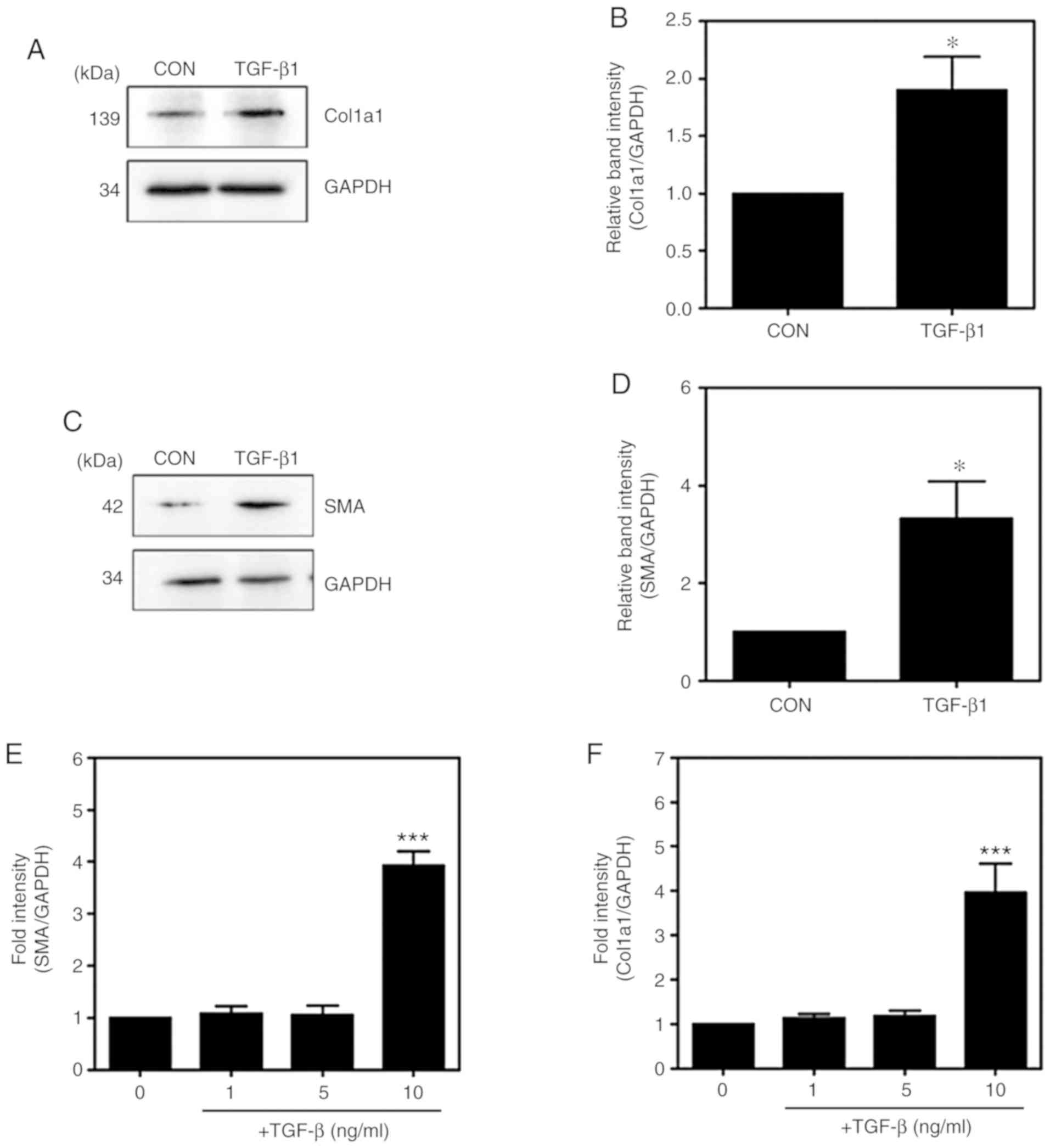
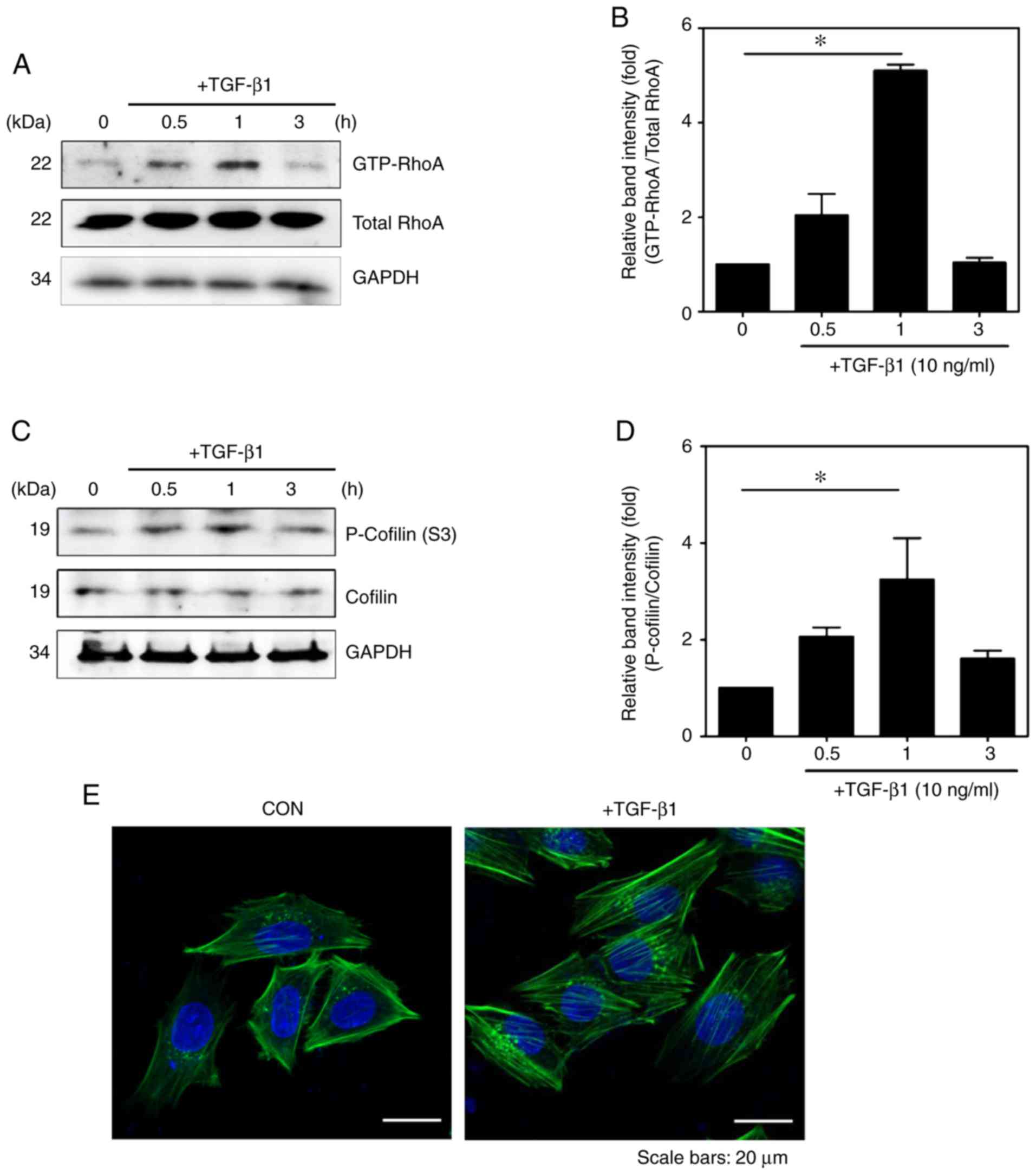
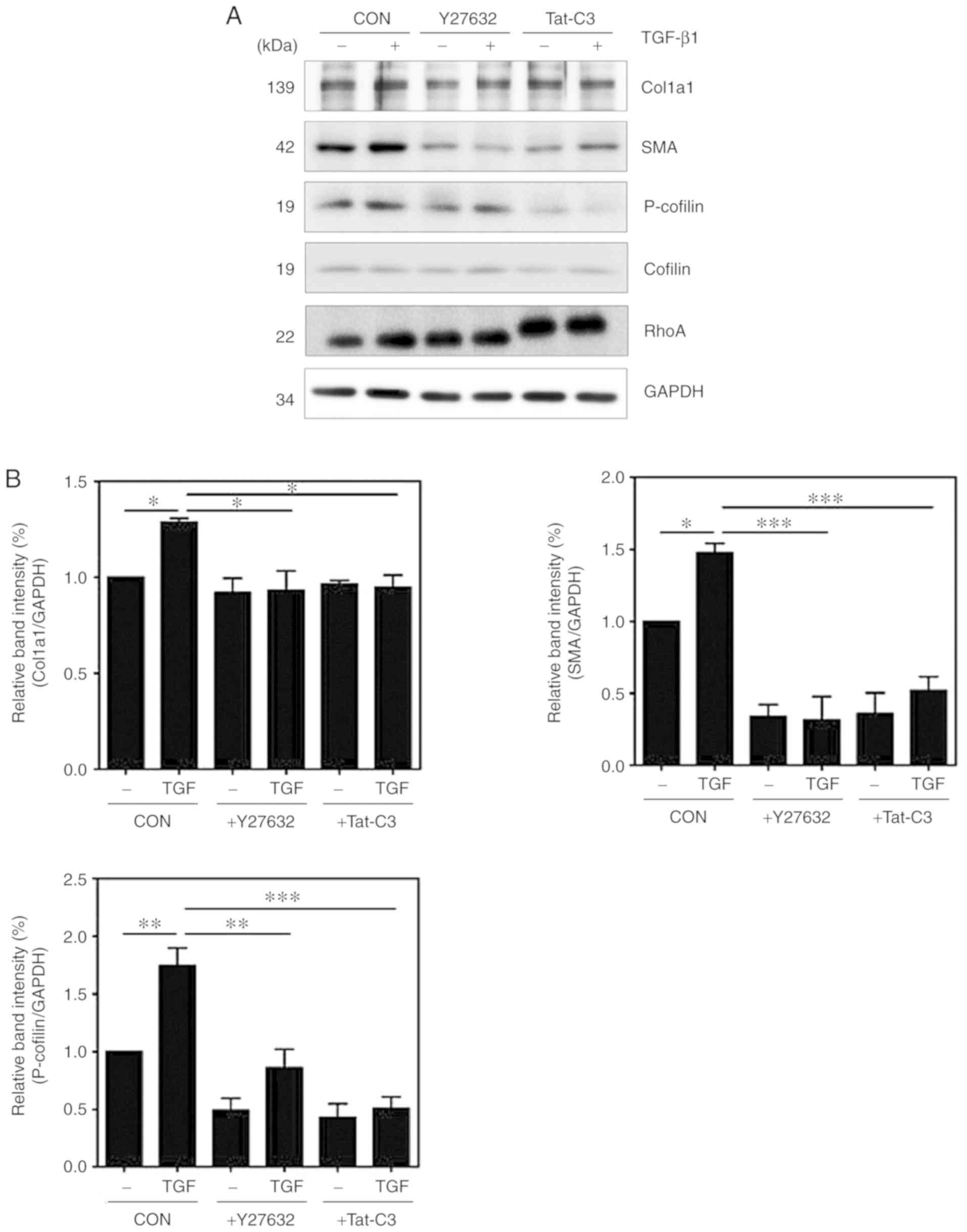
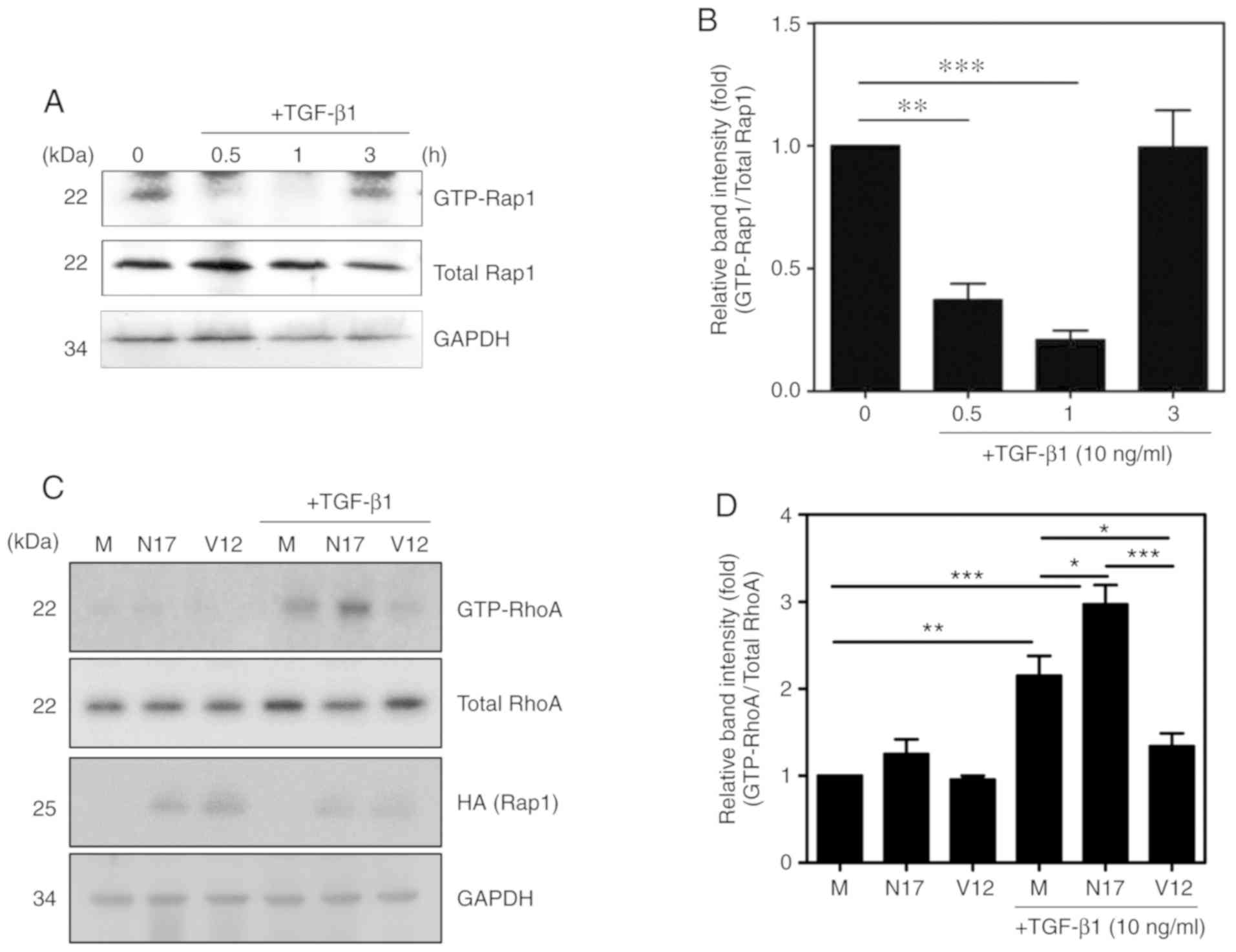
|
1
|
Friedman SL: Mechanisms of hepatic
fibrogenesis. Gastroenterology. 134:1655–1669. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Arriazu E, Ruiz de Galarreta M, Cubero FJ,
Varela-Rey M, Perez de Obanos MP, Leung TM, Lopategi A, Benedicto
A, Abraham-Enachescu I and Nieto N: Extracellular matrix and liver
disease. Antioxid Redox Signal. 21:1078–1097. 2014. View Article : Google Scholar :
|
|
3
|
Yang C, Zeisberg M, Mosterman B, Sudhakar
A, Yerramalla U, Holthaus K, Xu L, Eng F, Afdhal N and Kalluri R:
Liver fibrosis: Insights into migration of hepatic stellate cells
in response to extracellular matrix and growth factors.
Gastroenterology. 124:147–159. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Wang XM, Yu DM, McCaughan GW and Gorrell
MD: Fibroblast activation protein increases apoptosis, cell
adhesion, and migration by the LX-2 human stellate cell line.
Hepatology. 42:935–945. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Raftopoulou M and Hall A: Cell migration:
Rho GTPases lead the way. Dev Biol. 265:23–32. 2004. View Article : Google Scholar
|
|
6
|
Nobes CD and Hall A: Rho, rac, and cdc42
GTPases regulate the assembly of multimolecular focal complexes
associated with actin stress fibers, lamellipodia, and filopodia.
