Introduction
Diabetic nephropathy (DN) is a severe microvascular
complication in diabetic patients and has been the main cause of
end-stage renal disease (1). DN
is mainly manifested as the thickening of the glomerular basement
membrane (GBM), the proliferation of mesangial cells and
extracellular matrix, and renal tubulointerstitial fibrosis
(2). Furthermore, its
pathogenesis is complex, involving hereditary, metabolic and
hemodynamic changes, as well as inflammation. A recent study has
reported the correlation between DN and inflammation (3). Macrophages are a type of immune
cell and are derived from monocytes. Macrophages have multiple
functions, such as phagocytosing cell fragments, releasing
chemokines factors and secreting inflammatory mediators (4). Furthermore, it has been established
that macrophages play a key role in the occurrence and development
of DN (5,6).
Bruton's tyrosine kinase (BTK) is an intracellular
non-receptor tyrosine kinase, and is one of the five members of the
Tec tyrosine kinase family (7).
BTK is expressed in myeloid cells, such as macrophages and tissue
plasma cells. Moreover, BTK signaling molecules have been
well-documented and it has been considered as an important signal
in immunoregulation (8). It has
recently demonstrated that BTK is crucial for the proliferation,
development and differentiation of B cells (9). In addition, BTK may be involved in
human innate immunity, particularly in relation to B cell and
macrophage involvement (10). It
has also been demonstrated that BTK can conduct cell signals by
regulating Toll-like receptor (TLR)2 and TLR4 in macrophages
(11). Previous studies have
proved that BTK-regulated inflammation is involved in various renal
diseases, such as IgA nephropathy and Lupus nephritis (12,13). The BTK inhibitor PCI-32765, also
known as ibrutinib, has been widely used as an antitumor drug for
the treatment of chronic lymphocytic leukemia (14). It has been reported that
ibrutinib can reduce MAPK and NF-κB pathway activation, and then
decrease the release of inflammatory mediators in macrophages
(15). Moreover, a recent study
demonstrated that a BTK inhibitor may serve as an effective
therapeutic strategy in severe COVID-19, due to its excellent
anti-inflammatory effect (16).
The inflammasome is a multi-protein complex
expressed in myeloid cells, and is mainly composed of sensors,
apoptosis-associated speck like protein containing a caspase
recruitment domain (ASC), and the caspase protease. Inflammasomes
exist in the cytoplasm of multiple cell types and have the ability
to induce the innate immune responses by sensing damage signals and
microbial attacks (17). The
Nod-like receptor (NLR) family pyrin domain containing 3 (NLRP3)
inflammasome is the most widely studied inflammasome, and is
composed of NLRP3, ASC and pro-caspase-1 (18). Moreover, the NLRP3 inflammasome
can be activated and is known to participate in the development of
inflammation by cleaving the inactive cytokines, IL-1β precursor
and IL-18 precursor, into active IL-1β and IL-18 (19,20). Recent studies have reported that
the activation of the NLRP3 inflammasomes plays an important role
in diabetic kidney disease (DKD) (21,22). Additionally, inflammasome
activation marker expression levels, such as caspase-1, IL-1β and
IL-18, have been shown to be positively associated with the
severity of albuminuria in patients with DN (23,24).
Although the therapeutic potential of NLRP3 is
undisputed, currently no clinically approved therapies exist that
target the NLRP3 inflammasome directly, at least to the best of our
knowledge (25). Recently, BTK
has attracted increased attention as a regulator of NLRP3. It has
been demonstrated that the inhibition of BTK can reduce the
inflammatory response by decreasing the activation of the NLRP3
inflammasome in numerous diseases, such as ischemic brain injury,
diet-induced metabolic inflammation and polymicrobial sepsis
(26-28). However, the role of BTK in DN
remains to be elucidated. On this basis, the present study aimed to
investigate the effect of BTK on DN and its association with the
NLRP3 inflammasome in the kidneys.
Materials and methods
Recruitment
A total of 49 patients with type 2 DN and 18 healthy
individuals were recruited from the Department of Nephrology in The
First Affiliated Hospital of Anhui Medical University between
January 1, 2017 and January 1, 2020. All patients exhibited no
fever symptoms or infectious diseases, and were diagnosed via renal
biopsy. Renal tissue sections from patients with DN were provided
by the Kidney Pathology Center at The First Affiliated Hospital of
Anhui Medical University. The para-carcinoma tissues from patients
with renal carcinoma was used as the control group (29). The clinical characteristics of
all the study subjects are presented in Tables SI and SII. Serum samples from
patients and healthy individuals were provided by the Clinical
Laboratory of The First Affiliated Hospital of Anhui Medical
University. All the patients and healthy individuals had signed
informed consent forms and all experiments were approved by the
Ethics Committee of Anhui Medical University (approval number:
5101309).
Animals and experimental groups
A total of 28 male C57BL/6J mice (age, 6-8 weeks;
weight, 18-22 g) were purchased from the Experimental Animal Center
of Nanjing Medical University. The animal experiment was performed
in accordance with guidelines, 'Principles of Laboratory Animal
Care and Use in Research' (Ministry of Health, Beijing, China). The
experiments were approved by the Anhui Medical University Ethics
Committee (approval number: 2020064). A total of 30 BTK-floxed
(Macrophage-specific knockout) mouse frozen embryos were purchased
from The European Mouse Mutant Archive and resuscitated at the
Experimental Animal Center of Nanjing Medical University. The
embryos were removed from liquid nitrogen and were resuscitated in
a water bath for 2 min at 35°C, then immediately placed in a Petri
dish with thawing solution (0.1 M sucrose PBS with 6% glycerin) for
1 min, followed by diluent solution (0.1 M sucrose PBS with 3%
glycerin) for 3 min. The embryos were then transferred to the
washing solution (0.1 M sucrose PBS with 0% glycerin) for 3 min
twice. The embryos were then cultured in HTF medium (cat. no.
MR-070-D; EMD Millipore) in an incubator at 37°C with 5%
CO2. Following 24 h of culture at 37°C, the normal
embryos were selected and transplanted into the uterus of 15 female
C57BL/6J mice (age, 6-8 weeks; weight, 18-22 g, obtained from the
Experimental Animal Center of Nanjing Medical University) (30,31). The offspring from the female mice
(the BTK-floxed mice) were then crossed with 6 male and 9 female
CMV-cre transgenic mice (age, 6-8 weeks; weight, 18-22 g; C57BL/6J
genetic background, obtained from the Experimental Animal Center of
Nanjing Medical University), and the offspring were self-crossed to
obtain BTK knockout mice (BTK−/− mice).
Streptozotocin (STZ) was purchased from
Sigma-Aldrich (Merck KGaA). The mice were administered STZ daily,
at a dose of 50 mg/kg of body weight for 5 days. In total, 28 mice
were randomly divided into 4 groups as follows: i) Wild-type (WT)
group (WT littermate C57BL/6J mice; n=6); ii) STZ group (WT
littermate C57BL/6J mice + STZ; n=8); iii) BTK−/− group
(n=6); and iv) BTK−/− + STZ group (BTK−/− +
STZ; n=8). All mice were maintained under standard feeding
conditions (temperature, 20±2°C; humidity, 45-55%; 12-h light/dark
cycle) and were granted free access to food and water. After 12
weeks of rearing, all mice were anesthetized with an
intraperitoneal injection of sodium pentobarbital (50 mg/kg body
weight) and euthanized via exsanguination by drawing 1.2 ml blood.
All mice were observed for 20 min to verify death, according to the
criterion of breathing cessation and cardiac arrest. The
experimental humane endpoints that were established in the present
study, included loss of appetite, rapid weight loss (>20% of
body weight within a week), severe infections, weakness and organ
failure.
