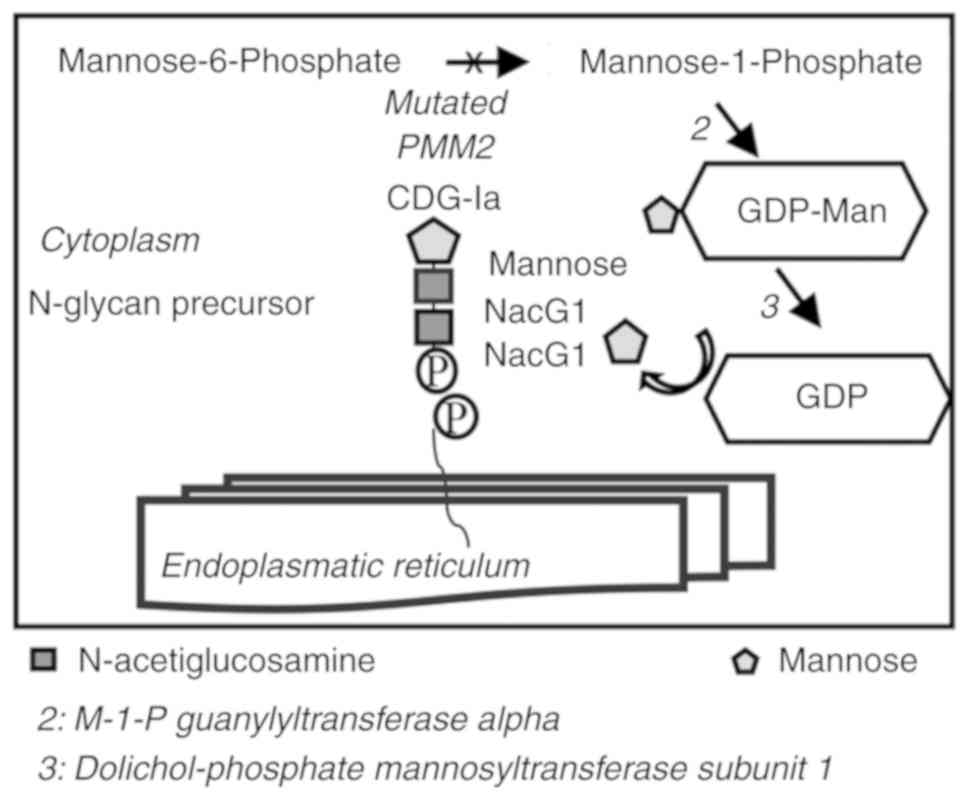
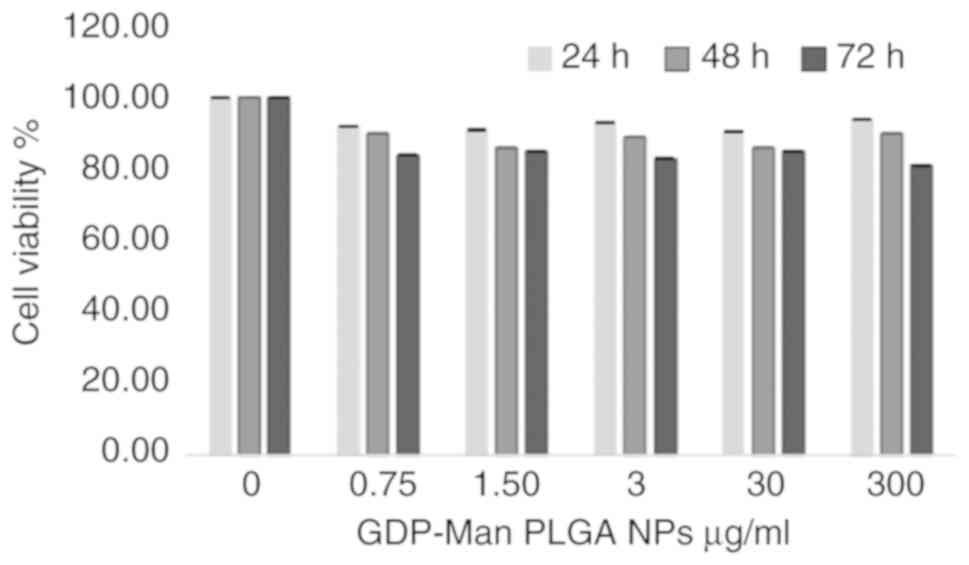
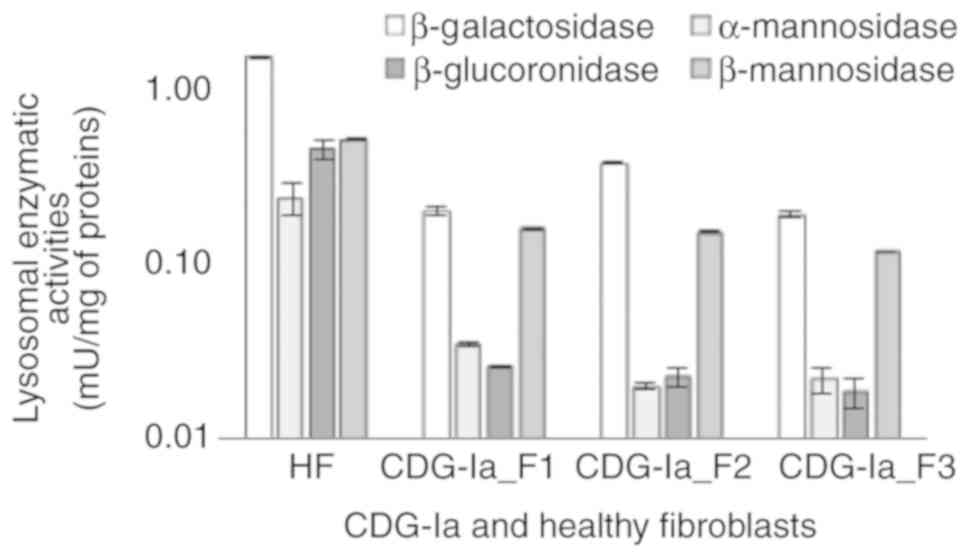
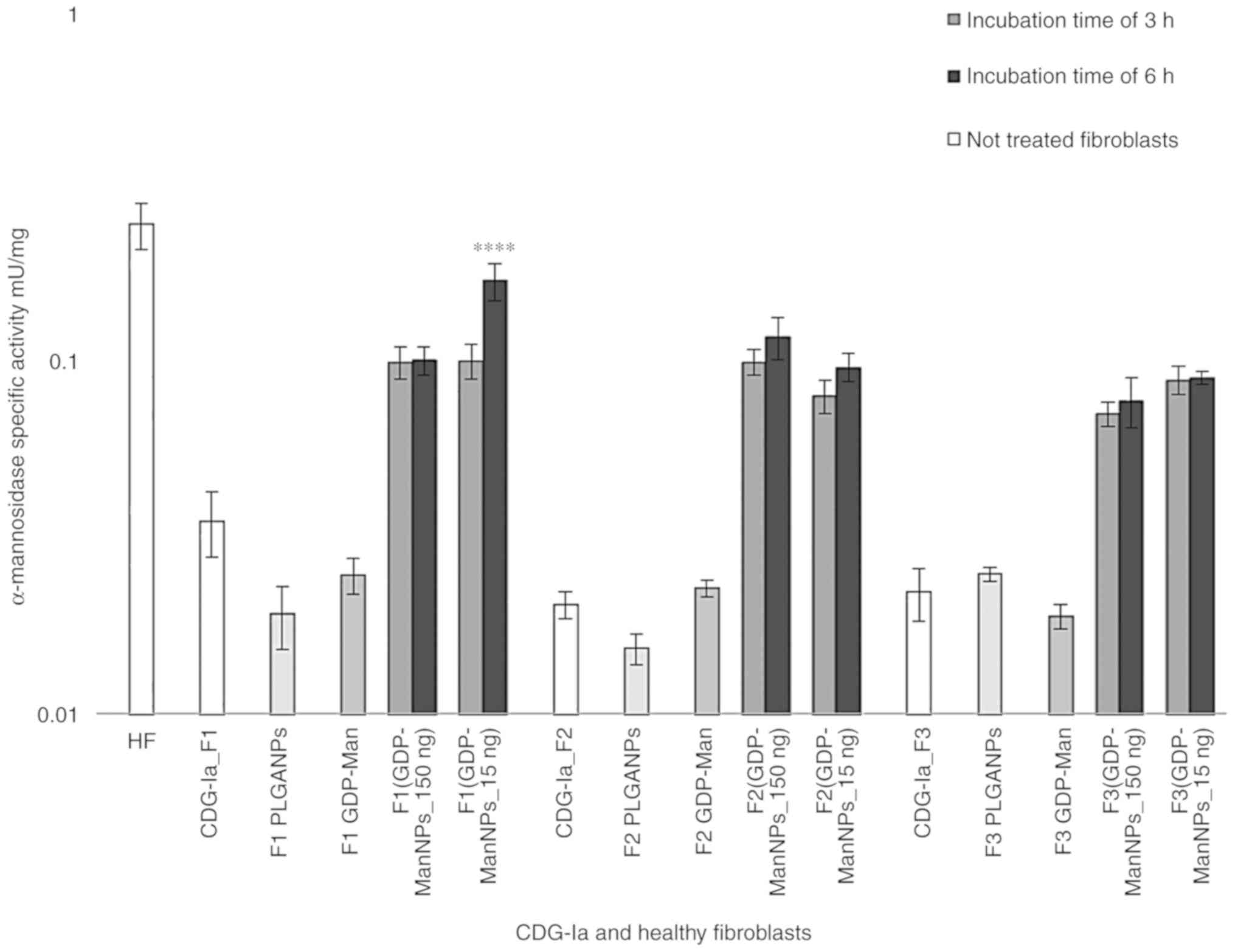
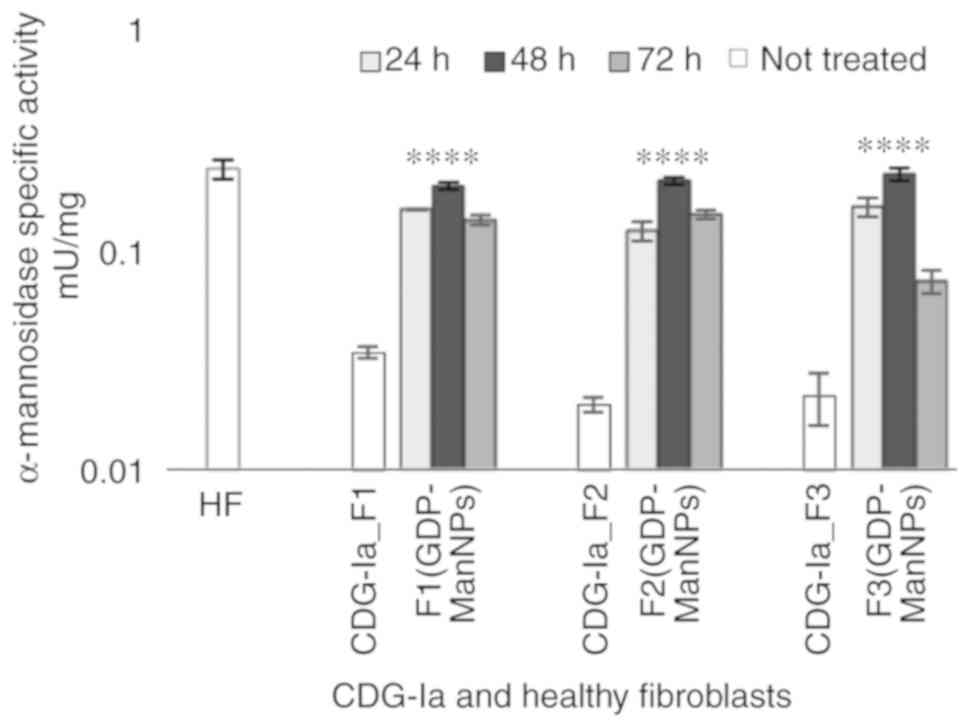
