Zinc is an essential metal being involved in a
variety of biological processes due to its function as a cofactor,
signaling molecule, and structural element. It is involved in the
regulation of carbohydrate and lipid metabolism, as well as the
functioning of the reproductive, cardiovascular, and nervous system
(1). At the same time, the most
critical role of zinc is demonstrated for the immune system.
Briefly, zinc regulates proliferation, differentiation, maturation,
and functioning of leukocytes and lymphocytes (2). Zinc plays a signaling role involved
in the modulation of inflammatory responses (3). It is also a component of nutritional
immunity (4). Correspondingly,
alteration of zinc status significantly affects immune response
resulting in increased susceptibility to inflammatory and
infectious diseases including acquired immune deficiency syndrome,
measles, malaria, tuberculosis, and pneumonia (5). Earlier data demonstrate that
populational Zn status is associated with the prevalence of
respiratory tract infections in children and adults (6,7).
In view of the high prevalence of zinc deficiency
worldwide (up to 17%), its impact on population health is
considered as a significant issue (8). Moreover, certain groups of people,
including infants, especially preterm ones, and elderly, are
considered to be at high risk of zinc deficiency and its adverse
effects (9).
Under zinc deficiency condition, organisms are more
susceptible to toxin-producing bacteria or enteroviral pathogens
that activate guanylate and adenylate cyclases, stimulating
chloride secretion, causing diarrhea and diminishing absorption of
nutrients, thus exacerbating an already compromised mineral status.
In addition, zinc deficiency may impair the absorption of water and
electrolytes, delaying the termination of normally self-limiting
gastrointestinal disease episodes (10). During chronic deficiency, the
production of pro-inflammatory cytokines increases, influencing the
outcome of a large number of inflammatory, metabolic,
neurodegenerative and immune diseases (11). Diseases such as rheumatoid
arthritis, diabetes (12),
atherosclerosis and obesity (13), impaired cognitive function
(14), as well as age-related
macular degeneration (AMD) may be due to zinc deficiency, worsening
chronic inflammation and triggering oxidative stress.
Coronaviridae were considered as the etiological
agent in 6-29% of respiratory infections (15,16), although the severity of the
disease varies significantly on the particular virus and its
virulence (17). The viruses from
the Coronaviridae family are zoonotic viruses that can be
transmitted from animals to humans. The bat is considered the
reservoir for these viruses, but other intermediate animals can
also transmit the virus to humans (18). COVID-19 is a coronavirus disease
caused by the novel 2019-nCoV virus (now called SARS-CoV-2) that
appeared for the first time in Wuhan, China at the end of 2019
(19). Despite a close relation
other two highly pathogenic coronaviruses, MERS-CoV and SARS-CoV
(20), SARS-CoV-2 expanded to the
majority of countries (21). On
11 March 2020, WHO characterized COVID-19 as a pandemic (22). Currently, the prevalence of
COVID-19 exceeds 1,521,200 cases resulting in 92,700 deaths
worldwide (23).
COVID-19 predominantly affects the respiratory
system resulting in pneumonia and acute respiratory distress
syndrome (24), leading to the
requirement of mechanical ventilation (25). In turn, advanced age, acute
respiratory distress syndrome (ARDS) and mechanical ventilation are
known to be associated with higher COVID-19 mortality (26). The risk is also increased by
modern life in which individuals are exposed to a multitude of
chemicals, even in low doses that in the long-term predispose to
chronic diseases and metabolic disturbances (27-31). Preexisting chronic metabolic
diseases including diabetes, cardiovascular diseases (32), and obesity (33) are considered as risk factors for
increased COVID-19 susceptibility and mortality. It is proposed
that the elderly are at higher risk of COVID-19 due to impaired
immune function (34).
In view of lack of clinical data on preventive
and/or therapeutic efficiency of zinc in COVID-19, as well as
primary involvement of the respiratory system, in this review, we
will discuss recent clinical data on the role of zinc in protection
against bronchopulmonary infections, as well as the existing
indications of the direct impact of zinc on nCoV-2019.
In view of the global COVID-19 pandemic, potential
protective effect of zinc is of particular interest. Zinc is
considered as the potential supportive treatment in therapy of
COVID-19 infection due to its immune modulatory effect, as well as
direct antiviral effect (36).
However, the existing data will be only mechanistically discussed
in this review, as direct data on anti-COVID-19 effects of zinc are
absent to date.
Another Zn-related approach to modulation of
COVID-19 may include targeting Zn ions in the structure of viral
proteins. Particularly, it has been demonstrated that
disulfiram-induced Zn2+ release from papain-like
protease in MERS-CoV and SARS-CoV resulting in protein
destabilization (45). In view of
the presence of similar critical Zn-containing sites, Zn-ejector
drugs (e.g., disulfiram) may be considered as potential antiviral
agents (46) and components of
targeted oxidation strategy in anti-SARS-CoV-2 treatment (47).
SARS-CoV-2 similarly to SARS-CoV requires
angiotensin-converting enzyme 2 (ACE2) for entry into target cells
(48). Therefore, modulation of
ACE2 receptor was considered as the potential therapeutic strategy
in COVID-19 treatment (49).
Speth et al (50)
demonstrated that zinc exposure (100 µM) was shown to reduce
recombinant human ACE-2 activity in rat lungs. Although this
concentration is close to physiological values of total zinc, the
modulating effect of zinc on SARS-CoV-2-ACE2 interaction seem to be
only hypothetical (51).
Despite limited data on the direct effect of zinc on
SARS-CoV-2 and COVID-19, its antiviral effects were demonstrated in
other viral diseases. Zinc was shown to have a significant impact
on viral infections through modulation of viral particle entry,
fusion, replication, viral protein translation and further release
for a number of viruses including those involved in respiratory
system pathology (37,61). Specifically, increasing
intracellular Zn levels through application of Zn ionophores such
as pyrithione and hinokitiol significantly alters replication of
picornavirus, the leading cause of common cold (62). These findings generally correspond
to the earlier indications of suppressive effect of zinc on
rhinovirus replication originating from the early 1970s (63). In addition, Zn treatment was shown
to increase interferon α (IFNα) production by leukocytes (64) and potentiate its antiviral
activity in rhinovirus-infected cells (65). As antiviral activity of IFNα is
mediated through JAK1/STAT1 downstream signaling and up-regulation
of antiviral enzymes [e.g., latent ribonuclease (RNaseL) and
protein kinase RNA-activated (PKR)] involved in viral RNA
degradation and inhibition of viral RNA translation (66), recent findings allow to propose
that these mechanisms may be stimulated by Zn2+.
These findings along with the existing data on the
role of zinc in immunity raised interest to the potential use of
zinc in prevention and/or treatment of common cold. A systematic
review by Singh and Das (67)
published in Cochrane database revealed a significant reduction in
common cold duration, as well as the incidence rate ratio of
developing common cold (IRR=0.64 (95% CI: 0.47-0.88), P=0.006) in
response to zinc supplementation. The results of meta-analysis
demonstrated that Zn supplementation in the dose >75 mg/day
significantly reduced duration of common colds (68), with Zn acetate being the most
effective form (69).
Certain studies also revealed the association
between Zn status and respiratory syncytial virus (RSV) infection.
Particularly, it has been demonstrated that whole blood zinc was
significantly lower in children with RSV pneumonia (70). Impaired zinc metabolism in
perinatal alcohol exposure is associated with immunosuppression and
altered alveolar macrophage activity resulting in increased
susceptibility to RSV infection (71). In turn, Zn compounds were shown to
inhibit respiratory syncytial virus replication and RSV plaque
formation with a more than 1,000-fold reduction at 10 µm Zn
preincubation (72).
It is also notable that zinc deficiency was
associated with higher mortality and adverse long-term outcome in
influenza-MRSA bacterial superinfection (73), also underlining the importance of
considering the risk of bacterial coinfection.
