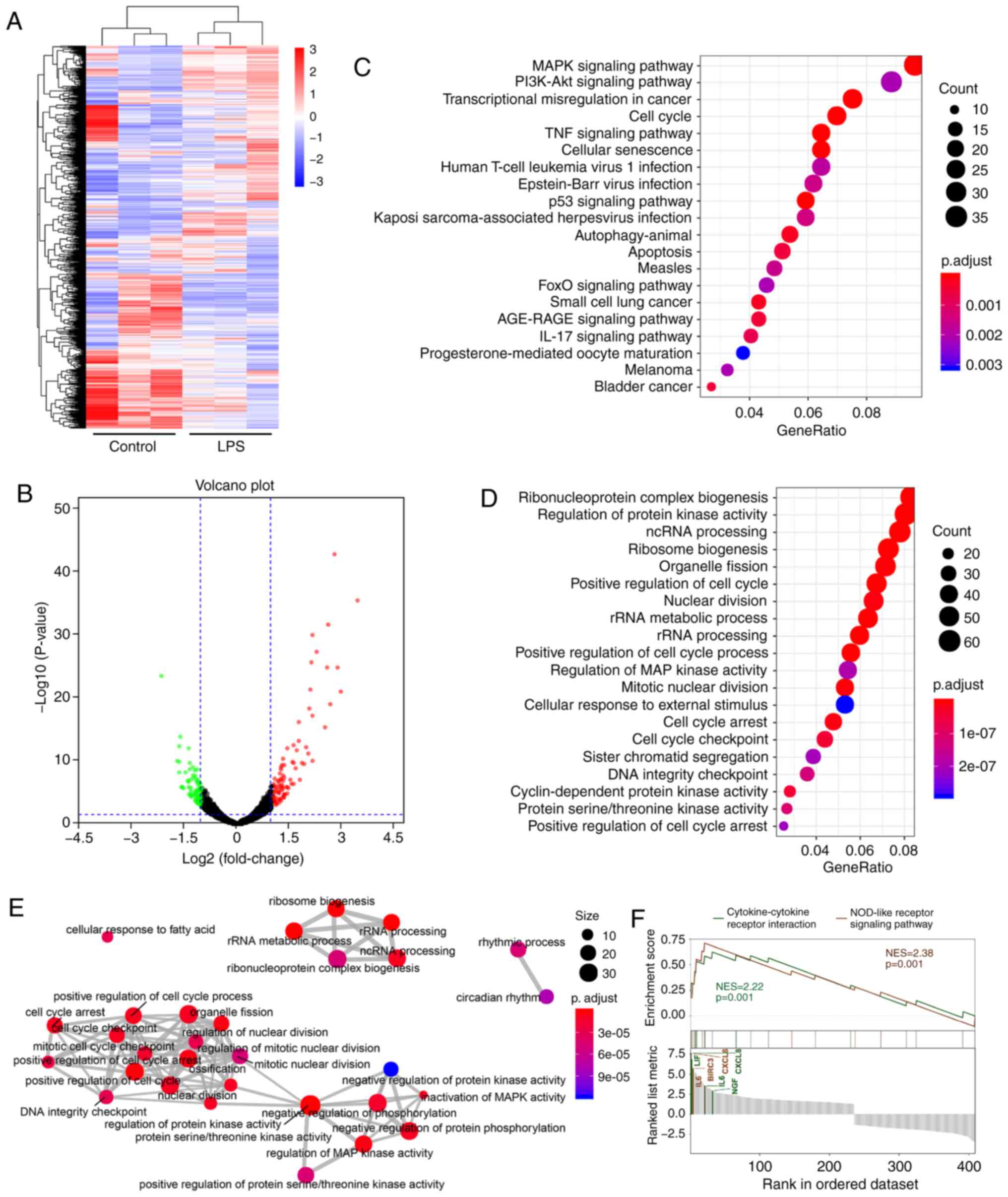
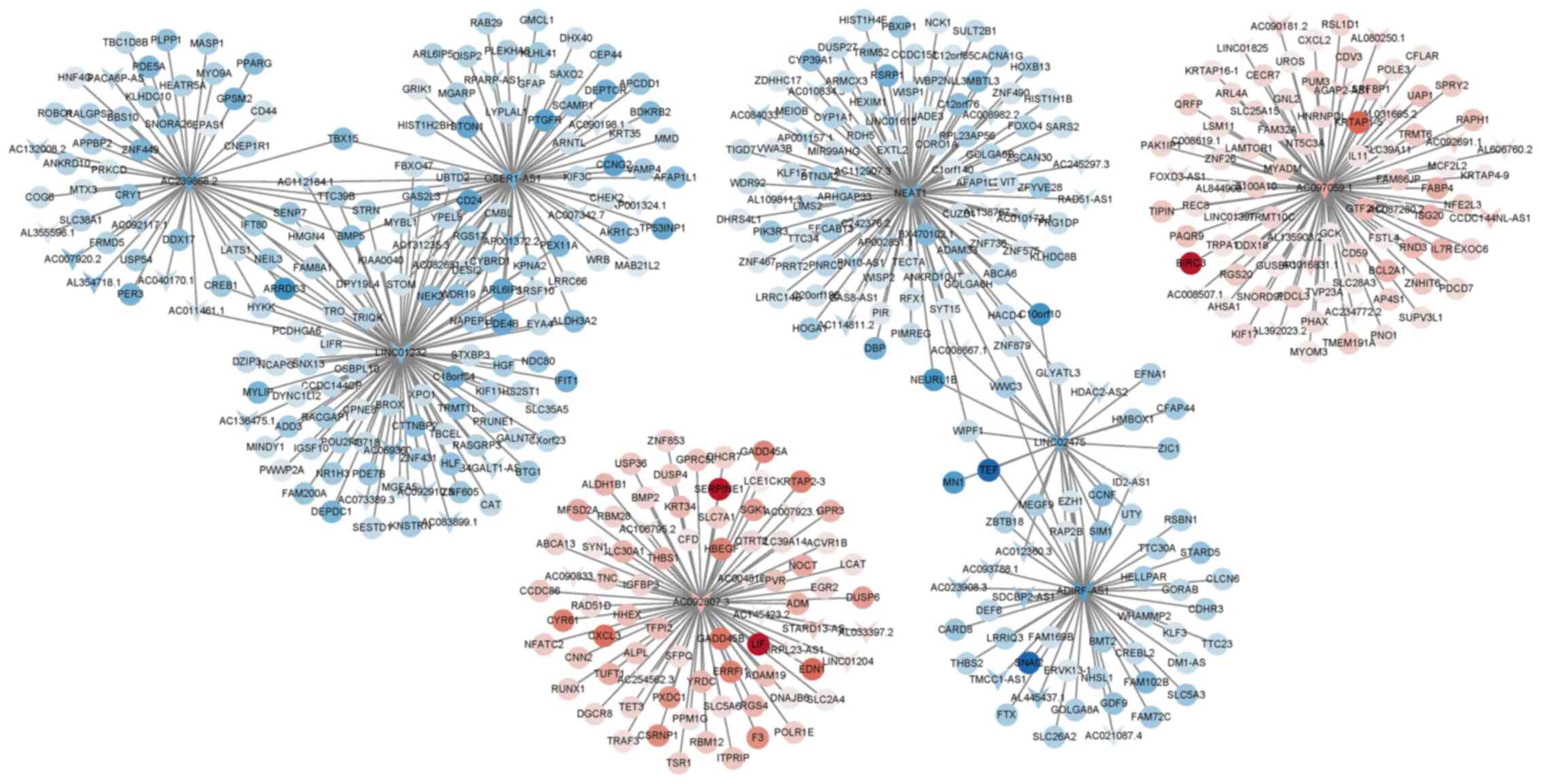
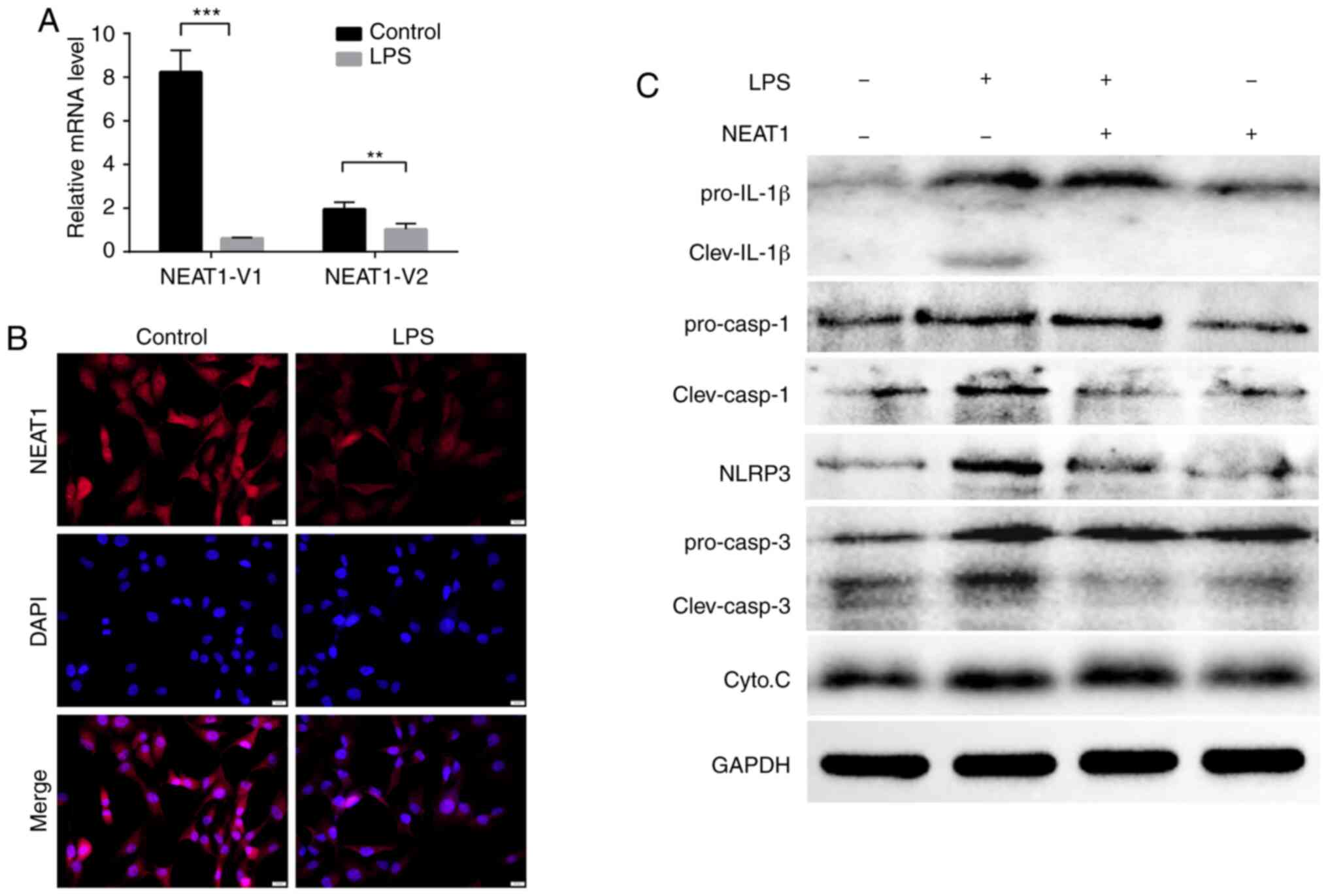
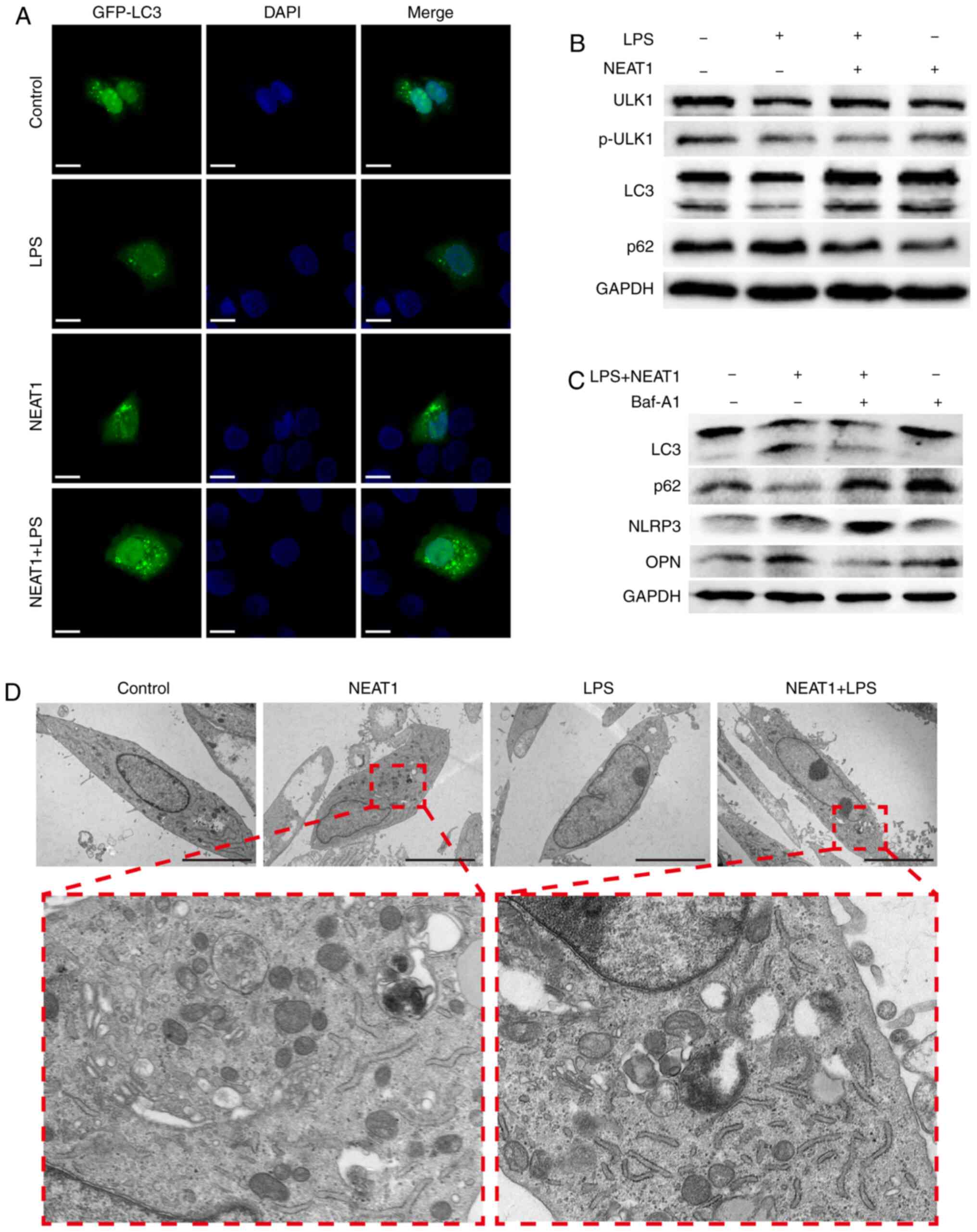
|
1
|
Redlich K and Smolen JS: Inflammatory bone
loss: Pathogenesis and therapeutic intervention. Nat Rev Drug
Discov. 11:234–250. 2012. View
Article : Google Scholar : PubMed/NCBI
|
|
2
|
Teitelbaum SL and Ross FP: Genetic
regulation of osteoclast development and function. Nat Rev Genet.
4:638–649. 2003. View
Article : Google Scholar : PubMed/NCBI
|
|
3
|
Zaidi M: Skeletal remodeling in health and
