Introduction
Prostate cancer has become the third leading cause
of death in male cancer patients (1). Clinically, most patients with
advanced prostate cancer receive androgen deprivation therapy (ADT)
targeting the androgen receptor (AR) (2). However, most tumors develop
metastatic castration-resistant prostate cancer (CRPC) after ADT
(3). Currently, treatments for
CRPC include second-generation androgen inhibitors,
chemotherapeutic agents and immunotherapy. However, these therapies
only increase the median overall survival of the patient by 4
months (4). Therefore, there is
an urgent need for more effective treatment for CRPC.
Notch is a family of evolutionarily highly conserved
transmembrane receptor proteins that are widely expressed in
numerous species, from invertebrates to mammals (5). Notch signaling consists of four
receptors (Notch 1-4) and five ligands. Growing evidence has found
that the Notch signaling pathway is involved in prostate cancer
cell proliferation, apoptosis, migration and invasion, and affects
angiogenesis and other processes (6). Increased AR plays a key role in
promoting prostate cancer development (7). Additionally, mutations in the AR
gene allow other endogenous hormones or antiandrogens as AR
agonists to promote prostate cancer cell proliferation (8,9).
Therefore, blocking and inhibition of the AR signaling pathway has
been a critical node for the treatment of CRPC. Increasing evidence
suggests that it exists as a mutual negative feedback regulation
mechanism between the AR and Notch pathways (10,11). The vascular system of tumors has
become a new and promising target for anti-tumor therapy. Vascular
endothelial growth factor (VEGF) has been considered as a key
factor in angiogenesis during malignant tumor metastasis (12). Hematogenous metastasis of prostate
cancer is the most common, but overexpression of activated Notch1
can inhibit the proliferation of prostate cancer cells (13). Prostate cancer secretes elevated
VEGF and this promotes endothelial cell proliferation and migration
through autocrine or paracrine pathways, which are closely related
to prostate cancer metastasis (14). In addition, Notch signaling is
important in determining how endothelial cells respond to VEGF
(15).
Activation of the Notch pathway by small activating
RNA (saRNA) can simultaneously regulate AR and VEGF, which could
provide new ideas for CRPC development and gene therapy, and the
present study has important theoretical value and clinical guiding
significance.
Materials and methods
saRNA technology synthesis
Three saRNAs (including saRNA-571, saRNA-947 and
saRNA-1480) were designed to target non-CpG islands and Alu regions
of the promoters of human Notch1 gene (GenBank: NM_017617), and
were chemically synthesized by the Bioneer Corporation. Then the
saRNA were screened for the highest activation rate of Notch1. The
sequence of the three saRNAs is listed in Table I.
 | Table ISequences of three saRNAs and
scramble RNA. |
Table I
Sequences of three saRNAs and
scramble RNA.
| saRNAs | Sequence |
|---|
| saRNA-571 |
5′-GCUGUUUCCAGAGUGCUCATT-3′ |
|
5′-UGAGCACUCUGGAAACAGCTT-3′ |
| saRNA-947 |
5′-GGCAUCGUGGUGGAGAAAUTT-3′ |
|
5′-AUUUCUCCACCACGAUGCCTT-3′ |
| saRNA-1480 |
5′-CCGCUUAUUCACAUGCAAATT-3′ |
|
5′-UUUGCAUGUGAAUAAGCGGTT-3′ |
| Scramble RNA |
5′-UUCUCCGAACGUGUCACGU-3′ |
|
5′-ACGUGACACGUUCGGAGAATT-3′ |
Cell culture and transfection
PC3 cells (prostate cancer cells from the bone
metastasis site cell line) were obtained from the Shanghai Cell
Bank of Chinese Academy of Sciences. PC3 cells were cultured in
DMEM (Gibco; Thermo Fisher Scientific, Inc.) supplemented with 10%
FBS (Gibco; Thermo Fisher Scientific, Inc.) and 100 U/ml
penicillin/streptomycin in a humidified CO2 incubator at
37°C. Cells were seeded into 12-well plates the day before cell
transfection, with a density of 2×105 cell/ml the next
day. Transfections of saRNA and scramble RNA were carried out at a
concentration of 25 nM/well using Lipofectamine 3000 reagent
(Invitrogen; Thermo Fisher Scientific, Inc.) and lasted for 24, 48
and 72 h, respectively.
Quantitative (q)PCR
PC3 cells were collected at 24, 48 and 72 h after
treating with saRNA and untreated cells were used as untreated
reference group. After washing twice with PBS, total RNA was
isolated using TRIzol® reagent (Invitrogen; Thermo
Fisher Scientific, Inc.), and then transcribed into cDNA by
PrimeScript RT Master Mix (Takara Biotechnology Co., Ltd.)
according to the following conditions: 25°C for 10 min, 37°C for 60
min, 70°C for 10 min. PCR amplification included an initial
denaturation step (95°C for 10 min), 38 cycles of denaturation
(95°C for 10 sec), and annealing (60°C for 1 min). The experimental
results were automatically calculated by reverse transcription
(RT)-qPCR analysis software CFX Manager 3.1 (Bio-Rad Laboratories,
Inc.). The primer sequences of Notch1, AR and
glyceraldehyde-3-phosphate dehydrogenase (GAPDH) are shown in
Table II. All experiments were
repeated 3 times independently. According to above methods, the
mRNA expression level of KISS1, MKK4 and KAI1 was calculated by
RT-qPCR. Table II lists the
primer sequences. The relative expression of each gene was
converted into 2-∆∆Cq (16).
 | Table IIPrimer sequences for quantitative
PCR. |
Table II
Primer sequences for quantitative
PCR.
| Target gene | Primer
sequence | Product size in
bp |
|---|
| Homo Notch1 |
5′-GAGGCGTGGCAGACTATGC-3′ | 140 |
|
5′-CTTGTACTCCGTCAGCGTGA-3′ | |
| Homo AR |
5′-CCAGGGACCATGTTTTGCC-3′ | 226 |
|
5′-CGAAGACGACAAGATGGACAA-3′ | |
| Homo KISS1 |
5′-CTCACTGGTTTCTTGGCAGC-3′ | 100 |
|
5′-GCCTGTGGGTCTAGAATTCCC-3′ | |
| Homo MKK4 |
5′-TGCAGGGTAAACGCAAAGCA-3′ | 93 |
|
5′-CTCCTGTAGGATTGGGATTCAGA-3′ | |
| Homo MTA1 |
5′-ACGCAACCCTGTCAGTCTG-3′ | 104 |
|
5′-GGGCAGGTCCACCATTTCC-3′ | |
| Homo KAI1 |
5′-TGTCCTGCAAACCTCCTCCA-3′ | 80 |
|
5′-CCATGAGCATAGTGACTGCCC-3′ | |
| GAPDH |
5′-CTCGCTTCGGCAGCACA-3′ | 90 |
|
5′-AACGCTTCACGAATTTGCGT-3′ | |
Western blotting analysis
Cells were collected at 24, 48 and 72 h after
treatment. Cells were rinsed twice with ice-cold PBS and were lysed
using 200 μl of RIPA lysis buffer (Beyotime Institute of
Biotechnology) per 1×106 cells. Lysates were allowed to
stand on ice for 5 min, transferred to a 1.5-ml centrifuge tube and
lysed for 30 min on ice. Cell lysates were clarified by
centrifugation at 12,000 × g for 15 min at 4°C and protein
concentrations were determined using the bicinchoninic acid protein
assay reagent (Pierce, Thermo Fisher Scientific, Inc.). Cell
lysates were denatured in a 100°C water for 5 min and then
collected by centrifugation at 13,000 × g at 4°C for 30 min. Cell
lysates (35 μg) were subjected to 12% SDS-PAGE gels and
electrophoretically transferred to Invitrolon™ polyvinylidene
difluoride membranes (Thermo Fisher Scientific, Inc.). Membranes
were blocked with 5% BSA (Beyotime Institute of Biotechnology) and
then incubated overnight with the appropriate primary antibodies
including anti-VEGF (1:1,000; cat. no. SAB1402390; Sigma-Aldrich;
Merck KGaA), anti-Notch1 (1:1,000; cat. no. 3608; Cell Signaling
Technology, Inc.), anti-VEGFR2 (1:1,000; cat. no. ab134191; Abcam),
anti-AR (1:1,000; cat. no. 5153S; Cell Signaling Technology, Inc.)
and anti-GAPDH (1:1,000; cat. no. 2118L; Cell Signaling Technology,
Inc.) followed by matching horseradish peroxidase-conjugated
secondary antibodies (1:2,000; cat. nos. ab205719 or ab7090;
Abcam). After washing each sample three times for 10 min with
TBS-0.05% Tween 20, Immobilon Western Chemiluminescent HRP
Substrate (EMD Millipore) and a ChemiDoc™ Touch Imaging System
(Bio-Rad Laboratories, Inc.) were used to visualize bands; the
intensity of bands was quantified with Image Lab™ Software (version
4.0; Bio-Rad Laboratories, Inc.).
