Research on cancer formation and treatment has
finally started to reduce the mortality of cancer patients. In
general, mortality rates decreased by 1.5% per year from 2001
through 2015, although these rates may differ depending on patient
sex, ethnic group and type of malignancy. Nevertheless, cancer
remains a significant global health problem, requiring constant
search for novel treatments (1).
The most common property of the malignant tumors is their ability
to invade adjacent tissue and to metastasize to distant sites
(2,3). This is due to increased expression
or increased activity of numerous proteases associated with
carcinogenesis (4-9). For example, cysteine proteases, such
as cathepsin B and cathepsin D, can take part in dissolving of
connective tissue and basement membrane needed for invasion and
metastasis. However, these become activated at the low pH of
lysosomes, requiring transporting into regions of tumor cell
invasion (10-13). Aspartate proteases and other
protease enzymes also serve a role in cancer development and
metastasis (14-16). Nevertheless, among all these
proteolytic events, the plasminogen and metalloproteinase family
members have an exceptional significance due to their ability to
cleave virtually any element of the extracellular matrix and
basement membrane (17-20).
The components of the plasminogen activation system
(PAS) and matrix metalloproteinase (MMP) families have been found
to be overexpressed in malignant tumors, revealing that tumors are
hijacking these systems and using them in the local invasion,
metastasis and angiogenesis (21-23). One of the hallmarks of this cancer
aggressiveness is the overexpression of proteolytic enzymes
(24-27). For instance, prostate cancer is
the most common non-cutaneous malignancy in men with >160,000
new cases in the United States each year, and the median age of
diagnosis is 68 years. This cancer grows slowly, and is initially
confined to the gland, requiring marginal or no treatment at all.
However, certain types of prostate cancer are aggressive, can
spread quickly and exhibit high proteolytic activity (28-31). This is also a case for other types
of cancer, such as breast, colon and oral carcinomas among others
(32-36).
In the present review, new findings on the role of
plasminogen activation system (PAS) and metalloproteinase families
in cancer formation and growth were summarized, and the study
explored whether targeting these two protein families can serve as
a possible treatment strategy.
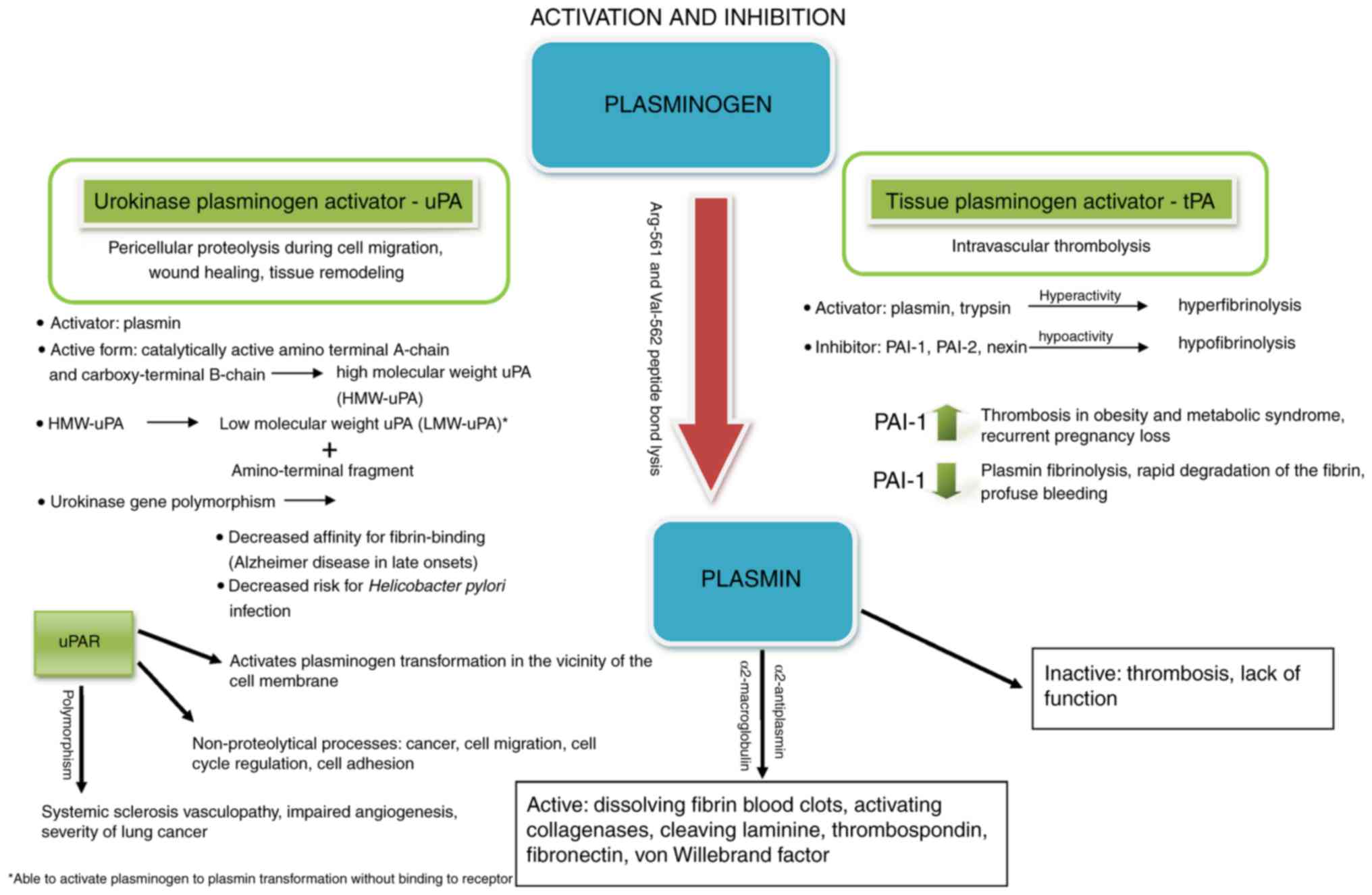
Plasminogen is synthetized as a zymogen in the liver
and released into the systemic circulation. In the circulation,
plasminogen is resistant to activation by adopting a closed
conformation. When bound to blood clots or to the cell surface,
plasminogen changes to an open form that can be cleaved into active
plasmin by a variety of enzymes. These enzymes are mainly
plasminogen activators, namely tissue plasminogen activator (tPA)
and urokinase plasminogen activator (uPA), while other enzymes
include kallikrein and factor XII. Plasminogen is converted into
plasmin by the cleavage of the peptide bond between Arg-561 and
Val-562 (37,38). Plasmin, a member of the serine
protease family, was originally considered to only act to dissolve
fibrin blood clots. However, in addition to fibrinolysis, plasmin
cleaves proteins in numerous other systems. For instance, it
activates collagenases, and cleaves fibronectin, thrombospondin,
laminin and von Willebrand factor (26,35,38). Two major forms of plasminogen are
detected in humans, including type I and type II. Notably, type I
plasminogen has two glycosylation moieties, namely N-linked to N289
and O-linked to T346, which is preferentially recruited to the cell
surface. Type II plasminogen contains only O-linked sugar to T346,
which appears to be more readily recruited to blood clots (39-41).
To prevent premature or unnecessary proteolysis, the
activity of plasmin needs to be tightly controlled. Two proteins,
α2-macroglobulin and α2-antiplasmin, inactivate plasmin, and the
underlying mechanism involves the cleavage of α2-macroglobulin at
the bait region by plasmin. This initiates a conformational change
resulting in the α2-macroglobulin-plasmin complex, where the active
site of plasmin is sterically shielded, decreasing the access of
plasmin to its substrates (42).
In addition, a major conformational change exposes a conserved
COOH-terminal receptor binding domain that allows the
α2-macroglobulin protease complex to bind to clearance receptors
and consequently be removed from the circulation (43). Furthermore, α2-antiplasmin forms
an irreversible complex with plasmin by attaching to Arg-376 in the
reactive center loop, resulting in the complete loss of plasmin
activity (42).
A rare disorder in humans known as plasminogen
deficiency type I may lead to thrombosis, as blood clots are not
effectively degraded. Plasminogen deficiency in humans is caused by
numerous mutations and polymorphisms in plasminogen gene and is
often manifested by ligneous conjunctivitis (44). Ligneous conjunctivitis is
characterized by fibrin-rich pseudomembranous lesions that develop
mainly on the underside of the eyelid, which can be
sight-threatening (45). It may
also influence the periodontal tissue, upper and lower respiratory
tract, kidneys, middle ear and female genitalia, and affects wound
healing (46-50).
The encoded preprotein of tPA is proteolytically
cleaved by plasmin or trypsin to produce heavy and light chains.
These chains are linked by disulfide bridges to form an active
heterodimeric enzyme that is able to activate plasminogen (53). Increased tPA activity triggers
hyperfibrinolysis, which causes excessive bleeding and an increase
of the vascular permeability (54-57), while decreased activity leads to
hypofibrinolysis, which can result in thrombosis or embolism
(58-60).
Similar to tPA, uPA is synthetized as a preprotein
and is activated by cleavage at a Lys-Ile bond by plasmin to form a
two-chain enzyme connected by a disulfide bond. In its active form,
uPA contains the amino terminal A-chain that is catalytically
active, and the carboxy-terminal B-chain. This two-chain derivative
form is referred to as high molecular weight uPA (HMW-uPA). HMW-uPA
can be cleaved at the A-chain into a short chain A (A1), forming
low molecular weight uPA (LMW-uPA) and an amino-terminal fragment.
