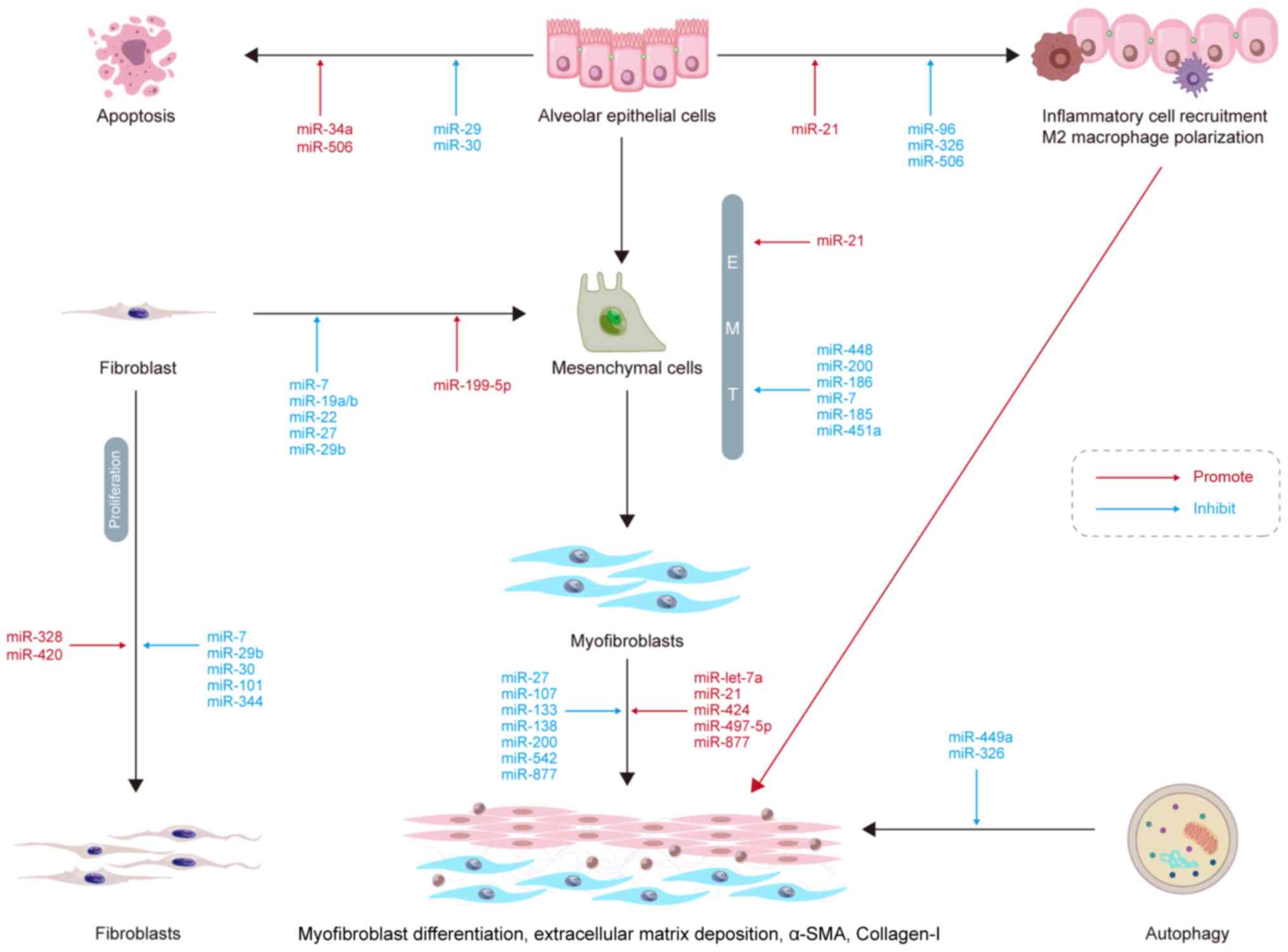
|
1
|
Raghu G, Remy-Jardin M, Myers JL, Richeldi
L, Ryerson CJ, Lederer DJ, Behr J, Cottin V, Danoff SK, Morell F,
et al: Diagnosis of idiopathic pulmonary fibrosis. An official
ATS/ERS/JRS/ALAT clinical practice guideline. Am J Respir Crit Care
Med. 198:e44–e68. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Lederer DJ and Martinez FJ: Idiopathic
pulmonary fibrosis. N Engl J Med. 378:1811–1823. 2018. View Article : Google Scholar
|
|
3
|
Schamberger AC, Schiller HB, Fernandez IE,
Sterclova M, Heinzelmann K, Hennen E, Hatz R, Behr J, Vašáková M,
Mann M, et al: Glutathione peroxidase 3 localizes to the epithelial
lining fluid and the extracellular matrix in interstitial lung
disease. Sci Rep. 6:299522016. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Zhang L, Wang Y, Pandupuspitasari NS, Wu
G, Xiang X, Gong Q, Xiong W, Wang CY, Yang P and Ren B: Endoplasmic
reticulum stress, a new wrestler, in the pathogenesis of idiopathic
pulmonary fibrosis. Am J Transl Res. 9:722–735. 2017.
|
|
5
|
Kirby T: Living with idiopathic pulmonary
fibrosis. Lancet Respir Med. 9:136–138. 2021. View Article : Google Scholar
|
|
6
|
Shenderov K, Collins SL, Powell JD and
Horton MR: Immune dysregulation as a driver of idiopathic pulmonary
fibrosis. J Clin Invest. 131:e1432262021. View Article : Google Scholar :
|
|
7
|
Guenther A, Krauss E, Tello S, Wagner J,
Paul B, Kuhn S, Maurer O, Heinemann S, Costabel U, Barbero MAN, et
al: The European IPF registry (eurIPFreg): Baseline characteristics
and survival of patients with idiopathic pulmonary fibrosis. Respir
Res. 19:1412018. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Gao J, Kalafatis D, Carlson L, Pesonen
IHA, Li CX, Wheelock Å, Magnusson JM and Sköld CM: Baseline
characteristics and survival of patients of idiopathic pulmonary
fibrosis: A longitudinal analysis of the Swedish IPF Registry.