Cell. 81:53–62. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Ridley AJ and Hall A: The small
GTP-binding protein rho regulates the assembly of focal adhesions
and actin stress fibers in response to growth factors. Cell.
70:389–399. 1992. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Nobes CD, Hawkins P, Stephens L and Hall
A: Activation of the small GTP-binding proteins rho and rac by
growth factor receptors. J Cell Sci. 108:225–233. 1995.PubMed/NCBI
|
|
9
|
Etienne-Manneville S and Hall A: Rho
GTPases in cell biology. Nature. 420:629–635. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Li MO, Wan YY, Sanjabi S, Robertson AK and
Flavell RA: Transforming growth factor-beta regulation of immune
responses. Annu Rev Immunol. 24:99–146. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Zhang YE: Non-Smad pathways in TGF-beta
signaling. Cell Res. 19:128–139. 2009. View Article : Google Scholar :
|
|
12
|
Edlund S, Landstrom M, Heldin CH and
Aspenstrom P: Transforming growth factor-beta-induced mobilization
of actin cytoskeleton requires signaling by small GTPases Cdc42 and
RhoA. Mol Boil Cell. 13:902–914. 2002. View Article : Google Scholar
|
|
13
|
Kim JS, Kim JG, Moon MY, Jeon CY, Won HY,
Kim HJ, Jeon YJ, Seo JY, Kim JI, Kim J, et al: Transforming growth
factor-beta1 regulates macrophage migration via RhoA. Blood.
108:1821–1829. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Papadimitriou E, Kardassis D, Moustakas A
and Stournaras C: TGFβ-induced early activation of the small GTPase
RhoA is Smad2/3-independent and involves Src and the guanine
nucleotide exchange factor Vav2. Cell Physiol Biochem. 28:229–238.
2011. View Article : Google Scholar
|
|
15
|
Li L, Wang JY, Yang CQ and Jiang W: Effect
of RhoA on transforming growth factor β1-induced rat hepatic
stellate cell migration. Liver Int. 32:1093–1102. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Yamamoto Y and Gaynor RB: IkappaB kinases:
Key regulators of the NF-kappaB pathway. Trends Biochem Sci.
29:72–79. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Kim HJ, Kim JG, Moon MY, Park SH and Park
JB: IκB kinase γ/nuclear factor-κB-essential modulator (IKKγ/NEMO)
facilitates RhoA GTPase activation, which, in turn, activates
Rho-associated KINASE (ROCK) to phosphorylate IKKβ in response to
transforming growth factor (TGF)-β1. J Boil Chem. 289:1429–1440.
2014. View Article : Google Scholar
|
|
18
|
Bos JL, de Bruyn K, Enserink J, Kuiperij
B, Rangarajan S, Rehmann H, Riedl J, de Rooij J, van Mansfeld F and
Zwartkruis F: The role of Rap1 in integrin-mediated cell adhesion.
Biochem Soc Trans. 31:83–86. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Moon MY, Kim HJ, Kim JG, Lee JY, Kim J,
Kim SC, Choi IG, Kim PH and Park JB: Small GTPase Rap1 regulates
cell migration through regulation of small GTPase RhoA activity in
response to transforming growth factor-β1. J Cell Physiol.
228:2119–2126. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Teo H, Ghosh S, Luesch H, Ghosh A, Wong
ET, Malik N, Orth A, de Jesus P, Perry AS, Oliver JD, et al:
Telomere-independent Rap1 is an IKK adaptor and regulates
NF-kappaB-dependent gene expression. Nat Cell Biol. 12:758–767.
2010. View
Article : Google Scholar : PubMed/NCBI
|
|
21
|
Ghosh AS and Tergaonkar V: Telomeres and
inflammation: Rap1 joins the ends? Cell Cycle. 9:3834–3835. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Vogel S, Piantedosi R, Frank J, Lalazar A,
Rockey DC, Friedman SL and Blaner WS: An immortalized rat liver
stellate cell line (HSC-T6): A new cell model for the study of
retinoid metabolism in vitro. J Lipid Res. 41:882–893.
2000.PubMed/NCBI
|
|
23
|
Park J, Kim JS, Jung KC, Lee HJ, Kim JI,
Kim J, Lee JY, Park JB and Choi SY: Exoenzyme Tat-C3 inhibits
association of zymosan particles, phagocytosis, adhesion, and
complement binding in macrophage cells. Mol Cells. 16:216–223.
