1. Introduction
Europe and the entire world have faced the second
wave of Coronavirus Disease 2019 (COVID-19) pandemic which was
characterized by an increased number of infections and related
deaths worldwide, thus still highlighting critical issues in the
management of this health emergency (1,2).
At the time of the writing of the present review article,
113,523,131 laboratory-confirmed COVID-19 cases and 2,519,454
COVID-19-related deaths have been recorded worldwide, highlighting
the impressive impact of this pandemic globally (3).
Despite the optimism deriving from the approval of
two new mRNA vaccines and of one recombinant vaccine against severe
acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infections by
the Food and Drug Administration (FDA) and the European Medicines
Agency (EMA) (5,6), a third wave of infections, already
observed in the United States, the United Kingdom and other
countries with greater proportions than the one just concluded, is
expected in the coming months (3,6,7).
In this regard, several governments worldwide have already begun to
adopt social distancing measures and the lockdown of collective
activities in order to avoid a drastic increase in the number of
infections (8,9). In addition, a great concern is also
represented by the need to differentiate COVID-19 cases from
seasonal flu that could clog hospital emergency services, slowing
down the diagnostic and therapeutic procedures for patients with
COVID-19 (10). These reasons
have led the scientific community to question which diagnostic
strategies are optimal in order to efficiently combat the imminent
increase in COVID-19 infections, as well as to perform differential
diagnoses between COVID-19 infections and seasonal flu.
In this context, population screening strategies
have been proposed and are currently being implemented for the
effective monitoring of the COVID-19 epidemiological curve and to
screen the immunized population; however, it is not yet clear which
is the most effective method for these surveillance programs
(11-14).
Since the beginning of the pandemic, immense efforts
have been made for the development of effective diagnostic
strategies which may be used to accurately identify
SARS-CoV-2-infected patients, thus limiting the risk of contagion
and promptly treating any respiratory symptoms, avoiding serious
consequences for individuals (15).
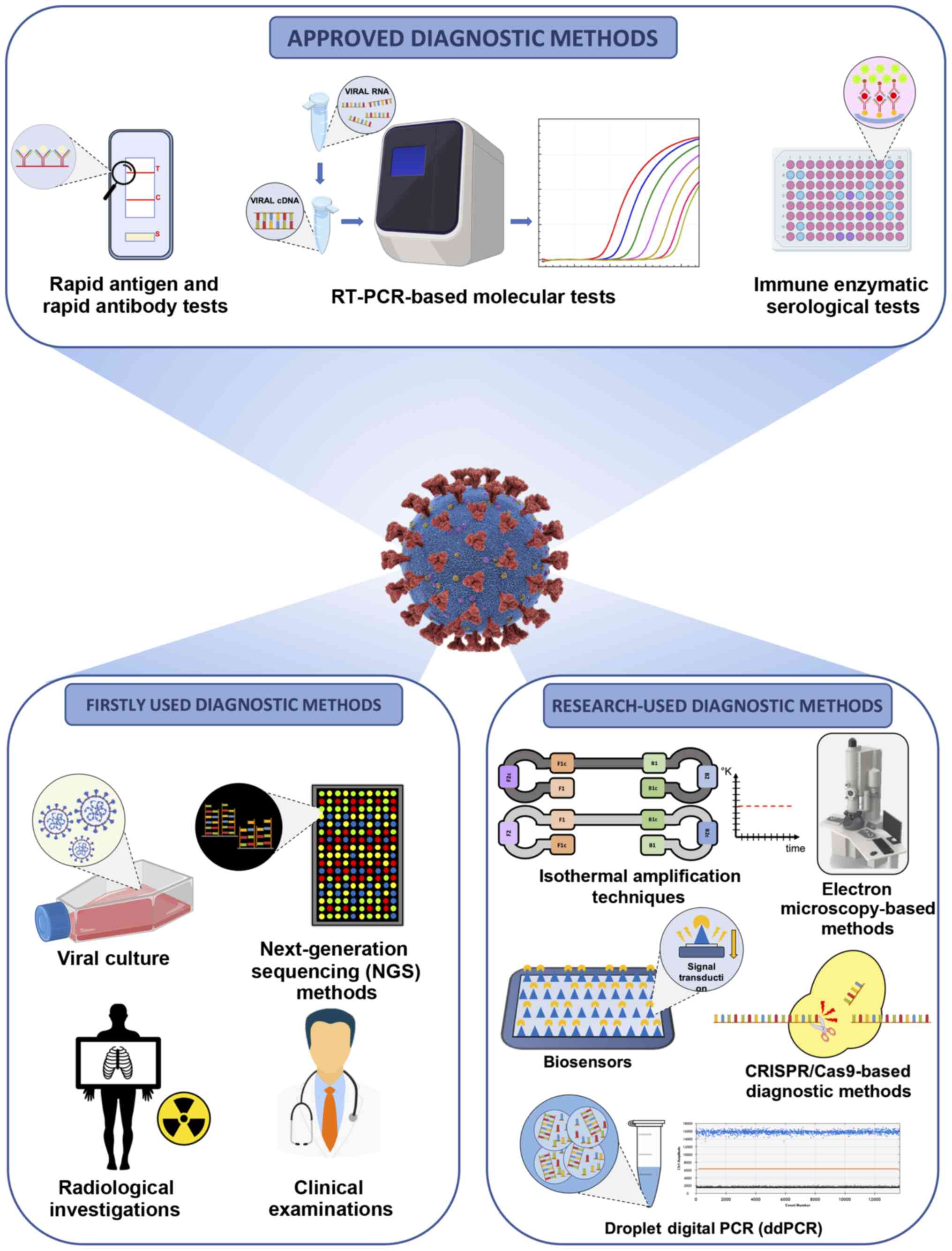
At present, several diagnostic methods have been
approved by regulatory agencies worldwide; however, there is still
confusion regarding the correct tests to be used based on the
patient's medical history or the investigation purpose (16). As regards Europe, there are 365
different commercialized devices CE-IVD- and FDA-approved or used
for research purposes. Of these devices, 168 are immunoassays, 192
are PCR-based methods, three are NGS-based methods and two
commercialized tools are based on different technologies (17). Therefore, it is evident that the
selection of the optimal diagnostic test can be difficult.
On these bases, the present review aimed to provide
the rationale for the correct use of SARS-CoV-2 diagnostic tools
currently available by setting out the decision-making principles
useful for the correct choice of the most appropriate test.
As will be discussed in the following paragraphs,
several diagnostic tests are currently available for the early
identification of SARS-CoV-2 infection, for the monitoring of the
presence of any infections among healthcare workers, for the
monitoring the incidence rates and the severity of the infection
and for the evaluation of the complete remission of patients with
COVID-19 (18-20).
Several parameters should be considered for the
selection of the optimal diagnostic test. A diagnostic test must
have good sensitivity and specificity rates; however, these
parameters are not the only features to be considered. Indeed, in
the case of the COVID-19 pandemic, an effective test should be
rapid, repeatable, based on technologies available at numerous
centers and keep costs limited in order to be carried out on a
large fraction of the population (21).
Therefore, it is evident that the selection of the
diagnostic test should be performed taking into account the
clinical or surveillance purpose of the investigation as well as
the possibility to repeat the test several times until the patients
are no longer positive.
For all these clinical and epidemiological needs,
three main types of tests for COVID-19 diagnosis are available: i)
Molecular RT-PCR swab tests; ii) serological tests; iii) rapid
antigen or antibody tests.
2. The right test, on the right sample, at
the right time
Although the molecular and structural
characteristics of SARS-CoV-2 were initially unknown, in a very
short period of time, research laboratories and biomedical
companies studied the main features of the virus, thus assisting
researchers world-wide in developing various diagnostic solutions
for a correct diagnosis of COVID-19 (22-24). Among such solutions, the most
commonly used and validated methodologies are rapid antigen or
antibody tests, immunoenzymatic serological tests and RT-PCR-based
molecular tests. Each of these three types of diagnostic tests can
be applied at a precise moment of infection. Of note, only kits,
reagents and molecular probes validated by the Centers for Diseases
Control and Prevention (CDC) and the World Health Organization
(WHO) and approved at the American and European level by the FDA
and by the EMA can be used for diagnostic purposes (25).
Although immunoenzymatic (either classic or rapid
methods) and molecular methods are the most widely used techniques
for the diagnosis of COVID-19, other approaches were used in the
early phase of the pandemic to identify positive patients and the
etiological agent of infection. Among these approaches, viral
culture and next-generation sequencing (NGS) methods were crucial
for the identification of the novel coronavirus and for the
characterization of its molecular structure. These two techniques
have made it possible to fully characterize the genome and the
viral protein structure, allowing the understanding of the viral
mechanisms of action, the mode of transmission, the clinical impact
and the development of any therapeutic strategies and diagnostic
tools (26,27).
Apart from these conventional strategies, other
diagnostic methods are under development and validation or are
currently finding application in research contexts. Among these
methods, digital PCR, isothermal amplification techniques, clusters
of regularly interspaced short palindromic repeats/Cas
(CRISPR/Cas)-based diagnostic methods, biosensors, electron
microscopy-based methods, etc., represent the armamentarium to
effectively diagnose COVID-19 infection and to effectively assess
the epidemiological spreading of the pandemic (15,28). Notably, clinical investigations
and radiological imaging have represented valid diagnostic
alternatives, particularly during the early stages of the pandemic
when there were still no validated molecular and serological tests
available (29) (Fig. 1).
Despite the availability of all these diagnostic
techniques, a correct diagnosis of COVID-19 infection can only be
established considering the test to be used, the type of sample to
be analyzed and the timing of the test itself. Therefore, it is
necessary to perform the correct test, at the correct time in the
correct biological sample (30,31).
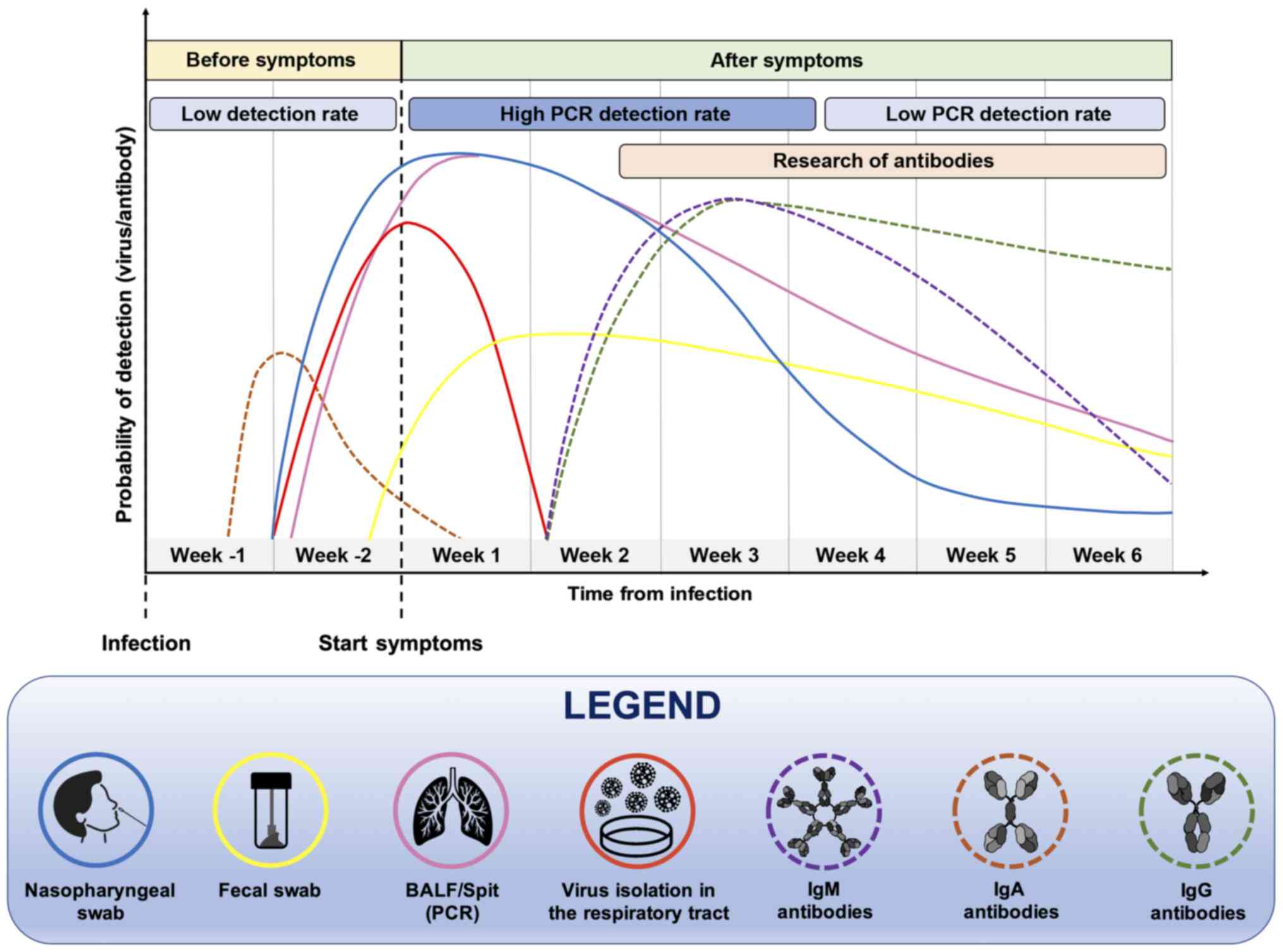
In particular, it is important to take into account
the moment of the suspected infection, the patient's medical
history, the symptoms and general clinical picture for a successful
outcome of the diagnostic test. Furthermore, the positivity of a
diagnostic test strongly depends on the moment at which it is
carried out (Fig. 2). Both
serological and molecular tests are not useful during the first
week of the supposed infection because the virus is still in its
incubation period and there are not yet sufficient copies of viral
RNA in circulation neither antibodies nor viral proteins
identifiable by serological tests (32,33). Therefore, before the onset of
symptoms, the probability of correctly determining the presence of
the virus, particularly using molecular tests, remains low
(32).
At two weeks after the presumed infection, and in
parallel with the onset of symptoms, molecular tests carried out on
nasopharyngeal swabs or bronchoalveolar lavage fluid (BALF) samples
will have a greater chance of being positive, as the virus is
actively replicating. However, this probability is reduced over
time, as a result of the elimination of the virus and the remission
of the disease. In the case of molecular tests, the probability of
a positive or negative result should be considered, as no
commercial diagnostic tools have a sensitivity of 100%,
particularly in cases of patients with low viral load as
asymptomatic or paucisymptomatic individuals (32,34).
As regards serological tests, the search for IgM and
IgG antibodies begins to yield positivity at approximately one
month after the presumed infection and the levels of these
immunoglobulins remain high for long periods of time. Of note, the
IgM serum levels decrease significantly after six weeks from the
onset of symptoms (35). The
detection of viral proteins and mucosal IgA antibodies is also very
important. Indeed, these markers increase in the early stages of
infection and can be used for the early diagnosis of COVID-19
infection (36). In summary, all
these data suggest that for a correct diagnosis of COVID-19
infection to be made, it is necessary to use the right test, at the
right time.
As already mentioned, the selection and the correct
handling of samples for both molecular and serological analyses are
fundamental for the diagnosis of COVID-19 infection. In particular,
the collection of samples and the pre-analytical phases are crucial
for the positive outcome of the diagnostic procedure (37,38). Different studies have
demonstrated that the positivity rate of molecular tests
significantly depends on the quality of the starting sample
influenced by sample collection, poor quality material, wrong
transport or storage, the presence of inhibitors, etc. (38). Technical troubleshooting will be
discussed in the following chapters.
The chief samples used for molecular analyses are
obtained from the respiratory tract. In particular, both
oropharyngeal and nasopharyngeal swabs represent good samples for
viral RNA extraction and amplification through RT-PCR; however,
previous studies have highlighted that BALF is the most appropriate
sample for SARS-CoV-2 molecular detection (39,40). Apart from these commonly analyzed
respiratory tract samples, other specimens may also be collected,
including nasal mid-turbinate swabs and nasal or nasopharyngeal
wash/aspirate (41). Of note,
the permanence of the virus in the respiratory tract is only
temporary, with a peak of positivity in the first three days of
infection, followed by a constant decrement of the rate of
positivity for molecular analyses until the 10th week following
symptom onset (42). To
ascertain SARS-CoV-2 positivity after a long period of time, other
biological samples are used. Among these, fecal samples represent
the optimal material to evaluate the permanence of viral RNA in
patients with negative nasopharyngeal swabs, but with clinical
symptoms attributable to COVID-19 infection (43). Indeed, a greater persistence of
SARS-CoV-2 in the gastrointestinal tract has been demonstrated,
allowing the detection of SARS-CoV-2 RNA even after more than a
month from infection (44).
Other studies have proposed diagnostic screening
based on the analysis of saliva and urine samples through
serological and molecular tests. However, the collection of these
samples is generally carried out by the patients themselves without
the surveillance of a healthcare professional, resulting in a
possible non-representative sample. In addition, the presence of
interfering substances or substances that degrade viral RNA or
human antibodies represents a considerable bias that significantly
limits the use of these samples in clinical practice (30).
All the types of biological samples used for
diagnostic purposes are presented in Table I. A detailed description of the
timing of use and the technique for which specific samples are
collected is also provided (Table
I).
 | Table IBiological samples and methods used
for an effective diagnosis of COVID-19 infection. |
Table I
Biological samples and methods used
for an effective diagnosis of COVID-19 infection.
| Type of
specimens | Collection
devices | Transport
conditions | Diagnostic methods
| Comments |
|---|
| Approved diagnostic
methods | Research-used
diagnostic methods |
|---|
| NP swab, OP swab,
and NP aspirate | Dacron or VTM
flocked swabs | Within 5 days, 4°C;
>5 days, −70°C | Real-time PCR | NGS - CRISPR/Cas
PCR - isothermal amplification techniques −ddPCR - viral
culture | |
| Sputum | Sterile vial | Within 48 h, 4°C;
>48 h, −70°C | Real-time PCR | NGS - CRISPR/Cas
PCR - isothermal amplification techniques - ddPCR - viral
culture | |
| Bronchial
washing | Sterile vial | Within 48 h, 4°C;
>48 h, −70°C | Real-time PCR | NGS - CRISPR/Cas
PCR - isothermal amplification techniques - ddPCR - viral
culture | Pathogens may be
diluted; however, the specimen can be used for diagnostic
testing |
| Tracheal aspirate
and transtracheal aspirate | Sterile vial | Within 48 h, 4°C;
>48 h, −70°C | Real-time PCR | NGS - CRISPR/Cas
PCR - isothermal amplification techniques - ddPCR - viral
culture | |
| Lung biopsy | Sterile vial | Within 48 h, 4°C;
>48 h, −70°C | Real-time PCR | NGS - CRISPR/Cas
PCR - isothermal amplification techniques - ddPCR - viral
culture | |
| Serum, plasma | Serum/plasma
collection tube: Adults, 3-5 ml; infants, 1 ml | Within 5 days, 4°C;
>5 days, −70°C | Rapid serological
test; immune enzymatic test | Biosensors | For the immune
enzymatic test, two samples are collected. The first sample is
collected between 1-7 days after symptom onset and the second is
collected 14 days after the onset of symptoms |
Overall, the type of sample should be collected
considering the timing of the infection. Collecting different
samples from different sites may be useful to avoid misdiagnosis of
asymptomatic patients negative for molecular tests. Otherwise, it
is possible to carry out repeated nasopharyngeal swabs in two or
three consecutive days in order to overcome the window period of
SARS-CoV-2 incubation thus being able to correctly diagnose a
patient infected with SARS-CoV-2 (45).