Respir Res. 22:402021. View Article : Google Scholar :
|
|
9
|
Maher TM, Costabel U, Glassberg MK, Kondoh
Y, Ogura T, Scholand MB, Kardatzke D, Howard M, Olsson J, Neighbors
M, et al: Phase 2 trial to assess lebrikizumab in patients with
idiopathic pulmonary fibrosis. Eur Respir J. 57:19024422021.
View Article : Google Scholar :
|
|
10
|
Di Martino E, Provenzani A, Vitulo P and
Polidori P: Systematic review and meta-analysis of pirfenidone,
nintedanib, and pamrevlumab for the treatment of idiopathic
pulmonary fibrosis. Ann Pharmacother. 55:723–731. 2021. View Article : Google Scholar
|
|
11
|
Raghu G, van den Blink B, Hamblin MJ,
Brown AW, Golden JA, Ho LA, Wijsenbeek MS, Vasakova M, Pesci A,
Antin-Ozerkis DE, et al: Effect of recombinant human pentraxin 2 vs
placebo on change in forced vital capacity in patients with
idiopathic pulmonary fibrosis: A randomized clinical trial. JAMA.
319:2299–2307. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Maher TM, van der Aar EM, Van de Steen O,
Allamassey L, Desrivot J, Dupont S, Fagard L, Ford P, Fieuw A and
Wuyts W: Safety, tolerability, pharmacokinetics, and
pharmacodynamics of GLPG1690, a novel autotaxin inhibitor, to treat
idiopathic pulmonary fibrosis (FLORA): A phase 2a randomised
placebo-controlled trial. Lancet Respir Med. 6:627–635. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Abuserewa ST, Duff R and Becker G:
Treatment of idiopathic pulmonary fibrosis. Cureus.
13:e153602021.PubMed/NCBI
|
|
14
|
Brigstock DR: Extracellular vesicles in
organ fibrosis: Mechanisms, therapies, and diagnostics. Cells.
10:15962021. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Yamada M: Extracellular vesicles: Their
emerging roles in the pathogenesis of respiratory diseases. Respir
Investig. 59:302–311. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Anderson HC: Vesicles associated with
calcification in the matrix of epiphyseal cartilage. J Cell Biol.
41:59–72. 1969. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
György B, Szabó TG, Pásztói M, Pál Z,
Misják P, Aradi B, László V, Pállinger E, Pap E, Kittel A, et al:
Membrane vesicles, current state-of-the-art: Emerging role of
extracellular vesicles. Cell Mol Life Sci. 68:2667–2688. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Pan BT, Teng K, Wu C, Adam M and Johnstone
RM: Electron microscopic evidence for externalization of the
transferrin receptor in vesicular form in sheep reticulocytes. J
Cell Biol. 101:942–948. 1985. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Menck K, Sivaloganathan S, Bleckmann A and
Binder C: Microvesicles in cancer: Small size, large potential. Int
J Mol Sci. 21:53732020. View Article : Google Scholar :
|
|
20
|
Pegtel DM and Gould SJ: Exosomes. Annu Rev
Biochem. 88:487–514. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Cocozza F, Grisard E, Martin-Jaular L,
Mathieu M and Théry C: SnapShot: Extracellular vesicles. Cell.
182:262–262.e1. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Zhang H, Freitas D, Kim HS, Fabijanic K,
Li Z, Chen H, Mark MT, Molina H, Martin AB, Bojmar L, et al:
Identification of distinct nanoparticles and subsets of
extracellular vesicles by asymmetric flow field-flow fractionation.
Nat Cell Biol. 20:332–343. 2018. View Article : Google Scholar :
|
|
23
|
Anand S, Samuel M and Mathivanan S:
Exomeres: A new member of extracellular vesicles family. Subcell
Biochem. 97:89–97. 2021. View Article : Google Scholar
|
|
24
|
Kučuk N, Primožič M, Knez Ž and Leitgeb M:
Exosomes engineering and their roles as therapy delivery tools,
therapeutic targets, and biomarkers. Int J Mol Sci. 22:95432021.
View Article : Google Scholar
|
|
25
|
Kodam SP and Ullah M: Diagnostic and
therapeutic potential of extracellular vesicles. Technol Cancer Res
Treat. 20:153303382110412032021. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Shao H, Im H, Castro CM, Breakefield X,
Weissleder R and Lee H: New technologies for analysis of
extracellular vesicles. Chem Rev. 118:1917–1950. 2018. View Article : Google Scholar :
|
|
27
|
Yang D, Zhang W, Zhang H, Zhang F, Chen L,
Ma L, Larcher LM, Chen S, Liu N, Zhao Q, et al: Progress
opportunity, and perspective on exosome isolation-efforts for
efficient exosome-based theranostics. Theranostics. 10:3684–3707.
