1. Introduction
In December 2019, the first patient with an atypical
form of pneumonia was diagnosed in China, a disease that would
later be referred to as COVID-19 by the World Health Organization
(WHO). One month later, in January 2020, the causative agent of the
COVID-19 disease, the SARS-COV-2 virus, was deciphered and
identified and its genome was published rapidly (1).
By mid-April, 2020, the extent of impact of COVID-19
on the global population was becoming evident. The most adversely
affected region per capita was Western Europe, the most seriously
affected population being the elderly, especially those with
extensive co-morbidities and weakened immune systems. The causes
for regional differences in COVID-19 will require months of
analysis, if not years, to ascertain. More focused approaches to
reduce the impacts of COVID-19 in the near-term, especially on the
most vulnerable populations, need to be pursued, in parallel with
longer-term approaches to lessen the impact of COVID-19 (or other
coronavirus-driven diseases) across all demographics.
As of mid-April, the very short-term measures that
have been implemented to reduce the transmission of SARS-CoV-2 are
the same as those that were taken to reduce the transmission of
SARS-CoV during the 2002-2003 SARS pandemic: good hygiene and
quarantine, with the latter being far more severe and extensive
than the quarantine implemented in 2002-2003. Other short-term
measures that have been implemented on a very sporadic basis are
what can be termed 'tactical treatments'. These tactical treatments
do not strengthen the immune system, but rather have the goal of
containing the adverse impacts of the viral attack. Some examples
of tactical treatments that have been tested are EIDD-2801,
remdesivir, favipiravir, chloroquine, hydroxychloroquine,
combination of lopinavir and ritonavir, tocilizumab, losartan, to
name a few. Tests so far have yielded very mixed results.
A second avenue being pursued to ameliorate/reduce
the effects of COVID-19 is development of vaccines to induce viral
immunity. The bulk of the present review addresses the status and
prospects of this vaccine approach.
The third leg of this triad of approaches to
ameliorate the ravaging effects (especially for the most
vulnerable) of COVID-19 is what can be called the 'strategic
treatment' approach (2). This
approach has the goal of strengthening the immune system to allow
its adaptive component to neutralize the incoming coronavirus
without the need for external support. Strategic treatments
eliminate tangible factors that contribute to weaken the immune
system. Some examples of such factors, based on existing studies,
include titanium dioxide, air pollution, chlorpyrifos, aluminum
sulfate, prenatal alcohol exposure, ZnO, oil fly ash, and many
others (3-9). Also the real life exposure scenario
in which we are exposed continuously to combination of stressors
influences in the long term the immune system and the response of
the organism to the viral stressors (10-14).
Tsatsakis et al (15) have shown how pollutants can
influence the immune system particularly by modulating AhR pathway
and how these effects can be prevented by more strict regulations.
While some positive impact may result in the short-term from
strategic treatments, their main impact will probably be for the
long-term, except for those whose immune systems have suffered
irreversible damage or those who have an overwhelming genetic
predisposition to immune dysfunction. Because strategic treatments
appropriate to any specific individual are difficult to identify,
and difficult to eliminate because of ingrained habits, some
emphasis needs to be placed on those types of treatments that do
not require severe lifestyle alterations.
This work aims to analyze the strategies and
challenges regarding the development of effective vaccines against
SARS-COV-2. The discovery of a vaccine against the novel
coronavirus is an important component of the three-pronged approach
described initially, given that the pandemic cannot seem to be
entirely stopped by social distancing and good hygiene practices.
The tactical therapies identified so far have not been entirely
effective, especially for the most vulnerable individuals, being
unable to prevent severe disability and ultimately death. While
healthcare systems are still struggling not to crumble under
pressure from coronavirus patients, research laboratories around
the world are competing to produce an effective vaccine against
SARS-CoV-2 as soon as possible, in order to be able to stop the
spread of the new coronavirus.
2. Vaccines: an overview
The development of a vaccine is a complex and
time-consuming process, which differs from the development of
conventional medicines. Normally, the period of development of a
vaccine is 12-15 years (16).
While the conventional medicines are oriented towards the treatment
of a disease whose symptoms have emerged, vaccines are intended for
use in persons not yet exhibiting disease symptoms, in order to
prevent the occurrence of diseases (17). Clinical trials to demonstrate the
effectiveness of a vaccine focus on demonstrating its ability to
prevent the disease, with minimal adverse reactions in the
short-term since truly long-term studies in humans are rarely, if
ever, conducted (especially on the order of decades), which implies
the need to enroll more people than in traditional drug studies
(18).
Traditional vaccine development methods, although
extremely effective in combating highly contagious diseases such as
measles, require large amounts of viruses or bacteria, which can
last for months. Those microorganisms then become the key element
in a vaccine, the so-called antigen, that warns the human immune
system that some foreign organisms have invaded the body and must
be eliminated.
Vaccines provide the immune system with the
necessary instructions for recognizing and mobilizing lines of
defense against disease-causing microorganisms, such as bacteria or
viruses. In classical vaccines, antigens (distinctive molecular
markers) are introduced into the body, originating from inactivated
or half-active bacteria or attenuated viruses. These antigens are
capable of causing the disease, but are still capable of activating
the immune system, and its cells develop antibodies. If the person
comes in contact with the native pathogen, the immune system will
already have the necessary antibodies ready and will multiply them
much faster because it has already been sensitized by
vaccination.
Risk factors for anti-SARS-COV-2 vaccine
efficacy
Global immune deficiency is a risk factor for
anti-COVID-19 vaccine efficacy, particularly in elderly who have
been exposed to a myriad of factors that contribute to weakening of
the immune system, as described previously. These factors also
result in diseases such as obesity/obesity-related: e.g., type II
diabetes, metabolic syndrome and immune-mediated cancers.
Mechanistic reasons for these diseases include weakness of antigen
recognition, decreased immune cell quantity and functionality,
increased level/length and timing of humoral immune alterations of
components, reduced initiation of cellular responses, and memory
cell disorders. Other associations with immunodeficiency include
age-dependent humoral and immune cell alterations;
immunosenescence; malnutrition (19); protein-energy-micronutrient
deficit and telomere shortening (20). In addition, past or current
treatments affect the scalable ineffectiveness of vaccines in both
older adults (21) and children
(22), especially in
immunocompromised (23).
Exposure to immunosuppressive drugs (24) or residence in developing countries
(25) with a low socioeconomic
status is associated with a higher viral mortality compared to
residence in developed countries (26). Current global obesity prevalence
in adult and children is also a risk factor for anti-SARS-CoV-2
vaccine inefficacy due to higher levels of IL-6 (27) and decreased IgG concentrations.
Parasitic infections, respiratory tract infections such as
complicated pneumonia (28), can
also affect subsequent immune response to anti-SARS-CoV-2
vaccination (29).
Adjuvant purity and safety, knowledge gaps of the
relative contribution of the innate and adaptive responses to
protection against individual pathogens, and the precise mode of
action of individual adjuvants (30) are existing negative factors in
vaccine efficacy. Anti-SARS-CoV-2 vaccines will be available by
vaccine market players according to current orphan drug (31) and vaccine legislation (32,33) due to the fight against SARS-CoV-2
pandemic. This means that design defect, failure to warn, and
negligence in testing may also affect vaccine efficacy. Under those
conditions, long-term safety testing of vaccines in humans will be
non-existent. There is no data on whether anti-SARS-CoV-2 vaccines
will be first licensed in developed countries (34) or if all vaccine production
facilities will be adequate to assure a reliable supply of new
anti-SARS-CoV-2 vaccines (35) in
a timely manner, and are compatible with specifications and
improved adult vaccine design strategy by WHO or immunobiography
(36) and heterogeneity of immune
responses in the elderly subpopulations (36).
Vaccine safety
The safety of the vaccine is initially assessed in
laboratory studies with mice or rabbits. If the animals do not show
signs of disease after receiving the vaccine, then the tests begin
in humans, and the number of subjects gradually increases (37). Duration of the clinical trial, on
average, for a classical vaccine (after preclinical stage - in
vitro and in vivo tests) is as follows (38,39):
In phase I, also called the first human test, the
vaccine is given to a small group of healthy volunteers (10 to
100). The purpose is not to test whether the vaccine protects
against the disease, but whether it is safe or whether it causes
severe side effects.
In phase II, the candidate vaccine is administered
to a larger group of subjects (100-1,000), and in phase III, to an
even larger group (1,000-100,000). Separate studies may be required
in adults, children and the elderly. It sometimes happens that
vaccines that seem safe when given to a small number of people,
show side effects when given to a larger number of people. This is
explained by the fact that any rare complication is less likely to
occur when the group of subjects is small. Continuous monitoring is
important in case of complications that occur with delayed effect
(40). During a pandemic, these
sequential studies may be shortened and partially overlapped, but
it is important that thousands of vaccinated people are followed
for several months before the general purpose vaccine is
approved.
If there are more severe signs of inflammation or if
the vaccine causes the immune system to malfunction in other ways,
the candidate vaccine may not be suitable for widespread use. When
such adverse effects are identified, delays in the production
process inevitably occur (41).
