Introduction
According to global cancer data, bladder cancer (BC)
is estimated to be the 9th most common type of cancer worldwide,
with ~400,000 new cases diagnosed annually (1,2).
The majority of BC cases are diagnosed as non-muscle invasive BC
(NMIBC), for which the mortality rate is generally low due to its
good prognosis; however, NMIBC often recurs and develops into MIBC
(3). MIBC is characterized by
rapid metastasis and progression, in addition to poor prognosis,
and is the main cause of BC-associated mortality (4-6),
with the 5-year overall survival rate being ~50% following surgery
(7). Therefore, it remains
urgent to identify novel molecular biomarkers and the underlying
molecular mechanism of BC tumorigenesis.
NF-κB can be activated following the stimulation of
various types of immune receptors, such as pattern recognition
receptors and toll-like receptors, and its activation is controlled
by the direct interaction with the IκB inhibitor family (8-10). The most well-established IκB
members are the proteins IκBα, IκBβ and IκBε, which keep NF-κB in
the cytoplasm of unstimulated cells (11). IκBζ is a newly identified IκB
family member protein, which is encoded by the NF-κB inhibitor ζ
(NFKBIZ) gene; it differs from IκBα, IκBβ and IκBε as it is
atypically localized in the nucleus rather than in the cytoplasm
(12). Notably, NFKBIZ has been
reported to be an important regulator of inflammation, cell
proliferation and survival (12-14). NFKBIZ is induced by toll-like
receptor 12 signaling and can coactivate both the canonical and
non-canonical NF-κB signaling pathways (15-17). Polymorphisms and mutations in
NFKBIZ are associated with numerous types of disease, including
psoriasis, spontaneous skin inflammation, invasive pneumococcal
disease and Crohn's disease (18-21); the majority of these diseases are
associated with inflammation. Furthermore, NFKBIZ has been
associated with cancer. For example, NFKBIZ expression is
upregulated in glioma and is associated with a poor prognosis
(22). In addition, NFKBIZ
expression is downregulated in mycosis fungoides, which is the most
common type of primary cutaneous T-cell lymphoma (23). Additionally, it has been
demonstrated that tumor formation in NFKBIZ-mutant mice is
significantly decreased, and low NFKBIZ expression in human
colorectal cancer cells affects cell proliferation (24). However, the mechanism underlying
the inhibition of tumor growth and whether NFKBIZ can regulate
other types of human carcinoma cells has not been elucidated.
Numerous signaling pathways are involved in the
occurrence and development of BC. Among them, the PI3K/AKT/mTOR
signaling pathway serves a crucial role in the pathogenesis of BC,
where it is usually overactivated and plays a central role in
stimulating cell proliferation, survival and metastasis (25-28). PTEN is an ubiquitous tumor
suppressor that serves an essential role in tumor cell
proliferation (25). Notably,
PTEN inactivation has been associated with the pathogenesis and
development of numerous types of solid tumors, such as colon,
gastric and lung cancer (29-31). PTEN inhibits the PI3K/AKT
signaling pathway, and a negative feedback association exists
between PTEN and phosphorylated (p)-PI3K, indicating that a
decrease in p-PI3K expression leads to an increase in PTEN
expression (27). Conversely,
the loss of PTEN function leads to the overactivation of the
PI3K/AKT signaling pathway, which has been demonstrated to
accelerate the growth, invasion and metastasis of BC (28). In addition, NF-κB and PTEN are
closely associated in numerous types of cancer, such as colon,
gastric and lung cancer (29-31), and NF-κB is regulated by
NFKBIZ.
Therefore, the present study aimed to explore the
association between NFKBIZ and PTEN, and whether NFKBIZ can affect
the development of BC by regulating PTEN and its downstream
PI3K/Akt signaling pathway. The expression levels of NFKBIZ in BC
were analyzed, and bioinformatics analysis was used to determine
the potential regulatory mechanism and clinical significance of
NFKBIZ in BC. In addition, functional experiments were performed to
investigate the function of NFKBIZ in BC. The current findings may
provide novel insights into the role of NFKBIZ in BC
tumorigenesis.
Materials and methods
Cell culture
The normal urinary tract epithelial cell line
(SV-HUC-1) and the BC cell lines (J82, UMUC-3, T24 and 5637) were
purchased from The Cell Bank of Type Culture Collection of The
Chinese Academy of Sciences. The identification of the BC cell
lines was conducted at the China Centre for Type Culture
Collection. SV-HUC-1 cells were cultured in F-12K medium (HyClone;
Cytiva) with 10% fetal calf serum (FCS; HyClone; Cytiva). J82 and
UMUC-3 cells were cultured in Minimum Essential Media (HyClone;
Cytiva) supplemented with 10% FCS. T24 and 5637 cells were cultured
in RPMI-1640 medium (HyClone; Cytiva) supplemented with 10% FCS.
Cells were maintained in a humidified atmosphere consisting of 5%
CO2 at 37°C.
Bioinformatics analysis and NFKBIZ
expression profile mining in The Cancer Genome Atlas (TCGA)
Relevant data or figures from TCGA database
(http://cancergenome.nih.gov) were
downloaded and analyzed using the Gene Expression Profiling
Interacting Analysis (GEPIA; http://gepia.cancer-pku.cn), UCSC Xena (http://xena.ucsc.edu) and UALCAN (http://ualcan.path.uab.edu) databases. The data from
GEPIA included NFKBIZ expression from 404 tumor and 28 adjacent
normal control tissues. The data from UCSC Xena included NFKBIZ
expression from 407 tumor and 29 adjacent normal control tissues.
The data from UALCAN included NFKBIZ expression from 408 tumor and
19 adjacent normal control tissues. NFKBIZ mRNA expression was also
explored at different stages, nodal metastasis status and
histological subtypes using the UALCAN database. Finally, the
'TCGAbiolinks' package in R language (https://www.rdocumentation.org/packages/TCGAbiolinks/versions/1.2.5)
was used to download the mRNA-reads count expression data from
TCGA-BLCA, including 408 BC and 19 normal bladder tissue samples.
An mRNA expression matrix was made with the raw counts of each RNA
in each sample. The 'Deseq2' package in R (http://www.bioconductor.org/packages/release/bioc/html/DESeq2.html)
was used to calculate the differential expression of mRNAs between
the BC tissues samples and paracancerous normal specimens. A
|fold-change| >2 and P<0.05 were used as the threshold
values. Survival analysis was performed using these differentially
expressed mRNAs using the 'ggsurv' package in R (https://www.rdocumentation.org/packages/GGally/versions/1.5.0/topics/ggsurv),
with P<0.05 used as the screening threshold. The survival curves
based on NFKBIZ expression were used to explore the effect of
NFKBIZ expression on survival and prognosis of patients with BC.
Kaplan-Meier analysis was used for survival analysis, and the
log-rank test was used to calculate the P-values.
Chemicals and lentivirus infection
Matrigel was purchased from BD Biosciences and the
PTEN inhibitor, VO-OHpic trihydrate (VO), was obtained from
MedChemExpress. The treatment condition of VO was 35 nM/l at room
temperature for 24 h. A lentiviral expression vector (pLV-NFKBIZ)
[OBiO Technology (Shanghai) Corp., Ltd.] was used for NFKBIZ gene
(NM_031419.4) delivery and stable overexpression. The MOI used to
infect T24 and 5637 cells was 40 and 20, respectively. T24 and 5637
cells (1×105) were seeded in 6-well plates and grown to
~50% confluency. Subsequently, the culture medium was removed, and
fresh culture medium containing lentiviral carrying NFKBIZ cDNA or
a negative control was added according to the MOI. The cells were
cultured in an incubator at 37°C with 5% CO2 for 18 h.
Next, the culture medium was removed and replaced with fresh
medium. After 48 h, culture medium containing 50 µg/ml
puromycin (Sigma-Aldrich; Merck KGaA) was added to remove any
non-transfected cells. The surviving cell clones were selected and
expanded. The lentiviruses were designated as pcDNA-NFKBIZ
(pSLenti-EF 1a-EGFP-P2A-Puro-CMV-NFKBIZ-3Flag). The empty vector
was used as a negative control (pcDNA-vector; pSLenti-EF1a-E
GFP-P2A-Puro-CMV-MCS-3Flag). Fluorescence microscopy
(magnification, ×40; IX73; Olympus Corporation) and western
blotting were used to evaluate infection efficiency.