Genotype identification
After euthanizing the mice, 3 mm mouse tail was
obtained and used for DNA extraction (cat. no. 10185ES50, Shanghai
Yeasen Biotechnology Co., Ltd.). Following amplification in a
QuantStudio™ 6 Flex Real-Time PCR System (Thermo Fisher Scientific,
Inc.), gene expression was detected by 1.5% agarose gel
electrophoresis. The PCR conditions were as follows: 95°C for 5
min, followed by 40 cycles of 95°C for 30 sec, 58°C for 30 sec and
72°C for 30 sec. The primers (Shanghai Sangon Biotech Co., Ltd.)
were as follows: BTK-FRT-tF1, CTG CAT AAG GCA GGT GCC ACT AA, and
BTK-FRT-tR1, CAT CAG AAG CAG GCC ACC CA; BTK-loxP-tF1, TTG CAT AAA
GGC AGC A AT ACA ACA G, and BTK-loxP-tR1, TAG CTC CAG AAC TCA ATG
ACA AAG A; lacZ3F4, CCG GTC GCT ACC ATT ACC AGT; IMP C-loxptR, ATG
GCG AGC TCA GAC CAT AAC; Cre KT 119, TGC CAC GAC CAA GTG ACA GCA
ATG, and Cre KT 120, ACC AGA GAC GGA AAT CCA TCG CTC.
Immunohistochemical staining
The paraffin-embedded kidney tissues from mice were
cut into 3-µm-thick sections. All sections were
deparaffinized, rehydrated and subjected to antigen retrieval in
0.01 M citrate buffer (pH 6.0) by microwaving. After blocking with
10% goat serum (cat. no. SL038; Beijing Solarbio Science &
Technology Co., Ltd.) at room temperature, the sections were
incubated with anti-CD68 (cat. no. ab213363; 1:300; Abcam),
anti-BTK (1:200; cat. no. 8547; Cell Signaling Technology, Inc.),
anti-F4/80 (1:200; cat. no. ab111101; Abcam), anti-collagen
(Col)-IV (1:200; cat. no. ab236640; Abcam), anti-fibronectin (FN;
1:200; cat. no. ab2413; Abcam), anti-Wilms Tumor 1 (WT1)
transcription factor (WT1; 1:200; cat. no. ab267377; Abcam) and
anti-Nephrin (1:200; cat. no. ab216341; Abcam) antibodies overnight
at 4°C. Secondary biotin-labeled goat anti-mouse/rabbit IgG
antibodies (1:200; cat. no. PV-6000; OriGene Technologies, Inc.)
were then added for 2 h at room temperature. Finally,
3,3-diaminobenzidine (cat. no. ZLI-9017; Sigma-Aldrich; Merck KGaA)
and hematoxylin at room temperature were used for staining. All
images were captured with a Zeiss microscope (Zeiss AG) and
analyzed using ImageJ 1.0 software (National Institutes of Health).
Six fields were selected for statistical analysis in each sample.
The mean value was calculated and used as the final sample of the
immunohistochemistry data. The mean values of all samples were used
for final statistical analysis.
ELISA
The levels of TNF-α, IL-1β and monocyte
chemoattractant protein-1 (MCP-1) in serum were detected with the
use of human and mouse ELISA kits (cat. no. P06804, P10749, P10148;
RayBiotech, Inc.). The urinary albumin excretion rate (UAER) was
detected using a mouse albumin ELISA kit (cat. no. ab108792,
Abcam). The detection methods were executed according to the
manufacturer's instructions.
Detection of HbA1c and blood glucose
HbA1c was detected using the Glycosylated hemoglobin
A1c Assay kit (cat. no. H464-1, Nanjing Jiancheng Bioengineering
Institute). The blood glucose levels were detected using the
glucose meter (Contour TS, Bayer). The detection methods were
executed according to the manufacturer's instructions.
Western blot (WB) analysis
Renal tissues were lysed in RIPA lysis buffer (cat.
no. P0013B, Beyotime Institute of Biotechnology). The mixture was
centrifuged at 15,294 × g at 4°C for 30 min and then the pellet was
removed. Protein samples were separated via 10 or 15% SDS-PAGE, and
transferred to the nitrocellulose membrane (cat. no. HATF00010,
Merck KGaA). The band distribution was observed via Ponceau
staining. The membrane was then blocked with 5% skim milk powder
(containing TBS-Tween-20) at room temperature for 2 h, followed by
incubation with the following primary antibodies at 4°C for 12 h:
Anti-β-actin (1:35,000, cat. no. 66009-1-Ig; ProteinTech Group,
Inc.), anti-TNF-α (1:1,000; cat. no. ARG10158; arigo
Biolaboratories Corp.), anti-IL-1β (1:1,000; cat. no. 12242; Cell
Signaling Technology, Inc.), anti-MCP-1 (1:1,000; cat. no.
ARG57649; arigo Biolaboratories Corp.), anti-BTK (1:1,000; cat. no.
ab25971; Abcam), anti-phosphorylated (p)-BTK (1:1,000; cat. no.
87457; Cell Signaling Technology, Inc.), anti-inducible nitric
oxide synthase (iNOS; 1:1,000; cat. no. ab178945; Abcam),
anti-p-NF-κB p65 (1:1,000; cat. no. 8242; Cell Signaling
Technology, Inc.), anti-NF-κB p65 (1:1,000; cat. no. 8242; Cell
Signaling Technology, Inc.), anti-nicotinamide adenine dinucleotide
phosphate oxidase 1 (NOX1) (cat. no. ab131088; 1:1,000; Abcam),
anti-nicotinamide adenine dinucleotide phosphate oxidase 4 (NOX4)
(1:1,000; cat. no. bs-1091R; Beijing Boaosen Biotechnology, Co.,
Ltd.), anti-NLRP3 (1:1,000; cat. no. ab214185; Abcam),
anti-caspase-1 (1:1,000; cat. no. ab138483; Abcam) and anti-ASC
(1:200; cat. no. sc-376916; Santa Cruz Biotechnology, Inc.). After
washing, the membrane was incubated with HRP-labeled secondary
antibodies (cat. nos. SA00001-1 and SA00001-2; 1:2,000; ProteinTech
Group, Inc.) at room temperature for 1 h. The signals were detected
with the use of a chemiluminescent gel imaging system and the
relative ratio was semi-quantified using ImageJ 1.0 software
(National Institutes of Health).
Reverse transcription-quantitative
(RT-qPCR)
TRIzol® reagent (Thermo Fisher
Scientific, Inc.) was used to extract the total RNA in renal
tissues. cDNA was synthesized from total RNA using a Reverse
Transcription kit (Vazyme Biotech Co., Ltd.). cDNA was amplified
and detected in a QuantStudio™ 6 Flex Real-Time PCR System (Thermo
Fisher Scientific, Inc.) using the miScript SYBR-Green PCR kit
(Qiagen GmbH) to determine the quantity of mRNA. The conditions
were as follows: 95°C for 10 min, followed by 40 cycles of 95°C for
10 sec, and 60°C for 20 sec. The expression levels of all genes
were standardized with the reference gene GAPDH using the
2−ΔΔCq method (32).
The primer sequences used were selected according to the previous
studies (15,33).
Histological staining
The kidney tissue was fixed in 4% paraformaldehyde
for 16 h at room temperature and then dehydrated through an ethanol
gradient. Subsequently, the kidney tissue was paraffin-embedded and
cut into 3-µm-thick sections. The sections were dewaxed and
stained with periodic acid-Schiff (PAS) staining kit (cat. no.
G1281; Beijing Solarbio Science & Technology Co., Ltd.) and the
Masson's staining kit (cat. no. G1340; Beijing Solarbio Science
& Technology Co., Ltd.). The staining methods were executed
according to the manufacturer's instructions. All images were
acquired using a light microscope (BX51, Olympus Corporation). The
glomerular mesangial expansion, tubulointerstitial injury index and
fibrosis area were evaluated and graded using ImageJ 1.0 software
(National Institutes of Health).
