Introduction
Myocardial infarction (MI) is one of the leading
causes of mortality worldwide. Oxidative stress and inflammation
play key roles in the development of MI (1). In ischemic tissues, reactive oxygen
species (ROS) activate the redox-sensitive signaling pathways,
damaging the cell components containing phospholipids and proteins
(2-4). Nuclear factor-κB (NF-κB) is a
complex linked to inflammatory responses and is involved in
cellular responses to a variety of stimuli such as stress, ROS,
cytokines and pathogens (5-7).
Signal-induced ubiquitylation and the subsequent degradation of
inhibitors of κB (IκBs) is initiated by the activation of IκB
kinase (IKK) (8). Once IκBs are
degraded, the NF-κB complex translocates to the nucleus to regulate
the expression of specific genes that have DNA-binding sites for
NF-κB (8). It has been
previously reported that the activation of NF-κB triggered by the
Toll-like receptor (TLR) pathway mediates early inflammation during
myocardial ischemia and reperfusion (9).
Therapy for MI is aimed at defining cardiomyocyte
death and improving prognosis. Accumulating evidence has indicated
that a number of traditional herbal drugs, such as grape seed
proanthocyanidins extract (GSPE), green tea catechins and
Scutellaria baicalensis extract (SbE) can protect cardiomyocytes
against ischemia and oxidative injury (10). Resveratrol (RES,
3,5,4′-trihydroxy-trans-stilbene) is a type of natural phenol
produced by several plants, including grapes, blueberries,
raspberries and mulberries. RES has protective potentials to
multi-targets related to cardiovascular diseases (11,12). RES is well-known for its
antioxidant, anti-inflammatory and anti-aging properties and
anti-coronavirus in vivo partially via scavenging radical
molecules (13,14). However, to date, the mechanisms
through which RES regulates endogenous antioxidants in the ischemic
myocardium remain largely unknown. Several lines of evidence have
indicated that numerous protective intracellular pathways of RES
are associated with the antioxidant response (14-16). Moreover, RES also exerts
anti-inflammatory effects in rat hearts subjected to MI and
reperfusion by inhibiting TLR4/NF-κB signaling (9). However, NF-κB promotes cell
survival and the transcription of various inflammatory cytokines,
and thus it may play both beneficial and detrimental roles in
cardiac infarct healing and post-MI remodeling. Therefore, it was
hypothesized that the cardioprotective effects of RES against
myocardial ischemic injury may be mediated via the inhibition of
NF-κB.
The present study demonstrated that the
administration of RES to mice with acute MI (AMI) and the treatment
of H9C2 cells subjected to oxygen-glucose deprivation (OGD) with
RES reduced the production of ROS and increased the activities of
cellular antioxidant systems, including superoxide dismutase (SOD),
glutathione (GSH) and glutathione peroxidase (GPx), but not
catalase (CAT). It was found that treatment with RES did not reduce
the myocardial infarct size in C57BL/6 mice, but successfully
suppressed myocardial cell apoptosis and increased left ventricular
systolic pressure (LVSP). More importantly, mice administered with
RES exhibited less production of phosphorylated NF-κB p65 (p-NF-κB
p65), phosphorylated IKK (p-IKK), interleukin (IL)-1β, IL-6, nerve
growth factor (NGF), andinsulin-like growth factor-1 (IGF-1). These
observations indicate that RES is a potential adjunctive
therapeutic drug for regulating redox homeostasis and inflammatory
responses post-MI; however, the effect of long-term administration
of RES in vivo needs to be further verified.
Materials and methods
Reagents
RES powder, nitrotetrazolium blue chloride (NBT),
2′,7′-dichlorofluorescein (DCF) and 4′,6-diamidino-2-phenylindole
(DAPI) were purchased from Sigma-Aldrich (Merck KGaA). Dulbecco's
modification of Eagle's medium (DMEM) and fetal bovine serum (FBS)
were purchased from Invitrogen (Thermo Fisher Scientific, Inc. Cell
lysis buffer for western blot analysis and immunoprecipitation, the
enhanced BCA protein assay kit, Cell Counting Kit-8 (CCK-8),
reactive oxygen species assay kit, catalase assay kit, total
glutathione assay kit, and glutathione peroxidase assay kit and
total superoxide dismutase assay kit were all purchased from the
Beyotime Institute of Biotechnology. The Annexin V-FITC apoptosis
detection kit was purchased from BD Pharmingen (BD Biosciences).
Caspase-3/7/9 antibody (cat. nos. 14220, 12827 and 9508), p-p65
(S536) antibody (cat. no. 3033), p-IKKα/β (Ser176/180) antibody
(cat. no. 2697), GAPDH antibody (cat. no. 5174), anti-rabbit IgG
(cat. no. 7074) and anti-mouse IgG (cat. no. 7076) were purchased
from Cell Signaling Technology, Inc. Antibodies to IL-1β
(sc-12742), IL-6 (sc-32296), NGF (sc-32300) and IGF-1 (sc-518040)
were purchased from Santa Cruz Biotechnology, Inc. The In
Situ Cell Death Detection kit was purchased from Roche Applied
Science. TRIzol reagent was purchased from Invitrogen (Thermo
Fisher Scientific, Inc.). The PrimeScrip RT reagent kit with gDNA
Eraser and SYBR Premix Ex Taq (Tli RNase H Plus) were purchased
from Takara Bio Inc.
Cell treatment and OGD
Rat H9C2 cardiac myoblasts purchased from the
American Type Culture Collection (ATCC) were cultured in
low-glucose DMEM supplemented with 10% FBS and maintained in a
humidified atmosphere consisting of 5% CO2 and 95% air
at 37°C. After acquiring 80% confluency, the H9C2 cells were
treated without or with 5-40 µM of RES for 24 h, and then
subjected to normoxia and OGD. After being washed 3 times with PBS,
the H9C2 cells were cultured with serum-free, low-glucose or
glucose-deprived DMEM. For OGD, the cells were placed in a 3-gas
incubator (Thermo Scientific Forma 3131; Thermo Fisher Scientific,
Inc.) at 37°C containing 1% O2, 5% CO2 and
94% N2.
Cell viability (CCK-8 assay)
Cell viability was assayed at 6-48 h following OGD
and RES treatment by CCK-8 assay. At the corresponding time point,
100 µl of CCK-8 solution were added to each well. After
being incubated at 37°C for 2 h, the OD value was detected at a 450
nm wavelength using a micro-plate spectrophotometer (Epoch; BioTek
Instruments, Inc.).
Cell apoptosis (Annexin V/PI
double-staining assay)
H9C2 cells were digested by 0.25% trypsin. The cells
(1×105) were then suspended in binding buffer. Double
staining with Annexin V-FITC/PI (BD 556547) was used to detect cell
apoptosis byflow cytometry (FCM, BD FACSCanto II).
Establishment of model of AMI
C57BL/6 mice (male, 20 g, 6-8 weeks old) were kept
under a controlled temperature (23±2°C) and humidity (50±5%) with a
12-h light/dark cycle (lights on at 7:00 a.m.) with free access to
food and water. After 2 weeks of adaptive feeding, mice were
anesthetized with 1% sodium pentobarbital (80 mg/kg) by an
intraperitoneal (i.p.) injection, and randomly assigned to 3
experimental groups as follows: i) The sham-operated (sham) group
(n=40): Silk was drilled underneath the left anterior descending
artery (LAD but not ligated, and the mice then received the vehicle
(0.9% NaCl, i.p.); ii) the AMI + vehicle group (n=50): AMI was
induced by ligating the LAD, and the successful ligation of the LAD
was evidenced by immediate regional cyanosis in the anterior
ventricular wall and the apex of the heart with color change
>40% of the left ventricle (LV) and confirmed by
electrocardiography (ECG), and the mice then received the vehicle
(0.9% NaCl, i.p.); iii) the AMI + RES group (n=50): RES (2
mg/kg/day, i.p.) was administered after the LAD ligation. On
post-operative days 1, 7 and 28, the mice were sacrificed by
decapitation under anesthesia with 1% sodium pentobarbital (80
mg/kg, i.p.), and the hearts were rapidly harvested for analysis.
The animal protocols were conducted according to the National
Institutes of Health (NIH) Guide for the care and use of animals in
laboratory experiments, and with the approval of the Institutional
Animal Care and Use Committee of Guangdong Medical University.
Assessment of infarct size
The myocardial infarct area was measured using the
NBT staining method as previously described (17). The ratio of the infarct area mass
to ventricle mass was used asa parameter for estimating the
myocardial infarct size.
Transmission electron microscopy
(TEM)
The H9C2 cells and mouse myocardium were routinely
processed by fixation in 2.5% glutaraldehyde at pH 7.2,
post-fixation in 1% OsO4 in 0.1 M cacodylate buffer, dehydration
through an ethanol series, embedding in epoxy resin, sectioning,
and post staining with uranyl acetate, and the ultra-thin sections
(70 nm) were then transferred into a copper grid for viewing under
a transmission electron microscope (JEM-1400; Jeol, Ltd.).