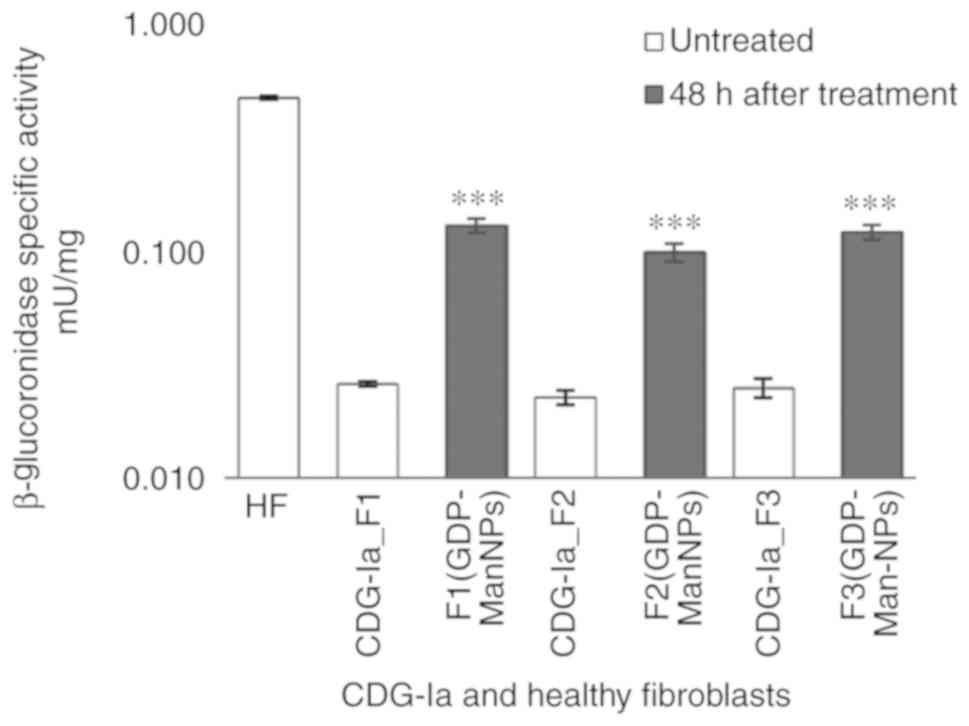
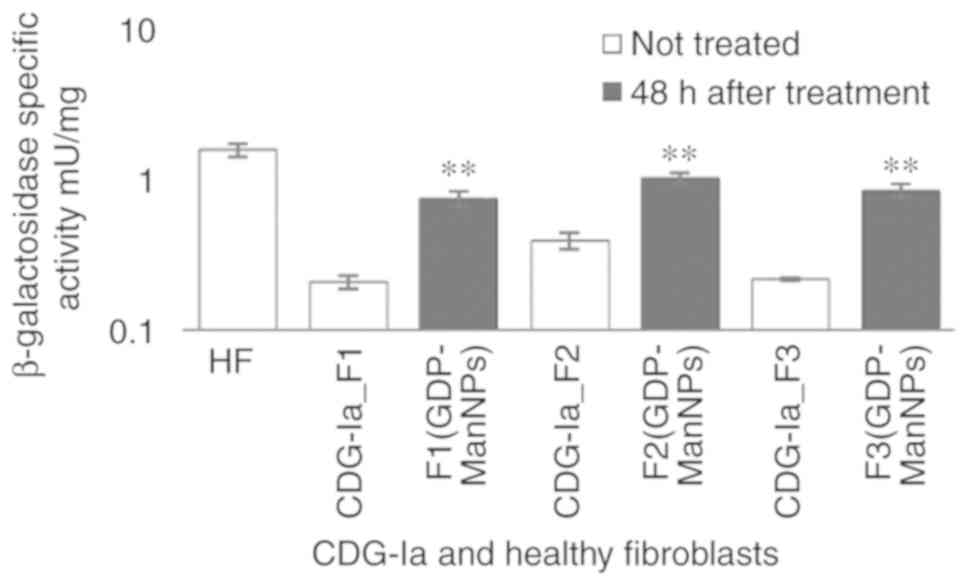
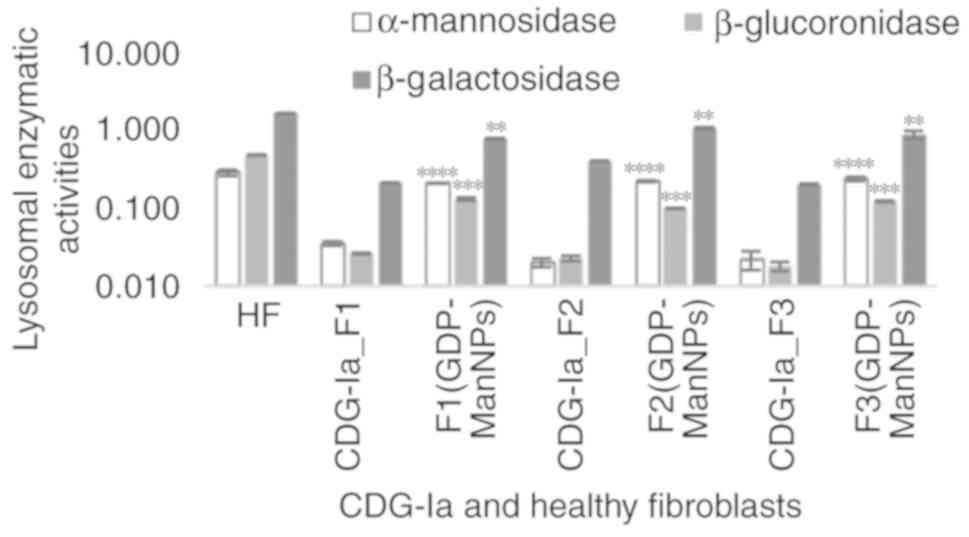
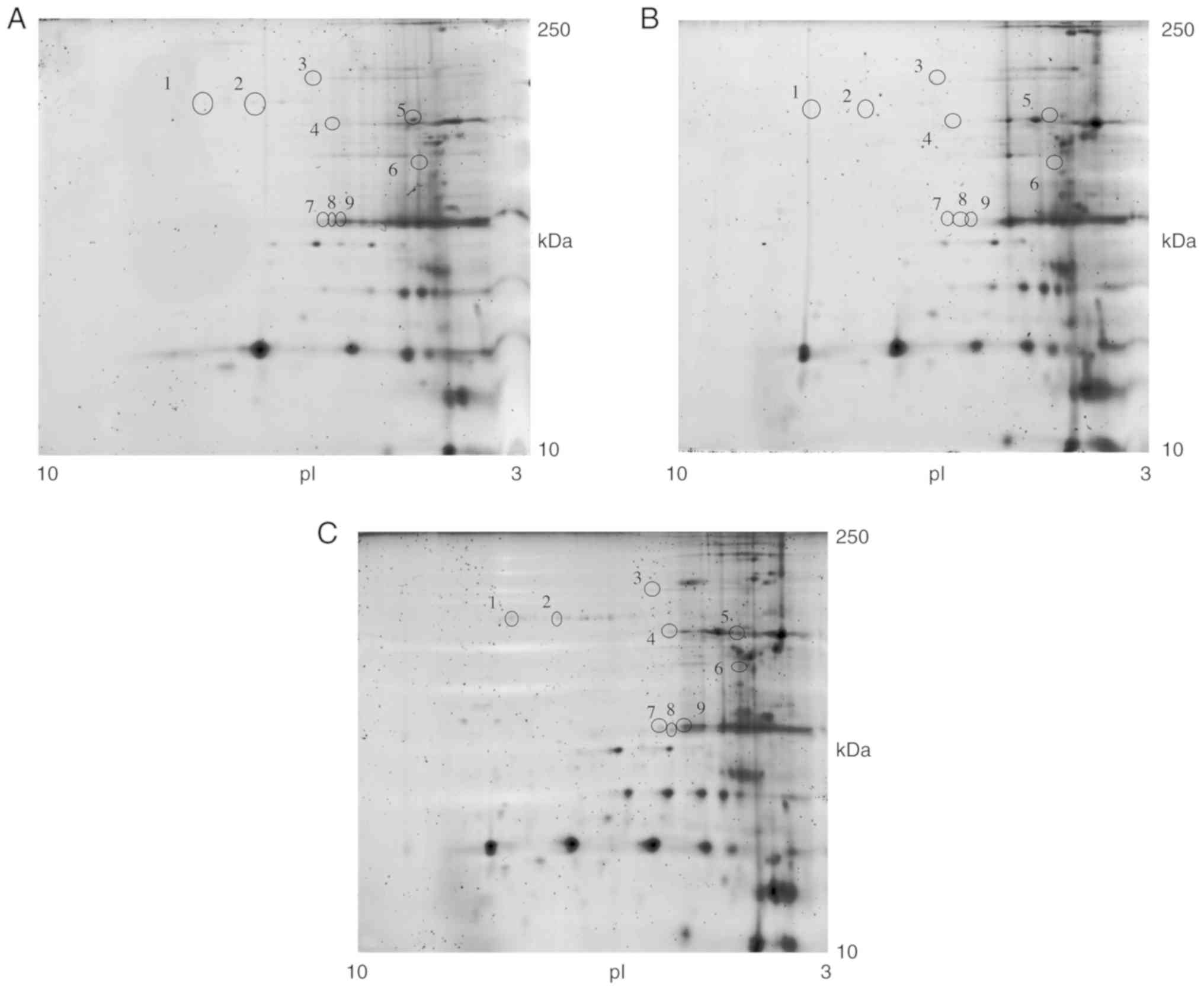