disease. Nat Med. 13:791–801. 2007. View
Article : Google Scholar : PubMed/NCBI
|
|
4
|
Matsuo K and Irie N: Osteoclast-osteoblast
communication. Arch Biochem Biophys. 473:201–209. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Karner CM and Long F: Wnt signaling and
cellular metabolism in osteoblasts. Cell Mol Life Sci.
74:1649–1657. 2017. View Article : Google Scholar :
|
|
6
|
Komori T: Regulation of proliferation,
differentiation and Functions of osteoblasts by Runx2. Histochem
Cell Biol. 20:16942019.
|
|
7
|
Abdallah BM, Jafari A, Zaher W, Qiu W and
Kassem M: Skeletal (stromal) stem cells: An update on intracellular
signaling path-ways controlling osteoblast differentiation. Bone.
70:28–36. 2015. View Article : Google Scholar
|
|
8
|
Nakagawa N, Kinosaki M, Yamaguchi K, Shima
N, Yasuda H, Yano K, Morinaga T and Higashio K: RANK is the
essential signaling receptor for osteoclast differentiation factor
in osteoclastogenesis. Biochem Biophys Res Commun. 253:395–400.
1998. View Article : Google Scholar
|
|
9
|
Asagiri M and Takayanagi H: The molecular
understanding of osteoclast differentiation. Bone. 40:251–264.
2007. View Article : Google Scholar
|
|
10
|
Kobayashi Y, Udagawa N and Takahashi N:
Action of RANKL and OPG for osteoclastogenesis. Crit Rev Eukaryot
Gene Expr. 19:61–72. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Liu C, Walter TS, Huang P, Zhang S, Zhu X,
Wu Y, Wedderburn LR, Tang P, Owens RJ, Stuart DI, et al: Structural
and functional insights of RANKL-RANK interaction and signaling. J
Immunol. 184:6910–6919. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Mundy GR: Osteoporosis and inflammation.
