Introduction
Sepsis is one of the leading causes of mortality and
morbidity in children and adults (1). An altered gut microflora is always
present in patients with sepsis, which induces infective and
non-infective complications (2).
In addition, intestinal epithelium dysfunction and dysbiosis,
induced by critical illnesses, result in increased translocation of
bacteria to the blood, which contributes to the adverse outcome of
sepsis (3).
Probiotics are regarded as the living
microorganisms, which, in adequate amounts, can bring health
benefits to the host (4). Among
them, the genera Lactobacillus and Bifidobacterium
are the most widely used (5). Up
to date, probiotics have been increasingly applied and studied in
clinical practice. It is believed that probiotics can reduce the
risk of disease (including diarrhea, allergic diseases and
inflammatory bowel disease) through competition for binding locus
and nutrients with pathogens, producing bacteriocins to kill
pathogens, synthesizing IgA to modify immune responses and reducing
inflammation by stimulating regulatory lymphocytes through
interleukin (IL)-10 and transforming growth factor signaling
(6–8). However, application of probiotics on
sepsis has been limited due to the theoretical risk of aggravating
bacteremia in patients with critical illnesses, although few data
exist that support this concern (9). A previous study from our group has
previously reported that prophylactic administration of a special
probiotic bacterial species in a septic mouse model effectively
reduced mortality (10). However,
the underlying mechanisms by which probiotics alleviated the
severity of sepsis remains unclear. In order to get a better
understanding of the role that the gut microbiota serve in the
process of sepsis, the present study performed a comprehensive
analysis of the microbiota alterations during sepsis.
In the past, the colonization patterns in septic
patients were investigated predominantly from culture-dependent
studies. However, most of the intestinal bacteria are obligate or
facultative anaerobic bacteria, which are technically challenging
to culture once exposed in oxygen (11). Therefore, previous studies merely
confirm the absence or presence of specific bacteria, without
providing a global view of microbiota alterations during sepsis.
Due to the development of bacterial 16S ribosomal DNA gene
sequencing (12), the present
study was able to decipher microbial diversity and reveal the
alterations of the gut microbiota structure during sepsis. The
present study aimed to investigate the effects of probiotic LGG on
the microbial diversity in septic mice, and to examine how this
contributes to the prevention and reversion of sepsis.
Materials and methods
Ethics statement
All procedures for animal care and use were approved
by the Animal Care Ethics Committee of the First Affiliated
Hospital, Zhejiang University (Hangzhou, China). Four-week-old male
C57BL6 mice were purchased from Zhejiang University and housed in
pathogen-free animal facilities under a standard 12-h light/dark
cycle. Standard mouse diet and water were given throughout the
study.
Probiotic administration and cecal
ligation and puncture (CLP) model
Four-week-old male C57BL6 mice (weight, 13.59±1.59
g) were administered daily by oral gavage with 200 µl of LGG
(2×109 CFU/ml, 2.9×107 CFU/g; Culturelle,
ConAgra Foods, Omaha, NE, USA; CLP+LGG group, n=23), or normal
saline (control sham group, n=20; CLP+saline group, n=18) 4 weeks
prior to CLP. To establish the murine septic peritonitis model, the
mice were anesthetized with isoflurane and bupivacaine: 3–4% for
induction and 1–3% for maintenance. A 1 cm incision was made on the
middle of abdomen, and the cecum was exteriorized through the
incision carefully. In order to induce mid-grade sepsis, the cecum
was ligated at middle of the bottom and distal pole of the cecum,
and was punctured from mesenteric toward anti-mesenteric direction
using a 23-gauge needle. A droplet of feces was extruded through
the holes, and the cecum was relocated into the abdominal cavity.
Finally, the fascia and skin incision were closed in layers. Sham
mice underwent the same operation except from the cecum ligation
and puncture procedures. In all mice, 1 ml pre-warmed normal saline
was injected subcutaneously following the CLP operation (13).
Serum sample preparation
Blood samples were obtained from mice at 24 h
post-CLP operation. In all instances, 1.5 ml of blood was
introduced in the tube not containing any anticoagulant substance.
The collected blood was left on the laboratory bench for 30 min at
room temperature prior to centrifugation (3,000 × g, 5 min, 4°C),
then the serum was carefully absorbed and transferred to a clean
tube, and then stored at −80°C as soon as possible.
ELISA
In brief, 10 µl serum sample was diluted in 40 µl
sample dilution buffer per ELISA (Nuoyang Biotech, Hangzhou, China)
plate well, and 100 µl horseradish peroxidise
(HRP)-conjugated detection antibody [IL-6, tumor necrosis factor
(TNF)-α, IL-22 and IL-2] was added. The ELISA kits used were as
follows: IL-6 (cat. no. CME0006-048), IL-22 (cat. no. CME0033-048)
IL-2 (cat. no. CME0001-048) (Nuoyang Biotech) and TNF-α (cat no.
ml002095; Shanghai Enzyme-linked Biotechnology Co., Ltd., Shanghai,
China; http://www.mlbio.cn/).
The plate was incubated for 1 h at 37°C and washed
five times. HRP-substrate goat anti-mouse IgG (cat. no. 31430;
1:100; Invitrogen; Thermo Fisher Scientific, Inc., Waltham, MA,
USA) was then added and incubated for 15 min at 37°C in the dark.
The enzyme reaction was then stopped with 50 µl stop
solution and absorbance was measured at 450 nm. Concentrations of
inflammatory factors were calculated using a standard curve
generated by standard solution in different concentration. Serum
levels of inflammatory factors are presented as pg/ml.
Histological analysis
Colon tissues were collected from mice at 24 h
post-CLP operation and fixed in 10% formalin for 24 h. The fixed
colon tissues were embedded in paraffin and sectioned at 4
µm thickness. The sections were stained with
hematoxylineosin (H&E) for histological observation to examine
the pathology of colon injury induced by CLP. Immunohistochemistry
staining for Ki67 and occludin were performed for measuring
proliferation and tight junction formation between intestinal
epithelial cells. For immunohistochemistry staining, the standard
protocol was performed. Briefly, sections were incubated with 3%
H2O2 5 min at room temperature to eliminate
the endogenous peroxidase activity, and then blocked with 5% bovine
serum albumin for 1 h at 37°C and then incubated with rabbit
anti-Ki67 polyclonal antibody (cat. no. PA5-19462; 1:100;
Invitrogen; Thermo Fisher Scientific, Inc.) or rabbit anti-occludin
polyclonal antibody (cat. no. 404700; 1:100; Invitrogen; Thermo
Fisher Scientific, Inc.) at 4°C overnight. This was followed by
washing three times with PBS for 5 min each. The sections were
incubated with horseradish peroxidase-conjugated mouse-anti-rabbit
IgG secondary antibody (cat. no. 31464; 1:100; Invitrogen; Thermo
Fisher Scientific, Inc.) for 1 h at 37°C. Colour was developed with
a 3,3′-diaminobenzidine kit (Nanjing KeyGen Biotech Co., Ltd.,
Nanjing, China) and microscopically examined with light microscope
(Motic BA210; Motic Instruments, Richmond, BC, Canada) using the
following calculation: Ki67-positive cells/100 crypts or
Occludin-positive cells, in five random ×100 magnified fields, five
mice per group, one section per mouse. Terminal
deoxynucleotidyl-transferase-mediated dUTP-biotin nick end-labeling
(TUNEL) assay (Invitrogen; Thermo Fisher Scientific, Inc.) was used
for evaluation of apoptosis in the CLP-treated colon, according to
the manufacturer's protocol (TUNEL-positive cells/100 crypts, in
five random ×100 magnified fields, five mice per group, one section
per mouse).
