Introduction
Asthma is a chronic airway disease that affects
>300,000,000 individuals worldwide (1). The prevalence of atopy and asthma
has markedly increased in developing countries in recent years
(2). The annual worldwide
mortality rate of asthma has been reported to be ~250,000 and it is
also a substantial economic burden (1).
Failure to clear bronchial secretions is the main
pathological reason for asthmatic lung disease. The primary cause
for asthma-associated mortality is intraluminal airway asphyxiation
of mucus plugs (3,4). Defective mucociliary clearance is
observed even in mild stable asthma (5-7). A
decrease in clearance causes acute exacerbation (8).
In total, 21 genes are reported to encode mucins in
the human genome. Mucin 5AC (MUC5AC) is expressed at high levels in
the airway system (9,10). The expression of MUC5AC varies in
airway diseases, including asthma, with an increase in the number
of goblet cells. Mucus may alter the normal structure and status of
goblet cells after failing to incorporate with MUC5AC. Without the
normal reaction between MUC5AC and mucus, the airway
viscoelasticity becomes vulnerable to plugging (11-13).
Current evidence shows that MUC5AC is regulated by
several factors. Previous studies have found that interleukin-13
can increase the expression of MUC5AC in epithelial cell lines and
in murine models (14-17). Epidermal growth factor receptor
(EGFR) signaling increases the expression of MUC5AC and the
expression of MUC5AC can be inhibited with an EGFR tyrosine kinase
inhibitor (17). Previous studies
have also shown that toll-like receptor 2 is related to the
expression of MUC5AC in asthmatic models (18) and corticosteroids have been
demonstrated to decrease mucus in airways and further decrease the
expression of MUC5AC (19).
In the pathogenesis of asthma, epigenetic
modifications are important environmental and genetic factors. In
particular, the reversible process of histamine acetylation is
mediated by histone acetyltransferases (HATs) and histone
decarboxylases (HDACs); histone acetylation is closely associated
with gene transcription and controlled by HATs.
P300 (EP300 or KAT3B) consists of 2,414 amino acids
and its full length is 300 kDa. It was first cloned in 1994
(20,21) in studies aimed at identifying
proteins that bind E1A, an adenoviral oncogenic transcription
factor. P300 was found to have HAT activity and to acetylate a
number of proteins (22). Studies
have shown that the gain-of-function in P300 can be induced by
mutation, the overexpression of P300 or several disease etiology
conditions, such as diabetes, obesity, thrombocytopenia or
hypertrophic heart diseases. Although, a P300 inhibitor has not
been used in clinical trials (23), P300 inhibitors are considered
potential therapeutic applications in several diseases.
In the present study, it was hypothesized that P300
may suppress the production of MUC5AC. The MUC5AC promoter was
constructed and its activity was investigated with dual-luciferase
assays. The study then observed the effects of the HAT protein P300
on the transcription of MUC5AC in adenocarcinomic human alveolar
basal epithelial cells and examined the associated mechanisms.
Materials and methods
Cell culture
The A549 lung carcinoma cell line and Beas-2b cells
were cultured in Dulbecco's modified Eagle's medium with 10% fetal
bovine serum (both Thermo Fisher Scientific, Inc., Waltham, MA,
USA) and antibiotics (50 U/ml penicillin and 50 µg/ml
streptomycin). The cells were maintained at 37°C in a 5%
CO2 humidified chamber.
Plasmids and small interfering RNA
(siRNA)
The DNA sequence (-1,300 to +48) of the MUC5AC
promoter region was amplified by polymerase chain reaction (PCR)
and digested with KpnI and BglII (Thermo Fisher
Scientific, Inc.). PCR was performed by using genomic DNA which was
extracted from the A549 cells using a TIANamp Genomic DNA kit
(Tiangen Biotech Co., Ltd., Beijing, China), DNA polymerase (LA
Taq), dNTPs, GC Buffer (Thermo Fisher Scientific, Inc.) and forward
and reverse primers. The forward primers contained a KpnI
restriction site, and the reverse primer contained a BglII
site. Sequences of primers were as follows: Forward,
5′-CGGGGTACCCTACCCATTCACATTTTCCCCATCC-3′ and reverse,
5′-GGAAGATCTGGGACCAAGCTGAGCTCTGC-3′. PCR was performed with the
following thermocycling conditions: 94°C for 5 min followed by 30
cycles of amplification (94°C, 30 sec; 60°C, 30 sec; 72°C, 2 min)
and followed by a final extension at 72°C for 10 min. Subsequently,
it was subcloned into the promoter-less luciferase expression
plasmid, pGL3-Basic (Promega Corporation, Madison, WI, USA). The
resulting plasmid was termed pGL-1300/+48. Truncated plasmids of
the MUC5AC promoter were constructed using pGL-1300/+48 as a
template. KpnI and XhoI double digestions were
performed on the MUC5AC promoter fragments of different lengths
obtained from the above PCR reaction, and pGL3-enhancer vector. The
vector and the target fragments were linked using a T4 ligase;
competent DH5α cells (Tiangen Biotech Co., Ltd.) were transformed,
positive clones were selected, and verification was conducted using
restriction endonuclease analysis and DNA sequencing. The
successfully constructed plasmids were named pGL-1300/+48, P2:
pGL-935/+48, P3: pGL-583/+48, P4: pGL-205/+48, P5: pGL-116/+48, and
P6: pGL-23/+48. The siRNAs were synthesized and purified (Shanghai
GenePharma, Co. Ltd., Shanghai, China). The targeted sequence was
designed to silence P300 gene transcription with the sequence
5′-GUCCUGGAUUAGGUUUGAUTT-3′. The control siRNA sequence was
5′-AUCAAACCUAAUCCAGGACTT-3′.
Transient transfection and luciferase
assays
The p300 mutant (p300 mut; P300Δ1472-1522) and
wild-type (p300 wt) plasmids were provided by Professor Zhou
Guoping (Fourth School of Clinical Medicine, Nanjing Medical
University, Nanjing, China) and were originally described by Boyes
et al (24). siRNA
transfection in the A549 cells was performed using
Lipofectamine® 3000 (Invitrogen; Thermo Fisher
Scientific, Inc.), according to the manufacturer's protocol.
