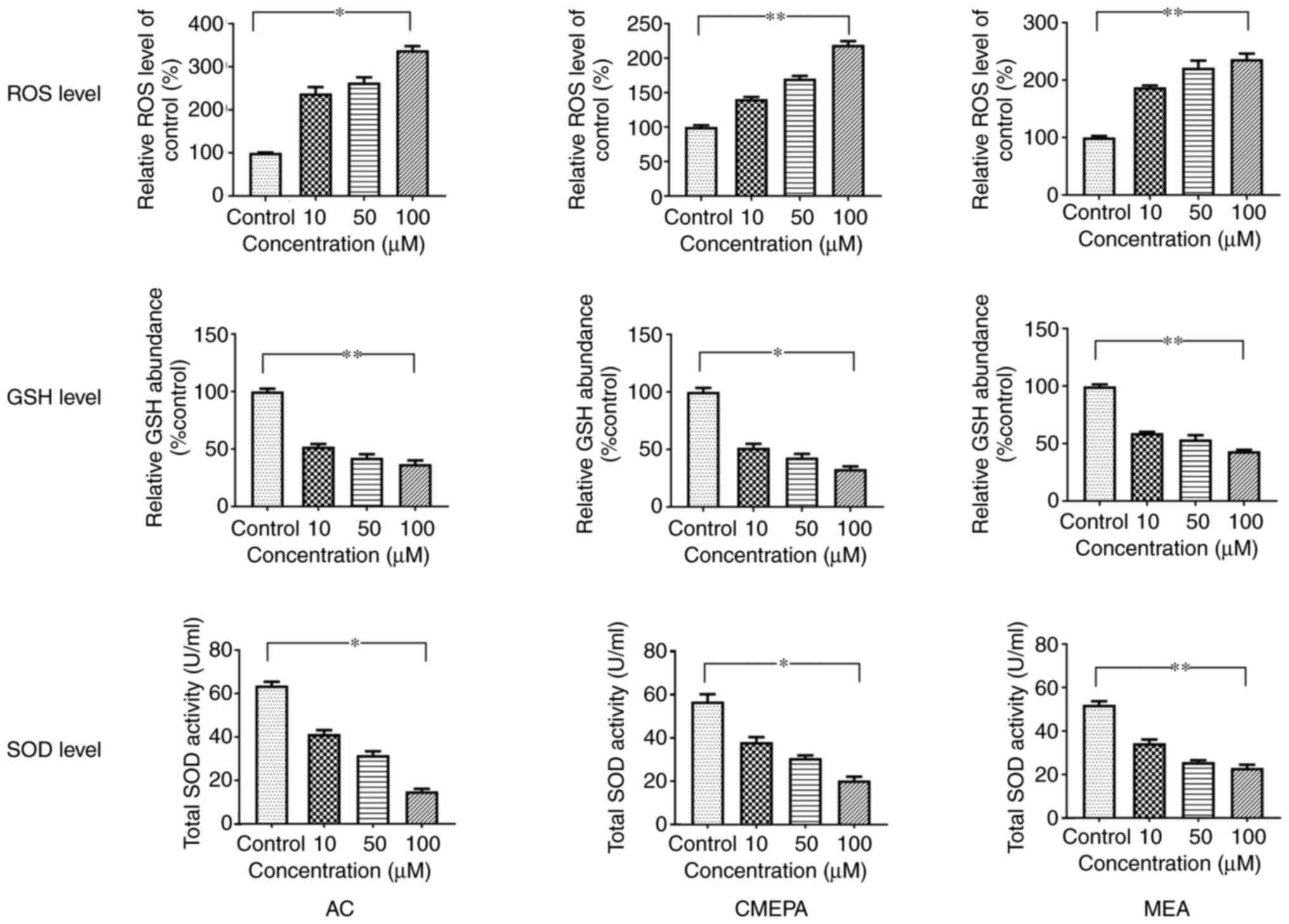
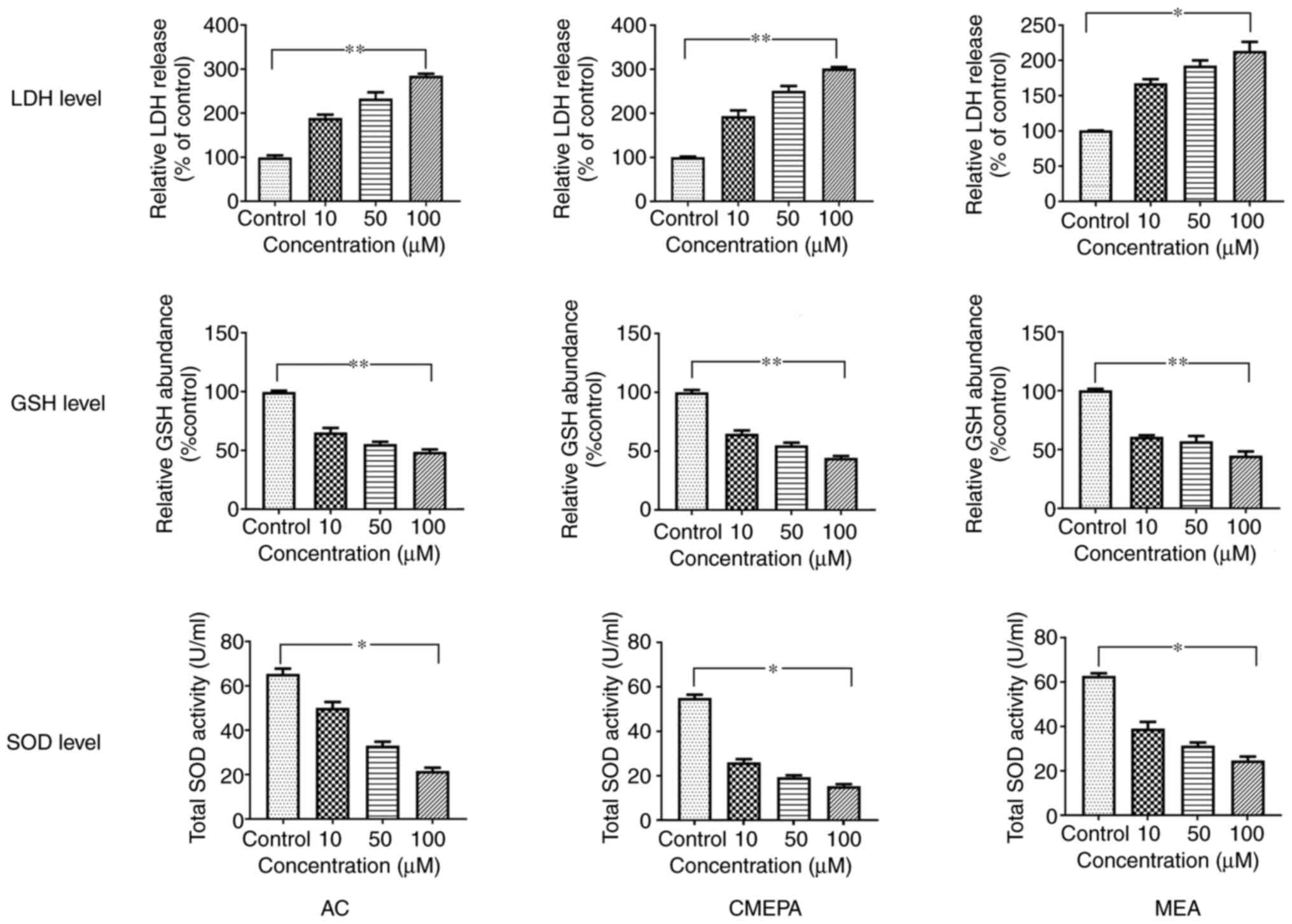
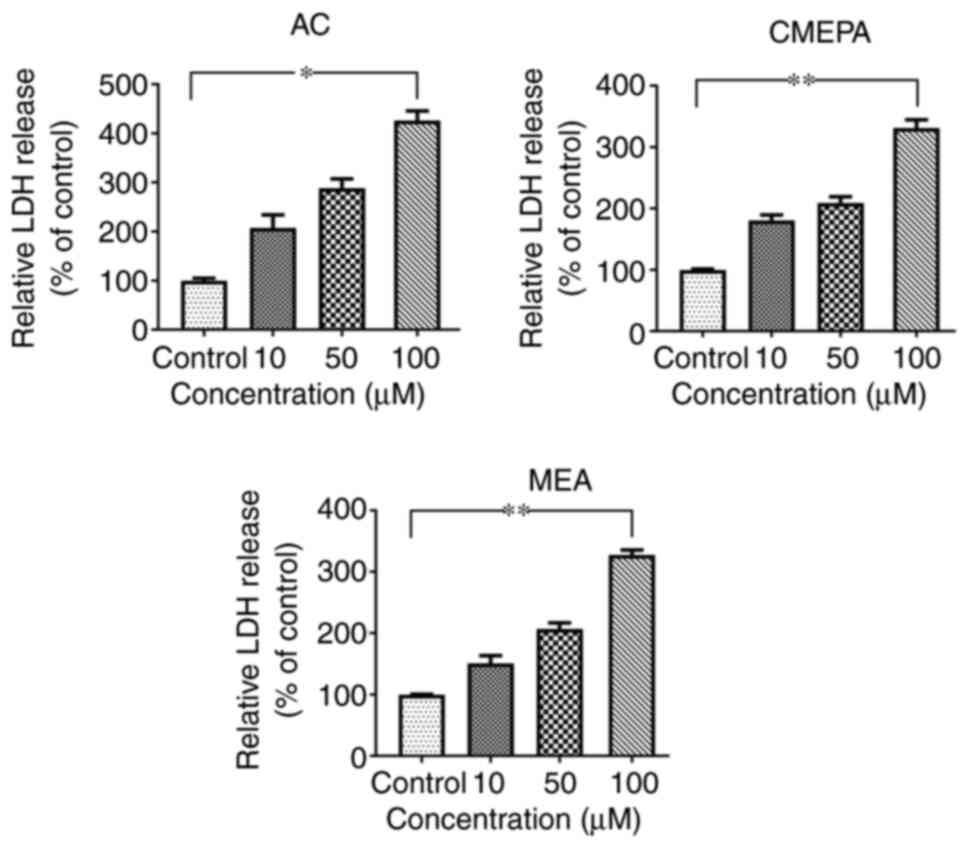
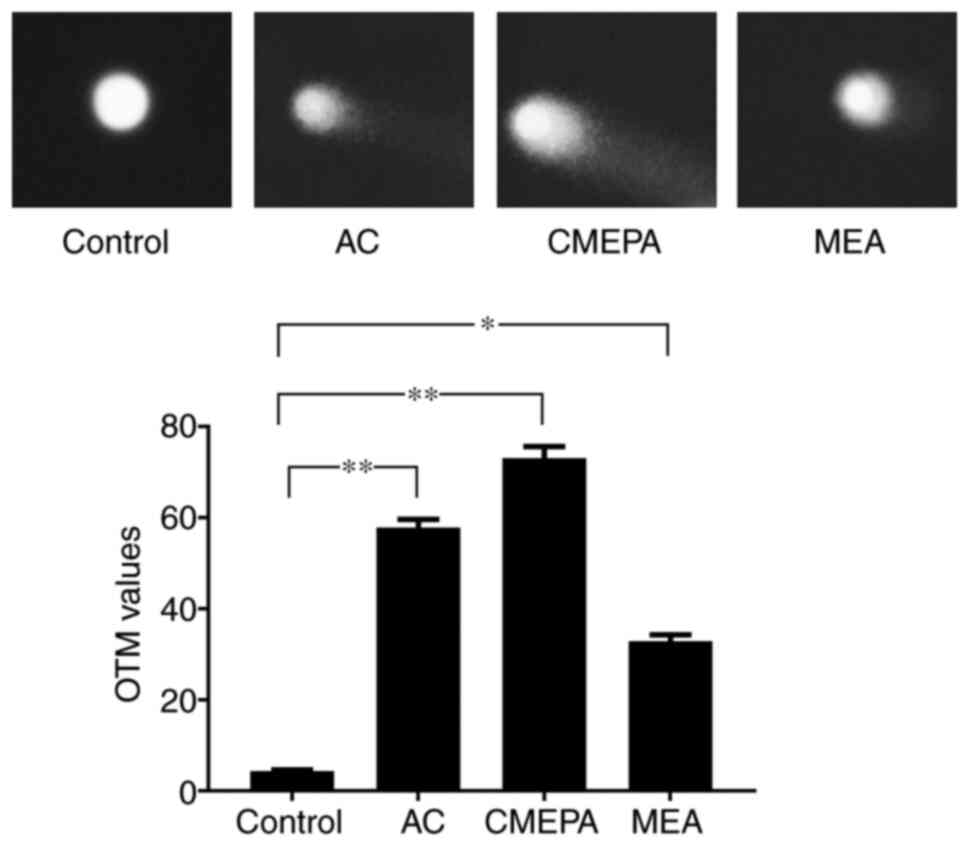
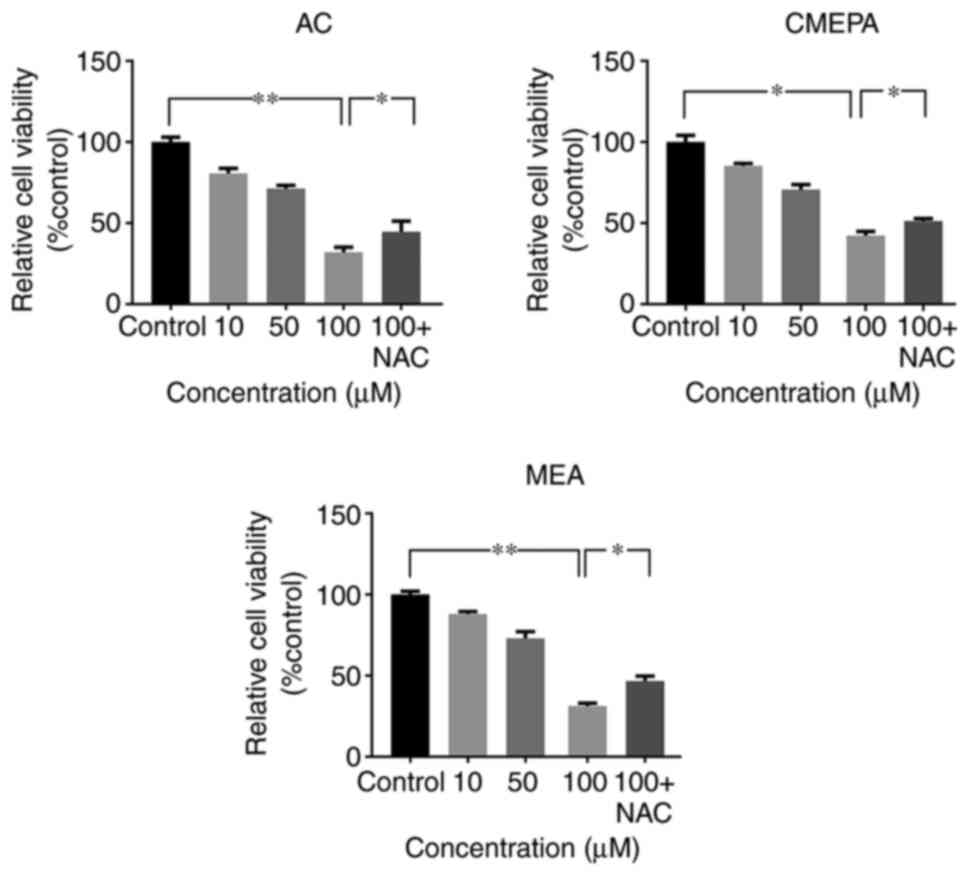
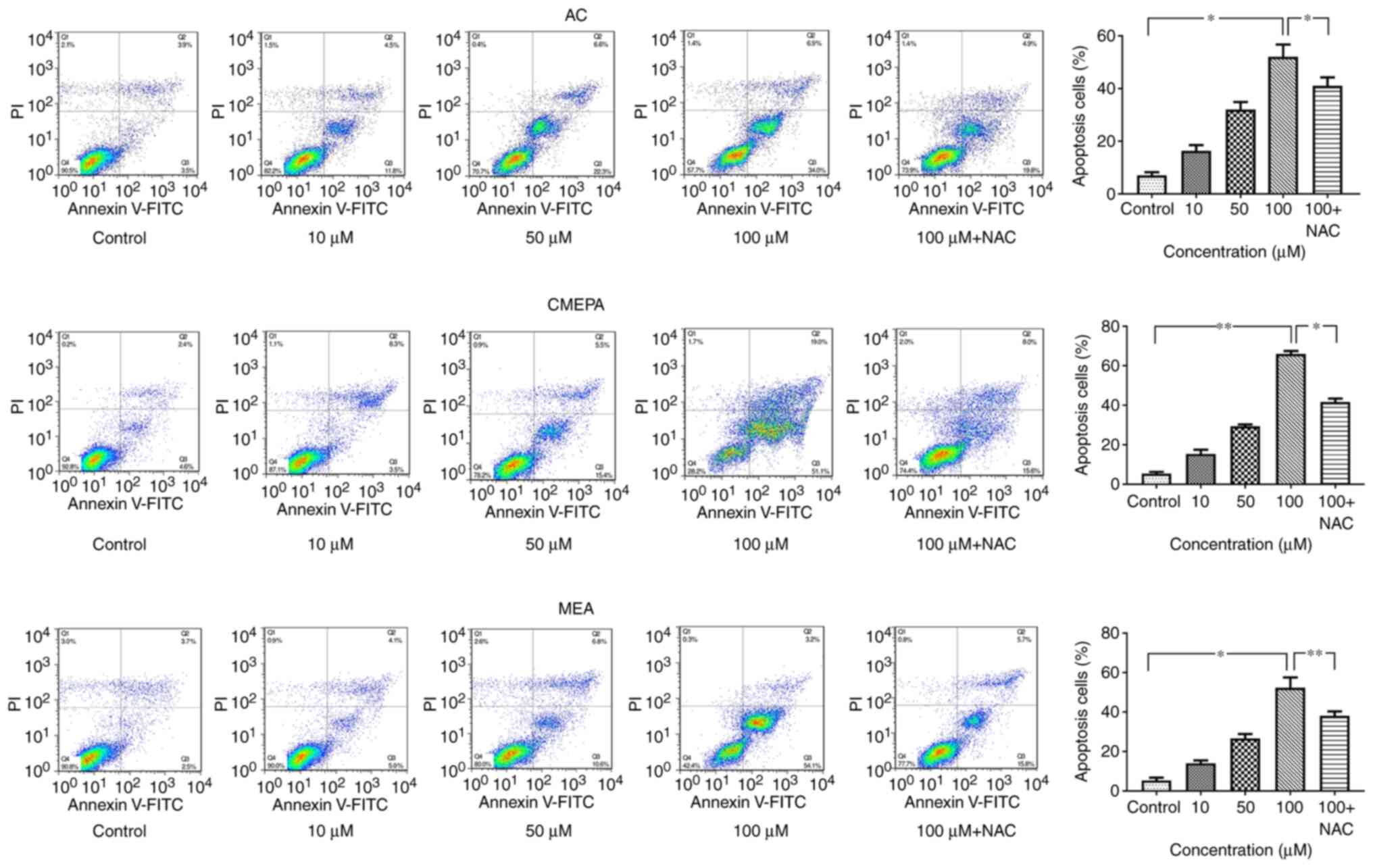
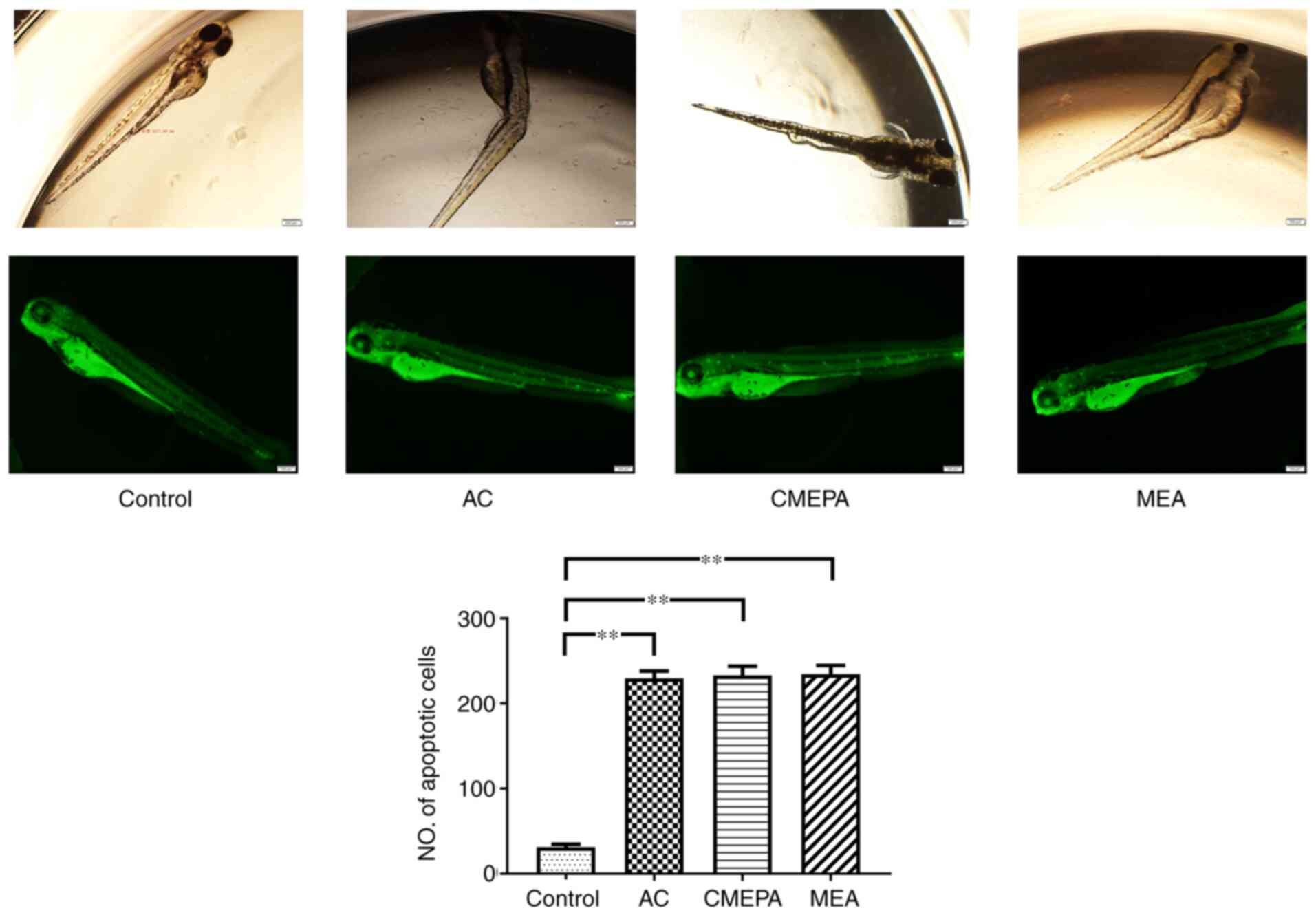
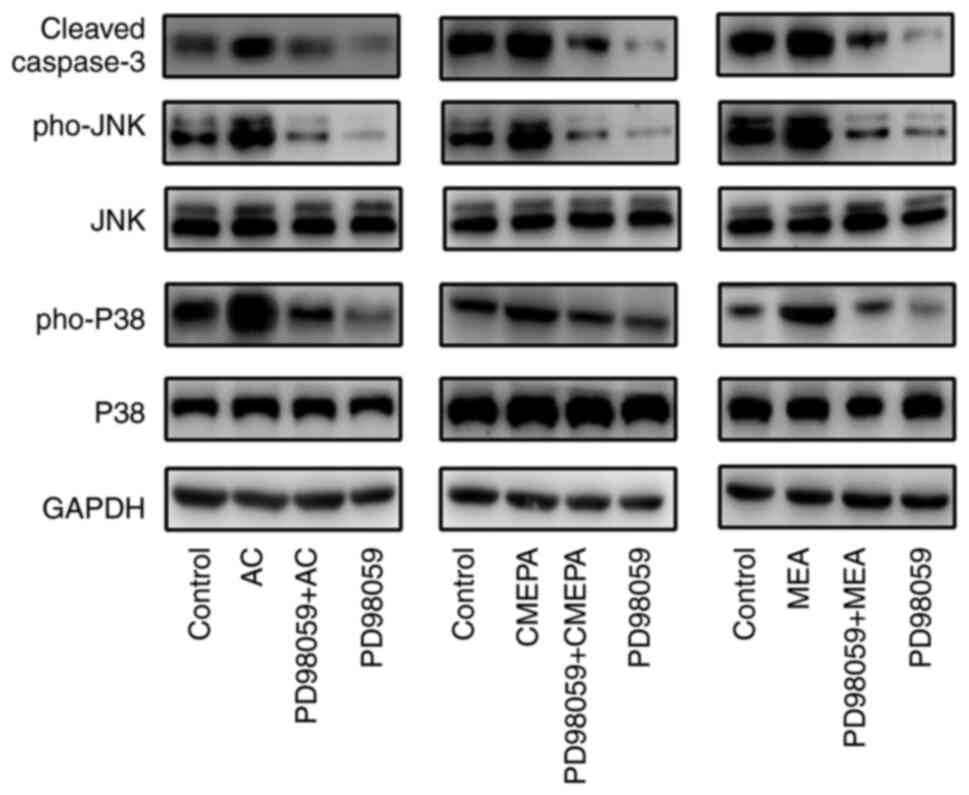
|
1
|
Xu C, Sun X, Niu L, Yang W, Tu W, Lu L,
Song S and Liu W: Enantioselective thyroid disruption in zebrafish
embryo-larvae via exposure to environmental concentrations of the
chloroacetamide herbicide acetochlor. Sci Total Environ.
653:1140–1148. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Li D, Li F, Zhao Y and Yuan J:
Solid-liquid stable equilibrium of the aqueous quaternary system
NH4SCN-(NH4)2S2O3-(NH4)2SO4-H2O at 303.15 K. J Chem Eng Data.