Patient tissues
A total of 34 pairs of fresh BC and adjacent tissue
specimens (>5 cm from the tumor) were obtained from patients (25
males and 9 females) with a median age of 65 years (range, 46-79
years) with BC who were diagnosed by the Pathology Department of
The Renmin Hospital of Wuhan University (Wuhan, China) between July
2019 and December 2019. A small part of the resected tissue was
removed within 30 min of surgical resection. The removed tissue was
divided into two parts: A part of the tissue was washed with saline
and then fixed with 4% paraformaldehyde (Beyotime Institute of
Biotechnology) for 48 h at room temperature for paraffin embedding,
and another part of the removed tissue was rapidly frozen in liquid
nitrogen (-196°C) and stored at −80°C until required for RNA
extraction. Most of the remaining resected tissues were sent to the
pathology department for further diagnosis. BC was defined by two
pathologists. The tumor stage and grade of all patients were
diagnosed according to the 2009 TNM staging system and the 2004
World Health Organization grading system (32,33), respectively. All patients
provided written informed consent and the research protocols were
approved by the Research Ethics Committee of Renmin Hospital of
Wuhan University (approval no. WDRY2019-K035).
Reverse transcription-quantitative PCR
(RT-qPCR)
Total RNA was extracted from BC samples using
TRIzol® reagent (Invitrogen; Thermo Fisher Scientific,
Inc.) and reverse transcribed into cDNA using a RevertAid First
Strand cDNA Synthesis kit (Thermo Fisher Scientific, Inc.)
according to the manufacturer's protocol. qPCR was subsequently
performed using a SYBR Green mix (Takara Bio, Inc.), at a final
volume of 20 µl, on an ABI 7500 Real-Time PCR Detection
system (Applied Biosystems; Thermo Fisher Scientific, Inc.). The
following primer sequences were used for qPCR: NFKBIZ forward,
5′-CCG ATT CGT TGT CTG ATG GAC C-3′ and reverse, 5′-GCA CTG CTC TCC
TGT TTG GGT T-3′; and GAPDH forward, 5′-CCT TCA TTG ACC TCA ACT
ACA-3′ and reverse, 5′-GCT CCT GGA AGA TGG TGA T-3′. The following
thermocycling conditions were used for qPCR: Initial denaturation
at 95°C for 5 min; followed by 40 cycles at 95°C for 10 sec and
60°C for 35 sec. The melting curve step occurred at 60-95°C, at
increments of 0.5 sec for 5 sec. The relative expression levels of
NFKBIZ were calculated using the 2−ΔΔCq method (34) and GAPDH served as the endogenous
reference gene to normalize the expression levels of target
mRNAs.
Western blotting
Western blotting was performed as previously
described (35). Briefly, total
protein was extracted from transfected T24 and 5637 cells using
lysis buffer (cat. no. P0287; Beyotime Institute of Biotechnology)
and the protein concentration was determined using BCA Protein
assay reagent (Thermo Fisher Scientific, Inc.). Equal amounts of
protein (10 µg protein/lane) were separated via 10% SDS-PAGE
(Invitrogen; Thermo Fisher Scientific, Inc.) and transferred onto
nitrocellulose membranes (EMD Millipore), then blocked in 5%
skimmed milk for 2 h at room temperature. The membranes were
incubated with the following primary antibodies (all Cell Signaling
Technology, Inc.) at 4°C overnight: Anti-NFKBIZ (1:1,000; cat. no.
9244S), anti-β-actin (1:2,000; cat. no. 4970), anti-PTEN (1:1,000;
cat. no. 9188), anti-PI3K (1:1,000; cat. no. 4249), anti-p-PI3K
(1:1,000; cat. no. 17366), anti-AKT (1:1,000; cat. no. 4685),
anti-p-AKT (1:1,000; cat. no. 4060), anti-mTOR (1:1,000; cat. no.
2983) and anti-p-mTOR (1:1,000; cat. no. 5536). Following the
primary antibody incubation, the membranes were incubated with
HRP-conjugated goat anti-mouse IgG (cat. no. ab205719) and goat
anti-rabbit IgG (cat. no. ab205718) (both 1:1,000; Abcam) secondary
antibodies for 2 h at room temperature. Protein bands were
visualized using the Clarity Western ECL kit (Bio-Rad Laboratories,
Inc.). β-actin was used as the internal loading control.
Densitometric analysis was performed using ImageJ software (v1.8.0;
National Institutes of Health).
Colony formation assay
Each group of transfected T24 and 5637 cells was
digested with pancreatin, counted and plated into 6-well plates
with complete medium at a density of 500 cells/well. During the
14-day incubation, the medium was changed once. Following
incubation, the medium was discarded and cells were washed with PBS
three times, fixed with 4% paraformaldehyde for 20 min at room
temperature, then stained with 0.1% crystal violet solution for
another 20 min at room temperature. Cells were subsequently washed
with PBS and air-dried. The number of colonies was counted in four
randomly selected fields of view using an Olympus digital camera
(Olympus Corporation).
Wound healing assay
The transfected T24 and 5637 cells from each group
were seeded into 6-well plates and cultured to 80-90% confluence.
Scratches were subsequently made in the cell monolayer with a
200-µl sterile pipette tip. Each well was washed 2-3 times
with PBS and the plates were then cultured in serum-free medium.
The width of the wound was captured at 0 and 24 h using a light
microscope (magnification, ×200; Nikon Corporation), and the area
of cell migration (µm2) was measured using ImageJ
software (v1.8.0).
Transwell invasion assay
A Matrigel chamber (BD Biosciences) was used for
determining the invasive ability of the cells. Briefly, the
transfected T24 and 5637 cells in each group were digested and
counted, and 3×104 cells/well were plated in serum-free
RPMI medium into the upper chamber of 24-well Transwell plates,
which had been precoated with 40 µl Matrigel (BD
biosciences) at 37°C for 1 h. Complete medium supplemented with 10%
FCS was plated into the lower chambers. Following incubation for 24
h at 37°C, the cells remaining in the upper chamber were removed,
while the invasive cells in the lower chamber were fixed with 4%
paraformaldehyde at room temperature for 30 min and stained with
0.1% crystal violet solution at room temperature for 15 min
(Sigma-Aldrich; Merck KGaA). The stained cells were counted in
three randomly selected fields of view under a light microscope
(magnification, ×200; CKX41; Olympus Corporation) for each
chamber.
Tumor xenograft model
A total of 6 female BALB/c nude mice (age, 4 weeks;
weight, 14±2 g) were obtained from the Animal Experiment Center of
Wuhan University. All animals were randomly divided into two groups
(n=3/group) and housed in a standard environment (25°C; 50%
humidity; 12-h light/dark cycle) with free access to food and
water. A volume of 200 µl PBS containing ~1×106
T24 cells transfected with pc-NC or pc-NFKBIZ were injected into
the right scapular region of each mouse. The tumor volumes were
measured every three days after injection and calculated using the
following formula: (Length x width2)/2. After 1 month,
the mice were sacrificed with CO2 (20% of the chamber
volume was displaced per minute with CO2), and the tumor
weight was measured immediately. The maximum tumor size observed in
the present study was 1.5 cm3. The tumors were
subsequently collected for further research. All animal protocols
were approved by the Animal Care and Use Committee of Renmin
Hospital of Wuhan University, and all animal experiments were
conducted in accordance with the National Institutes of Health
Guide for the Care and Use of Laboratory Animals.
Immunohistochemistry (IHC) assay
The paraffin-embedded tissue samples were cut into
5-µm-thick sections and the immunohistochemical expression
of Ki67, NFKBIZ and PTEN in tissue sections were determined using
Strept Actividin-Biotin Complex IHC staining (Wuhan Boster
Biological Technology, Ltd.). The tissue sections were
deparaffinized in xylene for 15 min at room temperature, rehydrated
with a graded series of alcohol and heated at 105°C for 10 min for
antigen retrieval in citric acid buffer (0.01 M). The slides were
subsequently washed with PBS for 10 min and then treated with 3%
hydrogen peroxide solution to inhibit endogenous peroxidase
activity, blocked with 10% normal goat serum (cat. no. AR0009;
Wuhan Boster Biological Technology, Ltd.) for 30 min at 37°C, and
subsequently incubated with the following primary antibodies:
Anti-Ki67 (1:200; cat. no. 9449; Cell Signaling Technology, Inc.),
anti-NFKBIZ (1:1,000; cat. no. 9244S; Cell Signaling Technology,
Inc.) and anti-PTEN (1:125; cat. no. 9188; Cell Signaling
Technology, Inc.) for 2 h at 37°C. The slides were washed three
times with PBS and incubated with HRP-conjugated anti-mouse IgG
secondary antibody (1:2,000; cat. no. sc-2005; Santa Cruz
Biotechnology, Inc.) for 1 h at room temperature. The slides were
subsequently stained using the diaminobenzidine substrate kit (cat.
no. SK-4100; Vector Laboratories, Inc.). The nuclei were stained
with hematoxylin for 4 min at room temperature, and the slides were
observed under an inverted light microscope (Olympus Corporation;
magnification, ×200). Brown staining was considered to be Ki67-,
PTEN- or NFKBIZ-positive. A total of five fields of view were
randomly selected from each slide, and the degree of positive
staining and the proportion of positive cells were recorded and
scored by blinded observation by two pathologists. The staining
intensity was classified as follows: 0, negative; 1, weak; 2,
moderate; and 3, strong. The scores 0 and 1 were defined as low
expression, and the scores 2 and 3 were defined as high
expression.