Nutr Rev. 65:S147–151. 2007. View Article : Google Scholar
|
|
13
|
Kadono H, Kido J, Kataoka M, Yamauchi N
and Nagata T: Inhibition of osteoblastic cell differentiation by
lipopolysaccha-ride extract from Porphyromonas gingivalis. Infect
Immun. 67:2841–2846. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Bandow K, Maeda A, Kakimoto K, Kusuyama J,
Shamoto M, Ohnishi T and Matsuguchi T: Molecular mechanisms of the
inhibitory effect of lipopolysaccharide (LPS) on osteoblast
differ-entiation. Biochem Biophys Res Commun. 402:755–761. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Daigang L, Jining Q, Jinlai L, Pengfei W,
Chuan S, Liangku H, Ding T, Zhe S, Wei W, Zhong L and Kun Z:
LPS-stimulated inflammation inhibits BMP-9-induced osteoblastic
differentiation through crosstalk between BMP/MAPK and Smad
signaling. Exp Cell Res. 341:54–60. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Huang RL, Yuan Y, Zou GM, Liu G, Tu J and
Li Q: LPS-stimulated inflammatory environment inhibits
BMP-2-induced osteoblastic differentiation through crosstalk
between TLR4/MyD88/NF-κB and BMP/Smad signaling. Stem Cells Dev.
23:277–289. 2014. View Article : Google Scholar
|
|
17
|
Yu X, Quan J, Long W, Chen H, Wang R, Guo
J, Lin X and Mai S: LL-37 inhibits LPS-induced inflammation and
stimulates the osteogenic differentiation of BMSCs via P2X7
receptor and MAPK signaling pathway. Exp Cell Res. 372:178–187.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Yao Z, Yang Z, Chen F, Jiang Y, Fu C, Wang
Y, Lu R and Wu H: Autophagy is essential for the endothelial
differentiation of breast cancer stemlike cells. Int J Mol Med.
45:255–264. 2020.
|
|
19
|
Zhang X, Yang Y, Li X, Zhang H, Gang Y and
Bai L: Alterations of autophagy in knee cartilage by treatment with
treadmill exercise in a rat osteoarthritis model. Int J Mol Med.
43:336–344. 2019.
|
|
20
|
Yang M, Feng C, Zhang Y, Liu C, Li B, Zhu
Q, Huang B and Zhou Y: Autophagy protects nucleus pulposus cells
from cyclic mechanical tensioninduced apoptosis. Int J Mol Med.
44:750–758. 2019.PubMed/NCBI
|
|
21
|
Liu J, Wang S, Zhang P, Said-Al-Naief N,
Michalek SM and Feng X: Molecular mechanism of the bifunctional
role of lipopolysaccharide in osteoclastogenesis. J Biol Chem.
284:12512–12523. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Orcel P, Feuga M, Bielakoff J and De
Vernejoul MC: Local bone injections of LPS and M-CSF increase bone
resorption by different pathways in vivo in rats. Am J Physiol.
264:E391–E397. 1993.PubMed/NCBI
|
|
23
|
Chiang CY, Kyritsis G, Graves DT and Amar
S: Interleukin-1 and tumor necrosis factor activities partially
account for calvarial bone resorption induced by local injection of
lipopolysaccharide. Infect Immun. 67:4231–4236. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Chen L, Yang Y, Bao J, Wang Z, Xia M, Dai
A, Tan J, Zhou L, Wu Y and Sun W: Autophagy negative-regulating Wnt
signaling enhanced inflammatory osteoclastogenesis from Pre-OCs in
vitro. Biomed Pharmacother. 126:1100932020. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Wang KC and Chang HY: Molecular mechanisms
of long noncoding RNAs. Mol Cell. 43:904–914. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Quinn JJ and Chang HY: Unique features of
long non-coding RNA biogenesis and function. Nat Rev Genet.
17:47–62. 2016. View Article : Google Scholar
|
|
27
|
Chen YG, Satpathy AT and Chang HY: Gene
regulation in the immune system by long noncoding RNAs. Nat
Immunol. 18:962–972. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Wang L, Wu F, Song Y, Li X, Wu Q, Duan Y
and Jin Z: Long noncoding RNA related to periodontitis interacts
with miR-182 to upregulate osteogenic differentiation in
periodontal mesenchymal stem cells of periodontitis patients. Cell
Death Dis. 7:e23272016. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Jin C, Jia L, Huang Y, Zheng Y, Du N, Liu
Y and Zhou Y: Inhibition of lncRNA MIR31HG promotes osteogenic
differentiation of human adipose-derived stem cells. Stem Cells.