2020. View Article : Google Scholar
|
|
28
|
Théry C, Zitvogel L and Amigorena S:
Exosomes: Composition, biogenesis and function. Nat Rev Immunol.
2:569–579. 2002. View
Article : Google Scholar : PubMed/NCBI
|
|
29
|
Trajkovic K, Hsu C, Chiantia S, Rajendran
L, Wenzel D, Wieland F, Schwille P, Brügger B and Simons M:
Ceramide triggers budding of exosome vesicles into multivesicular
endosomes. Science. 319:1244–1247. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
van Niel G, Porto-Carreiro I, Simoes S and
Raposo G: Exosomes: A common pathway for a specialized function. J
Biochem. 140:13–21. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Vietri M, Radulovic M and Stenmark H: The
many functions of ESCRTs. Nat Rev Mol Cell Biol. 21:25–42. 2020.
View Article : Google Scholar
|
|
32
|
Ju Y, Bai H, Ren L and Zhang L: The role
of exosome and the ESCRT pathway on enveloped virus infection. Int
J Mol Sci. 22:90602021. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Sun R, Liu Y, Lu M, Ding Q, Wang P, Zhang
H, Tian X, Lu P, Meng D, Sun N, et al: ALIX increases protein
content and protective function of iPSC-derived exosomes. J Mol Med
(Berl). 97:829–844. 2019. View Article : Google Scholar
|
|
34
|
Han Q, Lv L, Wei J, Lei X, Lin H, Li G,
Cao J, Xie J, Yang W, Wu S, et al: Vps4A mediates the localization
and exosome release of β-catenin to inhibit epithelial-mesenchymal
transition in hepatocellular carcinoma. Cancer Lett. 457:47–59.
2019. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Kunadt M, Eckermann K, Stuendl A, Gong J,
Russo B, Strauss K, Rai S, Kügler S, Falomir Lockhart L, Schwalbe
M, et al: Extracellular vesicle sorting of α-Synuclein is regulated
by sumoylation. Acta Neuropathol. 129:695–713. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Chang HM and Yeh ETH: SUMO: From bench to
bedside. Physiol Rev. 100:1599–1619. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
de Gassart A, Géminard C, Février B,
Raposo G and Vidal M: Lipid raft-associated protein sorting in
exosomes. Blood. 102:4336–4344. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Rana S and Zöller M: Exosome target cell
selection and the importance of exosomal tetraspanins: A
hypothesis. Biochem Soc Trans. 39:559–562. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
van Niel G, Charrin S, Simoes S, Romao M,
Rochin L, Saftig P, Marks MS, Rubinstein E and Raposo G: The
tetraspanin CD63 regulates ESCRT-Independent and -Dependent
endosomal sorting during melanogenesis. Dev Cell. 21:708–721. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Chairoungdua A, Smith DL, Pochard P, Hull
M and Caplan MJ: Exosome release of β-catenin: A novel mechanism
that antagonizes Wnt signaling. J Cell Biol. 190:1079–1091. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Doyle LM and Wang MZ: Overview of
extracellular vesicles, their origin, composition, purpose, and
methods for exosome isolation and analysis. Cells. 8:7272019.
View Article : Google Scholar :
|
|
42
|
Dang VD, Jella KK, Ragheb RRT, Denslow ND
and Alli AA: Lipidomic and proteomic analysis. of exosomes from
mouse cortical collecting duct cells. FASEB J. 31:5399–5408. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
43
|
O'Brien K, Breyne K, Ughetto S, Laurent LC
and Breakefield XO: RNA delivery by extracellular vesicles in
mammalian cells and its applications. Nat Rev Mol Cell Biol.
21:585–606. 2020. View Article : Google Scholar
|
|
44
|
Kalluri R and LeBleu VS: The biology,
function, and biomedical applications of exosomes. Science.
367:eaau69772020. View Article : Google Scholar :
|
|
45
|
Saad MH, Badierah R, Redwan EM and
El-Fakharany EM: A comprehensive insight into the role of exosomes
in viral infection: Dual faces bearing different functions.
Pharmaceutics. 13:14052021. View Article : Google Scholar :
|
|
46
|
Gurunathan S, Kang MH, Qasim M, Khan K and
Kim JH: Biogenesis, membrane trafficking, functions, and next
generation nanotherapeutics medicine of extracellular vesicles. Int
J Nanomedicine. 16:3357–3383. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Zhang Q, Higginbotham JN, Jeppesen DK,
Yang YP, Li W, McKinley ET, Graves-Deal R, Ping J, Britain CM,
Dorsett KA, et al: Transfer of functional cargo in exomeres. Cell
Rep. 27:940–954.e6. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Krol J, Loedige I and Filipowicz W: The
widespread regulation of microRNA biogenesis, function and decay.
Nat Rev Genet. 11:597–610. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Mohr AM and Mott JL: Overview of MicroRNA
biology. Semin Liver Dis. 35:3–11. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Valadi H, Ekström K, Bossios A, Sjöstrand
M, Lee JJ and Lötvall JO: Exosome-mediated transfer of mRNAs and
microRNAs is a novel mechanism of genetic exchange between cells.
Nat Cell Biol. 9:654–659. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Shao N, Xue L, Wang R, Luo K, Zhi F and
Lan Q: MiR-454-3p is an exosomal biomarker and functions as a tumor
suppressor in glioma. Mol Cancer Ther. 18:459–469. 2019. View Article : Google Scholar
|
|
52
|
Qiu Y, Li P, Zhang Z and Wu M: Insights
into exosomal Non-coding RNAs sorting mechanism and clinical
application. Front Oncol. 11:6649042021. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Schirle NT, Sheu-Gruttadauria J and MacRae
IJ: Structural basis for microRNA targeting. Science. 346:608–613.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Methods in MicroRNA biogenesis,
identification, function and decay. Methods. 152:1–2. 2019.
