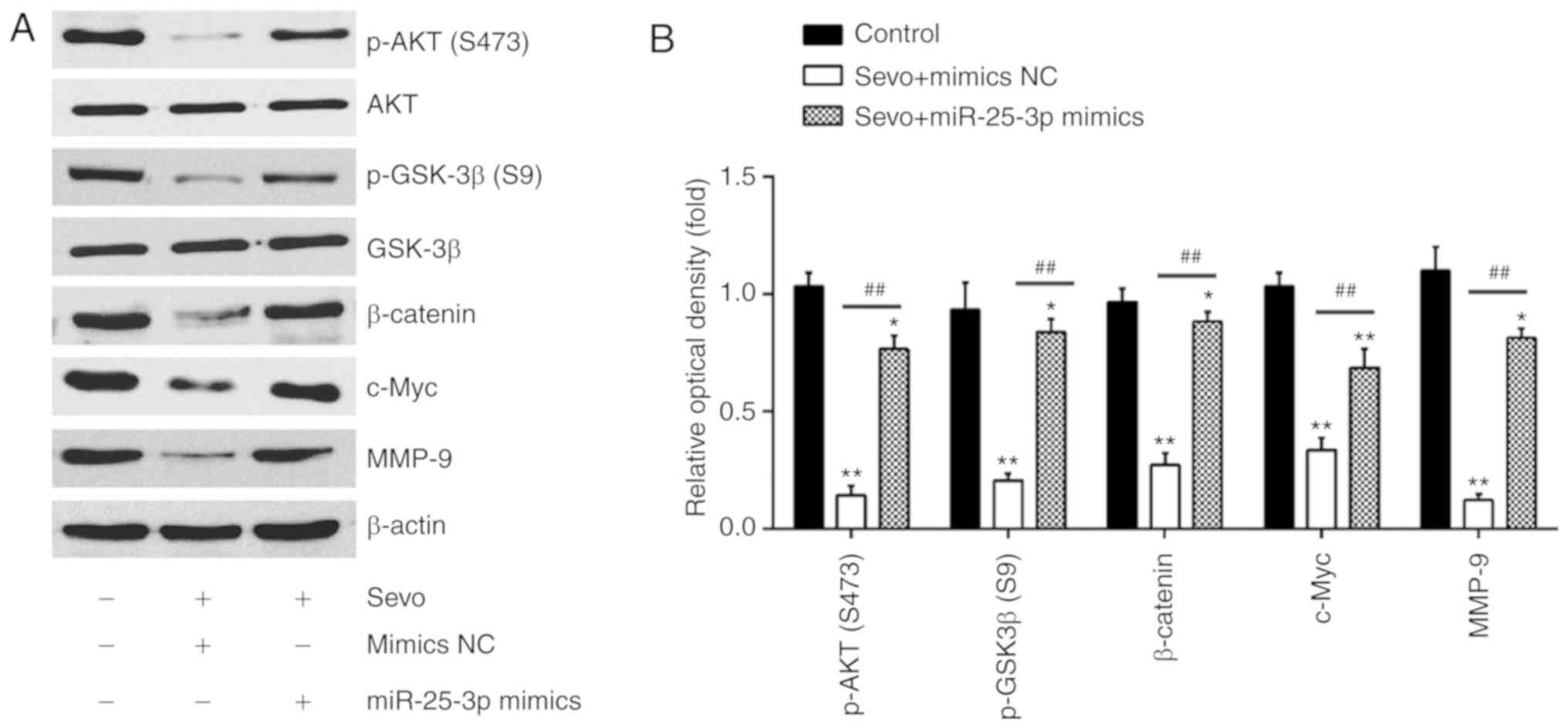
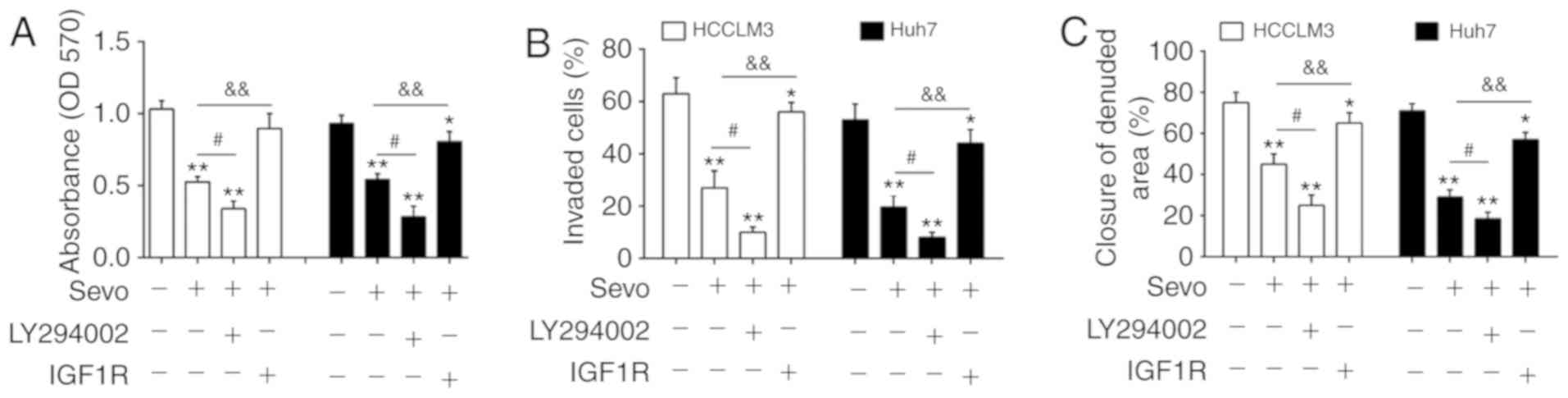
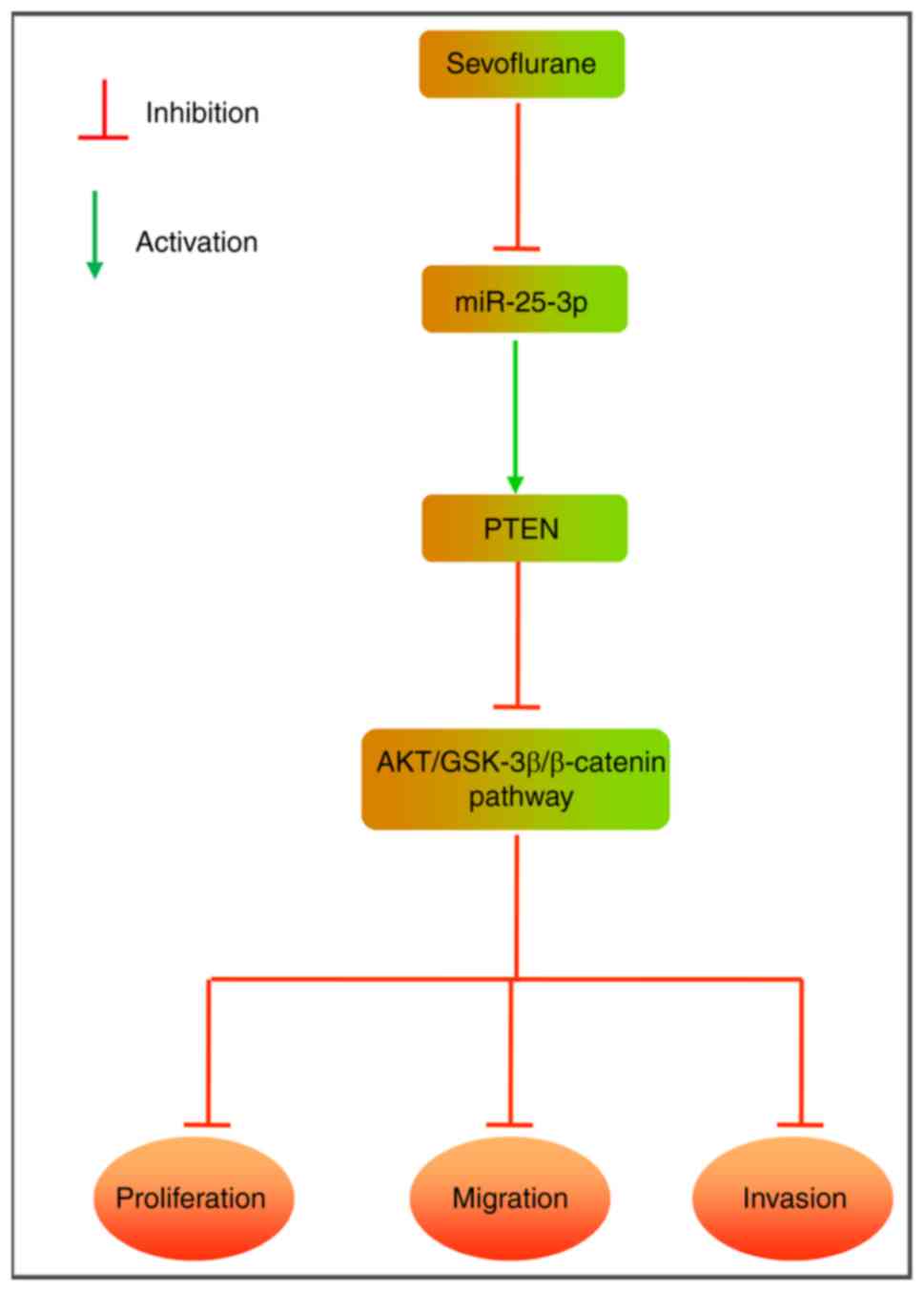
|
1
|
Ghouri YA, Mian I and Rowe JH: Review of
hepatocellular carcinoma: Epidemiology, etiology, and
carcinogenesis. J Carcinog. 16:12017. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
McGlynn KA and London WT: The global
epidemiology of hepatocellular carcinoma: Present and future. Clin
Liver Dis. 15:223–243. vii–x. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Tang ZY: Hepatocellular carcinoma-cause,
treatment and metastasis. World J Gastroenterol. 7:445–454. 2001.
View Article : Google Scholar
|
|
4
|
Tang Z, Zhou X, Lin Z, Yang B, Ma Z, Ye S,
Wu Z, Fan J, Liu Y, Liu K, et al: Surgical treatment of
hepatocellular carcinoma and related basic research with special
reference to recurrence and metastasis. Chin Med J (Engl).
112:887–891. 1999.
|
|
5
|
Liang H, Gu MN, Yang CX, Wang HB, Wen XJ
and Zhou QL: Sevoflurane inhibits proliferation, induces apoptosis,
