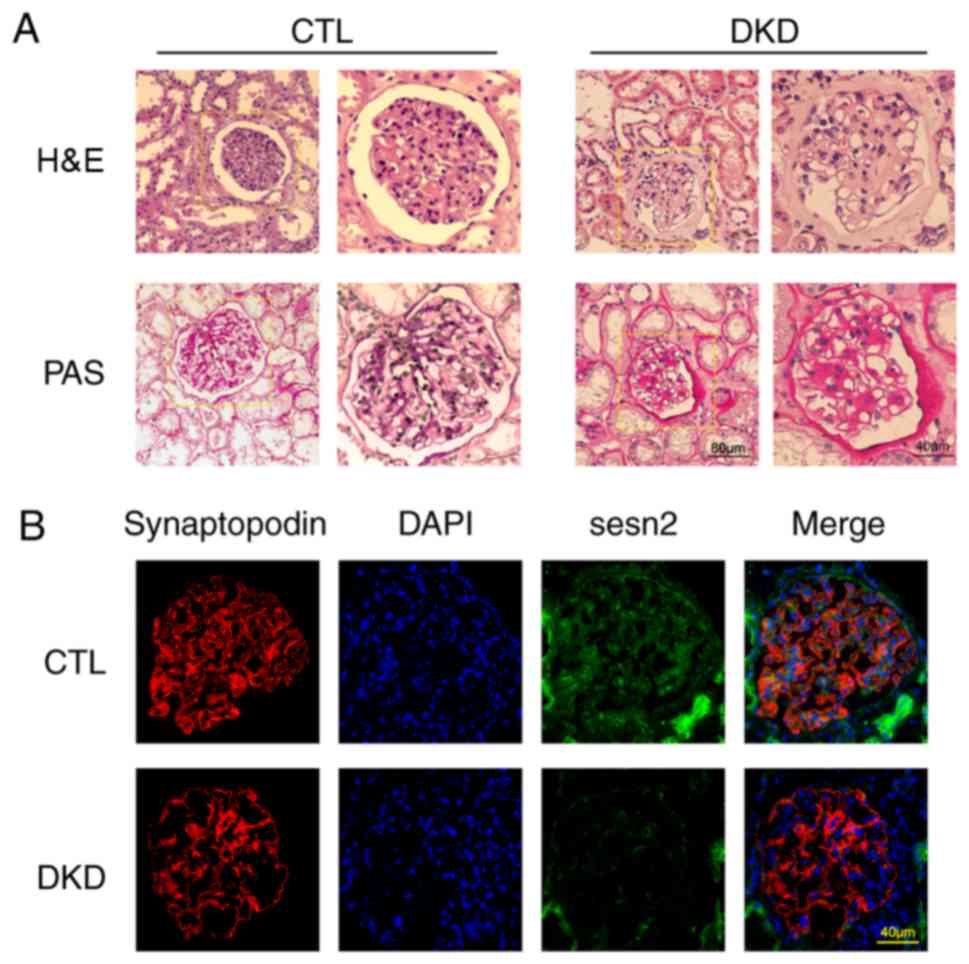
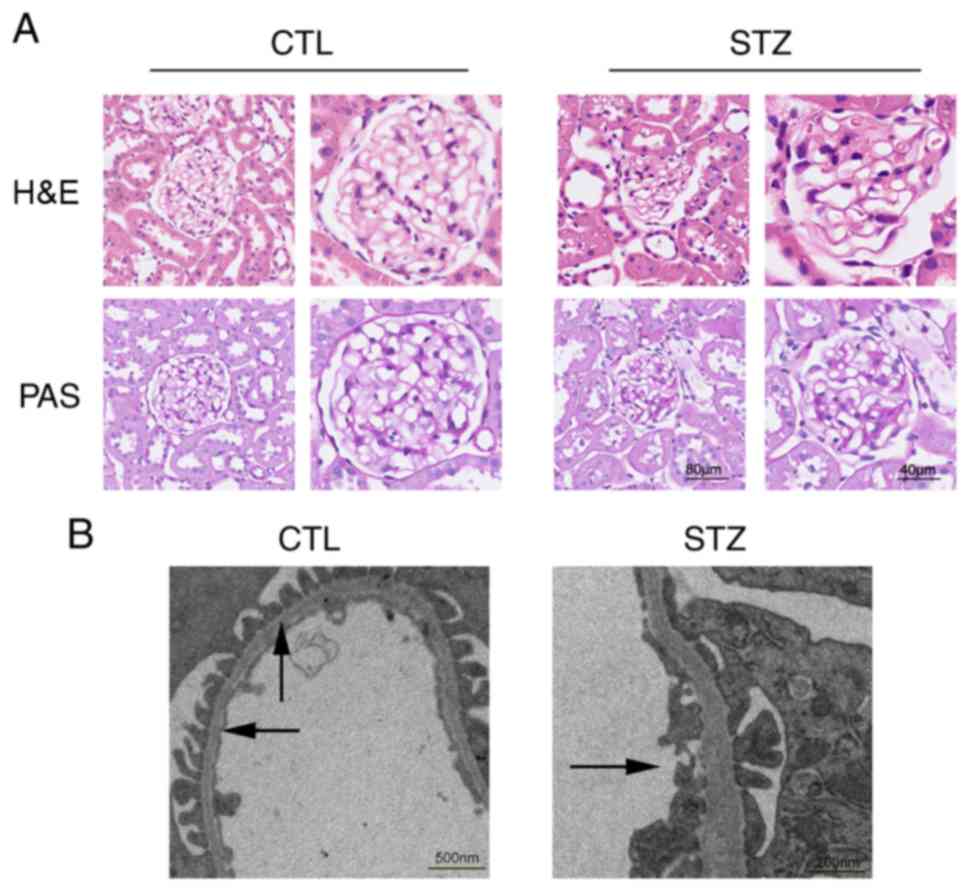