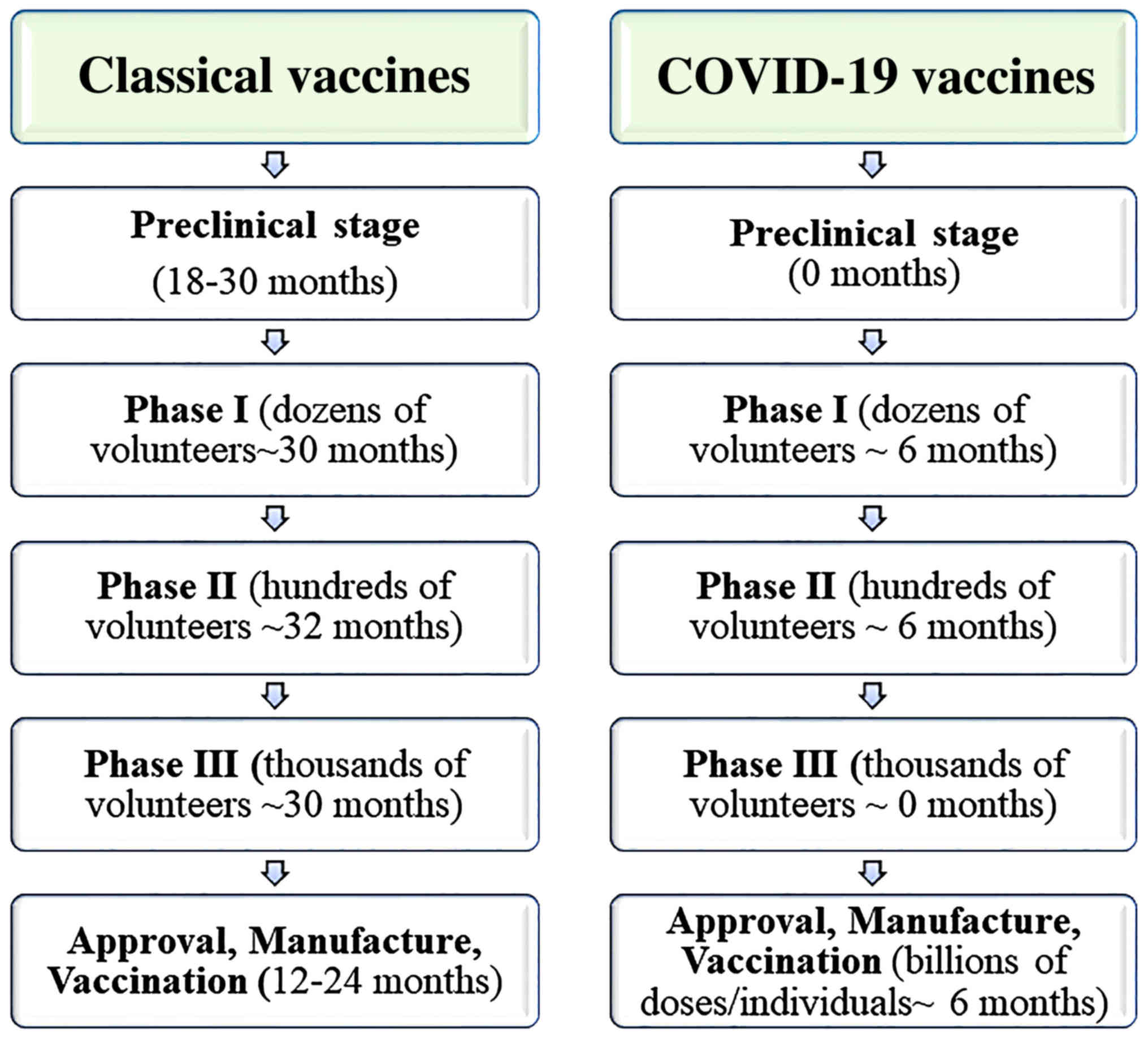
At present, only two potential vaccines against the SARS-CoV-2
virus are in the first phase and this by a derogation from the rule
- skipped animal studies (Fig.
1).
The first vaccine that does not follow all research
steps is called mRNA-1273 and will be tested in the first phase in
Seattle. There is an explanation for the speed of approval, which
is related to the fact that the coronavirus is not used in the
vaccine development. It is an experimental vaccine, made by a new
method, based on RNA. Basically, the RNA molecule in the vaccine
teaches the cells to produce the disease-specific antigen and
trains the body for the real enemy. But there is also a small
problem: to date, no vaccine based mRNA technology has been
approved for any disease, although a few have reached the second
phase of testing (42).
The second vaccine is Ad5-nCoV. The vaccine is
created by genetic engineering and tries to teach the body to
recognize the coronavirus protein S, which is delivered via a
type-5 adenovirus. Adenoviruses are a family of about one hundred
double-stranded DNA viruses that cause various diseases in humans
and animals, and type-5 is often used for genetic
recombination.
Although clinical trials have begun for these two
candidates and are likely to begin soon for other potential
vaccines, it is unlikely to move to phase II earlier than a few
months.
Vaccine efficacy
Vaccines stimulate immunity to the virus, but can
also stimulate inflammation in the body. This can manifest itself
either as pain or inflammation at the site where the subject was
injected, or as general symptoms such as fever or fatigue (symptoms
similar to COVID-19 disease). For most vaccines, these reactions
are mild and affect only a small proportion of subjects.
When a vaccine causes the production of antibodies,
in mice or in humans, this does not necessarily mean that the
vaccine will protect against the disease. Ideally, before moving on
to human studies, researchers should be able to demonstrate that
the vaccine will protect laboratory animals from the disease when
they are experimentally infected with the virus. In the case of
diseases caused by previous coronaviruses (SARS and MERS), it was
difficult to find an animal species that would be affected by the
disease in the same way as humans are affected. However, previous
research may help scientists speed up the animal testing process
for COVID-19. The protective effect of a human vaccine is assessed
using a series of studies similar to the series of safety studies.
If the vaccine elicits an immune system response, for example if
antibodies can be detected in the phase I study, then larger phase
II and phase III studies can be performed to see if the vaccine
protects against infection or disease.
Although COVID-19 is a new disease, research on SARS
and MERS has helped us understand how the human body responds to
coronaviruses and how the response of the immune system generates
protection against disease (43).
Restricted human studies can also be used to calculate the correct
dose and schedule for the vaccine. Some vaccines generate a strong
immune response after a single dose, but others require a booster
dose after a month or longer. This strategy also increases the
duration of studies.
To assess whether a vaccine prevents COVID-19 among
those exposed to the infection, it should be tested in phase III
studies, in a setting where the infection is actively occurring. In
response to the West African Ebola outbreak in 2014/15, the rVSV
Ebola vaccine went through all three phases of clinical development
in just 12 months, through the centralized coordination of WHO,
Médecins Sans Frontières. and manufacturer. During this period,
there were still cases of Ebola in Guinea, and the protection of
the vaccine was demonstrated by vaccinating the contacts of the
residual cases, as they appeared in the community (44).
Vaccine manufacturing
In the case of pandemic vaccines, tens or hundreds
of millions of doses are needed. This production process takes at
least six months, if the production lines already exist. Any new
vaccine involves a new production process, which involves several
quality control steps.
The manufacturer must ensure that each vaccine
produced is of consistent quality and requires repeated testing.
Moreover, because vaccine manufacturing is a biological process,
inevitably, some batches of vaccines will fail for reasons that are
not always clear, which can further delay production. There are
quite a few manufacturers in the world that can produce vaccines on
a large enough scale to meet the needs of a pandemic.
Vaccine regulation
Before initiating each stage of the human testing
process, the developer must provide evidence that the vaccine has
early indications of protection and is safe among those who have
been tested. Research ethics committees review clinical trial
plans, and authorities such as the European Medicines Agency (EMA)
and Food and Drug Administration (FDA) oversee the entire vaccine
development process before approving it for general use. These
assessments usually take several weeks or months (45). Although such approvals could be
shortened in the event of a pandemic, many potential COVID-19
vaccines use new technologies, so regulators will not be able to
rely on the experience of similar vaccines to speed up the process.
Developers of the COVID-19 vaccine were given a target to produce a
vaccine in 12-18 months, while historically, vaccines took 15-20
years to develop. There are diseases against which vaccines do not
yet exist, despite decades of work. For example, more than 100,000
children die each year from respiratory diseases caused by another
RNA virus, RSV (respiratory syncytial virus), which causes
pneumonia. Despite 50 years of research and 18 products developed,
there is currently no RSV vaccine available (46).
Numerous scientific research groups are working on
the development of a COVID-19 vaccine, using a number of different
approaches. Having many groups in the race is important, as most
vaccines entering clinical trials will fail for safety or efficacy
reasons.
3. The development of a COVID-19 vaccine:
current trends and prospects
SARS-COV-2: a brief summary
SARS-COV-2 is part of the coronavirus family, as is
SARS-CoV-1 (which generated the 2003 SARS epidemic) and MERS-CoV
(which generated the MERS epidemic in 2013) (47).
The new SARS-COV-2 coronavirus (severe acute
respiratory syndrome coronavirus 2) causes a respiratory infection
called COVID-19 (Coronavirus infectious disease - 2019), with an
average incubation period of 5 days (between 2 and 14 days)
(48). The disease is
characterized by predominant respiratory symptoms (cough,
difficulty breathing, fever) of moderate intensity in about 80% of
cases, but may have severe manifestations (bilateral interstitial
pneumonia), with progression to respiratory failure, acute
respiratory distress (ARDS), the main cause of mortality. There are
also cases with gastrointestinal manifestations (especially
diarrhea), and in some patients (especially in the young ones)
hypo/anosmia (loss of sense of smell) and hypo/dysgeusia
(alteration of the sense of taste) have been reported as early
symptoms (49).
Shortly after the publication of the genetic
structure of SARS-COV-2, the first two vaccines were developed.
SARS-COV-2 virus structure is composed of an RNA molecule,
surrounded by a series of structural and functional proteins
(50). Known structural proteins
are protein S (or spike - which leads to the characteristic
appearance), protein M (membrane), protein E (envelopes), protein N
(nucleocapsid). Of these, protein S has the role of attaching to
receptors in human cells and facilitating the fusion of viral
content with the cell. In the case of SARS-COV-2, the receptor is
the angiotensin converting enzyme 2 (ACE2), which is found in large
quantities in the respiratory tract and lung parenchyma (51-54). The initial infection of SARS-COV-2
occurs in the ciliary epithelial cells of the bronchi (55,56).