Nuclear run-on RT-qPCR
Notch1 transcriptional activity was measured by
nuclear run-on experiments, as previously described (17). PC3 cells were used to determine
transcriptional activity after transfection with
Notch1-saRNA-1480.
Cell cycle analysis
An AnnexinV detection kit (BD Biosciences) was used
to analyze cell cycle. PC3 cells were seeded in 6-well plates and
the density was 50% the next day. The serum-free medium was
replaced and synchronized for 24 h, and then the normal medium was
restored for subsequent experiments. The cells were transfected
with 25 nM saRNA using Lipofectamine 3000. Scramble RNAs were used
as reference. A total of 48 h after treatment, cells were
harvested, rinsed with PBS twice and fixed in 4% paraformaldehyde
phosphate buffer for 10 min at room temperature. Cold ethanol (750
μl) was added to resuspend the cells and fix at 4°C
overnight. After centrifugation at 300 × g for 5 min at 4°C, the
cells were resuspended in 1 ml PBS and allowed to stand at room
temperature for 30 min to recover. A total of 500 μl
propidium iodide (PI) staining solution was added into each tube,
incubated for 30 min at room temperature in the darkness. The
distribution of cell cycle was shown as the percentage of cells in
G0/G1, G2/M and S populations using a CytoFLEX flow cytometer
(Beckman Coulter, Inc.) and FlowJo software (version 11.0; FlowJo
LLC).
Apoptosis analysis
The culture supernatant and cells were collected,
washed twice with pre-cooled PBS and the supernatant was discarded.
After resuspending in 500 μl apoptosis positive control
solution, the cells were incubated on ice for 30 min. An
appropriate amount of pre-cooled 1X Binding Buffer was added,
followed by the same amount of untreated live cells. After adding
1.5 ml pre-cooled 1X Binding Buffer, the cells were divided into 3
tubes, one of which was a blank control tube and the two tubes were
single-dyed tubes. Then, 5 μl Annexin V-FITC or 10 μl
PI was added into the single-stained tube and the cells were
incubated at room temperature for 5 min in the dark. Finally, the
results were measured via flow cytometry.
Cell proliferation
The cells were seeded in 6-well plates, which was
transfected with saRNA the next day. Then the cells were seeded at
a density of 1×103/well in 96-well plates on the third
day in triplicate. Cell Counting Kit-8 values were determined at 0,
24, 48, 72 and 96 h according to the manufacturer's protocol. The
growth curves were plotted according to the results.
Colony formation assay
The cells were seeded in 6-well plates and
transfected with the corresponding saRNAs the next day. On the
third day, the cells were seeded at a density of 1×103
cell into 6-well plates and the medium was changed every 3 days.
After 2 weeks of culture, the cells were fixed with
paraformaldehyde 4°C for 20 min. After 20 min, crystal violet
stained for 15 min at room temperature and the count was observed
under a light microscope.
Cell migration and invasion assay
Transwell chambers (BD Biosciences) were inserted
into 6-well plates. In the invasion experiment, Matrigel was
diluted 40 times with PBS on ice and added to the upper chamber.
Then, cells transfected with saRNA for 48 h were seeded in 6-well
plates supplemented with serum-free medium. The migration assay was
performed in the same manner except without the Matrigel. After
incubating at 37°C for 1 h, the supernatant was aspirated. The
cells were centrifuged at 300 × g for for 5 min at 4°C, resuspended
and counted in serum-free medium. Then 2×102 inoculated
into the upper chamber of the Transwell chamber, and 600 μl
of complete medium in the lower chamber. After incubation for 24 h,
the paraformaldehyde was fixed for 20 min at 4°C and crystal violet
staining was performed for 15 min at room temperature. After 15
min, an image was captured under a light microscope.
Tube formation
Normally cultured cells were centrifuged at 300 × g
for 5 min at 4°C and inoculated into 6-well plates
(1×103 cells/well). The next day, different saRNAs were
transfected as required. After 48 h, the cells were digested,
centrifuged at 300 × g for 5 min at 4°C, adjusted to a cell density
of 5×104 cells/ml and plated into a 96-well plate in
which Matrigel matrix was pre-coated, and 0.1 ml of cell suspension
was added to each well, to give a final density of 5×103
cells/well. After 5-6 h of serum-free culture, the cells of each
group were observed under a light microscope; the total length of
blood vessels was calculated by Image J software (version 1.8.0,
National Institute of Health), and the angiogenic ability of each
group was analyzed.
In vivo antitumor activity of
Notch1-saRNA-1480
The PC3 cell line in the logarithmic growth phase
was prepared as a single cell suspension by adding serum-free
medium, and counted in a flow cytometer (the number of viable cells
was >95%). The cell density was adjusted to 7×107/ml
and 0.1 ml was inoculated into the left armpit of 10 male Balb/c
mice (weight, 20±2 g; Shanghai Slack Laboratory Animals Co., Ltd.)
at 5-6 weeks of age. All mice were reared under the following
conditions: Temperature, 21±2°C; relative humidity, 30-70%; light
and dark cycle, 12/12 h. All rats received food and water ad
libitum. The tumor formation of the mice was closely observed
every day after inoculation. From the fourth day of inoculation,
the long diameter (a) and short diameter (b) of the tumor were
measured with a vernier caliper every other day. The tumor volume
was calculated according to the formula V=axb2×0.5 and
the tumor growth curve was drawn. The saRNA powder (5OD) was
dissolved with 20 μl of ddH2O. A total of 14.4
μl of RNAiMax was added to 14.4 μl of Opti-MEM
(Gibco; Thermo Fisher Scientific, Inc.) and mixed well. After
adding 16 μl of the diluted saRNA solution, the cells were
allowed to stand at room temperature for 5-20 min. A total of four
days after tumor cell inoculation, intratumoral injection was
performed for 9 μl/3 days. After 5 weeks, all mice were
euthanized with sodium pentobarbital (150 mg/kg). Then, tumors were
removed and weighed. The current study complied with the ARRIVE
guidelines and the AVMA euthanasia guidelines 2013. To observe the
internal structure of the tumor, hematoxylin staining for 20 min at
room temperature and eosin staining for 1 min at room temperature
was performed. At the same time, the protein expression levels of
Notch1, AR, VEGFR2 and VEGF were also detected in tumor tissues as
described in the Western blotting analysis section. The present
study was approved by the Ethics Committee of Affiliated Hangzhou
First People's Hospital, Zhejiang University School of Medicine
(permit no. 2019421).
Statistical analysis
All statistical analyses were performed using
GraphPad Prism software 8.0 (GraphPad Software, Inc.). All values
were expressed as means ± SD. Comparisons between two groups were
analyzed using unpaired Student's t-test. Furthermore, multiple
comparisons were performed using one-way analysis of variance with
Tukey's post hoc test. P<0.05 was considered to indicate a
statistically significant difference.
