Introduction
Endometriosis is a non-malignant condition
characterised by the ectopic implantation and growth of endometrial
tissue in locations within the abdominal cavity, such as the
fallopian tubes, ovaries, peritoneum, vagina, bladder, bowel and
rectum. Ectopic endometrium responds to hormonal changes in the
same way as eutopic endometrium, by proliferating and shedding with
the menstrual cycle causing pain and inflammation in the affected
areas. Symptoms of endometriosis include dysmenorrhea, dyspareunia,
dysuria, dyschezia, infertility and chronic pelvic pain as well as
an increased risk of allergies, asthma, fibromyalgia, autoimmune
disease, hypothyroidism, multiple sclerosis and chronic fatigue
syndrome (1,2). Endometriosis is estimated to affect
around two million individuals in the UK (2).
Endometriosis is now becoming a well-documented risk
factor for ovarian cancer, occurring in up to 28% of ovarian cancer
patients (3-13). A 20-year study by Stewart et
al, of more than 21,000 patients seeking fertility treatment in
Western Australia has shown that endometriosis is particularly
associated with an increased rate of ovarian cancer in patients who
remained childless after fertility treatment (4). The study did not classify invasive
epithelial ovarian cancer any further; however, a similarly large
pooled analysis of 13 ovarian cancer risk factor studies examined
the relationship between endometriosis and the four major subtypes
of epithelial ovarian cancer: serous (high and low grade), clear
cell, endometrioid and mucinous (4). They found that the association
between endometriosis and ovarian cancer differed between subtypes.
Patients with low-grade serous (9.2%), endometrioid (13.9%) and
clear cell (20.2%) subtypes were more likely to have endometriosis
(compared to 6.2% of control cases). Mucinous epithelial ovarian
cancer was not at all associated with endometriosis (5). In the aforementioned studies, there
was no increased risk of ovarian cancer in patients with pelvic
inflammatory disorder, ovarian cysts, fibroids, breastfeeding,
weight, height, body-mass index and tubal ligation, highlighting
the unique relationship between endometriosis and ovarian cancer
(4,5,9).
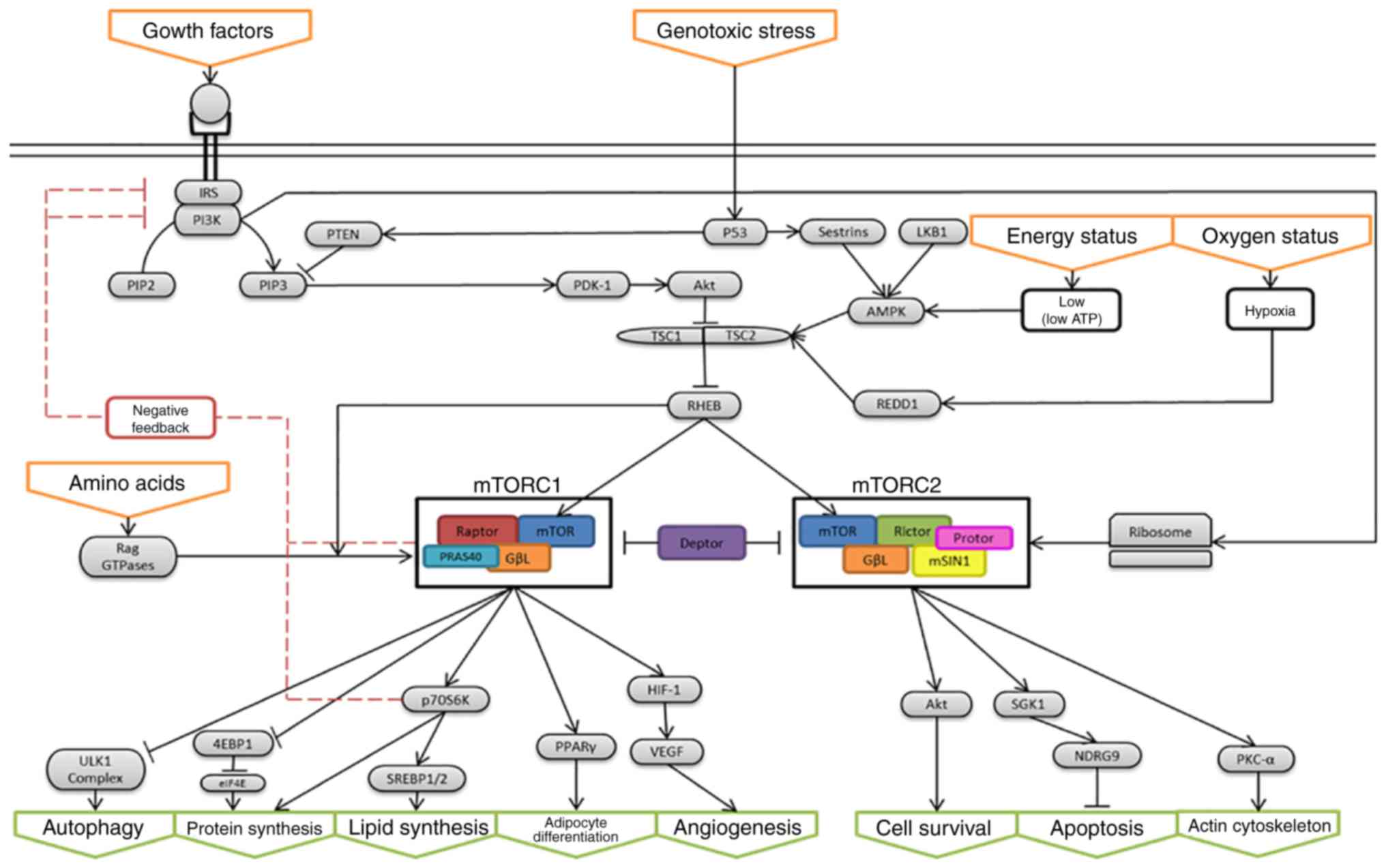
The mTOR pathway is a central regulator of growth,
proliferation, apoptosis and angiogenesis providing balance between
cellular resources such as amino acids and growth factors and
stresses such as hypoxia to control cellular behaviour accordingly.
The activities of this pathway are mediated through the mTOR
complex 1 (mTORC1) and mTOR complex 2 (mTORC2). mTORC1 contains
mTOR, Raptor, DEPTOR, GBL and PRAS
40. mTORC2 contains mTOR, Rictor, DEPTOR and GBL.
mTOR is a serine/threonine kinase and is the central catalytic
component of mTORC1 and mTORC2. DEPTOR is a component that inhibits
the activities of both mTORC1 and mTORC2 by direct interaction with
mTOR. Raptor is a protein component of mTORC1 only and controls
mTOR kinase activity. Rictor is a component of mTORC2 only. The
pathways, including upstream regulators and downstream effectors of
mTORC1 and mTORC2 are shown in Fig.
1, in an abbreviated manner. The mTOR pathway is inhibited via
Rapamycin, also known as sirolimus, which was first described in
1975. Since its discovery, a host of semi-synthetic
Rapamycin-related mTORC1 inhibitors known as rapalogues, including
Everolimus, Deforolimus and Temsirolimus have been developed.
Besides these, dual mTOR and PI3K inhibitors such as NVP-BEZ235 and
Resveratrol have also gained attention (14-16).
The genetic changes that characterise endometriosis
are poorly understood; however, the mTOR pathway is involved in
several ways. Leconte et al, showed an increased expression
of Akt and p70S6K in both the eutopic endometrium and endometriotic
lesion of patients with deep infiltrating endometriosis (DIE) in
comparison to the control endometrium (17). Afshar et al, showed changes
in 23 PI3K/Akt pathway-related genes and 26 MAP kinase
pathway-related genes in the eutopic endometrium of baboons with
endometriosis (18). Both the
PI3K and MAP kinase pathways can inactivate the TSC1/TSC2 complex
causing a release of its inhibitory effect on mTOR kinase activity
(19-21). Guo et al, identified higher
phosphorylated mTOR in ectopic endometrium than in eutopic or
control endometria (22).
