Introduction
Osteoporosis, a systemic metabolic bone disease,
which mainly occurs among elderly, thin, post-menopausal women
(1-4), with the characteristic features of
decreased bone mass, reduced bone quality and a deteriorated
microarchitecture, which leads to an increased risk of bone
fractures. The rate of bone resorption by osteoclasts overcomes the
rate of bone formation by osteoblasts, which causes an imbalance in
bone remodeling, thus leading to pathological bone disease
(5). Therefore, effective
strategies aimed at restoring bone formation capacity are required
in order to develop effective treatments for osteoporosis (2).
For bone formation, osteoblast progenitors undergo
proliferation and differentiation into mature osteoblasts. These
mature osteoblasts subsequently lay down the bone matrix and
mineralize it. Osteoblasts are produced by mesenchymal stem cell
(MSC) precursors via several microenvironment signaling pathways
(6-8), including the Wnt, transforming
growth factor-β (TGF-β) and bone morphogenetic protein (BMP)
pathways, which are involved in the bone cell differentiation
process and bone matrix synthesis. In canonical Wnt signaling, free
(unbound) β-catenin translocates to the nucleus, where it binds to
the T-cell specific transcription factor (TCF) and lymphoid
enhancer-binding factor (LEF) to activate TGF-β and BMP2 gene
transcription. In addition, Wnt signaling enhances the
transcription of osteoprotegerin (OPG) and decreases the level of
receptor activator of nuclear factor kappa-B ligand (RANKL), thus
determining a total decrease in the RANKL/OPG ratio, which
suppresses the differentiation of osteoclasts and enhances overall
bone formation (9). However, Wnt
signaling is deregulated or even blocked by different molecules in
osteoporosis. Dikkopf (DKK), Kremen and sclerostin factors inhibit
LRP5/6 co-factor, whereas secreted frizzled related protein (sFRP)
binds to Wnt factors. Therefore, the investigation of the
inhibitory agents and mechanisms of Wnt signaling antagonists as
well as the recovery of Wnt functions attenuate osteoporosis.
MicroRNAs (miRNAs or miRs) are a class of small,
endogenous RNAs of 15-25 nucleotides (nts) in length. miRNAs
modulate gene expression at the post-transcriptional level by
targeting specific mRNAs for degradation or translational
repression (10). With regard to
osteoblasts and osteogenesis, miRNAs have been reported to target
the mRNA of several regulators to elicit or hinder osteoblast
differentiation, depending on the specific targets (11-13). Notably, several miRNAs affect
osteogenesis by targeting Wnt signaling. For example, miR-29a binds
to multiple negative regulatory factors of Wnt signaling, including
Dkk1, Kremen2 and sFRP2, while its expression enhances bone
formation (14). In addition,
miR-218 is elicited through Wnt signaling and enhances bone
formation, binding to multiple antagonists of Wnt signaling,
including Dkk1, Sclerostin (SOST) and sFRP2 mRNA. The expression of
these miRNAs forms a positive feedback loop that optimizes Wnt
signaling to enhance osteoblast differentiation within various MSCs
(15,16). As a number of miRNAs have been
found to be dysregulated in osteoporosis or during MSC osteogenic
differentiation (17-19), it was hypothesized that there may
be more miRNAs targeting Wnt signaling antagonists to promote
osteoblast proliferation and osteogenesis.
Herein, multiple sets of online microarray
expression profiles that report the differentially expressed miRNAs
between normal bone and osteoporosis bone tissues, or between
osteogenic differentiated bone marrow-derived MSCs (BMSCs) and
non-differentiated BMSCs were downloaded and analyzed. The results
of the analysis indicated that miR-483-3p may be associated with
osteogenesis. Therefore, miR-483-3p was transfected into an
immortalized human osteoblast cell line, hFOB1.19, to generate
miR-483-3p-overexpressing cells in order to detect the specific
functions of miR-483-3p overexpression on osteoblast proliferation
and osteogenesis. Subsequently, online tools were used to search
among Wnt signaling antagonists for the direct downstream targets
of miR-483-3p, and DKK2 was predicted as the downstream target, and
the potential binding of miR-483-3p to DKK2 was then validated. The
specific role of DKK2 in osteoblast proliferation and osteogenesis
was investigated. Finally, the dynamic effects of miR-483-3p and
DKK2 were examined to investigate whether miR-483-3p exerts its
effects by targeting DKK2. The present study thus demonstrates that
the miR-483-3p/DKK2 axis modulates osteoblast proliferation and
osteogenesis through Wnt/β-catenin signaling. This axis may prove
to be a potential target for the treatment of osteoporosis.
Materials and methods
Clinical sampling
A total of 6 normal bone tissues (53.50±4.61,
female) were obtained from patients undergoing posterior lumbar
interbody fusion (PLIF) at L4/5. In addition, 6 bone tissues were
obtained from patients with osteoporosis [bone mineral density
(BMD) T score ≤-2.5, 59.00±5.48, female] undergoing PLIF at L4/5.
During the process of the surgery, part of the spinous process from
each patient was collected for tissue analysis. Osteoporosis was
diagnosed by a BMD T score of ≤-2.5 at the lumbar or femoral neck
by dual-energy X-ray absorptiometry (DXA) (20). No differences were observed in
age, BMI, serum vitamin D status, serum calcium and parathyroid
hormone (PTH) status between the controls and patients with
osteoporosis. Written informed consents were obtained from all
subjects. All clinical sampling was performed with the approval of
the Institutional Review Board and Ethics Committee of the Second
Xiangya Hospital [approval no. 2019(202)]. All experiments were
conducted in accordance with the approved guidelines.
Histopathological analysis was performed on bone tissue samples
using hematoxylin & eosin (H&E) staining and Masson's
staining following previously described methods (21).
H&E and Masson's staining of bone
sections
All bone samples were decalcified using
ethylenediamine tetraacetic acid (EDTA) for 3-5 days following 4%
formalin fixation and were then subjected to paraffin embedding,
sectioning H&E staining, and Masson's staining (Beyotime
Institute of Biotechnology, Inc.) according to previously described
methods (21). Three different
fields of vision of each section were observed using an optical
microscope (CKX53, Olympus Corporation) selected to calculate the
mean length and thickness of the trabecula in each sample. During
the decalcification process, the flexibility of all samples was
evaluated daily and the hardness was assessed by needling until all
samples were flexible and easily pierced by the needle (21).
Bioinformatics analysis and targeted gene
prediction
GEO datasets (GSE74209, GSE115197 and GSE19232) were
used for the analysis of miR-483-3p expression in
osteogenesis-related samples. The lncTar (http://www.cuilab.cn/lnctar) online tool was used to
predict the binding sites between miR-483-3p and 3′UTR of
Wnt/β-catenin signaling pathway-related genes.
Cell line and transfection
The human osteoblast 1.19 (hFOB1.19) (CRL-11372™)
immortalized human osteoblast cell line was obtained from ATCC and
cultured in a 1:1 mixture of Ham's F12 Medium and Dulbecco's
modified Eagle's medium supplemented with 2.5 mM L-glutamine
(without phenol red), 0.3 mg/ml G418, and 10% FBS (Invitrogen;
Thermo Fisher Scientific, Inc.).
The modulation of miR-348-3p expression was achieved
by transfection with 1 µg/ml miR-348-3p or anti-miR-348-3p
vector (Shanghai GenePharma Co., Ltd.). miR-NC vector or
anti-miR-NC vector was used as a control. DKK2 overexpression was
achieved by transfection with a DKK2 overexpression vector (DKK2
OE; Shanghai GenePharma Co., Ltd.). The empty vector was used as a
control. DDK2 knockdown was achieved by transfection with 20 nM
specific small interfering RNA (si-DKK2, sense,
UAUCCUUUAUGUGUCAAACTT and antisense, GUUUGACACAUAAAGGAUATT,
Shanghai GenePharma Co., Ltd.). si-NC was used as the negative
control (sense, UUCUCCGAACGUGUCACGUTT and antisense,
ACGUGACACGUUCGGAGAATT). All transfections were performed using
Lipofectamine 3000 transfection agent (Invitrogen; Thermo Fisher
Scientific, Inc.). After 48 h, the cells were harvested for use in
further experiments.