3. RT-PCR-based molecular tests
RT-PCR-based molecular methods represent the gold
standard techniques used worldwide to make a confirmatory diagnosis
of COVID-19 infection (46).
Since the complete sequencing of the SARS-CoV-2 genome (26), researchers of different countries
have begun to design molecular primers and probes specific to
SARS-CoV-2 RNA sequences in order to perform differential diagnosis
between COVID-19 infections and other pathologies with similar
symptoms, such as seasonal flu or bacterial infections (47,48). Of note, the whole genome sequence
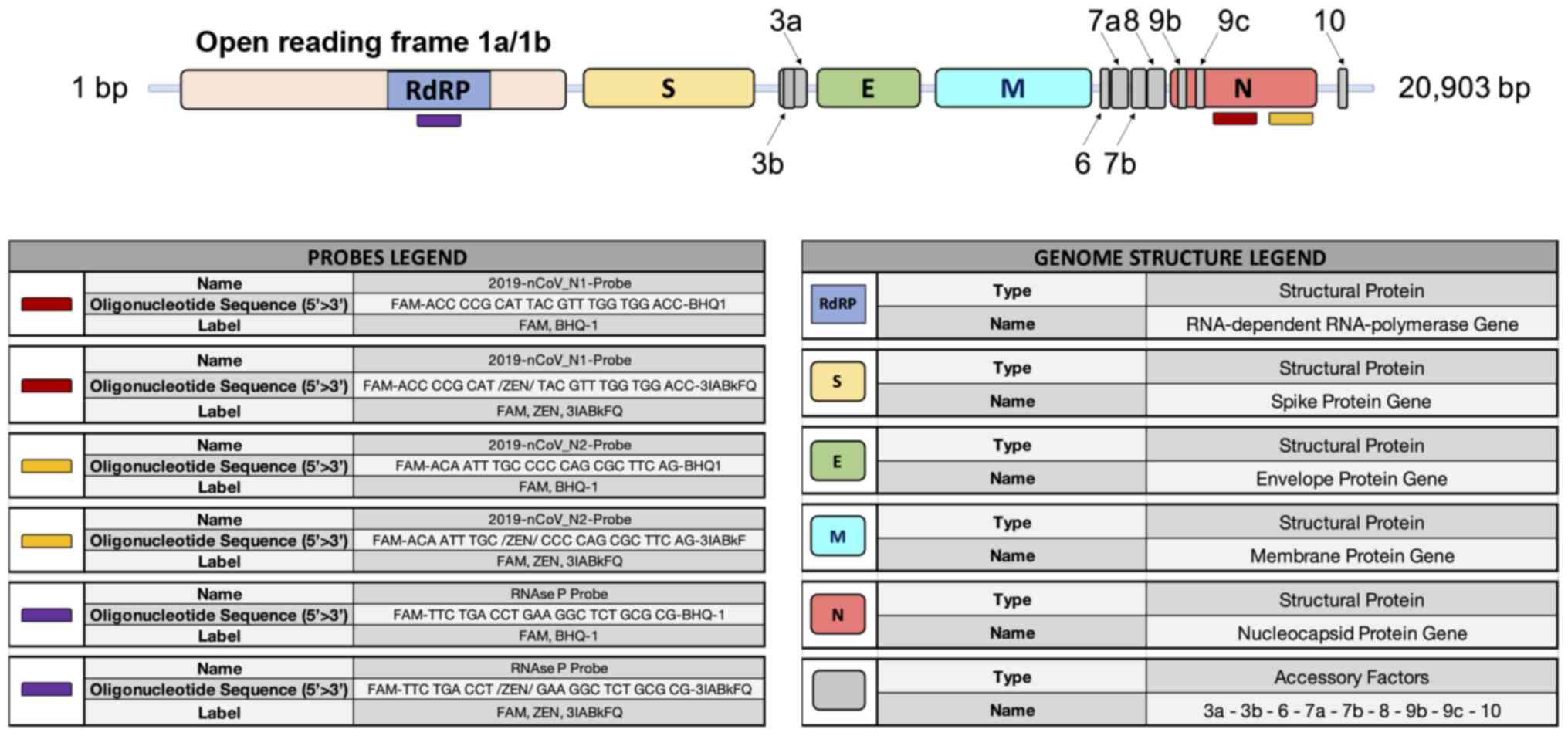
of SARS-CoV-2 is 29,903 bp in length containing the following
functional elements: A poly-A cap of 50 bases, the open reading
frame 1/ab (ORF1/ab) containing the coding sequences for the
RNA-dependent RNA polymerase (RdRP), the spike proteins, the
envelope proteins, the membrane and nucleocapsid proteins, and a
poly-A tail of 30 bp (49).
At present, several portions of the SARS-CoV-2
genome are used for the design of specific primers and probes,
including the genomic portions encoding for the RdRP gene, for
proteins constituting the nucleocapsid (N gene) and spike molecules
(S gene), for proteins of the envelope (E gene), for the membrane,
etc. (Fig. 3).
The CDC has made available a list of approved and
validated kits and reagents to clinically diagnose COVID-19
infection (50). Similarly, the
CDC has also made available a list of three primer pairs that can
be used for research purposes only, each working with two different
molecular probes, specific for two portions of the N gene and the
gene coding for viral RdRP, respectively (51).
RT-PCR-based molecular tests are considered the
optimal diagnostic option for wide surveillance strategies due to
the relatively low costs of the entire viral RNA extraction,
reverse transcription and amplification procedure, and the
availability of RT-PCR thermal cyclers in hospitals, research
institutes and private laboratories (52). Other advantages of RT-PCR methods
compared to other diagnostic techniques are the timesaving of the
procedure, the easy execution of the technique and the
non-necessity of highly trained personnel (53). In addition, a number of the
available RT-PCR kits are based on one-step amplification methods,
where the buffer of the nasopharyngeal swab is inserted into the
plate and the machine autonomously provides for the extraction,
reverse transcription, amplification and analysis of the samples.
These procedures ensure fast results guaranteeing excellent
reproducibility and standardization of the data obtained which are
less influenced by operator bias (54).
As already mentioned, the EMA has approved 192
PCR-based methods while the FDA has approved 235 different
molecular tests for both RT-PCR and the rapid detection of
SARS-CoV-2 RNA (17,55).
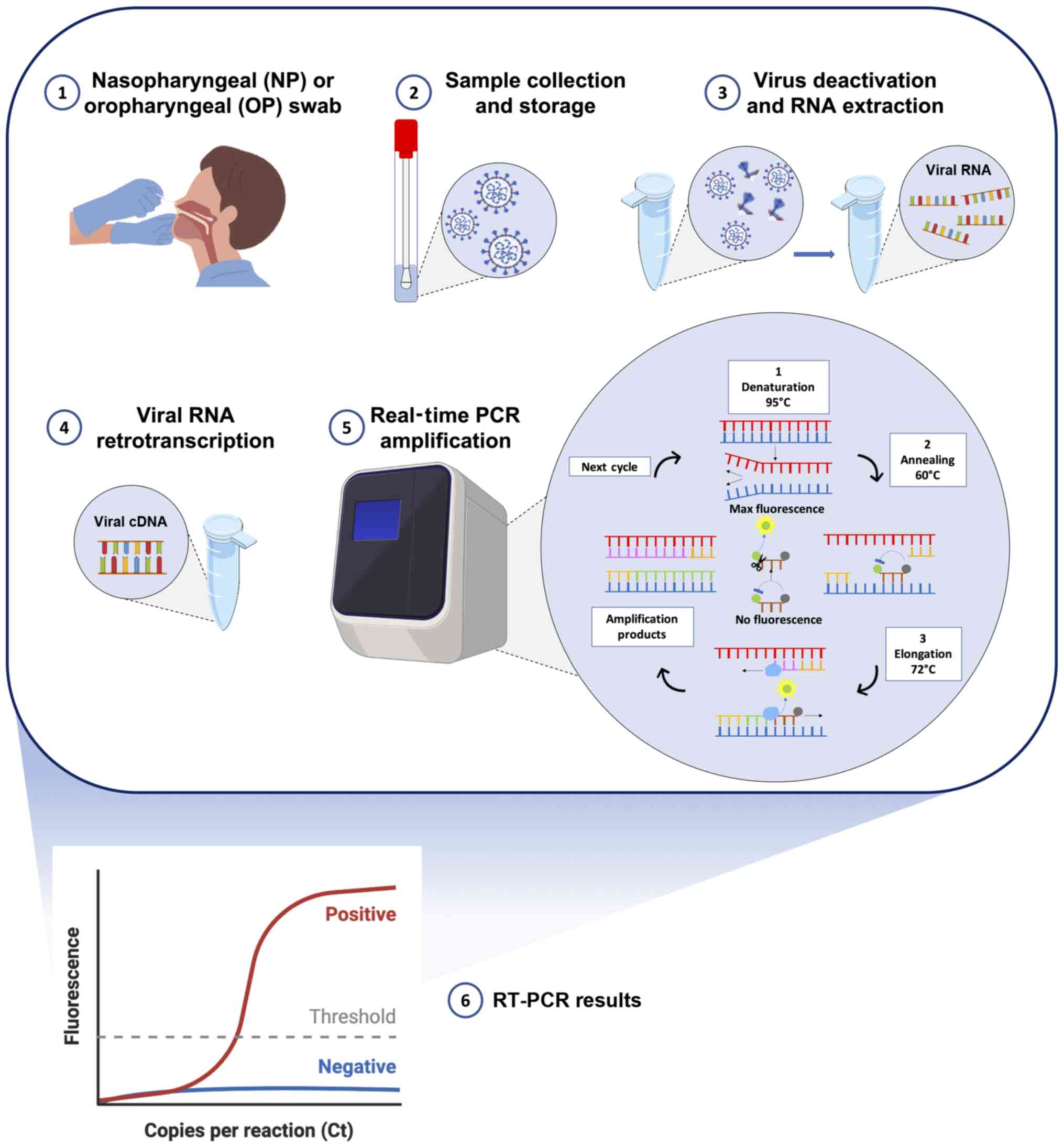
All these approved molecular tests detect two or
three fragments of SARS-CoV-2 RNA mainly using multiplex assays
based on standard RT-PCR protocols. Briefly, following the
collection of nasopharyngeal swabs from individuals with suspected
COVID-19 infection, the viral RNA is extracted using commercial
kits and lysing solutions with manual or automated extraction
protocols. Subsequently, the obtained RNA, containing both human
and viral RNA, can be directly analyzed by RT-PCR in the one-step
protocols or must be retrotranscribed into complementary DNA (cDNA)
before RT-PCR amplification. After obtaining the cDNA, the
SARS-CoV-2 targets are amplified through RT-PCR using TaqMan probes
specific for the two-three viral targets. In particular, the
exonuclease activity of the Taq polymerase (5′→3′ direction)
cleaves the probes annealed with the viral targets allowing the
emission and real-time detection of fluorescent signals. In this
manner, the intensity of the fluorescent signal is proportional to
the total amount of the amplified targets. However, all the
existing RT-PCR protocols are only qualitative and not quantitative
(Fig. 4) (56,57).
The entire analytical procedure is completed in 4-8
h, based on the type of RT-PCR protocol used (one-step or
two-steps). This makes it possible to establish the positivity of
an individual in a relatively short amount of time, allowing the
initiation of quarantine protocols that limit the spread of
infections (58,59). In addition, the majority of the
automated or semi-automated systems available are based on 48- or
96-well platforms in order to process a series of samples, thus
reducing the execution times, the costs of plastics and consumables
and procedural errors that may occur by analyzing the samples
individually (60).
RT-PCR-based methods ensure also a low limit of
detection (LoD) of SARS-CoV-2 RNA (61). Specifically, during the early
stages of the pandemic, when diagnostic techniques had not yet been
optimized and standardized, a significant fraction of
COVID-19-positive patients were identified as false-negative due to
the low sensitivity of the primers and probes used or the
inaccuracy of the whole RT-PCR procedure (false-negative rates
ranging from 38% at the day of symptoms onset to 67% before one day
from the onset of symptoms or to 66% after 16 days from the onset
of symptoms) (62). As will be
better described at the end of this chapter, in the case of
asymptomatic or paucisymptomatic patients, generally characterized
by a low viral load, the RT-PCR investigations could mistakenly
yield negative results. Currently, the commercially available
RT-PCR kits have partially solved the problem of the low
sensitivity of RT-PCR. Indeed, the methods available today have a
theoretical LoD that varies from 0.3 copies/µl to 100
copies/µl, depending on the diagnostic system used (63). However, it should be considered
that this limit is only theoretical; therefore, in clinical
practice, procedural errors or reaction interferers raise the LoD
by 10-fold (64).
Although RT-PCR represents the gold standard method
for the diagnosis of COVID-19 infection, this method is subject to
several limitations and criticisms that can lead to false-positive
or false-negative results, thus affecting the correct management of
the pandemic. As already mentioned, one of the main limits of
RT-PCR is its low sensitivity in correctly diagnosing samples with
low viral load, including swabs taken incorrectly or obtained from
asymptomatic or paucisymptomatic individuals (65). In addition, RT-PCR is affected by
contaminants and interferers contained in the sample or introduced
by the operator capable of inhibiting the reaction (65). Other limits also concern the
execution time of the analysis, which in non-automated systems, can
take up to 24 h to obtain a result that can be communicated to the
patient. Finally, RT-PCR is a method that is profoundly affected by
pre-analytical and analytical bias regarding the collection,
storage and handling of samples, therefore, careful attention
should be paid during the collection and management of samples
(66).
In order to reduce the errors in the interpretation
of molecular tests, the WHO has published specific recommendations
useful to make a correct diagnosis of COVID-19. In particular, a
molecular test must be conducted in two different biological
matrices, for example, a nasopharyngeal swab and a fecal swab, or
be performed on two consecutive nasopharyngeal swabs in order to
obtain reliable results. The approved tests include the analysis of
three different viral genes or two viral genes and a human control
gene and the use of specific negative and positive controls useful
to ascertain the presence of contamination or evaluate the
inhibition of the reaction (67).
Overall, RT-PCR represents the gold standard and
most widely used method worldwide to make an accurate diagnosis of
COVID-19 infection due to the rapidity of the method and the
availability of instruments in public and private hospitals and
laboratories. In addition, the RT-PCR tests currently available on
the market ensure good sensitivity and specificity rates for the
diagnosis of COVID-19 infection. The main RT-PCR diagnostic systems
currently available on the market, illustrating their main
technical features are presented in Table SI.
4. Rapid antigen and rapid antibody
tests
With the increase in the number of individuals with
a suspected COVID-19 infection, it became necessary to adopt more
rapid and low-cost diagnostic strategies to carry out extensive
surveillance campaigns (68-70). To cope with this emergency,
various rapid tests have been developed to detect viral antigens or
anti-SARS-CoV-2 human antibodies in salivary, nasal or
oropharyngeal swabs and blood samples. These tests are currently
adopted for the frequent monitoring of personnel operating in
at-risk environments such as schools or hospitals or to carry out
extensive screening strategies on populations where a new outbreak
of infection is suspected (70,71).
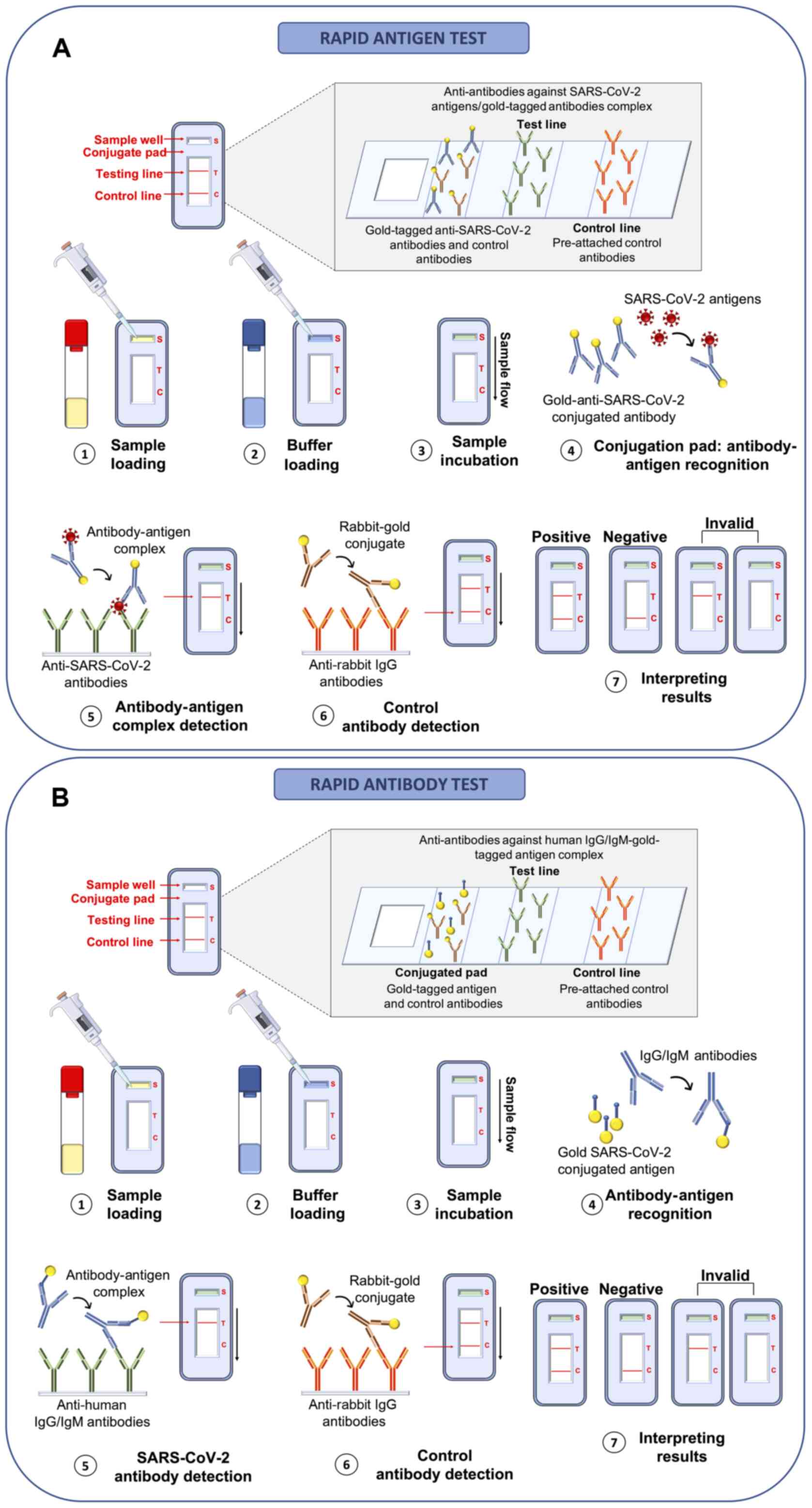
Compared to RT-PCR-based methods, rapid antigenic
and rapid antibody tests are characterized by more rapid execution
times of ~15-30 min, a lower cost and an easier procedure that does
not require the presence of highly trained personnel (72). These tests are mainly built on
platforms based on the principle of lateral flow immunoassay (LFIA)
for the direct detection of viral proteins (rapid antigen tests) or
human antibodies against SARS-CoV-2 antigens (rapid antibody
tests). As regards rapid antigen tests, these allow the
identification of COVID-19-positive individuals through the
detection of SARS-CoV-2 nucleocapsid or Spike proteins (viral
antigens) in swabs collected from the upper airways of the subject
with suspected infection. The principle behind these LFIA rapid
antigen tests is very simple and based on the bond between antigens
and antibodies that occurs on the surface of a porous membrane
where the swab buffer flows by capillarity. Briefly, the swab
buffer is loaded in the sample well of the cartridge and flows by
capillarity at the level of the conjugation pad containing control
rabbit antibodies and specific antibodies against SARS-CoV-2
antigens both linked to detector molecules (conjugated antibodies).
