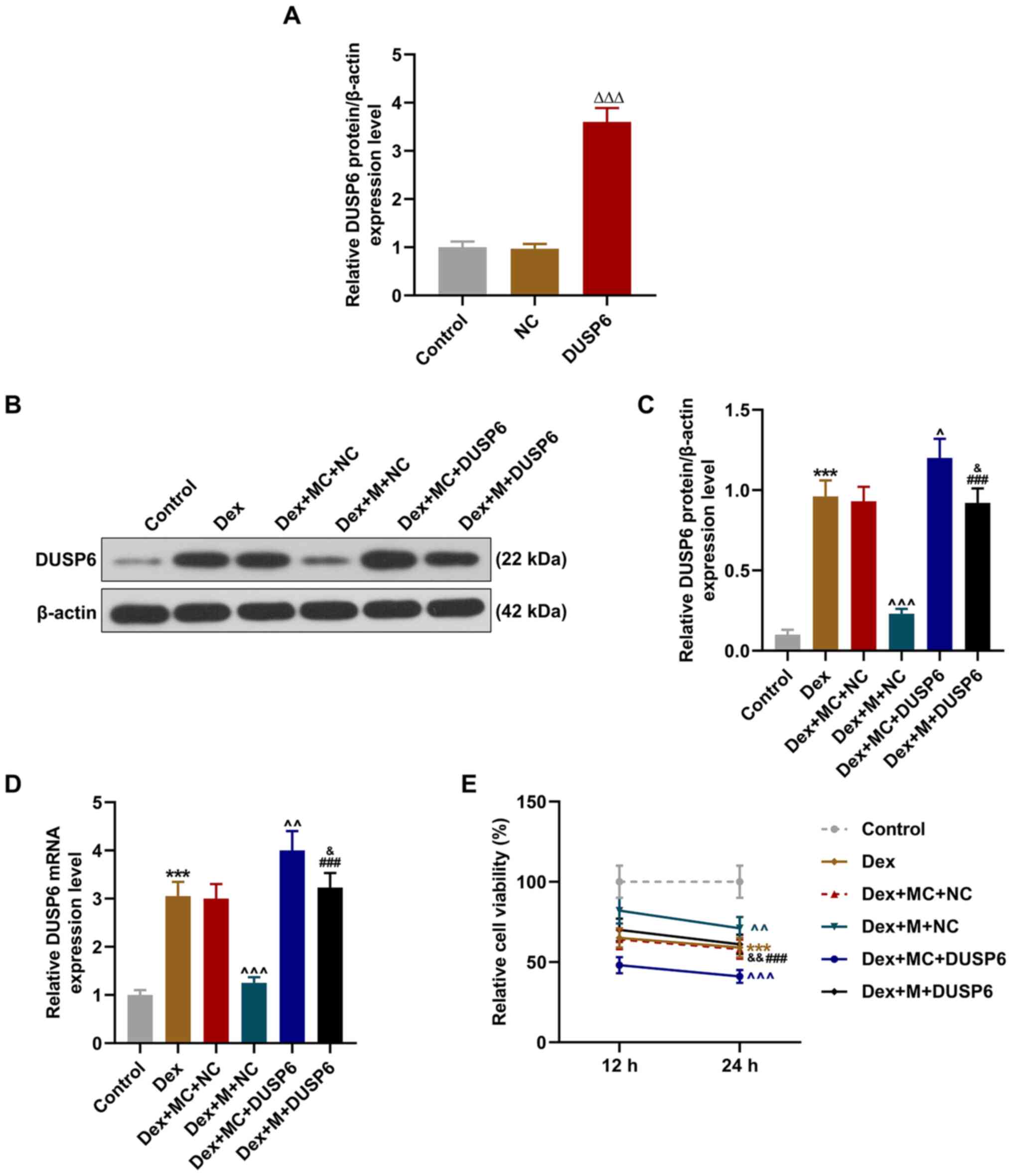
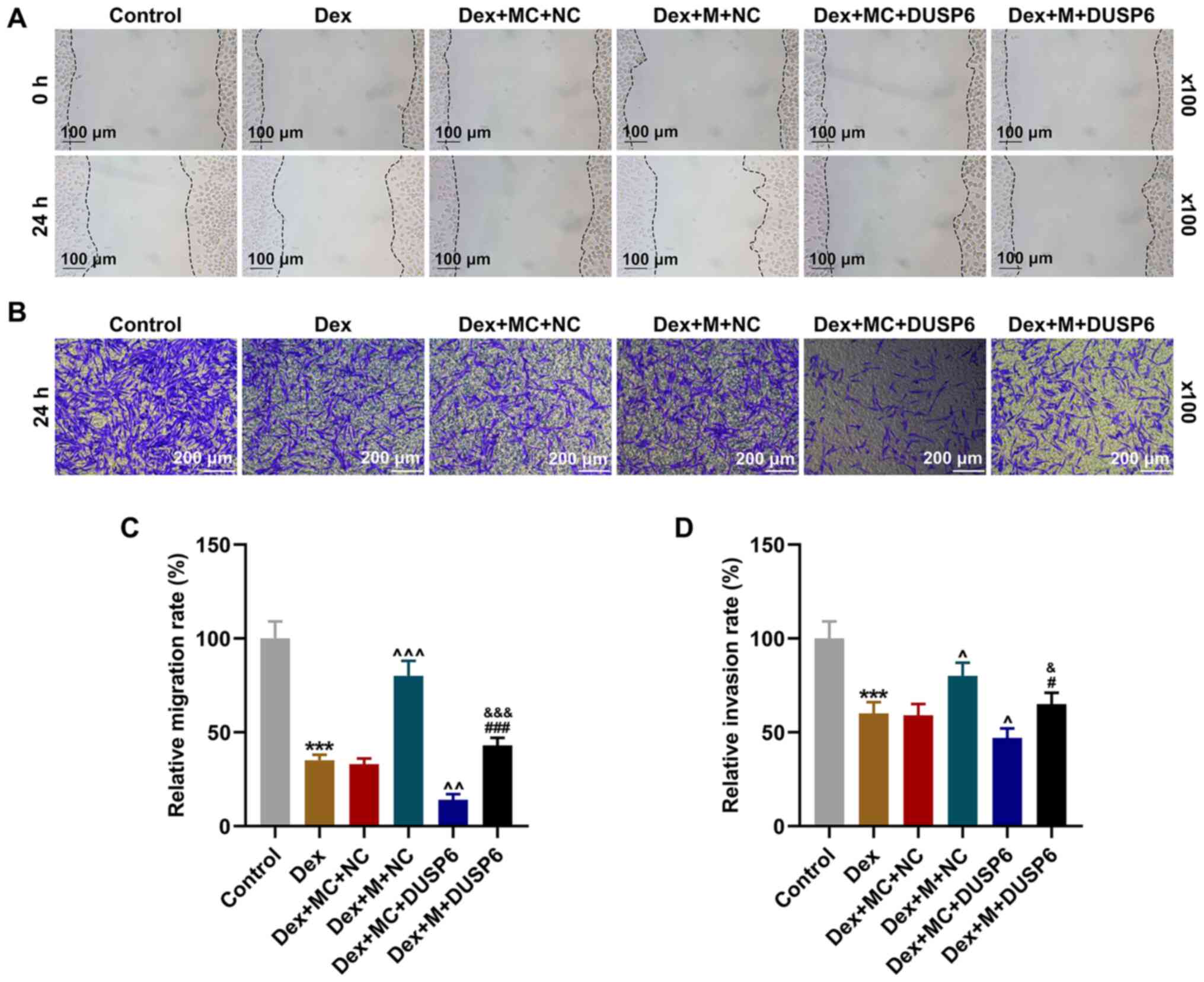
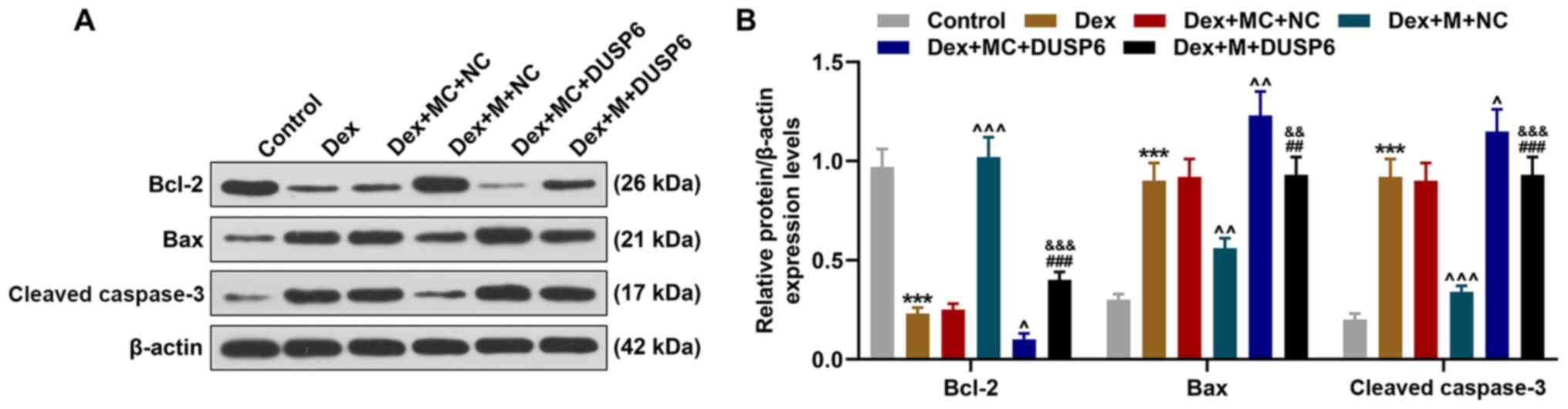
|
1
|
Shine S, Muhamud S and Demelash A:
Prevalence and associated factors of bronchial asthma among adult
patients in Debre Berhan Referral Hospital, Ethiopia 2018: A
cross-sectional study. BMC Res Notes. 12:6082019. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Ye C, Huang C, Zou M, Hu Y, Luo L, Wei Y,
Wan X, Zhao H, Li W, Cai S, et al: The role of secreted Hsp90α in
HDM-induced asthmatic airway epithelial barrier dysfunction. BMC
Pulm Med. 19:2182019. View Article : Google Scholar
|
|
3
|
Liu J, Zhang M, Niu C, Luo Z, Dai J, Wang
L, Liu E and Fu Z: Dexamethasone inhibits repair of human airway
epithelial cells mediated by glucocorticoid-induced leucine zipper
(GILZ). PLoS One. 8:e607052013. View Article : Google Scholar :
|
|
4
|
Xie B, Laxman B, Hashemifar S, Stern R,
Gilliam TC, Maltsev N and White SR: Chemokine expression in the