Terminal deoxynucleotidyl transferase
dUTP nick end labeling (TUNEL) staining assay
The mouse heart tissues were embedded in optimum
cutting temperature compound (OCT). Sections (5-µm-thick)
were prepared from the heart tissues and fixed with 4%
paraformaldehyde at room temperature for 30 min. The In Situ
Cell Death Detection kit (Roche Applied Science) was used for TUNEL
staining. Briefly, the heart sections were incubated in a 0.1%
Triton X-100 solution for 2 min on ice and then incubated with a
TUNEL reaction mixture at 37°C for 30 min. Finally, the sections
were incubated with 1 µg/ml DAPI at 37°C for 15 min and
observed using a laser scanning confocal microscope (TCS SP5 II;
Leica Microsystems GmbH).
The TUNEL-positive cells and total cells in 5
viewing fields of the heart sections were counted under a 10X
objective microscope lens.
Detection of LVSP
On post-operative days 7 and 28, the mice were
anesthetized with 1% sodium pentobarbital (80 mg/kg, i.p.). A
catheter was inserted into the carotid artery and advanced into the
LV, and LVSP was recorded using an acquisition and analysis system
(BL-420F; Chengdu Technology and Market Co., Ltd.).
Redox-system assay
The concentrations of total GSH, as well as the
activities of total SOD, GPx and CAT were measured using respective
kits. All assays were performed following the manufacturer's
protocols. The levels of ROS in the myocardium were determined
using a previously described method (18). Briefly, the reaction mixture (1
ml) containing Locke's buffer (pH 7.4), 0.2 ml homogenate of
myocardium (0.5 mg protein) and 10 µl of DCFH-DA (5 mM) was
incubated for 45 min at room temperature, and then measured using a
spectrofluorometer (EnSpire; PerkinElmer) with excitation at 484 nm
and emission at 530 nm. Finally, ROS production was measured from a
DCF-standard curve.
Reverse transcritpion-quantitative PCR
(RT-qPCR)
Total RNA was extracted from the myocardium using
TRIzol reagent following the manufacturer's protocol. The RNA
concentration was determined using UV spectrophotometry and
integrity was monitored by electrophoresis. The quantified total
RNA was used for reverse transcription to synthesize the first
strand cDNA using the PrimeScript RT reagent kit with a gDNA Eraser
(Perfect Real Time) kit; and qPCR was performed according to the
specifications of the SYBR Premix Ex Taq II (Tli RNase H Plus) kit
using the LightCycler 480 Real-Time PCR detection System (Roche
Diagnostics). In brief, the cycling conditions were as follows: A
pre-run at 95°C for 10 sec, 40 cycles of denaturation at 95°C for 5
sec, followed by a 60°C annealing for 25 sec and a 72°C extension
for 15 sec; the melting curve conditions were as follows: 1 Cycle
of denaturation at 95°C for 10 sec, then from 65°C change to 95°C
with a temperature change rate of 0.5°C/sec. GAPDH was used as
reference to normalize mRNA expression levels of target genes. The
qPCR reactions were performed in triplicate. Relative expression
levels of mRNAs are expressed as
2Cq(calibrator)−Cq(test) (19). The primers used for RT-qPCR are
listed in Table I.
 | Table ISequences of primers used for
RT-qPCR. |
Table I
Sequences of primers used for
RT-qPCR.
| Gene | Primer
sequences |
|---|
| GAPDH | Forward:
5′-TGCCCCCATGTTTGTGATG-3′ |
| Reverse:
5′-TGTGGTCATGAGCCCTTCC-3′ |
| IL-1β | Forward:
5′-CAAATCTCGCAGCAGCACATC-3′ |
| Reverse:
5′-TGTCCTCATCCTGGAAGGTC-3′ |
| IL-6 | Forward:
5′-GAAGTTCCTCTCTGCAAGA-3 |
| Reverse:
5′-ACAGGTCTGTTGGGAGTG-3′ |
| NGF | Forward:
5′-CTGGACTAAACTTCAGCATTC-3′ |
| Reverse:
5′-GTCTGGGGTCTACAGTGATG-3′ |
| IGF-1 | Forward:
5′-GATGCTCTTCAGTTCGTGTGTG-3′ |
| Reverse:
5′-TCTCCAGTCTCCTCAGATCAC-3′ |
Western blot analysis
Mouse myocardium was homogenized in liquid nitrogen,
and tissues were then lysed with cell lysis buffer for western blot
analysis and immunoprecipitation. The protein concentration was
determined using the BCA method. Briefly, proteins (10 µg)
were denatured and separated by 10% SDS-PAGE gels and
electroblotted onto PVDF membranes. The membranes were blocked with
TBS containing 5% skimmed milk and 0.1% Tween-20 for 1 h at room
temperature. After washing 3 times, the membranes were incubated
with anti-IL-1β (1:200), anti-IL-6 (1:200), anti-NGF (1:400),
anti-IGF-1 (1:400), anti-p-NF-κB p65 (1:1,000) and anti-GAPDH
(1:1,000) antibodies for at least 16 h at 4°C. Anti-rabbit/mouse
IgG (1:2,000) was incubated with the membranes at room temperature
for 1 h as the secondary antibody. Immunoreactions were detected
using an electrochemiluminescence (ECL) system according to the
manufacturer's instructions. The gray value was calculated using
ImageJ analysis software (National Institutes of Health). All
protein expression levels were normalized to GAPDH.
Statistical analysis
Data were analyzed using a Student's t-test or
one-way analysis of variance (ANOVA) and post-hoc analysis for
significance was performed with the Bonferroni method using
GraphPad Prism 5.0 (GraphPad Software, Inc.). Data are expressed as
the means ± standard error of means (SEM) and a P-value <0.05
was considered to indicate a statistically significant difference,
as previously described (20).
Results
RES promotes H9C2 cell survival and
reduces apoptosis
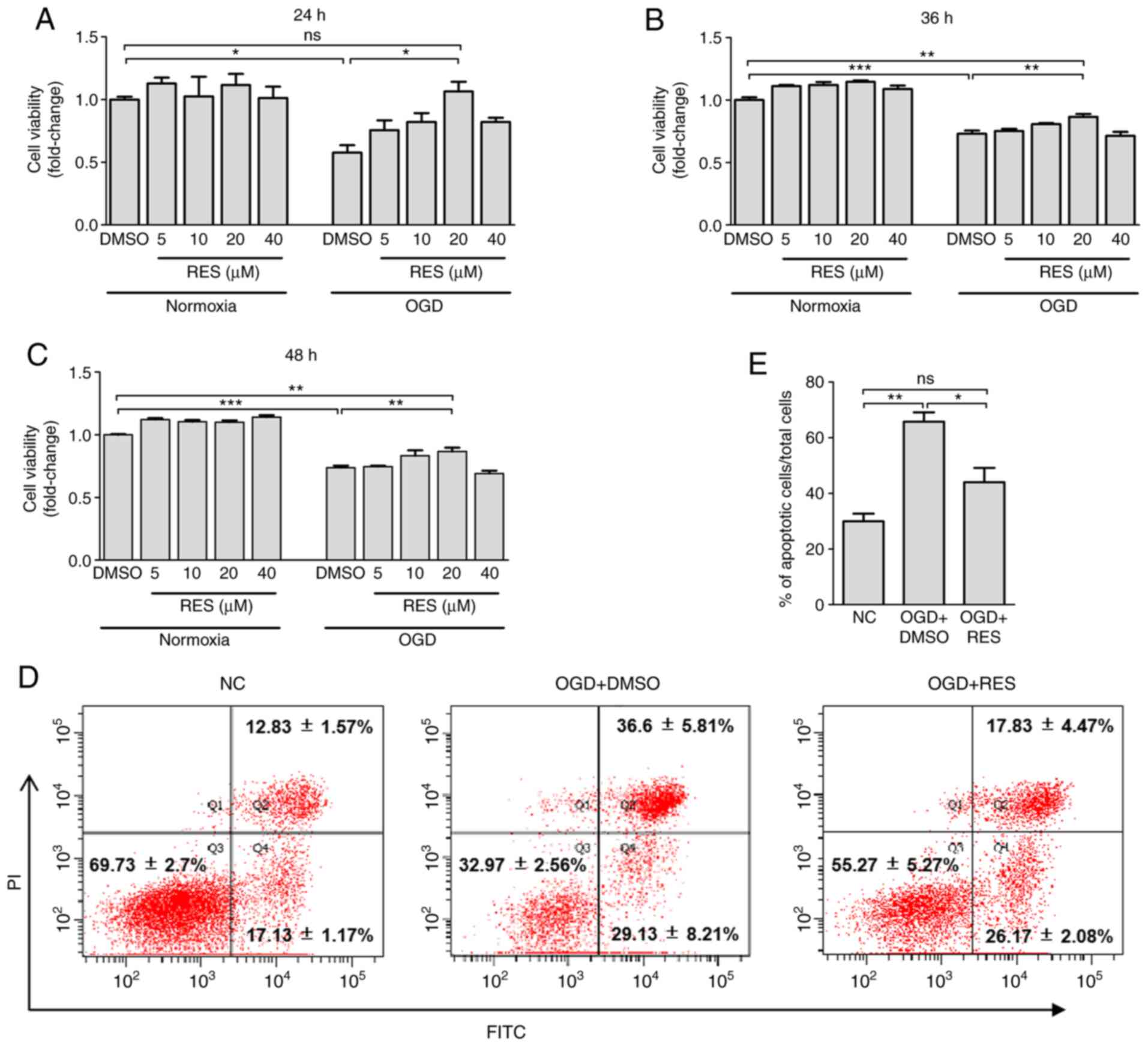
The results of CCK-8 assay revealed that exposure to
OGD for 24-48 h incurred a 26-42% decrease in cell viability
(Fig. 1A-C). Relative to
treatment with DMSO, 20 µM RES significantly increased cell
viability by 84.4, 18.6 and 17.5%, respectively at 24, 36 and 48 h
post-OGD, while RES at 5 and 40 µM failed to significantly
improve cell survival (Fig.
