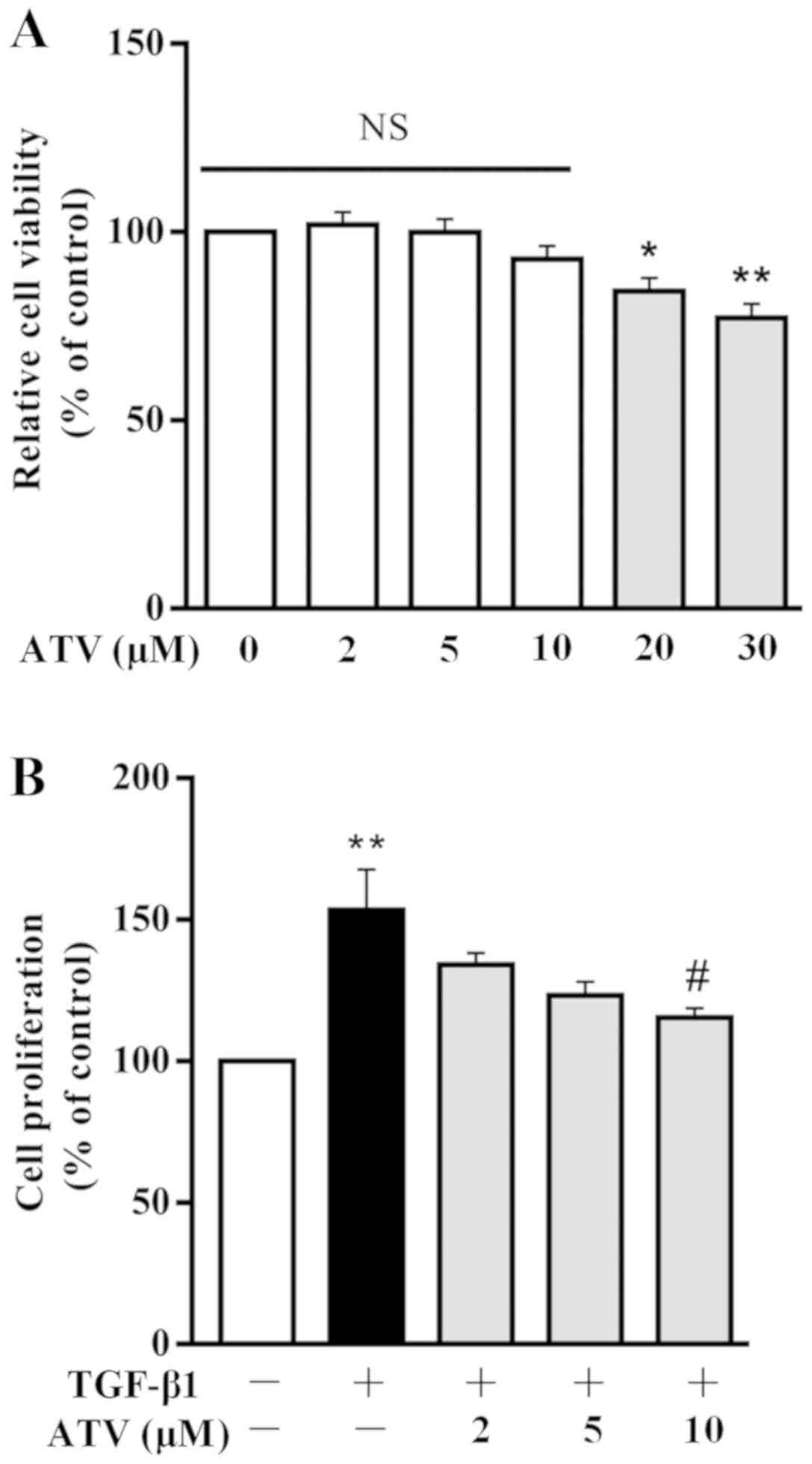
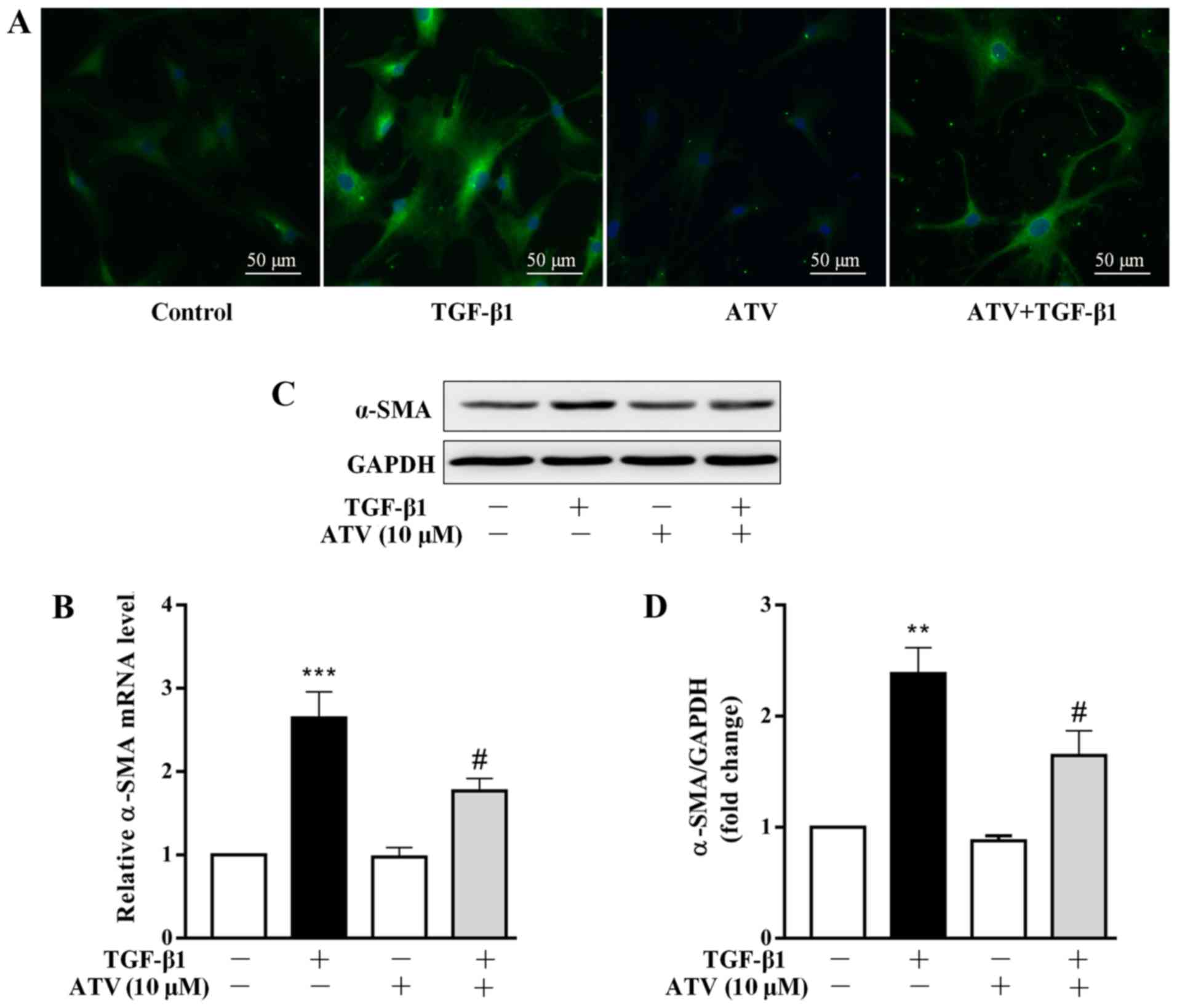
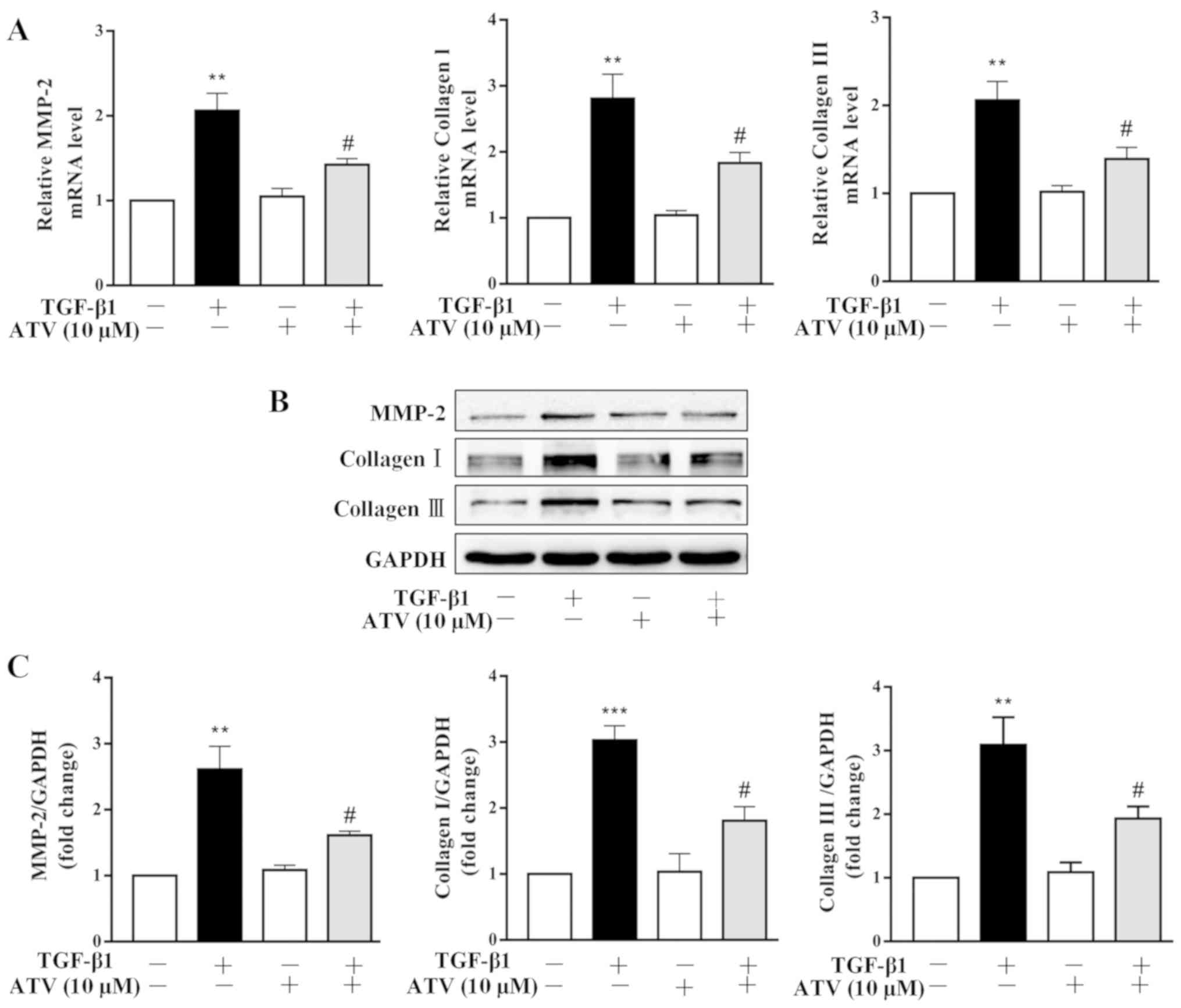
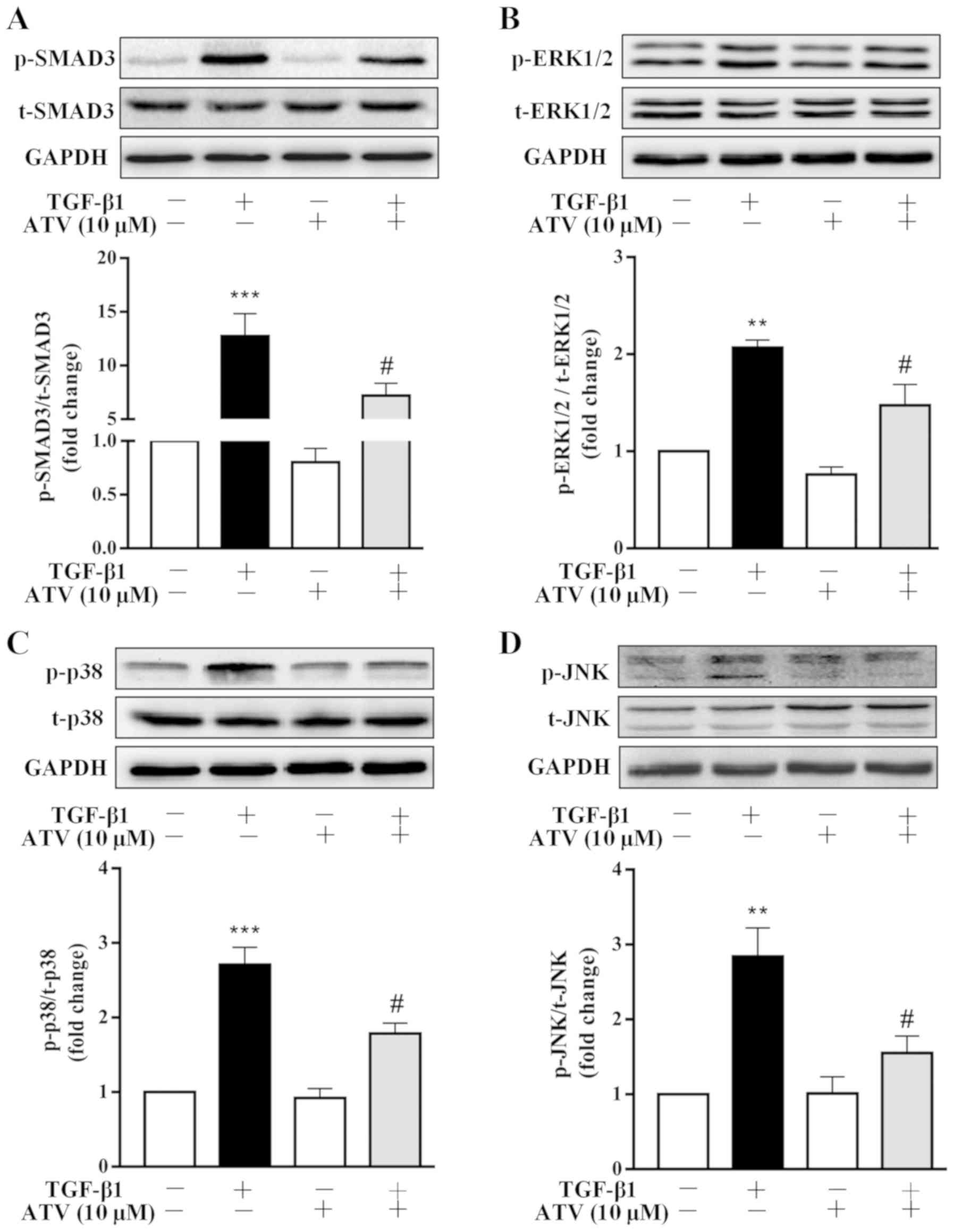
|
1
|
Rockey DC, Bell PD and Hill JA: Fibrosis-a
common pathway to organ injury and failure. N Engl J Med.
373:962015.
|
|
2
|
Davis J and Molkentin JD: Myofibroblasts:
Trust your heart and let fate decide. J Mol Cell Cardiol. 70:9–18.
2014. View Article : Google Scholar