Statistical analysis
Statistical analysis was performed using GraphPad
Prism 5 (GraphPad Software, Inc.) and SPSS 12.0 (SPSS, Inc.)
softwares, and quantitative data are presented as the mean ± SD.
The RT-qPCR data of tumor and non-tumor tissues from patients were
compared using paired t-test. An independent-sample t-test was used
to analyze the differences between two groups. One-way ANOVA with
Tukey's post-hoc test was used to analyze the statistical
differences among multiple groups. Categorical data were compared
using Fisher's exact test. Correlation analysis was preformed using
Spearman correlation coefficient algorithm. All experiments were
independently repeated in triplicate. P<0.05 was considered to
indicate a statistically significant difference.
Results
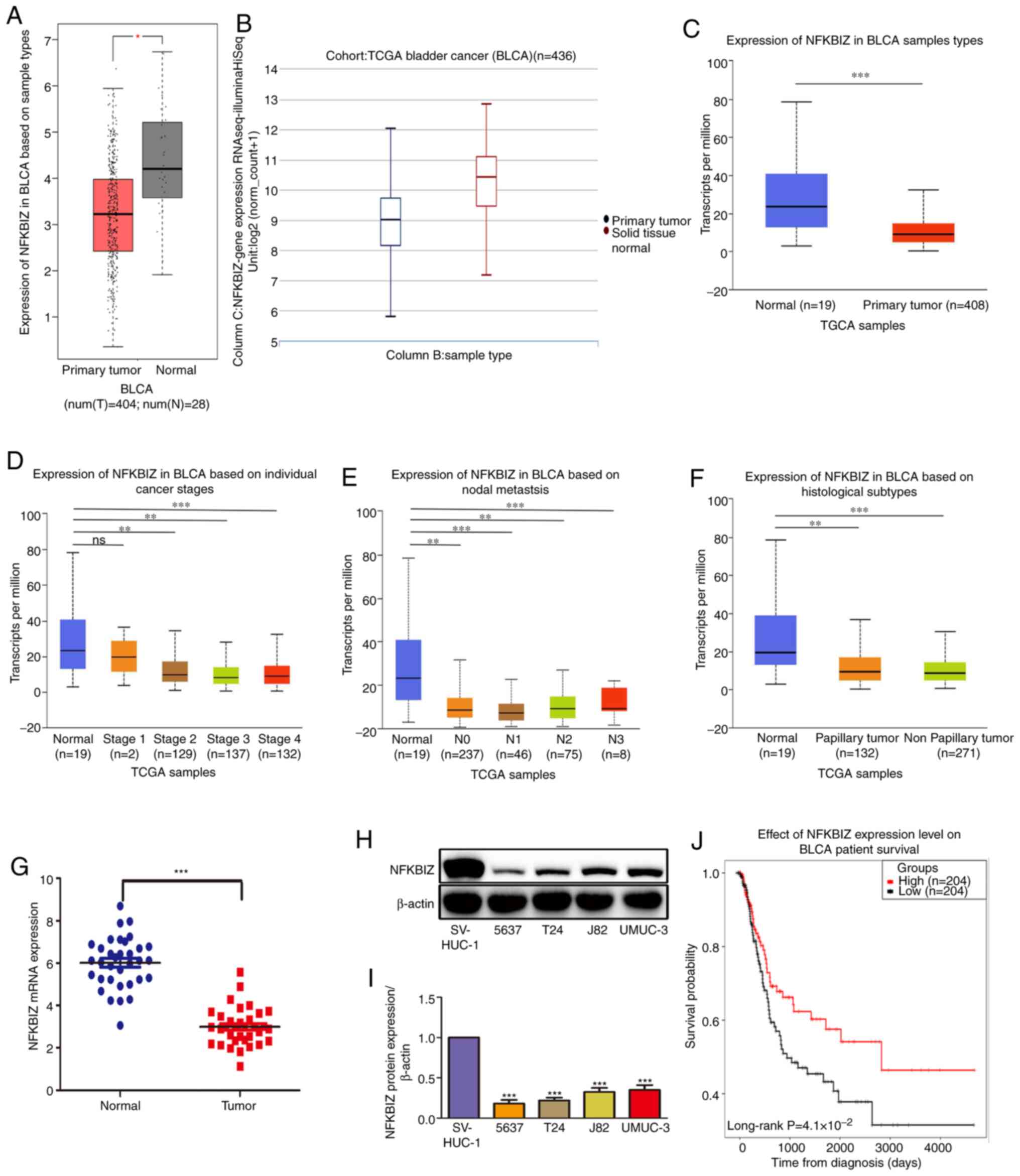
NFKBIZ expression is downregulated in
human BC tissues and is associated with a poor prognosis
As shown in Table
I, NFKBIZ expression in clinical patients with BC was
associated with tumor stage (P=0.005) and tumor grade (P=0.018).
However, no associations were observed between NFKBIZ expression
and other clinical features, such as sex (P=0.306), age (P=0.154),
tumor size (P=0.672) and tumor multiplicity (P>0.999). The
bioinformatics analysis revealed that NFKBIZ mRNA expression was
significantly downregulated in cancer tissues compared with in
normal tissues (Fig. 1A-C), and
at different stages, nodal metastasis status and histological
subtypes compared with normal tissues, which suggests that the low
NFKBIZ expression may be associated with a poor prognosis in
patients with BC (Fig. 1D-F). To
further validate the results obtained from databases, the
expression levels of NFKBIZ in clinical BC tissues and cell lines
were analyzed using RT-qPCR and western blotting, respectively. The
results revealed that the expression levels of NFKBIZ in BC tissues
and cell lines were significantly downregulated compared with in
adjacent non-tumor tissues and normal bladder epithelial cells,
respectively (Fig. 1G-I).
Additionally, survival analysis indicated that patients with BC
with high NFKBIZ expression had an increased overall survival rate
compared with those with low NFKBIZ expression (Fig. 1J). The current results suggested
that NFKBIZ may be act as a tumor suppressor gene and may serve an
important role in BC progression.
 | Table IAssociation between NFKBIZ expression
and clinical features of patients with bladder cancer (n=34). |
Table I
Association between NFKBIZ expression
and clinical features of patients with bladder cancer (n=34).