34:2707–2720. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
He S, Yang S, Zhang Y, Li X, Gao D, Zhong
Y, Cao L, Ma H, Liu Y, Li G, et al: LncRNA ODIR1 inhibits
osteogenic differentiation of hUC-MSCs through the
FBXO25/H2BK120ub/H3K4me3/OSX axis. Cell Death Dis. 10:9472019.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Li L, Wang XQ, Liu XT, Guo R and Zhang RD:
Integrative analysis reveals key mRNAs and lncRNAs in monocytes of
osteoporotic patients. Math Biosci Eng. 16:5947–5971. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Clemson CM, Hutchinson JN, Sara SA,
Ensminger AW, Fox AH, Chess A and Lawrence JB: An architectural
role for a nuclear noncoding RNA: NEAT1 RNA is essential for the
structure of paraspeckles. Mol Cell. 33:717–726. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Naganuma T, Nakagawa S, Tanigawa A, Sasaki
YF, Goshima N and Hirose T: Alternative 3′-end processing of long
noncoding RNA initiates construction of nuclear paraspeckles. EMBO
J. 31:4020–4034. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Luo Y, Hao T, Zhang J, Zhang M, Sun P and
Wu L: MicroRNA-592 suppresses the malignant phenotypes of thyroid
cancer by regulating lncRNA NEAT1 and downregulating NOVA1. Int J
Mol Med. 44:1172–1182. 2019.PubMed/NCBI
|
|
35
|
Liu R, Tang A, Wang X, Chen X, Zhao L,
Xiao Z and Shen S: Inhibition of lncRNA NEAT1 suppresses the
inflammatory response in IBD by modulating the intestinal
epithelial barrier and by exosome-mediated polarization of
macrophages. Int J Mol Med. 42:2903–2913. 2018.PubMed/NCBI
|
|
36
|
Zhang F, Wu L, Qian J, Qu B, Xia S, La T,
Wu Y, Ma J, Zeng J, Guo Q, et al: Identification of the long
noncoding RNA NEAT1 as a novel inflammatory regulator acting
through MAPK pathway in human lupus. J Autoimmun. 75:96–104. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
He F, Zhang C and Huang Q: Long noncoding
RNA nuclear enriched abundant transcript 1/miRNA-124 axis
correlates with increased disease risk, elevated inflammation,
deteriorative disease condition, and predicts decreased survival of
sepsis. Medicine (Baltimore). 98:e164702019. View Article : Google Scholar
|
|
38
|
Huang Q, Huang C, Luo Y, He F and Zhang R:
Circulating lncRNA NEAT1 correlates with increased risk, elevated
severity and unfavorable prognosis in sepsis patients. Am J Emerg
Med. 36:1659–1663. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Chen DD, Hui LL, Zhang XC and Chang Q:
NEAT1 contributes to ox-LDL-induced inflammation and oxidative
stress in macro-phages through inhibiting miR-128. J Cell Biochem.
Sep 11–2018.Epub ahead of print. View Article : Google Scholar
|
|
40
|
Huang-Fu N, Cheng JS, Wang Y, Li ZW and
Wang SH: Neat1 regulates oxidized low-density lipoprotein-induced
inflammation and lipid uptake in macrophages via paraspeckle
formation. Mol Med Rep. 17:3092–3098. 2018.
|
|
41
|
Zhang P, Cao L, Zhou R, Yang X and Wu M:
The lncRNA Neat1 promotes activation of inflammasomes in
macrophages. Nat Commun. 10:14952019. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar
|
|
43
|
Joshi-Tope G, Gillespie M, Vastrik I,
D'Eustachio P, Schmidt E, de Bono B, Jassal B, Gopinath GR, Wu GR,
Matthews L, et al: Reactome: A knowledgebase of biological
pathways. Nucleic Acids Res. 33:D428–D432. 2005. View Article : Google Scholar :
|
|
44
|
Yu G, Wang LG, Han Y and He QY:
ClusterProfiler: An R package for comparing biological themes among
gene clusters. Omics. 16:284–287. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Claude-Taupin A, Bissa B, Jia J, Gu Y and
Deretic V: Role of autophagy in IL-1β export and release from
cells. Seminars in cell & developmental biology. 83:36–41.
