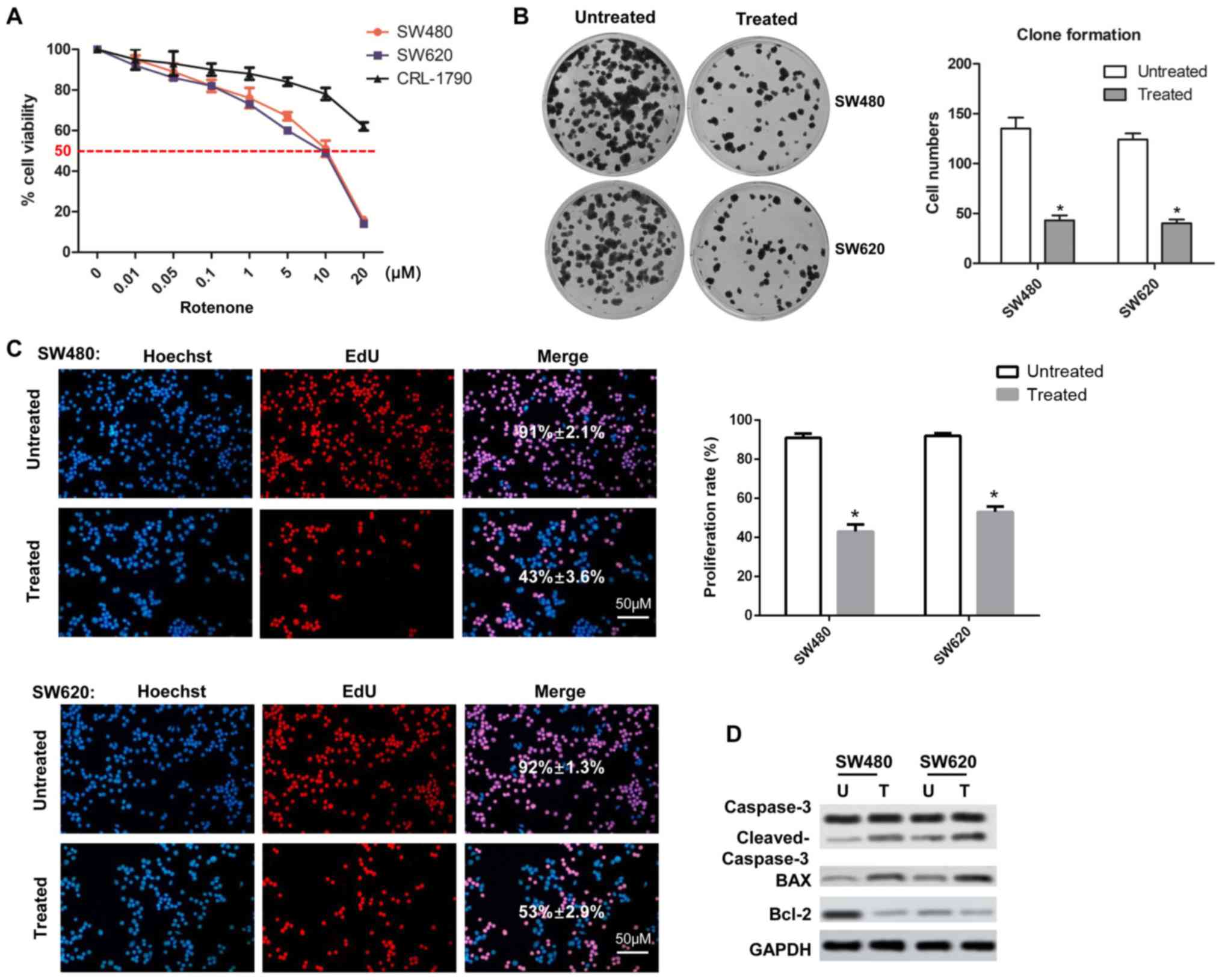
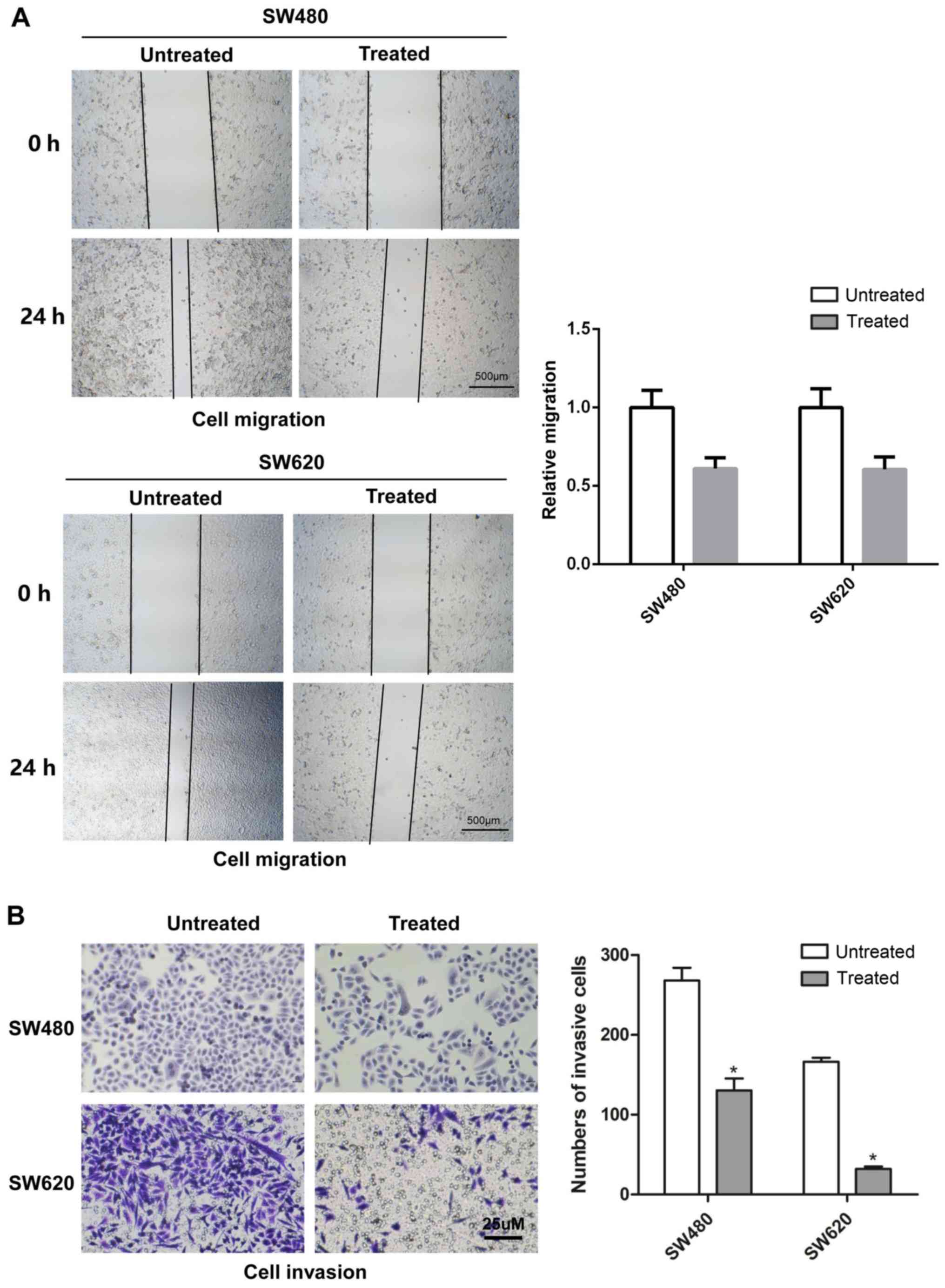