| Variable | Total | Low NFKBIZ
expression (n=28) | High NFKBIZ
expression (n=6) | P-value |
|---|
| Sex | | | | |
| Male | 25 | 22 | 3 | 0.306 |
| Female | 9 | 6 | 3 | |
| Age, years | | | | |
| ≥60 | 22 | 20 | 2 | 0.154 |
| <60 | 12 | 8 | 4 | |
| Tumor size, cm | | | | |
| ≥3 | 20 | 17 | 3 | 0.672 |
| <3 | 14 | 11 | 3 | |
| Multiplicity of
tumor | | | | |
| Single | 17 | 14 | 3 | >0.999 |
| Multiple | 17 | 14 | 3 | |
| Tumor grade | | | | |
| PUNLMP, low
grade | 8 | 4 | 4 | 0.018 |
| High grade | 26 | 24 | 2 | |
| Tumor stage | | | | |
| Ta, T1 | 10 | 5 | 5 | 0.005 |
| T2-T4 | 24 | 23 | 1 | |
| Lymph node
status | | | | |
| Negative | 26 | 22 | 4 | 0.609 |
| Positive | 8 | 6 | 2 | |
| Distant
metastasis | | | | |
| Absent | 34 | 28 | 6 | |
| Present | 0 | 0 | 0 | |
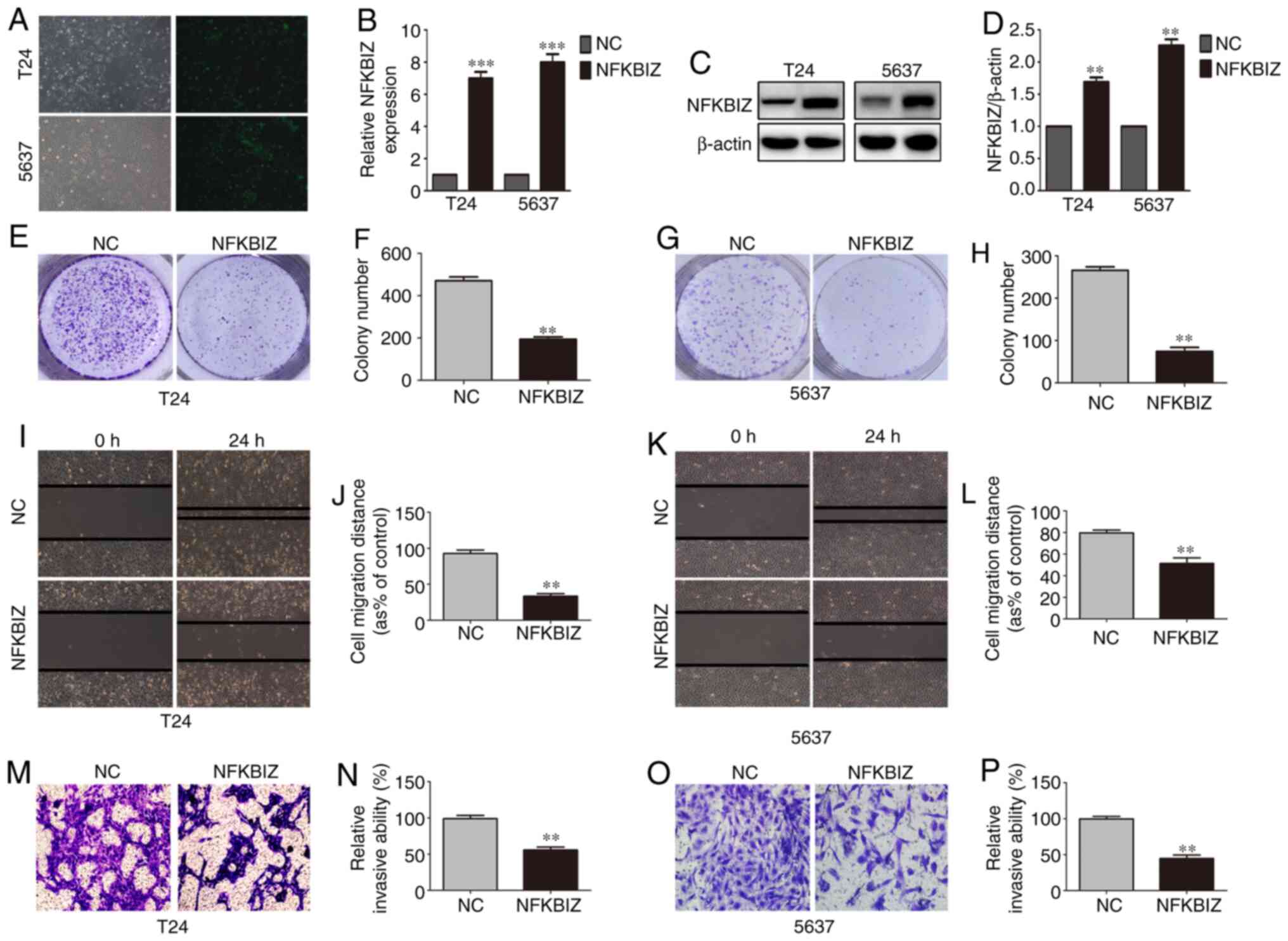
NFKBIZ suppresses cell proliferation,
migration and invasion
To investigate the biological role of NFKBIZ in BC
progression, T24 and 5637 cells were selected as in vitro
cell models, since NFKBIZ expression was the lowest in these cell
lines. The two cell lines were infected with lentivirus particles
against puromycin to induce the stable overexpression of NFKBIZ,
and the cell proliferative, migratory and invasive abilities were
determined. Fluorescence microscopy, RT-qPCR and western blot
analyses confirmed the transfection efficiency in the two cell
lines (Fig. 2A-D). According to
the results of the colony formation assay, the number of colonies
formed from T24 (Fig. 2E and F)
and 5637 (Fig. 2G and H) cells
in the pc-NFKBIZ group was significantly decreased compared with in
the pc-NC group. The results of the wound healing assay
demonstrated that compared with in the pc-NC group, the migration
rate of T24 (Fig. 2I and J) and
5637 (Fig. 2K and L) cells was
significantly inhibited in the pc-NFKBIZ group. Moreover, the
Transwell invasion assay results revealed that there were
significantly fewer invasive T24 (Fig. 2M and N) and 5637 (Fig. 2O and P) cells in the pc-NFKBIZ
group compared with in the pc-NC group. These results suggested
that BC cell proliferation, migration and invasion were suppressed
following the overexpression of NFKBIZ.
NFKBIZ expression affects BC growth in
vivo
To determine the effects of NFKBIZ expression on BC
growth in vivo, T24 cells were used to establish a xenograft
model. The tumor weights and volumes were significantly decreased
in the pc-NFKBIZ group compared with in the pc-NC group (Fig. 3A-C). In addition, the IHC results
demonstrated that Ki-67 expression (a proliferation marker) was
markedly decreased in the pc-NFKBIZ group compared with in the
pc-NC group (Fig. 3D). These
results indicated that BC growth was inhibited by the
overexpression of NFKBIZ in vivo.
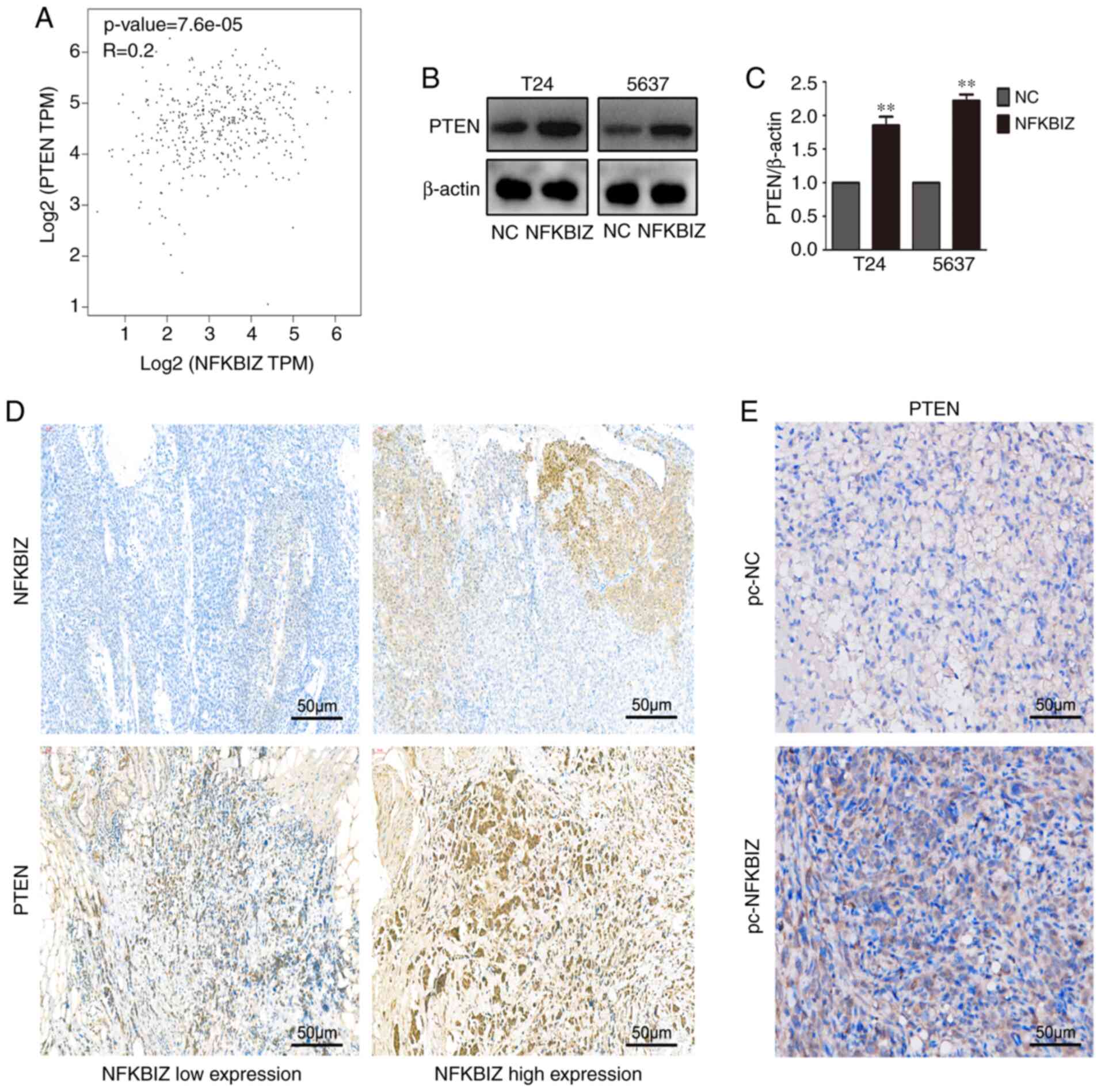
NFKBIZ and PTEN expression levels are
positively correlated
To investigate the potential mechanism of the
aforementioned biological functions of NFKBIZ in vivo and
in vitro, correlation analysis was performed. The results
identified a positive correlation between NFKBIZ and the
tumor-inhibiting factor, PTEN (Fig.