The binding of protein S to ACE2 is greatly favored
by the existence of the polybase cleavage site - a protein fragment
cleaved by the furin enzyme (commonly found in the human body),
allowing protein S fragments (S1, S2 and S3) to perform their
function. In addition to fusing the viral envelope with the cell
membrane, the virus also infects the human cell through endocytosis
(the cell membrane around the portion that binds to the virus forms
a vesicle inside the cell, detaches from the membrane and
introduces the virus into the cell, via that vesicle). Through the
process of entering the cell, the virus loses its envelope (which
fuses with the cell membrane) and the viral RNA is free in the
cytoplasm, carrying out two processes: replication and
transcription. First, the cellular components responsible for RNA
transcription produce viral proteins, and the RNA is copied.
Subsequently, these proteins and copies of the initial RNA are
'packed' into new virions, released from the cell to spread the
infection (57).
Humoral immunity consists of the production of
specific antibodies against these antigens. These are: i)
antibodies that appear rapidly during infection and disappear
rapidly (IgM); and ii) antibodies that appear slower and persist in
the body for a long time, even after the infection is cured (IgG)
(58).
Cellular immunity requires training certain immune
cells to recognize and destroy the virus. It has been observed in
SARS-COV-2 infection that the number of CD4+ and
CD8+ T lymphocytes is low in the peripheral blood
(because they are destroyed in the process of elimination of the
virus), but has high concentrations of activation markers (59). Regarding the SARS-CoV virus,
memory cells, capable of recognizing it, have been identified in
the blood of healed patients, several years after the infection.
The persistence of memory cells is an important element to consider
in the development of the anti-SARS-CoV-2 vaccine (60). Cases have been described where the
SARS-CoV-2 infection recurs after being considered cured (61).
On the other hand, there is evidence that
coronaviruses are equipped with mechanisms by which they can
sometimes evade immune attack. First, after infection of the cell,
the vesicles in which the virus is located are lacking in the
receptors that could recognize the virus as an 'invader' of the
body. Second, interferon synthesis (with antiviral function) is
inhibited by coronaviruses, and the presentation of viral antigens
by PCA is low in infected patients (62).
Strategies for COVID-19 vaccine
development
Current pandemic of COVID-19, caused by SARS-CoV-2,
is a global epidemiological problem, the solution of which will
require establishment of large-scale production of the vaccine.
Given the experience of previous coronavirus outbreaks, showing a
high variability of the virus, it is required to develop a vaccine
production platform, providing the scalability, technological
flexibility, and versatility. These vaccines must provide high
efficacy, safety, and tolerability.
This is due to the time required to prove that the
proposed vaccine is safe and effective as well as the time required
to produce millions of doses. Many 'candidate' vaccines, which
initially look promising, are likely to fail during the testing
process. The process of developing a new vaccine begins with a
so-called 'candidate' vaccine, which a team of scientists believes
to have potential. This is the fastest part of the process and
takes up to a few weeks. Thus, some candidate vaccines are being
developed in Russia against COVID-19. The Shemyakin and Ovchinnikov
Institute of Bioorganic Chemistry is developing
Liposome-encapsulated DNA-protein vaccine based on the COVID-19
Spike antigens and its DNA coding sequence and VLP vaccine based on
HSB antigen fused with COVID-19 Spike antigens. The vaccine
candidates against COVID-19 based on live attenuated recombinant
influenza vector platform are developed by FSBSI 'Chumakov Federal
Scientific Center for Research and Development of Immune- and
Biological Products of Russian Academy of Sciences' and
Smorodintsev Research Institute of Influenza (WHO National
Influenza Centre). The FSBSI 'Chumakov Federal Scientific Center
for Research and Development of Immune- and Biological Products of
Russian Academy of Sciences' is developing two vaccines based on an
attenuated and an inactivated strain of the COVID-19 virus isolated
in Centre Chumakov (personal communication of Professor
Egorov).
For efficient application of SARS-CoV-2 vaccines,
not only the production procedure needs to be developed, but also
requires a large-scale production followed by vaccination program
targeting muti-million population of different regions. Only such a
complex approach will allow combatting of the coronavirus disease
pandemic. New technologies do not require the work with live virus,
making their production much easier, but still needing further
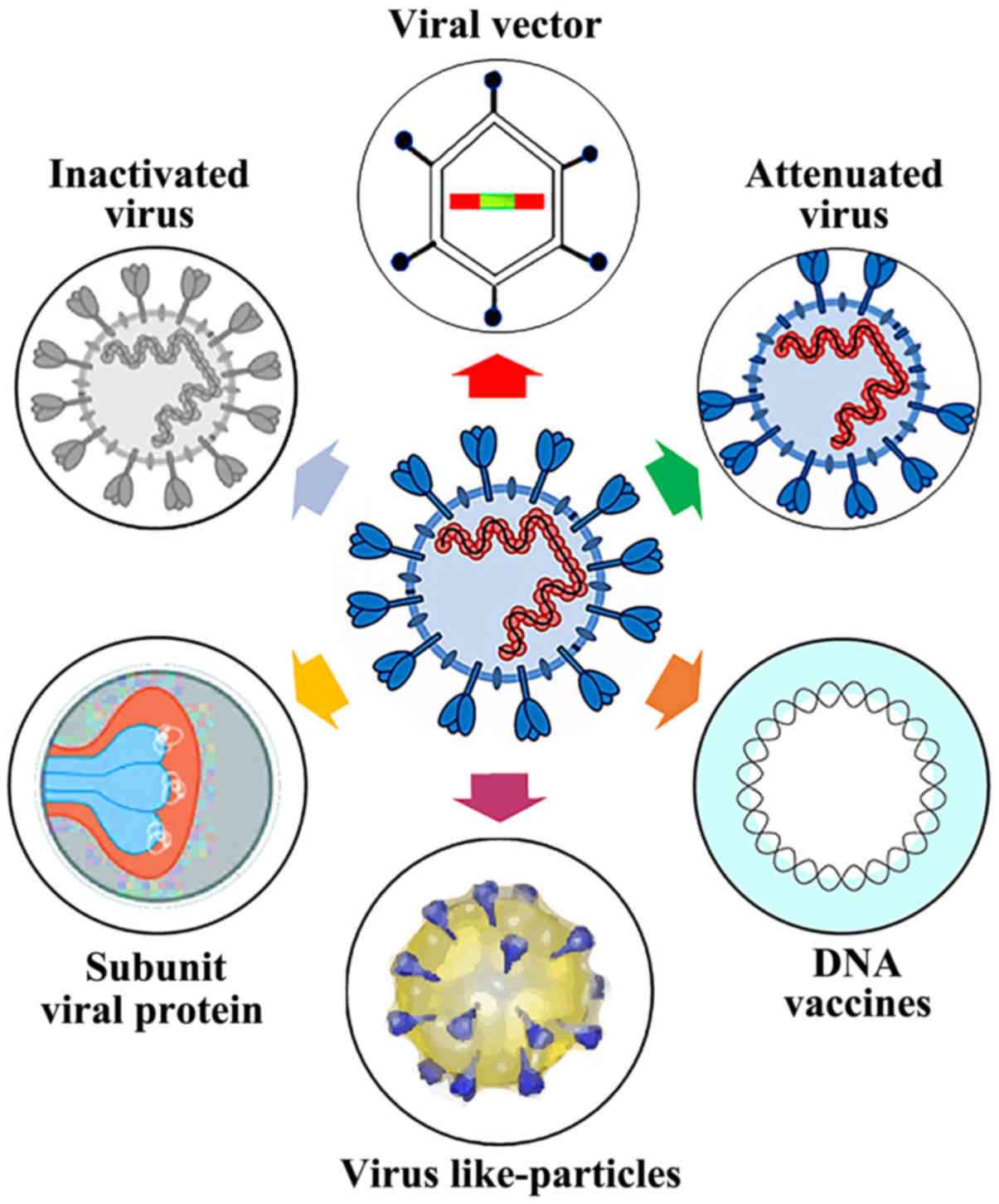
scientific and technological evidence (Fig. 2).
Type of vaccines against SARS-COV-2 RNA
vaccines
The mRNA (encoding RiboNucleic Acid) vaccine encodes
a stable prefused form (the form before being fused to the cell
membrane of the host cell) of Spike protein (S) (63). Protein complex S is required for
membrane fusion and host cell infection and has been the target of
vaccines against MERS and SARS.
Membrane fusion (viral and cellular) is an essential
step when encapsulated viruses enter cells. Fusion of the double
lipid layer requires catalysis to overcome a high kinetic barrier,
and viral fusion proteins are agents that perform this catalytic
function (64). Although
coronavirus uses many different proteins to replicate and invade
cells, protein S is the main surface protein that it uses to bind
to a receptor - this becomes a bridge to the human cell. After
protein S binds to the human cell receptor, the viral membrane
fuses with the human cell membrane, allowing the viral genome to
enter the human cells and initiate infection (49).
The mRNA vaccine against SARS-COV-2 is based on a
relatively new genetic method that does not require growing the
virus in the laboratory. The technique transforms the human body
into a 'living laboratory', because the whole process is no longer
carried out in the laboratory, but directly in the body that
received the vaccine (65).