early response to injury in human airway epithelial cells. PLoS
One. 13:e01933342018. View Article : Google Scholar :
|
|
5
|
Vareille M, Kieninger E, Edwards MR and
Regamey N: The airway epithelium: Soldier in the fight against
respiratory viruses. Clin Microbiol Rev. 24:210–229. 2011.
View Article : Google Scholar :
|
|
6
|
Gon Y and Hashimoto S: Role of airway
epithelial barrier dysfunction in pathogenesis of asthma. Allergol
Int. 67:12–17. 2018. View Article : Google Scholar
|
|
7
|
He Y, Shi J, Nguyen QT, You E, Liu H, Ren
X, Wu Z, Li J, Qiu W, Khoo SK, et al: Development of highly potent
glucocorticoids for steroid-resistant severe asthma. Proc Natl Acad
Sci USA. 116:6932–6937. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Adcock IM and Mumby S: Glucocorticoids.
Handb Exp Pharmacol. 237:171–196. 2017. View Article : Google Scholar
|
|
9
|
Jia S, Guo P, Ge X, Wu H, Lu J and Fan X:
Overexpression of indoleamine 2, 3-dioxygenase contributes to the
repair of human airway epithelial cells inhibited by dexamethasone
via affecting the MAPK/ERK signaling pathway. Exp Ther Med.
16:282–290. 2018.PubMed/NCBI
|
|
10
|
Svitich OA, Sobolev VV, Gankovskaya LV,
Zhigalkina PV and Zverev VV: The role of regulatory RNAs (miRNAs)
in asthma. Allergol Immunopathol (Madr). 46:201–205. 2018.
View Article : Google Scholar
|
|
11
|
Zhang H, Sun Y, Rong W, Fan L, Cai Y, Qu
Q, Gao Y and Zhao H: miR-221 participates in the airway epithelial
cells injury in asthma via targeting SIRT1. Exp Lung Res.
44:272–279. 2018. View Article : Google Scholar
|
|
12
|
Zhou H, Li J, Gao P, Wang Q and Zhang J:
miR-155: A Novel Target in Allergic Asthma. Int J Mol Sci.
17:17732016. View Article : Google Scholar :
|
|
13
|
Zhang X, Zhao X, Sun H, Yan Y, Huang L, Gu
W, Jiang W, Wang Y, Zhu C, Ji W, et al: The role of miR-29c/B7-H3
axis in children with allergic asthma. J Transl Med. 16:2182018.