4A). To determine whether the overexpression of NFKBIZ induced
the expression levels of PTEN, its expression was analyzed in both
T24 and 5637 cells using western blotting (Fig. 4B and C). Moreover, according to
the RT-qPCR results of clinical BC tissues, samples in the NFKBIZ
high expression and low expression groups were selected to conduct
PTEN or NFKBIZ IHC, and to investigate whether PTEN expression was
also high in the NFKBIZ high expression group or low in the NFKBIZ
low expression group, to further explore whether NFKBIZ expression
was positively correlated with PTEN expression. The IHC results
from human BC and nude mice tumor tissues revealed that the
overexpression of NFKBIZ upregulated PTEN expression (Fig. 4D and E). These results suggested
the existence of a positive correlation between the expression
levels of NFKBIZ and PTEN.
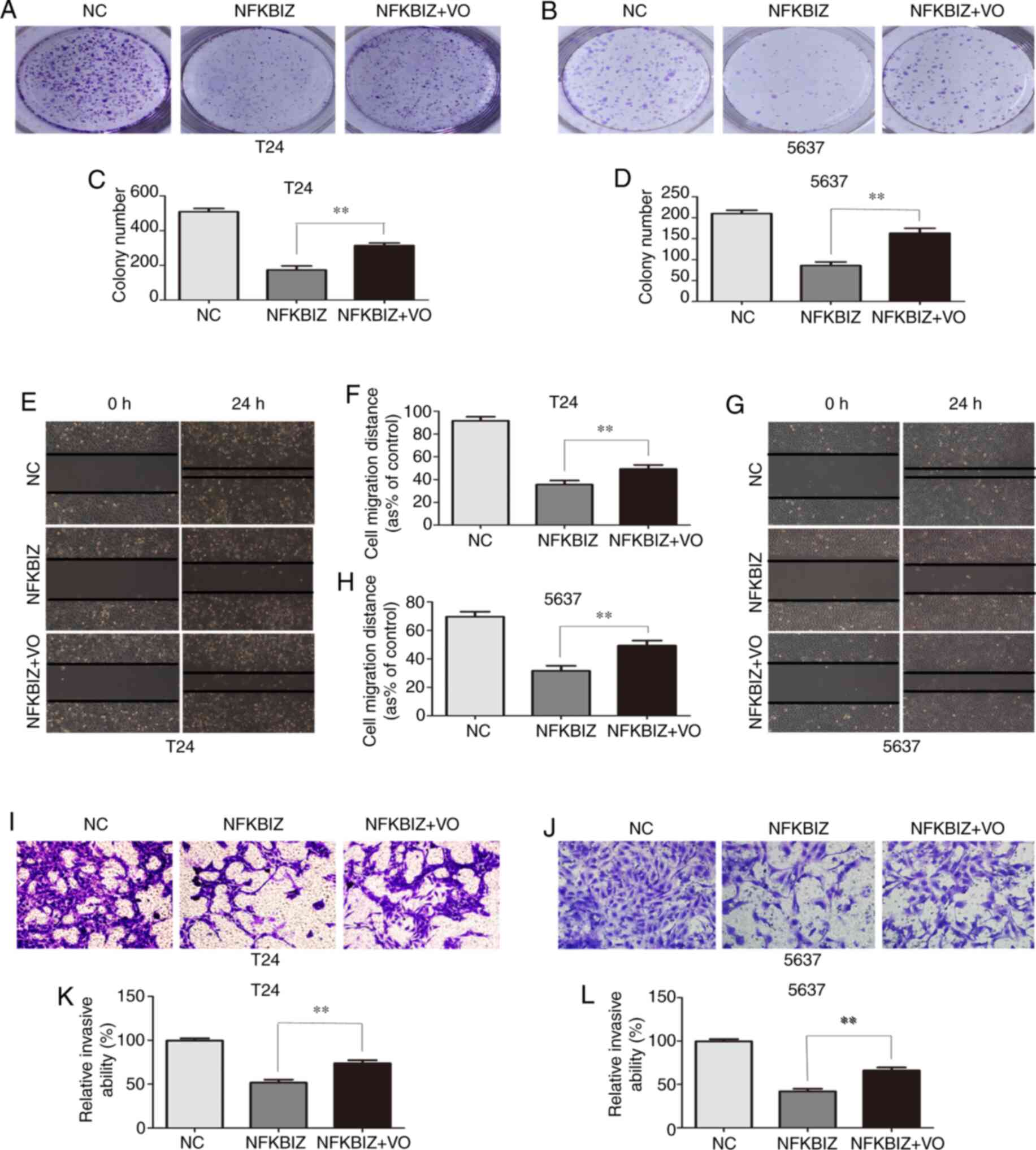
PTEN inhibition promotes BC cell
proliferation, migration and invasion
To further verify the positive correlation between
NFKBIZ and PTEN, NFKBIZ-overexpressing T24 and 5637 cells were
treated with the PTEN inhibitor VO (35 nM/l) at room temperature
for 24 h, to observe the effect on cell proliferation, migration
and invasion. According to the results of the colony formation
assay, the number of colonies formed from T24 (Fig. 5A and C) and 5637 (Fig. 5B and D) cells in the pc-NFKBIZ +
VO group was significantly increased compared with in the pc-NFKBIZ
group. The wound healing assay results demonstrated that, compared
with in the pc-NFKBIZ group, the migratory rate of T24 (Fig. 5E and F) and 5637 (Fig. 5G and H) cells was significantly
increased in the pc-NFKBIZ + VO group. In addition, the results of
the Transwell invasion assay revealed that the number of invasive
T24 (Fig. 5I and K) and 5637
(Fig. 5J and L) cells was
significantly increased in the pc-NFKBIZ + VO group compared with
in the pc-NFKBIZ group.
According to the aforementioned results, treatment
with VO enhanced the proliferative, migratory and invasive
abilities induced by NFKBIZ overexpression in T24 and 5637 cells.
Thus, the findings suggested that NFKBIZ may function as an
important tumor suppressor and may positively modulate the
tumor-inhibiting factor, PTEN, in BC.
NFKBIZ suppresses BC by regulating the
PTEN/PI3K/AKT signaling pathway in BC cells
The PI3K/AKT/mTOR signaling pathway serves an
important role in numerous types of cancer and regulates a wide
range of cellular processes, including survival, proliferation,
growth, metabolism, angiogenesis and metastasis (36). Thus, the present study determined
the effect of NFKBIZ overexpression on the signaling activity of
this pathway. The results revealed a significant downregulation in
the levels of key molecules involved in the AKT signaling pathway,
including p-PI3K, p-AKT and p-mTOR, in T24 and 5637 cells
overexpressing NFKBIZ; however, the expression levels of the total
proteins were unchanged (Fig.
6A-D). PTEN is directly upstream of the AKT signaling pathway
(23). To further determine
whether the activation of the PTEN/AKT/mTOR signaling pathway was
important for the oncogenic activity of NFKBIZ in BC cells,
NFKBIZ-overexpressing T24 and 5637 cells were treated with VO. The
treatment of T24 and 5637 cells overexpressing NFKBIZ with VO
rescued the inhibition of AKT signaling activity induced by the
overexpression of NFKBIZ (Fig.