LMW-uPA is able to activate plasminogen, however, it does not bind
to the uPA receptor (uPAR) (61,62). A C/T single-nucleotide
polymorphism (SNP) in codon 141 of urokinase (known as P141L) may
be associated with late onset Alzheimer’s disease, decreased
affinity for fibrin-binding (63)
and reduced risk of Helicobacter pylori infection (64).
uPAR, also known as cluster of differentiation 87
(CD87), was originally identified as a saturable binding site for
uPA and contains three domain glycoprotein bound to the cell
surface with a glycosylphosphatidylinositol (GPI) anchor. All
domains of uPAR are needed for high affinity binding of the
urokinase (21,27,35). Urokinase receptor anchors uPA and
therefore confines plasminogen activation in the vicinity of the
cell membrane. However, when uPA is bound to its receptor, it may
be cleaved in the proximity of the GPI anchor, and the uPAR is
released as a soluble receptor (65-67). Urokinase receptor has also been
suggested to be involved in non-proteolytic processes, such as
cancer, cell migration, cell cycle regulation or cell adhesion
(35,65-67). A previous study reported that
rs344781 (516 T/C) uPAR polymorphism was implicated in systemic
sclerosis vasculopathy, impaired angiogenesis (68) and the severity of lung cancer
(69).
Plasminogen activator inhibitor-1 (PAI-1), also
known as endothelial PAI or serine protease inhibitor E1, is a
fast-acting, high-affinity, principal inhibitor of tPA and uPA. The
other PAI, namely PAI-2, is only produced in physiologically
significant amounts during pregnancy and secreted by the placenta.
Protease nexin can also inhibit tPA and uPA, however, PAI-1 remains
a major inhibitor of plasmin-driven proteolysis (35,70-72). PAI-1 is overexpressed in various
diseases, such as obesity and metabolic syndrome, and has been
linked to risk of thrombosis in patients with these conditions
(36,73,74). Also, it has been reported that a
high activity of PAI-1 is associated with recurrent pregnancy loss
(75,76). By contrast, a low level of PAI-1
leads to excessive plasmin fibrinolysis that is unopposed by PAI-1
and rapid degradation of the fibrin, which may manifest in profuse
bleeding. Indeed, it was reported that a life-long bleeding
tendency was caused by undetectable PAI-1 activity and antigen
levels in a 76-year-old man, while severe menorrhagia has been
reported in patients with a low PAI-1 antigen level (77-79). Notably, in the case of low
activity of PAI-1, women have achieved pregnancy without
difficulty, but experienced antenatal bleeding and preterm labor
(80).
The promoter polymorphisms (844 A/G and 675 4G/5G)
in the PAI-1 gene yield higher plasma PAI-1 levels (81). Another SNP with substitution of
A15 to T15 and possibly V17 to Ile in the signal peptide leads to
lower PAI-1 activity compared with a control (78,82,83). In addition, a previous study
reported that a young Amish girl and certain members of her
extended immediate family had no PAI-1 antigen and PAI-1 activity.
In addition, a previous study reported that a young Caucasian girl
from an Amish congregation and certain members of her extended
family had no PAI-1 antigen and PAI-1 activity, leading to
excessive bleeding. They were found to be homozygous for a
dinucleotide insertion within exon 4 of PAI-1 gene, producing a
truncated, non-functional protein (78,79). The diverse function of PAS, as
discussed in the present study, is outlined in Fig. 1.
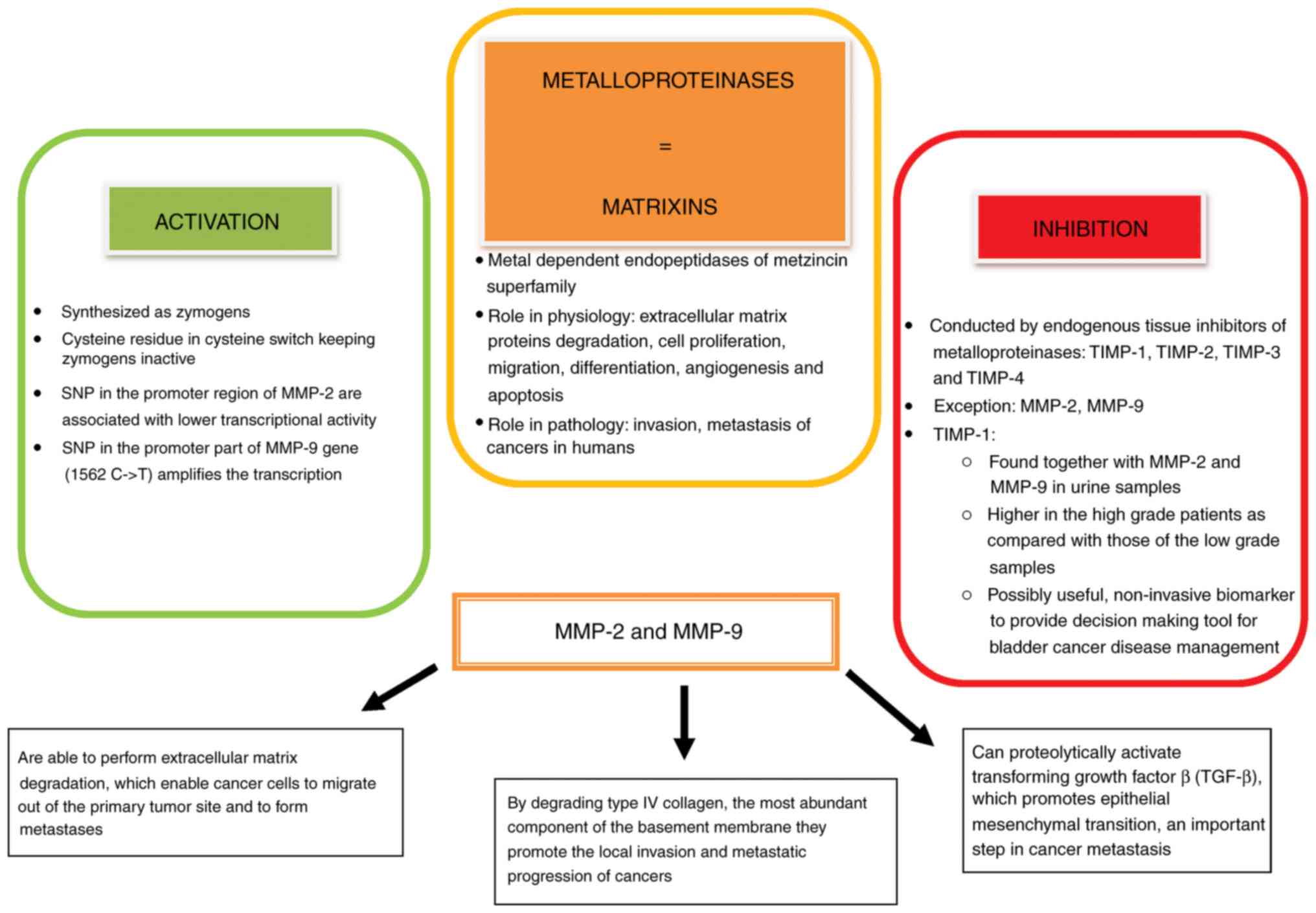
MMPs also known as matrixins, are metal-dependent
(Ca and Zn) endopeptidases that belong to a larger family known as
the metzincin superfamily (84-87). These enzymes degrade all types of
extracellular matrix proteins and are differentiated from other
endopeptidases by their dependence on metal ions as cofactors
(88,89). MMPs are synthesized as inactive
zymogens with a domain that must be removed to activate enzymes.
This domain is part of the cysteine switch, containing a cysteine
residue that prevents metals binding and keeps the enzyme in an
inactive form (90-93). Matrix metalloproteinases are also
involved in cell proliferation, migration, differentiation,
angiogenesis and apoptosis (92,94).
MMP-2 and MMP-9, two members of the 25-protein
family of MMPs, are considered to be critical in local invasion and
metastasis of cancer in humans (94,95). One of the most important functions
of these enzymes in cancer progression is their ability of
extracellular matrix degradation, which enables cancer cells to
migrate out of the primary tumor site and to form metastases. Both
MMP-2 and MMP-9 are capable of degrading type IV collagen, the most
abundant component of the basement membrane, which is an essential
step for the local invasion and metastatic progression of cancer
(95,96). In addition, MMP-2 and certain
other MMPs can proteolytically activate transforming growth factor
β, which promotes epithelial mesenchymal transition, an important
step in cancer metastasis (96,97). Diverse functions of
metalloproteinases are outlined in Fig. 2.
SNPs in the promoter region of MMP-2 (1306C->T
and 735C->T) are associated with lower transcriptional activity,
and the SNP in the promoter part of MMP-9 gene (1562C->T)
amplifies the transcription of this metalloproteinase gene. Others
SNPs in the coding region of the MMP-9 gene include 836A>G,
1721G>C and 2003G>A, which produce a missense amino acid
sequence, reducing the substrate and inhibitor binding ability of
MMP-9 (98,99).