The technique is actually based on messenger
ribonucleic acid (mRNA) fragments, the genetic material that is
copied from DNA and encodes proteins. Moderna company loads its
vaccine with viral mRNA, which encodes the proteins of the new
coronavirus, and then injects it into the body. Immune cells from
the lymph nodes process the mRNA and synthesize specific viral
protein antigens so that other immune cells recognize them
(66). mRNA is like a software
molecule in the body, which then produces viral proteins that can
generate an immune response (67). How the body processes the viral
protein is often very different from the way it processes the same
protein of the vaccine. Thus, one of the theoretical benefits of
introducing mRNA into a vaccine is that the body causes the viral
protein to behave in exactly the same way that the virus would have
directed the host to do (63)
(Table I).
 | Table IRNA candidate vaccines approved by
WHO as of April 2020 (68). |
Table I
RNA candidate vaccines approved by
WHO as of April 2020 (68).
| Candidate
vaccine | Producer | Current stage of
development | Viruses for which
the same strategy was used |
|---|
| LNP-encapsulated
mRNA | Moderna/NIAID | Clinical
evaluation, Phase I NCT04283461 | Multiple
candidates |
| LNP-encapsulated
mRNA encoding RBD | Fudan
University/Shanghai JiaoTong University/RNACure Biopharma | Pre-clinical | - |
| LNP encapsulated
mRNA cocktail encoding VLP | Fudan
University/Shanghai JiaoTong University/RNACure Biopharma | Pre-clinical | - |
| Replicating
Defective SARS-COV-2 derived RNAs | Centro Nacional
Biotecnología (CNB-CSIC), Spain | Pre-clinical | - |
| LNP-encapsulated
mRNA | University of
Tokyo/Daiichi-Sankyo | Pre-clinical | MERS |
|
Liposome-encapsulated mRNA | BIOCAD | Pre-clinical | - |
| mRNA | China CDC/Tongji
University/Stermina | Pre-clinical | - |
| mRNA |
Arcturus/Duke-NUS | Pre-clinical | Multiple
candidates |
| mRNA | BioNTech/Fosun
Pharma/Pfizer | Pre-clinical | - |
| saRNA | Imperial College
London | Pre-clinical | EBOV, LASV, MARV,
Inf (H7N9), RABV |
| mRNA | Curevac | Pre-clinical | RABV, LASV, YFV,
MERS, InfA, ZIKV |
DNA vaccines
DNA vaccines represent the latest direction of
development in the manufacture of vaccines. Vaccines obtained by
recombinant DNA technology are produced by genetic
modification.
The DNA encoding the target molecule is introduced
via a plasmid or viral vector in a suitable microorganism or cell
line, in which DNA is expressed and translated into protein. The
product is subsequently recovered by extraction and purification.
The injected DNA is a plasmid plus a promoter that provides
immunogenic protein synthesis (69).
An advantage of this type of vaccine is that it
stimulates both humoral and cellular immunity. Also, this type of
vaccine is stable and does not require maintenance under controlled
conditions (refrigeration), as is the case for conventional
vaccines. In contrast to live attenuated vaccines, the risks
arising from a potential inadequate attenuation of vaccine strains
are not present in DNA vaccines (70). Other advantages of this new
technology are the fact that the plasmids used are easy to
manufacture in large quantities, and the immunity conferred is
long-term. The disadvantages identified so far with regard to DNA
vaccines are due to the fact that this type of vaccine is limited
to protein immunogens (71).
A clinical trial for a new DNA vaccine against
Coronavirus has begun. This is called ChAdOx1 nCoV-19 and it was
initially developed to prevent MERS (72). This vaccine is based on an
adenovirus vaccine vector and on SARS-COV-2 spike protein (73). It has been modified so that it
cannot be reproduced in the human body and the genetic code that
transmits instructions for the production of Coronavirus Spike
protein has been added, allowing the adenovirus to produce this
protein after vaccination. The result is the formation of
antibodies against the Spike protein, known to be on the surface of
SARS-COV-2 (74).
The INO-4800 vaccine is another new DNA vaccine to
prevent SARS-COV-2 infection. It is entering human phase I testing
(75). The Phase I study will
enroll up to 40 healthy adult volunteers. Each participant will
receive two doses of INO-4800 every four weeks, and initial immune
responses and safety data from the study are expected by the end of
the summer. Preclinical data have shown promising results in immune
response in several animal models. Also, the preclinical results
for the INOVIO SARS-COV-2 vaccine were consistent with the results
of the Phase I study for the Middle East Respiratory Syndrome
(MERS) vaccine, caused by another type of coronavirus. Here, the
INOVIO DNA vaccine was well tolerated and induced high levels of
antibody response in 95% of subjects, generating T cell-mediated
responses in nearly 90% of study participants. Long-term antibody
response was maintained for up to 60 weeks in the case of the
anti-MERS vaccine (75) (Table II).
 | Table IIDNA candidate vaccines approved by
WHO as of 11 April 2020 (68). |
Table II
DNA candidate vaccines approved by
WHO as of 11 April 2020 (68).
| Candidate
vaccine | Producer | Current stage of
development | Viruses for which
the same strategy was used |
|---|
| DNA plasmid
vaccine | Inovio
Pharmaceuticals | Clinical trial | Lassa, Nipah,
HIV |
| Electroporation
device | | Phase I
NCT04336410 | Filovirus,
HPV
Cancer indications Zika, Hepatitis B |
| DNA plasmid
vaccine | Zydus Cadila | Pre-clinical | |
| DNA with
electroporation | Karolinska
Institute/Cobra Biologics (OPENCORONA Project) | Pre-clinical | |
| DNA plasmid
vaccine | Osaka
University/AnGes/Takara Bio | Pre-clinical | |
| DNA | Takis/Applied DNA
Sciences/Evvivax | Pre-clinical | |
| Plasmid DNA,
Needle-Free Delivery | Immunomic
Therapeutics, Inc./EpiVax, Inc./PharmaJet, Inc. | Pre-clinical | SARS |
Protein subunit
Subunit vaccines contain only certain antigenic
determinants of pathogenic microorganisms, and are obtained either
starting from conventional cultivation processes, or by recombinant
DNA technology (76).
Antigen determinants included in the vaccine
increase the efficiency of the immune response, and the presence of
a small number of pathogens reduces the risk of side effects.
Subunit vaccines mainly contain surface fragments of pathogens.
Because these types of structures are weak immunogens, to obtain
vaccines with suitable efficacy, the antigens are conjugated with
protein molecules (77).
Antigen purifiers sometimes lead to loss of
immunogenicity, requiring coupling with a protein carrier or with
an aluminum salt. However, adjuvants are associated with the onset
of local reactions to the vaccination site. Also, the duration the
immunity conferred by vaccination is lower, except for live
vaccines (78) (Table III).
 | Table IIIProtein subunit candidate vaccines
approved by WHO as of April 2020 (68). |
Table III
Protein subunit candidate vaccines
approved by WHO as of April 2020 (68).
| Candidate
vaccine | Producer | Current stage of
development | Viruses for which
the same strategy was used |
|---|
| Capsid-like
Particle AdaptVac | (PREVENT-nCoV
consortium) | Pre-clinical | |
| Drosophila S2
insect cell expression system VLPs ExpreS2ion | | Pre-clinical | |
| Drosophila S2
insect cell expression system VLPs | ExpreS2ion | Pre-clinical | |
| Peptide antigens
formulated in lipid nanoparticle formulation | IMV Inc | Pre-clinical | |
| S protein | WRAIR/USAMRIID | Pre-clinical | |
| S protein +
Adjuvant | National Institute
of Infectious Disease, Japan | Pre-clinical | Influenza |
| VLP recombinant
protein + adjuvant | Osaka
University/BIKEN/National Institutes of Biomedical Innovation,
Japan | Pre-clinical | |
| Native-like
trimeric subunit Spike protein vaccine | Clover
Biopharmaceuticals Inc./GSK/Dynavax | Pre-clinical | HIV, REV
Influenza |
| Microneedle arrays
S1 subunit | University of
Pittsburgh | Pre-clinical | MERS |
| Peptide | Vaxil Bio | Pre-clinical | |
| Adjuvanted protein
subunit (RBD) | Biological E
Ltd | Pre-clinical | |
| Peptide | Flow Pharma
Inc | Pre-clinical | Ebola, Marburg,
HIV, Zika, Influenza, HPV therapeutic vaccine, BreastCA
vaccine |
| S protein | AJ Vaccines | Pre-clinical | |
| Ii-Key peptide | Generex/EpiVax | Pre-clinical | Influenza, HIV,
SARS-CoV |
| S protein | EpiVax/Univ. of
Georgia | Pre-clinical | H7N9 |
| S protein
(baculovirus production) | Sanofi Pasteur | Pre-clinical | Influenza,
SARS-CoV |
| VLPrecombinant
protein nanoparticle vaccine + Matrix M | Novavax | Pre-clinical | RSV; CCHF, HPV,
VZV, EBOV |
| gp-96 backbone | Heat
Biologics/Univ. of Miami | Pre-clinical | NSCLC, HIV,
malaria, Zika |
| Molecular clamp
stabilized Spike protein | University of
Queensland/GSK/Dynavax | Pre-clinical | Nipah, influenza,
Ebola, Lassa |
| S1 or RBD
protein | Baylor College of
Medicine | Pre-clinical | - |
| Subunit protein,
plant produced |
iBio/CC-Pharming | | - |
| Recombinant
protein, nanoparticles (based on S protein and other epitopes) | Saint-Petersburg
Scientific Research Institute of Vaccines and Serums | Pre-clinical | - |
| SARS-COV-2 XWG-03
truncated S (spike) proteins Innovax/Xiamen University | GSK | Pre-clinical | HPV |
| Adjuvanted
microsphere peptide | VIDO-InterVac,
University of Saskatchewan | Pre-clinical | - |
| Synthetic Long
Peptide Vaccine candidate for S and M proteins | OncoGen | Pre-clinical | - |
Viral vectors
Along with traditional virus vaccines, viral vectors
are widely used, in which genome of one virus is used to deliver
the antigen of another virus, thus allowing development of a
platform technology of virus production. These technologies are
available for large-scale production of vaccines. Drawbacks of such
vaccines include a large variation of purification methods, need
for a reliable confirmation of purity and activity of the virus
(79) (Tables IV and V).