Transmission electron microscopy
(TEM)
Kidney tissue was quickly placed in 2%
glutaraldehyde for 24 h. The tissue was then fixed with 1% acetic
acid at room temperature for 2 h, followed by acetone (50, 70, 80,
90 and 100%; twice for 15 min each time) for dehydration; sections
were embedded in epoxy resin. The resin block was cut into
ultra-thin sections of 70 nm, stained with lead citrate at 37°C and
then observed under a transmission electron microscope (H-7700;
Hitachi, Ltd.). GBM thickness and foot process fusion were analyzed
using ImageJ 1.0 software (National Institutes of Health).
Statistical analysis
Data were statistically analyzed using SPSS 23.0
(IBM Corp.). Normally distributed data are presented as the mean ±
SD, and non-normally distributed data were expressed as the median
(p25, p75). Pearson's test was used to analyze the correlation
between normal distribution data. Spearman's test was used to
analyze the correlation between non-normal distribution data. The
homogeneity of variance was tested using the Levene method.
Differences between two groups were assessed using an independent
samples t-test. For multiple group comparisons, the significant
differences were analyzed by one-way ANOVA, followed by
Bonferroni's post hoc analysis. P<0.05 was considered to
indicate a statistically significant difference.
Results
BTK is markedly activated in renal
macrophages in the kidneys of patients with DN
In the present study, the clinical indicators of 49
patients with type 2 DN and 18 healthy individuals were collected
from the Department of Nephropathy and Clinical Laboratory at the
First Affiliated Hospital of Anhui Medical University. The results
demonstrated that the DN group had significantly higher levels of
haemoglobin A1c, systolic blood pressure (BP), diastolic BP, mean
arterial pressure, proteinuria, serum creatinine, blood urea
nitrogen, estimated glomerular filtration rate (eGFR) and serum
uric acid (Table SI). The role
of macrophage-mediated inflammation was evaluated by ELISA and
immunohistochemistry in patients with DN. The data revealed that
the TNF-α, IL-1β and MCP-1 serum levels were significantly
increased in the DN group (Fig.
S1A). Additionally, the TNF-α and CD68 expression levels were
significantly increased in the renal tissue from the DN group
(Fig. S1B and C). This
indicated that patients with DN exhibit more severe inflammation
and macrophage infiltration in the kidneys. Moreover, the results
also revealed that the DN group exhibited an increased BTK
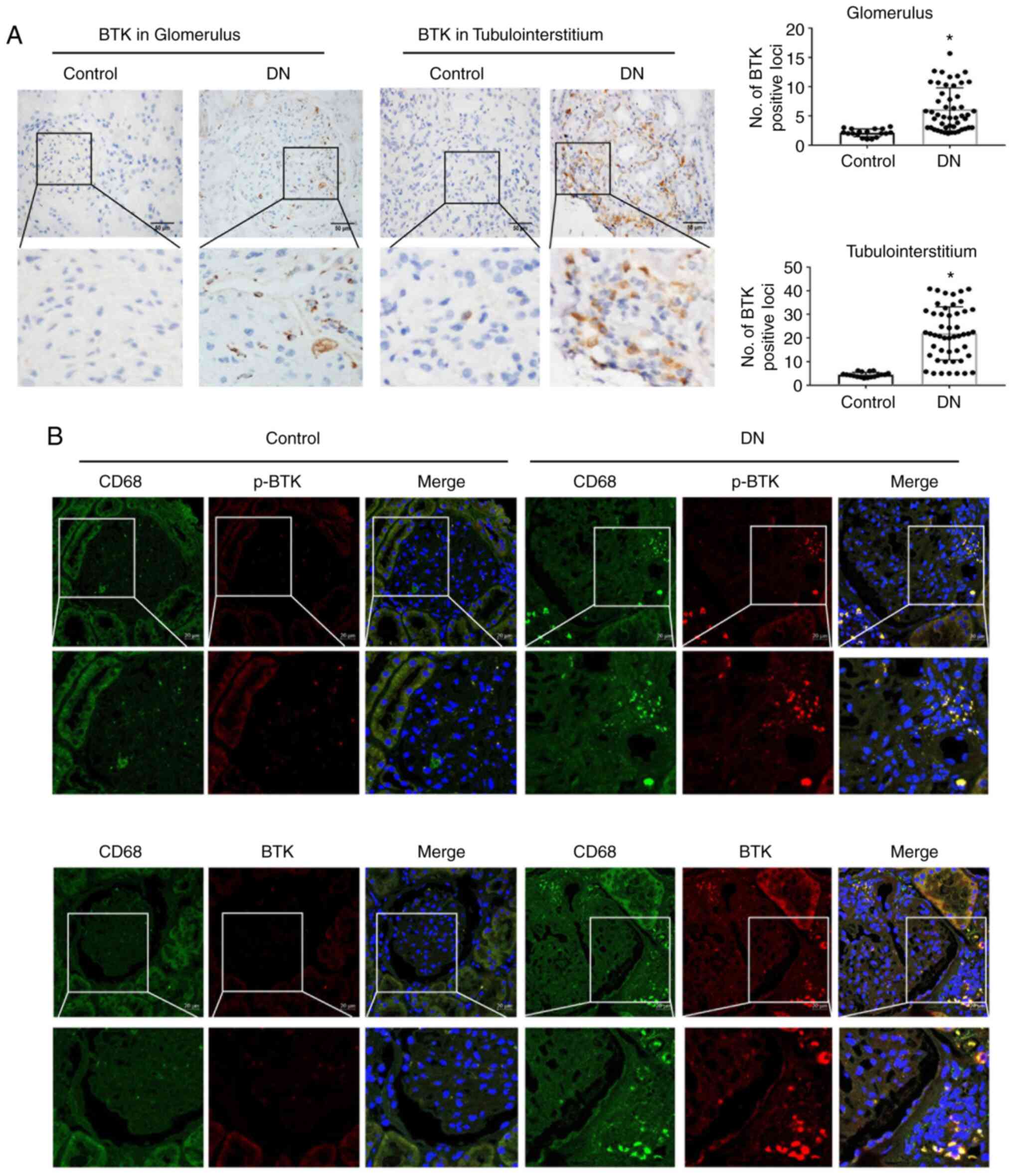
activation in comparison with the control group (Fig. 1A).
To further observe the activation of BTK in renal
macrophages, CD68 was used to label macrophages followed by
co-staining with BTK and p-BTK. The data indicated that the CD68
expression levels were increased in the DN group in comparison with
the control group. Moreover, the BTK and p-BTK expression levels
were increased in DN group (Fig.
1B). Furthermore, iNOS and NLRP3 were also notably activated in
renal macrophages in the kidneys of patients with DN (Fig. S2). This suggested that
macrophages exhibited an increased BTK activation in the kidneys of
patients with DN; thus, the activation of the NLRP3 inflammasome
may be involved in this process.