Despite the presence of experimental findings on the
protective effect of zinc supplementation against respiratory virus
infections, clinical and epidemiological data are still to be
elaborated and systematized.
Zinc is essential for the immune system and elderly
people have an increased probability for zinc deficiency (74). Low Zn status was considered as the
potential risk factor for pneumonia in elderly. Particularly,
subjects with high serum Zn (>70 µg/dl, i.e., approx.
10.8 µmol/l) were characterized by reduced incidence of
pneumonia [0.52 (0.36, 0.76), P<0.001], as well as lower disease
duration and antibiotic administration as compared to low-Zn
(<70 µg/ml) group (75), being also related to all-cause
mortality (76). Serum Zn levels
were 15% lower in cases of community-acquired pneumonia and
advanced age, being also associated with pneumonia severity as
evaluated by CURB-65 scores (77). The incidence of severe pneumonia
was significantly higher in Irani patients with low Zn status,
although the mean duration of fever, tachycardia, and tachypnea
only tended to be longer, although not significant (78). Correspondingly, serum Zn levels
were found deficient at the onset of acute respiratory failure with
the lowest values observed in septic shock patients. However, no
association between serum Zn values and day-30 mortality or period
of stay in intensive care unit was observed (79).
The results of systematic analysis also confirmed
the efficiency of intake of at least 75 mg/day Zn in reduction of
pneumonia symptom duration but not severity, with the response
being more pronounced in adults than in children (80). At the same time, certain studies
failed to reveal any improvement in pneumonia when administered
along with standard antibiotic treatment, although the period of
supplementation was only 4 days (81).
In Indian patients high plasma zinc levels were
found to be associated with reduced mortality from sepsis as well
as lower 48-h SOFA scores (84).
Moreover, persistent low serum Zn levels were associated with
increased risk of recurrent sepsis in critically ill patients
(85).
Altogether, the existing data demonstrate an
association between zinc status and pneumonia in adults and
elderly, as well as its complications including respiratory
failure, ventilator-induced injury, and sepsis.
Initial reports have postulated nearly exceptional
susceptibility of elderly to SARS-CoV-2 infection allowing to
propose natural resistance to COVID-19 in children (86). However, detailed analysis of the
pediatric COVID-19 cases (87)
and the emerging Russian experience indicate that children may be
also severely affected by SARS-CoV-2. In view of high incidence of
Zn deficiency in infants, the existing data on the association
between Zn status and pneumonia in children is also discussed.
High incidence of pneumonia in developing countries
has been considered as the consequence of zinc deficiency in the
population (7). The incidence of
low serum zinc in children with severe pneumonia was 80% (88). Correspondingly, a 2-fold lower
level of serum Zn was observed in pediatric acute lower respiratory
infection patients (89).
Significantly lower serum zinc levels were observed in children
with pneumonia complicated by sepsis, mechanical ventilation, and
cases of lethality (90).
Generally, indications of low zinc status in children with
pneumonia provide a rationale for preventive Zn
supplementation.
Particularly, Zn supplementation in developing
countries reduced pneumonia morbidity by 19% (RR=0.81; 95% CI:
0.73, 0.90), whereas a 15% decrease in pneumonia-specific mortality
was not significant (91). A
recent systematic review and meta-analysis published in Cochrane
database demonstrated that Zn supplementation significantly reduced
the incidence and prevalence of pneumonia in children by 13 and 41%
(92).
In contrast to the demonstrated preventive effects
of Zn supplementation, data on the therapeutic effect of zinc in
treatment of childhood pneumonia are conflicting (93). Despite the earlier observed
reduction of treatment failure risk (94) and case fatality [RR=0.67 (95% CI:
0.24-0.85)] (95) in children
with severe pneumonia, a more recent study demonstrated that Zn
supplementation in 2-24 months old children with radiologically
verified pneumonia did not result in significant improvement of
risk reduction of treatment failure (96). Moreover, Zn supplementation in
Zn-deficient children with pneumonia until achievement of normal
serum Zn levels did not improve clinical appearance of the disease
(97).
A number of studies revealed the potential
efficiency of Zn supplementation in prevention of non-specified
acute lower respiratory infections including bronchitis,
bronchiolitis, pneumonitis. Specifically, supplementation with 10
mg zinc gluconate in Zn-deficient children resulted in a nearly
twofold reduction of the number of episodes of acute lower
respiratory infections as well as the time to recovery (98). In addition, Zn supplementation (30
mg/day) in Thai children significantly reduced severity of acute
lower respiratory tract infections resulting in faster disease
cessation and shorter hospital stay (99). A detailed meta-analysis
demonstrated that Zn supplementation significantly decreased the
incidence of acute lower respiratory infection defined according to
specific clinical criteria in children aged <5 years (100).
In parallel, the impact of Zn supplementation in
relation to upper respiratory tract infections was also
demonstrated. Particularly, the number of upper respiratory tract
infections in Colombian children was reduced by 73% in response to
supplementation with 5 mg Zn in a 12-month randomized clinical
trial (101). Certain studies
also revealed protective effect of zinc supplementation against
both acute upper and lower respiratory diseases in children
(102,103).
Inflammation plays the key role in COVID-19
pathogenesis both at local (pneumonia) and systemic (cytokine
storm) levels, and the search for adequate anti-inflammatory agents
is of particular importance (104).
Although the role of zinc in regulation of
inflammatory response was discussed in detail in a number of
reviews (2,5), certain aspects of the regulatory
role of zinc in pneumonia pathogenesis and lung inflammation are
still to be elucidated. However, the existing data clearly
demonstrate that Zn ions may possess anti-inflammatory effects in
pneumonia thus limiting tissue damage and systemic effects.
Specifically, Zn deficiency in rats resulted in a
significant increase in proinflammatory TNFα and VCAM-1 expression
and lung tissue remodeling, being partially reversed by Zn
supplementation (105). Zn
deficiency also resulted in a significant alteration of lung
epithelial cell barrier function through up-regulation of TNFα,
IFNγ, and FasR signaling and cellular apoptosis in vitro
(106). Zn deficiency was shown
to up-regulate acute phase response-related genes through
stimulation of JAK-STAT signaling in lungs under septic conditions
(107). Zinc and nitric oxide
(NO)-metallothioneine (MT)-Zn pathways were shown to mediate lung
injury in response to LPS or hyperoxia (108).
In turn, Zn pretreatment significantly reduced
LPS-induced pulmonary endothelial cell damage and increased cell
viability in vitro, as well as improved respiratory function
as assessed by blood oxygen pressure and saturation (109). It has been demonstrated that Zn
pretreatment significantly decreases LPS-induced neutrophil
recruitment to the lungs thus reducing acute lung injury in mice
(110).
It is also notable that zinc deficiency is
associated with inflammatory alterations of lung extracellular
matrix predisposing to fibrosis (111). This finding is of particular
interest in view of the presence of interstitial pulmonary fibrosis
in COVID-19 patients (112).
Certain studies revealed protective effect of zinc
against lung injury in systemic inflammation including sepsis.
Experimental data demonstrate that Zn deficiency increases
susceptibility to systemic inflammation and sepsis-induced organ
damage including lungs in a murine model of polymicrobial sepsis
(113). In a model of
polymicrobial sepsis Zn deficiency resulted in increased NF-κB p65
mRNA expression and production in lungs resulting in up-regulation
of target genes IL-1β, TNFα, and ICAM-1 (114), whereas Zn supplementation
reduced neutrophil infiltration and MPO-mediated oxidative damage
(115,116). Modulation of ERK1/2 and NF-κB
pathways was shown to be critical for protective effect of zinc in
lungs under septic conditions (117).
Correspondingly, patients with sepsis were
character-ized by low serum Zn levels that may occur due to
increased ZIP8 (SLC39A8) mRNA expression. Moreover, serum Zn
concentrations inversely correlated with both disease severity and
proinflammatory cytokines IL-6, IL-8, and TNFα (118). Reciprocal regulation of ZIP8 and
NF-κB expression in response to TNFα or LPS exposure was
demonstrated in lung epithelia and alveolar macrophages (119). In addition, ZIP8-deficient mice
were characterized by increased airway neutrophil infiltration and
elevated CXCL1 and IL-23 production (120).
