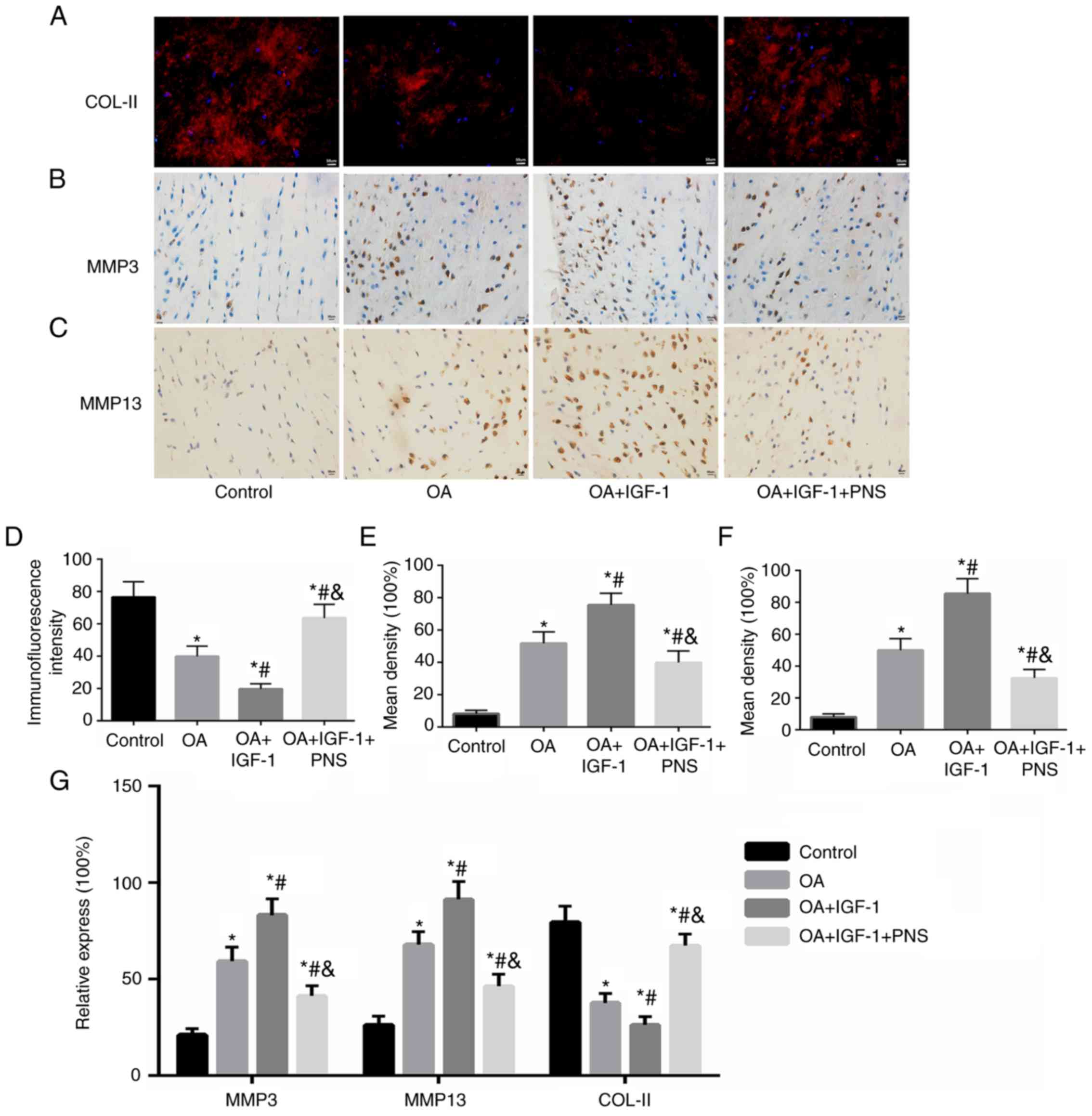
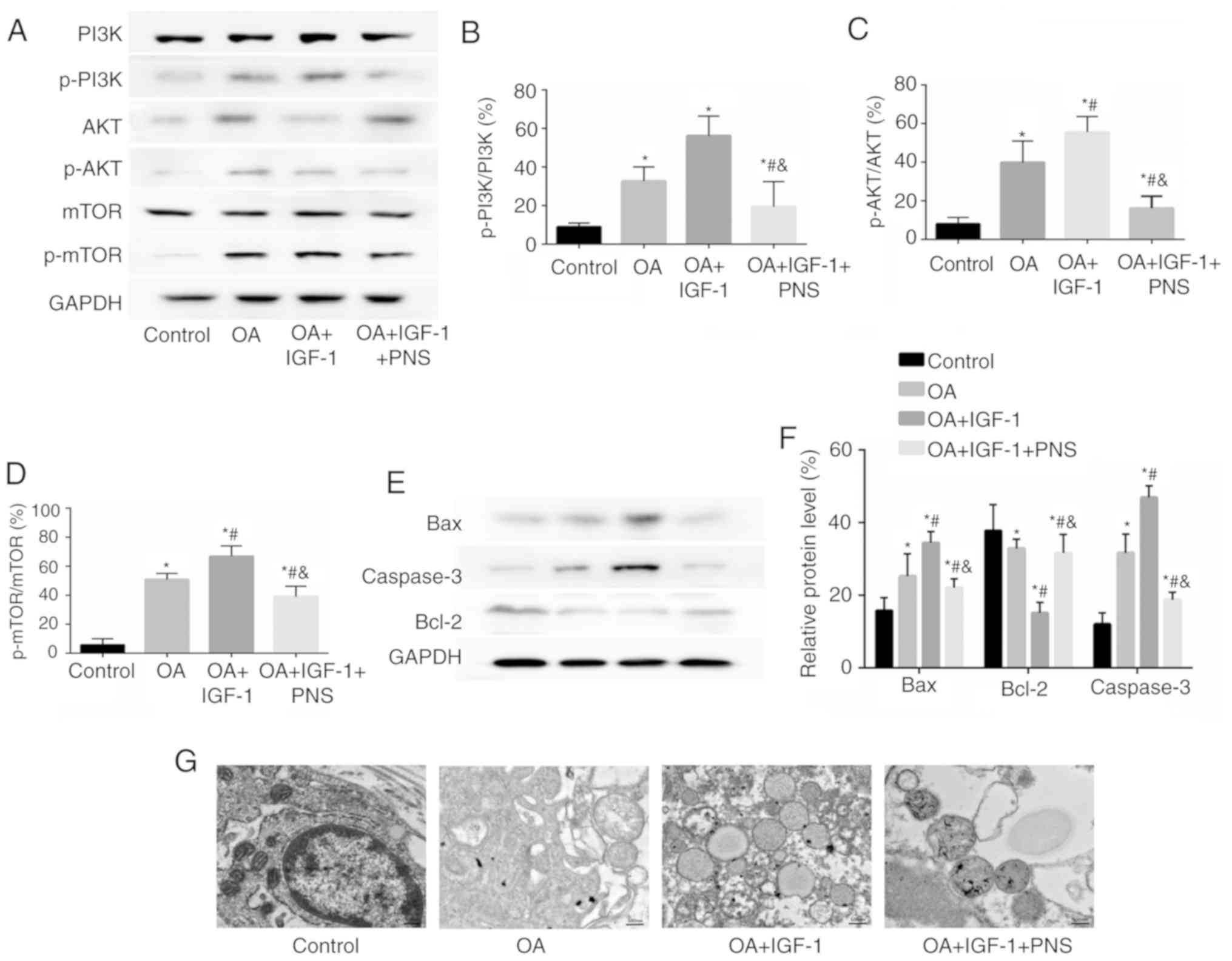
|
1
|
Guilak F, Nims RJ, Dicks A, Wu CL and
Meulenbelt I: Osteoarthritis as a disease of the cartilage
pericellular matrix. Matrix Biol. 71-72:40–50. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Rahmati M, Mobasheri A and Mozafari M:
Reply to 'Comment on: Inflammatory mediators in osteoarthritis: A
critical review of the state-of-the art, prospects, and future
challenges'. Bone. 105:3112017. View Article : Google Scholar
|
|
3
|
Englund M, Guermazi A, Gale D, Hunter DJ,
Aliabadi P, Clancy M and Felson DT: Incidental meniscal findings on
knee MRI in middle-aged and elderly persons. N Engl J Med.
359:1108–1115. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Prieto-Alhambra D, Judge A, Javaid MK,
Cooper C, Diez-Perez A and Arden NK: Incidence and risk factors for
clinically diagnosed knee, hip and hand osteoarthritis: Influences
of age, gender and osteoarthritis affecting other joints. Ann Rheum
Dis. 73:1659–1664. 2014. View Article : Google Scholar
|
|
5
|
Greene MA and Loeser RF: Aging-related
inflammation in osteoarthritis. Osteoarthritis Cartilage.