1A-C). However, compared with the normal control (NC) group,
non-significant differences in cell viability were observed at 6
and 12 h post-OGD (data not shown). Additionally, as shown by
Annexin-V/PI double-staining assay (Fig. 1D and E), exposure to OGD for 24 h
significantly increased the numbers of Annexin
V+/PI+ (late-stage apoptotic) cells. RES
significantly decreased OGD-induced cell apoptosis by 33.1%. Taken
together, these findings provide strong evidence that RES can
protect H9C2 cells against hypoxic injury.
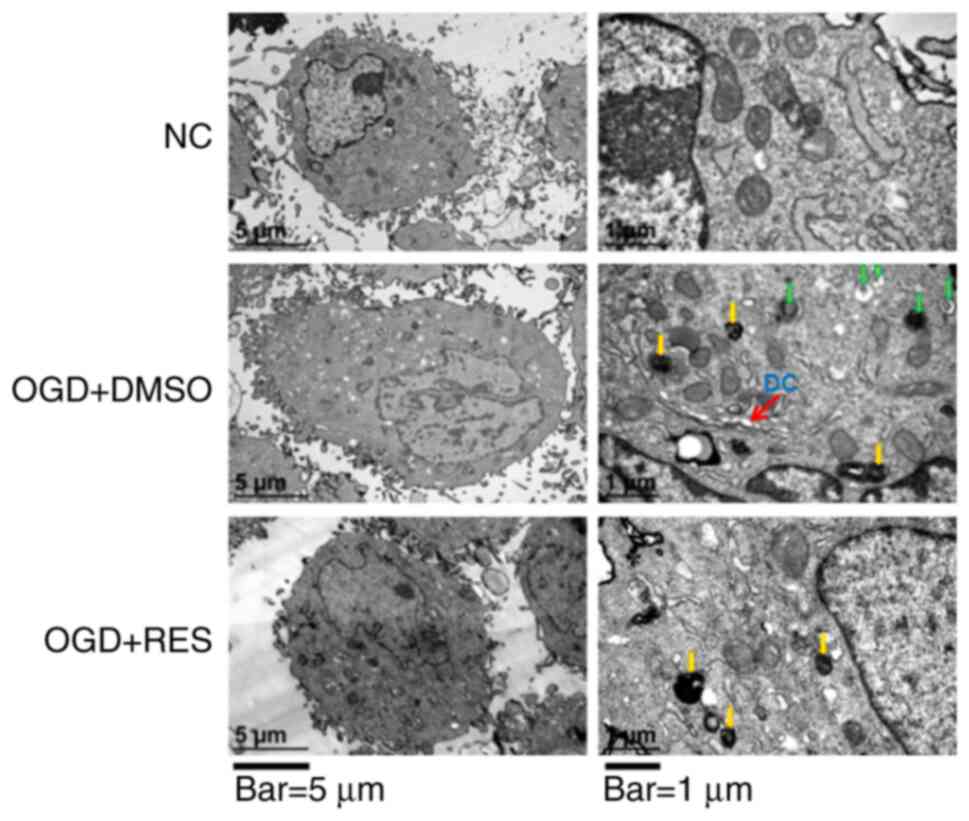
RES attenuates ultrastructural hypoxic
changes of H9C2 cells
Electron microscopy examination revealed that the
ultrastructure of the NC H9C2 cells was intact. Following exposure
to OGD for 24 h, the cell membrane became protruded and foamed, the
nuclear membrane cracked, and the nucleolus disappeared in the H9C2
cells (Fig. 2). In the OGD +
DMSO group, the cytoplasm was retracted, containing dilated
cisternae of endoplasmic reticulum, primary lysosomes and
autophagic vesicles (Fig. 2).
Treatment of the cells with RES (20 µM) reduced OGD-induced
cell damage, as the cell membrane and nuclear membrane appeared to
be intact, autophagic vesicles were reduced and the areas of the
cytoplasm became more spacious (Fig.
2). These findings indicate that RES protects cell against
ultrastructural damage induced during the earlystages of
hypoxia.
RES inhibits hypoxia-induced oxidative
stress in H9C2 cells
The effects of RES on ROS production were measured
by DCFH fluorescence. It was found that ROS production by the H9C2
cells peaked 24 h following exposure to OGD, and RES decreased ROS
generation in a concentration-dependent manner (Fig. 3A). The redox-regulation effect of
RES on the antioxidant system in H9C2 cells was also investigated.
Compared to the NC group, the level of GSH and the activities of
SOD, GPx and CAT in the OGD + DMSO group were significantly
decreased. Compared to the OGD + DMSO group, RES increased the
activities of SOD and GPx (Fig. 3B
and C), but had no effect on CAT activities and GSH contents
(Fig. 3D and E). These findings
suggest that RES inhibits hypoxia-mediated oxidative stress by
scavenging ROS and enhancing endogenous antioxidant systems.
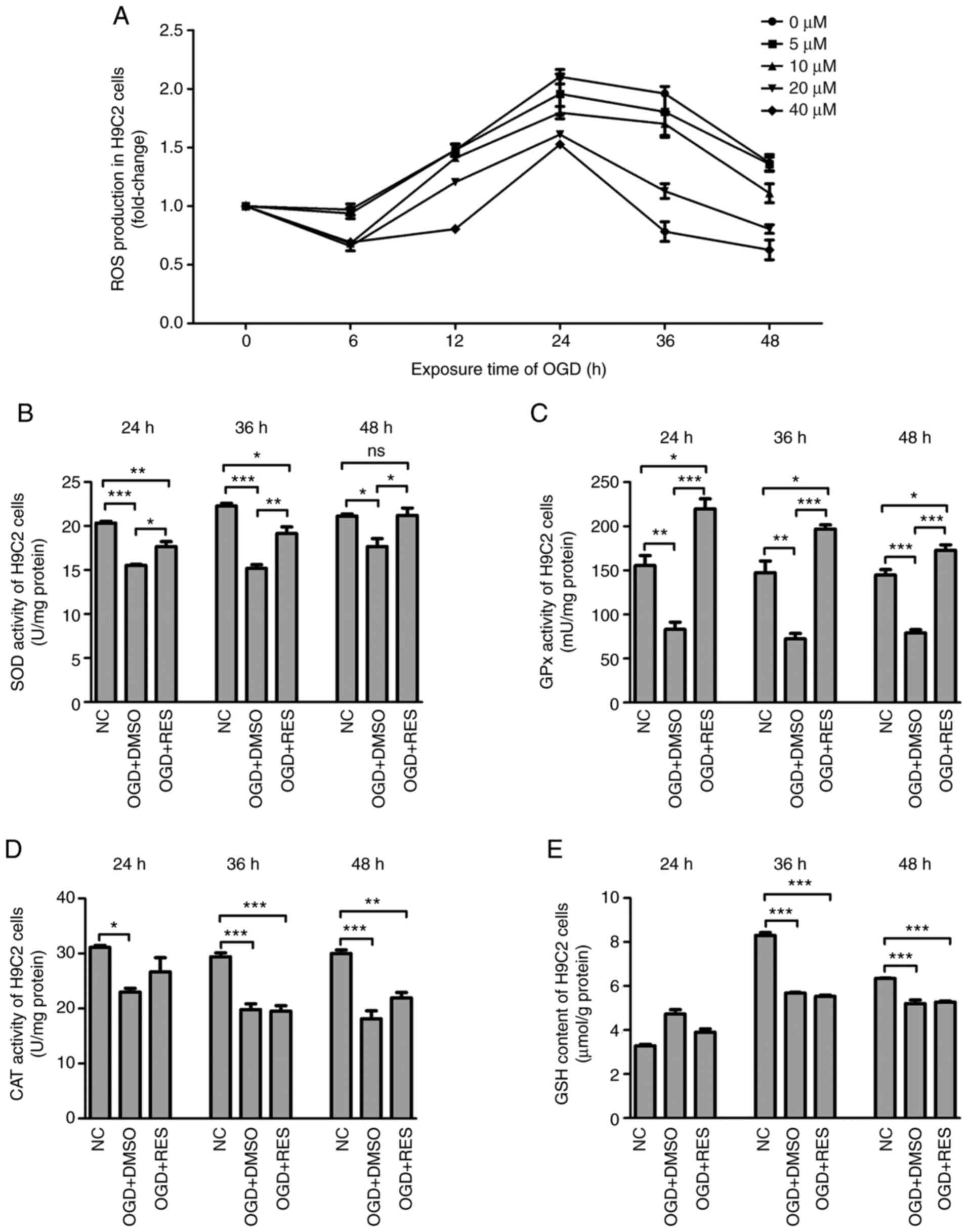 | Figure 3Effects of RES on redox-system of
H9C2 cells. (A) ROS production, (B) SOD activities, (C) GPx
activities, (D) CAT activities and (E) GSH contents were measured
in the H9C2 cells exposed to OGD at different time points. Data are
expressed as the means ± SEM (n=3 per group).