Results
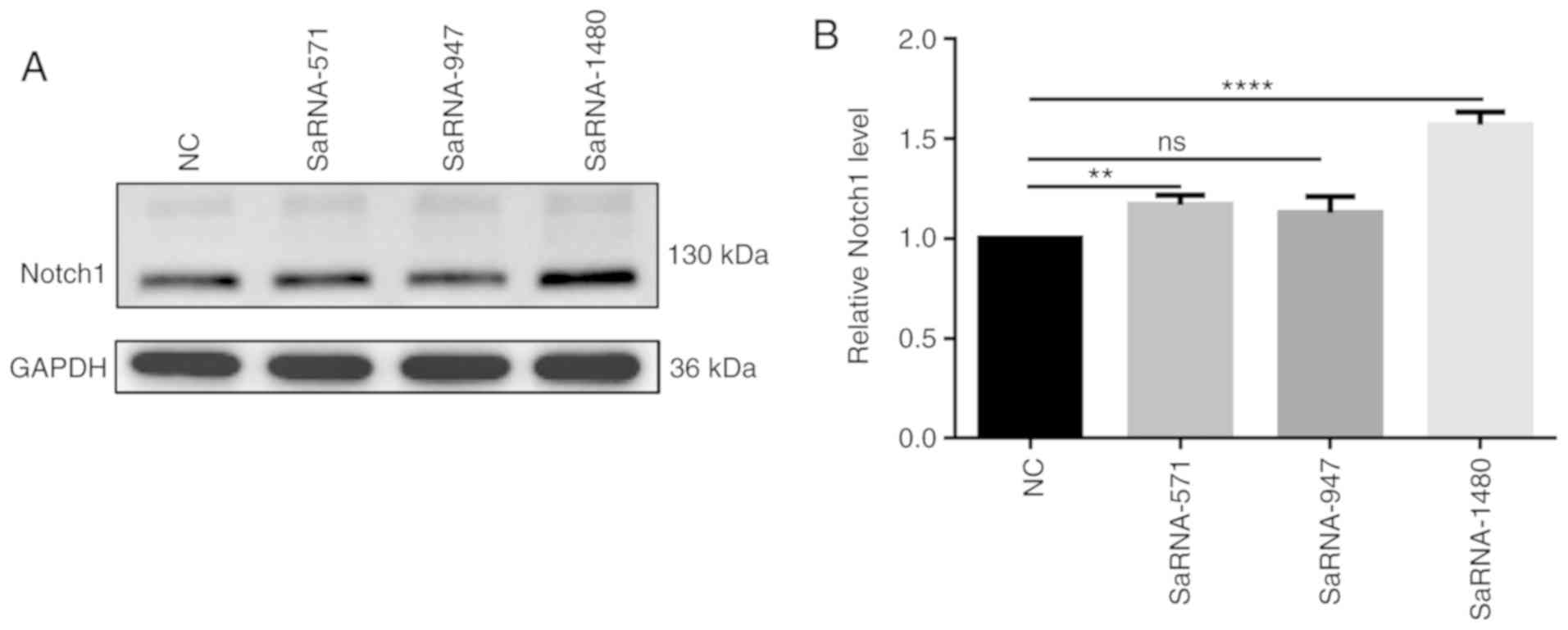
SaRNA-1480 has the highest functional
effect of activating Notch1
After transfection of saRNA-571, saRNA-947 and
saRNA-1480 into PC3 cells, the results of western blotting assay
showed the highest expression level of Notch1 in PC3 cells
transfected with saRNA-1480 (Fig. 1A
and B). Therefore, saRNA-1480 had the highest functional effect
of activating Notch1.
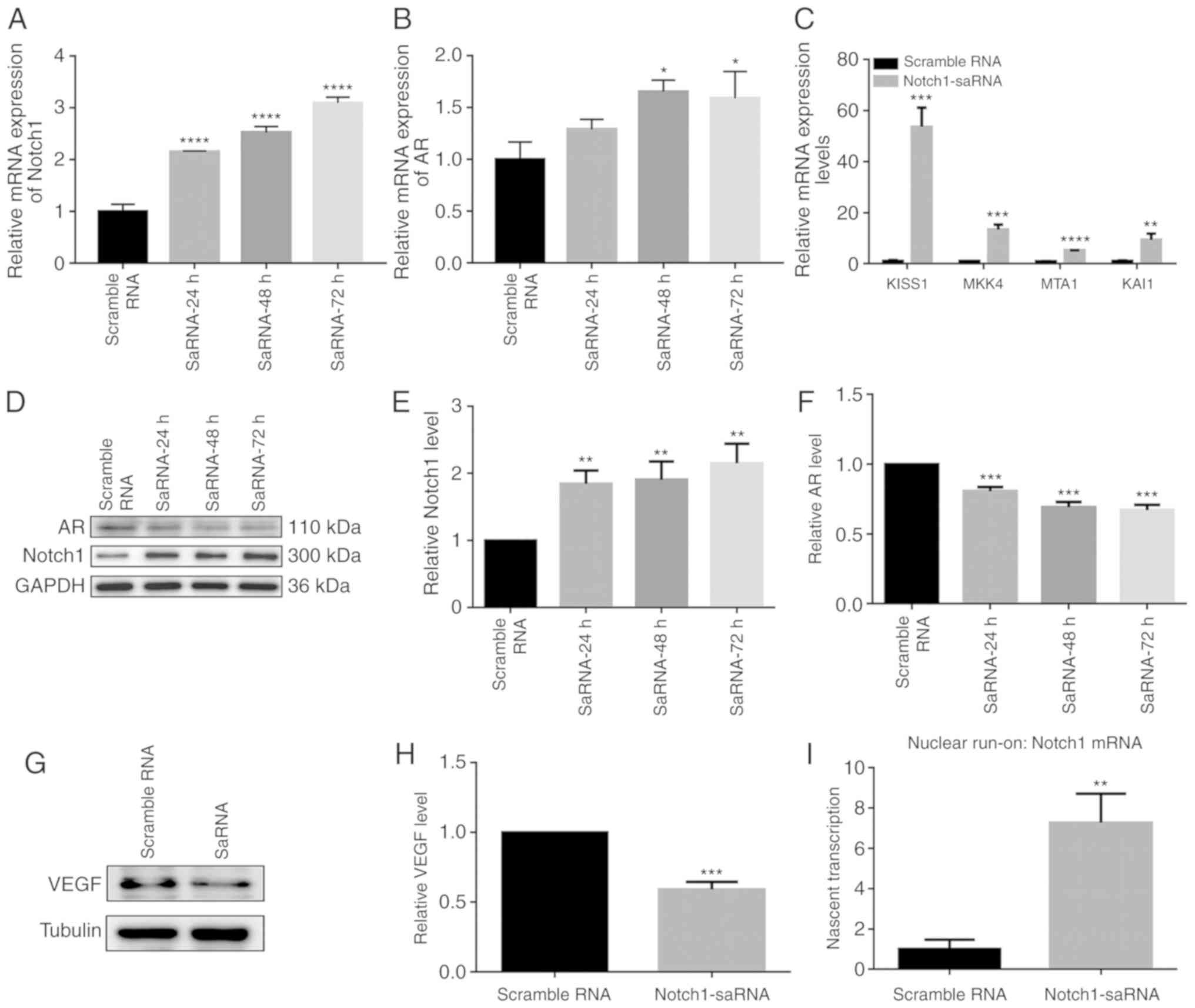
Expression levels of Notch1, AR, VEGF and
metastasis suppressor genes in the PC3 cells after transfection of
saRNA
Gene and protein expression after transfection of
saRNA in the PC3 cell line is shown in Fig. 2. RT-qPCR results demonstrated that
after saRNA transfection, the mRNA expression of Notch1 was
upregulated. Notch1 expression increased with increasing duration
(Fig. 2A). Previous studies found
that AR was expressed in PC3 cells (18,19). Furthermore, the mRNA expression of
AR was significantly upregulated by Notch1-saRNA (Fig. 2B). Moreover, compared with the
control group, RT-qPCR results showed that metastasis suppressor
genes including KISS1, MKK4, MTA1 and KAI1 were upregulated in
Fig. 2C. At the protein level,
Notch1-saRNA significantly increased the expression levels of AR
and Notch1 in Fig. 2D-F. Western
blotting analysis depicted a decrease in VEGF expression levels in
Notch1-saRNA compared with the scramble RNA (Fig. 2G and H).
Notch1-saRNA-1480 could activate Notch1
transcription
To determine whether Notch1-saRNA-1480 could
activate Notch1 transcription, nascent Notch1 mRNA transcription
was measured by a nuclear run-on experiment. After transfection of
Notch1-saRNA-1480 in PC3 cells, nascent Notch1 mRNA level was
significantly higher compared with the scramble RNA (Fig. 2I), indicating activation of
transcription.