View Article : Google Scholar :
|
|
14
|
Lu TX, Lim EJ, Wen T, Plassard AJ, Hogan
SP, Martin LJ, Aronow BJ and Rothenberg ME: MiR-375 is
downregulated in epithelial cells after IL-13 stimulation and
regulates an IL-13-induced epithelial transcriptome. Mucosal
Immunol. 5:388–396. 2012. View Article : Google Scholar
|
|
15
|
Ma R, Ma L, Weng W, Wang Y, Liu H, Guo R,
Gao Y, Tu J, Xu TL, Cheng J, et al: DUSP6 SUMOylation protects
cells from oxidative damage via direct regulation of Drp1
dephosphorylation. Sci Adv. 6:eaaz03612020. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Cheng Y, Zhu Y, Xu J, Yang M, Chen P, Xu
W, Zhao J, Geng L and Gong S: PKN2 in colon cancer cells inhibits
M2 phenotype polarization of tumor-associated macrophages via
regulating DUSP6-Erk1/2 pathway. Mol Cancer. 17:132018. View Article : Google Scholar :
|
|
17
|
Chen L, Wang Y, Luan H, Ma G, Zhang H and
Chen G: DUSP6 protects murine podocytes from high glucose-induced
inflammation and apoptosis. Mol Med Rep. 22:2273–2282. 2020.
View Article : Google Scholar
|
|
18
|
Gu W, Yuan Y, Wang L, Yang H, Li S, Tang Z
and Li Q: Long non-coding RNA TUG1 promotes airway remodelling by
suppressing the miR-145-5p/DUSP6 axis in cigarette smoke-induced
COPD. J Cell Mol Med. 23:7200–7209. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Lu M, Xu X, Xi B, Dai Q, Li C, Su L, Zhou
X, Tang M, Yao Y and Yang J: Molecular network-based identification
of competing endogenous RNAs in thyroid carcinoma. Genes (Basel).
9:442018. View Article : Google Scholar
|
|
20
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar
|
|
21
|
van den Berge M and Tasena H: Role of
microRNAs and exosomes in asthma. Curr Opin Pulm Med. 25:87–93.
2019. View Article : Google Scholar
|
|
22
|
Zhao L, Shi X, Wang N, Liu C and Wang J:
YAP1, targeted by miR-375, enhanced the pro-angiogenesis of airway
smooth muscle cells in asthma via STAT3 activation. Cell Cycle.
19:1275–1284. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Dolka I, Król M and Sapierzyński R:
Evaluation of apoptosis-associated protein (Bcl-2, Bax, cleaved
caspase-3 and p53) expression in canine mammary tumors: An
immunohistochemical and prognostic study. Res Vet Sci. 105:124–133.
2016. View Article : Google Scholar
|
|
24
|
Clifford RL, Patel J, MacIsaac JL, McEwen
LM, Johnson SR, Shaw D, Knox AJ, Hackett TL and Kobor MS: Airway
epithelial cell isolation techniques affect DNA methylation
profiles with consequences for analysis of asthma related
perturbations to DNA methylation. Sci Rep. 9:144092019. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Grainge C, Dennison P, Lau L, Davies D and
Howarth P: Asthmatic and normal respiratory epithelial cells
respond differently to mechanical apical stress. Am J Respir Crit
Care Med. 190:477–480. 2014. View Article : Google Scholar :
|
|
26
|
Wang L, Liu XH, Chen H, Chen ZY, Weng XD,
Qiu T and Liu L: Picroside II protects rat kidney against
ischemia/reperfusion-induced oxidative stress and inflammation by
the TLR4/NF-κB pathway. Exp Ther Med. 9:1253–1258. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Bordag N, Klie S, Jürchott K, Vierheller
J, Schiewe H, Albrecht V, Tonn JC, Schwartz C, Schichor C and
Selbig J: Glucocorticoid (dexamethasone)-induced metabolome changes
in healthy males suggest prediction of response and side effects.
Sci Rep. 5:159542015. View Article : Google Scholar :
|
|
28
|
Giles AJ, Hutchinson MND, Sonnemann HM,
Jung J, Fecci PE, Ratnam NM, Zhang W, Song H, Bailey R, Davis D, et
al: Dexamethasone-induced immunosuppression: Mechanisms and
implications for immunotherapy. J Immunother Cancer. 6:512018.
View Article : Google Scholar :
|
|
29
|
Bertoli S, Picard M, Bérard E, Griessinger
E, Larrue C, Mouchel PL, Vergez F, Tavitian S, Yon E, Ruiz J, et
al: Dexamethasone in hyperleukocytic acute myeloid leukemia.