MMPs are inhibited by four different specific
endogenous tissue inhibitors: TIMP-1, TIMP-2, TIMP-3 and TIMP-4.
All MMPs are inhibited by TIMPs when in their active form, with the
exception of MMP-2 and MMP-9, which can form complexes with TIMPs
in the latent form (99,100). Ricci et al (101) investigated MMP-2 and MMP-9, as
well as the inhibitors TIMP-1 and TIMP-2 in the urine and sera of
bladder cancer patients. The data revealed that urinary TIMP-1 was
significantly higher in high-grade patients, as compared with the
levels in the low-grade samples (101). The results also revealed a
significantly differing distribution of TIMP-1 expression between
Ta and T1 stage specimens, and the authors concluded that urinary
TIMP-1 can be a useful, non-invasive biomarker that may serve as a
decision making tool for bladder cancer disease management
(101). Three SNPs were reported
in TIMP-1, including 261 C->T, 328+16 C->T and 372 T->C,
(102), while six SNPs were
reported in TIMP-2, including rs2277698 T->C, rs2009196 C->G,
rs7342880 A->C, rs11654470 C->T, rs2003241 C->T, rs4789936
T->C (103).
The hallmark of metastatic disease is an altered
equilibrium between the synthesis and degradation of the
extracellular matrix proteins during pathological angiogenesis,
invasion and metastasis (35,102).
Angiogenesis is the physiological or pathological
development of revascularization from existing vessels. It is a
normal process during growth and development, and in wound healing;
however, angiogenesis is also an essential step in malignant tumor
growth (26,27,34,35). Sprouting angiogenesis occurs as
angiogenic growth factors activate receptors on endothelial cells,
followed by the release of proteases (PAS and MMP) that degrade the
basement membrane, permitting cells to escape in tandems to or from
the vessel walls (35,110). The endothelial cells then
proliferate into the surrounding matrix and form solid sprouts,
connecting neighboring vessels. These processes are basically the
same in both normal and aberrant angiogenesis (26,35,104).
It has previously been reported that receptor-bound
uPA on the cancer cell surface is overexpressed in final step of
malignant cell transformation that is responsible for invasion and
metastasis (107). In addition,
uPA has been implicated in the basement membrane and interstitial
protein degradation during carcinogenesis, and is responsible for
the disease progression (108-110). Therefore, inhibition of uPA may
be beneficial in the treatment of cancer by limiting or preventing
tumor growth and metastasis (26,111,112). Historically, one of the first
uPA specific inhibitors identified was amiloride, which limits
tumor growth and angiogenesis, as proven by in vitro and
in vivo models (26,108,110,111,113). Amiloride has been approved by
the Food and Drug Administration as a diuretic agent, however, it
is not currently used in anticancer therapy.
The availability of the high resolution structure of
uPA and sophisticated molecular modeling methods have aided the
development of inhibitors binding to uPA. The specificity pocket of
uPA is a part of this enzyme, which is responsible for the
recognition of uPA substrates, therefore binding to the specificity
pocket promises high specificity of inhibitors (35,67,105). Several 8-substituted
2-naphthamidine-based inhibitors with specific binding to uPA with
high potency and selectivity were developed by Wendt et al
(114). Among the multiple novel
inhibitors developed by the Bruncko et al (115) and Katz et al (116) groups, only a limited number of
inhibitors were translated into clinical practice. One of these is
WX-UK1, also known as ethyl 4-[3-(3-carbamimidoylphenyl)-
2-[[2,4,6-tri(propan-2-yl)phenyl]sulfonylamino]propanoyl]
piperazine-1-carboxylate or Mesupron®, which is one of
the most potent uPA inhibitors (0.41 µM), and is currently
in Phase I clinical trial for pancreatic and breast cancer, with
the possibility of application in ovarian and colon cancer as well
(117-120).
Another possibility for inhibiting uPA is through
the use of antibodies. For instance, a number of studies by
Ossowski (121,122) have reported that the ability to
invade the chick chorioallantoic membrane and metastasize from it
to the embryo was evidently reduced for human carcinoma cells
expressing a high amount of uPA during treatment with the antibody
against the active site of uPA, in comparison to non-treated
cells.
PAI-1 is also an attractive molecule for anticancer
therapy, since it is a fast-acting, specific and high-affinity
inhibitor of uPA (35,123). However, only active PAI-1 can be
used to control angiogenesis, invasion and metastasis of malignant
tumors. The half-life of wild-type PAI-1 is very short
(t1/2 = 1-2 h), after which it converts into its
inactive latent form. This transformation is associated with the
insertion of part of the reactive loop (P4-P10′) into the central
β-sheet of the PAI-1 molecule, where P1-P1′ amino acids are not
accessible for reaction with tPA or uPA (35,124). For that reason, wild-type PAI-1
cannot be used in anti-cancer therapy. Numerous engineered PAI-1
molecules with two or more mutations were developed, which
exhibited a half-life extended from 2 h to >700 h, opening a
possibility for their application in clinical settings (124-126). For instance, VLHL PAI-1
(t1/2 = >700 h) has been demonstrated to inhibit
angiogenesis and reduce tumor size in different ex vivo and
in vivo models (26,71,110).
Notably, PAI-1-deficient mice exhibited lower tumor
proliferation and higher apoptosis of implanted tumors when
compared with their wild-type counterparts. Additionally, an
elevated PAI-1 level is a predictive factor of poor prognosis in
invasive breast cancer in humans (34,127,128). When PAI-1 was administrated in
low concentrations, it was able to increase the metastatic
potential and angiogenesis (34,127-129). These observations contradict our
previous statements suggesting that PAI-1 is a potent inhibitor of
angiogenesis and tumor growth (26,125,128,130). This paradox can be explained by
the interaction of PAI-1 with vitronectin. This protein is a
multifunctional glycoprotein present in the plasma, platelets and
the extracellular matrix. PAI-1 binds to vitronectin and then binds
with uPA/uPAR, forming a four protein complex (namely
uPAR/uPA/PAI-1/vitronectin), which can be further combined with LDL
receptor-related protein. This interaction weakens the
PAI-1/vitronectin binding and triggers PAI-1/uPA/uPAR
internalization. PAI-1 and uPA are degraded, whereas uPAR is
recycled to the cell surface. Subsequently, uPAR can bind to uPA
again and form a new PAI-1/uPA/uPAR/vitronectin complex. This
process clears PAI-1 in the immediate vicinity of the cell, forcing
it to migrate toward increased concentration of PAI-1 (131,132). Thus, PAI-1 can have a dual
function as a pro-carcinogenic and anti-cancer agent, depending on
its concentration. PAI-1 in physiological concentration acts by
utilizing the vitro-nectin pathway. However, as it has been
demonstrated in our previous studies and by other groups, when
PAI-1 is present in much higher supraphysiologic concentrations, it
suppresses the vitronectin pathway and acts as a potent inhibitor
of angiogenesis by utilizing primarily its inhibitory properties to
block proteinase activity (133-136).
uPAR (or CD87) is a multidomain glycoprotein
attached to the cell membrane with a glycosylphosphatidylinositol
(GPI) anchor. When binding to uPA, it creates high proteolytic
activity in the proximity of the cell surface (137-139). Therefore, another approach to
prevent invasion or metastasis is to block the binding of uPA to
its receptor on the cancer cell surface. Similarly, limiting the
binding of uPA to vascular cells would reduce cancer-associated
angiogenesis. An octapeptide A6 (amino acids 136-143;
nonreceptor-binding domain) of uPA inhibits the interaction of uPA
with its receptor uPAR, and was reported to inhibit endothelial
cell motility and tumor cell invasion (117,140,141). Furthermore, the use of this
polypeptide has been reported in Phase I and II clinical studies of
gynecological cancer (142).
Mani et al (143)
synthesized a methyl 3-aminobenzoate derivative that binds to uPAR
with high affinity and demonstrated that this uPAR inhibitor lowers
the invasion ability of breast MDA-MB-231 cells. In addition,
female NSG mice inoculated with highly malignant TMD-MDA-MB-231
cells presented impaired metastasis to the lungs when treated with
this uPAR inhibitor (143). In a
subsequent study by Wang et al (144), the affinity of a large number of
derivatives was measured using structure-based virtual screening.
The synthesis of additional and cellular studies in cell lines
(A549, H460 and H1299) revealed that these compounds blocked
invasion, migration and adhesion, while the effects of these
compounds on invasion were consistent with their inhibition of uPAR
(144). Treatment of metastatic
liver cells with 1 or 4 g/ml lectin from Bandeiraea
simplicifolia seeds significantly inhibited the cell migration
and invasion, and downregulated MMP2, MMP9 and uPA production
(145). Furthermore, the
AKT/GSK-3b/β-catenin pathway, which is upstream of MMP2, MMP9 and
uPA, was found to be involved in the inhibition of cell migration
and invasion by lectin (145).
In a similar manner, quercetin functions on the uPA/uPAR system
(146).