View Article : Google Scholar
|
|
55
|
Zhang J, Li S, Li L, Li M, Guo C, Yao J
and Mi S: Exosome and exosomal microRNA: Trafficking, sorting, and
function. Genomics Proteomics Bioinformatics. 13:17–24. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Guduric-Fuchs J, O'Connor A, Camp B,
O'Neill CL, Medina RJ and Simpson DA: Selective extracellular
vesicle-mediated export of an overlapping set of microRNAs from
multiple cell types. BMC Genomics. 13:3572012. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Donzelli J, Proestler E, Riedel A,
Nevermann S, Hertel B, Guenther A, Gattenlöhner S, Savai R, Larsson
K and Saul MJ: Small extracellular vesicle-derived miR-574-5p
regulates PGE2-biosynthesis via TLR7/8 in lung cancer. J Extracell
Vesicles. 10:e121432021. View Article : Google Scholar
|
|
58
|
Melo SA, Sugimoto H, O'Connell JT, Kato N,
Villanueva A, Vidal A, Qiu L, Vitkin E, Perelman LT, Melo CA, et
al: Cancer exosomes perform Cell-Independent MicroRNA biogenesis
and promote tumorigenesis. Cancer Cell. 26:707–721. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Villarroya-Beltri C, Gutiérrez-Vázquez C,
Sánchez-Cabo F, Pérez-Hernández D, Vázquez J, Martin-Cofreces N,
Martinez-Herrera DJ, Pascual-Montano A, Mittelbrunn M and
Sánchez-Madrid F: Sumoylated hnRNPA2B1 controls the sorting of
miRNAs into exosomes through binding to specific motifs. Nat
Commun. 4:29802013. View Article : Google Scholar
|
|
60
|
Santangelo L, Giurato G, Cicchini C,
Montaldo C, Mancone C, Tarallo R, Battistelli C, Alonzi T, Weisz A
and Tripodi M: The RNA-Binding Protein SYNCRIP Is a component of
the hepatocyte exosomal machinery controlling MicroRNA sorting.
Cell Rep. 17:799–808. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Boateng E and Krauss-Etschmann S: miRNAs
in lung development and diseases. Int J Mol Sci. 21:27652020.
View Article : Google Scholar :
|
|
62
|
Bersimbaev R, Aripova A, Bulgakova O,
Kussainova A, Akparova A and Izzotti A: The plasma levels of
hsa-miR-19b-3p hsa-miR-125b-5p and hsamiR-320c in patients with
asthma, COPD and asthma-COPD overlap syndrome (ACOS). MicroRNA.
10:130–138. 2021. View Article : Google Scholar
|
|
63
|
Zeng Q and Zeng J: Inhibition of
miR-494-3p alleviates oxidative stress-induced cell senescence and
inflammation in the primary epithelial cells of COPD patients. Int
Immunopharmacol. 92:1070442021. View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Weidner J, Bartel S, Kılıç A, Zissler UM,
Renz H, Schwarze J, Schmidt-Weber CB, Maes T, Rebane A,
Krauss-Etschmann S and Rådinger M: Spotlight on microRNAs in
allergy and asthma. Allergy. 76:1661–1678. 2021. View Article : Google Scholar
|
|
65
|
Wardzyńska A, Pawełczyk M, Rywaniak J,
Makowska J, Jamroz-Brzeska J and Kowalski ML: Circulating miRNA
expression in asthmatics is age-related and associated with
clinical asthma parameters, respiratory function and systemic
inflammation. Respir Res. 22:1772021. View Article : Google Scholar
|
|
66
|
Zhong S, Golpon H, Zardo P and Borlak J:
MiRNAs in lung cancer. A systematic review identifies predictive
and prognostic miRNA candidates for precision medicine in lung
cancer. Transl Res. 230:164–196. 2021. View Article : Google Scholar
|
|
67
|
Cainap C, Balacescu O, Cainap SS and Pop
LA: Next generation sequencing technology in lung cancer diagnosis.
Biology (Basel). 10:8642021.
|
|
68
|
Yang G, Yang L, Wang W, Wang J, Wang J and
Xu Z: Discovery and validation of extracellular/circulating
microRNAs during idiopathic pulmonary fibrosis disease progression.
Gene. 562:138–144. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Zhang H, Song M, Guo J, Ma J, Qiu M and
Yang Z: The function of non-coding RNAs in idiopathic pulmonary
fibrosis. Open Med (Wars). 16:481–490. 2021. View Article : Google Scholar
|
|
70
|
Wang Q, Xie ZL, Wu Q, Jin ZX, Yang C and
Feng J: Role of various imbalances centered on alveolar epithelial
cell/fibroblast apoptosis imbalance in the pathogenesis of
idiopathic pulmonary fibrosis. Chin Med J (Engl). 134:261–274.