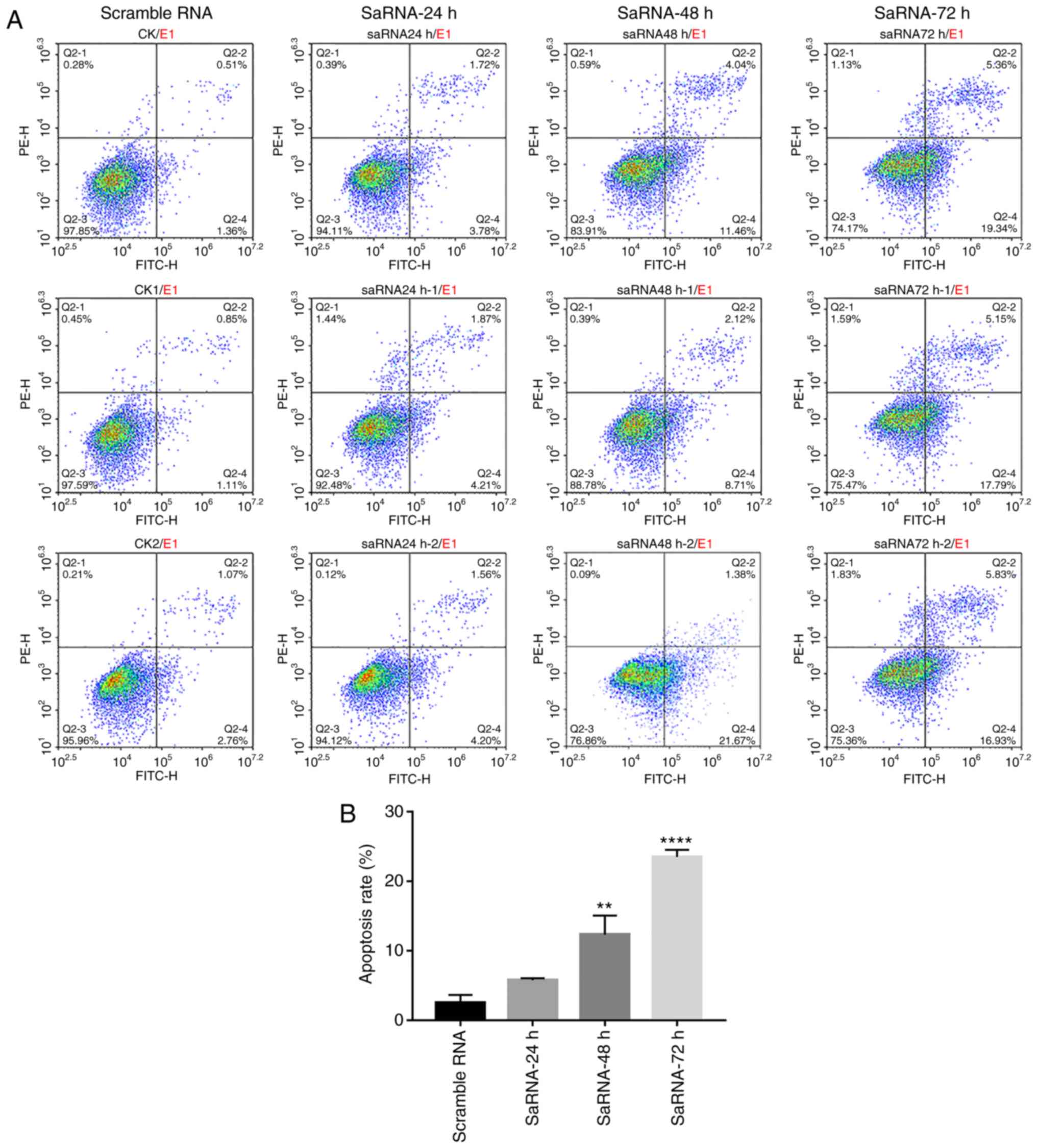
Cell apoptosis and cell cycle
distribution in the PC3 cells after Notch1-saRNA-1480
Apoptosis of PC3 cells after transfection of saRNA
was analyzed by flow cytometry (Fig.
3A and B). PC3 cells were labeled with PI and Annexin V, and
the results showed that Notch1-saRNA-1480 significantly increased
the proportion of apoptotic cells after transfection. As the
transfection time increased, the proportion of apoptotic cells
increased. A total of 24 h after transfection with
Notch1-saRNA-1480, the apoptotic rate was 5.78% compared with the
control group. The apoptosis rate was 12.35% compared with the
control group 48 h after transfection, which was 2.14 times that of
the 24-h apoptosis rate. A total of 72 h after transfection, the
apoptotic rate was 23.48% compared with the control group, which
was 4.06 times the 24-h apoptotic rate.
Notch1-saRNA-1480 could affect the cell cycle
distribution of PC3 cells. As shown in Fig. 3C and D, the percentage of G0/G1
phase cells in the Notch1-saRNA-1480 treated group increased, and
the proportion of S phase cells decreased with the increase of
transfection time. The proportion of cells in the G0/G1 phase
increased from 39.77% in 24 h to 41.99%, increased to 54.51% in 48
h and increased to 59.45% in 72 h. Correspondingly, the percentage
of cells in S phase decreased from 39.36 to 32.4% in 24 h,
decreased to 24.93% in 48 h, decreased to 18.14% in 72 h and only
0.47 times in 0 h S phase. This indicated that the cell cycle was
arrested at the G0/G1 checkpoint. These results were consistent
with previous studies (13,20,21).
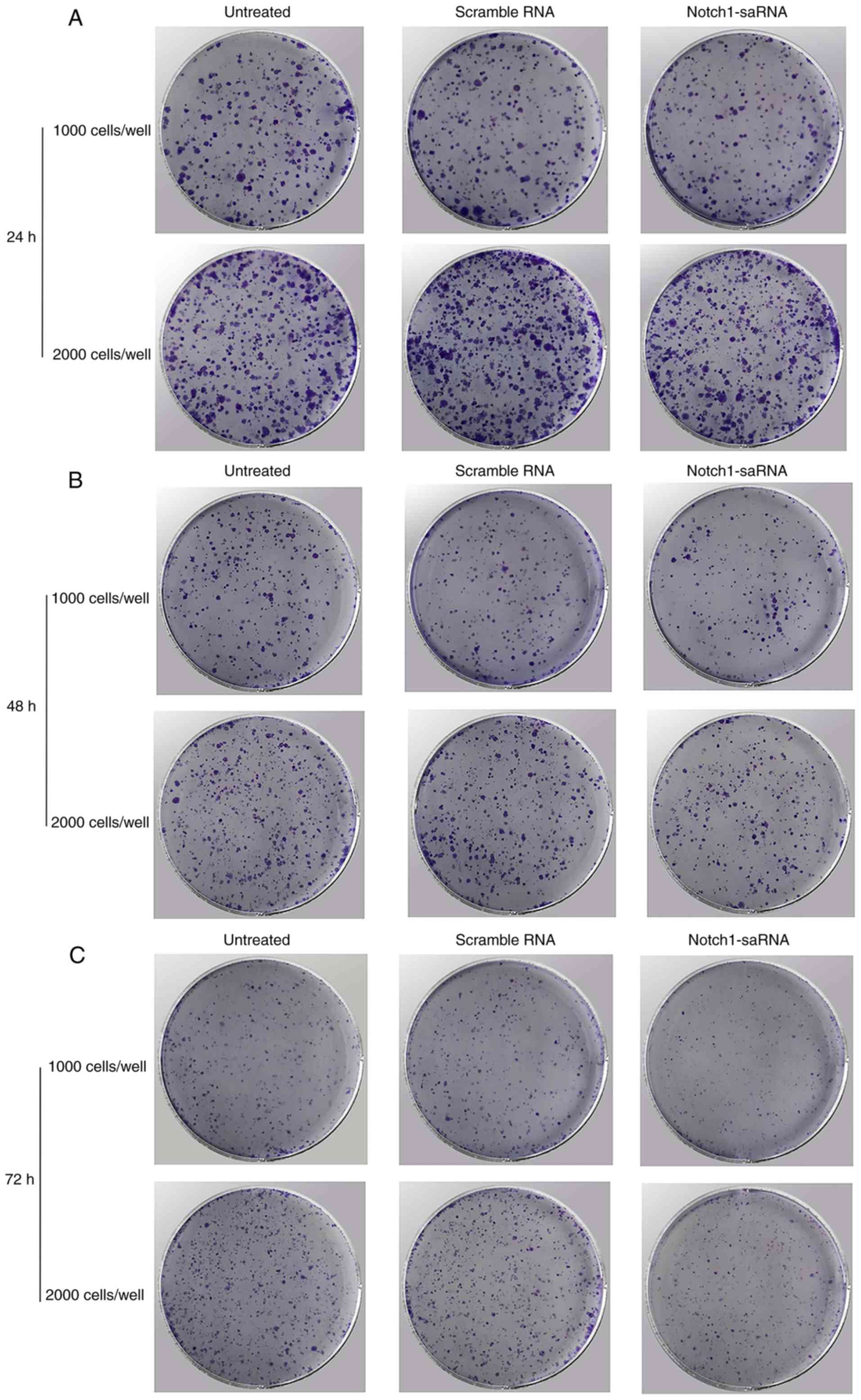
Notch1-saRNA-1480 could inhibit PC3 cell
proliferation and colony formation
Then whether Notch1-saRNA-1480 could affect cell
proliferation and colony formation was investigated. The results
depicted that Notch1-saRNA-1480 could inhibit cell proliferation
and colony formation of PC3 cells (Fig. 4A-C). As shown in the Fig. 4D, the growth rate of cells
transfected with Notch1-saRNA-1480 was significantly inhibited
compared with the control group. The transfected Notch1-saRNA-1480
group showed lower colony formation compared with the control
group.