2003.PubMed/NCBI
|
|
24
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar
|
|
25
|
Ellerbroek SM, Wennerberg K and Burridge
K: Serine phosphorylation negatively regulates RhoA in vivo. J Biol
Chem. 278:19023–19031. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Sit ST and Manser E: Rho GTPases and their
role in organizing the actin cytoskeleton. J Cell Sci. 124:679–683.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Spiering D and Hodgson L: Dynamics of the
Rho-family small GTPases in actin regulation and motility. Cell Adh
Migr. 5:170–180. 2011. View Article : Google Scholar :
|
|
28
|
Marra F, Arrighi MC, Fazi M, Caligiuri A,
Pinzani M, Romanelli RG, Efsen E, Laffi G and Gentilini P:
Extracellular signal-regulated kinase activation differentially
regulates platelet-derived growth factor's actions in hepatic
stellate cells, and is induced by in vivo liver injury in the rat.
Hepatology. 30:951–958. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Bataller R and Brenner DA: Liver fibrosis.
J Clin Invest. 115:209–218. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Bettermann K, Vucur M, Haybaeck J, Koppe
C, Janssen J, Heymann F, Weber A, Weiskirchen R, Liedtke C, Gassler
N, et al: TAK1 suppresses a NEMO-dependent but NF-kappaB-
independent pathway to liver cancer. Cancer Cell. 17:481–496. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Inokuchi S, Aoyama T, Miura K,
Osterreicher CH, Kodama Y, Miyai K, Akira S, Brenner DA and Seki E:
Disruption of TAK1 in hepatocytes causes hepatic injury,
inflammation, fibrosis, and carcinogenesis. Proc Natl Acad Sci USA.
107:844–849. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Luedde T, Beraza N, Kotsikoris V, van Loo
G, Nenci A, De Vos R, Roskams T, Trautwein C and Pasparakis M:
Deletion of NEMO/IKKgamma in liver parenchymal cells causes
steatohepatitis and hepatocellular carcinoma. Cancer Cell.
11:119–132. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Luedde T and Schwabe RF: NF-κB in the
liver-linking injury, fibrosis and hepatocellular carcinoma. Nat
Rev Gastroenterol Hepatol. 8:108–118. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Friedman SL: Molecular regulation of
hepatic fibrosis, an integrated cellular response to tissue injury.
J Biol Chem. 275:2247–2250. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Benyon RC and Arthur MJ: Mechanisms of
hepatic fibrosis. J Pediatr Gastroenterol Nutr. 27:75–85. 1998.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Lee UE and Friedman SL: Mechanisms of
hepatic fibrogenesis. Best Pract Res Clin Gastroenterol.
25:195–206. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Gressner AM, Weiskirchen R, Breitkopf K
and Dooley S: Roles of TGF-beta in hepatic fibrosis. Front Biosci.
7:d793–d807. 2002. View
Article : Google Scholar : PubMed/NCBI
|
|
38
|
Breitkopf K, Godoy P, Ciuclan L, Singer MV
and Dooley S: TGF-beta/Smad signaling in the injured liver. Z
Gastroenterol. 44:57–66. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Friedman SL: Liver fibrosis: From
mechanisms to treatment. Gastroenterol Clin Biol. 31:812–814. 2007.
View Article : Google Scholar
|
|
40
|
Ridley AJ: Rho GTPases and cell migration.
J Cell Sci. 114:2713–2722. 2001.PubMed/NCBI
|
|
41
|
Wang W, Eddy R and Condeelis J: The
cofilin pathway in breast cancer invasion and metastasis. Nat Rev
Cancer. 7:429–440. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
DesMarais V, Ghosh M, Eddy R and Condeelis
J: Cofilin takes the lead. J Cell Sci. 118:19–26. 2005. View Article : Google Scholar
|
|
43
|
Iwamoto H, Nakamuta M, Tada S, Sugimoto R,
Enjoji M and Nawata H: A p160ROCK-specific inhibitor, Y-27632,
attenuates rat hepatic stellate cell growth. J Hepatol. 32:762–770.