6E-G). These findings suggested that the overexpression of
NFKBIZ may suppress BC cell proliferation, migration and invasion
by inducing PTEN expression, and the mechanism may depend on
PTEN/PI3K/AKT/mTOR signaling.
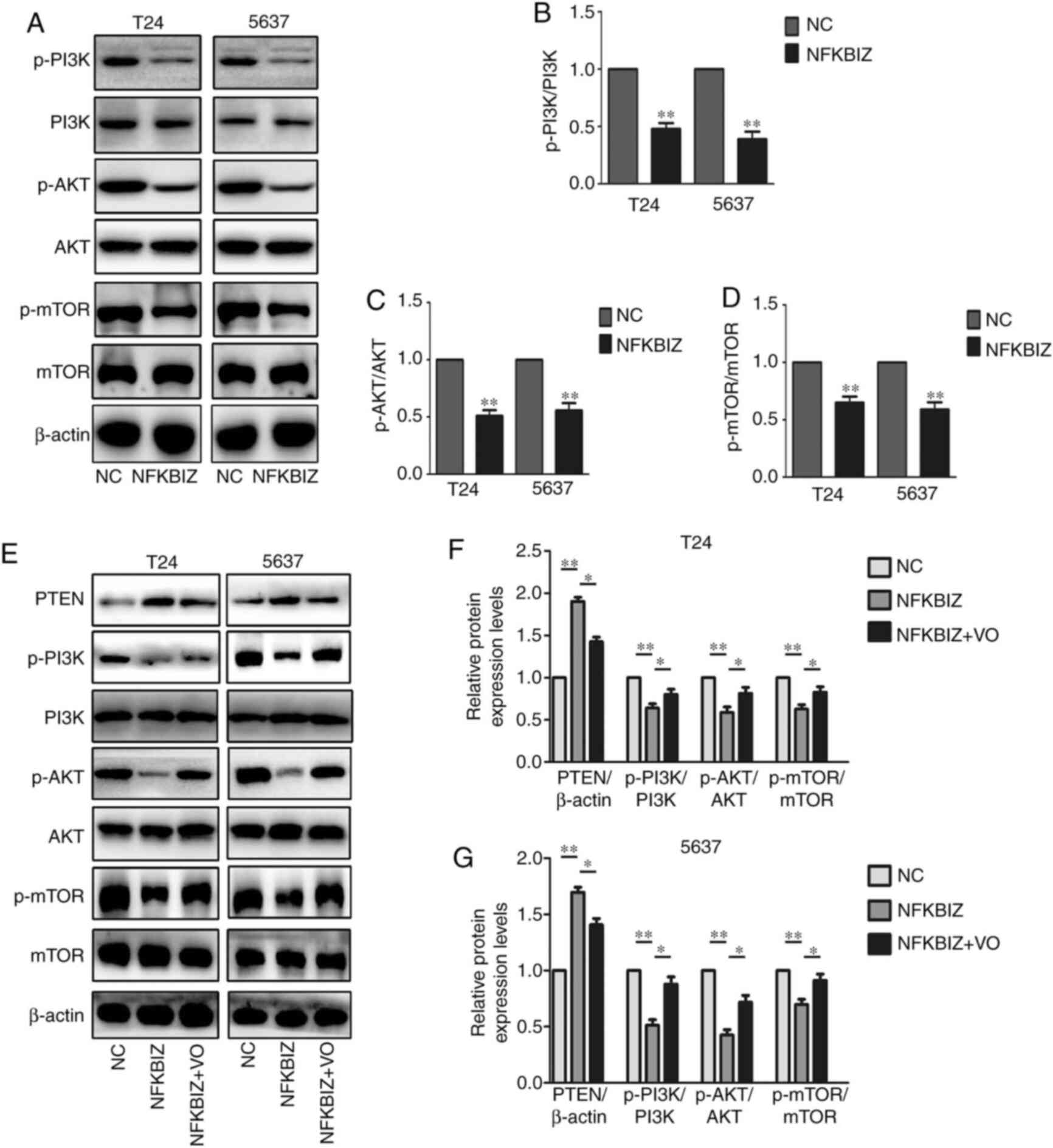 | Figure 6Overexpression of NFKBIZ induces BC
cell proliferation by regulating the PTEN/PI3K/Akt signaling
pathway in BC cells. (A) Representative western blot images and
quantitative analyses of related proteins, including (B)
p-PI3K/PI3K, (C) p-AKT/AKT and (D) p-mTOR/mTOR in transfected T24
and 5637 cells overexpressing NFKBIZ. **P<0.01 vs.
NC. (E) Representative western blot images and quantitative
analyses of related proteins, including PTEN, p-PI3K/PI3K,
p-AKT/AKT and p-mTOR/mTOR in transfected (F) T24 and (G) 5637 cells
overexpressing NFKBIZ and after VO treatment.
*P<0.05; **P<0.01. VO, VO-OHpic
trihydrate; NC, negative control; NFKBIZ, NF-κB inhibitor ζ; p,
phosphorylated; BC, bladder cancer. |
Discussion
BC is one of the most common and invasive malignant
tumors; however, the treatment of the disease is challenging.
Although some progress has been made in the early diagnosis and
comprehensive treatment of BC, the prognosis remains unsatisfactory
(37). Therefore, there is an
urgent requirement to elucidate the molecular mechanisms of the BC
regulatory network.
NFKBIZ, an inhibitor of the IκB protein, is
localized to the nucleus (12).
Several previous studies have reported that NFKBIZ serves a
potentially important role in modulating the progression of
numerous inflammatory diseases, such as spontaneous skin
inflammation, invasive pneumococcal disease and Crohn's disease
(19,20,38,39). Notably, inflammation is also
known to serve an important role in the development of cancer
(40). However, the underlying
pathological mechanisms of NFKBIZ in cancer remain elusive.
Therefore, the present study aimed to determine the role of NFKBIZ
in BC.
PTEN is a widely studied tumor suppressor gene
involved in the majority of human malignant tumors (25). PTEN signaling has been found to
inhibit the occurrence and development of cancer by inhibiting the
proliferation, migration and invasion of cancer cells (25). Therefore, the activation of the
PTEN signaling pathway has been considered to be a promising
therapeutic strategy for the clinical treatment of cancer (41). In a previous study, the
inactivation of the PTEN signaling pathway has been associated with
the invasiveness of BC (42).
PTEN, as an inhibitor of the PI3K/AKT signaling pathway, inhibits
the growth and invasion of tumor cells by directly inhibiting the
PI3K/AKT signaling pathway (43). In addition, the PI3K/AKT/mTOR
signaling pathway is a classical signaling pathway that has a
crucial role in regulating cell growth, metastasis and other
cellular processes in various types of cancer, including BC
(44-47). Previous studies have shown that
the PI3K/AKT/mTOR signaling pathway is activated in BC, and
patients with BC with simultaneous expression of PI3K/AKT/mTOR
exhibit a worse prognosis (48,49). NF-κB correlates with PTEN
(29-31) and is controlled by NFKBIZ
(12). To determine the
molecular mechanisms of NFKBIZ regulation over BC cell
proliferation, invasion and migration, the present study analyzed
the expression levels of proteins involved in the
PTEN/PI3K/AKT/mTOR signaling pathway.
In the present study, the expression levels of
NFKBIZ were significantly downregulated in BC samples and cell
lines, and positively associated with the overall survival rate of
patients. In addition, NFKBIZ expression at different stages, nodal
metastasis status and histological subtypes was significantly
decreased compared with in normal tissues, which suggested that low
NFKBIZ expression may be associated with a poor prognosis in
patients with BC. However, in 34 clinical samples, there was no
association between lymph node metastasis and NFKBIZ expression,
which was in contrast to the results obtained from the database
analysis and may be due to the limited number of clinical specimens
available for analysis. To verify whether NFKBIZ acted as a tumor
suppressor, lentiviruses overexpressing NFKBIZ were transfected
into BC cell lines to verify the results of the bioinformatics
analysis. Mechanistically, NFKBIZ suppressed BC cell proliferation,
migration and invasion. In addition, the data from the tumor
xenograft model studies revealed that BC growth was inhibited by
the overexpression of NFKBIZ, which provided further evidence for
the suggested tumor-suppressive role of NFKBIZ in BC. Ki67 has been
considered as a useful biomarker for BC, and its expression has
been associated with a poor prognosis (50). In the current study, the
upregulated expression levels of NFKBIZ were associated with the
downregulated expression levels of Ki67, which is suggestive of an
improved prognosis. Therefore, it may be inferred that the
expression levels of NFKBIZ may be downregulated as the stage of
the disease advances. Subsequently, the present study aimed to
determine which pathway affected the progression of BC. The results
of the current study revealed that PTEN expression was positively
correlated with NFKBIZ expression, which was proven using the GEPIA
database and IHC results obtained from clinical specimens. In
addition, further results revealed that NFKBIZ affected BC cells by
upregulating PTEN expression, which was followed by the
inactivation of the PI3K/AKT/mTOR signaling pathway. These effects
were reversed by the PTEN inhibitor VO. The current results
suggested that the regulation of NFKBIZ expression may be a novel
target in the treatment of BC.