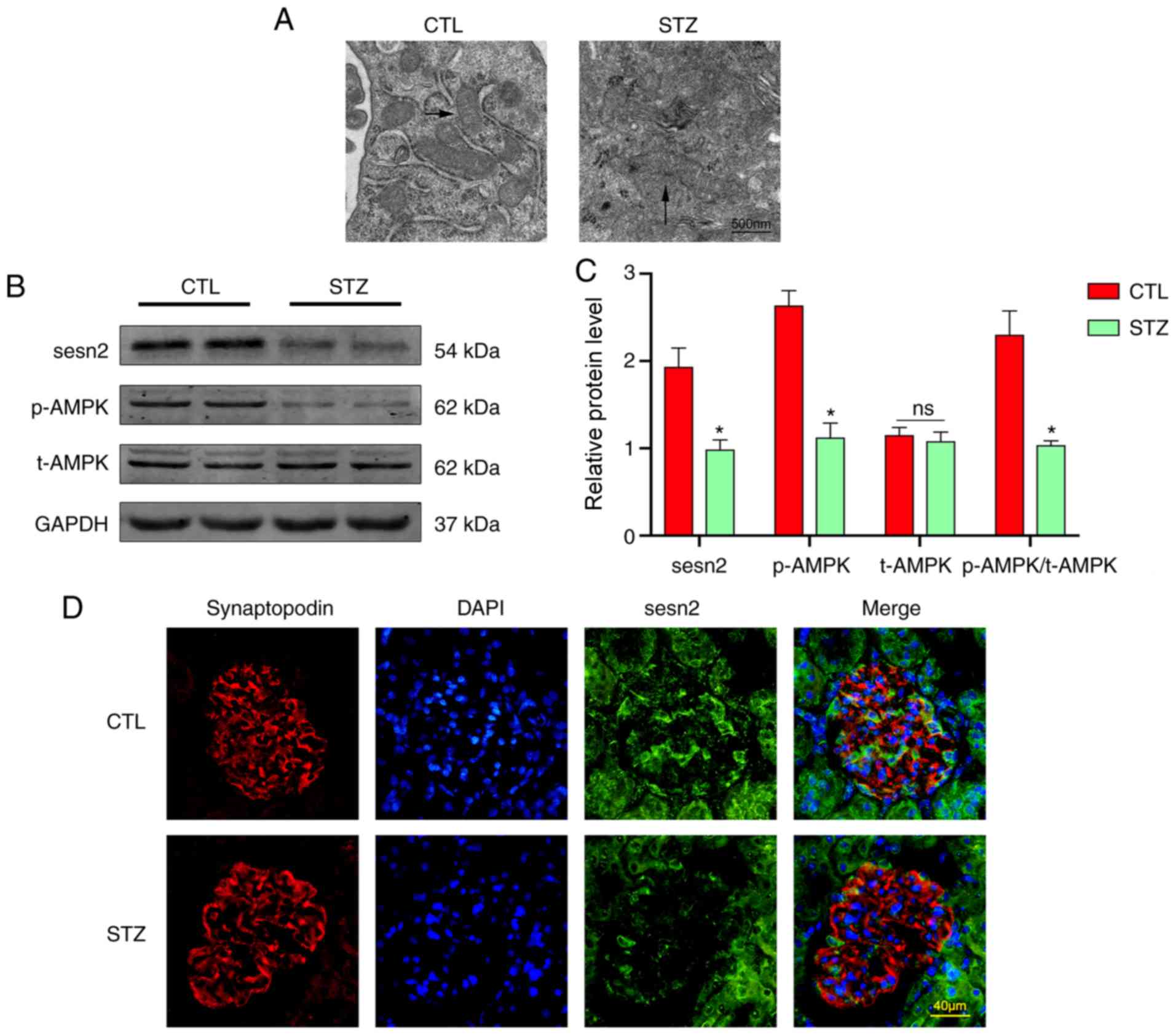
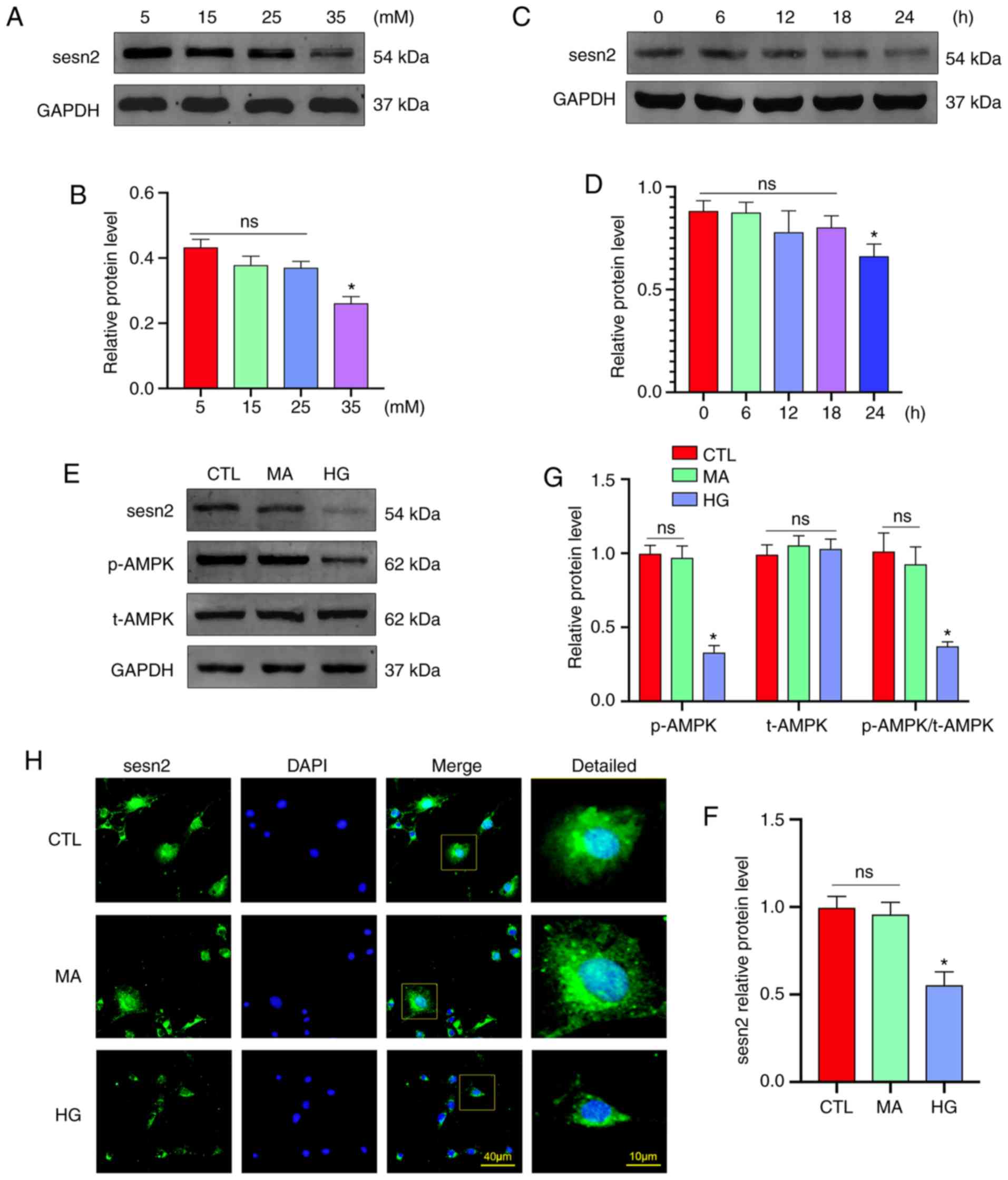