*P<0.05, **P<0.01,
***P<0.001; ns, not significant. RES, resveratrol;
OGD, oxygen-glucose deprivation; ROS, reactive oxygen species; SOD,
superoxide dismutase; GPx, glutathione peroxidase. |
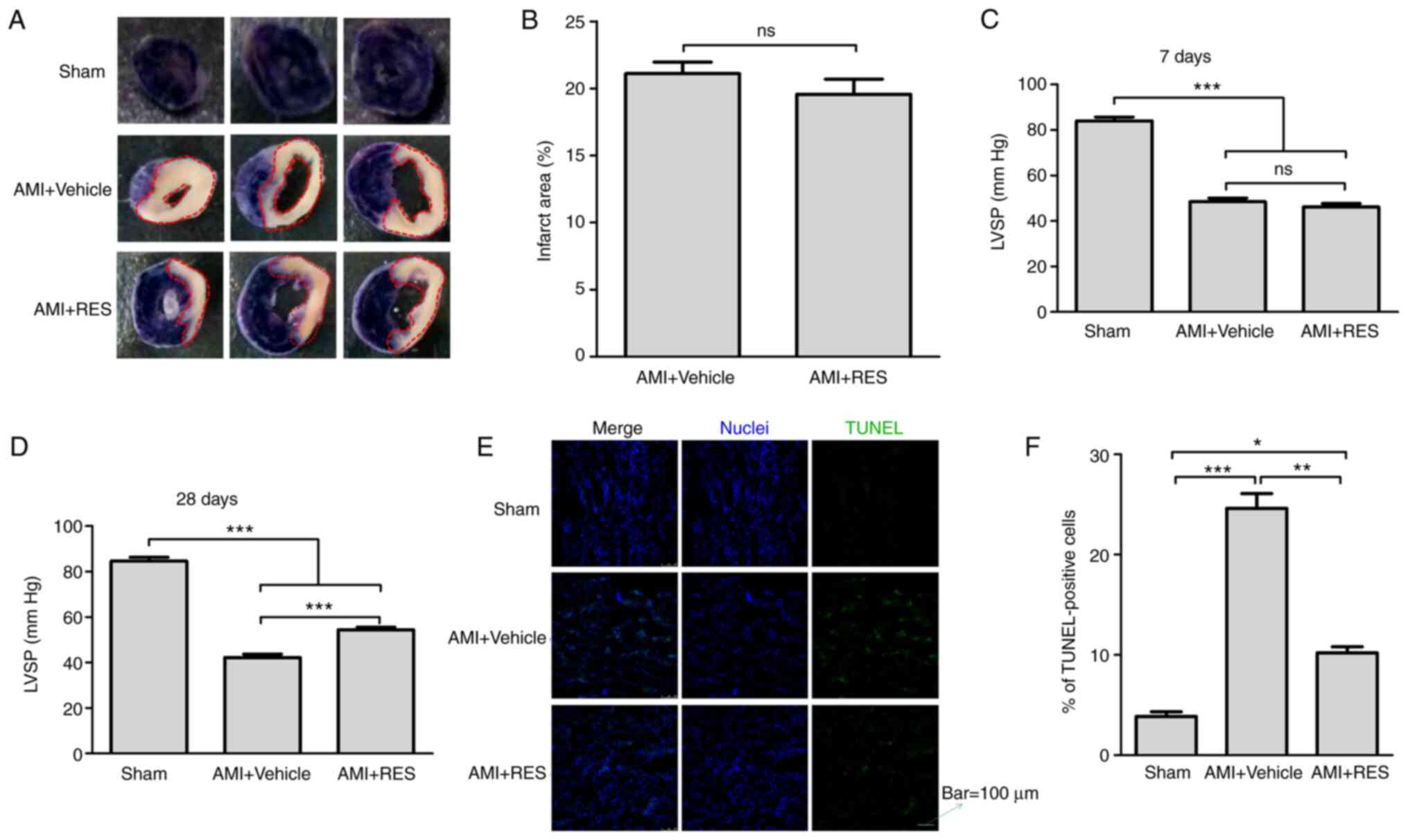
RES does not reduce the myocardial
infarct size, but prevents myocardial apoptosis and increases
LVSP
The myocardial infarct size was determined using the
NBT staining method (Fig. 4A).
It was found that the mice with AMI had an infarct area mass of
21.1±0.86% relative to the ventricle mass; however, RES did not
decrease the infarct size (Fig.
4B). In addition, the effect of RES on LVSP was examined in
order to evaluate the cardiac systolic function post-AMI. As shown
in Fig. 4C and D, significant
decreases in LVSP were detected in the AMI + vehicle group compared
with the sham group (48.5±1.6 vs. 84±1.7 mmHg at 7 days after
infarction; 42.2±1.5 vs. 84.6±1.6 mmHg at 28 days after infarction;
P<0.001). At 7 days after AMI, no differences were observed in
LVSP between the AMI + RES and AMI + vehicle groups (46.1±1.5 vs.
48.5±1.6 mmHg; P>0.05), while at 28 days after AMI, the LVSP in
the AMI + RES group (54.4±1.1 mmHg) was significantly higher than
that in the AMI + vehicle group (42.2±1.5 mmHg; P<0.001).
Myocardial apoptosis was detected by TUNEL staining.
As shown in Fig. 4E, a few
apoptotic cells were observed in the sham group. However, the
number of apoptotic cells in the AMI+DMSO and AMI + RES groups was
greater than that in the sham group. The percentage of apoptotic
cells was significantly decreased compared with the AMI + vehicle
group (10.3±0.6 vs. 24.6±1.5%; P<0.01; Fig. 4F).
RES attenuates ultrastructural damage due
to ischemic injury in the mouse myocardium
In the AMI + vehicle group, the dissolution of
myocardial myofilaments, disorganization of substantial sarcomere
Z-lines, myofibrillar disarray, dilated cisternae of the
endoplasmic reticulum, primary lysosomes and autophagic vesicles
were observed (Fig. 5). These
ultra-structural changes were not observed in the myocardium of the
NC and RES-treated AMI mice at 7 days post-infarction (Fig. 5). Collectively, RES alleviated
ultrastructural ischemic damage and improved myocardial
autophagy.