Alizarin Red staining
hFOB1.19 cells were fixed in 4% form-aldehyde
(Klinipath) for 10 min at room temperature, rinsed once with PBS,
rinsed twice with deionized water, and stained with Alizarin Red
solution (Sigma-Aldrich; Merck KGaA) for 40 min at 37°C to
visualize calcium crystals in the matrix. Images were captured
using a microscope (CKX53; Olympus Corporation) and analyzed using
ImageJ software (NIH).
Reverse transcription
quantitative-polymerase chain reaction (RT-qPCR)
Total RNA was extracted from target cells
(transfected or untransfected) using TRIzol reagent (Invitrogen;
Thermo Fisher Scientific, Inc.). DNA was eliminated by treating the
extracted RNA sample with DNase I (RNase free; Invitrogen; Thermo
Fisher Scientific, Inc.). First-strand cDNA was synthesized using
oligo(dT)20 and Superscript II reverse transcriptase (Invitrogen;
Thermo Fisher Scientific, Inc.). mRNA and miRNA expression levels
were detected using SYBR-Green PCR Master Mix (Qiagen, Inc.). The
qPCR thermocycling conditions were as follows: The initial
denaturation was first performed at 95°C for 2 min followed by
denaturation at 95°C for 15 sec and annealing and extension at 60°C
for 30 sec. The denaturation, annealing and extension were repeated
for 40 circles. The expression of GAPDH (for mRNA expression) or
RNU6B (for miRNA expression) was used as an endogenous control. The
relative expression levels were calculated using the
2−ΔΔCq method (22).
The primer sequences are listed in Table SI.
Cell counting kit-8 (CCK-8) assay for
cell viability
The transfected or untransfected target cells were
seeded in 96-well cell culture plates at a density of
1×104 cells/ml. Following 24 h of culture at 37°C, 10
µl of CCK-8 agent (03285; Sigma-Aldrich; Merck KGaA) were
added to each well to detect cell viability following a 2-h
incubation at 37°C. The optical density was determined at a
wavelength of 450 nm using a microplate reader (VICTOR Nivo;
PerkinElmer, Inc.).
5-Ethynyl-2′-deoxyuridine (EdU) assay of
DNA synthesis capacity
At 24 h following transfection, a Click-iT™ EdU Cell
Proliferation kit (C10337; Thermo Fisher Scientific, Inc.) was used
to stain transfected or untransfected cells according to the
manufacturer's instructions. Cells were then imaged and counted
under a microscope (CKX53; Japan Olympus Corporation).
Flow cytometric analysis of cell
apoptosis
Cell apoptosis was analyzed by flow cytometry using
an Annexin V-FITC/PI Apoptosis Detection kit (Beyotime Institute of
Biotechnology, Inc.) according to previously described methods
(23). Target cells (transfected
or untransfected) in each group were cultured for 24 h, digested,
and then washed 3 times with PBS. Cells were then resuspended with
Annexin V-FITC binding solution (195 µl), and Annexin V-FITC
(5 µl) was then added. Propidium iodide staining solution
(10 µl) was then added, and the cells were incubated for an
additional 10-20 min in the dark. Cell apoptosis was detected using
CellQuest software (BD Biosciences, Inc.).
Western blot analysis of protein
levels
Total proteins were extracted using RIPA lysis
buffer (Beyotime Institute of Biotechnology, Inc.) from target
cells (transfected or untransfected). The protein concentration was
determined using a bicinchoninic acid protein assay kit (Beyotime
Institute of Biotechnology, Inc.) loaded (50 µg per lane) on
10% sodium dodecyl sulfate (SDS)-polyacrylamide gels, and
transferred onto PVDF membranes (Thermo Fisher Scientific, Inc.).
The membranes were blocked for 2 h at 37°C with 5% non-fat milk in
Tris-buffered saline with Tween-20 (TBST) and then incubated
overnight at 4°C with the following primary antibodies: Ki-67
(dilution 1:1,000, 27309-1-AP; Wuhan Sanying Biotechnology),
cleaved-caspase-3 (dilution 1:1,000, ab2302; Abcam), caspase-3
(dilution 1:1,000, 19677-1-AP; Wuhan Sanying Biotechnology), WNT1
(dilution 1:1,000, 27935-1-AP; Wuhan Sanying Biotechnology),
β-catenin (dilution 1:1,000, 51067-2-AP; Wuhan Sanying
Biotechnology), cyclin D1 (dilution 1:1,000, 60186-1-Ig; Wuhan
Sanying Biotechnology), DKK2 (dilution 1:1,000, CSB-PA08279A0Rb;
CUSABIO), RANKL (dilution 1:1,000, sc-9073, Santa Cruz
Biotechnology, Inc.), OPG (dilution 1:1,000, sc-11383, Santa Cruz
Biotechnology, Inc.) and GAPDH (dilution 1:3,000, T0004; Affinity
Biosciences). The membranes were then incubated with an
HRP-conjugated secondary antibody (dilution 1:5,000, SA00001-1 or
SA00001-2, Wuhan Sanying Biotechnology) for 1 h at 37°C and then
coated with ECL luminescence reagent (Perkin-Elmer Inc.). GAPDH was
used as an internal normalization control.
Luciferase reporter assay for miR-483-3p
binding to DKK2 3′-UTR
The DKK2 3′-UTR was amplified by PCR and cloned into
downstream of the Renilla psiCHECK2 vector (Promega
Corporation), and named wt-DKK2 3′-UTR. The seed region of the DKK2
3′-UTR containing the predicted miR-483-3p binding site was mutated
to construct the mutant vector (mut-DKK2 3′-UTR). These reporter
vectors were then co-transfected into 293T cells (ATCC) with
miR-483-3p or anti-miR-483-3p and the luciferase activity was
determined at 48 h following transfection using the Dual Luciferase
Reporter assay system (Promega Corporation). Renilla
luciferase activity was normalized to Firefly luciferase activity
for each transfected well.
Data processing and statistical
analysis
Data processing and statistical analysis were
conducted using GraphPad software. Data are presented as the means
± SD, representing the results from at least 3 independent
experiments. Differences between 2 groups were analyzed for
statistical comparison using a Student′s t-test. Differences
between >2 groups were estimated for statistical comparisons
using one-way ANOVA followed by Tukey's multiple comparisons post
hoc test. Pearson's correlation coefficient analysis were used for
correlation analysis. P<0.05 was considered to indicate a
statistically significant difference.