Due to its involvement in growth factor and energy
sensing and its effect on protein synthesis and cell cycle
progression, the mTOR pathway is involved in a range of
malignancies. Although there is evidence for the involvement of the
mTOR pathway in endometriosis and ovarian cancer, its role in the
transformation from one condition to the other is poorly
understood. In the present study, we examined the expression of key
mTOR components in endometriosis and ovarian cancer and used an
ovarian cancer cell line of endometrioid origin as a preclinical
model to dissect further the mechanisms of action of mTOR
inhibitors.
Materials and methods
Clinical samples
Tissue samples from patients with endometriosis
(n=24), and non-affected controls (n=34) were obtained at the First
Department of Obstetrics and Gynaecology, ‘Papageorgiou’ Hospital,
University of Thessaloniki, Thessaloniki, Greece. Ethical approval
was obtained by the Ethics Committee of Papageorgiou Hospital. For
the circulating tumour cell (CTC) study, ovarian cancer blood
samples (n=3) were collected from ovarian cancer patients enrolled
on a clinical trial at Mount Vernon Hospital, London, UK, known as
CICATRIx. The present study was also approved by the National
Research Ethics Committee and the Research and Development
department of East and North Hertfordshire NHS Trust
(26/7/16-present).
Written informed consent was obtained from all the
patients. Patients were stage III/IV ovarian serous adenocarcinoma,
and enrolled on the trial to begin third-line dual chemotherapy
treatment. We confirm that all participants of this study from both
clinical settings have given their consent regarding inclusion of
material pertaining to themselves, as specified in the Declaration
of Helsinki. None of the patients and controls can be identified
via this manuscript as all cohorts are fully anonymized.
RNA isolation, cDNA synthesis and
quantitative RT-PCR
RNA was extracted from tissue lysate using the
GenElute™ mRNA MiniPrep kit (Sigma-Aldrich; Merck KGaA, Darmstadt,
Germany). cDNA was synthesised from mRNA using Superscript II
(Invitrogen; Thermo Fisher Scientific, Massachusetts, MN, USA).
cDNA concentration was normalised using RNA concentrations
determined by NanoDrop (Thermo Fisher Scientific, Pittsburgh, PA,
USA). Relative expression of the genes of interest was assessed by
quantitative PCR (qPCR) on an xxpress® (BJS
Biotechnologies, Middlesex, UK) using Kapa SYBR Fast Universal
Master Mix (KapaBiosystems-Sigma-Aldrich; Merck KGaA). Primers for
mTOR, DEPTOR, Rictor and Raptor and qRT-PCR quantification were
used as previously described (23). The following primers were used as
housekeeping genes: YWHAZ, forward:
5′-agacggaaggtgctgagaaa-3′, and reverse:
5′-gaagcattggggatcaagaa-3′; and RPL13A, forward:
5′-cctggtctgagcccaataaa-3′ and reverse:
5′-cttgctcccagcttcctatg-3′.
Tissue microarray
Paraffin-embedded ovarian tissue microarray slides
each containing 70 clinical samples were purchased from US Biomax
(Rockville, MD, USA). The slides were deparaffinised and
rehydrated, followed by antigen retrieval. Blocking was carried out
with 5% goat serum, followed by 24-h incubation with primary rabbit
monoclonal antibodies for mTOR (catalog no. 2983, Cell Signalling
Technology, Inc., Danvers, MA, USA; dilution 1:200), and DEPTOR
(catalog no. 11816, Cell Signalling Technology, Inc.; dilution
1:200). Following numerous washes with PBS, the slides were
incubated with HRP-conjugated secondary antibody for 60 min. The
slides were then washed and subjected to DAB staining,
counterstained with haematoxylin and washed with 0.1% sodium
bicarbonate. The slides were then analysed for immunoreactivity of
both proteins by light microscope (Zeiss, Jena, Germany) and
positive results were measured by the percentage of positive tumour
cells.
Cell line and treatments
MDAH-2774 cells were purchased from ATCC and are
human ovarian endometrioid adenocarcinoma-derived. MDAH-2774 cells
were grown in DMEM (Dulbecco’s Modified Eagle’s medium; Life
Technologies; Thermo Fisher Scientific) supplemented with 10% FBS
(Life Technologies; Thermo Fisher Scientific), 1%
penicillin/streptomycin (Life Technologies; Thermo Fisher
Scientific) and 1% L-glutamine (Life Technologies; Thermo Fisher
Scientific). MDAH-2774 cells were cultured at 37°C at 5%
CO2. We studied the effect of mTOR pathway inhibition on
the gene expression of mTOR, Rictor, DEPTOR, Raptor in vitro
using six inhibitory agents: Rapamycin (Sigma-Aldrich; Merck KGaA),
Everolimus (Sigma-Aldrich; Merck KGaA), Deforolimus (Selleckchem,
Houston, TX, USA), Temsirolimus (Sigma-Aldrich; Merck KGaA),
NVP-BEZ235 (Selleckchem) and resveratrol (Sigma-Aldrich; Merck
KGaA). Treatments were applied in varying concentrations as given
in Table I for 24, 48 and 72
h.
 | Table IDetails of the mTOR pathway
inhibitory agents used in this study.a |
Table I
Details of the mTOR pathway
inhibitory agents used in this study.a
| Inhibitor | Concentrations |
|---|
| Rapamycin | 20 nM | 100 nM |
| Everolimus | 20 nM | 100 nM |
| Deforolimus | 100 nM | 1,000 nM |
| Temsirolimous | 10 nM | 100 nM |
| Resveratrol | 25 µM | 50 µM |
| BEZ235 | 10 nM | 100 nM |
Immunofluorescence
Following treatments, MDAH-2774 cells were fixed for
10 min in 4% paraformaldehyde, followed by incubation with 10%
bovine serum albumin (BSA) for 1 h. MDAH-2774 cells were incubated
overnight at 4°C with mTOR primary rabbit monoclonal antibody
(catalog no. 2983, Cell Signalling Technology, Inc.), followed by
the addition of a secondary anti-rabbit Alexa Fluor®
488) (Thermo Fisher Scientific, Pittsburgh, PA, USA; dilution
1:400) and visualisation as previously described (20). ImageJ software was used to analyse
the staining intensity. For each image, 10 individual cells were
captured using the freehand tool, ensuring that the area for each
cell was kept approximately the same. A background measurement for
each image was calculated by taking an average measurement from 5
different areas across the image. This average background
measurement was subtracted from each of the images, producing the
final data that were used for statistical analysis. This process
was repeated for all 14 different images.
ImageStreamx Mark II flow cytometry
Whole blood (1 ml) from patient samples (n=3) was
mixed with 9 ml of red blood cell lysis (RBC) buffer (G
Biosciences, St. Louis, MO, USA), followed by incubation for 10 min
with gentle agitation. Following centrifugation at 1,260 × g for 10
min at 4°C the supernatant was discarded and 2 ml of RBC lysis
buffer was added. This was followed by incubation for 10 min at
room temperature with gentle agitation. Another centrifugation took
place as previously described and the pellet was resuspended with
1.5 ml of PBS. The solution containing CTCs (and some white blood
cells) was centrifuged at 1,450 × g for 3 min. The resulting cell
pellet was resuspended in 1 ml of ice-cold 4% paraformaldehyde for
7 min on ice and then centrifuged for 5 min at 250 × g. Subsequent
staining using specific antibodies against Rictor, Raptor and
DEPTOR diluted in FBS-PBS (1:200) were used as previously described
(24).
The qPCR data of the relative gene expression were
analysed using the ΔCq method whereby the Cq value of the
endogenous control was subtracted from the Cq value of the gene of
interest to calculate the ΔCq and an RQ (relative quantity) value
was calculated by finding 2−ΔΔCq (23). Where more than one reference gene
was used, the RQ values were averaged.
Statistical analysis
Data were presented as means ± SEM. All statistical
tests were performed using Graph Pad Prism software. If
homoscedasticity (variance) of data was proven, an unpaired,
two-tailed Student’s t-test was performed to assess significance in
all cases as no matched pairs of samples were used. If data were
not homoscedastic, an unpaired, two-tailed Student’s t-test with
Welch’s correction was performed to account for variance. Survival
information for TCGA ovarian samples (n=426) was extracted from
UCSC Xena browser repository (https://xenabrowser.net). Patient survival was
measured in days to death or end-of-study. The samples were
fractioned based on the mean gene expression value. The survival
Kaplan-Meier curves were plotted in R using survfit function
from bioconductor (https://bioconductor.org). P<0.05 was considered
statistically significant.