DNA extraction and polymerase chain
reaction (PCR) amplification
To ensure the abundance of LGG (normal/saline, n=5;
normal/LGG, n=5), DNA was extracted from fecal samples obtained
from a separate set of mice prior to CLP operation. Quantification
of LGG was performed by PCR using the following primers: LactoF,
5′-AGC AGT AGG GAA TCT TCC A; and LactoR, 5′-ATTYCACCGCTACACATG.
The PCR reaction was performed as follows: 95°C for 5 min, followed
by 35 cycles at 94°C for 15 sec, 53°C for 30 sec, and 72°C for 45
sec and a final extension at 72°C for 10 min. To analyze the
microbial diversity in mice with sepsis, fecal samples were
collected from mice at 24 h post-CLP operation and frozen at -80°C
immediately. Microbial DNA was extracted from fecal samples using a
Qiagen mini kit (Qiagen GmbH, Hilden, Germany) according to the
manufacturer's instructions. The V3-V4 region of the bacteria 16S
ribosomal RNA gene was PCR-amplified using primers 341F (5′-CCT AYG
GGR BGC ASC AG-3′) and 806R (5′-GGA CTA CNN GGG TAT CTA AT-3′). An
eight bp unique barcode sequence was attached to each sample. A
total of 50 µl PCR reactions contained 0.5 µl of
dNTPs (10 mmol/l), 0.5 µl each primer (5 µmol/l), 0.5
µl Fast-Pfu polymerase (Beijing Transgen Biotech Co., Ltd.,
Beijing, China), 5 µl 10× Fast-Pfu buffer, and 10 ng
template DNA. The PCR reaction was performed as follows: 98°C for
30 sec, followed by 15 cycles at 98°C for 10 sec, 65°C for 30 sec,
and 72°C for 30 sec and a final extension at 72°C for 5 min. PCR
products were pooled, purified using the AxyPrep DNA gel extraction
kit (Axygen Biosciences, Union City, CA, USA) according to the
manufacturer's protocol, and quantified using a fluorometric kit
(Quant-iT PicoGreen; Invitrogen; Thermo Fisher Scientific, Inc.).
Purified products were pooled in equimolar for library
construction. An Illumina miseq platform (Illumina Inc., San Diego,
CA, USA) was applied to construct the paired-end sequencing (2×250
bp) and sequencing libraries according to the standard
protocols.
Sequence analysis
Based on barcode and PCR primer, all raw reads were
screened using the Quantitative Insights Into Microbial Ecology
software (QIIME, version 1.17) (14). The 250 bp sequences were trimmed
at the site where a quality score <20 over a 10 bp sliding
window was received, and sequences were omitted according to the
following criteria: 1) Containing ambiguous sequences, 2) The
maximum nucleotide mismatches in primer matching was two, 3)
Sequences shorter than 150 bp. UCHIME was used to identify and
remove chimeras. USEARCH was applied to generate operational
taxonomic units (OTUs), reads with the maximum length in each OTU
were selected as representative sequences. Based on the bacterial
SILVA dataset (15),
representative sequences were assigned to different taxonomic
levels. The richness and diversity of gut bacteria were evaluated
using indexes chao1; simpson and shannon respective. Principal
coordinates analysis (PCoA) was conducted on the base of weighted
and unweighted uniFrac distance metrics analysis to assess the
interactions among bacterial communities of different samples.
Ribosomal database project classifier (http://rdp.cme.msu.edu/) was applied to annotate the
sequences.
Statistical analysis
The homogeneity of variances was verified using
Bartlett's test. One-way analysis of variance (with Bonferroni as a
post hoc test) was performed to compare the bacteremia,
inflammatory factors and histological study variables among
different groups. Survival studies were conducted via the log-rank
test. Two-tailed non-parametric Kruskal-Wallis test was used to
compare the differences in diversity indexes and microbial taxa.
The statistical analysis was conducted using GraphPad Prism 5
(GraphPad Software, Inc., La Jolla, CA, USA). P<0.05 was
considered to indicate a statistically significant difference.
Results
LGG pretreatment reduces mortality and
systemic inflammatory response in septic peritonitis
DNA isolated from the colon was analyzed by PCR to
quantify the abundance of LGG. There was a significant increase in
LGG in normal/LGG mice (9.24±0.06, log qPCR copy/fecal (g); n=5)
compared with normal/saline mice (8.16±0.11, log qPCR copy/fecal
(g); n=5; Table I). These data
indicated that LGG was able to survive and propagate in the
gastrointestinal tract following oral administration for 4
weeks.
 | Table ILactobacillus in the colon of
mice. |
Table I
Lactobacillus in the colon of
mice.
| Normal/LGG
(n=5) | Normal/saline
(n=5) | P-value |
|---|
|
Lactobacillus | 9.24±0.06 | 8.16±0.11 | <0.001 |
A previous study demonstrated that septic mice
pretreated with LGG had a markedly improved 7 day survival rate
compared with the CLP+saline mice (P=0.029). Five sham mice
survived (10).
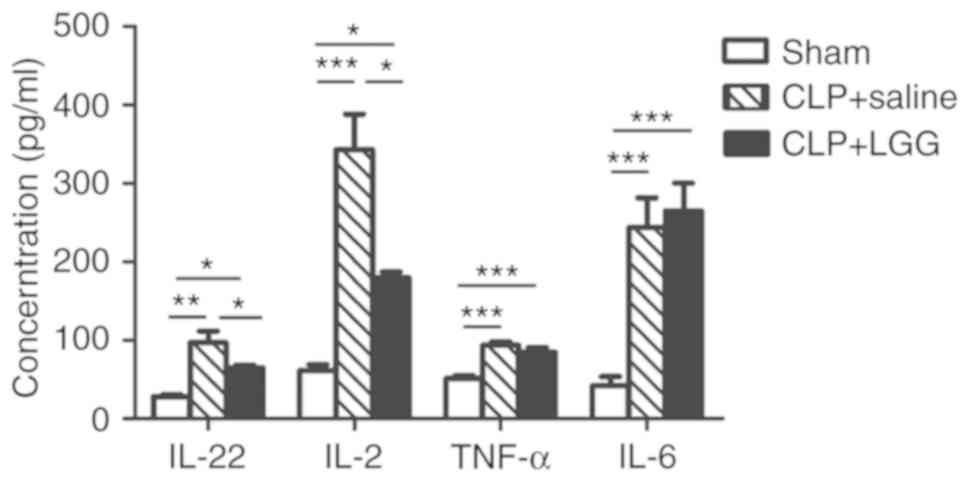
To evaluate the effect of LGG on reducing the
inflammatory response caused by sepsis, the levels of
proinflammatory cytokines IL-6, TNF-α, IL-2, and of the
anti-inflammatory cytokine IL-22, were detected by ELISA assay in
the serum of the mice. Serum levels of IL-22, IL-2, TNF-α and IL-6
were the lowest in sham mice (Fig.
1). IL-22 and IL-2 levels were the highest in CLP+saline mice
(Fig. 1). In the LGG-pretreated
mice, IL-22 and IL-2 levels remained elevated in the septic mice
compared with the sham mice (P<0.05; Fig. 1), but were decreased compared with
the CLP+saline mice (P<0.05; Fig.