23:1966–1971. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
McCulloc K, Litherland GJ and Rai TS:
Cellular senescence in osteoarthritis pathology. Aging Cell.
16:210–218. 2017. View Article : Google Scholar
|
|
7
|
Jeon OH, David N, Campisi J and Elisseeff
JH: Senescent cells and osteoarthritis: A painful connection. J
Clin Invest. 128:1229–1237. 2018. View
Article : Google Scholar : PubMed/NCBI
|
|
8
|
Calcinotto A, Kohli J, Zagato E,
Pellegrini L, Demaria M and Alimonti A: Cellular senescence: Aging,
cancer, and injury. Physiol Rev. 99:1047–1078. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Wodarz D: Effect of stem cell turnover
rates on protection against cancer and aging. J Theor Biol.
245:449–458. 2007. View Article : Google Scholar
|
|
10
|
Goessler UR, Bieback K, Bugert P, Heller
T, Sadick H, Hörmann K and Riedel F: In vitro analysis of integrin
expression during chondrogenic differentiation of mesenchymal stem
cells and chondrocytes upon dedifferentiation in cell culture. Int
J Mol Med. 17:301–307. 2006.PubMed/NCBI
|
|
11
|
Hou A, Chen P, Tang H, Meng H, Cheng X,
Wang Y, Zhang Y and Peng J: Cellular senescence in osteoarthritis
and anti-aging strategies. Mech Ageing Dev. 175:83–87. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Ashkavand Z, Malekinejad H and Vishwanath
BS: The patho-physiology of osteoarthritis. J Pharm Res. 7:132–138.
2013.
|
|
13
|
Muñoz-Espín D and Serrano M: Cellular
senescence: From physiology to pathology. Nat Rev Mol Cell Biol.
15:482–496. 2014. View
Article : Google Scholar : PubMed/NCBI
|
|
14
|
Levine B and Kroemer G: Autophagy in the
pathogenesis of disease. Cell. 132:27–42. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
LoPiccolo J, Blumenthal GM, Bernstein WB
and Dennis PA: Targeting the PI3K/Akt/mTOR pathway: Effective
combinations and clinical considerations. Drug Resist Update.
11:32–50. 2008. View Article : Google Scholar
|
|
16
|
Jones SA, Mills KH and Harris J: Autophagy
and inflammatory diseases. Immunol Cell Biol. 91:250–258. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Chen Z, Jin T and Lu Y: AntimiR-30b
inhibits TNF-α mediated apoptosis and attenuated cartilage
degradation through enhancing autophagy. Cell Physiol Biochem.
40:883–894. 2016. View Article : Google Scholar
|
|
18
|
Choi AM, Ryter SW and Levine B: Autophagy
in human health and disease. N Engl J Med. 368:651–662. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Green DR, Galluzzi L and Kroemer G:
Mitochondria and the autophagy-inflammation-cell death axis in
organismal aging. Science. 333:1109–1112. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Duan L, Xiong X, Hu J, Liu Y, Li J and
Wang J: Panax notoginseng saponins for treating coronary artery
disease: A functional and mechanistic overview. Front Pharmacol.
8:7022017. View Article : Google Scholar :
|
|
21
|
Haiping Z, Ziping H, Guangwen L, Sijia Z
and Yumin L: Therapeutic potential and cellular mechanisms of Panax
notoginseng on prevention of aging and cell senescence-associated
diseases. Aging Dis. 8:721–739. 2017. View Article : Google Scholar
|
|
22
|
Xu ZM, Li CB, Liu QL, Li P and Yang H:
Ginsenoside Rg1 prevents doxorubicin-induced cardiotoxicity through
the inhibition of autophagy and endoplasmic reticulum stress in
mice. Int J Mol Sci. 19:E36582018. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Zhou P, Weijie X, He S, Sun Y, Meng X, Sun
G and Sun X: Ginsenoside Rb1 as an anti-diabetic agent and its
underlying mechanism analysis. Cells. 8:2042019. View Article : Google Scholar :
|
|
24
|
Liu MW, Wei R, Su MX, Li H, Fang TW and
Zhang W: Effects of Panax notoginseng saponins on severe acute
pancreatitis through the regulation of mTOR/Akt and caspase-3
signaling pathway by upregulating miR-181b expression in rats. BMC
Complement Altern Med. 18:512018. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar
|
|
26
|
Kikuchi M, Matsuura K, Matsumoto Y,
Inagaki T and Ueda R: Bibliographical investigation of
complementary alternative medicines for osteoarthritis and
rheumatoid arthritis. Geriatr Gerontol Int. 9:29–40. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Marcus DM: Therapy: Herbals and
supplements for rheumatic diseases. Nat Rev Rheumatol. 5:299–300.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Wang T, Guo R, Zhou G, Zhou X, Kou Z, Sui
F, Li C, Tang L and Wang Z: Traditional uses, botany,
phytochemistry, pharmacology and toxicology of Panax notoginseng
(Burk) F H Chen: A review. J Ethnopharmacol. 188:234–258. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Rhule A, Navarro S, Smith JR and Shepherd
DM: Panax notoginseng attenuates LPS-induced pro-inflammatory
mediators in RAW264.7 cells. J Ethnopharmacol. 106:121–128. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Peng XD, Dai LL, Huang CQ, He CM, Yang B
and Chen LJ: Relationship between anti-fibrotic effect of Panax
notoginseng saponins and serum cytokines in rat hepatic fibrosis.
