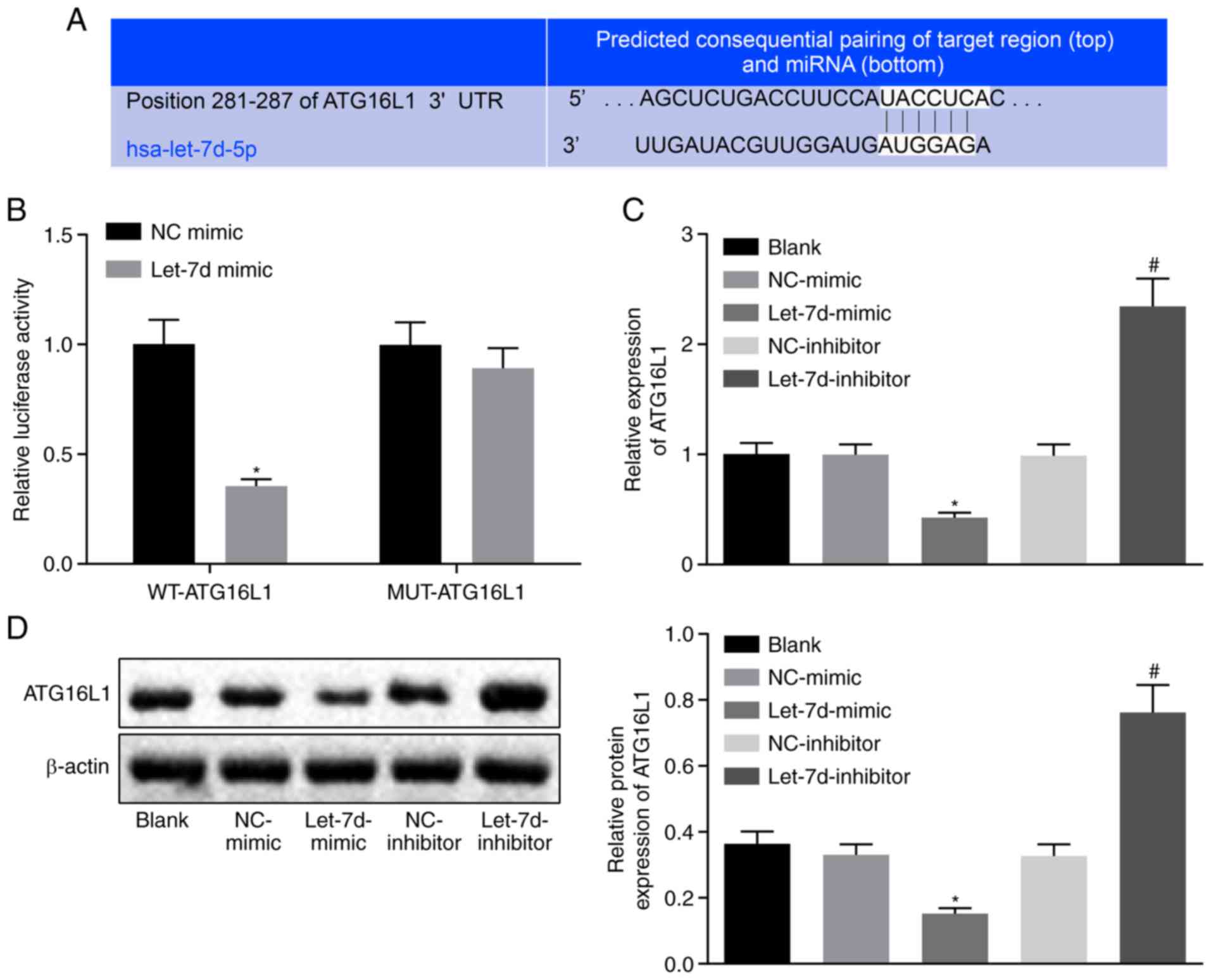
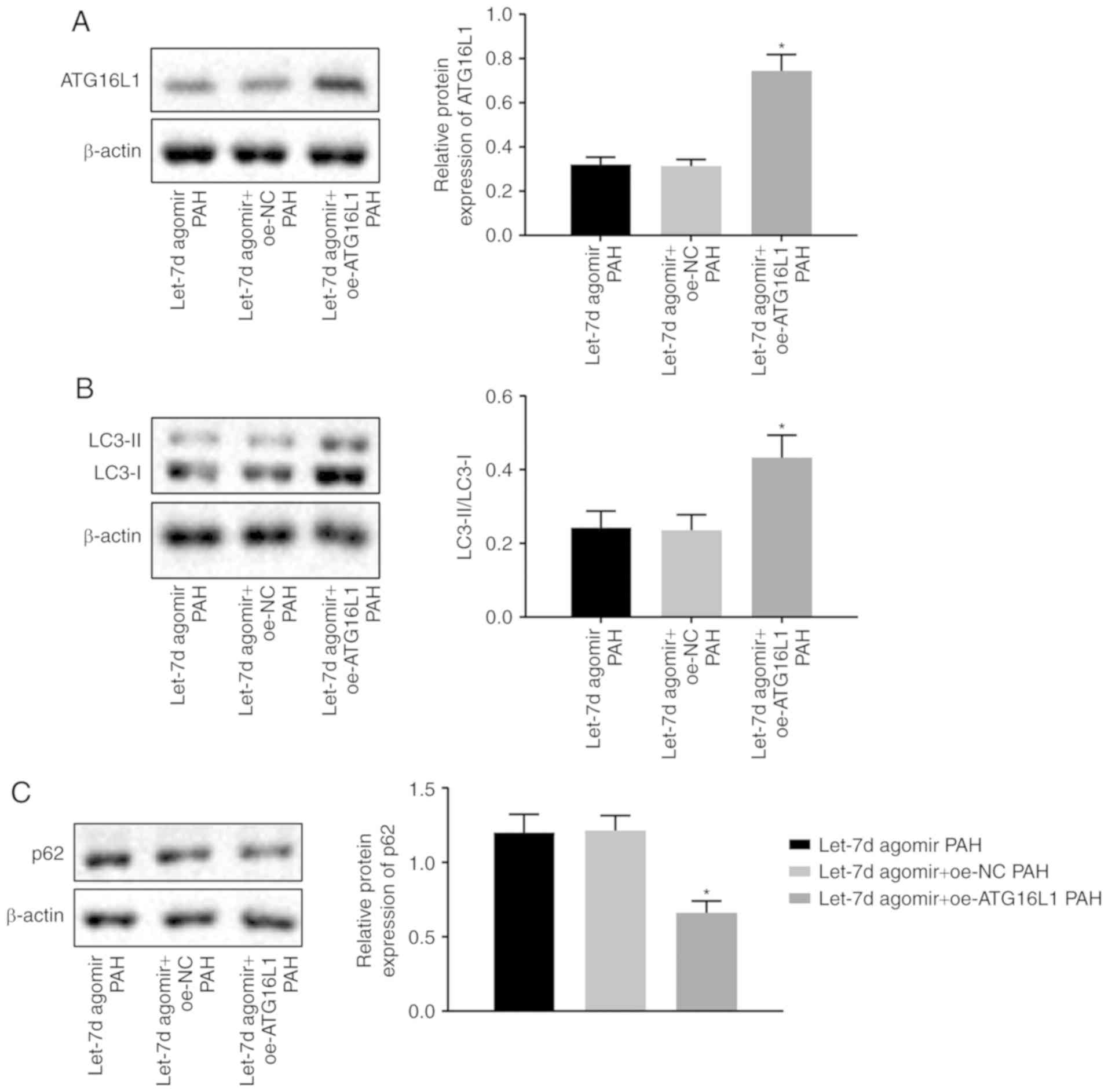
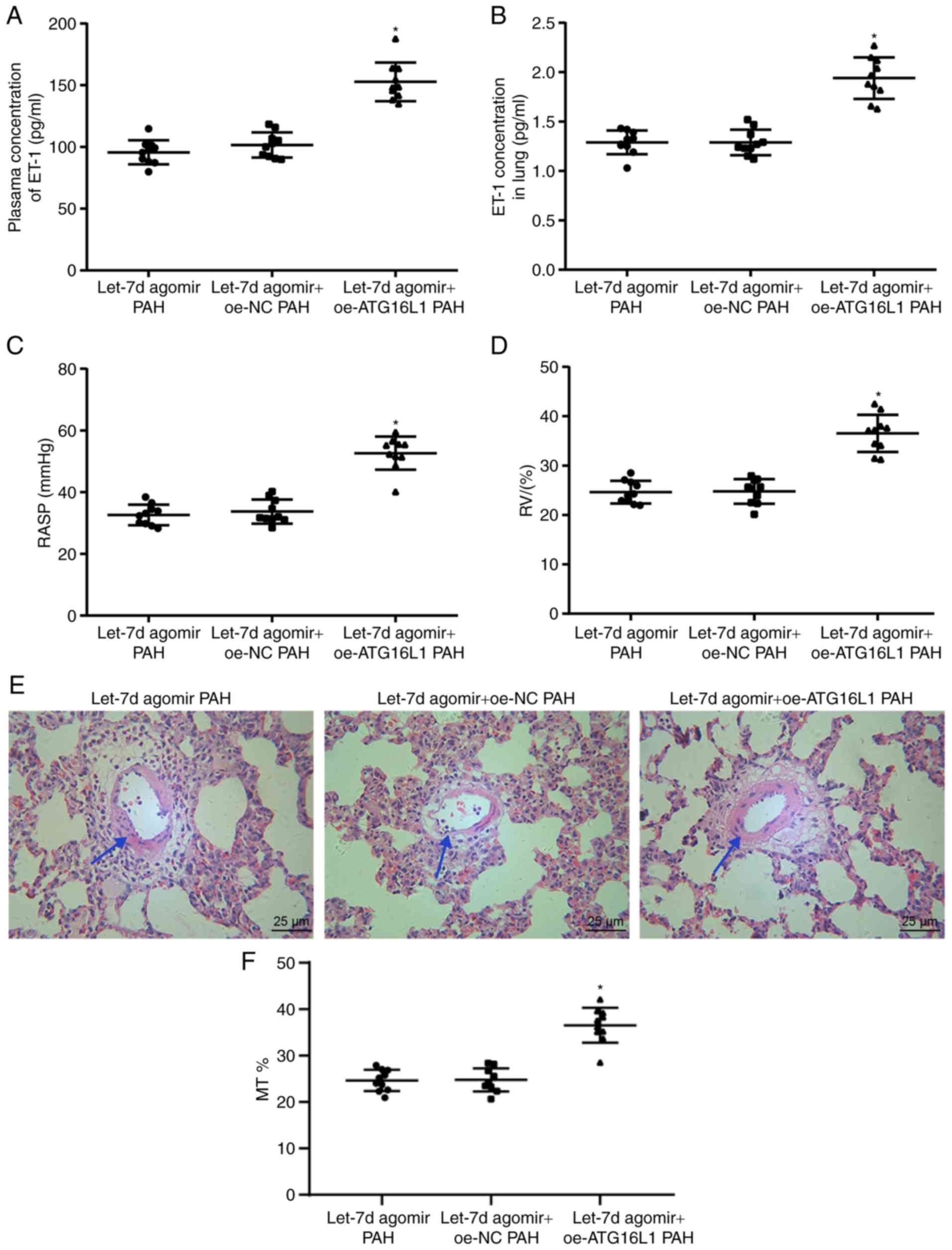
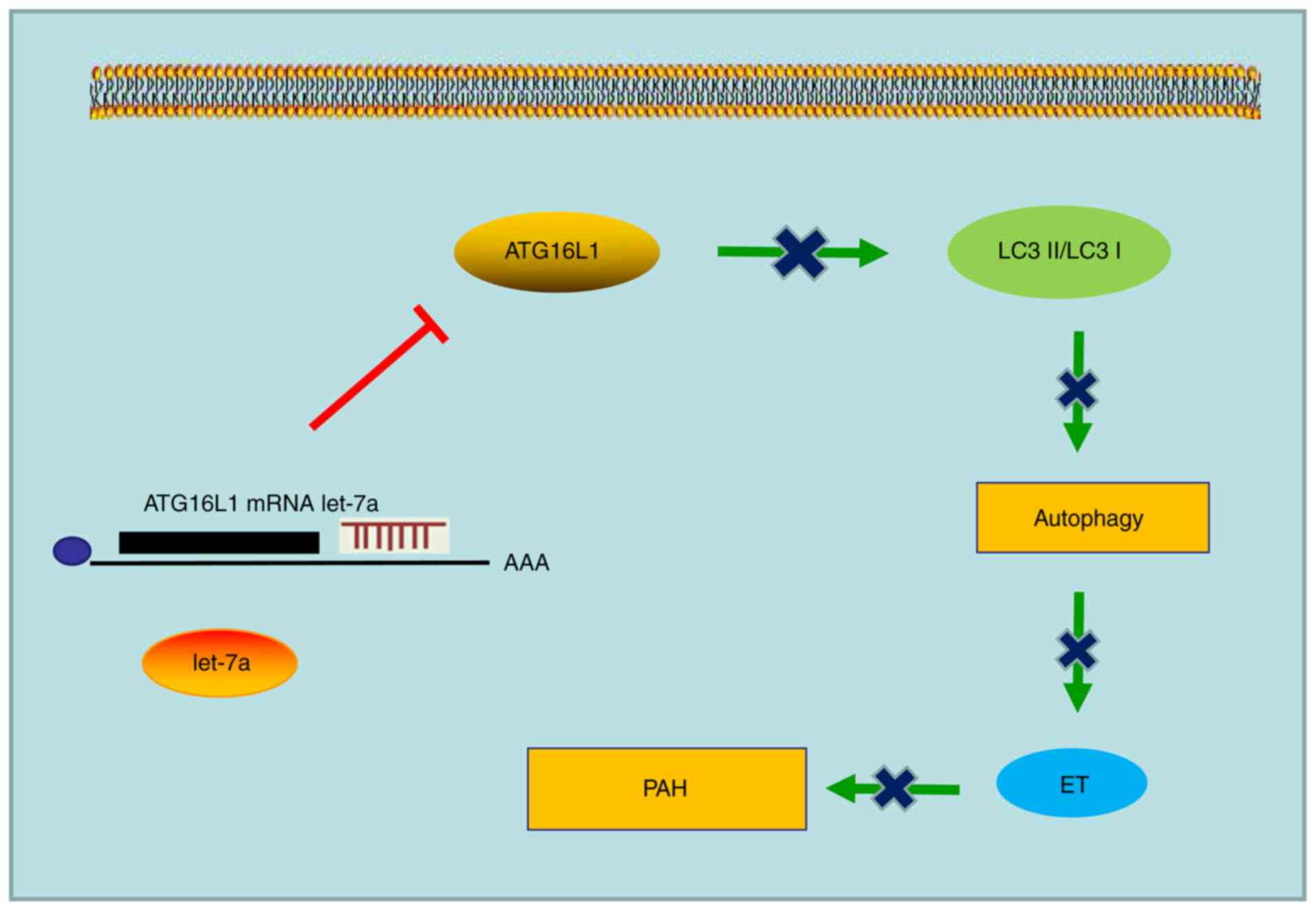
|
1
|
Steele P, Strange G, Wlodarczyk J, Dalton
B, Stewart S, Gabbay E and Keogh A: Hemodynamics in pulmonary
arterial hypertension (PAH): Do they explain long-term clinical
outcomes with PAH-specific therapy? BMC Cardiovasc Disord.
10:92010. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Preston IR, Roberts KE, Miller DP, Sen GP,
Selej M, Benton WW, Hill NS and Farber HW: Effect of warfarin
treatment on survival of patients with pulmonary arterial
hypertension (PAH) in the registry to evaluate early and long-term
PAH disease management (REVEAL). Circulation. 132:2403–2411. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Sikirica M, Iorga SR, Bancroft T and