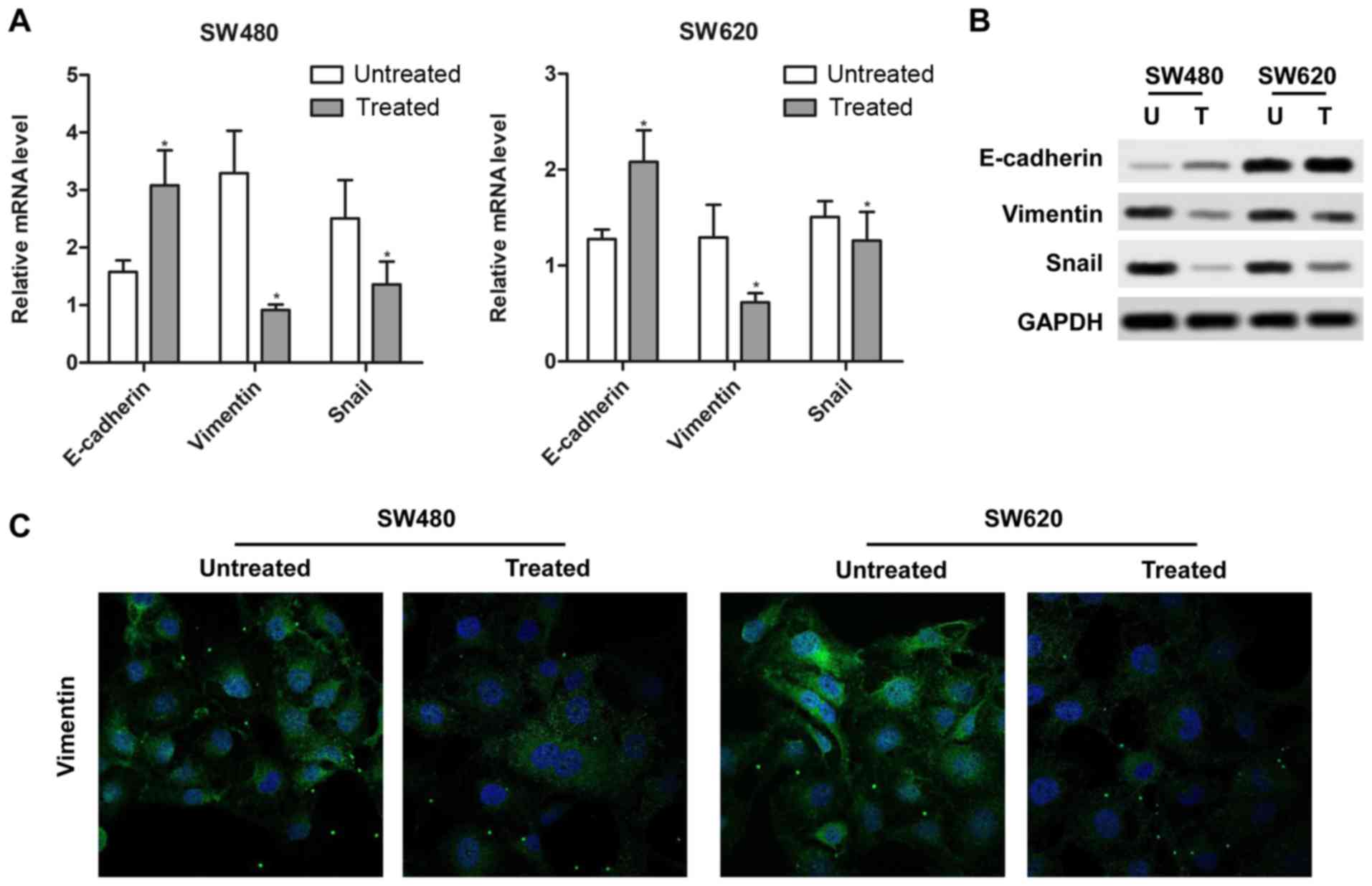
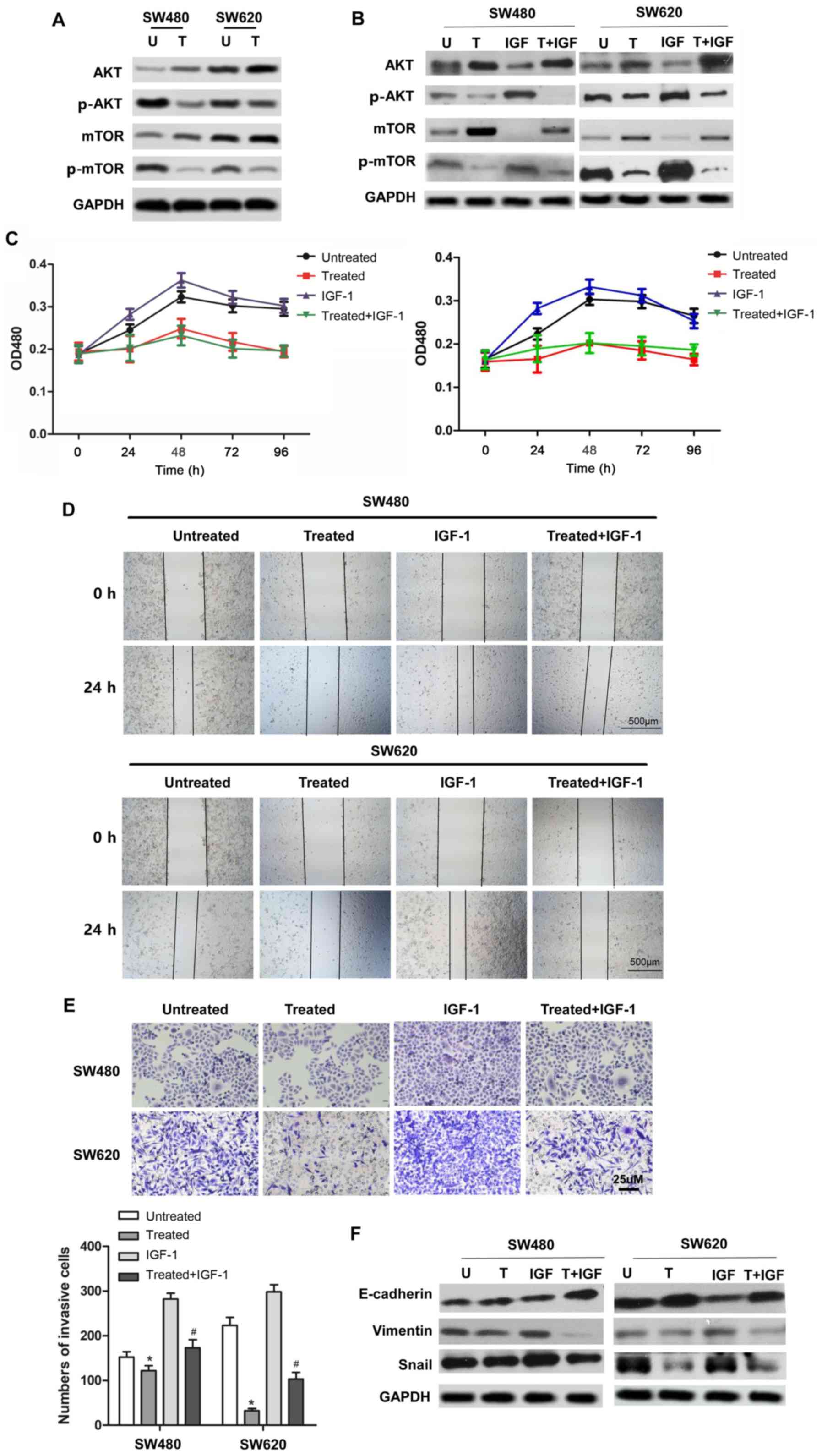