|
|
3
|
Schafer S, Viswanathan S, Widjaja AA, Lim
WW, Moreno-Moral A, DeLaughter DM, Ng B, Patone G, Chow K, Khin E,
et al: IL-11 is a crucial determinant of cardiovascular fibrosis.
Nature. 552:110–115. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Akhurst RJ and Hata A: Targeting the TGFβ
signalling pathway in disease. Nat Rev Drug Discov. 11:790–811.
2012. View
Article : Google Scholar : PubMed/NCBI
|
|
5
|
Wynn TA: Cellular and molecular mechanisms
of fibrosis. J Pathol. 214:199–210. 2008. View Article : Google Scholar
|
|
6
|
Eghbali M, Tomek R, Woods C and Bhambi B:
Cardiac fibro-blasts are predisposed to convert into myocyte
phenotype: Specific effect of transforming growth factor beta. Proc
Natl Acad Sci USA. 88:795–799. 1991. View Article : Google Scholar
|
|
7
|
Shi Y and Massague J: Mechanisms of
TGF-beta signaling from cell membrane to the nucleus. Cell.
113:685–700. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Schiller M, Javelaud D and Mauviel A:
TGF-beta-induced SMAD signaling and gene regulation: Consequences
for extracellular matrix remodeling and wound healing. J Dermatol
Sci. 35:83–92. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Evangelia P and Peter TD: TGFβ signaling
and cardiovascular diseases. Int J Bio Sci. 8:195–213. 2012.