Potash J: The economic burden of pulmonary arterial hypertension
(PAH) in the US on payers and patients. BMC Health Serv Res.
14:6762014. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Mehta J, Parthasarathy PT, Lockey R and
Kolliputi N: New hope for a microRNA therapy for pulmonary arterial
hypertension. Front Genet. 4:1372013. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Rubin LJ, Galie N, Grimminger F, Grunig E,
Humbert M, Jing ZC, Keogh A, Langleben D, Fritsch A, Menezes F, et
al: Riociguat for the treatment of pulmonary arterial hypertension:
A long-term extension study (PATENT-2). Eur Respir J. 45:1303–1313.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Meloche J, Le Guen M, Potus F, Vinck J,
Ranchoux B, Johnson I, Antigny F, Tremblay E, Breuils-Bonnet S,
Perros F, et al: MiR-223 reverses experimental pulmonary arterial
hypertension. Am J Physiol Cell Physiol. 309:C363–C372. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Potus F, Graydon C, Provencher S and
Bonnet S: Vascular remodeling process in pulmonary arterial
hypertension, with focus on miR-204 and miR-126 (2013 grover
conference series). Pulm Circ. 4:175–184. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Jiang L, Wang Y, Rong Y, Xu L, Chu Y,
Zhang Y and Yao Y: MiR-1179 promotes cell invasion through
SLIT2/ROBO1 axis in esophageal squamous cell carcinoma. Int J Clin
Exp Pathol. 8:319–327. 2015.PubMed/NCBI
|
|
9
|
Courboulin A, Paulin R, Giguere NJ,
Saksouk N, Perreault T, Meloche J, Paquet ER, Biardel S, Provencher
S, Cote J, et al: Role for miR-204 in human pulmonary arterial
hypertension. J Exp Med. 208:535–548. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Stevens HC, Deng L, Grant JS, Pinel K,
Thomas M, Morrell NW, MacLean MR, Baker AH and Denby L: Regulation
and function of miR-214 in pulmonary arterial hypertension. Pulm
Circ. 6:109–117. 2016. View
Article : Google Scholar : PubMed/NCBI
|
|
11
|
Ramberg H, Alshbib A, Berge V, Svindland A
and Tasken KA: Regulation of PBX3 expression by androgen and Let-7d
in prostate cancer. Mol Cancer. 10:502011. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Dai X and Cai Y: Down-regulation of
microRNA let-7d inhibits the proliferation and invasion of
trophoblast cells in preeclampsia. J Cell Biochem. 119:1141–1151.
