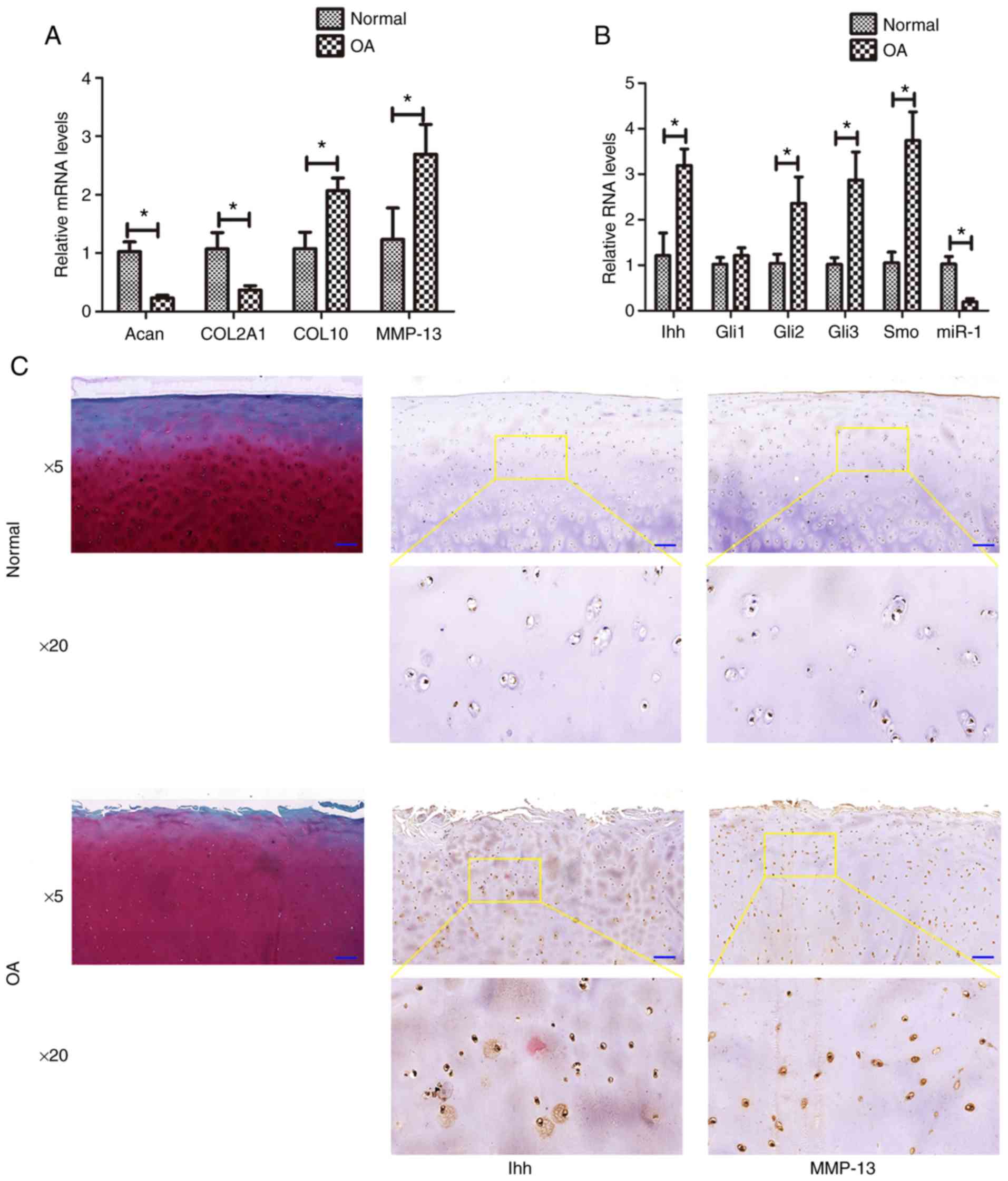
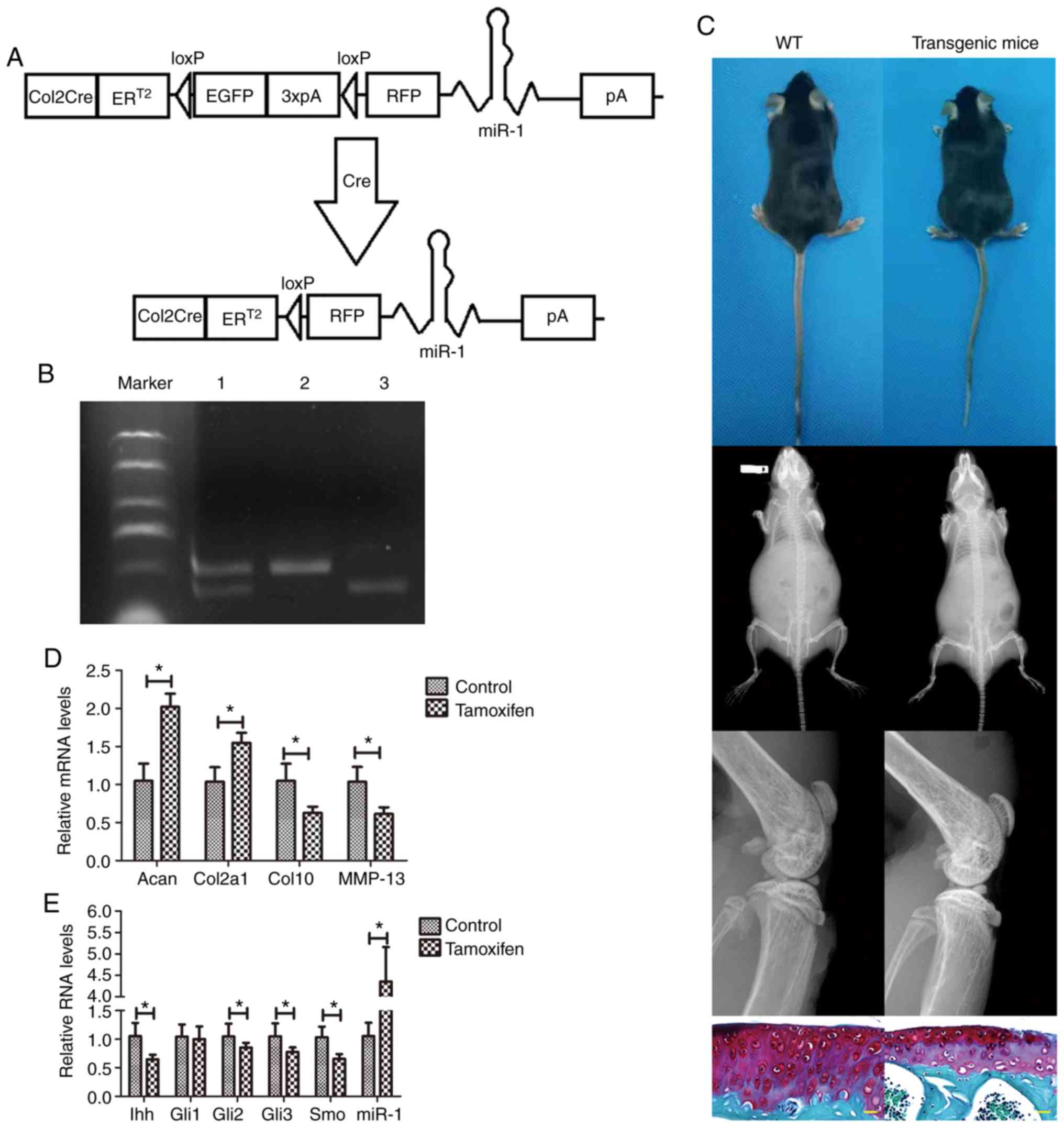
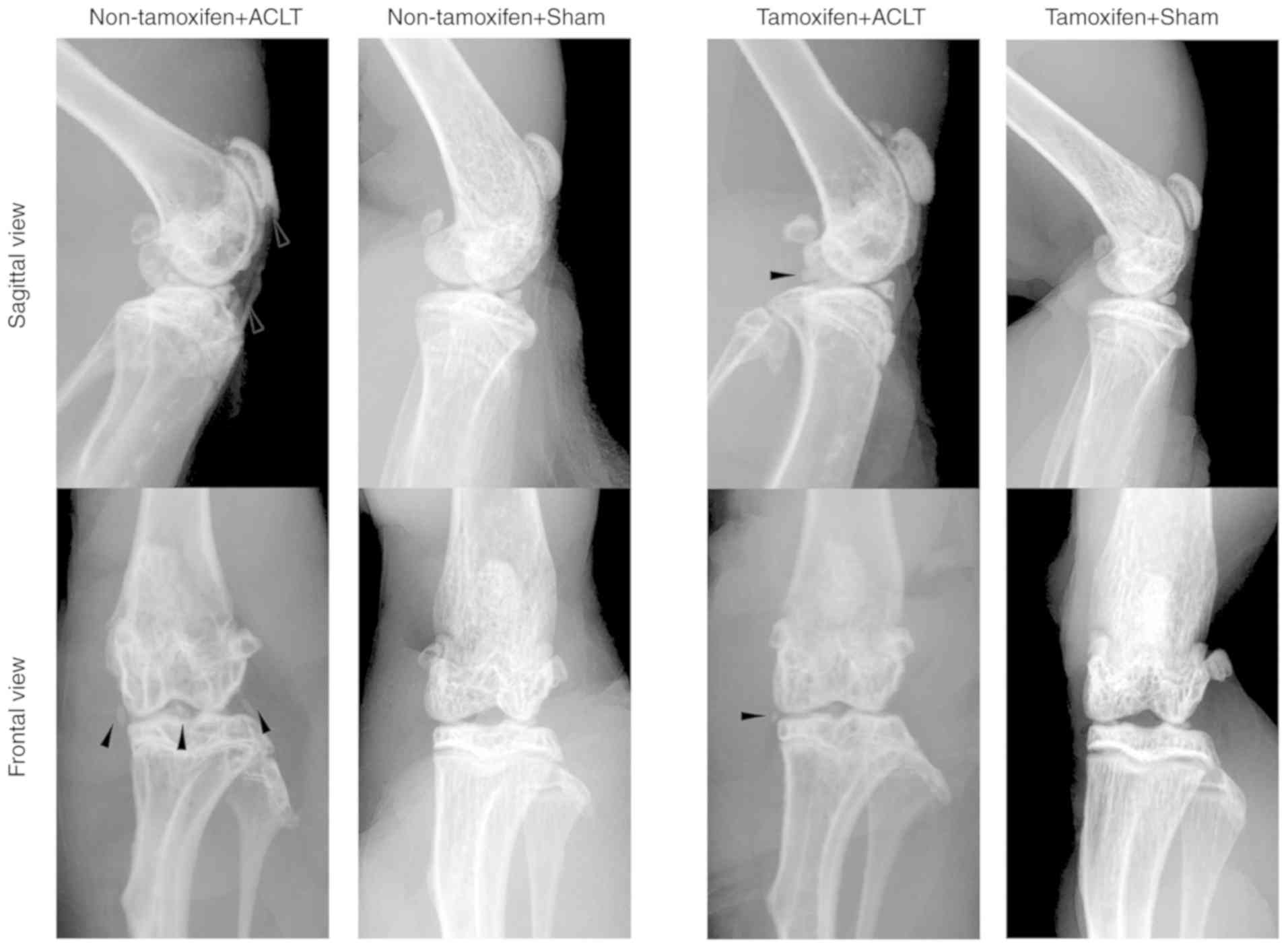
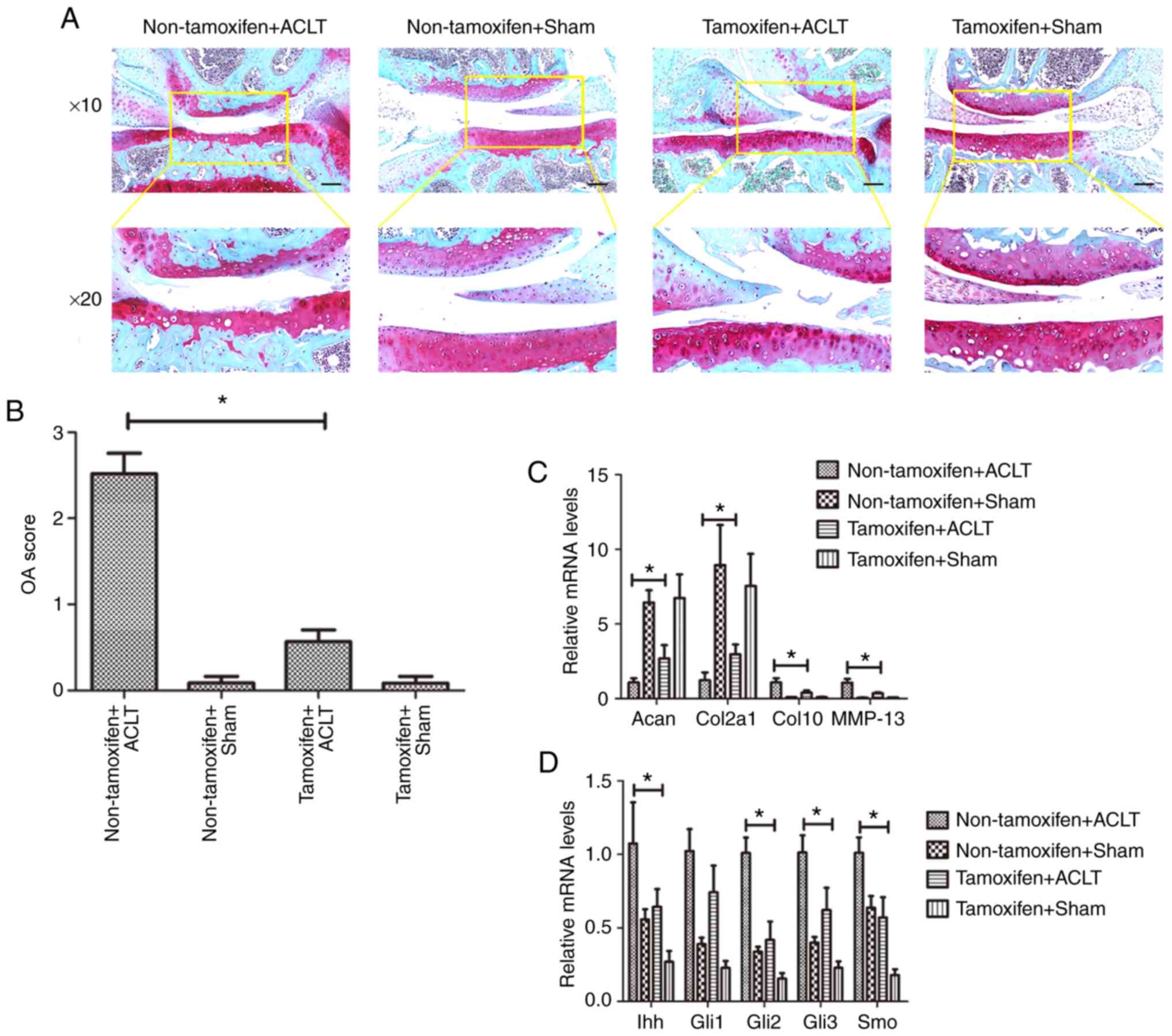
|
1
|
Glyn-Jones S, Palmer AJ, Agricola R, Price
AJ, Vincent TL, Weinans H and Carr AJ: Osteoarthritis. Lancet.
386:376–387. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Miller ME, Rejeski WJ, Messier SP and
Loeser RF: Modifiers of change in physical functioning in older
adults with knee pain: The observational arthritis study in seniors
(OASIS). Arthritis Rheum. 45:331–339. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Arden N and Nevitt MC: Osteoarthritis:
Epidemiology. Best Pract Res Clin Rheumatol. 20:3–25. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Berenbaum F: Osteoarthritis as an
inflammatory disease (osteoarthritis is not osteoarthrosis!).