Results
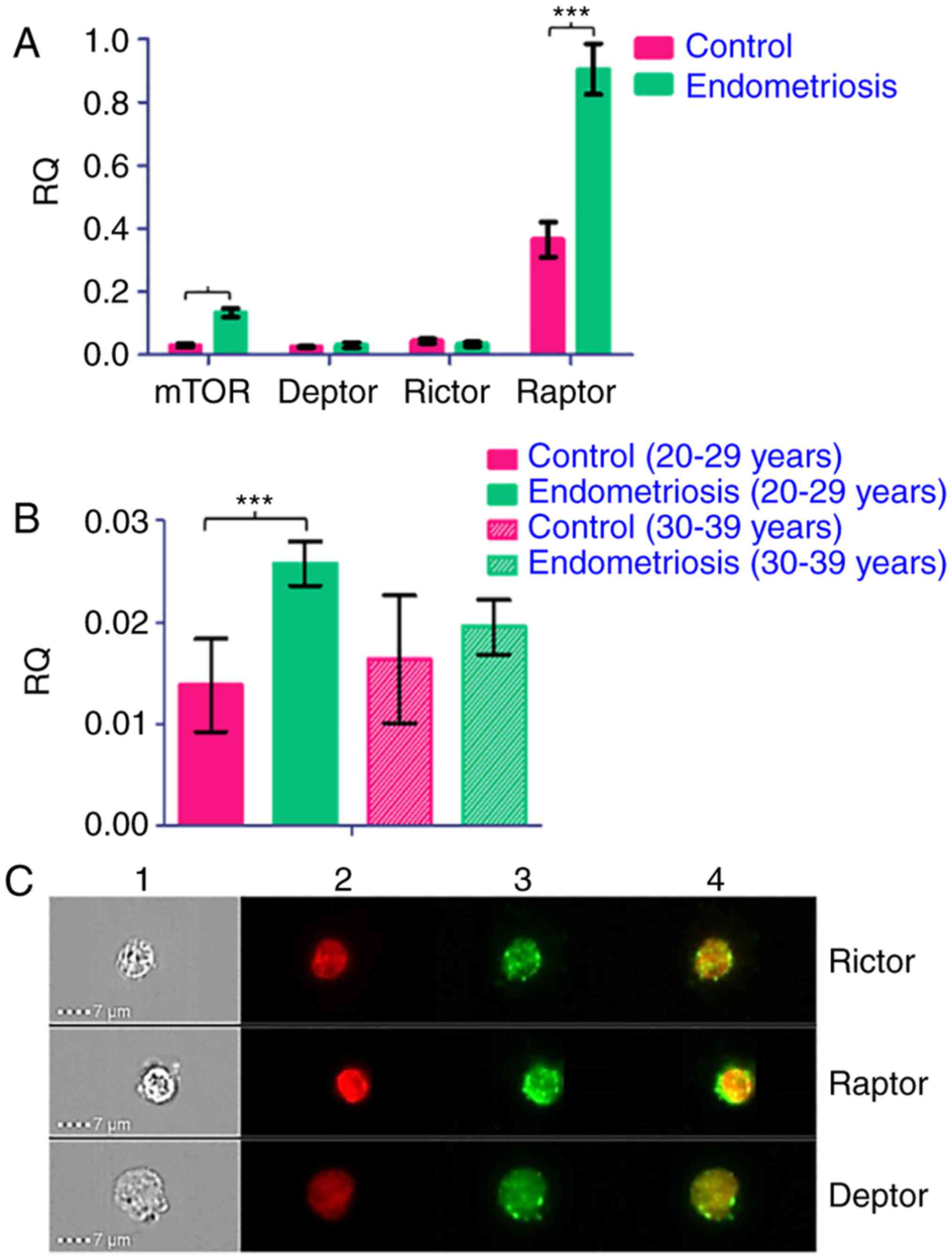
Expression of mTOR, DEPTOR, Rictor and
Raptor in endo- metriosis
qPCR for mTOR, DEPTOR, Rictor
and Raptor was carried out using the reference genes
RPL13A and YWHAZ in triplicate on cDNA synthesised
from the extracted RNA from tissue of endometriosis patients and
from non-affected controls. In endometriosis patients mTOR
and Raptor, but not DEPTOR, showed a significant
increase in expression (Fig. 2A,
P≤0.0001 for both groups). Rictor showed no significant
change overall but when segregated by age of patient (20-29 years
and 30-39 years) and grade points (<50 and >50) significant
changes were evident. Rictor showed a significant increase
in patients aged 20-29 years (P=0.0004) but not in patients aged
30-39 years (Fig. 2B). We
expanded our observations on the expression of mTOR components
using liquid biopsies from ovarian cancer patients given that
endometriosis is a risk factor for this malignancy. We observed
that circulating tumour cells (CTCs) express Rictor, Raptor and
DEPTOR (Fig. 2C).
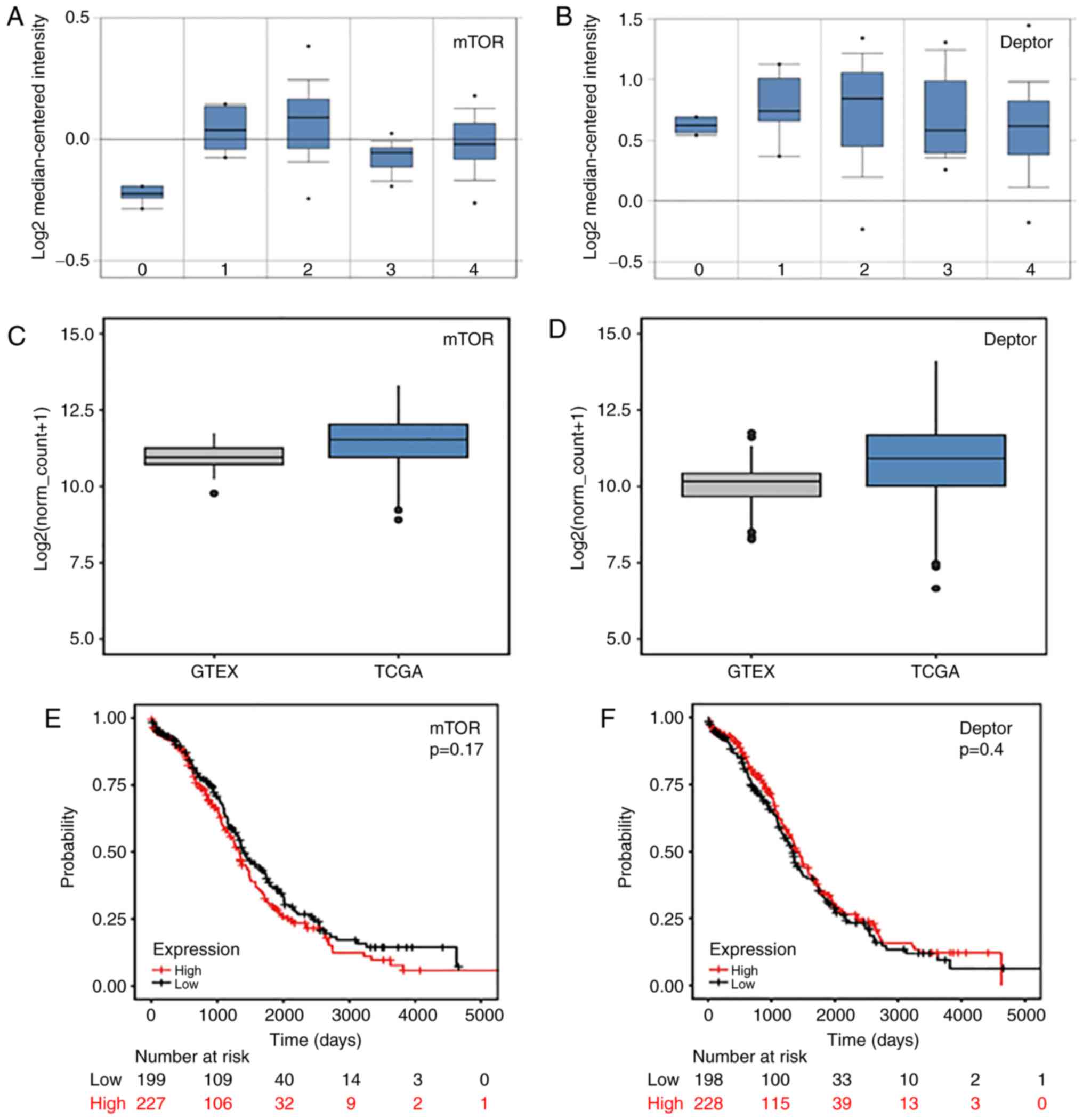
Differential expression of mTOR and
DEPTOR in ovarian cancer
We analysed the expression of mTOR and
DEPTOR using the available RNAseq and microarray data.
