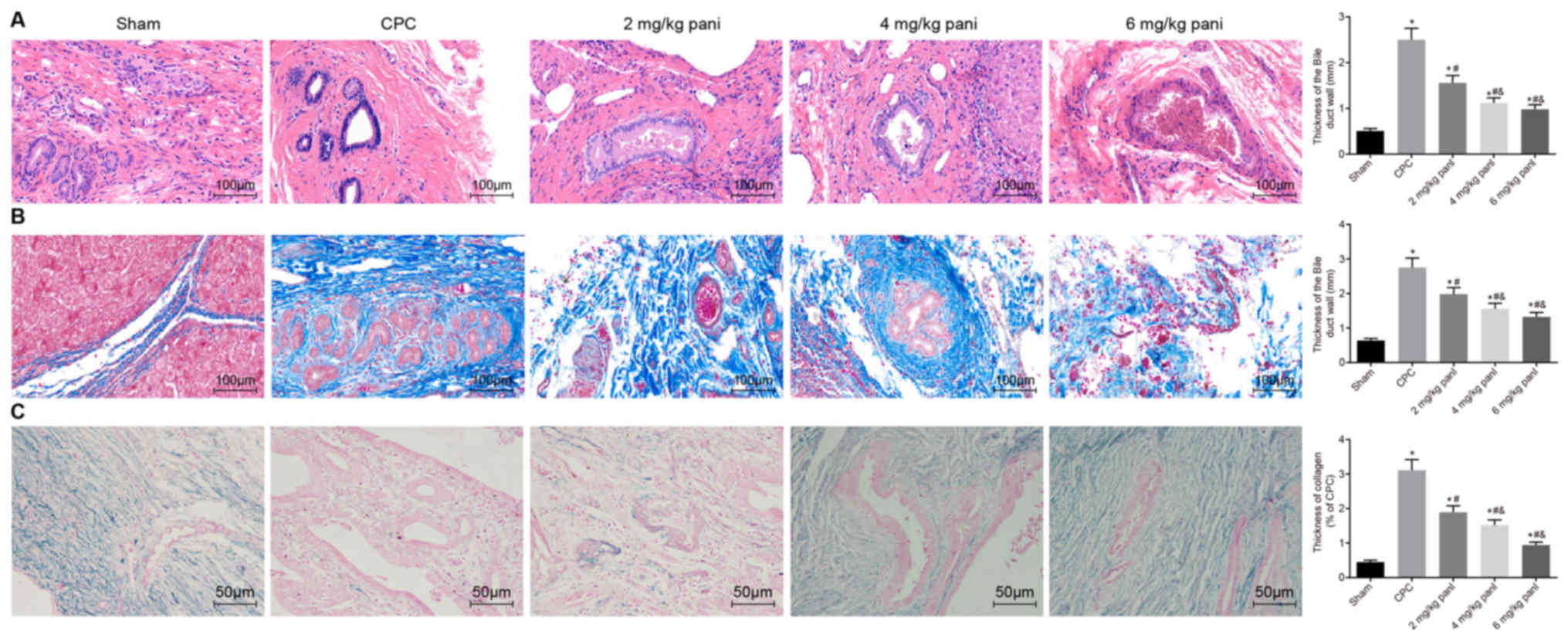
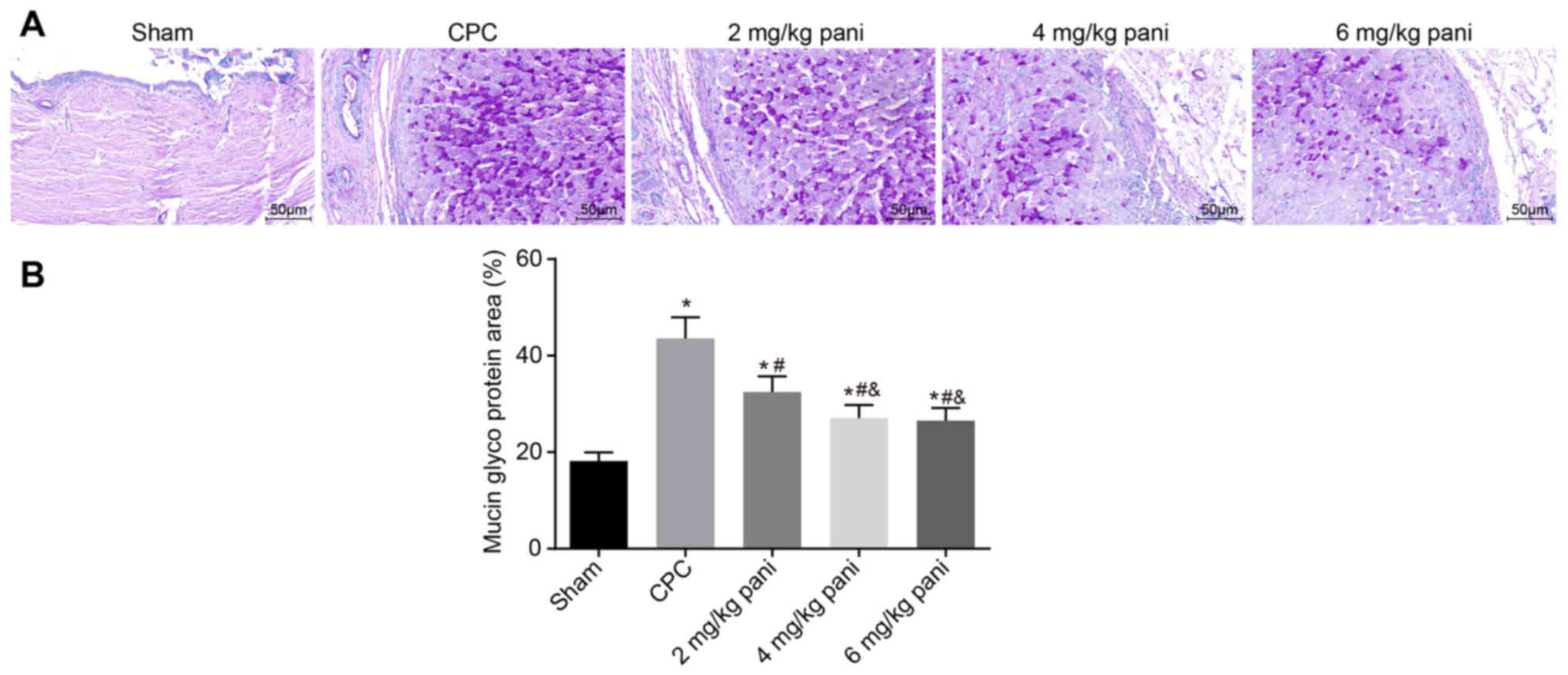
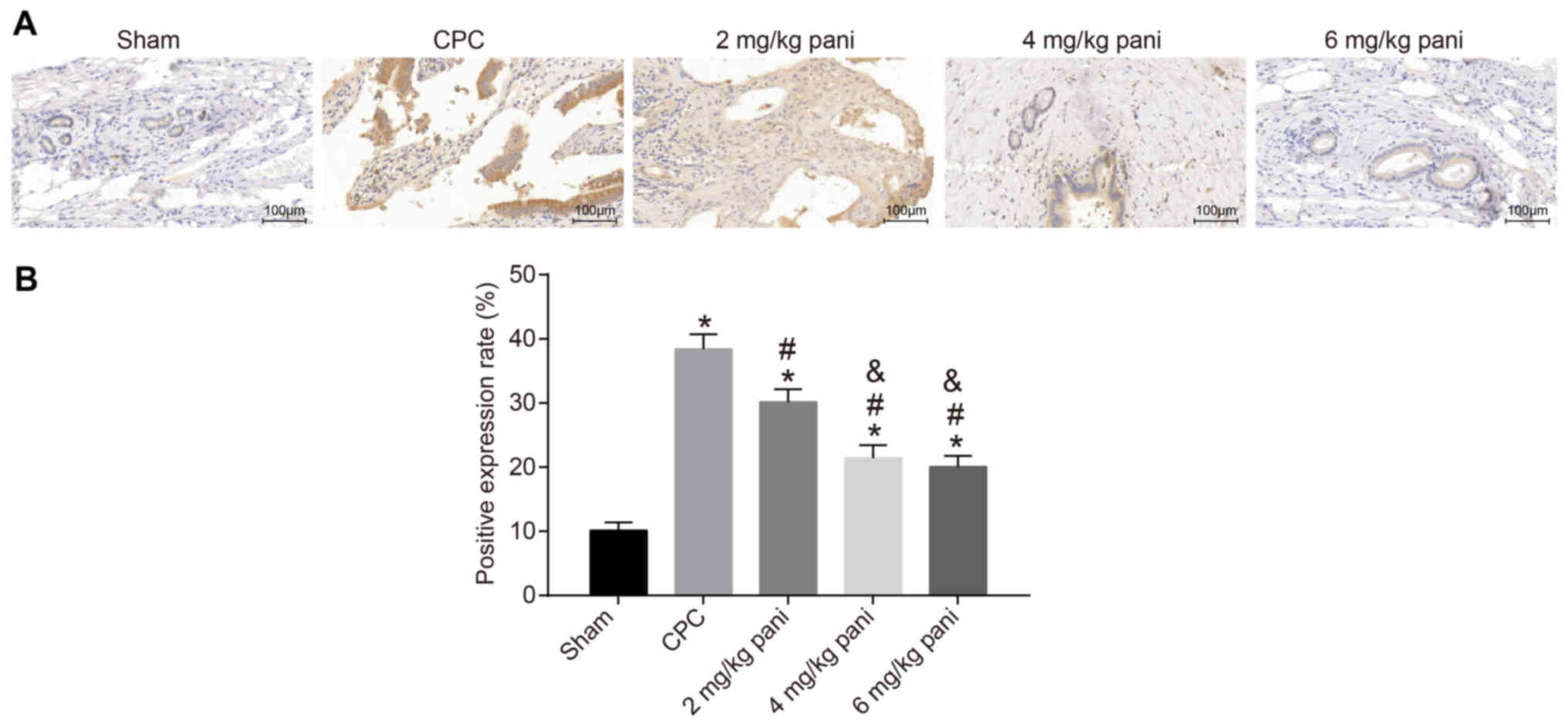
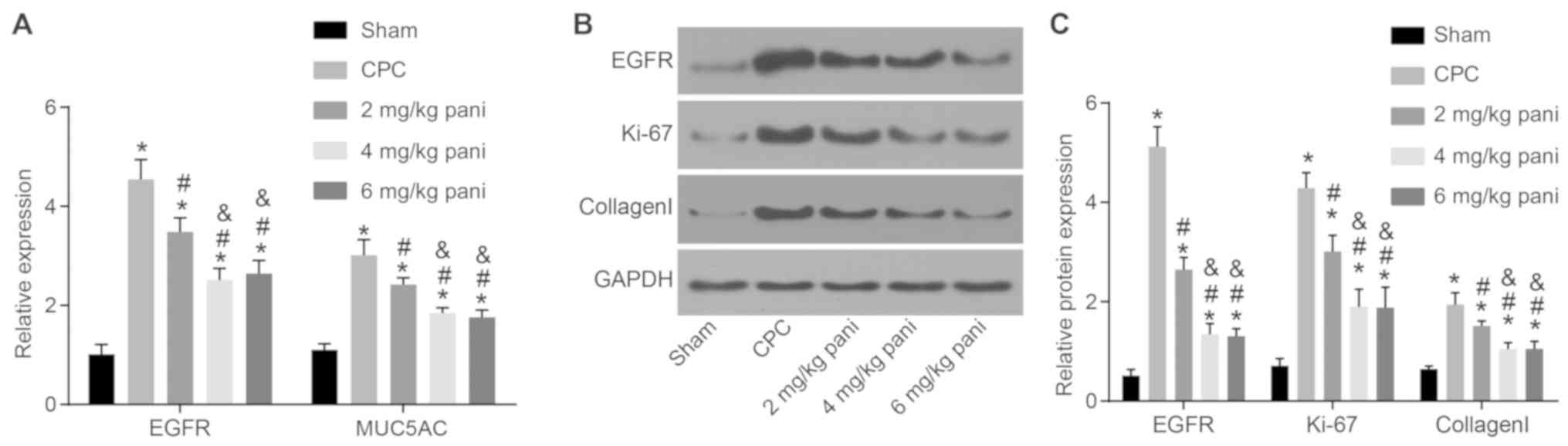
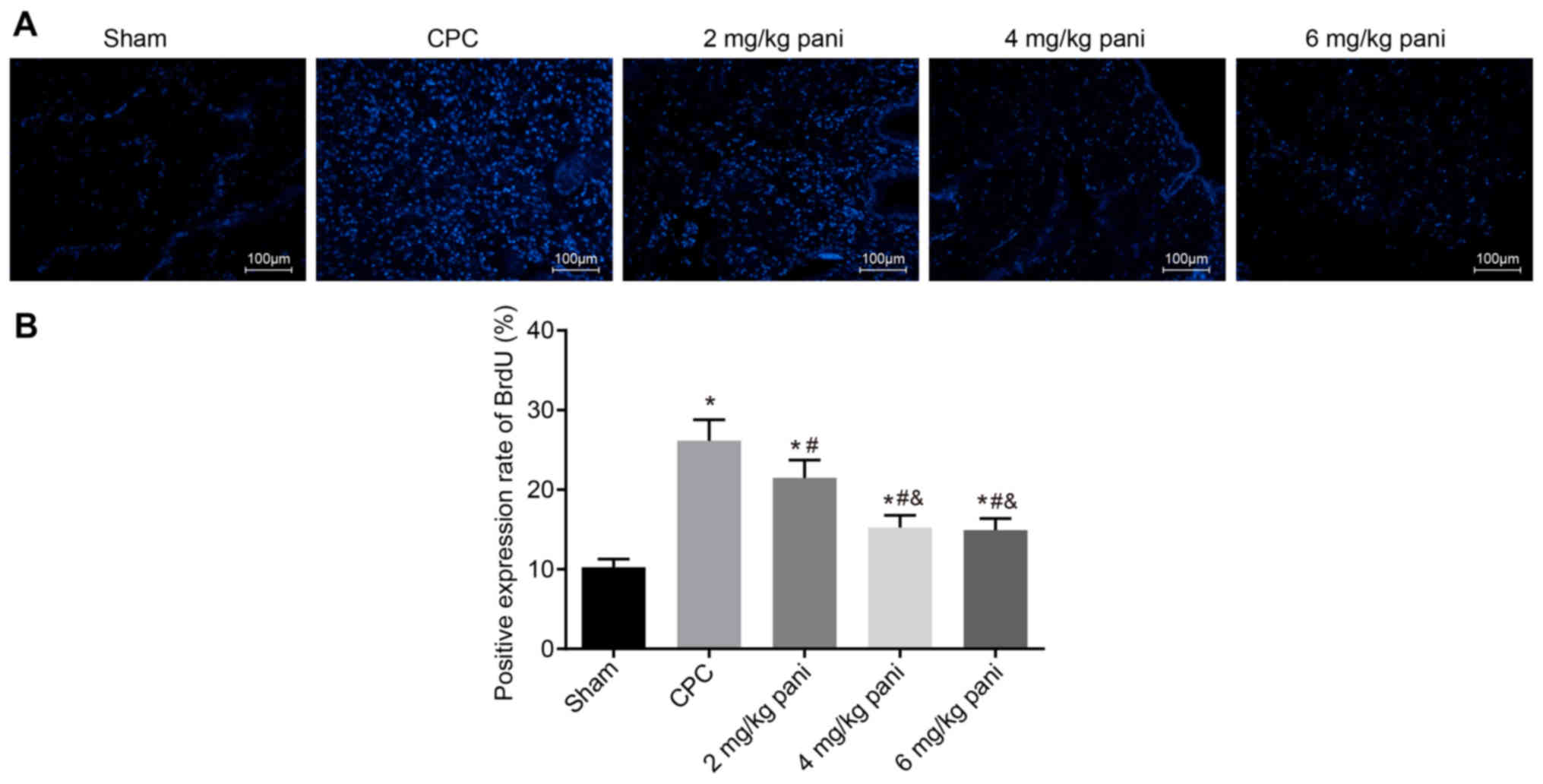
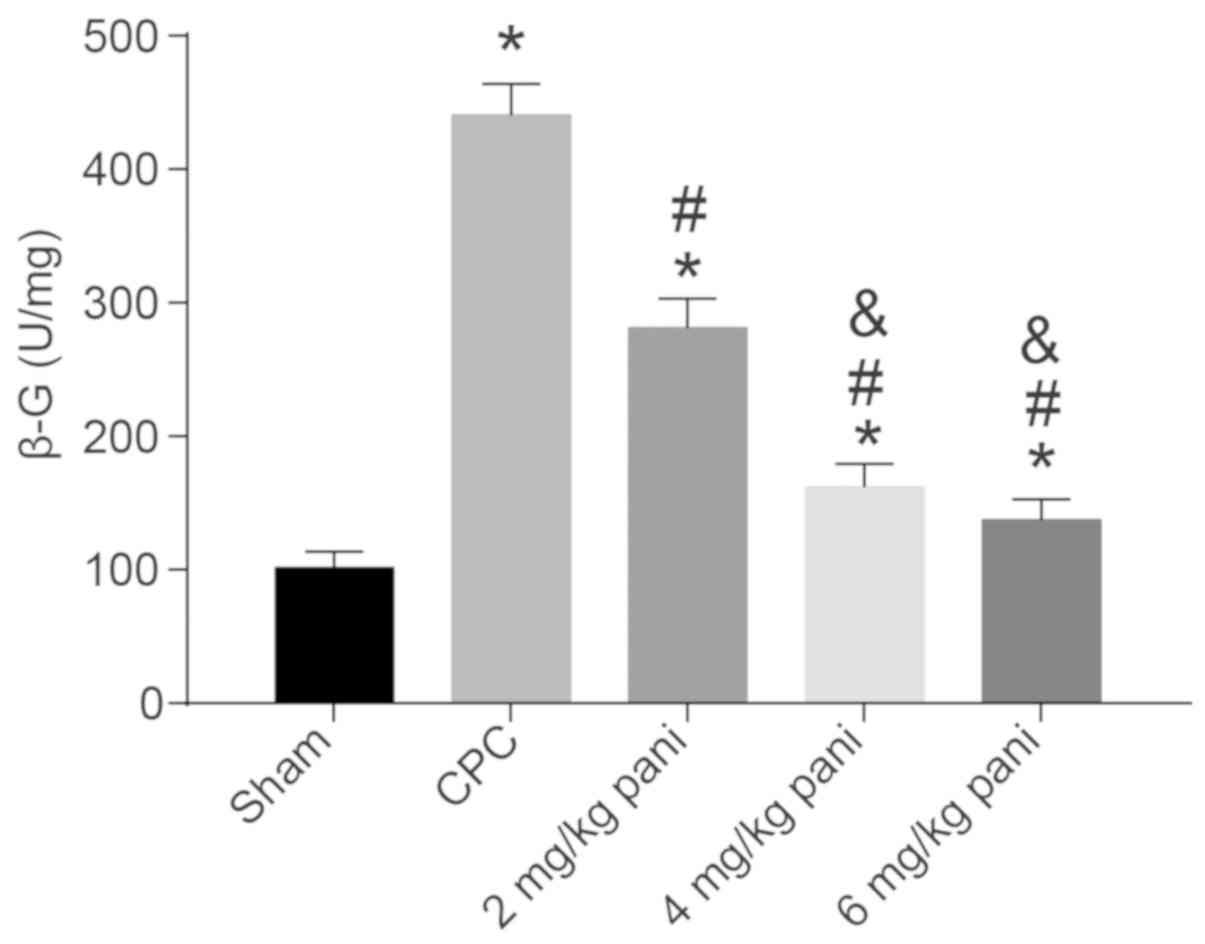