BTK expression is correlated with
clinical indicators and pathological changes in the kidneys of
patients with DN
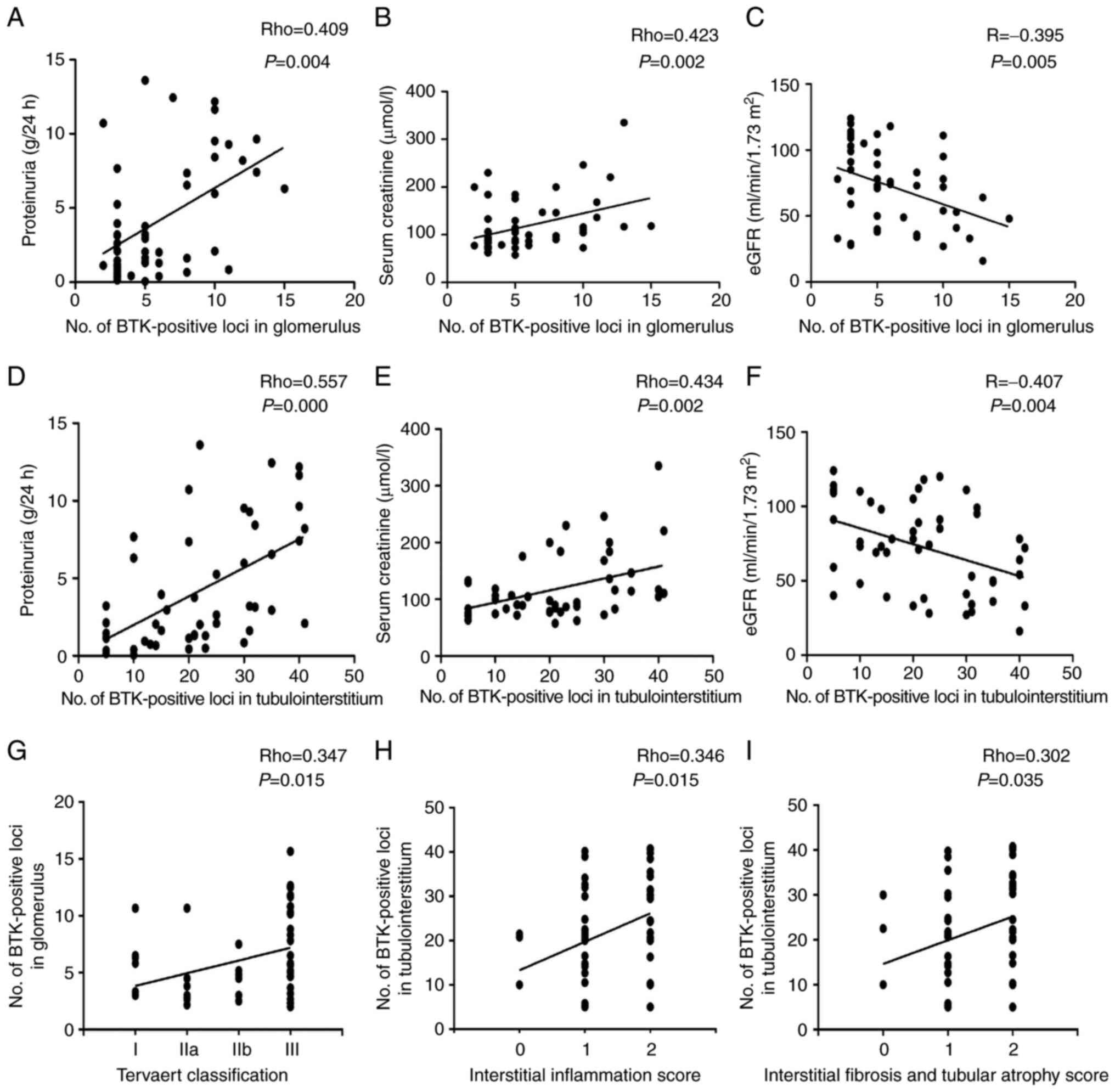
Correlation analysis was conducted to determine the
correlation between BTK expression and clinical indicators in
patients with DN. The data demonstrated that the expression level
of BTK positively correlated with proteinuria and serum creatinine
levels, and negatively correlated with eGFR levels (Fig. 2A-F). The pathological scores of
all renal patient tissues were also assessed (Table SII). The results depicted that
the expression level of BTK in the glomerulus correlated with the
Tervaert classification. In addition, renal BTK expression in the
tubulointerstitium also correlated with interstitial fibrosis, the
tubular atrophy score and the interstitial inflammation score
(Fig. 2G-I). This indicated that
BTK expression was significantly associated with disease
progression in patients with DN.
BTK knockout alleviates renal injury in
mice with STZ-induced diabetes
To further investigate the effects of BTK on DN, BTK
knockout mice were used to establish a diabetic model. Mouse
genotyping was initially performed (Fig. S3). After 12 weeks of rearing,
the general indicators and metabolic parameters were measured in
all mice. HbA1c was detected using an assay kit. The blood glucose
levels were detected using a Bayer glucose meter. UAER was detected
using a mouse albumin ELISA kit. The results identified that in
comparison with the WT group, blood glucose, HbA1c, kidney/body
weight and urinary protein excretion in the STZ group were
significantly increased, while the mouse body weight was
significantly decreased. The level of blood glucose, HbA1c, body
weight and the kidney/body ratio exhibited no changes in the
BTK−/− + STZ group compared with the STZ group. However,
the UAER was significantly reduced when BTK was knocked out
(Table I). These findings
suggest that BTK knockout may protect mice from diabetic kidney
injury.
 | Table IClinical indicators of mice
(n=28). |
Table I
Clinical indicators of mice
(n=28).
| Clinical
indicator | WT | STZ |
BTK−/− | BTK−/− +
STZ |
|---|
| Body weight | 28.88±0.57 | 22.56±1.93a | 27.16±1.02 | 23.66±1.35 |
| Kidney/body ratio
(%) | 0.62±0.04 | 1.00±0.11a | 0.70±0.06 | 0.97±0.06 |
| BG (mmol/l) | 6.51±0.68 | 22.17±1.69a | 7.22±0.60 | 22.76±2.85 |
| HbA1c (%) | 4.42±0.65 | 6.98±0.72a | 4.77±0.89 | 7.14±1.10 |
| UAER (µg/24
h) | 28.41±14.48 |
449.87±101.43a | 26.31±18.22 |
304.39±173.74b |
Subsequently, the changes in mouse renal injury were
detected. PAS staining was used to observe the pathologic changes
in mouse kidneys and Masson's trichrome staining was used to
evaluate the fibrosis area. The results demonstrated that the STZ
group exhibited more severe mesangial expansion and proliferation
than the WT group; this was significantly reversed in the
BTK−/−+STZ group (Fig.
3A). However, no marked differences amongst groups was observed
as regards the fibrosis area (Fig.
S4). ColIV and FN were used to observe the proliferation of the
mesangial matrix, while WT1 and Nephrin were used to evaluate
podocyte injury in the mouse kidney. It was found that the ColIV
and FN expression levels were increased in the STZ group (Fig. 3A). Moreover, the numbers of
WT1-positive cells and Nephrin-positive cells were significantly
reduced (Fig. 3B). However, in
the BTK−/− + STZ group, the number of macrophages and
the mesangial matrix were both decreased, and the number of
podocytes was significantly increased. A reduced F4/80 expression
was also observed in the BTK−/− + STZ group, as compared
with the STZ group (Fig. 3B).
These results suggested that BTK knockout decreased macrophage
infiltration and alleviated the accumulation of extracellular
matrix and podocyte injury in diabetic mice.