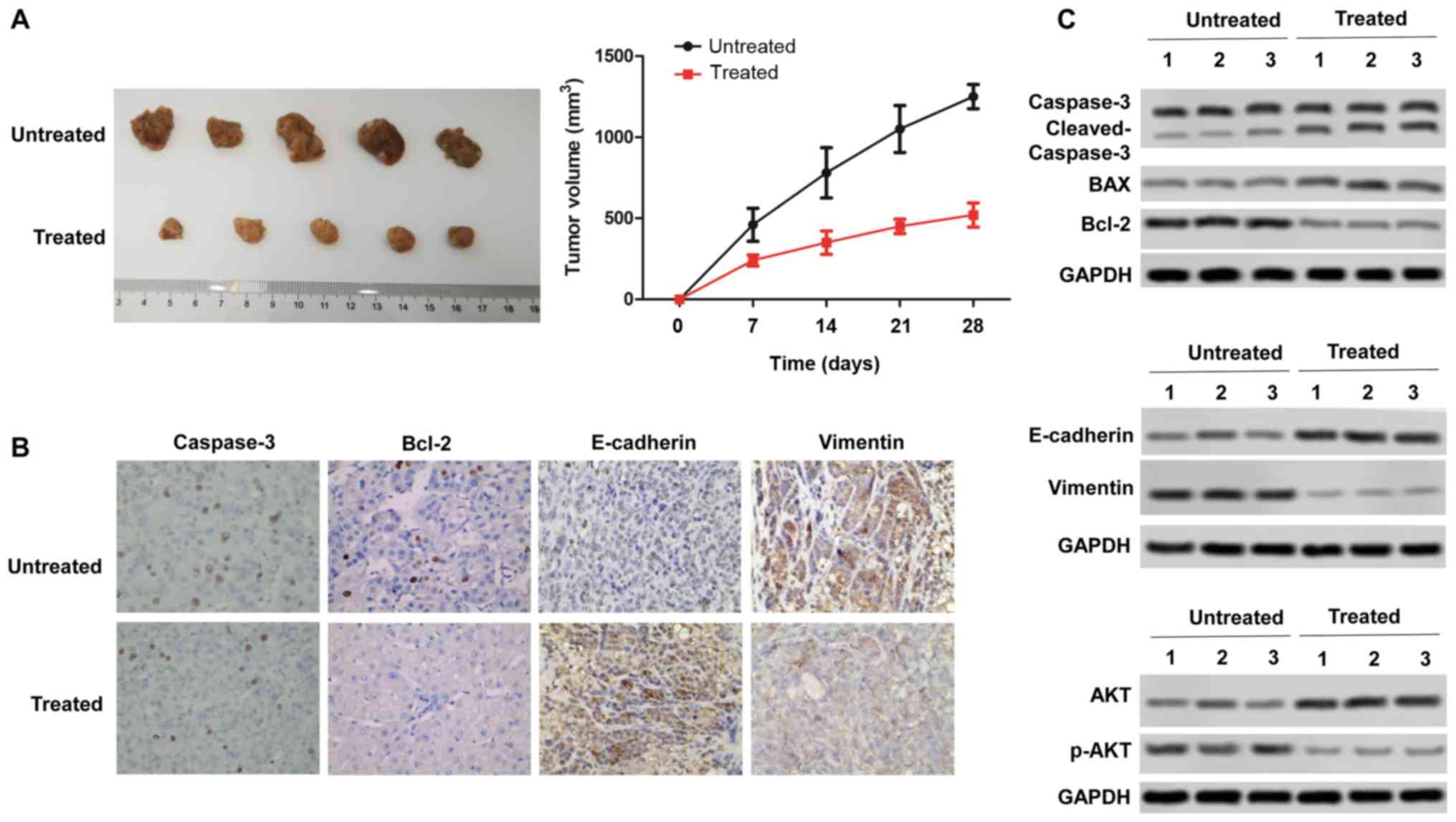
|
1
|
Ferlay J, Soerjomataram I, Dikshit R, Eser
S, Mathers C, Rebelo M, Parkin DM, Forman D and Bray F: Cancer
incidence and mortality worldwide: Sources methods and major
patterns in GLOBOCAN 2012. Int J Cancer. 136:E359–E386. 2015.