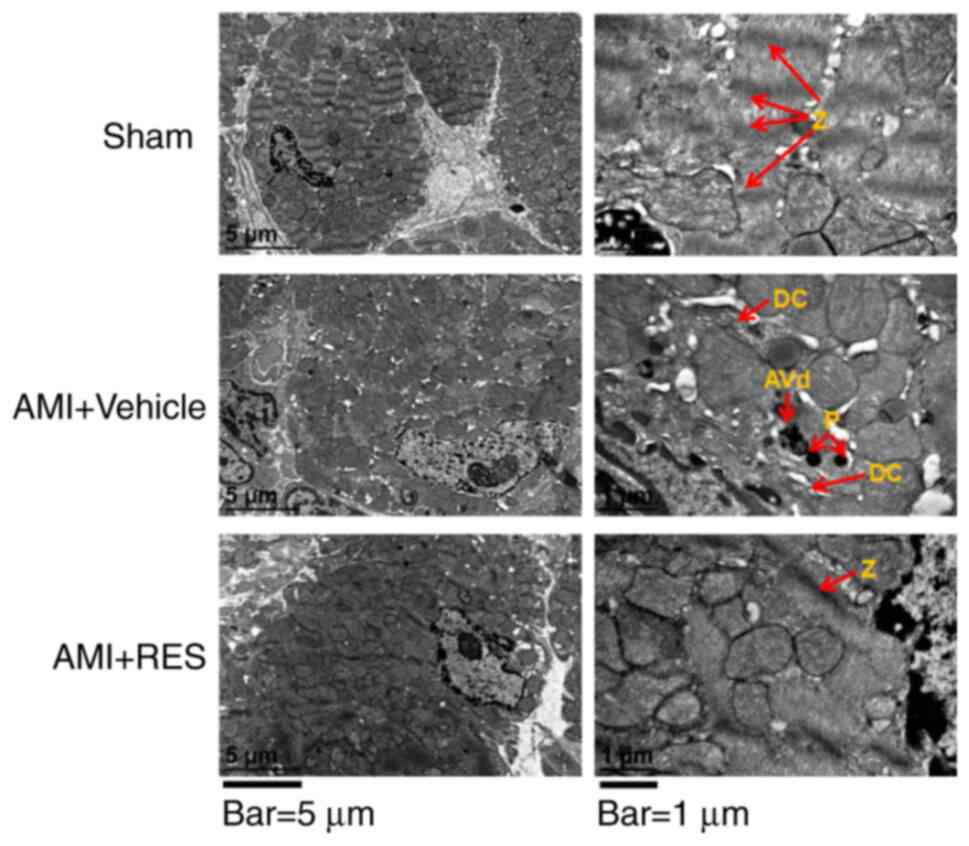 | Figure 5TEM images showing the
ultrastructural findings in mouse myocardium. Red arrows labeled
with the following indicate: Z, Z lines; DC, dilated cisternae of
endoplasmic reticulum; P, primary lysosome; AVd, degradative
autophagosomes. Scale bars, 5 µm (left panels) and 1
µm (right panels). Images taken using 2 different
magnifications, ×8,000 and ×30,000. RES, resveratrol; AMI, acute
myocardial infarction. |
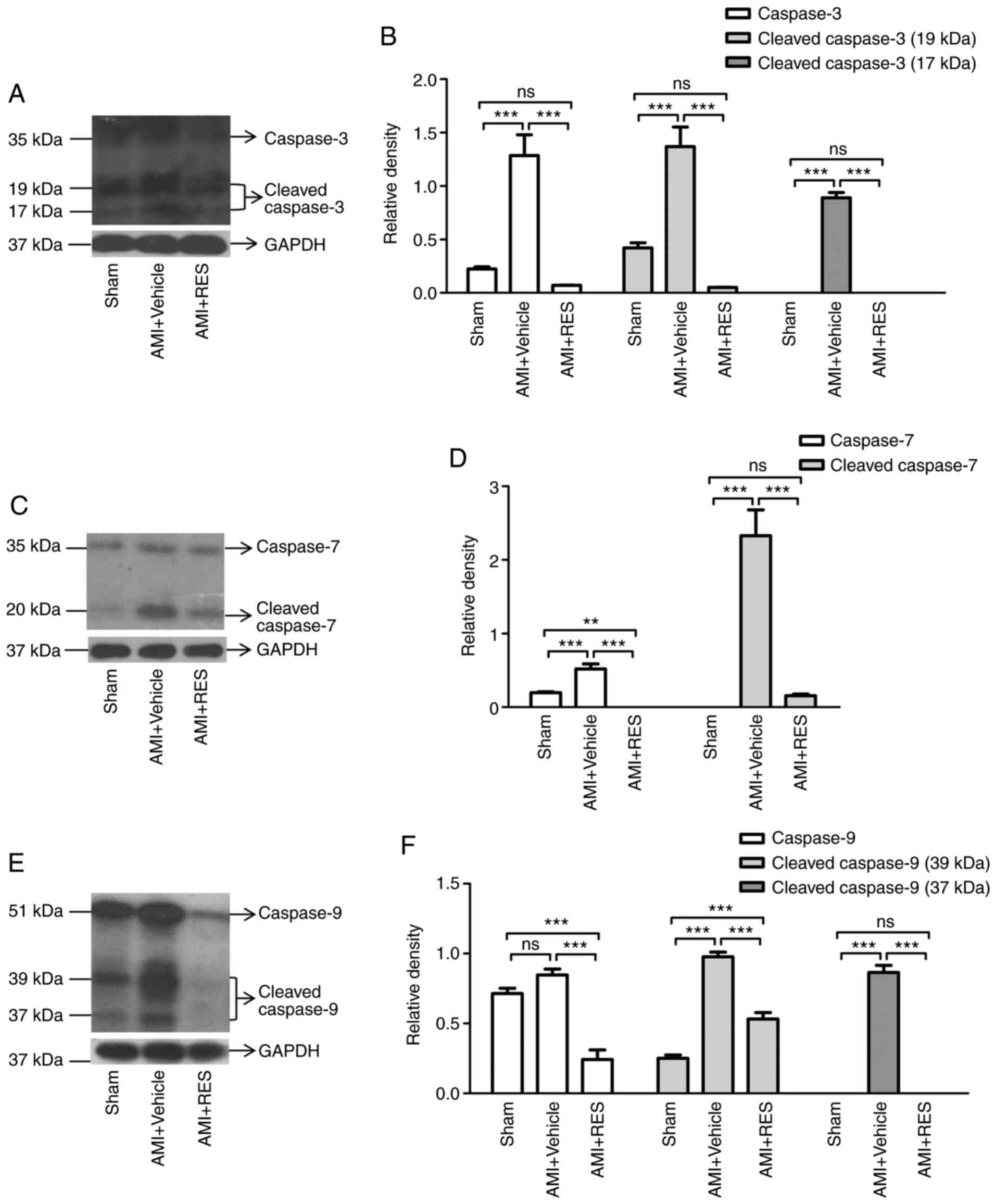
RES attenuates ischemia-induced
caspase-3/7/9 activation in the mouse myocardium
To further confirm whether RES attenuates
post-ischemic myocardial apoptosis, western blot analysis was
performed to detect the expression and activation of caspase-3/7/9.
It was found that the levels of procaspase-3/7/9 and cleaved
caspase-3/7/9 were significantly increased in the AMI + vehicle
group at 7 days after AMI (Fig.
6). Compared with the AMI + vehicle group, RES treatment
significantly decreased the levels of procaspase-3/7/9 and cleaved
caspase-3/7/9 (Fig. 6).
RES inhibits ischemia-induced oxidative
stress in the mouse myocardium
As shown in Fig.
7A, ROS generation in the AMI + vehicle group was upregulated
at 7 and 28 days after AMI. The administration of RES resulted in
significantly decreased ROS levels, which were even lower than
those of the sham group. As shown in Fig. 7B-D, the levels of GSH, and the
activities of SOD and GPx decreased post-AMI and RES treatment
reversed these effects. These findings further confirmed the
antioxidant properties of RES.
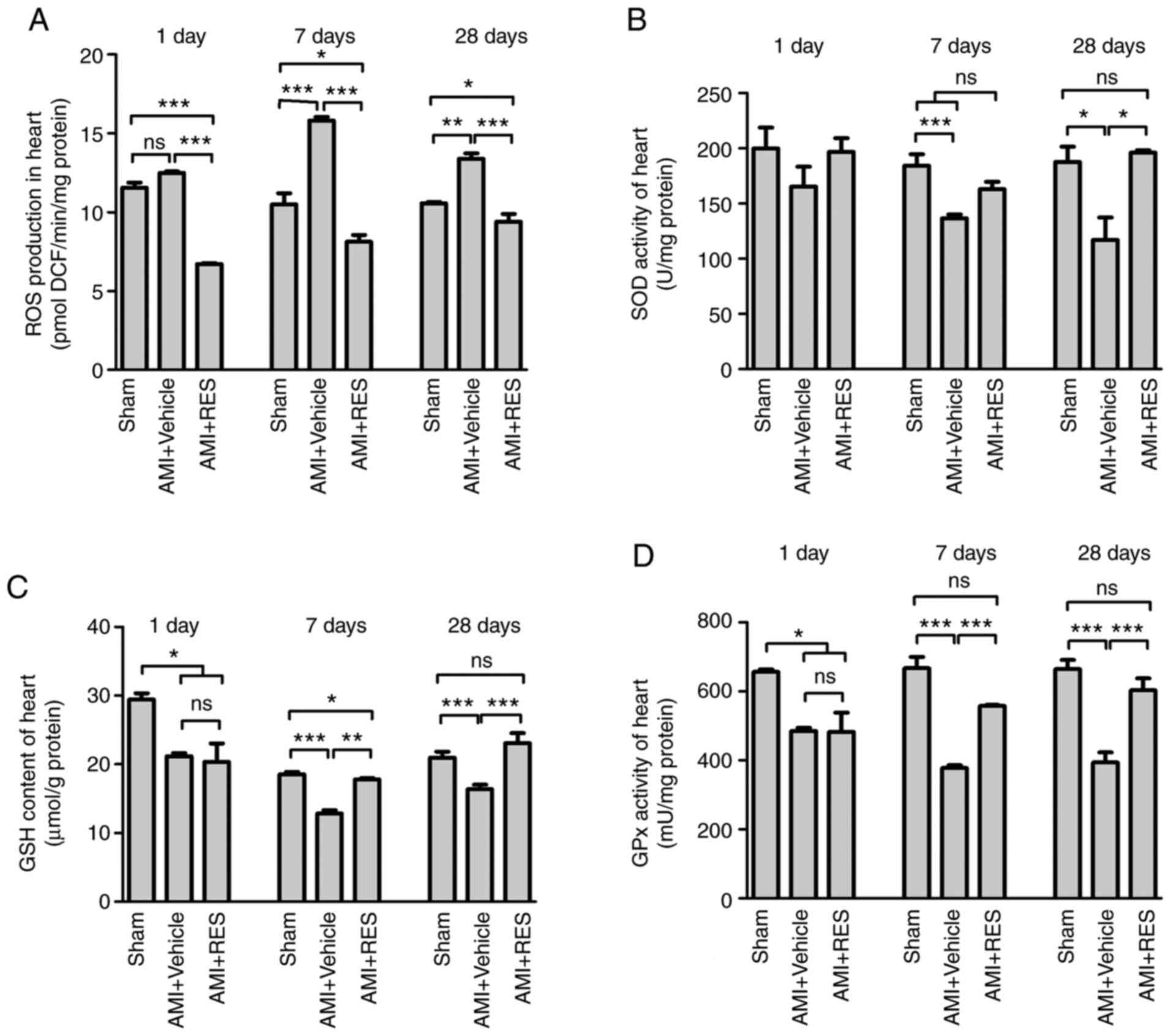 | Figure 7Effects of RES on the redox-system of
the mouse myocardium. (A) ROS production, (B) SOD activity, (C) GSH
content and (D) GPx activity were measured in the mouse myocardium
at 1, 7 and 28 days post-infarction. Data are expressed as the
means ± SEM (n=3 per group). *P<0.05,
**P<0.01, ***P<0.001; ns, not
significant. RES, resveratrol; AMI, acute myocardial infarction;
ROS, reactive oxygen species; SOD, superoxide dismutase; GPx,
glutathione peroxidase; GSH, glutathione. |
RES regulates NF-κB-dependent
inflammation
The effects of RES on NF-κB activation and
NF-κB-dependent pro-inflammatory pathways were investigated by
western blot analysis and RT-qPCR. No significant differences in
NF-κB-dependent pro-inflammatory pathways were observed between the
sham, AMI + vehicle and AMI + RES groups at 1 day after infarction
(data not shown). AMI upregulated the mRNA levels of IL-1β, IL-6,
NGF and IGF-1 at 7 and 28 days post-AMI (Fig. 8); however, RES suppressed the
mRNA levels of all these peptide factors, apart from NGF. The
results of western blot analysis revealed that RES significantly
reversed the ischemia-induced elevation of p-IKKα/β, p-NF-κB p65,
IL-1β, IL-6, NGF and IGF-1 levels at 7 and 28 days after AMI
(Fig. 9). These results imply
that RES is an important regulator of NF-κB-dependent
inflammation.