Transient transfection was performed following 24 h cell adaptation
on 96-well plates (1.5×104 cells/well).
In the ectopic overexpression experiments, the cells
were co-transfected with the P300 wt or P300 mut plasmids, and the
pGL-935/+48 reporter plasmid. Following transfection for 24 h, the
cells were collected for the luciferase assays. For the P300 siRNA
and P300 overexpression experiments, reporter plasmids containing
siRNA for P300 or the P300-overexpression plasmid were
co-transfected into A549 cells and harvested after 24 h.
Subsequently, the luciferase activity was measured with the Dual
Reporter assay system (Promega Corporation). All experiments were
conducted independently in triplicate.
RNA extraction and reverse
transcription-quantitative (RT-q)PCR analysis
RT-qPCR analysis was performed to confirm the
expression of MUC5AC in A549 cells. Following transfection for 24
h, the total R NA was extracted from the A549 cells using
TRIzol®. The RT-qPCR analysis was performed with the
SYBR PCR Master mix (Applied Biosystems; Thermo Fisher Scientific,
Inc.) and ABI 7500 Fast system (Applied Biosystems; Thermo Fisher
Scientific, Inc.). The primers were synthesized by Changzhou Bo
Hong Biological Engineering Co., Ltd. (Changzhou, China). MUC5AC
primer sequences were as follows: Forward,
5′-CTGTGAAGGTGGCTGACCAAGA3′ and reverse,
5′AAGGTGTAGTAGGTGCCGTCGAA-3′; GAPDH was selected as the control
reference with the following primer sequences: Forward,
5′-TGGTATCGTGGAAGGACTCATGAC3′ and reverse,
5′TGCCAGTGAGCTTCCCGTTCAGC-3′. cDNA was reverse transcribed by
primers using the PrimeScript RT reagent kit with gDNA Eraser
(Perfect Real Time) according to the manufacturer's protocol. The
reverse transcription cDNA products were stored at -20°C for PCR
amplification. According to the instruction of the SYBR Premix Ex
Taq II (Tli RnaseH Plus), the following reagents were added to the
PCR reaction mixture: 2 µl cDNA template, 0.8 µl
forward primer (10 µM), 0.8 µl reverse primer (10
µM), 10 µl SYBR® Premix EX Taq™ II (2X),
0.4 µl ROX Reference Dye II (50X) and 6 µl ddH2O to
make a 20-µl total reaction system. Detection and
quantification were performed as follows: Pre-denaturation, 95°C
for 30 sec; 40 cycles of denaturation at 95°C for 5 sec; and
extension, 60°C for 34 sec. Fluorescence data was collected at the
extension step. The relative expression of the MUC5AC gene was
analyzed with the 2−ΔΔCq method (25).
Immunofluorescence
The A549 cells were seeded on glass coverslips in
6-well plates (5×104 cells/well). When the cells reached
~50% confluence, the cells were washed twice with PBS and fixed
with 4% paraformaldehyde. Prior to staining with antibodies for
immunofluorescence, the cells were blocked with 0.2% Triton, 1%
bovine serum albumin and 1% goat serum (Sangon Biotech, Co., Ltd.,
Shanghai, China) for 30 min at room temperature. Following
blocking, the fixed cells were washed twice with PBS and incubated
with MUC5AC primary antibody (EPR16904; cat. no. ab198294; 1:250)
rabbit anti human monoclonal antibody (Abcam, Cambridge, UK) at 4°C
in a humid chamber overnight. The following morning, the cells were
incubated with fluorescein isothiocyanate-labeled goat anti-rabbit
immunoglobulin G (cat. no. ab150077; 1:500; Abcam) for 1 h at room
temperature and washed three times prior to observation under a
confocal laser fluorescent microscope (Zeiss 710; Carl Zeiss AG,
Oberkochen, Germany).
Statistical analysis
Statistical analysis was performed with SPSS
software (version 12.0; SPSS, Inc., Chicago, IL, USA). All data are
presented as the mean ± standard deviation. Student's t-test was
used to analyze the statistical significance between groups.
Differences between multiple groups were tested by one-way analysis
of variance followed by Tukey's post hoc test. P<0.05 was
considered to indicate a statistically significant difference.
Results
Identification of recombinant plasmid
pGL-1300/+48
The pGL-1300/+48 gene fragment was amplified by PCR.