1). TNF-α and IL-6 levels were higher in septic mice compared
with sham mice (P<0.001; Fig.
1), regardless of LGG pretreatment or not.
LGG pretreatment alleviates injury in
colon mucosa
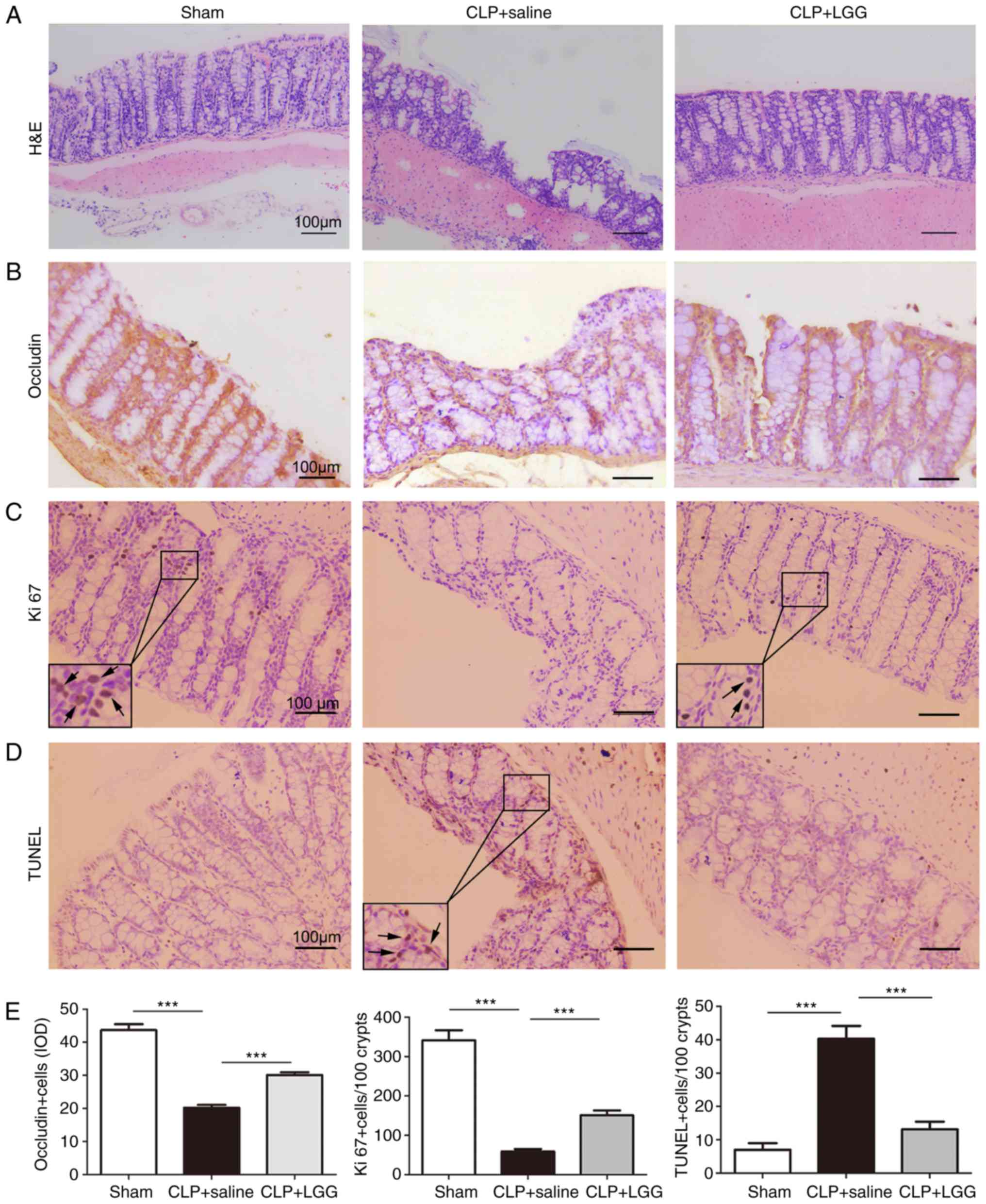
As evaluated by H&E staining and
histopathological analysis, colon tissues in sham mice were
histologically normal in all layers (Fig. 2A). In CLP+saline mice, the
epithelial structure was almost entirely corrupted and
disintegrated, accompanied with gland deformation and infiltration
of inflammatory cells (Fig. 2A).
However, with LGG pretreatment, the epithelial appearance was
regular, with less inflammatory cell infiltration in septic mice
(Fig. 2A). Occludin staining
revealed a significant decrease in tight junction formation in the
colonic tissue of both CLP+saline and CLP+LGG mice, compared with
the sham group (Fig. 2B).
However, the level of Occludin was much higher in CLP+LGG mice
compared with the CLP+saline mice (P<0.001; Fig. 2B and E). Colon mucosal injury was
obvious in septic mice. The damage in septic mice pretreated with
LGG was less obvious compared with the CLP+saline mice. Significant
differences existed in mucosal morphology and tight junction
formation among CLP+LGG mice, CLP+saline mice and sham mice.
LGG pretreatment normalizes cell
proliferation and apoptosis in colon
In the present study, colon mucosal injury was
obvious in septic mice, but the injury was alleviated by LGG
pretreatment. Therefore, cell proliferation marker and apoptosis of
the colonic epithelium were examined next, by Ki67 staining
(Fig. 2C) and TUNEL assay
(Fig. 2D), respectively.
Quantification of the results revealed a ~6-fold decrease of
Ki67-positive cells (P<0.001) and a significant increase of
TUNEL-positive cells (P<0.001) in CLP+saline mice compared with
the sham group (Fig. 2E).
Furthermore, the average numbers of Ki67-positive cells
dramatically increased (~3-fold), while TUNEL-positive cells
significantly decreased in CLP+LGG mice compared with CLP+saline
mice (P<0.001; Fig. 2E). These
results suggested that LGG pretreatment promoted the proliferation
and decreased the apoptosis of colon tissue in septic mice.
LGG pretreatment increases microbial
diversity in mice with sepsis
The coverage index was >97% per sample, which
means the 16SrRNA sequences detected in the present study were
sufficient to represent the majority of the bacteria present in the
samples. The OTUs, richness index (Chao 1) and diversity index
(Shannon and Simpson) were decreased in control septic mice
compared with sham mice (Table
II). With LGG pretreatment, all the four indexes increased in
septic mice (Table II). Although
no significant difference was observed in the four indexes, the
richness and diversity of bacteria in the CLP+LGG mice was much
closer to that of sham mice compared with CLP+saline mice (Table II). The result indicated that
sepsis decreased microbial richness and diversity, and the trend
could be reversed by LGG treatment.
 | Table IISummary of alpha analysis data. |
Table II
Summary of alpha analysis data.