Specifically, we compared the expression of the two genes in
various ovarian carcinomas using the Oncomine-curated dataset from
Hendrix et al (25)
(Fig. 2A and B). Both mTOR
and DEPTOR showed an increase in the expression level
compared to the control, across a variety of carcinomas, with mTOR
being significantly upregulated in the ovarian endometrioid
adenocarcinomas (Fig. 3A and B).
In order to remove any potential bias induced by the small sample
size of the Hendrix dataset (n=103), we examined the gene
expression in ovarian serous cystadenocarcinoma samples from TCGA
(n=428). As a control, we used the normal ovarian tissue samples
from GTEX (n=88). All the data were extracted from the UCSC Xena
repository. Ovarian samples from TCGA and GTEX were mapped,
processed, quantified, and normalised using the same pipeline as
described in Vivian et al (26). As previously observed in the
endometrioid dataset, both mTOR and DEPTOR showed a
statistically significant increase in the expression level in
cancer samples compared to normal (p-val=5.26e-13 and 3.43e-13,
respectively) (Fig. 3C and D). In
terms of overall survival, we examined the Kaplan-Meier plots for
the ovarian serous cystadenocarcinoma data from TCGA (n=426). The
samples were separated into high and low expression levels,
respectively, based on the mean expression across the entire
dataset (Fig. 3E and F). We
observed that in the case of mTOR, a higher expression level
is associated with a lower survival rate. By contrast,
DEPTOR suggests that an increase in expression level is
beneficial to the organisms’ survival. However, this result is not
statistically significant.
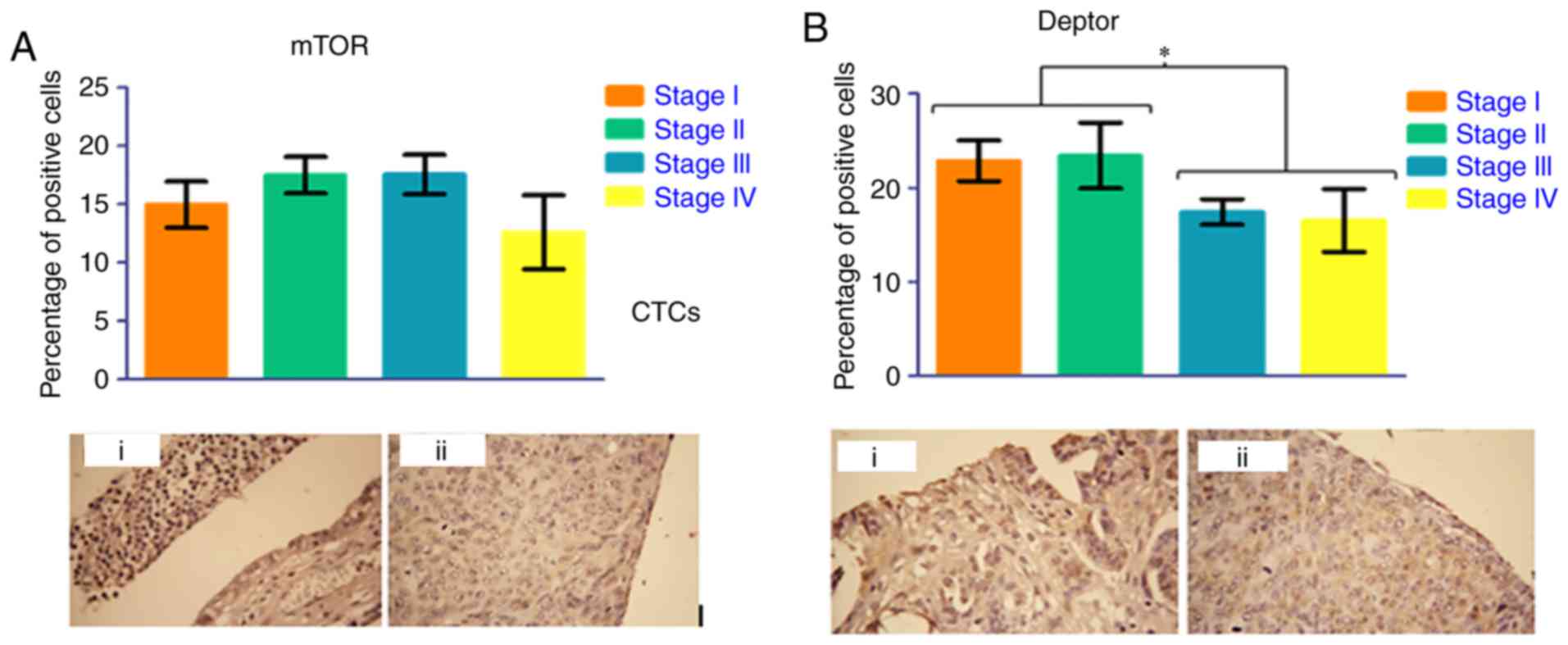
We expanded on these observations by assessing the
protein expression of the two key components of mTORC1 and mTORC2
complexes using tissue microarray. mTOR expression at the protein
level was identified throughout the stages of ovarian cancer with
very little fluctuation. DEPTOR, on the other hand, showed a
significant downregulation when we compared stages III and IV to
stages I and II (Fig. 4A and
B).
Effects of rapalogues on mTORC1 and
mTORC2 components in vitro
Based on our clinical findings, it is evident that
both mTOR and DEPTOR are involved in ovarian cancer.
For this reason, we investigated the effects of rapalogues on
mTORC1 and mTORC2 components in vitro, using the ovarian
cancer cell line of endometrioid origin MDAH-2774 as an
experimental model. MDAH-2774 cells were treated with Rapamycin, as
well as the rapalogues Everolimus, Deforolimus, and Temsirolimus,
NVP-BEZ235 and resveratrol. All the treatments led to a significant
increase in the gene expression of DEPTOR (Fig. 5).