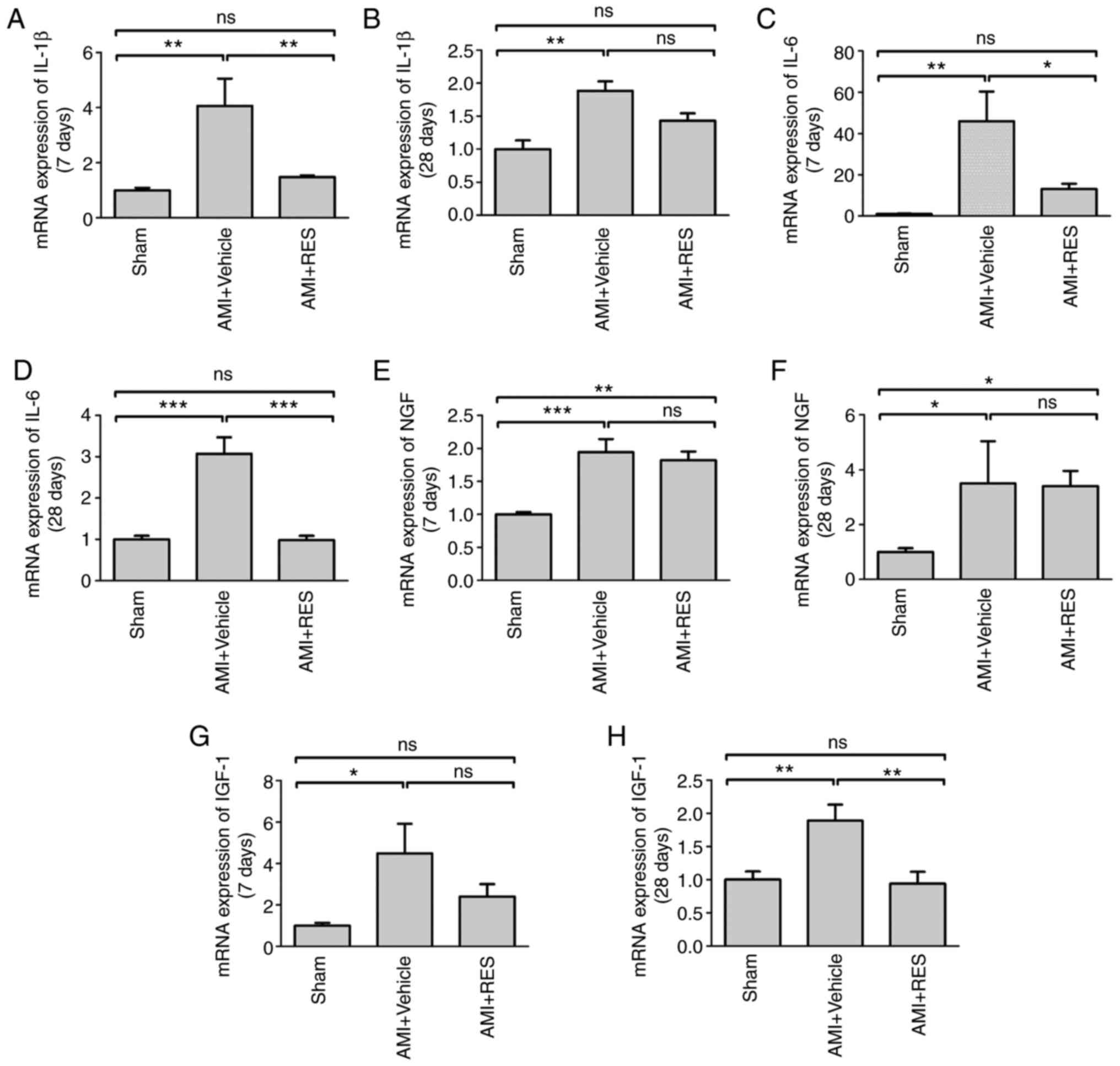 | Figure 8Effects of RES on inflammatory
pathways. mRNA levels of (A and B) IL-1β, (C and D) IL-6, (E and F)
NGF and (G and H) IGF-1 at 7 and 28 days post-AMI, respectively
were measured by RT-qPCR. Data are expressed as the fold change
relative to the sham group (n=8 per group). *P<0.05,
**P<0.01, ***P<0.001; ns, not
significant. RES, resveratrol; AMI, acute myocardial infarction;
IL, interleukin; NGF, nerve growth factor; IGF-1, insulin-like
growth factor-1. |
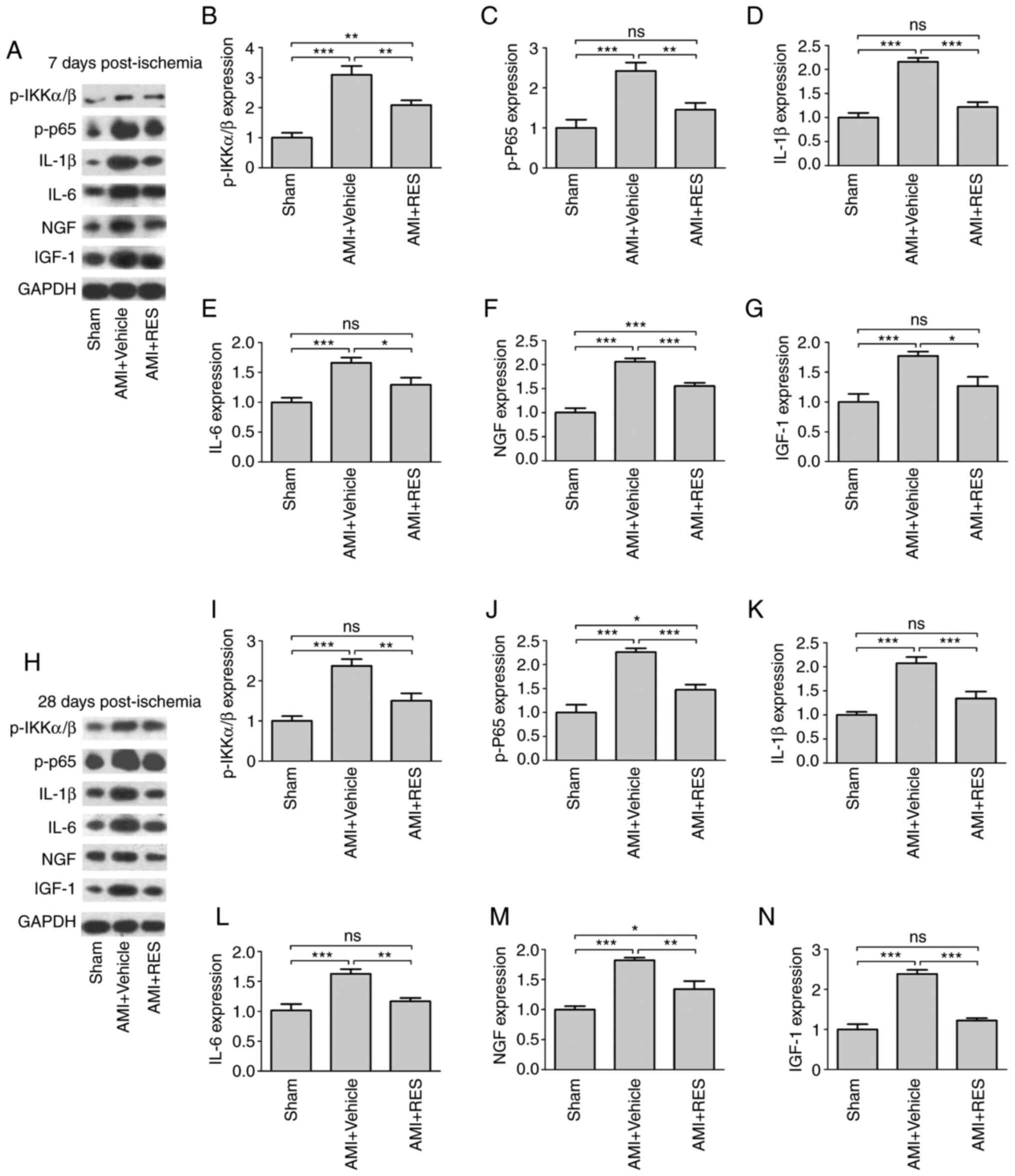 | Figure 9Effects of RES on NF-κB-dependent
inflammation. The expression of p-IKKα/β, p-p65, IL-1β, IL-6, NGF
and IGF-1 was determined in the mouse myocardium at (A-G) 7 days
and (H-N) 28 days by western blot analysis. Data are expressed as
the fold change relative to the sham group (n=8 per group).