| Sham | CLP+saline | CLP+LGG |
|---|
| OTUs |
1,299.60±166.56 |
1,058.75±199.53 |
1,184.63±343.76 |
| Coverage | 99% | 99% | 99% |
| Chao1 |
2,151.26±230.87 |
1,800.81±325.82 |
1,953.06±569.96 |
| Shannon | 6.27±0.39 | 5.29±0.72 | 5.84±1.12 |
| Simpson | 0.97±0.02 | 0.92±0.06 | 0.94±0.07 |
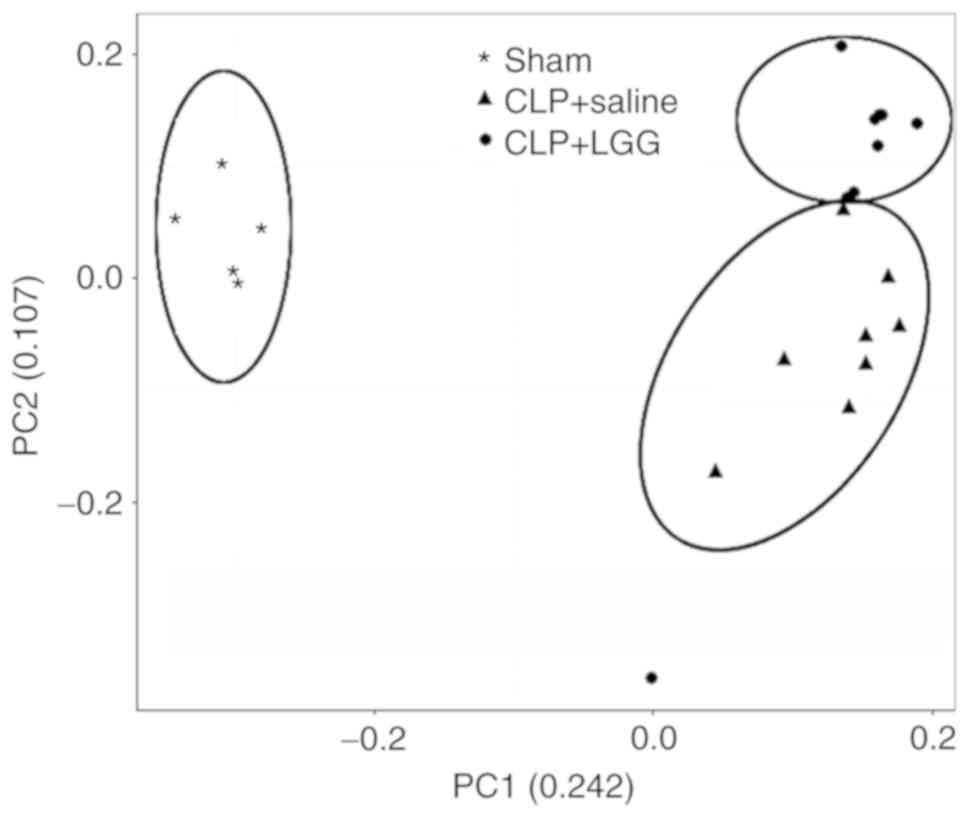
Principal coordinates analysis among
three groups
The weighted unifrac PCoA plot was used to compare
the similarities of microbiota structure among three groups. The
PCoA plot revealed a significantly separate clustering in
microbiota structure among the three groups. The distance between
CLP+LGG and sham mice was closer compared with the distance between
CLP+saline and sham mice (PC1 and PC2 were 0.242 and 0.107,
respectively; Fig. 3).
Bacterial taxonomic differences between
septic mice with different treatment
The microbial profiles of the experimental mouse
groups represented 10 phyla, among which, Bacteroide, Firmicutes
and Verrucomicrobia were the dominant phyla in sham mice, while
Bacteroide, Proteobactria and Firmicutes composed the majority of
phyla in both control CLP+saline and CLP+LGG mice (Fig. 4A and B). Compared with sham mice,
the level of Firmicutes was lower, Proteobactria (P<0.01) and
Deferribacteres (P<0.05) were higher, and the ratio of
Bacteroidetes/Firmicutes (B/F) increased in CLP+saline mice. With
LGG pretreatment, both bacteria composition and B/F ratio in septic
mice reversed to the level of sham mice (P>0.05; Fig. 4C and D). The majority of OTUs that
were detected among the three groups of mouse microbiomes were
assigned to 28 bacterial families. Among these detected taxa,
Staphylococcaceae and Enterococcaceae only existed in septic mice,
whereas Prevotellaceae was only present in sham mice.
Verrucomicrobiaceae (P<0.05), S24-7 (P<0.05), F16
(P<0.001) and Alcaligenaceae (P<0.001) were remarkably lower
and Enterobacteriaceae (P<0.01), Bacteroidaceae (P<0.01),
Erysipelotrichaceae, Deferribacteraceae (P<0.05), Clostridiaceae
(P<0.05) and Pseudomonadaceae (P<0.01) were higher in
CLP+saline mice compared with sham mice. With LGG treatment,
Staphylococcaceae disappeared, Enterococcaceae decreased and the
other bacteria reversed to the levels of shams (P>0.05; Fig. 4E and F). At genus level,
Akkermansia (P>0.05), Coprococcus (P<0.01), Sutterella
(P<0.001), Oscillospira (P>0.05), Lactobacillus (P<0.05)
and Desulfovibrio (P<0.01) decreased, and Bacteroides
(P<0.01), Pseudomonas (P<0.01), Parabacteroides, Ruminococcus
(P<0.05), Mucispirillum (P<0.05) and Vagococcus (P<0.05)
increased in CLP+saline mice compared with sham mice. With LGG
pretreatment, no significant difference was observed between septic
and sham mice in most of the bacteria (Fig. 4G and H).