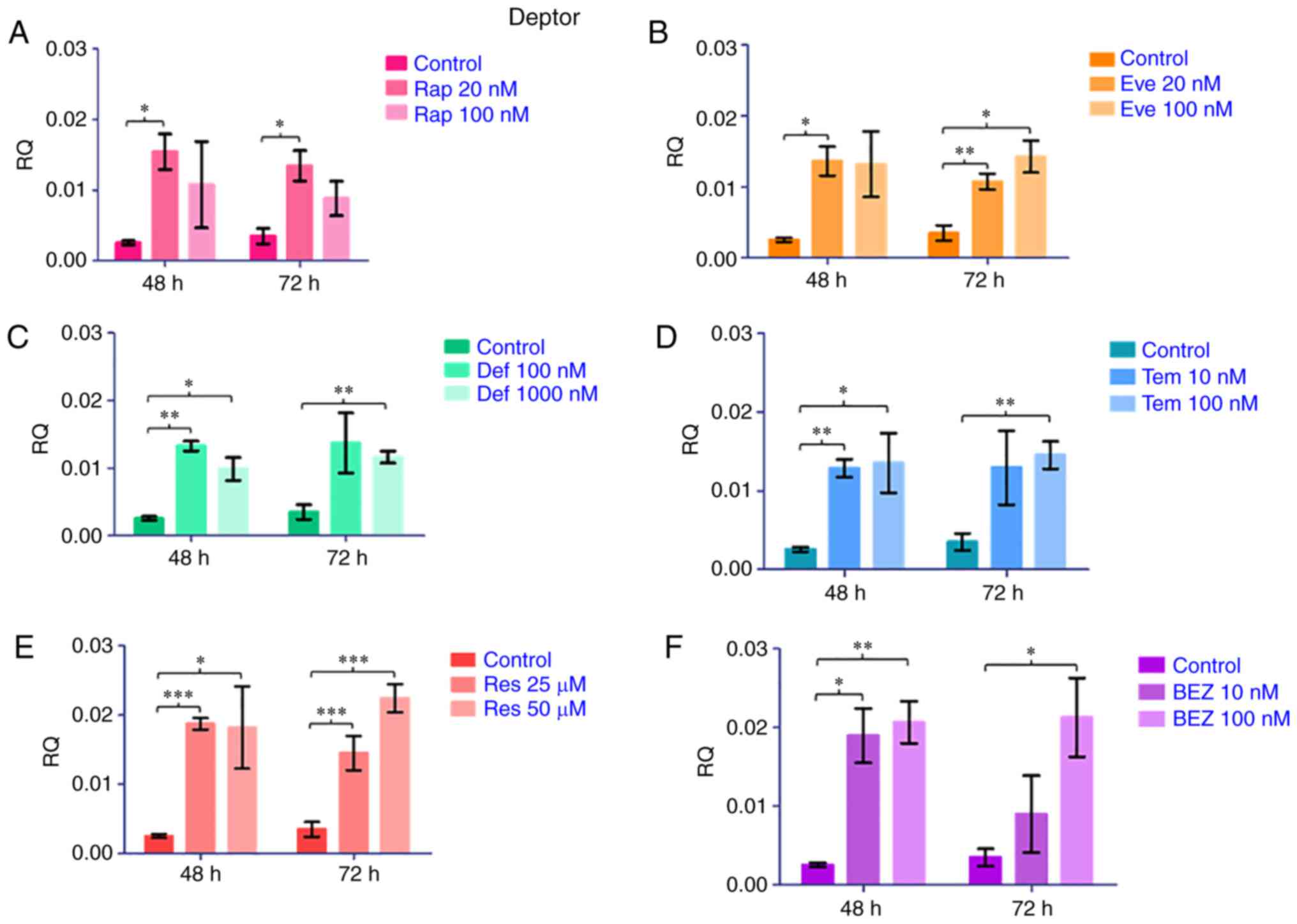 | Figure 5Relative DEPTOR expression was
measured by qPCR in MDAH-2774 cells treated with Rap (A, 20 and 100
nM), Eve (B, 20 and 100 nM), Def (C, 100 and 1,000 nM), Tem (D, 10
nM and 100 nM), Res (E, 25 and 50 µM), BEZ (F, 10 and 100
nM) and carrier (DMSO) only control for 48 and 72 h. cDNA was
synthesised from extracted RNA from 3 biological replicates for
each condition *P<0.05, **P<0.01,
***P<0.001. |
Subsequently, we assessed the expression of
Raptor. MDAH-2774 cells showed an overall trend towards
decreasing Raptor expression in response to mTOR pathway
inhibition (Fig. 6). Rapamycin
and Everolimus treatment induced no change in expression at any
time point or in response to high or low concentrations in
MDAH-2774 cells. Deforolimous treatment (1,000 nM) induced a
decrease in raptor expression after 48 h of treatment in comparison
to the controls (Fig. 6C,
P=0.0067). Temsirolimous treatment (10 nM) induced a decrease in
raptor expression after 48 and 72 h of treatment in MDAH-2774 cells
in comparison to the controls (Fig.
6D, P=0.0457 and 0.0101, respectively). In addition, 100 nM Tem
treatment induced a decrease in Raptor expression after 48 h
of treatment (Fig. 6D, P=0.0115).
Resveratrol treatment (25 µM) resulted in a decreased raptor
expression after 72 h in comparison to controls and 50 µM
Res treatment had the same effect after 48-h treatment (Fig. 6E, P=0.0412 and 0.0318,
respectively). BEZ treatment (10 nM) induced a decrease in
Raptor expression after 48 h of treatment and 100 nM BEZ
treatment resulted in decreased raptor expression after 72 h in
comparison to controls (Fig. 6F,
P=0.012 and 0.0298, respectively).
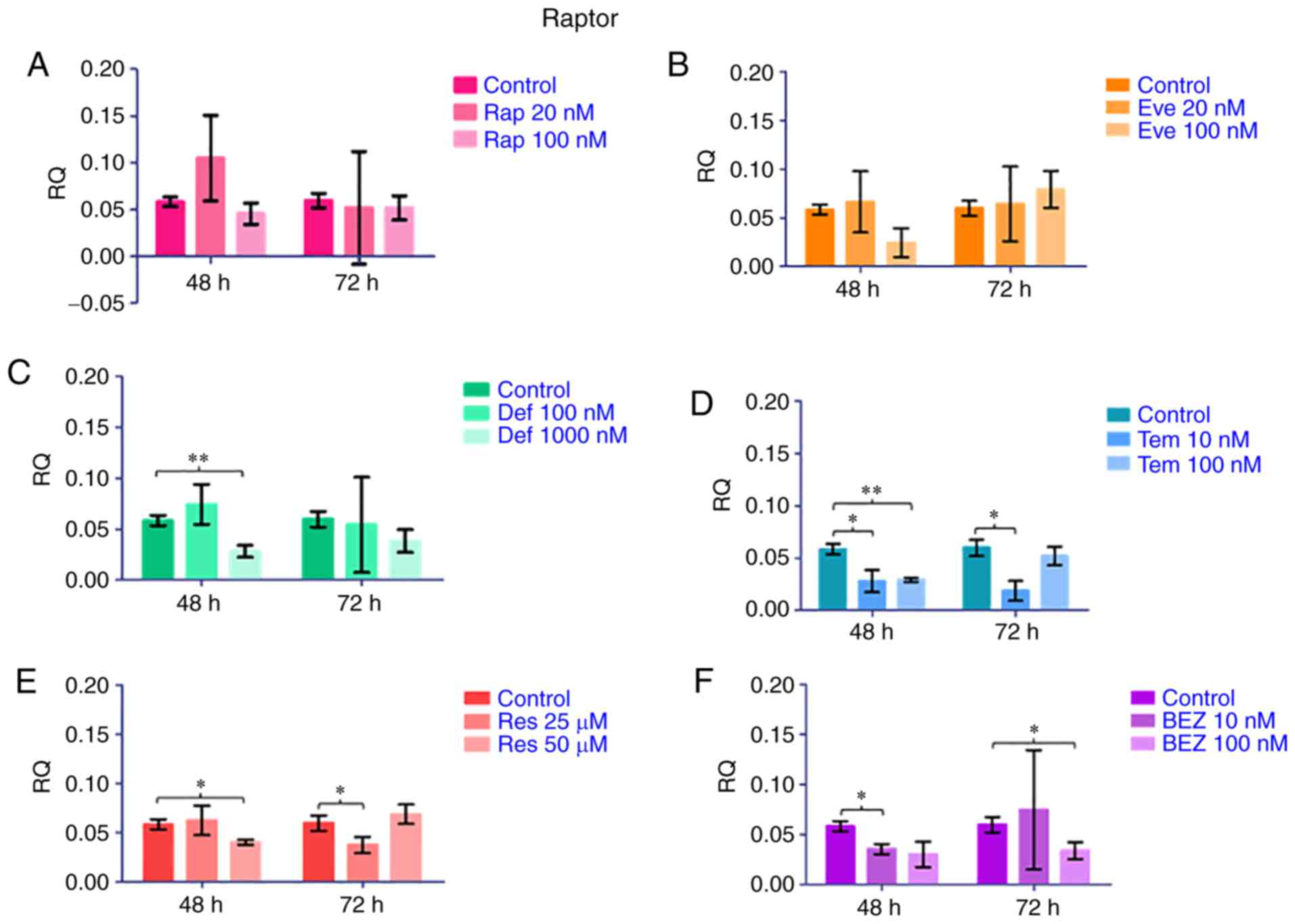 | Figure 6Relative Raptor expression was
measured by qPCR in MDAH-2774 cells treated with Rap (A, 20 and 100
nM), Eve (B, 20 and 100 nM), Def (C, 100 and 1,000 nM), Tem (D, 10
and 100 nM), Res (E, 25 and 50 µM), BEZ (F, 10 and 100 nM)
and carrier (DMSO) only control for 48 and 72 h.