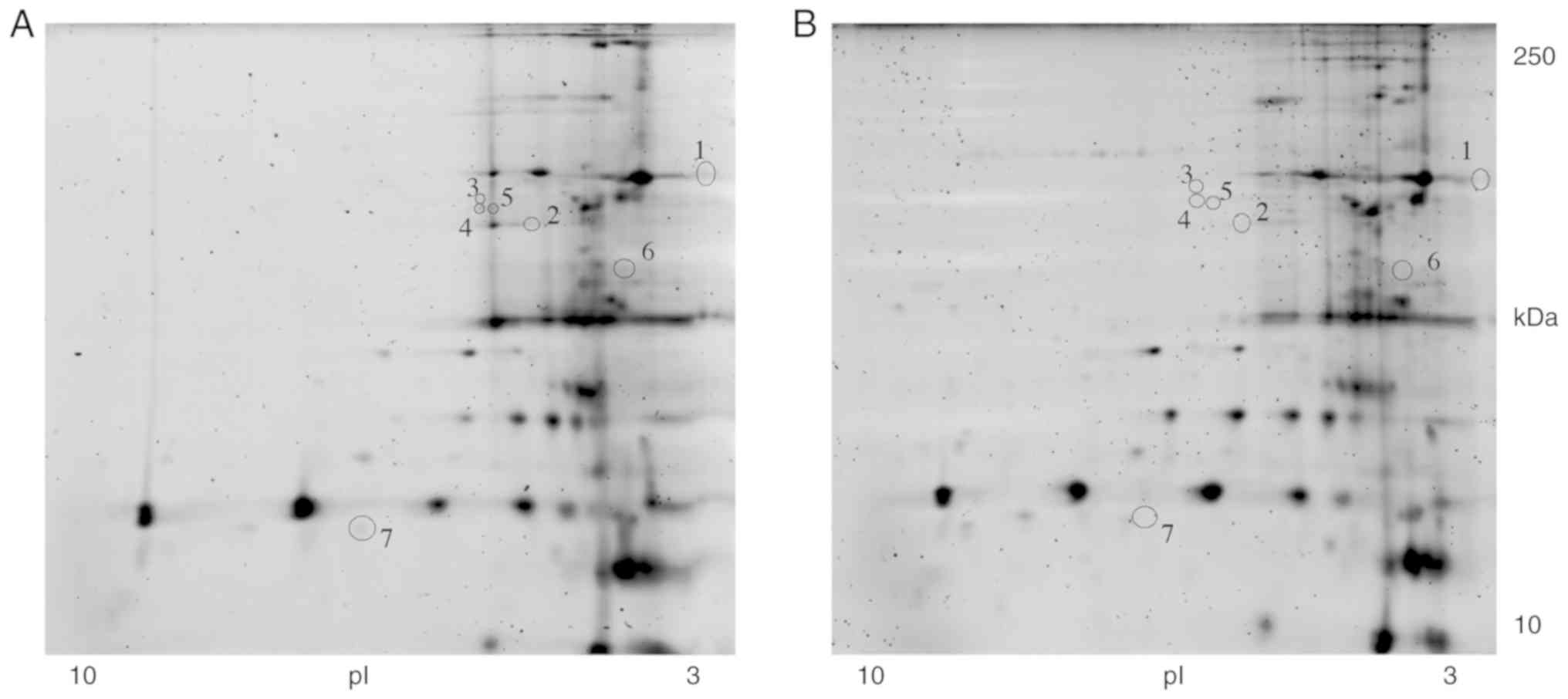
|
1
|
Barone R, Carchon H, Jansen E, Pavone L,
Fiumara A, Bosshard NU, Gitzelmann R and Jaeken J: Lysosomal enzyme
activities in serum and leukocytes from patients with
carbohydrate-deficient glycoprotein syndrome type IA
(phosphomannomutase deficiency). J Inherit Metab Dis. 21:167–172.
1998. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Jaeken J: Congenital disorders of
glycosylation. Ann NY Acad Sci. 1214:190–198. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Ichisaka S, Ohno K, Yuasa I, Nanba E,
Sakuraba H and Suzuki Y: Increased expression of
beta-hexosaminidase alpha chain in cultured skin fibroblasts from
patients with carbohydrate-deficient glycoprotein syndrome type I.