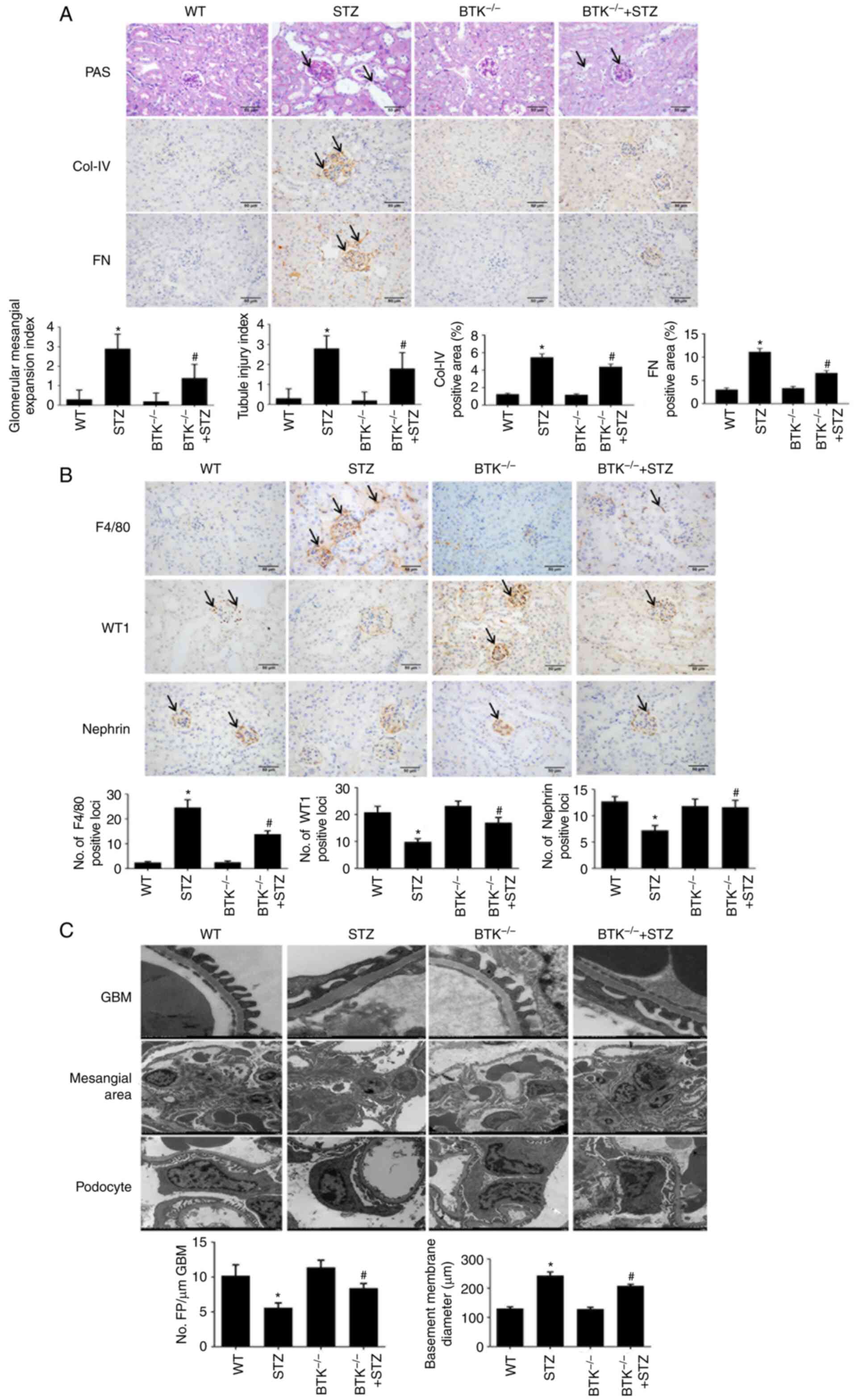 | Figure 3BTK knockout alleviates the
pathological changes in mice with STZ-induced diabetes. (A)
Glomerular mesangial expansion and tubular injury were detected
using PAS staining. Immunohistochemistry of ColIV and FN expression
was performed in mouse kidneys (scale bar, 50 µm). (B)
Immunohistochemistry of F4/80, WT1 and FN expression levels in
mouse kidneys (scale bar, 50 µm). (C) The thickness of GBM,
mesangial expansion and podocyte injury were detected via
transmission electron microscopy (*P<0.05 vs. WT
group; #P<0.05 vs. STZ group). WT, wild-type; BTK,
Bruton's tyrosine kinase; FN, fibronectin; STZ, Streptozotocin;
WT1, WT1 transcription factor; ColIV, collagen IV; PAS, periodic
acid-Schiff; GBM, glomerular basement membrane. |
Finally, TEM was used to observe the ultrastructural
changes in the mouse kidneys. It was identified that, when compared
with the WT group, the STZ group exhibited further extracellular
matrix proliferation, thickened basement membrane and severe
podocyte fusion. The number of podocytes was also decreased, and
organelle loss was increased. However, these phenomena were all
alleviated in the BTK−/− + STZ group (Fig. 3C). Thus, these results indicated
that BTK knockout alleviated renal injury in diabetic mice.
BTK knockout alleviates renal
inflammation in diabetic mice
The change in the inflammatory response was also
evaluated in the mice. The results of ELISA demonstrated that the
TNF-α, IL-1β and MCP-1 levels were increased in serum from the STZ
group. However, this change was significantly reduced in the
BTK−/− + STZ group (Fig.
4A). Subsequently, changes in inflammatory mediators were
detected in the mouse kidneys. The data indicated that the mRNA and
protein expression levels of TNF-α, IL-1β and MCP-1 were increased
in the STZ group (Fig. 4B-D).
Moreover, these changes were significantly reduced when BTK was
knocked out. The immunohistochemistry results also revealed that
the expression levels of TNF-α, IL-1β and MCP-1 were significantly
decreased in the BTK−/− + STZ group compared with the
STZ group (Fig. 4C). On the
whole, the results indicated that diabetic mice exhibited
additional severe inflammation and macrophage infiltration in serum
and kidney, while BTK knockout alleviated this phenomenon.
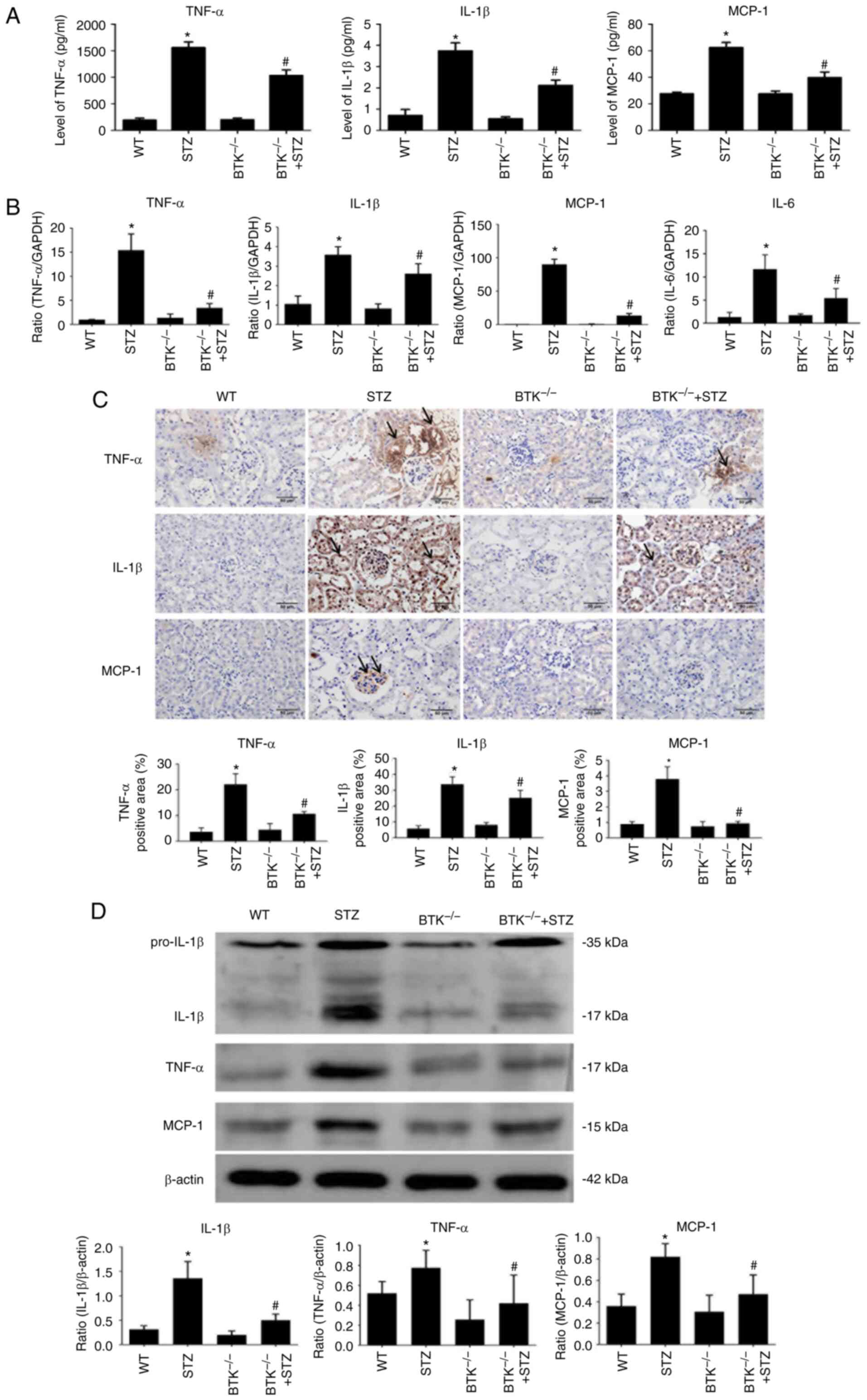 | Figure 4BTK knockout alleviates kidney
inflammation in diabetic mice. (A) TNF-α, IL-1β and MCP-1 levels of
in mouse serum were detected using ELISA kits. (B) TNF-α, IL-1β,
MCP-1 and IL-6 mRNA expression levels in mouse kidneys were
detected by RT-qPCR. (C) TNF-α, IL-1β and MCP-1
immunohistochemistry analysis of protein expression was conducted
in mouse kidneys (scale bar, 50 µm). (D) Protein expression
levels of TNF-α, IL-1β and MCP-1 were detected by western blot
analysis (*P<0.05 vs. WT group; #P<0.05
vs. STZ group). WT, wild-type; BTK, Bruton's tyrosine kinase; STZ,
Streptozotocin; MCP-1, monocyte chemoattractant protein-1; RT-qPCR,
reverse transcription-quantitative PCR. |
BTK knockout suppresses the activation of
the NLRP3 inflammasome in the kidneys of diabetic mice
To further observe the mechanisms of BTK in renal
inflammation, WB analysis was conducted to observe the activation
of the NLRP3 inflammasome in the mouse kidneys. The WB analysis
data demonstrated that the expression levels of p-BTK/BTK, iNOS and
p-p65 were all increased in the STZ group. However, when BTK was
knocked out, the expression levels of iNOS and p-p65 were
significantly decreased (Fig. 5A and
B). This suggested that BTK knockout reduced macrophage
activation in the kidneys of diabetic mice. The change in oxidative
stress in the mouse kidneys was also evaluated. According to these
results, NOX1 and NOX4 expression levels were increased in the STZ
group compared with the WT group. However, BTK deletion
significantly alleviated this effect (Fig. 5C). Moreover, it was found that
the expression levels of NLRP3, caspase-1 and ASC were
significantly decreased in the BTK−/− + STZ group
compared with the STZ group (Fig.