Osteoarthritis Cartilage. 21:16–21. 2013. View Article : Google Scholar
|
|
5
|
Araldi E and Schipani E: MicroRNA-140 and
the silencing of osteoarthritis. Genes Dev. 24:1075–1080. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Miyaki S, Sato T, Inoue A, Otsuki S, Ito
Y, Yokoyama S, Kato Y, Takemoto F, Nakasa T, Yamashita S, et al:
MicroRNA-140 plays dual roles in both cartilage development and
homeostasis. Genes Dev. 24:1173–1185. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Zhang X, Wang C, Zhao J, Xu J, Geng Y, Dai
L, Huang Y, Fu SC, Dai K and Zhang X: MiR-146a facilitates
osteoarthritis by regulating cartilage homeostasis via targeting
Camk2d and Ppp3r2. Cell Death Dis. 8:e27342017. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Guan YJ, Li J, Yang X, Du S, Ding J, Gao
Y, Zhang Y, Yang K and Chen Q: Evidence that miR-146a attenuates
aging- and trauma-induced osteoarthritis by inhibiting Notch1,
IL-6, and IL-1 mediated catabolism. Aging Cell. 17:e127522018.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Li YP, Wei XC, Li PC, Chen CW, Wang XH,
Jiao Q, Wang DM, Wei FY, Zhang JZ and Wei L: The role of miRNAs in
cartilage homeostasis. Curr Genomics. 16:393–404. 2015. View Article : Google Scholar
|
|
10
|
Bagga S, Bracht J, Hunter S, Massirer K,
Holtz J, Eachus R and Pasquinelli AE: Regulation by let-7 and lin-4
miRNAs results in target mRNA degradation. Cell. 122:553–563. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Eulalio A, Rehwinkel J, Stricker M,
Huntzinger E, Yang SF, Doerks T, Dorner S, Bork P, Boutros M and
Izaurralde E: Target-specific requirements for enhancers of
decapping in miRNA-mediated gene silencing. Genes Dev.
21:2558–2570. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Valencia-Sanchez MA, Liu J, Hannon GJ and
Parker R: Control of translation and mRNA degradation by miRNAs and
siRNAs. Genes Dev. 20:515–524. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Yuan X, Guo Y, Chen D, Luo Y, Chen D, Miao
J and Chen Y: Long non-coding RNA MALAT1 functions as miR-1 sponge
to regulate Connexin 43-mediated ossification of the posterior
longitudinal ligament. Bone. 127:305–314. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Li P, Wei X, Guan Y, Chen Q, Zhao T, Sun C
and Wei L: MicroRNA-1 regulates chondrocyte phenotype by repressing
histone deacetylase 4 during growth plate development. FASEB J.
28:3930–3941. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Zhou J, Wei X and Wei L: Indian Hedgehog,
a critical modulator in osteoarthritis, could be a potential
therapeutic target for attenuating cartilage degeneration disease.
Connect Tissue Res. 55:257–261. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Lin AC, Seeto BL, Bartoszko JM, Khoury MA,
Whetstone H, Ho L, Hsu C, Ali SA and Alman BA: Modulating hedgehog
signaling can attenuate the severity of osteoarthritis. Nat Med.
15:1421–1425. 2009. View
Article : Google Scholar : PubMed/NCBI
|
|
17
|
Kirsch T, Swoboda B and Nah H: Activation
of annexin II and V expression, terminal differentiation,
mineralization and apoptosis in human osteoarthritic cartilage.
Osteoarthritis Cartilage. 8:294–302. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Pfander D, Swoboda B and Kirsch T:
Expression of early and late differentiation markers (proliferating
cell nuclear antigen, syndecan-3, annexin VI, and alkaline
phosphatase) by human osteoarthritic chondrocytes. Am J Pathol.
159:1777–1783. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Zhang C, Wei X, Chen C, Cao K, Li Y, Jiao
Q, Ding J, Zhou J, Fleming BC, Chen Q, et al: Indian hedgehog in
synovial fluid is a novel marker for early cartilage lesions in
human knee joint. Int J Mol Sci. 15:7250–7265. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Orfanidou T, Iliopoulos D, Malizos KN and
Tsezou A: Involvement of SOX-9 and FGF-23 in RUNX-2 regulation in
osteoarthritic chondrocytes. J Cell Mol Med. 13:3186–3194. 2009.
View Article : Google Scholar
|
|
21
|
Wei F, Zhou J, Wei X, Zhang J, Fleming BC,
Terek R, Pei M, Chen Q, Liu T and Wei L: Activation of Indian
hedgehog promotes chondrocyte hypertrophy and upregulation of
MMP-13 in human osteoarthritic cartilage. Osteoarthritis Cartilage.