2018. View Article : Google Scholar
|
|
46
|
Harris J, Hartman M, Roche C, Zeng SG,
O'Shea A, Sharp FA, Lambe EM, Creagh EM, Golenbock DT, Tschopp J,
et al: Autophagy controls IL-1beta secretion by targeting
pro-IL-1beta for degradation. J Biol Chem. 286:9587–9597. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Krakauer T: Inflammasomes, autophagy, and
cell death: The trinity of innate host defense against
intracellular bacteria. Mediators Inflamm. 2019:24712152019.
View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Gao C, Chen J, Fan F, Long Y, Tang S,
Jiang C, Wang J and Xu Y and Xu Y: RIPK2-mediated autophagy and
negatively regulated ROS-NLRP3 inflammasome signaling in GMCs
stimulated with high glucose. Mediators Inflamm. 2019:62075632019.
View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Chakravarty D, Sboner A, Nair SS,
Giannopoulou E, Li R, Hennig S, Mosquera JM, Pauwels J, Park K,
Kossai M, et al: The oestrogen receptor alpha-regulated lncRNA
NEAT1 is a critical modulator of prostate cancer. Nat Commun.
5:53832014. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Fu JW, Kong Y and Sun X: Long noncoding
RNA NEAT1 is an unfavorable prognostic factor and regulates
migration and invasion in gastric cancer. J Cancer Res Clin Oncol.
142:1571–1579. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Li S, Shuch BM and Gerstein MB:
Whole-genome analysis of papillary kidney cancer finds significant
noncoding alterations. PLoS Genetics. 13:e10066852017. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Ning L, Li Z, Wei D, Chen H and Yang C:
LncRNA, NEAT1 is a prognosis biomarker and regulates cancer
progression via epithelial-mesenchymal transition in clear cell
renal cell carcinoma. Cancer Biomark. 19:75–83. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Taiana E, Favasuli V, Ronchetti D,
Todoerti K, Pelizzoni F, Manzoni M, Barbieri M, Fabris S,
Silvestris I, Gallo Cantafio ME, et al: Long non-coding RNA NEAT1
targeting impairs the DNA repair machinery and triggers anti-tumor
activity in multiple myeloma. Leukemia. 34:234–244. 2020.
View Article : Google Scholar
|
|
54
|
Zeng C, Liu S, Lu S, Yu X, Lai J, Wu Y,
Chen S, Wang L, Yu Z, Luo G and Li Y: The c-Myc-regulated lncRNA
NEAT1 and paraspeckles modulate imatinib-induced apoptosis in CML
cells. Mol Cancer. 17:1302018. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Zeng C, Xu Y, Xu L, Yu X, Cheng J, Yang L,
Chen S and Li Y: Inhibition of long non-coding RNA NEAT1 impairs
myeloid differentiation in acute promyelocytic leukemia cells. BMC
Cancer. 14:6932014. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Adriaens C, Standaert L, Barra J, Latil M,
Verfaillie A, Kalev P, Boeckx B, Wijnhoven PW, Radaelli E, Vermi W,
et al: p53 induces formation of NEAT1 lncRNA-containing
paraspeckles that modulate replication stress response and
chemosensitivity. Nat Med. 22:861–868. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Mello SS, Sinow C, Raj N and Attardi LD:
Neat1 is a p53-inducible lincRNA essential for transformation
suppression. Genes Dev. 31:1095–1108. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Jen J, Tang YA, Lu YH, Lin CC, Lai WW and
Wang YC: Oct4 transcriptionally regulates the expression of long
non-coding RNAs NEAT1 and MALAT1 to promote lung cancer
progression. Mol Cancer. 16:1042017. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Zheng X, Zhang Y, Liu Y, Fang L, Li L, Sun
J, Pan Z, Xin W and Huang P: HIF-2α activated lncRNA NEAT1 promotes
hepa-tocellular carcinoma cell invasion and metastasis by affecting
the epithelial-mesenchymal transition. J Cell Biochem.
119:3247–3256. 2018. View Article : Google Scholar
|
|
60
|
Zhong J, Jiang L, Huang Z, Zhang H, Cheng
C, Liu H, He J, Wu J, Darwazeh R, Wu Y and Sun X: The long
non-coding RNA Neat1 is an important mediator of the therapeutic
effect of bexarotene on traumatic brain injury in mice. Brain Behav
Immun. 65:183–194. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Barutcu AR, Hong D, Lajoie BR, McCord RP,
van Wijnen AJ, Lian JB, Stein JL, Dekker J, Imbalzano AN and Stein
GS: RUNX1 contributes to higher-order chromatin organization and
gene regulation in breast cancer cells. Biochim Biophys Acta.