In the present study, VO downregulated PTEN
expression and indirectly activated the PI3K/Akt/mTOR signaling
pathway, and then reversed the anticancer effects of NFKBIZ
overexpression, which is consistent with previous studies (51-53). Additionally, another study has
revealed that high concentration of VO can inhibit hepatocarcinoma
cell proliferation and induce senescence (54). Whether high concentration of VO
can directly inhibit BC cell proliferation should be further
explored in future studies.
In conclusion, to the best of our knowledge, the
findings of the present study revealed for the first time that
NFKBIZ may serve as a tumor suppressor to inhibit tumor growth both
in vitro and in vivo. In addition, NFKBIZ expression
was positively correlated with PTEN expression, which affected BC
tumorigenesis through the PTEN-mediated inhibition of the
PI3K/AKT/mTOR signaling pathway. Furthermore, the results
demonstrated that upregulated expression levels of NFKBIZ were an
indicator of an improved prognosis in patients with BC. Therefore,
the data of the present study revealed the molecular mechanism of
the NFKBIZ-mediated regulation of BC, and the NFKBIZ/PTEN/PI3K/AKT
axis may represent a novel clinical marker and therapeutic target
for BC.
Availability of data and materials
All data generated or analyzed during this study are
included in this published article.
Authors' contributions
TX designed the study. TX, WMY, JZN and XY performed
the experiments and drafted the manuscript. SMZ, KY and TB
participated in data analysis. TX, TR and FC were involved in the
discussion and interpretation of the results. TX and FC were
responsible for confirming the authenticity of the data. All
authors have read and approved the final manuscript.
Ethics approval and consent to
participate
All patients provided written informed consent and
the research protocols were approved by the Research Ethics
Committee of Renmin Hospital of Wuhan University (approval no.
WDRY2019-K035; Wuhan, China). All animal protocols were approved by
the Animal Care and Use Committee of Renmin Hospital of Wuhan
University, and all animal experiments were conducted in accordance
with the National Institutes of Health Guide for the Care and Use
of Laboratory Animals.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
Acknowledgments
Not applicable.
Funding
The present study was supported by grants from the National
Natural Science Foundation of China (grant. nos. 81870471 and
81800617) and the Science and Technology Major Project of Hubei
Province (grant. no. 2019AEA170).
References
|
1
|
Siegel RL, Miller KD and Jemal A: Cancer
statistics, 2018. CA Cancer J Clin. 68:7–30. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Antoni S, Ferlay J, Soerjomataram I, Znaor
A, Jemal A and Bray F: Bladder cancer incidence and mortality: A
global overview and recent trends. Eur Urol. 71:96–108. 2017.
View Article : Google Scholar
|
|
3
|
Song BN, Kim SK, Mun JY, Choi YD, Leem SH
and Chu IS: Identification of an immunotherapy-responsive molecular
subtype of bladder cancer. EBioMedicine. 50:238–245. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Chou R, Selph SS, Buckley DI, Gustafson
KS, Griffin JC, Grusing SE and Gore JL: Treatment of
muscle-invasive bladder cancer: A systematic review. Cancer.
122:842–851. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Jemal A, Siegel R, Ward E, Murray T, Xu J
and Thun MJ: Cancer statistics, 2007. CA Cancer J Clin. 57:43–66.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Levi F, La Vecchia C, Randimbison L and
Franceschi S: Incidence of infiltrating cancer following
superficial bladder carcinoma. Int J Cancer. 55:419–421. 1993.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Witjes JA, Compérat E, Cowan NC, De Santis
M, Gakis G, Lebret T, Ribal MJ, Van der Heijden AG and Sherif A;
European Association of Urology: EAU guidelines on muscle-invasive
and metastatic bladder cancer: Summary of the 2013 guidelines. Eur
Urol. 65:778–792. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Zhu G, Xu Y, Cen X, Nandakumar KS, Liu S
and Cheng K: Targeting pattern-recognition receptors to discover
new small molecule immune modulators. Eur J Med Chem. 144:82–92.
2018. View Article : Google Scholar
|
|
9
|
Herrero-Beaumont G, Pérez-Baos S,
Sánchez-Pernaute O, Roman-Blas JA, Lamuedra A and Largo R:
Targeting chronic innate inflammatory pathways, the main road to
prevention of osteoarthritis progression. Biochem Pharmacol.
165:24–32. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Napetschnig J and Wu H: Molecular basis of
NF-κB signaling. Annu Rev Biophys. 42:443–468. 2013. View Article : Google Scholar :
|
|
11
|
Baldwin AS Jr: The NF-kappa B and I kappa
B proteins: New discoveries and insights. Annu Rev Immunol.
14:649–683. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Willems M, Dubois N, Musumeci L, Bours V
and Robe PA: IκBζ: An emerging player in cancer. Oncotarget.
7:66310–66322. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Horber S, Hildebrand DG, Lieb WS,
Lorscheid S, Hailfinger S, Schulze-Osthoff K and Essmann F: The
Atypical Inhibitor of NF-κB, IκBζ, controls macrophage
interleukin-10 expression. J Biol Chem. 291:12851–12861. 2016.
View Article : Google Scholar
|
|
14
|
Wu Z, Zhang X, Yang J, Wu G, Zhang Y, Yuan
Y, Jin C, Chang Z, Wang J, Yang X and He F: Nuclear protein
IkappaB-zeta inhibits the activity of STAT3. Biochem Biophys Res
Commun. 387:348–352. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Nogai H, Wenzel SS, Hailfinger S, Grau M,
Kaergel E, Seitz V, Wollert-Wulf B, Pfeifer M, Wolf A, Frick M, et
al: IκB-ζ controls the constitutive NF-κB target gene network and
survival of ABC DLBCL. Blood. 122:2242–2250. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Yamamoto M, Yamazaki S, Uematsu S, Sato S,
Hemmi H, Hoshino K, Kaisho T, Kuwata H, Takeuchi O, Takeshige K, et
al: Regulation of Toll/IL-1-receptor-mediated gene expression by
the inducible nuclear protein IkappaBzeta. Nature. 430:218–222.
2004. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Krappmann D: Shaping oncogenic NF-κB
activity in the nucleus. Blood. 122:2146–2147. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Coto-Segura P, Gonzalez-Lara L, Batalla A,
Eiris N, Queiro R and Coto E: NFKBIZ and CW6 in adalimumab response
among psoriasis patients: Genetic association and alternative
transcript analysis. Mol Diagn Ther. 23:627–633. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Kim Y, Lee YS, Yang JY, Lee SH, Park YY
and Kweon MN: The resident pathobiont Staphylococcus xylosus in
Nfkbiz-deficient skin accelerates spontaneous skin inflammation.