*P<0.05, **P<0.01,
***P<0.001. |
We also assessed the phosphorylation status of
PRAS40 and the protein expression of GβL on MDAH-2774 cells treated
with Rapamycin, Deforolimus, Resveratrol, Everolimus and
Temsirolimus. Little change was observed at 48 h; however, a
reduction of phospho-PRAS40 following treatment with Rapamycin,
Deforolimus, Everolimus and Temsirolimus was evident. Similarly, a
slight decrease in GβL following rapamycin treatment was also
identified (data not shown).
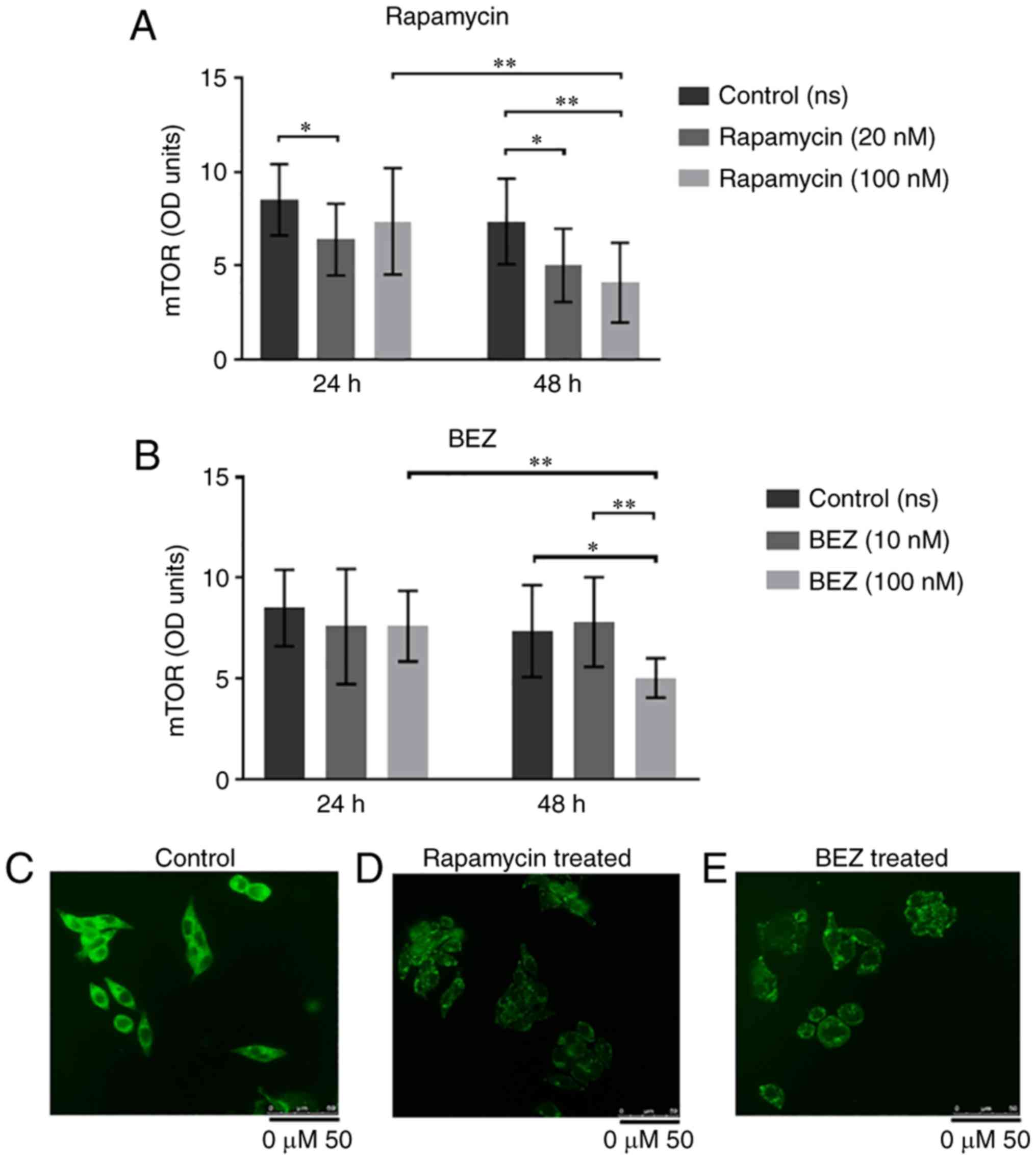
Interestingly, although the same treatments did not
significantly alter mTOR mRNA expression (data not shown), the dual
kinase inhibitor NVP-BEZ235 as well rapamycin significantly
decreased its protein expression at 48 h (Fig. 7). It is evident, therefore, that
the differential expression of mTOR and DEPTOR is crucial for the
activity of the complexes.
Discussion
In this study we provide evidence of differential
expression of mTOR components in patients with endometriosis and
ovarian cancer, and how rapalogues, including dual kinase
inhibitors, can alter the expression of mTOR and DEPTOR.
Collectively our data suggest a crucial role for DEPTOR in ovarian
cancer. In patients with endometriosis there was a significant
increase in the expression of mTOR and Raptor as compared to the
controls. Rictor did not show a significant change in overall
expression, indicating the involvement of mTORC1 rather than
mTORC2. However, Rictor was significantly increased only in those
aged 20-29 years. Proliferative diseases of the endometrium such as
endometriosis and cancer are estrogen-dependent. It is possible
that steroids affect the expression and subsequent activity of mTOR
pathway. In a recent study, it was shown that the activation of
protein synthesis in mouse uterine epithelial cells by
estradiol-17β is mediated via a PKC-ERK1/2-mTOR signalling pathway
(27). In breast cancer, Rictor’s
expression was higher in estrogen receptor-positive cases than in
estrogen receptor-negative cases. Similarly, there is a direct link
between mTORC1 and ERα, since mTORC1 directly phosphorylates and
activates ERα upon estrogen stimulation (28). In a large meta-analysis involving
444,255 patients from 1,625 relevant studies, endometriosis was
strongly associated with the increased risk of ovarian cancer, and
endometriosis-associated ovarian cancer (EAOC) showed favourable
characteristics including early-stage disease, and a specific
histology such as endometrioid or clear cell carcinoma (29).
The shift or change from endometriosis to ovarian
cancer has been suggested to involve the mTOR pathway (30-35). Deletion of PTEN, a tumour
suppressor gene upstream of mTOR and involved in the PI3
kinase/Akt/mTOR axis in mice, induced ovarian endometrioid
adenocarcinoma. In addition, the combination of K-Ras
phosphorylation and PTEN deletion induced the phosphorylation of
Akt, mTOR and p70S6 kinase indicating that the mTOR pathway, either
by PI3 kinase/Akt or Ras/MAP kinase activation is involved in the
transformation of endometriosis to ovarian cancer (30). This is further supported by the
frequent downregulation of PTEN found in ovarian cancers (31,32) and increased K-Ras activation in
endometriosis (33,34). In a more recent study using
targeted next generation sequencing, it was shown that the
PI3K-AKT-mTOR pathway may promote cell malignant transformation
towards EAOC (35).
In the present study, a common pattern of expression
was seen between ovarian cancer and endometriosis. We observed an
increase in mTOR and Raptor expression in endometriosis, suggesting
that mTOR complex 1 is involved in this condition as Raptor is an
exclusive component of mTORC1. Previous findings have shown that,
DEPTOR is involved in the pathogenesis of human malignancies,
primarily via its inhibitory role towards mTORC1 and mTORC2
(36,37). In addition, DEPTOR is
underexpressed in many types of cancer including that of the
prostate, bladder, and cervix, but it is overexpressed in multiple
myelomas, thyroid cancers, and taxol-resistant ovarian cancer cell
lines (28,36,38).
In the present study, although we did not observe
any marked changes in the expression of DEPTOR in either
endometriosis or ovarian cancer patients, there was a significant
decrease in its protein expression when we compared stages I and II
to III and IV in ovarian cancer patients. This is consistent with
the notion that deregulation of DEPTOR can contribute towards a
‘hyperactive’ mTOR pathway in cancer (39). DEPTOR can also be of prognostic
value towards overall survival, since upregulation correlates
positively with better prognosis. These data corroborate a previous
study showing that DEPTOR expression is an independent prognostic
marker for thyroid carcinoma (38). It has also been considered as a
predictive biomarker for therapeutic response in MM patients
treated with thalidomide (40).