The product and the pGL vector were digested using restriction
endonucleases BglII and KpnI. The recombinant plasmid
pGL-1300/+48 was constructed by connecting the digested products
with DNA ligase. The recombinant plasmid was then identified by
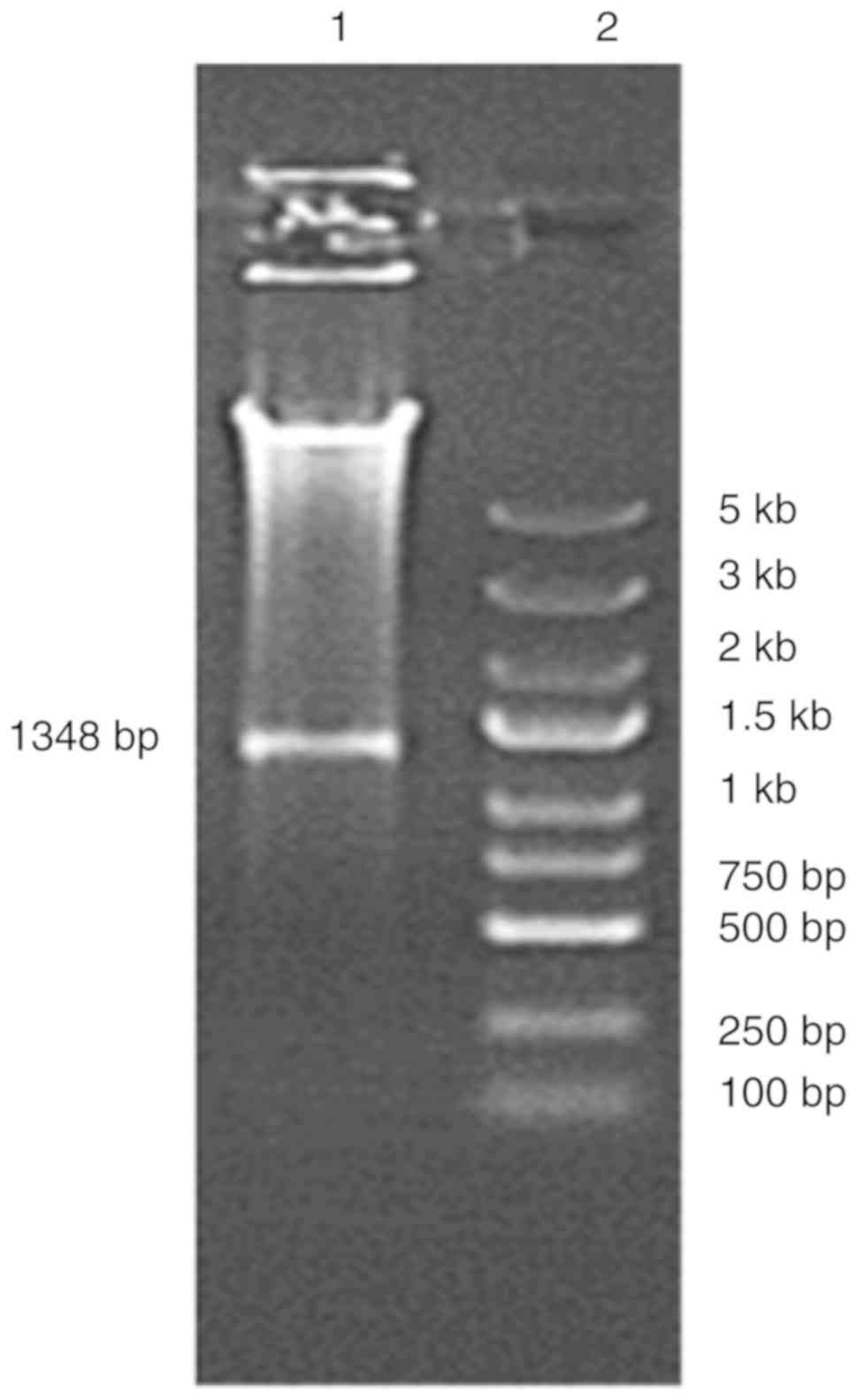
PCR. The endonuclease digestion result is presented in Fig. 1. The PCR product and the digested
product had a size of ~1,350 bp, as expected. The results indicated
that the recombinant plasmid pGL-1300/+48 was successfully
constructed. Through agarose gel electrophoresis, the PCR products
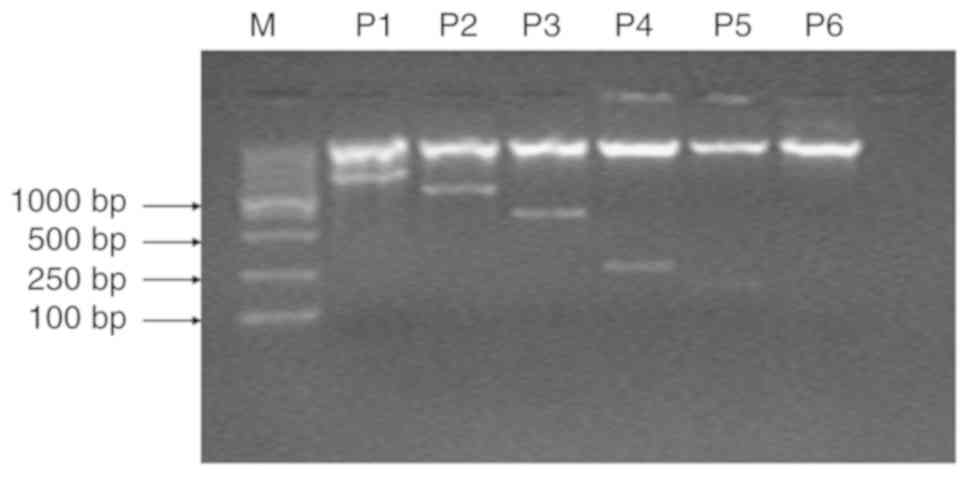
showed bands at ~983, 631, 253, 164 and 71 bp (Fig. 2). The electrophoresis results of
the PCR products were as predicted.
Verification of the recombinant
luciferase reporter plasmid
The electrophoresis results of the recombinant
luciferase reporter plasmid containing the MUC5AC promoter
fragments with restriction enzyme digestions are shown in Fig. 2. The results were as expected, and
further sequencing was conducted. The sequencing results were
consistent with the designed DNA fragment sequences, which further
confirmed the successful construction of a recombinant plasmid of
MUC5AC promoter fragments (Fig.
3).