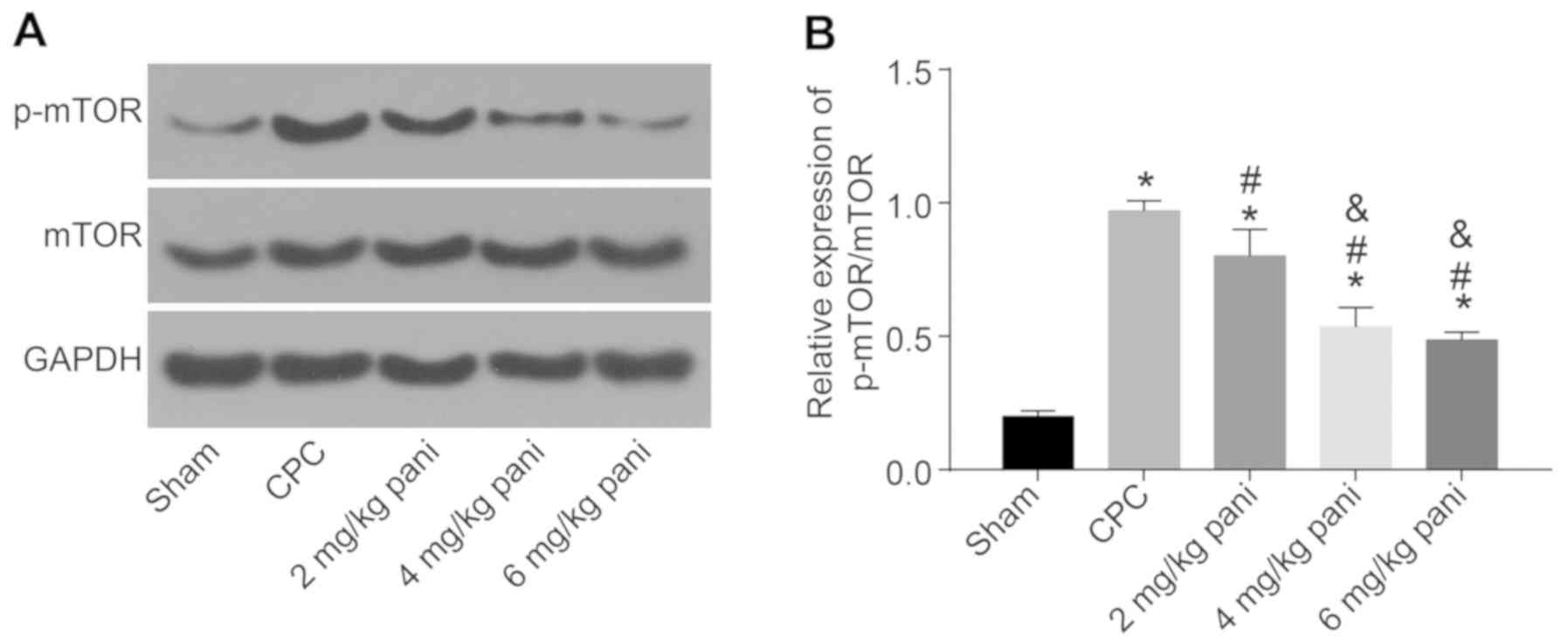
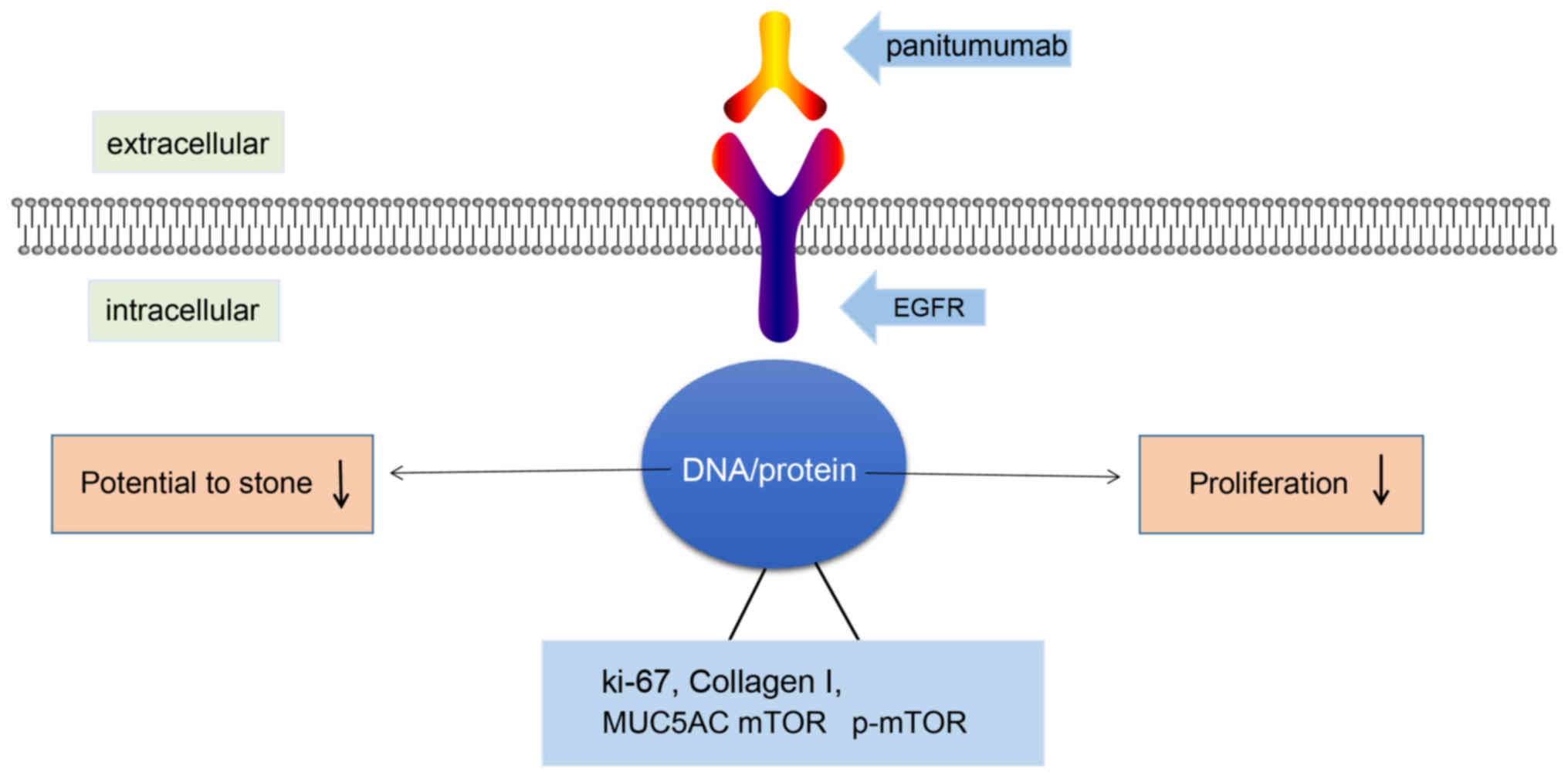
|
1
|
Jiang L, Jiang LS, Yan LN, Li FY, Wang W,
Cheng NS and Wen TF: Effects of epidermal growth factor receptor
inhibitor genistein on proliferative cholangitis in rats. J Surg
Res. 162:59–67. 2010. View Article : Google Scholar
|
|
2
|
Li FY, Cheng NS, Cheng JQ, Mao H, Zhou Y,
Jiang LS and Li N: Proliferating cell nuclear antigen shRNA
treatment attenuates chronic proliferative cholangitis in rats. J
Gastroenterol Hepatol. 24:920–926. 2009. View Article : Google Scholar
|
|
3
|
Li FY, Cheng NS, Mao H, Jiang LS, Cheng
JQ, Li QS and Munireddy S: Significance of controlling chronic
proliferative cholangitis in the treatment of hepatolithiasis.
World J Surg. 33:2155–2160. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Asano T, Shoda J, Ueda T, Maruyama T,
Kawamoto T, Sugimoto Y, Ichikawa A and Tanaka N: Involvement of
prostaglandin E2 and its specific receptor subtype EP4 in chronic
proliferative cholangitis in the bile ducts of patients with
hepatolithiasis. Gastroenterology. 124:A246. 2003. View Article : Google Scholar
|
|
5
|
Koh YX, Chiow AK, Chok AY, Lee LS, Tan SS
and Ibrahim S: Recurrent pyogenic cholangitis: Disease
characteristics and patterns of recurrence. ISRN Surg.