Inhibitors targeting MMP activity were initially
intended to target the catalytic domain of these enzymes. The most
clear approach was to synthetize polypeptides derived from the
sequences of the amino acids of the MMP substrates, preventing
hydrolysis of the scissile bond of proteins (147). Batimastat (INN/USAN, codenamed
BB-94), a broad-spectrum, competitive, injectable drug was the
first MMP inhibitor (MMPI) to go into clinical trials, acting as an
angiogenesis inhibitor. While this agent reached Phase III trials,
it was never marketed due to side effects (injection into the
peritoneum caused peritonitis), poor solubility and very low oral
bioavailability (147-149). Marimastat, another MMPI drug,
exhibited some effects in delaying disease progression; however, it
also had a dose-limiting toxicity. Patients experienced significant
musculoskeletal pain and inflammation, and the clinical study was
discontinued (147,150). The small molecular inhibitors of
MMPs failed for a variety of reasons, despite preclinical data
demonstrating their great potential for therapeutic use, leading to
cancelled phase I trials on Prinomastat, and cancelled phase III
trials on Batimastat, Marimastat, Tanomastat and CGS 27023A, which
is a doxycycline tetracycline derivative (147).
The mainstream of the presently used
chemotherapeutic agents attack all dividing cells, leading to the
adverse effects that are typically associated with cancer
treatment, such as hair loss or loss of appetite. For future
therapeutics to be clinically successful, they need to be highly
selective for a specific protein or proteins, and able to act on
the cancerous tissues without causing adverse systemic effects.
Despite unsuccessful early clinical studies on the inhibition of
proteolysis in cancer (particularly MMPs), this approach still has
great potential for therapeutic use. Inhibition of proteolysis in
cancerous tissue has the ability to attenuate tumor invasion,
angiogenesis and migration, which are the most fundamental
properties of malignant tumors, thus deserving future development
(147,153). This includes application of
inhibitors at the time and stage of malignancy for which regulation
of pathological proteolysis will be most beneficial. For instance,
inhibiting cancer proteolytic enzymes during or after surgery can
prevent cancer cell implantation and consequently cancer reemission
(113). Inhibiting both uPA and
MMPs is another beneficial approach, since tumors overexpress these
groups of enzymes; therefore, employing this simple concept can be
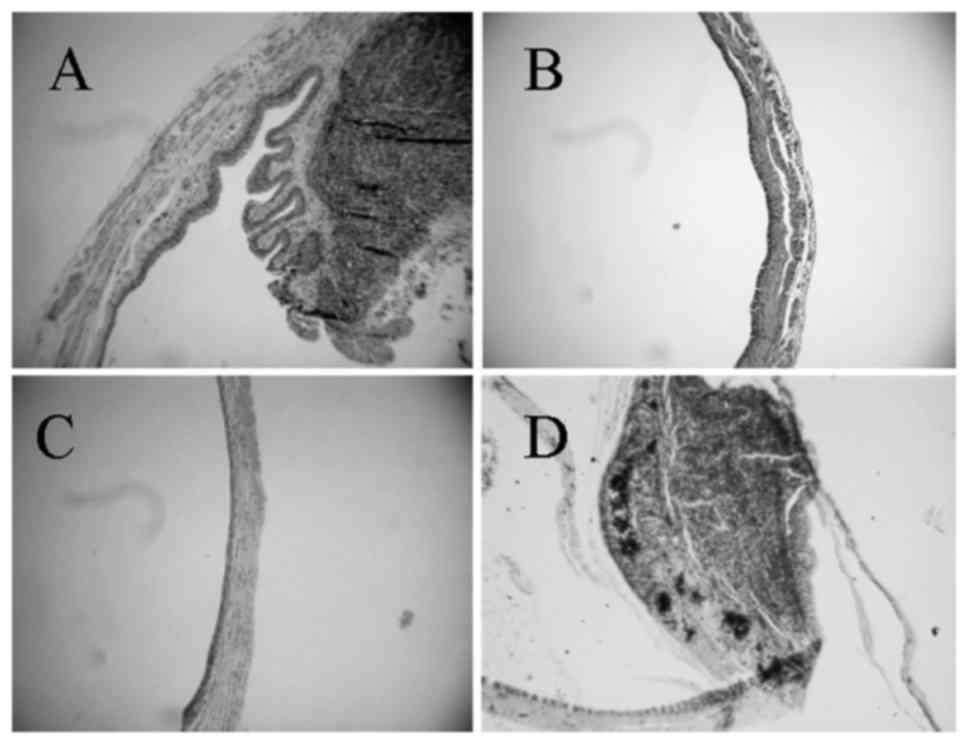
equally beneficial as cancer cell killing (Fig. 3). Epigallocatechin gallate, an
inhibitor of uPA and MMPs, has previously been used, and this agent
had a weak lethality, but prevented cancer cell implantation into
the bladder wall following surgery and was slightly better in
reducing cancer growth when compared with mitomycin C (Fig. 4) (113). Inhibition of PAS and MMPs
simultaneously by a cocktail of inhibitors or a single inhibitor
that is active against both proteolytic enzymes in all other types
of cancer remains unexplored, but is a promising therapeutic
approach deserving future development.
No funding was received.
Not applicable.
MWS, ESJ and JJ conceived the review and analyzed
the relevant literature. MWS and JJ wrote the first draft of the
manuscript. MT and DM collected literature and wrote the
manuscript. ESJ collected literature and critically revised the
manuscript. JJ and MT created the figures. All authors read and
approved the final manuscript.
Not applicable.
Not applicable.
The authors declare that they have no competing
interests.
Not applicable.
|
1
|
Rosso T, Bertuccio P, La Vecchia C, Negri
E and Malvezzi M: Cancer mortality trend analysis in Italy,
1980–2010 and predictions for 2015. Tumori. 101:664–675. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Jankun J, Merrick HW and Goldblatt PJ:
Expression and localization of elements of the plasminogen
activation system in benign breast disease and breast cancers. J
Cell Biochem. 53:135–144. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Kiziridou AD, Toliou T, Stefanou D and
Agnantis N: u-PA expression in benign, borderline and malignant
ovarian tumors. Anticancer Res. 22:985–990. 2002.PubMed/NCBI
|
|
4
|
Safavi F and Rostami A: Role of serine
proteases in inflammation: Bowman-Birk protease inhibitor (BBI) as
a potential therapy for autoimmune diseases. Exp Mol Pathol.
93:428–433. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Van Hove I, Lemmens K, Van de Velde S,
Verslegers M and Moons L: Matrix metalloproteinase-3 in the central
nervous system: A look on the bright side. J Neurochem.
123:203–216. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
van der Vorst EP, Keijbeck AA, de Winther
MP and Donners MM: A disintegrin and metalloproteases: Molecular
scissors in angiogenesis, inflammation and atherosclerosis.
Atherosclerosis. 224:302–308. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Zheng D, Chen H, Bartee MY, Williams J,
Davids JA, Huang E, Moreb J and Lucas A: Virus-derived
anti-inflammatory proteins: Potential therapeutics for cancer.
Trends Mol Med. 18:304–310. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Avgeris M, Mavridis K and Scorilas A:
Kallikrein-related peptidases in prostate, breast, and ovarian
cancers: from pathobiology to clinical relevance. Biol Chem.
393:301–317. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Frank A, David V, Aurelie TR, Florent G,
William H and Philippe B: Regulation of MMPs during melanoma
progression: From genetic to epigenetic. Anticancer Agents Med
Chem. 12:773–782. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Pulz LH and Strefezzi RF: Proteases as
prognostic markers in human and canine cancers. Vet Comp Oncol.
15:669–683. 2017. View Article : Google Scholar
|
|
11
|
Liu WL, Liu D, Cheng K, Liu YJ, Xing S,
Chi PD, Liu XH, Xue N, Lai YZ, Guo L and Zhang G: Evaluating the
diagnostic and prognostic value of circulating cathepsin S in
gastric cancer. Oncotarget. 7:28124–28138. 2016.PubMed/NCBI
|
|
12
|
Pišlar A, Perišić Nanut M and Kos J:
Lysosomal cysteine peptidases-Molecules signaling tumor cell death
and survival. Semin Cancer Biol. 35:168–179. 2015. View Article : Google Scholar
|
|
13
|
Wallin H, Abrahamson M and Ekstrom U:
Cystatin C properties crucial for uptake and inhibition of
intracellular target enzymes. J Biol Chem. 288:17019–17029. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Eatemadi A, Aiyelabegan HT, Negahdari B,
Mazlomi MA, Daraee H, Daraee N, Eatemadi R and Sadroddiny E: Role
of protease and protease inhibitors in cancer pathogenesis and
treatment. Biomed Pharmacother. 86:221–231. 2017. View Article : Google Scholar
|
|
15
|
Hahlbrock A, Goesswein D, Kunzel J, Wünsch
D and Stauber RH: Threonine Aspartase1: An unexplored protease with
relevance for oral oncology? Oral Oncol. 54:e10–e12. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Werner AB, Tait SW, de Vries E, Eldering E
and Borst J: Requirement for aspartate-cleaved bid in apoptosis
signaling by DNA-damaging anti-cancer regimens. J Biol Chem.