Zn-mediated respiratory protection was also
demonstrated in models of toxic atmospheric pollutant exposure.
Particularly, Zn deficiency in agricultural organic dust-exposed
animals aggravated neutrophil migration and proinflammatory
cytokine (TNFα, IL-6, CXCL1) overproduction, as well as increased
IL-23 and CXCL1 expression by macrophages due to NF-κB activation
(121). In turn, Zn
supplementation in cigarette smoke exposed mice significantly
reduced the number of alveolar macrophages in bronchoalveolar
lavage (122).
The observed anti-inflammatory effects of Zn in lung
tissue seem to be mainly mediated by inhibition of NF-κB signaling
through PKA-induced inhibition of Raf-1 and IκB kinase β (IKKβ)
(123,124) or A20-dependent inhibition
(125). Moreover, Zn-induced
modulation of T-cell activity may also play a significant role in
limiting inflammatory response (126,127). Lastly, zinc was shown to
normalize the overproduction of proinflammatory cytokines induced
by zinc deficiency on the epigenetic level (124,128).
Although COVID-19 is characterized by viral
pneumonia caused by SARS-CoV-2 virus, bacterial co-infection may
represent a significant issue due its high incidence in H1N1
influenza-associated pneumonia (129). Specifically, human coronavirus
NL63 was associated with increased adherence of S.
pneumoniae to epithelial cells (130). In turn, Streptococcus
pneumoniae infection is considered as the most common cause of
pneumonia.
A number of studies demonstrated antibacterial
effect of zinc oxide nanoparticles (140). Particularly, ZnO was shown to
inhibit both growth and biofilm formation by S. pneumoniae
(141). Similar effect was
observed for other bacterial agents involved in etiology of
pneumonia, including K. pneumoniae (142), methicillin-resistant S.
aureus (143), and P.
aeruginosa (144). However,
the potential antibacterial application of ZnO-(NPs) may be limited
due to their toxicity to human lung cells (145), as well as impairment of
phagocytic activity of macrophages in bronchi and lungs (146).
The obtained data demonstrate that adequate zinc
status of the individual increases immune reactivity.
Correspondingly, inadequate zinc supply may predispose to
infectious diseases of upper and lower respiratory tract. Although
the therapeutic effects of Zn are considered as inconsistent, the
existing evidence-based data indicate efficiency of Zn
supplementation and improvement of Zn status in prevention of
pneumonia and its complications due to anti-inflammatory effect of
zinc.
Certain indirect indications of the potential
antiviral effect of Zn against nCoV-2019 exist, although their
biomedical relevance is yet to be studied. In view of recent data
on clinical course of the disease, it appears that adequate Zn
status may possess protective effect as adjuvant therapy of
COVID-19 through reducing lung inflammation, improvement of
mucociliary clearance, prevention of ventilator-induced lung
injury, modulation of antibacterial and antiviral immunity
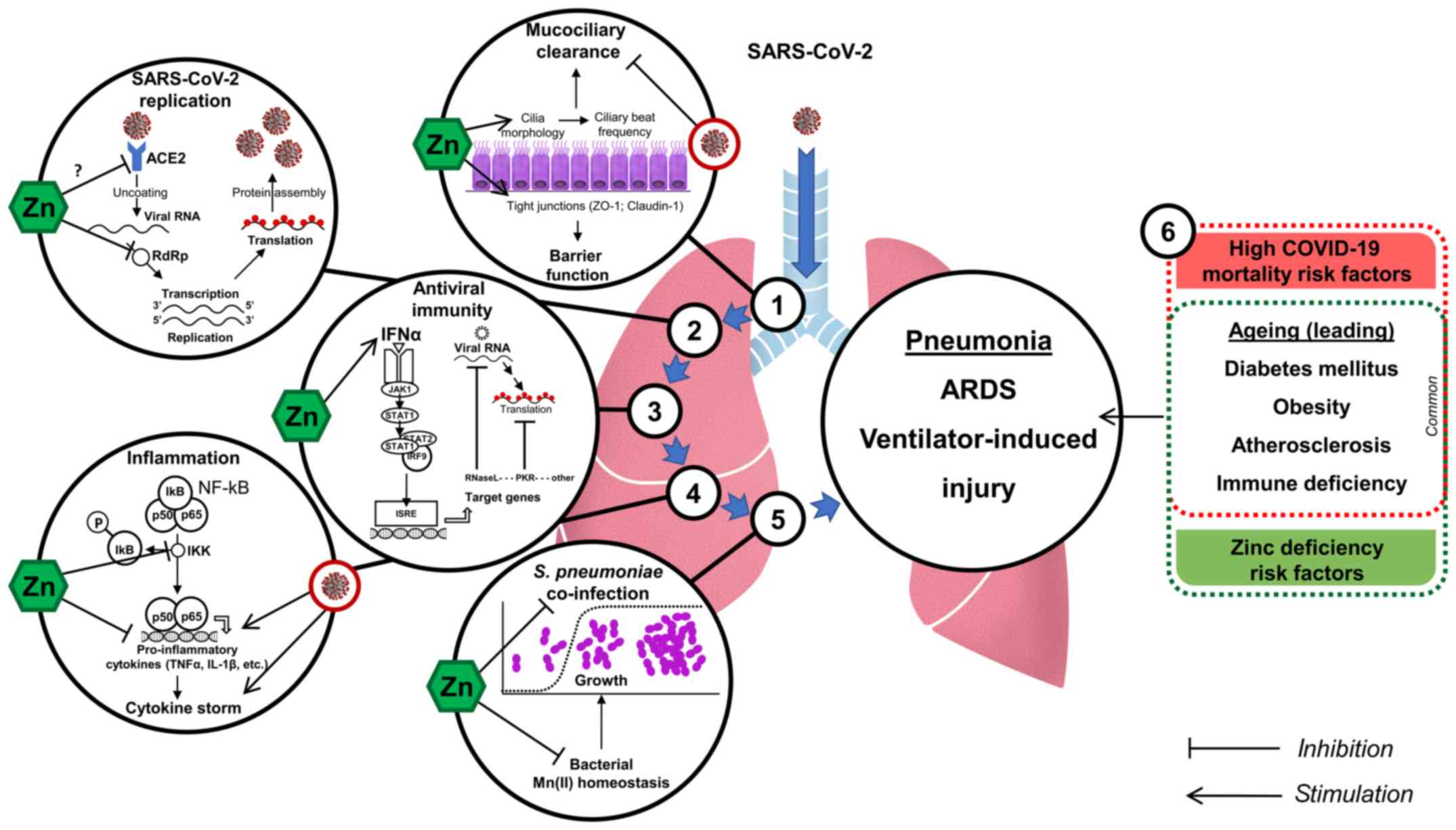
especially in elderly (Fig. 1).
Further clinical and experimental studies are strongly required to
elucidate the potential role of Zn deficiency in COVID-19
susceptibility, as well as effects of Zn supplementation, and the
underlying mechanisms.
The study was partially supported by the Russian
Ministry of Science and Higher Education, Project no.
0856-2020-0008. MA was supported by NIH grants nos. NIEHS R0110563,
R01ES07331 and NIEHS R01ES020852.
Not applicable.
Conceptualization: AVS, LR, MA, JA, AT, AAT;
validation, research, resources, data reviewing, and writing: AVS,
LR, OPA, MA, VAG, SIA, AAS, DP, DAS, JA, AT, AAT; figure
preparation and edition: AAT; review and editing: AVS, LR, MA, JA,
AT, AAT. All authors read and approved the final manuscript.
Not applicable.
Not applicable.
DAS is the Editor-in-Chief for the journal, but had
no personal involvement in the reviewing process, or any influence
in terms of adjudicating on the final decision, for this article.