5D). These results thus suggested that BTK knockout decreased
oxidative stress and the activation of the NLRP3 inflammasome in
the kidneys of diabetic mice.
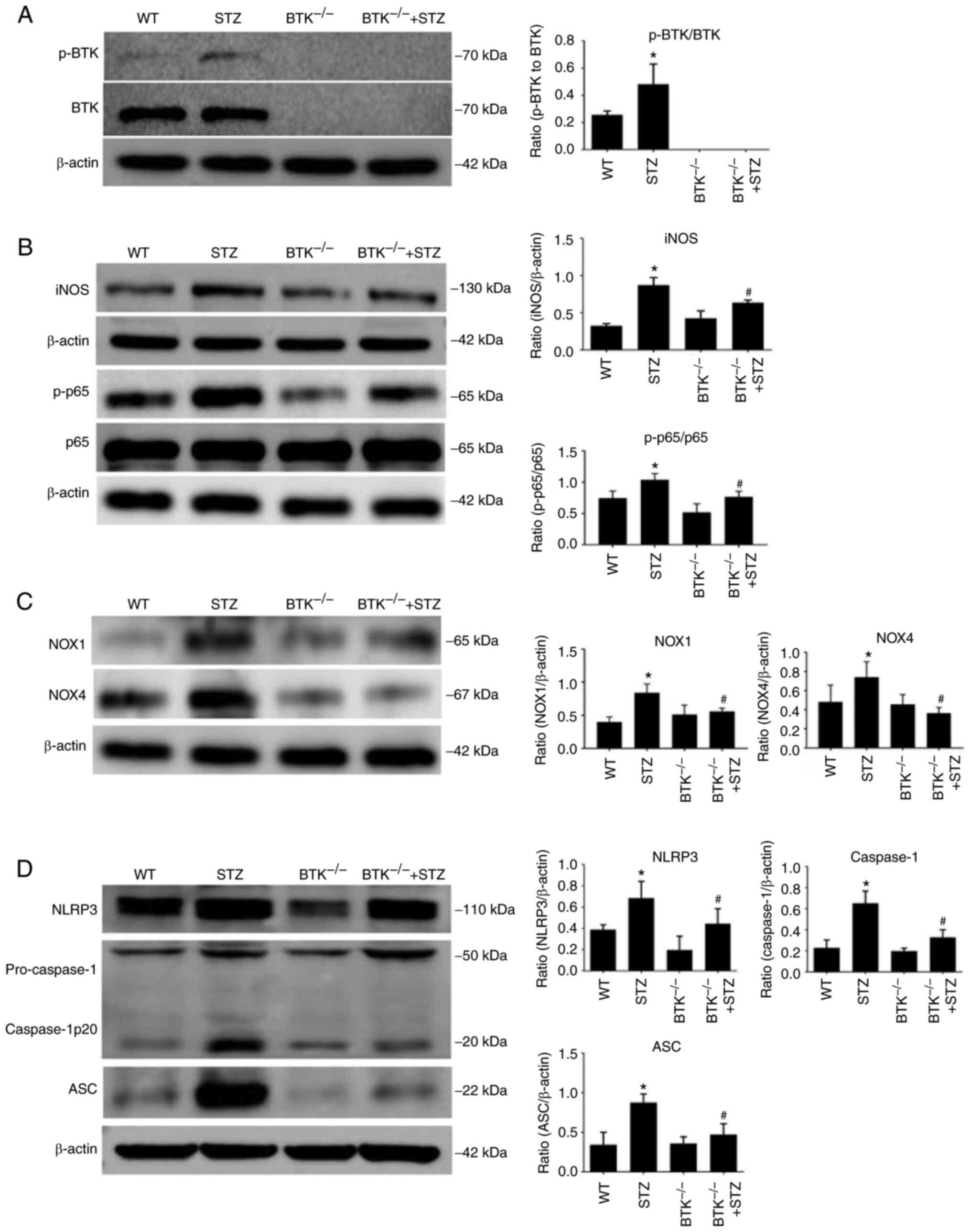 | Figure 5BTK knockout suppresses the oxidative
stress and the activation of the NLRP3 inflammasome in the kidneys
of diabetic mice. (A) p-BTK/BTK expression levels in mouse kidneys
were detected via WB analysis. (B) iNOS, p-p65 and p65 expression
levels in mouse kidneys were detected via WB analysis. (C) NOX1 and
NOX4 expression levels in mouse kidneys were detected via WB
analysis. (D) NLRP3, caspase-1 and ASC expression levels in mouse
kidneys were detected via WB analysis (*P<0.05 vs. WT
group; #P<0.05 vs. STZ group). WT, wild-type; BTK,
Bruton's tyrosine kinase; STZ, Streptozotocin; p-, phosphorylated;
NLRP3, NLR family pyrin domain containing 3; iNOS, inducible nitric
oxide synthase; ASC, apoptosis-associated speck-like protein
containing a caspase recruitment domain; WB, western blot; p-p65,
phosphorylated-NF-κB p65; NOX1, nicotinamide adenine dinucleotide
phosphate oxidase 1; NOX4, nicotinamide adenine dinucleotide
phosphate oxidase 4. |
Discussion
DN is a severe complication of diabetes mellitus,
and its treatment remains challenging (1). Previous studies have reported that
the development of DN is closely associated with inflammation, and
that macrophages are the main immune cell type involved in this
process (5,6). In the early stages of DN, patients
present an increase in serum inflammatory mediator levels.
Subsequently, macrophages can be recruited into the kidneys by
these mediators, such as C-C motif chemokine ligand 2 and MCP-1
(34,35). This process was verified in the
present study and culminated in an increased macrophage
infiltration in kidneys. The results of the present demonstrated
that either in the serum of patients with DN or in the serum of
diabetic mice, inflammatory mediator expression levels were
significantly increased. Moreover, macrophage infiltration and
activation were increased in the kidneys of patients with DN and
diabetic mice. It was also identified that the levels of
inflammatory cytokines released by macrophages in kidneys were
increased. These findings indicated that macrophages can be
recruited into the kidneys and activated by hyperglycemia, leading
to the aggravation of inflammation.