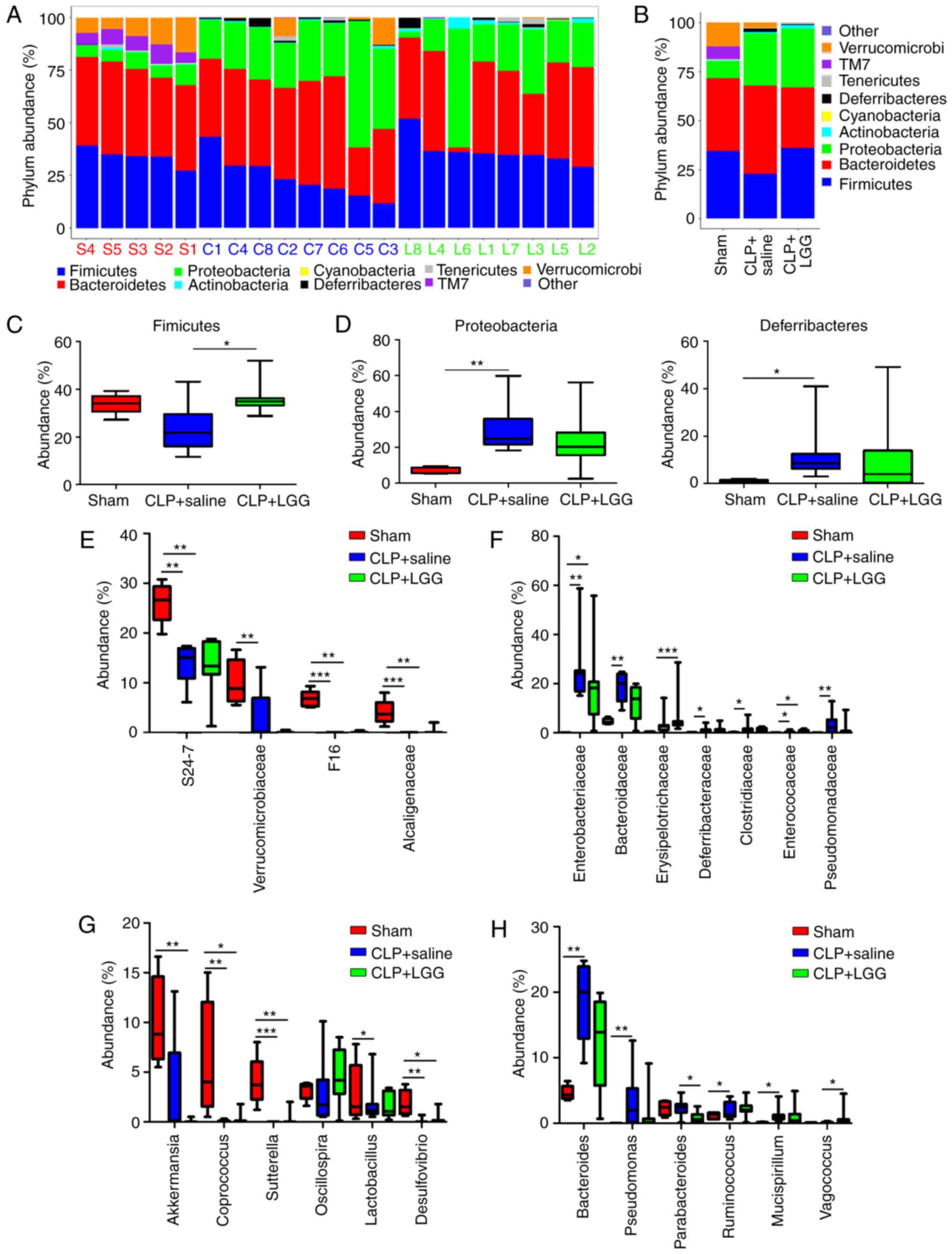 | Figure 4Comparison of the major microbiomes at
the (A–D) phyla, (E and F) family and (G and H) genus levels
between sham mice (red), CLP+saline (blue) and CLP+LGG mice (green)
respectively. (A) Comparison of the abundances of bacterial phyla
of each sample. (B) Comparison of the average abundance of each
bacterial phylum in the sham, CLP+saline and CLP+LGG mice,
respectively. (C and D) Significant differences among the
abundances of discriminatory phyla among three groups. (E and G)
Enrichment in sham mice; (F and H) Enrichment in CLP+saline mice;
the upper and lower ranges of the box represent the 75 and 25%
quartiles, respectively. Sham mice, n=5; CLP+saline mice, n=8;
CLP+LGG mice, n=8. *P<0.05, **P<0.01
and ***P<0.001 with comparisons indicated by lines.
LGG, Lactobacillus rhamnosus GG; CLP, cecal ligation and
puncture. |
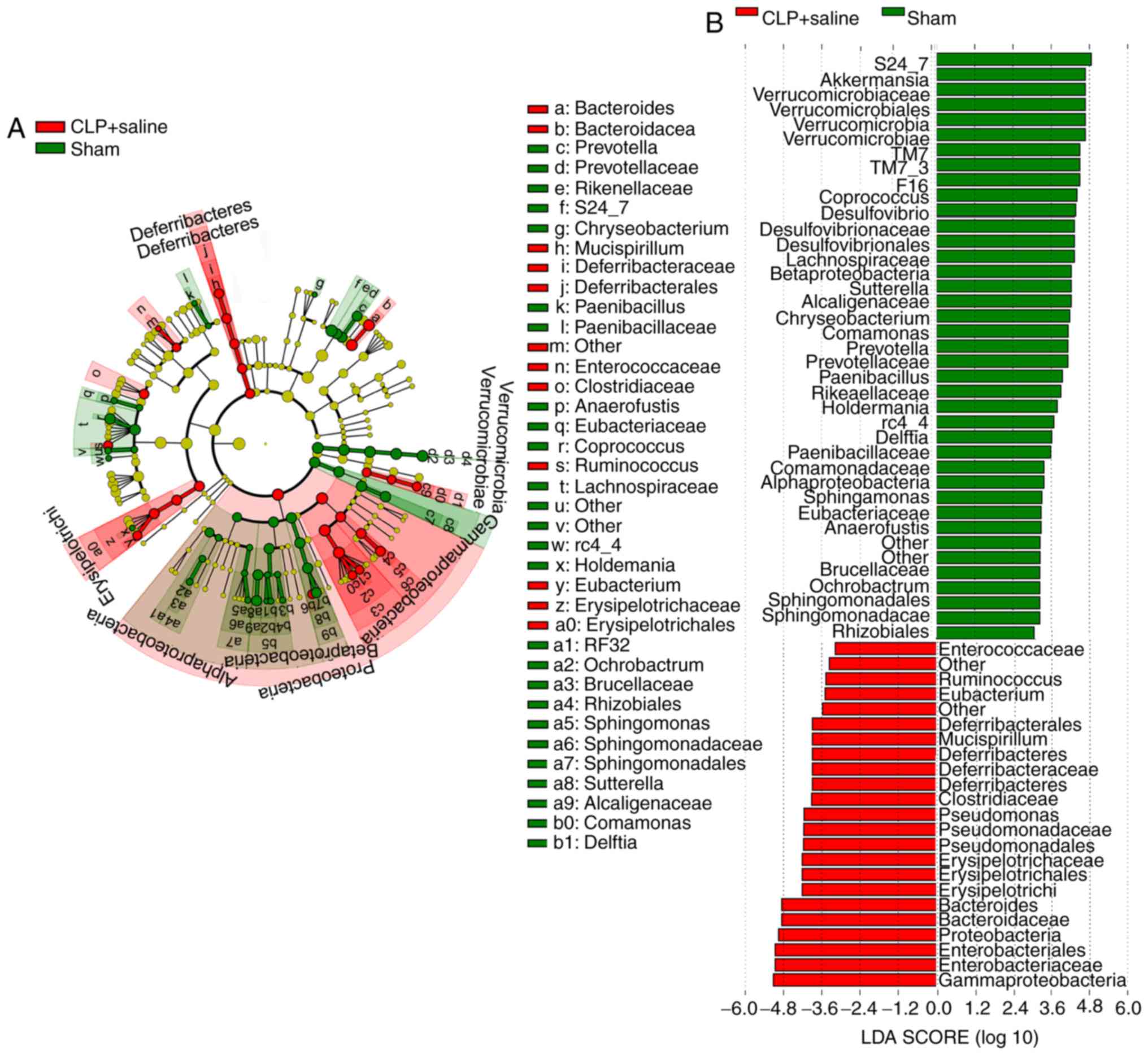