Transcription activation activity of the
expression of MUC5AC
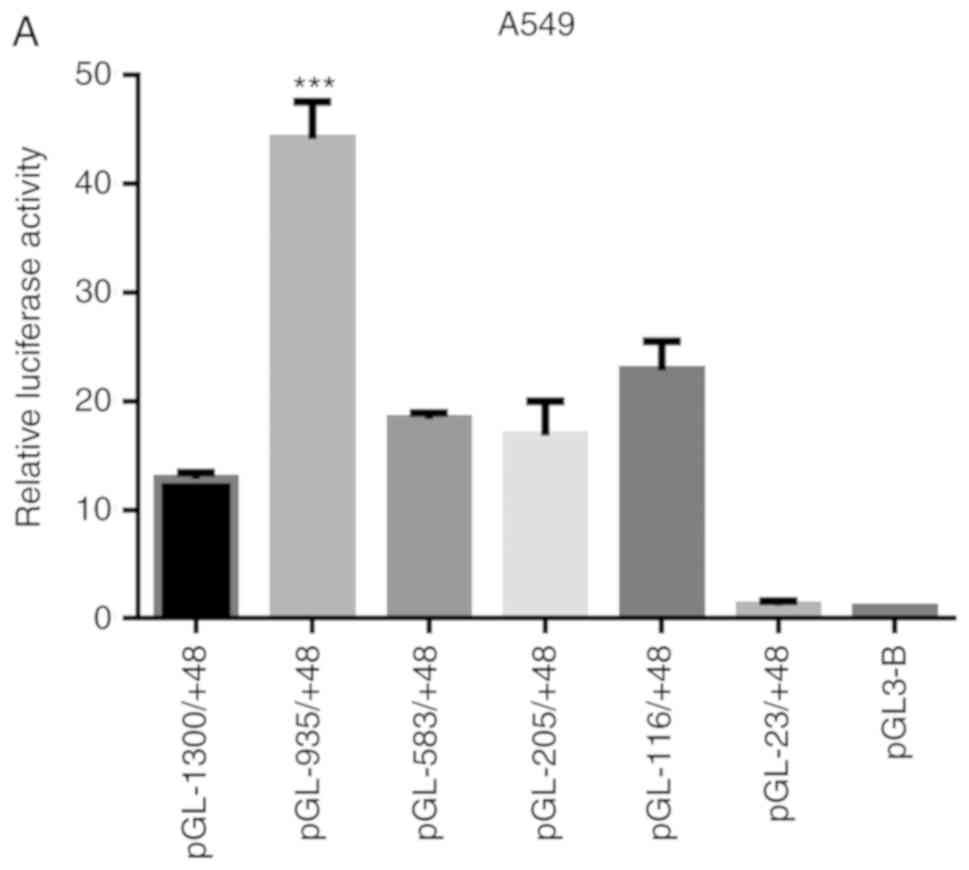
The reporter gene plasmids containing regulatory
sequences of different lengths of the human MUC5AC promoter region
were co-transfected with the internal control plasmid -TK into A549
and Beas-2b cells, respectively. The transfected cells were
stimulated for 30 min, and the specific luciferase activity was
detected. The results showed that the luciferase activity of the
recombinant pGL-MUC5AC-935/+48 plasmid was significantly induced
(Fig. 4A and B).
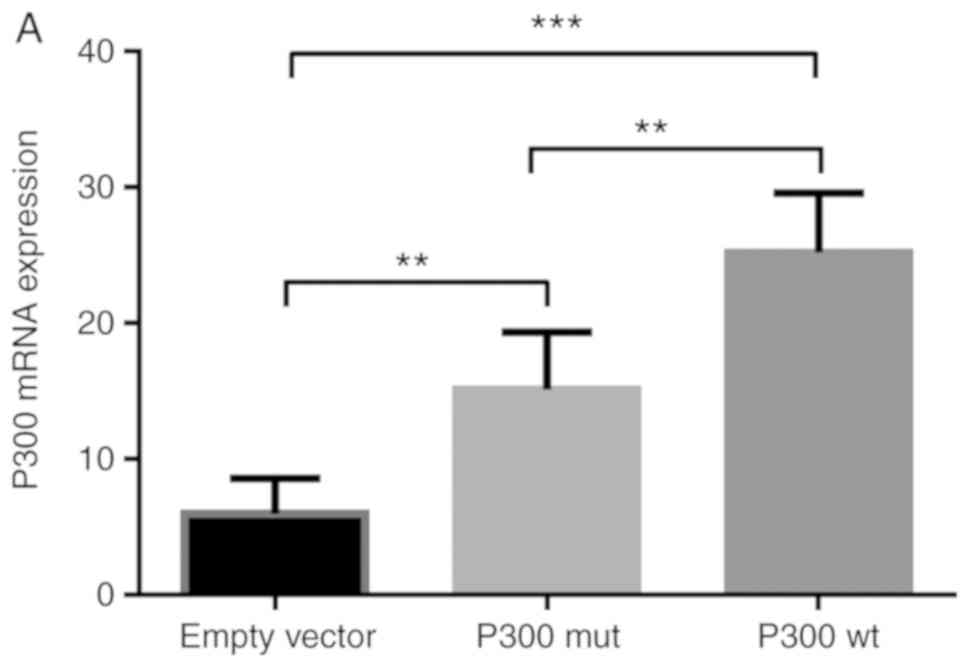
mRNA expression of P300
To demonstrate transfection success, the empty
vector control, the P300 expression plasmid (P300 wt) and the P300
siRNA, in addition to the control plasmids, were transfected into
A549 cells respectively. The transfected cells were stimulated for
30 min and the mRNA expression of P300 was detected. The results
showed that P300 wt markedly increased the mRNA expression of P300,
whereas P300 siRNA inhibited the expression of P300 (Fig. 5A and B).
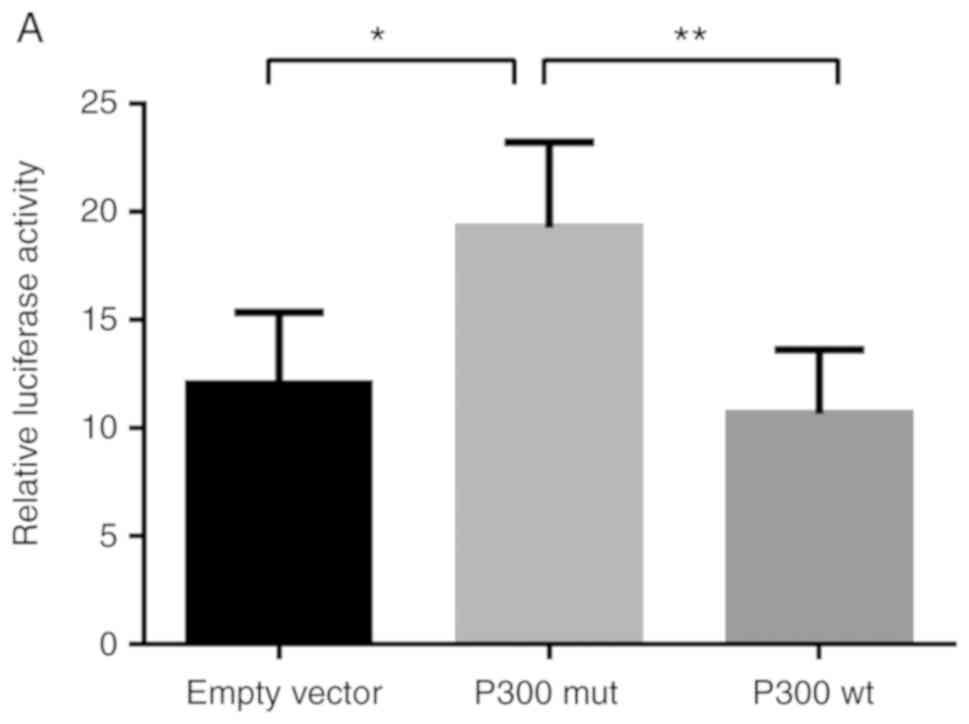
P300 inhibits MUC5AC promoter
activity
In order to determine how P300 regulates MUC5AC
promoter activity, the MUC5AC plasmid pGL-935/+48, the empty vector
control, P300 wt, P300 siRNA, and the control plasmids were
co-transfected into A549 cells, respectively (Fig. 6). By overexpressing P300, the
promoter activity was significantly decreased in the A549 cells
(Fig. 6A). However, P300 siRNA
markedly increased the promoter activity of MUC5AC (Fig. 6B). Taken together, P300 inhibited
the expression of MUC5AC by suppressing its promoter activity.