Notch1-saRNA-1480 could inhibit PC3 cell
migration, invasion and epithelial-mesenchymal transition
(EMT)
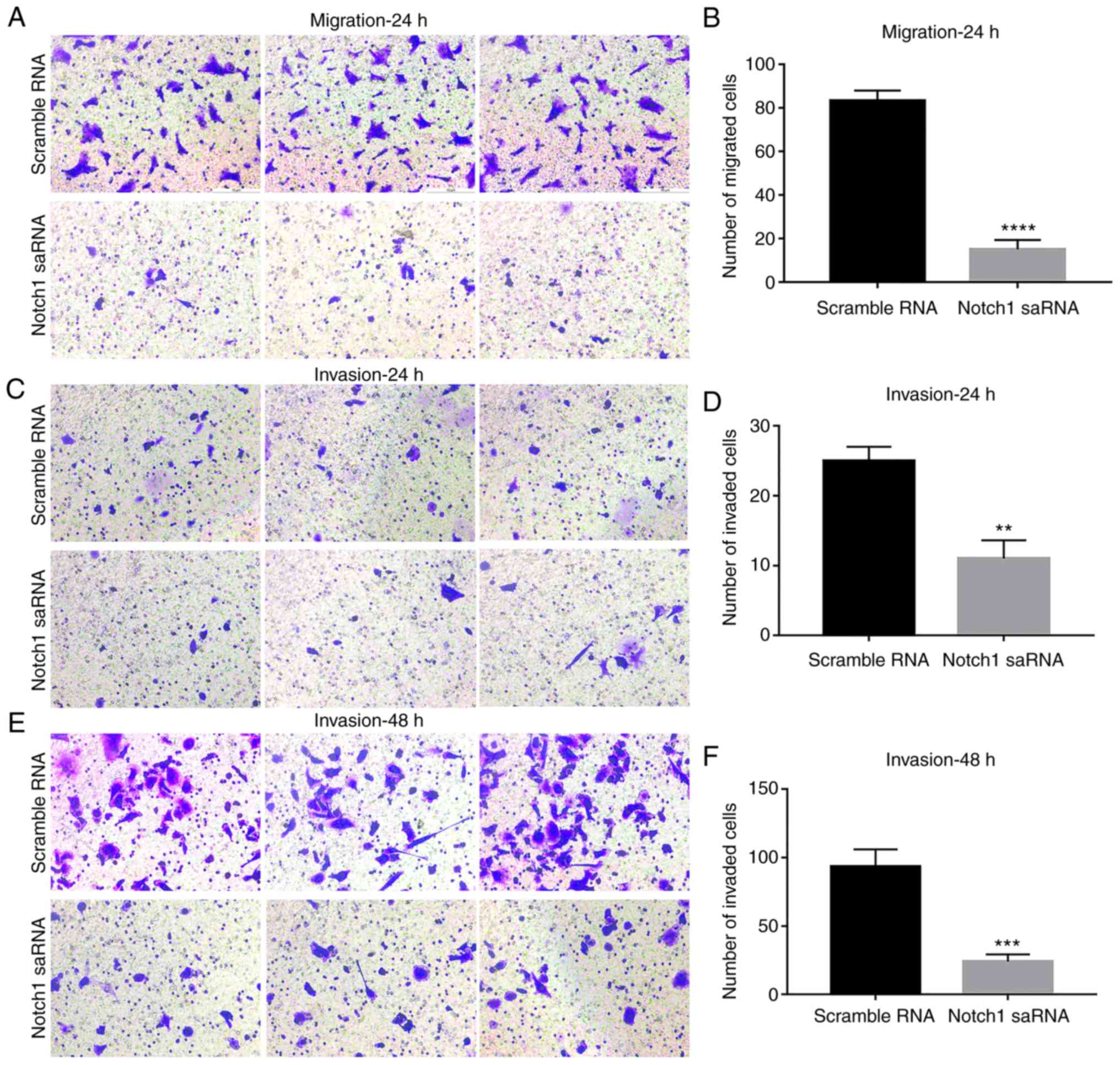
To assess the cell migration and invasion ability of
PC3 cells after Notch1-saRNA-1480, transwell analysis was
performed. After the crystal violet staining, the transwell chamber
was imaged and the number of migrated or invaded cells was
calculated for significant analysis. The results showed that the
ability of PC3 cell migration after Notch1-saRNA-1480 was
significantly decreased compared with the control group (Fig. 5A and B). With increasing time, the
number of PC3 cell invasion was significantly higher than of that
PC3 cells after Notch1-saRNA-1480 (Fig. 5C-F).
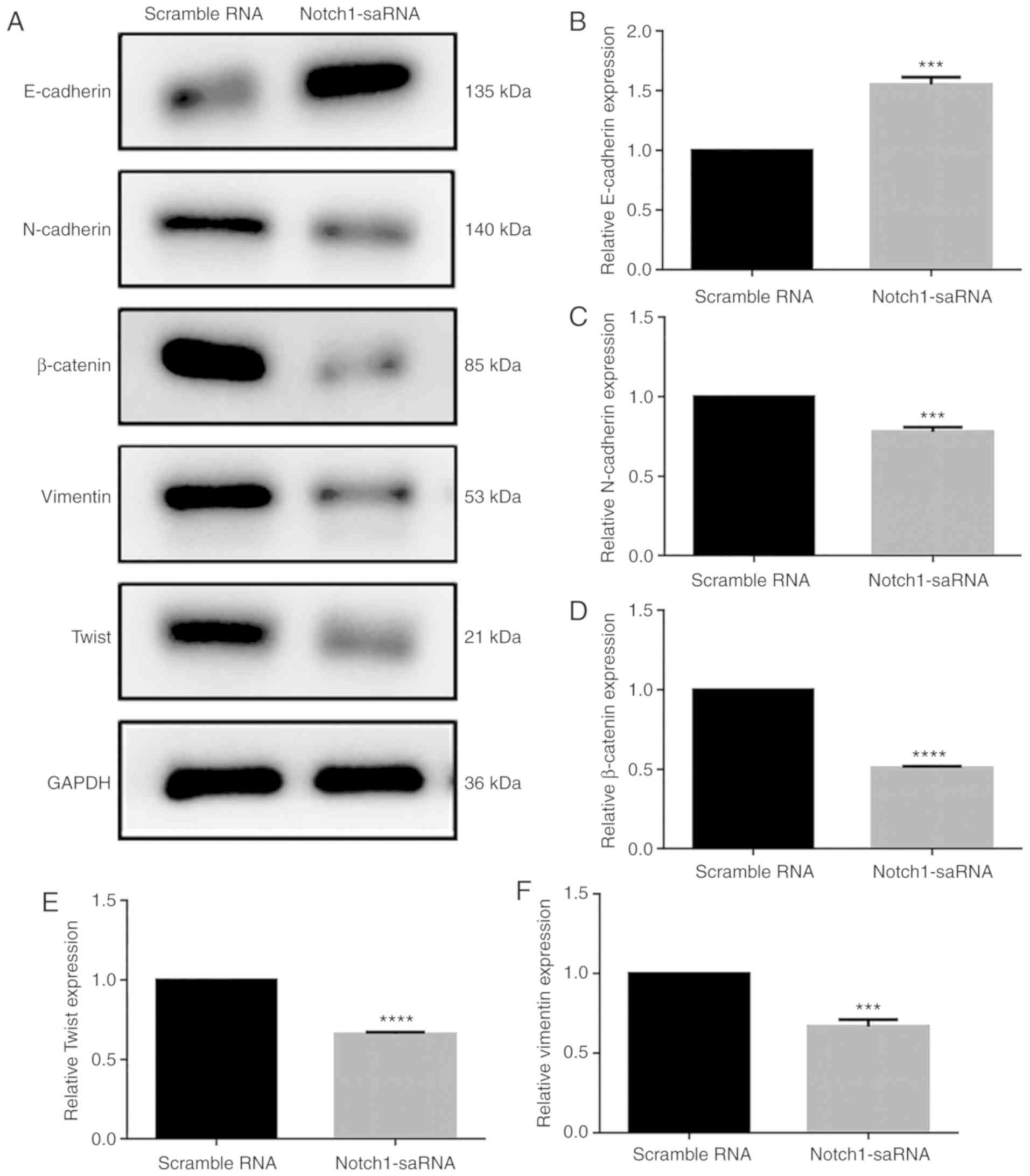
Next, whether Notch1-saRNA-1480 could affect the
expression levels of EMT related proteins was observed (Fig. 6A). The results showed that
Notch1-saRNA-1480 significantly increased E-cadherin expression
(Fig. 6B) and inhibited the
expression levels of N-cadherin (Fig.