60:82–88. 2015. View Article : Google Scholar
|
|
3
|
Saha S, Dutta D, Karmakar R and Ray DP:
Structure-toxicity relationship of chloroacetanilide herbicides:
Relative impact on soil microorganisms. Environ Toxicol Pharmacol.
34:307–314. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Tu W, Niu L, Liu W and Xu C: Embryonic
exposure to butachlor in zebrafish (Danio rerio): Endocrine
disruption, developmental toxicity and immunotoxicity. Ecotoxicol
Environ Saf. 89:189–195. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Ranke J: Persistence of antifouling agents
in the marine biosphere. Environ Sci Technol. 36:1539–1545. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Fu L, Lu X, Tan J, Wang L and Chen J:
Multiresidue determination and potential risks of emerging
pesticides in aquatic products from Northeast China by LC-MS/MS. J
Environ Sci (China). 63:116–125. 2018. View Article : Google Scholar
|
|
7
|
Tang XY, Yang Y, Tam NF, Tao R and Dai YN:
Pesticides in three rural rivers in Guangzhou, China:
Spatiotemporal distribution and ecological risk. Environ Sci Pollut
Res Int. 26:3569–3577. 2019. View Article : Google Scholar
|
|
8
|
Yu J, Xu EG, Ren Y, Jin S, Zhang T, Liu J
and Li Z: Mixture toxicity of bensulfuron-methyl and acetochlor to
red swamp crayfish (Procambarus clarkii): Behavioral, morphological
and histological effects. Int J Environ Res Public Health.
14:14662017. View Article : Google Scholar
|
|
9
|
Dearfield KL, McCarroll NE, Protzel A,
Stack HF, Jackson MA and Waters MD: A survey of EPA/OPP and open