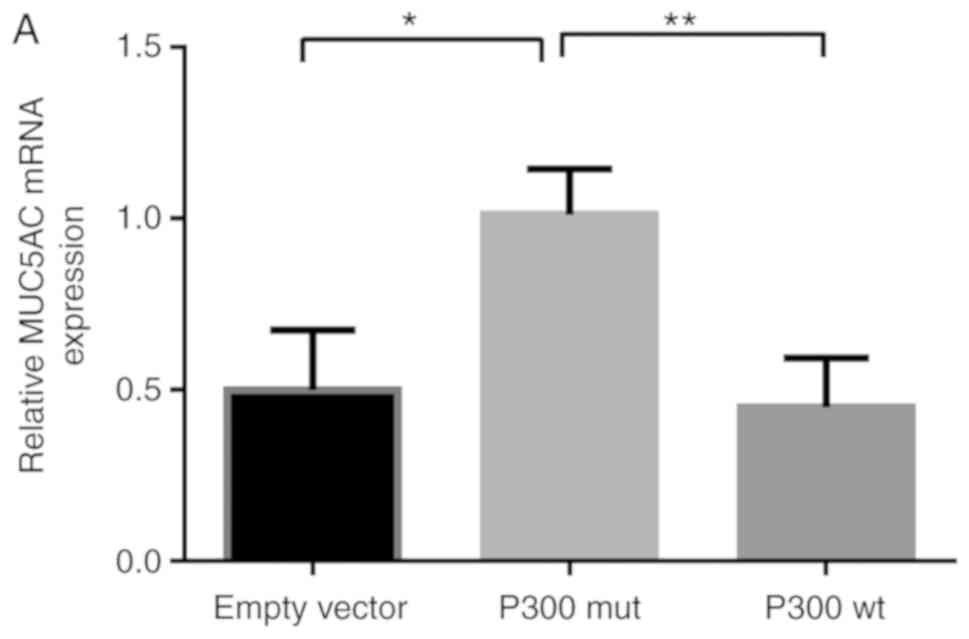
P300 decreases the mRNA level of
MUC5AC
To determine whether P300 affects MUC5AC gene
transcription, the co-transfected A549 cell RNA was isolated and
the expression of MUC5AC was detected by RT-qPCR analysis (Fig. 7A and B). The mRNA expression of
MUC5AC was significantly decreased following co-transfection with
the P300 wt expression plasmid compared with the control. By
contrast, P300 siRNA increased the mRNA levels of MUC5AC, which
suggests that P300 decreased the mRNA expression of MUC5AC.
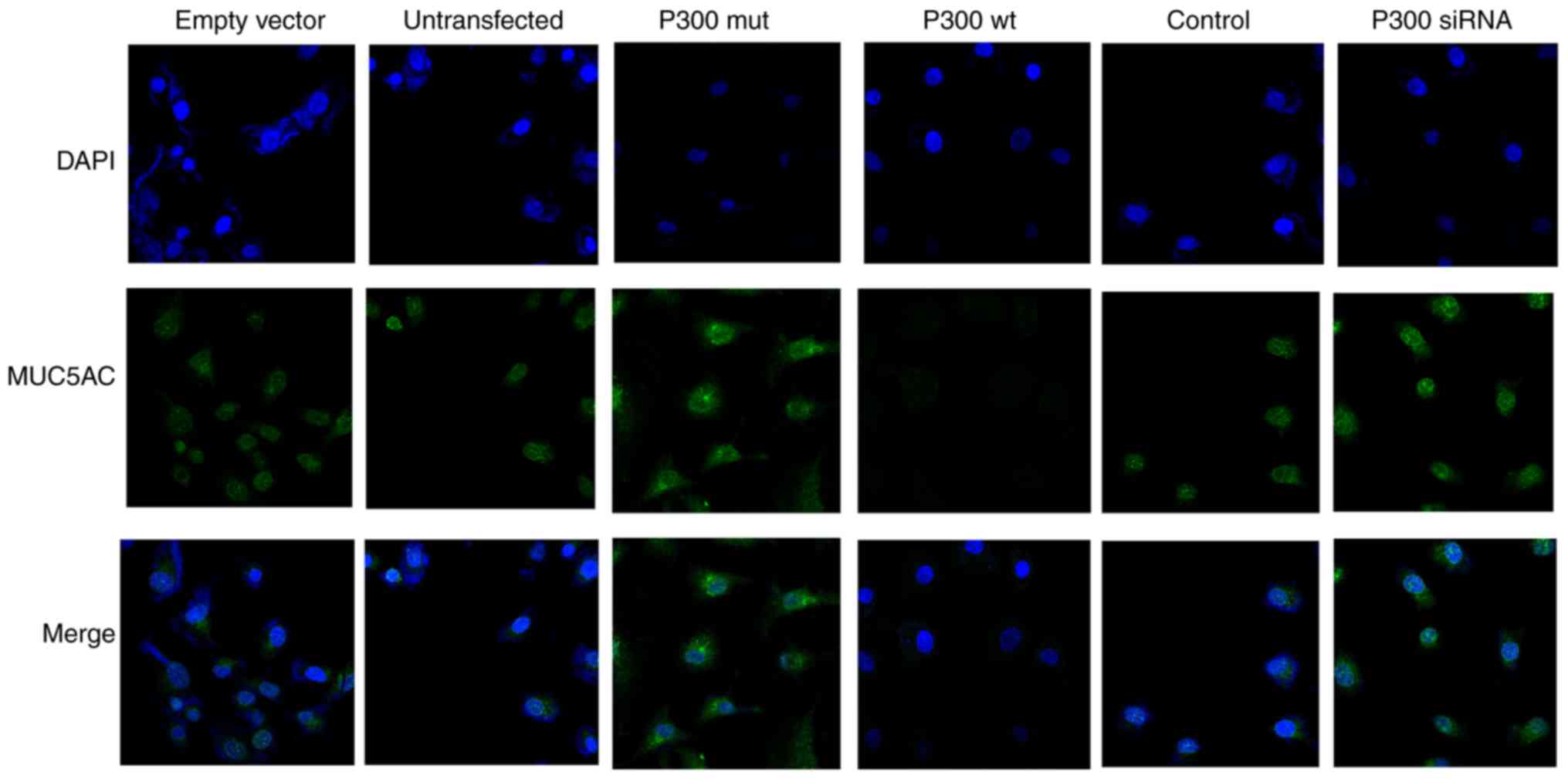
P300 decreases the protein expression
level of MUC5AC
The empty vector, untransfected control, the P300
expression plasmid and P300 siRNA, together with their control
plasmids, were transfected into A549 cells. Immunofluorescence was
then performed to detect the protein expression of MUC5AC in each
group. The A549 cells were stained with immunofluorescent MUC5AC
(green) antibody, and the nuclei were stained with DAPI (blue). The
results showed that the P300 expression plasmid downregulated the
protein levels of MUC5AC, whereas P300 siRNA upregulated the
protein levels of MUC5AC (Fig.
8).
Discussion
Mucosal airway hypersecretion is one of the most
serious pathophysiological features of chronic airway inflammatory
diseases. In cystic fibrosis, the thick and dehydrated airway
mucus, which is difficult to clear, makes patients prone to chronic
inflammation and bacterial infections. It is beneficial to reduce
the impact of lung infections and improve lung function by clearing
the airways of mucus (26).
Excessive mucus production and secretion in airways are associated
with disabling symptoms (cough and sputum), decline in lung
function, exacerbations and mortality in patients with chronic
obstructive pulmonary disease (COPD) (27). With an increasing number of
individuals suffering from mucosal airway hypersecretion, it is
becoming increasingly urgent to develop an effective treatment. It
is well known that mucus plugging is the main cause of acute asthma
(3). In patients with asthma,
bronchial secretion deposits are frequently observed (28). At present, anticholinergics,
corticosteroids or other medicines are applied to decrease mucus
hypersecretion in the clinical setting. However, these treatments
have a number of side effects. Therefore, attempts to identify
novel efficient therapeutic methods for airway inflammatory
diseases are ongoing (29).
Mucins are high-molecular-weight glycoproteins
secreted by goblet cells, which cover the airways, the esophagus
and other glandular organs (30).
At present, 21 mucin-like genes have been identified and sorted
into two categories; membrane-bound and secreted mucins (31). Secreted mucins have terminal
cysteine-rich domains and form disulfide bonds for polymers that
may be detected on gels (9,10,32).
MUC5AC and MUC5B are two typical polymers of airway
mucins (9,10). MUC5B, a tenacious secretory mucin,
is produced in the lung airway system in normal and pathological
conditions. MUC5AC is also a gel-forming mucin and secreted at high