2018. View Article : Google Scholar
|
|
13
|
Bao MH, Feng X, Zhang YW, Lou XY, Cheng Y
and Zhou HH: Let-7 in cardiovascular diseases, heart development
and cardiovascular differentiation from stem cells. Int J Mol Sci.
14:23086–23102. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Han J, Pan XY, Xu Y, Xiao Y, An Y, Tie L,
Pan Y and Li XJ: Curcumin induces autophagy to protect vascular
endothelial cell survival from oxidative stress damage. Autophagy.
8:812–825. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Kim JK, Yuk JM, Kim SY, Kim TS, Jin HS,
Yang CS and Jo EK: MicroRNA-125a inhibits autophagy activation and
antimicrobial responses during mycobacterial infection. J Immunol.
194:5355–5365. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Nishimura T, Kaizuka T, Cadwell K, Sahani
MH, Saitoh T, Akira S, Virgin HW and Mizushima N: FIP200 regulates
targeting of Atg16L1 to the isolation membrane. EMBO Rep.
14:284–291. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Chen R, Li X, He B and Hu W: MicroRNA-410
regulates autophagy-related gene ATG16L1 expression and enhances
chemosensitivity via autophagy inhibition in osteosarcoma. Mol Med
Rep. 15:1326–1334. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Magne J, Gustafsson P, Jin H, Maegdefessel
L, Hultenby K, Wernerson A, Eriksson P, Franco-Cereceda A, Kovanen
PT, Goncalves I and Ehrenborg E: ATG16L1 expression in carotid
atherosclerotic plaques is associated with plaque vulnerability.
Arterioscler Thromb Vasc Biol. 35:1226–1235. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Chin KM, Rubin LJ, Channick R, Di Scala L,
Gaine S, Galie N, Ghofrani HA, Hoeper MM, Lang IM, McLaughlin VV,
et al: Association of N-terminal pro brain natriuretic peptide and
long-term outcome in patients with pulmonary arterial hypertension.
Circulation. 139:2440–2450. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Ruiz-Irastorza G, Garmendia M, Villar I,
Egurbide MV and Aguirre C: Pulmonary hypertension in systemic lupus
erythematosus: Prevalence, predictors and diagnostic strategy.
Autoimmun Rev. 12:410–415. 2013. View Article : Google Scholar
|
|
21
|
Zhai C, Shi W, Feng W, Zhu Y, Wang J, Li
S, Yan X, Wang Q, Zhang Q, Chai L, et al: Activation of AMPK
prevents monocrotaline-induced pulmonary arterial hypertension by
suppression of NF-kappaB-mediated autophagy activation. Life Sci.
208:87–95. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Lee SJ, Smith A, Guo L, Alastalo TP, Li M,
Sawada H, Liu X, Chen ZH, Ifedigbo E, Jin Y, et al: Autophagic
protein LC3B confers resistance against hypoxia-induced pulmonary
hypertension. Am J. Respir Crit Care Med. 183:649–658. 2011.