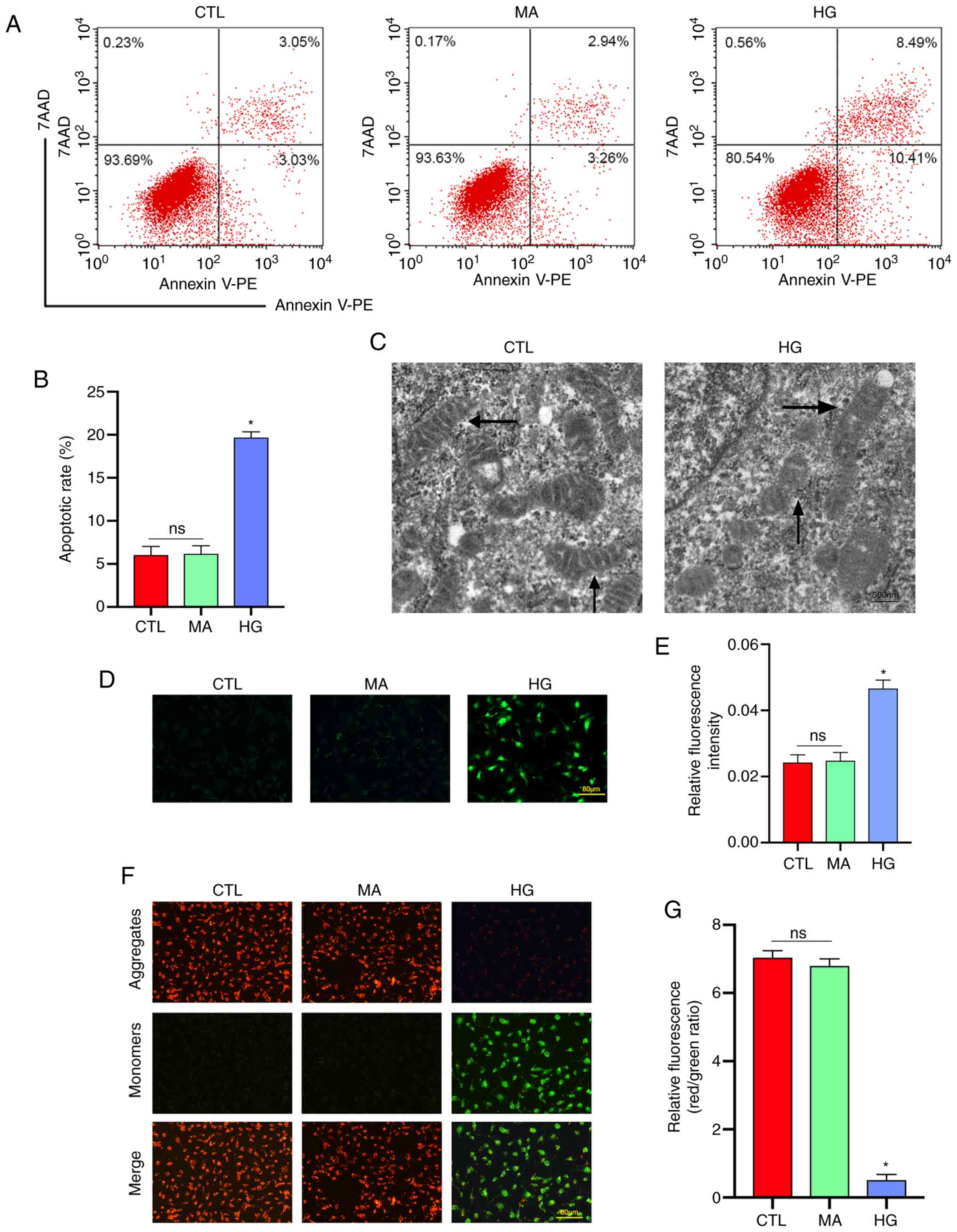
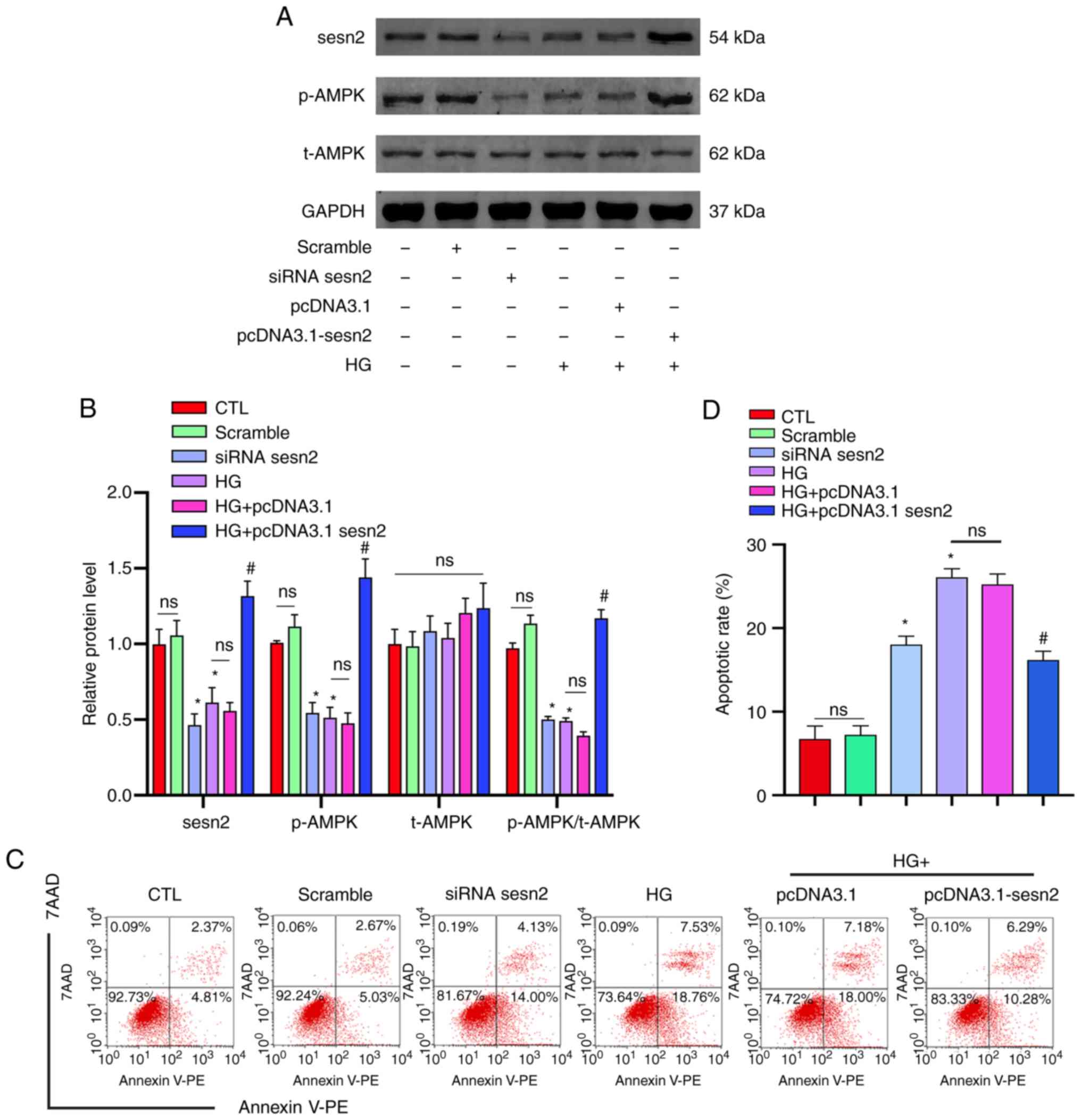