*P<0.05, **P<0.01,
***P<0.001; ns, not significant. RES, resveratrol;
AMI, acute myocardial infarction; IL, interleukin; NGF, nerve
growth factor; IGF-1, insulin-like growth factor-1. |
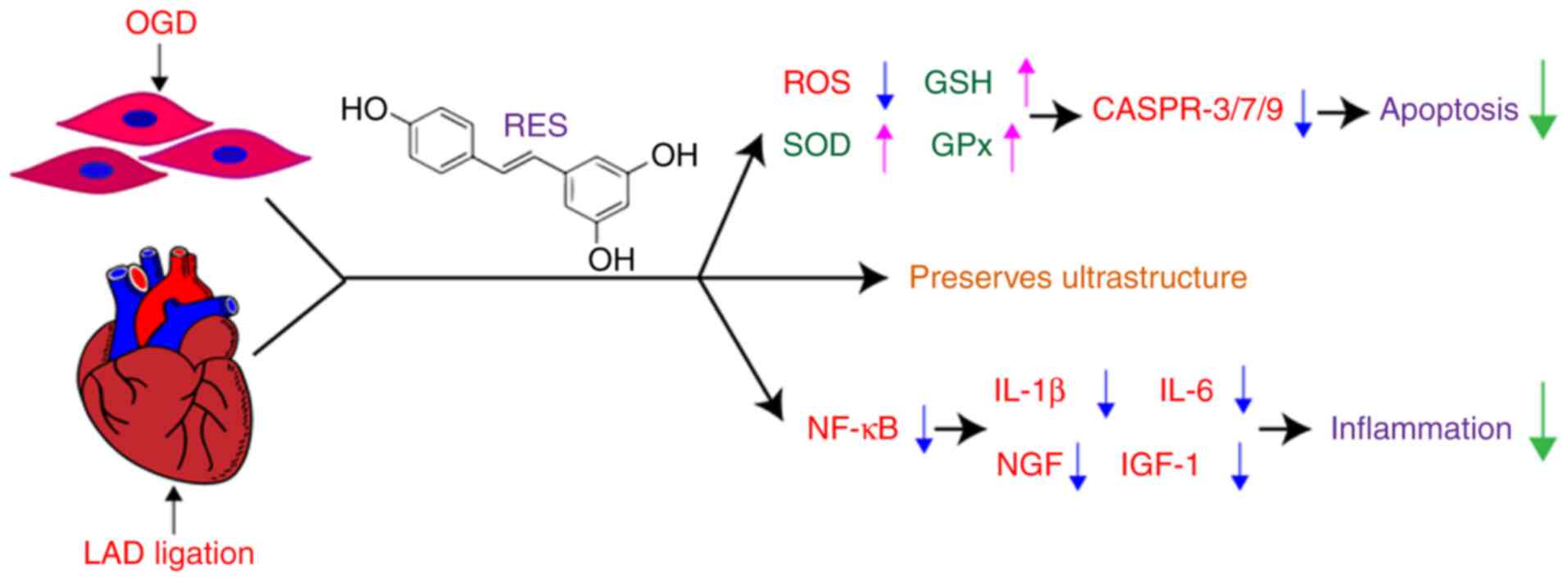
The present study demonstrates that RES attenuates
oxidative stress and NF-κB-dependent inflammation, and decreases
the apoptosis of cardiomyocytes following myocardial
hypoxia/ischemia (Fig. 10).
Discussion
Oxidative stress involves an attack on cells when a
biological system cannot readily detoxify the redox reactive
intermediates or repair the resulting damage. The heart is one of
the major organs that is vulnerable to be affected by ROS.
Oxidative stress participates in the pathological process of AMI
(21). The major ROS include
superoxide, hydrogen peroxide (H2O2),
hydroxyl anions and hydroxyl radicals. H2O2,
which can freely diffuse across the plasma membrane, is relatively
stable under physiological conditions (22). Thus, scavenging
H2O2 is essential to maintaining redox
homeostasis. Superoxide is usually rapidly dismutated to
H2O2 by SOD. H2O2 can
be scavenged to H2O and O2 by CAT and GPx
(23-25).
There is accumulating evidence to indicate that RES
not only prevents biomolecule oxidation by being oxidized itself,
but also enhances endogenous antioxidant defense system. Recently,
Aguilar-Alonso et al (26) found that RES treatment decreased
the levels of nitric oxide (NO) and malonaldehyde (MDA) in
cardiomyocytes during the aging process in rats without significant
changes in CAT and SOD levels. The present study demonstrated the
antioxidant effects of RES in myocardial ischemic injury.
Specifically, the present study demonstrated that RES regulated the
production and the balance of ROS in cardiomyocytes (Figs. 1 and 3). The enzyme activities of SOD and
GPx, and the GSH content in the mouse myocardium were markedly
reduced from the 7th day onward after AMI (Fig. 7), suggesting that the excessive
ROS in myocardium may not be scavenged in time. It was found RES
exerted protective effects against myocardial ischemic injury in
association with enhanced endogenous antioxidant defense systems.
These findings suggest that RES may have relatively limited ability
to remove H2O2.
ROS act as either a signaling molecule or a mediator
of inflammation in the infarcted myocardium (27). Inflammation plays a central role
in cardiac repair and the pathogenesis of post-infarction
remodeling and heart failure (28-31). NF-κB has been identified as a
redox-sensitive transcription factor associated with immune
responses (2). Upon activation,
NF-κB translocates to the nucleus to regulate the transcription of
proinflammatory cytokines (32).
On the one hand, the increase in the levels of pro-inflammatory
cytokines, such as IL-1β and IL-6 induces endothelial cell adhesion
molecule synthesis and activates leukocyte integrins, leading to
the recruitment of inflammatory cells into the infarcted areas
(29), thus aggravating
oxidative stress, myocardial infarction and consequent ventricular
dysfunction. On the other hand, IL-1β and IL-6 can trigger the
expression of growth factors, such as NGF and IGF-1, which promote
cardiac repair and preserve cardiac function by preventing negative
cardiac remodeling following MI (33-35).
The present study demonstrated that RES treatment
inhibited NF-κB signaling pathway activation, and decreased the
expression levels of IL-1β, IL-6, NGF and IGF-1 in the myocardium
of mice with AMI (Figs. 8 and
9). Moreover, a previous study
by the authors provided evidence that RES attenuated macrophage
infiltration in the infarcted zone of the heart (36). Thus, it was hypothesized that RES
may have the potential to reverse myocardial fibrosis and
pathologic remodeling in the late stage of AMI. Cardiac remodeling
has been confirmed to be involved in the pathogenesis and
progression of heart failure following MI. Ahmet et al
(37) found that long-term
dietary RES supplementation reduced cardiovascular structural and
functional deterioration in chronic heart failure, which verifies
our hypothesis.
Multiple lines of compelling evidence indicate that
RES possesses antioxidant and anti-inflammatory properties.
However, whether antioxidant therapy is a valid means of arresting
inflammation in patients remains controversial. Chekalina (38) examined the effect of RES on
parameters of central hemodynamics and myocardial ischemia in
patients with stable coronary heart disease (CHD), and demonstrated
that RES treatment effectively relieved myocardial ischemia and
improved LV diastolic function in the patients with CHD. These
findingsindicate that RES is applicable for the prevention and
therapy of myocardial ischemia. Hence, it is necessary to further
explore the long-term effects and underlying mechanisms of RES in
patients with AMI.
In conclusion, the present study suggests that RES
can potentially be used as a therapeutic and preventive drug for
myocardial hypoxic/ischemic injury. However, taking into account
the timeliness of RES treatment, it is suggested that optimizing
the therapeutic time window of RES against myocardial
hypoxic/ischemic injury in patients with CHD may enhance the
beneficial effects of RES.
Availability of data and materials
All data generated or analyzed during this study are
included in this published article or are available from the
corresponding author on reasonable request.
Authors' contributions
WL, CC and YZ conceived the study. WL, YH and JZ
designed the experiments. YH, XL, TC and YY carried out the
experiments and collected the data. YH, JZ and WL wrote and edited
the manuscript. All authors read and approved the final
manuscript.