However, to date, the exact role of DEPTOR remains controversial
due to its ability to function as an oncogene and as a
tumour-suppressor gene, depending on its cellular or tissue
distribution (40).
Our study opens prospects for the use of DEPTOR,
Rictor, and Raptor as potential biomarkers. Over the past years,
the concept of liquid biopsies has been introduced as an
alternative to conventional tissue biopsy. Circulating tumour cells
(CTCs) are cells that invade into the bloodstream from the primary
tumours in the abdominal cavity (41,42). These cells are different to normal
circulating blood cells and express tumour-specific
characteristics. CTCs are of diagnostic value in various types of
cancer, but the clinical value of CTCs in ovarian cancer remains to
be determined.
In order to gain better insight, we have used the
MDAH-2774 endometrioid ovarian cancer cell line as a preclinical
model to study the mTOR pathway in basal state and upon inhibition
using a wide repertoire of rapalogues, resveratrol and a dual
kinase inhibitor. The findings showed that, inhibition of the mTOR
pathway causes a shift of the DEPTOR/mTOR gene ratio. Moreover,
rapamycin and NVP-BEZ downregulated the protein expression of mTOR.
We hypothesise that DEPTOR overexpression counteracts mTOR activity
acting as a potential drug target. Previous studies have already
demonstrated that use of rapalogues can be of therapeutic potential
in ovarian cancer. Everolimus inhibited the proliferation of human
ovarian cancer cells, and prolonged survival in an ovarian cancer
mouse model in vivo (43).
A more detailed analysis of mTORC1 and mTORC2 signalling must be
carried out to fully explore the extent to which each pathway is
active in endometriosis and ovarian cancer using appropriate in
vivo models with a view to identify the most appropriate drug
targets. Indeed, rapalogues, which robustly inhibit mTORC1
signalling but have little effect on mTORC2 signalling have not
demonstrated any advances in cancer treatment that was expected
from them, with few licenced uses for malignancy (14). Moreover, performing colocalization
experiments on DEPTOR with mTORC1 and mTORC2 components under basal
conditions and in the presence of mTOR inhibitors allows us to
further understand the stoichiometry of DEPTOR within the mTOR
complexes.
The present study is a proof-of-concept pilot study.
This research can be extended in the future with the use of primary
cell lines, pending ethical approval. Moreover, future studies
would provide better insight into the role of mTOR inhibitors
(including dual inhibitors and resveratrol) in the expression of
key components such as Protor or mSIN1 at the gene or protein
level. Moreover, to the best of our knowledge, we have mapped for
the first time, the expression of DEPTOR, Rictor and Raptor in
CTCs. Future studies should demonstrate whether their expression
can be used as a novel biomarker for diagnostic or prognostic
purposes.
Upregulation of DEPTOR is a positive prognostic
marker in ovarian cancer and is increased in response to mTOR
pathway inhibition suggesting that it functions as a tumour
suppressor gene in endometrioid ovarian carcinoma. Collectively,
our data suggest the mTOR pathway as a potential connection between
endometriosis and ovarian cancer and may be a potential target in
the treatment of both conditions (44).
Acknowledgments
Not applicable.
Funding
This study was funded by GRACE, Department of
Gynaecological Oncology, The Royal Surrey County Hospital, Egerton
Road, Guildford, Surrey GU2 7XX; and the Cancer Treatment and
ResearchTrust (CTRT), Mount Vernon Hospital, Rickmansworth Road,
Northwood, Middlesex, HA6 2RN, UK.
Availability of data and materials
All data generated or analyzed during this study are
available from the corresponding author on reasonable request.
Authors’ contributions
EM and MH contributed to the conception and design
of the study. KRB, CS EM, JJ, EK and JK contributed to data
acquisition and analysis. TG, GW, GP, DT, JC and EK were involved
in data analysis and interpretation. TG, GW, JC and EK were
involved in the drafting, writing and revision of the manuscript
for intellectual content. All authors read and approved the final
manuscript.
Ethics approval and consent to
participate
Ethical approval was obtained by the Ethics
Committee of Papageorgiou Hospital, Thessaloniki, Greece and the
Research and Development department of East and North Hertfordshire
NHS Trust, England, UK. Written informed consent was obtained from
all the patients.
Patient consent of publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Sinaii N, Cleary SD, Ballweg ML, Nieman LK
and Stratton P: High rates of autoimmune and endocrine disorders,
fibromyalgia, chronic fatigue syndrome and atopic diseases among
women with endometriosis: A survey analysis. Hum Reprod.
17:2715–2724. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
NHS Choices Endometriosis-NHS Choices.
http://www.nhs.uk/conditions/endometriosis/pages/introduction.aspx.
Accessed June 1, 2016.
|
|
3
|
Sainz de la Cuesta R, Eichhorn JH, Rice
LW, Fuller AF Jr, Nikrui N and Goff BA: Histologic transformation
of benign endometriosis to early epithelial ovarian cancer. Gynecol
Oncol. 60:238–244. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Stewart LM, Holman CD, Aboagye-Sarfo P,
Finn JC, Preen DB and Hart R: In vitro fertilization,
endometriosis, nulliparity and ovarian cancer risk. Gynecol Oncol.
128:260–264. 2013. View Article : Google Scholar
|
|
5
|
Pearce CL, Templeman C, Rossing MA, Lee A,
Near AM, Webb PM, Nagle CM, Doherty JA, Cushing-Haugen KL, Wicklund
KG, et al: Association between endometriosis and risk of
histological subtypes of ovarian cancer: A pooled analysis of
case-control studies. Lancet Oncol. 13:385–394. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Wu AH, Pearce CL, Tseng CC, Templeman C
and Pike MC: Markers of inflammation and risk of ovarian cancer in
Los Angeles County. Int J Cancer. 124:1409–1415. 2009. View Article : Google Scholar
|
|
7
|
Merritt MA, Green AC, Nagle CM and Webb
PM; Australian Cancer Study (Ovarian Cancer); Australian Ovarian
Cancer Study Group: Talcum powder, chronic pelvic inflammation and
NSAIDs in relation to risk of epithelial ovarian cancer. Int J
Cancer. 122:170–176. 2008. View Article : Google Scholar
|
|
8
|
Melin A, Sparén P, Persson I and Bergqvist
A: Endometriosis and the risk of cancer with special emphasis on
ovarian cancer. Hum Reprod. 21:1237–1242. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Borgfeldt C and Andolf E: Cancer risk
after hospital discharge diagnosis of benign ovarian cysts and
endometriosis. Acta Obstet Gynecol Scand. 83:395–400. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Rossing MA, Cushing-Haugen KL, Wicklund
KG, Doherty JA and Weiss NS: Risk of epithelial ovarian cancer in
relation to benign ovarian conditions and ovarian surgery. Cancer
Causes Control. 19:1357–1364. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Brinton LA, Gridley G, Persson I, Baron J
and Bergqvist A: Cancer risk after a hospital discharge diagnosis
of endometriosis. Am J Obstet Gynecol. 176:572–579. 1997.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Brinton LA, Lamb EJ, Moghissi KS, Scoccia
B, Althuis MD, Mabie JE and Westhoff CL: Ovarian cancer risk
associated with varying causes of infertility. Fertil Steril.
82:405–414. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Brinton LA, Sakoda LC, Sherman ME,
Frederiksen K, Kjaer SK, Graubard BI, Olsen JH and Mellemkjaer L:
Relationship of benign gynecologic diseases to subsequent risk of
ovarian and uterine tumors. Cancer Epidemiol Biomarkers Prev.