View Article : Google Scholar
|
|
23
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar
|
|
24
|
Montani D, Souza R, Binkert C, Fischli W,
Simonneau G, Clozel M and Humbert M: Endothelin-1/endothelin-3
ratio: A potential prognostic factor of pulmonary arterial
hypertension. Chest. 131:101–108. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Humbert M, Sitbon O, Chaouat A, Bertocchi
M, Habib G, Gressin V, Yaici A, Weitzenblum E, Cordier JF, Chabot
F, et al: Survival in patients with idiopathic, familial, and
anorex-igen-associated pulmonary arterial hypertension in the
modern management era. Circulation. 122:156–163. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Lee WT, Ling Y, Sheares KK, Pepke-Zaba J,
Peacock AJ and Johnson MK: Predicting survival in pulmonary
arterial hypertension in the UK. Eur Respir J. 40:604–611. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Caruso P, Dempsie Y, Stevens HC, McDonald
RA, Long L, Lu R, White K, Mair KM, McClure JD, Southwood M, et al:
A role for miR-145 in pulmonary arterial hypertension: Evidence
from mouse models and patient samples. Circ Res. 111:290–300. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Huleihel L, Ben-Yehudah A, Milosevic J, Yu
G, Pandit K, Sakamoto K, Yousef H, LeJeune M, Coon TA, Redinger CJ,
et al: Let-7d microRNA affects mesenchymal phenotypic properties of
lung fibroblasts. Am J Physiol Lung Cell Mol Physiol.
306:L534–L542. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Pandit KV, Corcoran D, Yousef H,
Yarlagadda M, Tzouvelekis A, Gibson KF, Konishi K, Yousem SA, Singh
M, Handley D, et al: Inhibition and role of let-7d in idiopathic
pulmonary fibrosis. Am J Respir Crit Care Med. 182:220–229. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Liang H, Pan Z, Zhao X, Liu L, Sun J, Su
X, Xu C, Zhou Y, Zhao D, Xu B, et al: LncRNA PFL contributes to
cardiac fibrosis by acting as a competing endogenous RNA of let-7d.
Theranostics. 8:1180–1194. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Wang X, Wang HX, Li YL, Zhang CC, Zhou CY,
Wang L, Xia YL, Du J and Li HH: MicroRNA Let-7i negatively
regulates cardiac inflammation and fibrosis. Hypertension.
66:776–785. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Zhao B, Han H, Chen J, Zhang Z, Li S, Fang
F, Zheng Q, Ma Y, Zhang J, Wu N and Yang Y: MicroRNA let-7c
inhibits migration and invasion of human non-small cell lung cancer
by targeting ITGB3 and MAP4K3. Cancer Lett. 342:43–51. 2014.
View Article : Google Scholar
|
|
33
|
Wang L, Guo LJ, Liu J, Wang W, Yuan JX,
Zhao L, Wang J and Wang C: MicroRNA expression profile of pulmonary
artery smooth muscle cells and the effect of let-7d in chronic
thrombo-embolic pulmonary hypertension. Pulm Circ. 3:654–664. 2013.
View Article : Google Scholar
|
|
34
|
Jiang F: Autophagy in vascular endothelial
cells. Clin Exp Pharmacol Physiol. 43:1021–1028. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Guo L, Zhao J, Qu Y, Yin R, Gao Q, Ding S,
Zhang Y, Wei J and Xu G: MicroRNA-20a inhibits autophagic process
by targeting ATG7 and ATG16L1 and favors mycobacterial survival in
macrophage cells. Front Cell Infect Microbiol. 6:1342016.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Yang X, Zhong X, Tanyi JL, Shen J, Xu C,
Gao P, Zheng TM, DeMichele A and Zhang L: Mir-30d regulates
multiple genes in the autophagy pathway and impairs autophagy
process in human cancer cells. Biochem Biophys Res Commun.
431:617–622. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Xu Y, Huang X, Xie J, Chen Y, Fu J and
Wang L: Let-7i-induced Atg4B suppression is essential for autophagy
of placental trophoblast in preeclampsia. J Cell Physiol.
232:2581–2589. 2017. View Article : Google Scholar
|
|
38
|
Shao D, Park JE and Wort SJ: The role of
endothelin-1 in the pathogenesis of pulmonary arterial
hypertension. Pharmacol Res. 63:504–511. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Jacobs ME, Wingo CS and Cain BD: An
emerging role for microRNA in the regulation of endothelin-1. Front
Physiol. 4:222013. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Li D, Yang P, Xiong Q, Song X, Yang X, Liu
L, Yuan W and Rui YC: MicroRNA-125a/b-5p inhibits endothelin-1
expression in vascular endothelial cells. J Hypertens.
28:1646–1654. 2010. View Article : Google Scholar : PubMed/NCBI
|