Results
Selection and verification of
miR-483-3p
By cross-checking the data from several sets of
online microarray expression profiles, it was found that miR-483-3p
may be closely related to the osteoblast proliferation and
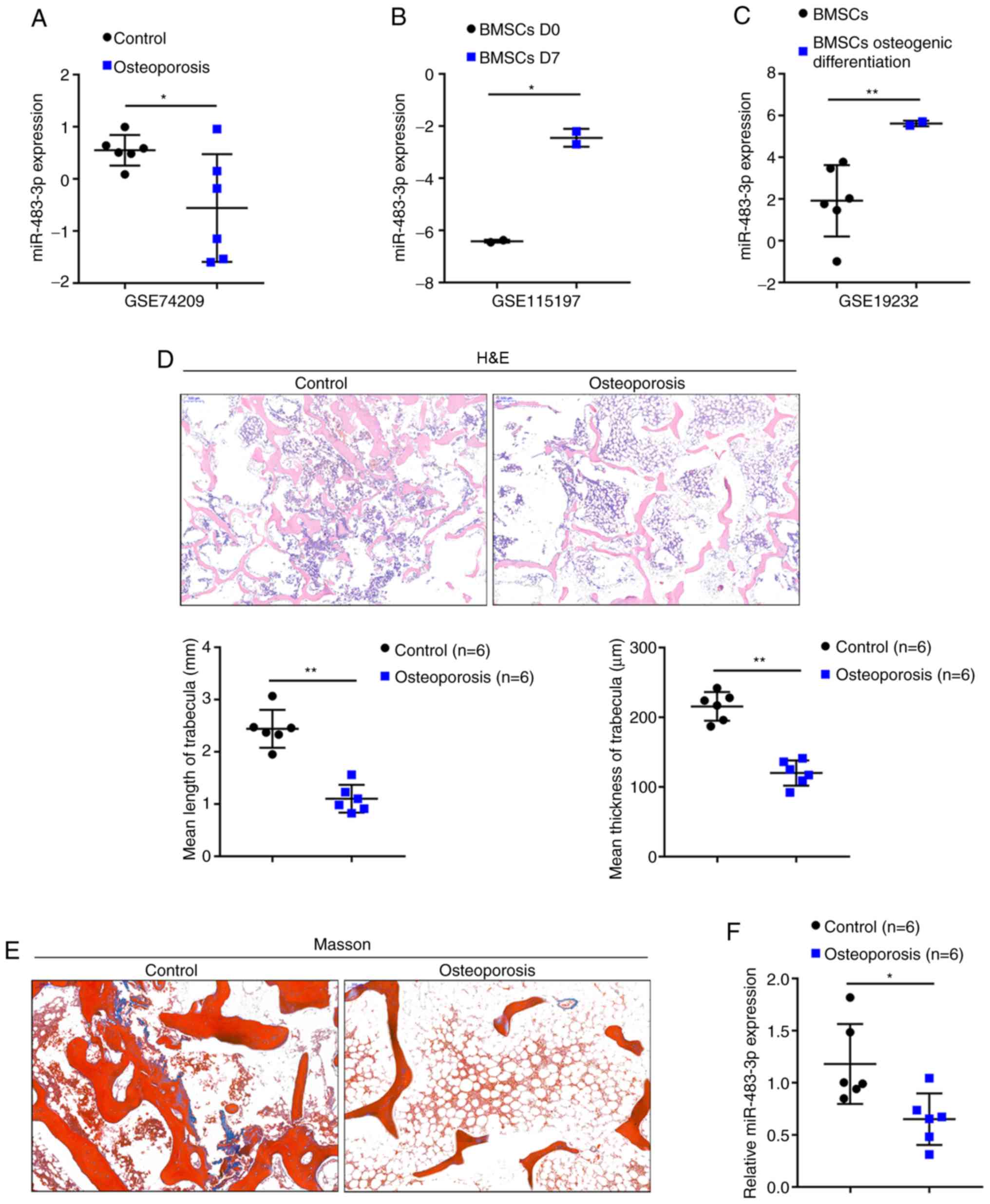
osteoblast differentiation of BMSCs. According to GSE74209, the
expression of miR-483-3p was significantly decreased in the femur
tissue samples of patients with osteoporosis compared to those in
normal bone tissue samples (Fig.
1A). According to GSE115197, the expression of miR-483-3p was
significantly increased in the BMSCs following 7 days of osteoblast
differentiation induction compared to day 0 (Fig. 1B). According to GSE19232, the
expression of miR-483-3p was significantly increased in
osteogenically differentiated BMSCs compared to undifferentiated
BMSCs (Fig. 1C). In summary, the
expression of miR-483-3p was found to be downregulated in
osteoporosis, in which osteogenesis was impaired; however,
miR-483-3p expression was upregulated in osteogenically
differentiated BMSCs.
To further examine miR-483-3p expression in
osteoporosis, osteoporosis and normal lumbar bone tissue samples
were obtained. Representative images of H&E and Masson's
staining of the osteoporosis and normal lumbar bone tissues are
shown in Fig. 1D and E. H&E
staining revealed thinner and decreased trabecular bone in the
osteoporotic bone tissues compared to the normal bone tissues
(control group) (Fig. 1D).
Moreover, the collagen in osteoporosis bone tissues was lightly
colored and was sparsely arranged compared to normal bone tissues
(control group) (Fig. 1E).
Consistent with the above-mentioned online data, miR-483-3p
expression was found to be decreased in 6 osteoporotic bone tissues
compared to 6 normal bone tissues (Fig. 1F). Taken together, these findings
suggest that miR-483-3p may contribute to the pathogenesis of
osteoporosis in a osteogenic differentiation-related manner.
miR-483-3p plays a role in hFOB1.19 cell
proliferation and osteogenic differentiation
To examine the specific effects of miR-483-3p on
osteoblasts, miR-483-3p was transfected into osteoblasts to
generate miR-483-3p-overexpressing hFOB1.19 cells and RT-qPCR was
performed to verify the transfection efficiency (Fig. 2A) and the related indexes. As
regards osteoblast proliferation, miR-483-3p overexpression
significantly promoted cell viability (Fig. 2B) and DNA synthesis capacity
(Fig. 2C); however, it inhibited
cell apoptosis (Fig. 2D). In
addition, the protein level of the proliferation marker, Ki-67, was
increased, while that of the apoptosis-related cleaved
caspase-3/caspase-3 ratio was decreased following the
overexpression of miR-483-3p (Fig.
2E). As regards osteoblast osteogenesis, miR-483-3p
overexpression significantly decreased the bone resorption-related
factor RANKL/OPG ratio (Fig. 2E);
however, it upregulated osteogenesis marker-related mRNA
expression, including alkaline phosphatase (ALP), runt-related
transcription factor 2 (RUNX2), and osteocalcin (OCN) (Fig. 2F). In addition, miR-483-3p
overexpression significantly increased the Wnt1, β-catenin and
cyclin D1 protein levels (Fig.
2E). Alizarin Red staining revealed that DKK2 knockdown
increased hFOB1.19 cell mineralization (Fig. 2G) suggesting that miR-483-3p may
enhance osteoblast proliferation and osteogenesis via Wnt/β-catenin
signaling.