literature on selected pesticide chemicals. II. Mutagenicity and
carcinogenicity of selected chloroacetanilides and related
compounds. Mutat Res. 443:183–221. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Liu Y, Fang K, Zhang X, Liu T and Wang X:
Enantioselective toxicity and oxidative stress effects of
acetochlor on earthworms (Eisenia fetida) by mediating the
signaling pathway. Sci Total Environ. 766:1426302021. View Article : Google Scholar
|
|
11
|
Jiang J, Wu S, Liu X, Wang Y, An X, Cai L
and Zhao X: Effect of acetochlor on transcription of genes
associated with oxidative stress, apoptosis, immunotoxicity and
endocrine disruption in the early life stage of zebrafish. Environ
Toxicol Pharmacol. 40:516–523. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Li W, Zha J, Li Z, Yang L and Wang Z:
Effects of exposure to acetochlor on the expression of thyroid
hormone related genes in larval and adult rare minnow (Gobiocypris
rarus). Aquat Toxicol. 94:87–93. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Yang M, Hu J, Li S, Ma Y, Gui W and Zhu G:
Thyroid endocrine disruption of acetochlor on zebrafish (Danio
rerio) larvae. J Appl Toxicol. 36:844–852. 2016. View Article : Google Scholar
|
|
14
|
Coscollà C, López A, Yahyaoui A, Colin P,
Robin C, Poinsignon Q and Yusà V: Human exposure and risk
assessment to airborne pesticides in a rural French community. Sci
Total Environ. 584-585:856–868. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Wan N and Lin G: Parkinson's disease and
pesticides exposure: New findings from a comprehensive study in
nebraska, USA. J Rural Health. 32:303–313. 2016. View Article : Google Scholar
|
|
16
|
Liu H, Chu T, Chen L, Gui W and Zhu G: In
vivo cardiovascular toxicity induced by acetochlor in zebrafish
larvae. Chemosphere. 181:600–608. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Kale VM, Miranda SR, Wilbanks MS and Meyer
SA: Comparative cytotoxicity of alachlor, acetochlor, and
metolachlor herbicides in isolated rat and cryopreserved human
hepatocytes. J Biochem Mol Toxicol. 22:41–50. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Coleman S, Linderman R, Hodgson E and Rose
RL: Comparative metabolism of chloroacetamide herbicides and
selected metabolites in human and rat liver microsomes. Environ
Health Perspect. 108:1151–1157. 2000.