and blocks cell cycle progression of lung carcinoma cells. Asian
Pac J Cancer Prev. 12:3415–3420. 2011.PubMed/NCBI
|
|
6
|
Liu J, Yang L, Guo X, Jin G, Wang Q, Lv D,
Liu J, Chen Q, Song Q and Li B: Sevoflurane suppresses
proliferation by upregulating microRNA-203 in breast cancer cells.
Mol Med Rep. 18:455–460. 2018.PubMed/NCBI
|
|
7
|
Yang X, Zheng YT and Rong W: Sevoflurane
induces apoptosis and inhibits the growth and motility of colon
cancer in vitro and in vivo via inactivating Ras/Raf/MEK/ERK
signaling. Life Sci. 239:1169162019. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Ciechanowicz S, Zhao H, Chen Q, Cui J, Mi
E, Mi E, Lian Q and Ma D: Differential effects of sevoflurane on
the metastatic potential and chemosensitivity of non-small-cell
lung adenocarcinoma and renal cell carcinoma in vitro. Br J
Anaesth. 120:368–375. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Ecimovic P, McHugh B, Murray D, Doran P
and Buggy DJ: Effects of sevoflurane on breast cancer cell function
in vitro. Anticancer Res. 33:4255–4260. 2013.PubMed/NCBI
|
|
10
|
Ambros V: The functions of animal
microRNAs. Nature. 431:350–355. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Bartel DP: MicroRNAs: Genomics,
biogenesis, mechanism, and function. Cell. 116:281–297. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Fan L, Wu Y, Wang J, He J and Han X:
Sevoflurane inhibits the migration and invasion of colorectal
cancer cells through regulating ERK/MMP-9 pathway by up-regulating
miR-203. Eur J Pharmacol. 850:43–52. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Sun SQ, Ren LJ, Liu J, Wang P and Shan SM:
Sevoflurane inhibits migration and invasion of colorectal cancer
cells by regulating microRNA-34a/ADAM10 axis. Neoplasma.
66:887–895. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Song G, Tian L, Cheng Y, Liu J, Wang K, Li
S and Li T: Antitumor activity of sevoflurane in HCC cell line is
mediated by miR-29a-induced suppression of Dnmt3a. J Cell Biochem.