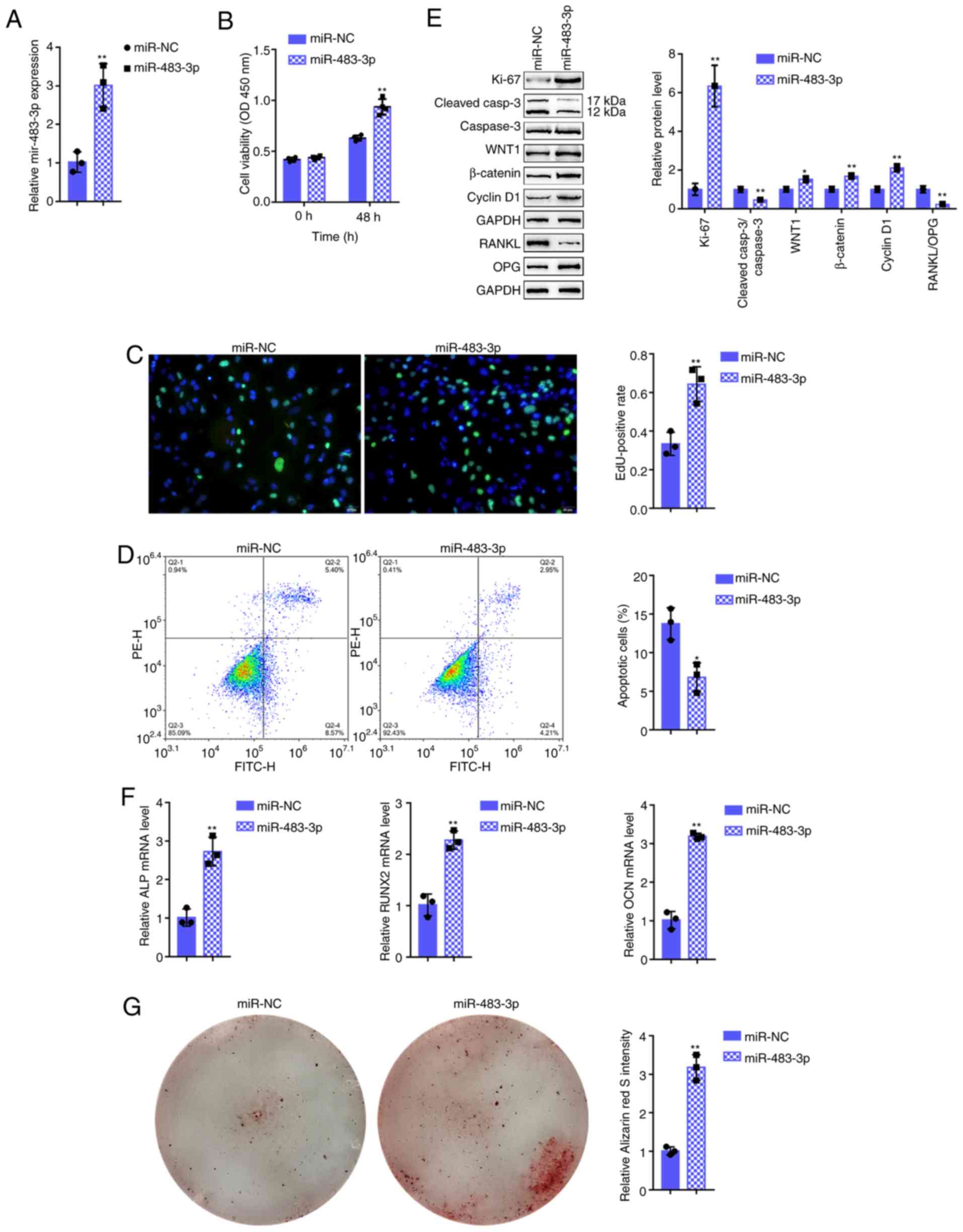 | Figure 2Effects of miR-483-3p on hFOB1.19
cell proliferation and osteogenic differentiation. (A) miR-483-3p
overexpression was generated in hFOB1.19 cells by transfection with
miR-483-3p, as confirmed by RT-qPCR. hFOB1.19 cells were
transfected with miR-483-3p and examined for (B) cell viability by
CCK-8 assay; (C) DNA synthesis capacity by EdU assay, and (D) cell
apoptosis by flow cytometric assay. (E) The protein levels of
Ki-67, cleaved-caspase-3, caspase-3, WNT1, β-catenin, cyclin D1,
RANKL and OPG were examined by western blot analysis, and (F) the
mRNA expression of osteogenesis markers, including ALP, RUNX2, OCN
and OPN was determined by RT-qPCR. (G) The mineralization of
hFOB1.19 cells was determined by Alizarin Red staining.
*P<0.05, **P<0.01. |
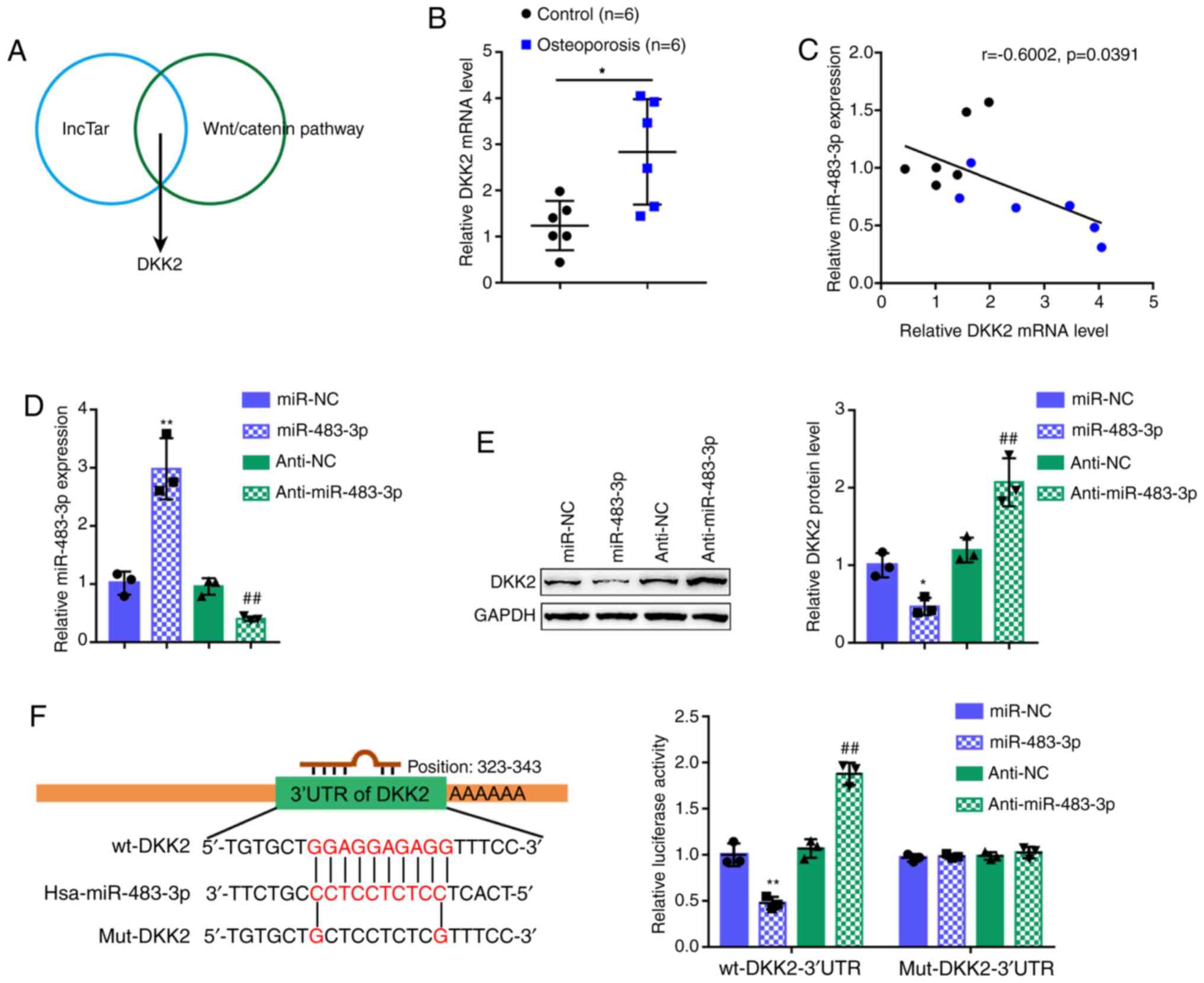
miR-483-3p directly binds to DKK2
Considering that miR-483-3p regulates Wnt1,
β-catenin and cyclin D1 protein levels, the lncTar online tool was
employed to predict the downstream targets of miR-483-3p in the
Wnt/β-catenin signaling pathway. Following a cross-check, DKK2 was
predicted by all the online tools as a direct downstream target of
miR-483-3p (Fig. 3A). Before
investigating the specific effects of DKK2 on osteoblast
proliferation and osteogenesis, DKK2 expression was first examined
in 6 normal bone tissues and 6 osteoporotic bone tissues. In
contrast to miR-483-3p expression, DKK2 expression was
significantly increased in the osteoporotic bone tissue samples
(Fig. 3B). In the tissue samples,
Pearson's correlation analysis revealed a negative correlation
between miR-483-3p and DKK2 expression (Fig. 3C). To confirm the regulation of
DKK2 by miR-483-3p, miR-483-3p/anti-miR-483-3p we transfected into
the cells to generate cells with miR-483-3p overexpression or
inhibition and RT-qPCR was then performed to verify the
transfection efficiency (Fig.