levels in asthmatic inflammation or COPD. Increasing evidence has
demonstrated that MUC5AC may have a less important role in human
airway systems in normal conditions. Therefore, suppression of the
production of MUC5AC may be a therapeutic method for secretory
chronic airway inflammatory diseases (4). In our previous studies, mucosal
airway hypersecretion was investigated in A549 lung adenocarcinoma
cells, human NCI-H292 airway epithelial cells and normal human
bronchial epithelial (NHBE) cells (33-35). In the present study, A549 alveolar
only basal epithelial cells were used to investigate mucosal airway
hypersecretion, owing to their high stability, medium culture
conditions and low cost. Furthermore, this cell line has been
investigated widely and it is known to express MUC5AC, MUC1 and
MUC5B (36).
Histone acetylation, particularly by HATs, is
essential for gene transcription and protein expression in
asthmatic diseases. The decreased activity of HDACs is likely to
cause increased secretion of inflammatory factors, thus inducing
asthma (37). P300 has HAT
activity, which can acetylate a number of proteins that regulate
lung function and glandular secretion. Additionally, P300 exerts
its effect through its binding domains with different proteins.
P300 is able to interact with >411 proteins, to induce a signal
response or influence gene expression. In addition, P300 and other
acetyltransferases can influence protein-coupled structures by
protein lysine acetylation. P300 is involved in the integration of
protein complexes and regulating DNA element functions (3).
In the present study, the MUC5AC gene promoter was
cloned and a luciferase reporter with different lengths was
successfully constructed. The results demonstrated that the core
promoter area was in the region of -935 to -583 bp upstream of the
MUC5AC gene. In addition, the expression of MUC5AC was reduced by
P300. Through a literature review and analyses using relevant
software, it was demonstrated that the region of the core promoter
contained nuclear factor (NF)-κB, and transcription factor Sp1
(Sp1) binding sites. Each binding site can bind with different
transcription factors, which influence MUC5AC gene transcription
(38). There are several studies
showing that Sp1 and NF-κb (39,40) are important ubiquitous
transcription factors, which regulate the transcription of MUC5AC
and mucous metaplasia by interacting with gene promoters (19). Furthermore, studies have shown
that glucocorticoids can influence gene expression and activity by
integrating with the ligand-activated glucocorticoid receptor and
activating glucocorticoid-responsive elements (GREs) in the
promoter region. The MUC5AC gene also has GRE regions in its 5′
sequence and may be similarly influenced by glucocorticoids. All
signals influencing P300 (activity, structure or recruitment), may
have an impact on gene transcription to further affect the gene
expression of MUC5AC. P300 is ubiquitously expressed in multiple
tissues/organs and is involved in numerous physiological processes.