The other authors declare that they have no competing
interests.
Not applicable.
|
1
|
Prasad AS: Discovery of Zinc for Human
Health and Biomarkers of Zinc Deficiency. Molecular, Genetic, and
Nutritional Aspects of Major and Trace Minerals. Collins JF:
Academic Press; Cambridge: pp. 241–260. 2017, View Article : Google Scholar
|
|
2
|
Wessels I, Maywald M and Rink L: Zinc as a
gatekeeper of immune function. Nutrients. 9:12862017. View Article : Google Scholar
|
|
3
|
Maywald M, Wessels I and Rink L: Zinc
signals and immunity. Int J Mol Sci. 18:22222017. View Article : Google Scholar :
|
|
4
|
Haase H and Rink L: Multiple impacts of
zinc on immune function. Metallomics. 6:1175–1180. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Gammoh NZ and Rink L: Zinc in infection
and inflammation. Nutrients. 9:6242017. View Article : Google Scholar :
|
|
6
|
Aftanas LI, Bonitenko EYu, Varenik VI,
Grabeklis AR, Kiselev MF, Lakarova EV, Nechiporenko SP, Nikolaev
VA, Skalny AV and Skalnaya MG: Element status of population of
Central Federal Region. Element status of population of Russia.
Part II. Skalny AV and Kiselev MF: ELBI-SPb; Saint Petersburg: pp.
4302011
|
|
7
|
Walker CLF, Rudan I, Liu L, Nair H,
Theodoratou E, Bhutta ZA, O'Brien KL, Campbell H and Black RE:
Global burden of childhood pneumonia and diarrhoea. Lancet.
381:1405–1416. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Bailey RL, West KP Jr and Black RE: The
epidemiology of global micronutrient deficiencies. Ann Nutr Metab.
66(Suppl 2): 22–33. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Yasuda H and Tsutsui T: Infants and
elderlies are susceptible to zinc deficiency. Sci Rep. 6:218502016.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Wapnir RA: Zinc deficiency, malnutrition
and the gastrointestinal tract. J Nutr. 130(Suppl): 1388S–1392S.
2000. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Bonaventura P, Benedetti G, Albarède F and
Miossec P: Zinc and its role in immunity and inflammation.
Autoimmun Rev. 14:277–285. 2015. View Article : Google Scholar
|
|
12
|
Chabosseau P and Rutter GA: Zinc and
diabetes. Arch Biochem Biophys. 611:79–85. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Olechnowicz J, Tinkov A, Skalny A and
Suliburska J: Zinc status is associated with inflammation,
oxidative stress, lipid, and glucose metabolism. J Physiol Sci.
68:19–31. 2018. View Article : Google Scholar :
|
|
14
|
Kozlowski H, Luczkowski M, Remelli M and
Valensin D: Copper, zinc and iron in neurodegenerative diseases
(Alzheimer's, Parkinson's and prion diseases). Coord Chem Rev.
256:2129–2141. 2012. View Article : Google Scholar
|
|
15
|
Berry M, Gamieldien J and Fielding BC:
Identification of new respiratory viruses in the new millennium.
Viruses. 7:996–1019. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Peiris JSM: Coronaviruses. Clinical
Virology. Richman DD, Whitley RJ and Hayden FG: 4th edition. ASM
Press; Washington: pp. 1244–1265. 2016
|
|
17
|
Docea AO, Tsatsakis A, Albulescu D,
Cristea O, Zlatian O, Vinceti M, Moschos SA, Tsoukalas D, Goumenou
M, Drakoulis N, et al: A new threat from an old enemy: Re-emergence
of coronavirus (Review). Int J Mol Med. 45:1631–1643.
2020.PubMed/NCBI
|
|
18
|
Goumenou M, Spandidos DA and Tsatsakis A:
[Editorial] Possibility of transmission through dogs being a
contributing factor to the extreme Covid 19 outbreak in North
Italy. Mol Med Rep. 21:2293–2295. 2020.PubMed/NCBI
|
|
19
|
Lai CC, Shih TP, Ko WC, Tang HJ and Hsueh
PR: Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2)
and coronavirus disease-2019 (COVID-19): The epidemic and the
challenges. Int J Antimicrob Agents. 55:1059242020. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Liu J, Cao R, Xu M, Wang X, Zhang H, Hu H,
Li Y, Hu Z, Zhong W and Wang M: Hydroxychloroquine, a less toxic
derivative of chloroquine, is effective in inhibiting SARS-CoV-2
infection in vitro. Cell Discov. 6:162020. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Khachfe HH, Chahrour M, Sammouri J, Salhab
H, Makki B and Fares MY: An epidemiological study on COVID-19: A
rapidly spreading disease. Cureus. 12:e73132020.PubMed/NCBI
|
|
22
|
World Health Organization (WHO):
Coronavirus disease 2019. Events as they happen. WHO; Geneva: 2020,
https://www.who.int/emergencies/diseases/novel-coronavirus-2019/events-as-they-happen.
Updated April 9, 2020.
|
|
23
|
World Health Organization (WHO):
Coronavirus disease (COVID-2019). Situation report - 81. WHO;
Geneva: 2020, https://www.who.int/docs/default-source/coronaviruse/situation-reports/20200410-sitrep-81-covid-19.pdf.
Accessed April 10, 2020.
|
|
24
|
Rothan HA and Byrareddy SN: The
epidemiology and pathogenesis of coronavirus disease (COVID-19)
outbreak. J Autoimmun. 109:1024332020. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Ñamendys-Silva SA: Respiratory support for
patients with COVID-19 infection. Lancet Respir Med. 8:e182020.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Yang X, Yu Y, Xu J, Shu H, Xia J, Liu H,
Wu Y, Zhang L, Yu Z, Fang M, et al: Clinical course and outcomes of
critically ill patients with SARS-CoV-2 pneumonia in Wuhan, China:
a single-centered, retrospective, observational study. Lancet
Respir Med. Feb 24–2020.Epub ahead of print. View Article : Google Scholar
|
|
27
|
Docea AO, Goumenou M, Calina D, Arsene AL,
Dragoi CM, Gofita E, Pisoschi CG, Zlatian O, Stivaktakis PD,
Nikolouzakis TK, et al: Adverse and hormetic effects in rats
exposed for 12 months to low dose mixture of 13 chemicals: RLRS
part III. Toxicol Lett. 310:70–91. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Hernández AF, Docea AO, Goumenou M,
Sarigiannis D, Aschner M and Tsatsakis A: Application of novel
technologies and mechanistic data for risk assessment under the
real-life risk simulation (RLRS) approach. Food Chem Toxicol.
137:1111232020. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Fountoucidou P, Veskoukis AS, Kerasioti E,
Docea AO, Taitzoglou IA, Liesivuori J, Tsatsakis A and Kouretas D:
A mixture of routinely encountered xenobiotics induces both redox
adaptations and perturbations in blood and tissues of rats after a
long-term low-dose exposure regimen: The time and dose issue.
Toxicol Lett. 317:24–44. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Tsatsakis AM, Kouretas D, Tzatzarakis MN,
Stivaktakis P, Tsarouhas K, Golokhvast KS, Rakitskii VN, Tutelyan
VA, Hernandez AF, Rezaee R, et al: Simulating real-life exposures
to uncover possible risks to human health: A proposed consensus for
a novel methodological approach. Hum Exp Toxicol. 36:554–564. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Tsatsakis A, Tyshko NV, Docea AO,
Shestakova SI, Sidorova YS, Petrov NA, Zlatian O, Mach M, Hartung T
and Tutelyan VA: The effect of chronic vitamin deficiency and long
term very low dose exposure to 6 pesticides mixture on neurological
outcomes - A real-life risk simulation approach. Toxicol Lett.