20:755–763. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Zhou J, Chen Q, Lanske B, Fleming BC,
Terek R, Wei X, Zhang G, Wang S, Li K and Wei L: Disrupting the
Indian hedgehog signaling pathway in vivo attenuates surgically
induced osteoarthritis progression in Col2a1-CreERT2; Ihhfl/fl
mice. Arthritis Res Ther. 16:R112014. View
Article : Google Scholar : PubMed/NCBI
|
|
23
|
Guo L, Wei X, Zhang Z, Wang X, Wang C, Li
P, Wang C and Wei L: Ipriflavone attenuates the degeneration of
cartilage by blocking the Indian hedgehog pathway. Arthritis Res
Ther. 21:1092019. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Lorenz J and Grassel S: Experimental
osteoarthritis models in mice. Methods Mol Biol. 1194:401–419.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Jeon OH, Kim C, Laberge RM, Demaria M,
Rathod S, Vasserot AP, Chung JW, Kim DH, Poon Y, David N, et al:
Local clearance of senescent cells attenuates the development of
post-traumatic osteoarthritis and creates a pro-regenerative
environment. Nat Med. 23:775–781. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Altman R, Asch E, Bloch D, Bole G,
Borenstein D, Brandt K, Christy W, Cooke TD, Greenwald R, Hochberg
M, et al: Development of criteria for the classification and
reporting of osteoarthritis. Classification of osteoarthritis of
the knee. Diagnostic and therapeutic criteria committee of the
American rheumatism association. Arthritis Rheum. 29:1039–1049.
1986. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar
|
|
28
|
Glasson SS, Chambers MG, Van Den Berg WB
and Little CB: The OARSI histopathology initiative-recommendations
for histological assessments of osteoarthritis in the mouse.
Osteoarthritis Cartilage. 18(Suppl 3): S17–S23. 2010. View Article : Google Scholar
|
|
29
|
Ntziachristos V, Bremer C and Weissleder
R: Fluorescence imaging with near-infrared light: New technological
advances that enable in vivo molecular imaging. Eur Radiol.
13:195–208. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Peterson JD, Labranche TP, Vasquez KO,
Kossodo S, Melton M, Rader R, Listello JT, Abrams MA and Misko TP:
Optical tomographic imaging discriminates between disease-modifying
anti-rheumatic drug (DMARD) and non-DMARD efficacy in collagen
antibody-induced arthritis. Arthritis Res Ther. 12:R1052010.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Selbach M, Schwanhausser B, Thierfelder N,
Fang Z, Khanin R and Rajewsky N: Widespread changes in protein
synthesis induced by microRNAs. Nature. 455:58–63. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Chen JF, Mandel EM, Thomson JM, Wu Q,
Callis TE, Hammond SM, Conlon FL and Wang DZ: The role of
microRNA-1 and microRNA-133 in skeletal muscle proliferation and
differentiation. Nat Genet. 38:228–233. 2006. View Article : Google Scholar
|
|
33
|
Young B, Minugh-Purvis N, Shimo T,
St-Jacques B, Iwamoto M, Enomoto-Iwamoto M, Koyama E and Pacifici
M: Indian and sonic hedgehogs regulate synchondrosis growth plate
and cranial base development and function. Dev Biol. 299:272–282.
2006. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Aigner T, Reichenberger E, Bertling W,
Kirsch T, Stoss H and von der Mark K: Type X collagen expression in
osteoarthritic and rheumatoid articular cartilage. Virchows Arch B
Cell Pathol Incl Mol Pathol. 63:205–211. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Clements DN, Carter SD, Innes JF, Ollier
WE and Day PJ: Analysis of normal and osteoarthritic canine
cartilage mRNA expression by quantitative polymerase chain
reaction. Arthritis Res Ther. 8:R1582006. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Little CB, Barai A, Burkhardt D, Smith SM,
Fosang AJ, Werb Z, Shah M and Thompson EW: Matrix metalloproteinase
13-deficient mice are resistant to osteoarthritic cartilage erosion
but not chondrocyte hypertrophy or osteophyte development.
Arthritis Rheum. 60:3723–3733. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Aigner T, Dietz U, Stoss H and von der
Mark K: Differential expression of collagen types I, II, III, and X
in human osteophytes. Lab Invest. 73:236–243. 1995.PubMed/NCBI
|
|
38
|
Tchetina EV, Squires G and Poole AR:
Increased type II collagen degradation and very early focal
cartilage degeneration is associated with upregulation of
chondrocyte differentiation related genes in early human articular
cartilage lesions. J Rheumatol. 32:876–886. 2005.PubMed/NCBI
|