Linear discriminant analysis (LDA) effect
size (LEfSe) reveals microbiota structure of the three groups and
their predominant bacteria
LEfSe was used to compare the microbiota phylotypes
among the three groups. The sham microbiome had a huge
preponderance of Verrucomicrobia, Akkermansia, S24-7, F16 and TM_7,
whereas the CLP+saline microbiome had a preponderance of
Bacteroide, Enterobacteriales, Enterococcaceae, Deferribacteres,
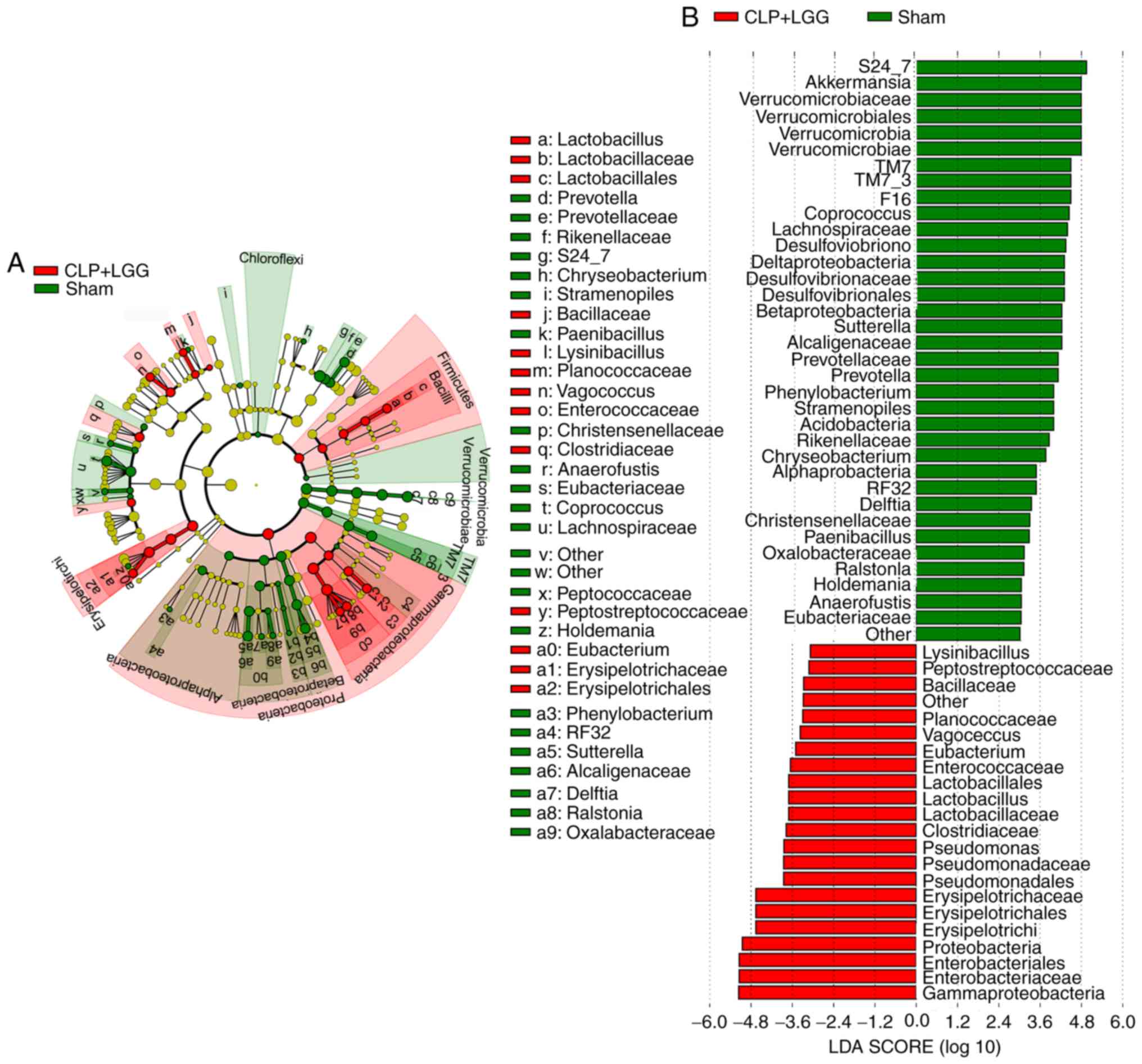
Pseudomonales and Erysipelotrichi (Fig. 5). Lactobacillales, Pseudomonales
and Erysipelotrichi were enriched in CLP+LGG mice (Fig. 6). Compared with control septic
mice, Deferribacteres disappeared, and Verrucomicrobia and
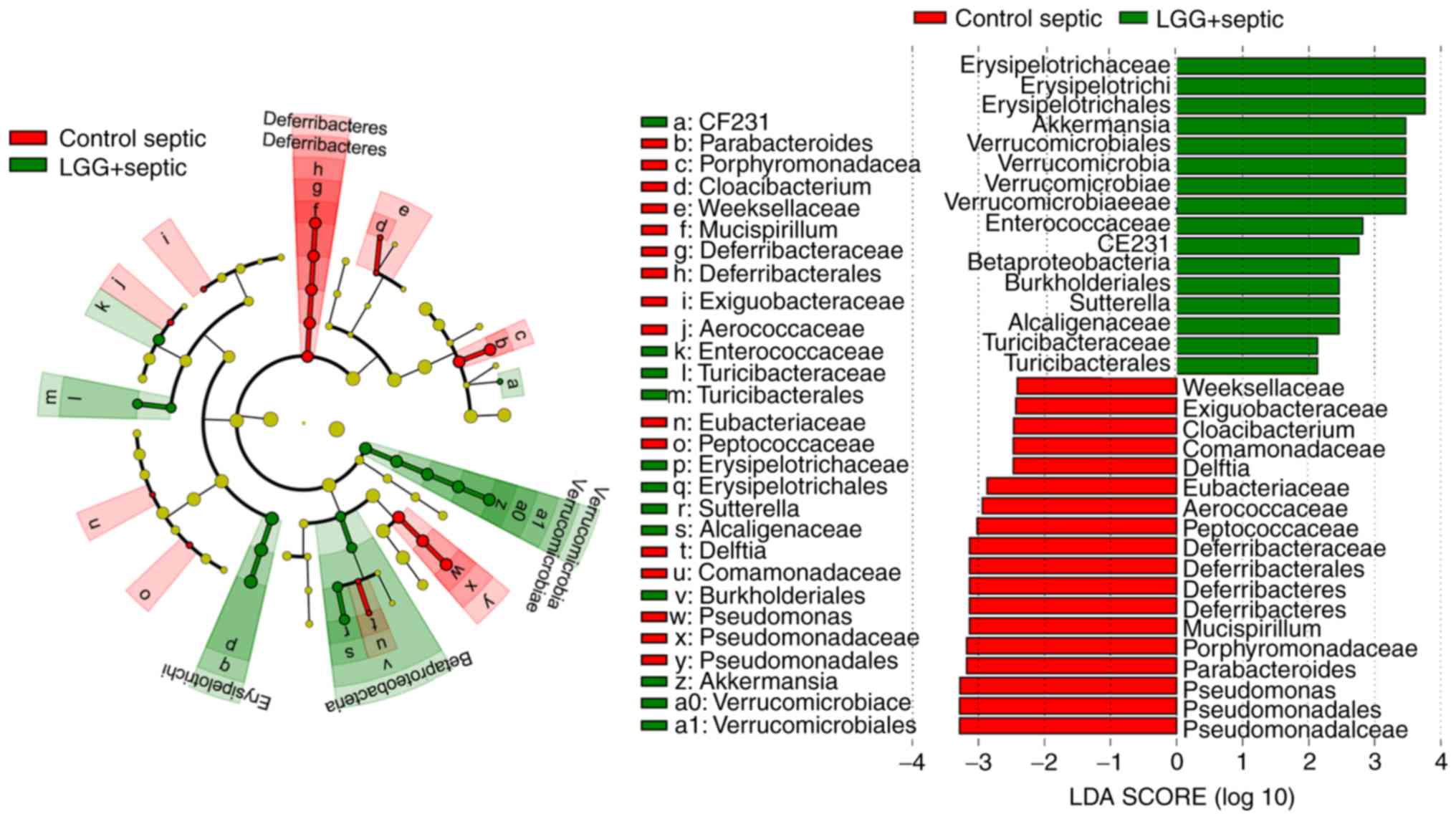
Akkermansia appeared in septic mice treated with LGG (Fig. 7). These results suggested that
sepsis-induced microbiota dysbiosis can be reversed by LGG
pretreatment.