315:96–106. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Wu C, Chen X, Cai Y, Zhou X, Xu S, Huang
H, Wu C, Chen X, Cai Y, Zhou X, et al: Risk factors associated with
acute respiratory distress syndrome and death in patients with
coronavirus disease 2019 pneumonia in Wuhan. JAMA Intern Med. Mar
13–2020.Epub ahead of print. View Article : Google Scholar
|
|
33
|
Huang R, Zhu L, Xue L, Liu L, Yan X, Wang
J, Zhang B, Xu T, Ji F, et al: Clinical findings of patients with
coronavirus disease 2019 in Jiangsu province, China: A
retrospective, multi-center study. SSRN, 2020. https://ssrn.com/abstract=3548785.
Accessed Febryary 28, 2020.
|
|
34
|
Jiang F, Deng L, Zhang L, Cai Y, Cheung CW
and Xia Z: Review of the clinical characteristics of coronavirus
disease 2019 (COVID-19). J Gen Intern Med. Mar 4–2020.Epub ahead of
print. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Haase H and Rink L: The immune system and
the impact of zinc during aging. Immun Ageing. 6:92009. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Zhang L and Liu Y: Potential interventions
for novel coronavirus in China: A systematic review. J Med Virol.
92:479–490. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Read SA, Obeid S, Ahlenstiel C and
Ahlenstiel G: The role of zinc in antiviral immunity. Adv Nutr.
10:696–710. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Sandstead HH and Prasad AS: Zinc intake
and resistance to H1N1 influenza. Am J Public Health. 100:970–971.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
te Velthuis AJ, van den Worm SH, Sims AC,
Baric RS, Snijder EJ and van Hemert MJ: Zn(2+) inhibits coronavirus
and arterivirus RNA polymerase activity in vitro and zinc
ionophores block the replication of these viruses in cell culture.
PLoS Pathog. 6:e10011762010. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Wang M, Cao R, Zhang L, Yang X, Liu J, Xu
M, Shi Z, Hu Z, Zhong W and Xiao G: Remdesivir and chloroquine
effectively inhibit the recently emerged novel coronavirus
(2019-nCoV) in vitro. Cell Res. 30:269–271. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Liu J, Zheng X, Tong Q, Li W, Wang B,
Sutter K, Trilling M, Lu M, Dittmer U and Yang D: Overlapping and
discrete aspects of the pathology and pathogenesis of the emerging
human pathogenic coronaviruses SARS-CoV, MERS-CoV, and 2019-nCoV. J
Med Virol. 92:491–494. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Xue J, Moyer A, Peng B, Wu J, Hannafon BN
and Ding WQ: Chloroquine is a zinc ionophore. PLoS One.
9:e1091802014. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Guastalegname M and Vallone A: Could
chloroquine/hydroxy-chloroquine be harmful in Coronavirus disease
2019 (COVID-19) treatment? Clin Infect Dis. Mar 24–2020.Epub ahead
of print. View Article : Google Scholar
|
|
44
|
Dabbagh-Bazarbachi H, Clergeaud G, Quesada
IM, Ortiz M, O'Sullivan CK and Fernández-Larrea JB: Zinc ionophore
activity of quercetin and epigallocatechin-gallate: From Hepa 1-6
cells to a liposome model. J Agric Food Chem. 62:8085–8093. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Lin MH, Moses DC, Hsieh CH, Cheng SC, Chen
YH, Sun CY and Chou CY: Disulfiram can inhibit MERS and SARS
coro-navirus papain-like proteases via different modes. Antiviral
Res. 150:155–163. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Sargsyan K, Chen T, Grauffel C and Lim C:
Identifying COVID-19 drug-sites susceptible to clinically safe
Zn-ejector drugs using evolutionary/physical principles. OSF
Preprints, 2020. https://osf.io/snuqf/.
Accessed February 13, 2020.
|
|
47
|
Xu L, Tong J, Wu Y, Zhao S and Lin BL:
Targeted oxidation strategy (TOS) for potential inhibition of
Coronaviruses by disulfiram - a 70-year old anti-alcoholism drug.
ChemRxiv. In Press.
|
|
48
|
Hoffmann M, Kleine-Weber H, Krüger N,
Mueller MA, Drosten C and Pöhlmann S: The novel coronavirus 2019
(2019-nCoV) uses the SARS-coronavirus receptor ACE2 and the
cellular protease TMPRSS2 for entry into target cells. bioRxiv. In
Press.
|
|
49
|
Zhang H, Penninger JM, Li Y, Zhong N and
Slutsky AS: Angiotensin-converting enzyme 2 (ACE2) as a SARS-CoV-2
receptor: Molecular mechanisms and potential therapeutic target.
Intensive Care Med. 46:586–590. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Speth R, Carrera E, Jean-Baptiste M,
Joachim A and Linares A: Concentration-dependent effects of zinc on
angiotensin-converting enzyme-2 activity (1067.4). FASEB J.
28(Suppl 1): 1067.42014.
|
|
51
|
Chilvers MA, McKean M, Rutman A, Myint BS,
Silverman M and O'Callaghan C: The effects of coronavirus on human
nasal ciliated respiratory epithelium. Eur Respir J. 18:965–970.
2001. View Article : Google Scholar
|
|
52
|
Maret W: Analyzing free zinc(II) ion
concentrations in cell biology with fluorescent chelating
molecules. Metallomics. 7:202–211. 2015. View Article : Google Scholar
|
|
53
|
Essaidi-Laziosi M, Brito F, Benaoudia S,
Royston L, Cagno V, Fernandes-Rocha M, Piuz I, Zdobnov E, Huang S,
Constant S, et al: Propagation of respiratory viruses in human
airway epithelia reveals persistent virus-specific signatures. J
Allergy Clin Immunol. 141:2074–2084. 2018. View Article : Google Scholar
|
|
54
|
Pittet LA, Hall-Stoodley L, Rutkowski MR
and Harmsen AG: Influenza virus infection decreases tracheal
mucociliary velocity and clearance of Streptococcus pneumoniae. Am
J Respir Cell Mol Biol. 42:450–460. 2010. View Article : Google Scholar :
|
|
55
|
Darma A, Ranuh RG, Merbawani W,
Setyoningrum RA, Hidajat B, Hidayati SN, Andaryanto A and Sudarmo
SM: Zinc supplementation effect on the bronchial cilia length, the
number of cilia, and the number of intact bronchial cell in zinc
deficiency rats. Indones Biomed J. 12:78–84. 2020. View Article : Google Scholar
|
|
56
|
Woodworth BA, Zhang S, Tamashiro E,
Bhargave G, Palmer JN and Cohen NA: Zinc increases ciliary beat
frequency in a calcium-dependent manner. Am J Rhinol Allergy.
24:6–10. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Truong-Tran AQ, Carter J, Ruffin R and
Zalewski PD: New insights into the role of zinc in the respiratory
epithelium. Immunol Cell Biol. 79:170–177. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Roscioli E, Jersmann HP, Lester S, Badiei
A, Fon A, Zalewski P and Hodge S: Zinc deficiency as a
codeterminant for airway epithelial barrier dysfunction in an ex
vivo model of COPD. Int J Chron Obstruct Pulmon Dis. 12:3503–3510.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Wittekindt OH: Tight junctions in
pulmonary epithelia during lung inflammation. Pflugers Arch.
469:135–147. 2017. View Article : Google Scholar :
|
|
60
|
Günzel D and Yu AS: Claudins and the
modulation of tight junction permeability. Physiol Rev. 93:525–569.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Ishida T: Review on the role of
Zn2+ ions in viral pathogenesis and the effect of
Zn2+ ions for host cell-virus growth inhibition. Am J
Biomed Sci Res. 2:AJBSR.MS.ID.000566. 2019. View Article : Google Scholar
|
|
62
|
Krenn BM, Gaudernak E, Holzer B, Lanke K,
Van Kuppeveld FJM and Seipelt J: Antiviral activity of the zinc
ionophores pyrithione and hinokitiol against picornavirus
infections. J Virol. 83:58–64. 2009. View Article : Google Scholar :
|
|
63
|
Korant BD, Kauer JC and Butterworth BE:
Zinc ions inhibit replication of rhinoviruses. Nature. 248:588–590.