279:28771–28780. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Verollet C, Charrière GM, Labrousse A,
Cougoule C, Le Cabec V and Maridonneau-Parini I: Extracellular
proteolysis in macrophage migration: Losing grip for a
breakthrough. Eur J Immunol. 41:2805–2813. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Roycik MD, Fang X and Sang QX: A fresh
prospect of extracellular matrix hydrolytic enzymes and their
substrates. Curr Pharm Des. 15:1295–1308. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Christiaens V and Lijnen HR: Role of the
fibrinolytic and matrix metalloproteinase systems in development of
adipose tissue. Arch Physiol Biochem. 112:254–259. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Riddick AC, Shukla CJ, Pennington CJ, Bass
R, Nuttall RK, Hogan A, Sethia KK, Ellis V, Collins AT, Maitland
NJ, et al: Identification of degradome components associated with
prostate cancer progression by expression analysis of human
prostatic tissues. Br J Cancer. 92:2171–2180. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Alfano D, Franco P, Vocca I, Gambi N, Pisa
V, Mancini A, Caputi M, Carriero MV, Iaccarino I and Stoppelli MP:
The urokinase plasminogen activator and its receptor: Role in cell
growth and apoptosis. Thromb Haemost. 93:205–211. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Carriero MV and Stoppelli MP: The
urokinase-type plasminogen activator and the generation of
inhibitors of urokinase activity and signaling. Curr Pharm Des.
17:1944–1961. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Liu Y, Wang Y, Teng Z, Chen J, Li Y, Chen
Z, Li Z and Zhang Z: Matrix metalloproteinase 9 expression and
survival of patients with osteosarcoma: A meta-analysis. Eur J
Cancer Care (Engl). 26:e123642017. View Article : Google Scholar
|
|
24
|
Hadler-Olsen E, Winberg JO and
Uhlin-Hansen L: Matrix metal-loproteinases in cancer: Their value
as diagnostic and prognostic markers and therapeutic targets.
Tumour Biol. 34:2041–2051. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Woodward JK, Holen I, Coleman RE and
Buttle DJ: The roles of proteolytic enzymes in the development of
tumour-induced bone disease in breast and prostate cancer. Bone.
41:912–927. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Swiercz R, Keck RW, Skrzypczak-Jankun E,
Selman SH and Jankun J: Recombinant PAI-1 inhibits angiogenesis and
reduces size of LNCaP prostate cancer xenografts in SCID mice.
Oncol Rep. 8:463–470. 2001.PubMed/NCBI
|
|
27
|
Rabbani SA and Xing RH: Role of urokinase
(uPA) and its receptor (uPAR) in invasion and metastasis of
hormone-dependent malignancies. Int J Oncol. 12:911–920.
1998.PubMed/NCBI
|
|
28
|
Gupta S, Gupta A, Saini AK, Majumder K,
Sinha K and Chahal A: Prostate cancer: How young is too young. Curr
Urol. 9:212–215. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Johnston TJ, Shaw GL, Lamb AD, Parashar D,
Greenberg D, Xiong T, Edwards AL, Gnanapragasam V, Holding P,
Herbert P, et al: Mortality among men with advanced prostate cancer
excluded from the protect trial. Eur Urol. 71:381–388. 2017.
View Article : Google Scholar :
|
|
30
|
Litwin MS and Tan HJ: The diagnosis and
treatment of prostate cancer: A review. JAMA. 317:2532–2542. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Macedo F, Ladeira K, Pinho F, Saraiva N,
Bonito N, Pinto L and Goncalves F: Bone metastases: An overview.
Oncol Rev. 11:3212017. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Hildenbrand R, Allgayer H, Marx A and
Stroebel P: Modulators of the urokinase-type plasminogen activation
system for cancer. Exp Opin Investig Drugs. 19:641–652. 2010.
View Article : Google Scholar
|
|
33
|
Honkavuori M, Talvensaari-Mattila A,
Puistola U, Turpeenniemi-Hujanen T and Santala M: High serum TIMP-1
is associated with adverse prognosis in endometrial carcinoma.
Anticancer Res. 28:2715–2719. 2008.PubMed/NCBI
|
|
34
|
Isogai C, Laug WE, Shimada H, Declerck PJ,
Stins MF, Durden DL, Erdreich-Epstein A and DeClerck YA:
Plasminogen activator inhibitor-1 promotes angiogenesis by
stimulating endothelial cell migration toward fibronectin. Cancer
Res. 61:5587–5594. 2001.PubMed/NCBI
|
|
35
|
Jankun J and Skrzypczak-Jankun E: Yin and
yang of the plasminogen activator inhibitor. Pol Arch Med Wewn.
119:410–417. 2009.PubMed/NCBI
|
|
36
|
Kodaman N, Aldrich MC, Sobota R,
Asselbergs FW, Brown NJ, Moore JH and Williams SM: Plasminogen
activator inhibitor-1 and diagnosis of the metabolic syndrome in a
west african population. J Am Heart Assoc. 5:e0038672016.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Forsgren M, Råden B, Israelsson M, Larsson
K and Hedén LO: Molecular cloning and characterization of a
full-length cDNA clone for human plasminogen. FEBS Lett.
213:254–260. 1987. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Miyata T, Iwanaga S, Sakata Y and Aoki N:
Plasminogen Tochigi: Inactive plasmin resulting from replacement of
alanine-600 by threonine in the active site. Proc Natl Acad Sci
USA. 79:6132–6136. 1982. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Borisov OV, Field M, Ling VT and Harris
RJ: Characterization of oligosaccharides in recombinant tissue
plasminogen activator produced in Chinese hamster ovary cells: Two
decades of analytical technology development. Anal Chem.
81:9744–9754. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Hatton MW, Day S, Ross B, Southward SM,
DeReske M and Richardson M: Plasminogen II accumulates five times
faster than plasminogen I at the site of a balloon
de-endothelializing injury in vivo to the rabbit aorta: Comparison
with other hemostatic proteins. J Lab Clin Med. 134:260–266. 1999.
View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Mølgaard L, Ponting CP and Christensen U:
Glycosylation at Asn-289 facilitates the ligand-induced
conformational changes of human Glu-plasminogen. FEBS Lett.
405:363–368. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Abdul S, Leebeek FW, Rijken DC and Uitte
de Willige S: Natural heterogeneity of α2-antiplasmin: Functional
and clinical consequences. Blood. 127:538–545. 2016. View Article : Google Scholar
|
|
43
|
Stefansson S, Lawrence DA and Argraves WS:
Plasminogen activator inhibitor-1 and vitronectin promote the
cellular clearance of thrombin by low density lipoprotein
receptor-related proteins 1–2. J Biol Chem. 271:8215–8220. 1996.
View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Ainsworth S, Carter S, Fisher C, Dawson J,
Makrides L, Nuttall T and Mason SL: Ligneous membranitis in
Scottish Terriers is associated with a single nucleotide
polymorphism in the plasminogen (PLG) gene. Anim Genet. 46:707–710.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Silva GB, Bariani C, Mendonça EF and
Batista AC: Clinical manifestations due to severe plasminogen
deficiency: A case report. J Dent Child(Chic). 73:179–182.
2006.
|
|
46
|
Celkan T: Plasminogen deficiency. J Thromb
Thrombolysis. 43:132–138. 2017. View Article : Google Scholar
|
|
47
|
Sivolella S, De Biagi M, Sartori MT,
Berengo M and Bressan E: Destructive membranous periodontal disease
(ligneous gingivitis): A literature review. J Periodontol.
83:465–476. 2012. View Article : Google Scholar
|
|
48
|
Lotan TL, Tefs K, Schuster V, Miller J,
Manaligod J, Filstead A, Yamada SD and Krausz T: Inherited
plasminogen deficiency presenting as ligneous vaginitis: A case
report with molecular correlation and review of the literature. Hum
Pathol. 38:1569–1575. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Gunhan O, Günhan M, Berker E, Gürgan CA
and Yildirim H: Destructive membranous periodontal disease
(Ligneous peri-odontitis). J Periodontol. 70:919–925. 1999.
View Article : Google Scholar
|
|
50
|
Cohen SR: Ligneous conjunctivitis: An
ophthalmic disease with potentially fatal tracheobronchial
obstruction. Laryngeal and tracheobronchial features. Ann Otol
Rhinol Laryngol. 99:509–512. 1990. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Foley JH, Kim PY, Mutch NJ and Gils A:
Insights into thrombin activatable fibrinolysis inhibitor function
and regulation. J Thromb Haemost. 11(Suppl 1): S306–S315. 2013.
View Article : Google Scholar
|
|
52
|
Kolev K, Longstaff C and Machovich R:
Fibrinolysis at the fluid-solid interface of thrombi. Curr Med Chem
Cardiovasc Hematol Agents. 3:341–355. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Bode W and Renatus M: Tissue-type
plasminogen activator: Variants and crystal/solution structures
demarcate structural determinants of function. Curr Opin Struct
Biol. 7:865–872. 1997. View Article : Google Scholar
|
|
54
|
Marcos-Contreras OA, Martinez de
Lizarrondo S, Bardou I, Orset C, Pruvost M, Anfray A, Frigout Y,
Hommet Y, Lebouvier L, Montaner J, et al: Hyperfibrinolysis
increases blood-brain barrier permeability by a plasmin- and
bradykinin-dependent mechanism. Blood. 128:2423–2434. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Chapman MP, Moore EE, Moore HB, Gonzalez
E, Gamboni F, Chandler JG, Mitra S, Ghasabyan A, Chin TL, Sauaia A,
et al: Overwhelming tPA release, not PAI-1 degradation, is
responsible for hyperfibrinolysis in severely injured trauma
patients. J Trauma Acute Care Surg. 80:16–23; discussion 23–15,
2016. PubMed/NCBI
|
|
56
|
Cardenas JC, Matijevic N, Baer LA, Holcomb
JB, Cotton BA and Wade CE: Elevated tissue plasminogen activator
and reduced plasminogen activator inhibitor promote
hyperfibrinolysis in trauma patients. Shock. 41:514–521. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Genét GF, Ostrowski SR, Sørensen AM and
Johansson PI: Detection of tPA-induced hyperfibrinolysis in whole
blood by RapidTEG, KaolinTEG, and functional fibrinogenTEG in
healthy individuals. Clin Appl Thromb Hemost. 18:638–644. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Prabhudesai A, Shetty S, Ghosh K and
Kulkarni B: Dysfunctional fibrinolysis and cerebral venous
thrombosis. Blood Cells Mol Dis. 65:51–55. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Talens S, Malfliet JJ, Rudež G, Spronk HM,
Janssen NA, Meijer P, Kluft C, de Maat MP and Rijken DC: Biological
variation in tPA-induced plasma clot lysis time. Thromb Haemost.