BTK is an important factor in innate immunity and
has been closely associated with inflammation (8,11). It has been previously revealed
that high glucose levels may promote the phosphorylation of BTK,
leading to the release of inflammatory mediators via the NF-κB
signaling pathway (15). It was
also demonstrated in the present study that BTK was activated in
renal macrophages in patients with DN. Moreover, BTK was associated
with clinical indicators and pathological changes, which indicated
that BTK played a crucial role in the disease progression of DN. In
in vivo experiments, it was identified that BTK knockout
reduced UAER, the accumulation of extracellular matrix and
macrophage infiltration in diabetic mice. The fibrotic lesions were
also observed by Masson's staining, although no significant changes
were observed in any of the groups analyzed. This may be attributed
to an early diabetic pathology change in mouse kidneys observed at
12 weeks of feeding, which manifested through mesangial matrix
proliferation and tubulointerstitial damage. Podocytes are
important intrinsic cells in the kidney and are known as the most
vulnerable cells in the kidney (36). Podocyte loss and fusion of foot
process can occur in early stage DN, and it is also the main cause
of proteinuria (37,38). Therefore, it was considered that
podocyte injury could be a favorable choice to evaluate early renal
injury, thus WT1 and Nephrin were used the present study as
podocyte markers (39). The
results of the present study demonstrated that BTK knockout
protected the diabetic mice from podocyte injury, further confirmed
through TEM. It was also noted that the thickened basement
membrane, proliferated extracellular matrix and podocyte injury
were alleviated in BTK knockout diabetic mice. Collectively, these
results suggested that BTK knockout alleviated kidney injury in
diabetic mice.
The NLRP3 inflammasome is a multiprotein complex
that regulates innate immune responses to infection and cell stress
(18). In recent studies, it was
reported that the activation of NLRP3 may be associated with
disease progression in patients with DKD (21,22), Currently, there are no existing
clinically approved therapies that target the NLRP3 inflammasome
directly (24). BTK has been
proven to serve as a critical regulator of NLRP3 inflammasome
activation (25). It has been
observed that the regulatory effects of BTK inhibition on the NLRP3
inflammasome vary in different mouse models. BTK deficiency has
been shown to promote NLRP3 inflammasome activation and induce
IL-1β-mediated colitis, but it also alleviates other diseases, such
as ischemic brain injury, diet-induced metabolic inflammation and
polymicrobial sepsis (26-28). On this basis, the association
between BTK and the NLRP3 inflammasome in DN was examined in the
present study. It was revealed that no significant side-effects
were noted in BTK knockout mice. Moreover, it was noted that BTK
knockout markedly reduced the NLRP3, caspase-1 and ASC expression
levels in the kidneys of diabetic mice. Those results suggested
that the protective effect of BTK knockout was mainly attributed to
the reduced activation of the NLRP3 inflammasome.
In conclusion, the present study demonstrated that
there was a notable association between BTK and the clinical
indicators and pathological changes of patients with DN. Moreover,
BTK knockout reduced inflammation in diabetic mice, and this
protection was a result of the suppression of NLRP3 inflammasome
activation. However, the lack of analysis of IL-6 protein and NF-κB
mRNA expression also present a limitation of the present study. To
date, to the best of our knowledge, there is no effective treatment
available for DN. BTK inhibitor had been widely used in clinical
practice. The present study demonstrated that BTK deletion
effectively reduced the activation of the NLRP3 inflammasome; thus,
this may prove to be a potential method for the treatment of DN.
The findings presented herein may have important clinical
significance; BTK inhibitor may be an effective therapeutic
strategy against DN. The authors aim to perform further studies in
the future in order to provide further suggestions which may aid in
the development of novel treatment strategies for DN.
Supplementary Data
Availability of data and materials
All data generated or analyzed during this study are
included in this published article.
Authors' contributions
JZ and YGW conceived and designed the study. JC and
YYL collected the data and performed the animal experiments. JZ and
JC analyzed the data and wrote the manuscript. YGW and LLX
conducted the animal experiments and reviewed the manuscript. JZ,
LLX and YGW confirmed the authenticity of all the raw data. All
authors have read and approved the final manuscript.
Ethics approval and consent to
participate
All patients and healthy individuals had signed
informed consent forms and all experiments were approved by the
Ethics Committee of Anhui Medical University (approval number:
5101309). The animal experiments were performed in accordance with
guidelines, 'Principles of Laboratory Animal Care and Use in
Research' (Ministry of Health, Beijing, China). The experiments
were approved by the Anhui Medical University Ethics Committee
(approval number: 2020064).
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
Acknowledgements
The authors would like to thank the Research and
Experimental Center of Anhui Medical University, for providing
experimental technical support.
References
|
1
|
Cole JB and Florez JC: Genetics of
diabetes mellitus and diabetes complications. Nat Rev Nephrol.
16:377–390. 2020. View Article : Google Scholar
|
|
2
|
Tervaert TW, Mooyaart AL, Amann K, Cohen
AH, Cook HT, Drachenberg CB, Ferrario F, Fogo AB, Haas M, de Heer
E, et al: Pathologic classification of diabetic nephropathy. J Am
Soc Nephrol. 21:556–563. 2010. View Article : Google Scholar
|
|
3
|
Liang G, Song L, Chen Z, Qian Y, Xie J,
Zhao L, Lin Q, Zhu G, Tan Y, Li X, et al: Fibroblast growth factor
1 ameliorates diabetic nephropathy by an anti-inflammatory
mechanism. Kidney Int. 93:95–109. 2018. View Article : Google Scholar
|
|
4
|
Guilliams M, Thierry GR, Bonnardel J and
Bajenoff M: Establishment and maintenance of the macrophage niche.
Immunity. 52:434–451. 2020. View Article : Google Scholar
|
|
5
|
Klessens CQF, Zandbergen M, Wolterbeek R,
Bruijn JA, Rabelink TJ, Bajema IM and IJpelaar DHT: Macrophages in
diabetic nephropathy in patients with type 2 diabetes. Nephrol Dial
Transplant. 32:1322–1329. 2017.
|
|
6
|
Fu J, Akat KM, Sun Z, Zhang W, Schlondorff
D, Liu Z, Tuschl T, Lee K and He JC: Single-Cell RNA profiling of
glomerular cells shows dynamic changes in experimental diabetic
kidney disease. J Am Soc Nephrol. 30:533–545. 2019. View Article : Google Scholar
|
|
7
|
Smith CI, Islam TC, Mattsson PT, Mohamed
AJ, Nore BF and Vihinen M: The Tec family of cytoplasmic tyrosine
kinases: Mammalian Btk, Bmx, Itk, Tec, Txk and homologs in other
species. BioEssays. 23:436–446. 2001. View Article : Google Scholar
|
|
8
|
Weber ANR, Bittner Z, Liu X, Dang TM,
Radsak MP and Brunner C: Bruton's Tyrosine Kinase: An emerging key
player in innate immunity. Front Immunol. 8:14542017. View Article : Google Scholar
|
|
9
|
Pal Singh S, Dammeijer F and Hendriks RW:
Role of Bruton's tyrosine kinase in B cells and malignancies. Mol
Cancer. 17:572018. View Article : Google Scholar
|
|
10
|
Haselmayer P, Camps M, Liu-Bujalski L,
Nguyen N, Morandi F, Head J, O'Mahony A, Zimmerli SC, Bruns L,
Bender AT, et al: Efficacy and pharmacodynamic modeling of the BTK
inhibitor evobrutinib in autoimmune disease models. J Immunol.