 | Table IVReplicating viral vector vaccines
approved by WHO as of 11 April 2020 (68). |
Table IV
Replicating viral vector vaccines
approved by WHO as of 11 April 2020 (68).
| Candidate
vaccine | Producer | Current stage of
development | Viruses for which
the same strategy was used |
|---|
| Measles Vector | Zydus Cadila | Pre-clinical | - |
| Measles Vector | Institute
Pasteur/Themis/University of Pittsburg Center for Vaccine
Research | Pre-clinical | West nile, chik,
Ebola, Lassa, Zika |
| Horsepox vector
expressing S protein | Tonix
Pharma/Southern Research | Pre-clinical | Smallpox,
monkeypox |
| Live viral vectored
vaccine based on attenuated influenza virus backbone
(intranasal) | BiOCAD and IEM | Pre-clinical | - |
| Influenza vector
expressing RBD | University of Hong
Kong | Pre-clinical | - |
| VSV vector
expressing S protein | IAVI/Batavia | Pre-clinical | Ebola, Marburg,
Lassa |
 | Table VNon-replicating viral vector
candidate vaccines approved by WHO as of 11 April 2020 (68). |
Table V
Non-replicating viral vector
candidate vaccines approved by WHO as of 11 April 2020 (68).
| Candidate
vaccine | Producer | Current stage of
development | Viruses for which
the same strategy was used |
|---|
| Adenovirus type 5
vector (Ad5-nCoV) | CanSino Biological
Inc/Beijing Institute of Biotechnology | Clinical - Phase
I
ChiCTR2000031781
Clinical - Phase II
ChiCTR2000030906 | Ebola |
| ChAdOx1 | Advent-IRBM,
Pomezia - Italy, and Jenner Institute of the University of
Oxford | Phase I/II
NCT04324606 | MERS, influenza,
TB, Chikungunya, Zika, MenB, plague |
| MVA encoded
VLP |
GeoVax/BravoVax | Pre-clinical | LASV, EBOV, MARV,
HIV |
| Ad26 (alone or with
MVA boost) | Janssen
Pharmaceutical Companies | Pre-clinical | Ebola, HIV,
RSV |
| MVA-S encoded | DZIF - German
Center for Infection Research | Pre-clinical | Many |
| Adenovirus based
NasoVAX expressing SARS2-CoV spike protein | Altimmune | Pre-clinical | Influenza |
| Ad5 S (GREVAX™
platform) | Greffex | Pre-clinical | MERS |
| Oral vaccine
platform | Vaxart | Pre-clinical | InfA, CHIKV, LASV,
NORV, EBOV, RVF, HBV, VEE |
| MVA expressing
structural proteins | Centro Nacional
Biotecnología (CNB-CSIC), Spain | Pre-clinical | HIV, HCV,
chikungunya, Ebola, zika, malaria, leishmania |
VLPs (virus-like particles)
Vaccine development based on the recombinant
proteins and virus-like particles (VLPs) is a more innovative
approach. Antiviral vaccines are usually developed on the basis of
surface proteins that form VLPs. Production of VLPs in the cells
with further reconstruction into the stable and immunogenic forms
is a multi-stage process. Substantial issues appear for the VLP
production for non-enveloped viruses. Nucleic acid based vaccines
are very interesting from the platform technology viewpoint,
allowing the same process to be used for the production of
different antigens. They elicit both humoral and cell immune
responses (80).
Production of these vaccines may be based on E.
coli cell fermentation and corresponding technologies of
extraction and purification of plasmids ensuring their structural
integrity (81) (Table VI).
 | Table VIVLP (virus like particle) candidates
vaccines approved by WHO as of April 2020 (68). |
Table VI
VLP (virus like particle) candidates
vaccines approved by WHO as of April 2020 (68).
| Candidate
vaccine | Producer | Current stage of
development | Viruses for which
the same strategy was used |
|---|
| Virus-like
particle, based on RBD displayed on virus-like particles | Saiba GmbH | Pre-clinical | Flu, rotavirus,
norovirus, West Nile virus, cancer |
| Plant-derived
VLP | Medicago Inc. | Pre-clinical | |
| ADDomer™
multiepitope display | Imophoron Ltd and
Bristol University's Max Planck Centre | | |
Inactivated virus
Another category of vaccines is vaccines containing
whole microorganisms, but inactivated by chemical or physical
methods. This type of vaccine has the advantage of higher
stability; however, effectiveness is lower and requires reminders
of immune system. Vaccines with inactivated live virus require
stabilization of the structure in the dry form, separate supply of
the solvent, and cold-chain transportation. These factors
complicate the production process and lead to increase of their
cost (82) (Table VII).
 | Table VIIInactivated virus candidate vaccines
approved by WHO as of 11 April 2020 (68). |
Table VII
Inactivated virus candidate vaccines
approved by WHO as of 11 April 2020 (68).
| Candidate
vaccine | Producer | Current stage of
development | Viruses for which
the same strategy was used |
|---|
| Inactivated +
alum | Sinovac | Pre-clinical | SARS |
| Inactivated | Beijing Institute
of Biological Products/Wuhan Institute of Biological Products | Pre-clinical | |
| TBD | Osaka
University/BIKEN/NIBIOHN | Pre-clinical | |
Live attenuated virus
Live attenuated vaccines were the first vaccines
utilized. These types of vaccines are obtained through cultivation
of microorganisms under suboptimal conditions or through successive
passage in cultures, techniques that determine attenuation of
virulence while maintaining the capacity to induce the immune
response (83).
Although characterized as very effective, live
attenuated vaccines have the disadvantages of risks related to the
possible occurrence of mutations, leading to the virulence
reversal, as well as contra-indication in the case of
immunocompromised persons (Table
VIII).
 | Table VIIILive attenuated virus candidate
vaccines approved by WHO as of 11 April 2020 (68). |
Table VIII
Live attenuated virus candidate
vaccines approved by WHO as of 11 April 2020 (68).
| Candidate
vaccine | Producer | Current stage of
development | Viruses for which
the same strategy was used |
|---|
| Deoptimized live
attenuated vaccines | Codagenix/Serum
Institute of India | Pre-clinical | HAV, InfA, ZIKV,
FMD, SIV, RSV, DENV |
Others
The LV-SMENP-DC Vaccine (Lentiviral Minigene
Vaccine, Shenzhen Geno-Immune Medical Institute) entered Phase I
clinical trial in March 2020. It is a vaccine based on dendritic
cells modified with a lentiviral vector expressing synthetic
minigens based on selected viral protein domains, and is
administered with antigen-specific cytotoxic T lymphocytes
(84).
Pathogen-specific aAPC vaccine (Shenzhen Geno-Immune
Medical Institute), Phase 1 clinical trial (85) is based on lentiviral vector
modified artificial antigen presenting cells (aAPCs) expressing
synthetic minigens based on selected viral protein domains.
4. Discussion
The challenges of COVID-19 vaccines.
Where do we stand?
The development and manufacture of a COVID-19
vaccine is an urgent issue, but it is likely to take many months to
resolve. Although many companies have announced that the COVID-19
vaccine will be ready soon, this will be quite difficult to do in
reality (72).
The main reason is that before being put on the
market, the vaccine should be safe, both in the short-term and in
the long-term. This is very important because, in the history of
vaccine production, there have been situations of contamination
with other viruses, fortunately without major consequences
(16). For example, one third of
the polio vaccines administered in the US between 1955 and 1963
also contained simian virus 40 (SV40) and more recently, rotavirus
vaccines have been discovered to also have swine circoviruses
(86-88).
In order to avoid such situations, everything to be
tested on humans should first be checked for purity, and then
sterile production lines provided. This takes time. In rare cases,
certain antibodies generated by immunization may promote an
aggravated form of the disease (a situation called ADE, antibody
dependent enhancement) (89).
When these antibodies re-establish contact with the virus, they
will actually help it enter the cells and cause infection. ADE has
been described in many viral infections (influenza, Dengue, Zika,
etc.), but also in coronaviruses. The mechanism of ADE has not been
confirmed for coronavirus in humans (90).
Several animal studies have shown that some types of
anti-SARS and anti-MERS vaccines, although effective in generating
antibodies, can lead to more severe forms of disease when the virus
is subsequently inoculated (91).
The second reason is that the vaccine must not only
be safe, but also effective. It must be able to determine the
synthesis of antibodies of a certain type, at a certain
concentration (titer) and to provide protection for a reasonable
time.
However, vaccines never generate immunity to all
vaccinated people (92). The
causes are complex and vary from genetic and immunological factors,
to the quality of the vaccines themselves and how they are
administered.
Age is an important aspect and some influenza
studies have shown that aging of the immune system dramatically
decreases the effectiveness of vaccination (93). Therefore, for any future
anti-SARS-COV-2 vaccine, all these aspects should be evaluated, and
the primary immunization failures minimized by adjusting the doses
or number of administrations.
Assuming that the vaccine will generate an effective
immune response to a sufficient number of individuals among those
vaccinated, the time frame of vaccine protection is questionable
(94). For example, after measles
vaccination, a small percentage of those who initially respond
well, lose their protective antibody status within a few years, a
phenomenon called secondary immunization failure (95).
This is why it takes time to check the
post-vaccination persistence of anti-COVID antibodies. Last but not
least, both safety and efficacy are significantly dependent on the
type of vaccine, i.e. the technology or platform used.