108:640–646. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Wang J, Li J and Liu Q: Association
between platelet activation and fibrinolysis in acute stroke
patients. Neurosci Lett. 384:305–309. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Simone TM and Higgins PJ: Low molecular
weight antagonists of plasminogen activator inhibitor-1:
Therapeutic potential in cardiovascular disease. Mol Med Ther.
1:1012012. View Article : Google Scholar
|
|
62
|
Higazi AA, Upson RH, Cohen RL, Manuppello
J, Bognacki J, Henkin J, McCrae KR, Kounnas MZ, Strickland DK,
Preissner KT, et al: Interaction of single-chain urokinase with its
receptor induces the appearance and disappearance of binding
epitopes within the resultant complex for other cell surface
proteins. Blood. 88:542–551. 1996.PubMed/NCBI
|
|
63
|
Finckh U, van Hadeln K, Müller-Thomsen T,
Alberici A, Binetti G, Hock C, Nitsch RM, Stoppe G, Reiss J and Gal
A: Association of late-onset Alzheimer disease with a genotype of
PLAU, the gene encoding urokinase-type plasminogen activator on
chromosome 10q22.2. Neurogenetics. 4:213–217. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Goto Y, Hagikura S, Katsuda N and Hamajima
N: A C to T polymorphism of urokinase plasminogen activator (P141L)
is associated with Helicobacter pylori infection. Asian Pac J
Cancer Prev. 12:803–806. 2011.PubMed/NCBI
|
|
65
|
Kriegbaum MC, Persson M, Haldager L,
Alpízar-Alpízar W, Jacobsen B, Gårdsvoll H, Kjær A and Ploug M:
Rational targeting of the urokinase receptor (uPAR): Development of
antagonists and non-invasive imaging probes. Curr Drug Targets.
12:1711–1728. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Romer J, Nielsen BS and Ploug M: The
urokinase receptor as a potential target in cancer therapy. Curr
Pharm Des. 10:2359–2376. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Ploug M, Gardsvoll H, Jorgensen TJ,
Lønborg Hansen L and Dano K: Structural analysis of the interaction
between urokinase-type plasminogen activator and its receptor: A
potential target for anti-invasive cancer therapy. Biochem Soc
Trans. 30:177–183. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
68
|
Manetti M, Allanore Y, Revillod L, Fatini
C, Guiducci S, Cuomo G, Bonino C, Riccieri V, Bazzichi L, Liakouli
V, et al: A genetic variation located in the promoter region of the
UPAR (CD87) gene is associated with the vascular complications of
systemic sclerosis. Arthritis Rheum. 63:247–256. 2011. View Article : Google Scholar
|
|
69
|
Shih CM, Kuo WH, Lin CW, Chen W, Cheng WE,
Chen SC and Lee YL: Association of polymorphisms in the genes of
the urokinase plasminogen activation system with susceptibility to
and severity of non-small cell lung cancer. Clin Chim Acta.
412:194–198. 2011. View Article : Google Scholar
|
|
70
|
Flevaris P and Vaughan D: The role of
plasminogen activator inhibitor type-1 in fibrosis. Semin Thromb
Hemost. 43:169–177. 2017. View Article : Google Scholar
|
|
71
|
Jankun J, Yang J, Zheng H, Han FQ,
Al-Senaidy A and Skrzypczak-Jankun E: Remarkable extension of PAI-1
half-life surprisingly brings no changes to its structure. Int J
Mol Med. 29:61–64. 2012.
|
|
72
|
Rabieian R, Boshtam M, Zareei M, Kouhpayeh
S, Masoudifar A and Mirzaei H: Plasminogen activator inhibitor
type-1 as a regulator of fibrosis. J Cell Biochem. 119:17–27. 2018.
View Article : Google Scholar
|
|
73
|
Piao L, Jung I, Huh JY, Miyata T and Ha H:
A novel plasminogen activator inhibitor-1 inhibitor, TM5441,
protects against high-fat diet-induced obesity and adipocyte injury
in mice. Br J Pharmacol. 173:2622–2632. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
74
|
Srikanthan K, Feyh A, Visweshwar H,
Shapiro JI and Sodhi K: Systematic review of metabolic syndrome
biomarkers: A panel for early detection, management, and risk
stratification in the west virginian population. Int J Med Sci.
13:25–38. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
75
|
Magdoud K, Herbepin VG, Touraine R, Almawi
WY and Mahjoub T: Plasminogen activator inhibitor 1 4G/5G and
-844G/A variants in idiopathic recurrent pregnancy loss. Am J
Reprod Immunol. 70:246–252. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
76
|
Honig A, Engel JB, Segerer SE, Kranke P,
Häusler S and Wurfel W: Pregnancy-triggered antiphospholipid
syndrome in a patient with multiple late miscarriages. Hum Reprod.
25:2753–2754. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
77
|
Jankun J and Skrzypczak-Jankun E: Bleeding
diathesis is associated with an A15T heterozygous mutation in exon
2 of the plasminogen activator inhibitor type 1. Exp Ther Med.
1:575–577. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
78
|
Mehta R and Shapiro AD: Plasminogen
activator inhibitor type 1 deficiency. Haemophilia. 14:1255–1260.
2008. View Article : Google Scholar
|
|
79
|
Schleef RR, Higgins DL, Pillemer E and
Levitt LJ: Bleeding diathesis due to decreased functional activity
of type 1 plasminogen activator inhibitor. J Clin Invest.
83:1747–1752. 1989. View Article : Google Scholar : PubMed/NCBI
|
|
80
|
Heiman M, Gupta S and Shapiro AD: The
obstetric, gynaecological and fertility implications of homozygous
PAI-1 deficiency: Single-centre experience. Haemophilia.
20:407–412. 2014. View Article : Google Scholar
|
|
81
|
Lin S, Huiya Z, Bo L, Wei W and Yongmei G:
The plasminogen activator inhibitor-1 (PAI-1) gene -844 A/G and
-675 4G/5G promoter polymorphism significantly influences plasma
PAI-1 levels in women with polycystic ovary syndrome. Endocrine.
36:503–509. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
82
|
Fay WP, Parker AC, Condrey LR and Shapiro
AD: Human plasminogen activator inhibitor-1 (PAI-1) deficiency:
Characterization of a large kindred with a null mutation in the
PAI-1 gene. Blood. 90:204–208. 1997.PubMed/NCBI
|
|
83
|
Jankun J and Skrzypczak-Jankun E: Val17Ile
single nucleotide polymorphisms similarly as Ala15Thr could be
related to the lower secretory dynamics of PAI-1 secretion:
Theoretical evidence. Curr Mol Med. 11:512–516. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
84
|
Miao C, Liang C, Zhu J, Xu A, Zhao K, Hua
Y, Zhang J, Chen W, Suo C, Zhang C, et al: Prognostic role of
matrix metal-loproteinases in bladder carcinoma: A systematic
review and meta-analysis. Oncotarget. 8:32309–32321.
2017.PubMed/NCBI
|
|
85
|
Turunen SP, Tatti-Bugaeva O and Lehti K:
Membrane-type matrix metalloproteases as diverse effectors of
cancer progression. Biochim Biophys Acta. 1864.1974–1988. 2017.
|
|
86
|
Van Lint P and Libert C: Chemokine and
cytokine processing by matrix metalloproteinases and its effect on
leukocyte migration and inflammation. J Leuk Biol. 82:1375–1381.
2007. View Article : Google Scholar
|
|
87
|
Verma RP and Hansch C: Matrix
metalloproteinases (MMPs): Chemical-biological functions and
(Q)SARs. Bioorg Med Chem. 15:2223–2268. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
88
|
Swarnakar S, Paul S, Singh LP and Reiter
RJ: Matrix metal-loproteinases in health and disease: Regulation by
melatonin. J Pineal Res. 50:8–20. 2011. View Article : Google Scholar
|
|
89
|
Zitka O, Kukacka J, Krizkova S, Huska D,
Adam V, Masarik M, Prusa R and Kizek R: Matrix metalloproteinases.
Curr Med Chem. 17:3751–3768. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
90
|
Fisher KE, Fei Q, Laird ER, Stock JL,
Allen MR, Sahagan BG and Strick CA: Engineering autoactivating
forms of matrix metalloproteinase-9 and expression of the active
enzyme in cultured cells and transgenic mouse brain. Biochemistry.