1859:1389–1397. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Lo PK, Zhang Y, Wolfson B, Gernapudi R,
Yao Y, Duru N and Zhou Q: Dysregulation of the BRCA1/long
non-coding RNA NEAT1 signaling axis contributes to breast
tumorigenesis. Oncotarget. 7:65067–65089. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Henning P, Conaway HH and Lerner UH:
Retinoid receptors in bone and their role in bone remodeling. Front
Endocrinol (Lausanne). 6:312015. View Article : Google Scholar
|
|
64
|
Menéndez-Gutiérrez MP and Ricote M: The
multi-faceted role of retinoid X receptor in bone remodeling. Cell
Mol Life Sci. 74:2135–2149. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Menéndez-Gutiérrez MP, Rőszer T, Fuentes
L, Núñez V, Escolano A, Redondo JM, De Clerck N, Metzger D,
Valledor AF and Ricote M: Retinoid X receptors orchestrate
osteoclast differentiation and postnatal bone remodeling. J Clin
Invest. 125:809–823. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Nowak B, Matuszewska A, Filipiak J,
Nikodem A, Merwid-Ląd A, Pieśniewska M, Fereniec-Gołębiewska L,
Kwiatkowska J and Szeląg A: The influence of bexarotene, a
selective agonist of the retinoid receptor X (RXR), and tazarotene,
a selective agonist of the retinoid acid receptor (RAR), on bone
metabolism in rats. Adv Med Sci. 61:85–89. 2016. View Article : Google Scholar
|
|
67
|
Wang Y, Moser AH, Shigenaga JK, Grunfeld C
and Feingold KR: Downregulation of liver X receptor-alpha in mouse
kidney and HK-2 proximal tubular cells by LPS and cytokines. J
Lipid Res. 46:2377–2387. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
68
|
Huang Y, Wang H, Hao Y, Lin H, Dong M, Ye
J, Song L, Wang Y, Li Q, Shan B, et al: Myeloid PTEN promotes
chemotherapy-induced NLRP3-inflammasome activation and antitumour
immunity. Nat Cell Biol. 22:716–727. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
69
|
He H, Jiang H, Chen Y, Ye J, Wang A, Wang
C, Liu Q, Liang G, Deng X, Jiang W and Zhou R: Oridonin is a
covalent NLRP3 inhibitor with strong anti-inflammasome activity.
Nat Commun. 9:25502018. View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Yao X, Zhang C, Xing Y, Xue G, Zhang Q,
Pan F, Wu G, Hu Y, Guo Q, Lu A, et al: Remodelling of the gut
microbiota by hyperactive NLRP3 induces regulatory T cells to
maintain homeostasis. Nat Commun. 8:18962017. View Article : Google Scholar : PubMed/NCBI
|
|
71
|
Wang X, Jiang W, Yan Y, Gong T, Han J,
Tian Z and Zhou R: RNA viruses promote activation of the NLRP3
inflammasome through a RIP1-RIP3-DRP1 signaling pathway. Nat
Immunol. 15:1126–1133. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
72
|
Hoss F, Rodriguez-Alcazar JF and Latz E:
Assembly and regulation of ASC specks. Cell Mol Life Sci.
74:1211–1229. 2017. View Article : Google Scholar
|
|
73
|
Afonina IS, Zhong Z, Karin M and Beyaert
R: Limiting inflammation-the negative regulation of NF-kappaB and
the NLRP3 inflammasome. Nat Immunol. 18:861–869. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
74
|
Kong Y, Huang T, Zhang H, Zhang Q, Ren J,
Guo X, Fan H and Liu L: The lncRNA NEAT1/miR-29b/Atg9a axis
regulates IGFBPrP1-induced autophagy and activation of mouse
hepatic stellate cells. Life Sci. 237:1169022019. View Article : Google Scholar : PubMed/NCBI
|
|
75
|
Liu F, Ai FY, Zhang DC, Tian L, Yang ZY
and Liu SJ: LncRNA NEAT1 knockdown attenuates autophagy to elevate
5-FU sensitivity in colorectal cancer via targeting miR-34a. Cancer
Med. 9:1079–1091. 2020. View Article : Google Scholar
|
|
76
|
Cao Z, Wang Y, Long Z and He G:
Interaction between autophagy and the NLRP3 inflammasome. Acta
Biochim Biophys Sin (Shanghai). 51:1087–1095. 2019. View Article : Google Scholar
|