In the case of a positive sample, the link between viral antigens
and conjugated antibodies take place at the level of the
conjugation pad. Subsequently, the buffer containing conjugated
control rabbit antibodies and the antigen-conjugated antibody
complex flows to the test line where other antibodies specific for
viral antigens are immobilized. In the case of a positive sample,
the binding between antibodies immobilized in the test line and the
antigen-conjugated antibody complex gives rise to a colorimetric
reaction indicating the positivity of the sample. Finally, the
buffer flows further to the control line where anti-antibodies
specific for the conjugated control rabbit antibodies are
immobilized; if the test is performed correctly, the reaction
between these two molecules also gives rise to a colorimetric
reaction. To be trusted, the test control line must always be
positive (73,74) (Fig. 5A).
Similarly, rapid antibody tests use the same LFIA
principle; however, human IgA or IgG and IgM against SARS-CoV-2
antigens are searched for. In particular, the blood (or saliva)
sample is loaded inside the sample well and flows to the conjugated
pad containing gold-tagged viral antigens and gold-tagged control
antibodies. In this test, anti-SARS-CoV-2 human antibodies (IgA,
IgG or IgM) bind the gold-tagged antigens and the sample flows to
the test line where anti-human IgA, IgG or IgM antibodies are
pre-attached to the membrane. In the case of a positive sample, the
IgA, IgG or IgM-gold-tagged antigen complex binds to the human
anti-antibodies immobilized in the test line determining a
colorimetric reaction which indicates the positivity of the sample.
Finally, the gold-tagged control rabbit antibodies flow to the
control line binding anti-rabbit antibodies thus giving rise to a
confirmatory colorimetric reaction (75,76) (Fig. 5B).
Both rapid antigen and rapid antibody tests yield
results readable to the naked eye in a very short period of time
and can be performed at the 'point-of-care' without any specific
instruments or sample processing. Of note, these tests yield
qualitative, but not quantitative results; therefore, it is only
possible to establish whether the individual is positive or not for
COVID-19 infection without assessing the viral load. In addition,
in the case of rapid antibody tests, it is possible to establish
whether the patient is carrying anti-SARS-CoV-2 antibodies;
however, it is not possible to establish whether the patient has an
active SARS-CoV-2 infection or already resolved disease (77,78).
Overall, among the main advantages of rapid antigen
and rapid antibody tests are the low cost, the possibility of
carrying out the test directly at the point-of-care and the high
speed and easy execution that ensure a positive or negative result
in ~30 min. However, these tests suffer from important limitations
mainly related to a low sensitivity and specificity of 56.2 and
99.5%, respectively (79).
Indeed, a recent review of the literature demonstrated that rapid
antigen tests had a false negative rate of 27.9%, while no
false-positive results were observed (68). As regards rapid antibody tests, a
recent study comparing three different kits demonstrated a
false-positive rate ranging from 51.6 to 28.1%, and a
false-negative rate ranging from 0 to 4.2% (80).
As regards rapid antibody tests, these are very
straight-forward and can be quickly performed, and provide useful
information about the stage of infection. Indeed, in the case of
negativity, only the control band is colored. In the case of an
ongoing infection, the IgM and control bands are stained. In the
case of a recent infection, both the IgM and IgG bands are stained,
since both antibodies are present in the bloodstream. Following the
remission of the disease, for a period of time that varies from
patient to patient, only the IgG band is positive together with the
control one, and in the case of re-infection, all the three bands
are positive again (Fig. S1).
The test is considered invalid when none of the lines stain or when
the test lines are stained but the control line is not (81).
The limited sensitivity and the related high
false-negative results are mainly related to the low viral load and
the low antibody response observed in some patients; however, as
already mentioned, the probability of obtaining a positive test
depends also on the time of the presumed infection and on the test
execution time. Indeed, although the viral antigens are found in
the samples after a short time from infection, their permanence and
stability in the biological sample are limited; therefore, it is
not always possible to correctly identify these proteins (82). Similarly, rapid antibody tests
are mainly designed to identify IgM and IgG antibodies, which are
not produced by the body immediately, but begin to be found in the
bloodstream after the third week of the suspected infection.
Therefore, it is important to use the most appropriate test
considering the time of the presumed infection. More recently, the
use of rapid tests for the detection of IgA to accelerate the
diagnosis of COVID-19 infection has been proposed (83).
Overall, rapid antigen and rapid antibody tests are
widely used for screening strategies on large portions of the
population (70,84); however, they do not ensure a
precise diagnosis of COVID-19. Therefore, these tests should be
always confirmed by RT-PCR analyses. Details of rapid antigen and
rapid anti-body tests currently approved by international agencies
are reported in Table SII.
5. Immunoenzymatic serological tests
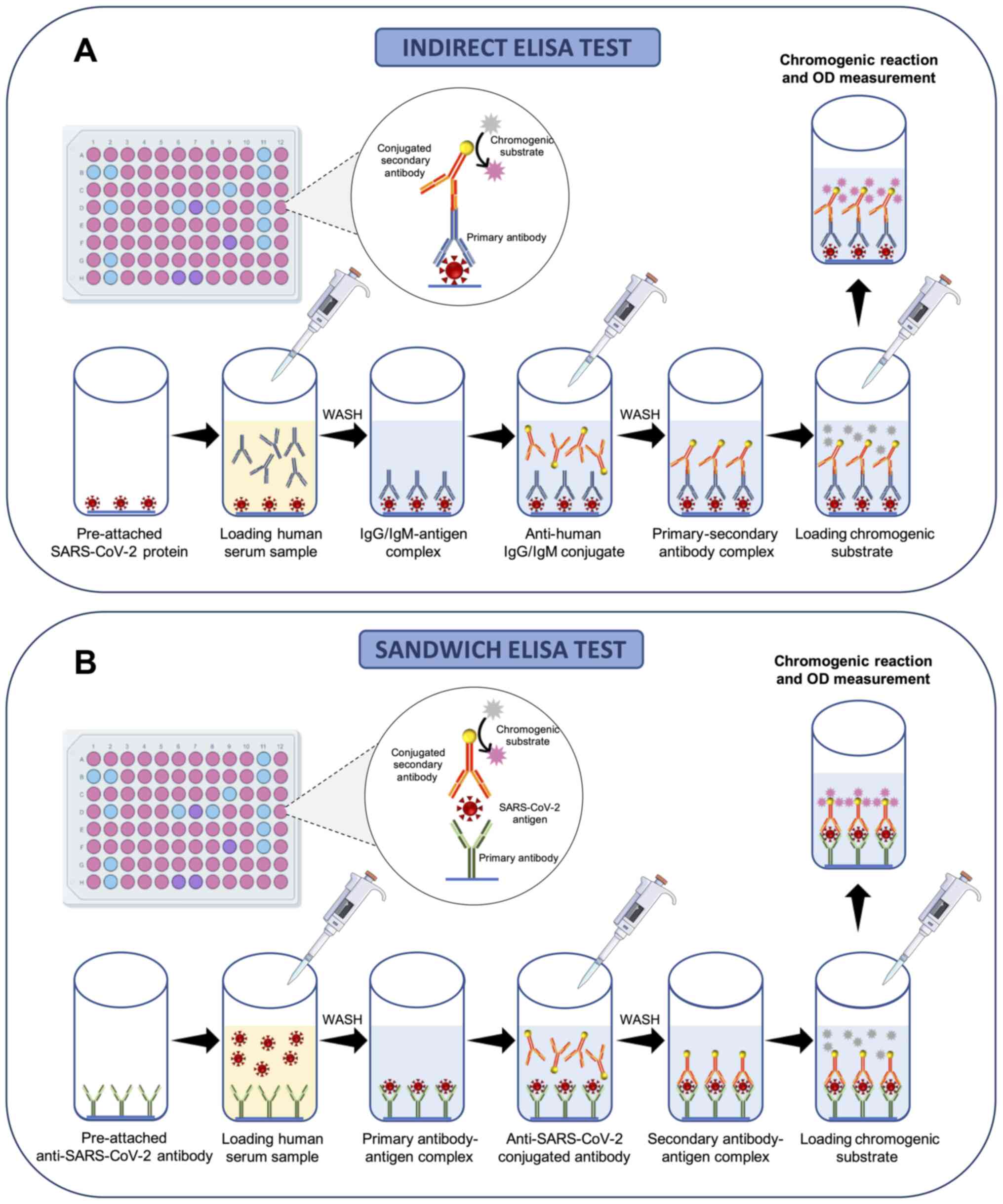
Most of the immunoenzymatic serological tests used
for COVID-19 investigations are based on the principle of indirect
enzyme-linked immunosorbent assay (ELISA). Of note, ELISA is a
colorimetric, chemiluminescent or fluorescent microwell plate-based
assay used for the quantitation and detection of human proteins,
immunoglobulins, antigens and other peptides through the binding
between the target protein and a specific antibody that results in
a detectable signal (85). This
technique allows researchers to obtain highly specific and
sensitive results in a relatively short time ranging from 1 to 5 h
(85).
Briefly, 96-well commercial COVID-19 ELISA indirect
tests contain immobilized viral antigens at the bottom of each well
that are recognized and bound by anti-SARS-CoV-2 antibodies present
in the serum of patients following proper dilutions. A series of
washes is then made to remove the serum and unbounded antibodies;
subsequently, a conjugated anti-body specific for human
immunoglobulins is added to each well. Following further washing, a
chromogenic (or fluorescent or chemiluminescent) substrate is
finally added and metabolized in the presence of antibodies against
SARS-CoV-2 and enzyme-conjugated anti-human antibodies. This
process gives rise to a colorimetric reaction easily detectable by
optical densitometry (or fluorescence or chemiluminescence) whose
intensity is indicative of the quantity of IgG, IgM or IgA
anti-body presents in the sample (Fig. 6A).
Similarly, 96-well commercial COVID-19 ELISA
sandwich tests contain immobilized antibodies against SARS-CoV-2
antigens at the bottom of each well able to bind antigens contained
in the serum samples of patients. Subsequently, a series of washes
is performed to remove the serum and unbounded antigens and a
conjugated antibody specific for SARS-CoV-2 antigens is added to
each well. Finally, following further washing, a chromogenic
substrate is added and metabolized in the case of positive samples
giving rise to a colorimetric reaction (Fig. 6B).
As regards the management of the COVID-19 pandemic,
ELISAs are currently used for the detection of IgM and IgG
antibodies (86) specific for
SARS-CoV-2 antigens or for the identification of viral Spike
proteins (87). More recently,
other ELISAs have been developed for the detection of human IgA
antibodies whose determination is of fundamental importance as they
are the first antibodies to be produced following exposure to the
virus (86).
In a very short period of time, several COVID-19
ELISAs have been developed and some of these have been approved by
international agencies (Table
SIII). These tests are mainly adopted to monitor the
immunological status of patients or for the immunosurveillance of
specific work categories, such as healthcare workers or school
personnel (88,89). In particular, ELISA tests for the
detection of IgG and IgM are often performed on COVID-19 patients
who obtained a negative result in molecular tests conducted on
nasopharyngeal swabs in order to ascertain the seroconversion of
patients and the acquisition of immunocompetence against COVID-19
infection (90). Contrariwise,
ELISAs for the detection of viral proteins and anti-SARS-CoV-2
human IgA can be used for diagnostic purposes or for large
screening strategies, as these molecules are rapidly founded in
clinical samples (83,91). Of note, ELISAs for the detection
of viral antigens are based on sandwich ELISA instead of indirect
ELISA (92).
Several ELISAs have been approved for the management
of COVID-19 infection, allowing the identification of individuals
exposed to the SARS-CoV-2 virus (Table SIII); however, these tests are
not able to confirm the infectious status of the subject. Indeed,
in the case of an ELISA-positive result (either IgA, IgM, IgG or
viral antigens), it is necessary to perform molecular confirmatory
analyses on nasopharyngeal swabs (93). However, ELISA-based serological
tests are much more reliable than rapid antigen or antibody tests;
the sensitivity and specificity values of these tests range from
75.6 to 100%, and 85.7 to 100%, respectively, albeit important
variations in these values may be related to both the manufacturer
or the human antibody or viral antigen tested (94). Indeed, generally, the search for
IgG is more accurate compared to that of IgM or IgA (this latter is
less sensitive) (94). Of note,
both sensitivity and specificity depend on the timing of infection
and the timing of test execution.
Overall, SARS-CoV-2 ELISAs represent a good clinical
option for large screening and surveillance campaigns mainly
adopted for specific work categories due to the rapidity of this
method, the possibility of analyzing multiple samples in one round
and the availability of automated or semi-automated systems that
allow a precise quantitation of viral antigens or human
antibodies.
6. Alternative methods for the effective
diagnosis of COVID-19 infection
The COVID-19 pandemic has prompted research groups
worldwide to develop novel technologies for the diagnosis of
COVID-19 infection or to readapt existing diagnostic systems
according to the characteristics of the new SARS-CoV-2 virus.
When the infection had not yet assumed the dimension
of a global pandemic, traditional culture methods, clinical
investigations and NGS techniques were among the first methods used
to diagnose COVID-19 infection. Apart from these approaches, other
methods have been developed, including biosensors, CRISPR/Cas-based
tests, nucleic acid isothermal amplification methods, electron
microscopy, etc. (25,95).
Viral culture and electron
microscopy
Viral culture has represented the fundamental method
that allows the identification of SARS-CoV-2 as a novel causative
agent of human pneumonia (96).
Despite the difficult realization and the long period of times
necessary to obtain a viral culture in vitro, viral isolates
represent a milestone for the discovery of novel viral infections
(96,97). As regards SARS-CoV-2 infection,
viral culture was fundamental in the initial phase of the outbreak
prior to the development of other diagnostic assays. Zhu et
al (96) (2020) were the
first to obtain SARS-CoV-2 viral isolates from clinical specimens
and to observe cytopathic effects using transmission electron
microscopy. Briefly, the authors of that study inoculated 150
µl of BALF supernatant obtained from a COVID-19 patient into
pathogen-free human airway epithelial cells. Following 2 h of
incubation at 37°C, infected epithelial cells were washed with
phosphate-buffered saline and incubated at 37°C for a long time
period. To assess the efficacy of infection and the production of
novel viral particles, the authors of that study collected cells
supernatant every two days for molecular analyses and observed
cytopathic effects under light microscopy. Finally, infected cells
were prepared for electron microscopy observation (96).
Subsequently, following the study by Zhu et
al (96), other research
groups isolated SARS-CoV-2 with an aim to study the structural
features and molecular interaction with infected cells (98,99). For these purposes, other cell
lines have been used, including the Vero and LLC-MK2 cell lines; by
using electron microscopy and cells infected with clinical
specimens obtained from COVID-19 patients, it was possible to
identify the ultrastructural details of the virus, the interaction
between virus and cells and the resulting cytopathic effects
(100).
Of note, electron microscopy is one of the
pioneering methods for the discovery of novel pathogens, allowing
the identification of their structural features. As regards viral
infections, two main applications of electron microscopy exist:
Solid-phase immune electron microscopy (SPIEM) and immunolabeling
electron microscopy (IEM), which are based on the observation of
cells blocked in the surface of a grid and on the observation of
antibody-antigen complex occurring in infected cells, respectively
(101,102).
Overall, viral culture and electron microscopy are
important techniques used to observe the main characteristics of
the virus. In the case of SARS-CoV-2, these two methods allowed the
identification of the typical structure of coronaviruses
characterized by a nucleocapsid enclosed within a crown-like
envelope composed of spike proteins. As regards the cytopathic
effects, both methods displayed a broad range of cellular
alterations mainly represented by the formation of plaques
characterized by a net-like structure or fused cells. These
plaques, composed of multinucleated syncytial cells, also present
deformed cilia with a granular formation and a disordered polarity.
SARS-CoV-2-infected cells also exhibited double-membrane vesicles
and degraded mitochondria. Finally, viral infections also led to
the expansion of the endoplasmic reticulum and an increased number
of secretory vesicles (96).
Overall, these techniques have made it possible to
establish the main characteristics of the novel SARS-CoV-2 virus,
which then allowed researchers to develop the diagnostic systems
currently used, as well as to propose the first therapeutic
approaches for the treatment of COVID-19 infection. Despite the
importance of both viral culture and electron microscopy, both
methods present some issues that limit their use in clinical
settings. Indeed, viral culture is time-consuming and requires
specific equipment and high biosafety levels. For these reasons,
the CDC recommends SARS-CoV-2 viral culture only for research
studies carried out in laboratories equipped with level 3 biosafety
cabinets (103). On the other
hand, electron microscopy is not widely used as it requires costly
instruments and highly trained personnel with specific skills in
sample preparation and electron microscopy image analysis. In
addition, this technique is characterized by low diagnostic
sensitivity and specificity and optimal results can be obtained
only if appropriate viral cultures are available (104).