41:8289–8297. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
91
|
Marchenko GN, Ratnikov BI, Rozanov DV,
Godzik A, Deryugina EI and Strongin AY: Characterization of matrix
metalloproteinase-26, a novel metalloproteinase widely expressed in
cancer cells of epithelial origin. Biochem J. 356:705–718. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
92
|
Merchant N, Nagaraju GP, Rajitha B,
Lammata S, Jella KK, Buchwald ZS, Lakka SS and Ali AN: Matrix
metalloproteinases: Their functional role in lung cancer.
Carcinogenesis. 38:766–780. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
93
|
Morgunova E, Tuuttila A, Bergmann U,
Isupov M, Lindqvist Y, Schneider G and Tryggvason K: Structure of
human pro-matrix metalloproteinase-2: Activation mechanism
revealed. Science. 284:1667–1670. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
94
|
Su SC, Hsieh MJ, Yang WE, Chung WH, Reiter
RJ and Yang SF: Cancer metastasis: Mechanisms of inhibition by
melatonin. J Pineal Research. 62:2017. View Article : Google Scholar
|
|
95
|
Webb AH, Gao BT, Goldsmith ZK, Irvine AS,
Saleh N, Lee RP, Lendermon JB, Bheemreddy R, Zhang Q, Brennan RC,
et al: Inhibition of MMP-2 and MMP-9 decreases cellular migration,
and angiogenesis in in vitro models of retinoblastoma. BMC Cancer.
17:4342017. View Article : Google Scholar : PubMed/NCBI
|
|
96
|
Eiro N, Fernandez-Gomez J, Sacristan R,
Sacristán R, Fernandez-Garcia B, Lobo B, Gonzalez-Suarez J, Quintas
A, Escaf S and Vizoso FJ: Stromal factors involved in human
prostate cancer development, progression and castration resistance.
J Cancer Res Clin Oncol. 143:351–359. 2017. View Article : Google Scholar
|
|
97
|
Gialeli C, Theocharis AD and Karamanos NK:
Roles of matrix metalloproteinases in cancer progression and their
pharmacological targeting. FEBS J. 278:16–27. 2011. View Article : Google Scholar
|
|
98
|
Grieu F, Li WQ and Iacopetta B: Genetic
polymorphisms in the MMP-2 and MMP-9 genes and breast cancer
phenotype. Breast Cancer Res Treat. 88:197–204. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
99
|
Singh R, Srivastava P, Srivastava A and
Mittal RD: Matrix metalloproteinase (MMP-9 and MMP-2) gene
polymorphisms influence allograft survival in renal transplant
recipients. Nephrol Dial Transplant. 25:3393–3401. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
100
|
Ghaffarpour S, Ghazanfari T, Kabudanian
Ardestani S, Pourfarzam S, Fallahi F, Shams J, Mirsharif ES,
Mohseni Majd AM and Faghihzadeh S: Correlation between MMP-9 and
MMP-9/ TIMPs complex with pulmonary function in sulfur mustard
exposed civilians: Sardasht-Iran cohort study. Arch Iran Med.
20:74–82. 2017.PubMed/NCBI
|
|
101
|
Ricci S, Bruzzese D and DI Carlo A:
Evaluation of MMP-2, MMP-9, TIMP-1, TIMP-2, NGAL and MMP-9/NGAL
complex in urine and sera from patients with bladder cancer. Oncol
Lett. 10:2527–2532. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
102
|
Hinterseher I, Krex D, Kuhlisch E, Schmidt
KG, Pilarsky C, Schneiders W, Saeger HD and Bergert H: Tissue
inhibitor of metalloproteinase-1 (TIMP-1) polymorphisms in a
Caucasian population with abdominal aortic aneurysm. World J Surg.
31:2248–2254. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
103
|
Guo Xu P, Jin W, Wang T, Fan J, Hao D,
Jing Z, Han S, Du C, Jiang JD, et al: TIMP-2 SNPs rs7342880 and
rs4789936 are linked to risk of knee osteoarthritis in the Chinese
Han Population. Oncotarget. 8:1166–1176. 2017.
|
|
104
|
Birbrair A, Zhang T, Wang ZM, Messi ML,
Olson JD, Mintz A and Delbono O: Type-2 pericytes participate in
normal and tumoral angiogenesis. Am J Physiol Cell Physiol.
307:C25–C38. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
105
|
Lah TT, Durán Alonso MB and Van Noorden
CJ: Antiprotease therapy in cancer: Hot or not. Exp Opin Biol Ther.
6:257–279. 2006. View Article : Google Scholar
|
|
106
|
Wong MS, Sidik SM, Mahmud R and Stanslas
J: Molecular targets in the discovery and development of novel
antimetastatic agents: Current progress and future prospects. Clin
Exp Pharmacol Physiol. 40:307–319. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
107
|
Jankun J, Maher VM and McCormick JJ:
Malignant transformation of human fibroblasts correlates with
increased activity of receptor-bound plasminogen activator. Cancer
Res. 51:1221–1226. 1991.PubMed/NCBI
|
|
108
|
Jankun J, Selman SH, Aniola J and
Skrzypczak-Jankun E: Nutraceutical inhibitors of urokinase:
Potential applications in prostate cancer prevention and treatment.
Oncol Rep. 16:341–346. 2006.PubMed/NCBI
|
|
109
|
Kamat AM and Lamm DL: Chemoprevention of
bladder cancer. Urol Clin North Am. 29:157–168. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
110
|
Swiercz R, Skrzypczak-Jankun E, Merrell
MM, Selman SH and Jankun J: Angiostatic activity of synthetic
inhibitors of urokinase type plasminogen activator. Oncol Rep.
6:523–526. 1999.PubMed/NCBI
|
|
111
|
Jankun J, Selman SH, Swiercz R and
Skrzypczak-Jankun E: Why drinking green tea could prevent cancer.
Nature. 387:5611997. View
Article : Google Scholar : PubMed/NCBI
|
|
112
|
Kemberling JK, Hampton JA, Keck RW, Gomez
MA and Selman SH: Inhibition of bladder tumor growth by the green
tea derivative epigallocatechin-3-gallate. J Urol. 170:773–776.
2003. View Article : Google Scholar : PubMed/NCBI
|
|
113
|
Jankun J, Keck RW and Selman SH:
Epigallocatechin-3-gallate prevents tumor cell implantation/growth
in an experimental rat bladder tumor model. Int J Oncol.
44:147–152. 2014. View Article : Google Scholar
|
|
114
|
Wendt MD, Geyer A, McClellan WJ, Rockway
TW, Weitzberg M, Zhao X, Mantei R, Stewart K, Nienaber V,
Klinghofer V and Giranda VL: Interaction with the S1 beta-pocket of
urokinase: 8-heterocycle substituted and 6,8-disubstituted
2-naphthamidine urokinase inhibitors. Bioorg Med Chem Lett.
14:3063–3068. 2004.PubMed/NCBI
|
|
115
|
Bruncko M, McClellan WJ, Wendt MD, Sauer
DR, Geyer A, Dalton CR, Kaminski MA, Weitzberg M, Gong J, Dellaria
JF, et al: Naphthamidine urokinase plasminogen activator inhibitors
with improved pharmacokinetic properties. Bioorg Med Chem Lett.
15:93–98. 2005. View Article : Google Scholar
|
|
116
|
Katz BA, Sprengeler PA, Luong C, Verner E,
Elrod K, Kirtley M, Janc J, Spencer JR, Breitenbucher JG, Hui H, et
al: Engineering inhibitors highly selective for the S1 sites of
Ser190 trypsin-like serine protease drug targets. Chem Biol.
8:1107–1121. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
117
|
Schmitt M, Harbeck N, Brünner N, Jänicke
F, Meisner C, Mühlenweg B, Jansen H, Dorn J, Nitz U, Kantelhardt EJ
and Thomssen C: Cancer therapy trials employing level-of-evidence-1
disease forecast cancer biomarkers uPA and its inhibitor PAI-1. Exp
Rev Mol Diagn. 11:617–634. 2011. View Article : Google Scholar
|
|
118
|
Zengel P, Ramp D, Mack B, Zahler S,
Berghaus A, Muehlenweg B, Gires O and Schmitz S: Multimodal therapy
for synergic inhibition of tumour cell invasion and tumour-induced
angiogenesis. BMC Cancer. 10:922010. View Article : Google Scholar : PubMed/NCBI
|
|
119
|
Setyono-Han B, Stürzebecher J, Schmalix
WA, Muehlenweg B, Sieuwerts AM, Timmermans M, Magdolen V, Schmitt
M, Klijn JG and Foekens JA: Suppression of rat breast cancer
metastasis and reduction of primary tumour growth by the small
synthetic urokinase inhibitor WX-UK1. Thromb Haemost. 93:779–786.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
120
|
Ertongur S, Lang S, Mack B, Wosikowski K,
Muehlenweg B and Gires O: Inhibition of the invasion capacity of
carcinoma cells by WX-UK1, a novel synthetic inhibitor of the
urokinase-type plasminogen activator system. Int J Cancer.
110:815–824. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
121
|
Ossowski L: Invasion of connective tissue
by human carcinoma cell lines: Requirement for urokinase, urokinase
receptor, and interstitial collagenase. Cancer Res. 52:6754–6760.