2013:5360812013. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Li F, Cheng J, He S, Li N, Zhang M, Dong
J, Jiang L, Cheng N and Xiong X: The practical value of applying
chemical biliary duct embolization to chemical hepatectomy for
treatment of hepatolithiasis. J Surg Res. 127:131–138. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Okines AF, Ashley SE, Cunningham D, Oates
J, Turner A, Webb J, Saffery C, Chua YJ and Chau I: Epirubicin,
oxaliplatin, and capecitabine with or without panitumumab for
advanced esophagogastric cancer: Dose-finding study for the
prospective multicenter, randomized, phase II/III REAL-3 trial. J
Clin Oncol. 28:3945–3950. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Di Nicolantonio F, Martini M, Molinari F,
Sartore-Bianchi A, Arena S, Saletti P, De Dosso S, Mazzucchelli L,
Frattini M, Siena S and Bardelli A: Wild-type BRAF is required for
response to panitumumab or cetuximab in metastatic colorectal
cancer. J Clin Oncol. 26:5705–5712. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Peeters M, Price TJ, Cervantes A, Sobrero
AF, Ducreux M, Hotko Y, Andre T, Chan E, Lordick F, Punt CJ, et al:
Randomized phase III study of panitumumab with fluorouracil,
leucovorin, and irinotecan (FOLFIRI) compared with FOLFIRI alone as
second-line treatment in patients with metastatic colorectal
cancer. J Clin Oncol. 28:4706–4713. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Cohenuram M and Saif MW: Panitumumab the
first fully human monoclonal antibody: From the bench to the
clinic. Anticancer Drugs. 18:7–15. 2007. View Article : Google Scholar
|
|
11
|
Keating GM: Panitumumab: A review of its
use in metastatic colorectal cancer. Drugs. 70:1059–1078. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Lacouture ME, Mitchell EP, Piperdi B,
Pillai MV, Shearer H, Iannotti N, Xu F and Yassine M: Skin toxicity
evaluation protocol with panitumumab (STEPP), a phase II,
open-label, randomized trial evaluating the impact of a pre-Emptive
Skin treatment regimen on skin toxicities and quality of life in
patients with metastatic colorectal cancer. J Clin Oncol.
28:1351–1357. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Hecht JR, Patnaik A, Berlin J, Venook A,
Malik I, Tchekmedyian S, Navale L, Amado RG and Meropol NJ:
Panitumumab monotherapy in patients with previously treated
metastatic colorectal cancer. Cancer. 110:980–988. 2007. View Article : Google Scholar
|
|
14
|
Li F, Zhou Y, Cheng N, Mao H, Jiang L, Li
N, Li Q, de Jong MC and Pawlik TM: Epidermal growth factor receptor
as a target for anti-proliferative treatment of proliferative
cholangitis in hepatolithiasis. J Surg Res. 166:87–94. 2011.
View Article : Google Scholar
|
|
15
|
Ji YY, Wang ZD, Wang SF, Wang BT, Yang ZA,
Zhou XR, Lei NN and Yue WN: Ischemic preconditioning ameliorates
intestinal injury induced by ischemia-reperfusion in rats. World J
Gastroenterol. 21:8081–8088. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar
|
|
17
|
Li N, Xiao LJ, Chen SW, Li L, Xiao BL,
Chen WB, Gao XK and Gu SJ: Mucus histochemical study of bilirubin
cholangiolithiasis in rabbit model. Hua Xi Yi Ke Da Xue Xue Bao.
20:417–420. 1989.In Chinese. PubMed/NCBI
|
|
18
|
Ho KJ, Lin XZ, Yu SC, Chen JS and Wu CZ:
Cholelithiasis in Taiwan. Gallstone characteristics, surgical
incidence, bile lipid composition, and role of beta-glucuronidase.
Dig Dis Sci. 40:1963–1973. 1995. View Article : Google Scholar
|
|
19
|
Li FY, Cheng NS, Cheng JQ, Mao H, Jiang
LS, Li QS and Zhou Y: Practical value of applying cdc2 kinase shRNA
to chronic proliferative cholangitis in treatment of
hepatolithiasis. Hepatogastroenterology. 56:1477–1482. 2009.
|
|
20
|
Li FY, Cheng NS, Cheng JQ, Mao H, Jiang
LS, Li N and He S: Treatment of chronic proliferative cholangitis
with c-myc shRNA. World J Gastroenterol. 15:95–101. 2009.
View Article : Google Scholar :
|
|
21
|
Leone F, Cavalloni G, Pignochino Y,
Sarotto I, Ferraris R, Piacibello W, Venesio T, Capussotti L, Risio
M and Aglietta M: Somatic mutations of epidermal growth factor
receptor in bile duct and gallbladder carcinoma. Clin Cancer Res.