Some technologies are very new and therefore require
more careful testing. Others are old but need to be adapted for
COVID-19. Another aspect is not only the ability of a company to
develop the technology, but also its large-scale production
capacity so that it is quickly accessible globally. Because there
is no commercially available anti-SARS-COV-2 vaccine precedent for
the proposed platforms, completely new production lines, capable of
generating billions of doses in a few months, must be considered.
And this must be done without stopping the production of current
vaccines, already included in the official protocols and
guidelines. At present, this capacity will be reached with
difficulty and will represent an unprecedented effort. DNA and RNA
vaccines are based on the principle of insertion of these nucleic
acids in some cells of the vaccinated ones, forcing them to make
immunogenic viral proteins (96).
Although some recent data seem encouraging, these concepts have
questionable efficiency in humans. Non-replicative vectors are
actually common viruses (e.g., adenovirus) genetically modified to
display SARS-CoV proteins on the outer surface (97). But they are so common that many of
us have already met with them throughout our lives and as a result,
we already have immunity and neutralize them before they do their
job.
The attenuated viruses would be variants of
SARS-CoV-2 made less or not at all pathogenic by genetic
engineering. They are by far the most immunogenic, but there is a
risk that they will become pathogenic after mutations (98).
Inactivated viruses, viral fragments, and synthetic
peptides are all relatively weakly immunogenic.
The analysis of these variables is normally done
over 10 years, in the form of a trial. The stages of a trial are
inflexible, standardized, intensely regulated for the purpose of
maximum safety and efficacy of the final vaccine.
The first stage is the preclinical research, in
which the technology that will be included in the vaccine is
selected and then the efficacy and safety profile of human cells
(in vitro) and animal models (in vivo) is tested. If
the expected anti-infectious effect is seen in vitro, and
cells do not die excessively, then in vivo animal studies
are conducted. Mice are usually the perfect candidate for in
vivo studies, because they are about 85% genetically similar to
humans, relatively inexpensive, and do not raise major ethical
issues (99). However, one of the
notable differences between the human and the mouse is fixed in the
case of the ACE2 receptor gene (to which SARS viruses bind during
infection). Mice simply do not make the respiratory syndromes, they
eliminate the virus relatively quickly from the body. This is why
it is necessary for either the genetically modified mouse to have a
human ACE2 receptor, or to use ferrets and monkeys, and this makes
everything much more expensive and more difficult to access.
The preclinical stage lasts between 1½ and 2½ years
and is by far the most selective. It is estimated that less than
20% of the vaccines tested are able to progress to human tests.
Some fail because the product does not work, others because they
can no longer find financing (1,100).
After obtaining the vaccine, the first people
vaccinated will be the highest risk categories. For everyone else,
the term is sometime in 2022, possibly long after we have naturally
acquired immunity.
There are still many unknowns related to coronavirus
immunity and that is why obtaining a vaccine is difficult. Future
mutations in SARS-COV-2 could occur at any time, which
automatically means a high risk of any vaccine becoming useless. No
vaccine has been obtained for any other coronavirus so far, and no
one can guarantee its success yet. It is possible that we will have
the anti-COVID vaccine in a very short time, without having the
benefits from long-term testing results. The vaccine will probably
not be available to all of us for 18 months.
5. Conclusion and future perspectives
Scientists began working on coronavirus vaccines
during SARS and MERS outbreaks, but their efforts did not
materialize because of a myriad of difficulties. Since this
extremely severe current coronavirus pandemic, COVID-19, the spread
of the outbreak appears much broader than was the case for SARS.
There is also the possibility of the disease becoming endemic and
seasonal in its appearance, according to some investigators. This
explains why many research groups and companies are undertaking
efforts to develop an effective vaccine against SARS-CoV-2 all over
the world, also speeding up all the usual phases needed to develop
and test a vaccine in the human.
An important feature in the landscape of vaccine
research and development for SARS-COV-2 is represented by the
varied range of evaluated technological platforms, including
nucleic acids (DNA and RNA), virus-like particles, peptides, viral
vector (replicative and non-replicative), recombinant proteins,
live attenuated viruses and inactivated viruses. Many of these
platforms are not currently the basis of vaccines already
authorized, but experience in areas such as oncology encourages
developers to exploit new opportunities for increased development
and manufacturing speeds.
There is not an effective therapy for severe
COVID-19, and social distancing is extremely 'costly' from a social
and economic perspective. Therefore, the development of an
effective vaccine, along with efforts to implement immune-enhancing
strategic treatments and shorter-term efforts to identify tactical
repurposed treatments, should be considered major public health
priorities. We hope to get this key tool for disease prevention,
and to do this quite soon.
It is unknown whether there ever will be a
successful SARS-CoV-2 vaccine, but the efforts of the scientific
community in attempting to develop such a vaccine is without
precedent. Thus, COVID-19 vaccines are a never ending story.
Funding
No funding was received.
Availability of data and materials
Not applicable.
Authors' contributions
Conceptualization: DC, AT, AOD and DAS; validation,
research, resources, data reviewing, and writing: DP, AME, AAI,
AGG, MIS, RK, FC, MV; review and editing: DC, AOD, AT, DP and RK.
All authors read and approved the final manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
DAS is the Editor-in-Chief for the journal, but had
no personal involvement in the reviewing process, or any influence
in terms of adjudicating on the final decision, for this article.
The other authors declare that they have no competing
interests.
Acknowledgments
Not applicable.
References
|
1
|
Yuen KS, Ye ZW, Fung SY, Chan CP and Jin
DY: SARS-CoV-2 and COVID-19: The most important research questions.
Cell Biosci. 10:402020. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Kostoff RN: Combining Tactical and
Strategic Treatments for COVID-19. Georgia Institute of Technology;
2020, http://hdl.handle.net/1853/62523
Accessed March 23 2020.
|
|
3
|
Hashem MM, Abo-El-Sooud K, Abd-Elhakim YM,
Badr YA, El-Metwally AE and Bahy-El-Dien A: The long-term oral
exposure to titanium dioxide impaired immune functions and
triggered cytotoxic and genotoxic impacts in rats. J Trace Elem Med
Biol. 60:1264732020. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Torres M, Carranza C, Sarkar S, Gonzalez
Y, Osornio Vargas A, Black K, Meng Q, Quintana-Belmares R,
Hernandez M, Angeles Garcia JJF, et al: Urban airborne particle
exposure impairs human lung and blood Mycobacterium tuberculosis
immunity. Thorax. 74:675–683. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Goumenou M, Sarigiannis D, Tsatsakis A,
Anesti O, Docea AO, Petrakis D, Tsoukalas D, Kostoff R, Rakitskii
V, Spandidos DA, et al: COVID-19 in Northern Italy: An integrative
overview of factors possibly influencing the sharp increase of the
outbreak (Review). Mol Med Rep. 22:20–32. 2020.PubMed/NCBI
|
|
6
|
Omran GA: Hematological and immunological
impairment following in-utero and postnatal exposure to aluminum
sulfate in female offspring of albino rats. Immunopharmacol
Immunotoxicol. 41:40–47. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Reid N, Moritz KM and Akison LK: Adverse
health outcomes associated with fetal alcohol exposure: A
systematic review focused on immune-related outcomes. Pediatr
Allergy Immunol. 30:698–707. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Engin AB, Nikitovic D, Neagu M,
Henrich-Noack P, Docea AO, Shtilman MI, Golokhvast K and Tsatsakis
AM: Mechanistic understanding of nanoparticles' interactions with
extracellular matrix: The cell and immune system. Part Fibre
Toxicol. 14:222017. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Delfosse VC, Tasat DR and Gioffré AK: In
vivo short-term exposure to residual oil fly ash impairs pulmonary
innate immune response against environmental mycobacterium
infection. Environ Toxicol. 30:589–596. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Tsatsakis A, Docea AO, Constantin C,
Calina D, Zlatian O, Nikolouzakis TK, Stivaktakis PD, Kalogeraki A,
Liesivuori J, Tzanakakis G, et al: Genotoxic, cytotoxic, and
cytopathological effects in rats exposed for 18 months to a mixture
of 13 chemicals in doses below NOAEL levels. Toxicol Lett.
316:154–170. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Docea AO, Calina D, Goumenou M, Neagu M,
Gofita E and Tsatsakis A: Study design for the determination of
toxicity from long-term-low-dose exposure to complex mixtures of
pesticides, food additives and lifestyle products. Toxicol Lett.
258:S1792016. View Article : Google Scholar
|
|
12
|
Docea AO, Gofita E, Goumenou M, Calina D,
Rogoveanu O, Varut M, Olaru C, Kerasioti E, Fountoucidou P,
Taitzoglou I, et al: Six months exposure to a real life mixture of
13 chemicals' below individual NOAELs induced non monotonic
sex-dependent biochemical and redox status changes in rats. Food
Chem Toxicol. 115:470–481. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Docea AO, Goumenou M, Calina D, Arsene AL,
Dragoi CM, Gofita E, Pisoschi CG, Zlatian O, Stivaktakis PD,
Nikolouzakis TK, et al: Adverse and hormetic effects in rats
exposed for 12 months to low dose mixture of 13 chemicals: RLRS
part III. Toxicol Lett. 310:70–91. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Fountoucidou P, Veskoukis AS, Kerasioti E,
Docea AO, Taitzoglou IA, Liesivuori J, Tsatsakis A and Kouretas D:
A mixture of routinely encountered xenobiotics induces both redox
adaptations and perturbations in blood and tissues of rats after a
long-term low-dose exposure regimen: The time and dose issue.