1974. View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Cakman I, Kirchner H and Rink L: Zinc
supplementation reconstitutes the production of interferon-α by
leukocytes from elderly persons. J Interferon Cytokine Res.
17:469–472. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Berg K, Bolt G, Andersen H and Owen TC:
Zinc potentiates the antiviral action of human IFN-α tenfold. J
Interferon Cytokine Res. 21:471–474. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Lin FC and Young HA: Interferons: Success
in anti-viral immunotherapy. Cytokine Growth Factor Rev.
25:369–376. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Singh M and Das RR: Zinc for the common
cold. Cochrane Database Syst Rev. 2013:CD0013642013.
|
|
68
|
Hemilä H: Zinc lozenges and the common
cold: A meta-analysis comparing zinc acetate and zinc gluconate,
and the role of zinc dosage. JRSM Open. 8:20542704176942912017.
View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Hemilä H: Zinc lozenges may shorten the
duration of colds: A systematic review. Open Respir Med J. 5:51–58.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Che Z and Sun J: Investigation on
relationship between whole blood zinc and Fe elements with children
pneumonia caused by respiratory syncytial virus. Int J Lab Med.
37:2401–2402. 2016.
|
|
71
|
Johnson JK, Harris FL, Ping XD, Gauthier
TW and Brown LAS: Role of zinc insufficiency in fetal alveolar
macrophage dysfunction and RSV exacerbation associated with fetal
ethanol exposure. Alcohol. 80:5–16. 2019. View Article : Google Scholar
|
|
72
|
Suara RO and Crowe JE Jr: Effect of zinc
salts on respiratory syncytial virus replication. Antimicrob Agents
Chemother. 48:783–790. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
73
|
Kaynar AM, Andreas A, Maloy A, Austin W,
Pitt BR, Gopal R and Alcorn JF: Zinc deficiency worsens the
long-term outcome and exacerbates inflammation in a murine model of
influenza-MRSA superinfection. Am J Respir Crit Care Med.
199:A41302019.
|
|
74
|
Haase H, Mocchegiani E and Rink L:
Correlation between zinc status and immune function in the elderly.
Biogerontology. 7:421–428. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
75
|
Barnett JB, Hamer DH and Meydani SN: Low
zinc status: A new risk factor for pneumonia in the elderly? Nutr
Rev. 68:30–37. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
76
|
Meydani SN, Barnett JB, Dallal GE, Fine
BC, Jacques PF, Leka LS and Hamer DH: Serum zinc and pneumonia in
nursing home elderly. Am J Clin Nutr. 86:1167–1173. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
77
|
Bhat MH, Rather AB, Dhobi GN, Koul AN,
Bhat FA and Hussain A: Zinc levels in community acquired pneumonia
in hospitalized patients; a case control study. Egypt J Chest Dis
Tuberc. 65:485–489. 2016. View Article : Google Scholar
|
|
78
|
Saleh P, Sadeghpour A,
Mirza-Aghazadeh-Attari M, Hatampour M, Naghavi-Behzad M and Tabrizi
A: Relationship between plasma levels of zinc and clinical course
of pneumonia. Tanaffos. 16:40–45. 2017.PubMed/NCBI
|
|
79
|
Linko R, Karlsson S, Pettilä V, Varpula T,
Okkonen M, Lund V, Ala-Kokko T and Ruokonen E; FINNALI Study Group:
Serum zinc in critically ill adult patients with acute respiratory
failure. Acta Anaesthesiol Scand. 55:615–621. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
80
|
Saigal P and Hanekom D: Does zinc improve
symptoms of viral upper respiratory tract infection? EBP. 23:37–39.
2020.
|
|
81
|
Sharafi S and Allami A: Efficacy of zinc
sulphate on in-hospital outcome of community-acquired pneumonia in
people aged 50 years and over. Int J Tuberc Lung Dis. 20:685–688.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
82
|
Boudreault F, Pinilla-Vera M, Englert JA,
Kho AT, Isabelle C, Arciniegas AJ, Barragan-Bradford D, Quintana C,
Amador-Munoz D, Guan J, et al: MICU Registry: Zinc deficiency
primes the lung for ventilator-induced injury. JCI Insight.
2:e865072017. View Article : Google Scholar
|
|
83
|
Chen X, Bian J and Ge Y: Zinc-deficient
diet aggravates ventilation-induced lung injury in rats. J Biomed
Res. 26:59–65. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
84
|
Visalakshy J, Surendran S, Pillai MPG,
Rajendran A and Sherif AA: Could plasma zinc be a predictor for
mortality and severity in sepsis syndrome? Int J Res Med Sci.
5:3929–3934. 2017. View Article : Google Scholar
|
|
85
|
Hoeger J, Simon TP, Beeker T, Marx G,
Haase H and Schuerholz T: Persistent low serum zinc is associated
with recurrent sepsis in critically ill patients - A pilot study.
PLoS One. 12:e01760692017. View Article : Google Scholar : PubMed/NCBI
|
|
86
|
Lee PI, Hu YL, Chen PY, Huang YC and Hsueh
PR: Are children less susceptible to COVID-19? J Microbiol Immunol
Infect. Feb 25–2020.Epub ahead of print. View Article : Google Scholar
|
|
87
|
Dong Y, Mo X, Hu Y, Qi X, Jiang F, Jiang Z
and Tong S: Epidemiological characteristics of 2143 pediatric
patients with 2019 coronavirus disease in China. Pediatrics. In
Press.
|
|
88
|
Kumar N, Jayaprakash S and Kavitha D: Low
serum zinc level - a possible marker of severe pneumonia. JMSCR.
5:21554–21570. 2017. View Article : Google Scholar
|
|
89
|
Islam SN, Kamal MM, Rahmatullah R, Sadi
SKS and Ahsan M: Serum zinc levels in children with acute
respiratory infections: Association with sociodemography and
nutritional status. Clin Nutr Exp. 22:11–18. 2018. View Article : Google Scholar
|
|
90
|
Saleh NY and Abo El Fotoh WMM: Low serum
zinc level: The relationship with severe pneumonia and survival in
critically ill children. Int J Clin Pract. 72:e132112018.
View Article : Google Scholar : PubMed/NCBI
|
|
91
|
Yakoob MY, Theodoratou E, Jabeen A, Imdad
A, Eisele TP, Ferguson J, Jhass A, Rudan I, Campbell H, Black RE,
et al: Preventive zinc supplementation in developing countries:
Impact on mortality and morbidity due to diarrhea, pneumonia and
malaria. BMC Public Health. 11(Suppl 3): S232011. View Article : Google Scholar : PubMed/NCBI
|
|
92
|
Lassi ZS, Moin A and Bhutta ZA: Zinc
supplementation for the prevention of pneumonia in children aged 2
months to 59 months. Cochrane Database Syst Rev.
12:CD0059782016.PubMed/NCBI
|
|
93
|
Das RR, Singh M and Shafiq N: Short-term
therapeutic role of zinc in children <5 years of age
hospitalised for severe acute lower respiratory tract infection.
Paediatr Respir Rev. 13:184–191. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
94
|
Basnet S, Shrestha PS, Sharma A, Mathisen
M, Prasai R, Bhandari N, Adhikari RK, Sommerfelt H,
Valentiner-Branth P, Strand TA, Zinc Severe Pneumonia and Study
Group: A randomized controlled trial of zinc as adjuvant therapy
for severe pneumonia in young children. Pediatrics. 129:701–708.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
95
|
Srinivasan MG, Ndeezi G, Mboijana CK,
Kiguli S, Bimenya GS, Nankabirwa V and Tumwine JK: Zinc adjunct
therapy reduces case fatality in severe childhood pneumonia: A
randomized double blind placebo-controlled trial. BMC Med.