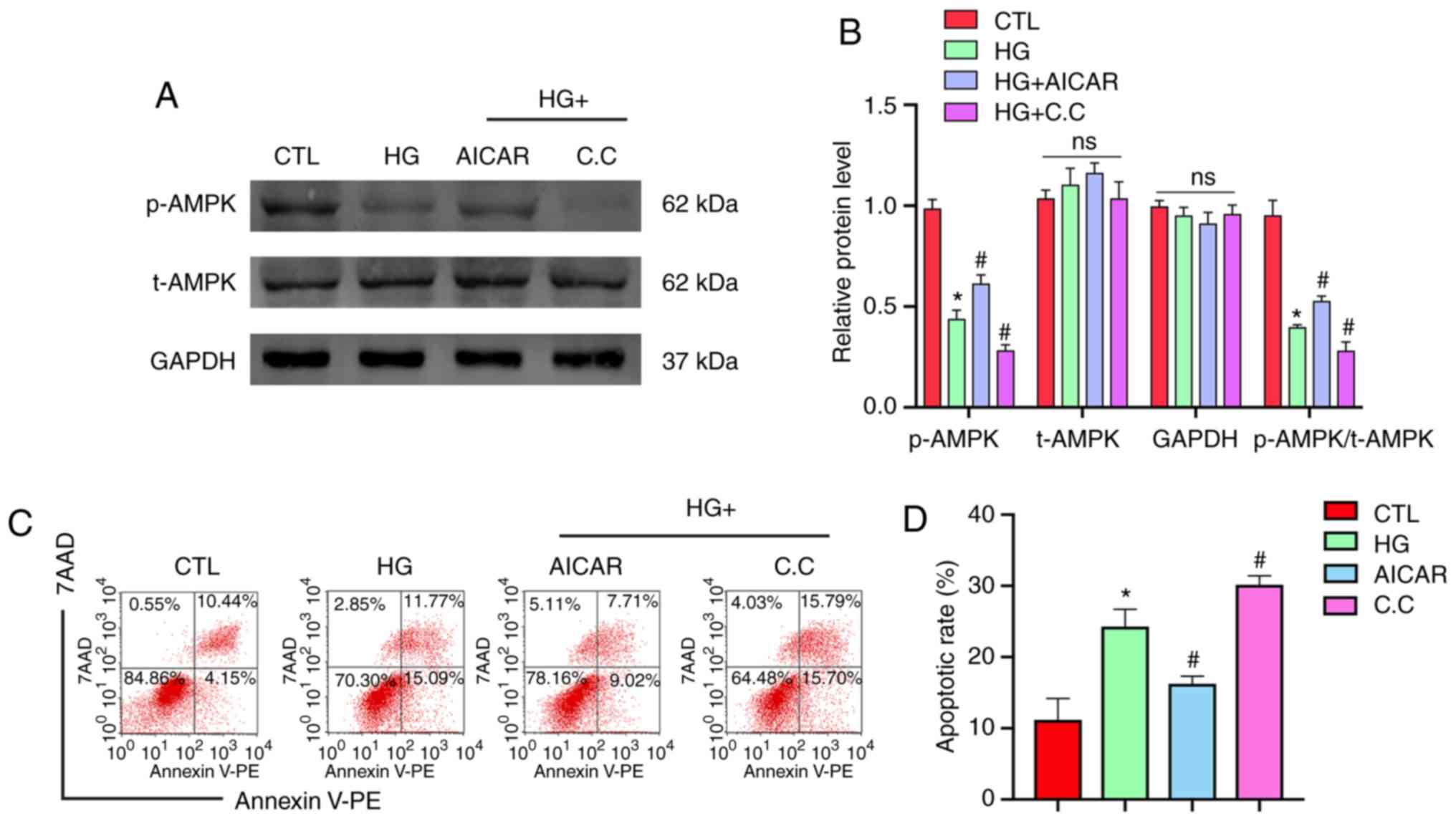
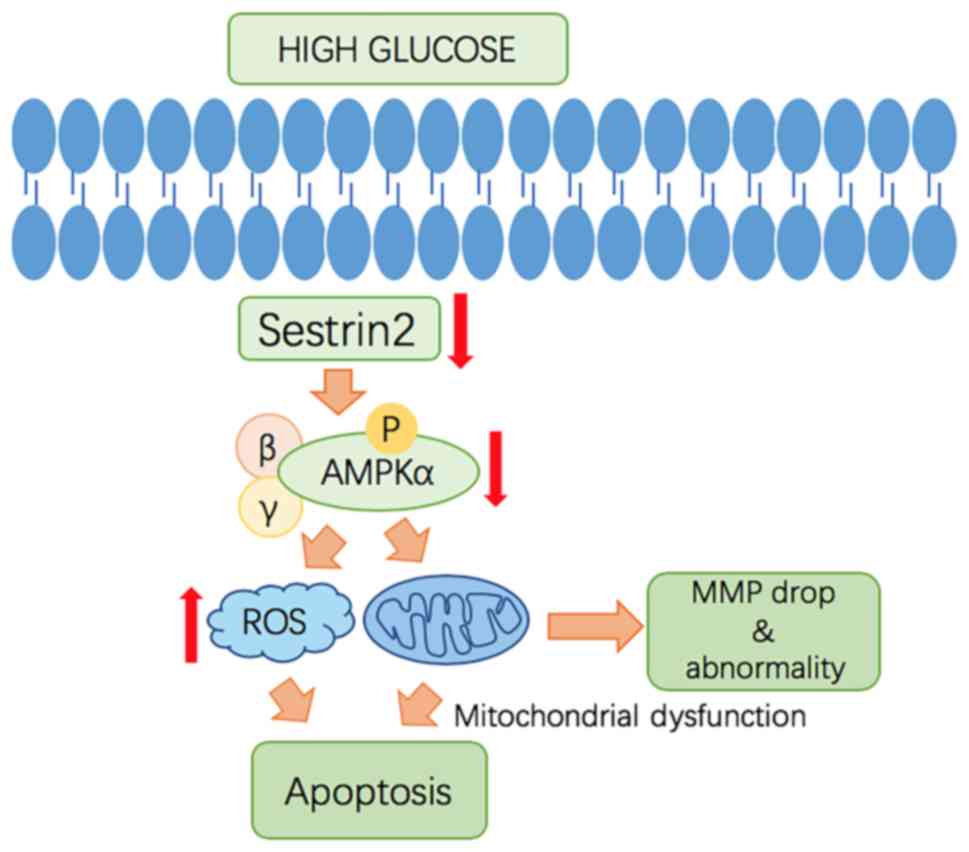
|
1
|
Locatelli F, Pozzoni P and Del Vecchio L:
Renal replacement therapy in patients with diabetes and end-stage
renal disease. J Am Soc Nephrol. 15(Suppl 1): S25–S29. 2004.