Brain Dev. 20:302–306. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Schwarze SR, Ho A, Vocero-Akbani A and
Dowdy SF: In vivo protein transduction: Delivery of a biologically
active protein into the mouse. Science. 285:1569–1572. 1999.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Elson-Schwab L, Garner OB, Schuksz M,
Crawford BE, Esko JD and Tor Y: Guanidinylated neomycin delivers
large, bioactive cargo into cells through a heparan
sulfate-dependent pathway. J Biol Chem. 282:13585–13591. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Snyder EL and Dowdy SF: Cell penetrating
peptides in drug delivery. Pharm Res. 21:389–393. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Freeze HH: Towards a therapy for
phosphomannomutase 2 deficiency, the defect in CDG-Ia patients.
Biochim Biophys Acta. 1792:835–840. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Eklund EA, Merbouh N, Ichikawa M,
Nishikawa A, Clima JM, Dorman JA, Norberg T and Freeze HH:
Hydrophobic Man-1-P derivatives correct abnormal glycosylation in
type I congenital disorder of glycosylation fibroblasts.
Glycobiology. 15:1084–1093. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Mayatepek E, Schröder M, Kohlmüller D,
Bieger WP and Nützenadel W: Continuous mannose infusion in
carbohydrate-deficient glycoprotein syndrome type I. Acta Paediatr.
86:1138–1140. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Kjaergaard S, Kristiansson B, Stibler H,
Freeze HH, Schwartz M, Martinsson T and Skovby F: Failure of
short-term mannose therapy of patients with carbohydrate-deficient
glycoprotein syndrome type 1A. Acta Paediatr. 87:884–888. 1998.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Mayatepek E and Kohlmüller D: Mannose
supplementation in carbohydrate-deficient glycoprotein syndrome
type I and phosphomannomutase deficiency. Eur J Pediatr.
157:605–606. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Brasil S, Pascoal C, Francisco R,
Marques-da-Silva D, Andreotti G, Videira PA, Morava E, Jaeken J and
Dos Reis Ferreira V: CDG therapies: From bench to bedside. Int J
Mol Sci. 19:E13042018. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Parveen S, Misra R and Sahoo SK:
Nanoparticles: A boon to drug delivery, therapeutics, diagnostics
and imaging. Nanomedicine. 8:147–166. 2012. View Article : Google Scholar
|
|
14
|
Tancini B, Tosi G, Bortot B, Dolcetta D,
Magini A, De Martino E, Urbanelli L, Ruozi B, Forni F, Emiliani C,
et al: Use of poly-lactide-co-glycolide-nanoparticles for lysosomal
delivery of a therapeutic enzyme in glycogenosis type II
fibroblasts. J Nanosci Nanotechnol. 15:2657–2666. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Costantino L, Gandolfi F, Tosi G, Rivasi
F, Vandelli MA and Forni F: Peptide-derivatized biodegradable
nanoparticles able to cross the blood-brain barrier. J Control
Release. 108:84–96. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Tosi G, Bortot B, Ruozi B, Dolcetta D,
Vandelli MA, Forni F and Severini GM: Potential use of polymeric
nanoparticles for drug delivery across the blood-brain barrier.
Curr Med Chem. 20:2212–2225. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Bondioli L, Costantino L, Ballestrazzi A,
Lucchesi D, Boraschi D, Pellati F, Benvenuti S, Tosi G and Vandelli
MA: PLGA nanopar-ticles surface decorated with the sialic acid,
N-acetylneuraminic acid. Biomaterials. 31:3395–3403. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Rosca ID, Watari F and Uo M: Microparticle
formation and its mechanism in single and double emulsion solvent
evaporation. J Control. 99:271–280. 2004.
|
|
19
|
Ruozi B, Belletti D, Forni F, Sharma A,
Muresanu D, Mössler H, Vandelli MA, Tosi G and Sharma HS: Poly
(D,L-lactide-co-glycolide) nanoparticles loaded with cerebrolysin
display neuroprotective activity in a rat model of concussive head
injury. CNS Neurol Disord Drug Targets. 13:1475–1482. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Joshi DP, Lan-Chun-Fung YL and Pritchard
JG: Determination of poly(vinyl alcohol) via its complex with boric
acid and iodine. Anal Chim Acta. 104:153–160. 1979. View Article : Google Scholar
|
|
21
|
Belletti D, Grabrucker AM, Pederzoli F,
Menrah I, Vandelli MA, Tosi G, Duskey TJ, Forni F and Ruozi B:
Hybrid nanoparticles as a new technological approach to enhance the
delivery of cholesterol into the brain. Int J Pharm. 543:300–310.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Pirard M, Achouri Y, Collet JF, Schollen
E, Matthijs G and Van Schaftingen E: Kinetic properties and
tissular distribution of mammalian phosphomannomutase isozymes.