View Article : Google Scholar
|
|
2
|
Jaganathan SK, Vellayappan MV, Narasimhan
G and Supriyanto E: Role of pomegranate and citrus fruit juices in
colon cancer prevention. World J Gastroenterol. 20:4618–4625. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Chen J, Elfiky A, Han M, Chen C and Saif
MW: The role of Src in colon cancer and its therapeutic
implications. Clin Colorectal Cancer. 13:5–13. 2014. View Article : Google Scholar
|
|
4
|
Aggarwal BB, Takada Y and Oommen OV: From
chemoprevention to chemotherapy: Common targets and common goals.
Expert Opin Investig Drugs. 13:1327–1338. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Lee HY, Suh YA, Kosmeder JW, Pezzuto JM,
Hong WK and Kurie JM: Deguelin-induced inhibition of
cyclooxygenase-2 expression in human bronchial epithelial cells.
Clin Cancer Res. 10:1074–1079. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Dell'Eva R, Ambrosini C, Minghelli S,
Noonan DM, Albini A and Ferrari N: The Akt inhibitor deguelin, is
an angiopreventive agent also acting on the NF-kappaB pathway.
Carcinogenesis. 28:404–413. 2007. View Article : Google Scholar
|
|
7
|
Gerhäuser C, Mar W, Lee SK, Suh N, Luo Y,
Kosmeder J, Luyengi L, Fong HH, Kinghorn AD, Moriarty RM, et al:
Rotenoids mediate potent cancer chemopreventive activity through
transcriptional regulation of ornithine decarboxylase. Nat Med.
1:260–266. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Chun KH, Kosmeder JW II, Sun S, Pezzuto
JM, Lotan R, Hong WK and Lee HY: Effects of deguelin on the
phosphatidylinositol 3-kinase/Akt pathway and apoptosis in
premalignant human bronchial epithelial cells. J Natl Cancer Inst.
95:291–302. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Shang HS, Chang JB, Lin JH, Lin JP, Hsu
SC, Liu CM, Liu JY, Wu PP, Lu HF, Au MK and Chung JG: Deguelin
inhibits the migration and invasion of U-2 OS human osteosarcoma
cells via the inhibition of matrix metalloproteinase-2/-9 in vitro.
Molecules. 19:16588–16608. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Liu YP, Lee JJ, Lai TC, Lee CH, Hsiao YW,
Chen PS, Liu WT, Hong CY, Lin SK, Ping Kuo MY, et al: Suppressive
function of low-dose deguelin on the invasion of oral cancer cells
by downregulating tumor necrosis factor alpha-induced nuclear
factor-kappa B signaling. Head Neck. 38(Suppl 1): E524–E534. 2016.
View Article : Google Scholar
|
|
11
|
Deng YT, Huang HC and Lin JK: Rotenone
induces apoptosis in MCF-7 human breast cancer cell-mediated ROS
through JNK and p38 signaling. Mol Carcinog. 49:141–151. 2010.
|
|
12
|
Lee JH, Lee DH, Lee HS, Choi JS, Kim KW
and Hong SS: Deguelin inhibits human hepatocellular carcinoma by
antiangiogenesis and apoptosis. Oncol Rep. 20:129–134.
2008.PubMed/NCBI
|
|
13
|
Hu W, Tian H, Yue W, Li L, Li S, Gao C, Si
L, Qi L, Lu M, Hao B and Shan S: Rotenone induces apoptosis in
human lung cancer cells by regulating autophagic flux. IUBMB Life.