NGS
Apart from viral culture and electron microscopy,
NGS has represented a key method for the identification of
SARS-CoV-2 and for the development of almost all of the currently
adopted molecular diagnostic methods. Through NGS, it was possible
to fully characterize the entire genome of SARS-CoV-2, thus
establishing that it belongs to the β-coronavirus genus (49). The de novo sequencing of
SARS-CoV-2 was performed using the nanopore technology through a
sequence-independent single-primer amplification approach (105,106).
At present, NGS is not used for diagnostic purposes,
but for molecular epidemiology and for the discovery of novel
molecular variants. Indeed, its diagnostic application is limited
due to the high costs of the analysis, the requirement of expensive
technologies and the need for highly trained personnel with
molecular and bioinformatics skills (25). Despite the high procedural costs,
some companies have proposed commercial tests for the sequencing of
SARS-CoV-2 through NGS platforms. In particular, the commercial
kits available are mainly based on NGS techniques coupled with
hybrid capture methods (BioCat, Arbor Biosciences and Swift tests).
These platforms are built with biotinylated RNA probes that
hybridize SARS-CoV-2 RNA fragments. Subsequently, the biotinylated
probes are amplified through PCR using streptavidin-coated beads
(107,108).
Apart from these platforms, more complex NGS tools
have been developed to detect mutations in the sequence of
SARS-CoV-2 genome, thus identifying novel emerging strains
important from an epidemiological point of view and for the
development of novel vaccines. Among these tools, amplicon-based
metagenomic sequencing represents the most powerful approach with
which to rapidly identify and comprehensively characterize
SARS-CoV-2 and other pathogens. In particular, metagenomic
sequencing allows the identification of the normal microbiome of
patients, while amplicon-based sequencing allows the amplification
and the subsequent sequencing of SARS-CoV-2 viral RNA. Together,
amplicon-based sequencing and metagenomic sequencing are able to
correctly diagnose COVID-19 infection, thus also identifying
secondary infections due to other pathogens aggravating the health
status of patients (109).
As already mentioned, amplicon-based and metagenomic
approaches based on the sequence-independent single primer
amplification (SISPA) method are able to detect mutations occurring
in the sequence of SARS-CoV-2 and potentially associated with
vaccine inefficacy or resistance to antiviral therapies (110,111). Examples of this type of
sequencing technique are the MinION and IDbyDNA platforms produced
by Oxford Nanopore Technologies and Illumina, respectively
(112). As regards the IDbyDNA
platform, it ensures the collection of >13 million reads of
which >8 million are unique reads with an average length of ~75
bp and an in-depth coverage. This shotgun sequencing allows the
generation of high-quality library score and Q score ensuring the
correct identification of single variants in the SARS-CoV-2
sequence (113).
Similarly, MinION technology ensures the collection
of millions of short and ultra-long reads (4,000 bp in length)
obtaining output data up to 30 Gb. This technology is based on a
portable platform that allows the real-time analysis of clinical
samples with limited costs (114). This technology was effectively
used for the analysis of the SARS-CoV-2 genome by using primers for
16 conserved binding sites of coronavirus allowing the
reconstruction of the whole genome of SARS-CoV-2 through the
generation of 1,000 bp reads with overlapping regions each other
(75).
Overall, NGS whole-genome sequencing is the most
powerful method for the molecular characterization of SARS-CoV-2,
for the identification of novel variants during genomic
surveillance screening and for the development of genome-based
therapeutic approaches (115-117).
Clinical investigations and imaging
techniques
During the early stages of the infection, when the
causative agent was still unknown, the diagnosis of COVID-19 was
predominantly clinical based on the observation of the patient's
respiratory and extra-respiratory symptoms and on the use of
radiological imaging techniques (118,119).
Of note, a significant fraction (~50-75%) of
COVID-19 patients is asymptomatic or paucisymptomatic, presenting
mild symptoms for a limited period of time, while other patients
(~10%) present severe respiratory symptoms resulting in acute
respiratory distress syndrome (ARDS) responsible for dyspnea,
interstitial pneumonitis, multiorgan dysfunctions and in some
cases, even death (120,121).
Such severe manifestations are often observed in patients with
pre-existing comorbidities, such as diabetes, cardiovascular
diseases, hypertension and cancer, and are mediated by specific
host cell entry mediators (122-126).
Among the COVID-19 symptoms, there are not only
respiratory manifestations, but also other systemic symptoms.
According to a recent comprehensive review, the main respiratory
symptoms include, but are not limited to dyspnea (19-64%), cough
(69-82%), rhinorrhea (4-24%) and a sore throat (5-14%). Other
common symptoms are fever (44-98%), headaches (5-14%) and diarrhea
(2-5%) (127). However, the
most common symptoms reported by patients with COVID-19 are anosmia
and ageusia, together with fatigue (128,129).
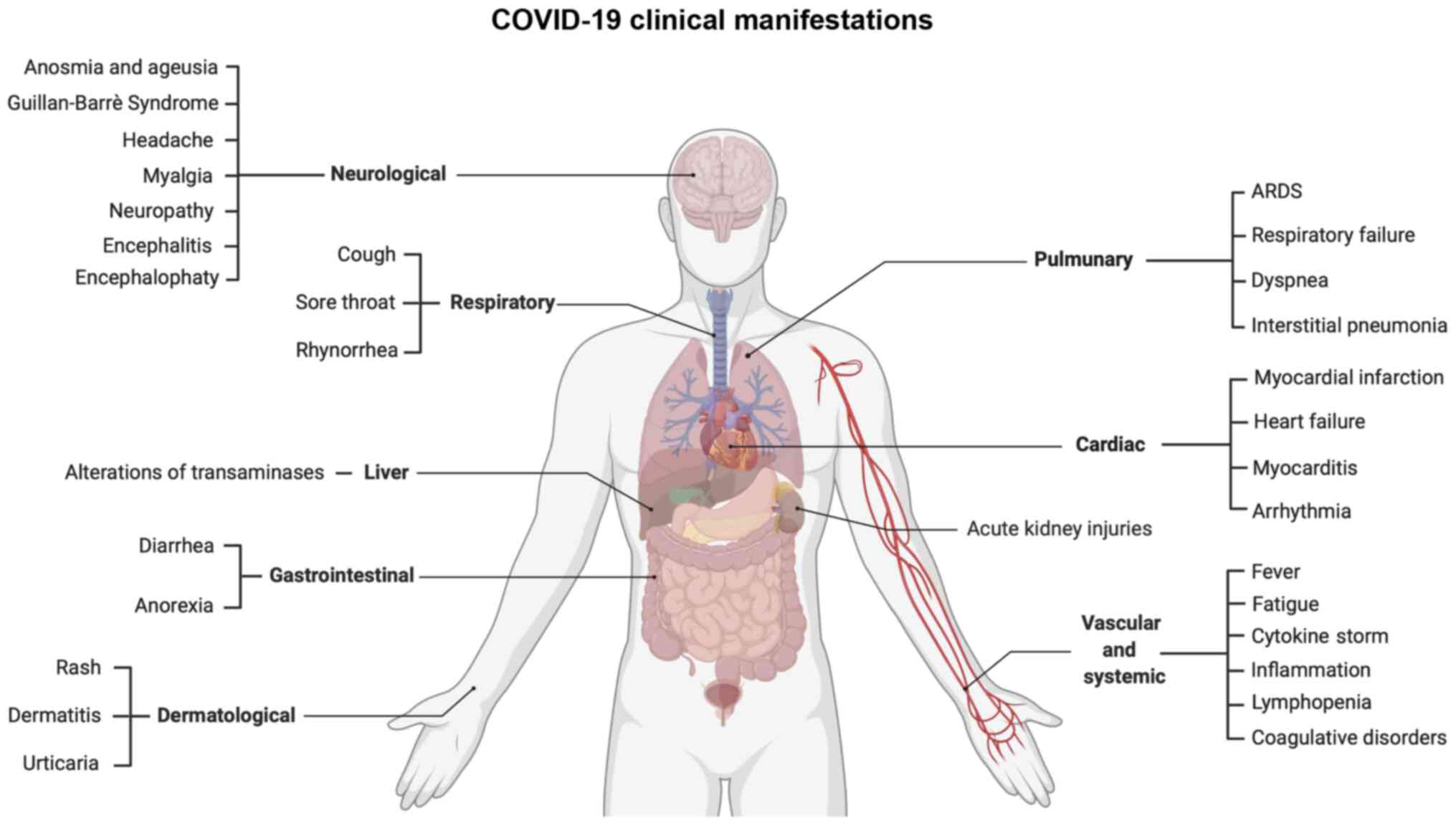
Apart from these commonly reported symptoms, a
plethora of clinical manifestations has been reported, ranging from
cardiovascular to neurological disorders and from gastrointestinal
to systemic symptoms (Fig. 7).
In particular, SARS-CoV-2 exhibits a neuroinvasive behavior via the
retrograde transsynaptic invasion of the central nervous system.
SARS-CoV-2 is able to bind specific receptors in the cells
constituting the olfactory bulb or pulmonary and airways sensorial
receptors. Among the most frequent neurological symptoms, there are
headaches, neuropathy, myalgia, encephalitis, encephalopathy, etc.
(130).
Other common symptoms are related to systemic
inflammation responsible for the alteration of coagulative and
hematological parameters and for the so-called 'cytokine storm'
observed in patients with COVID-19 with severe respiratory
symptoms. In fact, inflammation leads to the increase of
fibrin/fibrinogen debris and D-dimer associated with coagulopathy.
In rare cases, such alterations may induce disseminated
intravascular coagulopathy that requires anticoagulant prophylactic
or curative treatments (131).
In addition, inflammation is also responsible for lymphopenia and
T-cell exhaustion (132).
Strictly related to these hematological alterations are
cardiovascular disorders, such as acute myocardial injury, coronary
syndrome, cardiomyopathy, myocarditis, arrhythmias, etc. (133).
Among gastrointestinal disorders, COVID-19 infection
is responsible for nausea, vomiting, weight loss, anorexia and more
frequently, diarrhea. Among these symptoms, hepatic manifestations
mainly represented by increasing levels of transaminases are also
included (134). Finally, other
symptoms affect the kidneys, skin, endocrine system, etc. (135).
Overall, COVID-19 symptoms are mild in 80-90% of
positive individuals; however, a small fraction of patients
experiences severe symptoms that require hospitalization.
Approximately 5% of patients develop interstitial pneumonia
associated with respiratory failure, cytokine storm and multiorgan
failure that could lead to patient death (127,136).
Therefore, the careful clinical evaluation of all
these symptoms, together with radiological and laboratory data,
helps the clinicians to correctly diagnose COVID-19 infection, and
thus to the timely commencement of effective therapeutic protocols
(24).
As already mentioned, radiological investigations
help clinicians to correctly diagnose COVID-19 infection in the
case of a suspicious case of pneumonia. Among radiological imaging
techniques, chest X-ray (CXR) and computed tomography (CT) are the
most powerful methods for the diagnosis of COVID-19 pneumonia
(137,138).
CXR is usually used for the detection of pulmonary
abnormalities following lung injuries due to infectious or
neoplastic diseases (139).
During the first phase of the COVID-19 outbreak, CXR was widely
used to detect multifocal opacities affecting mainly lung
interstitial space and alveoli in patients with COVID-19-related
symptomatology (140). In
particular, CXR is mainly adopted for patients suspected of
COVID-19 infection with moderate or severe symptomatology who
usually exhibit interstitial opacities (71.7%), or alveolar
opacities (60.5%) frequently affecting both lungs (64.5%) (141). These radiological findings
become more severe over time with the progression of symptoms and
are mostly found in elderly patients with previous pulmonary
parenchyma alterations (such as patients with chronic obstructive
pulmonary disease) who exhibit both bilateral interstitial and
alveolar opacities (142).
Despite the low cost and the rapid radiological
findings obtained through CRX, some pulmonary abnormalities are not
clearly detectable through this technique. Therefore, besides CRX,
CT scan is frequently adopted to better display lung abnormalities
mainly represented by bilateral interstitial ground-glass opacities
(143). In particular, the CT
scan has a great resolution power with a sensitivity of ~95-100%,
although the specificity is very low, as this method does not allow
for the discrimination of pulmonary alterations associated with
other etiological agents other than SARS-CoV-2 (144).
The CT scan has been demonstrated to be an important
technique for the diagnosis of asymptomatic patients with COVID-19
negative for molecular tests due to low viral load (145,146). In fact, the CT scan is able to
discriminate the presence of morphological abnormalities in the
lung already during the early stages of infection; however, this
technique is not effective in correctly diagnosing a COVID-19
infection characterized by the absence of respiratory symptoms or
where there is no involvement of the lung parenchyma (147). Other limits of the CT scan are
represented by the needs of two radiologists' evaluations and the
risk of cumulative radiations. This latter issue is addressed
through the use of low-dose CT scan or ultra-low-dose CT scan often
used for the long-term monitoring of COVID-19 patients with severe
respiratory symptoms (148).
Overall, the radiological hallmarks of COVID-19
infection are the bilateral interstitial ground-glass opacities
that may also affect the peripheral areas and the alveolar
parenchyma. These alterations together with clinical symptoms, lead
clinicians to perform a diagnosis of COVID-19 infection.
Biosensor COVID-19 testing
techniques
To effectively diagnose COVID-19 infection rapidly
and directly at the point-of-care, several electrochemical
biosensing systems have been developed. These biosensor-based tools
use the principle of impedance and electrochemical reactions
occurring when viral RNA or proteins bind specific probes or
antibodies. Different types of biosensor platforms are currently
available for the diagnosis of COVID-19. These include
electrochemical biosensors, colorimetric biosensors,
fluorescence-based biosensors, surface-enhanced Raman scattering
(SERS) biosensors, quartz crystal microbalance (QCM) biosensors,
localized surface plasmon resonance (LSPR) and other platforms that
ensure the accurate and fast detection of SARS-CoV-2 particles
(149-152).
Among these platforms, the most commonly used are
electrochemical biosensors and SERS adopted as point-of-care
platforms due to the limited size of instruments, the low cost and
the easy execution of the procedure (153).
Electrochemical biosensors can be used for the
detection of SARS-CoV-2 proteins or for the detection of viral RNA.
Seo et al (154) (2020)
described an innovative biosensor for the detection of a low
concentration of SARS-CoV-2 spike protein (LoD, 1 fg/ml) built with
a graphene sensor with immobilized anti-SARS-CoV-2 spike
antibodies. This type of field-effect transistor biosensor (FET)
allowed the effective identification of SARS-CoV-2 in different
types of samples (PBS, transport medium, viral culture medium,
etc.) through the evaluation of electrical performance after S
protein-antibody interaction suggesting its application in clinical
settings (154).
As regards electrochemical systems for viral RNA
detection, Zhao et al (155) (2020) developed an
ultrasensitive electrochemical biosensor built with calixarene
functionalized graphene oxide containing specific probes for
SARS-CoV-2 RNA. This system allows the detection of viral RNA
without nucleic acid amplification systems through the use of
capture probes and label probes specific for SARS-CoV-2 RNA that
through a calixarene substrate are able to detect electrochemical
mediators, including toluidine blue and gold nanoparticles,
generated by the binding between probes and the viral RNA. The
device is portable and the analysis of data can be performed on
smartphone apps as a point-of-care testing (155).
Several other types of biosensors have been
produced; however, their detailed description is beyond the scope
of the present review. Overall, these biosensors exhibited a
sensitivity ranging from 86.43 to 93.75%, and a specificity of
90.63-100% depending on the platform used (156). As already mentioned, biosensors
present several advantages mainly represented by high sensitivity
and specificity, low costs of analysis, rapid execution time, the
optimal LoD and the possibility of developing miniaturized
platforms used directly in the point-of-care.
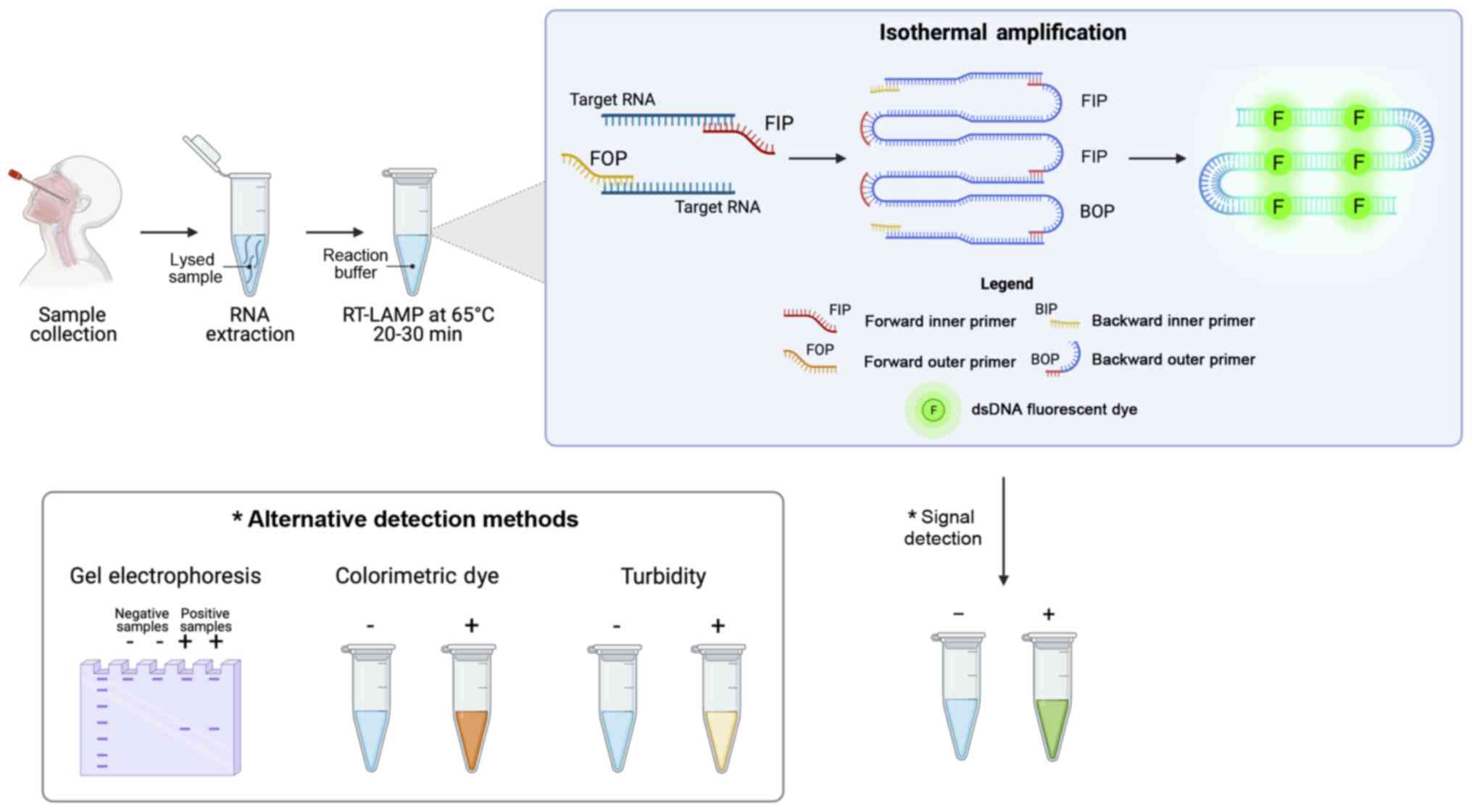
Loop-mediated isothermal amplification
(LAMP) COVID-19 testing methods
LAMP systems are among the most commonly used
alternative nucleic acid amplification methods for the diagnosis of
infectious diseases. These techniques are characterized by an easy
execution of the assay that does not require specific equipment, a
rapid and highly sensitive detection of targets and an easy
interpretation of results (157). As regards SARS-CoV-2, several
RT-LAMP systems have been developed for the effective detection of
viral RNA, highlighting how these methods can be coupled with other
diagnostic techniques including NGS, digital detection systems,
biosensors, etc. (158).