Sci Rep. 7:63482017. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Sangil A, Arranz MJ, Guerri-Fernandez R,
Perez M, Monzon H, Payeras A, Andres M, Torviso J, Ibanez L, Garau
J and Calbo E: Genetic susceptibility to invasive pneumococcal
disease. Infect Genet Evol. 59:126–131. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Kerami Z, Duijvis NW, Vogels EW, van
Dooren FH, Moerland PD and Te Velde AA: Effect of interleukin-17 on
gene expression profile of fibroblasts from Crohn's disease
patients. J Crohns Colitis. 8:1208–1216. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Brennenstuhl H, Armento A, Braczysnki AK,
Mittelbronn M and Naumann U: IκBζ, an atypical member of the
inhibitor of nuclear factor kappa B family, is induced by
Y-irradiation in glioma cells, regulating cytokine secretion and
associated with poor prognosis. Int J Oncol. 47:1971–1980. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
van Kester MS, Borg MK, Zoutman WH,
Out-Luiting JJ, Jansen PM, Dreef EJ, Vermeer MH, van Doorn R,
Willemze R and Tensen CP: A meta-analysis of gene expression data
identifies a molecular signature characteristic for tumor-stage
mycosis fungoides. J Invest Dermatol. 132:2050–2059. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Kakiuchi N, Yoshida K, Uchino M, Kihara T,
Akaki K, Inoue Y, Kawada K, Nagayama S, Yokoyama A, Yamamoto S, et
al: Frequent mutations that converge on the NFKBIZ pathway in
ulcerative colitis. Nature. 577:260–265. 2020. View Article : Google Scholar
|
|
25
|
Jiang BH and Liu LZ: PI3K/PTEN signaling
in angiogenesis and tumorigenesis. Adv Cancer Res. 102:19–65. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Wang X, Huang H and Young KH: The PTEN
tumor suppressor gene and its role in lymphoma pathogenesis. Aging
(Albany NY). 7:1032–1049. 2015. View Article : Google Scholar
|
|
27
|
Carracedo A and Pandolfi PP: The PTEN-PI3K
pathway: Of feedbacks and cross-talks. Oncogene. 27:5527–5541.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Yang X, Cheng Y, Li P, Tao J, Deng X,
Zhang X, Gu M, Lu Q and Yin C: A lentiviral sponge for miRNA-21
diminishes aerobic glycolysis in bladder cancer T24 cells via the
PTEN/PI3K/AKT/mTOR axis. Tumour Biol. 36:383–391. 2015. View Article : Google Scholar
|
|
29
|
Zununi Vahed S, Barzegari A, Rahbar Saadat
Y, Goreyshi A and Omidi Y: Leuconostoc mesenteroides-derived
anticancer pharmaceuticals hinder inflammation and cell survival in
colon cancer cells by modulating NF-κB/AKT/PTEN/MAPK pathways.
Biomed Pharmacother. 94:1094–1100. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Zhou W, Fu XQ, Zhang LL, Zhang J, Huang X,
Lu XH, Shen L, Liu BN, Liu J, Luo HS, et al: The
AKT1/NF-kappaB/Notch1/PTEN axis has an important role in
chemoresistance of gastric cancer cells. Cell Death Dis.
4:e8472013. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Yang Z, Fang S, Di Y, Ying W, Tan Y and Gu
W: Modulation of NF-κB/miR-21/PTEN pathway sensitizes non-small
cell lung cancer to cisplatin. PLoS One. 10:e01215472015.
View Article : Google Scholar
|
|
32
|
Sobin LH, Gospodarowicz MK and Wittekind
C: TNM Classification of Malignant tumors. 7th edition.
Wiley-Blackwell; pp. 262–265. 2009
|
|
33
|
Sauter G, Algaba F, Amin MB, Busch C,
Cheville J, Gasser T, Grignon D, Hofstaedter F, Lopez-Beltran A and
Epstein J: Tumours of the urinary system: Non-invasive urothelial
neoplasias. World Health Organization Classification of Tumours:
Pathology and Genetics of Tumours of the Urinary System and Male
Genital Organs. Eble JN, Sauter G, Epstein JI and Sesterhenn I:
IARC Press; Lyon: pp. 89–157. 2004
|
|
34
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar
|
|
35
|
Yang K, Xiao Y, Xu T, Yu W, Ruan Y, Luo P
and Cheng F: Integrative analysis reveals CRHBP inhibits renal cell
carcinoma progression by regulating inflammation and apoptosis.
Cancer Gene Ther. 27:607–618. 2020. View Article : Google Scholar :
|
|
36
|
Ersahin T, Tuncbag N and Cetin-Atalay R:
The PI3K/AKT/mTOR interactive pathway. Mol Biosyst. 11:1946–1954.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Kluth LA, Black PC, Bochner BH, Catto J,
Lerner SP, Stenzl A, Sylvester R, Vickers AJ, Xylinas E and Shariat
SF: Prognostic and prediction tools in bladder cancer: A
comprehensive review of the literature. Eur Urol. 68:238–253. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Ishiguro-Oonuma T, Ochiai K, Hashizume K
and Morimatsu M: The role of IFN-γ in regulating Nfkbiz expression
in epidermal keratinocytes. Biomed Res. 36:103–107. 2015.
View Article : Google Scholar
|
|
39
|
Muromoto R, Tawa K, Ohgakiuchi Y, Sato A,
Saino Y, Hirashima K, Minoguchi H, Kitai Y, Kashiwakura JI, Shimoda
K, et al: IκB-ζ expression requires both TYK2/STAT3 activity and
IL-17-regulated mRNA stabilization. Immunohorizons. 3:172–185.
2019. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Coussens LM and Werb Z: Inflammation and
cancer. Nature. 420:860–867. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Keniry M and Parsons R: The role of PTEN
signaling perturbations in cancer and in targeted therapy.
Oncogene. 27:5477–5485. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Puzio-Kuter AM, Castillo-Martin M, Kinkade
CW, Wang X, Shen TH, Matos T, Shen MM, Cordon-Cardo C and
Abate-Shen C: Inactivation of p53 and Pten promotes invasive
bladder cancer. Genes Dev. 23:675–680. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Tamura M, Gu J, Tran H and Yamada KM: PTEN
gene and integrin signaling in cancer. J Natl Cancer Inst.
91:1820–1828. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Corti F, Nichetti F, Raimondi A, Niger M,
Prinzi N, Torchio M, Tamborini E, Perrone F, Pruneri G, Di
Bartolomeo M, et al: Targeting the PI3K/AKT/mTOR pathway in biliary
tract cancers: A review of current evidences and future
perspectives. Cancer Treat Rev. 72:45–55. 2019. View Article : Google Scholar
|
|
45
|
Ediriweera MK, Tennekoon KH and Samarakoon
SR: Role of the PI3K/AKT/mTOR signaling pathway in ovarian cancer:
Biological and therapeutic significance. Semin Cancer Biol.
59:147–160. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Xing X, Zhang L, Wen X, Wang X, Cheng X,
Du H, Hu Y, Li L, Dong B, Li Z and Ji J: PP242 suppresses cell
proliferation, metastasis, and angiogenesis of gastric cancer
through inhibition of the PI3K/AKT/mTOR pathway. Anticancer Drugs.
25:1129–1140. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Sathe A and Nawroth R: Targeting the
PI3K/AKT/mTOR pathway in bladder cancer. Methods Mol Biol.
1655:335–350. 2018. View Article : Google Scholar
|
|
48
|
Hou T, Zhou L, Wang L, Kazobinka G, Zhang
X and Chen Z: CLCA4 inhibits bladder cancer cell proliferation,
migration, and invasion by suppressing the PI3K/AKT pathway.
Oncotarget. 8:93001–93013. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Li Z, Hong S and Liu Z: lncRNA LINC00641
predicts prognosis and inhibits bladder cancer progression through
miR-197-3p/KLF10/PTEN/PI3K/AKT cascade. Biochem Biophys Res Commun.
503:1825–1829. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Zhang XG, Zhang T, Li CY, Zhang MH and
Chen FM: CD164 promotes tumor progression and predicts the poor
prognosis of bladder cancer. Cancer Med. 7:3763–3772. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Li JY, Huang WX, Zhou X, Chen J and Li Z:
Numb inhibits epithelial-mesenchymal transition via
RBP-Jκ-dependent Notch1/PTEN/FAK signaling pathway in tongue
cancer. BMC Cancer. 19:3912019. View Article : Google Scholar
|
|
52
|
Li X, Dai Y and Xu J: miR-21 promotes
pterygium cell proliferation through the PTEN/AKT pathway. Mol Vis.
24:485–494. 2018.
|
|
53
|
Qin J, Fu M, Wang J, Huang F, Liu H,
Huangfu M, Yu D, Liu H, Li X, Guan X and Chen X: PTEN/AKT/mTOR
signaling mediates anticancer effects of epigallocatechin-3-gallate
in ovarian cancer. Oncol Rep. 43:1885–1896. 2020.PubMed/NCBI
|
|
54
|
Augello G, Puleio R, Emma MR, Cusimano A,
Loria GR, McCubrey JA, Montalto G and Cervello M: A PTEN inhibitor
displays preclinical activity against hepatocarcinoma cells. Cell
Cycle. 15:573–583. 2016. View Article : Google Scholar : PubMed/NCBI
|