View Article : Google Scholar
|
|
2
|
Szrejder M and Piwkowska A: AMPK
signalling: Implications for podocyte biology in diabetic
nephropathy. Biol Cell. 111:109–120. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Jefferson JA, Alpers CE and Shankland SJ:
Podocyte biology for the bedside. Am J Kidney Dis. 58:835–845.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Higgins GC and Coughlan MT: Mitochondrial
dysfunction and mitophagy: The beginning and end to diabetic
nephropathy? Br J Pharmacol. 171:1917–1942. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Yuan Y, Huang S, Wang W, Wang Y, Zhang P,
Zhu C, Ding G, Liu B, Yang T and Zhang A: Activation of peroxisome
proliferator-activated receptor-ү coactivator 1α ameliorates
mitochondrial dysfunction and protects podocytes from
aldosterone-induced injury. Kidney Int. 82:771–789. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Granata S, Dalla Gassa A, Tomei P, Lupo A
and Zaza G: Mitochondria: A new therapeutic target in chronic
kidney disease. Nutr Metab (Lond). 12:492015. View Article : Google Scholar
|
|
7
|
Budanov AV, Lee JH and Karin M: Stressin’
Sestrins take an aging fight. EMBO Mol Med. 2:388–400. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Lee JH, Budanov AV and Karin M: Sestrins
orchestrate cellular metabolism to attenuate aging. Cell Metab.
18:792–801. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Budanov AV, Sablina AA, Feinstein E,
Koonin EV and Chumakov PM: Regeneration of peroxiredoxins by
p53-regulated sestrins, homologs of bacterial AhpD. Science.
304:596–600. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Budanov AV and Karin M: p53 target genes
sestrin1 and sestrin2 connect genotoxic stress and mTOR signaling.
Cell. 134:451–460. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Lee JH, Budanov AV, Park EJ, Birse R, Kim
TE, Perkins GA, Ocorr K, Ellisman MH, Bodmer R, Bier E and Karin M:
Sestrin as a feedback inhibitor of TOR that prevents age-related
pathologies. Science. 327:1223–1228. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Woo HA, Bae SH, Park S and Rhee SG:
Sestrin 2 is not a reductase for cysteine sulfinic acid of
peroxiredoxins. Antioxid Redox Signal. 11:739–745. 2009. View Article : Google Scholar
|
|
13
|
Bae SH, Sung SH, Oh SY, Lim JM, Lee SK,
Park YN, Lee HE, Kang D and Rhee SG: Sestrins activate Nrf2 by
promoting p62-dependent autophagic degradation of Keap1 and prevent
oxidative liver damage. Cell Metab. 17:73–84. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Budanov AV, Shoshani T, Faerman A, Zelin
E, Kamer I, Kalinski H, Gorodin S, Fishman A, Chajut A, Einat P, et
al: Identification of a novel stress-responsive gene Hi95 involved
in regulation of cell viability. Oncogene. 21:6017–6031. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Maiuri MC, Malik SA, Morselli E, Kepp O,
Criollo A, Mouchel PL, Carnuccio R and Kroemer G: Stimulation of
autophagy by the p53 target gene Sestrin2. Cell Cycle. 8:1571–1576.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Lee JH, Budanov AV, Talukdar S, Park EJ,
Park HL, Park HW, Bandyopadhyay G, Li N, Aghajan M, Jang I, et al:
Maintenance of metabolic homeostasis by Sestrin2 and Sestrin3. Cell
Metab. 16:311–321. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Dugan LL, You YH, Ali SS, Diamond-Stanic
M, Miyamoto S, DeCleves AE, Andreyev A, Quach T, Ly S, Shekhtman G,
et al: AMPK dysregulation promotes diabetes-related reduction of
superoxide and mitochondrial function. J Clin Invest.
123:4888–4899. 2013. View
Article : Google Scholar : PubMed/NCBI
|
|
18
|
Shrestha B and Dunn L: The declaration of
helsinki on medical research involving human subjects: A review of
seventh revision. J Nepal Health Res Conc. 17:548–552. 2020.
|
|
19
|
Hu J, Yang Q, Chen Z, Liang W, Feng J and
Ding G: Small GTPase Arf6 regulates diabetes-induced cholesterol
accumulation in podocytes. J Cell Physiol. 234:23559–23570. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Cammisotto PG and Bendayan M: Adiponectin
stimulates phosphorylation of AMP-activated protein kinase alpha in
renal glomeruli. J Mol Histol. 39:579–584. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Cai X, Bao L, Ren J, Li Y and Zhang Z:
Grape seed procyanidin B2 protects podocytes from high
glucose-induced mitochondrial dysfunction and apoptosis via the
AMPK-SIRT1-PGC-1α axis in vitro. Food Funct. 7:805–815. 2016.