120:18152–18161. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Roesslein M, Frick M, Auwaerter V, Humar
M, Goebel U, Schwer C, Geiger KK, Pahl HL, Pannen BH and Loop T:
Sevoflurane-mediated activation of p38-mitogen-activated
stresskinase is independent of apoptosis in Jurkat T-cells. Anesth
Analg. 106:1150–1160, table of contents. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Loop T, Scheiermann P, Doviakue D,
Musshoff F, Humar M, Roesslein M, Hoetzel A, Schmidt R, Madea B,
Geiger KK, et al: Sevoflurane inhibits
phorbolmyristate-acetate-induced activator protein-1 activation in
human T lymphocytes in vitro: Potential role of the p38-stress
kinase pathway. Anesthesiology. 101:710–721. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Mei LL, Wang WJ, Qiu YT, Xie XF, Bai J and
Shi ZZ: MiR-125b-5p functions as a tumor suppressor gene partially
by regulating HMGA2 in esophageal squamous cell carcinoma. PLoS
One. 12:e01856362017. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2 (-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar
|
|
19
|
Yao Y, Sun F and Lei M: MiR-25 inhibits
sepsis-induced cardiomyocyte apoptosis by targetting PTEN. Biosci
Rep. 38:BSR201715112018. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Xu Q, Xu HX, Li JP, Wang S, Fu Z, Jia J,
Wang L, Zhu ZF, Lu R and Yao Z: Growth differentiation factor 15
induces growth and metastasis of human liver cancer stem-like cells
via AKT/GSK-3β/β-catenin signaling. Oncotarget. 8:16972–16987.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Yi W, Li D, Guo Y, Zhang Y, Huang B and Li
X: Sevoflurane inhibits the migration and invasion of glioma cells
by upregulating microRNA-637. Int J Mol Med. 38:1857–1863. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Su ZX, Zhao J, Rong ZH, Geng WM, Wu YG and
Qin CK: Upregulation of microRNA-25 associates with prognosis in
hepatocellular carcinoma. Diagn Pathol. 9:472014. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Wang C, Wang X, Su Z, Fei H, Liu X and Pan
Q: MiR-25 promotes hepatocellular carcinoma cell growth, migration
and invasion by inhibiting RhoGDI1. Oncotarget. 6:36231–36244.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Zhang J, Gong X, Tian K, Chen D, Sun J,
Wang G and Guo M: MiR-25 promotes glioma cell proliferation by
targeting CDKN1C. Biomed Pharmacother. 71:7–14. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Xu LF, Wu ZP, Chen Y, Zhu QS, Hamidi S and
Navab R: MicroRNA-21 (miR-21) regulates cellular proliferation,
invasion, migration, and apoptosis by targeting PTEN, RECK and
Bcl-2 in lung squamous carcinoma, Gejiu City, China. PLoS One.
9:e1036982014. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Song MS, Salmena L and Pandolfi PP: The
functions and regu-lation of the PTEN tumour suppressor. Nat Rev
Mol Cell Biol. 13:283–296. 2012. View
Article : Google Scholar : PubMed/NCBI
|
|
27
|
Park NR, Cha JH, Jang JW, Bae SH, Jang B,
Kim JH, Hur W, Choi JY and Yoon SK: Synergistic effects of CD44 and
TGF-β1 through AKT/GSK-3β/β-catenin signaling during
epithelial-mesenchymal transition in liver cancer cells. Biochem
Biophys Res Commun. 477:568–574. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Liang H, Gu M, Yang C, Wang H, Wen X and
Zhou Q: Sevoflurane inhibits invasion and migration of lung cancer
cells by inactivating the p38 MAPK signaling pathway. J Anesth.
26:381–392. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Müller-Edenborn B, Roth-Z'graggen B,
Bartnicka K, Borgeat A, Hoos A, Borsig L and Beck-Schimmer B:
Volatile anesthetics reduce invasion of colorectal cancer cells
through down-regulation of matrix metalloproteinase-9.