Biochem Biophys Res Commun. 388:31–34. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Qiang H, Zhang C, Shi ZB, Yang HQ and Wang
KZ: Protective effects and mechanism of Panax notoginseng saponins
on oxidative stress-induced damage and apoptosis of rabbit bone
marrow stromal cells. Chin J Integr Med. 16:525–530. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Youle RJ and Narendra DP: Mechanisms of
mitophagy. Nat Rev Mol Cell Biol. 12:9–14. 2011. View Article : Google Scholar
|
|
33
|
Zhang Y, Vasheghani F, Li YH, Blati M,
Simeone K, Fahmi H, Lussier B, Roughley P, Lagares D, Pelletier JP,
et al: Cartilage-specific deletion of mTOR upregulates autophagy
and protects mice from osteoarthritis. J Ann Rheum Dis.
74:1432–1440. 2015. View Article : Google Scholar
|
|
34
|
López de Figueroa P, Lotz MK, Blanco FJ
and Caramés B: Autophagy activation protects from mitochondrial
dysfunction in human chondrocytes. Arthritis Rheumatol. 67:966–976.
2015. View Article : Google Scholar
|
|
35
|
Jung CH, Ro SH, Cao J, Otto NM and Kim DH:
mTOR regulation of autophagy. FEBS Lett. 584:1287–1295. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Xue JF, Shi ZM, Zou J and Li XL:
Inhibition of PI3K/AKT/mTOR signaling pathway promotes autophagy of
articular chondrocytes and attenuates inflammatory response in rats
with osteoarthritis. Biomed Pharmacother. 89:1252–1261. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Pal B, Endisha H, Zhang Y and Kapoor M:
Mtor: A potential therapeutic target in osteoarthritis? Drugs R D.
15:27–36. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Caramés B, Hasegawa A, Taniguchi N, Miyaki
S, Blanco FJ and Lotz M: Autophagy activation by rapamycin reduces
severity of experimental osteoarthritis. Ann Rheum Dis. 71:575–581.
2012. View Article : Google Scholar :
|
|
39
|
Kapoor M, Martelpelletier J, Lajeunesse D,
Pelletier JP and Fahmi H: Role of proinflammatory cytokines in the
patho-physiology of osteoarthritis. Nat Rev Rheumatol. 7:33–42.
2011. View Article : Google Scholar
|
|
40
|
Gianluigi Z, Paola T, Paolo R, Simona G,
Luigino B and Antonio L: Systemic and nonrenal adverse effects
occurring in renal transplant patients treated with mTOR
inhibitors. Clin Dev Immunol. 2013:4032802013.
|
|
41
|
Fosang AJ and Little CB: Drug insight:
Aggrecanases as therapeutic targets for osteoarthritis. Nat Clin
Pract Rheumatol. 4:420–427. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Creel PA: Management of mTOR inhibitor
side effects. J Clin J Oncol Nurs. 13(Suppl): S19–S23. 2009.
View Article : Google Scholar
|
|
43
|
Mandl LA: Osteoarthritis year in review
2018: Clinical. Osteoarthr Cartilage. 27:359–364. 2019. View Article : Google Scholar
|
|
44
|
Miyaki S and Lotz MK: Extracellular
vesicles in cartilage homeostasis and osteoarthritis. Curr Opin
Rheumatol. 30:129–135. 2018. View Article : Google Scholar :
|
|
45
|
Rai MF, Cotton R and Sandell LJ:
Chemokines modulate chondrocyte homeostasis: Implications in
osteoarthritis. Osteoarthr Cartilage. 20(Suppl): S2402012.
View Article : Google Scholar
|