6C), β-catenin (Fig. 6D),
Twist (Fig. 6E) and vimentin
(Fig. 6F). Above results
indicated that Notch1-saRNA-1480 could inhibit the EMT process of
PC3 cells.
Notch1-saRNA-1480 could inhibit PC3 cell
angiogenesis
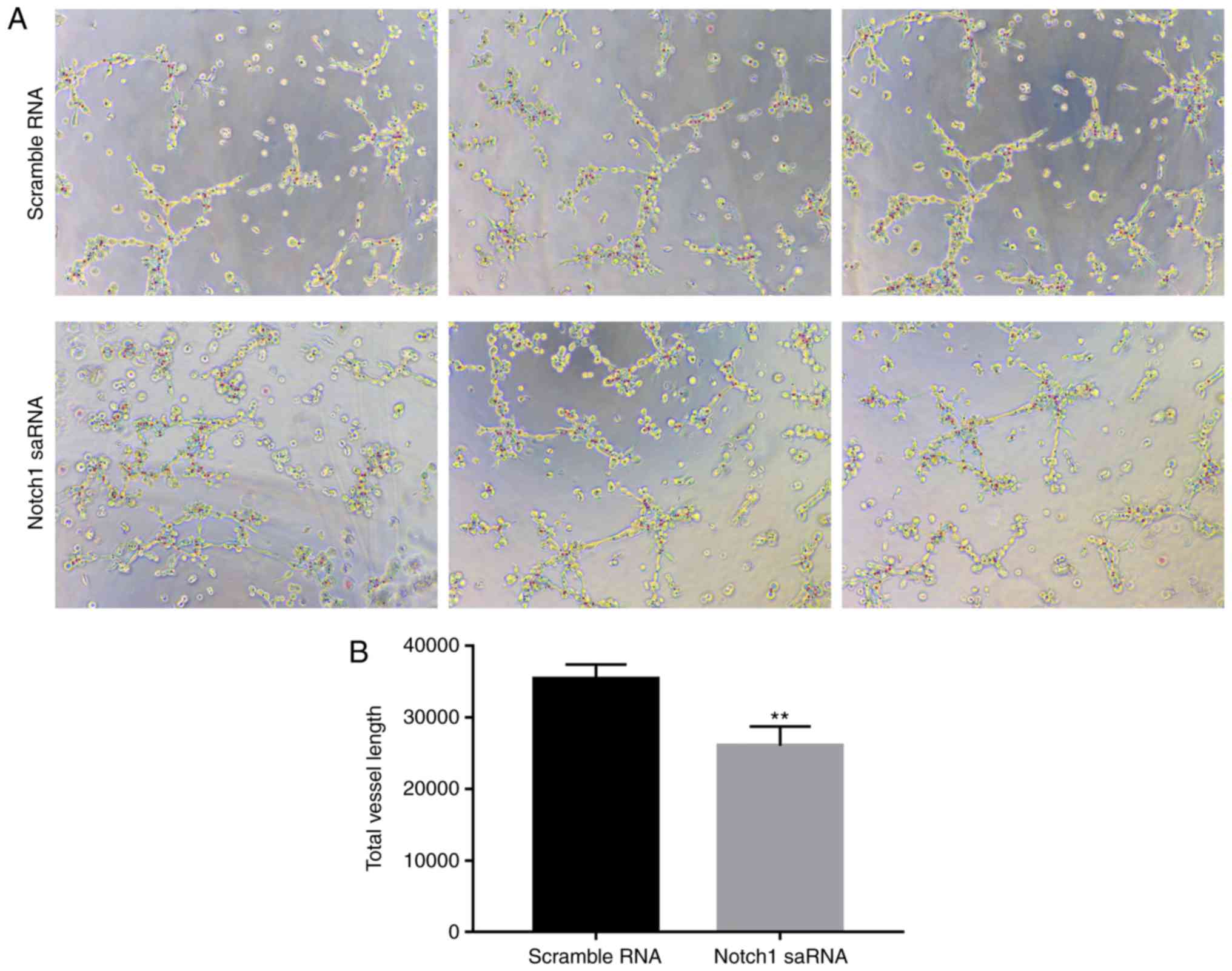
Then a PC3 cell angiogenesis test was performed.
Compared with the control group, total vessel length in PC3 cell
after Notch1-saRNA-1480 treatment was decreased. The results
indicated that Notch1-saRNA-1480 could inhibit PC3 cell
angiogenesis in Fig. 7.
In vivo anti-tumor activity of
Notch1-saRNA-1480
A total of 10 nude mice were randomly divided into
two groups, including the negative control group and the
Notch1-saRNA-1480 group. The administration of the tumor cells was
started 4 days after the inoculation and performed by intratumoral
injection. It was administered once every 3 days for a total of 5
weeks. The tumor diameter was measured every other day. The nude
mice were sacrificed on the 35th day and the tumor tissue was taken
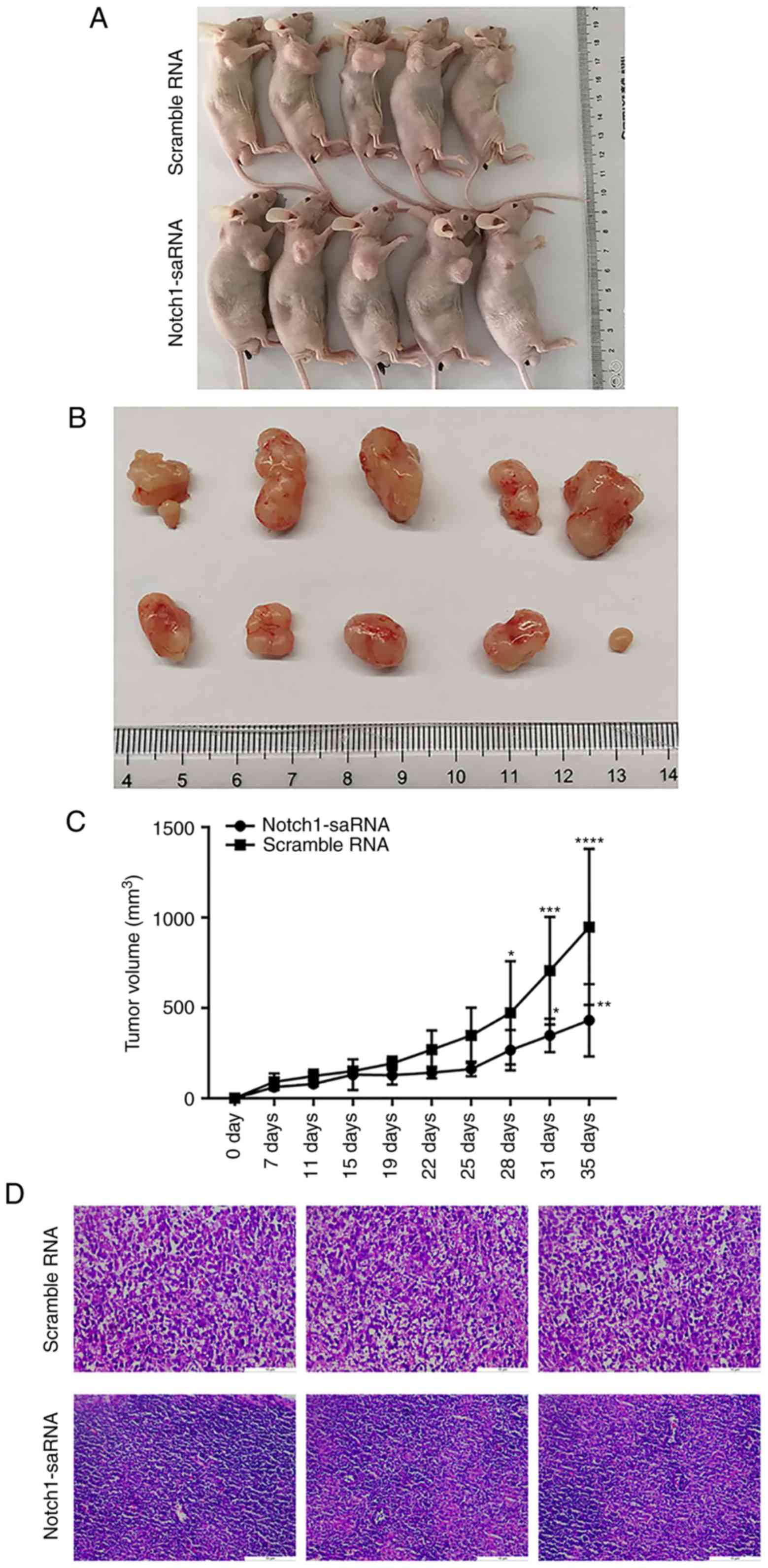
out for subsequent experiments. Photographs of mice sacrificed and
corresponding tumor mass images are shown in Fig. 8A, and the volume of the tumor is
recorded in Fig. 8B. Tumor volume
statistics of the Notch1-saRNA-1480 treated group and the control
group showed that the tumor diameter of the Notch1-saRNA-1480
treated group was smaller than that of the control group, and there
was a significant difference. Additionally, Fig. 8C exhibits the tumor growth curve.