10:142012. View Article : Google Scholar : PubMed/NCBI
|
|
96
|
Bagri NK, Bagri N, Jana M, Gupta AK,
Wadhwa N, Lodha R, Kabra SK, Chandran A, Aneja S, Chaturvedi MK, et
al: Efficacy of oral zinc supplementation in radiologically
confirmed pneumonia: Secondary analysis of a randomized controlled
trial. J Trop Pediatr. 64:110–117. 2018. View Article : Google Scholar
|
|
97
|
Yuan X, Qian SY, Li Z and Zhang ZZ: Effect
of zinc supplementation on infants with severe pneumonia. World J
Pediatr. 12:166–169. 2016. View Article : Google Scholar
|
|
98
|
Shah UH, Abu-Shaheen AK, Malik MA, Alam S,
Riaz M and Al-Tannir MA: The efficacy of zinc supplementation in
young children with acute lower respiratory infections: A
randomized double-blind controlled trial. Clin Nutr. 32:193–199.
2013. View Article : Google Scholar
|
|
99
|
Rerksuppaphol S and Rerksuppaphol L: A
randomized controlled trial of zinc supplementation in the
treatment of acute respiratory tract infection in Thai children.
Pediatr Rep. 11:79542019. View Article : Google Scholar : PubMed/NCBI
|
|
100
|
Roth DE, Richard SA and Black RE: Zinc
supplementation for the prevention of acute lower respiratory
infection in children in developing countries: Meta-analysis and
meta-regression of randomized trials. Int J Epidemiol. 39:795–808.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
101
|
Martinez-Estevez NS, Alvarez-Guevara AN
and Rodriguez-Martinez CE: Effects of zinc supplementation in the
prevention of respiratory tract infections and diarrheal disease in
Colombian children: A 12-month randomised controlled trial.
Allergol Immunopathol (Madr). 44:368–375. 2016. View Article : Google Scholar
|
|
102
|
Aggarwal R, Sentz J and Miller MA: Role of
zinc administration in prevention of childhood diarrhea and
respiratory illnesses: A meta-analysis. Pediatrics. 119:1120–1130.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
103
|
Khera D, Singh S, Purohit P, Sharma P and
Singh K: Prevalence of Zinc deficiency and effect of Zinc
supplementation on prevention of acute respiratory infections: A
non randomized open label study. SSRN. 2018, https://ssrn.com/abstract=3273670.
Accessed October 26, 2018. View Article : Google Scholar
|
|
104
|
Mehta P, McAuley DF, Brown M, Sanchez E
and Tattersall RS: COVID-19: Consider cytokine storm syndromes and
immuno-suppression. Lancet. 395:1033–1034. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
105
|
Biaggio VS, Pérez Chaca MV, Valdéz SR,
Gómez NN and Gimenez MS: Alteration in the expression of
inflammatory parameters as a result of oxidative stress produced by
moderate zinc deficiency in rat lung. Exp Lung Res. 36:31–44. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
106
|
Bao S and Knoell DL: Zinc modulates
cytokine-induced lung epithelial cell barrier permeability. Am J
Physiol Lung Cell Mol Physiol. 291:L1132–L1141. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
107
|
Liu MJ, Bao S, Napolitano JR, Burris DL,
Yu L, Tridandapani S and Knoell DL: Zinc regulates the acute phase
response and serum amyloid A production in response to sepsis
through JAK-STAT3 signaling. PLoS One. 9:e94934PubMed/NCBI
|
|
108
|
St Croix CM, Leelavaninchkul K, Watkins
SC, Kagan VE and Pitt BR: Nitric oxide and zinc homeostasis in
acute lung injury. Proc Am Thorac Soc. 2:236–242. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
109
|
Krones CJ, Klosterhalfen B, Butz N, Hoelzl
F, Junge K, Stumpf M, Peiper C, Klinge U and Schumpelick V: Effect
of zinc pretreatment on pulmonary endothelial cells in vitro and
pulmonary function in a porcine model of endotoxemia. J Surg Res.
123:251–256. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
110
|
Wessels I, Pupke JT, von Trotha KT,
Gombert A, Himmelsbach A, Fischer HJ, Jacobs MJ, Rink L and Grommes
J: Zinc supplementation ameliorates lung injury by reducing
neutrophil recruitment and activity. Thorax. 75:253–261. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
111
|
Biaggio VS, Salvetti NR, Pérez Chaca MV,
Valdez SR, Ortega HH, Gimenez MS and Gomez NN: Alterations of the
extracellular matrix of lung during zinc deficiency. Br J Nutr.
108:62–70. 2012. View Article : Google Scholar
|
|
112
|
Luo W, Yu H, Gou J, Li X, Sun Y, Li J and
Liu L: Clinical pathology of critical patient with novel
Coronavirus pneumonia (COVID-19). Preprints.
2020:20200204072020.
|
|
113
|
Knoell DL, Julian MW, Bao S, Besecker B,
Macre JE, Leikauf GD, DiSilvestro RA and Crouser ED: Zinc
deficiency increases organ damage and mortality in a murine model
of polymicrobial sepsis. Crit Care Med. 37:1380–1388. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
114
|
Bao S, Liu MJ, Lee B, Besecker B, Lai JP,
Guttridge DC and Knoell DL: Zinc modulates the innate immune
response in vivo to polymicrobial sepsis through regulation of
NF-kappaB. Am J Physiol Lung Cell Mol Physiol. 298:L744–L754. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
115
|
Nowak JE, Harmon K, Caldwell CC and Wong
HR: Prophylactic zinc supplementation reduces bacterial load and
improves survival in a murine model of sepsis. Pediatr Crit Care
Med. 13:e323–e329. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
116
|
Ganatra HA, Varisco BM, Harmon K, Lahni P,
Opoka A and Wong HR: Zinc supplementation leads to immune
modulation and improved survival in a juvenile model of murine
sepsis. Innate Immun. 23:67–76. 2017. View Article : Google Scholar
|
|
117
|
Slinko S, Piraino G, Hake PW, Ledford JR,
O'Connor M, Lahni P, Solan PD, Wong HR and Zingarelli B: Combined
zinc supplementation with proinsulin C-peptide treatment decreases
the inflammatory response and mortality in murine polymicrobial
sepsis. Shock. 41:292–300. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
118
|
Besecker BY, Exline MC, Hollyfield J,
Phillips G, Disilvestro RA, Wewers MD and Knoell DL: A comparison
of zinc metabolism, inflammation, and disease severity in
critically ill infected and noninfected adults early after
intensive care unit admission. Am J Clin Nutr. 93:1356–1364. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
119
|
Liu MJ, Bao S, Gálvez-Peralta M, Pyle CJ,
Rudawsky AC, Pavlovicz RE, Killilea DW, Li C, Nebert DW, Wewers MD,
et al: ZIP8 regulates host defense through zinc-mediated inhibition
of NF-κB. Cell Rep. 3:386–400. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
120
|
Hall SC, Smith DR, Katafiasz DM, Bailey KL
and Knoell DL: Novel role of zinc homeostasis in IL-23 regulation
and host defense following bacterial infection. J Immunol.
202(Suppl 1): 62–6. 2019.
|
|
121
|
Knoell DL, Smith DA, Sapkota M, Heires AJ,
Hanson CK, Smith LM, Poole JA, Wyatt TA and Romberger DJ:
Insufficient zinc intake enhances lung inflammation in response to
agricultural organic dust exposure. J Nutr Biochem. 70:56–64. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
122
|
Lang CJ, Hansen M, Roscioli E, Jones J,
Murgia C, Leigh Ackland M, Zalewski P, Anderson G and Ruffin R:
Dietary zinc mediates inflammation and protects against wasting and
metabolic derangement caused by sustained cigarette smoke exposure
in mice. Biometals. 24:23–39. 2011. View Article : Google Scholar
|
|
123
|
von Bülow V, Dubben S, Engelhardt G, Hebel
S, Plümäkers B, Heine H, Rink L and Haase H: Zinc-dependent
suppression of TNF-α production is mediated by protein kinase
A-induced inhibition of Raf-1, IκB kinase β, and NF-κB. J Immunol.