View Article : Google Scholar
|
|
22
|
Madhavi YV, Gaikwad N, Yerra VG, Kalvala
AK, Nanduri S and Kumar A: Targeting AMPK in diabetes and diabetic
complications: Energy homeostasis, autophagy and mitochondrial
health. Curr Med Chem. 26:5207–5229. 2019. View Article : Google Scholar
|
|
23
|
Ma Y and Li J: Metabolic shifts during
aging and pathology. Compr Physiol. 5:667–686. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Butt A and Riaz S: Study of protein
profiling of human urine in diabetic hypertensive nephropathy
versus normal healthy controls. Diabetes Technol Ther. 12:379–386.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Piwkowska A, Rogacka D, Audzeyenka I,
Jankowski M and Angielski S: High glucose concentration affects the
oxidant-antioxidant balance in cultured mouse podocytes. J Cell
Biochem. 112:1661–1672. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Ma Y, Yang Q, Chen X, Liang W, Ren Z and
Ding G: c-Abl contributes to glucose-promoted apoptosis via p53
signaling pathway in podocytes. Diabetes Res Clin Pract.
113:171–178. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Redza-Dutordoir M and Averill-Bates DA:
Activation of apoptosis signalling pathways by reactive oxygen
species. Biochim Biophys Acta. 1863.2977–2992. 2016.
|
|
28
|
Galvan DL, Green NH and Danesh FR: The
hallmarks of mitochondrial dysfunction in chronic kidney disease.
Kidney Int. 92:1051–1057. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Halestrap AP: What is the mitochondrial
permeability transition pore? J Mol Cell Cardiol. 46:821–831. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Orrenius S, Gogvadze V and Zhivotovsky B:
Calcium and mitochondria in the regulation of cell death. Biochem
Biophys Res Commun. 460:72–81. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Green DR and Reed JC: Mitochondria and
apoptosis. Science. 281:1309–1312. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Quan N, Wang L, Chen X, Luckett C, Cates
C, Rousselle T, Zheng Y and Li J: Sestrin2 prevents age-related
intolerance to post myocardial infarction via AMPK/PGC-1α pathway.
J Mol Cell Cardiol. 115:170–178. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Hwang HJ, Kim JW, Chung HS, Seo JA, Kim
SG, Kim NH, Choi KM, Baik SH and Yoo HJ: Knockdown of Sestrin2
increases lipopolysaccharide-induced oxidative stress, apoptosis,
and fibrotic reactions in H9c2 cells and heart tissues of Mice via
an AMPK-dependent mechanism. Mediators Inflamm. 2018:6209140. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Kim HJ, Joe Y, Kim SK, Park SU, Park J,
Chen Y, Kim J, Ryu J, Cho GJ, Surh YJ, et al: Carbon monoxide
protects against hepatic steatosis in mice by inducing sestrin-2
via the PERK-eIF2α-ATF4 pathway. Free Radic Biol Med. 110:81–91.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Liu X, Niu Y, Yuan H, Huang J and Fu L:
AMPK binds to Sestrins and mediates the effect of exercise to
increase insulin-sensitivity through autophagy. Metabolism.
64:658–665. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Egan DF, Shackelford DB, Mihaylova MM,
Gelino S, Kohnz RA, Mair W, Vasquez DS, Joshi A, Gwinn DM, Taylor
R, et al: Phosphorylation of ULK1 (hATG1) by AMP-activated protein
kinase connects energy sensing to mitophagy. Science. 331:456–461.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Toyama EQ, Herzig S, Courchet J, Lewis TL
Jr, Losón OC, Hellberg K, Young NP, Chen H, Polleux F, Chan DC and
Shaw RJ: Metabolism. AMP-activated protein kinase mediates
mitochondrial fission in response to energy stress. Science.
351:275–281. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Zong H, Ren JM, Young LH, Pypaert M, Mu J,
Birnbaum MJ and Shulman GI: AMP kinase is required for
mitochondrial biogenesis in skeletal muscle in response to chronic
energy deprivation. Proc Natl Acad Sci USA. 99:15983–15987. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Jager S, Handschin C, St-Pierre J and
Spiegelman BM: AMP-activated protein kinase (AMPK) action in
skeletal muscle via direct phosphorylation of PGC-1alpha. Proc Natl
Acad Sci USA. 104:12017–12022. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Canto C and Auwerx J: PGC-1alpha, SIRT1
and AMPK, an energy sensing network that controls energy
expenditure. Curr Opin Lipidol. 20:98–105. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Ventura-Clapier R, Garnier A and Veksler
V: Transcriptional control of mitochondrial biogenesis: The central
role of PGC-1alpha. Cardiovasc Res. 79:208–217. 2008. View Article : Google Scholar : PubMed/NCBI
|