202:2888–2906. 2019. View Article : Google Scholar
|
|
11
|
Rip J, de Bruijn MJW, Appelman MK, Pal
Singh S, Hendriks RW and Corneth OBJ: Toll-Like receptor signaling
drives BTK-mediated autoimmune disease. Front Immunol. 10:952019.
View Article : Google Scholar
|
|
12
|
Wei J, Wang Y, Qi X and Wu Y: Enhanced
Bruton's tyrosine kinase activity in the kidney of patients with
IgA nephropathy. Int Urol Nephrol. 53:1399–1415. 2021. View Article : Google Scholar
|
|
13
|
Kong W, Deng W, Sun Y, Huang S, Zhang Z,
Shi B, Chen W, Tang X, Yao G, Feng X and Sun L: Increased
expression of Bruton's tyrosine kinase in peripheral blood is
associated with lupus nephritis. Clin Rheumatol. 37:43–49. 2018.
View Article : Google Scholar
|
|
14
|
Jain N, Keating M, Thompson P, Ferrajoli
A, Burger J, Borthakur G, Takahashi K, Estrov Z, Fowler N, Kadia T,
et al: Ibrutinib and venetoclax for first-line treatment of CLL. N
Engl J Med. 380:2095–2103. 2019. View Article : Google Scholar
|
|
15
|
Fan Z, Wang Y, Xu X and Wu Y: Inhibitor of
Bruton's tyrosine kinases, PCI-32765, decreases pro-inflammatory
mediators' production in high glucose-induced macrophages. Int
Immunopharmacol. 58:145–153. 2018. View Article : Google Scholar
|
|
16
|
Roschewski M, Lionakis MS, Sharman JP,
Roswarski J, Goy A, Monticelli MA, Roshon M, Wrzesinski SH, Desai
JV, Zarakas MA, et al: Inhibition of Bruton tyrosine kinase in
patients with severe COVID-19. Sci Immunol. 5:eabd01102020.
View Article : Google Scholar
|
|
17
|
Rathinam VA and Fitzgerald KA:
Inflammasome complexes: Emerging mechanisms and effector functions.
Cell. 165:792–800. 2016. View Article : Google Scholar
|
|
18
|
Wang L and Hauenstein AV: The NLRP3
inflammasome: Mechanism of action, role in disease and therapies.
Mol Aspects Med. 76:1008892020. View Article : Google Scholar
|
|
19
|
Hooftman A, Angiari S, Hester S, Corcoran
SE, Runtsch MC, Ling C, Ruzek MC, Slivka PF, McGettrick AF, Banahan
K, et al: The immunomodulatory metabolite itaconate modifies NLRP3
and inhibits inflammasome activation. Cell Metab. 32:468–478.e7.
2020. View Article : Google Scholar
|
|
20
|
Liu D, Yang P, Gao M, Yu T, Shi Y, Zhang
M, Yao M, Liu Y and Zhang X: NLRP3 activation induced by neutrophil
extracellular traps sustains inflammatory response in the diabetic
wound. Clin Sci (Lond). 133:565–582. 2019. View Article : Google Scholar
|
|
21
|
Han Y, Xu X, Tang C, Gao P, Chen X, Xiong
X, Yang M, Yang S, Zhu X, Yuan S, et al: Reactive oxygen species
promote tubular injury in diabetic nephropathy: The role of the
mitochondrial ros-txnip-nlrp3 biological axis. Redox Biol.
16:32–46. 2018. View Article : Google Scholar
|
|
22
|
Mulay SR: Multifactorial functions of the
inflammasome component NLRP3 in pathogenesis of chronic kidney
diseases. Kidney Int. 96:58–66. 2019. View Article : Google Scholar
|
|
23
|
Tang SCW and Yiu WH: Innate immunity in
diabetic kidney disease. Nat Rev Nephrol. 16:206–222. 2020.
View Article : Google Scholar
|
|
24
|
Ram C, Jha AK, Ghosh A, Gairola S, Syed
AM, Murty US, Naidu VGM and Sahu BD: Targeting NLRP3 inflammasome
as a promising approach for treatment of diabetic nephropathy:
Preclinical evidences with therapeutic approaches. Eur J Pharmacol.
885:1735032020. View Article : Google Scholar
|
|
25
|
Weber ANR: Targeting the NLRP3
inflammasome via BTK. Front Cell Dev Biol. 9:6304792021. View Article : Google Scholar
|
|
26
|
Ito M, Shichita T, Okada M, Komine R,
Noguchi Y, Yoshimura A and Morita R: Bruton's tyrosine kinase is
essential for NLRP3 inflammasome activation and contributes to
ischaemic brain injury. Nat Commun. 6:73602015. View Article : Google Scholar
|
|
27
|
Purvis GSD, Collino M, Aranda-Tavio H,
Chiazza F, O'Riordan CE, Zeboudj L, Mohammad S, Collotta D, Verta
R, Guisot NE, et al: Inhibition of Bruton's TK regulates macrophage
NF-κB and NLRP3 inflammasome activation in metabolic inflammation.
Br J Pharmacol. 177:4416–4432. 2020.
|
|
28
|
O'Riordan CE, Purvis GSD, Collotta D,
Krieg N, Wissuwa B, Sheikh MH, Ferreira Alves G, Mohammad S,
Callender LA, Coldewey SM, et al: X-Linked immunodeficient mice
with no functional bruton's tyrosine kinase are protected from
sepsis-induced multiple organ failure. Front Immunol.
11:5817582020. View Article : Google Scholar
|
|
29
|
Li XQ, Chang DY, Chen M and Zhao MH:
Deficiency of C3a receptor attenuates the development of diabetic
nephropathy. BMJ Open Diabetes Res Care. 7:e0008172019. View Article : Google Scholar
|
|
30
|
Liu J, Lee GY, Biggers JD, Toth TL and
Toner M: Low cryoprotectant concentration rapid vitrification of
mouse oocytes and embryos. Cryobiology. 98:233–238. 2021.
View Article : Google Scholar
|
|
31
|
Weinerman R, Ord T, Bartolomei MS,
Coutifaris C and Mainigi M: The superovulated environment,
independent of embryo vitrification, results in low birthweight in
a mouse model. Biol Reprod. 97:133–142. 2017. View Article : Google Scholar
|
|
32
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar
|
|
33
|
Birnbaum Y, Bajaj M, Yang HC and Ye Y:
Combined SGLT2 and DPP4 Inhibition Reduces the Activation of the
Nlrp3/ASC inflammasome and attenuates the development of diabetic
nephropathy in mice with type 2 diabetes. Cardiovasc Drugs Ther.
32:135–145. 2018. View Article : Google Scholar
|
|
34
|
Moreno JA, Gomez-Guerrero C, Mas S, Sanz
AB, Lorenzo O, Ruiz-Ortega M, Opazo L, Mezzano S and Egido J:
Targeting inflammation in diabetic nephropathy: A tale of hope.
Expert Opin Investig Drugs. 27:917–930. 2018. View Article : Google Scholar
|
|
35
|
Wen Y and Crowley SD: The varying roles of
macrophages in kidney injury and repair. Curr Opin Nephrol
Hypertens. 29:286–292. 2020. View Article : Google Scholar
|
|
36
|
Nagata M: Podocyte injury and its
consequences. Kidney Int. 89:1221–1230. 2016. View Article : Google Scholar
|
|
37
|
Agrawal S, He JC and Tharaux PL: Nuclear
receptors in podocyte biology and glomerular disease. Nat Rev
Nephrol. 17:185–204. 2021. View Article : Google Scholar
|
|
38
|
Zhou L, Chen X, Lu M, Wu Q, Yuan Q, Hu C,
Miao J, Zhang Y, Li H, Hou FF, et al: Wnt/β-catenin links oxidative
stress to podocyte injury and proteinuria. Kidney Int. 95:830–845.
2019. View Article : Google Scholar
|
|
39
|
Puelles VG, Bertram JF and Moeller MJ:
Quantifying podocyte depletion: Theoretical and practical
considerations. Cell Tissue Res. 369:229–236. 2017. View Article : Google Scholar
|