12:1680–1685. 2006. View Article : Google Scholar
|
|
22
|
Miyata H, Sasaki T, Kuwahara K, Serikawa M
and Chayama K: The effects of ZD1839 (Iressa), a highly selective
EGFR tyrosine kinase inhibitor, as a radiosensitiser in bile duct
carcinoma cell lines. Int J Oncol. 28:915–921. 2006.
|
|
23
|
Gajadhar AS, Bogdanovic E, Munoz DM and
Guha A: In situ analysis of mutant EGFRs prevalent in glioblastoma
multiforme reveals aberrant dimerization, activation, and
differential response to anti-EGFR targeted therapy. Mol Cancer
Res. 10:428–440. 2012. View Article : Google Scholar
|
|
24
|
Peeters M, Balfour J and Arnold D: Review
article: Panitumumab-a fully human anti-EGFR monoclonal antibody
for treatment of metastatic colorectal cancer. Aliment Pharmacol
Ther. 28:269–281. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Imano M, Satou T, Itoh T, Takeyama Y,
Yasuda A, Peng YF, Shinkai M, Haji S, Yasuda C, Nakai T, et al: An
immunohisto-chemical study of osteopontin in pigment gallstone
formation. Am Surg. 76:91–95. 2010.PubMed/NCBI
|
|
26
|
Kim S, Schein AJ and Nadel JA: E-cadherin
promotes EGFR-mediated cell differentiation and MUC5AC mucin
expression in cultured human airway epithelial cells. Am J Physiol
Lung Cell Mol Physiol. 289:L1049–L1060. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Barbier D, Garcia-Verdugo I, Pothlichet J,
Khazen R, Descamps D, Rousseau K, Thornton D, Si-Tahar M, Touqui L,
Chignard M and Sallenave JM: Influenza A induces the major secreted
airway mucin MUC5AC in a protease-EGFR-extracellular regulated
kinase-Sp1-dependent pathway. Am J Respir Cell Mol Biol.
47:149–157. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Shao MX and Nadel JA: Dual oxidase
1-dependent MUC5AC mucin expression in cultured human airway
epithelial cells. Proc Natl Acad Sci USA. 102:767–772. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Bullwinkel J, Baron-Luhr B, Ludemann A,
Wohlenberg C, Gerdes J and Scholzen T: Ki-67 protein is associated
with ribosomal RNA transcription in quiescent and proliferating
cells. J Cell Physiol. 206:624–635. 2006. View Article : Google Scholar
|
|
30
|
Salehi F, Agur A, Scheithauer BW, Kovacs
K, Lloyd RV and Cusimano M: Ki-67 in pituitary neoplasms: A
review-part I. Neurosurgery. 65:429–437. 2009. View Article : Google Scholar
|
|
31
|
Wang P, He Y, Ma X, Sun B, Huang B, Zhu C
and Liu Y: Expression and significance of COX-2 and Ki-67 in
hepatolithiasis with bile duct carcinoma. Med Sci Monit.
21:2943–2949. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Liang H, Li X, Chen B, Wang B, Zhao Y,
Zhuang Y, Shen H, Zhang Z and Dai J: A collagen-binding EGFR
single-chain Fv antibody fragment for the targeted cancer therapy.
J Control Release. 209:101–109. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Attallah AM, Mosa TE, Omran MM, Abo-Zeid
MM, El-Dosoky I and Shaker YM: Immunodetection of collagen types I,
II, III, and IV for differentiation of liver fibrosis stages in
patients with chronic HCV. J Immunoassay Immunochem. 28:155–168.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Wu D, Peng F, Zhang B, Ingram AJ, Kelly
DJ, Gilbert RE, Gao B, Kumar S and Krepinsky JC: EGFR-PLCgamma1
signaling mediates high glucose-induced PKCbeta1-Akt activation and
collagen I upregulation in mesangial cells. Am J Physiol Renal
Physiol. 297:F822–F834. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Su YC, Cheng TC, Leu YL, Roffler SR, Wang
JY, Chuang CH, Kao CH, Chen KC, Wang HE and Cheng TL: PET imaging
of beta-glucuronidase activity by an activity-based 124I-trapping
probe for the personalized glucuronide prodrug targeted therapy.
Mol Cancer Ther. 13:2852–2863. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Chen M, Cheng KW, Chen YJ, Wang CH, Cheng
TC, Chang KC, Kao AP and Chuang KH: Real-time imaging of intestinal
bacterial β-glucuronidase activity by hydrolysis of a fluorescent
probe. Sci Rep. 7:31422017. View Article : Google Scholar
|
|
37
|
Guertin DA and Sabatini DM: Defining the
role of mTOR in cancer. Cancer Cell. 12:9–22. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Feldman ME, Apsel B, Uotila A, Loewith R,
Knight ZA, Ruggero D and Shokat KM: Active-site inhibitors of mTOR
target rapamycin-resistant outputs of mTORC1 and mTORC2. PLoS Biol.
7:e382009. View Article : Google Scholar
|
|
39
|
Schmid K, Bago-Horvath Z, Berger W, Haitel
A, Cejka D, Werzowa J, Filipits M, Herberger B, Hayden H and
Sieghart W: Dual inhibition of EGFR and mTOR pathways in small cell
lung cancer. Br J Cancer. 103:622–628. 2010. View Article : Google Scholar
|