Studies on P300 have mainly focused on viral infections, cancer and
neurodegenerative diseases (41).
Consistent with these diseases, P300 is important in mucosal airway
hypersecretion. Differences in the mechanisms of P300 in these
diseases remain to be elucidated. Therefore, the task of
investigating the clinical applicability of targeting the
expression of P300 as a potential treatment for mucosal airway
hypersecretion involves understanding how the protein receives
signals in cells, what induces its recruitment in a given signal
transduction pathway, and what determines the final outcome of its
individual activity. Therefore, further investigations are required
focusing on the pathways that regulate the P300 pathway, and
investigating the effects of MUC5AC in lung inflammatory secretion
diseases.
In conclusion, the findings of the present study
indicate that P300 inhibited the gene expression of MUC5AC in A549
cells, which may be a novel therapeutic target for chronic airway
inflammatory diseases. However, further investigations are required
to identify the signal pathways influencing P300.
Funding
The present study was supported by the Medical
Innovation Team of Jiangsu Province (grant no. CXTDB2017016), the
Major Program of Wuxi Health and Family Planning Commission (grant
no. Z201606) and the General Program of Wuxi Municipal Health
Bureau (grant no. MS201506).
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
SX carried out the experiments, collected and
analyzed the data and wrote the manuscript. YH designed the
experimental study and assisted with revising the manuscript. JS
was involved in experiments and analysis. LL and JQ evaluated the
data, and revised and edited the manuscript. All authors read and
approved the final manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
Acknowledgments
The authors would like to thank Professor Joan Boyes
(The Institute of Cancer Research, London, UK) for providing the
P300 wt plasmid and P300 mut plasmid (P300mut-P300Δ1472-1522), and
Professor Zhou Guoping (The Fourth School of Clinical Medicine,
Nanjing Medical University) for providing the A549 cells.
References
|
1
|
Masoli M, Fabian D, Holt S, Beasley R and
Global Initiative; for Asthma (GINA) Program: The global burden of
asthma: Executive summary of the GINA dissemination committee
report. Allergy. 59:469–478. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
D'Amato G, Holgate ST, Pawankar R, Ledford
DK, Cecchi L, Al-Ahmad M, Al-Enezi F, Al-Muhsen S, Ansotegui I,
Baena-Cagnani CE, et al: Meteorological conditions, climate change,
new emerging factors, and asthma and related allergic disorders. A
statement of the world allergy organization. World Allergy Organ J.
8:252015.PubMed/NCBI
|
|
3
|
Kuyper LM, Paré PD, Hogg JC, Lambert RK,
Ionescu D, Woods R and Bai TR: Characterization of airway plugging
in fatal asthma. Am J Med. 115:6–11. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Evans CM, Kim K, Tuvim MJ and Dickey BF:
Mucus hypersecretion in asthma: Causes and effects. Curr Opin Pulm
Med. 15:4–11. 2009. View Article : Google Scholar :
|
|
5
|
Bateman JR, Pavia D, Sheahan NF, Agnew JE
and Clarke SW: Impaired tracheobronchial clearance in patients with
mild stable asthma. Thorax. 38:463–467. 1983. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Agnew JE, Bateman JR, Pavia D and Clarke
SW: Radionuclide demonstration of ventilatory abnormalities in mild
asthma. Clin Sci (Lond). 66:525–531. 1984. View Article : Google Scholar
|
|
7
|
O'Riordan TG, Zwang J and Smaldone GC:
Mucociliary clearance in adult asthma. Am Rev Respir Dis.
146:598–603. 1992. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Messina MS, O'riordan TG and Smaldone GC:
Changes in mucociliary clearance during acute exacerbations of
asthma. Am Rev Respir Dis. 143:993–997. 1991. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Thornton DJ, Rousseau K and McGuckin MA:
Structure and function of the polymeric mucins in airways mucus.
Annu Rev Physiol. 70:459–486. 2008. View Article : Google Scholar
|
|
10
|
Rose MC and Voynow JA: Respiratory tract
mucin genes and mucin glycoproteins in health and disease. Physiol
Rev. 86:245–278. 2006. View Article : Google Scholar
|
|
11
|
Bonser LR and Erle DJ: Airway mucus and
asthma: The role of MUC5AC and MUC5B. J Clin Med. 6:E1122017.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Jeffery PK and Li D: Airway mucosa:
Secretory cells, mucus and mucin genes. Eur Respir J. 10:1655–1662.
1997. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Woodruff PG, Modrek B, Choy DF, Jia G,
Abbas AR, Ellwanger A, Koth LL, Arron JR and Fahy JV: T-helper type
2-driven inflammation defines major subphenotypes of asthma. Am J
Respir Crit Care Med. 180:388–395. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Bonser LR, Zlock L, Finkbeiner W and Erle
DJ: Epithelial tethering of MUC5AC-rich mucus impairs mucociliary
transport in asthma. J Clin Invest. 126:2367–2371. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Kuperman DA, Lewis CC, Woodruff PG,
Rodriguez MW, Yang YH, Dolganov GM, Fahy JV and Erle DJ: Dissecting
asthma using focused transgenic modeling and functional genomics. J
Allergy Clin Immunol. 116:305–311. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Zhen G, Park SW, Nguyenvu LT, Rodriguez
MW, Barbeau R, Paquet AC and Erle DJ: IL-13 and epidermal growth
factor receptor have critical but distinct roles in epithelial cell
mucin production. Am J Respir Cell Mol Biol. 36:244–253. 2007.