Discussion
Critical illness and its treatment create a hostile
environment in the gastrointestinal tract by altering the
microbiota. Altered mucosal oxygen gradient (16) and increased nitrate production
(17) caused by critical illness
favor the growth and invasion of opportunistic pathogens, such as
Pseudomonas and Escherichia coli in Proteobacteria
phylum, and Staphylococcus and Enterococcus in
Firmicutes phylum (18–20), resulting in the release of
cytokines, cell apoptosis and corruption of epithelial tight
junctions (21). With loss of
intestinal barrier function, the gut is unable to prevent the
translocation of pathogens and toxins into the blood and
extraintestinal organs, leading to or aggravating sepsis and
mortality (22). The present
study demonstrated that, after the onset of sepsis, there was an
appearance of opportunistic pathogens Staphylococcaceae and
Enterococcaceae, and a disappearance of beneficial Prevotellaceae.
High relative abundance of potentially pathogenic commensals, such
as Enterobacteriaceae, Bacteroidaceae, Erysipelotrichaceae,
Deferribacteraceae, Clostridiaceae and Pseudomonadaceae, was
associated with more severe immune responses during sepsis,
demonstrated by higher serum levels of proinflammatory cytokines
(IL-22, IL-2, TNF-α and IL-6), epithelial cell apoptosis and
disruption of tight junctions. With LGG pretreatment, opportunistic
pathogens decreased or even disappeared, while beneficial bacteria,
such as Verrucomicrobiaceae, increased, epithelial cell apoptosis
was inhibited, and proliferation and cell tight junction formation
was promoted. The present results suggest that prophylactic LGG
therapy could be effective in reducing mortality from sepsis via
the normalization of altered gut flora, inhibiting systemic
inflammation and maintaining the mucosal barrier function.
Bacteroidetes and Firmicutes are the two dominant
phylum in the human and mouse microbiome. Changes in the relative
abundance of Bacteroidetes and Firmicutes have been determined to
affect energy balance. Firmicutes was related with energy harvest
and storage, while Bacteroidetes has a capacity to consume energy
(23). The present results
revealed a higher B/F ratio in CLP+saline mice compared with sham
mice. With LGG treatment, the B/F ratio decreased. One potential
explanation for this may be that the high degree of systemic
inflammation caused by sepsis is a high energy consuming process,
and therefore, during this process, both energy harvest and storage
decreased. As a result, Firmicutes decreased and Bacteroidetes
increased. Prophylactic LGG therapy partly reversed the B/F ratio
and therefore rebalanced the energy intake and expenditure, and
reduced mortality in sepsis.
Vollaard et al (24) reported the concept of colonization
resistance (CR). In this concept, the anaerobic microbiome limits
the concentration of opportunistic pathogenic (mostly aerobic)
bacteria in the gastrointestinal tract (24). Analysis of fecal microbiome
revealed that total anaerobic bacteria counts decreased notably in
patients with severe systemic inflammatory response syndrome
(25). Research linking gut
translocation of bacteria to the development of postoperative
sepsis indicated that Gram-negative facultative anaerobic bacteria
were the dominant microflora in patients with postoperative sepsis,
whereas obligate anaerobes counts decreased, suggesting that the
imbalance between obligate anaerobes and facultative anaerobes is
closely related to the incidence of infectious complications in
critically ill patients (26). In
the present study, facultative anaerobic bacteria, including
Enterobacteriaceae, Bacteroidaceae and Erysipelotrichaceae,
increased significantly and obligate anaerobes, such as
Prevotellaceae, disappeared in control septic mice. Prophylactic
LGG therapy rebalanced the ratio of obligate anaerobes and
facultative anaerobes, and therefore enhanced the ability of
colonization resistance in the host.
Evidence suggests that several specific members of
microbiota serve important roles in rebalancing dysbiosis and
preventing disease (27,28). For example, Coprococcus produces
butyrate to provide an energy source for epithelial cells (29) and induces the differentiation of
colonic regulatory T cells to suppress inflammatory and allergic
responses (30). Akkermansia has
been described to have an anti-inflammatory property. It has been
reported that Akkermansia administration could raise the levels of
endocannabinoids in intestinal cells that control the gut peptide
secretion, gut barrier, and inflammation (31). In addition, Akkermansia,
Coprococcus, Lactobacillus, Oscillibacter have been correlated
negatively with lipopolysaccharide (LSP) in feces and hepatic
function features (including plasma glucose and total lipids),
whereas the LPS-producer Bacteroides were positively correlated
with fecal LPS and hepatic function in mice with liver diseases
(32). In the present study,
Akkermansia, Coprococcus, Sutterella, Oscillospira, Lactobacillus
and Desulfovibrio were decreased and Bacteroides were increased in
control septic mice compared with sham mice at the genus level.
With LGG treatment, no significant difference was observed between
septic and sham mice, indicating that LGG may increase the gut
barrier function, decrease the levels of LPS, reduce inflammation
and improve hepatic function through increasing Akkermansia,
Coprococcus, Lactobacillus, Oscillibacter and decreasing
Bacteroides.
In summary, the present study demonstrated that
prophylactic LGG therapy reduced mortality through attenuating
inflammatory responses, and increasing gut barrier integrity and
function. LGG pretreatment increased the diversity of intestinal
microbiota and expression of beneficial bacterium. Among them,
Prevotellaceae, Lactobacillaceae, Staphylococcaceae,
Enterococcaceae, Enterobacteriaceae, Bacteroidaceae,
Deferribacteraceae can be regarded as the key bacteria in sepsis
treatment. Prevotellaceae may be exploited as a potential probiotic
in sepsis treatment.
Funding
The present study was supported by the Natural
Science Foundation of China (grant no. 81771498) and by Diagnosis
and treatment of gastrointestinal dysfunction in elderly patients
with integrated traditional Chinese and Western Medicine
(2017-XK-A31).
Availability of data and materials
The analyzed datasets generated during the study are
available from the corresponding author on reasonable request. The
data that support the findings of survival studies are available
from reference 10.
Authors' contributions
LC performed the histological examination of the
colon, 16SrRNA analysis and was a major contributor in writing the
manuscript. HL and JL performed the LGG administration and CLP
model establishment. YC performed the ELISA analysis. YY was the
corresponding author of this manuscript and took part in revising
the manuscript. All authors read and approved the final
manuscript.
Ethics approval and consent to
participate
All procedures for animal care and use were approved
by the Animal Care Ethics Committee of the First Affiliated
Hospital, Zhejiang University (Hangzhou, China).
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
Acknowledgments
Not applicable.
References
|
1
|
Martin GS: Sepsis, severe sepsis and
septic shock: Changes in incidence, pathogens and outcomes. Expert
Rev Anti Infect Ther. 10:701–706. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Dickson RP: The microbiome and critical
illness. Lancet Respir Med. 4:59–72. 2016. View Article : Google Scholar :
|
|
3
|
Klingensmith NJ and Coopersmith CM: The
gut as the motor of multiple organ dysfunction in critical illness.