The SARS-CoV-2 RT-LAMP detection systems are based
on autocycling strand displacement DNA synthesis-mediated Bst DNA
polymerase and six primers, which work at isothermal temperatures
ranging from 60 to 65°C, thus avoiding the use of a costly PCR
thermocycler. RT-LAMP amplification is able to amplify RNA
fragments up to 106-109 copies in ~30-60 min. The final
amplification products can be visualized by agarose gel
electrophoresis, by fluorescence labeling, turbidity, or
colorimetry for an immediate readout of the data obtained and a
prompt diagnosis of COVID-19 infection (Fig. 8).
Overall, SARS-CoV-2 RT-LAMP tests have a high
sensitivity and specificity and some of these have been approved
for diagnostic purposes by different national and international
agencies. Some studies have reported a sensitivity of ~100% for the
detection of ORF1ab SARS-CoV-2 gene, highlighting a higher
diagnostic accuracy compared to other validated RT-PCR diagnostic
methods. In addition, the specificity rate is higher than that
obtained through RT-PCR, as the use of more than six primers in the
RT-LAMP ensures the correct diagnosis of SARS-CoV-2 infection
(25).
Among the most commonly used RT-LAMP methods there
is The ID NOW™ COVID-19 assay (Abbott Laboratories), which has been
approved by FDA with Emergency Use Authorization (EUA). This
RT-LAMP-based system ensures high-sensitive results in ~5 min
through the identification of the SARS-CoV-2 RdRp gene. Abbott ID
NOW™ and other similar systems represent effective point-of-care
testing, which have improved the management of COVID-19 infection
thanks to their rapid results and limited costs (159).
Through the development of novel integrated
diagnostic tools, RT-LAMP has been coupled with other diagnostic
systems improving their applicability in clinical settings. In
particular, Broughton et al (160) developed an RT-LAMP system
combined with CRISPR/Cas12 lateral flow assay, which ensures a
better display of the results and a significant reduction of the
execution times.
Similarly, other LAMP systems have been combined
with recombinase polymerase amplification (RPA) techniques,
resulting in a high-sensitive method termed as RAMP. In particular,
RAMP consists of two consecutive isothermal enzymatic steps that
are RPA and LAMP. Firstly, RPA is performed at 37°C for 20 min in
order to amplify all viral targets; subsequently, RPA amplicons are
detected through isothermal LAMP performed for 30 min at 65°C. The
amplification data are finally displayed by using colorimetric dyes
or are monitored in real-time by using intercalating fluorescent
dyes, such as SYBR-Green (161). At present, a RAMP detection
system is used in research contexts for the effective detection of
the SARS-CoV-2 ORF1ab gene with a sensitivity of 100% (162). If adequately validated, both
RT-LAMP and RAMP may represent an effective alternative to nucleic
acid amplification systems (163-165).
CRISPR/Cas-based COVID-19 testing
methods
The CRISPR/Cas system is a bacterial nucleoprotein
complex that confers resistance to external plasmids or phages
nucleic acids acting as a type of prokaryotic immune system. The
CRISPR/Cas system has revolutionized the field of molecular biology
allowing the development of genome editing, a technology that has
received the Nobel Prize 2020 for Chemistry (166,167). The applications and
potentialities of CRISPR/Cas systems are almost unlimited, ranging
from genome editing to diagnostics and from therapies to gene
regulation (168).
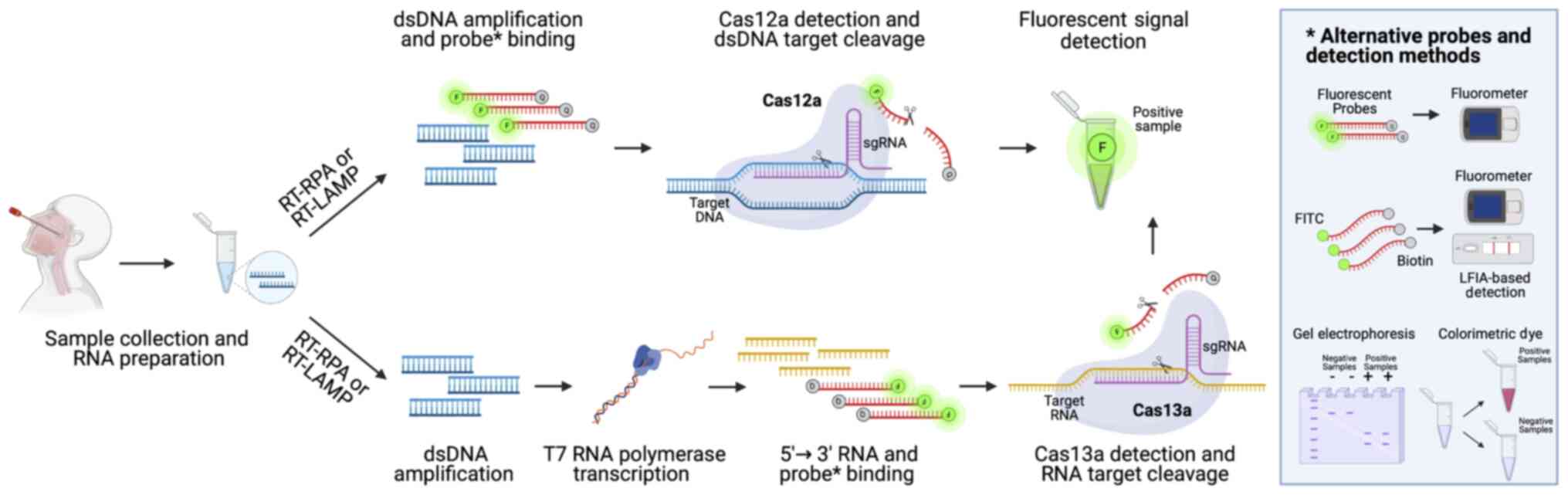
As mentioned in above, CRISPR/Cas technology has
been used for the effective diagnosis of COVID-19 associated with
LAMP isothermal amplification (169). The technology behind the
SARS-CoV-2 diagnostic CRISPR/Cas systems is very simple and based
on the use of CRISPR sequences with guide RNAs (gRNAs) specific for
SARS-CoV-2 viral RNA, Cas nuclease proteins, target nucleic acid
fragments and fluorescent probes or colorimetric dyes. At present,
two main CRISPR/Cas technologies are used for the effective
detection of the viral genome, i.e., CRISPR/Cas13a and
CRISPR/Cas12a that are able to bind and cleave RNA and DNA targets,
respectively. Other systems, not yet approved, but with a higher
level of accuracy, use also FnCas9 nuclease (170).
Following viral RNA extraction, the sample is
retrotranscribed into cDNA using RT-RPA or RT-LAMP, as described in
above [please see section entitled 'Loop-mediated isothermal
amplification (LAMP) COVID-19 testing methods']. Subsequently, the
obtained cDNA can be directly analyzed through CRISPR/Cas12a
detection system or can be transcribed into RNA using T7
transcriptase. In this latter case, the RNA obtained is then
processed through the CRISPR/Cas13a nucleoprotein complex able to
cleave labeled targeted viral RNA, thus allowing the detection of a
positive signal in case of COVID-19 infection. If the CRISPR/Cas12a
detection system is used, the double-strand DNA bound by a probe
labeled with a fluorescent reporter and a quencher is cleaved by
Cas12a generating a readable signal. The data generated by using
both CRISPR/Cas13a or CRISPR/Cas12a systems can be observed via
different methods by using electrophoresis gel, lateral flow strips
or instruments able to detect real-time fluorescent signals
occurring when probes are cleaved by Cas proteins (159) (Fig. 9).
Some SARS-CoV-2 CRISPR/Cas detection systems have
been already approved by international agencies and can be used as
point-of-care testing due to their smart execution that does not
require specific laboratory equipment, the rapidity of the
procedure and the low cost of the assay (171).
Among the most commonly used systems, there is the
Sherlock™ CRISPR SARS-CoV-2 assay (Sherlock Biosciences) that has
been approved by the US FDA for emergency use. This system based on
Cas13a nuclease activity is composed of two different stages. The
first one is an RT-LAMP, where viral RNA is reverse transcribed
into cDNA and amplified through a strand-displacing DNA polymerase
(LAMP). The second step consists of RNA transcription and
collateral cleavage mediated by the CRISPR/Cas13a complex able to
target a specific SARS-CoV-2 sequence. The cleavage of SARS-CoV-2
RNA results in a fluorescent signal easily detectable (172,173). This system has exhibited a more
rapid execution time compared with RT-PCR, with 100% sensitivity
and specificity to correctly diagnose COVID-19 in clinical samples,
a minimal footprint of instruments and a LoD of 6.75
copies/µl (174).
Other CRISPR/Cas-based systems have proven similar
accuracy. Hou et al (175) (2020) developed a CRISPR/Cas13a
system able to produce high-sensitive results within 40 min with a
LoD of 7.5 copies per reaction. Similarly, Curti et al
(176) (2020) developed an
ultrasensitive, rapid, and portable CRISPR/Cas12a-based assay for
the effective detection of SARS-CoV-2 RNA demonstrating a LoD of 10
copies/µl.
Overall, as shown for RT-LA MP methods,
CRISPR/Cas-based assays (176)
represent a promising alternative for the effective diagnosis of
COVID-19, ensuring high sensitivity and specificity rates, and low
costs of analysis.
Digital PCR COVID-19 testing methods
Digital PCR represents a technological evolution of
the currently adopted RT-PCR methods (177). Digital PCR has been widely used
for different applications, including mutational analysis, viral
load detection, microbiological investigations, copy number variant
analysis, single-cell analysis, analysis of liquid biopsy samples
and detection of low expressed targets (177-183).
Different digital PCR platforms have been developed
by different companies. Among these, droplet digital PCR (ddPCR)
represents one of the most accurate systems currently available for
the effective detection of viral infections. Of note, ddPCR (and
other digital PCR systems in general) was initially developed to
find application in microbiological diagnostics, especially in that
of the virus, where it is of fundamental importance to assess the
viral load of an infected individual in order to establish its
contagiousness and prognosis (177). Subsequently, ddPCR was applied
to various clinical fields and currently represents one of the most
sensitive and accurate methods for the diagnosis of numerous
pathologies, including COVID-19 infection (178,184,185).
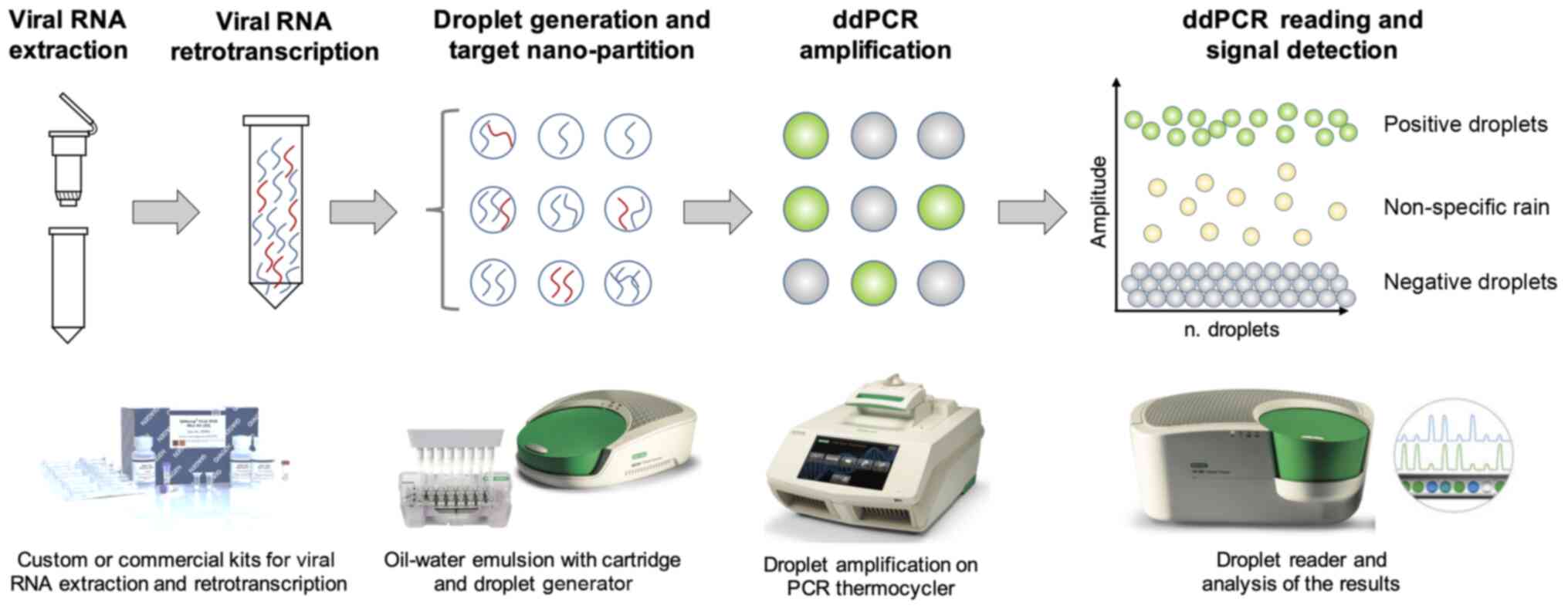
Of note, digital PCR uses the principles of the
sample micro-partitions and DNA ultra-dilutions performed on solid
supports or through the water-oil emulsion of the reaction mix.
This latter example is the principle of ddPCR, where the reaction
mix is nano-partitioned in >20,000 oil-water droplets containing
the target nucleic acid, specific primers and probes, Taq
polymerase and the amplification buffer necessary for the PCR
amplification. The workflow is similar to that observed for RT-PCR;
following sample collection, viral RNA is extracted through custom
or commercial protocols. Subsequently, viral RNA can be directly
processed in ddPCR by using one-step protocols or needs a previous
RT step to obtain cDNA. The ddPCR protocol consists of the
preparation of the reaction mix and the subsequent generation of
oil-water emulsion droplets performed through specific cartridges
and a droplet generator. The thousands of droplets thus generated
are subsequently amplified through a classic RT-PCR amplification
protocol which amplifies target DNA contained in each individual
droplet resulting in hundreds of separate amplification reactions
in a single well. Finally, the amplified droplets are read through
a droplet reader which uses capillary tubes where the droplets flow
separately and are excited by a laser beam that in case of
positivity will generate a fluorescent emission signal detected by
a CCD camera (186) (Fig. 10).
As regards SARS-CoV-2 RNA detection, ddPCR ensures
a greater sensitivity, specificity and accuracy compared to other
molecular systems, including RT-PCR (178,187). Indeed, the elevated
false-negative rate and the lack of availability of quantitative
RT-PCR methods significantly limit the diagnostic potential of
RT-PCR. Conversely, ddPCR is able to accurately assess SARS-CoV-2
viral load, thus allowing clinicians to effectively diagnose
COVID-19 infection, particularly in asymptomatic or
paucisymptomatic patients (188). In addition, ddPCR may be used
for the monitoring of COVID-19 patients, thus allowing the
discharge of patients who have resolved the infection and to
guarantee correct social isolation measures useful for limiting the
spread of infections (189).
Apart from the advantages in terms of sensitivity
and specificity, ddPCR has been proven to have a higher robustness
compared to RT-PCR, also in terms of stability and reproducibility
of the method. Indeed, RT-PCR efficiency is affected by several
pre-analytical and analytical biases due to the source of
specimens, the storage of samples, the sampling timing, the
sampling modalities, the extraction protocols and the nucleic acid
quality (38,64). Conversely, ddPCR has exhibited a
lower LoD compared to RT-PCR and it is also less susceptible to PCR
interferers (178,190).
The better accuracy of the ddPCR translates into
the optimal performance in terms of LoD. Indeed, in an experimental
setting ddPCR has an LoD of 2.1 and 1.8 copies/reaction for ORF1ab
and N SARS-CoV-2 genes, respectively, compared to RT-PCR, which has
an LoD of 1,039 copies/reaction for ORF1ab and 873.2
copies/reaction for N gene. Overall, ddPCR has a 500-fold greater
sensitivity compared to RT-PCR (188).
These data suggest how ddPCR may be considered a
turning point method for the effective diagnosis of COVID-19
infection, due to its greater sensitivity, specificity and accuracy
compared to other existing molecular methods.
7. Conclusions
The COVID-19 pandemic has highlighted the
importance of laboratory diagnostics in the management of a health
emergency that has deeply affected the social, economic and health
fabric of the entire world. At present, nucleic acid amplification
methods represent the gold standard for the diagnosis of COVID-19
infection with several RT-PCR-based tests approved by different
national and international regulatory agencies. These tests,
together with clinical and radiological investigations, have
significantly improved the ability to correctly and rapidly
diagnose a COVID-19 infection compared to the first diagnostic
methods mainly represented by viral culture analysis and de
novo sequencing of the SARS-CoV-2 genome. However, despite the
high sensitivity of RT-PCR, patients with a low viral load are not
often correctly diagnosed. In addition, this technique requires
confirmatory analyses, trained personnel and expensive instruments
and reagents that limit its application, especially in low-income
countries. For these purposes, other methods have been developed to
overcome the limitations of RT-PCR. Among these methods,
CRISPR/Cas-based assays and isothermal amplification methods
represent promising low-cost diagnostic strategies that can be used
for the effective diagnosis of COVID-19 infection in middle- and
low-income countries. Moreover, more sensitive molecular methods,
including ddPCR and biosensors, are beginning to be approved by
international agencies and used for the diagnosis of COVID-19
infection, as well as for the monitoring of viral load in
hospitalized patients or quarantined individuals.
Apart from these widely used diagnostic strategies,
other techniques have been developed for the screening of
SARS-CoV-2 infection in the population or in high-risk
environments, such as hospitals and schools. Among these, rapid
antigen and rapid antibody tests and immunoenzymatic serological
tests represent the most used techniques for the monitoring of
COVID-19 infection spreading. Of note, despite the low-cost of such
techniques, the low sensitivity and specificity of LFIAs and ELISAs
do not allow clinicians to formulate a precise diagnosis of
COVID-19 infection that should be confirmed with other techniques.