1992.PubMed/NCBI
|
|
122
|
Ossowski L: In vivo invasion of modified
chorioallantoic membrane by tumor cells: The role of cell
surface-bound urokinase. J Cell Biol. 107:2437–2445. 1988.
View Article : Google Scholar : PubMed/NCBI
|
|
123
|
Jankun J, Keck RW, Skrzypczak-Jankun E and
Swiercz R: Inhibitors of urokinase reduce size of prostate cancer
xenografts in severe combined immunodeficient mice. Cancer Res.
57:559–563. 1997.PubMed/NCBI
|
|
124
|
Berkenpas MB, Lawrence DA and Ginsburg D:
Molecular evolution of plasminogen activator inhibitor-1 functional
stability. EMBO J. 14:2969–2977. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
125
|
Chorostowska-Wynimko J, Swiercz R,
Skrzypczak-Jankun E, Wojtowicz A, Selman SH and Jankun J: A novel
form of the plasminogen activator inhibitor created by cysteine
mutations extends its half-life: Relevance to cancer and
angiogenesis. Mol Cancer Ther. 2:19–28. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
126
|
Tucker HM, Mottonen J, Goldsmith EJ and
Gerard RD: Engineering of plasminogen activator inhibitor-1 to
reduce the rate of latency transition. Nat Struct Biol. 2:442–445.
1995. View Article : Google Scholar : PubMed/NCBI
|
|
127
|
Binder BR and Mihaly J: The plasminogen
activator inhibitor ‘paradox’ in cancer. Immunol Lett. 118:116–124.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
128
|
Beaulieu LM, Whitley BR, Wiesner TF,
Rehault SM, Palmieri D, Elkahloun AG and Church FC: Breast cancer
and metabolic syndrome linked through the plasminogen activator
inhibitor-1 cycle. Bioessays. 29:1029–1038. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
129
|
Duffy MJ: Urokinase plasminogen activator
and its inhibitor, PAI-1, as prognostic markers in breast cancer:
From pilot to level 1 evidence studies. Clin Chem. 48:1194–1197.
2002.PubMed/NCBI
|
|
130
|
Chorostowska-Wynimko J, Swiercz R,
Skrzypczak-Jankun E, Selman SH and Jankun J: Plasminogen activator
inhibitor type-1 mutants regulate angiogenesis of human umbilical
and lung vascular endothelial cells. Oncol Rep. 12:1155–1162.
2004.PubMed/NCBI
|
|
131
|
Masset A, Maillard C, Sounni NE, Jacobs N,
Bruyére F, Delvenne P, Tacke M, Reinheckel T, Foidart JM, Coussens
LM and Noël A: Unimpeded skin carcinogenesis in K14–HPV16
transgenic mice deficient for plasminogen activator inhibitor. Int
J Cancer. 128:283–293. 2011. View Article : Google Scholar
|
|
132
|
Mazar AP, Henkin J and Goldfarb RH: The
urokinase plasminogen activator system in cancer: Implications for
tumor angiogenesis and metastasis. Angiogenesis. 3:15–32. 1999.
View Article : Google Scholar
|
|
133
|
Wyganowska-Świątkowska M and Jankun J:
Plasminogen activation system in oral cancer: Relevance in
prognosis and therapy (Review). Int J Oncol. 47:16–24. 2015.
View Article : Google Scholar
|
|
134
|
Chen SC, Henry DO, Hicks DG, Reczek PR and
Wong MK: Intravesical administration of plasminogen activator
inhibitor type-1 inhibits in vivo bladder tumor invasion and
progression. J Urol. 181:336–342. 2009. View Article : Google Scholar
|
|
135
|
Yamakawa S, Asai T, Uchida T, Matsukawa M,
Akizawa T and Oku N: (-)-Epigallocatechin gallate inhibits
membrane-type 1 matrix metalloproteinase, MT1-MMP, and tumor
angiogenesis. Cancer Lett. 210:47–55. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
136
|
Stefansson S, Petitclerc E, Wong MK,
McMahon GA, Brooks PC and Lawrence DA: Inhibition of angiogenesis
in vivo by plasminogen activator inhibitor-1. J Biol Chem.
276:8135–8141. 2001. View Article : Google Scholar
|
|
137
|
Su SC, Lin CW, Yang WE, Fan WL and Yang
SF: The urokinase-type plasminogen activator (uPA) system as a
biomarker and therapeutic target in human malignancies. Exp Opin
Ther Targets. 20:551–566. 2016. View Article : Google Scholar
|
|
138
|
Ulisse S, Baldini E, Sorrenti S and
D’Armiento M: The urokinase plasminogen activator system: A target
for anti-cancer therapy. Curr Cancer Drug Targets. 9:32–71. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
139
|
Iwamoto J, Mizokami Y, Takahashi K,
Nakajima K, Ohtsubo T, Miura S, Narasaka T, Takeyama H, Omata T,
Shimokobe K, et al: Expressions of urokinase-type plasminogen
activator, its receptor and plasminogen activator inhibitor-1 in
gastric cancer cells and effects of Helicobacter pylori. Scand J
Gastroenterol. 40:783–793. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
140
|
Haj-Yehia A, Nassar T, Sachais BS, Kuo A,
Bdeir K, Al-Mehdi AB, Mazar A, Cines DB and Higazi AA:
Urokinase-derived peptides regulate vascular smooth muscle
contraction in vitro and in vivo. FASEB J. 14:1411–1422. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
141
|
Guo Y, Higazi AA, Arakelian A, Sachais BS,
Cines D, Goldfarb RH, Jones TR, Kwaan H, Mazar AP and Rabbani SA: A
peptide derived from the nonreceptor binding region of urokinase
plasminogen activator (uPA) inhibits tumor progression and
angiogenesis and induces tumor cell death in vivo. FASEB J.
14:1400–1410. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
142
|
Berkenblit A, Matulonis UA, Kroener JF,
Dezube BJ, Lam GN, Cuasay LC, Brünner N, Jones TR, Silverman MH and
Gold MA: A6, a urokinase plasminogen activator (uPA)-derived
peptide in patients with advanced gynecologic cancer: A phase I
trial. Gynecol Oncol. 99:50–57. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
143
|
Mani T, Wang F, Knabe WE, Sinn AL, Khanna
M, Jo I, Sandusky GE, Sledge GW Jr, Jones DR, Khanna R, et al:
Small-molecule inhibition of the uPAR.uPA interaction: Synthesis,
biochemical, cellular, in vivo pharmacokinetics and efficacy
studies in breast cancer metastasis. Bioorg Med Chem. 21:2145–2155.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
144
|
Wang F, Eric Knabe W, Li L, Jo I, Mani T,
Roehm H, Oh K, Li J, Khanna M and Meroueh SO: Design, synthesis,
biochemical studies, cellular characterization, and structure-based
computational studies of small molecules targeting the urokinase
receptor. Bioorg Med Chem. 20:4760–4773. 2012. View Article : Google Scholar :
|
|
145
|
Jian Q, Yang Z, Shu J, Liu X, Zhang J and
Li Z: Lectin BS-I inhibits cell migration and invasion via
AKT/GSK-3β/β-catenin pathway in hepatocellular carcinoma. J Cell
Mol Med. 22:315–329. 2018. View Article : Google Scholar
|
|
146
|
Li H and Chen C: Quercetin has
antimetastatic effects on gastric cancer cells via the interruption
of uPA/uPAR function by modulating NF-κb, PKC-δ, ERK1/2, and AMPKα.
Integr Cancer Ther. 17:511–523. 2018. View Article : Google Scholar
|
|
147
|
Cathcart J, Pulkoski-Gross A and Cao J:
Targeting matrix metalloproteinases in cancer: Bringing new life to
old ideas. Genes Dis. 2:26–34. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
148
|
Rothenberg ML, Nelson AR and Hande KR: New
drugs on the horizon: Matrix metalloproteinase inhibitors.
Oncologist. 3:271–274. 1998.
|
|
149
|
Rothenberg ML, Nelson AR and Hande KR: New
drugs on the horizon: Matrix metalloproteinase inhibitors. Stem
cells. 17:237–240. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
150
|
Coussens LM, Fingleton B and Matrisian LM:
Matrix metal-loproteinase inhibitors and cancer: Trials and
tribulations. Science. 295:2387–2392. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
151
|
Martens E, Leyssen A, Van Aelst I, Fiten
P, Piccard H, Hu J, Descamps FJ, Van den Steen PE, Proost P, Van
Damme J, et al: A monoclonal antibody inhibits gelatinase B/MMP-9
by selective binding to part of the catalytic domain and not to the
fibronectin or zinc binding domains. Biochim Biophys Acta.
1770:178–186. 2007. View Article : Google Scholar
|
|
152
|
Kaimal R, Aljumaily R, Tressel SL, Pradhan
RV, Covic L, Kuliopulos A, Zarwan C, Kim YB, Sharifi S and Agarwal
A: Selective blockade of matrix metalloprotease-14 with a
monoclonal antibody abrogates invasion, angiogenesis, and tumor
growth in ovarian cancer. Cancer Res. 73:2457–2467. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
153
|
Shay G, Lynch CC and Fingleton B: Moving
targets: Emerging roles for MMPs in cancer progression and
metastasis. Matrix Biol. 44–46:200–206. 2015. View Article : Google Scholar
|