: The Hippo
pathway in organ size control, tissue regeneration and stem cell
self-renewal. Nat Cell Biol. 13:877–883. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Moroishi T, Park HW, Qin B, Chen Q, Meng
Z, Plouffe SW, Taniguchi K, Yu FX, Karin M, Pan D, et al: A
YAP/TAZ-induced feedback mechanism regulates Hippo pathway
homeostasis. Genes Dev. 29:1271–1284. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Hong JH, Hwang ES, Mcmanus MT, Amsterdam
A, Tian Y, Kalmukova R, Mueller E, Benjamin T, Spiegelman BM, Sharp
PA, et al: TAZ, a transcriptional modulator of mesen-chymal stem
cell differentiation. Science. 309:1074–1078. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Cui CB, Cooper LF, Yang X, Karsenty G and
Aukhil I: Transcriptional coactivation of bone-specific
transcription factor Cbfa1 by TAZ. Mol Cell Biol. 23:1004–1013.
2003. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Kawano S, Maruyama J, Nagashima S, Inami
K, Qiu W, Iwasa H, Nakagawa K, Ishigami-Yuasa M, Kagechika H,
Nishina H and Hata Y: A cell-based screening for TAZ activators
identifies ethacridine, a widely used antiseptic and abortifacient,
as a compound that promotes dephosphorylation of TAZ and inhibits
adipogenesis in C3H10T1/2 cells. J Biochem. 158:413–423. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Jang EJ, Jeong H, Kang JO, Kim NJ, Kim MS,
Choi SH, Yoo SE, Hong JH, Bae MA and Hwang ES: TM-25659 enhances
osteogenic differentiation and suppresses adipogenic
differentiation by modulating the transcriptional co-activator TAZ.
Br J Pharmacol. 165:1584–1594. 2012. View Article : Google Scholar :
|
|
28
|
Byun MR, Kim AR, Hwang JH, Kim KM, Hwang
ES and Hong JH: FGF2 stimulates osteogenic differentiation through
ERK induced TAZ expression. Bone. 58:72–80. 2014. View Article : Google Scholar
|
|
29
|
Byun MR, Kim AR, Hwang JH, Sung MK, Lee
YK, Hwang BS, Rho JR, Hwang ES and Hong JH: Phorbaketal A
stimulates osteoblast differentiation through TAZ mediated Runx2
activation. FEBS Lett. 586:1086–1092. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Wen Y, Lan J, Huang H, Yu M, Cui J, Liang
J, Jiang B and Xu X: Application of eGFP to label human periodontal
ligament stem cells in periodontal tissue engineering. Arch Oral
Biol. 57:1241–1250. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Jiang B, Wen Y, Huang H, Cui J, Liang J,
Ma X, Lan J and Xu X: Study of labeling human periodontal ligament
stem cells with enhanced green fluorescent protein by lentivirus
vector infection. Hua Xi Kou Qiang Yi Xue Za Zhi. 30:82–86. 2012.In
Chinese. PubMed/NCBI
|
|
32
|
Essers J, Theil AF, Baldeyron C, van
Cappellen WA, Houtsmuller AB, Kanaar R and Vermeulen W: Nuclear
dynamics of PCNA in DNA replication and repair. Mol Cell Biol.
25:9350–9359. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2−ΔΔC T method. Methods. 25:402–408.
2001. View Article : Google Scholar
|
|
34
|
Daley GQ: The promise and perils of stem
cell therapeutics. Cell Stem Cell. 10:740–749. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Seo BM, Miura M, Gronthos S, Bartold PM,
Batouli S, Brahim J, Young M, Robey PG, Wang CY and Shi S:
Investigation of multipotent postnatal stem cells from human
periodontal ligament. Lancet. 364:149–155. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Khanna-Jain R, Vuorinen A, Sándor GK,
Suuronen R and Miettinen S: Vitamin D3 metabolites
induce osteogenic differentiation in human dental pulp and human
dental follicle cells. J Steroid Biochem Mol Biol. 122:133–141.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Hlaing SM, Garcia LA, Contreras JR, Norris
KC, Ferrini MG and Artaza JN: 1,25-Vitamin D3 promotes
cardiac differentiation through modulation of the WNT signaling
pathway. J Mol Endocrinol. 53:303–317. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Reichrath J: Vitamin D and the skin: An
ancient friend, revisited. Exp Dermatol. 16:618–625. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Feng Y, Sun Y, Jia W and Zhang C:
Platelet-rich plasma and 1,25(OH)2 vitamin D3
synergistically stimulate osteogenic differentiation of adult human
mesenchymal stem cells. Biotechnol Lett. 32:635–642. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Atkins GJ, Anderson PH, Findlay DM,
Welldon KJ, Vincent C, Zannettino AC, O’Loughlin PD and Morris HA:
Metabolism of vitamin D3 in human osteoblasts: Evidence for
autocrine and paracrine activities of 1 alpha,25-dihydroxyvitamin
D3. Bone. 40:1517–1528. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Shiba H, Uchida Y, Kamihagi K, Sakata M,
Fujita T, Nakamura S, Takemoto T, Kato Y and Kurihara H:
Transforming growth factor-beta1 and basic fibroblast growth factor
modulate osteo-calcin and osteonectin/SPARC syntheses in
vitamin-D-activated pulp cells. J Dent Res. 80:1653–1659. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Shigetani Y, Ohkura N, Yoshiba K, Ohshima
H, Hosoya A, Yoshiba N and Okiji T: GaAlAs laser-induced pulp
mineralization involves dentin matrix protein 1 and osteopontin
expression. Oral Dis. 22:399–405. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Campos JM, Prati AJ, Cirano FR, Pimentel
SP, Pastore GP, Pecorari VG, Ribeiro FV, Casati MZ and Casarin RC:
Smoking modulates gene expression of type I collagen, bone
sialoprotein, and osteocalcin in human alveolar bone. J Oral
Maxillofac Surg. 73:2123–2131. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Meng Z, Moroishi T and Guan KL: Mechanisms
of Hippo pathway regulation. Genes Dev. 30:1–17. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Byun MR, Hwang JH, Kim AR, Kim KM, Hwang
ES, Yaffe MB and Hong JH: Canonical Wnt signalling activates TAZ
through PP1A during osteogenic differentiation. Cell Death Differ.
21:854–863. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Yi T and Weiss SJ: Snail/Slug-YAP/TAZ
complexes cooperatively regulate mesenchymal stem cell function and
bone formation. Cell Cycle. 16:399–405. 2017. View Article : Google Scholar
|
|
47
|
Yang JY, Sun WC, An JH, Ju YJ, Sang WK,
Kim SY, Kim JE and Chan SS: Osteoblast-targeted overexpression of
TAZ increases bone mass in vivo. PLoS One. 8:e565852013. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Xiao H, Tong R, Yang B, Lv Z, Du C, Peng
C, Ding C, Cheng S, Zhou L, Xie H, et al: TAZ regulates cell
proliferation and sensitivity to vitamin D3 in intrahepatic
cholangiocarcinoma. Cancer Lett. 381:370–379. 2016. View Article : Google Scholar : PubMed/NCBI
|