View Article : Google Scholar
|
|
10
|
Chung CC, Kao YH, Yao CJ, Lin YK and Chen
YJ: A comparison of left and right atrial fibroblasts reveals
different collagen production activity and stress-induced
mitogen-activated protein kinase signalling in rats. Acta Physiol
(Oxf). 220:432–445. 2017. View Article : Google Scholar
|
|
11
|
Molkentin JD, Bugg D, Ghearing N, Dorn LE,
Kim P, Sargent MA, Gunaje J, Otsu K and Davis J:
Fibroblast-specific genetic manipulation of p38 mitogen-activated
protein kinase in vivo reveals its central regulatory role in
fibrosis. Circulation. 136:549–561. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
McFarlane SI, Muniyappa R, Francisco R and
Sowers JR: Clinical review 145: Pleiotropic effects of statins:
Lipid reduction and beyond. J Clin Endocrinol Metab. 87:1451–1458.
2002. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Wang CY, Liu PY and Liao JK: Pleiotropic
effects of statin therapy: Molecular mechanisms and clinical
results. Trends Mol Med. 14:37–44. 2008. View Article : Google Scholar
|
|
14
|
Ludman A, Venugopal V, Yellon DM and
Hausenloy DJ: Statins and cardioprotection-more than just lipid
lowering? Pharmacol Ther. 122:30–43. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Liao JK: Effects of statins on
3-hydroxy-3-methylglutaryl coenzyme a reductase inhibition beyond
low-density lipoprotein cholesterol. Am J Cardiol. 96:24F–33F.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Chen M, Li H, Wang G, Shen X, Zhao S and
Su W: Atorvastatin prevents advanced glycation end products
(AGEs)-induced cardiac fibrosis via activating peroxisome
proliferator-activated receptor gamma (PPAR-γ). Metabolism.
65:441–453. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Choi SY, Park JS, Roh MS, Kim CR, Kim MH
and Serebruany V: Inhibition of angiotensin II-induced cardiac
fibrosis by atorvastatin in adiponectin knockout mice. Lipids.
52:415–422. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Fang T, Guo B, Xue L and Wang L:
Atorvastatin prevents myocardial fibrosis in spontaneous
hypertension via inter-leukin-6 (IL-6)/signal transducer and
activator of transcription 3 (STAT3)/endothelin-1 (ET-1) pathway.
Med Sci Monit. 25:318–323. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Wang Q, Cui W, Zhang HL, Hu HJ, Zhang YN,
Liu DM and Liu J: Atorvastatin suppresses aldosterone-induced
neonatal rat cardiac fibroblast proliferation by inhibiting ERK1/2
in the genomic pathway. J Cardiovasc Pharmacol. 61:520–527. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Gao G, Jiang S, Ge L, Zhang S, Zhai C,
Chen W and Sui S: Atorvastatin improves doxorubicin-induced cardiac
dysfunction by modulating Hsp70, Akt, and MAPK signaling pathways.
J Cardiovasc Pharmacol. 73:223–231. 2019. View Article : Google Scholar
|
|
21
|
Xiao H, Ma X, Feng W, Fu Y, Lu Z, Xu M,
Sheng Q, Zhu Y and Zhang Y: Metformin attenuates cardiac fibrosis
by inhibiting the TGFbeta1-Smad3 signalling pathway. Cardiovasc
Res. 87:504–513. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar
|
|
23
|
Yi X, Li X, Zhou Y, Ren S, Wan W, Feng G
and Jiang X: Hepatocyte growth factor regulates the TGF-β1-induced
proliferation, differentiation and secretory function of cardiac
fibroblasts. Int J Mol Med. 34:381–390. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Van Nieuwenhoven FA and Turner NA: The
role of cardiac fibro-blasts in the transition from inflammation to
fibrosis following myocardial infarction. Vascul Pharmacol.
58:182–188. 2013. View Article : Google Scholar
|
|
25
|
Abdalla M, Goc A, Segar L and Somanath PR:
Akt1 mediates α-smooth muscle actin expression and myofibroblast
differentiation via myocardin and serum response factor. J Biol
Chem. 288:33483–33493. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Lan TH, Huang XQ and Tan HM: Vascular
fibrosis in atherosclerosis. Cardiovasc Pathol. 22:401–407. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Park S, Nguyen NB, Pezhouman A and
Ardehali R: Cardiac fibrosis: Potential therapeutic targets. Transl
Res. 209:121–137. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Jugdutt BI: Remodeling of the myocardium
and potential targets in the collagen degradation and synthesis
pathways. Curr Drug Targets Cardiovasc Haematol Disord. 3:1–30.