68:388–393. 2016. View
Article : Google Scholar : PubMed/NCBI
|
|
14
|
Chen Y and Ding YY: LINC00467 enhances
head and neck squamous cell carcinoma progression and
epithelial-mesenchymal transition process via miR-299-5p/ubiquitin
specific protease-48 axis. J Gene Med. e318411–Mar;2020.Epub ahead
of print.
|
|
15
|
Iwadate Y: Epithelial-mesenchymal
transition in glioblastoma progression. Oncol Lett. 11:1615–1620.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Boreddy SR and Srivastava SK: Deguelin
suppresses pancreatic tumor growth and metastasis by inhibiting
epithelial-to-mesen-chymal transition in an orthotopic model.
Oncogene. 32:3980–3991. 2013. View Article : Google Scholar
|
|
17
|
Wang X, Liu H, Wang X and An Y: Clinical
significance of migration and invasion inhibitor protein expression
in non-small-cell lung cancer. Oncol Lett. 8:2417–2422. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Zhao D, Han W, Liu X, Cui D and Chen Y:
Deguelin inhibits epithelial-to-mesenchymal transition and
metastasis of human non-small cell lung cancer cells by regulating
NIMA-related kinase 2. Thorac Cancer. 8:320–327. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Miao HL, Lei CJ, Qiu ZD, Liu ZK, Li R, Bao
ST and Li MY: MicroRNA-520c-3p inhibits hepatocellular carcinoma
cell proliferation and invasion through induction of cell apoptosis
by targeting glypican-3. Hepatol Res. 44:338–348. 2014. View Article : Google Scholar
|
|
20
|
Zeng C, Pan F, Jones LA, Lim MM, Griffin
EA, Sheline YI, Mintun MA, Holtzman DM and Mach RH: Evaluation of
5-ethynyl-2′-deoxyuridine staining as a sensitive and reliable
method for studying cell proliferation in the adult nervous system.
Brain Res. 1319:21–32. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar
|
|
22
|
Kang HW, Kim JM, Cha MY, Jung HC, Song IS
and Kim JS: Deguelin, an Akt inhibitor, down-regulates NF-κB
signaling and induces apoptosis in colon cancer cells and inhibits
tumor growth in mice. Dig Dis Sci. 57:2873–2882. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Xia ZS, Wu D, Zhong W, Lu XJ, Yu T and
Chen QK: Wip1 gene silencing enhances the chemosensitivity of human
colon cancer cells. Oncol Lett. 14:1875–1883. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Ding YL, Wang QS, Zhao WM and Xiang L:
Expression of smoothened protein in colon cancer and its prognostic
value for postoperative liver metastasis. Asian Pac J Cancer Prev.
13:4001–4005. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Goncalves AP, Videira A, Maximo V and
Soares P: Synergistic growth inhibition of cancer cells harboring
the RET/PTC1 oncogene by staurosporine and rotenone involves
enhanced cell death. J Biosci. 36:639–648. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Agarwal NR, Maurya N, Pawar JS and Ghosh
I: A combined approach against tumorigenesis using glucose
deprivation and mitochondrial complex 1 inhibition by rotenone.
Cell Biol Int. 40:821–831. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Matsumoto R, Mori S, Kita Y, Toda H,
Sasaki K, Arigami T, Matsushita D, Kurahara H, Maemura K and
Natsugoe S: Multiple liver metastases with synchronous gastric and
transverse colon cancer diagnosed by gastric perforation
successfully treated by SOX plus bevacizumab and completely
resected by surgery: A case report. Surg Case Rep. 6:512020.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Li S, Hou X, Wu C, Han L, Li Q, Wang J and
Luo S: MiR-645 promotes invasiveness, metastasis and tumor growth
in colorectal cancer by targeting EFNA5. Biomed Pharmacother.
125:1098892020. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Valastyan S and Weinberg RA: Tumor
metastasis: Molecular insights and evolving paradigms. Cell.
147:275–292. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Bates RC and Mercurio AM: The
epithelial-mesenchymal tansition (EMT) and colorectal cancer
progression. Cancer Biol Ther. 4:365–370. 2005. View Article : Google Scholar : PubMed/NCBI
|