However, the use of such point-of-care testing made it possible to
implement effective health surveillance systems that allowed the
effective management of the COVID-19 pandemic thus limiting the
number of infections.
At present, other techniques are essential for the
genomic surveillance of the pandemic. Indeed, NGS techniques and
SARS-CoV-2 whole-genome sequencing are fundamental for the precise
characterization of novel genetic variants of SARS-CoV-2. However,
the high costs of these methods limit their application in clinical
practice.
Overall, in a short period of time, the scientific
community has developed several methods useful for correctly
diagnosing a suspected case of COVID-19 infection. However, in
order to perform a correct diagnosis of COVID-19, it is necessary
to take into account, not only the test to be used, but also the
patient's medical history, the time of the suspicious SARS-CoV-2
exposure, the type of sample to be collected and analyzed and how
to interpret the result. Only by integrating all these elements, it
will be possible to formulate a correct diagnosis of COVID-19
infection and effectively manage the COVID-19 pandemic.
Supplementary Data
Availability of data and materials
The datasets used and/or analyzed during the
current study are available from the corresponding author on
reasonable request. The original contributions/references of
studies presented in the study are publicly available. These data
can be found at: www.pubmed.com and https://www.who.int.
Authors' contributions
LF, DAS and ML conceptualized the study. LF and GG
wrote and prepared the draft of the manuscript. AT, DAS and ML
provided critical revisions. LF and GG drew the figures and prepare
the tables. LF and ML confirm the authenticity of all the raw data.
All authors contributed to manuscript revision and approved the
final version of the manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
DAS is the Editor-in-Chief for the journal, but had
no personal involvement in the reviewing process, or any influence
in terms of adjudicating on the final decision, for this article.
The other authors declare that they have no competing
interests.
Acknowledgments
Not applicable.
Funding
No funding was received.
References
|
1
|
Bontempi E: The europe second wave of
COVID-19 infection and the Italy 'strange' situation. Environ Res.
193:1104762021. View Article : Google Scholar
|
|
2
|
Cacciapaglia G, Cot C and Sannino F:
Second wave COVID-19 pandemics in Europe: A temporal playbook. Sci
Rep. 10:155142020. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
COVID-19 Dashboard by the Center for
Systems Science and Engineering (CSSE) Jonhs Hopkins University
(JHU). https://gisanddata.maps.arcgis.com/apps/opsdashboard/index.html#/bda7594740fd40299423467b48e9ecf6.
Accessed February 27, 2021.
|
|
4
|
Polack FP, Thomas SJ, Kitchin N, Absalon
J, Gurtman A, Lockhart S, Perez JL, Pérez Marc G, Moreira ED,
Zerbini C; C4591001 Clinical Trial Group; et al: Safety and
Efficacy of the BNT162b2 mRNA Covid-19 Vaccine. N Engl J Med.
383:2603–2615. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
European Medicine Agency (EMA): COVID-19
Vaccines. https://www.ema.europa.eu/en/human-regulatory/overview/public-health-threats/coronavirus-disease-covid-19/treatments-vaccines/covid-19-vaccines.
Accessed February 27, 2021.
|
|
6
|
Dong E, Du H and Gardner L: An interactive
web-based dashboard to track COVID-19 in real time. Lancet Infect
Dis. 20:533–534. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Pullano G, Di Domenico L, Sabbatini CE,
Valdano E, Turbelin C, Debin M, Guerrisi C, Kengne-Kuetche C, Souty
C, Hanslik T, et al: Underdetection of cases of COVID-19 in France
threatens epidemic control. Nature. 590:134–139. 2021. View Article : Google Scholar
|
|
8
|
O'Dowd A: Covid-19: Avoid indoor mixing
over Christmas or risk third wave, warns iSAGE. BMJ. 371:m48322020.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Farsalinos K, Poulas K, Kouretas D,
Vantarakis A, Leotsinidis M, Kouvelas D, Docea AO, Kostoff R,
Gerotziafas GT, Antoniou MN, et al: Improved strategies to counter
the COVID-19 pandemic: Lockdowns vs. primary and community
healthcare. Toxicol Rep. 8:1–9. 2021. View Article : Google Scholar
|
|
10
|
Liu S and Pan C: Differentiating diagnosis
of COVID-19 or influenza in patients based on laboratory data
during flu season. EClinicalMedicine. 26:1005112020. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
García-Basteiro AL, Chaccour C, Guinovart
C, Llupià A, Brew J, Trilla A and Plasencia A: Monitoring the
COVID-19 epidemic in the context of widespread local transmission.
Lancet Respir Med. 8:440–442. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Wu AHB: Screening the General Population
for SARS-CoV-2 Virus and COVID-19 Antibodies: A Counterargument. J
Appl Lab Med. 5:1107–1110. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Rostami A, Sepidarkish M, Leeflang MMG,
Riahi SM, Nourollahpour Shiadeh M, Esfandyari S, Mokdad AH, Hotez
PJ and Gasser RB: SARS-CoV-2 seroprevalence worldwide: a systematic
review and meta-analysis. Clin Microbiol Infect. 27:331–340. 2021.
View Article : Google Scholar
|
|
14
|
Wells PM, Doores KJ, Couvreur S, Nunez RM,
Seow J, Graham C, Acors S, Kouphou N, Neil SJD, Tedder RS, et al:
Estimates of the rate of infection and asymptomatic COVID-19
disease in a population sample from SE England. J Infect.
81:931–936. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Ji T, Liu Z, Wang G, Guo X, Akbar Khan S,
Lai C, Chen H, Huang S, Xia S, Chen B, et al: Detection of
COVID-19: A review of the current literature and future
perspectives. Biosens Bioelectron. 166:1124552020. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Sanyaolu A, Okorie C, Marinkovic A,
Ayodele O, Abbasi AF, Prakash S, Ahmed M, Kayode D, Jaferi U and
Haider N: Navigating the diagnostics of COVID-19. SN Compr Clin
Med. Jul 25–2020.Epub ahead of print. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
European Commission (EC): Current
performance of COVID-19 test methods and devices and proposed
performance criteria. EC; Brussels: 2020, https://ec.europa.eu/docsroom/documents/40805.
Accessed January 2, 2021.
|
|
18
|
Taleghani N and Taghipour F: Diagnosis of
COVID-19 for controlling the pandemic: A review of the
state-of-the-art. Biosens Bioelectron. 174:1128302021. View Article : Google Scholar
|
|
19
|
Jung J, Jang H, Kim HK, Kim J, Kim A and
Ko KP: The Importance of Mandatory COVID-19 Diagnostic Testing
Prior to Release from Quarantine. J Korean Med Sci. 35:e3142020.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Winter AK and Hegde ST: The important role
of serology for COVID-19 control. Lancet Infect Dis. 20:758–759.
2020. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Vandenberg O, Martiny D, Rochas O, van
Belkum A and Kozlakidis Z: Considerations for diagnostic COVID-19
tests. Nat Rev Microbiol. 19:171–183. 2021. View Article : Google Scholar
|
|
22
|
Tromberg BJ, Schwetz TA, Pérez-Stable EJ,
Hodes RJ, Woychik RP, Bright RA, Fleurence RL and Collins FS: Rapid
scaling up of Covid-19 diagnostic testing in the United States -
The NIH RADx Initiative. N Engl J Med. 383:1071–1077. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Guglielmi G: The explosion of new
coronavirus tests that could help to end the pandemic. Nature.
583:506–509. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Tsatsakis A, Calina D, Falzone L, Petrakis
D, Mitrut R, Siokas V, Pennisi M, Lanza G, Libra M, Doukas SG, et
al: SARS-CoV-2 pathophysiology and its clinical implications: An
integrative overview of the pharmacotherapeutic management of
COVID-19. Food Chem Toxicol. 146:1117692020. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Alpdagtas S, Ilhan E, Uysal E, Sengor M,
Ustundag CB and Gunduz O: Evaluation of current diagnostic methods
for COVID-19. APL Bioeng. 4:0415062020. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Wu F, Zhao S, Yu B, Chen YM, Wang W, Song
ZG, Hu Y, Tao ZW, Tian JH, Pei YY, et al: A new coronavirus
associated with human respiratory disease in China. Nature.
579:265–269. 2020. View Article : Google Scholar : PubMed/NCBI
Erratum. Nature. 580:E72020.
|
|
27
|
Zhu N, Zhang D, Wang W, Li X, Yang B, Song
J, Zhao X, Huang B, Shi W, Lu R, et al: China novel coronavirus
investigating and research team: a novel coronavirus from patients
with pneumonia in China 2019. N Engl J Med. 382:727–733. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Kumar R, Nagpal S, Kaushik S and
Mendiratta S: COVID-19 diagnostic approaches: Different roads to
the same destination. Virusdisease. 31:97–105. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Brun AL, Gence-Breney A, Trichereau J,
Ballester MC, Vasse M, Chabi ML, Mellot F and Grenier PA: COVID-19
pneumonia: High diagnostic accuracy of chest CT in patients with
intermediate clinical probability. Eur Radiol. 31:1969–1977. 2021.
View Article : Google Scholar
|
|
30
|
Martinez RM: Clinical samples for
SARS-CoV-2 detection: Review of the early literature. Clin
Microbiol Newsl. 42:121–127. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Rao GG, Agarwal A and Batura D: Testing
times in Coronavirus disease (COVID-19): A tale of two nations. Med
J Armed Forces India. 76:243–249. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Jarvis KF and Kelley JB: Temporal dynamics
of viral load and false negative rate influence the levels of
testing necessary to combat COVID19 spread. medRxiv. View Article : Google Scholar
|
|
33
|
Rode OĐ, Kurolt IC, Puljiz I, Čivljak R,
Balent NC, Laškaj R, Tiljak MK, Mikulić R and Markotić A: Antibody
response and the clinical presentation of patients with COVID-19 in
Croatia: The importance of a two-step testing approach. Eur J Clin
Microbiol Infect Dis. 40:261–268. 2021. View Article : Google Scholar
|
|
34
|
Bisoffi Z, Pomari E, Deiana M, Piubelli C,
Ronzoni N, Beltrame A, Bertoli G, Riccardi N, Perandin F, Formenti
F, et al: Sensitivity, specificity and predictive values of
molecular and serological tests for COVID-19: a longitudinal study
in emergency room. Diagnostics (Basel). 10:6692020. View Article : Google Scholar
|
|
35
|
Krajewski R, Gołębiowska J, Makuch S,
Mazur G and Agrawal S: Update on serologic testing in COVID-19.
Clin Chim Acta. 510:746–750. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Guo L, Ren L, Yang S, Xiao M, Chang D,
Yang F, Dela Cruz CS, Wang Y, Wu C, Xiao Y, et al: Profiling early
humoral response to diagnose novel coronavirus disease (COVID-19).
Clin Infect Dis. 71:778–785. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Chirumbolo S: Swabbing Thoroughly People
for COVID-19 Positivity. Insights on the Main Bio-analytical and
Microbiology Bias and Concerns. Curr Microbiol. 77:3680–3684. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Lippi G, Simundic AM and Plebani M:
Potential preanalytical and analytical vulnerabilities in the
laboratory diagnosis of coronavirus disease 2019 (COVID-19). Clin
Chem Lab Med. 58:1070–1076. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Ora J, Puxeddu E, Cavalli F, Giorgino FM,
Girolami A, Chiocchi M, Sergiacomi G, Federici M and Rogliani P:
Does bronchoscopy help the diagnosis in COVID-19 infection? Eur
Respir J. 56:20016192020. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Tong Y, Bao A, Chen H, Huang J, Lv Z, Feng
L, Cheng Y, Wang Y, Bai L, Rao W, et al: Necessity for detection of
SARS-CoV-2 RNA in multiple types of specimens for the discharge of
the patients with COVID-19. J Transl Med. 18:4112020. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
McFee DRB: COVID-19 Laboratory Testing/CDC
Guidelines. Dis Mon. 66:1010672020. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Fu Y, Li Y, Guo E, He L, Liu J, Yang B, Li
F, Wang Z, Li Y, Xiao R, et al: Dynamics and correlation among
viral positivity, seroconversion, and disease severity in COVID-19:
a retrospective study. Ann Intern Med. 8:M20–M3337. 2020.
|
|
43
|
Mesoraca A, Margiotti K, Viola A, Cima A,
Sparacino D and Giorlandino C: Evaluation of SARS-CoV-2 viral RNA
in fecal samples. Virol J. 17:862020. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Ng SC and Tilg H: COVID-19 and the
gastrointestinal tract: More than meets the eye. Gut. 69:973–974.
2020. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Long DR, Gombar S, Hogan CA, Greninger AL,
O'Reilly-Shah V, Bryson-Cahn C, Stevens B, Rustagi A, Jerome KR,
Kong CS, et al: Occurrence and timing of subsequent severe acute
respiratory syndrome coronavirus 2 reverse-transcription polymerase
chain reaction positivity among initially negative patients. Clin
Infect Dis. 72:323–326. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Mahendiratta S, Batra G, Sarma P, Kumar H,
Bansal S, Kumar S, Prakash A, Sehgal R and Medhi B: Molecular
diagnosis of COVID-19 in different biologic matrix, their
diagnostic validity and clinical relevance: A systematic review.
Life Sci. 258:1182072020. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
da Silva SJR, Silva CTAD, Guarines KM,
Mendes RPG, Pardee K, Kohl A and Pena L: Clinical and Laboratory
Diagnosis of SARS-CoV-2, the Virus Causing COVID-19. ACS Infect
Dis. 6:2319–2336. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Li D, Zhang J and Li J: Primer design for
quantitative real-time PCR for the emerging Coronavirus SARS-CoV-2.
Theranostics. 10:7150–7162. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Chan JF, Kok KH, Zhu Z, Chu H, To KK, Yuan
S and Yuen KY: Genomic characterization of the 2019 novel
human-pathogenic coronavirus isolated from a patient with atypical
pneumonia after visiting Wuhan. Emerg Microbes Infect. 9:221–236.
2020. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Centers for Disease Control and Prevention
(CDC): CDC's Diagnostic Test for COVID-19 Only and Supplies.
https://www.cdc.gov/coronavirus/2019-ncov/lab/virus-requests.html.
Accessed January 5, 2021.
|
|
51
|
Centers for Disease Control and Prevention
(CDC): Research Use Only 2019-Novel Coronavirus (2019-nCoV)
Real-time RT-PCR Primers and Probes. https://www.cdc.gov/coronavirus/2019-ncov/lab/rt-pcr-panel-primer-probes.html.
Accessed January 5, 2021.
|
|
52
|
Dorlass EG, Monteiro CO, Viana AO, Soares
CP, Machado RRG, Thomazelli LM, Araujo DB, Leal FB, Candido ED,
Telezynski BL, et al: Lower cost alternatives for molecular
diagnosis of COVID-19: Conventional RT-PCR and SYBR Green-based
RT-qPCR. Braz J Microbiol. 51:1117–1123. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Neilan AM, Losina E, Bangs AC, Flanagan C,
Panella C, Eskibozkurt GE, Mohareb A, Hyle EP, Scott JA, Weinstein
MC, et al: Clinical Impact, Costs, and Cost-Effectiveness of
Expanded SARS-CoV-2 Testing in Massachusetts. Clin Infect Dis. Sep
18–2020.Epub ahead of print. View Article : Google Scholar
|
|
54
|
Smyrlaki I, Ekman M, Lentini A, Rufino de
Sousa N, Papanicolaou N, Vondracek M, Aarum J, Safari H,
Muradrasoli S, Rothfuchs AG, et al: Massive and rapid COVID-19
testing is feasible by extraction-free SARS-CoV-2 RT-PCR. Nat
Commun. 11:48122020. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
U.S Food & Drug: COVID-19 tests and
collection kits authorized by the FDA in 2020: Infographic.
https://www.fda.gov/medical-devices/coronavirus-covid-19-and-medical-devices/covid-19-tests-and-collection-kits-authorized-fda-2020-infographic.
Accessed January 8, 2021.
|
|
56
|
Fomsgaard AS and Rosenstierne MW: An
alternative workflow for molecular detection of SARS-CoV-2 - escape
from the NA extraction kit-shortage, Copenhagen, Denmark, March
2020. Euro Surveill. 25:20003982020. View Article : Google Scholar
|
|
57
|
Udugama B, Kadhiresan P, Kozlowski HN,
Malekjahani A, Osborne M, Li VYC, Chen H, Mubareka S, Gubbay JB and
Chan WCW: Diagnosing COVID-19: The Disease and Tools for Detection.
ACS Nano. 14:3822–3835. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Ravi N, Cortade DL, Ng E and Wang SX:
Diagnostics for SARS-CoV-2 detection: A comprehensive review of the
FDA-EUA COVID-19 testing landscape. Biosens Bioelectron.
165:1124542020. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Wells CR, Townsend JP, Pandey A, Moghadas
SM, Krieger G, Singer B, McDonald RH, Fitzpatrick MC and Galvani
AP: Optimal COVID-19 quarantine and testing strategies. Nat Commun.
12:3562021. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Bustin SA and Nolan T: RT-qPCR Testing of
SARS-CoV-2: A Primer. Int J Mol Sci. 21:30042020. View Article : Google Scholar :
|
|
61
|
Böger B, Fachi MM, Vilhena RO, Cobre AF,
Tonin FS and Pontarolo R: Systematic review with meta-analysis of
the accuracy of diagnostic tests for COVID-19. Am J Infect Control.
49:21–29. 2021. View Article : Google Scholar
|
|
62
|
Kucirka LM, Lauer SA, Laeyendecker O, Boon
D and Lessler J: Variation in false-negative rate of reverse
transcriptase polymerase chain reaction-based SARS-CoV-2 tests by
time since exposure. Ann Intern Med. 173:262–267. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Afzal A: Molecular diagnostic technologies
for COVID-19: Limitations and challenges. J Adv Res. 26:149–159.
2020. View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Tahamtan A and Ardebili A: Real-time
RT-PCR in COVID-19 detection: Issues affecting the results. Expert
Rev Mol Diagn. 20:453–454. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Pan Y, Long L, Zhang D, Yuan T, Cui S,
Yang P, Wang Q and Ren S: Potential false-negative nucleic acid
testing results for severe acute respiratory syndrome coronavirus 2
from thermal inactivation of samples with low viral loads. Clin
Chem. 66:794–801. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Li Y, Yao L, Li J, Chen L, Song Y, Cai Z
and Yang C: Stability issues of RT-PCR testing of SARS-CoV-2 for
hospitalized patients clinically diagnosed with COVID-19. J Med
Virol. 92:903–908. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
67
|
World Health Organization (WHO):
Recommendations for laboratories testing by PCR for presence of
SARS coronavirus-RNA. WHO; Geneva: 2020, https://www.who.int/csr/sars/coronarec-ommendations/en/.