|
|
19
|
Zerin T, Song HY and Kim YS: Extracellular
signal-regulated kinase pathway play distinct role in
acetochlor-mediated toxicity and intrinsic apoptosis in A549 cells.
Toxicol In Vitro. 29:85–92. 2015. View Article : Google Scholar
|
|
20
|
Apostolou P, Toloudi M, Kourtidou E,
Mimikakou G, Vlachou I, Chatziioannou M and Papasotiriou I: Use of
the comet assay technique for quick and reliable prediction of in
vitro response to chemotherapeutics in breast and colon cancer. J
Biol Res (Thessalon). 21:142014. View Article : Google Scholar
|
|
21
|
Hong Y, Han HJ, Lee H, Lee D, Ko J, Hong
ZY, Lee JY, Seok JH, Lim HS, Son WC and Sohn I: Deep learning
method for comet segmentation and comet assay image analysis. Sci
Rep. 10:189152020. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Kimmel CB, Ballard WW, Kimmel SR, Ullmann
B and Schilling TF: Stages of embryonic development of the
zebrafish. Dev Dyn. 203:253–310. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Plemel JR, Caprariello AV, Keough MB,
Henry TJ, Tsutsui S, Chu TH, Schenk GJ, Klaver R, Yong VW and Stys
PK: Unique spectral signatures of the nucleic acid dye acridine
orange can distinguish cell death by apoptosis and necroptosis. J
Cell Biol. 216:1163–1181. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Tucker B and Lardelli M: A rapid apoptosis
assay measuring relative acridine orange fluorescence in zebrafish
embryos. Zebrafish. 4:113–116. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Iman V, Mohan S, Abdelwahab SI, Karimian
H, Nordin N, Fadaeinasab M, Noordin MI and Noor SM: Anticancer and
anti-inflammatory activities of girinimbine isolated from Murraya
koenigii. Drug Des Devel Ther. 11:103–121. 2016. View Article : Google Scholar
|
|
26
|
Cagnol S and Chambard JC: ERK and cell
death: Mechanisms of ERK-induced cell death-apoptosis, autophagy
and senescence. FEBS J. 277:2–21. 2010. View Article : Google Scholar
|
|
27
|
Yin XH, Li SN, Zhang L, Zhu GN and Zhuang
HS: Evaluation of DNA damage in Chinese toad (Bufo bufo
gargarizans) after in vivo exposure to sublethal concentrations of
four herbicides using the comet assay. Ecotoxicology. 17:280–286.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Daam MA, Moutinho MF, Espindola ELG and
Schiesari L: Lethal toxicity of the herbicides acetochlor, ametryn,
glyphosate and metribuzin to tropical frog larvae. Ecotoxicology.
28:707–715. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Wang H, Meng Z, Zhou L, Cao Z, Liao X, Ye
R and Lu H: Effects of acetochlor on neurogenesis and behaviour in
zebrafish at early developmental stages. Chemosphere. 220:954–964.
2019. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Huang T, Huang Y, Huang Y, Yang Y, Zhao Y
and Martyniuk CJ: Toxicity assessment of the herbicide acetochlor
in the human liver carcinoma (HepG2) cell line. Chemosphere.
243:1253452020. View Article : Google Scholar
|
|
31
|
Kocsis Z, Marcsek ZL, Jakab MG, Szende B
and Tompa A: Chemopreventive properties of trans-resveratrol
against the cytotoxicity of chloroacetanilide herbicides in vitro.