Ethics approval and consent to
participate
The animal protocols followed the guidelines of the
Institutional Animal Care and Use Committee of Guangdong Medical
University, and the experiments were conducted according to the
National Institutes of Health (NIH) Guide for the care and use of
animals in laboratory experiments. The study was approved by the
Institutional Animal Care and Use Committee of Guangdong Medical
University.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
Acknowledgments
Not applicable.
References
|
1
|
Neri M, Fineschi V, Di Paolo M, Pomara C,
Riezzo I, Turillazzi E and Cerretani D: Cardiac oxidative stress
and inflammatory cytokines response after myocardial infarction.
Curr Vasc Pharmacol. 13:26–36. 2015. View Article : Google Scholar
|
|
2
|
Morgan MJ and Liu ZG: Crosstalk of
reactive oxygen species and NF-κB signaling. Cell Res. 21:103–115.
2011. View Article : Google Scholar
|
|
3
|
Pagliaro P and Penna C: Redox signalling
and cardioprotection: Translatability and mechanism. Br J
Pharmacol. 172:1974–1995. 2015. View Article : Google Scholar :
|
|
4
|
Hansen JM, Jones DP and Harris C: The
redox theory of development. Antioxid Redox Signal. 32:715–740.
2020. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Blaser H, Dostert C, Mak TW and Brenner D:
TNF and ROS crosstalk in inflammation. Trends Cell Biol.
26:249–261. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Choudhury S, Ghosh S, Gupta P, Mukherjee S
and Chattopadhyay S: Inflammation-induced ROS generation causes
pancreatic cell death through modulation of Nrf2/NF-κB and SAPK/JNK
pathway. Free Radic Res. 49:1371–1383. 2015. View Article : Google Scholar
|
|
7
|
Wan J, Shan Y, Fan Y, Fan C, Chen S, Sun
J, Zhu L, Qin L, Yu M and Lin Z: NF-κB inhibition attenuates
LPS-induced TLR4 activation in monocyte cells. Mol Med Rep.
14:4505–4510. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Hinz M and Scheidereit C: The IκB kinase
complex in NF-κB regulation and beyond. EMBO Rep. 15:46–61. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Li J, Xie C, Zhuang J, Li H, Yao Y, Shao C
and Wang H: Resveratrol attenuates inflammation in the rat heart
subjected to ischemia-reperfusion: Role of the TLR4/NF-κB signaling
pathway. Mol Med Rep. 11:1120–1126. 2015.
|
|
10
|
Ahmad R, Javed S and Bhandari U:
Antiapoptotic potential of herbal drugs in cardiovascular
disorders: An overview. Pharm Biol. 48:358–374. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Bonnefont-Rousselot D: Resveratrol and
cardiovascular diseases. Nutrients. 8:2502016. View Article : Google Scholar :
|
|
12
|
Stephan LS, Almeida ED, Markoski MM,
Garavaglia J and Marcadenti A: Red wine, resveratrol and atrial
fibrillation. Nutrients. 9:11902017. View Article : Google Scholar :
|
|
13
|
Annunziata G, Sanduzzi Zamparelli M,
Santoro C, Ciampaglia R, Stornaiuolo M, Tenore GC, Sanduzzi A and
Novellino E: May polyphenols have a role against coronavirus
infection? An overview of in vitro evidence. Front Med (Lausanne).
7:2402020. View Article : Google Scholar
|
|
14
|
Mao ZJ, Lin H, Hou JW, Zhou Q, Wang Q and
Chen YH: A meta-analysis of resveratrol protects against myocardial
ischemia/reperfusion injury: Evidence from small animal studies and
insight into molecular mechanisms. Oxid Med Cell Longev.
2019:57938672019. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Shen X, Wang M, Bi X, Zhang J, Wen S, Fu G
and Xia L: Resveratrol prevents endothelial progenitor cells from
senescence and reduces the oxidative reaction via PPAR-γ/HO-1
pathways. Mol Med Rep. 14:5528–5534. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Sun ZM, Guan P, Luo LF, Qin LY, Wang N,
Zhao YS and Ji ES: Resveratrol protects against CIH-induced
myocardial injury by targeting Nrf2 and blocking NLRP3 inflammasome
activation. Life Sci. 245:1173622020. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Ma L, Chuang CC, Weng W, Zhao L, Zheng Y,
Zhang J and Zuo L: Paeonol protects rat heart by improving regional
blood perfusion during no-reflow. Front Physiol. 7:2982016.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Shinomol GK and Muralidhara: Differential
induction of oxidative impairments in brain regions of male mice
following subchronic consumption of Khesari dhal (Lathyrus sativus)
and detoxified Khesari dhal. Neurotoxicology. 28:798–806. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar
|
|
20
|
Boos DD and Stefanski LA: P-value
precision and reproducibility. Am Stat. 65:213–221. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Misra MK, Sarwat M, Bhakuni P, Tuteja R
and Tuteja N: Oxidative stress and ischemic myocardial syndromes.
Med Sci Monit. 15:RA209–RA219. 2009.PubMed/NCBI
|
|
22
|
Bienert GP, Møller AL, Kristiansen KA,
Schulz A, Møller IM, Schjoerring JK and Jahn TP: Specific
aquaporins facilitate the diffusion of hydrogen peroxide across
membranes. J Biol Chem. 282:1183–1192. 2007. View Article : Google Scholar
|
|
23
|
Luna CM, Pastori GM, Driscoll S, Groten K,
Bernard S and Foyer CH: Drought controls on
H2O2 accumulation, catalase (CAT) activity
and CAT gene expression in wheat. J Exp Bot. 56:417–423. 2005.
View Article : Google Scholar
|
|
24
|
Bagulho A, Vilas-Boas F, Pena A, Peneda C,
Santos FC, Jerónimo A, de Almeida RFM and Real C: The extracellular
matrix modulates H2O2 degradation and redox
signaling in endothelial cells. Redox Biol. 6:454–460. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Rhee SG, Woo HA and Kang D: The role of
peroxiredoxins in the transduction of H2O2
signals. Antioxid Redox Signal. 28:537–557. 2018. View Article : Google Scholar
|
|
26
|
Aguilar-Alonso P, Vera-López O,
Brambila-Colombres E, Segura-Badilla O, Avalos-López R,
Lazcano-Hernández M and Navarro-Cruz AR: Evaluation of oxidative
stress in cardiomyocytes during the aging process in rats treated
with resveratrol. Oxid Med Cell Longev. 2018:13904832018.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Mittal M, Siddiqui MR, Tran K, Reddy SP
and Malik AB: Reactive oxygen species in inflammation and tissue
injury. Antioxid Redox Signal. 20:1126–1167. 2014. View Article : Google Scholar :
|
|
28
|
Frangogiannis NG: Targeting the
inflammatory response in healing myocardial infarcts. Curr Med
Chem. 13:1877–1893. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Frangogiannis NG: The inflammatory
response in myocardial injury, repair, and remodelling. Nat Rev
Cardiol. 11:255–265. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Christia P and Frangogiannis NG: Targeting
inflammatory pathways in myocardial infarction. Eur J Clin Invest.
43:986–995. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Anzai T: Inflammatory mechanisms of
cardiovascular remodeling. Circ J. 82:629–635. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Zhang L, Chen Y, Yue Z, He Y, Zou J, Chen
S, Liu M, Chen X, Liu Z, Liu X, et al: The p65 subunit of NF-κB
involves in RIP140-mediated inflammatory and metabolic
dysregulation in cardiomyocytes. Arch Biochem Biophys. 554:22–27.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Mahmoud AI, O'Meara CC, Gemberling M, Zhao
L, Bryant DM, Zheng R, Gannon JB, Cai L, Choi WY, Egnaczyk GF, et
al: Nerves regulate cardiomyocyte proliferation and heart
regeneration. Dev Cell. 34:387–399. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Liu Y and Wu JH: GW26-e0203 effects of
nerve growth factor on late reperfusion after myocardial
infarction. J Am Coll Cardiol. 66(16 Suppl): C28–C29. 2015.
View Article : Google Scholar
|
|
35
|
Jackson R, Tilokee EL, Latham N, Mount S,
Rafatian G, Strydhorst J, Ye B, Boodhwani M, Chan V, Ruel M, et al:
Paracrine engineering of human cardiac stem cells with insulin-like
growth factor 1 enhances myocardial repair. J Am Heart Assoc.
4:e0021042015. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Xin P, Pan Y, Zhu W, Huang S, Wei M and
Chen C: Favorable effects of resveratrol on sympathetic neural
remodeling in rats following myocardial infarction. Eur J
Pharmacol. 649:293–300. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Ahmet I, Tae HJ, Lakatta EG and Talan M:
Long-term low dose dietary resveratrol supplement reduces
cardiovascular structural and functional deterioration in chronic
heart failure in rats. Can J Physiol Pharmacol. 95:268–274. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Chekalina NI: Resveratrol has a positive
effect on parameters of central hemodynamics and myocardial
ischemia in patients with stable coronary heart disease. Wiad Lek.
70:286–291. 2017.PubMed/NCBI
|