Accessed January 26, 2021.
|
|
68
|
Cerutti F, Burdino E, Milia MG, Allice T,
Gregori G, Bruzzone B and Ghisetti V: Urgent need of rapid tests
for SARS CoV-2 antigen detection: Evaluation of the SD-Biosensor
antigen test for SARS-CoV-2. J Clin Virol. 132:1046542020.
View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Olalekan A, Iwalokun B, Akinloye OM,
Popoola O, Samuel TA and Akinloye O: COVID-19 rapid diagnostic test
could contain transmission in low- and middle-income countries. Afr
J Lab Med. 9:12552020. View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Augustine R, Das S, Hasan ASA, Abdul Salam
S, Augustine P, Dalvi YB, Varghese R, Primavera R, Yassine HM, et
al: Rapid antibody-based COVID-19 mass surveillance: relevance,
challenges, and prospects in a pandemic and post-pandemic world. J
Clin Med. 9:33722020. View Article : Google Scholar :
|
|
71
|
Albert E, Torres I, Bueno F, Huntley D,
Molla E, Fernández-Fuentes MÁ, Martínez M, Poujois S, Forqué L,
Valdivia A, et al: Field evaluation of a rapid antigen test
(Panbio™ COVID-19 Ag Rapid Test Device) for COVID-19 diagnosis in
primary healthcare centres. Clin Microbiol Infect.
27:472.e7–472.e10. 2021. View Article : Google Scholar
|
|
72
|
Pilarowski G, Lebel P, Sunshine S, Liu J,
Crawford E, Marquez C, Rubio L, Chamie G, Martinez J, Peng J, et
al: Performance characteristics of a rapid SARS-CoV-2 antigen
detection assay at a public plaza testing site in San Francisco. J
Infect Dis. Jan 4–2021.Epub ahead of print. View Article : Google Scholar :
|
|
73
|
Lee JH, Choi M, Jung Y, Lee SK, Lee CS,
Kim J, Kim J, Kim NH, Kim BT and Kim HG: A novel rapid detection
for SARS-CoV-2 spike 1 antigens using human angiotensin converting
enzyme 2 (ACE2). Biosens Bioelectron. 171:1127152021. View Article : Google Scholar
|
|
74
|
Diao B, Wen K, Zhang J, Chen J, Han C,
Chen Y, Wang S, Deng G, Zhou H and Wu Y: Accuracy of a nucleocapsid
protein antigen rapid test in the diagnosis of SARS-CoV-2
infection. Clin Microbiol Infect. 27:289.e1–289.e4. 2021.
View Article : Google Scholar
|
|
75
|
Carter LJ, Garner LV, Smoot JW, Li Y, Zhou
Q, Saveson CJ, Sasso JM, Gregg AC, Soares DJ, Beskid TR, et al:
Assay Techniques and Test Development for COVID-19 Diagnosis. ACS
Cent Sci. 6:591–605. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
76
|
Huang C, Wen T, Shi FJ, Zeng XY and Jiao
YJ: Rapid detection of igm antibodies against the SARS-CoV-2 virus
via colloidal gold nanoparticle-based lateral-flow assay. ACS
Omega. 5:12550–12556. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
77
|
Mak GC, Cheng PK, Lau SS, Wong KK, Lau CS,
Lam ET, Chan RC and Tsang DN: Evaluation of rapid antigen test for
detection of SARS-CoV-2 virus. J Clin Virol. 129:1045002020.
View Article : Google Scholar : PubMed/NCBI
|
|
78
|
Lanser L, Bellmann-Weiler R, Öttl KW,
Huber L, Griesmacher A, Theurl I and Weiss G: Evaluating the
clinical utility and sensitivity of SARS-CoV-2 antigen testing in
relation to RT-PCR Ct values. Infection. 13:1–3. 2020.
|
|
79
|
Dinnes J, Deeks JJ, Adriano A, Berhane S,
Davenport C, Dittrich S, Emperador D, Takwoingi Y, Cunningham J,
Beese S, et al: Cochrane COVID-19 Diagnostic Test Accuracy Group:
Rapid, point-of-care antigen and molecular-based tests for
diagnosis of SARS-CoV-2 infection. Cochrane Database Syst Rev.
8:CD0137052020.
|
|
80
|
Montesinos I, Gruson D, Kabamba B, Dahma
H, Van den Wijngaert S, Reza S, Carbone V, Vandenberg O, Gulbis B,
Wolff F, et al: Evaluation of two automated and three rapid lateral
flow immunoassays for the detection of anti-SARS-CoV-2 antibodies.
J Clin Virol. 128:1044132020. View Article : Google Scholar : PubMed/NCBI
|
|
81
|
Prazuck T, Colin M, Giachè S, Gubavu C,
Seve A, Rzepecki V, Chevereau-Choquet M, Kiani C, Rodot V, Lionnet
E, et al: Evaluation of performance of two SARS-CoV-2 Rapid IgM-IgG
combined antibody tests on capillary whole blood samples from the
fingertip. PLoS One. 15:e02376942020. View Article : Google Scholar : PubMed/NCBI
|
|
82
|
Jacobs J, Kühne V, Lunguya O, Affolabi D,
Hardy L and Vandenberg O: Implementing COVID-19 (SARS-CoV-2) rapid
diagnostic tests in Sub-Saharan Africa: a review. Front Med
(Lausanne). 7:5577972020. View Article : Google Scholar
|
|
83
|
La Rosa Fabián C and Urquizo Briceño L:
Anti-SARS-Cov-2 IgA in Current Scenario of IgM and IgG Rapid Test:
A New Alternative for the Diagnostic of COVID-19. SN Compr Clin
Med. 26:1–3. 2020.
|
|
84
|
Canetti D, Dell'Acqua R, Riccardi N, Della
Torre L, Bigoloni A, Muccini C, Bruzzesi E, Ranzenigo M, Chiurlo M,
Racca S, et al: SARS-CoV-2 IgG/IgM Rapid Test as a Diagnostic Tool
in Hospitalized Patients and Healthcare Workers, at a large
Teaching Hospital in northern Italy, during the 2020 COVID-19
Pandemic. New Microbiol. 43:161–165. 2020.PubMed/NCBI
|
|
85
|
Alhajj M and Farhana A: Enzyme Linked
Immunosorbent Assay. StatPearls. StatPearls Publishing; Treasure
Island, FL: 2020
|
|
86
|
Tré-Hardy M, Wilmet A, Beukinga I,
Favresse J, Dogné JM, Douxfils J and Blairon L: Analytical and
clinical validation of an ELISA for specific SARS-CoV-2 IgG, IgA,
and IgM anti-bodies. J Med Virol. 93:803–811. 2021. View Article : Google Scholar
|
|
87
|
Freeman B, Lester S, Mills L, Rasheed MAU,
Moye S, Abiona O, Hutchinson GB, Morales-Betoulle M, Krapinunaya I,
Gibbons A, et al: Validation of a SARS-CoV-2 spike protein ELISA
for use in contact investigations and serosurveillance. bioRxiv.
View Article : Google Scholar
|
|
88
|
Korth J, Wilde B, Dolff S, Anastasiou OE,
Krawczyk A, Jahn M, Cordes S, Ross B, Esser S, Lindemann M, et al:
SARS-CoV-2-specific antibody detection in healthcare workers in
Germany with direct contact to COVID-19 patients. J Clin Virol.
128:1044372020. View Article : Google Scholar : PubMed/NCBI
|
|
89
|
Alharbi SA, Almutairi AZ, Jan AA and
Alkhalify AM: Enzyme-linked immunosorbent assay for the detection
of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2)
IgM/IgA and IgG antibodies among healthcare workers. Cureus.
12:e102852020.PubMed/NCBI
|
|
90
|
Stringhini S, Wisniak A, Piumatti G, Azman
AS, Lauer SA, Baysson H, De Ridder D, Petrovic D, Schrempft S,
Marcus K, et al: Seroprevalence of anti-SARS-CoV-2 IgG antibodies
in Geneva, Switzerland (SEROCoV-POP): A population-based study.
Lancet. 396:313–319. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
91
|
Vashist SK: In vitro diagnostic assays for
COVID-19: recent advances and emerging trends. Diagnostics (Basel).
10:2022020. View Article : Google Scholar
|
|
92
|
Tanaka Y, Kamimura K, Nakamura R,
Ohkoshi-Yamada M, Koseki Y, Mizusawa T, Ikarashi S, Hayashi K, Sato
H, Sakamaki A, et al: Usefulness of ultrasonography to assess the
response to steroidal therapy for the rare case of type 2b
immunoglobulin G4-related sclerosing cholangitis without
pancreatitis: A case report. World J Clin Cases. 8:5821–5830. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
93
|
AvŞar T, ÇaliŞ Ş, Yilmaz B, Demİrcİ
OtluoĞlu G, Holyavkİn C and Kili CT: Genome-wide identification of
Chiari malformation type I associated candidate genes and
chromosomal variations. Turk J Biol. 44:449–456. 2020. View Article : Google Scholar
|
|
94
|
La Marca A, Capuzzo M, Paglia T, Roli L,
Trenti T and Nelson SM: Testing for SARS-CoV-2 (COVID-19): A
systematic review and clinical guide to molecular and serological
in-vitro diagnostic assays. Reprod Biomed Online. 41:483–499. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
95
|
Sheikhzadeh E, Eissa S, Ismail A and
Zourob M: Diagnostic techniques for COVID-19 and new developments.
Talanta. 220:1213922020. View Article : Google Scholar : PubMed/NCBI
|
|
96
|
Zhu N, Wang W, Liu Z, Liang C, Wang W, Ye
F, Huang B, Zhao L, Wang H, Zhou W, et al: Morphogenesis and
cytopathic effect of SARS-CoV-2 infection in human airway
epithelial cells. Nat Commun. 11:39102020. View Article : Google Scholar : PubMed/NCBI
|
|
97
|
Leland DS and Ginocchio CC: Role of cell
culture for virus detection in the age of technology. Clin
Microbiol Rev. 20:49–78. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
98
|
Jain R, Sarkale P, Mali D, Shete AM, Patil
DY, Majumdar T, Suryawanshi A, Patil S, Mohandas S and Yadav PD:
Inactivation of SARS-CoV-2 by gamma irradiation. Indian J Med Res.
Nov 27–2020.Epub ahead of print. PubMed/NCBI
|
|
99
|
Park WB, Kwon NJ, Choi SJ, Kang CK, Choe
PG, Kim JY, Yun J, Lee GW, Seong MW, Kim NJ, et al: Virus Isolation
from the First Patient with SARS-CoV-2 in Korea. J Korean Med Sci.
35:e842020. View Article : Google Scholar : PubMed/NCBI
|
|
100
|
Zhao J, Zhou H, Huang W, Zhou J, Qiu M,
Deng Z, Chen L, Weng Y, Cai L, Gu Y, et al: Cell morphological
analysis of SARS-CoV-2 infection by transmission electron
microscopy. J Thorac Dis. 12:4368–4373. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
101
|
Akilesh S, Nicosia RF, Alpers CE,
Tretiakova M, Hsiang TY, Gale M Jr and Smith KD: Characterizing
viral infection by electron microscopy: lessons from the
coronavirus disease 2019 pandemic. Am J Pathol. 191:222–227. 2021.
View Article : Google Scholar
|
|
102
|
Goldsmith CS, Tatti KM, Ksiazek TG, Rollin
PE, Comer JA, Lee WW, Rota PA, Bankamp B, Bellini WJ and Zaki SR:
Ultrastructural characterization of SARS coronavirus. Emerg Infect
Dis. 10:320–326. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
103
|
Bain W, Lee JS, Watson AM and
Stitt-Fischer MS: Practical guidelines for collection, manipulation
and inactivation of SARS-CoV-2 and COVID-19 clinical specimens.
Curr Protoc Cytom. 93:e772020.PubMed/NCBI
|
|
104
|
Wu SY, Yau HS, Yu MY, Tsang HF, Chan LWC,
Cho WCS, Shing Yu AC, Yuen Yim AK, Li MJW, Wong YKE, et al: The
diagnostic methods in the COVID-19 pandemic, today and in the
future. Expert Rev Mol Diagn. 20:985–993. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
105
|
Chan JF, Yuan S, Kok KH, To KK, Chu H,
Yang J, Xing F, Liu J, Yip CC, Poon RW, et al: A familial cluster
of pneumonia associated with the 2019 novel coronavirus indicating
person-to-person transmission: A study of a family cluster. Lancet.
395:514–523. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
106
|
Wang M, Fu A, Hu B, Tong Y, Liu R, Liu Z,
Gu J, Xiang B, Liu J, Jiang W, et al: Nanopore targeted sequencing
for the accurate and comprehensive detection of SARS-CoV-2 and
other respiratory viruses. Small. 16:e20021692020. View Article : Google Scholar : PubMed/NCBI
|
|
107
|
Addetia A, Xie H, Roychoudhury P, Shrestha
L, Loprieno M, Huang ML, Jerome KR and Greninger AL: Identification
of multiple large deletions in ORF7a resulting in in-frame gene
fusions in clinical SARS-CoV-2 isolates. J Clin Virol.
129:1045232020. View Article : Google Scholar : PubMed/NCBI
|
|
108
|
Di Giallonardo F, Duchene S, Puglia I,
Curini V, Profeta F, Cammà C, Marcacci M, Calistri P, Holmes EC and
Lorusso A: Genomic Epidemiology of the First Wave of SARS-CoV-2 in
Italy. Viruses. 12:14382020. View Article : Google Scholar :
|
|
109
|
Moore SC, Penrice-Randal R, Alruwaili M,
Randle N, Armstrong S, Hartley C, Haldenby S, Dong X, Alrezaihi A,
Almsaud M, et al: Amplicon-Based Detection and Sequencing of
SARS-CoV-2 in Nasopharyngeal Swabs from Patients With COVID-19 and
Identification of Deletions in the Viral Genome That Encode
Proteins Involved in Interferon Antagonism. Viruses. 12:11642020.
View Article : Google Scholar :
|
|
110
|
Kames J, Holcomb DD, Kimchi O, DiCuccio M,
Hamasaki-Katagiri N, Wang T, Komar AA, Alexaki A and Kimchi-Sarfaty
C: Sequence analysis of SARS-CoV-2 genome reveals features
important for vaccine design. Sci Rep. 10:156432020. View Article : Google Scholar : PubMed/NCBI
|
|
111
|
Conti P, Caraffa A, Gallenga CE, Kritas
SK, Frydas I, Younes A, Di Emidio P, Tetè G, Pregliasco F and
Ronconi G: The British variant of the new coronavirus-19
(Sars-Cov-2) should not create a vaccine problem. J Biol Regul
Homeost Agents. 35:1–4. 2021.
|
|
112
|
Hourdel V, Kwasiborski A, Balière C,
Matheus S, Batéjat CF, Manuguerra JC, Vanhomwegen J and Caro V:
rapid genomic characterization of SARS-CoV-2 by direct
amplicon-based sequencing through comparison of MinION and Illumina
iSeq100TM system. Front Microbiol. 11:5713282020. View Article : Google Scholar :
|
|
113
|
illumina: Comprehensive Workf low for
Detecting Coronavirus Using Illumina Benchtop Systems - A Shotgun
Metagenomics Sequencing Workflow for Effective Detection and
Characterization of Coronavirus Strains. https://www.illumina.com/content/dam/illumina-marketing/documents/products/appnotes/ngs-coronavirus-app-note-1270-2020-001.pdf.
Accessed January 20, 2021.
|
|
114
|
Oxford Nanopore Technologies MinION -
Powerful, real-time, long-read sequencing in the palm of your hand.
https://nanoporetech.com/sites/default/files/s3/literature/MinION-Mk1C-brochure.pdf.
Accessed January 20, 2021.
|
|
115
|
Meredith LW, Hamilton WL, Warne B,
Houldcroft CJ, Hosmillo M, Jahun AS, Curran MD, Parmar S, Caller
LG, Caddy SL, et al: Rapid implementation of SARS-CoV-2 sequencing
to investigate cases of health-care associated COVID-19: A
prospective genomic surveillance study. Lancet Infect Dis.
20:1263–1271. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
116
|
Wen F, Yu H, Guo J, Li Y, Luo K and Huang
S: Identification of the hyper-variable genomic hotspot for the
novel coronavirus SARS-CoV-2. J Infect. 80:671–693. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
117
|
Lam TT: Tracking the Genomic Footprints of
SARS-CoV-2 Transmission. Trends Genet. 36:544–546. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
118
|
Landete P, Quezada Loaiza CA,
Aldave-Orzaiz B, Muñiz SH, Maldonado A, Zamora E, Sam Cerna AC, Del
Cerro E, Alonso RC and Couñago F: Clinical features and
radiological manifestations of COVID-19 disease. World J Radiol.
12:247–260. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
119
|
Chen H, Ai L, Lu H and Li H: Clinical and
imaging features of COVID-19. Radiol Infect Dis. 7:43–50. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
120
|
Al-Qahtani M, AlAli S, AbdulRahman A,
Salman Alsayyad A, Otoom S and Atkin SL: The prevalence of
asymptomatic and symptomatic COVID-19 in a cohort of quarantined
subjects. Int J Infect Dis. 102:285–288. 2021. View Article : Google Scholar
|
|
121
|
Yanes-Lane M, Winters N, Fregonese F,
Bastos M, Perlman-Arrow S, Campbell JR and Menzies D: Proportion of
asymptomatic infection among COVID-19 positive persons and their
transmission potential: A systematic review and meta-analysis. PLoS
One. 15:e02415362020. View Article : Google Scholar : PubMed/NCBI
|
|
122
|
Katopodis P, Kerslake R, Davies J, Randeva
HS, Chatha K, Hall M, Spandidos DA, Anikin V, Polychronis A,
Robertus JL, et al: COVID 19 and SARS CoV 2 host cell entry
mediators: Expression profiling of TMRSS4 in health and disease.
Int J Mol Med. 47:642021. View Article : Google Scholar
|
|
123
|
Vivarelli S, Falzone L, Torino F,
Scandurra G, Russo G, Bordonaro R, Pappalardo F, Spandidos DA,
Raciti G and Libra M: Immune-checkpoint inhibitors from cancer to
COVID-19: A promising avenue for the treatment of patients with
COVID-19 (Review). Int J Oncol. 58:145–157. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
124
|
Vivarelli S, Falzone L, Grillo CM,
Scandurra G, Torino F and Libra M: Cancer Management during
COVID-19 Pandemic: Is Immune Checkpoint Inhibitors-Based
Immunotherapy Harmful or Beneficial? Cancers (Basel). 12:22372020.
View Article : Google Scholar
|
|
125
|
Abdi A, Jalilian M, Sarbarzeh PA and
Vlaisavljevic Z: Diabetes and COVID-19: A systematic review on the
current evidences. Diabetes Res Clin Pract. 166:1083472020.