Anesthesiology. 117:293–301. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Liang H, Yang CX, Zhang B, Wang HB, Liu
HZ, Lai XH, Liao MJ and Zhang T: Sevoflurane suppresses
hypoxia-induced growth and metastasis of lung cancer cells via
inhibiting hypoxia-inducible factor-1α. J Anesth. 29:821–830. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Hurmath FK, Mittal M, Ramaswamy P,
Umamaheswara Rao GS and Dalavaikodihalli Nanjaiah N: Sevoflurane
and thiopental preconditioning attenuates the migration and
activity of MMP-2 in U87MG glioma cells. Neurochem Int. 94:32–38.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Kvolik S, Glavas-Obrovac L, Bares V and
Karner I: Effects of inhalation anesthetics halothane, sevoflurane,
and isoflurane on human cell lines. Life Sci. 77:2369–2383. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Kvolik S, Dobrosevic B, Marczi S, Prlic L
and Glavas-Obrovac L: Different apoptosis ratios and gene
expressions in two human cell lines after sevoflurane anaesthesia.
Acta Anaesthesiol Scand. 53:1192–1199. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Zhao X, Jin Y, Li H, Jia Y and Wang Y:
Sevoflurane impairs learning and memory of the developing brain
through post-transcriptional inhibition of CCNA2 via
microRNA-19-3p. Aging (Albany NY). 10:3794–3805. 2018. View Article : Google Scholar
|
|
35
|
Xu C, Niu JJ, Zhou JF and Wei YS:
MicroRNA-96 is responsible for sevoflurane-induced cognitive
dysfunction in neonatal rats via inhibiting IGF1R. Brain Res Bull.
144:140–148. 2019. View Article : Google Scholar
|
|
36
|
Shao CZ and Xia KP: Sevoflurane anesthesia
represses neurogenesis of hippocampus neural stem cells via
regulating microRNA-183-mediated NR4A2 in newborn rats. J Cell
Physiol. 234:3864–3873. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Otsuki T, Ishikawa M, Hori Y, Goto G and
Sakamoto A: Volatile anesthetic sevoflurane ameliorates
endotoxin-induced acute lung injury via microRNA modulation in
rats. Biomed Rep. 3:408–412. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Gramantieri L, Fornari F, Callegari E,
Sabbioni S, Lanza G, Croce CM, Bolondi L and Negrini M: MicroRNA
involvement in hepatocellular carcinoma. J Cell Mol Med.
12:2189–2204. 2008. View Article : Google Scholar
|
|
39
|
Liu C, Yu J, Yu S, Lavker RM, Cai L, Liu
W, Yang K, He X and Chen S: MicroRNA-21 acts as an oncomir through
multiple targets in human hepatocellular carcinoma. J Hepatol.
53:98–107. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Chen H, Pan H, Qian Y, Zhou W and Liu X:
MiR-25-3p promotes the proliferation of triple negative breast
cancer by targeting BTG2. Mol Cancer. 17:42018. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Zhao H, Wang Y, Yang L, Jiang R and Li W:
MiR-25 promotes gastric cancer cells growth and motility by
targeting RECK. Mol Cell Biochem. 385:207–213. 2014. View Article : Google Scholar
|
|
42
|
Wang F, Qi X, Li Z, Jin S, Xie Y and Zhong
H: lncRNA CADM1-AS1 inhibits cell-cycle progression and invasion
via PTEN/AKT/GSK-3β axis in hepatocellular carcinoma. Cancer Manag
Res. 11:3813–3828. 2019. View Article : Google Scholar :
|
|
43
|
He J, Mu M, Luo Y, Wang H, Ma H, Guo S,
Fang Q, Qian Z, Lu H and Song C: MicroRNA-20b promotes
proliferation of H22 hepatocellular carcinoma cells by targeting
PTEN. Oncol Lett. 17:2931–2936. 2019.PubMed/NCBI
|
|
44
|
Han Y, Chen M, Wang A and Fan X:
STAT3-induced upregulation of lncRNA CASC11 promotes the cell
migration, invasion and epithelial-mesenchymal transition in
hepatocellular carcinoma by epigenetically silencing PTEN and
activating PI3K/AKT signaling pathway. Biochem Biophys Res Commun.
508:472–479. 2019. View Article : Google Scholar
|
|
45
|
Wan W, Wan W, Long Y, Li Q, Jin X, Wan G,
Zhang F, Lv Y, Zheng G, Li Z and Zhu Y: MiR-25-3p promotes
malignant phenotypes of retinoblastoma by regulating PTEN/Akt
pathway. Biomed Pharmacother. 118:1091112019. View Article : Google Scholar : PubMed/NCBI
|