Tumor growth in the Notch1-saRNA-1480 treated group was inhibited
compared with the control group.
The results of hematoxylin and eosin staining showed
that the tumor cells in the control group were evenly distributed
as a whole, arranged in a different order and the nuclei were
deeply stained. In the Notch1-saRNA group, there were few tumors
and a large number of macrophages appeared (Fig. 8D).
The expression of Notch1, AR and VEGFR2 protein in
tumor tissues was detected by western blotting (Fig. 8E). The results showed that the
expression of Notch1 was increased in the saRNA group (Fig. 8F). Furthermore, the expression of
VEGFR2 and AR proteins in the Notch1-saRNA-1480 treated group was
significantly downregulated (Fig. 8G
and H). The study also found that Notch1-saRNA significantly
inhibited the expression of VEGF (Fig. 8I).
Discussion
Currently, the treatment of prostate cancer includes
radiation therapy, surgery and chemotherapy. If treated with
conventional chemotherapy, it leads to the resistance and
development of androgen-independent prostate cancer, which further
complicates the situation. It has been reported that ADT treatment
significantly increases the expression of AR in prostate cancer
cells (9), and AR reactivation is
also observed in recurrent diseases. VEGF and Notch1 have been
shown to play important roles in epithelial-mesenchymal transition
(EMT) (22). Activation of Notch1
is known to be involved in the development and progression of human
malignancies. Emerging evidence suggests that the acquisition of
the EMT phenotype is associated with induction of cancer stem cells
or cancer stem cell-like phenotypes and contributes to tumor
recurrence and drug resistance (23). E-cadherin is one of the important
indicators of EMT. Moreover, E-cadherin-saRNA can induce migration
and invasion of PC3 cells, which is related to the relocalization
of β-catenin from the nucleus to the plasma membrane and
β-catenin-mediated transactivation (24). In this experiment, it was found
that Notch1-saRNA also had a similar effect, which can inhibit the
migration, invasion and EMT process of PC3 cells. The saRNA could
activate the related genes in cancer cells during the cell cycle
and apoptosis. For example, p21 saRNA could cause the changes in
the proliferation of T24 cells in a time- and dose-dependent
manner. Furthermore, the same p21 saRNA could induce cell cycle
arrest and cell apoptosis in the G1 phase (25). In this experiment,
Notch1-saRNA-1480 also induced cell cycle changes and promoted
apoptosis.
Notch signaling plays a key role in angiogenesis
(26). The activin receptor-like
kinase 1 signaling pathway acts in concert with the Notch signaling
pathway to inhibit angiogenesis. The Notch signaling pathway
together induces the target genes HEY1 and HEY2, thereby inhibiting
the VEGF signaling pathway, cell formation and endothelial cell
formation (27). One study found
that emodin inhibits VEGF expression in PC3 cells via the Notch
signaling pathway, thereby reducing angiogenesis (20,28). In the present study,
Notch1-saRNA-1480 could induce Notch1 protein expression, which in
turn affected cell apoptosis. To investigate the relationship
between Notch1 and VEGF, the expression of the VEGF protein was
examined. Under the stimulation of Notch1-saRNA-1480, the
expression of VEGF decreased and then the angiogenesis experimental
results showed that Notch1-saRNA-1480 could inhibit the ability of
PC3 cells to form tubes, thereby destroying the formation of blood
vessels. The treatment of prostate cancer is linked to vascular
targeted therapy and the simultaneous regulation of AR and VEGF is
achieved by activation of Notch1 (29,30).
The protein expression of Notch1 was increased 15
days after transfection of Notch1-saRNA-1480 in PC3 cells, which
contrasted with the short silencing effect of siRNA. Therefore, the
current study concluded that saRNA may act on the promoter region
of the target gene. Demethylation of histones is induced, altering
chromatin structure and leading to long-term increases in gene
expression (31-33). Then in vivo experiments
were conducted. Nude mice were injected with Notch1-saRNA-1480
after subcutaneous tumor formation. The experimental results showed
that the tumor diameter was reduced. Western blot analysis showed
that the expression levels of VEGFR2 and VEGF were both decreased
in the tumor tissues. Finally, the anticancer effect of
Notch1-saRNA-1480 was successfully validated on xenograft animal
models. Based on this, the next step is to focus on clinically
relevant studies to investigate the effects of Notch-saRNA-1480 on
prostate cancer. The present study believes this is an important
step in the future possible application in humans.
Notch1-saRNA-1480 could significantly inhibit the
proliferation of PC3 cells in vitro and the growth of tumor
in vivo, which is related to the inhibition of AR and VEGF
pathways. However, the current study lacks analysis on the
expression of Notch1-saRNA-1480 and clinicopathological features in
patients with prostate cancer. Thus, the next step is to perform
clinically relevant studies to investigate the effects of
Notch-saRNA-1480 on prostate cancer.
Funding
The current study was funded by the Zhejiang
Province Natural Science Foundation (grant nos. LY17H050002 and
Y2111329); Zhejiang Province Science and Technology Plan Projects
(grant no. 2014C37016); Zhejiang Province Chinese Medicine Science
and Technology Plan Projects (grant nos. 2016ZB099, 2013ZA107,
2011ZB099 and 2020ZA087); Zhejiang Province Medicine and Health
Science and Technology Plan Projects (grant nos. 2011KYB066 and
2015KYB295); Hangzhou Science and Technology Plan Projects (grant
nos. 2017A05, 2018A09, 20110833B05 and 20110733Q12).
Availability of data and materials
The datasets analyzed during the current study are
available from the corresponding author on reasonable request.
Authors' contributions
GD, LM conceived and designed the study. KJ, PJ
conducted most of the experiments and data analysis, and wrote the
manuscript. HH, KC and JS participated in data acquisition and
helped to draft the manuscript. All authors reviewed and approved
the manuscript.
Ethics approval and consent to
participate
The study was approved by the Ethics Committee of
Affiliated Hangzhou First People's Hospital, Zhejiang University
School of Medicine (permit no. 2019421).
Patient consent for publication
Not applicable.