3D). Consistent with their negative correlation, miR-483-3p
overexpression decreased DDK2 protein expression, while miR-483-3p
inhibition increased DKK2 protein expression (Fig. 3E).
The putative binding of miR-483-3p to DKK2 was then
verified using a luciferase reporter assay. Two different types of
DKK2 3′-UTR reporter vectors were constructed, wild-type (wt-DKK2
3′-UTR) and mutant (mut-DKK2 3′-UTR) (Fig. 3F). These vectors were
co-transfected into 293T cells with miR-483-3p/anti-miR-483-3p, and
examined for luciferase activity. miR-483-3p overexpression
decreased wt-DKK2 3′-UTR luciferase activity, whereas miR-483-3p
inhibition increased wt-DKK2 3′-UTR luciferase activity. In the
mut-DKK2 3′-UTR group, the mutation of the predicted
miR-483-3p-binding site eliminated the miR-483-3p-induced
alterations in luciferase activity (Fig. 3F). In summary, these findings
demonstrate that miR-483-3p targets DKK2 to decrease the DKK2
protein level.
Effects of DKK2 on hFOB1.19 cell
proliferation and osteogenic differentiation
As miR-483-3p targets to inhibit DKK2, it was
hypothesized that DKK2 knockdown may play a similar role to that of
miR-483-3p overexpression in osteoblast proliferation and
osteogenesis. Therefore, si-DKK2 was transfected into the hFOB1.19
cells to generate cells in which DKK2 was knocked down and RT-qPCR
was then performed to verify the transfection efficiency (Fig. 4A). As was expected, DKK2 knockdown
significantly enhanced cell viability and DNA synthesis capacity,
while it inhibited cell apoptosis (Fig. 4B-D). Consistently, the expression
of the Ki-67 proliferation marker was increased, whereas the
cleaved-caspace3/caspase-3 apoptotic ratio was decreased by DKK2
knockdown (Fig. 4E). As regards
osteogenesis, DKK2 knockdown significantly decreased the bone
resorption-related factor, RANKL/OPG ratio (Fig. 4E), whereas it increased
osteogenesis marker mRNA expression, including ALP, RUNX2 and OCN
(Fig. 4F). Since DKK2 functions
as a Wnt/β-catenin signaling antagonist, DKK2 knockdown
significantly increased the Wnt1, β-catenin and cyclin D1 protein
levels (Fig. 4E). Alizarin Red
staining revealed that DKK2 knockdown increased hFOB1.19 cell
mineralization (Fig. 4G),
suggesting that DKK2 knockdown also promotes osteoblast
proliferation and osteogenesis through Wnt/β-catenin signaling.
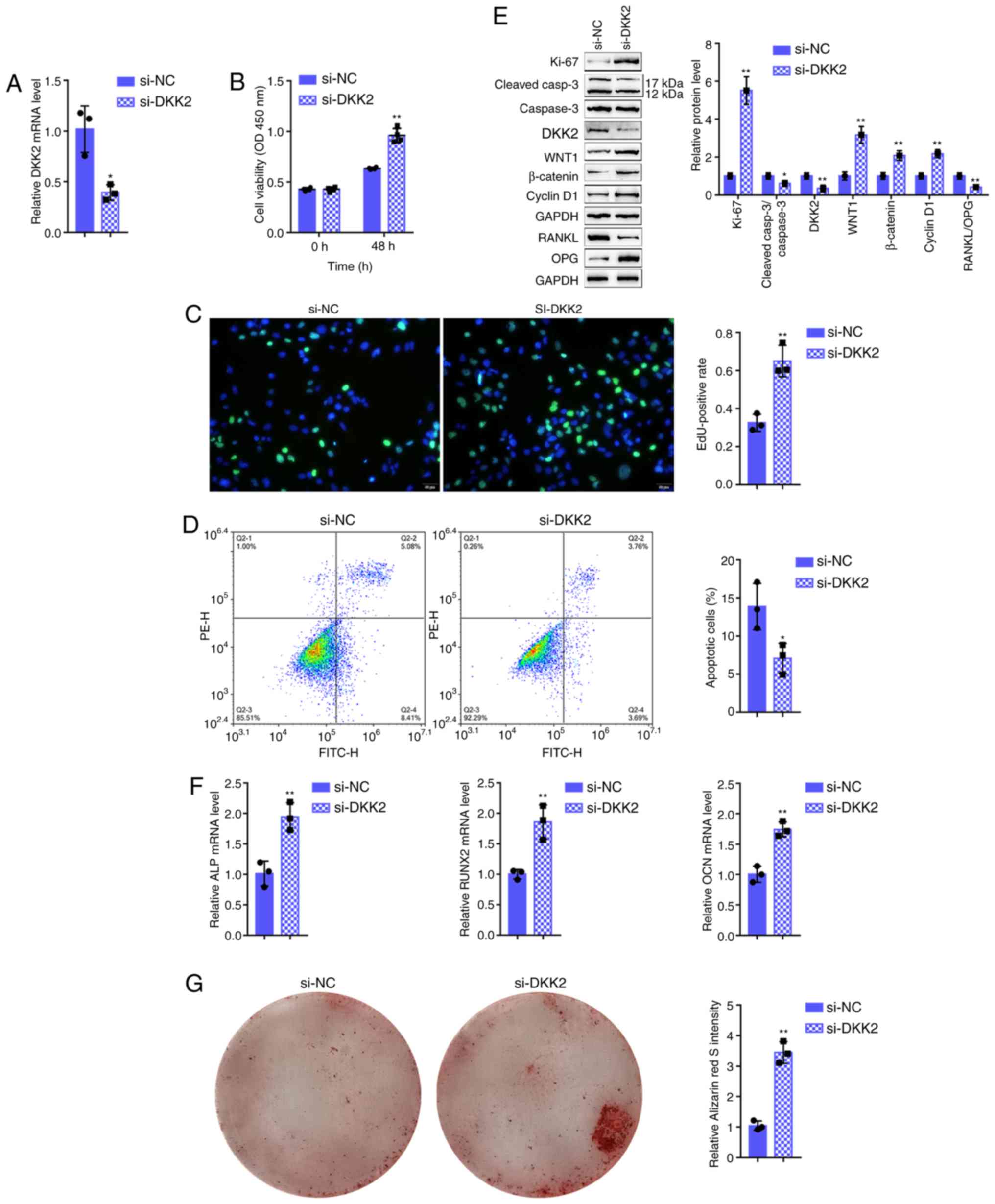 | Figure 4Effects of DKK2 on hFOB1.19 cell
proliferation and osteogenic differentiation. (A) DKK2 knockdown
was generated in hFOB1.19 cells by transfection with si-DKK2, as
confirmed by RT-qPCR. hFOB1.19 cells were transfected with si-DKK2
and examined for (B) cell viability by CCK-8 assay; (C) DNA
synthesis capacity by EdU assay, and (D) cell apoptosis by flow
cytometric assay. (E) The protein levels of Ki-67,
cleaved-caspase-3, caspase-3, WNT1, β-catenin, cyclin D1, RANKL and
OPG were determined by western blot analysis, and (F) the mRNA
expression of osteogenesis markers, including ALP, RUNX2, OCN and
OPN, was determined by RT-qPCR. (G) The mineralization of hFOB1.19
cells was determined by Alizarin Red staining.