Toxicol Lett. 317:24–44. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Tsatsakis A, Petrakis D, Nikolouzakis TK,
Docea AO, Calina D, Vinceti M, Goumenou M, Kostoff RN, Mamoulakis
C, Aschner M, et al: COVID-19, an opportunity to reevaluate the
correlation between long-term effects of anthropogenic pollutants
on viral epidemic/pandemic events and prevalence. Food Chem
Toxicol. In Press.
|
|
16
|
Han S: Clinical vaccine development. Clin
Exp Vaccine Res. 4:46–53. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Kamal AM, Mitrut P, Docea AO, Soşoi S,
Kamal KC, Mitrut R, Mărgăritescu D, Călina D, Banciu C, Tica OS, et
al: Double therapy with pegylated Interferon and Ribavirin for
chronic hepatitis C. A pharmacogenenetic guide for predicting
adverse events. Farmacia. 65:877–884. 2017.
|
|
18
|
Black S: The costs and effectiveness of
large Phase III pre-licensure vaccine clinical trials. Expert Rev
Vaccines. 14:1543–1548. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Wouters-Wesseling W, Rozendaal M, Snijder
M, Graus Y, Rimmelzwaan G, De Groot L and Bindels J: Effect of a
complete nutritional supplement on antibody response to influenza
vaccine in elderly people. J Gerontol A Biol Sci Med Sci.
57:M563–M566. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Tsoukalas D, Fragkiadaki P, Docea AO,
Alegakis AK, Sarandi E, Vakonaki E, Salataj E, Kouvidi E, Nikitovic
D, Kovatsi L, et al: Association of nutraceutical supplements with
longer telomere length. Int J Mol Med. 44:218–226. 2019.PubMed/NCBI
|
|
21
|
Ventura MT, Casciaro M, Gangemi S and
Buquicchio R: Immunosenescence in aging: Between immune cells
depletion and cytokines up-regulation. Clin Mol Allergy. 15:212017.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Savy M, Edmond K, Fine PE, Hall A, Hennig
BJ, Moore SE, Mulholland K, Schaible U and Prentice AM: Landscape
analysis of interactions between nutrition and vaccine responses in
children. J Nutr. 139:2154S–2218S. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Arvas A: Vaccination in patients with
immunosuppression. Turk Pediatri Ars. 49:181–185. 2014. View Article : Google Scholar
|
|
24
|
Pickering LK, Baker CJ, Kimberlin DW and
Long SS: Red Book: 2012 Report of the Committee on Infectious
Diseases.
|
|
25
|
Keusch GT: Nutritional effects on response
of children in developing countries to respiratory tract pathogens:
Implications for vaccine development. Rev Infect Dis. 13(Suppl 6):
S486–S491. 1991. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Opal SM, Girard TD and Ely EW: The
immunopathogenesis of sepsis in elderly patients. Clin Infect Dis.
41(Suppl 7): S504–S512. 2005. View
Article : Google Scholar : PubMed/NCBI
|
|
27
|
Eliakim A, Schwindt C, Zaldivar F, Casali
P and Cooper DM: Reduced tetanus antibody titers in overweight
children. Autoimmunity. 39:137–141. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Calina D, Roșu L, Roșu AF, Ianoşi G,
Ianoşi S, Zlatian O, Mitruț R, Docea AO, Rogoveanu O, Mitruț P, et
al: Etiological diagnosis and pharmacotherapeutic management of
parap-neumonic pleurisy. Farmacia. 64:946–952. 2016.
|
|
29
|
Skalny AV, Rink L, Ajsuvakova OP, Aschner
M, Gritsenko VA, Alekseenko SI, Svistunov AA, Petrakis D, Spandidos
DA, Aaseth J, et al: Zinc and respiratory tract infections:
Perspectives for COVID-19 (Review). Int J Mol Med. 46:17–26.
2020.
|
|
30
|
Stanberry LR and Strugnell R: Vaccines of
the future. Understanding Modern Vaccines: perspectives in
Vaccinology. Garçon N, Stern PL and Cunningham AL: Elsevier; pp.
151–199. 2011
|
|
31
|
Scharf SF: Orphan drugs: The question of
products liability. Am J Law Med. 10:491–513. 1985.PubMed/NCBI
|
|
32
|
Haffner ME and Kelsey JV: Evaluation of
orphan products by the U.S. Food and Drug Administration. Int J
Technol Assess Health Care. 8:647–657. 1992. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Olliario P: Will the fight against
tropical diseases benefit from orphan drug status? Trop Med Int
Health. 2:113–115. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Mahoney RT and Maynard JE: The
introduction of new vaccines into developing countries. Vaccine.
17:646–652. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Milstien J, Batson A and Meaney W: A
systematic method for evaluating the potential viability of local
vaccine producers. Vaccine. 15:1358–1363. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Franceschi C, Salvioli S, Garagnani P, de
Eguileor M, Monti D and Capri M: Immunobiography and the
heterogeneity of immune responses in the elderly: A focus on
inflamation and trained immunity. Front Immunol. 8:9822017.
View Article : Google Scholar
|
|
37
|
DeStefano F, Bodenstab HM and Offit PA:
Principal controversies in vaccine safety in the United States.
Clin Infect Dis. 69:726–731. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Pronker ES, Weenen TC, Commandeur H,
Claassen EH and Osterhaus AD: Risk in vaccine research and
development quantified. PLoS One. 8:e577552013. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Goetz KB, Pfleiderer M and Schneider CK:
First-in-human clinical trials with vaccines - what regulators
want. Nat Biotechnol. 28:910–916. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Guerra Mendoza Y, Garric E, Leach A,
Lievens M, Ofori-Anyinam O, Pirçon JY, Stegmann JU, Vandoolaeghe P,
Otieno L, Otieno W, et al: Safety profile of the RTS, S/AS01
malaria vaccine in infants and children: Additional data from a
phase III randomized controlled trial in sub-Saharan Africa. Hum
Vaccin Immunother. 15:2386–2398. 2019. View Article : Google Scholar :
|
|
41
|
Peeples L: News feature: Avoiding pitfalls
in the pursuit of a COVID-19 vaccine. Proc Natl Acad Sci USA.
117:8218–8221. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Schlake T, Thess A, Fotin-Mleczek M and
Kallen KJ: Developing mRNA-vaccine technologies. RNA Biol.
9:1319–1330. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Yi Y, Lagniton PNP, Ye S, Li E and Xu RH:
COVID-19: What has been learned and to be learned about the novel
coronavirus disease. Int J Biol Sci. 16:1753–1766. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Wojda TR, Valenza PL, Cornejo K, McGinley
T, Galwankar SC, Kelkar D, Sharpe RP, Papadimos TJ and Stawicki SP:
The Ebola Outbreak of 2014-2015: From Coordinated Multilateral
Action to Effective Disease Containment, Vaccine Development, and
Beyond. J Glob Infect Dis. 7:127–138. 2015. View Article : Google Scholar
|
|
45
|
Grenham A and Villafana T: Vaccine
development and trials in low and lower-middle income countries:
Key issues, advances and future opportunities. Hum Vaccin
Immunother. 13:2192–2199. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Mazur NI, Higgins D, Nunes MC, Melero JA,
Langedijk AC, Horsley N, Buchholz UJ, Openshaw PJ, McLellan JS,
Englund JA, et al: Respiratory Syncytial Virus Network (ReSViNET)
Foundation: The respiratory syncytial virus vaccine landscape:
Lessons from the graveyard and promising candidates. Lancet Infect
Dis. 18:e295–e311. 2018. View Article : Google Scholar
|
|
47
|
Docea AO, Tsatsakis A, Albulescu D,
Cristea O, Zlatian O, Vinceti M, Moschos SA, Tsoukalas D, Goumenou
M, Drakoulis N, et al: A new threat from an old enemy: Re-emergence
of coronavirus (Review). Int J Mol Med. 45:1631–1643.
2020.PubMed/NCBI
|
|
48
|
Goumenou M, Spandidos DA and Tsatsakis A:
[Editorial] Possibility of transmission through dogs being a
contributing factor to the extreme Covid-19 outbreak in North
Italy. Mol Med Rep. 21:2293–2295. 2020.PubMed/NCBI
|
|
49
|
Chen Y, Liu Q and Guo D: Emerging
coronaviruses: Genome structure, replication, and pathogenesis. J
Med Virol. 92:418–423. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Yang P and Wang X: COVID-19: A new
challenge for human beings. Cell Mol Immunol. 17:555–557. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Hoffmann M, Kleine-Weber H, Schroeder S,
Krüger N, Herrler T, Erichsen S, Schiergens TS, Herrler G, Wu NH,
Nitsche A, et al: SARS-CoV-2 cell entry depends on ACE2 and TMPRSS2
and is blocked by a clinically proven protease inhibitor. Cell.
181:271–280.e8. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Wang Q, Zhang Y, Wu L, Niu S, Song C,
Zhang Z, Lu G, Qiao C, Hu Y, Yuen KY, et al: Structural and
functional basis of SARS-CoV-2 entry by using human ACE2. Cell. Apr
7–2020.Epub ahead of print.
|
|
53
|
Li Y, Zhou W, Yang L and You R:
Physiological and pathological regulation of ACE2, the SARS-CoV-2
receptor. Pharmacol Res. 157:1048332020. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Lukassen S, Chua RL, Trefzer T, Kahn NC,
Schneider MA, Muley T, Winter H, Meister M, Veith C, Boots AW, et
al: SARS-CoV-2 receptor ACE2 and TMPRSS2 are primarily expressed in
bronchial transient secretory cells. EMBO J. 4:e1051142020.
|
|
55
|
Leung JM, Yang CX, Tam A, Shaipanich T,
Hackett TL, Singhera GK, Dorscheid DR and Sin DD: ACE-2 expression
in the small airway epithelia of smokers and COPD patients:
Implications for COVID-19. Eur Respir J. Apr 8–2020.Epub ahead of
print. View Article : Google Scholar
|
|
56
|
Mousavizadeh L and Ghasemi S: Genotype and
phenotype of COVID-19: Their roles in pathogenesis. J Microbiol
Immunol. Mar 31–2020.Epub ahead of print.
|
|
57
|
Woo PC, Huang Y, Lau SK and Yuen KY:
Coronavirus genomics and bioinformatics analysis. Viruses.