View Article : Google Scholar
|
|
17
|
Takeyama K, Dabbagh K, Lee HM, Agustí C,
Lausier JA, Ueki IF, Grattan KM and Nadel JA: Epidermal growth
factor system regulates mucin production in airways. Proc Natl Acad
Sci USA. 96:3081–3086. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Kraft M, Adler KB, Ingram JL, Crews AL,
Atkinson TP, Cairns CB, Krause DC and Chu HW: Mycoplasma pneumoniae
induces airway epithelial cell expression of MUC5AC in asthma. Eur
Respir J. 31:43–46. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Chen Y, Nickola TJ, DiFronzo NL,
Colberg-Poley AM and Rose MC: Dexamethasone-mediated repression of
MUC5AC gene expression in human lung epithelial cells. Am J Respir
Cell Mol Biol. 34:338–347. 2006. View Article : Google Scholar
|
|
20
|
Bannister AJ and Kouzarides T: The CBP
co-activator is a histone acetyltransferase. Nature. 384:641–643.
1996. View
Article : Google Scholar : PubMed/NCBI
|
|
21
|
Ogryzko VV, Schiltz RL, Russanova V,
Howard BH and Nakatani Y: The transcriptional coactivators P300 and
CBP are histone acetyltransferases. Cell. 87:953–959. 1996.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Ito K, Caramori G, Lim S, Oates T, Chung
KF, Barnes PJ and Adcock IM: Expression and activity of histone
deacetylases (HDAC) in human asthmatic airways. Am J Respir Crit
Care Med. 166:392–396. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Dancy BM and Cole PA: Protein lysine
acetylation by P300/CBP. Chem Rev. 115:2419–2452. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Boyes J, Byfield P, Nakatani Y and Ogryzko
V: Regulation of activity of the transcription factor GATA-1 by
acetylation. Nature. 396:594–598. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(−Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar
|
|
26
|
Martin C, Frija-Masson J and Burgel PR:
Targeting mucus hyper-secretion: New therapeutic opportunities for
COPD? Drugs. 74:1073–1089. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Button BM and Button B: Structure and
function of the mucus clearance system of the lung. Cold Spring
Harb Perspect Med. 3:a0097202013. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Dunnill MS: The pathology of asthma, with
special reference to changes in the bronchial mucosa. J Clin
Pathol. 13:27–33. 1960. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Rogers DF and Barnes PJ: Treatment of
airway mucus hypersecretion. Ann Med. 38:116–125. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Di Valentin E, Crahay C, Garbacki N,
Hennuy B, Guéders M, Noël A, Foidart JM, Grooten J, Colige A,
Piette J and Cataldo D: New asthma biomarkers: Lessons from murine
models of acute and chronic asthma. Am J Physiol Lung Cell Mol
Physiol. 296:L185–L197. 2009. View Article : Google Scholar
|
|
31
|
Yu H, Li Q, Kolosov VP, Perelman JM and
Zhou X: Interlenkin-13 induces mucin 5AC production involving
STAT6/SPDEF in human airway epithelial cells. Cell Commun Adhes.
17:83–92. 2010. View Article : Google Scholar
|
|
32
|
Hattrup CL and Gendler SJ: Structure and
function of the cell surface (tethered) mucins. Annu Rev Physiol.
70:431–457. 2008. View Article : Google Scholar
|
|
33
|
Lee JW, Kim YI, Im CN, Kim SW, Kim SJ, Min
S, Joo YH, Yim SV and Chung N: Grape seed proanthocyanidin inhibits
mucin synthesis and viral replication by suppression of AP-1 and
NF-κB via p38 MAPKs/JNK signaling pathways in respiratory syncytial
virus-infected A549 cells. J Agric Food Chem. 65:4472–4483. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Al-Sawalha N, Pokkunuri I, Omoluabi O, Kim
H, Thanawala VJ, Hernandez A, Bond RA and Knoll BJ: Epinephrine
activation of the β2-adrenoceptor is required for IL-13-induced
mucin production in human bronchial epithelial cells. PLoS One.
10:e01325592015. View Article : Google Scholar
|
|
35
|
Song WY, Song YS, Ryu HW, Oh SR, Hong J
and Yoon DY: Tilianin inhibits MUC5AC expression mediated via
down-regulation of EGFR-MEK-ERK-Sp1 signaling pathway in NCI-H292
human airway cells. J Microbiol Biotechnol. 27:49–56. 2017.
View Article : Google Scholar
|
|
36
|
Berger JT, Horger T and Voynow JA:
Respiratory carcinoma cell lines express MUC5/5AC mRNA and secrete
MUC5/5AC mucins(C). In: 5th International Workshop on Carcinoma
Associated Mucins; Cambridge, UK. 1998;
|
|
37
|
Cui ZL, Gu W, Ding T, Peng XH, Chen X,
Luan CY, Han RC, Xu WG and Guo XJ: Histone modifications of Notch1
promoter affect lung CD4+T cell differentiation in
asthmatic rats. Int J Immunopathol Pharmacol. 26:371–381. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Hewson CA, Edbrooke MR and Johnston SL:
PMA induces the MUC5AC respiratory mucin in human bronchial
epithelial cells, via PKC, EGF/TGF-alpha, Ras/Raf, MEK, ERK and
Sp1-dependent mechanisms. J Mol Biol. 344:683–695. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Perrais M, Pigny P, Copin MC, Aubert JP
and Van Seuningen I: Induction of MUC2 and MUC5AC mucins by factors
of the epidermal growth factor (EGF) family is mediated by EGF
receptor/Ras/Raf/extracellular signal-regulated kinase cascade and
Sp1. J Biol Chem. 277:32258–32267. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Chen Y, Garvin LM, Nickola TJ, Watson AM,
Colberg-Poley AM and Rose MC: IL-1β induction of MUC5AC gene
expression is mediated by CREB and NF-κB and repressed by
dexamethasone. Am J Physiol Lung Cell Mol Physiol. 306:L797–L807.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Janknecht R: The versatile functions of
the transcriptional coactivators P300 and CBP and their roles in
disease. Histol Histopathol. 17:657–668. 2002.PubMed/NCBI
|