*P<0.05, **P<0.01. |
Dynamic effects of miR-483-3p and DKK2 on
hFOB1.19 cell proliferation and osteogenic differentiation
To investigate whether miR-483-3p exerts its
functions by targeting DKK2, the dynamic effects of miR-483-3p and
DKK2 on osteoblast proliferation and osteogenesis were examined.
hFOB1.19 cells were co-transfected with miR-483-3p and DKK2 OE, and
the related indexes were determined. The effects of miR-483-3p
overexpression were similar to the above-mentioned observations
(Fig. 5A-E). DKK2 overexpression
significantly inhibited both cell viability and DNA synthesis, but
promoted cell apoptosis (Fig.
5A-C). Regard to osteogenesis, DKK2 overexpression increased
the RANKL/OPG ratio (Fig. 5D) but
decreased the mRNA levels of ALP, RUNX2, and OCN (Fig. 5E). Consistent with these cellular
functions, DKK2 overexpression decreased the Ki-67, Wnt1, β-catenin
and cyclin D1 protein levels, whereas it increased the
cleaved-caspase-3/caspase-3 ratio and DKK2 protein levels (Fig. 5D). Alizarin Red staining revealed
that miR-483-3p overexpression increased hFOB1.19 cell
mineralization, which was decreased by DDK2 overexpression
(Fig. 5F). Taken together, these
data demonstrate that DKK2 overexpression attenuates the functions
of miR-483-3p overexpression, suggesting that miR-483-3p targets
DKK2 to promote osteoblast proliferation and osteogenesis.
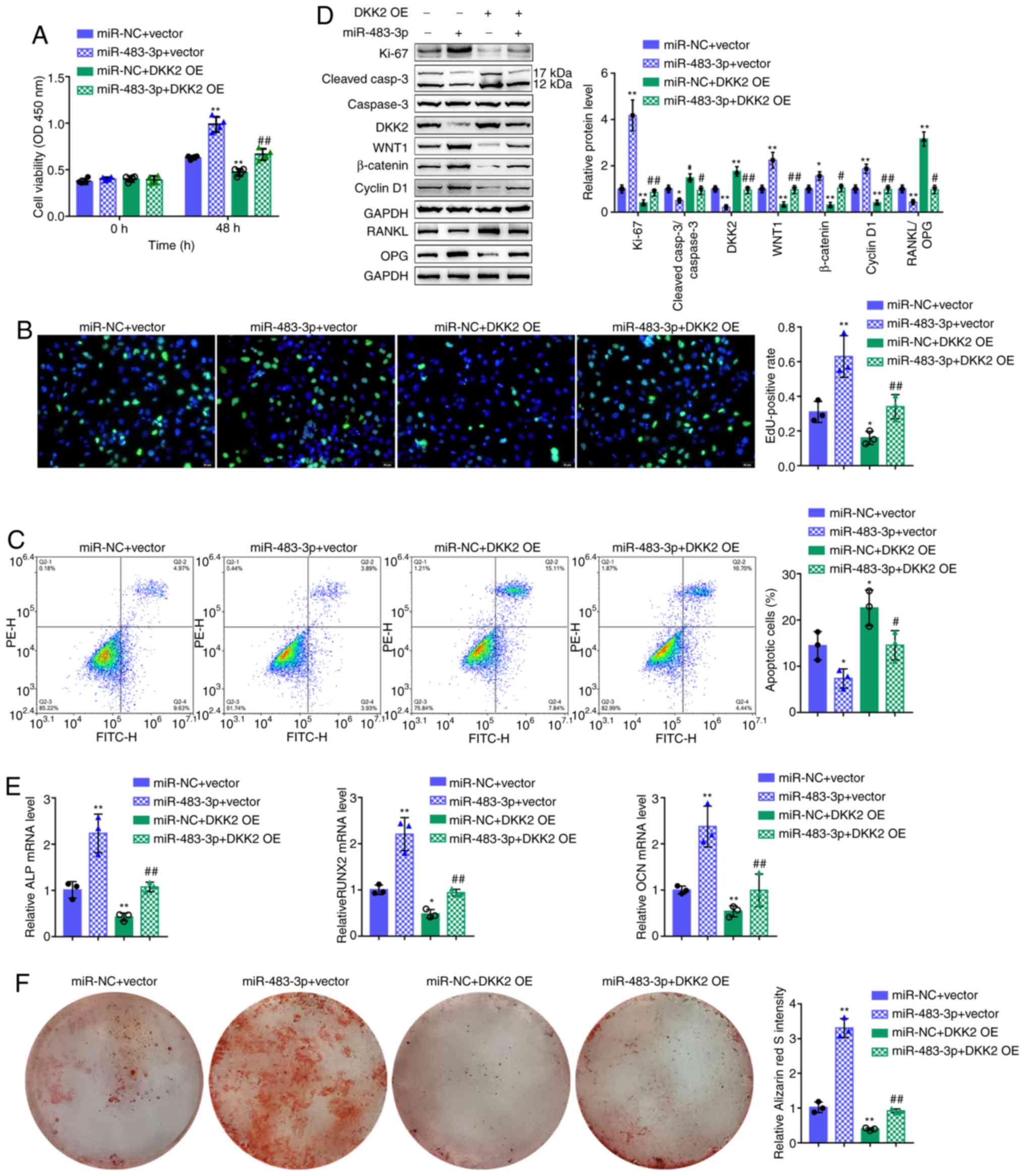 | Figure 5Dynamic effects of miR-483-3p and
DKK2 on hFOB1.19 cell proliferation and osteogenic differentiation
of hFOB1.19 cells co-transfected with miR-483-3p and DKK2 OE and
examined for (A) cell viability by CCK-8 assay; (B) DNA synthesis
capacity by EdU assay and (C) cell apoptosis by flow cytometry
assay. (D) The protein levels of Ki-67, cleaved-caspase-3,
caspase-3, DKK2, WNT1, β-catenin, cyclin D1, RANKL and OPG were
detected by western blot analysis, and (E) the mRNA expression of
osteogenesis markers, including ALP, RUNX2, OCN and OPN were
determined by RT-qPCR. (F) The mineralization of hFOB1.19 cells was
determined by Alizarin Red staining. *P<0.05,
**P<0.01, compared to the control group;
#P<0.05, ##P<0.01, compared to the
miR-NC + DKK2 OE group. |
Discussion
The present study demonstrated that miR-483-3p
expression was considerably decreased in osteoporotic bone tissue
samples. In miR-483-3p-overexpressing human osteoblasts, cell
viability, DNA synthesis capacity and osteogenesis were promoted.
Moreover, the Wnt1, β-catenin and cyclin D1 protein levels were
increased, whereas cell apoptosis was inhibited in
miR-483-3p-overexpressing human osteoblasts. The Wnt signaling
antagonist, DKK2, is a direct downstream target of miR-483-3p. In
the present study, miR-483-3p negatively regulated DKK2 protein
levels. In human osteoblasts, DKK2 knockdown exerted an effect
similar to that of miR-483-3p overexpression. DKK2 overexpression
inhibited cell viability, DNA synthesis capacity and osteogenesis.
DDK2 overexpression also decreased the Wnt1, β-catenin and cyclin
D1 protein levels, as well as the RANKL/OPG ratio in human
osteoblasts, and it promoted human osteoblast apoptosis. Thus,
these finding demonstrate that DKK2 overexpression attenuates the
functions of miR-483-3p overexpression, suggesting that miR-483-3p
exerts its effects through DKK2 and the Wnt/β-catenin pathway.