Crit Care Clin. 32:203–312. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Sánchez B, Delgado S, Blanco-Míguez A,
Lourenço A, Gueimonde M and Margolles A: Probiotics, gut
microbiota, and their influence on host health and disease. Mol
Nutr Food Res. 61:2017. View Article : Google Scholar
|
|
5
|
Krumbeck JA, Maldonado-Gomez MX,
Ramer-Tait AE and Hutkins RW: Prebiotics and synbiotics: Dietary
strategies for improving gut health. Curr Opin Gastroenterol.
32:110–119. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Arboleya S, Watkins C, Stanton C and Ross
RP: Gut bifidobacteria populations in human health and aging. Front
Microbiol. 7:12042016. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Sakai F, Hosoya T, Ono-Ohmachi A, Ukibe K,
Ogawa A, Moriya T, Kadooka Y, Shiozaki T, Nakagawa H, Nakayama Y
and Miyazaki T: Lactobacillus gasseri SBT2055 induces TGF-β
expression in dendritic cells and activates TLR2 signal to produce
IgA in the small intestine. PLoS One. 9:e1053702014. View Article : Google Scholar
|
|
8
|
Jo SG, Noh EJ, Lee JY, Kim G, Choi JH, Lee
ME, Song JH, Chang JY and Park JH: Lactobacillus curvatus WiKim38
isolated from kimchi induces IL-10 production in dendritic cells
and alleviates DSS-induced colitis in mice. J Microbiol.
54:503–509. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Besselink MG, van Santvoort HC, Buskens E,
Boermeester MA, van Goor H, Timmerman HM, Nieuwenhuijs VB, Bollen
TL, van Ramshorst B, Witteman BJ, et al: Probiotic prophylaxis in
predicted severe acute pancreatitis: A randomised, double-blind,
placebo-controlled trial. Lancet. 371:651–659. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Chen L, Xu K, Gui Q, Chen Y, Chen D and
Yang Y: Probiotic pre-administration reduces mortality in a mouse
model of cecal ligation and puncture-induced sepsis. Exp Ther Med.
12:1836–1842. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
von Martels JZ, Sadaghian Sadabad M,
Bourgonje AR, Blokzijl T, Dijkstra G, Faber KN and Harmsen HJM: The
role of gut microbiota in health and disease: In vitro modeling of
host-microbe interactions at the aerobe-anaerobe interphase of the
human gut. Anaerobe. 44:3–12. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Das S, Dash HR, Mangwani N, Chakraborty J
and Kumari S: Understanding molecular identification and polyphasic
taxonomic approaches for genetic relatedness and phylogenetic
relationships of microorganisms. J Microbiol Methods. 103:80–100.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Rittirsch D, Huber-Lang MS, Flierl MA and
Ward PA: Immunodesign of experimental sepsis by cecal ligation and
puncture. Nat Protoc. 4:31–36. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Caporaso JG, Kuczynski J, Stombaugh J,
Bittinger K, Bushman FD, Costello EK, Fierer N, Peña AG, Goodrich
JK, Gordon JI, et al: QIIME allows analysis of high-throughput
community sequencing data. Nat Methods. 7:335–336. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Glöckner FO, Yilmaz P, Quast C, Gerken J,
Beccati A, Ciuprina A, Bruns G, Yarza P, Peplies J, Westram R and
Ludwig W: 25 years of serving the community with ribosomal RNA gene
reference databases and tools. J Biotechnol. 261:169–176. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Albenberg L, Esipova TV, Judge CP,
Bittinger K, Chen J, Laughlin A, Grunberg S, Baldassano RN, Lewis
JD, Li H, et al: Correlation between intraluminal oxygen gradient
and radial partitioning of intestinal microbiota. Gastroenterology.
147:1055–1063. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Winter SE, Winter MG, Xavier MN,
Thiennimitr P, Poon V, Keestra AM, Laughlin RC, Gomez G, Wu J,
Lawhon SD, et al: Host-derived nitrate boosts growth of E. coli in
the inflamed gut. Science. 339:708–711. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Lupp C, Robertson ML, Wickham ME, Sekirov
I, Champion OL, Gaynor EC and Finlay BB: Host-mediated inflammation
disrupts the intestinal microbiota and promotes the overgrowth of
Enterobacteriaceae. Cell Host Microbe. 2:119–129. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Honda K and Littman DR: The microbiome in
infectious disease and inflammation. Annu Rev Immunol. 30:759–795.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Grootjans J, Lenaerts K, Derikx JP,
Matthijsen RA, de Bruïne AP, van Bijnen AA, van Dam RM, Dejong CH
and Buurman WA: Human intestinal ischemia-reperfusion-induced
inflammation characterized: Experiences from a new translational
model. Am J Pathol. 176:2283–2291. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Alverdy JC, Laughlin RS and Wu L:
Influence of the critically ill state on host-pathogen interactions
within the intestine: Gut-derived sepsis redefined. Crit Care Med.
31:598–607. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Mittal R and Coopersmith CM: Redefining
the gut as the motor of critical illness. Trends Mol Med.
20:214–223. 2014. View Article : Google Scholar :
|
|
23
|
Turnbaugh PJ, Ley RE, Mahowald MA, Magrini
V, Mardis ER and Gordon JI: An obesity-associated gut microbiome
with increased capacity for energy harvest. Nature. 444:1027–1031.
2006. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Vollaard EJ and Clasener HA: Colonization
resistance. Antimicrob Agents Chemother. 38:409–414. 1994.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Shimizu K, Ogura H, Goto M, Asahara T,
Nomoto K, Morotomi M, Yoshiya K, Matsushima A, Sumi Y, Kuwagata Y,
et al: Altered gut flora and environment in patients with severe
SIRS. J Trauma. 60:126–133. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
O'Boyle CJ, MacFie J, Mitchell CJ,
Johnstone D, Sagar PM and Sedman PC: Microbiology of bacterial
translocation in humans. Gut. 42:29–35. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Lynch SV and Pedersen O: The human
intestinal microbiome in health and disease. N Engl J Med.
375:2369–2379. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Shreiner AB, Kao JY and Young VB: The gut
microbiome in health and in disease. Curr Opin Gastroenterol.
31:69–75. 2015. View Article : Google Scholar :
|
|
29
|
Donohoe DR, Garge N, Zhang X, Sun W,
O'Connell TM, Bunger MK and Bultman SJ: The microbiome and butyrate
regulate energy metabolism and autophagy in the mammalian colon.
Cell Metab. 13:517–526. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Furusawa Y, Obata Y, Fukuda S, Endo TA,
Nakato G, Takahashi D, Nakanishi Y, Uetake C, Kato K, Kato T, et
al: Commensal microbe-derived butyrate induces the differentiation
of colonic regulatory T cells. Nature. 504:446–450. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Everard A, Belzer C, Geurts L, Ouwerkerk
JP, Druart C, Bindels LB, Guiot Y, Derrien M, Muccioli GG, Delzenne
NM, et al: Cross-talk between Akkermansia muciniphila and
intestinal epithelium controls diet-induced obesity. Proc Natl Acad
Sci USA. 110:9066–9071. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Xie G, Wang X, Liu P, Wei R, Chen W,
Rajani C, Hernandez BY, Alegado R, Dong B, Li D and Jia W:
Distinctly altered gutmicrobiota in the progression of liver
disease. Oncotarget. 7:19355–19366. 2016.PubMed/NCBI
|