2:1804–1820. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Liu W, Fontanet A, Zhang PH, Zhan L, Xin
ZT, Baril L, Tang F, Lv H and Cao WC: Two-year prospective study of
the humoral immune response of patients with severe acute
respiratory syndrome. J Infect Dis. 193:792–795. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Ganji A, Farahani I, Khansarinejad B,
Ghazavi A and Mosayebi G: Increased expression of CD8 marker on
T-cells in COVID-19 patients. Blood Cells Mol Dis. 83:1024372020.
View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Farsalinos K, Niaura R, Le Houezec J,
Barbouni A, Tsatsakis A, Kouretas D, Vantarakis A and Poulas K:
Editorial: Nicotine and SARS-CoV-2: COVID-19 may be a disease of
the nicotinic cholinergic system. Toxicol Rep. Apr 30–2020.Epub
ahead of print. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Zhou G and Zhao Q: Perspectives on
therapeutic neutralizing antibodies against the Novel Coronavirus
SARS-CoV-2. Int J Biol Sci. 16:1718–1723. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Kikkert M: Innate immune evasion by human
respiratory RNA viruses. J Innate Immun. 12:4–20. 2020. View Article : Google Scholar :
|
|
63
|
Jackson NAC, Kester KE, Casimiro D,
Gurunathan S and DeRosa F: The promise of mRNA vaccines: A biotech
and industrial perspective. NPJ Vaccines. 5:112020. View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Zhang C, Maruggi G, Shan H and Li J:
Advances in mRNA Vaccines for infectious diseases. Front Immunol.
10:5942019. View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Pardi N, Hogan MJ, Porter FW and Weissman
D: mRNA vaccines - a new era in vaccinology. Nat Rev Drug Discov.
17:261–279. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Maruggi G, Zhang C, Li J, Ulmer JB and Yu
D: mRNA as a transformative technology for vaccine development to
control infectious diseases. Mol Ther. 27:757–772. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Kramps T and Probst J: Messenger RNA-based
vaccines: Progress challenges, applications. Wiley Interdiscip Rev
RNA. 4:737–749. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
68
|
World Health Organization (WHO): DRAFT
landscape of COVID-19 candidate vaccines. WHO; Geneva: 2020,
https://www.who.int/blueprint/priority-diseases/key-action/Novel_Coronavirus_Landscape_nCoV_11April2020.PDF?ua=1.
Accessed April 11 2020.
|
|
69
|
Li L and Petrovsky N: Molecular mechanisms
for enhanced DNA vaccine immunogenicity. Expert Rev Vaccines.
15:313–329. 2016. View Article : Google Scholar :
|
|
70
|
Hobernik D and Bros M: DNA Vaccines-How
Far From Clinical Use? Int J Mol Sci. 19:E36052018. View Article : Google Scholar : PubMed/NCBI
|
|
71
|
Ferraro B, Morrow MP, Hutnick NA, Shin TH,
Lucke CE and Weiner DB: Clinical applications of DNA vaccines:
Current progress. Clin Infect Dis. 53:296–302. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
72
|
Thanh Le T, Andreadakis Z, Kumar A, Gómez
Román R, Tollefsen S, Saville M and Mayhew S: The COVID-19 vaccine
development landscape. Nat Rev Drug Discov. Apr 9–2020.Epub ahead
of print. View Article : Google Scholar : PubMed/NCBI
|
|
73
|
US National Library of Medicine,
ClinicalTrials.gov: A Study of a Candidate COVID-19 Vaccine
(COV001). https://clinicaltrials.gov/ct2/show/NCT04324606.
Accessed March 27 2020.
|
|
74
|
Clinical Trials Arena: University of
Oxford starts enrolment for Covid-19 vaccine trial. https://www.clinicaltrialsarena.com/news/oxford-university-covid-19-vaccine-trial/
Accessed March 30, 2020.
|
|
75
|
Clinical Trials Arena: Inovio commences
Phase I trial of DNA vaccine for Covid-19. https://www.clinicaltrialsarena.com/news/inovio-SARS-COV-2-vaccine-trial/.
Accessed April 7, 2020.
|
|
76
|
Zhang N, Tang J, Lu L, Jiang S and Du L:
Receptor-binding domain-based subunit vaccines against MERS-CoV.
Virus Res. 202:151–159. 2015. View Article : Google Scholar :
|
|
77
|
Lee NH, Lee JA, Park SY, Song CS, Choi IS
and Lee JB: A review of vaccine development and research for
industry animals in Korea. Clin Exp Vaccine Res. 1:18–34. 2012.
View Article : Google Scholar
|
|
78
|
Wang M, Jiang S and Wang Y: Recent
advances in the production of recombinant subunit vaccines in
Pichia pastoris. Bioengineered. 7:155–165. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
79
|
Choi Y and Chang J: Viral vectors for
vaccine applications. Clin Exp Vaccine Res. 2:97–105. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
80
|
Sarkar B, Islam SS, Zohora US and Ullah
MA: Virus like particles - A recent advancement in vaccine
development. Korean J Microbiol. 55:327–343. 2019.
|
|
81
|
Huang X, Wang X, Zhang J, Xia N and Zhao
Q: Escherichia coli-derived virus-like particles in vaccine
development. NPJ Vaccines. 2:32017. View Article : Google Scholar :
|
|
82
|
Sridhar S, Brokstad KA and Cox RJ:
Influenza vaccination strategies: Comparing inactivated and live
attenuated influenza vaccines. Vaccines (Basel). 3:373–389. 2015.
View Article : Google Scholar
|
|
83
|
Lauring AS, Jones JO and Andino R:
Rationalizing the development of live attenuated virus vaccines.
Nat Biotechnol. 28:573–579. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
84
|
US National Library of Medicine,
ClinicalTrials.gov: Immunity and Safety of Covid-19 Synthetic
Minigene Vaccine. https://clinicaltrials.gov/ct2/show/NCT04276896.
Accessed February 19, 2020.
|
|
85
|
US National Library of Medicine,
ClinicalTrials.gov: Safety and Immunity of Covid-19 aAPC Vaccine.
https://clinicaltrials.gov/ct2/show/NCT04299724.
Accessed March 9, 2020.
|
|
86
|
Dang-Tan T, Mahmud SM, Puntoni R and
Franco EL: Polio vaccines, Simian Virus 40, and human cancer: The
epidemiologic evidence for a causal association. Oncogene.
23:6535–6540. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
87
|
Strickler HD, Rosenberg PS, Devesa SS,
Hertel J, Fraumeni JF Jr and Goedert JJ: Contamination of
poliovirus vaccines with simian virus 40 (1955-1963) and subsequent
cancer rates. JAMA. 279:292–295. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
88
|
Baylis SA, Finsterbusch T, Bannert N,
Blümel J and Mankertz A: Analysis of porcine circovirus type 1
detected in Rotarix vaccine. Vaccine. 29:690–697. 2011. View Article : Google Scholar
|
|
89
|
Smatti MK, Al Thani AA and Yassine HM:
Viral-induced enhanced disease illness. Front Microbiol.
9:29912018. View Article : Google Scholar : PubMed/NCBI
|
|
90
|
Tetro JA: Is COVID-19 receiving ADE from
other coronaviruses? Microbes Infect. 22:72–73. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
91
|
Xu J, Jia W, Wang P, Zhang S, Shi X, Wang
X and Zhang L: Antibodies and vaccines against Middle East
respiratory syndrome coronavirus. Emerg Microbes Infect. 8:841–856.
2019. View Article : Google Scholar : PubMed/NCBI
|
|
92
|
Colgrove J: Immunity for the people: The
challenge of achieving high vaccine coverage in American history.
Public Health Rep. 122:248–257. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
93
|
Lord JM: The effect of ageing of the
immune system on vaccination responses. Hum Vaccin Immunother.
9:1364–1367. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
94
|
Boda D, Docea AO, Calina D, Ilie MA,
Caruntu C, Zurac S, Neagu M, Constantin C, Branisteanu DE,
Voiculescu V, et al: Human papilloma virus: Apprehending the link
with carcinogenesis and unveiling new research avenues (Review).
Int J Oncol. 52:637–655. 2018.PubMed/NCBI
|
|
95
|
Wiedermann U, Garner-Spitzer E and Wagner
A: Primary vaccine failure to routine vaccines: Why and what to do?
Hum Vaccin Immunother. 12:239–243. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
96
|
Leitner WW, Ying H and Restifo NP: DNA and
RNA-based vaccines: Principles, progress and prospects. Vaccine.
18:765–777. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
97
|
Fehr AR and Perlman S: Coronaviruses: An
overview of their replication and pathogenesis. Methods Mol Biol.
1282:1–23. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
98
|
Perlman S and Netland J: Coronaviruses
post-SARS: Update on replication and pathogenesis. Nat Rev
Microbiol. 7:439–450. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
99
|
Vandamme TF: Use of rodents as models of
human diseases. J Pharm Bioallied Sci. 6:2–9. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
100
|
Fogel DB: Factors associated with clinical
trials that fail and opportunities for improving the likelihood of
success: A review. Contemp Clin Trials Commun. 11:156–164. 2018.
View Article : Google Scholar : PubMed/NCBI
|