View Article : Google Scholar : PubMed/NCBI
|
|
126
|
Huang S, Wang J, Liu F, Liu J, Cao G, Yang
C, Liu W, Tu C, Zhu M and Xiong B: COVID-19 patients with
hypertension have more severe disease: A multicenter retrospective
observational study. Hyperte Res. 43:824–831. 2020. View Article : Google Scholar
|
|
127
|
Pascarella G, Strumia A, Piliego C, Bruno
F, Del Buono R, Costa F, Scarlata S and Agrò FE: COVID-19 diagnosis
and management: A comprehensive review. J Intern Med. 288:192–206.
2020. View Article : Google Scholar : PubMed/NCBI
|
|
128
|
Torre La G, Massetti AP, Antonelli G,
Fimiani C, Fantini M, Marte M, Faticoni A, Previte CM, Turriziani
O, Pugliese F, et al: Covid-Sapienza Collaborative Group: Anosmia
and Ageusia as Predictive Signs of COVID-19 in Healthcare Workers
in Italy: A Prospective Case-Control Study. J Clin Med. 9:28702020.
View Article : Google Scholar
|
|
129
|
Tanasa IA, Manciuc C, Carauleanu A,
Navolan DB, Bohiltea RE and Nemescu D: Anosmia and ageusia
associated with coronavirus infection (COVID-19) - what is known?
Exp Ther Med. 20:2344–2347. 2020.PubMed/NCBI
|
|
130
|
Pennisi M, Lanza G, Falzone L, Fisicaro F,
Ferri R and Bella R: SARS-CoV-2 and the Nervous System: From
Clinical Features to Molecular Mechanisms. Int J Mol Sci.
21:54752020. View Article : Google Scholar :
|
|
131
|
Connors JM and Levy JH: COVID-19 and its
implications for thrombosis and anticoagulation. Blood.
135:2033–2040. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
132
|
Diao B, Wang C, Tan Y, Chen X, Liu Y, Ning
L, Chen L, Li M, Liu Y, Wang G, et al: Reduction and functional
exhaustion of T cells in patients with coronavirus disease 2019
(COVID-19). Front Immunol. 11:8272020. View Article : Google Scholar : PubMed/NCBI
|
|
133
|
Basu-Ray I, Almaddah Nk, Adeboye A and
Soos MP: Cardiac Manifestations Of Coronavirus. StatPearls.
StatPearls Publishing; Treasure Island, FL: 2020
|
|
134
|
Garland V, Kumar AB and Borum ML:
Gastrointestinal and hepatic manifestations of COVID-19: evolving
recognition and need for increased understanding in vulnerable
populations. J Natl Med Assoc. Aug 15–2020.Epub ahead of print.
|
|
135
|
Baj J, Karakuła-Juchnowicz H, Teresiński
G, Buszewicz G, Ciesielka M, Sitarz E, Forma A, Karakuła K, Flieger
W, Portincasa P, et al: COVID-19: Specific and non-specific
clinical manifestations and symptoms: the current state of
knowledge. J Clin Med. 9:17532020. View Article : Google Scholar :
|
|
136
|
Renu K, Prasanna PL and Valsala
Gopalakrishnan A: Coronaviruses pathogenesis, comorbidities and
multi-organ damage - A review. Life Sci. 255:1178392020. View Article : Google Scholar : PubMed/NCBI
|
|
137
|
Chang MC, Lee W, Hur J and Park D: Chest
Computed Tomography Findings in Asymptomatic Patients with
COVID-19. Respiration. 99:748–754. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
138
|
Jędrusik P, Gaciong Z, Sklinda K,
Sierpiński R, Walecki J and Gujski M: Diagnostic role of chest
computed tomography in coronavirus disease 2019. Pol Arch Intern
Med. 130:520–528. 2020.
|
|
139
|
Kang EY, Staples CA, McGuinness G, Primack
SL and Müller NL: Detection and differential diagnosis of pulmonary
infections and tumors in patients with AIDS: Value of chest
radiography versus CT. AJR Am J Roentgenol. 166:15–19. 1996.
View Article : Google Scholar : PubMed/NCBI
|
|
140
|
Larici AR, Cicchetti G, Marano R, Merlino
B, Elia L, Calandriello L, Del Ciello A, Farchione A, Savino G,
Infante A, et al: Multimodality imaging of COVID-19 pneumonia: From
diagnosis to follow-up. A comprehensive review. Eur J Radiol.
131:1092172020. View Article : Google Scholar : PubMed/NCBI
|
|
141
|
Ippolito D, Maino C, Pecorelli A,
Allegranza P, Cangiotti C, Capodaglio C, Mariani I, Giandola T,
Gandola D, Bianco I, et al: Chest X-ray features of SARS-CoV-2 in
the emergency department: A multicenter experience from northern
Italian hospitals. Respir Med. 170:1060362020. View Article : Google Scholar : PubMed/NCBI
|
|
142
|
Li P, Chen L, Liu Z, Pan J, Zhou D, Wang
H, Gong H, Fu Z, Song Q, Min Q, et al: Clinical features and
short-term outcomes of elderly patients with COVID-19. Int J Infect
Dis. 97:245–250. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
143
|
Cui N, Zou X and Xu L: Preliminary CT
findings of coronavirus disease 2019 (COVID-19). Clin Imaging.
65:124–132. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
144
|
Kovács A, Palásti P, Veréb D, Bozsik B,
Palkó A and Kincses ZT: The sensitivity and specificity of chest CT
in the diagnosis of COVID-19. Eur Radiol. 13:1–6. 2020.
|
|
145
|
Zhao W, He L, Tang H, Xie X, Tang L and
Liu J: The Relationship Between Chest Imaging Findings and the
Viral Load of COVID-19. Front Med (Lausanne). 7:5585392020.
View Article : Google Scholar
|
|
146
|
Meng H, Xiong R, He R, Lin W, Hao B, Zhang
L, Lu Z, Shen X, Fan T, Jiang W, et al: CT imaging and clinical
course of asymptomatic cases with COVID-19 pneumonia at admission
in Wuhan, China. J Infect. 81:e33–e39. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
147
|
Mehrabi S, Fontana S, Mambrin F, Nguyen
HQ, Righi E, Tacconelli E and Mansueto G: Pitfalls of computed
tomography in the coronavirus 2019 (COVID-19) era: a new
perspective on ground-glass opacities. Cureus.
12:e81512020.PubMed/NCBI
|
|
148
|
Tofighi S, Najafi S, Johnston SK and
Gholamrezanezhad A: Low-dose CT in COVID-19 outbreak: Radiation
safety, image wisely, and image gently pledge. Emerg Radiol.
27:601–605. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
149
|
Qiu G, Gai Z, Tao Y, Schmitt J,
Kullak-Ublick GA and Wang J: Dual-functional plasmonic photothermal
biosensors for highly accurate severe acute respiratory syndrome
coronavirus 2 detection. ACS Nano. 14:5268–5277. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
150
|
Pandey LM: Design of engineered surfaces
for prospective detection of SARS-CoV-2 using quartz crystal
micro-balance-based techniques. Expert Rev Proteomics. 17:425–432.
2020. View Article : Google Scholar : PubMed/NCBI
|
|
151
|
Maddali H, Miles CE, Kohn J and O'Carroll
DM: Optical Biosensors for Virus Detection: Prospects for
SARS-CoV-2/COVID-19. ChemBioChem. 22:1176–1189. 2021. View Article : Google Scholar
|
|
152
|
Cui F and Zhou HS: Diagnostic methods and
potential portable biosensors for coronavirus disease 2019. Biosens
Bioelectron. 165:1123492020. View Article : Google Scholar : PubMed/NCBI
|
|
153
|
Samson R, Navale GR and Dharne MS:
Biosensors: frontiers in rapid detection of COVID-19. 3 Biotech.
10:3852020.
|
|
154
|
Seo G, Lee G, Kim MJ, Baek SH, Choi M, Ku
KB, Lee CS, Jun S, Park D, Kim HG, et al: Rapid detection of
COVID-19 causative virus (SARS-CoV-2) in human nasopharyngeal swab
specimens using field-effect transistor-based biosensor. ACS Nano.
14:5135–5142. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
155
|
Zhao H, Liu F, Xie W, Zhou TC, OuYang J,
Jin L, Li H, Zhao CY, Zhang L, Wei J, et al: Ultrasensitive
supersandwich-type electrochemical sensor for SARS-CoV-2 from the
infected COVID-19 patients using a smartphone. Sens Acuators B
Chem. 327:1288992021. View Article : Google Scholar
|
|
156
|
Choi JR: Development of point-of-care
biosensors for COVID-19. Front Chem. 8:5172020. View Article : Google Scholar : PubMed/NCBI
|
|
157
|
Lu R, Wu X, Wan Z, Li Y, Jin X and Zhang
C: A novel reverse transcription loop-mediated isothermal
amplification method for rapid detection of SARS-CoV-2. Int J Mol
Sci. 21:28262020. View Article : Google Scholar :
|
|
158
|
Yuan X, Yang C, He Q, Chen J, Yu D, Li J,
Zhai S, Qin Z, Du K, Chu Z, et al: Current and perspective
diagnostic techniques for COVID-19. ACS Infect Dis. 6:1998–2016.
2020. View Article : Google Scholar : PubMed/NCBI
|
|
159
|
Huang WE, Lim B, Hsu CC, Xiong D, Wu W, Yu
Y, Jia H, Wang Y, Zeng Y, Ji M, et al: RT-LAMP for rapid diagnosis
of coronavirus SARS-CoV-2. Microb Biotechnol. 13:950–961. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
160
|
Broughton JP, Deng X, Yu G, Fasching CL,
Servellita V, Singh J, Miao X, Streithorst JA, Granados A,
Sotomayor-Gonzalez A, et al: CRISPR-Cas12-based detection of
SARS-CoV-2. Nat Biotechnol. 38:870–874. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
161
|
Song J, Liu C, Mauk MG, Rankin SC, Lok JB,
Greenberg RM and Bau HH: Two-Stage Isothermal Enzymatic
Amplification for Concurrent Multiplex Molecular Detection. Clin
Chem. 63:714–722. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
162
|
El-Tholoth M, Bau HH and Song J: A Single
and Two-Stage, Closed-Tube, Molecular Test for the 2019 Novel
Coronavirus (COVID-19) at Home, Clinic, and Points of Entry.
ChemRxiv. View Article : Google Scholar
|
|
163
|
Smithgall MC, Scherberkova I, Whittier S
and Green DA: Comparison of Cepheid Xpert Xpress and Abbott ID Now
to Roche cobas for the Rapid Detection of SARS-CoV-2. J Clin Virol.
128:1044282020. View Article : Google Scholar : PubMed/NCBI
|
|
164
|
Rhoads DD, Cherian SS, Roman K, Stempak
LM, Schmotzer CL and Sadri N: Comparison of Abbott ID Now, DiaSorin
Simplexa, and CDC FDA Emergency Use Authorization Methods for the
Detection of SARS-CoV-2 from Nasopharyngeal and Nasal Swabs from
Individuals Diagnosed with COVID-19. J Clin Microbiol.
58:e00760–e20. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
165
|
Basu A, Zinger T, Inglima K, Woo KM, Atie
O, Yurasits L, See B and Aguero-Rosenfeld ME: Performance of Abbott
ID Now COVID-19 Rapid Nucleic Acid Amplification Test Using
Nasopharyngeal Swabs Transported in Viral Transport Media and Dry
Nasal Swabs in a New York City Academic Institution. J Clin
Microbiol. 58:e01136–e20. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
166
|
Westermann L, Neubauer B and Köttgen M:
Nobel Prize 2020 in Chemistry honors CRISPR: A tool for rewriting
the code of life. Pflugers Arch. 473:1–2. 2021. View Article : Google Scholar :
|
|
167
|
Doudna JA and Charpentier E: Genome
editing. The new frontier of genome engineering with CRISPR-Cas9.
Science. 346:12580962014. View Article : Google Scholar : PubMed/NCBI
|
|
168
|
Pickar-Oliver A and Gersbach CA: The next
generation of CRISPR-Cas technologies and applications. Nat Rev Mol
Cell Biol. 20:490–507. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
169
|
Broughton JP, Deng X, Yu G, Fasching CL,
Singh J, Streithorst J, Granados A, Sotomayor-Gonzalez A, Zorn K,
Gopez A, et al: Rapid detection of 2019 novel coronavirus
SARS-CoV-2 using a CRISPR-based DETECTR lateral flow assay.
medRxiv. https://doi.org/10.1101/2020.03.06.20032334.
|
|
170
|
Javalkote VS, Kancharla N, Bhadra B,
Shukla M, Soni B, Goodin M, Bandyopadhyay A and Dasgupta S:
CRISPR-based assays for rapid detection of SARS-CoV-2. Methods. Oct
9–2020.Epub ahead of print. View Article : Google Scholar : PubMed/NCBI
|
|
171
|
van Dongen JE, Berendsen JTW, Steenbergen
RDM, Wolthuis RMF, Eijkel JCT and Segerink LI: Point-of-care
CRISPR/Cas nucleic acid detection: Recent advances, challenges and
opportunities. Biosens Bioelectron. 166:1124452020. View Article : Google Scholar : PubMed/NCBI
|
|
172
|
Kellner MJ, Koob JG, Gootenberg JS,
Abudayyeh OO and Zhang F: SHERLOCK: Nucleic acid detection with
CRISPR nucleases. Nat Protoc. 14:2986–3012. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
173
|
Joung J, Ladha A, Saito M, Segel M,
Bruneau R, Huang MW, Kim NG, Yu X, Li J, Walker BD, et al:
Point-of-care testing for COVID-19 using SHERLOCK diagnostics.
medRxiv. View Article : Google Scholar
|
|
174
|
Food and Drug Administration (FDA):
Sherlock™ CRISPR SARS-CoV-2 kit - Instructions for use. https://www.fda.gov/media/137746/download. Accessed
February 2, 2021.
|
|
175
|
Hou T, Zeng W, Yang M, Chen W, Ren L, Ai
J, Wu J, Liao Y, Gou X, Li Y, et al: Development and evaluation of
a rapid CRISPR-based diagnostic for COVID-19. PLoS Pathog.
16:e10087052020. View Article : Google Scholar
|
|
176
|
Curti L, Pereyra-Bonnet F and Gimenez CA:
An Ultrasensitive, Rapid, and Portable Coronavirus SARS-CoV-2
Sequence Detection Method Based on CRISPR-Cas12. bioRxiv.
https://doi.org/10.1101/2020.02.29.971127.
|
|
177
|
Salipante SJ and Jerome KR: Digital PCR-An
Emerging Technology with Broad Applications in Microbiology. Clin
Chem. 66:117–123. 2020. View Article : Google Scholar
|
|
178
|
Falzone L, Musso N, Gattuso G, Bongiorno
D, Palermo CI, Scalia G, Libra M and Stefani S: Sensitivity
assessment of droplet digital PCR for SARS-CoV-2 detection. Int J
Mol Med. 46:957–964. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
179
|
Falzone L, Gattuso G, Lombardo C, Lupo G,
Grillo CM, Spandidos DA, Libra M and Salmeri M: Droplet digital PCR
for the detection and monitoring of Legionella pneumophila. Int J
Mol Med. 46:1777–1782. 2020.PubMed/NCBI
|
|
180
|
Crimi S, Falzone L, Gattuso G, Grillo CM,
Candido S, Bianchi A and Libra M: Droplet Digital PCR Analysis of
Liquid Biopsy Samples Unveils the Diagnostic Role of
hsa-miR-133a-3p and hsa-miR-375-3p in Oral Cancer. Biology (Basel).
9:3792020.
|
|
181
|
Salemi R, Falzone L, Madonna G, Polesel J,
Cinà D, Mallardo D, Ascierto PA, Libra M and Candido S: MMP-9 as a
Candidate Marker of Response to BRAF Inhibitors in Melanoma
Patients With BRAFV600E Mutation Detected in Circulating-Free DNA.
Front Pharmacol. 9:8562018. View Article : Google Scholar :
|
|
182
|
Battaglia R, Palini S, Vento ME, La
Ferlita A, Lo Faro MJ, Caroppo E, Borzì P, Falzone L, Barbagallo D,
Ragusa M, et al: Identification of extracellular vesicles and
characterization of miRNA expression profiles in human blastocoel
fluid. Sci Rep. 9:842019. View Article : Google Scholar : PubMed/NCBI
|
|
183
|
Bizouarn F: Clinical applications using
digital PCR. Methods Mol Biol. 1160:189–214. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
184
|
Olmedillas-López S, García-Arranz M and
García-Olmo D: Current and Emerging Applications of Droplet Digital
PCR in Oncology. Mol Diagn Ther. 21:493–510. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
185
|
Denis JA, Nectoux J, Lamy PJ, Rouillac Le
Sciellour C, Guermouche H, Alary AS, Kosmider O, Sarafan-Vasseur N,
Jovelet C, Busser B, et al: Development of digital PCR molecular
tests for clinical practice: Principles, practical implementation
and recommendations. Ann Biol Clin (Paris). 76:505–523. 2018.
|
|
186
|
Pinheiro LB, Coleman VA, Hindson CM,
Herrmann J, Hindson BJ, Bhat S and Emslie KR: Evaluation of a
droplet digital polymerase chain reaction format for DNA copy
number quantification. Anal Chem. 84:1003–1011. 2012. View Article : Google Scholar :
|
|
187
|
Alteri C, Cento V, Antonello M, Colagrossi
L, Merli M, Ughi N, Renica S, Matarazzo E, Di Ruscio F, Tartaglione
L, et al: Detection and quantification of SARS-CoV-2 by droplet
digital PCR in real-time PCR negative nasopharyngeal swabs from
suspected COVID-19 patients. PLoS One. 15:e02363112020. View Article : Google Scholar : PubMed/NCBI
|
|
188
|
Suo T, Liu X, Feng J, Guo M, Hu W, Guo D,
Ullah H, Yang Y, Zhang Q, Wang X, et al: ddPCR: A more accurate
tool for SARS-CoV-2 detection in low viral load specimens. Emerg
Microbes Infect. 9:1259–1268. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
189
|
Liu C, Shi Q, Peng M, Lu R, Li H, Cai Y,
Chen J, Xu J and Shen B: Evaluation of droplet digital PCR for
quantification of SARS-CoV-2 Virus in discharged COVID-19 patients.
Aging (any NY). 12:20997–21003. 2020. View Article : Google Scholar
|
|
190
|
Vasudevan HN, Xu P, Servellita V, Miller
S, Liu L, Gopez A, Chiu CY and Abate AR: Digital droplet PCR
accurately quantifies SARS-CoV-2 viral load from crude lysate
without nucleic acid purification. Sci Rep. 11:7802021. View Article : Google Scholar : PubMed/NCBI
|