The role of miRNAs in osteogenesis has been widely
investigated. Various miRNAs, such as miR-98, miR-335, miR-433 and
miR-542, have been implicated in the promotion of osteogenesis. In
particular, miR-98 binds to SOST mRNA. leading to osteoblast
differentiation (24). miR-335
and miR-433 expression bind to Dkk1 mRNA in its 3′UTR, thus
promoting osteoblastogenesis (25,26). During the process of osteoblast
differentiation, miR-335 is highly expressed in pre-osteoblasts,
thereby promoting the response of these cells to the Wnt pathway
and enhancing osteogenesis (26).
miR-542 promotes osteogenesis by targeting sFRP1 mRNA. In rats, the
expression of miR-542, which prevents ovariectomy (OVX)-induced
osteoporosis, is decreased in post-menopausal osteoporotic
patients, suggesting that miR-542 contributes to the pathological
processes of osteoporosis (27).
Of note, it has been reported that miR-483-5p may target IGF2 to
promote osteogenic differentiation, thus indicating its involvement
in osteoporosis pathology (28).
The present study demonstrated that miR-483-3p expression was
inhibited in osteoporotic bone tissue samples, whereas it enhanced
in osteogenically differentiated BSMCs, suggesting that miR-483-3p
is involved in osteogenesis.
As was expected, miR-483-3p overexpression enhanced
cell viability and DNA synthesis capacity, but suppressed cell
apoptosis. Moreover, miR-483-3p overexpression increased the Ki-67
protein level, but decreased the cleaved-caspase-3/caspase-3 ratio.
As is well known, Ki-67 is a nuclear protein associated with and
necessary for cell proliferation. The inactivation of Ki-67 results
in the suppression of ribosomal RNA synthesis (29). Thus, Ki-67 is used as a
proliferating cell marker (30).
As regards caspase-3, it is the most important executioner caspase.
Once the apoptotic signaling events, either extrinsic (death
ligand) or intrinsic (mitochondrial), occur, caspase-3 is cleaved
(31), which kills cells
indiscriminately (32).
Therefore, the upregulation of the cleaved-caspase-3/caspase-3
ratio is a reliable marker of apoptotic cells. In the present
study, miR-483-3p overexpression increased Ki-67 expression,
whereas it decreased the cleaved-caspase-3/caspase-3 ratio, further
indicating that miR-483-3p overexpression enhances the capacity of
human osteoblasts to proliferate.
Human osteoblast proliferation, pre-osteoblast
differentiation into mature osteoblasts and new bone matrix
formation via mature osteoblasts are all critical steps of bone
formation by osteoblasts. In the present study, miR-483-3p
overexpression upregulated the mRNA expression of osteogenesis
markers, including ALP, RUNX2 and OCN, and it increased the Wnt1,
β-catenin and cyclin D1 protein levels. ALP is a typical protein
marker of osteoblast-phenotype and osteogenic differentiation, and
ALP activity is the most well-known biochemical marker of
osteoblast activity. The osteoblast phenotype is characterized by
two stages. In stage 1, ALP is detected during cell proliferation
and matrix maturation (5,33). In stage 2, matrix mineralization
occurs and late bone markers, such as OCN, are acquired. Multiple
anabolic signaling pathways, such as Wnt and RUNX2 pathways, play
positive roles in regulating bone formation (33). In addition, RANKL and OPG are two
vital factors produced by osteoblasts which modulate the maturation
and differentiation of osteoclasts, which regulate bone resorption
(34). In the present study, as
miR-483-3p overexpression increased the mRNA or protein expression
of these key factors, it was inferred that the overexpression of
miR-483-3p not only promotes osteoblast proliferation, but also
significantly promotes the differentiation of pre-osteoblasts into
mature osteoblasts and new bone matrix formation via mature
osteoblasts, thereby enhancing bone formation by osteoblasts.
As regards the molecular mechanisms, the positive
miR-483-3p regulation of Wnt1, β-catenin and cyclin D1 suggests
that miR-483-3p may target Wnt signaling antagonist(s) to exert a
promoting effect on Wnt signaling. The Wnt signaling pathway is
regulated by two main classes of modulators, which are classified
based on their crosstalk with Wnt signaling or its receptors
(35). Secreted frizzled-related
proteins (SFRPs) belong to the class that not only bind to Wnt
protein, but also antagonize Wnt signaling (36). The other class consists of the DKK
protein family, which binds to the Wnt receptor complex (37,38). In the present study, the lncTar
online tool was used to search these antagonists for the potential
direct downstream targets of miR-483-3p and DKK2 was identified as
a potential downstream target. The experimental results confirmed
the putative miR-483-3p binding to and the negative regulation of
DKK2. Through interacting with LRPs, DKKs suppress canonical Wnt
signaling (39). The elevated
expression of DKK2 has been reported to induce an abnormal
phenotype of osteoarthritic osteoblasts and poor in vitro
mineralization (40). In the
present study, consistent with previous observations (34), the knockdown of DKK2 in human
osteoblasts led to enhanced cell proliferation and enhanced
osteogenesis, while the overexpression of DKK2 led to opposite
results. More importantly, the overexpression of DKK2 significantly
attenuated the promoting effects of miR-483-3p overexpression on
osteoblast proliferation and osteogenesis, suggesting that
miR-483-3p exerts its effects by targeting DKK2.
In conclusion, these findings demonstrate that the
miR-483-3p/DKK2 axis modulates the osteoblast-mediated bone
formation process by regulating osteoblast proliferation,
pre-osteoblast differentiation into mature osteoblasts, and new
bone matrix formation via mature osteoblasts. As regards the
shortcoming of the present study, the function of the
miR-483-3p/DKK2 axis warrants further in vivo and clinical
investigations.
Supplementary Data
Acknowledgments
Not applicable.
Funding
The present study was supported by the Innovative
Research and Development project of Hunan Development and Reform
Commission (no. 2019412).
Availability of data and materials
All data generated or analyzed during this study are
included in this published article or are available from the
corresponding author on reasonable request.
Authors' contributions
BZ, KP, GW and WC were involved in the investigative
aspects of the study. BZ and FC were involved in the writing of the
manuscript. FC and YK were involved the design of the study. PL and
FC were involved in data collection and analysis. All authors have
read and approved the final manuscript.
Ethics approval and consent to
participate
Written informed consents were obtained from all
subjects. All clinical sampling was performed with the approval of
the Institutional Review Board and Ethics Committee of the Second
Xiangya Hospital [approval no. 2019(202)].
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Armas LA and Recker RR: Pathophysiology of
osteoporosis: New mechanistic insights. Endocrinol Metab Clin North
Am. 41:475–486. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Bidwell JP, Alvarez MB, Hood M Jr and
Childress P: Functional impairment of bone formation in the
pathogenesis of osteoporosis: The bone marrow regenerative
competence. Curr Osteoporos Rep. 11:117–125. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Khosla S, Melton LJ III and Riggs BL: The
unitary model for estrogen deficiency and the pathogenesis of
osteoporosis: Is a revision needed? J Bone Miner Res. 26:441–451.
2011. View
Article : Google Scholar
|
|
4
|
Xie Z and Chen Y, Gurbuz S, Zhang B, Li Y,
Bai F and Chen Y: Effects of teriparatide in Chinese and Caucasian
women with osteoporosis: Bridging study on efficacy. Clin Interv
Aging. 14:959–968. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
An J, Yang H, Zhang Q, Liu C, Zhao J,
Zhang L and Chen B: Natural products for treatment of osteoporosis:
The effects and mechanisms on promoting osteoblast-mediated bone
formation. Life Sci. 147:46–58. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Komori T: Signaling networks in
RUNX2-dependent bone development. J Cell Biochem. 112:750–755.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Guntur AR and Rosen CJ: IGF-1 regulation
of key signaling pathways in bone. Bonekey Rep. 2:4372013.
View Article : Google Scholar :
|
|
8
|
Kobayashi Y, Uehara S, Udagawa N and
Takahashi N: Regulation of bone metabolism by Wnt signals. J
Biochem. 159:387–392. 2016. View Article : Google Scholar :
|
|
9
|
Takahashi N, Maeda K, Ishihara A, Uehara S
and Kobayashi Y: Regulatory mechanism of osteoclastogenesis by
RANKL and Wnt signals. Front Biosci (Landmark Ed). 16:21–30. 2011.