Int J Hyg Environ Health. 208:211–218. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Liu HT, Li WM, Xu G, Li XY, Bai XF, Wei P,
Yu C and Du YG: Chitosan oligosaccharides attenuate hydrogen
peroxide-induced stress injury in human umbilical vein endothelial
cells. Pharmacol Res. 59:167–175. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Thorpe GW, Reodica M, Davies MJ, Heeren G,
Jarolim S, Pillay B, Breitenbach M, Higgins VJ and Dawes IW:
Superoxide radicals have a protective role during H2O2 stress. Mol
Biol Cell. 24:2876–2884. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Kim H, Lee SW, Baek KM, Park JS and Min
JH: Continuous hypoxia attenuates paraquat-induced cytotoxicity in
the human A549 lung carcinoma cell line. Exp Mol Med. 43:494–500.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Adeoye O, Olawumi J, Opeyemi A and
Christiania O: Review on the role of glutathione on oxidative
stress and infertility. JBRA Assist Reprod. 22:61–66. 2018.
|
|
36
|
Nita M and Grzybowski A: The role of the
reactive oxygen species and oxidative stress in the pathomechanism
of the age-related ocular diseases and other pathologies of the
anterior and posterior eye segments in adults. Oxid Med Cell
Longev. 2016:31647342016. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Kumar P, Nagarajan A and Uchil PD:
Analysis of cell viability by the lactate dehydrogenase assay. Cold
Spring Harb Protoc. 2018:2018.
|
|
38
|
Schieber M and Chandel NS: ROS function in
redox signaling and oxidative stress. Curr Biol. 24:R453–R462.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Lee VY, McClintock DS, Santore MT,
Budinger GR and Chandel NS: Hypoxia sensitizes cells to nitric
oxide-induced apoptosis. J Biol Chem. 277:16067–16074. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Fattori V, Pinho-Ribeiro FA, Borghi SM,
Alves-Filho JC, Cunha TM, Cunha FQ, Casagrande R and Verri WA Jr:
Curcumin inhibits superoxide anion-induced pain-like behavior and
leukocyte recruitment by increasing Nrf2 expression and reducing
NF-κB activation. Inflamm Res. 64:993–1003. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Cadet J and Wagner JR: DNA base damage by
reactive oxygen species, oxidizing agents, and UV radiation. Cold
Spring Harb Perspect Biol. 5:a0125592013. View Article : Google Scholar
|
|
42
|
Singh A, Kukreti R, Saso L and Kukreti S:
Oxidative stress: Role and response of short guanine tracts at
genomic locations. Int J Mol Sci. 20:42582019. View Article : Google Scholar :
|
|
43
|
Dizdaroglu M and Jaruga P: Mechanisms of
free radical-induced damage to DNA. Free Radic Res. 46:382–419.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Cooke MS, Evans MD, Dizdaroglu M and Lunec
J: Oxidative DNA damage: Mechanisms, mutation, and disease. FASEB
J. 17:1195–1214. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Lobo V, Patil A, Phatak A and Chandra N:
Free radicals, antioxidants and functional foods: Impact on human
health. Pharmacogn Rev. 4:118–126. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Pallepati P and Averill-Bates DA: Mild
thermotolerance induced at 40°C protects HeLa cells against
activation of death receptor-mediated apoptosis by hydrogen
peroxide. Free Radic Biol Med. 50:667–679. 2011. View Article : Google Scholar
|
|
47
|
Pallepati P and Averill-Bates DA:
Activation of ER stress and apoptosis by hydrogen peroxide in HeLa
cells: Protective role of mild heat preconditioning at 40°C.
Biochim Biophys Acta. 1813:1987–1999. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Runić R, Lockwood CJ, LaChapelle L,
Dipasquale B, Demopoulos RI, Kumar A and Guller S: Apoptosis and
Fas expression in human fetal membranes. J Clin Endocrinol Metab.
83:660–666. 1998.
|
|
49
|
Pang W, Zhang Y, Zhao N, Darwiche SS, Fu X
and Xiang W: Low expression of Mfn2 is associated with
mitochondrial damage and apoptosis in the placental villi of early
unexplained miscarriage. Placenta. 34:613–618. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Kumagai K, Otsuki Y, Ito Y, Shibata MA,
Abe H and Ueki M: Apoptosis in the normal human amnion at term,
independent of Bcl-2 regulation and onset of labour. Mol Hum
Reprod. 7:681–689. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Voss AK and Strasser A: The essentials of
developmental apoptosis. F1000Res. 9:F10002020. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Brill A, Torchinsky A, Carp H and Toder V:
The role of apoptosis in normal and abnormal embryonic development.
J Assist Reprod Genet. 16:512–519. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Ding Z, Zhou H, McCauley N, Ko G, Zhang KK
and Xie L: In ovo hyperglycemia causes congenital limb defects in
chicken embryos via disruption of cell proliferation and apoptosis.
Biochim Biophys Acta Mol Basis Dis. 1866:1659552020. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Honjo Y and Ichinohe T: Stage-specific
effects of ionizing radiation during early development. Int J Mol
Sci. 21:39752020. View Article : Google Scholar :
|
|
55
|
Takahashi M: Oxidative stress and redox
regulation on in vitro development of mammalian embryos. J Reprod
Dev. 58:1–9. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Cebral E, Carrasco I, Vantman D and Smith
R: Preimplantation embryotoxicity after mouse embryo exposition to
reactive oxygen species. Biocell. 31:51–59. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Wang R, Liu K, Zhang Y, Chen X and Wang X:
Evaluation of the developmental toxicity induced by E804 in
zebrafish embryos. Front Pharmacol. 11:322020. View Article : Google Scholar : PubMed/NCBI
|