14:2929–2935. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Laplante M and Sabatini DM: mTOR signaling
in growth control and disease. Cell. 149:274–293. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Zoncu R, Efeyan A and Sabatini DM: mTOR:
From growth signal integration to cancer, diabetes and ageing. Nat
Rev Mol Cell Biol. 12:21–35. 2011. View
Article : Google Scholar
|
|
16
|
Rogers-Broadway KR, Chudasama D, Pados G,
Tsolakidis D, Goumenou A, Hall M and Karteris E: Differential
effects of rapalogues, dual kinase inhibitors on human ovarian
carcinoma cells in vitro. Int J Oncol. 49:133–143. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Leconte M, Nicco C, Ngô C, Chéreau C,
Chouzenoux S, Marut W, Guibourdenche J, Arkwright S, Weill B,
Chapron C, et al: The mTOR/AKT inhibitor temsirolimus prevents deep
infiltrating endometriosis in mice. Am J Pathol. 179:880–889. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Afshar Y, Hastings J, Roqueiro D, Jeong
JW, Giudice LC and Fazleabas AT: Changes in eutopic endometrial
gene expression during the progression of experimental
endometriosis in the baboon Papio anubis. Biol Reprod. 88:442013.
View Article : Google Scholar
|
|
19
|
Potter CJ, Pedraza LG and Xu T: Akt
regulates growth by directly phosphorylating Tsc2. Nat Cell Biol.
4:658–665. 2002. View
Article : Google Scholar : PubMed/NCBI
|
|
20
|
Manning BD, Tee AR, Logsdon MN, Blenis J
and Cantley LC: Identification of the tuberous sclerosis complex-2
tumor suppressor gene product tuberin as a target of the
phos-phoinositide 3-kinase/akt pathway. Mol Cell. 10:151–162. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Ma XM and Blenis J: Molecular mechanisms
of mTOR-mediated translational control. Nat Rev Mol Cell Biol.
10:307–318. 2009. View
Article : Google Scholar : PubMed/NCBI
|
|
22
|
Guo J, Gao J, Yu X, Luo H, Xiong X and
Huang O: Expression of DJ-1 and mTOR in eutopic and ectopic
endometria of patients with endometriosis and adenomyosis. Gynecol
Obstet Invest. 79:195–200. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Foster H, Coley HM, Goumenou A, Pados G,
Harvey A and Karteris E: Differential expression of mTOR signalling
components in drug resistance in ovarian cancer. Anticancer Res.
30:3529–3534. 2010.PubMed/NCBI
|
|
24
|
Chudasama D, Bo V, Hall M, Anikin V,
Jeyaneethi J, Gregory J, Pados G, Tucker A, Harvey A, Pink R and
Karteris E: Identification of cancer biomarkers of prognostic value
using specific gene regulatory networks (GRN): A novel role of
RAD51AP1 for ovarian and lung cancers. Carcinogenesis. 39:407–417.
2018. View Article : Google Scholar :
|
|
25
|
Hendrix ND, Wu R, Kuick R, Schwartz DR,
Fearon ER and Cho KR: Fibroblast growth factor 9 has oncogenic
activity and is a downstream target of Wnt signaling in ovarian
endometrioid adenocarcinomas. Cancer Res. 66:1354–1362. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Vivian J, Rao AA, Nothaft FA, Ketchum C,
Armstrong J, Novak A, Pfeil J, Narkizian J, Deran AD,
Musselman-Brown A, et al: Toil enables reproducible, open source,
big biomedical data analyses. Nat Biotechnol. 35:314–316. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Wang Y, Zhu L, Kuokkanen S and Pollard JW:
Activation of protein synthesis in mouse uterine epithelial cells
by estradiol-17β is mediated by a PKC-ERK1/2-mTOR signaling
pathway. Proc Natl Acad Sci USA. 112:1382–1391. 2015. View Article : Google Scholar
|
|
28
|
Alayev A, Salamon RS, Berger SM, Schwartz
NS, Cuesta R, Snyder RB and Holz MK: mTORC1 directly phosphorylates
and activates ERα upon estrogen stimulation. Oncogene.
35:3535–3543. 2016. View Article : Google Scholar
|
|
29
|
Kim HS, Kim TH, Chung HH and Song YS: Risk
and prognosis of ovarian cancer in women with endometriosis: A
meta-analysis. Br J Cancer. 110:1878–1890. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Dinulescu DM, Ince TA, Quade BJ, Shafer
SA, Crowley D and Jacks T: Role of K-ras and Pten in the
development of mouse models of endometriosis and endometrioid
ovarian cancer. Nat Med. 11:63–70. 2005. View Article : Google Scholar
|
|
31
|
Laudański P, Kowalczuk O,
Klasa-Mazurkiewicz D, Milczek T, Rysak-Luberowicz D, Garbowicz M,
Baranowski W, Charkiewicz R, Szamatowicz J and Chyczewski L:
Selective gene expression profiling of mTOR-associated tumor
suppressor and oncogenes in ovarian cancer. Folia Histochem
Cytobiol. 49:317–324. 2011. View Article : Google Scholar
|
|
32
|
De Marco C, Rinaldo N, Bruni P, Malzoni C,
Zullo F, Fabiani F, Losito S, Scrima M, Marino FZ, Franco R, et al:
Multiple genetic alterations within the PI3K pathway are
responsible for AKT activation in patients with ovarian carcinoma.
PLoS One. 8:e553622013. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Shahrabi-Farahani M, Shahbazi S, Mahdian R
and Amini-Moghaddam S: K-Ras 4A Transcript variant is up-regulated
in eutopic endometrium of endometriosis patients during
proliferative phase of menstrual cycle. Arch Gynecol Obstet.
292:225–229. 2015. View Article : Google Scholar
|
|
34
|
Amemiyaa S, Sekizawaa A, Otsukaa J,
Tachikawa T, Saito H and Okai T: Malignant transformation of
endometriosis and genetic alterations of K-ras and microsatellite
instability. Int J Gynaecol Obstet. 86:371–376. 2004. View Article : Google Scholar
|
|
35
|
Er TK, Su YF, Wu CC, Chen CC, Wang J,
Hsieh TH, Herreros-Villanueva M, Chen WT, Chen YT, Liu TC, et al:
Targeted next-generation sequencing for molecular diagnosis of
endometriosis-associated ovarian cancer. J Mol Med (Berl).
94:835–847. 2016. View Article : Google Scholar
|
|
36
|
Peterson TR, Laplante M, Thoreen CC,
Sancak Y, Kang SA, Kuehl WM, Gray NS and Sabatini DM: DEPTOR is an
mTOR inhibitor frequently overexpressed in multiple myeloma cells
and required for their survival. Cell. 137:873–886. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Kazi AA, Hong-Brown L, Lang SM and Lang
CH: Deptor knockdown enhances mTOR Activity and protein synthesis
in myocytes and ameliorates disuse muscle atrophy. Mol Med.
17:925–936. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Pei L, Xie P, Zhou E, Yang Q, Luo Y and
Tang Z: Overexpression of DEP domain containing mTOR-interacting
protein correlates with poor prognosis in differentiated thyroid
carcinoma. Mol Med Rep. 4:817–823. 2011.PubMed/NCBI
|
|
39
|
Wang Z, Zhong J, Inuzuka H, Gao D, Shaik
S, Sarkar FH and Wei W: An evolving role for DEPTOR in tumor
development and progression. Neoplasia. 14:368–375. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Catena V and Fanciulli M: Deptor: Not only
a mTOR inhibitor. J Exp Clin Cancer Res. 36(12): 2017
|
|
41
|
Chudasama DY, Freydina DV, Freidin MB,
Leung M, Montero Fernandez A, Rice A, Nicholson AG, Karteris E,
Anikin V and Lim E: Inertia based microfluidic capture and
characterisation of circulating tumour cells for the diagnosis of
lung cancer. Ann Transl Med. 4:4802016. View Article : Google Scholar
|
|
42
|
Chudasama D, Burnside N, Beeson J,
Karteris E, Rice A and Anikin V: Perioperative detection of
circulating tumour cells in patients with lung cancer. Oncol Lett.
14:1281–1286. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Mabuchi S, Altomare DA, Cheung M, Zhang L,
Poulikakos PI, Hensley HH, Schilder RJ, Ozols RF and Testa JR:
RAD001 inhibits human ovarian cancer cell proliferation, enhances
cisplatin-induced apoptosis, and prolongs survival in an ovarian
cancer model. Clin Cancer Res. 13:4261–4270. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Grunt TW and Mariani GL: Novel approaches
for molecular targeted therapy of breast cancer: Interfering with
PI3K/AKT/mTOR signaling. Curr Cancer Drug Targets. 13:188–204.
2013. View Article : Google Scholar
|