179:4180–4186. 2007. View Article : Google Scholar
|
|
124
|
Wessels I, Haase H, Engelhardt G, Rink L
and Uciechowski P: Zinc deficiency induces production of the
proinflammatory cytokines IL-1β and TNFα in promyeloid cells via
epigenetic and redox-dependent mechanisms. J Nutr Biochem.
24:289–297. 2013. View Article : Google Scholar
|
|
125
|
Prasad AS, Bao B, Beck FW and Sarkar FH:
Zinc-suppressed inflammatory cytokines by induction of A20-mediated
inhibition of nuclear factor-κB. Nutrition. 27:816–823. 2011.
View Article : Google Scholar
|
|
126
|
Wellinghausen N, Martin M and Rink L: Zinc
inhibits interleukin-1-dependent T cell stimulation. Eur J Immunol.
27:2529–2535. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
127
|
Rosenkranz E, Metz CH, Maywald M, Hilgers
RD, Weßels I, Senff T, Haase H, Jäger M, Ott M, Aspinall R, et al:
Zinc supplementation induces regulatory T cells by inhibition of
Sirt-1 deacetylase in mixed lymphocyte cultures. Mol Nutr Food Res.
60:661–671. 2016. View Article : Google Scholar
|
|
128
|
Kahmann L, Uciechowski P, Warmuth S,
Plümäkers B, Gressner AM, Malavolta M, Mocchegiani E and Rink L:
Zinc supplementation in the elderly reduces spontaneous
inflammatory cytokine release and restores T cell functions.
Rejuvenation Res. 11:227–237. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
129
|
Kim H: Outbreak of novel coronavirus
(COVID-19): What is the role of radiologists? Eur Radiol. Feb
18–2020.Epub ahead of print. View Article : Google Scholar :
|
|
130
|
Golda A, Malek N, Dudek B, Zeglen S,
Wojarski J, Ochman M, Kucewicz E, Zembala M, Potempa J and Pyrc K:
Infection with human coronavirus NL63 enhances streptococcal
adherence to epithelial cells. J Gen Virol. 92:1358–1368. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
131
|
Eijkelkamp BA, Morey JR, Neville SL, Tan
A, Pederick VG, Cole N, Singh PP, Ong CY, Gonzalez de Vega R,
Clases D, et al: Dietary zinc and the control of Streptococcus
pneumoniae infection. PLoS Pathog. 15:e10079572019. View Article : Google Scholar : PubMed/NCBI
|
|
132
|
Coles CL, Sherchand JB, Khatry SK, Katz J,
Leclerq SC, Mullany LC and Tielsch JM: Zinc modifies the
association between nasopharyngeal Streptococcus pneumoniae
carriage and risk of acute lower respiratory infection among young
children in rural Nepal. J Nutr. 138:2462–2467. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
133
|
Strand TA, Hollingshead SK, Julshamn K,
Briles DE, Blomberg B and Sommerfelt H: Effects of zinc deficiency
and pneumococcal surface protein A immunization on zinc status and
the risk of severe infection in mice. Infect Immun. 71:2009–2013.
2003. View Article : Google Scholar : PubMed/NCBI
|
|
134
|
Strand TA, Briles DE, Gjessing HK, Maage
A, Bhan MK and Sommerfelt H: Pneumococcal pulmonary infection,
septicaemia and survival in young zinc-depleted mice. Br J Nutr.
86:301–306. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
135
|
Hamza SA, Mousa SM, Taha SE, Adel LA,
Samaha HE and Hussein DA: Immune response of 23-valent pneumococcal
poly-saccharide vaccinated elderly and its relation to frailty
indices, nutritional status, and serum zinc levels. Geriatr
Gerontol Int. 12:223–229. 2012. View Article : Google Scholar
|
|
136
|
Mansouri F, Vaziri S, Janbakhsh A, Sayad
B, Najafi F, Karimivafa SM, Kashef M and Azizi M: The effect of
zinc on the Immune responses of pneumococcal vaccination in
elderly. Int J Med Microbiol. 10:67–73. 2016.
|
|
137
|
Osendarp SJ, Prabhakar H, Fuchs GJ, van
Raaij JM, Mahmud H, Tofail F, Santosham M and Black RE:
Immunization with the heptavalent pneumococcal conjugate vaccine in
Bangladeshi infants and effects of zinc supplementation. Vaccine.
25:3347–3354. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
138
|
Jacobsen FE, Kazmierczak KM, Lisher JP,
Winkler ME and Giedroc DP: Interplay between manganese and zinc
homeostasis in the human pathogen Streptococcus pneumoniae.
Metallomics. 3:38–41. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
139
|
McDevitt CA, Ogunniyi AD, Valkov E,
Lawrence MC, Kobe B, McEwan AG and Paton JC: A molecular mechanism
for bacterial susceptibility to zinc. PLoS Pathog. 7:e10023572011.
View Article : Google Scholar : PubMed/NCBI
|
|
140
|
Pasquet J, Chevalier Y, Pelletier J,
Couval E, Bouvier D and Bolzinger MA: The contribution of zinc ions
to the antimicrobial activity of zinc oxide. Colloids Surf A
Physicochem Eng Asp. 457:263–274. 2014. View Article : Google Scholar
|
|
141
|
Bhattacharyya P, Agarwal B, Goswami M,
Maiti D, Baruah S and Tribedi P: Zinc oxide nanoparticle inhibits
the biofilm formation of Streptococcus pneumoniae. Antonie van
Leeuwenhoek. 111:89–99. 2018. View Article : Google Scholar
|
|
142
|
Reddy LS, Nisha MM, Joice M and Shilpa PN:
Antimicrobial activity of zinc oxide (ZnO) nanoparticle against
Klebsiella pneumoniae. Pharm Biol. 52:1388–1397. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
143
|
Kadiyala U, Turali-Emre ES, Bahng JH,
Kotov NA and VanEpps JS: Unexpected insights into antibacterial
activity of zinc oxide nanoparticles against methicillin resistant
Staphylococcus aureus (MRSA). Nanoscale. 10:4927–4939. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
144
|
Ann LC, Mahmud S, Bakhori SKM, Sirelkhatim
A, Mohamad D, Hasan H, Seeni A and Rahman RA: Antibacterial
responses of zinc oxide structures against Staphylococcus aureus,
Pseudomonas aeruginosa and Streptococcus pyogenes. Ceram Int.
40:2993–3001. 2014. View Article : Google Scholar
|
|
145
|
Sahu D, Kannan GM, Vijayaraghavan R, Anand
T and Khanum F: Nanosized zinc oxide induces toxicity in human lung
cells. ISRN Toxicol. 2013:3160752013. View Article : Google Scholar : PubMed/NCBI
|
|
146
|
Lin CD, Kou YY, Liao CY, Li CH, Huang SP,
Cheng YW, Liao WC, Chen HX, Wu PL, Kang JJ, et al: Zinc oxide
nanoparticles impair bacterial clearance by macrophages.
Nanomedicine (Lond). 9:1327–1339. 2014. View Article : Google Scholar
|
|
147
|
Bayle L, Chimalapati S, Schoehn G, Brown
J, Vernet T and Durmort C: Zinc uptake by Streptococcus pneumoniae
depends on both AdcA and AdcAII and is essential for normal
bacterial morphology and virulence. Mol Microbiol. 82:904–916.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
148
|
Brown LR, Caulkins RC, Schartel TE, Rosch
JW, Honsa ES, Schultz-Cherry S, Meliopoulos VA, Cherry S and
Thornton JA: Increased zinc availability enhances initial
aggregation and biofilm formation of Streptococcus pneumoniae.
Front Cell Infect Microbiol. 7:2332017. View Article : Google Scholar :
|
|
149
|
Skalnaya MG and Skalny AV: Essential trace
elements in human health: a physician's view. Publishing House of
Tomsk State University; Tomsk: 2018
|