2021. View Article : Google Scholar
|
|
71
|
Mao C, Zhang J, Lin S, Jing L, Xiang J,
Wang M, Wang B, Xu P, Liu W, Song X and Lv C: Mi RNA -30a inhibits
AEC s-II apoptosis by blocking mitochondrial fission dependent on
Drp-1. J Cell Mol Med. 18:2404–2416. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
72
|
Matsushima S and Ishiyama J: MicroRNA-29c
regulates apoptosis sensitivity via modulation of the cell-surface
death receptor, Fas, in lung fibroblasts. Am J Physiol Lung Cell
Mol Physiol. 311:L1050–L1061. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
73
|
Shetty SK, Tiwari N, Marudamuthu AS,
Puthusseri B, Bhandary YP, Fu J, Levin J, Idell S and Shetty S: p53
and miR-34a feedback promotes lung epithelial injury and pulmonary
fibrosis. Am J Pathol. 187:1016–1034. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
74
|
Milanovic M, Fan DNY, Belenki D, Däbritz
JHM, Zhao Z, Yu Y, Dörr JR, Dimitrova L, Lenze D, Monteiro Barbosa
IA, et al: Senescence-associated reprogramming promotes cancer
stemness. Nature. 553:96–100. 2018. View Article : Google Scholar
|
|
75
|
Wolters PJ, Collard HR and Jones KD:
Pathogenesis of idiopathic pulmonary fibrosis. Annu Rev Pathol.
9:157–179. 2014. View Article : Google Scholar :
|
|
76
|
Hussen BM, Shoorei H, Mohaqiq M, Dinger
ME, Hidayat HJ, Taheri M and Ghafouri-Fard S: The impact of
Non-coding RNAs in the epithelial to mesenchymal transition. Front
Mol Biosci. 8:6651992021. View Article : Google Scholar : PubMed/NCBI
|
|
77
|
Sun J, Li Q, Lian X, Zhu Z, Chen X, Pei W,
Li S, Abbas A, Wang Y and Tian L: MicroRNA-29b mediates lung
mesenchymal-epithelial transition and prevents lung fibrosis in the
silicosis model. Mol Ther Nucleic Acids. 14:20–31. 2019. View Article : Google Scholar
|
|
78
|
Qi Y, Zhao A, Yang P, Jin L and Hao C:
MiR-34a-5p Attenuates EMT through targeting SMAD4 in silica-induced
pulmonary fibrosis. J Cell Mol Med. 24:12219–12224. 2020.
View Article : Google Scholar :
|
|
79
|
Jeong MH, Kim HR, Park YJ, Chung KH and
Kim HS: Reprogrammed lung epithelial cells by decrease of miR-451a
in extracellular vesicles contribute to aggravation of pulmonary
fibrosis. Cell Biol Toxicol. Aug 30–2021.Epub ahead of print.
View Article : Google Scholar
|
|
80
|
Liu Y, Nie H, Ding Y, Hou Y, Mao K and Cui
Y: MiRNA, a new treatment strategy for pulmonary fibrosis. Curr
Drug Targets. 22:793–802. 2021. View Article : Google Scholar
|
|
81
|
Rajasekaran S, Rajaguru P and Sudhakar
Gandhi PS: MicroRNAs as potential targets for progressive pulmonary
fibrosis. Front Pharmacol. 6:2542015. View Article : Google Scholar : PubMed/NCBI
|
|
82
|
Wei P, Xie Y, Abel PW, Huang Y, Ma Q, Li
L, Hao J, Wolff DW, Wei T and Tu Y: Transforming growth factor
(TGF)-β1-induced miR-133a inhibits myofibroblast differentiation
and pulmonary fibrosis. Cell Death Dis. 10:6702019. View Article : Google Scholar
|
|
83
|
Xiao T, Zou Z, Xue J, Syed BM, Sun J, Dai
X, Shi M, Li J, Wei S, Tang H, et al: LncRNA H19-mediated M2
polarization of macro-phages promotes myofibroblast differentiation
in pulmonary fibrosis induced by arsenic exposure. Environ Pollut.
268(Pt A): 1158102021. View Article : Google Scholar
|
|
84
|
Wang P, Xiao T, Li J, Wang D, Sun J, Cheng
C, Ma H, Xue J, Li Y, Zhang A and Liu Q: MiR-21 in EVs from
pulmonary epithelial cells promotes myofibroblast differentiation
via glycolysis in arsenic-induced pulmonary fibrosis. Environ
Pollut. 286:1172592021. View Article : Google Scholar : PubMed/NCBI
|
|
85
|
Huang Y, Xie Y, Abel PW, Wei P, Plowman J,
Toews ML, Strah H, Siddique A, Bailey KL and Tu Y: TGF-β1-induced
miR-424 promotes pulmonary myofibroblast differentiation by
targeting Slit2 protein expression. Biochem Pharmacol.
180:1141722020. View Article : Google Scholar
|
|
86
|
Chen X, Shi C, Wang C, Liu W, Chu Y, Xiang
Z, Hu K, Dong P and Han X: The role of miR-97-5p in myofibroblast
differentiation of LR-MSCs and pulmonary fibrogenesis. Sci Rep.
7:409582017. View Article : Google Scholar
|
|
87
|
Wang C, Gu S, Cao H, Li Z, Xiang Z, Hu K
and Han X: MiR-877-3p targets Smad7 and is associated with
myofibroblast differentiation and bleomycin-induced lung fibrosis.
Sci Rep. 6:301222016. View Article : Google Scholar : PubMed/NCBI
|
|
88
|
Akbari Dilmaghnai N, Shoorei H, Sharifi G,
Mohaqiq M, Majidpoor J, Dinger ME, Taheri M and Ghafouri-Fard S:
Non-coding RNAs modulate function of extracellular matrix proteins.