2003. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Souders CA, Bowers SL and Baudino TA:
Cardiac fibroblast: The renaissance cell. Circ Res. 105:1164–1176.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Li P, Wang D, Lucas J, Oparil S, Xing D,
Cao X, Novak L, Renfrow MB and Chen YF: Atrial natriuretic peptide
inhibits transforming growth factor beta-induced Smad signaling and
myofibroblast transformation in mouse cardiac fibroblasts. Circ
Res. 102:185–192. 2008. View Article : Google Scholar
|
|
31
|
Rizvi F, Siddiqui R, DeFranco A, Homar P,
Emelyanova L, Holmuhamedov E, Ross G, Tajik AJ and Jahangir A:
Simvastatin reduces TGF-β1-induced SMAD2/3-dependent human
ventricular fibroblasts differentiation: Role of protein
phosphatase activation. Int J Cardiol. 270:228–236. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Bujak M and Frangogiannis NG: The role of
TGF-beta signaling in myocardial infarction and cardiac remodeling.
Cardiovasc Res. 74:184–195. 2007. View Article : Google Scholar
|
|
33
|
Li RK, Li G, Mickle DA, Weisel RD, Merante
F, Luss H, Rao V, Christakis GT and Williams WG: Overexpression of
transforming growth factor-beta1 and insulin-like growth factor-I
in patients with idiopathic hypertrophic cardiomyopathy.
Circulation. 96:874–881. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Travers JG, Kamal FA, Robbins J, Yutzey KE
and Blaxall BC: Cardiac fibrosis: The fibroblast awakens. Circ Res.
118:1021–1040. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Petrov VV, Fagard RH and Lijnen PJ:
Stimulation of collagen production by transforming growth
factor-beta1 during differentiation of cardiac fibroblasts to
myofibroblasts. Hypertension. 39:258–263. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Li B, Chen H, Yang X, Wang Y, Qin L and
Chu Y: Knockdown of eIF3a ameliorates cardiac fibrosis by
inhibiting the TGF-β1/Smad3 signaling pathway. Cell Mol Biol
(Noisy-le-grand). 62:97–101. 2016.
|
|
37
|
Zhang M, Pan X, Zou Q, Xia Y, Chen J, Hao
Q, Wang H and Sun D: Notch3 Ameliorates cardiac fibrosis after
myocardial infarction by inhibiting the TGF-β1/Smad3 pathway.
Cardiovasc Toxicol. 16:316–324. 2016. View Article : Google Scholar
|
|
38
|
Zhao M, Zheng S, Yang J, Wu Y, Ren Y, Kong
X, Li W and Xuan J: Suppression of TGF-β1/Smad signaling pathway by
sesamin contributes to the attenuation of myocardial fibrosis in
spontaneously hypertensive rats. PLoS One. 10:e01213122015.
View Article : Google Scholar
|
|
39
|
Khalil H, Kanisicak O, Prasad V, Correll
RN, Fu X, Schips T, Vagnozzi RJ, Liu R, Huynh T, Lee SJ, et al:
Fibroblast-specific TGF-β-Smad2/3 signaling underlies cardiac
fibrosis. J Clin Invest. 127:3770–3783. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Massagué J: TGFβ signalling in context.
Nat Rev Mol Cell Biol. 13:616–630. 2012. View Article : Google Scholar
|
|
41
|
Schmierer B and Hill CS: TGFbeta-SMAD
signal transduction: Molecular specificity and functional
flexibility. Nat Rev Mol Cell Biol. 8:970–982. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Lee SJ, Park K, Ha SD, Kim WJ and Moon SK:
Gleditsia sinensis thorn extract inhibits human colon cancer cells:
The role of ERK1/2, G2/M-phase cell cycle arrest and p53
expression. Phytother Res. 24:1870–1876. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Yin Y, Guan Y, Duan J, Wei G, Zhu Y, Quan
W, Guo C, Zhou D, Wang Y, Xi M and Wen A: Cardioprotective effect
of Danshensu against myocardial ischemia/reperfusion injury and
inhibits apoptosis of H9c2 cardiomyocytes via Akt and ERK1/2
phosphorylation. Eur J Pharmacol. 699:219–226. 2013. View Article : Google Scholar
|
|
44
|
Zhang W and Liu HT: MAPK signal pathways
in the regulation of cell proliferation in mammalian cells. Cell
Res. 12:9–18. 2002. View Article : Google Scholar : PubMed/NCBI
|