2000. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Kato M, Iwamoto H, Higashi N, Sugimoto R,
Uchimura K, Tada S, Sakai H, Nakamuta M and Nawata H: Role of Rho
small GTP binding protein in the regulation of actin cytoskeleton
in hepatic stellate cells. J Hepatol. 31:91–99. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Tada S, Iwamoto H, Nakamuta M, Sugimoto R,
Enjoji M, Nakashima Y and Nawata H: A selective ROCK inhibitor,
Y27632, prevents dimethylnitrosamine-induced hepatic fibrosis in
rats. J Hepatol. 34:529–536. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Bos JL, de Rooij J and Reedquist KA: Rap1
signalling: Adhering to new models. Nat Rev Mol Cell Boil.
2:369–377. 2001. View Article : Google Scholar
|
|
47
|
Fujita H, Fukuhara S, Sakurai A, Yamagishi
A, Kamioka Y, Nakaoka Y, Masuda M and Mochizuki N: Local activation
of Rap1 contributes to directional vascular endothelial cell
migration accompanied by extension of microtubules on which RAPL, a
Rap1-associating molecule, localizes. J Biol Chem. 280:5022–5031.
2005. View Article : Google Scholar
|
|
48
|
Reedquist KA, Ross E, Koop EA, Wolthuis
RM, Zwartkruis FJ, van Kooyk Y, Salmon M, Buckley CD and Bos JL:
The small GTPase, Rap1, mediates CD31-induced integrin adhesion. J
Cell Biol. 148:1151–1158. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Katagiri K, Hattori M, Minato N, Irie SK,
Takatsu K and Kinashi T: Rap1 is a potent activation signal for
leukocyte function-associated antigen 1 distinct from protein
kinase C and phosphatidylinositol-3-OH kinase. Mol Cell Biol.
20:1956–1969. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Tohyama Y, Katagiri K, Pardi R, Lu C,
Springer TA and Kinashi T: The critical cytoplasmic regions of the
alphaL/beta2 integrin in Rap1-induced adhesion and migration. Mol
Boil Cell. 14:2570–2582. 2003. View Article : Google Scholar
|
|
51
|
Jeon CY, Kim HJ, Lee JY, Kim JB, Kim SC
and Park JB: p190RhoGAP and Rap-dependent RhoGAP (ARAP3) inactivate
RhoA in response to nerve growth factor leading to neurite
outgrowth from PC12 cells. Exp Mol Med. 42:335–344. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Krugmann S, Williams R, Stephens L and
Hawkins PT: ARAP3 is a PI3K- and rap-regulated GAP for RhoA. Curr
Boil. 14:1380–1384. 2004. View Article : Google Scholar
|
|
53
|
Mao R, Fan Y, Mou Y, Zhang H, Fu S and
Yang J: TAK1 lysine 158 is required for TGF-β-induced
TRAF6-mediated Smad-independent IKK/NF-κB and JNK/AP-1 activation.
Cell Signal. 23:222–227. 2011. View Article : Google Scholar
|
|
54
|
Sorrentino A, Thakur N, Grimsby S,
Marcusson A, von Bulow V, Schuster N, Zhang S, Heldin CH and
Landstrom M: The type I TGF-beta receptor engages TRAF6 to activate
TAK1 in a receptor kinase-independent manner. Nat Cell Boil.
10:1199–1207. 2008. View Article : Google Scholar
|
|
55
|
Takaesu G, Surabhi RM, Park KJ,
Ninomiya-Tsuji J, Matsumoto K and Gaynor RB: TAK1 is critical for
IkappaB kinase-mediated activation of the NF-kappaB pathway. J Mol
Boil. 326:105–115. 2003. View Article : Google Scholar
|
|
56
|
Wang C, Deng L, Hong M, Akkaraju GR, Inoue
J and Chen ZJ: TAK1 is a ubiquitin-dependent kinase of MKK and IKK.
Nature. 412:346–351. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Qi W, Chen X, Holian J, Mreich E, Twigg S,
Gilbert RE and Pollock CA: Transforming growth factor-beta1
differentially mediates fibronectin and inflammatory cytokine
expression in kidney tubular cells. Am J Physiol Renal Physiol.
291:F1070–F1077. 2006. View Article : Google Scholar : PubMed/NCBI
|