Haematologica. 103:988–998. 2018. View Article : Google Scholar :
|
|
30
|
Bonfiglio V, Reibaldi M, Fallico M, Russo
A, Pizzo A, Fichera S, Rapisarda C, Macchi I, Avitabile T and Longo
A: Widening use of dexamethasone implant for the treatment of
macular edema. Drug Des Devel Ther. 11:2359–2372. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Pohlmann D, Vom Brocke GA, Winterhalter S,
Steurer T, Thees S and Pleyer U: Dexamethasone inserts in
noninfectious uveitis: a single-center experience. Ophthalmology.
125:1088–1099. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Piastra M, Pizza A, Gaddi S, Luca E,
Genovese O, Picconi E, De Luca D and Conti G: Dexmedetomidine is
effective and safe during NIV in infants and young children with
acute respiratory failure. BMC Pediatr. 18:2822018. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Abaya R, Jones L and Zorc JJ:
Dexamethasone compared to prednisone for the treatment of children
with acute asthma exacerbations. Pediatr Emerg Care. 34:53–58.
2018. View Article : Google Scholar
|
|
34
|
Ji Y, Yang X and Su H: Overexpression of
microRNA-375 impedes platelet-derived growth factor-induced
proliferation and migration of human fetal airway smooth muscle
cells by targeting Janus kinase 2. Biomed Pharmacother. 98:69–75.
2018. View Article : Google Scholar
|
|
35
|
Ling H, Fabbri M and Calin GA: MicroRNAs
and other non-coding RNAs as targets for anticancer drug
development. Nat Rev Drug Discov. 12:847–865. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Wu CP, Bi YJ, Liu DM and Wang LY:
Hsa-miR-375 promotes the progression of inflammatory bowel disease
by upregulating TLR4. Eur Rev Med Pharmacol Sci. 23:7543–7549.
2019.
|
|
37
|
Wang T, Chen D, Wang P, Xu Z and Li Y:
miR-375 prevents nasal mucosa cells from apoptosis and ameliorates
allergic rhinitis via inhibiting JAK2/STAT3 pathway. Biomed
Pharmacother. 103:621–627. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Jin Y, Liu Y, Zhang J, Huang W, Jiang H,
Hou Y, Xu C, Zhai C, Gao X, Wang S, et al: The expression of
miR-375 is associated with carcinogenesis in three subtypes of lung
cancer. PLoS One. 10:e01441872015. View Article : Google Scholar :
|
|
39
|
Novaleski CK, Carter BD, Sivasankar MP,
Ridner SH, Dietrich MS and Rousseau B: Apoptosis and vocal fold
disease: clinically relevant implications of epithelial cell death.
J Speech Lang Hear Res. 60:1264–1272. 2017. View Article : Google Scholar
|
|
40
|
Hassan M, Watari H, AbuAlmaaty A, Ohba Y
and Sakuragi N: Apoptosis and molecular targeting therapy in
cancer. BioMed Res Int. 2014:1508452014. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Xu G, Kuang G, Jiang W, Jiang R and Jiang
D: Polydatin promotes apoptosis through upregulation the ratio of
Bax/Bcl-2 and inhibits proliferation by attenuating the β-catenin
signaling in human osteosarcoma cells. Am J Transl Res. 8:922–931.
2016.
|
|
42
|
Hsu SF, Lee YB, Lee YC, Chung AL, Apaya
MK, Shyur LF, Cheng CF, Ho FM and Meng TC: Dual specificity
phosphatase DUSP6 promotes endothelial inflammation through
inducible expression of ICAM-1. FEBS J. 285:1593–1610. 2018.
View Article : Google Scholar
|
|
43
|
Beaudry K, Langlois MJ, Montagne A, Cagnol
S, Carrier JC and Rivard N: Dual-specificity phosphatase 6 deletion
protects the colonic epithelium against inflammation and promotes
both proliferation and tumorigenesis. J Cell Physiol.
234:6731–6745. 2019. View Article : Google Scholar
|
|
44
|
Carson WF IV, Salter-Green SE, Scola MM,
Joshi A, Gallagher KA and Kunkel SL: Enhancement of macrophage
inflammatory responses by CCL2 is correlated with increased miR-9
expression and downregulation of the ERK1/2 phosphatase Dusp6. Cell
Immunol. 314:63–72. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Zhang Z, Kobayashi S, Borczuk AC, Leidner
RS, Laframboise T, Levine AD and Halmos B: Dual specificity
phosphatase 6 (DUSP6) is an ETS-regulated negative feedback
mediator of oncogenic ERK signaling in lung cancer cells.
Carcinogenesis. 31:577–586. 2010. View Article : Google Scholar :
|