Biomed Pharmacother. 136:1112402021. View Article : Google Scholar : PubMed/NCBI
|
|
89
|
Li J, Zhang X, Wang T, Li J, Su Q, Zhong
C, Chen Z and Liang Y: The MIR155 host
gene/microRNA-627/HMGB1/NF-κB loop modulates fibroblast
proliferation and extracellular matrix deposition. Life Sci.
269:1190852021. View Article : Google Scholar
|
|
90
|
Wang YC, Xie H, Zhang YC, Meng QH, Xiong
MM, Jia MW, Peng F and Tang DL: Exosomal miR-107 antagonizes
profibrotic phenotypes of pericytes by targeting a pathway
involving HIF-1 α/Notch1/PDGFR β/YAP1/Twist1 axis in vitro. Am J
Physiol-Heart Circ Physiol. 320:H520–H534. 2021. View Article : Google Scholar
|
|
91
|
Phan THG, Paliogiannis P, Nasrallah GK,
Giordo R, Eid AH, Fois AG, Zinellu A, Mangoni AA and Pintus G:
Emerging cellular and molecular determinants of idiopathic
pulmonary fibrosis. Cell Mol Life Sci. 78:2031–2057. 2021.
View Article : Google Scholar
|
|
92
|
Zhang S, Jia X, Zhang Q, Zhang L, Yang J,
Hu C, Shi J, Jiang X, Lu J and Shen H: Neutrophil extracellular
traps activate lung fibroblast to induce polymyositis-related
interstitial lung diseases via TLR9-miR-7-Smad2 pathway. J Cell Mol
Med. 24:1658–1669. 2020. View Article : Google Scholar
|
|
93
|
Chen YC, Chen BC, Yu CC, Lin SH and Lin
CH: MiR-19a, -19b, and -26b Mediate CTGF expression and pulmonary
fibroblast differentiation. J Cell Physiol. 231:2236–2248. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
94
|
Yi M, Liu B, Tang Y, Li F, Qin W and Yuan
X: Irradiated human umbilical vein endothelial cells undergo
endothelial-mesenchymal transition via the Snail/miR-199a-5p axis
to promote the differentiation of fibroblasts into myofibroblasts.
Biomed Res Int. 2018:41358062018. View Article : Google Scholar : PubMed/NCBI
|
|
95
|
Desai O, Winkler J, Minasyan M and Herzog
EL: The role of immune and inflammatory cells in idiopathic
pulmonary fibrosis. Front Med (Lausanne). 5:432018. View Article : Google Scholar
|
|
96
|
Heukels P, Moor CC, von der Thüsen JH,
Wijsenbeek MS and Kool M: Inflammation and immunity in IPF
pathogenesis and treatment. Respir Med. 147:79–91. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
97
|
Zhu M, An Y, Zhang X, Wang Z and Duan H:
Experimental pulmonary fibrosis was suppressed by microRNA-506
through NF-kappa-mediated apoptosis and inflammation. Cell Tissue
Res. 378:255–265. 2019. View Article : Google Scholar
|
|
98
|
Zhou L, Li P, Zhang M, Han B, Chu C, Su X,
Li B, Kang H, Ning J, Zhang B, et al: Carbon black nanoparticles
induce pulmonary fibrosis through NLRP3 inflammasome pathway
modulated by miR-96 targeted FOXO3a. Chemosphere. 241:1250752020.
View Article : Google Scholar
|
|
99
|
Mo Y, Zhang Y, Wan R, Jiang M, Xu Y and
Zhang Q: MiR-21 mediates nickel nanoparticle-induced pulmonary
injury and fibrosis. Nanotoxicology. 14:1175–1197. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
100
|
Tan S and Chen S: The mechanism and effect
of autophagy, apoptosis, and pyroptosis on the progression of
silicosis. Int J Mol Sci. 22:81102021. View Article : Google Scholar :
|
|
101
|
Zhao H, Wang Y, Qiu T, Liu W and Yao P:
Autophagy, an important therapeutic target for pulmonary fibrosis
diseases. Clin Chim Acta. 502:139–147. 2020. View Article : Google Scholar
|
|
102
|
Lv X, Li K and Hu Z: Autophagy and
pulmonary fibrosis. Adv Exp Med Biol. 1207:569–579. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
103
|
Xu T, Yan W, Wu Q, Xu Q, Yuan J, Li Y, Li
P, Pan H and Ni C: MiR-326 inhibits inflammation and promotes
autophagy in Silica-Induced pulmonary fibrosis through targeting
TNFSF14 and PTBP1. Chem Res Toxicol. 32:2192–2203. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
104
|
Han R, Ji X, Rong R, Li Y, Yao W, Yuan J,
Wu Q, Yang J, Yan W, Han L, et al: MiR-449a regulates autophagy to
inhibit silica-induced. pulmonary fibrosis through targeting Bcl2.
J Mol Med (Berl). 94:1267–1279. 2016. View Article : Google Scholar
|
|
105
|
Zareba L, Szymanski J, Homoncik Z and
Czystowska-Kuzmicz M: EVs from BALF-Mediators of inflammation and
potential biomarkers in lung diseases. Int J Mol Sci. 22:36512021.