View Article : Google Scholar
|
|
10
|
Landgraf P, Rusu M, Sheridan R, Sewer A,
Iovino N, Aravin A, Pfeffer S, Rice A, Kamphorst AO, Landthaler M,
et al: A mammalian microRNA expression atlas based on small RNA
library sequencing. Cell. 129:1401–1414. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Sun X, Guo Q, Wei W, Robertson S, Yuan Y
and Luo X: Current progress on MicroRNA-based gene delivery in the
treatment of osteoporosis and osteoporotic fracture. Int J
Endocrinol. 2019:67826532019. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Liu H, Liu Q, Wu XP, He HB and Fu L:
MiR-96 regulates bone metabolism by targeting osterix. Clin Exp
Pharmacol Physiol. 45:602–613. 2018. View Article : Google Scholar
|
|
13
|
Xia Z, Chen C, Chen P, Xie H and Luo X:
MicroRNAs and their roles in osteoclast differentiation. Front Med.
5:414–419. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Kapinas K, Kessler C, Ricks T, Gronowicz G
and Delany AM: MiR-29 modulates Wnt signaling in human osteoblasts
through a positive feedback loop. J Biol Chem. 285:25221–25231.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Hassan MQ, Maeda Y, Taipaleenmaki H, Zhang
W, Jafferji M, Gordon JA, Li Z, Croce CM, van Wijnen AJ, Stein JL,
et al: MiR-218 directs a Wnt signaling circuit to promote
differentiation of osteoblasts and osteomimicry of metastatic
cancer cells. J Biol Chem. 287:42084–42092. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Zhang WB, Zhong WJ and Wang L: A
signal-amplification circuit between miR-218 and Wnt/β-catenin
signal promotes human adipose tissue-derived stem cells osteogenic
differentiation. Bone. 58:59–66. 2014. View Article : Google Scholar
|
|
17
|
Xu X, Jiang H, Li X, Wu P, Liu J, Wang T,
Zhou X, Xiong J and Li W: Bioinformatics analysis on the
differentiation of bone mesenchymal stem cells into osteoblasts and
adipocytes. Mol Med Rep. 15:1571–1576. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Martin EC, Qureshi AT, Dasa V, Freitas MA,
Gimble JM and Davis TA: MicroRNA regulation of stem cell
differentiation and diseases of the bone and adipose tissue:
Perspectives on miRNA biogenesis and cellular transcriptome.
Biochimie. 124:98–111. 2016. View Article : Google Scholar
|
|
19
|
Tornero-Esteban P, Rodriguez-Rodriguez L,
Abasolo L, Tomé M, López-Romero P, Herranz E, González MA, Marco F,
Moro E, Fernández-Gutiérrez B and Lamas JR: Signature of microRNA
expression during osteogenic differentiation of bone marrow MSCs
reveals a putative role of miR-335-5p in osteoarthritis. BMC
Musculoskelet Disord. 16:1822015. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Xie F, Zhou B, Wang J, Liu T, Wu X, Fang
R, Kang Y and Dai R: Microstructural properties of trabecular bone
autografts: Comparison of men and women with and without
osteoporosis. Arch Osteoporos. 13:182018. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Lu J, Yang J, Zheng Y, Chen X and Fang S:
Extracellular vesicles from endothelial progenitor cells prevent
steroid-induced osteoporosis by suppressing the ferroptotic pathway
in mouse osteoblasts based on bioinformatics evidence. Sci Rep.
9:161302019. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar
|
|
23
|
Zhang F, Cao K, Du G, Zhang Q and Yin Z:
MiR-29a promotes osteoblast proliferation by downregulating DKK-1
expression and activating Wnt/β-catenin signaling pathway. Adv Clin
Exp Med. 28:1293–1300. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Ye W, Wang Y, Mei B, Hou S, Liu X, Wu G,
Qin L, Zhao K and Huang Q: Computational and functional
characterization of four SNPs in the SOST locus associated with
osteoporosis. Bone. 108:132–144. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Tang X, Lin J, Wang G and Lu J:
MicroRNA-433-3p promotes osteoblast differentiation through
targeting DKK1 expression. PLoS One. 12:e01798602017. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Zhang J, Tu Q, Bonewald LF, He X, Stein G,
Lian J and Chen J: Effects of miR-335-5p in modulating osteogenic
differentiation by specifically downregulating Wnt antagonist DKK1.
J Bone Miner Res. 26:1953–1963. 2011. View
Article : Google Scholar : PubMed/NCBI
|
|
27
|
Zhang X, Zhu Y, Zhang C, Liu J, Sun T, Li
D, Na Q, Xian CJ, Wang L and Teng Z: MiR-542-3p prevents
ovariectomy-induced osteoporosis in rats via targeting SFRP1. J
Cell Physiol. 233:6798–6806. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Li K, Chen S, Cai P, Chen K, Li L, Yang X,
Yi J, Luo X, Du Y and Zheng H: MiRNA-483-5p is involved in the
pathogenesis of osteoporosis by promoting osteoclast
differentiation. Mol Cell Probes. 49:1014792020. View Article : Google Scholar
|
|
29
|
Rahmanzadeh R, Huttmann G, Gerdes J and
Scholzen T: Chromophore-assisted light inactivation of pKi-67 leads
to inhibition of ribosomal RNA synthesis. Cell Prolif. 40:422–430.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Scholzen T and Gerdes J: The Ki-67
protein: From the known and the unknown. J Cell Physiol.
182:311–322. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Boatright KM and Salvesen GS: Mechanisms
of caspase-activation. Curr Opin Cell Biol. 15:725–731. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Ghavami S, Hashemi M, Ande SR, Yeganeh B,
Xiao W, Eshraghi M, Bus CJ, Kadkhoda K, Wiechec E, Halayko AJ and
Los M: Apoptosis and cancer: Mutations within caspasegenes. J Med
Genet. 46:497–510. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Franceschi RT, Ge C, Xiao G, Roca H and
Jiang D: Transcriptional regulation of osteoblasts. Ann NY Acad
Sci. 1116:196–207. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Ren H, Ren H, Li X, Yu D, Mu S, Chen Z and
Fu Q: Effects of intermedin on proliferation, apoptosis and the
expression of OPG/RANKL/M-CSF in the MC3T3-E1 osteoblast cell line.
Mol Med Rep. 12:6711–6717. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Sharma G, Sharma AR, Seo EM and Nam JS:
Genetic polymorphism in extracellular regulators of Wnt signaling
pathway. Biomed Res Int. 2015:8475292015. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Kawano Y and Kypta R: Secreted antagonists
of the Wnt signal-ling pathway. J Cell Sci. 116:2627–2634. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Caraci F, Busceti C, Biagioni F, Aronica
E, Mastroiacovo F, Cappuccio I, Battaglia G, Bruno V, Caricasole A,
Copani A and Nicoletti F: The Wnt antagonist, Dickkopf-1, as a
target for the treatment of neurodegenerative disorders. Neurochem
Res. 33:2401–2406. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Veeck J and Dahl E: Targeting the Wnt
pathway in cancer: The emerging role of Dickkopf-3. Biochim Biophys
Acta. 1825:18–28. 2012.
|
|
39
|
Zorn AM: Wnt signalling: Antagonistic
dickkopfs. Curr Biol. 11:R592–R595. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Chan TF, Couchourel D, Abed E, Delalandre
A, Duval N and Lajeunesse D: Elevated Dickkopf-2 levels contribute
to the abnormal phenotype of human osteoarthritic osteoblasts. J
Bone Miner Res. 26:1399–1410. 2011. View Article : Google Scholar : PubMed/NCBI
|