Biochem J. 339:201–207. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Barone R, Carrozzi M, Parini R, Battini R,
Martinelli D, Elia M, Spada M, Lilliu F, Ciana G, Burlina A, et al:
A nationwide survey of PMM2-CDG in Italy: High frequency of a mild
neurological variant associated with the L32R mutation. J Neurol.
262:154–164. 2015. View Article : Google Scholar
|
|
24
|
Vega AI, Pérez-Cerdá C, Abia D, Gámez A,
Briones P, Artuch R, Desviat LR, Ugarte M and Pérez B: Expression
analysis revealing destabilizing mutations in phosphomannomutase 2
deficiency (PMM2-CDG): Expression analysis of PMM2-CDG mutations. J
Inherit Metab Dis. 34:929–939. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Grünewald S, Schollen E, Van Schaftingen
E, Jaeken J and Matthijs G: High residual activity of PMM2 in
patients' fibroblasts: Possible pitfall in the diagnosis of CDG-Ia
(phosphomannomutase deficiency). Am J Hum Genet. 68:347–354. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Bortot B, Cosentini D, Faletra F, Biffi S,
De Martino E, Carrozzi M and Severini GM: PMM2-CDG: Phenotype and
genotype in four affected family members. Gene. 531:506–509. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Monaco I, Arena F, Biffi S, Locatelli E,
Bortot B, La Cava F, Marini GM, Severini GM, Terreno E and Comes
Franchini M: Synthesis of lipophilic core-shell
Fe3O4 @SiO2 @Au nanoparticles and
polymeric entrapment into nanomicelles: A novel nanosystem for in
vivo active targeting and magnetic resonance-photoacoustic dual
imaging. Bioconjug Chem. 28:1382–1390. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Carcoforo P, Ura B, Mischiati C,
Squerzanti M, Lanzara V, Cervellati C, Calza R, De Laureto PP,
Frare E, Portinari M, et al: Comparative proteomic analysis of
ductal breast carcinoma demonstrates an altered expression of
chaperonins and cytoskeletal proteins. Mol Med Rep. 7:1700–1704.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Sahay G, Alakhova DY and Kabanov AV:
Endocytosis of nano-medicines. J Control Release. 145:182–195.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Hu Q, Gu G, Liu Z, Jiang M, Kang T, Miao
D, Tu Y, Pang Z, Song Q, Yao L, et al: F3 peptide-functionalized
PEG-PLA nanoparticles co-administrated with tLyp-1 peptide for
anti-glioma drug delivery. Biomaterials. 34:1135–1145. 2013.
View Article : Google Scholar
|
|
31
|
Rush JS, Panneerselvam K, Waechter CJ and
Freeze HH: Mannose supplementation corrects GDP-mannose deficiency
in cultured fibroblasts from some patients with Congenital
Disorders of Glycosylation (CDG). Glycobiology. 10:829–835. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Kleinert P, Kuster T, Arnold D, Jaeken J,
Heizmann CW and Troxler H: Effect of glycosylation on the protein
pattern in 2-D-gel electrophoresis. Proteomics. 7:15–22. 2007.
View Article : Google Scholar
|
|
33
|
Cartiera MS, Johnson KM, Rajendran V,
Caplan MJ and Saltzman WM: The uptake and intracellular fate of
PLGA nano-particles in epithelial cells. Biomaterials.
30:2790–2798. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar
|
|
35
|
Van Scherpenzeel M, Willems E and Lefeber
DJ: Clinical diagnostics and therapy monitoring in the congenital
disorders of glycosylation. Glycoconj J. 33:345–358. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Chan B, Clasquin M, Smolen GA, Histen G,
Powe J, Chen Y, Lin Z, Lu C, Liu Y, Cang Y, et al: A mouse model of
a human congenital disorder of glycosylation caused by loss of
PMM2. Hum Mol Genet. 25:2182–2193. 2016. View Article : Google Scholar : PubMed/NCBI
|