View Article : Google Scholar : PubMed/NCBI
|
|
106
|
Shaba E, Landi C, Carleo A, Vantaggiato L,
Paccagnini E, Gentile M, Bianchi L, Lupetti P, Bargagli E, Prasse A
and Bini L: Proteome characterization of BALF extracellular
vesicles in idiopathic pulmonary fibrosis: Unveiling undercover
molecular pathways. Int J Mol Sci. 22:56962021. View Article : Google Scholar :
|
|
107
|
Rodríguez M, Silva J, López-Alfonso A,
López-Muñiz MB, Peña C, Domínguez G, García JM, López-Gónzalez A,
Méndez M, Provencio M, et al: Different exosome cargo from
plasma/bronchoalveolar lavage in non-small-cell lung cancer. Genes
Chromosomes Cancer. 53:713–724. 2014.PubMed/NCBI
|
|
108
|
Liu B, Jiang T, Hu X, Liu Z, Zhao L, Liu
H, Liu Z and Ma L: Downregulation of microRNA-30a in
bronchoalveolar lavage fluid from idiopathic pulmonary fibrosis
patients. Mol Med Rep. 18:5799–5806. 2018.PubMed/NCBI
|
|
109
|
Zhu L, Chen Y, Chen M and Wang W:
Mechanism of miR-204-5p in exosomes derived from bronchoalveolar
lavage fluid on the progression of pulmonary fibrosis via AP1S2.
Ann Transl Med. 9:10682021. View Article : Google Scholar : PubMed/NCBI
|
|
110
|
Guiot J, Demarche S, Henket M, Paulus V,
Graff S, Schleich F, Corhay JL, Louis R and Moermans C: Methodology
for Sputum Induction and Laboratory Processing. J Vis Exp.
130:566122017.
|
|
111
|
Pastor L, Vera E, Marin JM and Sanz-Rubio
D: Extracellular Vesicles from Airway Secretions: New insights in
lung diseases. Int J Mol Sci. 22:5832021. View Article : Google Scholar :
|
|
112
|
Guiot J, Henket M, Corhay JL, Moermans C
and Louis R: Sputum biomarkers in IPF: Evidence for raised gene
expression and protein level of IGFBP-2, IL-8 and MMP-7. PLoS One.
12:e01713442017. View Article : Google Scholar : PubMed/NCBI
|
|
113
|
Njock MS, Guiot J, Henket MA, Nivelles O,
Thiry M, Dequiedt F, Corhay JL, Louis RE and Struman I: Sputum
exosomes: Promising biomarkers for idiopathic pulmonary fibrosis.
Thorax. 74:309–312. 2019. View Article : Google Scholar
|
|
114
|
Trappe A, Donnelly SC, McNally P and
Coppinger JA: Role of extracellular vesicles in chronic lung
disease. Thorax. 76:1047–1056. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
115
|
Hua Y, Ding Y, Hou Y, Liu Y, Mao K, Cui Y
and Nie H: Exosomal MicroRNA: Diagnostic marker and therapeutic
tool for lung diseases. Curr Pharm Des. 27:2934–2942. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
116
|
Minnis P, Kane R, Anglin R, Walsh S,
Worrel J, Khan F, Lumsden RV, Whitty S and Keane MP: Serum exosomes
from IPF patients display a fibrotic. miRNA profile that correlates
to clinical measures of disease severity. Eur Respir Rev. 46(Suppl
59): PA38452015.
|
|
117
|
Makiguchi T, Yamada M, Yoshioka Y, Sugiura
H, Koarai A, Chiba S, Fujino N, Tojo Y, Ota C, Kubo H, et al: Serum
extra-cellular vesicular miR-21-5p is a predictor of the prognosis
in idiopathic pulmonary fibrosis. Respir Res. 17:1102016.
View Article : Google Scholar
|
|
118
|
Lacedonia D, Scioscia G, Soccio P, Conese
M, Catucci L, Palladino GP, Simone F, Quarato CMI, Di Gioia S, Rana
R, et al: Downregulation of exosomal let-7d and miR-16 in
idiopathic pulmonary fibrosis. BMC Pulm Med. 21:1882021. View Article : Google Scholar : PubMed/NCBI
|
|
119
|
Kuse N, Kamio K, Azuma A, Matsuda K,
Inomata M, Usuki J, Morinaga A, Tanaka T, Kashiwada T, Atsumi K, et
al: Exosome-derived microRNA-22 ameliorates. pulmonary fibrosis by
regulating fibroblast-to-myofibroblast differentiation in vitro and
in vivo. J Nippon Med Sch. 87:118–128. 2020. View Article : Google Scholar
|
|
120
|
Inomata M, Kamio K, Azuma A, Matsuda K,
Usuki J, Morinaga A, Tanaka T, Kashiwada T, Atsumi K, Hayashi H, et
al: Rictor-targeting exosomal microRNA-16 ameliorates lung fibrosis
by inhibiting the mTORC2-SPARC axis. Exp Cell Res. 398:1124162021.
View Article : Google Scholar
|
|
121
|
Vasse GF, Nizamoglu M, Heijink IH,
Schlepütz M, van Rijn P, Thomas MJ, Burgess JK and Melgert BN:
Macrophage-stroma interactions in fibrosis: Biochemical,
biophysical, and cellular perspectives. J Pathol. 254:344–357.
2021. View Article : Google Scholar : PubMed/NCBI
|
|
122
|
Kishore A and Petrek M: Roles of
macrophage polarization and macrophage-derived. miRNAs in pulmonary
fibrosis. Front Immunol. 12:6784572021. View Article : Google Scholar
|
|
123
|
Yao MY, Zhang WH, Ma WT, Liu QH, Xing LH
and Zhao GF: MicroRNA-328 in. exosomes derived from M2 macrophages
exerts a promotive effect on the progression of pulmonary fibrosis
via FAM13A in a rat model. Exp Mol Med. 51:1–16. 2019. View Article : Google Scholar
|
|
124
|
Wang D, Hao C, Zhang L, Zhang J, Liu S, Li
Y, Qu Y, Zhao Y, Huang R, Wei J and Yao W: Exosomal miR-125a-5p
derived from silica-exposed. macrophages induces fibroblast
transdifferentiation. Ecotoxicol Environ Saf. 192:1102532020.