Competing interests
The authors declare no conflicts of interest.
Acknowledgments
Not applicable.
Abbreviations:
|
CRPC
|
castration-resistant prostate
cancer
|
|
AR
|
androgen receptor
|
|
RT-qPCR
|
reverse transcription-quantitative
PCR
|
|
VEGF
|
vascular endothelial growth factor
|
|
saRNA
|
small activating RNA
|
References
|
1
|
Siegel RL, Miller KD and Jemal A: Cancer
statistics, 2017. CA Cancer J Clin. 67:7–30. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Filson CP, Marks LS and Litwin MS:
Expectant management for men with early stage prostate cancer. CA
Cancer J Clin. 65:265–282. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Chen WY, Tsai YC, Yeh HL, Suau F, Jiang
KC, Shao AN, Huang J and Liu YN: Loss of SPDEF and gain of TGFBI
activity after androgen deprivation therapy promote EMT and bone
metastasis of prostate cancer. Sci Signal. 10:eaam68262017.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Stoyanova T, Riedinger M, Lin S,
Faltermeier CM, Smith BA, Zhang KX, Going CC, Goldstein AS, Lee JK,
Drake JM, et al: Activation of Notch1 synergizes with multiple
pathways in promoting castration-resistant prostate cancer. Proc
Natl Acad Sci USA. 113:E6457–E6466. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Carvalho FL, Simons BW, Eberhart CG and
Berman DM: Notch signaling in prostate cancer: A moving target.
Prostate. 74:933–945. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Deng G, Ma L, Meng Q, Ju X, Jiang K, Jiang
P and Yu Z: Notch signaling in the prostate: Critical roles during
development and in the hallmarks of prostate cancer biology. J
Cancer Res Clin Oncol. 142:531–547. 2016. View Article : Google Scholar
|
|
7
|
Nabbi A, McClurg UL, Thalappilly S, Almami
A, Mobahat M, Bismar TA, Binda O and Riabowol KT: ING3 promotes
prostate cancer growth by activating the androgen receptor. BMC
Med. 15:1032017. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Miyamoto H, Meming EM and Chang C:
Androgen deprivation therapy for prostate cancer: Current status
and future prospects. Prostate. 61:332–353. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Stanbrough M, Bubley GJ, Ross K, Golub TR,
Rubin MA, Penning TM, Febbo PG and Balk SP: Increased expression of
genes converting adrenal androgens to testosterone in
androgen-independent prostate cancer. Cancer Res. 66:2815–2825.
2006. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Wang Y, Wu X, Ou L, Yang X, Wang X, Tang
M, Chen E and Luo C: PLCε knockdown inhibits prostate cancer cell
proliferation via suppression of Notch signalling and nuclear
translocation of the androgen receptor. Cancer Lett. 362:61–69.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Belandia B, Powell SM, García-Pedrero JM,
Walker MM, Bevan CL and Parker MG: Hey1, a mediator of notch
signaling, is an androgen receptor corepressor. Mol Cell Biol.
25:1425–1436. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Sadick H, Naim R, Gössler U, Hörmann K and
Riedel F: Angiogenesis in hereditary hemorrhagic telangiectasia:
VEGF165 plasma concentration in correlation to the VEGF expression
and microvessel density. Int J Mol Med. 15:15–19. 2005.
|
|
13
|
Shou J, Ross S, Koeppen H, de Sauvage FJ
and Gao WQ: Dynamics of notch expression during murine prostate
development and tumorigenesis. Cancer Res. 61:7291–7297.
2001.PubMed/NCBI
|
|
14
|
D'Amore PA and Ng YS: Won't you be my
neighbor? Local induction of arteriogenesis. Cell. 110:289–292.
2002. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Siekmann AF, Covassin L and Lawson ND:
Modulation of VEGF signalling output by the Notch pathway.
Bioessays. 30:303–313. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar
|
|
17
|
Roberts TC, Hart JR, Kaikkonen MU,
Weinberg MS, Vogt PK and Morris KV: Quantification of nascent
transcription by bromouridine immunocapture nuclear run-on RT-qPCR.
Nat Protoc. 10:1198–1211. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Alimirah F1, Chen J, Basrawala Z, Xin H
and Choubey D: DU-145 and PC-3 human prostate cancer cell lines
express androgen receptor: Implications for the androgen receptor
functions and regulation. FEBS Lett. 580:2294–2300. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Davis R, Jia D, Cinar B, Sikka SC, Moparty
K, Zhau HE, Chung LW, Agrawal KC and Abdel-Mageed AB: Functional
androgen receptor confers sensitization of androgen-independent
prostate cancer cells to anticancer therapy via caspase activation.
Biochem Biophys Res Commun. 309:937–945. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Deng G, Ju X, Meng Q, Yu ZJ and Ma LB:
Emodin inhibits the proliferation of PC3 prostate cancer cells in
vitro via the Notch signaling pathway. Mol Med Rep. 12:4427–4433.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Zhang L, Sha J, Yang G, Huang X, Bo J and
Huang Y: Activation of Notch pathway is linked with
epithelial-mesenchymal transition in prostate cancer cells. Cell
Cycle. 16:999–1007. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Yang JH, Wylie-Sears J and Bischoff J:
Opposing actions of Notch1 and VEGF in post-natal cardiac valve
endothelial cells. Biochem Biophys Res Commun. 374:512–516. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Bao B, Wang Z, Ali S, Kong D, Li Y, Ahmad
A, Banerjee S, Azmi AS, Miele L and Sarkar FH: Notch-1 induces
epithelial-mesenchymal transition consistent with cancer stem cell
phenotype in pancreatic cancer cells. Cancer Lett. 307:26–36. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Mao Q, Zheng X, Yang K, Qin J, Bai Y, Jia
X, Li Y and Xie L: Suppression of migration and invasion of PC3
prostate cancer cell line via activating E-cadherin expression by
small activating RNA. Cancer Invest. 28:1013–1018. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Yang K, Zheng XY, Qin J, Wang YB, Bai Y,
Mao QQ, Wan Q, Wu ZM and Xie LP: Up-regulation of p21WAF1/Cip1 by
saRNA induces G1-phase arrest and apoptosis in T24 human bladder
cancer cells. Cancer Lett. 265:206–214. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Liu ZJ, Shirakawa T, Li Y, Soma A, Oka M,
Dotto GP, Fairman RM, Velazquez OC and Herlyn M: Regulation of
Notch1 and Dll4 by vascular endothelial growth factor in arterial
endothelial cells: Implications for modulating arteriogenesis and
angiogenesis. Mol Cell Biol. 23:14–25. 2003. View Article : Google Scholar :
|
|
27
|
Larrivée B, Prahst C, Gordon E, del Toro
R, Mathivet T, Duarte A, Simons M and Eichmann A: ALK1 signaling
inhibits angiogenesis by cooperating with the Notch pathway. Dev
Cell. 22:489–500. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Yang J, Wang C, Zhang Z, Chen X, Jia Y,
Wang B and Kong T: Curcumin inhibits the survival and metastasis of
prostate cancer cells via the Notch-1 signaling pathway. APMIS.
125:134–140. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Sahara M, Hansson EM, Wernet O, Lui KO,
Später D and Chien KR: Manipulation of a VEGF-Notch signaling
circuit drives formation of functional vascular endothelial
progenitors from human pluripotent stem cells. Cell Res.
25:1482015. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Zang M, Hu L, Zhang B, Zhu Z, Li J, Zhu Z,
Yan M and Liu B: Luteolin suppresses angiogenesis and vasculogenic
mimicry formation through inhibiting Notch1-VEGF signaling in
gastric cancer. Biochem Biophys Res Commun. 490:913–919. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Esquela-Kerscher A and Slack FJ:
Oncomirs-microRNAs with a role in cancer. Nat Rev Cancer.
6:259–269. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Elbashir SM, Harborth J, Lendeckel W,
Yalcin A, Weber K and Tuschl T: Duplexes of 21-nucleotide RNAs
mediate RNA interference in cultured mammalian cells. Nature.
411:494–498. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Chen Z, Place RF, Jia ZJ, Pookot D, Dahiya
R and Li LC: Antitumor effect of dsRNA-induced p21(WAF1/CIP1) gene
activation in human bladder cancer cells. Mol Cancer Ther.
7:698–703. 2008. View Article : Google Scholar : PubMed/NCBI
|