View Article : Google Scholar
|
|
125
|
Guiot J, Cambier M, Boeckx A, Henket M,
Nivelles O, Gester F, Louis E, Malaise M, Dequiedt F, Louis R, et
al: Macrophage-derived exosomes attenuate fibrosis. in airway
epithelial cells through delivery of antifibrotic miR-142-3p.
Thorax. 75:870–881. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
126
|
Cruz FF and Rocco PRM: The potential of
mesenchymal stem cell therapy for chronic lung disease. Expert Rev
Respir Med. 14:31–39. 2020. View Article : Google Scholar
|
|
127
|
Yang S, Liu P, Jiang Y, Wang Z, Dai H and
Wang C: Therapeutic applications of. mesenchymal stem cells in
idiopathic pulmonary fibrosis. Front Cell Dev Biol. 9:6396572021.
View Article : Google Scholar
|
|
128
|
Ntolios P, Manoloudi E, Tzouvelekis A,
Bouros E, Steiropoulos P, Anevlavis S, Bouros D and Froudarakis M:
Longitudinal outcomes of patients. Enrolled. in a phase Ib clinical
trial of the adipose-derived stromal cells-stromal vascular
fraction in idiopathic pulmonary fibrosis. Clin Respir J.
12:2084–2089. 2018. View Article : Google Scholar
|
|
129
|
Hade MD, Suire CN and Suo Z: Mesenchymal
stem cell-derived exosomes: Applications in regenerative medicine.
Cells. 10:19592021. View Article : Google Scholar : PubMed/NCBI
|
|
130
|
Wan X, Chen S, Fang Y, Zuo W, Cui J and
Xie S: Mesenchymal stem cell-derived. extracellular vesicles
suppress the fibroblast proliferation by downregulating FZD6
expression in fibroblasts via micrRNA-29b-3p in idiopathic
pulmonary fibrosis. J Cell Physiol. 235:8613–8625. 2020. View Article : Google Scholar
|
|
131
|
Lei X, He N, Zhu L, Zhou M, Zhang K, Wang
C, Huang H, Chen S, Li Y, Liu Q, et al: Mesenchymal stem
cell-derived extracellular vesicles Attenuate radiation-induced
lung injury via miRNA-214-3p. Antioxid Redox Signal. 35:849–862.
2021. View Article : Google Scholar
|
|
132
|
Zhou J, Lin Y, Kang X, Liu Z, Zhang W and
Xu F: MicroRNA-186 in extracellular vesicles. from bone marrow
mesenchymal stem cells alleviates idiopathic pulmonary fibrosis via
interaction with SOX4 and DKK1. Stem Cell Res Ther. 12:962021.
View Article : Google Scholar
|
|
133
|
Ibrahim A, Ibrahim A and Parimon T:
Diagnostic and therapeutic applications of extracellular vesicles
in interstitial lung diseases. Diagnostics (Basel). 11:872021.
View Article : Google Scholar
|
|
134
|
Parimon T, Yao C, Habiel DM, Ge L, Bora
SA, Brauer R, Evans CM, Xie T, Alonso-Valenteen F, Medina-Kauwe LK,
et al: Syndecan-1 promotes lung fibrosis by. regulating epithelial
reprogramming through extracellular vesicles. JCI Insight.
4:e1293592019. View Article : Google Scholar
|
|
135
|
Feng Z, Zhou J, Liu Y, Xia R, Li Q, Yan L,
Chen Q, Chen X, Jiang Y, Chao G, et al: Epithelium- and
endothelium-derived exosomes regulate the. alveolar macrophages by
targeting RGS1 mediated calcium signaling-dependent immune
response. Cell Death Differ. 28:2238–2256. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
136
|
Dinh PC, Paudel D, Brochu H, Popowski KD,
Gracieux MC, Cores J, Huang K, Hensley MT, Harrell E, Vandergriff
AC, et al: Inhalation of lung spheroid cell secretome and exosomes
promotes lung repair in pulmonary fibrosis. Nat Commun.
11:10642020. View Article : Google Scholar :
|
|
137
|
Kadota T, Fujita Y, Araya J, Watanabe N,
Fujimoto S, Kawamoto H, Minagawa S, Hara H, Ohtsuka T, Yamamoto Y,
et al: Human bronchial epithelial cell-derived. Extracellular.
vesicle therapy for pulmonary fibrosis via inhibition of TGF-β-WNT
crosstalk. J Extracell Vesicles. 10:e121242021. View Article : Google Scholar
|
|
138
|
Liu Z, Yan J, Tong L, Liu S and Zhang Y:
The role of exosomes from BALF in lung disease. J Cell Physiol. Aug
13–2021.Epub ahead of print.
|
|
139
|
Tieu A, Hu K, Gnyra C, Montroy J,
Fergusson DA, Allan DS, Stewart DJ, Thébaud B and Lalu MM:
Mesenchymal stromal cell extracellular vesicles as. Therapy. for
acute and chronic respiratory diseases: A meta-analysis. J
Extracell Vesicles. 10:e121412021. View Article : Google Scholar
|